Targeted synergistic therapy for bone and brain disorders using hydrogel microneedles loaded with exosomes and drugs
Abstract
This review highlights an innovative co-delivery system using hydrogel microneedles (MNs) to encapsulate exosomes and drugs, presenting a transformative strategy for treating interconnected bone and brain disorders. By offering potential strategies to address the blood-brain barrier, MNs enable minimally invasive delivery of exosomes, natural vesicles rich in microRNAs and cytokines, facilitating spatiotemporal control over therapeutic release. This system enhances regenerative outcomes in diabetic wounds, myocardial infarction, osteochondral defects, and neurological conditions via multi-mechanistic actions including immunomodulation, angiogenesis, and tissue repair. However, clinical translation remains challenging due to issues in exosome standardization, microneedle mechanical properties, and long-term biosafety.
Keywords
INTRODUCTION
Regenerative medicine faces challenges such as persistent inflammation, insufficient angiogenesis, and dysregulated extracellular matrix (ECM) remodeling[1,2]. Conventional approaches, including systemic drug delivery and cell transplantation are hampered by off-target effects, poor bioavailability, and immune rejection[3,4]. To overcome these limitations, hydrogel–exosome-based synergistic systems have emerged as a promising platform for localized therapy [Figure 1]. Building on this concept, hydrogel microneedles (MNs) offer an advanced strategy for the co-delivery of exosomes and therapeutic agents by enabling minimally invasive penetration and deeper tissue diffusion. Hydrogel MNs leverage the biocompatibility and structural versatility of hydrogels to enable controlled release, while exosomes serve as natural nanocarriers mediating intercellular communication [Figure 2][5,6]. Their combination allows spatiotemporally precise delivery via the porous hydrogel network, enhancing the loading and release efficiency of both exosomes and drugs[6-8]. Furthermore, mechanical properties can be tuned via crosslinking density, and functionalization with targeting motifs or responsive linkers enhances delivery precision [Figure 2][9]. Advanced techniques, such as three-dimensional (3D) printing, further enable the creation of scaffolds with graded release profiles[10,11]. However, a pivotal challenge remains with the blood-brain barrier (BBB). While hydrogel MNs show promise for brain disorders, transcending the BBB remains a critical frontier, necessitating future designs that incorporate receptor-mediated transcytosis or advanced nanocarriers[12,13]. Concurrently, exosomes derived from mesenchymal stem cells (MSCs) exhibit unique advantages in chronic inflammatory microenvironments due to their inherent immunomodulatory and proangiogenic properties [Figure 3][14]. To enhance targeting precision, strategies such as tissue-specific exosomes or surface engineering modifications have been developed to improve lesion-specific accumulation[15,16]. Furthermore, the emerging concept of the "Bone-Brain Axis", which highlights bidirectional skeletal-neurological crosstalk, underscores the potential of this platform for simultaneously treating pathophysiologically linked disorders. Engineering strategies further amplify outcomes, exemplified by miR-21-5p overexpression enhancing anti-fibrotic effects[17], or cluster of differentiation 44 (CD44)-targeting peptides improving tissue accumulation[18]. Co-loading exosomes with drugs synergistically inhibits inflammation and promotes angiogenesis[19].
Figure 1. Comparison of traditional bone therapies and hydrogel-exosome synergistic systems. Traditional bone therapies such as autografts, allografts, and synthetic materials present several limitations. Autografts, though biocompatible, often lead to donor site pain and carry a risk of bacterial infection. Allografts and synthetic scaffolds may offer structural support but are prone to immune rejection due to foreign material implantation. In contrast, hydrogel-exosome synergistic systems provide a promising alternative. These systems utilize hydrogels as smart carriers that respond to environmental stimuli such as pH and temperature, allowing for controlled exosome release. Exosomes enriched with proteins such as CD81 and CD9 enhance cellular communication and tissue regeneration. The combined system supports osteogenesis, angiogenesis, and immune regulation through interactions with osteoblasts, macrophages, and fibroblasts.
Figure 2. Comparison of traditional bone repair and hydrogel-exosome synergy for bone regeneration. This figure compares traditional bone repair methods with the innovative hydrogel-exosome system. The top panel illustrates three traditional approaches: Autografts, which offer high compatibility but may cause donor site complications; Allografts, which reduce the need for donor tissue but carry immune rejection risks; and Synthetic Materials, which provide temporary support but lack bioactivity. The bottom panel highlights the Hydrogel-Exosome Synergy, where exosomes deliver crucial signals for osteoblast proliferation, immune modulation, and angiogenesis, while hydrogels provide a controlled, biodegradable environment that ensures targeted release and 3D printability. In addition to these regenerative benefits, exosomes may carry oncogenic microRNAs or proteins that could activate tumor-related pathways, representing a potential oncogenic risk that must be carefully evaluated in clinical translation. This combination promotes effective bone remodeling. Challenges including exosome yield, hydrogel degradation, and safety concerns such as tumorigenic potential are emphasized, suggesting directions for future research to optimize these systems for clinical use.
Figure 3. Biological characteristics, source selection, and engineering strategies of exosomes for bone repair. (A) Molecular characteristics and mechanisms: Exosomes exhibit low immunogenicity, excellent biocompatibility, and high stability in circulation. They facilitate intercellular communication to promote bone regeneration and repair. (B) Source selection and functional differences: Mesenchymal stem cell (MSC)-derived exosomes (such as those derived from BMSCs, hUCMSCs, and ADSCs) possess robust osteogenic properties, activating paracrine pathways and recruiting bone marrow stromal cells. Furthermore, these exosomes promote the phenotypic polarization of M2 macrophages, which effectively enhances bone angiogenesis and healing. Advanced engineering strategies, including gene regulation (e.g., CRISPR for overexpressing specific miRNAs), targeted delivery, and utilizing their ability to penetrate the blood-marrow barrier, further augment their therapeutic efficacy in clinical translation.
The synergy between exosomes and hydrogels operates through multiple mechanisms. In the contexts of inflammation and angiogenesis, their coordinated actions include the inhibition of Nuclear factor-κB (NF-κB) by exosomal miR-146a combined with hydrogel-mediated delivery of antimicrobial peptides (AMPs)[20,21]. In angiogenesis, exosomal vascular endothelial growth factor (VEGF) and hydrogel-released basic fibroblast growth factor (bFGF) jointly activate the phosphoinositide 3-kinase/protein kinase B (PI3K/AKT) pathway to enhance neovascularization[22]. Moreover, hydrogel-based reactive oxygen species (ROS) scavenging alleviates hypoxia, thereby amplifying exosomal therapeutic effects[23,24]. Metabolic reprogramming is another important mechanism. For instance, exosomes combined with AMP-activated protein kinase (AMPK) agonists promote M2 macrophage polarization, while hydrogels maintain local concentrations, thereby sustaining the effect for over 72 h[25]. This synergy promotes collagen deposition and accelerates healing through peroxisome proliferator-activated receptor gamma (PPAR-γ) activation. Preclinical studies have shown efficacy in diabetic wounds, myocardial infarction, and bone/cartilage repair[26]. However, clinical translation faces obstacles, including exosome batch heterogeneity and particle size fluctuations, which require standardized quality control systems[27,28]. The mechanical strength of microneedles must be balanced with penetration force via structural design. Additionally, long-term biosafety concerns, including immune risks associated with exosomal heterogeneity and potential toxicity from hydrogel degradation, must be evaluated more thoroughly in large-animal models to ensure clinical safety[29].
In the context of global aging, this minimally invasive, multi-mechanism strategy holds significant promise for transforming the treatment of refractory diseases such as diabetic foot ulcers and myocardial infarction[26]. Through the deep integration of materials science, molecular biology, and clinical medicine, this strategy has the potential to revolutionize the management of difficult-to-heal wounds.
HYDROGEL MICRONEEDLE DELIVERY ADVANTAGES
Hydrogel types and characteristics
Natural polymer hydrogels (chitosan, gelatin, hyaluronic acid)
Natural polymer hydrogels, owing to their high similarity to the ECM, serve as biocompatible carriers for delivering neuroregulatory or osteoimmunomodulatory exosomes across systemic barriers, facilitating localized repair at the skeletal and neurological interface [Table 1]. For instance, chitosan forms a degradable network through β-1,4 glycosidic bond crosslinking; its amino groups enable electrostatic adsorption of negatively charged exosomes, facilitating targeted delivery to inflammation sites[30,31]. In osteoporotic models with cognitive decline, such systems increased bone mass and improved hippocampal plasticity[32].
Comprehensive summary of hydrogel microneedle systems for synergistic exosome and drug delivery
| Category | Subtype/Component | Composition & design | Key properties & release kinetics | Therapeutic outcomes & mechanisms |
| Hydrogel matrix | Chitosan-based | Chitosan/β-glycerophosphate thermosensitive hydrogel; porous 3D network | Gelation at 37 °C; sustained release via swelling | ↑ Neovascularization (CD31+ density); ↑ collagen deposition; restored epidermal thickness; inhibited NF-κB |
| GelMA | Gelatin methacrylate; photocrosslinked porous structure | Tunable modulus (2-15 kPa); ROS-responsive (3×release under inflammation) | Protected exosome integrity; enhanced fibroblast migration and axonal sprouting; on-demand release | |
| Pluronic F127 | Thermosensitive triblock copolymer | Shear-thinning; semi-solid gel at body temperature; sustained retention | Minimally invasive injection; extended exosome retention in joint cavities | |
| OSA-CMCS | Oxidized hyaluronic acid-carboxymethyl chitosan (Schiff base) | High swelling ratio; self-healing (G' > G''); pH-sensitive | Maintained moist environment; prolonged exosomal retention via CD44 receptor uptake | |
| Microneedle design | Core-shell structure | Microfluidics-fabricated; shell: vancomycin/curcumin; core: exosomes | Sequential release: rapid drug release → sustained exosome delivery; ROS-accelerated degradation | Early anti-infection/anti-inflammation + late-stage regeneration; tripled release rate |
| Layered structure | 3D-printed bilayer scaffold: upper (cartilage: GelMA), lower (bone: β-TCP) | Gradient porosity and stiffness; spatiotemporal release | Chondrogenesis (SOX9, miR-140) + osteogenesis (BMP-2/RUNX2); increased BV/TV | |
| Barbed structure | 3D-printed SilMA with backward-facing barbs | Enhanced tensile modulus; secure tissue interlocking | Superior tissue adhesion and in situ retention; ensured mechanical stability | |
| Exosome Source | MSC-derived | miR-21-5p, TGF-β, TSG6; hypoxia-preconditioned or genetically engineered | Paracrine delivery; enhanced VEGF expression | Angiogenesis (PI3K/AKT); M2 macrophage polarization; immunomodulation (↓IL-6, TNF-α) |
| Tissue-specific | Cardiomyocyte (Hsp70, miR-199a-3p); Chondrocyte (SOX9, miR-140-5p) | Inherited tissue-homing capability | Reduced apoptosis; inhibited MMP13; activated tissue-specific repair pathways | |
| Engineered (Targeted) | Surface modified (e.g., CD44-peptide, Lamp2b-CD44); loaded with miRNAs (e.g., miR-146a) | Enhanced targeting and circulation half-life; improved BBB penetration | Specific accumulation at lesions; prolonged therapeutic effect; enhanced tumor suppression | |
| Synergistic therapy | Exo + curcumin | Co-loaded in core-shell MN; outer curcumin, inner exosomes | Temporal release: rapid anti-inflammation → sustained regeneration | Dual "anti-inflammatory-angiogenesis" effect via PI3K/AKT/mTOR; ↓healing time |
| Exo + conductive hydrogel | PPy-Gel hydrogel loaded with MSC exosomes | Combined electrical conductivity + exosome release | Improved electrical conduction velocity; reduced arrhythmia; enhanced angiogenesis | |
| Exo + antimicrobial peptide | LL-37 + MSC exosomes in ROS-sensitive hydrogel | ROS-triggered release in infected microenvironment | Biofilm disruption + immunomodulation (M2 polarization); bacterial reduction | |
| Manufacturing tech | 3D Bioprinting | Layer-by-layer deposition using CAD models | Customizable scaffolds; patient-specific design; mimics ECM | Supports osteoblast adhesion; enables controlled exosome delivery; creates gradient structures |
| Microfluidics | Precise control of flows for synthesis and encapsulation | Uniform microspheres; enhanced exosome stability; high encapsulation efficiency | Protects exosomal cargo; enables precise core-shell structure fabrication | |
| Clinical translation | Preclinical efficacy | ADSC-exosomes + metformin in MN patch; Conductive hydrogel + exosomes | Multicenter preclinical trial data; sustained release over days | Higher diabetic ulcer healing rate; ↑LVEF, ↓fibrotic area, ↓ventricular arrhythmias |
| Key Challenges | Exosome heterogeneity (batch, size); MN mechanical strength; long-term immune safety | Requires standardization (ISO 10993, ICH Q1A) | Batch variability; potential immunogenicity; degradation product toxicity | |
| Future outlook | CRISPR-edited exosomes; AI-driven QC; 3D printing with biosensors | Smart responsive (photo/magnetic); real-time monitoring | Personalized and dynamic therapy; precise spatiotemporal control |
Gelatin hydrogels, particularly gelatin methacrylate (GelMA), exhibit a tunable elastic modulus (2-15 kPa) matching the dermal layer[33]. Notably, photopolymerized GelMA hydrogels preserve exosome integrity and have been adapted to deliver neuroprotective factors such as brain-derived neurotrophic factor (BDNF) to injury sites, enhancing fibroblast migration and axonal sprouting in spinal cord injury models[34]. Hyaluronic acid (HA) hydrogels are favored for intra-articular delivery due to their hydrophilicity and viscoelasticity. Specifically, oxidized hyaluronic acid-carboxymethyl chitosan (OSA-CMCS) hydrogel, formed via Schiff base reactions, maintains a moist wound environment[35]. HA hydrogels also facilitate brain-targeted exosome delivery via CD44 receptor-mediated uptake, prolonging retention in rheumatoid arthritis and neurodegenerative joint syndromes while modulating glial activation and osteoclastogenesis[36].
Synthetic/composite hydrogels
Synthetic hydrogels enable precise control over structure and responsiveness. A key example is Pluronic F127, a temperature-sensitive triblock copolymer that forms a porous 3D matrix for sustained exosome release [Table 1][37,38]. In models of traumatic brain injury with secondary osteoporosis, shear-thinning Pluronic-based hydrogels support injectable microneedle delivery into both periosteal and subcortical regions for dual-site modulation. GelMA hydrogels offer tunable strength and degradation based on the degree of methacrylation[39]. Notably, under near-infrared (NIR) light stimulation, ROS-responsive GelMA enables on-demand exosome release, tripling release rates under inflammatory conditions, allowing dynamic intervention in oxidative stress microenvironments. GelMA also supports 3D printing of complex scaffolds, such as bilayer designs for simultaneous cartilage and bone regeneration.
Microneedle system design optimization
Structural design
Microneedle geometry (height, base diameter, cone angle) critically influences penetration efficiency[40,41]. Porous structures fabricated via freeze-drying or 3D printing enable uniform drug/exosome loading. For example, porous GelMA microneedles allow controlled exosome release via swelling behavior [Figure 4][42,43]. Layered and core-shell structures enable sequential release. Typically, the outer layer rapidly releases drugs, while the inner layer provides sustained exosome release [Table 1][44]. In diabetic wound repair, hydrophobic drugs encapsulated in polylactic-co-glycolic acid (PLGA) exhibit sustained release, while exosomes in the hydrogel outer layer are released rapidly. Microfluidically engineered core-shell systems can encapsulate antimicrobials in the shell and exosomes in the core, with shell degradation accelerating release in ROS-rich microenvironments[45].
Figure 4. Mechanisms, sources, and engineering of exosomes, and microneedle delivery systems for bone regeneration. (A) Molecular Mechanisms of Exosomes: Exosomes promote bone healing through miRNA regulation, protein delivery, and lipid-mediated uptake. These processes enhance osteogenesis, angiogenesis, and immune modulation. (B) Sources and Functions: Exosomes from MSCs, immune cells, and neurovascular cells serve distinct roles. MSC-derived exosomes boost osteogenic signaling; immune cell-derived exosomes regulate inflammation; neurovascular-derived exosomes support vascularization and graft integration. (C) Engineering Strategies: Genetic editing enhances exosome bioactivity, while targeted delivery improves retention in bone defects. Therapeutic loading with factors such as BMP-2 and IGF-1 enhances regeneration while minimizing side effects. (D) Minimally Invasive Penetration and Deep Tissue Diffusion: Design optimization of microneedles, including geometric parameters (height, base diameter, cone angle) and gradient stiffness structures, enables efficient skin penetration. Porous hydrogel structures and photothermal-responsive systems facilitate the controlled and sustained release of exosomes into deep tissues.
Mechanical performance can be enhanced by introducing stiffness gradients. A representative design integrates a polycaprolactone (PCL)-reinforced core with a chitosan outer coating to improve mechanical strength and penetration depth[46]. Barbed structures enhance tissue adhesion stability. For example, 3D-printed silicone-based methacrylate (SilMA) microneedles with a high tensile modulus ensure in situ anchoring on fibrous surfaces [Table 1]. Light- or magnetically-controlled systems modified with gold nanoparticles achieve NIR-triggered drug release, with magnetic fields guiding delivery to target areas.
Loading strategy
Natural hydrogels (chitosan, gelatin) offer biocompatibility and tunable loading via porous structures, achieving high encapsulation efficiency and controlled release through swelling. Thermosensitive gels such as Pluronic F127 solidify at body temperature, supporting injectable systems with sustained exosome retention[38]. Functional modifications enhance targeting and responsiveness. For instance, arginine-glycine-aspartic acid (RGD)-modified GelMA improves fibroblast adhesion and collagen deposition, while boronate ester-based hydrogels increase release in ROS-rich environments[47]. Staged release systems utilize multilayer microneedles to sequentially deliver anti-inflammatory agents and regenerative factors. In diabetic wounds, such strategies reduce healing time, lower infection rates, and significantly increase neovascularization and collagen deposition[48-50].
Microenvironment-responsive design
Stimuli-responsive microneedles enable precise and localized release. pH-sensitive chitosan hydrogels degrade faster in acidic environments, tripling release rates and achieving higher drug accumulation in infected wounds[51]. ROS-responsive systems (based on thioether or boronate bonds) exhibit accelerated degradation under oxidative stress, enhancing antimicrobial and regenerative effects, such as reducing bacterial load and promoting M2 macrophage polarization in diabetic ulcers [Figure 5][52].
Figure 5. Engineered intelligent hydrogel platforms for bone tissue reconstruction. (A) Material Innovation: Different polymeric materials are utilized to engineer hydrogels, including natural polymers, synthetic polymers, and composite materials, each contributing distinct properties for bone repair. Natural polymers provide biocompatibility, synthetic polymers allow customization of mechanical and chemical properties, and composite materials combine the advantages of both. (B) Hydrogel-Induced Bone Defect Repair: These hydrogel platforms promote bone repair through mechanisms such as cell proliferation, differentiation, angiogenesis, and gene expression regulation. Additionally, they exhibit antibacterial properties that help combat infections during the repair process. (C) Release Regulation: The hydrogels are designed to regulate the controlled release of therapeutic agents (e.g., growth factors) in response to pH, ROS, and MMP activity, further enhancing bone regeneration. These platforms maintain a favorable environment for tissue healing and remodeling. (D) Hydrogel Manufacturing Technology: The production of these hydrogels utilizes advanced technologies such as 3D bioprinting for custom shapes and structures and microfluidics for precise material synthesis and formulation, ensuring uniformity and efficiency in hydrogel preparation for clinical applications.
3D printing technology application
3D printing enables precise structural control and functional customization [Figure 6]. For example, SilMA-based arrays with barbed structures show strong mechanical adhesion[53]. In diabetic models, 3D-printed methacrylated hyaluronic acid microneedles co-loaded with exosomes and sitagliptin have been reported to double healing rates, enhance vascularization, and restore epidermal thickness[54,55]. Layered scaffolds mimicking gradient tissue interfaces can support stem cell differentiation and achieve new bone formation through controlled porosity and stiffness gradients[56]. Patient-specific constructs guided by computed tomography (CT) imaging can match defect geometries and significantly improve osteogenesis and matrix remodeling[57]. Stability evaluations confirm that 3D-printed microneedles maintain encapsulation efficiency and mechanical integrity, complying with ISO 10993 and ICH Q1A guidelines for scale-up[55].
Figure 6. Biomaterials, release mechanisms, and manufacturing technologies for hydrogel-based delivery systems in bone repair.(A) Biomaterials for Hydrogel-Based Systems: Various materials are employed for hydrogel-based delivery systems, including natural materials (e.g., GelMA, Chitosan), synthetic materials (e.g., Pluronic F127, PEG/HA), and composite materials (e.g., Black Phosphorus, Bioactive Glass). These materials are chosen for their biocompatibility, antibacterial properties, and ability to support cell proliferation and differentiation, enhance angiogenesis, and promote bone matrix formation. (B) Release Mechanisms in Hydrogel-Based Exosome Delivery: Several strategies, including MMP-Responsive, ROS-Sensitive, and pH-Responsive mechanisms, regulate the release of therapeutic exosomes. These mechanisms are activated by specific factors such as MMP-9 upregulation, elevated ROS levels, and acidic microenvironments at injury sites, facilitating targeted delivery, regulating exosome release, and preventing premature loss. (C) Advanced Manufacturing Techniques: 3D bioprinting and microfluidics are employed to fabricate hydrogel-based delivery systems. 3D bioprinting enables customizable, patient-specific scaffolds, while microfluidics allows precise exosome encapsulation and stability. These techniques optimize exosome bioactivity, enable controlled delivery, and protect therapeutic cargo for effective bone healing.
Mechanical property optimization
Mechanical optimization encompasses both material innovation and structural design. The crosslinking density in hydrogels, such as Pluronic F127 and GelMA, can be precisely tuned. F127 generates semi-solid gels that facilitate exosome retention in joint cavities, whereas porous GelMA degrades in ROS-rich environments, thereby accelerating release without compromising structural integrity[58]. Stiffness gradient designs enhance penetration capability and fracture strength[59]. Dynamic responsive behaviors are also critical; shear-thinning hydrogels exhibit low viscosity during injection but rapidly regain elasticity thereafter, making them ideal for dynamic tissues[60]. Computational tools, such as finite element analysis, facilitate design refinement. For instance, a dual-layer 3D-printed scaffold has been shown to enhance stem cell differentiation and promote new bone formation[57]. Preclinical validation in rodents confirms that optimized microneedles restore epidermal thickness, promote angiogenesis, and enhance collagen deposition[61]. Future efforts should leverage multi-omics and smart materials to fine-tune mechanical strength, penetration efficiency, and functionality in complex disease microenvironments[62].
Biocompatibility and safety
Biocompatibility is essential for clinical translation. Chitosan-based thermosensitive hydrogels show low cytotoxicity (meeting ISO 10993 standards), support cell proliferation, and enhance angiogenesis in diabetic wounds. OSA-CMCS hydrogels exhibit hemolysis rates below safety thresholds and self-healing capability, ensuring post-injection stability[63,64]. PLGA degradation may generate acidic by-products that induce inflammation, necessitating optimization of the composition ratio. GelMA hydrogels loaded with exosomes can fully degrade without residues or immune responses. In animal models, microneedles improve cardiac ejection fraction, reduce fibrosis, and show no serum toxicity[65]. For example, PLGA-poly (ethylene glycol) (PEG)-PLGA hydrogels restore cartilage thickness in rabbits without inducing inflammation[66]. Future designs should incorporate antioxidant groups and controlled degradation properties to enhance biocompatibility and support standardized manufacturing to minimize exosome heterogeneity[67].
Delivery efficiency enhancement mechanisms
Delivery efficiency can be enhanced through structural and functional innovations [Figure 6]. Natural polymer hydrogels (chitosan, gelatin) with porous structures support exosome encapsulation and thermosensitive release. Pluronic F127 and GelMA further improve mechanical strength and responsiveness; The shear-thinning behavior of F127 enables injectable delivery[68]. RGD peptides and ROS-sensitive modifications enhance targeting capability. RGD improves fibroblast adhesion and collagen deposition, while phenylboronate esters significantly increase release rates in ROS-rich environments[69]. Core-shell structures, created through microfluidics, facilitate sequential release of drugs and exosomes, thereby promoting regeneration[45]. Intelligent responsive systems integrate photothermal and magnetic control strategies. Exosomes modified with gold nanoparticles enable NIR-triggered drug release, with magnetic fields guiding delivery to tumors[70]. pH- and enzyme-dual-responsive microspheres allow controlled swelling and release via matrix metalloproteinase (MMP)-mediated degradation [Figure 6][71]. 3D-printed barb structures improve tissue adhesion[53]. Mechanical optimization via PCL/chitosan gradients supports deep penetration and biocompatibility[46]. In diabetic wound models, these strategies enhance angiogenesis, collagen deposition, and epidermal thickness, while reducing infection [72]. Integrated multimodal responsiveness and personalized design represent a promising path for improving bioavailability and therapeutic precision[73].
Mechanisms to enhance delivery efficacy
Minimally invasive penetration and deep tissue diffusion
The hydrogel matrix facilitates uniform cargo loading and release. Initially, the outer layer releases antibacterial agents, followed by the release of exosomes from the core. The shell degrades more rapidly in microenvironments enriched with ROS[74]. Intelligent responsive microneedles integrate photothermal and magnetic control. Exosomes modified with gold nanoparticles enable NIR-triggered drug release, with magnetic fields guiding delivery to target areas[70]. Biomimetic barbed structures via 3D printing (e.g., SilMA-based arrays) enhance tissue adhesion[53]. Photothermal-responsive hydrogels enable controlled exosome release, and magnetic guidance further improves targeting. Preclinical results show increased neovascularization, enhanced collagen deposition, recovery of epidermal thickness, and reduced infection[75].
Spatiotemporal controlled release mechanism
Natural polymer hydrogels (chitosan, gelatin) with thermosensitive properties support minimally invasive injection and sustained release. Their porous structures enable efficient exosome loading. Synthetic composite hydrogels further optimize mechanical performance and responsiveness through regulation of crosslinking density[76]. In contrast, unstructured carriers typically exhibit significantly shorter retention under the same conditions, further emphasizing the superior release control of the designed system. Functional modifications enhance responsiveness; specifically, RGD peptides improve fibroblast adhesion and collagen deposition[69], while ROS-sensitive boronic ester bonds accelerate degradation and release in oxidative environments. Temporal release designs utilize dual-layer or multilayer loading to achieve synergistic effects, with the outer layer enabling rapid release of curcumin to inhibit inflammation and the inner layers maintaining sustained exosome release to promote regeneration. This strategy effectively shortens healing time and reduces infection rates in diabetic wounds[76]. Preclinical data confirm improved healing outcomes, including enhanced neovascularization and collagen synthesis. Future research should focus on intelligent materials and multi-omics approaches to improve delivery precision and standardize exosome production[77].
Microenvironment-responsive delivery
ROS-sensitive hydrogels degrade via boronic ester bonds in tumor microenvironments, tripling release rates; Loaded exosomes deliver microRNA (miRNA) to inhibit tumor proliferation[78]. pH-sensitive designs degrade in acidic environments, thereby enhancing drug delivery[22]. Thermoresponsive hydrogels form gels at physiological temperature, enabling prolonged retention of exosomes. Biomimetic targeting strategies direct drugs to infection sites, reducing biofilm thickness[79]. DNA hydrogels, triggered by tumor-associated MMPs, release nucleases and exosomes, achieving tumor inhibition in vivo. Photothermal-responsive materials combined with magnetic guidance further enhance targeted release[80]. These innovations enable precise release under pathological conditions, improving therapeutic outcomes[73]. In diabetic wounds, ROS-responsive microneedles increase exosome release, improve neovascularization and collagen deposition, and restore epidermal thickness.
EXOSOMES’ THERAPEUTIC POTENTIAL AND ENGINEERING STRATEGIES
Sources and functions of exosomes
Mesenchymal stem cell-derived exosomes
MSC-derived exosomes exhibit immunomodulatory effects, promote angiogenesis, and support tissue repair [Figure 3]. Notably, exosomal miR-21-5p activates the PI3K/AKT pathway, stimulating osteogenesis and conferring neuroprotection to hippocampal neurons under oxidative stress, thereby demonstrating a bone-brain axis connection [Table 1][81,82]. These exosomes modulate inflammatory mediators and deliver miRNAs that influence osteoclast activity and neuronal survival[83,84]. Engineering strategies can further boost therapeutic effects by enhancing VEGF expression and neovascularization[85]. Surface modification with targeting ligands prolongs circulation and improves lesion-specific delivery. Clinically, MSC exosomes deliver miR-140-5p in osteoarthritis to inhibit MMP13 and upregulate collagen type II alpha 1 (Col2A1), restoring cartilage thickness[86]. In nerve regeneration, they deliver BDNF and miR-219 to promote axonal regeneration and improve motor function (as assessed by the Basso, Beattie, and Bresnahan score) in spinal cord injury models. Their multi-mechanistic synergistic effects include macrophage polarization, activation of the Wnt/β-catenin pathway for fibroblast migration and collagen deposition, and antioxidant ROS clearance. In myocardial repair, MSC exosomes incorporated into conductive hydrogels restore electrical conduction via connexin 43 (Cx43) and promote angiogenesis.
Tissue-specific exosomes
Tissue-specific exosomes inherit unique molecular signatures from their parental cells, enabling targeted therapeutic interventions[87,88]. For instance, cardiomyocyte-derived exosomes deliver heat shock protein 70 (Hsp70) and miR-199a-3p to activate PI3K/AKT signaling, thereby reducing apoptosis, improving left ventricular ejection fraction (LVEF), and promoting angiogenesis[89-92]. Similarly, adipocyte-derived exosomes, enriched with VEGF, platelet-derived growth factor (PDGF), and miR-29a, modulate ECM degradation and stimulate fibroblast function, thereby accelerating diabetic wound closure and enhancing neovascularization[93,94]. Chondrocyte-derived exosomes carry SRY-box transcription factor 9 (SOX9) and miR-140-5p to inhibit A disintegrin and metalloproteinase with thrombospondin motifs 5 (ADAMTS-5) and activate Wnt/β-catenin signaling, facilitating cartilage repair[95], while neuronal exosomes transporting BDNF, nerve growth factor (NGF), and miR-219 reduce glial scar formation and improve functional recovery after spinal cord injury[96,97]. Furthermore, certain exosomes demonstrate context-dependent functions. Tumor cell-derived exosomes, for example, typically promote progression through immune suppression and oncogenic miRNAs such as miR-21, yet they can also be engineered for targeted chemotherapy, such as the delivery of doxorubicin[98,99]. Hepatocyte-derived exosomes exert anti-fibrotic effects by transferring miR-4500 and hepatocyte growth factor (HGF), modulating the transforming growth factor-beta/small mothers against decapentaplegic homolog (Smad) and nuclear factor erythroid 2-related factor 2/heme oxygenase-1 (Nrf2/HO-1) pathways to alleviate fibrosis and oxidative stress[100,101]. Skin cell-derived exosomes (from keratinocytes/fibroblasts) promote wound healing via miR-31 and keratinocyte growth factor (KGF), enhancing fibroblast migration, collagen synthesis, and targeted delivery to inflamed tissues through CD44 receptors [102,103]. Placental exosomes, enriched in soluble fms-like tyrosine kinase-1 (sFlt-1) and interleukin-10 (IL-10), inhibit pathological angiogenesis and inflammation in preeclampsia, improving both maternal and fetal outcomes[104,105].
Apoptotic neutrophil-derived extracellular vesicles
Apoptotic neutrophil-derived extracellular vesicles (Anx-N-EVs) play a crucial role in resolving inflammation by delivering pro-apoptotic and anti-inflammatory signals. These vesicles are characterized by the surface expression of phosphatidylserine (PS) and annexin A5, which enhance macrophage efferocytosis through PS receptor recognition[106,107]. Mechanistically, Anx-N-EVs deliver miR-146a-5p and TGF-β1 to inhibit NF-κB signaling, reducing tumor necrosis factor-alpha (TNF-α) and interleukin-6 (IL-6) secretion while promoting M2 macrophage polarization and increasing IL-10/TGF-β production[108,109]. They also activate the PI3K/AKT pathway via T cell immunoglobulin and mucin domain-containing protein 4 (TIM-4)-PS interaction, further enhancing phagocytic efficiency[106,107]. In diabetic ulcer models, Anx-N-EVs combined with hydrogels significantly increase CD31+ vessel density and accelerate epithelialization. In rheumatoid arthritis, miR-146a-5p targets TNF receptor-associated factor 6 (TRAF6) and interleukin-1 receptor-associated kinase 1 (IRAK1), reducing MMP13 expression and collagen degradation, while other miRNAs such as miR-155 suppress mitogen-activated protein kinase (MAPK) signaling, lowering interleukin-1 beta (IL-1β) and interleukin-12 (IL-12) levels[110].
Additionally, Anx-N-EVs transport specialized pro-resolving mediators such as lipoxin A4 (LXA4), which activates ALX/FPR2 receptors on macrophages to inhibit ROS production and enhance IL-10 and TGF-β expression[111]. In infectious wound models, this is reflected by increased LXA4 and decreased prostaglandin E2 (PGE2) levels, promoting inflammation resolution[112]. Beyond immunomodulation, Anx-N-EVs reduce nicotinamide adenine dinucleotide phosphate (NADPH) oxidase activity and myeloperoxidase (MPO) release, thereby inhibiting neutrophil extracellular trap (NET) formation (NETosis). In sepsis models, they decrease circulating NETs and suppress IL-1β-dependent pyroptosis by reducing gasdermin D N-terminal (GSDMD-NT) cleavage, thereby alleviating systemic inflammation. They also deliver immunosuppressive molecules and promote regulatory T cell (Treg) expansion. In experimental autoimmune encephalomyelitis (EAE), Anx-N-EVs reduce Th17 cells and increase Tregs, mitigating central nervous system (CNS) inflammation, while surface human leukocyte antigen G (HLA-G) binds to killer-cell immunoglobulin-like receptors (KIRs) on natural killer (NK) cells to suppress cytotoxicity.
Engineered extracellular vesicle technologies
Gene modification strategies
Gene modification strategies enable precise regulation of gene expression in extracellular vesicle (EV) parent cells, enhancing targeting, cargo loading, and functional modulation[113]. Overexpression of specific miRNAs, such as miR-21-5p or miR-146a, significantly improves EV therapeutic efficacy. For instance, TNF-α-preconditioned mesenchymal stem cell-derived EVs (TG-exo) enriched with miR-21-5p inhibited apoptosis via programmed cell death protein 4 (PDCD4) and modulated microglial activation, thereby protecting retinal ganglion cells. Similarly, miR-146a-loaded EVs suppressed NF-κB signaling, alleviating synovial inflammation in rheumatoid arthritis. Regulation of transcription factors further enhances EV functionality. Hypoxia-inducible factor 1-alpha (HIF-1α) overexpression promotes angiogenesis and immune evasion through VEGF pathway activation, while SOX9-modified exosomes support cartilage repair via Wnt/β-catenin signaling, though excessive SOX9 may pose oncogenic risks[114,115].
Surface ligand modification
Surface engineering improves EV targeting by displaying ligands such as CD44-binding peptides and immune checkpoint regulators. CD44, highly expressed in tumors and inflamed tissues, facilitates EV homing; modifications via gene fusion (e.g., Lamp2b-CD44) or chemical conjugation enhance tissue-specific accumulation[116]. For example, CD44-modified EVs carrying temozolomide increased BBB permeability and suppressed glioblastoma growth[117,118]. Immune checkpoint molecules, including CD47 and programmed death-ligand 1 (PD-L1), extend circulation and enhance immune evasion. CD47-modified EVs inhibit phagocytosis, reducing inflammation, while PD-L1-modified EVs suppress T cell activation and increase tumor-infiltrating lymphocytes[119-122]. Dual-modified exosomes exhibit enhanced targeting and immune evasion, significantly inhibiting tumor growth in lung cancer models[123,124].
Hybrid nanomaterial systems
Hybrid systems integrate EVs with synthetic nanomaterials to augment delivery capacity[125]. Fusion of exosomes with liposomes or nanoparticles yields composite carriers with improved targeting and loading. For instance, doxorubicin-loaded liposomes fused with tumor-derived exosomes formed a CD44-targeted "Trojan Horse" system, enhancing drug penetration and antitumor efficacy[126,127]. Exosome-nanoparticle composites enable theranostic applications[128]. EGFR-targeted exosome-gold nanoparticles facilitate CT-guided photothermal therapy, promoting tumor apoptosis, while magnetic nanoparticles allow field-directed accumulation, boosting drug delivery[129]. Biomimetic modifications, such as lipid chain encoding in Lamp2b or viral protein fusion, improve stability and cellular uptake, enhancing delivery in diabetic and tumor models[130,131]. Multifunctional carriers combining pH-sensitive liposomes and targeting peptides enable selective drug release in acidic microenvironments, reducing recurrence[132-134].
MECHANISTIC INNOVATIONS IN SYNERGISTIC THERAPY
Multi-mechanism synergistic effects
Simultaneous activation of anti-inflammatory and regenerative processes
In osteoarthritis, exosome-diclofenac co-delivery suppresses MMP13 via miR-140-5p, promotes COL2A1 synthesis, and inhibits PGE2-mediated inflammation, thereby restoring cartilage thickness[135]. Conductive hydrogels incorporating exosomes and neurotrophic factors co-activate extracellular signal-regulated kinase 1/2 (ERK1/2), increasing NeuN+ neurons and myelin basic protein-positive (MBP+) myelin, and improving motor function in spinal cord injury models [Table 1][136]. These systems also modulate neuroinflammation by reducing ionized calcium-binding adapter molecule 1 (Iba1)+ microglia and glial fibrillary acidic protein (GFAP)+ astrocytes, and enhance synaptic plasticity via upregulation of synapsin-1 and postsynaptic density protein 95 (PSD-95)[137,138]. The exosome-drug-hydrogel MN system exerts therapeutic effects through synergistic anti-inflammatory and regenerative mechanisms. MSC exosomes deliver miR-21-5p and TGF-β to suppress NF-κB signaling, reduce pro-inflammatory cytokines, and promote M2 macrophage polarization, while anti-inflammatory agents such as curcumin inhibit cyclooxygenase-2 (COX-2)/inducible nitric oxide synthase (iNOS) and PGE2 production[139].
In diabetic wounds, a temporal release system (in which curcumin is rapidly released from the outer layer and exosomes are sustainedly released from the inner layer) reduces IL-6, increases VEGF and CD31+ vessel density, and activates the PI3K/AKT/mTOR pathway, thereby enhancing fibroblast migration, collagen deposition, and restoration of epidermal thickness [Table 1][140]. Engineered exosomes (e.g., miR-146a-enriched or CD44-targeted) further amplify these effects. For example, tumor necrosis factor-stimulated gene 6 (TSG-6)-modified exosomes incorporated into chitosan hydrogels reduce IL-1β, activate the Wnt/β-catenin pathway, and improve angiogenesis and LVEF in myocardial infarction[141].
Metabolic and epigenetic mechanisms further contribute to synergy. Exosomal miR-21-5p inhibits glycolysis via PDCD4, while drugs such as metformin activate AMPK to promote oxidative phosphorylation, forming an anti-inflammatory feedback loop[142,143]. In cancer, exosomes loaded with stimulator of interferon genes (STING) agonists activate cyclic GMP-AMP synthase (cGAS)-STING/type I interferon signaling and inhibit signal transducer and activator of transcription 3 (STAT3), reversing immunosuppression[144]. These strategies have demonstrated efficacy in chronic wounds, myocardial repair, and osteoarthritis. For example, exosome-antimicrobial peptide co-delivery reduces bacterial load and increases bone volume/total volume (BV/TV) in diabetic bone defects[145]. Clinical trials further support the utility of microneedles combined with exosomes and metformin in enhancing the healing of diabetic foot ulcers, increasing vessel density, and promoting collagen deposition. Furthermore, conductive hydrogels containing exosomes have been shown to improve LVEF and reduce fibrosis in patients with myocardial infarction[146].
Regulation of angiogenesis
Angiogenesis is central to tissue regeneration in ischemic or damaged contexts. MSC exosomes carry VEGF and FGF, activating the PI3K/AKT pathway to promote endothelial cell migration and lumen formation[147]. Modified exosomes (e.g., hypoxia-preconditioned or miR-21-overexpressing) further enhance VEGF secretion and angiogenic capacity[148]. Hydrogel MNs enable sustained exosome release. Core-shell designs allow rapid release of anti-inflammatory agents (e.g., curcumin) while maintaining sustained exosome release, increasing CD31+ vessel density in diabetic models[147]. Photothermal and magnetic responsive systems enable spatiotemporal control, with NIR irradiation triggering release and magnetic fields guiding delivery, thereby elevating VEGF concentration at target sites[149].
The synergistic effects of combined therapeutics enhance efficacy. For example, curcumin-loaded exosomes inhibit NF-κB and activate HIF-1α/VEGF, which improves LVEF in cases of myocardial infarction. Additionally, conductive hydrogels synchronize electrical stimulation with VEGF delivery, resulting in increased vessel density and maturity, as evidenced by the alpha-smooth muscle actin-positive (α-SMA+)/CD31+ ratio[150,151]. Smart hydrogels (ROS- or pH-sensitive) enable microenvironment-responsive release, promoting vascularization in wounds or inhibiting abnormal angiogenesis in tumors[152-154]. Preclinical studies have confirmed accelerated healing, reduced infection, and restoration of epidermal thickness in diabetic wounds, alongside improved LVEF and reduced fibrosis in myocardial repair[152]. Future work should clarify miRNA-drug metabolite interactions via multi-omics and optimize responsive materials for precise angiogenic modulation[155]. Additional mechanisms include immune-mediated angiogenesis, where exosomes containing IL-10 promote M2 macrophage polarization and the secretion of VEGF and TGF-β[156] [Figure 7]. In osteoarthritis, exosomes carrying insulin-like growth factor 1 (IGF-1) increase vessel density via the PI3K/AKT/mTOR pathway and suppress synovitis. Exosome-mediated transfer of mitochondrial DNA (mtDNA) enhances oxidative phosphorylation in endothelial cells, leading to increased vascular density and lumen diameter in ischemic models[157].
Figure 7. Hydrogel-exosome synergy for enhanced bone regeneration. The hydrogel scaffold, designed with a 3D microporous structure, serves as a carrier for bioactive molecules and cells. It is loaded with growth factors such as BMP-2, bFGF, and stem cells to stimulate bone healing. Exosomes are encapsulated within the hydrogel, carrying bioactive cargos including IL-10, miRNA, and PDGFA to target cells in a controlled manner to support osteoblast differentiation and promote bone matrix formation. Upon release, exosomes interact with macrophages, guiding their polarization toward the M2 phenotype, and with osteoblasts to promote osteogenesis and angiogenesis through VEGF and BMP signaling. Additionally, exosomes aid in restoring osteocalcin expression, and these exosome-driven pathways mitigate the negative impact of AGEs (advanced glycation end-products) on bone healing.
Metabolic reprogramming
Metabolic reprogramming bridges inflammation control and energy homeostasis to promote tissue repair. MSC exosomes deliver miR-21-5p to inhibit the PTEN/PI3K/AKT pathway, shifting macrophages from glycolysis (M1 phenotype) to oxidative phosphorylation (M2 phenotype), thereby increasing IL-10 and reducing TNF-α/IL-6[147]. This shift enhances fatty acid oxidation via PPAR-γ, supporting tissue repair.
In diabetic wounds, co-delivery of exosomes and metformin activates AMPK and inhibits mTOR, reducing glycolysis while enhancing oxidative phosphorylation (OXPHOS) in macrophages, decreasing cytokines and increasing TGF-β to accelerate closure[158]. Engineered exosomes can activate the mitochondrial unfolded protein response (UPRmt), upregulating Hsp60/Hsp10 and caseinolytic mitochondrial matrix peptidase proteolytic subunit (ClpP), enhancing mitophagy and OXPHOS[159]. In osteoarthritis, these effects restore mitochondrial membrane potential, increase adenosine triphosphate (ATP) production, and reduce IL-1β levels by inhibiting the pyrin domain-containing protein 3 (NLRP3) inflammasome[160].
Hypoxia-preconditioned exosomes activate HIF-1α and miR-210, enhancing VEGF expression and angiogenesis[148]. In myocardial infarction, this effect increases vessel density and reduces infarct size. Anti-infection strategies combine AMPs with exosomes to modulate macrophage metabolism[161]. Specifically, inhibition of M1-associated glycolysis and promotion of M2-associated arginine metabolism reduce bacterial load and enhance vascularization in Staphylococcus aureus infections [Table 1].
Metabolic-epigenetic crosstalk further enhances repair mechanisms, as exosomal miR-375 inhibits pyruvate dehydrogenase kinase 1 (PDK1)-mediated glycolysis and activates sirtuin 1 (SIRT1)-dependent mitochondrial biogenesis, thereby boosting OXPHOS and collagen synthesis during diabetic skin repair[162,163]. Preclinical data confirm efficacy in diverse models. For instance, exosomal miR-124-3p inhibits pyruvate kinase M2 (PKM2), reduces amyloid-beta (Aβ) deposition, and improves cognition in Alzheimer’s disease[164]. AMPK/SIRT1 activation promotes fatty acid oxidation, reduces fibrosis, and alleviates oxidative stress in liver disease[165].
CLINICAL APPLICATION CASES AND EFFICACY EVALUATION
Diabetic wound healing
Temporal release systems
Temporal release systems represent a key innovation in diabetic wound therapy, orchestrating anti-inflammatory and regenerative phases through smart material design. For example, a chitosan-based temperature-sensitive hydrogel crosslinked with β-glycerophosphate forms a 3D network that gels rapidly at body temperature, enabling minimally invasive injection. These systems, typically structured as bilayers or core-shells, rapidly release anti-inflammatory agents from the outer layer to curb early inflammation, while the inner layer provides sustained exosome release to foster tissue regeneration[44]. The porous structure efficiently loads exosomes and controls release kinetics via swelling[166]. In diabetic wound models, this system increased neovascularization, promoted collagen deposition, and restored epidermal thickness to normal levels[167]. Composite hydrogels allow for mechanical and responsive tuning. F127 forms a semi-solid gel for exosome retention, while GelMA achieves photocrosslinked porosity and ROS-responsive degradation, enhancing release rates and balancing inflammation-repair dynamics[168]. Functional modifications such as RGD peptides or ROS-sensitive linkers further enhance performance; RGD-GelMA microneedles increased fibroblast adhesion and collagen synthesis, while phenylboronic ester linkers enabled faster ROS-triggered drug release[169]. Preclinical data confirm that temporal release systems reduce healing time and infection rates in diabetic wounds. Multicenter clinical trials have demonstrated that chitosan MNs loaded with MSC exosomes and curcumin significantly shorten wound closure time, promote neovascularization, enhance collagen deposition, and restore near-normal epidermal thickness[155]. Mechanistically, curcumin inhibits NF-κB phosphorylation, reducing IL-6 and TNF-α, while exosomal miR-21-5p activates the PI3K/AKT pathway, enhancing VEGF secretion and collagen type I alpha 1 (COL1A1) expression[170].
Smart-responsive systems integrate photothermal/magnetic control mechanisms. Exosomes modified with gold nanoparticles enable NIR-triggered release, while magnetic fields guide delivery to increase drug concentration at target sites[70]. Dual pH/enzyme-responsive microspheres and MMP-sensitive layers allow deep penetration and staged release, while SilMA microneedles with barbed tips ensure stable dermal retention[53,71]. In diabetic rats, insulin and MSC-derived exosome co-loaded microneedles improved wound closure and neovascularization compared with single-drug treatments. Matching the phase transition temperature (Tm) to the wound site enhanced dermal drug concentration, and the system sustained controlled exosome release while maintaining skin-compatible elasticity[171].
Recent studies have further optimized material and structural design. A chitosan/silk composite hydrogel loaded with hypoxia-preconditioned MSC exosomes reversed macrophage dysfunction via the miR-4645-5p/SREBP2 axis, improving diabetic foot ulcer healing[172]. 3D-printed peptide DNA microneedles with engineered exosomes simultaneously cleared ROS, regulated the immune microenvironment, and promoted neuroregeneration, enabling full-thickness skin regeneration[173]. In LepRdb/db mice, a Q5Exo hydrogel with exosomes shortened wound closure time comparably to subcutaneous injection[174]. Autofluorescence enabled monitoring of delivery, avoiding repeated injections, and histological analysis showed enhanced re-epithelialization and type II collagen expression.
Other systems also demonstrated efficacy, with radial-structured microneedles containing adipose tissue-derived exosomes enhancing wound healing[175], and composite hydrogels loaded with disulfiram-containing exosomes shortening healing time, increasing epidermal thickness and vascular density, boosting M2 macrophage infiltration, and decreasing IL-1β[176,177]. Marine-derived exosome (MENV) hydrogels restored epidermal thickness, normalized collagen volume, downregulated NLRP3/caspase-1, restored near-normal white blood cell (WBC) levels, and reversed T helper 2 (Th2) infiltration, with concurrent hair follicle regeneration in infected diabetic wounds[174,178].
In a Staphylococcus aureus-infected full-thickness rat skin defect model, the OD/GM/QCS@Exo hydrogel (prepared with catechol-modified oxidized hyaluronic acid (OD), methacrylated gelatin (GM), and quaternized chitosan (QCS) and loaded with adipose mesenchymal stem cell-derived exosomes (Exos)) achieved rapid and complete wound closure. Histological analysis showed that epithelial thickness and the collagen deposition rate reached healthy skin levels, accompanied by increased CD31+ vascular density[179]. This system inhibits bacterial proliferation and early inflammatory storms while sequentially releasing exosomes and QCS to activate angiogenesis and ECM reconstruction, confirming its clinical translational potential[180].
Recent preclinical studies indicate that a multifunctional hydrogel integrating MnO2/ε-poly-L-lysine (ε-PL) nanosheets, fibroblast growth factor 2 (FGF2), and M2-derived exosomes (M2-Exos) significantly promotes chronic wound healing via multi-component synergy, with MnO2 catalyzing H2O2 decomposition to alleviate oxidative stress, FGF2 accelerating re-epithelialization (1.8-fold increase in epithelial coverage), and exosomes promoting neovascularization, as evidenced by an increased CD31+ area[178,180]. In diabetic rat models with full-thickness skin defects, exosome-loaded microneedles achieved high wound closure rates quickly, surpassing blank controls and exosome-only groups. Histological analysis further revealed increased granulation tissue thickness and improved collagen fiber alignment[181]. Collectively, these smart, synergistic systems show strong translational potential for diabetic wound repair[182]. Future research should focus on optimizing microenvironment responsiveness via multi-omics and intelligent materials, overcoming delivery barriers, and standardizing the production process to address exosome heterogeneity.
Anti-infection synergy
Anti-infection combination therapy for diabetic wounds achieves efficient sterilization and tissue repair through integrated mechanisms. A hydrogel microneedle system loaded with AMPs and MSC exosomes inhibits biofilm formation and modulates the immune response. The peptides disrupt bacterial membranes, while the exosomes deliver miR-21-5p and TGF-β1 to promote M2 polarization, angiogenesis, and collagen remodeling[183]. Responsive hydrogels enable targeted delivery. ROS/pH-sensitive systems degrade in response to elevated H₂O₂, releasing exosomes and insulin to lower glucose and inflammation. In diabetic rats, this approach achieved rapid wound closure, with reduced infection and enhanced epidermal thickness[184]. Nanomaterials further enhance this synergistic effect. A chitosan hydrogel with silver nanoparticles (AgNPs) and MSC exosomes combines broad-spectrum antibacterial activity with immunomodulatory effects, reducing bacterial load, increasing VEGF expression, and enhancing fibroblast migration[185]. Photodynamic therapy-exosome combinations also show efficacy. A thermosensitive hydrogel containing porphyrin photosensitizers and MSC exosomes generates ROS under laser irradiation to kill bacteria, while exosomal miR-126-3p activates PI3K/AKT to promote endothelial lumen formation[186]. In diabetic foot ulcers, this strategy shortened healing time versus traditional therapy, without significant scarring[187]. Preclinical studies have demonstrated that calcium alginate-based composite hydrogels with platelet exosomes (pEVs) and gentamicin synergize through pEV immunomodulation and antibiotic-mediated bactericidal effects, reducing wound infection and accelerating epithelialization. This system downregulates TNF-α and IL-6 by inhibiting NF-κB and activates STAT3 to promote epidermal stem cell proliferation[188]. Intelligent DNA hydrogels degrade in response to tumor-associated MMPs, releasing exosomes and CpG oligodeoxynucleotides (CpG ODNs) to achieve antibacterial effects and immune activation in diabetic infection models[189]. Flow cytometry analysis showed an increased proportion of M2 macrophages, higher CD31+ vascular density, and near-normal collagen deposition[190]. Material design innovations include oxidized sodium alginate hydrogels with MENVs and ε-polylysine. MENV’s natural anti-inflammatory components (e.g., vitamin C, flavonoids) synergize with ε-polylysine’s antibacterial activity, reducing bacterial colonization and promoting M2 polarization (as indicated by an increased CD206+ cell proportion). In diabetic mice, this system achieved significant wound closure and near-normal hair follicle regeneration[191]. In summary, anti-infection synergy integrates bacterial eradication, exosome-mediated immune modulation, controlled release via responsive materials, and nanoparticle-mediated localized delivery to reduce systemic toxicity[192]. Future research should combine engineered exosomes with intelligent materials to improve targeting and outcomes[193].
Myocardial infarction repair
Conductive hydrogels enhance electrical signal transmission
Conductive hydrogels mimic native myocardial electrophysiology, facilitating electrical signal propagation and functional recovery post-infarction. When integrated with exosome delivery, they significantly enhance cardiac repair and angiogenesis. For instance, a polypyrrole-gelatin (PPy-Gel) hydrogel exhibits cardiomyocyte-like conductivity, enabling efficient action potential transmission[194]. Loaded with MSC exosomes, it accelerates conduction velocity in the infarct zone and reduces the incidence of arrhythmias in myocardial infarction models[195]. Mechanistically, the conductive hydrogel promotes cardiomyocyte adhesion and differentiation via integrin β1/ focal adhesion kinase (FAK), while exosomal miR-133a inhibits the expression of fibrotic genes[196].
Regulation of the immune microenvironment
Regulation of the immune microenvironment balances myocardial repair and scar formation by promoting M2 macrophage polarization and inhibiting fibrosis. Exosomes deliver miR-146a-5p and TGF-β1 to synergistically induce M2 polarization[197]. Flow cytometry shows increased CD206+ macrophages and IL-10 secretion in infarcted areas[198]. miR-146a-5p inhibits NF-κB by targeting TRAF6/IRAK1, reducing TNF-α[199]. Co-delivery of anti-fibrotic drugs and exosomes effectively inhibits TGF-β/Smad signaling. Masson’s trichrome staining reveals reduced fibrotic areas and collagen deposition[200]. Immunohistochemistry shows decreased α-SMA+ fibroblasts and increased VEGF expression, promoting angiogenesis[201].
Bone and cartilage regeneration
Double-layer 3D-printed scaffolds
Double-layer 3D-printed scaffolds replicate the osteochondral interface, providing mechanical and biochemical gradients for tissue-specific regeneration. The upper cartilage layer (GelMA, black phosphorus, human umbilical cord mesenchymal stem cell (hUCMSC)-derived exosomes) delivers exosomal TGF-β1 and miR-140-3p to upregulate SOX9 and inhibit MMP13, facilitating chondrogenesis. The lower osteogenic layer (β-tricalcium phosphate (β-TCP), black phosphorus, exosomes) promotes bone formation via phosphate-mediated bone morphogenetic protein-2 (BMP-2)/Runt-related transcription factor 2 (RUNX2) activation, elevating alkaline phosphatase (ALP) activity and mineral deposition[202,203] [Figure 7]. This biomimetic gradient alleviates interfacial shear stress, guides MSC differentiation, and exerts immunomodulatory effects, shifting macrophages toward M2 phenotype[204]. In vivo, full defect closure was observed at 6 weeks. Micro-CT showed increased bone volume fraction, and magnetic resonance imaging (MRI) T2 mapping indicated regenerated cartilage matching native tissue[205]. Histological analysis revealed an orderly collagen matrix. The scaffold accurately matched defect shapes and enabled personalized regeneration via spatiotemporal exosome-phosphate regulation[206,207].
Autophagy activation strategy
Autophagy activation enhances osteochondral regeneration by regulating cellular metabolism, inflammation, and immune responses. Exosomes from activating transcription factor 5 (ATF5)-overexpressing MSCs activate UPRmt, promoting autophagy, suppressing IL-1β-induced cytokines, and improving ECM homeostasis by upregulating Col2A1 and downregulating MMP13. In osteoarthritis, Gel@Exo-ATF5-modified hydrogel restored cartilage thickness and reduced OARSI scores[208]. In diabetic bone defects, Silk-6/ε-PL@Exo hydrogels activate the hexokinase 2 (HK2)-NF-κB pathway to stimulate osteogenesis, promoting vascularization and immunomodulation [Figure 8]. Exosomal miR-21-5p targets multiple signaling pathways (PDCD4/NF-κB, PTEN/PI3K/AKT, TGFBR2/Smad), promoting tube formation, reducing fibrosis, and inducing M2 macrophage polarization[209]. Synergistic strategies further enhance efficacy. AMP-RAD/EXO hydrogels provide antibacterial protection and facilitate exosome-stimulated osteogenesis, significantly increasing bone volume fraction, trabecular thickness, and trabecular number[210]. Sequential delivery systems, such as curcumin-loaded hUCMSC-derived exosome hydrogels, reduce IL-6 and induce mitophagy via miR-378/PDK1, improving collagen organization. Adipose-derived MSC exosomes (AMSC-EVs) in Pluronic F127 hydrogel promote osteogenesis via protein kinase R-like endoplasmic reticulum kinase (PERK) and activating transcription factor 4 (PERK/ATF4) activation and modulate immune responses by reducing CD86 expression (M1) while increasing CD206 expression (M2)[211].
Figure 8. Hydrogel-exosome synergy in stem cell regulation for bone regeneration. Hydrogel contribution: Hydrogels provide critical mechanical cues such as stiffness, porosity, and topography that influence stem cell attachment and differentiation. They facilitate exosome integration by mimicking the extracellular matrix (ECM), supporting cell-material interactions essential for osteogenesis and angiogenesis. Furthermore, hydrogels enable the controlled release of molecules such as BMP-2 and bFGF, which enhance bone formation. Exosome Contribution: Exosomes play a crucial role in the regulation of key molecular factors, including miRNAs such as miR-140-3p, miR-375, and miR-1260a, which modulate osteogenesis and angiogenesis. They also activate essential pathways such as PI3K, AKT, MAPK/ERK, and VEGF signaling to promote endothelial migration, osteoblast function, and vascularization. Molecular Regulators: The interactions between hydrogel and exosome systems, particularly in the activation of PI3K/AKT and MAPK/ERK pathways, support osteoblast survival, migration, and differentiation, while promoting angiogenesis and immune modulation. Impact on Bone Regeneration: The combined hydrogel-exosome system significantly enhances bone regeneration by supporting osteoblast differentiation, improving vascularization, and regulating immune responses, providing an integrated solution for effective bone tissue engineering.
CONCLUSION
Challenges in hydrogel microneedles-exosome therapy
Despite promising preclinical results, the clinical translation of hydrogel MN-exosome therapies faces challenges in manufacturing scalability, mechanical durability, and long-term safety [Figure 9 and 10]. Exosome production suffers from batch heterogeneity and low yield; although microfluidic isolation shows promise, standardized mass production remains difficult[212]. Engineering MNs that reliably penetrate pathological tissues requires innovations in materials and structural design[213]. Safety concerns include immunogenic responses to allogeneic exosomes, biological impacts of hydrogel degradation byproducts, and the uncertain long-term fate and function of exosomal cargo in vivo[214,215]. Addressing these challenges requires rigorous large-animal studies and standardized regulatory frameworks.
Figure 9. Preclinical validation and clinical translation bottlenecks in hydrogel-exosome bone and nerve regeneration therapy. (A) Exosome-Mediated Vascular and Bone Repair: Exosomes carrying miRNAs play a vital role in promoting vascular repair and bone regeneration. Delivery of exosomes loaded with therapeutic miRNAs enhances vascular regeneration and bone healing by stimulating endothelial cells and osteoblast differentiation. (B) Multimodal Imaging and Analytical Techniques: This panel illustrates the use of advanced imaging technologies, such as photoacoustic imaging and single-cell sequencing, to track and monitor the distribution and effects of hydrogel-exosome systems in animal models. These techniques enable high-resolution monitoring of tissue regeneration processes in vivo. Additionally, the figure highlights the dual-therapeutic potential of this system in promoting both bone healing and nerve regeneration. Specifically, in vivo animal models reveal that internalized exosomes can effectively reduce intracellular ROS levels to mitigate oxidative stress, while also facilitating neural cell differentiation for nerve regeneration. (C) Exosome Isolation Methods: Exosomes are isolated from biological samples using microfluidics, ultracentrifugation, ultrafiltration membranes, immunoseparation, or polymer precipitation. These methods ensure high yield and purity of exosomes for therapeutic applications, which are essential for their effective loading into hydrogels and subsequent delivery to target bone defects.
Figure 10. Challenges and evaluation strategies for hydrogel microneedle (MN)-exosome in bone regeneration. This figure outlines the animal models commonly employed to evaluate the efficacy of these therapies, including both small animal models, such as diabetic rat femoral defects, and large animal models, such as goat spinal fusion models. Small animal models are cost-effective but may not fully replicate human healing processes, while large animals offer better anatomical relevance but involve higher costs and longer study durations. The figure also highlights various evaluation techniques, including micro-CT for 3D bone volume and structure assessment, photoacoustic imaging for real-time monitoring of vascularization, and single-cell sequencing for detailed cellular analysis, offering insights into the effects of exosomes on osteoblasts and immune cells. Additionally, the figure addresses challenges in the clinical translation of hydrogel MN-exosome therapies. Issues such as the standardization of exosome sources and isolation methods, hydrogel degradation leading to inflammatory byproducts, mechanical durability affecting tissue penetration, and potential tumor risks associated with exosomal cargo, such as oncogenic miRNAs, need to be overcome. Furthermore, regulatory uncertainty remains a significant hurdle, as no universally accepted classification currently exists for exosome-hydrogel combination products. To address this, regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have begun issuing draft guidance documents outlining expectations for exosome-based therapies, focusing on source characterization, manufacturing consistency, and safety profiling. Leveraging such guidance can help developers align preclinical and clinical development strategies. Moreover, the absence of standardized efficacy validation protocols also impedes clinical translation. Future directions should include the development of consensus guidelines for potency assays, long-term safety studies, and clinical trial design, potentially coordinated through international consortia or working groups. These efforts, combined with advancements in manufacturing and quality control, are essential for the safe and effective clinical translation of hydrogel-exosome therapies.
Future directions of hydrogel microneedles-exosome therapy
Future breakthroughs should integrate CRISPR-based gene editing with patient-specific exosome libraries for therapeutic precision, combine photothermal- and magnetic-responsive systems for spatiotemporal release, and develop artificial intelligence (AI)-driven quality control to optimize drug-exosome ratios and production parameters [Figure 11]. Key barriers to clinical translation include low recovery rates, high costs, and the lack of standardized quality-control frameworks for exosomes[216]. Robust models based on miRNA content are needed to improve batch consistency and predict outcomes. Multi-omics analysis must clarify the synergistic effects of combination therapies to support regulatory approval[215]. Interdisciplinary technologies merging molecular biology, materials science, and AI are crucial. Integrating real-time biosensing with microneedles will enable dynamic, patient-specific treatment adjustments Furthermore, advanced concepts such as cascade exosome signaling and organ-on-a-chip models will be critical for future personalized screening and therapeutic amplification [Figure 11]. Prioritizing high-throughput exosome sorting, personalized formulations, and advanced microneedle engineering will help overcome patient heterogeneity, especially in bone regeneration and related orthopedic disorders. Machine learning and biosensing can further optimize therapeutic efficacy and safety [Figure 12]. In summary, hydrogel MNs loaded with exosomes and drugs enhance efficacy via minimally invasive, localized, and sustained delivery, promoting exosome-mediated targeting and regeneration. This approach improves wound healing, reduces inflammation, and enhances tissue regeneration compared with monotherapies in diabetic wounds and myocardial infarction. By addressing low bioavailability and systemic side effects, it advances precision medicine. Technological innovations and interdisciplinary collaboration improve bench-to-bedside feasibility through AI personalization, smart microneedles, and biosensing[217]. Future work must elucidate molecular mechanisms to support regulatory approval and predict patient-specific outcomes, ultimately delivering novel therapies for diabetic wounds, myocardial infarction, and osteoarthritis.
Figure 11. Advanced intelligent designs and personalized strategies for hydrogel-exosome-based bone regeneration. Advanced Intelligent Designs include two systems: the Closed-Loop Feedback system, which modulates exosome release based on glucose levels using smart sensors and hydrogels, tailored for diabetic patients but facing sensor accuracy and material stability challenges; and the Cascade Exosome Signaling system, where primary exosomes release secondary exosomes to boost angiogenesis and osteogenesis, though stability and scalability need to be improved. Personalized Strategies feature a patient-specific bioink approach, combining autologous MSC-derived exosomes with 3D-printed hydrogels for better biocompatibility, though challenges with MSC differentiation remain. Organ-on-a-Chip Models can simulate bone defects for personalized screening, but struggle with construction complexity and environment simulation. The Clinical Translation Roadmap outlines three stages: Short-term (1-3 years) for standardization and Phase I trials, Mid-term (≈5 years) for therapy combinations and expanded indications, and Long-term (≈10 years) for AI-driven designs. Challenges include batch variability, optimization, and data integration, emphasizing the innovations, strategies, and obstacles in advancing hydrogel-exosome systems.
Figure 12. Smart exosome-hydrogel system and personalized treatment strategy for bone regeneration. This figure illustrates a comprehensive strategy combining a smart exosome-hydrogel system with a personalized treatment approach for bone regeneration. On the left, the Smart Exosome-Hydrogel System is depicted. It shows the release of exosomes, activated by specific stimuli, to promote bone regeneration by targeting osteoblast activation while incorporating blood glucose monitoring for adaptive therapeutic responses. The system aims to improve bone healing through controlled, on-demand exosome release. On the right, the Personalized Treatment Strategy is outlined, emphasizing the use of 3D printing, microfluidic chips, and artificial intelligence (AI) for individualized care. The 3D printing approach customizes scaffolds for precise bone healing, while microfluidic chips facilitate dynamic testing of different treatments. AI is employed to analyze patient-specific data and optimize therapy for effective bone regeneration, ensuring treatment is tailored to the unique needs of each patient.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study, manuscript drafting, and critical revision: Shi, H.; Chen, Y.; Chen, M.
Performed data collection, literature review, and data analysis: Huang, Z.; Shen, C.; Xiao, J.; Zhao, H.
Assisted in figure preparation, technical support, and data verification: Zou, Z.; Zhang, W.; Feng, K.; Ou, Z.
Provided resources, administered the project, and supervised experimental protocols: Jia, L.; Wang, Q.; Li, B.
Contributed to manuscript editing and provided institutional support: Shi, L.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (No. 82101956), the Science and Technology Development Fund of Macau Special Administrative Region (Nos. 0114/2022/A, 0018/2024/RIA1), the High-Level Key Discipline Construction Project of the State Administration of Traditional Chinese Medicine (zyyzdxk-2023123), the Integration Development Strategy Project of Jinan City and Colleges (JNSX2024046), the Clinical-Basic Joint Innovation Team Project of Shandong First Medical University (CX202408), the Natural Science Foundation of Shandong Province (ZR2024MH189), the University Youth Innovation Team of Shandong Province (2023KJ176), the Young Talent Lifting Project for Science and Technology in Shandong Province (SDAST2024QTA044), the Shandong Province Traditional Chinese Medicine Science and Technology Project (M-2023106, M-2022253), and the Medicine and Health Science and Technology Project of Shandong Province (202404070291). The literature review and related research were also supported by the National Social Science Fund of China (Grant No. 0059082101, Project No. 23XTY007: "A Study on the Scientific Literacy Promotion Program for Silver-Aged Physical Education").
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Golebiowska, A. A.; Intravaia, J. T.; Sathe, V. M.; Kumbar, S. G.; Nukavarapu, S. P. Decellularized extracellular matrix biomaterials for regenerative therapies: advances, challenges and clinical prospects. Bioact. Mater. 2024, 32, 98-123.
2. Xu, M.; Su, T.; Jin, X.; et al. Inflammation-mediated matrix remodeling of extracellular matrix-mimicking biomaterials in tissue engineering and regenerative medicine. Acta. Biomater. 2022, 151, 106-17.
3. Majumder, J.; Taratula, O.; Minko, T. Nanocarrier-based systems for targeted and site specific therapeutic delivery. Adv. Drug. Deliv. Rev. 2019, 144, 57-77.
4. Zhao, Z.; Ukidve, A.; Kim, J.; Mitragotri, S. Targeting strategies for tissue-specific drug delivery. Cell 2020, 181, 151-67.
5. Hannafon, B. N.; Ding, W. Q. Intercellular communication by exosome-derived microRNAs in cancer. Int. J. Mol. Sci. 2013, 14, 14240-69.
6. Bhatnagar, D.; Simon, M.; Rafailovich, M. H. Hydrogels for regenerative medicine. In: Parveen FK, editors. Recent advances in biopolymers. InTech; 2016.
7. Chen, Y.; Wang, X.; Tao, S.; et al. Research advances in smart responsive-hydrogel dressings with potential clinical diabetic wound healing properties. Mil. Med. Res. 2023, 10, 37.
8. Liu, M.; Zeng, X.; Ma, C.; et al. Injectable hydrogels for cartilage and bone tissue engineering. Bone. Res. 2017, 5, 17014.
9. Zhao, X.; Lang, Q.; Yildirimer, L.; et al. Photocrosslinkable gelatin hydrogel for epidermal tissue engineering. Adv. Healthc. Mater. 2016, 5, 108-18.
10. Levingstone, T. J.; Matsiko, A.; Dickson, G. R.; O'Brien, F. J.; Gleeson, J. P. A biomimetic multi-layered collagen-based scaffold for osteochondral repair. Acta. Biomater. 2014, 10, 1996-2004.
11. Manferdini, C.; Cavallo, C.; Grigolo, B.; et al. Specific inductive potential of a novel nanocomposite biomimetic biomaterial for osteochondral tissue regeneration. J. Tissue. Eng. Regen. Med. 2016, 10, 374-91.
12. Jella, K. K.; Nasti, T. H.; Li, Z.; Malla, S. R.; Buchwald, Z. S.; Khan, M. K. Exosomes, their biogenesis and role in inter-cellular communication, tumor microenvironment and cancer immunotherapy. Vaccines 2018, 6, 69.
13. Liu, J.; Ren, L.; Li, S.; et al. The biology, function, and applications of exosomes in cancer. Acta. Pharm. Sin. B. 2021, 11, 2783-97.
14. Wu, J.; Chen, L. H.; Sun, S. Y.; Li, Y.; Ran, X. W. Mesenchymal stem cell-derived exosomes: the dawn of diabetic wound healing. World. J. Diabetes. 2022, 13, 1066-95.
15. Wang, X.; Chen, Y.; Zhao, Z.; et al. Engineered exosomes with ischemic myocardium-targeting peptide for targeted therapy in myocardial infarction. J. Am. Heart. Assoc. 2018, 7, e008737.
16. Hu, S.; Qiao, L.; Cheng, K. Generation and manipulation of exosomes. Methods. Mol. Biol. 2021, 2158, 295-305.
17. Liu, R. H.; Ning, B.; Ma, X. E.; Gong, W. M.; Jia, T. H. Regulatory roles of microRNA-21 in fibrosis through interaction with diverse pathways (Review). Mol. Med. Rep. 2016, 13, 2359-66.
18. Lv, Q.; Deng, J.; Chen, Y.; Wang, Y.; Liu, B.; Liu, J. Engineered human adipose stem-cell-derived exosomes loaded with miR-21-5p to promote diabetic cutaneous wound healing. Mol. Pharm. 2020, 17, 1723-33.
19. Li, D.; Zhang, C.; Gao, Z.; et al. Curcumin-loaded macrophage-derived exosomes effectively improve wound healing. Mol. Pharm. 2023, 20, 4453-67.
20. Puthia, M.; Butrym, M.; Petrlova, J.; et al. A dual-action peptide-containing hydrogel targets wound infection and inflammation. Sci. Transl. Med. 2020, 12, eaax6601.
21. Selvadoss, A.; Baby, H. M.; Zhang, H.; Bajpayee, A. G. Harnessing exosomes for advanced osteoarthritis therapy. Nanoscale 2024, 16, 19174-91.
22. Garbern, J. C.; Minami, E.; Stayton, P. S.; Murry, C. E. Delivery of basic fibroblast growth factor with a pH-responsive, injectable hydrogel to improve angiogenesis in infarcted myocardium. Biomaterials 2011, 32, 2407-16.
23. Li, L.; Xiao, B.; Mu, J.; et al. A MnO2 Nanoparticle-dotted hydrogel promotes spinal cord repair via regulating reactive oxygen species microenvironment and synergizing with mesenchymal stem cells. ACS. Nano. 2019, 13, 14283-93.
24. Chen, J.; Pan, C.; Gao, Y.; Chen, Q.; An, X.; Liu, Z. Reactive oxygen species scavenging injectable hydrogel potentiates the therapeutic potential of mesenchymal stem cells in skin flap regeneration. ACS. Appl. Mater. Interfaces. 2024, 16, 17120-8.
25. Kwak, G.; Cheng, J.; Kim, H.; et al. Sustained exosome‐guided macrophage polarization using hydrolytically degradable PEG Hydrogels for cutaneous wound healing: identification of key proteins and MiRNAs, and sustained release formulation. Small 2022, 18, 2200060.
26. Fan, M.; Pi, J.; Zou, C.; et al. Hydrogel-exosome system in tissue engineering: a promising therapeutic strategy. Bioact. Mater. 2024, 38, 1-30.
27. Du, S.; Zhou, X.; Zheng, B. Beyond traditional medicine: EVs-loaded hydrogels as a game changer in disease therapeutics. Gels 2024, 10, 162.
28. Cao, J.; Wu, B.; Yuan, P.; Liu, Y.; Hu, C. Advances in research of hydrogel microneedle-based delivery systems for disease treatment. Pharmaceutics 2024, 16, 1571.
29. Nelson, B. C.; Maragh, S.; Ghiran, I. C.; et al. Measurement and standardization challenges for extracellular vesicle therapeutic delivery vectors. Nanomed 2020, 15, 2149-70.
30. Li, S.; Tian, X.; Fan, J.; Tong, H.; Ao, Q.; Wang, X. Chitosans for tissue repair and organ three-dimensional (3D) bioprinting. Micromachines 2019, 10, 765.
31. Liu, X.; Ma, L.; Mao, Z.; Gao, C. Chitosan-based biomaterials for tissue repair and regeneration. In Chitosan for biomaterials II. Berlin, Heidelberg: Springer Berlin Heidelberg; 2011. pp. 81-127.
32. Wang, C.; Liang, C.; Wang, R.; et al. The fabrication of a highly efficient self-healing hydrogel from natural biopolymers loaded with exosomes for the synergistic promotion of severe wound healing. Biomater. Sci. 2020, 8, 313-24.
33. Yue, K.; Trujillo-de, Santiago. G.; Alvarez, M. M.; Tamayol, A.; Annabi, N.; Khademhosseini, A. Synthesis, properties, and biomedical applications of gelatin methacryloyl (GelMA) hydrogels. Biomaterials 2015, 73, 254-71.
34. Hassannejad, Z.; Zadegan, S. A.; Vaccaro, A. R.; Rahimi-Movaghar, V.; Sabzevari, O. Biofunctionalized peptide-based hydrogel as an injectable scaffold for BDNF delivery can improve regeneration after spinal cord injury. Injury 2019, 50, 278-85.
35. Yu, Z.; Li, Q.; He, X.; et al. A multifunctional hydrogel based on nature polysaccharide fabricated by Schiff base reaction. Eur. Polym. J. 2023, 197, 112330.
36. Singh, R.; Malhotra, H.; Jadhav, K.; et al. Intelligently actuating dual-barrier hyaluronic acid-functionalized inflammation-responsive nanohydrogel for targeted rheumatoid arthritis therapy. ACS. Appl. Mater. Interfaces. 2025, 17, 40012-34.
37. Jaquilin, P. J. R.; Oluwafemi, O. S.; Thomas, S.; Oyedeji, A. O. Recent advances in drug delivery nanocarriers incorporated in temperature-sensitive Pluronic F-127-A critical review. J. Drug. Deliv. Sci. Technol. 2022, 72, 103390.
38. Shriky, B.; Kelly, A.; Isreb, M.; et al. Pluronic F127 thermosensitive injectable smart hydrogels for controlled drug delivery system development. J. Colloid. Interface. Sci. 2020, 565, 119-30.
39. O'connell, C. D.; Zhang, B.; Onofrillo, C.; et al. Tailoring the mechanical properties of gelatin methacryloyl hydrogels through manipulation of the photocrosslinking conditions. Soft. Matter. 2018, 14, 2142-51.
40. Piccolo, L.; Bornillo, K.; Micheli, S.; et al. A penetration efficiency model for the optimization of solid conical microneedles’ geometry. J. Micromech. Microeng. 2024, 34, 025009.
41. Abdullah, A. C.; Ahmadinejad, E.; Tasoglu, S. Optimizing solid microneedle design: a comprehensive ML-augmented DOE approach. ACS. Meas. Sci. Au. 2024, 4, 504-14.
42. Grenier, J.; Duval, H.; Lv, P.; et al. Interplay between crosslinking and ice nucleation controls the porous structure of freeze-dried hydrogel scaffolds. Biomater. Adv. 2022, 139, 212973.
43. Grenier, J.; Duval, H.; Barou, F.; Lv, P.; David, B.; Letourneur, D. Mechanisms of pore formation in hydrogel scaffolds textured by freeze-drying. Acta. Biomater. 2019, 94, 195-203.
44. Sun, D.; Zhuang, X.; Xiang, X.; et al. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol. Ther. 2010, 18, 1606-14.
45. Perez, R. A.; Kim, H. Core-shell designed scaffolds for drug delivery and tissue engineering. Acta. Biomater. 2015, 21, 2-19.
46. Gupta, D.; Singh, A. K.; Bellare, J. Natural bone inspired core-shell triple-layered gel/PCL/gel 3D printed scaffolds for bone tissue engineering. Biomed. Mater. 2023, 18, 065027.
47. Qi, H.; Wang, B.; Wang, M.; Xie, H.; Chen, C. A pH/ROS-responsive antioxidative and antimicrobial GelMA hydrogel for on-demand drug delivery and enhanced osteogenic differentiation in vitro. Int. J. Pharm. 2024, 657, 124134.
48. Allen, Jr. RJ, Soares MA, Haberman ID, et al. Combination therapy accelerates diabetic wound closure. PLoS. ONE. 2014, 9, e92667.
49. Wang, Y.; Wu, Y.; Long, L.; et al. Inflammation-responsive drug-loaded hydrogels with sequential hemostasis, antibacterial, and anti-inflammatory behavior for chronically infected diabetic wound treatment. ACS. Appl. Mater. Interfaces. 2021, 13, 33584-99.
50. Wang, C.; Wang, M.; Xu, T.; et al. Engineering bioactive self-healing antibacterial exosomes hydrogel for promoting chronic diabetic wound healing and complete skin regeneration. Theranostics 2019, 9, 65-76.
51. Zhang, X.; Liu, Y.; Wang, Z.; et al. pH-responsive and self-adaptive injectable sodium alginate/carboxymethyl chitosan hydrogel accelerates infected wound healing by bacteriostasis and immunomodulation. Carbohydr. Polym. 2025, 354, 123322.
52. Zhao, H.; Huang, J.; Li, Y.; et al. ROS-scavenging hydrogel to promote healing of bacteria infected diabetic wounds. Biomaterials 2020, 258, 120286.
53. Han, D.; Morde, R. S.; Mariani, S.; et al. 4D printing of a bioinspired microneedle array with backward‐facing barbs for enhanced tissue adhesion. Adv. Funct. Mater. 2020, 30, 1909197.
54. Detamornrat, U.; Mcalister, E.; Hutton, A. R. J.; Larrañeta, E.; Donnelly, R. F. The role of 3D printing technology in microengineering of microneedles. Small. 2022, 18, 2106392.
55. Li, R.; Zhang, L.; Jiang, X.; et al. 3D-printed microneedle arrays for drug delivery. J. Controlled. Release. 2022, 350, 933-48.
56. Zhou, X.; Esworthy, T.; Lee, S.; et al. 3D printed scaffolds with hierarchical biomimetic structure for osteochondral regeneration. Nanomed. Nanotechnol. Biol. Med. 2019, 19, 58-70.
57. Ren, Y.; Zhang, C.; Liu, Y.; et al. Advances in 3D printing of highly bioadaptive bone tissue engineering scaffolds. ACS. Biomater. Sci. Eng. 2023, 10, 255-70.
58. Sharun, K.; Banu, S. A.; Mamachan, M.; et al. Thermoresponsive and injectable pluronic F127 hydrogel for loading adipose-derived mesenchymal stem cells. Discov. Med. 2024, 36, 294.
59. Ozkan, O.; Sasmazel, H. T. Antibacterial performance of PCL-chitosan core-shell scaffolds. J. Nanosci. Nanotechnol. 2018, 18, 2415-21.
60. Guvendiren, M.; Lu, H. D.; Burdick, J. A. Shear-thinning hydrogels for biomedical applications. Soft. Matter. 2012, 8, 260-72.
61. Zeitter, S.; Sikora, Z.; Jahn, S.; et al. Microneedling: matching the results of medical needling and repetitive treatments to maximize potential for skin regeneration. Burns 2014, 40, 966-73.
62. Zhu, Y. F.; He, M. Q.; Lin, C. S.; et al. Multifunctional nanocarrier drug delivery systems: from diverse design to precise biomedical applications. Adv. Healthcare. Mater. 2025, 15, e02178.
63. Su, F.; Wang, Y.; Liu, X.; et al. Biocompatibility and in vivo degradation of chitosan based hydrogels as potential drug carrier. J. Biomater. Sci. Polym. Ed. 2018, 29, 1515-28.
64. Wang, X.; Song, R.; Johnson, M.; et al. An injectable chitosan-based self-healable hydrogel system as an antibacterial wound dressing. Materials 2021, 14, 5956.
65. Yuan, J.; Yang, H.; Liu, C.; et al. Microneedle patch loaded with exosomes containing MicroRNA‐29b prevents cardiac fibrosis after myocardial infarction. Adv. Healthcare. Mater. 2023, 12, 2202959.
66. Yu, X.; Pan, Q.; Zheng, Z.; et al. pH-responsive and porous vancomycin-loaded PLGA microspheres: evidence of controlled and sustained release for localized inflammation inhibition in vitro. RSC. Adv. 2018, 8, 37424-32.
67. Shiekh, P. A.; Singh, A.; Kumar, A. Exosome laden oxygen releasing antioxidant and antibacterial cryogel wound dressing OxOBand alleviate diabetic and infectious wound healing. Biomaterials 2020, 249, 120020.
68. Chen, M. H.; Wang, L. L.; Chung, J. J.; Kim, Y.; Atluri, P.; Burdick, J. A. Methods to assess shear-thinning hydrogels for application as injectable biomaterials. ACS. Biomater. Sci. Eng. 2017, 3, 3146-60.
69. Ahmadi, Z.; Yadav, S.; Kar, A. K.; et al. An injectable self-assembling hydrogel based on RGD peptidomimetic β-sheets as multifunctional biomaterials. Biomater. Adv. 2022, 133, 112633.
70. Roma-Rodrigues, C.; Raposo, L.; Cabral, R.; Paradinha, F.; Baptista, P.; Fernandes, A. Tumor microenvironment modulation via gold nanoparticles targeting malicious exosomes: implications for cancer diagnostics and therapy. Int. J. Mol. Sci. 2017, 18, 162.
71. Zhou, J.; Wang, M.; Ying, H.; et al. Extracellular matrix component shelled nanoparticles as dual enzyme-responsive drug delivery vehicles for cancer therapy. ACS. Biomater. Sci. Eng. 2018, 4, 2404-11.
72. Liang, C.; Wang, R.; He, T.; et al. Revolutionizing diabetic wound healing: the power of microneedles. Chinese. J. Plastic. Reconstruct. Surgery. 2023, 5, 185-94.
73. Sun, Q.; Tao, S.; Bovone, G.; et al. Versatile mechanically tunable hydrogels for therapeutic delivery applications. Adv. Healthcare. Mater. 2024, 13, 2304287.
74. Xu, X.; Saw, P. E.; Tao, W.; et al. ROS‐responsive polyprodrug nanoparticles for triggered drug delivery and effective cancer therapy. Adv. Mater. 2017, 29, 1700141.
75. Hui, L.; Qin, S.; Yang, L. Upper critical solution temperature polymer, photothermal agent, and erythrocyte membrane coating: an unexplored recipe for making drug carriers with spatiotemporally controlled cargo release. ACS. Biomater. Sci. Eng. 2016, 2, 2127-32.
76. Martinez, J. O.; Chiappini, C.; Ziemys, A.; et al. Engineering multi-stage nanovectors for controlled degradation and tunable release kinetics. Biomaterials 2013, 34, 8469-77.
77. Yang, G.; Waheed, S.; Wang, C.; Shekh, M.; Li, Z.; Wu, J. Exosomes and their bioengineering strategies in the cutaneous wound healing and related complications: current knowledge and future perspectives. Int. J. Biol. Sci. 2023, 19, 1430-54.
78. Su, Z.; Chen, M.; Xiao, Y.; et al. ROS-triggered and regenerating anticancer nanosystem: an effective strategy to subdue tumor's multidrug resistance. J. Controlled. Release. 2014, 196, 370-83.
79. Sun, J.; Ju, F.; Jin, J.; et al. M2 macrophage membrane-mediated biomimetic-nanoparticle carrying COX‐siRNA targeted delivery for prevention of tendon adhesions by inhibiting inflammation. Small 2023, 19, 2300326.
80. Yang, J.; Chen, Y.; Li, Y.; Yin, X. Magnetic resonance imaging-guided multi-drug chemotherapy and photothermal synergistic therapy with pH and NIR-stimulation release. ACS. Appl. Mater. Interfaces. 2017, 9, 22278-88.
81. Yang, C.; Liu, X.; Zhao, K.; et al. miRNA-21 promotes osteogenesis via the PTEN/PI3K/Akt/HIF-1α pathway and enhances bone regeneration in critical size defects. Stem. Cell. Res. Ther. 2019, 10, 65.
82. Fu, M.; Fang, L.; Xiang, X.; Fan, X.; Wu, J.; Wang, J. Microarray analysis of circRNAs sequencing profile in exosomes derived from bone marrow mesenchymal stem cells in postmenopausal osteoporosis patients. J. Clin. Lab. Anal. 2021, 36, e23916.
83. Wang, X.; Zhou, Y.; Gao, Q.; et al. The role of exosomal microRNAs and oxidative stress in neurodegenerative diseases. Oxid. Med. Cell. Longev. 2020, 2020, 1-17.
84. Zhou, X.; Ye, C.; Jiang, L.; et al. The bone mesenchymal stem cell-derived exosomal miR-146a-5p promotes diabetic wound healing in mice via macrophage M1/M2 polarization. Mol. Cell. Endocrinol. 2024, 579, 112089.
85. Boccardo, S.; Gaudiello, E.; Melly, L.; et al. Engineered mesenchymal cell-based patches as controlled VEGF delivery systems to induce extrinsic angiogenesis. Acta. Biomater. 2016, 42, 127-35.
86. Joo, H. S.; Suh, J. H.; Lee, H. J.; Bang, E. S.; Lee, J. M. Current knowledge and future perspectives on mesenchymal stem cell-derived exosomes as a new therapeutic agent. Int. J. Mol. Sci. 2020, 21, 727.
87. Marote, A.; Teixeira, F. G.; Mendes-pinheiro, B.; Salgado, A. J. MSCs-derived exosomes: cell-secreted nanovesicles with regenerative potential. Front. Pharmacol. 2016, 7, 231.
88. Xue, C.; Shen, Y.; Li, X.; et al. Exosomes derived from hypoxia-treated human adipose mesenchymal stem cells enhance angiogenesis through the PKA signaling pathway. Stem. Cells. Dev. 2018, 27, 456-65.
89. Zhu, F.; Chen, Y.; Li, J.; et al. Human umbilical cord mesenchymal stem cell‐derived exosomes attenuate myocardial infarction injury via miR‐24‐3p‐promoted M2 macrophage polarization. Adv. Biol. 2022, 6, 2200074.
90. Sahoo, S.; Losordo, D. W. Exosomes and cardiac repair after myocardial infarction. Circ. Res. 2014, 114, 333-44.
91. Liu, W.; Feng, Y.; Wang, X.; et al. Human umbilical vein endothelial cells-derived exosomes enhance cardiac function after acute myocardial infarction by activating the PI3K/AKT signaling pathway. Bioengineered 2022, 13, 8850-65.
92. Ribeiro-Rodrigues, T. M.; Laundos, T. L.; Pereira-Carvalho, R.; et al. Exosomes secreted by cardiomyocytes subjected to ischaemia promote cardiac angiogenesis. Cardiovasc. Res. 2017, 113, 1338-50.
93. Hsu, H.; Wang, A. Y. L.; Loh, C. Y. Y.; Pai, A. A.; Kao, H. Therapeutic potential of exosomes derived from diabetic adipose stem cells in cutaneous wound healing of db/db mice. Pharmaceutics 2022, 14, 1206.
94. Zhao, B.; Zhang, X.; Zhang, Y.; et al. Human exosomes accelerate cutaneous wound healing by promoting collagen synthesis in a diabetic mouse model. Stem. Cells. Dev. 2021, 30, 922-33.
95. Tao, S.; Yuan, T.; Zhang, Y.; Yin, W.; Guo, S.; Zhang, C. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics 2017, 7, 180-95.
96. Shen, Y.; Cai, J. The importance of using exosome-loaded miRNA for the treatment of spinal cord injury. Mol. Neurobiol. 2022, 60, 447-59.
97. Feng, J.; Zhang, Y.; Zhu, Z.; Gu, C.; Waqas, A.; Chen, L. Emerging exosomes and exosomal MiRNAs in spinal cord injury. Front. Cell. Dev. Biol. 2021, 9, 703989.
98. Whiteside, T. L. Exosomes and tumor-mediated immune suppression. J. Clin. Investig. 2016, 126, 1216-23.
99. Tan, S.; Xia, L.; Yi, P.; et al. Exosomal miRNAs in tumor microenvironment. J. Exp. Clin. Cancer. Res. 2020, 39, 67.
100. Sato, K.; Meng, F.; Glaser, S.; Alpini, G. Exosomes in liver pathology. J. Hepatol. 2016, 65, 213-21.
101. Jiao, Y.; Xu, P.; Shi, H.; Chen, D.; Shi, H. Advances on liver cell‐derived exosomes in liver diseases. J. Cell. Mol. Med. 2020, 25, 15-26.
102. Prasai, A.; Jay, J. W.; Jupiter, D.; Wolf, S. E.; El, Ayadi. A. Role of exosomes in dermal wound healing: a systematic review. J. Investig. Dermatol. 2022, 142, 662-78.e8.
103. Zhou, X.; Brown, B. A.; Siegel, A. P.; et al. Exosome-mediated crosstalk between keratinocytes and macrophages in cutaneous wound healing. ACS. Nano. 2020, 14, 12732-48.
104. Sabapatha, A.; Gercel‐taylor, C.; Taylor, D. D. Specific isolation of placenta‐derived exosomes from the circulation of pregnant women and their immunoregulatory consequences. Am. J. Reprod. Immunol. 2006, 56, 345-55.
105. Chang, X.; Yao, J.; He, Q.; Liu, M.; Duan, T.; Wang, K. Exosomes from women with preeclampsia induced vascular dysfunction by delivering sFlt (soluble Fms-like tyrosine kinase)-1 and sEng (soluble endoglin) to endothelial cells. Hypertension 2018, 72, 1381-90.
107. Miyanishi, M.; Tada, K.; Koike, M.; Uchiyama, Y.; Kitamura, T.; Nagata, S. Identification of Tim4 as a phosphatidylserine receptor. Nature 2007, 450, 435-9.
109. Zhou, Y.; Bréchard, S. Neutrophil extracellular vesicles: a delicate balance between pro-inflammatory responses and anti-inflammatory therapies. Cells 2022, 11, 3318.
110. Alexander, M.; O'connell, R. Exosomal miRNAs regulate inflammatory responses (IRM11P.624). J. Immunol. 2015, 194, 132.3.
111. Maderna, P.; Cottell, D. C.; Toivonen, T.; et al. FPR2/ALX receptor expression and internalization are critical for lipoxin A4 and annexin‐derived peptide‐stimulated phagocytosis. FASEB. J. 2010, 24, 4240-9.
112. Loynes, C. A.; Lee, J. A.; Robertson, A. L.; et al. PGE2 production at sites of tissue injury promotes an anti-inflammatory neutrophil phenotype and determines the outcome of inflammation resolution in vivo. Sci. Adv. 2018, 4, eaar8320.
113. Liu, Q.; Li, D.; Pan, X.; Liang, Y. Targeted therapy using engineered extracellular vesicles: principles and strategies for membrane modification. J. Nanobiotechnol. 2023, 21, 334.
114. Sun, J.; Shen, H.; Shao, L.; et al. HIF-1α overexpression in mesenchymal stem cell-derived exosomes mediates cardioprotection in myocardial infarction by enhanced angiogenesis. Stem. Cell. Res. Ther. 2020, 11, 373.
115. Larsimont, J.; Youssef, K. K.; Sánchez-Danés, A.; et al. Sox9 controls self-renewal of oncogene targeted cells and links tumor initiation and invasion. Cell. Stem. Cell. 2015, 17, 60-73.
116. Mattheolabakis, G.; Milane, L.; Singh, A.; Amiji, M. M. Hyaluronic acid targeting of CD44 for cancer therapy: from receptor biology to nanomedicine. J. Drug. Targeting. 2015, 23, 605-18.
117. Meng, L.; Wang, C.; Lu, Y.; et al. Targeted regulation of blood-brain barrier for enhanced therapeutic efficiency of hypoxia-modifier nanoparticles and immune checkpoint blockade antibodies for glioblastoma. ACS. Appl. Mater. Interfaces. 2021, 13, 11657-71.
118. Kim, S.; Rait, A.; Kim, E.; Demarco, J.; Pirollo, K. F.; Chang, E. H. Encapsulation of temozolomide in a tumor-targeting nanocomplex enhances anti-cancer efficacy and reduces toxicity in a mouse model of glioblastoma. Cancer. Lett. 2015, 369, 250-8.
119. Hsu, Y.; Acuña, M.; Tahara, S. M.; Peng, C. Reduced phagocytosis of colloidal carriers using soluble CD47. Pharm. Res. 2003, 20, 1539-42.
120. Zhang, X.; Wang, C.; Wang, J.; et al. PD‐1 blockade cellular vesicles for cancer immunotherapy. Adv. Mater. 2018, 30, 1707112.
121. Dai, X.; Wang, Z.; Fan, M.; et al. PD-L1 antibodies-armed exosomal vaccine for enhanced cancer immunotherapy by simultaneously in situ activating T cells and blocking PD-1/PD-L1 axis. Extracellular. Vesicle. 2022, 1, 100012.
122. Chao, M. P.; Weissman, I. L.; Majeti, R. The CD47-SIRPα pathway in cancer immune evasion and potential therapeutic implications. Curr. Opin. Immunol. 2012, 24, 225-32.
123. Kim, D. H.; Kim, H.; Choi, Y. J.; et al. Exosomal PD-L1 promotes tumor growth through immune escape in non-small cell lung cancer. Exp. Mol. Med. 2019, 51, 1-13.
124. Chen, G.; Huang, A. C.; Zhang, W.; et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature 2018, 560, 382-6.
125. Mohammadi, M. R.; Nojoomi, A.; Mozafari, M.; Dubnika, A.; Inayathullah, M.; Rajadas, J. Nanomaterials engineering for drug delivery: a hybridization approach. J. Mater. Chem. B. 2017, 5, 3995-4018.
126. Gomes, E. R.; Carvalho, A. T.; Barbosa, T. C.; et al. Fusion of tumor-derived exosomes with long-circulating and pH-sensitive liposomes loaded with doxorubicin for the treatment of breast cancer. AAPS. PharmSciTech. 2022, 23, 255.
127. Qiao, L.; Hu, S.; Huang, K.; et al. Tumor cell-derived exosomes home to their cells of origin and can be used as Trojan horses to deliver cancer drugs. Theranostics 2020, 10, 3474-87.
128. Barjesteh, T.; Mansur, S.; Bao, Y. Inorganic nanoparticle-loaded exosomes for biomedical applications. Molecules 2021, 26, 1135.
129. Wang, J.; Chen, P.; Dong, Y.; et al. Designer exosomes enabling tumor targeted efficient chemo/gene/photothermal therapy. Biomaterials 2021, 276, 121056.
130. Yang, Y.; Hong, Y.; Nam, G. H.; Chung, J. H.; Koh, E.; Kim, I. S. Virus‐mimetic fusogenic exosomes for direct delivery of integral membrane proteins to target cell membranes. Adv. Mater. 2017, 29, 1605604.
131. Zubarev, I.; Vladimirtsev, D.; Vorontsova, M.; et al. Viral membrane fusion proteins and RNA sorting mechanisms for the molecular delivery by exosomes. Cells 2021, 10, 3043.
132. Hung, M. E.; Leonard, J. N. Stabilization of exosome-targeting peptides via engineered glycosylation. J. Biol. Chem. 2015, 290, 8166-72.
133. Chan, M.; Chang, Z.; Huang, C. F.; Lee, L. J.; Liu, R.; Hsiao, M. Integrated therapy platform of exosomal system: hybrid inorganic/organic nanoparticles with exosomes for cancer treatment. Nanoscale. Horiz. 2022, 7, 352-67.
134. Nie, W.; Wu, G.; Zhang, J.; et al. Responsive exosome nano-bioconjugates for synergistic cancer therapy. Angew. Chem. Int. 2019, 59, 2018-22.
135. Liang, Y.; Xu, X.; Li, X.; et al. Chondrocyte-targeted MicroRNA delivery by engineered exosomes toward a cell-free osteoarthritis therapy. ACS. Appl. Mater. Interfaces. 2020, 12, 36938-47.
136. Fan, L.; Liu, C.; Chen, X.; et al. Exosomes-loaded electroconductive hydrogel synergistically promotes tissue repair after spinal cord injury via immunoregulation and enhancement of myelinated axon growth. Adv. Sci. 2022, 9, 2105586.
137. Chen, Y.; Lin, J.; Yan, W. A Prosperous application of hydrogels with extracellular vesicles release for traumatic brain injury. Front. Neurol. 2022, 13, 908468.
138. Li, S.; Xu, J.; Qian, Y.; Zhang, R. Hydrogel in the treatment of traumatic brain injury. Biomater. Res. 2024, 28, 0085.
139. Arabpour, M.; Saghazadeh, A.; Rezaei, N. Anti-inflammatory and M2 macrophage polarization-promoting effect of mesenchymal stem cell-derived exosomes. Int. Immunopharmacol. 2021, 97, 107823.
140. Kant, V.; Gopal, A.; Kumar, D.; et al. Curcumin-induced angiogenesis hastens wound healing in diabetic rats. J. Surg. Res. 2015, 193, 978-88.
141. Tan, X.; Zhang, J.; Heng, Y.; et al. Locally delivered hydrogels with controlled release of nanoscale exosomes promote cardiac repair after myocardial infarction. J. Controlled. Release. 2024, 368, 303-17.
142. Peng, H.; Hou, M.; Wu, Z.; et al. Plasma exosomal miR-122 regulates the efficacy of metformin via AMPK in type 2 diabetes and hepatocellular carcinoma. Heliyon 2022, 8, e11503.
143. Morrissey, S. M.; Zhang, F.; Ding, C.; et al. Tumor-derived exosomes drive immunosuppressive macrophages in a pre-metastatic niche through glycolytic dominant metabolic reprogramming. Cell. Metab. 2021, 33, 2040-58.e10.
144. Mcandrews, K. M.; Che, S. P.; Lebleu, V. S.; Kalluri, R. Effective delivery of STING agonist using exosomes suppresses tumor growth and enhances antitumor immunity. J. Biol. Chem. 2021, 296, 100523.
145. Afewerki, S.; Bassous, N.; Harb, S.; et al. Advances in dual functional antimicrobial and osteoinductive biomaterials for orthopaedic applications. Nanomed. Nanotechnol. Biol. Med. 2020, 24, 102143.
146. Wang, H.; Gui, B.; Chen, Y.; et al. Black-phosphorus-reinforced injectable conductive biodegradable hydrogel for the delivery of ADSC-derived exosomes to repair myocardial infarction. ACS. Appl. Mater. Interfaces. 2024, 16, 58286-98.
147. Lyu, S.; Liu, Q.; Yuen, H.; et al. A differential-targeting core-shell microneedle patch with coordinated and prolonged release of mangiferin and MSC-derived exosomes for scarless skin regeneration. Mater. Horiz. 2024, 11, 2667-84.
148. Namazi, H.; Mohit, E.; Namazi, I.; et al. Exosomes secreted by hypoxic cardiosphere‐derived cells enhance tube formation and increase pro‐angiogenic miRNA. J. Cell. Biochem. 2018, 119, 4150-60.
149. Meikle, S.; Piñeiro, Y.; Bañobre, López. M.; Rivas, J.; Santin, M. Surface functionalization superparamagnetic nanoparticles conjugated with thermoresponsive poly(epsilon-lysine) dendrons tethered with carboxybetaine for the mild hyperthermia-controlled delivery of VEGF. Acta. Biomater. 2016, 40, 235-42.
150. Li, X.; Qu, K.; Zhou, B.; et al. Electrical stimulation of neonatal rat cardiomyocytes using conductive polydopamine-reduced graphene oxide-hybrid hydrogels for constructing cardiac microtissues. Colloid. Surface. B. 2021, 205, 111844.
151. Chen, R. R.; Silva, E. A.; Yuen, W. W.; Mooney, D. J. Spatio-temporal VEGF and PDGF delivery patterns blood vessel formation and maturation. Pharm. Res. 2006, 24, 258-64.
152. Dai, F.; Zhang, J.; Chen, F.; et al. A multi‐responsive hydrogel combined with mild heat stimulation promotes diabetic wound healing by regulating inflammatory and enhancing angiogenesis. Adv. Sci. 2024, 11, 2408783.
153. Darge, H. F.; Andrgie, A. T.; Hanurry, E. Y.; et al. Localized controlled release of bevacizumab and doxorubicin by thermo-sensitive hydrogel for normalization of tumor vasculature and to enhance the efficacy of chemotherapy. Int. J. Pharm. 2019, 572, 118799.
154. Jiang, L.; Yang, X.; Zhang, Y.; et al. Novel ROS-scavenging hydrogel with enhanced anti-inflammation and angiogenic properties for promoting diabetic wound healing. Biomater. Adv. 2023, 144, 213226.
155. Tao, S.; Guo, S.; Li, M.; Ke, Q.; Guo, Y.; Zhang, C. Chitosan wound dressings incorporating exosomes derived from MicroRNA-126-overexpressing synovium mesenchymal stem cells provide sustained release of exosomes and heal full-thickness skin defects in a diabetic rat model. Stem. Cells. Transl. Med. 2017, 6, 736-47.
156. Li, H.; Feng, Y.; Zheng, X.; et al. M2-type exosomes nanoparticles for rheumatoid arthritis therapy via macrophage re-polarization. J. Controlled. Release. 2022, 341, 16-30.
157. Zhang, K.; Zhao, X.; Chen, X.; et al. Enhanced therapeutic effects of mesenchymal stem cell-derived exosomes with an injectable hydrogel for hindlimb ischemia treatment. ACS. Appl. Mater. Interfaces. 2018, 10, 30081-91.
158. Li, M.; Wang, T.; Tian, H.; Wei, G.; Zhao, L.; Shi, Y. Macrophage-derived exosomes accelerate wound healing through their anti-inflammation effects in a diabetic rat model. Artif. Cells. Nanomed. Biotechnol. 2019, 47, 3793-803.
159. Yi, H.; Chang, J. Y.; Shong, M. The mitochondrial unfolded protein response and mitohormesis: a perspective on metabolic diseases. J. Mol. Endocrinol. 2018, 61, R91-R105.
160. Nomura, J.; So, A.; Tamura, M.; Busso, N. Intracellular ATP decrease mediates NLRP3 inflammasome activation upon nigericin and crystal stimulation. J. Immunol. 2015, 195, 5718-24.
161. Marín V, Buccini DF, Arboleda V JW, Cardoso MH, Franco OL. Expanding therapeutic strategies for intracellular bacterial infections through conjugates of apoptotic body-antimicrobial peptides. Drug. Discov. Today. 2023, 28, 103444.
162. Ryall, J. G.; Cliff, T.; Dalton, S.; Sartorelli, V. Metabolic reprogramming of stem cell epigenetics. Cell. Stem. Cell. 2015, 17, 651-62.
163. Wang, Z.; Zhao, F.; Xu, C.; et al. Metabolic reprogramming in skin wound healing. Burns. Trauma. 2024, 12, tkad047.
164. Ke, J.; Ding, J.; Xu, Y.; et al. Engineering microglial exosome-mediated microRNA-124-3p delivery for Alzheimer's disease combinational therapy. Biomater. Sci. 2026, 14, 186-97.
165. An, H.; Jang, Y.; Choi, J.; Hur, J.; Kim, S.; Kwon, Y. New Insights into AMPK, as a potential therapeutic target in metabolic dysfunction-associated steatotic liver disease and hepatic fibrosis. Biomol. Ther. 2024, 33, 18-38.
166. Gao, Y.; Yuan, Z.; Yuan, X.; et al. Bioinspired porous microspheres for sustained hypoxic exosomes release and vascularized bone regeneration. Bioact. Mater. 2022, 14, 377-88.
167. Edwards, N.; Feliers, D.; Zhao, Q.; Stone, R.; Christy, R.; Cheng, X. An electrochemically deposited collagen wound matrix combined with adipose-derived stem cells improves cutaneous wound healing in a mouse model of type 2 diabetes. J. Biomater. Appl. 2018, 33, 553-65.
168. Zhang, Y.; Chen, H.; Li, J. Recent advances on gelatin methacrylate hydrogels with controlled microstructures for tissue engineering. Int. J. Biol. Macromol. 2022, 221, 91-107.
169. Mauri, E.; Sacchetti, A.; Rossi, F. The synthesis of RGD-functionalized hydrogels as a tool for therapeutic applications. J. Vis. Exp. 2016, 54445.
170. Mo, Y.; Guo, R.; Zhang, Y.; Xue, W.; Cheng, B.; Zhang, Y. Controlled dual delivery of angiogenin and curcumin by electrospun nanofibers for skin regeneration. Tissue. Eng. Part. A. 2017, 23, 597-608.
171. Wei, Z.; Ren, J.; Hu, J.; Wei, H. Polyethylene glycol-polyester based temperature-sensitive hydrogel delivering mesenchymal stem cell-derived exosomes enhances acute skin wound healing. Front. Bioeng. Biotechnol. 2025, 13, 1730631.
172. Shi, Y.; Wang, S.; Wang, K.; et al. Relieving macrophage dysfunction by inhibiting SREBP2 activity: a hypoxic mesenchymal stem cells‐derived exosomes loaded multifunctional hydrogel for accelerated diabetic wound healing. Small 2024, 20, 2309276.
173. Govender, M.; Indermun, S.; Choonara, Y. E. 3D bioprinted microneedles: merging drug delivery and scaffold science for tissue-specific applications. Expert. Opin. Drug. Deliv. 2024, 21, 1559-72.
174. Britton, D.; Almanzar, D.; Xiao, Y.; et al. Exosome loaded protein hydrogel for enhanced gelation kinetics and wound healing. ACS. Appl. Bio. Mater. 2024, 7, 5992-6000.
175. Cho, B. S.; Kim, J. O.; Ha, D. H.; Yi, Y. W. Exosomes derived from human adipose tissue-derived mesenchymal stem cells alleviate atopic dermatitis. Stem. Cell. Res. Ther. 2018, 9, 187.
176. Sun, X.; Ding, H.; Li, X.; Wu, Y.; Huang, X. Disulfiram-loaded nanovesicles hydrogel promotes healing of diabetic wound. J. Transl. Med. 2024, 22, 1066.
177. Wang, K.; Dong, R.; Tang, J.; et al. Exosomes laden self-healing injectable hydrogel enhances diabetic wound healing via regulating macrophage polarization to accelerate angiogenesis. Chem. Eng. J. 2022, 430, 132664.
178. Li, X.; Qu, S.; Ouyang, Q.; et al. A multifunctional composite nanoparticle with antibacterial activities, anti-inflammatory, and angiogenesis for diabetic wound healing. Int. J. Biol. Macromol. 2024, 260, 129531.
179. Lv, Y.; Li, L.; Zhang, J.; et al. Visible-light cross-linkable multifunctional hydrogels loaded with exosomes facilitate full-thickness skin defect wound healing through participating in the entire healing process. ACS. Appl. Mater. Interfaces. 2024, 16, 25923-37.
180. Xiong, Y.; Chen, L.; Liu, P.; et al. All-in-one: multifunctional hydrogel accelerates oxidative diabetic wound healing through timed‐release of exosome and fibroblast growth factor. Small 2021, 18, 2104229.
181. Shi, Q.; Qian, Z.; Liu, D.; et al. GMSC-derived exosomes combined with a chitosan/silk hydrogel sponge accelerates wound healing in a diabetic rat skin defect model. Front. Physiol. 2017, 8, 904.
182. Feng, J.; Yao, Y.; Wang, Q.; et al. Exosomes: potential key players towards novel therapeutic options in diabetic wounds. Biomed. Pharmacother. 2023, 166, 115297.
183. Geng, X.; Qi, Y.; Liu, X.; Shi, Y.; Li, H.; Zhao, L. A multifunctional antibacterial and self-healing hydrogel laden with bone marrow mesenchymal stem cell-derived exosomes for accelerating diabetic wound healing. Biomater. Adv. 2022, 133, 112613.
184. Jing, Y.; Huang, T.; Zhao, B.; et al. A ROS/glucose stimulated-responsive ADSCs-derived exosomes-release hydrogel system for diabetic wound healing. Chem. Eng. J. 2024, 487, 150561.
185. You, C.; Li, Q.; Wang, X.; et al. Silver nanoparticle loaded collagen/chitosan scaffolds promote wound healing via regulating fibroblast migration and macrophage activation. Sci. Rep. 2017, 7, 10489.
186. Hu, X.; Zhang, H.; Wang, Y.; et al. Synergistic antibacterial strategy based on photodynamic therapy: progress and perspectives. Chem. Eng. J. 2022, 450, 138129.
187. Losi, P.; Briganti, E.; Errico, C.; et al. Fibrin-based scaffold incorporating VEGF- and bFGF-loaded nanoparticles stimulates wound healing in diabetic mice. Acta. Biomater. 2013, 9, 7814-21.
188. Shafei, S.; Khanmohammadi, M.; Heidari, R.; et al. Exosome loaded alginate hydrogel promotes tissue regeneration in full‐thickness skin wounds: an in vivo study. J. Biomed. . Mater. Res. 2019, 108, 545-56.
189. Zhou, L.; Jiao, X.; Liu, S.; et al. Functional DNA-based hydrogel intelligent materials for biomedical applications. J. Mater. Chem. B. 2020, 8, 1991-2009.
190. Khoshnoudi, P.; Sabiza, S.; Khosravi, M.; Mohamadian, B. Exploring effect of M2 macrophages on experimental full‐thickness wound healing in streptozotocin‐induced diabetic rats. Int. J. Exp. Pathol. 2023, 105, 13-20.
191. Tu, Z.; Chen, M.; Wang, M.; et al. Engineering bioactive M2 macrophage-polarized anti-inflammatory, antioxidant, and antibacterial scaffolds for rapid angiogenesis and diabetic wound repair. Adv. Funct. Mater. 2021, 31, 2100924.
192. Hu, Y.; Ruan, X.; Lv, X.; et al. Biofilm microenvironment-responsive nanoparticles for the treatment of bacterial infection. Nano. Today. 2022, 46, 101602.
193. Schwarz, G.; Ren, X.; Xie, W.; Guo, H.; Jiang, Y.; Zhang, J. Engineered exosomes: a promising drug delivery platform with therapeutic potential. Front. Mol. Biosci. 2025, 12, 1583992.
194. Zhang, L.; Li, T.; Yu, Y.; et al. An injectable conductive hydrogel restores electrical transmission at myocardial infarct site to preserve cardiac function and enhance repair. Bioact. Mater. 2023, 20, 339-54.
195. Yan, C.; Wang, X.; Wang, Q.; et al. A novel conductive polypyrrole-chitosan hydrogel containing human endometrial mesenchymal stem cell-derived exosomes facilitated sustained release for cardiac repair. Adv. Healthcare. Mater. 2024, 13, 2304207.
196. Yaping, X.; Guotian, Y.; Dandan, J.; Jintao, D.; Xinyi, L.; Zhikun, G. Fibroblast-derived exosomal miRNA-133 promotes cardiomyocyte-like differentiation. Acta. Histochem. 2022, 124, 151931.
197. Chen, C.; Cai, S.; Wu, M.; et al. Role of cardiomyocyte-derived exosomal MicroRNA-146a-5p in macrophage polarization and activation. Dis. Markers. 2022, 2022, 1-13.
198. Ben-Mordechai, T.; Holbova, R.; Landa-Rouben, N.; et al. Macrophage subpopulations are essential for infarct repair with and without stem cell therapy. J. Am. Coll. Cardiol. 2013, 62, 1890-901.
199. Li, S.; Yue, Y.; Xu, W.; Xiong, S. MicroRNA-146a represses mycobacteria-induced inflammatory response and facilitates bacterial replication via targeting IRAK-1 and TRAF-6. PLoS. ONE. 2013, 8, e81438.
200. Wang, M.; Wang, C.; Chen, M.; et al. Efficient angiogenesis-based diabetic wound healing/skin reconstruction through bioactive antibacterial adhesive ultraviolet shielding nanodressing with exosome release. ACS. Nano. 2019, 13, 10279-93.
201. Elzamly, S.; Agina, H. A.; Elbalshy, A. E.; Abuhashim, M.; Saad, E.; Abd, Elmageed. Z. Y. Integration of VEGF and α-SMA expression improves the prediction accuracy of fibrosis in chronic hepatitis C liver biopsy. Appl. Immunohistochem. Mol. Morphol. 2017, 25, 261-70.
202. Chen, J.; Chen, H.; Li, P.; et al. Simultaneous regeneration of articular cartilage and subchondral bone in vivo using MSCs induced by a spatially controlled gene delivery system in bilayered integrated scaffolds. Biomaterials 2011, 32, 4793-805.
203. Sun, T.; Feng, Z.; He, W.; et al. Novel 3D-printing bilayer GelMA-based hydrogel containing BP, β-TCP and exosomes for cartilage-bone integrated repair. Biofabrication 2023, 16, 015008.
204. Wang, Y.; Wang, J.; Gao, R.; et al. Biomimetic glycopeptide hydrogel coated PCL/nHA scaffold for enhanced cranial bone regeneration via macrophage M2 polarization-induced osteo-immunomodulation. Biomaterials 2022, 285, 121538.
205. Battaglia, M.; Rimondi, E.; Monti, C.; et al. Validity of T2 mapping in characterization of the regeneration tissue by bone marrow derived cell transplantation in osteochondral lesions of the ankle. Eur. J. Radiol. 2011, 80, e132-9.
206. Levingstone, T. J.; Thompson, E.; Matsiko, A.; Schepens, A.; Gleeson, J. P.; O’brien, F. J. Multi-layered collagen-based scaffolds for osteochondral defect repair in rabbits. Acta. Biomater. 2016, 32, 149-60.
207. Xu, J.; Ji, J.; Jiao, J.; et al. 3D printing for bone-cartilage interface regeneration. Front. Bioeng. Biotechnol. 2022, 10, 828921.
208. Ma, T.; Xu, G.; Gao, T.; et al. Engineered exosomes with ATF5-modified mRNA loaded in injectable thermogels alleviate osteoarthritis by targeting the mitochondrial unfolded protein response. ACS. Appl. Mater. Interfaces. 2024, 16, 21383-99.
209. Liu, Y.; Lin, S.; Xu, Z.; et al. High‐performance hydrogel-encapsulated engineered exosomes for supporting endoplasmic reticulum homeostasis and boosting diabetic bone regeneration. Adv. Sci. 2024, 11, 2309491.
210. Xu, H.; Feng, J.; Dai, N.; et al. Self-assembling peptide hydrogel scaffold integrating stem cell-derived exosomes for infected bone defects. J. Biomater. Sci. Polym. Ed. 2024, 35, 1511-22.
211. Lotz, M. K.; Caramés, B. Autophagy and cartilage homeostasis mechanisms in joint health, aging and OA. Nat. Rev. Rheumatol. 2011, 7, 579-87.
212. Contreras-naranjo, J. C.; Wu, H.; Ugaz, V. M. Microfluidics for exosome isolation and analysis: enabling liquid biopsy for personalized medicine. Lab. Chip. 2017, 17, 3558-77.
213. Biswas, A. A.; Dhondale, M. R.; Agrawal, A. K.; Serrano, D. R.; Mishra, B.; Kumar, D. Advancements in microneedle fabrication techniques: artificial intelligence assisted 3D-printing technology. Drug. Deliv. Transl. Res. 2024, 14, 1458-79.
214. Jangam, T. C.; Desai, S. A.; Patel, V. P.; Pagare, N. B.; Raut, N. D. Exosomes as therapeutic and diagnostic tools: advances, challenges, and future directions. Cell. Biochem. Biophys. 2025, 83, 2805-29.
215. Batrakova, E. V.; Kim, M. S. Development and regulation of exosome‐based therapy products. WIREs. Nanomed. Nanobiotechnol. 2016, 8, 744-57.
216. Verma, N.; Arora, S. Navigating the global regulatory landscape for exosome-based therapeutics: challenges, strategies, and future directions. Pharmaceutics 2025, 17, 990.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






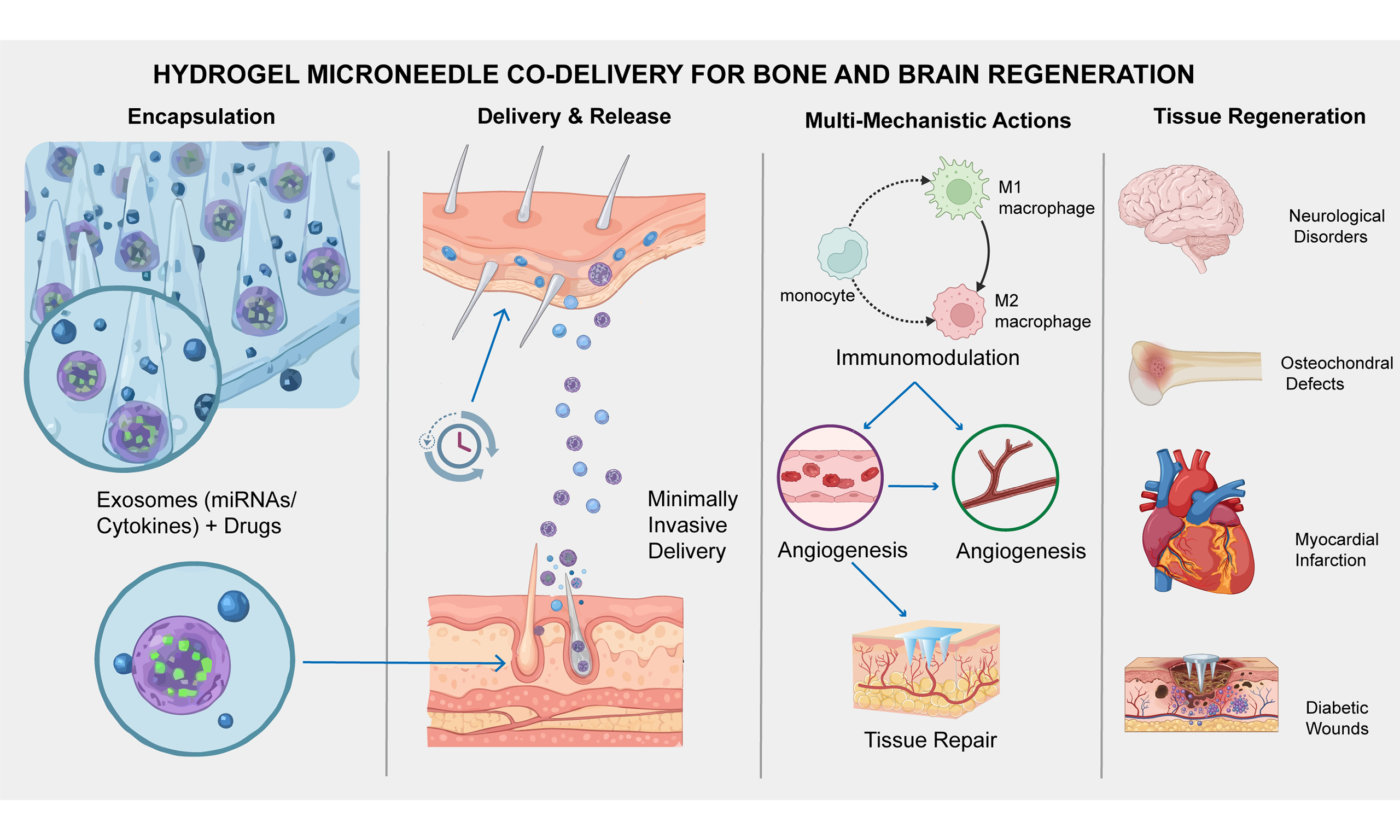
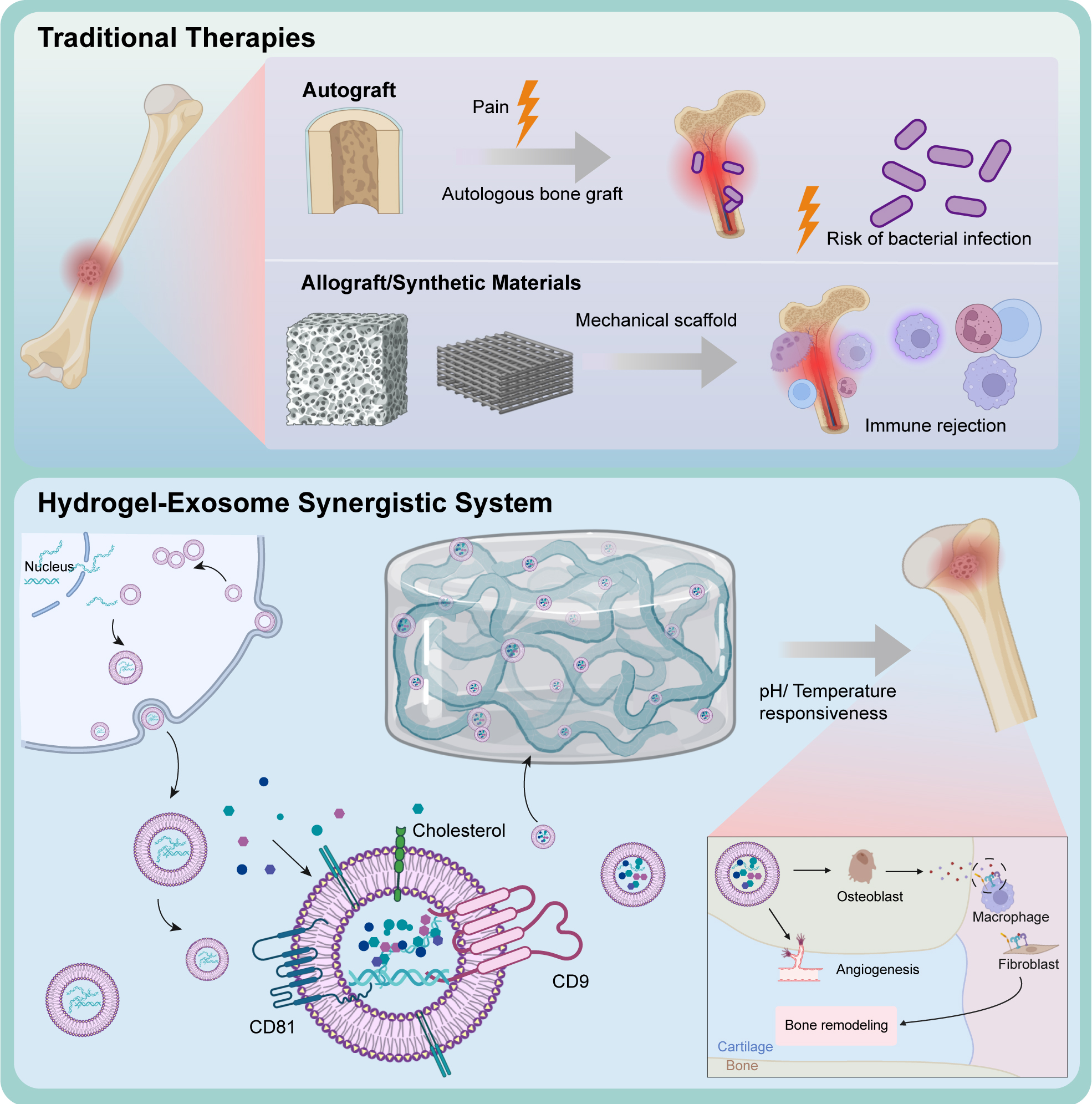
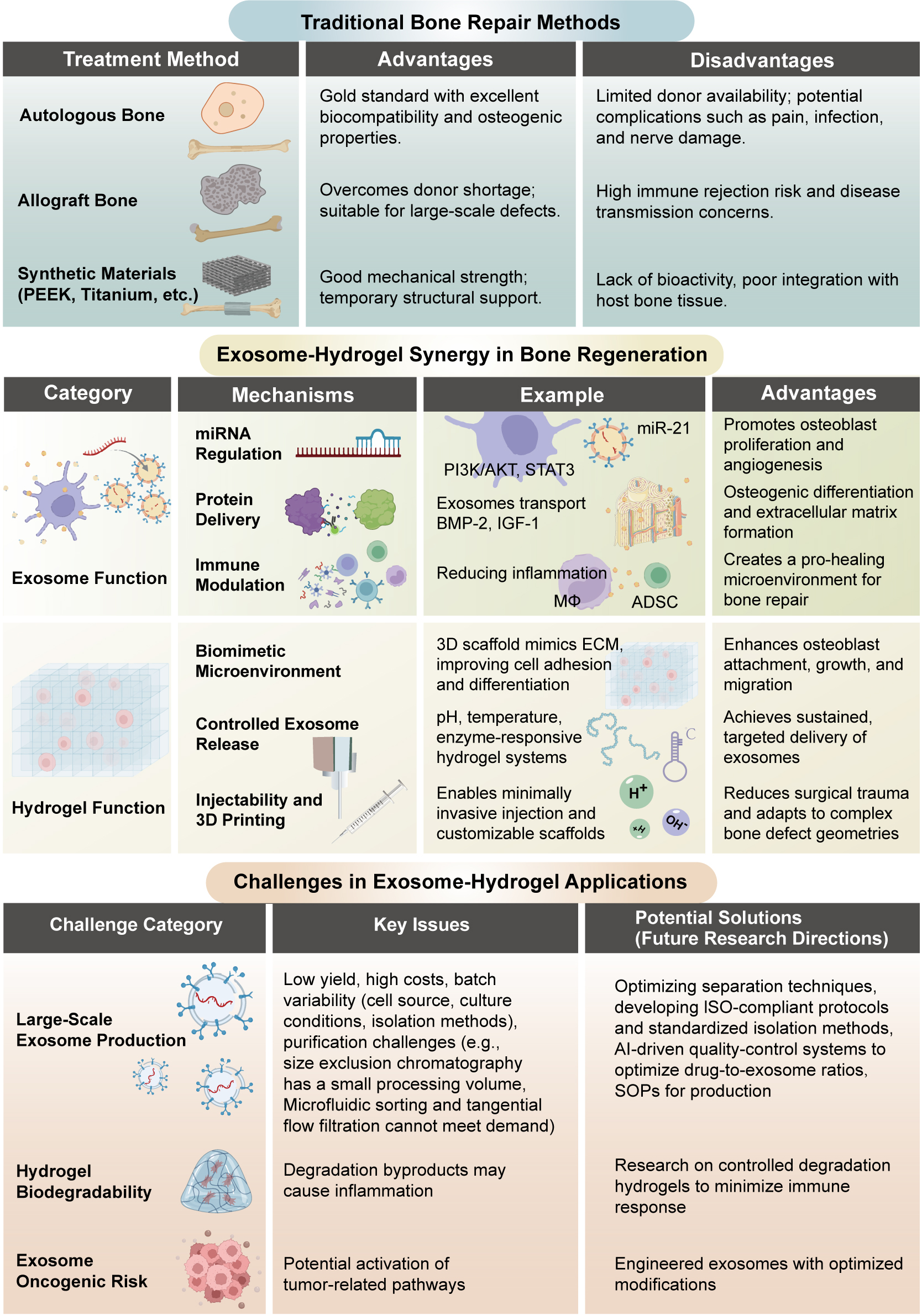
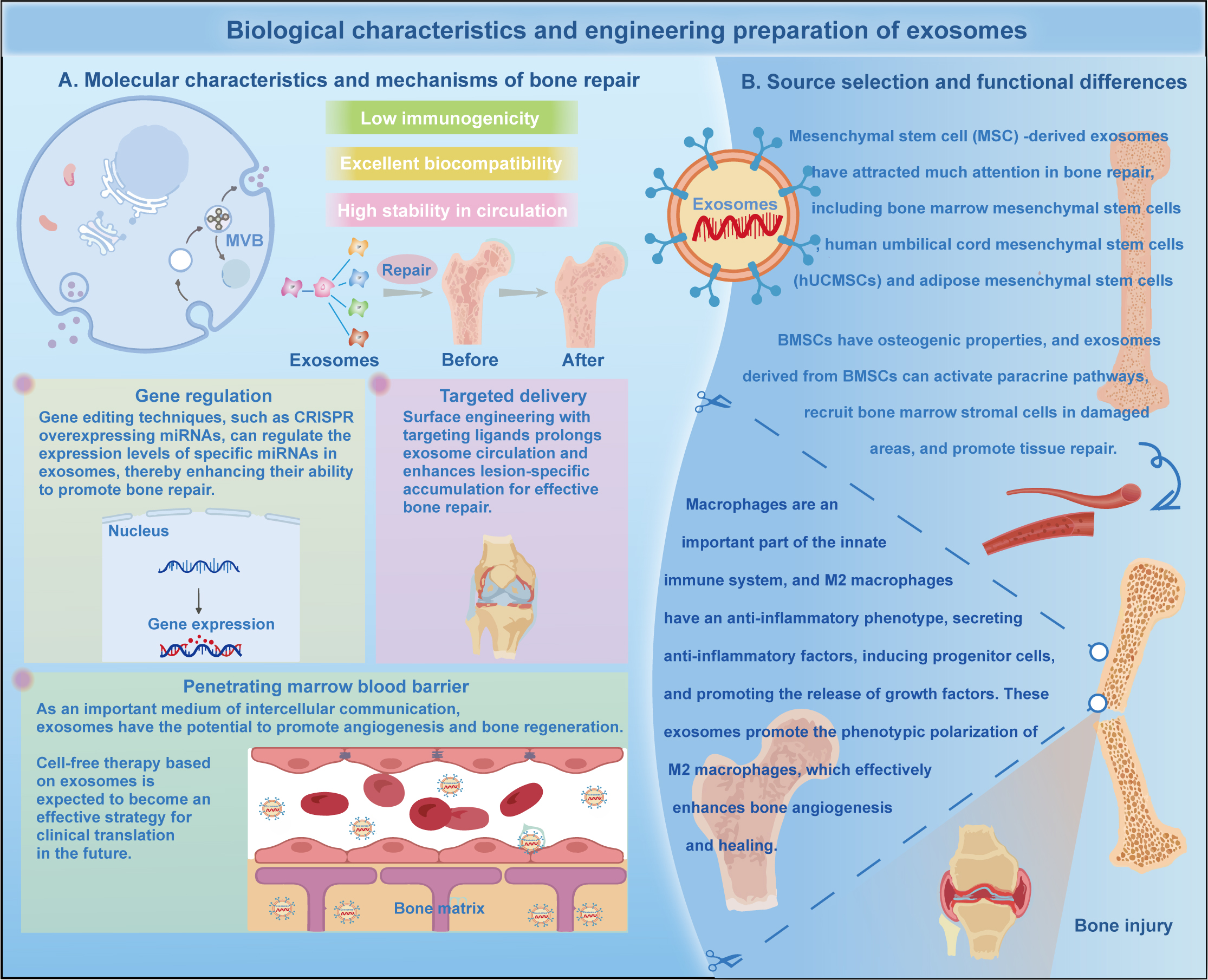
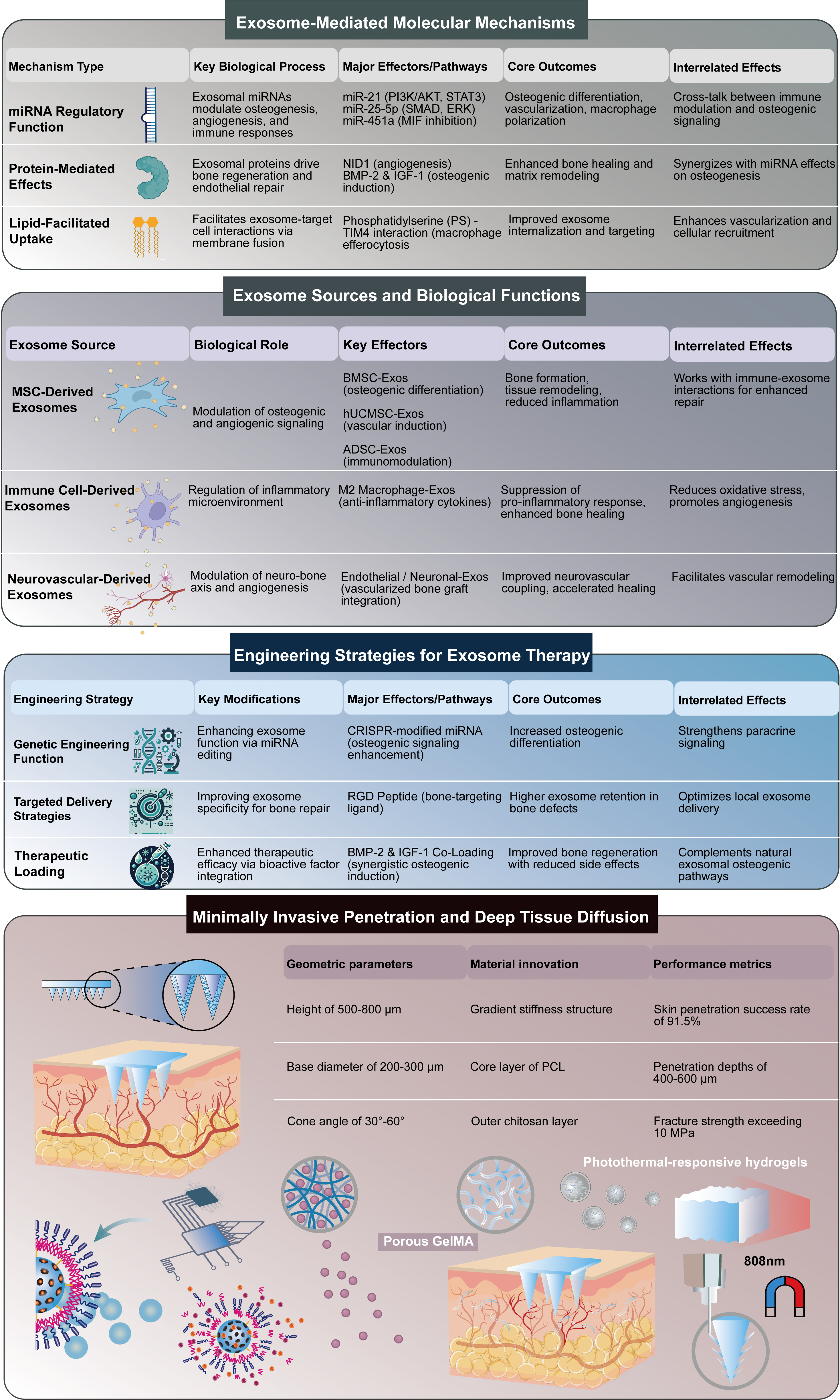
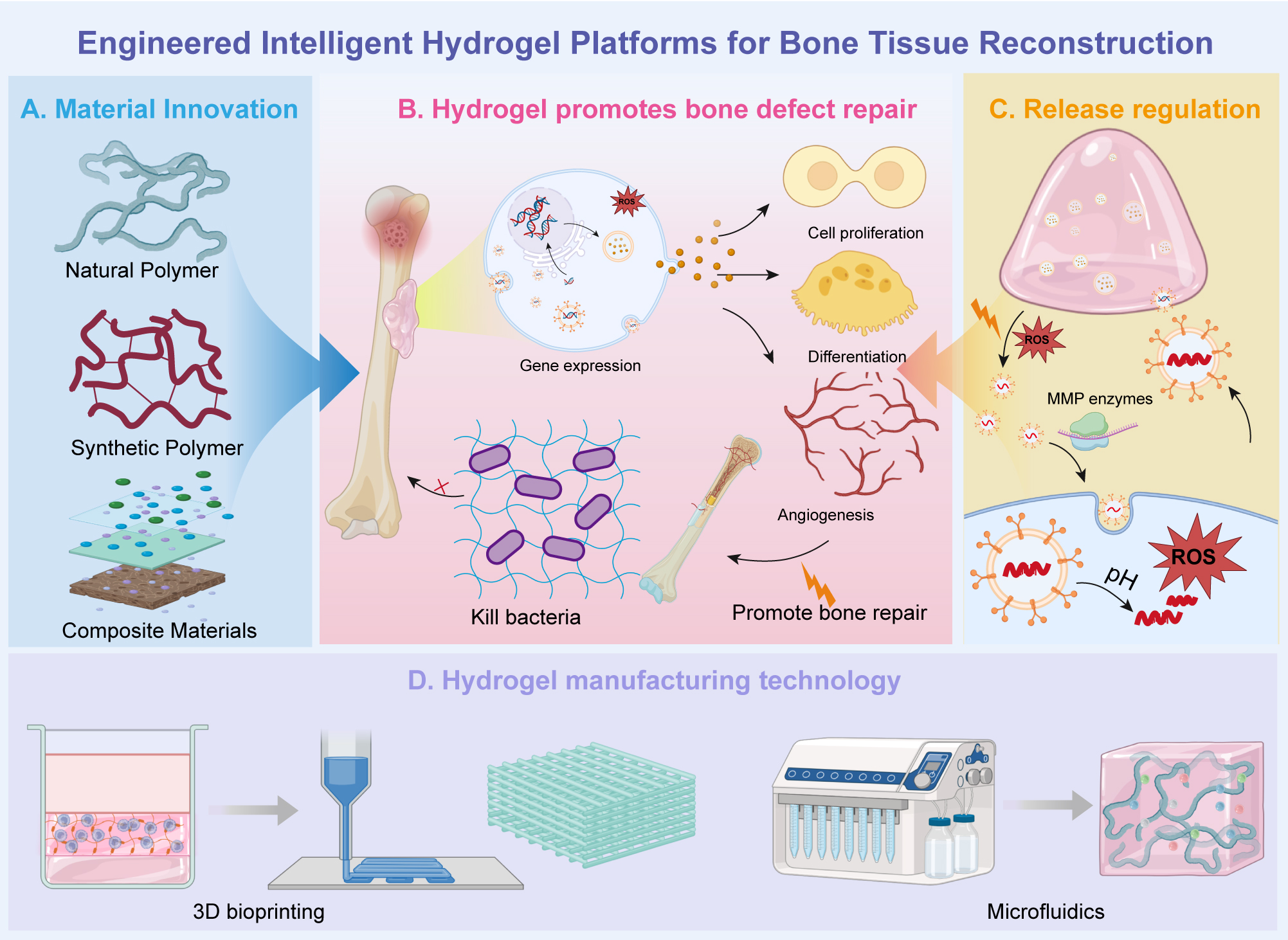
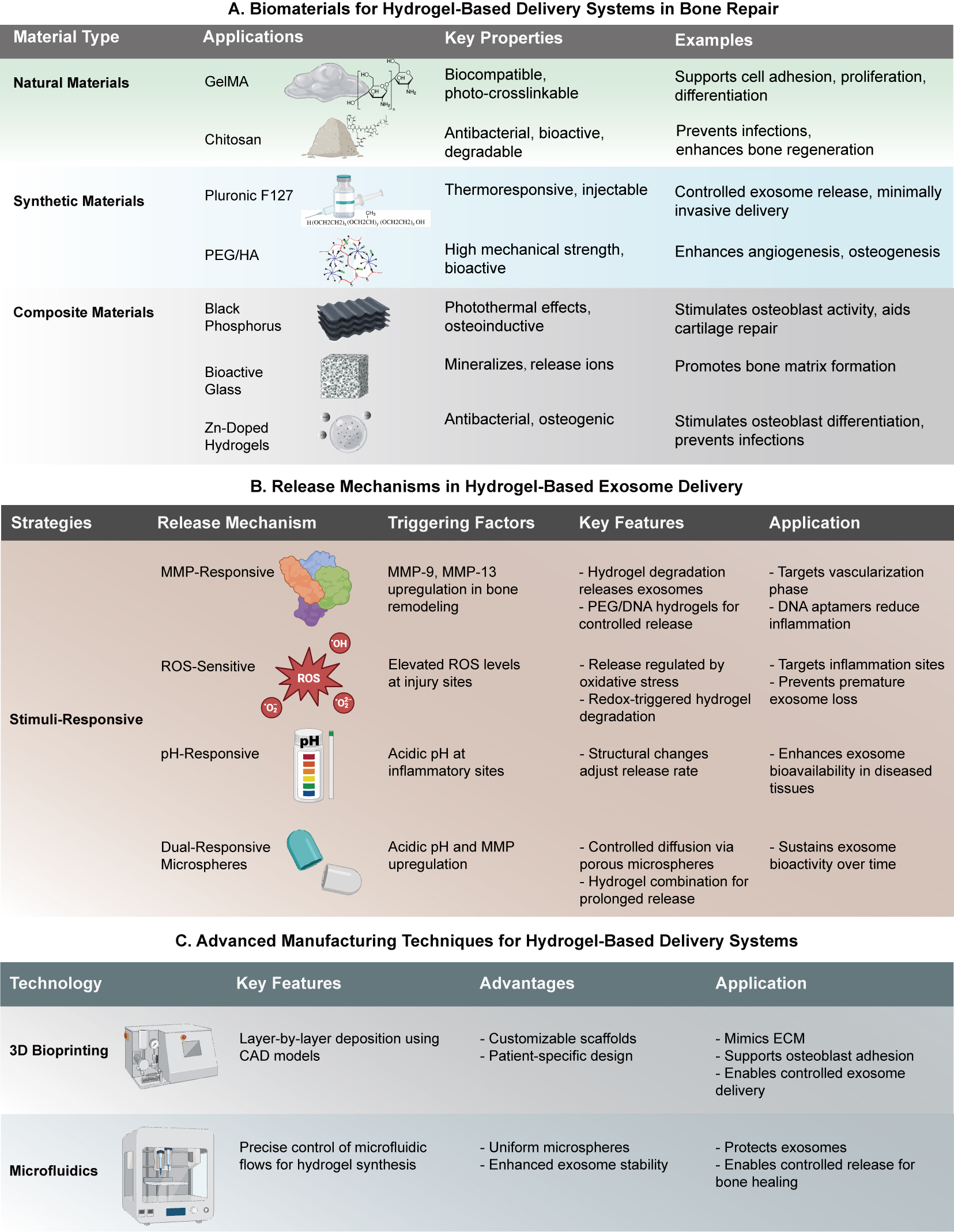
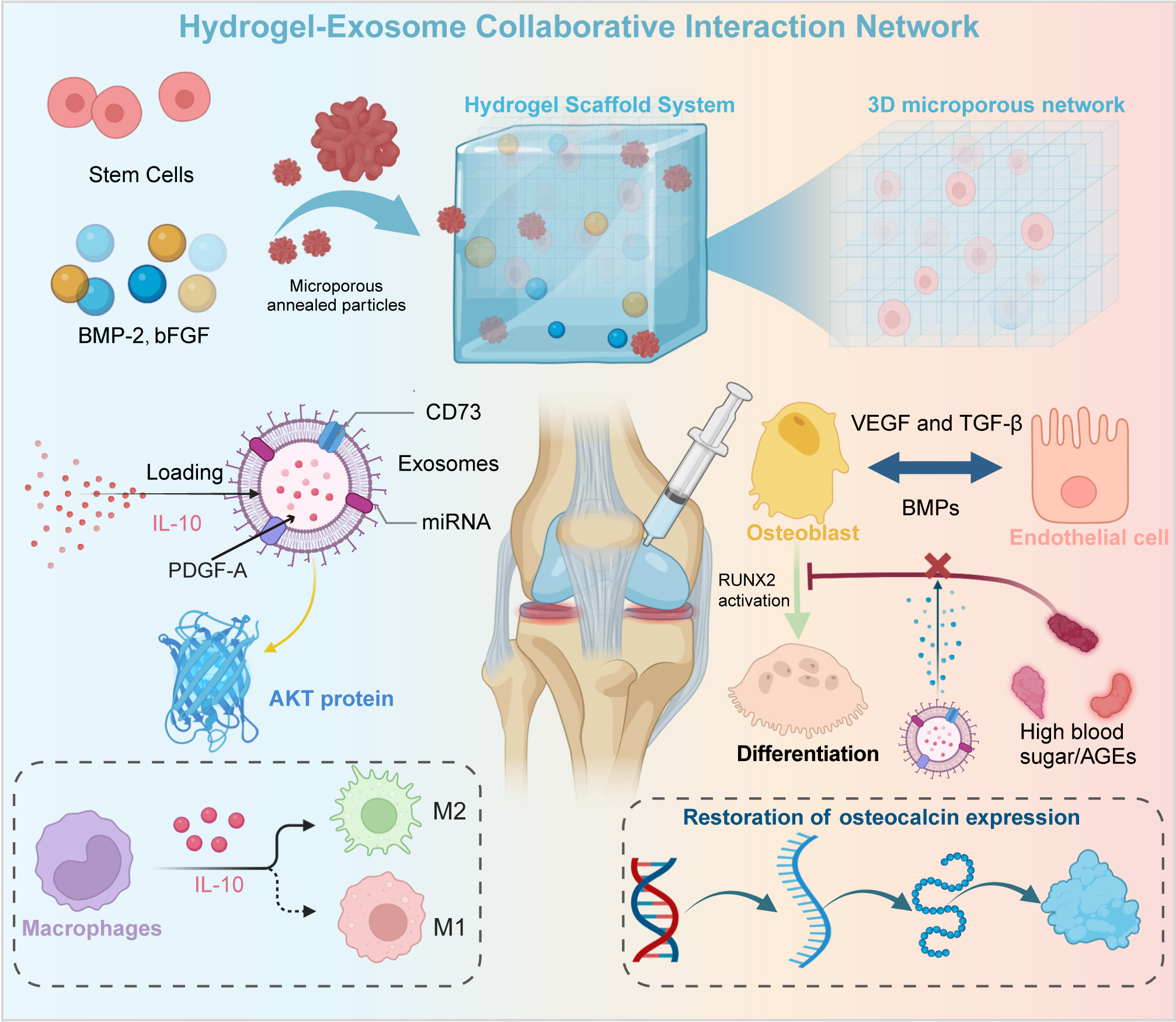
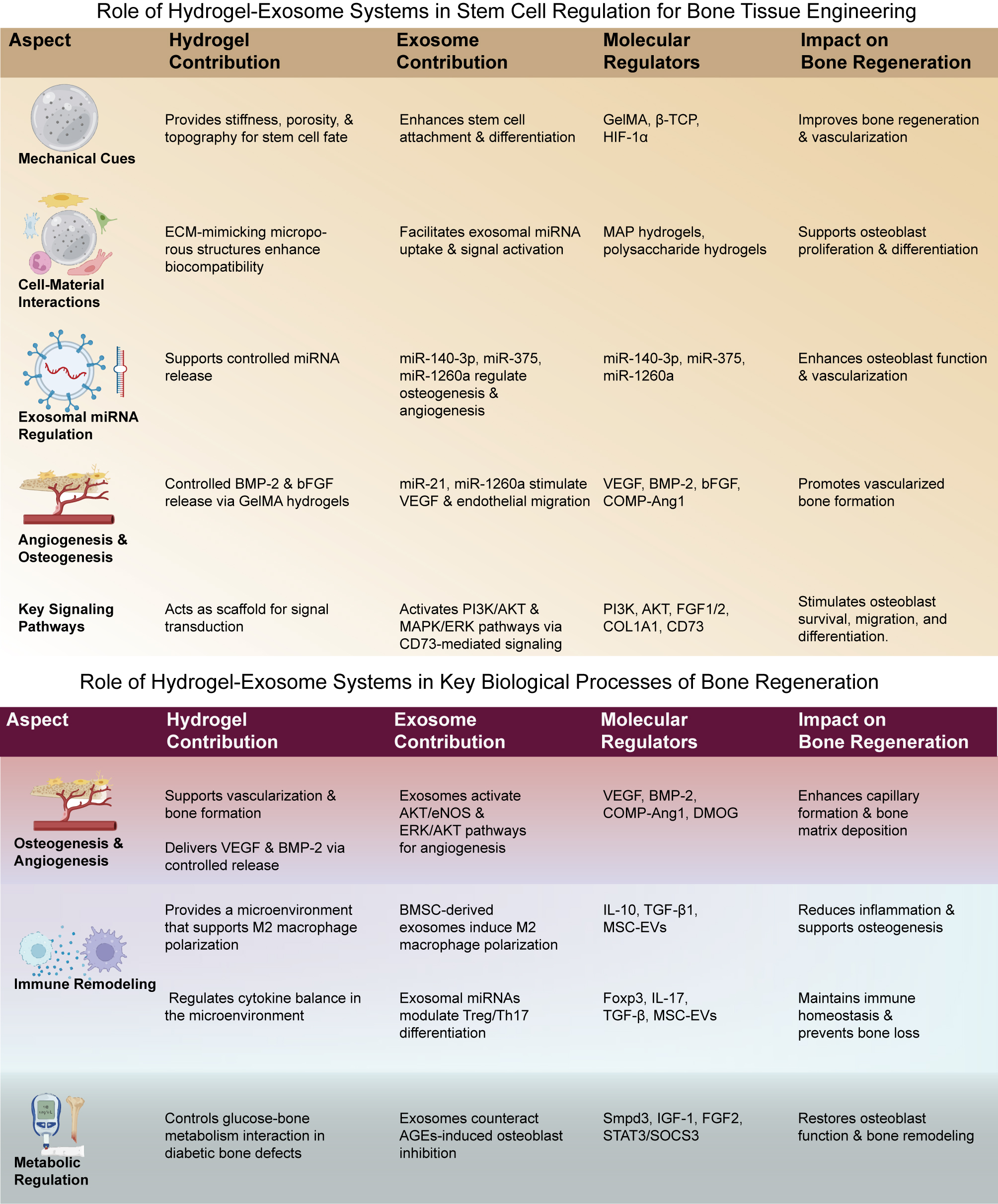
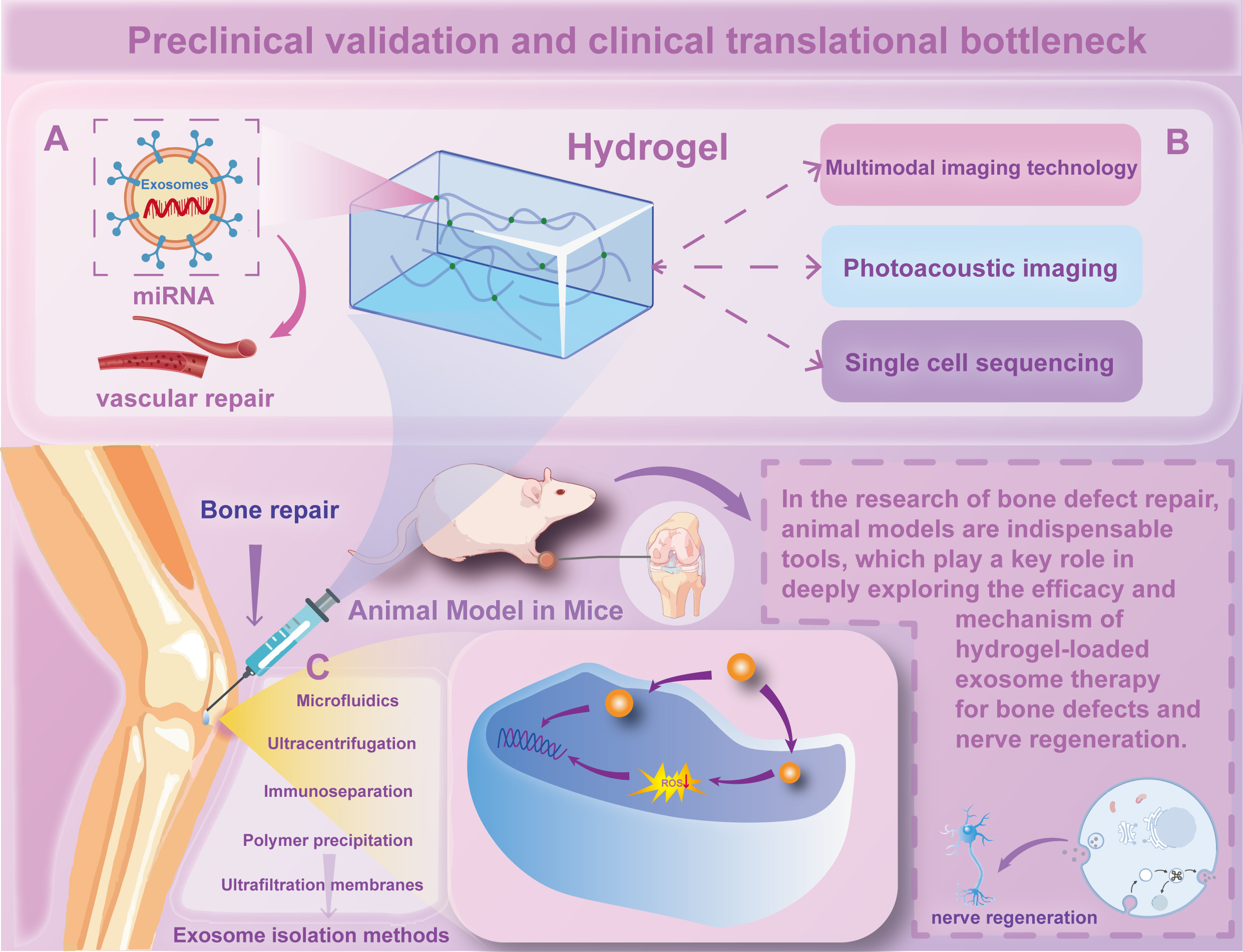
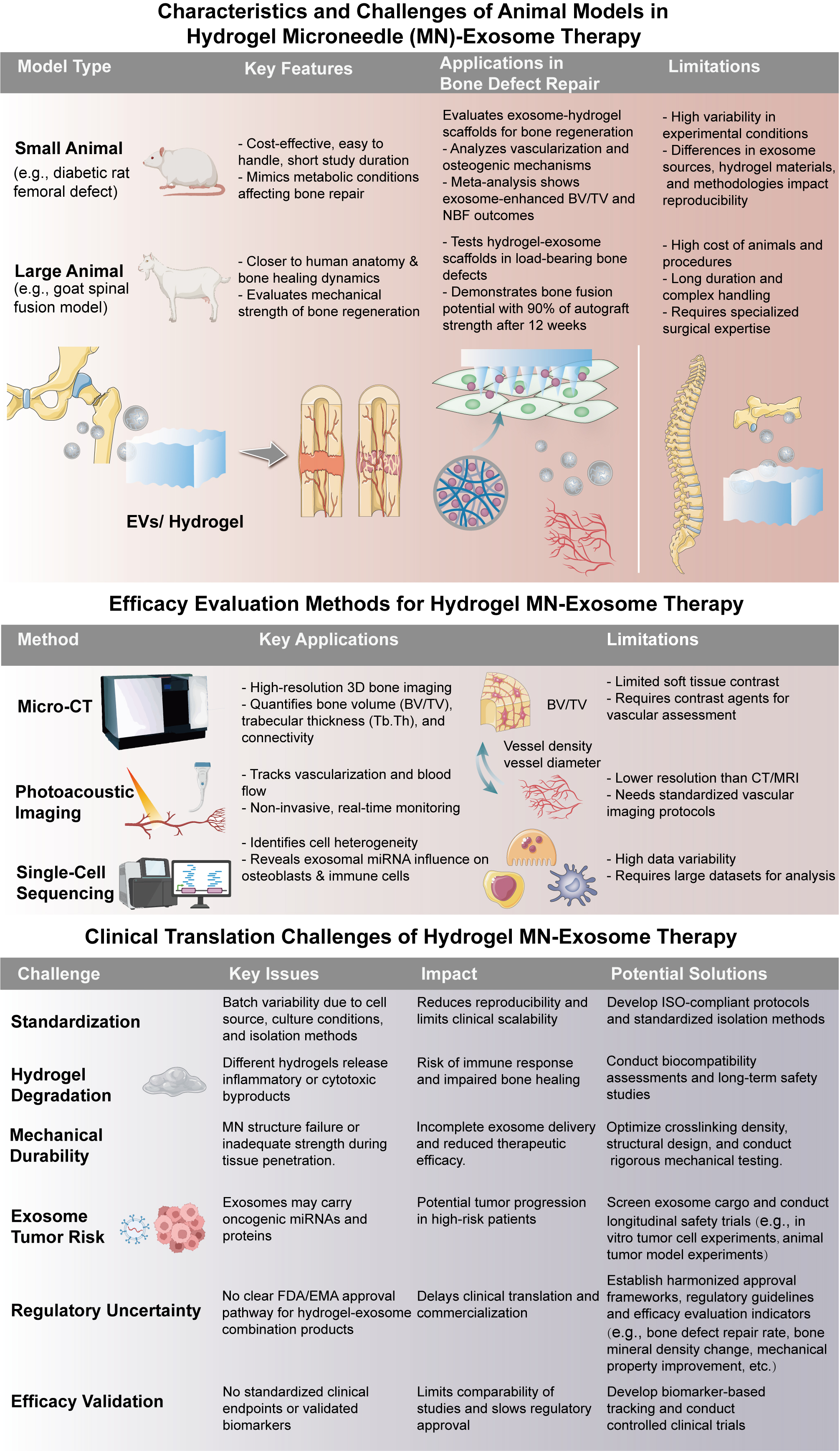
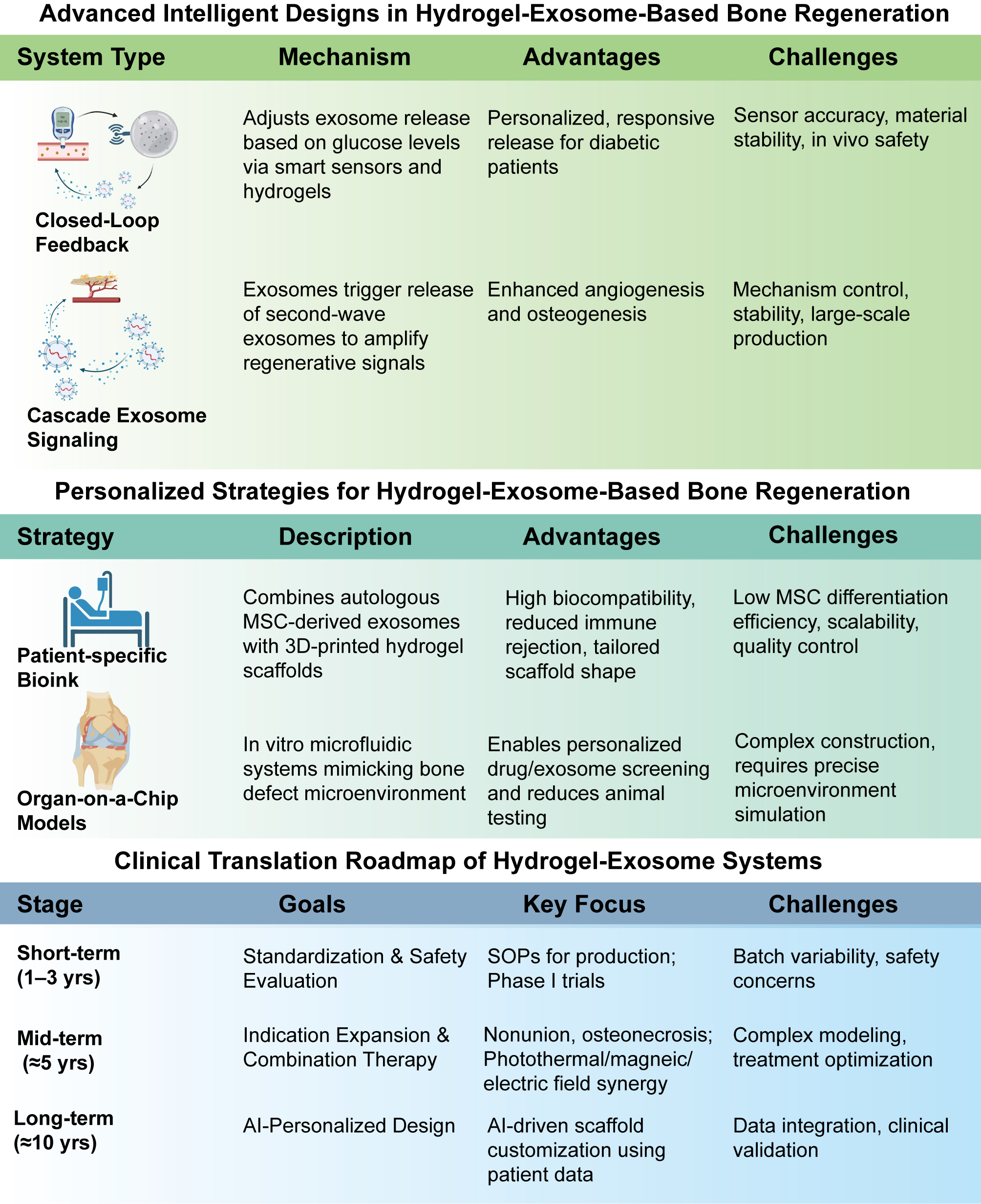
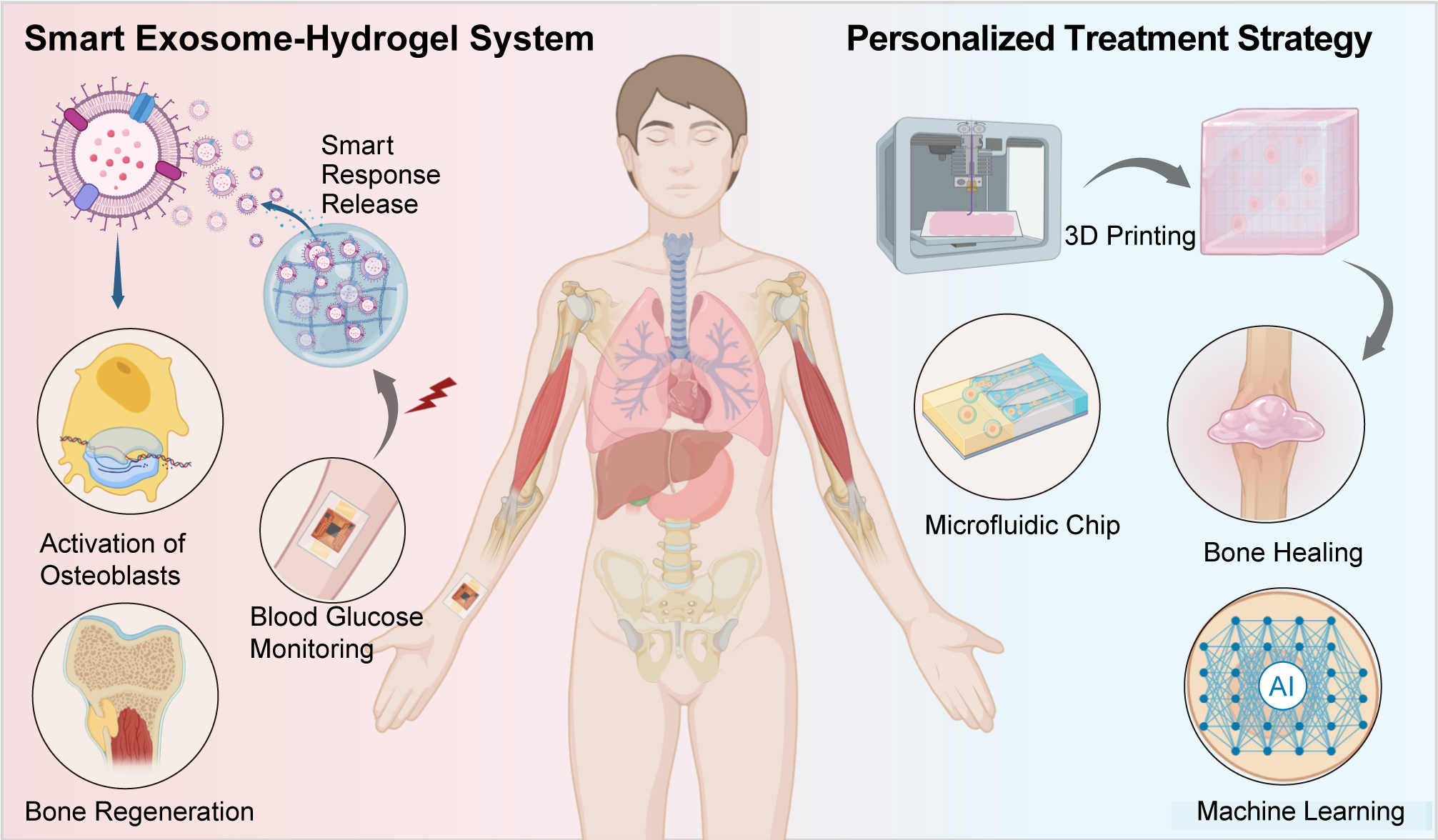












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].