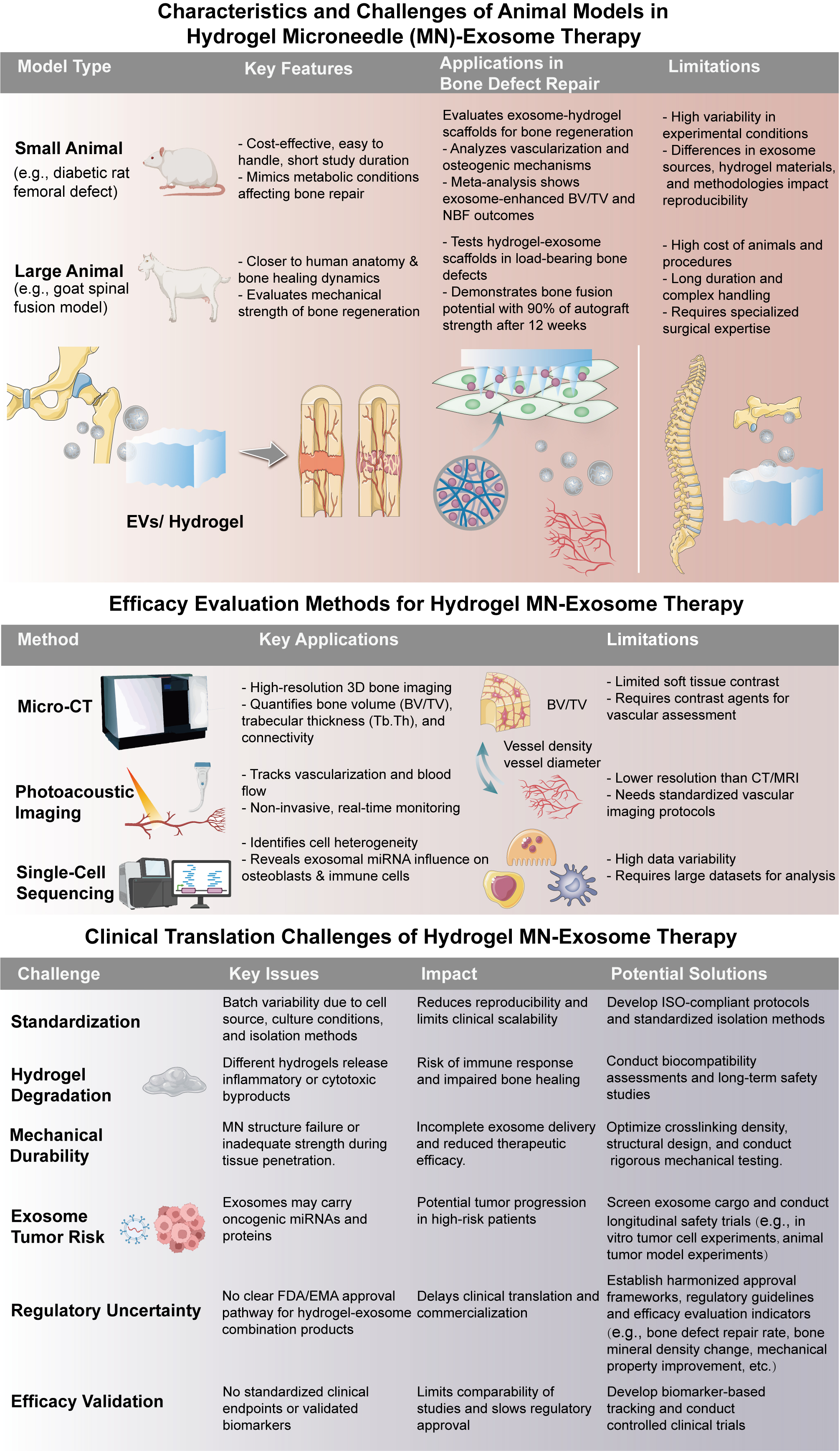
fig10
Figure 10. Challenges and evaluation strategies for hydrogel microneedle (MN)-exosome in bone regeneration. This figure outlines the animal models commonly employed to evaluate the efficacy of these therapies, including both small animal models, such as diabetic rat femoral defects, and large animal models, such as goat spinal fusion models. Small animal models are cost-effective but may not fully replicate human healing processes, while large animals offer better anatomical relevance but involve higher costs and longer study durations. The figure also highlights various evaluation techniques, including micro-CT for 3D bone volume and structure assessment, photoacoustic imaging for real-time monitoring of vascularization, and single-cell sequencing for detailed cellular analysis, offering insights into the effects of exosomes on osteoblasts and immune cells. Additionally, the figure addresses challenges in the clinical translation of hydrogel MN-exosome therapies. Issues such as the standardization of exosome sources and isolation methods, hydrogel degradation leading to inflammatory byproducts, mechanical durability affecting tissue penetration, and potential tumor risks associated with exosomal cargo, such as oncogenic miRNAs, need to be overcome. Furthermore, regulatory uncertainty remains a significant hurdle, as no universally accepted classification currently exists for exosome-hydrogel combination products. To address this, regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have begun issuing draft guidance documents outlining expectations for exosome-based therapies, focusing on source characterization, manufacturing consistency, and safety profiling. Leveraging such guidance can help developers align preclinical and clinical development strategies. Moreover, the absence of standardized efficacy validation protocols also impedes clinical translation. Future directions should include the development of consensus guidelines for potency assays, long-term safety studies, and clinical trial design, potentially coordinated through international consortia or working groups. These efforts, combined with advancements in manufacturing and quality control, are essential for the safe and effective clinical translation of hydrogel-exosome therapies.