Functional metal nanozyme-hydrogel for biomedical applications
Abstract
The metal nanozyme-hydrogel composite system, as a new class of soft intelligent biomaterial, has attracted much attention due to its great potential in simulating the activity of natural enzymes, regulating the pathological microenvironment and achieving synergistic treatment. Metal nanozymes (such as precious metals, transition metal oxides, metal-organic frameworks, etc.) can simulate the activities of various redox enzymes and precisely regulate the homeostasis of reactive oxygen species. The hydrogel matrix provides it with an ideal biocompatible scaffold, a controllable release platform and a macroscopic functional form. The combination of the two endows the composite material with multifunctional synergy. This review systematically summarizes the construction strategies of metal nanozyme-hydrogel systems, with a focus on elaborating the engineering design of nanozymes, the design mechanism of hydrogel matrices, and the application of advanced manufacturing technologies such as three-dimensional printing and injectable gels in achieving macro-structure regulation. Based on this, this article delves deeply into the key applications of this composite system in the biomedical field, including reshaping the tumor microenvironment to enhance combined therapy, comprehensive treatment of infectious wounds (especially diabetic wounds), and achieving anti-inflammatory and repair effects in functional tissues such as osteoarthritis and intestinal inflammation. Finally, we analyzed the core challenges faced by this field and looked forward to its future development direction, aiming to provide new ideas for the design and clinical transformation of the next generation of intelligent biomaterials.
Keywords
INTRODUCTION
Malignant tumors, chronic wounds, and other persistent inflammatory diseases represent severe challenges to global public health[1]. Although their etiologies vary, these diseases are commonly associated with a severe disruption of the local pathological microenvironment (PME) during their progression. This imbalance is primarily driven by the dysregulation of redox signaling pathways, manifesting as disrupted homeostasis of reactive oxygen species (ROS) and consequent oxidative stress[2-4]. Under physiological conditions, a dynamic balance is maintained between cellular ROS production and endogenous antioxidant defense systems[5]. However, under pathological conditions, this homeostasis is disrupted, and dysregulated ROS levels can lead to cell dysfunction or death by directly damaging biological macromolecules such as nucleic acids, membrane lipids, and proteins. This dual nature of ROS, acting as essential signaling mediators at physiological levels yet promoting damage when overproduced, highlights its paradoxical role in biology[6]. More importantly, dysregulated ROS levels can further induce aberrant redox-dependent signal transduction[7-9]. Ultimately, these pathological processes manifest as persistent oxidative stress, chronic inflammation, dysregulated cell death, immune evasion, and a severe impairment of tissue regeneration[2,10].
Crucially, emerging evidence indicates that ROS regulation in therapy should not be regarded as a unidirectional scavenging process. Excessive depletion of ROS may also be detrimental, as basal ROS levels are indispensable for fundamental physiological processes such as cell proliferation, immune activation, angiogenesis, and tissue remodeling. Thus, effective therapeutic intervention requires maintaining ROS concentrations within a disease- and stage-specific therapeutic window, rather than simply minimizing ROS levels. This concept underscores the necessity for dynamic and precisely controlled redox modulation strategies capable of adapting to different pathological phases. Consequently, the precise modulation of the PME and the restoration of redox homeostasis within this optimal range have emerged as key therapeutic targets and important research directions in disease treatment[11].
However, conventional strategies for regulating ROS often fail to fully meet clinical needs[12]. Against this backdrop, nanozymes have emerged as a class of artificial nanomaterials with enzyme-like catalytic activity, capable of catalyzing ROS-related reactions and modulating PME[13]. Since the team of Gao et al. first reported in 2007 that Fe3O4 nanoparticles (NPs) have peroxidase (POD)-like activity[14], the field of nanozymes has attracted considerable research interest and rapidly developed into a prominent research topic at the interface of chemistry, materials science, and biomedicine.
Among the nanozyme family, metal-based nanozymes are one of the most extensively studied and structurally diverse branches. They have attracted much attention due to their advantages such as high catalytic activity, excellent stability, relatively low cost and facile surface functionalization[15]. The core of their catalytic activity lies in the unique and flexible electronic structures and the tunable oxidation states of the metal centers. For instance, transition metal or rare earth metal ions such as iron (Fe), copper (Cu), manganese (Mn), cerium (Ce), etc., can act as redox-active centers, shuttling electrons through Fe2+/Fe3+, Ce3+/Ce4+, Mn2+/Mn4+ redox couples, thereby mimicking the redox functions of the active sites of natural metalloenzymes[16]. These materials not only include traditional noble metals and metal oxides, but also encompass emerging architectures such as metal-organic frameworks (MOFs), Prussian blue (PB) and single-atom nanozymes (SANs). Metal-based nanozymes can, depending on their composition, structure and reaction conditions, mediate either ROS clearance (e.g., anti-inflammatory and antioxidative effects) or ROS generation (e.g., tumor catalytic therapy and antibacterial action) by simulating the activities of multiple enzymes, including superoxide dismutase (SOD), catalase (CAT), POD, and glutathione peroxidase (GPx), thereby contributing to the precise regulation of the PME[17,18].
However, free metal-based nanozymes still face many challenges in complex physiological environments, such as being easily cleared by the reticuloendothelial system, having a limited retention time at the lesion site, being prone to agglomeration and loss of catalytic activity, and having potential biological toxicity. To overcome these obstacles, hydrogel-based systems have emerged as an ideal delivery vehicle and functional matrix[19]. Hydrogels are three-dimensional (3D) cross-linked networks formed by physical or chemical cross-linking of hydrophilic polymers (e.g., polysaccharide-based matrices such as chitosan, sodium alginate and cellulose derivatives), featuring high water content, good biocompatibility, and tissue-like physicochemical properties reminiscent of the natural extracellular matrix (ECM)[20,21].
The combination of metal-based nanozymes and hydrogel matrices is not merely a simple physical encapsulation, but rather a functional synergy and enhancement at both the molecular and macroscopic scales. To achieve this synergy, the rational construction of metal nanozyme-hydrogel composite systems should follow several fundamental design principles. At the molecular level, covalent anchoring of nanozymes to the hydrogel skeleton at the molecular scale through chemical bonds or in situ growth of nanozyme within the network can minimize leakage and enhance catalytic activity through improved substrate accessibility and interfacial charge transfer[22]. More importantly, through rational molecular design, hydrogels can be endowed with stimuli-responsive behavior to specific microenvironmental signals, such as pH, temperature, light and ultrasound[23]. This responsiveness transforms hydrogels from passive carriers or scaffolds into active, intelligent biocatalytic reactors. Consequently, such systems enable the on-demand release and in-situ activation of nanozymes at the lesion site. At the macroscopic scale, metal-based nanozymes endow conventional hydrogels with additional biocatalytic functions, transforming them into active microenvironment regulators. By integrating advanced fabrication technologies such as 3D printing and injectable in situ-forming hydrogels, the macroscopic morphology and microstructure of the metal-based nanozyme-hydrogel composite system can be tailored to meet the complex demands of various clinical scenarios (such as injectable filling, microsphere delivery, customized implantation, and intelligent dressings)[24].
In recent years, research on metal-based nanozyme-hydrogel composite systems has grown rapidly, demonstrating considerable application potential in the biomedical field [Figure 1]. However, current studies in this field remain relatively fragmented, and there is still a lack of reviews that systematically clarify the intrinsic links between construction strategy, functional synergy and clinical application. In view of this, this review systematically summarizes the latest research progress on metal-based nanozyme-hydrogel systems. First, we focus on detailing the construction strategies and functional synergy mechanism within the composite system, covering the engineering design of different types of metal nanozymes, the regulation of hydrogel matrices, and macroscopic structural design enabled by advanced manufacturing technologies. Subsequently, we discuss the application of this system in key biomedical fields, including tumor microenvironment remodeling and combined therapy, comprehensive treatment of infectious wounds, as well as anti-inflammatory and the repair/regeneration of functional tissues (such as bone and the intestine). Finally, we summarize the core challenges currently faced in this field and outline future directions for development and clinical translation.
Figure 1. Schematic illustration of the components, catalytic activity, and stimulus-responsive properties of metal-based nanozyme-engineered hydrogels. CNT: Carbon nanotube; MOF: metal-organic framework; CNE: composite nanozyme; SAzymes: single-atom nanozymes; PEG: polyethylene glycol; PVA: polyvinyl alcohol; ROS: reactive oxygen species; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; POD: peroxidase; GOx: glucose oxidation; UV: ultraviolet.
CONSTRUCTION AND FUNCTION OF METAL NANOZYME-HYDROGEL SYSTEMS
Engineering metallic nanozymes
Metal-based nanozymes are a type of artificial enzyme system that can partially mimic or even approach the functions of natural oxidoreductases. They can precisely regulate the level of ROS through various enzyme-like activities such as SOD, CAT, POD, and GPx.
Precious metal nanozymes
Precious metal nanozymes such as gold (Au), platinum (Pt), palladium (Pd), and ruthenium (Ru) have become widely studied nanozyme systems due to their tunable catalytic activity, high chemical stability, and excellent electronic conductivity[25]. Pt-based nanozymes (such as Pt NPs and Pt-Cu alloys) typically exhibit strong CAT and SOD-like activity, effectively decomposing H2O2 into H2O and O2, thereby alleviating tissue hypoxia and oxidative damage[26]. Au nanozymes are promising antioxidants that have been proven to effectively eliminate ROS in the tumor microenvironment[27].
It is worth noting that ruthenium-based nanozymes exhibit multi-enzyme-like activity and can form a cascade catalytic reaction, thereby achieving therapeutic effects on tumors[28]. Lin et al. constructed multi-enzyme ruthenium nanozyme-hydrogels. In this system, Ru⁰/Ru4+ nanozymes are covalently embedded into the methylpropenylated gelatin network. Ru⁰ is responsible for adsorbing and removing NO, while Ru4+ exhibits excellent catalytic activity in ROS removal [Figure 2A][29]. However, despite the excellent catalytic performance of noble metal nanozymes, their low clearance rate and potential accumulation in circulation and tissues remain the main challenges in long-term biosafety studies[30].
Figure 2. Engineering strategies and functional mechanisms of metallic nanozymes in metal nanozyme-hydrogel systems. (A) Noble Metal Nanozyme: Schematic illustration of the RuNZs’ multienzymatic activities, highlighting NO adsorption at Ru0 sites and ROS scavenging mediated by Ru4+ centers. Reproduced with permission from Ref.[29]. Copyright 2024, Wiley; (B) Metal Oxide Nanozyme: Schematic representation of the ROS-scavenging performance of F/Rgel. Reproduced with permission from Ref.[33]. Copyright 2025, Nature; Figure 2B is reproduced from (Zhang, F., 2025) under the CC BY-NC-ND license. No modifications were made; (C) Bio-Hybrid Nanozyme: Schematic illustration of the preparation of Mito-Fenozyme. Reproduced with permission from Ref.[34]. Copyright 2021, Wiley; (D) Metal-Organic Framework: Schematic illustration of the preparation of the MOF. Reproduced with permission from Ref.[45]. Copyright 2022, American Chemical Society; (E) Single-Atom Nanozyme: Illustration depicting the synthesis process of FeSA@FibBLG. Reproduced with permission from Ref.[47]. Copyright 2024, Nature. ROS: Reactive oxygen species; MOF: metal-organic framework; FeSA@FibBLG: single-iron atom-anchored amyloid hydrogel catalytic platform based on β -lactoglobulin self-assembled fibers; NZs: nanozymes; CAT: catalase; SOD: superoxide dismutase; FTn: ferritin; TPP: triphenylphosphonium; DMF: N,N-dimethylformamide; BLG: β-lactoglobulin; FibBLG: nitrogen-rich site fiber structure; HRP: horseradish peroxidase.
Transition metal oxide nanozymes
Transition metal oxide nanozymes have attracted much attention due to their reversible multivalent state transformation and good biological metabolic compatibility. For instance, CeO2 nanozymes can achieve self-regenerative redox catalysis through adjustable valence states (Ce3+/Ce4+ cycles), thereby maintaining ROS clearance capacity and antioxidant homeostasis for a long time[31,32]. The Zhang team developed a dynamic phase regulation ε -polylysine-CeO2 complex hydrogel (εPL-CeO2 F/R gel), which achieves programmed regulation of the infection and repair phases through a Schiff base dynamic cross-linking network[33]. This system utilizes the reversible electron transfer of Ce3+/Ce4+ of CeO2 nanozyme to rapidly eliminate excessive ROS (including H2O2, •OH and O2•-, etc.) as shown in Figure 2B, significantly alleviating the oxidative stress-inflammatory cycle and restoring the homeostasis of the wound microenvironment. CeO2 nanozyme simultaneously possesses CAT-like activity, capable of decomposing endogenous H2O2 into O2, thereby alleviating hypoxia.
In addition, Zhang et al. constructed mitochondrial-targeted hybrid nanozymes (Mito-Fenozyme) using biomimetic strategies, which also achieved the functionalization of transition metal oxide systems[34]. This platform uses ferritin (FTn) heavy chain as the protein scaffold, generates MnO2 nanoclusters in situ through H2O2 oxidation of Mn2+, and then achieves mitochondrial-specific localization, as shown in Figure 2C. The obtained Mito-Fenozyme has dual enzyme activities of SOD and CAT, and can achieve the cascade transformation from O2•- to H2O2 and then to O2 within cells, significantly reducing mitochondrial oxidative damage during myocardial ischemia-reperfusion. These studies indicate that the catalysis of transition metal oxide nanozymes depends on the valence state transition of metals and the structure of crystal defects. They can not only achieve continuous antioxidation through cycles such as Ce3+/Ce4+ or Mn4+/Mn2+[34], but also realize microenvironment adaptive response by combining with intelligent hydrogel networks. Their excellent biocompatibility and ion metabolism characteristics make them have broad application potential in anti-inflammatory, anti-tumor and other aspects.
Prussian blue and metal-organic frameworks
Prussian blue is a clinically approved ferricyanide complex with inherent CAT, SOD and POD-like activities, which can effectively eliminate various ROS and alleviate inflammatory responses[35,36]. For instance, Prussian blue nanozymes (PB NZs) can effectively reduce ROS levels and inhibit the expression of inflammatory factors in ischemia-reperfusion models[35], and it also has good biocompatibility and safety. In addition, through ion doping (such as Zn, Mn, Cu, Co, etc.) or surface functionalization, its multi-enzyme cascade activity and specific targeting ability can be further enhanced[37].
In contrast, MOFs are a typical class of porous nanomaterials, composed of organic linkers and metal ions[38-40], and have become an ideal platform for constructing artificial enzymes due to their high specific surface area, inherent porosity and highly programmable chemical environment[41,42]. Its unsaturated metal nodes can serve as catalytically active centers, while the functionalized organic ligands and the adjustable pore structure jointly create an efficient and selective reaction microenvironment, which greatly promotes substrate diffusion and electron transfer[43,44]. Against this backdrop, a team designed a MOF-818 nanozyme/hydrogel system (MOF/Gel) with antioxidant oxidase mimicking activity based on the antioxidant properties of the metal-organic framework MOF-818[45]. This system takes the MOF-818 constructed with Zn2+/Cu2+ coordination nodes and organic ligands as the core, and is in situ synthesized at 100 °C to form a highly crystallized octahedral structure. Its metal center simultaneously exhibits SOD-like and CAT-like activities. On the one hand, it catalyzes the disproportionation of O2•- to generate H2O2; on the other hand, it decomposes H2O2 to generate O2 and H2O, thereby achieving dual-channel clearance of ROS [Figure 2D]. In the chronic wound model of diabetes, MOF/Gel can continuously regulate the local oxidative stress level and promote the natural transition from the inflammatory phase to the proliferative phase.
Other novel nanozymes
In recent years, in order to achieve highly selective catalysis like enzymes, the design of novel nanozymes with clear active center structures has become a research hotspot. Among them, SANs and High-Entropy Nanozymes (HEzymes) offer two highly promising strategies. On the one hand, SANs have a clear geometric structure and highly dispersed active sites, making it an ideal model system[46]. For instance, researchers constructed a single-iron atom-anchored amyloid hydrogel catalytic platform based on β -lactoglobulin self-assembled fibers (FeSA@FibBLG)[47]. This system was prepared by a simple wet chemical impregnation method, β-lactoglobulin (BLG) self-assembled at high temperature (90 °C, 5 h) under acidic conditions to form a nitrogen-rich site fiber structure (FibBLG), and then Fe3+ ions were introduced to coordinate with nitrogen atoms to form Fe-N4 active centers [Figure 2E]. This single-atom coordination structure features a stable and uniform electronic distribution, which can effectively simulate the catalytic characteristics of the active center. Animal experiments have shown that after oral administration, the blood alcohol concentration of mice decreased by 55.8% within 300 min, and no acetaldehyde accumulation or liver damage was caused, verifying its excellent biocompatibility[47]. On the other hand, unlike SANs, which enhance selectivity through structural refinement, HEzymes adopt a different strategy, that is, they utilize the complex active interfaces and synergistic interactions composed of multi-metal components to improve catalytic performance. For instance, a study prepared FePtCoNiCu HEzymes by the low-temperature oil phase method and embedded them in a hydrogel matrix to construct a portable and low-cost on-site visual biosensing platform[48]. This type of HEzymes also exhibits strong POD-like activity and can generate hydroxyl radicals by efficiently converting H2O2. Density functional theory calculations show that the strong electronic coupling among its various metal components promotes charge transfer through the D-orbital rearrangement near the Fermi level, thereby significantly improving the catalytic efficiency. Furthermore, HEzymes was fixed in alginate saline gel and combined with smartphone imaging to construct a portable, rapid and visual on-site colorimetric platform, which can be used for sensitive biological monitoring (such as biothiols, acetylcholinesterase, etc.).
The above-mentioned nanozymes each have their unique catalytic advantages and application limitations. Noble metal nanozymes (Au, Pt) typically exhibit high catalytic activity and stability, but the long-term bioaccumulation caused by their high cost and non-degradability is the main obstacle to their clinical transformation. Transition metal oxides (CeO2, MnO2) have shown great potential in the fields of antioxidants and anti-inflammation (such as wound repair) due to their excellent biocompatibility, controllable multivalent cycling and metabolizability[49]. However, their catalytic activity is usually weaker than that of precious metals and is easily affected by local pH and H2O2 concentrations. MOFs and PB analogues offer highly configurable platforms. Their high porosity makes them easy to load other drugs, but their structural stability is a key issue that requires fine regulation. Their stability in the physiological environment is insufficient, and they degrade too quickly. SANs and HEzymes achieve high atomic utilization rate and natural enzyme-like catalytic mechanisms through well-defined active sites and synergistic electron effects, making them ideal models for mechanism research. However, their complex preparation processes and high costs limit their large-scale application.
Therefore, in practical applications, the selection of nanozymes is not about pursuing a single strongest activity, but should be based on a comprehensive consideration of specific scenarios, for instance, acute and short-term antibacterial applications may place more emphasis on catalytic efficiency (such as Pt)[50]; For the long-term regulation of chronic inflammation, priority should be given to biological safety and metabolizability.
Dynamic catalysis of metal nanozymes
With the integration of energy science and materials science, the design of nanozymes has gradually expanded from static catalysis to energy-driven synergistic catalysis systems. By introducing exogenous energy fields such as photothermal (PTT) and photodynamic (PDT), dynamic intervention of the pathological microenvironment can be achieved while regulating the reaction rate and selectivity[51-54]. Recently, Zhang’s team reported an intelligent conductive hydrogel (HEPP) system with multi-responsiveness and anti-inflammatory properties[55]. This hydrogel triggers the release of embedded nanozymes (PTPPG) through the microenvironment, and simultaneously combines the self-cascading regulatory mechanisms of PDT, PTT and ROS to achieve multi-dimensional therapeutic functions. Specifically, PTPPG nanozymes possess activities such as CAT and POD, and can generate thermal and excited-state oxygen through the PDT/PTT effect under light (808 nm), rapidly eliminating pathogens[55]. Meanwhile, its CAT-like activity can decompose H2O2 and continuously release oxygen, effectively alleviating the hypoxia state that is widespread in diabetic wounds.
Hydrogel matrix
Hydrogels are flexible materials with high water content and 3D cross-linked network structures[56]. Through molecular design, they can achieve response regulation to biological signals and precise integration of nanozymes[20,57]. A reasonable network structure can not only effectively protect the activity of nanozymes, prevent agglomeration and inactivation, but also optimize the catalytic efficiency by adjusting the stability of the microenvironment, etc.[19]. In recent years, hydrogel engineering has gradually evolved from simple physical embedding to stimulus response and multi-functional collaborative design[58,59], to match the dynamic reaction characteristics of nanozyme in complex physiological environments[23,60].
There are various types of hydrogels. Common systems include natural polymer systems (such as chitosan, gelatin, hyaluronic acid, alginate and cellulose, etc.) and synthetic polymer systems. Natural hydrogels exhibit excellent biocompatibility and bioactivity, while synthetic hydrogels, composed of polymer monomers, possess well-defined chemical properties and tunable mechanical performance. They have different matrix sources and cross-linking mechanisms[61-63]. This complementary nature motivates the development of hybrid hydrogel systems that integrate the advantages of both.
Beyond serving as conventional carriers, hydrogels act as multifunctional regulatory platforms that fundamentally govern the behavior and performance of metal nanozymes in biological settings. These hydrophilic polymeric networks exert multi-level control over nanozyme function: enhancing substrate diffusion and active-site exposure, maintaining structural integrity and catalytic stability, mediating targeted, responsive retention and release, and sustaining long-term redox reactions. In the subsequent sections, we delineate these key regulatory roles to establish the fundamental rationale for the synergistic design of nanozyme-hydrogel systems.
Enhancing substrate accessibility
Substrate accessibility represents a core determinant of nanozyme catalytic efficiency, and free nanozymes often suffer from limited mass transport, aggregation-induced active-site shielding, and high diffusion resistance in physiological environments[64]. Hydrogels address these limitations by constructing ordered, porous microenvironments that facilitate substrate transport and active-site exposure[65].
Li et al. developed a structured hydrogel (TCH) incorporating the porous organic cage RCC1 for the treatment of fungal keratitis[66]. TCH was formed via condensation between RCC1 and oxidized gellan gum, whose porous architecture creates interconnected channels that reduce diffusion resistance for substrates present in bodily fluids or corneal tissue fluid. This design enables efficient substrate delivery to nanozyme active sites, directly enhancing substrate accessibility. Moreover, the synergistic combination of the RCC1 cage-like motif and the hydrogel 3D network firmly immobilizes and uniformly disperses nanozymes, avoiding aggregation commonly observed in free nanozyme systems. Through spatial confinement, the RCC1-based hydrogel ensures full exposure of nanozyme active sites, fundamentally strengthening substrate-active site contact efficiency and boosting overall catalytic performance.
Responsive release and prolonged in vivo retention
A key advantage of hydrogel-nanozyme systems is spatiotemporal control over nanozyme distribution, which improves local bioavailability, extends therapeutic windows, and reduces off-target toxicity. Responsive hydrogels sense pathological microenvironmental cues to achieve on-demand degradation and controlled nanozyme release, thereby prolonging in vivo retention at target sites[67].
A research team developed a glucose /H2O2 dual-responsive sound-sensitive hydrogel in response to the characteristic of difficult healing of implant infections in diabetic patients[68]. This system uses carboxyphenylboronic acid (PBA) modified chitosan (CS-PBA) as the skeleton. After activation, it forms a borate ester cross-linked network with polyvinyl alcohol (PVA) as shown in Figure 3A. Endow the hydrogel with dual sensitivity to glucose and H2O2. This hydrogel can dynamically degrade and release MnO2 nanozymes and sonosensitists in a high-sugar and high-oxidation environment. Under the action of ultrasound, it triggers the sonodynamic effect, generating ROS to kill bacteria and release antigens. Meanwhile, Mn2+ promotes the maturation of dendritic cells and activates the systemic immune response. This design fully demonstrates the unique advantages of the combination of hydrogel materials and nanozymes in immune regulation and antibioticfree antiinfective therapy achieved via responsive release and prolonged in vivo retention.
Figure 3. Design and fabrication strategies of hydrogel matrices in metal nanozyme–hydrogel systems. (A) Schematic illustration of the fabrication process of the CPA hydrogel. Reproduced with permission from Ref.[68]. Copyright 2024, Elsevier; (B) The synthesis of the supramolecular hydrogel. Reproduced with permission from Ref.[70]. Copyright 2025, Elsevier. CPA: Composite polymeric aquogel; PBA: phenylboronic acid; CS-PBA: carboxyphenylboronic acid modified chitosan; PVA: polyvinyl alcohol; BPQD@MOF: black phosphorus quantum dots@metal-organic framework; NCECS: nitrided chitosan.
Maintaining structural and catalytic stability
As a stabilizing carrier for nanozymes, hydrogels rely on network structural design and intermolecular interaction regulation to maintain nanozyme structural integrity and sustained catalytic activity[69]. For nanozymes with multi-enzyme catalytic activities in particular, their stable catalytic function hinges on the dual stabilization of structure and activity provided by the hydrogel carrier.
The BPQD@MOF multi-enzyme cascade nanozyme hydrogel designed by Li et al. further validates the dual stabilization effects of hydrogel polymer matrices on nanozymes and their application potential in performance regulation[70]. As illustrated in Figure 3B, this system, with PVA, agarose and nitrided chitosan (NCECS) as the main components, constructs a stable and self-healing bone repair matrix through a hierarchically organized hydrogen-bonding network. Strong hydrogen bonds (type I) form a skeletal support structure between NCECS, Agarose, and PVA, while reversible hydrogen bonds (type II) contribute to dynamic self-repairing behavior, and the BPQD@MOF nanozymes are stably immobilized within the hydrogel network through type III interactions with PVA segments, which prevents nanozyme aggregation, leaching and structural collapse at the microscopic level, thus achieving the microstructural stabilization of the nanozymes. Such a hierarchical hydrogen-bonding network not only imparts the material with outstanding mechanical flexibility and ductility, but also effectively maintains the catalytic stability of nanozymes via their stable immobilization and sustained controlled release, avoiding the attenuation of catalytic activity resulting from structural instability or premature release of nanozymes. This multi-network structure endows the material with excellent mechanical flexibility and ductility, and enables the continuous release of nanozymes. BPQD@MOF simultaneously possesses multi-enzyme activities such as PTT, glucose oxidation (GOx), and ROS clearance (SOD/CAT), achieving local glucose consumption, oxygen generation, and ROS clearance, thereby promoting M2 polarization and osteogenic regeneration of macrophages. Through the precise structural design of the hydrogel polymeric matrix, this system realizes the dual regulation of nanozymes’ structural stability and catalytic stability. It provides a multi-dimensional stabilized catalytic therapeutic strategy for diabetic bone defect repair, and further corroborates the core regulatory value of hydrogels in nanozyme stabilization applications.
Sustaining the redox cycle
Redox cycle is one of the core regulatory mechanisms for metal nanozyme-hydrogel systems to achieve long-acting and precise therapy. Hydrogels provide a confined, adaptive microenvironment that supports reversible metal valence transitions, accelerates electron transfer, and sustains closed-loop redox cycling for overcoming single-turnover catalytic limitations[71,72].
Huang et al. established the FDPF thermosensitive hydrogel system centered on Fe-Co bimetallic nanozymes[73]. This system constructs a closed-loop redox cycle through the dynamic valence state transitions of Fe2+/Fe3+ and Co2+/Co3+: H2O2 in the bacterial microenvironment binds to Co2+, triggering the oxidative transition of Co2+/Co3+ and driving the decomposition of H2O2 to generate •OH. Meanwhile, Co3+ is reduced back to Co2+ via electron transfer with Fe ions, and the synchronous valence state transition of Fe ions synergistically accelerates electron transfer to ensure the continuous operation of the cycle. The thermosensitive hydrogel forms a spatially confined network through gelation at 37 °C, stably immobilizing nanozymes to avoid aggregation or leakage of metal active sites. It also adapts to the acidic microenvironment of infected sites, providing favorable reaction conditions for valence state transitions. Combined with the photothermal effect of polydopamine (PDA) under 808 nm near-infrared (NIR) irradiation, the efficiency of electron transfer and cycle intensity are further enhanced. The •OH continuously produced by this redox cycle, synergistically with NO derived from D-Arg, achieves efficient treatment of subcutaneous abscesses and bacterial corneal ulcers. The hydrogel provides a suitable microenvironment for the valence cycle of metal nanozymes, breaks the limitation of single-turnover catalysis, continuously drives the redox cycle, and sustains the catalytic activity of nanozymes in the long term.
Advanced manufacturing for macro-structural control
In order to enable the nanozyme-hydrogel system to perform optimally in complex physiological environments, it is crucial to precisely regulate its macroscopic structure. Advanced manufacturing technologies (such as 3D printing and injectable in-situ gels) can achieve geometric customization of materials, optimization of pore structure and construction of functional gradients at the macroscopic scale, thereby meeting the needs of different tissue repair and disease treatment.
3D printing
3D printing technology features high flexibility and controllability[74]. Through algorithm design and layer-by-layer addition of manufacturing techniques[75], it enables the highly controllable construction of nanozymes and hydrogel precursors in space, providing programmable morphological and functional regulation capabilities for composite materials[76-78].
For instance, Jin’s team developed an antioxidant nanozyme-hepatocyte spheroid 3D composite scaffold (HS/N-Au@composite) for the treatment of acute liver failure[79]. The system prepared hyaluronic acid/gelatin/sodium alginate hydrogel loaded with NAC-modified gold nanoclusters (NAC-Au NCs) by 3D printing technology (N-Au@hydrogel) and combined with biological hydrogel-encapsulated hepatocyte spheroids containing acellular liver matrix (dECM), thrombin and fibrinogen (HS@dECM) [Figure 4A]. The obtained HS/N-Au@composite not only provides biomimetic mechanical support and cell adhesion microenvironment, but also significantly alleviates ROS-induced liver necrosis through the antioxidant and anti-inflammatory effects of Au nanozyme, promoting the functional recovery of transplanted cells and liver regeneration. This strategy has broken through the bottlenecks of low cell utilization and immune rejection in traditional liver transplantation, providing new ideas for stem cell therapy and organ regeneration.
Figure 4. Advanced manufacturing strategies for metal nanozyme-hydrogel systems. (A) Schematic illustration of the preparation of the HS/N-Au@ composite system for the treatment of acute liver failure. Reproduced with permission from Ref.[79]. Copyright 2025, Elsevier; (B) Schematic representation of the surgical procedure. Reproduced with permission from Ref.[80]. Copyright 2025, Elsevier; (C) Schematic illustration of the composition and formation mechanism of the oriented CuTA-CMHT hydrogel. Reproduced with permission from Ref.[81]. Copyright 2024, Elsevier; Figure 4C is reproduced from (Dong, L., 2024) under the CC BY-NC-ND license. No modifications were made; (D) In situ injection of MS@MCL into a rat model to induce local lactate depletion and promote NP regeneration. Reproduced with permission from Ref.[84]. Copyright 2022, Elsevier; Figure 4D is reproduced from (Shen, J., 2022) under the CC BY-NC-ND license. No modifications were made; (E) Fabrication and construction of the heterogeneous SponGel MS, which is inspired by the natural cartilage matrix. Reproduced with permission from Ref.[85]. Copyright 2025, Wiley. CuTA-CMHT: Copper-tannic acid nanozyme and hyaluronic acid-tannic acid composite with carboxymethyl cellulose-methacrylate; MS@MCL: MnO2-Chitosan-LOX (MCL) nanozyme was abundantly coupled to the porous microspheres via condensation reaction; NP: nanoparticle; SponGel MS: injectable chondroid heterogeneous biphasic microsphere; NAC: N-acetyl-L-cysteine; 3D: three-dimensional; hADMSCs: human adipose-derived mesenchymal stem cells; HLCs: hepatocyte-like cells; PDMS: polydimethylsiloxane; dECM: decellularized extracellular matrix; HS@dECM: hepatocyte spheroids@decellularized extracellular matrix; UV: ultraviolet; IVDD: intervertebral disc degeneration; NPCs: nucleus pulposus cells; SDF-1: stromal cell-derived factor 1; EGCG: epigallocatechin gallate.
Similar concepts have also been applied to the repair of intestinal defects and the treatment of corneal ulcers. For example, Su et al. prepared GDMA/Prussian Blue nanozyme wet adhesion hydrogel (GDMA@PB hydrogel) by 3D bioprinting[80]. This material has both strong wet adhesion and immune regulation capabilities, and can closely adhere and achieve anti-inflammatory repair in intestinal perforation models, as shown in Figure 4B. Prussian Blue nanozyme endows it with SOD and CAT-like activities, promotes epithelial regeneration and alleviates tissue inflammation by inducing M2-type polarization of macrophages. Furthermore, the directional structure CuTA-CMHT hydrogel proposed by the Dong team utilized high-precision digital lithography (DLP) to achieve controllable manufacturing of micro-groove array structures, as shown in Figure 4C[81]. This oriented hydrogel simulates the orthogonal arrangement of collagen fibers in the corneal stroma, significantly enhancing the cell adhesion, migration and orientation regeneration capabilities. The integrated CuTA nanozyme not only inhibits bacteria and eliminates ROS, but also regulates the TGF-β and NF-κB signaling pathways, thereby preventing scar formation and achieving functional corneal repair. These studies demonstrate that 3D printing technology can achieve precise macroscopic geometric construction and microscopic functional partitioning in nanozyme-hydrogel systems, providing the possibility for customized regeneration of complex tissues.
Injectable and in-situ gelation systems
For deep or complex geometric tissue injuries, injectable and in situ gel systems offer a minimally invasive and environmentally adaptive solution. Such systems are usually injected in the form of sols or microspheres and can achieve rapid gelation in vivo through temperature, pH, light or ion triggering, forming a 3D support network of adhesion without damaging the surrounding tissues[60,82,83].
For instance, the Shen team constructed an MnO2-lactate oxidase (LOX) nanozyme functionalized microsphere (MS@MCL) with injectable and long-lasting metabolic regulation capabilities[84]. The system uses microfluidic technology (microfluidic fabrication) to prepare uniform-sized and pore-adjustable hyaluronic acid methacrylate (HAMA) microspheres as the matrix, achieving precise control of morphology, structure and rheological properties. A dynamic catalytic microreactor with both oxygen production and lactic acid consumption functions was constructed by firmly immobilizing MnO2-LOX composite nanozyme on the surface of microspheres through chemical bonding. Thanks to the fluidity and plasticity endowed by microfluidic control, this material is in a fluid state before injection and can be directly injected into complex or narrow defect areas through fine needles. It rapidly disperses in the body and adheres to the interface of surrounding tissues, forming a uniformly distributed functional microenvironment as shown in Figure 4D. After injection, LOX catalyzes the oxidation of local lactic acid to pyruvate and H2O2, while MnO2 further catalyzes the decomposition of H2O2 to produce O2, thereby forming a lactic acid-oxygen self-circulation system, achieving long-term metabolic correction in an inflammatory microenvironment of hypoxia and high lactic acid.
Furthermore, Feng et al. developed an injectable chondroid heterogeneous biphasic microsphere (SponGel MS), whose unique structure simultaneously possesses flow injectability and in situ tissue shaping ability [Figure 4E][85]. This material is composed of a stable sponge-like scaffold phase and a dynamic hydrogel phase, among them, the hydrogel phase is cross-linked through Schiff base reaction, which can be gradually degraded in vivo and dynamically release MnO2@EGCG nanozyme and chemokine SDF-1, thereby achieving continuous anti-inflammatory effect and stem cell recruitment after injection; The sponge phase is prepared through low-temperature free radical reactions, forming a support network with high porosity and mechanical toughness, providing a stable scaffold for cell adhesion, aggregation and cartilage differentiation. Thanks to its excellent injectable property, SponGel MS can directly fill complex-shaped cartilage defect sites through minimally invasive methods. In animal models, it effectively inhibits inflammatory responses (down-regulation of IL-1β and COL-1 expression) and promotes the regeneration and integration of hyaluronic cartilage, demonstrating significant application potential in cartilage tissue repair.
3D printing and injectable gels represent two distinct yet complementary clinical translational strategies. The core advantage of 3D printing (especially high-precision technologies such as DLP) lies in its unparalleled structural controllability, which enables the manufacturing of complex scaffolds with bionic microstructures (such as micro-grooves) and functional zoning[86]. It is an ideal choice for achieving personalized customization of implants (such as corneal and cartilage scaffolds). However, its limitations lie in the fact that the manufacturing process is usually layer-by-layer and relatively slow, which imposes strict requirements on the rheological properties of bio-inks and has poor adaptability to deep or irregular tissue defects. In contrast, injectable in situ gel systems (such as microspheres and thermosensitive gels) have significant advantages of being minimally invasive, easy to operate, and capable of closely adhering to irregular defect shapes through flow, making them the preferred choice for treating deep tissue injuries (such as myocardial infarction and bone defect filling). However, the main challenge lies in the fact that the in-situ formed gel network usually has insufficient mechanical strength (such as stiffness and toughness), and its final morphology and internal pore structure are difficult to be precisely controlled in vivo. At the same time, the uncertainty of crosslinking degree before and after injection may lead to problems such as syringe blockage or precursor diffusion[75]. In the future, synergistically integrating the two techniques could compensate for their respective limitations, thereby advancing the clinical translation of composite systems.
APPLICATION OF METAL NANOZYME-HYDROGELS IN THE BIOMEDICAL FIELD
Tumor microenvironment remodeling and combination therapy
The inherent aberrant physicochemical characteristics of the tumor microenvironment (TME), including hypoxia, mild acidosis, elevated levels of H2O2, and glutathione (GSH), are key factors contributing to the failure of conventional therapies such as radiotherapy, chemotherapy, and PDT, as well as to tumor immune evasion[87-93]. These factors collectively establish a robust defensive barrier that significantly compromises treatment efficacy and shelters tumor cells. To overcome this challenge, metal-based nanozyme-hydrogel hybrid materials have emerged. Leveraging their diverse and unique catalytic activities, these systems enable precise intervention and multi-targeted remodeling of the TME.
Hypoxia reversal and reactive ROS storm
Hypoxia is a prevalent characteristic of solid tumors, primarily caused by aberrant vasculature resulting from rapid tumor proliferation. This oxygen-deficient condition significantly compromises the efficacy of various therapeutic modalities, including radiotherapy and PDT[93]. To address this limitation, hydrogels incorporated with metal-based nanozymes exhibiting CAT-like activity, such as copper selenide NPs or MOFs, can sustainably decompose endogenous H2O2 abundantly present in the TME to generate oxygen in situ, thereby effectively alleviating tumor hypoxia. This process not only directly enhances the outcomes of oxygen-dependent therapies like radiotherapy and PDT but also supplies molecular oxygen as a substrate for subsequent catalytic reactions. Furthermore, by co-loading nanozymes with POD-like activity within the same hydrogel system, chemotherapy-derived or exogenously supplied H2O2 can be utilized to initiate chemodynamic therapy (CDT), generating highly toxic hydroxyl radicals (•OH) that induce tumor cell death[94-96].
To overcome the limitations posed by hypoxia in solid tumors, Li et al. devised a synthesis strategy utilizing hemin as a precursor to fabricate axially chlorinated single-atom Fe/N-doped carbon nanodots (Cl-FeNCDs) via a one-pot solvothermal approach[97]. The resulting Cl-FeNCDs exhibit type I photodynamic properties, capable of efficiently generating superoxide anion radicals (O2•-) under 625 nm light irradiation even under hypoxic conditions. Notably, Cl-FeNCDs demonstrate a neutrophil-mimicking enzyme cascade functionality, encompassing SOD-, POD-, and myeloperoxidase-like activities. The photoinduced O2•- effectively triggers subsequent enzymatic cascades, addressing the constraint of substrate scarcity in oxygen-deprived tumor microenvironments. For efficient delivery, the team encapsulated Cl-FeNCDs into bovine serum albumin (BSA), forming biocompatible nanocomplexes (CDs@BSA), which were further incorporated into a photocrosslinkable and injectable hydrogel system. This hydrogel facilitates localized retention and activation within the postsurgical cavity, enabling effective suppression of triple-negative breast cancer (TNBC) recurrence through a topically applied, neutrophil-inspired photo-enzymatic cascade mechanism [Figure 5A]. In the postoperative triple-negative breast cancer model study, CDS@BSA-GEL was demonstrated to not only effectively eliminate residual tumor cells but also reverse the immunosuppressive microenvironment via immune stimulation triggered by immunogenic cell death (ICD), thereby significantly inhibiting tumor recurrence and prolonging survival. This strategy offers novel perspectives for oxygen-dependent anticancer therapies and paves the way for promising approaches in postoperative immunotherapy to prevent tumor recurrence, highlighting considerable potential for clinical translation [Figure 5B-D].
Figure 5. Tumor microenvironment remodeling and combination therapeutic strategies enabled by metal nanozyme-hydrogel systems. (A) Schematic Illustration of a Photoenzymatic Cascade Therapy (PECT): Carbon Nanodot-Based Hydrogel for Preventing Tumor Recurrence after Surgery; (B) Body weight, tumor volume, and survival curves after different treatments. ****P < 0.0001; (C) Representative photographs of recurrent tumors after different treatments and H&E and TUNEL staining at the 24th day; (D) Timeline for treatment and recurrence monitoring following tumor resection. This figure quoted with permission from Ref.[97]. Copyright 2025, American Chemical Society. Cl-FeNCDs: Axially chlorinated single-atom Fe/N-doped carbon nanodots; BSA: bovine serum albumin; CDs@BSA: encapsulation of Cl-FeNCDs in bovine serum albumin yields stable nanocomplexes; PDT: photodynamic; POD: peroxidase; MPO: myeloperoxidase; ROS: reactive oxygen species; GSDME: gasdermin E; CTL: cytotoxic T lymphocyte; DC: dendritic cell; DAMPs: damage-associated molecular patterns; + L: 625 nm light irradiation; H&E: Hematoxylin and Eosin; TUNEL: terminal deoxynucleotidyl transferase dUTP nick end labeling.
In a similar approach, Xu et al. developed an injectable self-healing hydrogel with pH-responsive properties, integrating gold NP-decorated titanium/iron bimetallic organic framework tetragonal nanosheets (Au/TF-MOF TNS) and the chemotherapeutic agent doxorubicin (DOX) for synergistic sonodynamic/chemo dynamic/starvation/chemo-therapy (SDT/CDT/ST/CT)[95]. Under the acidic tumor microenvironment, the hydrogel enables controlled release of both Au/TF-MOF and DOX. Au/TF-MOF functioned as nanozymes, producing O2 to alleviate tumor hypoxia, initiating the Fenton reaction to generate ∙OH for CDT, and depleting intracellular glucose to achieve starvation therapy. Furthermore, Au/TF-MOF acted as sonosensitizers, converting ambient O2 into singlet oxygen (1O2) upon ultrasound irradiation, thereby elevating intracellular oxidative stress and triggering apoptosis. Both in vitro and in vivo antitumor studies demonstrated that the nanocomposite hydrogel effectively suppressed tumor growth and achieved highly efficient synergistic therapeutic outcomes.
Metabolic intervention and immune activation
High concentrations of GSH serve as vital antioxidants within tumor cells, efficiently scavenging therapy-induced ROS, and are a key factor in resistance to CDT and chemotherapy[98]. Metal nanozymes with GSH-depleting capabilities [such as Cu-Fe3O4 nanoclusters (NCs), in which Fe2+ consumes GSH] can effectively disrupt the antioxidant defense system of tumor cells, thereby amplifying the ROS-mediated therapeutic effects. To achieve localized and sustained metabolic intervention within the tumor microenvironment, Zhang et al. dispersed Cu-Fe3O4 nanoclusters and artesunate (AS) into sodium alginate solution, followed by the addition of Ca2+ to induce the formation of a 3D porous network structure, constructing an injectable hydrogel capable of in situ stabilization[99]. Delivery of Cu-Fe3O4 nanoclusters through the hydrogel enables localized and sustained release of Cu2+ and Fe2+/Fe3+. Intracellular accumulation of Fe2+ promotes lipid peroxidation and GSH depletion, ultimately leading to inactivation of GPx, impairing the cell’s ability to clear ROS and thereby initiating ferroptosis. DCFH-DA fluorescence staining demonstrated significantly elevated ROS levels in tumor cells treated with NCs, inducing mitochondrial membrane potential depolarization (JC-1), GSH depletion (ThiolTrackerTM Violet), and extensive apoptosis, as observed by a cell apoptosis staining kit (Annexin V-FITC/PI) and flow cytometry. In vivo experiments involved in situ injection of the hydrogel into a murine osteosarcoma model, which significantly inhibited tumor growth and prolonged mouse survival. Compared with the control group, the NCs-AS-ALG group showed a marked reduction in tumor volume and significantly improved survival rates[99]. Histological staining (H&E, TUNEL, Ki67) revealed that the hydrogel could induce tumor cell apoptosis and inhibit proliferation. Immunofluorescence staining of tumor tissues further confirmed elevated ROS levels and activation of ferroptosis-associated molecular mechanisms in vivo, consistent with in vitro findings.
In addition to the direct tumoricidal effects achieved via CDT and chemotherapy, recent studies indicate that localized immunomodulation plays a pivotal role in enhancing antitumor responses[100]. Miao et al. developed an injectable hydrogel (CFe/βCP+Cis hydrogel) with excellent tumor-site retention, controlled cisplatin release capability, and significant tumor immune microenvironment remodeling potential[101]. The researchers co-dispersed porous single-atom iron-carbon nanozymes (CFe) and cisplatin into a β-CD-g-PEGMA (βCP) polymer brush hydrogel network[101]. This system effectively reversed the immunosuppressive state by promoting the polarization of tumor-associated macrophages from the M2 phenotype to the M1 phenotype. Flow cytometry and immunohistochemistry results demonstrated a significant decrease in CD206+ (M2 marker) cell populations and an upregulation of CD86+ (M1 marker), while CD8+ cytotoxic T lymphocyte infiltration into the tumor was markedly enhanced, indicating activation of an adaptive immune response. Further investigations revealed that bone marrow-derived macrophages (BMDMs) treated with the CFe/βCP hydrogel secreted significantly elevated levels of proinflammatory cytokines such as TNF-α and IL-6[101]. Moreover, their conditioned medium effectively inhibited tumor cell viability, suggesting that this material can mediate indirect antitumor effects via immune cell activation. To comprehensively evaluate the in vivo antitumor efficacy of the CFe/βCP+Cis hydrogel, the researchers established a subcutaneous CT26 tumor-bearing BALB/c mouse model[101]. Fourteen days after initiation of treatment, a single local injection of the CFe/βCP+Cis hydrogel (Group G6) achieved sustained tumor growth inhibition. Compared with monotherapy groups receiving only CFe (Groups G3/G4) or the hydrogel matrix alone (Group G2), the CFe/βCP group (G5) exhibited significant tumor control, with a tumor inhibition rate as high as 68%, greatly exceeding that of the untreated group (G1) or single-agent control groups[101].
Comprehensive treatment of infected wounds
Treatment strategies for infected diabetic wounds
Diabetes mellitus is a metabolic disorder characterized by elevated blood sugar levels[102,103]. The healing of normal skin wounds goes through four stages in sequence: hemostasis, inflammation, proliferation and remodeling. However, in the diabetic state, this process is significantly hindered by the combined effect of persistent hyperglycemia, inflammatory imbalance and oxidative stress, resulting in typical chronic and difficult-to-heal wounds[104]. The core pathology lies in the fact that immune dysregulation and oxidative damage-driven inflammation block the physiological transition to the proliferative phase[105]. Therefore, the treatment strategy needs to be precisely intervened in stages according to the healing course to restore the homeostasis of the microenvironment and promote functional regeneration.
In the early stage of infection, inhibiting pathogen proliferation and eliminating excessive ROS are the keys to breaking the vicious cycle of inflammation[4,106,107]. For instance, the Tu team designed a multifunctional MnO2 nanozyme composite hydrogel (PPGA)[108], which is composed of polyethylene glycol methacrylate - glycidyl methacrylate -acrylamide copolymer (PEGMA-GMA-AAm) cross-linked with highly branched polylysine-modified MnO2 nanosheets. Further loading of pravastatin sodium to promote NO production. This system can simultaneously eliminate multiple ROS, continuously supply oxygen and release NO, achieving a synergistic effect of antibacterial, anti-inflammatory and immune regulation. In the diabetic skin wound model infected with methicillin-resistant Staphylococcus aureus (MRSA), PPGA significantly downregulated the levels of IL-1β, IL-6 and TNF-α, and induced macrophages to polarize to M2 type, thereby accelerating inflammation regression and tissue regeneration [Figure 6A]. Similarly, the zinc-based polymetallic oxonate nanozyme (Zn-POM)-based nanozyme functionalized hydrogel proposed by Pu et al. catalyzes the oxidation of glucose to gluconic acid and H2O2 through GOx, while Zn-POM acts as a CAT/SOD mimicase to further decompose H2O2 and eliminate ROS. Achieve precise intervention in the high-sugar microenvironment [Figure 6B][109].
Figure 6. Multifunctional treatment strategies for chronic infected wounds based on metal nanozyme-hydrogel systems. (A) An antibacterial hydrogel integrating ROS scavenging, O2 generation and NO release eradicates MRSA infection, relieve inflammation and promotes healing in diabetic wounds. Reproduced with permission from Ref.[108]. Copyright 2022, Elsevier; (B) Schematic diagram of producing H2O2 and scavenging ROS. Reproduced with permission from Ref.[109]. Copyright 2024, Springer; Figure 6B is reproduced from (Pu, C., 2024) under the CC BY-NC-ND license. No modifications were made; (C) Schematic illustration of PdNPs extraction from the traditional Chinese medicine Bletilla striata, preparation of PDAs, and their subsequent microfluidic encapsulation into HAMA hydrogel microcarriers. Reproduced with permission from Ref.[111]. Copyright 2025, Springer; Figure 6C is reproduced from (Che, J., 2024) under the CC BY-NC-ND license. No modifications were made; (D) PdNP + PDA@HAMA hydrogel microcarriers markedly enhance diabetic wound healing compared with controls, as evidenced by faster wound closure and improved histological regeneration on H&E and Masson’s staining (12 rats each group). Reproduced with permission from Ref.[111]. Copyright 2025, Springer; Figure 6D is reproduced from (Che, J., 2024) under the CC BY-NC-ND license. No modifications were made; (E) Multifaceted functions of the MZGB hydrogel during diabetic wound healing. Reproduced with permission from Ref.[112]. Copyright 2025, Elsevier; (F) Dynamic, phase-adaptive regulatory roles of the gel in promoting scarless wound healing. Reproduced with permission from Ref.[33]. Copyright 2025, Nature. ROS: Reactive oxygen species; MRSA: methicillin-resistant Staphylococcus aureus; NPs: nanoparticles; PDAs: polydopamines; HAMA: hyaluronic acid methacrylate; H&E: Hematoxylin and Eosin; MZGB: Mn-ZIF@GOx/BC; HBPL: hyperbranched poly-L-lysine; PEGMA-GMA-AAm: polyethylene glycol methacrylate - glycidyl methacrylate -acrylamide copolymer; IL: interleukin; CXCL: Chemokine (C-X-C motif) ligand; TNF: tumor necrosis factor; CAT: catalase; SOD: superoxide dismutase; PBS: phosphate buffered saline; HE: Hematoxylin and Eosin; ATP: adenosine triphosphate; MCs: mast cells.
After the inflammation and oxidative stress of the wound are initially controlled, the treatment goal shifts to promoting a smooth transition from the inflammatory phase to the proliferative phase. The core of this stage lies in reshaping the immune microenvironment (such as inducing macrophage polarization) and providing support for cell proliferation[45,110]. For instance, Che et al. used the extract of the traditional Chinese medicine Bletilla striata as a reducing agent and stabilizer to green synthesize active Pd NPs, and co-embedded them with PDA in HAMA hydrogel. The PdNP+PDA@HAMA complex microcarrier system [Figure 6C][111] was constructed. This system can effectively eliminate ROS, regulate the macrophage phenotype, restore fibroblast function, and has shown excellent pro-healing effects in animal experiments. In the full-thickness wound model of diabetes in mice, as shown in Figure 6D-i, compared with the PBS control group and the single group containing only HAMA, PdNP or PDA, the wound healing ability of PdNP+PDA@HAMA complex microvector was outstanding, and the healing rate on the 9th day was significantly better than that of PBS and the single component control [Figure 6D-ii]. Histological analysis revealed that HE staining showed that in the PdNP+PDA@HAMA group, granulation tissue was most closely connected to the surrounding skin, and regeneration was obvious; Mason tricolor staining further revealed that this group had the richest collagen deposition and orderly fiber arrangement, thereby accelerating tissue repair and improving the structure and function of the skin after healing [Figure 6D-iii]. In addition, some research teams have further combined redox homeostasis with mitochondrial function repair. For instance, the Yan team developed the Mn-ZIF@GOx/BC (MZGB) hydrogel system[112]. This system uniformly disperses the Mn-ZIF@GOx (MZG) nanozyme with SOD/CAT cascade activity in the bacterial cellulose (BC) matrix, and forms a stable composite network through hydrogen bonds and electrostatic interactions. It utilizes glucose oxidation in the body to produce gluconic acid, reducing local pH, while decomposing excessive ROS and providing O2, thereby achieving redox homeostasis and metabolic self-regulation, as shown in Figure 6E. MZGB not only restores mitochondrial function and ATP levels, but also promotes angiogenesis and epithelial regeneration by inducing M2-type polarization, and demonstrates ROS-independent antibacterial properties. Therefore, it collaboratively regulates immunity and redox homeostasis, significantly promoting angiogenesis and diabetic wound repair.
Ideal wound healing not only requires a fast speed but also the near-physiological recovery of function and appearance. The key lies in effectively inhibiting excessive fibrosis in the later stage of repair to prevent scar formation[113]. Recently, the dynamic adaptive regulation of hydrogels (F/R gels) reported by Zhang et al. demonstrated a new direction for precise regulation in the late healing stage [Figure 6F][33]. This system forms a hierarchical drug release system through dynamic cross-linking of Schiff bases. Initially, ε-polylysine and CeO2 nanozyme are released to fight bacteria and eliminate ROS. Subsequent remodeling of the growth and fibroblast subtypes effectively inhibits scar formation. In mouse and rabbit ear diabetic wound models, this system achieved rapid and scarless functional skin regeneration.
Internal environment-triggered nanozyme-hydrogels are used in treating bacterial infection wounds
Bacterial infections, especially chronic wounds that form biofilms, have significant differences in their internal microenvironment (such as pH value, concentrations of specific enzymes and metabolites) from normal tissues[114]. The internal environment-responsive nanozyme-hydrogel designed on this basis can achieve precise and on-demand release of antibacterial drugs, thereby effectively eradicating infections and promoting tissue regeneration.
The key signaling molecules of the microenvironment of infected wounds provide multiple trigger targets for intelligent nanozyme-hydrogels. The main trigger types include pH response and microenvironmental-specific response, etc.[115]. At the site of bacterial infection, the accumulation of metabolic products leads to changes in the local pH value, thereby causing alterations in the acidity or alkalinity of the environment[116]. Therefore, pH response is the most common triggering mechanism. pH-responsive hydrogels can intelligently release nanozymes or change their structure and activity under the stimulation of the infected site to achieve the purpose of capturing and killing bacteria. For instance, a research team has constructed a MOF nanozyme composite cryogel (CSG-MX), in which Fe-MIL88NH2 nanozyme is grafted onto glycidyl methacrylate functionalized dialdehyde chitosan through Schiff base reaction[117]. CSG-MX possesses high hydrophilicity, acid-enhanced positive charge, and pH-responsive nanozyme release and rebinding capabilities. Thanks to the negative potential of bacteria, the influence of infection on pH, and the pH-responsive reversible release and enzyme activity of nanozymes (such as POD and OXD mimicase activity), CSG-MX can achieve intelligent adaptive capture and killing of bacteria in the presence of low concentrations of H2O2 (100 μM) [Figure 7A].
Figure 7. Internal microenvironment-triggered metal nanozyme-hydrogel systems for antibacterial therapy and infected wound repair. (A) Schematic Diagram of CSG-MX preparation and its subsequent applications. Reproduced with permission from Ref.[117]. Copyright 2021, American Chemical Society; (B) Schematic illustration of the underlying antibacterial mechanism. Reproduced with permission from Ref.[118]. Copyright 2024, Oxford University Press; (C) Proposed mechanisms through which OBG@CG facilitates diabetic wound repair. Reproduced with permission from Ref.[121]. Copyright 2024, Elsevier; (D) SEM images and live-dead images of bacteria after different treatments. Reproduced with permission from Ref.[121]. Copyright 2024, Elsevier; (E) Mechanism illustration of CuCD1-based microenvironment responsive disruption of bacterial biofilm. Reproduced with permission from Ref.[122]. Copyright 2025, Elsevier; (F) Tissue-regenerative effects of CPAN-AMP in infected diabetic wounds. Reproduced with permission from Ref.[120]. Copyright 2025, Wiley. CSG-MX: Chitosan-based cryogel - MOF nanozyme composite; MOF: metal-organic framework; OBG@CG: OHA/borax-gelatin hydrogel@Cu2-xSe-BSA-GOx; SEM: scanning electron microscopy; CPAN-AMP: conductive polyaniline-antimicrobial peptide composite hydrogel; POD: peroxidase; GSH: glutathione; HIF: hypoxia-inducible factor; VEGF: vascular endothelial growth factor; CS-GMA: chitosan-g-glycidyl methacrylate; APS: ammonium persulfate; TEMED: tetramethylethylenediamine; BLK: blank; Gox: glucose oxidase.
The more sophisticated design makes use of multiple signals in the infection microenvironment. For example, A programmed self-activating antibacterial hydrogel in response to bacteria has been developed. This system consists of pH-responsive H2O2 self-supplied complex nanozyme (MSCO) and pH/enzyme-sensitive LOX micelles jointly encapsulated in L-arginine-modified chitosan and phenylboronic acid-modified oxidized glucan hydrogels[118]. In the infection microenvironment, bacterial metabolic products (such as lactic acid) cause a decrease in pH[119]. On the one hand, this activates the protonation of the guanidine and amino groups of CA to destroy the bacterial cell wall. On the other hand, MSCO dissociates to release Cu2+ and H2O2. H2O2 mediates the Fenton reaction catalyzed by Cu2+ and the POD-like activity of MoS2, efficiently generating •OH. Meanwhile, the LOX released by PPEL catalyzes the decomposition of lactic acid, generating more H2O2 and forming a positive feedback effect. Furthermore, H2O2 and Cu2+ also trigger the self-driven cascade release of NO, weakening the bacterial resistance to ROS [Figure 7B].
It is worth noting that biofilms are the core obstacle that makes chronic infections difficult to cure. Nanozymes-hydrogels triggered by the internal environment have shown great potential in destroying the structure of biofilms. In addition to the traditional pH value or enzyme sensitivity reaction systems, researchers have also developed smart materials that respond to metabolic signals in infectious wounds. The glucose activation mechanism is a typical representative among them. It directly utilizes the pathological feature of high glucose concentration in diabetic wounds to achieve disease-specific therapeutic regulation[120]. For the variable microenvironment of infectious diabetic wounds, a study has constructed a double-dynamic bond cross-linked hydrogel (OBG@CG) loaded with GOx and pH-responsive self-assembled Cu2-xSe-BSA nanozyme[121]. In the acute infection period, acidic conditions trigger Cu2-xSe-BSA nanozyme self-assembly, activate its POD-like activity, catalyze H2O2 to produce •OH, and efficiently attack bacterial biofilms [Figure 7C]. During the wound recovery period, when the pH value increases, Cu2-xSe-BSA depolymerizes, terminating the production of ROS, and the released Cu2+ can promote collagen production and angiogenesis. The antibacterial effect of the material was confirmed by scanning electron microscopy (SEM) and live/dead staining images of bacteria. After treatment, the bacterial morphology was damaged and a large number of bacteria died [Figure 7D]. In addition, other response strategies targeting the biofilm microenvironment are equally effective. For instance, the Cu-doped carbon dot nanozyme (CuCD1) designed by researchers emphasizes responsive clearance of the bacterial biofilm microenvironment[122]. This nanozyme features an ultra-small size and a positively charged surface, enabling it to penetrate biofilms in slightly acidic environments and catalyze the generation of endogenous H2O2 to •OH. This enhanced oxidative stress destroys the membrane structure of bacteria, achieving the eradication of biofilms [Figure 7E]. Researchers further loaded CuCD1 into carboxymethyl chitosan/oxidized glucan cross-linked hydrogels. This composite system can continuously kill bacteria and promote collagen deposition.
Furthermore, a recently reported glucose-activated programmable hydrogel (CPAN-AMP) with self-switching enzyme-like activity demonstrated the potential of metabolic signal-driven multimodal reaction systems in complex wound environments[120]. This system consists of Au-MoS2 -phenylboronic acid (PBA) nanozyme, nitroimidazole-modified sodium alginate microcapsules loaded with insulin and PBA-chitosan network. In hyperglycemia, CPAN-AMP oxidizes glucose to generate ROS for antibacterial action. The resulting hypoxia, together with nitroreductase and bioreductants, drives amphiphilic alginate to reduce nitroimidazole into hydrophilic aminoimidazole, disrupting capsule amphiphilicity and triggering insulin release. Under normoglycemia, CPAN-AMP switches to oxygen generation, preventing hypoxia-responsive capsule transformation and relieving tissue hypoxia. This programmable hydrogel enables feedback glycemic control and orderly wound repair [Figure 7F].
External environment-triggered nanozyme-hydrogels are used for the treatment of bacterial infection wounds
In addition to responding to the physiological signals on the wound surface, precisely manipulating the nanozyme-hydrogel system using external physical fields (such as light, heat, and sound) provides a powerful non-invasive treatment tool for addressing deep infections and biofilm issues. This external triggering strategy has outstanding advantages such as controllable time and space and adjustable dosage[123,124]. The external stimuli applied to the treatment of bacterial infection wounds are mainly divided into two categories: NIR light and ultrasound.
Combining internal microenvironment responses with external stimuli can achieve more precise and efficient synergistic antibacterial treatment. For instance, a research team has developed an alginate (Alg/CuP) hydrogel based on copper hydrogen phosphate (CuP) nanozyme[125]. This system ingeniously takes advantage of the pH differences among various wounds: in acidic infected wounds, it exhibits POD-like activity to produce •OH for bactericidal purposes; In alkaline diabetic wounds, it switches to CAT-like activity to produce oxygen and promote angiogenesis. Meanwhile, this hydrogel has excellent NIR photothermal conversion capability. The mild thermal effect produced by near-infrared light irradiation can significantly enhance its pH-responsive catalytic activity and Cu2+ release, thereby synergically accelerating the healing of various refractory wounds [Figure 8A].
Figure 8. External environment-triggered metal nanozyme-hydrogel systems for antibacterial therapy and infected wound healing. (A) Multifunctional, pH-switchable CuP nanozyme-composite alginate hydrogel exhibiting mild photothermal-enhanced catalytic activity and therapeutic ion release, significantly accelerating the healing of various refractory wounds. Reproduced with permission from Ref.[125]. Copyright 2023, American Chemical Society; (B) Schematic illustration of the antibacterial effect of the hydrogel. Reproduced with permission from Ref.[127]. Copyright 2023, Wiley; (C) Schematic illustration of a NIR-responsive nanozyme-hydrogel combines ROS generation, photothermal effects, antibacterial activity and antioxidation to promote healing of bacteria-infected wounds. Reproduced with permission from Ref.[128]. Copyright 2025, Elsevier; (D) Schematic diagram of the MoS2@TA/Fe NSs’ POD- and CAT-like activities. Reproduced with permission from Ref.[131]. Copyright 2022, Elsevier; Figure 8D is reproduced from (Li, Y., 2022) under the CC BY-NC-ND license. No modifications were made; (E) Stimuli-responsive hydrogel dressings integrating bacterial-trapping, antimicrobial, and antibiofilm functions to accelerate wound healing. Reproduced with permission from Ref.[132]. Copyright 2025, Elsevier; (F) The adhesion, rheological properties and microstructure of the hydrogel. Reproduced with permission from Ref.[132]. Copyright 2025, Elsevier. NIR: Near-infrared; ROS: reactive oxygen species; NSs: nanozymes; POD: peroxidase; CAT: catalase; PTT: photothermal; bFGF: basic fibroblast growth factor; CDT: chemodynamic therapy; EGCG: epigallocatechin gallate; OD-AB: modified dextran; PDA: polydopamine; CMCS-PEI: carboxymethyl chitosan - polyethylenimine; OACPPIh: developed using OD-AB, CMCS-PEI, PDA, and iron-hydrated nanoparticles; NPs: nanoparticles.
The PTT can not only directly kill bacteria but also significantly enhance the catalytic efficiency of nanozymes[126]. A study has constructed a PNMn hydrogel with multi-enzyme activities (POD, CAT, OXD) and excellent NIR-II photothermal performance. Its photothermal effect can enhance enzymatic catalytic reactions, thereby generating ROS more effectively[127]. In addition, this hydrogel also has the property of bacterial capture, which can enrich bacteria in the ROS generation region and further enhance the antibacterial effect, as shown in Figure 8B. Similarly, a research team functionalized the injection of PVA-alginic acid hydrogel with Ag@MXene nanozyme[128]. MXenes (transition metal carbides and nitrides) are a class of emerging two-dimensional materials[129]. In this design, Ag nanoclusters not only provide more catalytically active sites but also efficiently convert NIR light energy into thermal energy. This photothermal-enhanced catalytic effect jointly constructs the NIR-responsive ROS generation antibacterial platform [Figure 8C].
The ideal nanozyme-hydrogel system should be capable of dynamically regulating ROS levels according to therapeutic needs: effectively generating ROS in the antibacterial stage and eliminating excessive ROS in the subsequent anti-inflammatory stage[130]. Hydrogels based on tannic acid-metal-modified MoS2 double nanozymes (MoS2@TA/Fe NSs) have this function[131]. Under acidic conditions, it generates ROS through POD-like activity; In a neutral environment, it switches to CAT-like activity, which can not only eliminate
In addition, for different clinical needs, good adhesion and mechanical adaptability of hydrogels are crucial for frequently moving parts such as joints. A team designed an intelligent stimulus-responsive hydrogel with glycocalyx-mimetic structure[132]. This system functions through a sophisticated cascade reaction; it captures bacteria by taking advantage of the characteristics of the simulated glycocalyx-mimetic structure, and then, under NIR irradiation, the photothermal effect of PDA synergistically kills bacteria with cations. Meanwhile, the photothermal effect can also increase the CAT-like activity of hydrated iron nanozyme by eliminating ROS and producing oxygen [Figure 8E]. This hydrogel exhibits excellent tissue adhesion, can firmly adhere to pigskin and even the joints of moving hands, and can withstand compression and tensile deformation [Figure 8F]. Its self-healing property also ensures long-term stable coverage of dynamic wounds.
Ultrasound, as an external stimulus capable of penetrating deep tissues, features non-invasiveness and high penetration depth[133-135]. Therefore, it is often used to activate photosensitizers for SDT. In response to the complex pathological features commonly present in diabetic infected wounds, such as hypoxia, high H2O2 (leading to oxidative stress), and biofilm barriers, researchers have developed a sophisticated ultrasound-responsive nanozyme-hydrogel system. For example, a team developed a multifunctional antibacterial hydrogel dressing (PPCN@Pt-AMPs/HGel) for treating diabetic infected wounds[136]. The system was based on gelatin and sodium alginate, loaded with the nano-sonosensitizer PN-224, Pt nanozymes with CAT-like activity, and antimicrobial peptides (AMPs) targeting biofilms, as shown in Figure 9A. Under low-intensity ultrasound irradiation, this system exhibits a cascade effect of synergistic treatment; the CAT-like activity of Pt nanozyme catalyzes the decomposition of excess H2O2 in the wound bed into O2. This process not only alleviates the oxidative stress of the wound, but more importantly, the O2 produced, as a key substrate, greatly alleviates the hypoxic state of the wound bed, thereby significantly enhancing the SDT efficacy of the sonosensitized agent PCN-224 and enabling it to efficiently generate ROS to kill bacteria. Meanwhile, the biofilm-targeting ability of AMPs further assists in the clearance of infections. In MRSA-infected diabetic rat models, the PPCN@Pt-AMPs/HGel (US) treatment group showed the fastest wound healing rate and the highest healing quality [Figure 9B], confirming its significant effect in promoting wound healing in infectious diabetes.
Figure 9. Ultrasound-responsive metal nanozyme-hydrogel systems for enhanced antibacterial therapy and infected wound healing. (A) An ultrasound-activated PPCN@Pt-AMPs/HGel dressing alleviates H2O2-mediated oxidative stress and hypoxia while enhancing sonodynamic therapy to promote healing of diabetic-infected wounds. Reproduced with permission from Ref.[136]. Copyright 2025, Elsevier; (B) Representative wound photographs at multiple time points and corresponding healing curves, illustrating residual wound area over time under different treatments (n = 3). US: with the ultrasound irradiation at 0.8 W/cm2 for 5 min. WO: without ultrasound irradiation.
Another study developed a hydrogel based on platinum NP assemblies (PNAs) and revealed a new role of ultrasound in nanozyme catalysis[137]. PNAs are high-density platinum NP assemblies synthesized using metal-organic coordination polymers with dynamic covalent bond hybridization as templates. Mechanistically, under the action of ultrasound, this system not only exhibits traditional CAT-like and POD-like activities to modulate ROS levels but also facilitates macrophage polarization from the pro-inflammatory M1 phenotype to the regenerative M2 phenotype [Figure 9C]. This metabolic reprogramming translates into enhanced tissue repair signals; specifically, immunofluorescence analysis revealed that the expressions of epidermal growth factor (EGF) and vascular endothelial growth factor (VEGF) were significantly upregulated in the treated wounds, indicating effective promotion of angiogenesis and epithelialization [Figure 9D]. This therapeutic efficacy is fundamentally driven by a novel glutathione reductase (GR)-mimicking activity: ultrasound induces a surface plasmon resonance effect on dense Pt NPs, which promotes the electron transfer of coenzyme reduced coenzyme II (NADPH) and enables the catalytic reduction of oxidized GSH to reduced GSH [Figure 9E]. The regeneration of GSH is crucial for regulating cellular redox homeostasis. Ultimately, quantitative assessment confirmed that the GelMA + PNAs + US treatment effectively alleviated oxidative stress (reduced ROS and HIF-1α) while enhancing repair factors, achieving high-quality tissue repair [Figure 9F].
In conclusion, whether it is the internal stimulation of the wound microenvironment or the application of an external physical field, the core objective is to achieve on-demand and precise regulation of the functions of the nanozyme-hydrogel system (such as ROS generation/clearance, drug release, and O2 supply). Internal stimuli (such as pH response) endow dressings with intelligent adaptive capabilities, enabling them to automatically activate antibacterial functions when infections occur and even switch their enzyme activity patterns at different pathological stages (such as hyperglycemia/hypoglycemia). External stimuli (such as light and sound) offer a higher level of active control and have superior controllability in terms of time and space. Through external triggering, not only can non-invasive treatment of deep tissues be achieved, but it can also be coordinated with internal responses, significantly enhancing the catalytic activity of pH responses through photothermal effects, or enhancing multi-enzyme synergistic antibacterial effects. The ideal smart dressing precisely integrates internal and external stimulus responses, ingeniously connecting the efficient antibacterial/anti-biofilm stage with the subsequent anti-inflammatory, antioxidant and pro-regeneration stages (such as promoting angiogenesis and collagen deposition), shifting from simple sterilization to the regulation of microenvironment homeostasis, thereby providing a more powerful and precise solution for the healing of chronic infected wounds.
Nanozyme-hydrogels are used for anti-inflammation and repair of other functional tissues
Related to bone and joint
Although osteoarthritis (OA), rheumatoid arthritis (RA), and bone defects are respectively related to mechanical wear, immune abnormalities, and trauma/infection in terms of etiology, all three are accompanied by obvious inflammatory responses, inflammatory cell infiltration, and continuous destruction of osteochondral cartilage during the onset process. Excessive accumulation of ROS and abnormal upregulation of inflammatory factors are their common pathological driving forces. It directly leads to joint tissue damage, bone loss and functional degeneration[12,138,139]. In recent years, the composite system of nanozymes and hydrogels has demonstrated unique advantages in reshaping the local microenvironment, promoting bone regeneration and delaying the progression of lesions due to its antioxidant, anti-inflammatory and sustained-release properties.
The multi-enzyme cascade activity of nanozymes can effectively eliminate excessive ROS in the lesion area, alleviate inflammatory responses, protect cells and tissues, and thereby regulate the microenvironment of bone regeneration[140]. For instance, Wang et al. reported a therapeutic strategy based on manganese trioxide (Mn3O4) nanozymes for alleviating cartilage degeneration in OA[141]. This nanozyme has multi-enzyme activities similar to those of SOD and CAT, which can effectively eliminate the accumulated ROS in the joint cavity, thereby reducing the damage of oxidative stress to chondrocytes and inhibiting inflammatory responses. To achieve local and persistent delivery, the research team embedded Mn3O4 nanozymes into injectable chondroitin sulfate (CS) hydrogels. In the medial meniscus instability surgery-induced OA mouse model, the Mn3O4@CS hydrogel treatment group demonstrated a significant cartilage protective effect. The Micro-CT image Figure 10A-i shows that the joint surface destruction and osteophyte formation in the treatment group were significantly improved. HE staining [Figure 10A-ii] confirmed the reduction of cartilage destruction and inflammatory cell infiltration, while fanred-O staining Figure 10A-iii showed that the cartilage thickness and integrity of the treatment group were effectively maintained, indicating that this composite system could significantly slow the progression of OA.
Figure 10. Anti-inflammatory and regenerative applications of metal nanozyme-hydrogel systems in bone and joint tissues. (A) Mn3O4@CS hydrogel alleviated cartilage degeneration in a mice model. (i) Representative micro-CT images for each group, and (ii) H&E and (iii) Safranin O staining used to evaluate cartilage degeneration. Reproduced with permission from Ref.[141]. Copyright 2023, Wiley; (B) Schematic illustration of the fabrication of a DNA hydrogel co-loaded with MSC-derived mitochondria and nanozymes, and its application in RA therapy. Reproduced with permission from Ref.[142]. Copyright 2025, Elsevier; Figure 10B is reproduced from (Wang, F., 2025) under the CC BY-NC-ND license. No modifications were made; (C) Schematic representation of the hydrogel platform designed for RA treatment. Reproduced with permission from Ref.[144]. Copyright 2025, Nature; (D) Proposed mechanism of ROS scavenging by the DAGQD@Cu SAN. Reproduced with permission from Ref.[144]. Copyright 2025, Nature; Figure 10D is reproduced from (He, H., 2025) under the CC BY-NC-ND license. No modifications were made; (E) (i) An anatomic illustration of hydrogel adhesion within the joint; (ii) FESEM and (iii) metallographic microscopy images of the hydrogel-cartilage cross-section. Reproduced with permission from Ref.[144]. Copyright 2025, Nature. Figure 10E is reproduced from (He, H., 2025) under the CC BY-NC-ND license. No modifications were made. CS: Chondroitin sulfate; ROS: reactive oxygen species; SAN: single-atom nanozyme; CTL: control; DMM: destabilization of the medial meniscus; MSC: mesenchymal stem cell; RGD: Arg-Gly-Asp; RA: rheumatoid arthritis; IL: interleukin; TNF: tumor necrosis factor; M1: M1 macrophage; M2: M2 macrophage; iNOS: inducible nitric oxide synthase; CD206: cluster of differentiation 206; DA-HA: dopamine-modified hyaluronic acid; KGN: kartogenin; SOX9: SRY-box transcription factor 9; GQD: graphene quantum dot; CAT: catalase; PBS: phosphate buffered saline; DAGQD: dopamine-derived graphene quantum dot.
In the more complex microenvironment of RA, a single anti-inflammatory or antioxidant strategy is often insufficient to repair existing tissue damage. In response to this, Wang et al. designed a polymer-modified DNA hydrogel platform for the synergistic delivery of Prussian blue nanozyme and active mitochondria to achieve the dual goals of anti-inflammation and repair[142]. This DNA hydrogel has excellent biocompatibility, injectability, self-healing and shear-thinning properties, making it suitable for intra-articular administration. In this system, Prussian blue nanozyme is responsible for eliminating excessive exogenous ROS in the RA microenvironment and down-regulating pro-inflammatory cytokines to alleviate the inflammatory response. Meanwhile, the delivered healthy mitochondria replenish the function of damaged cells through mitochondrial transfer, inhibit the production of endogenous ROS and restore cellular energy metabolism, as shown in Figure 10B. This strategy that combines eliminating exogenous sources with inhibiting endogenous sources has significantly promoted the proliferation of chondrocytes in animal experiments, alleviated RA symptoms, and repaired joint tissues.
In addition, in the bone immune microenvironment of RA, the efficacy of stem cell therapy is often limited by ROS and hypoxic stress. To address this challenge, Zhao’s team developed a nanozyme-enhanced self-repairing hydrogel as an H2O2 -driven Oxygenerator to reshape the RA microenvironment and improve osseointegration at the prosthesis interface[143]. This system, with manganese cobalt oxide nanozyme as its core, can not only eliminate excessive ROS but also synergistically utilize endogenous H2O2 to generate dissolved oxygen, thereby alleviating local hypoxia and oxidative stress. As a delivery carrier of bone marrow mesenchymal stem cells, this hydrogel protects the implanted cells from ROS and hypoxia-mediated death, significantly enhances their osteogenic potential, and simultaneously downregulates local inflammatory factors, ultimately achieving better bone integration and arthritis relief.
In addition to anti-inflammatory and regenerative properties, hydrogels can also be endowed with lubricating functions to reduce joint friction, which is crucial for the treatment of OA and RA. For instance, He et al. developed an injectable bioadhesive and lubricating hydrogel for the full-cycle treatment of RA as shown in Figure 10C[144]. The hydrogel is loaded with a multifunctional Cu single-atom nanozyme. This nanozyme efficiently eliminates various ROS such as H2O2 and •OH through a dual catalytic mechanism of Cu single atoms (providing SOD/ CAT-like activity) and the catechol-quinone redox pair of dopamine, as shown in Figure 10D, thereby effectively inhibiting the inflammatory microenvironment. In addition, hydrogels can continuously release chondroietin, recruit stem cells and promote their differentiation into chondrocytes to repair damaged cartilage. This design achieves lubrication and anti-inflammatory protection for early-stage RA to cartilage repair for advanced-stage RA, providing a highly promising full-cycle treatment strategy. This system consists of a dopamine-modified hyaluronic acid (DA-HA) and sulfonated hyaluronic acid (SO3--HA) network. Inspired by mussel adhesion proteins, DA endows the hydrogel with strong bioadhesion, enabling it to firmly adhere to the surface of articular cartilage [Figure 10E] for long-term retention. The SO3--HA network simulates the composition of synovial fluid, providing excellent lubricity and significantly reducing the coefficient of friction.
Intestinal-related
In recent years, the composite system of nanozymes and hydrogels has gradually become a potential strategy for the treatment of inflammatory bowel disease (IBD). Unlike the application in relatively closed joint cavities and fluid environments, the intestinal environment features acid-base gradients, digestive enzymes, food flow, and complex flora, making it extremely easy for drugs to be diluted or inactivated. Therefore, in IBD, in addition to providing in situ retention and sustained-release protection, it is also necessary to have certain oral stability, mucosal adhesion and inflammatory site targeting, so as to ensure the stability and local effective concentration of nanozym-hydrogel in the complex intestinal environment[145-147].
During the course of IBD, nanozymes can effectively eliminate excessive ROS in the lesion area, reduce the levels of inflammatory factors, and alleviate oxidative stress and tissue damage. Specifically, the multi-enzyme activity of nanozymes can be administered through the rectum or orally, effectively eliminating excessive ROS in intestinal lesions, thereby blocking inflammatory signaling pathways and reducing tissue damage and apoptosis. For instance, the research team designed a thermosensitive hydrogel containing Mn3O4 nanozymes, which can target the inflamed colon[148]. In the mouse colitis model, this composite system can form a gel in situ at the inflammatory site, continuously remove ROS, significantly reduce the levels of pro-inflammatory factors, and improve intestinal function[148].
To enhance the retention and targeting efficiency of nanozyme in the intestinal tract, the excellent biocompatibility, injectability and sustained-release performance of hydrogels make them an ideal platform. Through sustained-release action, hydrogels can maintain the local concentration of nanozymes in the lesion area and prolong the therapeutic effect. For instance, Zhang et al. reported a complex system in which Mn3O4 nanozymes were immobilized on kaolin and further embedded into PDA hydrogels[149]. The PDA coating enables the system to rapidly form adhesions in the colon and firmly remain in the damaged and inflamed areas with its strong mucosal adhesion, enhancing the efficiency of local retention and removal of ROS and restoring the expression of barrier-associated tight junction proteins.
Gut microbial species play an important role in host health[150]. The occurrence of IBD is closely related to the imbalance of intestinal flora. Merely eliminating ROS is not enough. The hydrogel system that combines nanozyme with probiotics or specific inorganic components can not only reduce inflammation but also reshape the micro-ecological environment and promote the recovery of intestinal homeostasis. For example, Zhou et al. constructed a dual-targeted Mn@CeO2 nanozyme-modified probiotic hydrogel microsphere (MnCe@LR/AMs)[151]. In this study, Mn@CeO2 nanozymes with high SOD and CAT-like activities were first coated on the surface of Lactobacillus reuteri, and then it was prepared by electrostatic spraying, which overcomes the gastrointestinal barrier [Figure 11A]. Sodium alginate facilitated dual targeting of MnCe@LR/AMs by combining charge-mediated interactions with mannose receptor-mediated binding at inflammation-associated regions [Figure 11B and C]. The negative charge of alginate enables it to electrostatically adsorb onto positively charged inflammatory mucosal areas. Meanwhile, the mannose residues in its structure can specifically bind to the highly expressed mannose receptors on macrophages at the inflammatory site. In the dextran sulfate sodium colitis model, MnCe@LR/AMs effectively repaired the intestinal barrier through the synergistic effect of nanozymes (ROS clearance) and probiotics (colonization), and improved immune homeostasis by regulating the polarization of macrophages from M1 type to M2 type. At the same time, it significantly increases the relative abundance of beneficial bacteria (such as Lactobacillus reuteri), improves amino acid metabolism, and thereby reshapes the intestinal microecology[151].
Figure 11. Intestinal anti-inflammatory and regenerative applications of metal nanozyme-hydrogel systems. (A) Schematic illustration of a nanocoating-hydrogel microsphere system that enhances intestinal colonization of probiotics. Reproduced with permission from Ref.[151]. Copyright 2025, American Chemical Society; Figure 11A is reproduced from (Zhou, P., 2025) under the CC BY-NC-ND license. No modifications were made; (B) Schematic diagram showing MnCe@LR/AMs targeting colonic inflammation via electrostatic adsorption and mannose receptor-mediated recognition. Reproduced with permission from Ref.[151]. Copyright 2025, American Chemical Society; Figure 11B is reproduced from (Zhou, P., 2025) under the CC BY-NC-ND license. No modifications were made; (C) MnCe@LR/AMs derived from Mn@CeO2 nanozymes tolerate gastric acid, target inflamed colonic sites, scavenge ROS, repair the intestinal barrier, and modulate gut microbiota, metabolism, and immune responses. Reproduced with permission from Ref.[151]. Copyright 2025, American Chemical Society; Figure 11C is reproduced from (Zhou, P., 2025) under the CC BY-NC-ND license. No modifications were made; (D) Schematic illustration showing that W-PB gel treats IBD by efficiently scavenging ROS, alleviating inflammation, and modulating the gut microbiota. Reproduced with permission from Ref.[152]. Copyright 2025, Elsevier; (E) Experimental flowchart of the treatment on IBD mice model. Reproduced with permission from Ref.[152]. Copyright 2025, Elsevier; (F) H&E staining and AB/PAS histological staining. Reproduced with permission from Ref.[152]. Copyright 2025, Elsevier. MnCe@LR/AMs: Dual-targeted Mn@CeO2 nanozyme-modified probiotic hydrogel microsphere; ROS: reactive oxygen species; W-PB: tungsten-doped Prussian blue; IBD: inflammatory bowel disease; H&E: Hematoxylin and Eosin; AB: Alcian blue; PAS: periodic acid-Schiff; DSPE: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine; PEG: polyethylene glycol; LR: L. reuteri; ZO-1: Zonula occludens-1; CAT: catalase; IL: interleukin; TNF: tumor necrosis factor; M1: M1 macrophage; M2: M2 macrophage; IFN: interferon; DSS: dextran sulfate sodium; PB: Prussian blue; 5-ASA: 5-aminosalicylic acid.
Similarly, Luo et al. designed a tungsten-doped Prussian blue (W-PB) nanozyme and encapsulated it in an alginate/chitosan electrostatically cross-linked hydrogel as shown in Figure 11D[152]. This hydrogel protects nanozyme from degradation in the gastrointestinal environment and achieves pH-responsive release in the weakly alkaline environment of the colon. This system reshapes the intestinal microecology through spectral antioxidation and specific antibacterial properties to alleviate colitis: W-PB nanozyme itself can efficiently eliminate excessive reactive oxygen and nitrogen species, reduce oxidative stress, and inhibit the STING pathway to weaken inflammation. The released tungsten ions can specifically inhibit the excessive proliferation of pathogenic Enterobacteriaceae by competitively replacing molybdenum cofactors. In the dextran sulfate sodium-induced colitis model, as shown in Figure 11E, oral W-PB hydrogel treatment demonstrated significant efficacy. Histological analysis [Figure 11F] further confirmed that H&E staining showed that the colonic mucosal structure in the W-PB gel group was intact and there was no obvious infiltration of immune cells. Alcian blue/Periodic acid-Schiff staining also indicated that its goblet cell structure and mucin secretion were effectively protected, verifying the advantages of this strategy in repairing the intestinal barrier and reshaping the microecology.
In addition to the basic antioxidant and microecological regulation functions, some nanozyme-hydrogel systems also have additional functions such as imaging, anti-fibrosis and reduction of parenteral complications, providing new ideas for the multi-dimensional treatment of IBD. For instance, Cao et al. achieved an integrated diagnosis and treatment of inflammation and anti-fibrosis guided by computed tomography imaging by using dextran-coated CeO2 nanozyme[153]. The ZnCeγO2/Se oral hydrogel designed by Kan et al. can achieve dual tissue protection by activating the Nrf2 antioxidant axis and inhibiting ferroptosis[154].
Moreover, Wu et al. constructed inulin hydrogel that combines PB nanozyme with nattokinase[155]. After oral administration, it can not only eliminate ROS and lyse intestinal microthrombi, but also alleviate extraintestinal complications related to ulcerative colitis (such as anxiety and depression phenotypes) by regulating the microbiota-gut-brain axis.
Others
Prolonged hyperglycemia can lead to organ failure and tissue damage, thereby triggering various complications. Therefore, accurate and timely monitoring of blood glucose levels is of paramount importance for the prevention and management of diabetes. With advances in clinical testing, home-based health monitoring, and personalized treatment, convenient glucose detection methods have attracted widespread attention[156]. On one hand, glucose detection using blood-alternative samples such as sweat, interstitial fluid, tears, saliva, and urine has been extensively investigated; on the other hand, visual, continuous, and non-invasive glucose monitoring technologies are also continually advancing and being refined[157-161].
Building upon these shared design principles, metal nanozyme-hydrogel systems have been extensively integrated into wearable biosensing platforms, where their catalytic responsiveness can be directly translated into measurable electrical or optical signals. Driven by these advances, wearable devices are becoming increasingly visible in healthcare[162,163]. Yang et al. developed a wearable non-enzymatic electrochemical sensor for sweat glucose detection. The sensor is functionalized with a highly hydrophilic gel that rapidly concentrates sweat molecules on its surface. AuNPs subsequently oxidize glucose in the sweat to gluconolactone and H2O2, enabling real-time and non-invasive monitoring of glucose. Moreover, ZIF-8-C further catalyzes the oxidation of the generated H2O2 into hydroxyl radicals, thereby endowing the wearable glucose sensor with high antibacterial activity[159]. In addition, colorimetric sensors have been developed as a convenient and intuitive technology for detecting various analytes. In a complementary study, Fei et al. designed and introduced a Fe3O4@MXene-Au nanocomposite with exceptional peroxidase-like activity for the first time in the field of nanozymes[160]. Based on the enzyme-mimicking properties of this material, they developed a colorimetric biosensor for glucose detection, which exhibited high selectivity toward glucose and was successfully applied to the analysis of human blood samples with satisfactory recovery rates. Furthermore, a portable detection platform integrated with an agarose hydrogel and smartphone-based readout was constructed, demonstrating promising potential for on-site and visual monitoring of glucose.
CONCLUSION AND OUTLOOK
The metal nanozyme-hydrogel composite system represents an emerging intersection of biomimetic catalysis and biomaterials engineering. As detailed in this review, through well-established top-down or bottom-up strategies, various metal nanozymes have been successfully integrated into hydrogel networks, achieving multifunctional synergy. The core advantage of this system lies in the fact that the hydrogel matrix not only provides structural protection to mitigate deactivation, dilution, aggregation, and nonspecific clearance in complex biological environments, but its adjustable 3D network also facilitates mass transport, including substrate diffusion and product removal. Conversely, nanozymes, as built-in catalytic engines, endow hydrogels with enhanced biocatalytic activities, such as reactive oxygen species regulation, immune regulation, and tissue regeneration capabilities. At the application level, 3D printing technology makes it possible to construct personalized implants with bionic structures, while injectable in situ gels have become an ideal choice for filling deep or irregular tissue defects. Their promising potential in tumor co-therapy, infected wound management, and biosensing fully demonstrates their value as next-generation intelligent diagnosis and treatment platforms. However, from laboratory proof-of-concept to clinically applicable therapies, this field still faces a series of severe core challenges.
Critical challenges in catalytic efficacy and biosafety
The primary challenge lies in the gap of catalytic efficacy: despite the continuous improvement of design strategies, the catalytic efficiency and substrate specificity of most nanozymes are still far inferior to those of natural enzymes. Furthermore, the selection of an optimal nanozyme material necessitates a complex balance between competing performance metrics. Precious metal nanozymes (e.g., Pt, Pd) often exhibit superior catalytic activity and stability but face challenges related to high cost, potential long-term bioaccumulation, and limited biodegradability. Iron-based nanozymes (e.g., Fe3O4) generally offer better biocompatibility, lower cost, and potential metabolic pathways, yet their catalytic activity may be lower or more condition-dependent. MOF-based nanozymes provide highly tunable structures and multifunctionality but can suffer from instability in acidic microenvironments or complex degradation profiles. Therefore, maximizing therapeutic outcomes requires carefully weighing catalytic efficiency against biosafety, manufacturing scalability, and overall cost-effectiveness.
Beyond intrinsic material properties, biological barriers present further impediments. Once inside the body, the surface of the nanozyme will rapidly adsorb proteins to form a protein corona. This dynamic biomolecular mask can occlude active sites, leading to a sharp decline in catalytic activity.
Equally critical is the long-term biosafety of the delivery system. Hydrogels, as delivery carriers, can markedly prolong the residence time of metal nanozymes at lesion sites, thereby effectively increasing local concentrations and extending the therapeutic window. However, enhanced local retention may also promote chronic accumulation and consequently raise potential safety concerns. Current studies on metal nanozyme-hydrogel systems largely focus on in vitro cytocompatibility, hemolysis, and short-term oxidative stress as acute safety readouts for preliminary validation, whereas comparatively limited attention has been paid to the long-term degradation behavior of hydrogels, the in vivo metabolic fate of nanozymes, and the resulting systemic safety issues, including chronic immune responses, tissue fibrosis, or long-term organ accumulation. Moreover, the competing demands between hydrogel mechanical stability and timely, predictable biodegradation further complicate the balance between therapeutic performance and long-term biosafety in clinical translation. Non-biodegradable precious metal nanozymes (such as Pt, Au) may accumulate for a long time in certain organs, and their potential chronic toxicity remains unclear. Some Fe- or Mn-based nanozymes and certain metal organic frameworks may offer improved biodegradability and clearance, depending on composition, dose, and local microenvironment; however, their degradation rates, local concentrations of metal ions, valence state changes, and long-term impacts on cellular homeostasis and the immune microenvironment urgently need to be systematically and deeply evaluated[164]. Establishing comprehensive profiles of biodistribution, biodegradation, clearance, and long-term immunological consequences is the prerequisite for solving this problem.
Engineering hurdles: environmental robustness and scalability
Furthermore, the robustness of current intelligent response designs remains to be validated under clinically relevant, multi-factor pathological microenvironments. Most systems rely on responses to single signals such as pH or H2O2, while the real pathological microenvironment (such as tumors) is a complex network dynamically interwoven with hypoxia, acidity, multiple enzymes and oxidative stress signals. This overly simplified response logic may lead to premature activation of the material, non-specific release or failure at critical moments. Finally, controllable manufacturing and large-scale production are engineering obstacles that must be overcome. Scaling up the successful 3D printing or microfluidic processes in the laboratory, while ensuring the uniform distribution of nanozymes in the gel, stable mechanical properties and high repeatability between batches, and establishing a complete quality control system covering the key performance parameters of materials, is the inevitable path to achieving the transformation from samples to products. Moreover, sterilization and long-term storage stability are decisive yet often overlooked factors for clinical translation. Standard sterilization techniques (e.g., autoclaving, ethylene oxide, or gamma irradiation) pose risks of altering hydrogel mechanics (via polymer degradation or excessive crosslinking) and compromising nanozyme activity (via surface ligand stripping or aggregation). Consequently, future engineering efforts must focus on optimizing sterilization protocols that balance sterility with functional integrity. Regarding storage, developing advanced lyophilization (freeze-drying) technologies with appropriate cryoprotectants is essential to prevent moisture-induced degradation and nanozyme leaching, thereby enabling the production of products with stable mechanical and catalytic performance.
Future paradigms: AI-driven design and therapeutic integration
Facing these challenges, the future development will present several clear and exciting trends. First of all, the design paradigm will undergo a fundamental transformation from bionic simulation to artificial intelligence and computation-driven rational design. One of the key driving forces behind this transformation is Machine Learning, a highly watched and rapidly developing branch in the field of artificial intelligence. Machine learning has demonstrated powerful capabilities in multiple fields such as natural language processing, image recognition, and medical diagnosis[165-167]. In the field of materials science, it is becoming a revolutionary tool for mining patterns, predicting performance and accelerating discovery from massive amounts of data[168]. By combining machine learning with high-throughput computing, the optimal solution can be reverse-engineered from a vast array of material combinations. Meanwhile, by using operando spectroscopy technology and density functional theory calculations, it is expected to uncover the black box of catalysis, precisely analyze the reaction mechanism at the atomic level, and thereby guide the design of high-performance nanozymes with activity and selectivity approaching those of natural enzymes. In addition, the Safety-by-Design concept will become the core principle of material Design. The development of composite materials that are fully biodegradable and whose degradation products (such as specific metal ions) have biobeneficial functions (such as promoting angiogenesis and immune regulation) will achieve a transformation from passive carriers or scaffolds to active microenvironment modulators. Finally, the deep integration with advanced biological therapies will open up new frontiers. For instance, using nanozyme-hydrogels as adjunctive platforms in CAR-T cell or stem cell therapy may help improve local immunosuppression or hypoxia, thereby supporting efficacy in solid tumors or tissue regeneration.
In conclusion, the future of metal nanozyme-hydrogel composite systems depends on our deeper understanding of the mechanisms of nanoscale-to-macroscopic interface interactions and the deep integration of interdisciplinary fields. Through continuous exploration on this path, we are expected to witness these smart materials eventually break through the bottleneck of transformation and become a key force in revolutionizing future medicine. Ultimately emerging as a core material that empowers intelligent diagnosis and treatment and innovates clinical therapy.
DECLARATIONS
Authors’ contribution
Supervised the review: Wan, W.; Zhao, D.; Zhang, K.
Conceived the topic, defined the scope, and established the overall framework: Wang, T.; Liu, Y.; Zhang, X.; Yu, L.
Performed the literature search, collected key references, and organized the materials: Wang, T.; Liu, Y.; Zhang, X.; Yu, L.; Wang, J.; Tian, Y.; Zhuo, Y.
Synthesized the evidence and wrote the initial draft: Wang, T.; Liu, Y.; Zhang, X.; Yu, L.
Prepared the figures and tables with input from Wang, T. and Liu, Y.: Zhang, X.; Wang, J.
Provided critical revisions and refined the manuscript for important intellectual content: Wan, W.; Zhao, D.; Zhang, K.
All authors discussed the content and commented on the manuscript. Wang, T., Liu, Y., Zhang, X., and Yu, L. contributed equally to this work. Wan, W., Zhao, D. and Zhang, K. are the corresponding authors.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version 5.2) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the Natural Science Foundation of Liaoning Province, Grant No. 2024-MS-075; and the Liaoning Province Key R&D Project (No. 2024JH2/102400049).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Bray, F.; Laversanne, M.; Sung, H.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer. J. Clin. 2024, 74, 229-63.
2. Wu, K.; El Zowalaty, A. E.; Sayin, V. I.; Papagiannakopoulos, T. The pleiotropic functions of reactive oxygen species in cancer. Nat. Cancer. 2024, 5, 384-99.
3. Hong, Y.; Boiti, A.; Vallone, D.; Foulkes, N. S. Reactive oxygen species signaling and oxidative stress: transcriptional regulation and evolution. Antioxidants 2024, 13.
4. Hunt, M.; Torres, M.; Bachar-Wikstrom, E.; Wikstrom, J. D. Cellular and molecular roles of reactive oxygen species in wound healing. Commun. Biol. 2024, 7, 1534.
5. Feng, G.; Zhang, H.; Liu, H.; et al. Natural flavonoid-derived enzyme mimics dhknase balance the two-edged reactive oxygen species function for wound healing and inflammatory bowel disease therapy. Research 2024, 7, 0464.
6. Kasai, S.; Shimizu, S.; Tatara, Y.; Mimura, J.; Itoh, K. Regulation of Nrf2 by mitochondrial reactive oxygen species in physiology and pathology. Biomolecules 2020, 10.
7. Juan, C. A.; Pérez de la Lastra, J. M.; Plou, F. J.; Pérez-Lebeña, E. The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. Int. J. Mol. Sci. 2021, 22.
8. Zuo, J.; Zhang, Z.; Luo, M.; et al. Redox signaling at the crossroads of human health and disease. MedComm 2022, 3, e127.
9. Schieber, M.; Chandel, N. S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453-62.
10. Yang, J.; Luo, J.; Tian, X.; Zhao, Y.; Li, Y.; Wu, X. Progress in understanding oxidative stress, aging, and aging-related diseases. Antioxidants 2024, 13.
11. Li, B.; Ming, H.; Qin, S.; et al. Redox regulation: mechanisms, biology and therapeutic targets in diseases. Signal. Transduct. Target. Ther. 2025, 10, 72.
12. Gao, X.; Zhang, J.; Gong, Y.; Yan, L. The biomedical applications of nanozymes in orthopaedics based on regulating reactive oxygen species. J. Nanobiotechnol. 2024, 22, 569.
13. Jiang, D.; Ni, D.; Rosenkrans, Z. T.; Huang, P.; Yan, X.; Cai, W. Nanozyme: new horizons for responsive biomedical applications. Chem. Soc. Rev. 2019, 48, 3683-704.
14. Gao, L.; Zhuang, J.; Nie, L.; et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577-83.
15. Zhang, R.; Yan, X.; Gao, L.; Fan, K. Nanozymes expanding the boundaries of biocatalysis. Nat. Commun. 2025, 16, 6817.
16. Fu, Q.; Wei, C.; Wang, M. Transition-metal-based nanozymes: synthesis, mechanisms of therapeutic action, and applications in cancer treatment. ACS. Nano. 2024, 18, 12049-95.
17. Qiu, M.; Man, C.; Zhao, Q.; et al. Nanozymes meet hydrogels: fabrication, progressive applications, and perspectives. Adv. Colloid. Interface. Sci. 2025, 338, 103404.
18. Makkar, S.; Sarawagi, N.; Priyadarshi, N.; et al. Recent advancements in nanozyme hydrogel based system for enhanced sensing applications. Biosens. Bioelectron.:. X. 2024, 21, 100548.
19. Kurian, A. G.; Singh, R. K.; Sagar, V.; Lee, J. H.; Kim, H. W. Nanozyme-engineered hydrogels for anti-inflammation and skin regeneration. Nanomicro. Lett. 2024, 16, 110.
20. Rijns, L.; Rutten, M. G. T. A.; Vrehen, A. F.; Aldana, A. A.; Baker, M. B.; Dankers, P. Y. W. Mimicking the extracellular world: from natural to fully synthetic matrices utilizing supramolecular biomaterials. Nanoscale 2024, 16, 16290-312.
21. Zhao, Y.; Dong, X.; Li, Y.; et al. Integrated cross-scale manipulation and modulable encapsulation of cell-laden hydrogel for constructing tissue-mimicking microstructures. Research 2024, 7, 0414.
22. Li, H.; Liu, Z.; Zhang, P.; Zhang, D. The recent research progress in the application of the nanozyme-hydrogel composite system for drug delivery. Drug. Delivery. 2024, 31, 2417986.
23. Yu, H.; Gao, R.; Liu, Y.; Fu, L.; Zhou, J.; Li, L. Stimulus-responsive hydrogels as drug delivery systems for inflammation targeted therapy. Adv. Sci. 2024, 11, e2306152.
24. Chen, Z.; Song, S.; Zeng, H.; Ge, Z.; Liu, B.; Fan, Z. 3D printing MOF nanozyme hydrogel with dual enzymatic activities and visualized glucose monitoring for diabetic wound healing. Chem. Eng. J. 2023, 471, 144649.
25. Wang, X.; Shu, C.; Wang, G.; et al. Recent progress of noble metal-based nanozymes: structural engineering and biomedical applications. Nanoscale 2025, 17, 10557-80.
26. Zhang, Y.; Gao, W.; Ma, Y.; et al. Integrating Pt nanoparticles with carbon nanodots to achieve robust cascade superoxide dismutase-catalase nanozyme for antioxidant therapy. Nano. Today. 2023, 49, 101768.
27. Lee, H.; Chuang, Y. C.; Tsai, J.; et al. Gold nanodandelions as nanozymes and reactive oxygen species scavengers in tumor microenvironment components. ACS. Appl. Nano. Mater. 2023, 6, 21359-70.
28. Zhong, S.; Zhang, Z.; Zhao, Q.; et al. Lattice expansion in ruthenium nanozymes improves catalytic activity and electro-responsiveness for boosting cancer therapy. Nat. Commun. 2024, 15, 8097.
29. Lin, P.; Qian, Z.; Liu, S.; et al. A single-cell RNA sequencing guided multienzymatic hydrogel design for self-regenerative repair in diabetic mandibular defects. Adv. Mater. 2024, 36, e2410962.
30. Kus-Liśkiewicz, M.; Fickers, P.; Ben Tahar, I. Biocompatibility and cytotoxicity of gold nanoparticles: recent advances in methodologies and regulations. Int. J. Mol. Sci. 2021, 22.
31. Bai, Y.; Li, Y.; Li, Y.; Tian, L. Advanced biological applications of cerium oxide nanozymes in disease related to oxidative damage. ACS. Omega. 2024, 9, 8601-14.
32. Cai, Y.; Ren, W.; Du, C.; et al. Cerium oxide nanozymes: exploring synthesis, catalytic activity, and biomedical potential. Small 2025, 21, e2504436.
33. Zhang, F.; Zhang, H.; Wang, S.; et al. A dynamically phase-adaptive regulating hydrogel promotes ultrafast anti-fibrotic wound healing. Nat. Commun. 2025, 16, 3738.
34. Zhang, Y.; Khalique, A.; Du, X.; et al. Biomimetic design of mitochondria-targeted hybrid nanozymes as superoxide scavengers. Adv. Mater. 2021, 33, e2006570.
35. Long, M.; Wang, L.; Kang, L.; et al. Prussian blue nanozyme featuring enhanced superoxide dismutase-like activity for myocardial ischemia reperfusion injury treatment. ACS. Nano. 2025, 19, 4561-81.
36. Ding, H.; Long, M.; Wu, Y.; et al. Subcellular distribution of Prussian blue nanozymes dictates enzymatic activity and macrophage polarization for effective colitis therapy. Biomaterials 2026, 325, 123622.
37. Zhang, Y.; Yuan, X.; Guo, X.; et al. All-in-one zinc-doped prussian blue nanozyme for efficient capture, separation, and detection of copper ion (Cu2+) in complicated matrixes. Small 2024, 20, e2306961.
38. Dheyab, M. A.; Abdullah, W.; Aziz, A. A.; et al. Two birds, one stone: smart MOF nanozymes for dual catalysis and theranostics. Small 2025, 21, e09992.
39. Fan, Z.; He, P.; Bai, H.; et al. Green recycling of waste poly(ethylene terephthalate) into Ni-MOF nanorod for simultaneous interfacial solar evaporation and photocatalytic degradation of organic pollutants. EcoMat 2023, 6, e12422.
40. Fan, X.; Pan, M.; Li, X.; Kong, L.; Kuchmizha, A.; Xu, H. Research progress of MOF electrochromic materials. Resources. Chemicals. and. Materials. 2024, 3, 230-45.
41. Liu, C.; Chen, Y.; Xu, X.; Yin, M.; Zhang, H.; Su, W. Utilizing macrophages missile for sulfate-based nanomedicine delivery in lung cancer therapy. Research 2024, 7, 0448.
42. Islam, M. R.; Afroj, S.; Tan, S.; Eichhorn, S. J.; Novoselov, K. S.; Karim, N. Inkjet-printed metal–organic frameworks for smart e-textile supercapacitors. EcoMat 2025, 7, e70020.
43. Li, Y.; Chai, H.; Yuan, Z.; et al. Metal–organic framework-engineered enzyme/nanozyme composites: preparation, functionality, and sensing mechanisms. Chem. Eng. J. 2024, 496, 153884.
44. Lee, D.; Lee, S.; Choi, I.; Kim, M. Positional functionalizations of metal-organic frameworks through invasive ligand exchange and additory MOF-on-MOF strategies: a review. Smart. Mol. 2024, 2, e20240002.
45. Chao, D.; Dong, Q.; Yu, Z.; et al. Specific nanodrug for diabetic chronic wounds based on antioxidase-mimicking MOF-818 nanozymes. J. Am. Chem. Soc. 2022, 144, 23438-47.
46. Sun, Q.; Liu, S.; Li, Z.; et al. Spatial coordination structure-driven enzyme-like selectivity in single-atom nanozymes. Adv. Mater. 2025, 37, e2508125.
47. Su, J.; Wang, P.; Zhou, W.; et al. Single-site iron-anchored amyloid hydrogels as catalytic platforms for alcohol detoxification. Nat. Nanotechnol. 2024, 19, 1168-77.
48. Fan, C.; Pan, Y.; Wu, D.; et al. FePtCoNiCu high-entropy nanozymes embedded in a hydrogel matrix as a portable, rapid, and visual on-site biosensing platform. Anal. Chem. 2025, 97, 22368-79.
49. Nandhini, J.; Karthikeyan, E.; Rajeshkumar, S. Green synthesis of zinc oxide nanoparticles: eco-friendly advancements for biomedical marvels. Resour. Chem. Mater. 2024, 3, 294-316.
50. He, X.; Wang, Z.; Mao, B.; et al. Trimetallic Pt-Pd-Au alloy nanozymes for multimodal synergistic therapy to overcome deep-seated drug-resistant infections via ROS cascade. Bioact. Mater. 2025, 51, 841-57.
51. Zhu, X.; Zhang, Y.; He, Y.; et al. Self-enriching nanozyme with photothermal-cascade amplification for tumor microenvironment-responsive synergistic therapy and enhanced photoacoustic imaging. Mater. Today. Bio. 2025, 34, 102230.
52. Lyu, S.; Li, L.; Gao, J.; Liu, D.; Song, F. Metallo-supramolecular nanofibers based on type-I photosensitizer for synergistic antibacterial therapy. Smart. Mol. 2024, 2, e20240037.
53. Gu, H.; Sun, W.; Du, J.; Fan, J.; Peng, X. Dual-acceptor engineering of donor-acceptor type molecules for all-round boosting anti-tumor phototherapy. Smart. Mol. 2024, 2, e20230014.
54. Zhao, Z.; Chen, T.; Li, J.; Xue, X.; Ge, J.; Wang, P. A NIR light activated self-reporting carbon dots assembly as phototheranostics for tumor photodynamic therapy. Smart. Mol. 2025, 3, e20240049.
55. Zhang, T.; Meng, Z.; Yu, H.; Ding, P.; Kai, T. An intelligent and conductive hydrogel with multiresponsive and ROS scavenging properties for infection prevention and anti-inflammatory treatment assisted by electrical stimulation for diabetic wound. Adv. Sci. 2025, 12, e2500696.
57. Li, X.; Li, L.; Liu, X.; Hu, Y. Hydrogel actuators: from building blocks selection, anisotropic structures design to multifunctional applications. Soft. Sci. 2025, 5, 41.
58. Cao, H.; Duan, L.; Zhang, Y.; Cao, J.; Zhang, K. Current hydrogel advances in physicochemical and biological response-driven biomedical application diversity. Signal. Transduct. Target. Ther. 2021, 6, 426.
59. Tang, Z.; Deng, L.; Zhang, J.; et al. Intelligent hydrogel-assisted hepatocellular carcinoma therapy. Research 2024, 7, 0477.
60. Ma, X.; Sekhar, K. P. C.; Zhang, P.; Cui, J. Advances in stimuli-responsive injectable hydrogels for biomedical applications. Biomater. Sci. 2024, 12, 5468-80.
61. Gholamali, I.; Vu, T. T.; Jo, S. H.; Park, S. H.; Lim, K. T. Exploring the progress of hyaluronic acid hydrogels: synthesis, characteristics, and wide-ranging applications. Materials 2024, 17.
62. Hong, F.; Qiu, P.; Wang, Y.; et al. Chitosan-based hydrogels: from preparation to applications, a review. Food. Chem. X. 2024, 21, 101095.
63. Kavand, A.; Noverraz, F.; Gerber-Lemaire, S. Recent advances in alginate-based hydrogels for cell transplantation applications. Pharmaceutics 2024, 16.
64. Liu, L.; Jiang, H.; Wang, X. Alkaline phosphatase-responsive Zn2+ double-triggered nucleotide capped gold nanoclusters/ alginate hydrogel with recyclable nanozyme capability. Biosens. Bioelectron. 2021, 173, 112786.
65. Jiao, L.; Xu, W.; Yan, H.; et al. A dopamine-induced Au hydrogel nanozyme for enhanced biomimetic catalysis. Chem. Commun. 2019, 55, 9865-8.
66. Li, N.; Qi, X.; Ma, L.; et al. Non-lytic nanozyme hydrogel for the treatment of fungal keratitis by silent pathogen eradication and host inflammation suppression. Chem. Eng. J. 2026, 527, 171844.
67. Li, Q.; Dong, M.; Han, Q.; et al. Enhancing diabetic wound healing with a pH-responsive nanozyme hydrogel featuring multi-enzyme-like activities and oxygen self-supply. J. Control. Release. 2024, 365, 905-18.
68. Xu, K.; Zou, Y.; Lin, C.; et al. Cascade catalysis nanozyme for interfacial functionalization in combating implant infections associated with diabetes via sonodynamic therapy and adaptive immune activation. Biomaterials 2024, 311, 122649.
69. Zhu, X.; Mao, X.; Wang, Z.; Feng, C.; Chen, G.; Li, G. Fabrication of nanozyme@DNA hydrogel and its application in biomedical analysis. Nano. Res. 2016, 10, 959-70.
70. Li, D.; Ai, Z.; Tian, Y.; Wang, Y.; Zhang, C. Bioactive prosthetic interface constructed with cascading multi-nanozyme hydrogel to induce M2 macrophage polarization and heal diabetic bone defects. Biomaterials 2026, 325, 123624.
71. Liu, Y.; Ye, R.; Yang, J.; et al. Multifunctional oral hydrogel containing dual-active Pt/Mn3O4 nanozyme for synergistic pyroptosis suppression and gut microenvironment reprogramming to alleviate radiation intestinal injury. Small 2026, 22, e11251.
72. Aggarwal, M.; Ghosh, P.; Mohithraj, K. P.; et al. Hydrothermal synthesis of light-responsive adhesive protein polymeric dot nanogel for wound healing. ACS. Appl. Mater. Interfaces. 2026, 18, 7803-17.
73. Huang, L.; Feng, Y.; Chen, H.; et al. Metal valence transition strategy-engineered bimetallic nanozyme thermosensitive hydrogel for deep-tissue therapy of subcutaneous abscess wounds and bacterial corneal ulcers. J. Colloid. Interface. Sci. 2025, 700, 138613.
74. Zhang, X.; Qu, H.; Li, X.; et al. Autonomous atmospheric water harvesting over a wide RH Range enabled by super hygroscopic composite aerogels. Adv. Mater. 2024, 36, e2310219.
75. Zhou, X.; Yu, X.; You, T.; et al. 3D printing-based hydrogel dressings for wound healing. Adv. Sci. 2024, 11, e2404580.
76. Liu, B.; Liu, Q.; Feng, J. Operando colorations from real-time growth of 3D-printed nanoarchitectures. Adv. Mater. 2024, 36, e2404977.
77. Lian, L.; Xie, M.; Luo, Z.; et al. Rapid volumetric bioprinting of decellularized extracellular matrix bioinks. Adv. Mater. 2024, 36, e2304846.
78. Hafa, L.; Breideband, L.; Ramirez Posada, L.; et al. Light sheet-based laser patterning bioprinting produces long-term viable full-thickness skin constructs. Adv. Mater. 2024, 36, e2306258.
79. Jin, Y.; Zhang, J.; Chen, X.; et al. 3D printing incorporating gold nanozymes with mesenchymal stem cell-derived hepatic spheroids for acute liver failure treatment. Biomaterials 2025, 315, 122895.
80. Su, Y.; Ju, J.; Shen, C.; et al. In situ 3D bioprinted GDMA/Prussian blue nanozyme hydrogel with wet adhesion promotes macrophage phenotype modulation and intestinal defect repair. Mater. Today. Bio. 2025, 31, 101636.
81. Dong, L.; Fan, Z.; Fang, B.; et al. Oriented cellulose hydrogel: directed tissue regeneration for reducing corneal leukoplakia and managing fungal corneal ulcers. Bioact. Mater. 2024, 41, 15-29.
82. Liu, J.; Du, C.; Huang, W.; Lei, Y. Injectable smart stimuli-responsive hydrogels: pioneering advancements in biomedical applications. Biomater. Sci. 2023, 12, 8-56.
83. Wang, Z.; Zhai, B.; Sun, J.; et al. Recent advances of injectable in situ-forming hydrogels for preventing postoperative tumor recurrence. Drug. Delivery. 2024, 31, 2400476.
84. Shen, J.; Chen, A.; Cai, Z.; et al. Exhausted local lactate accumulation via injectable nanozyme-functionalized hydrogel microsphere for inflammation relief and tissue regeneration. Bioact. Mater. 2022, 12, 153-68.
85. Feng, Q.; Li, Q.; Lin, Z.; et al. Injectable microspheres with cartilage-like structure facilitate inflammation inhibition and tissue regeneration. Adv. Healthc. Mater. 2025, 14, e2404877.
86. Liu, X.; Gao, M.; Li, B.; et al. Bioinspired capillary transistors. Adv. Mater. 2024, 36, e2310797.
87. Wang, S.; Zeng, N.; Zhang, Q.; Chen, M.; Huang, Q. Nanozyme hydrogels for self-augmented sonodynamic/photothermal combination therapy. Front. Oncol. 2022, 12, 888855.
88. Wang, S.; Fei, H.; Ma, Y.; et al. Cu-doped polypyrrole hydrogel with tumor catalyst activity for NIR-II thermo-radiotherapy. Front. Bioeng. Biotechnol. 2023, 11, 1225937.
89. Wang, S.; Zhang, Q.; Zeng, N.; Qi, P.; Huang, C.; Huang, Q. Injectable hydrogel system for camptothecin initiated nanocatalytic tumor therapy with high performance. Front. Oncol. 2022, 12, 904960.
90. Wang, Z.; Zeng, W.; Chen, Z.; Suo, W.; Quan, H.; Tan, Z. J. An intratumoral injectable nanozyme hydrogel for hypoxia-resistant thermoradiotherapy. Colloids. Surf. B. Biointerfaces. 2021, 207, 112026.
91. Hao, Y.; Liu, Y.; Wu, Y.; et al. A robust hybrid nanozyme@hydrogel platform as a biomimetic cascade bioreactor for combination antitumor therapy. Biomater. Sci. 2020, 8, 1830-9.
92. Wu, H.; Liu, L.; Song, L.; Ma, M.; Gu, N.; Zhang, Y. Enhanced tumor synergistic therapy by injectable magnetic hydrogel mediated generation of hyperthermia and highly toxic reactive oxygen species. ACS. Nano. 2019, 13, 14013-23.
93. Wu, C. Y.; Hsu, Y. H.; Chen, Y.; et al. Robust O2 supplementation from a trimetallic nanozyme-based self-sufficient complementary system synergistically enhances the starvation/photothermal therapy against hypoxic tumors. ACS. Appl. Mater. Interfaces. 2021, 13, 38090-104.
94. Liu, Y.; Ding, L.; Chen, G.; Wang, P.; Wang, X. A thermo-sensitive hydrogel with prominent hemostatic effect prevents tumor recurrence via anti-anoikis-resistance. J. Nanobiotechnol. 2024, 22, 496.
95. Xu, S.; Zhang, H.; Qian, Z.; Yuan, W. pH-Responsive injectable self-healing hydrogels loading Au nanoparticles-decorated bimetallic organic frameworks for synergistic sonodynamic-chemodynamic-starvation-chemo therapy of cancer. J. Colloid. Interface. Sci. 2024, 675, 746-60.
96. Lu, S.; Wu, Y.; Liu, Y.; Sun, X.; Li, J.; Li, J. Multifunctional photothermal hydrogel in the second near-infrared window for localized tumor therapy. ACS. Appl. Bio. Mater. 2023, 6, 4694-702.
97. Li, W.; Li, H.; Lin, C.; et al. Axially chlorinated carbon nanodots for hypoxia-tolerant neutrophil-mimicking photoenzymatic cascade therapy against postoperative tumor recurrence. ACS. Nano. 2025, 19, 25501-14.
98. Lin, L. S.; Song, J.; Song, L.; et al. Simultaneous fenton-like ion delivery and glutathione depletion by MnO2-based nanoagent to enhance chemodynamic therapy. Angew. Chem. Int. Ed. Engl. 2018, 57, 4902-6.
99. Zhang, Y.; Zhang, N.; Xing, J.; et al. In situ hydrogel based on Cu-Fe3O4 nanoclusters exploits oxidative stress and the ferroptosis/cuproptosis pathway for chemodynamic therapy. Biomaterials 2024, 311, 122675.
100. Santollani, L.; Maiorino, L.; Zhang, Y. J.; et al. Local delivery of cell surface-targeted immunocytokines programs systemic antitumor immunity. Nat. Immunol. 2024, 25, 1820-9.
101. Miao, D.; Luo, R.; Li, Y.; et al. Injectable functional porous carbon-loaded hydrogel with long-term retention and synergistic anti-tumor properties for tumor therapy. Adv. Mater. 2025, 37, e2502455.
102. Huan, Z.; Li, J.; Luo, Z.; Yu, Y.; Li, L. Hydrogel-encapsulated pancreatic islet cells as a promising strategy for diabetic cell therapy. Research 2024, 7, 0403.
103. Wang, Y.; Feng, C.; Yu, B.; et al. Enhanced effects of intermittent fasting by magnetic fields in severe diabetes. Research 2024, 7, 0468.
105. Dawi, J.; Tumanyan, K.; Tomas, K.; et al. Diabetic foot ulcers: pathophysiology, immune dysregulation, and emerging therapeutic strategies. Biomedicines 2025, 13, 1076.
106. Lopes, F. B.; Sarandy, M. M.; Novaes, R. D.; Valacchi, G.; Gonçalves, R. V. OxInflammatory responses in the wound healing process: a systematic review. Antioxidants 2024, 13, 823.
107. Xiong, Y.; Chu, X.; Yu, T.; et al. Reactive oxygen species-scavenging nanosystems in the treatment of diabetic wounds. Adv. Healthc. Mater. 2023, 12, e2300779.
108. Tu, C.; Lu, H.; Zhou, T.; et al. Promoting the healing of infected diabetic wound by an anti-bacterial and nano-enzyme-containing hydrogel with inflammation-suppressing, ROS-scavenging, oxygen and nitric oxide-generating properties. Biomaterials 2022, 286, 121597.
109. Pu, C.; Wang, Y.; Xiang, H.; et al. Zinc-based polyoxometalate nanozyme functionalized hydrogels for optimizing the hyperglycemic-immune microenvironment to promote diabetic wound regeneration. J. Nanobiotechnol. 2024, 22, 611.
110. Peng, Y.; Guo, Y.; Ge, X.; et al. Construction of programmed time-released multifunctional hydrogel with antibacterial and anti-inflammatory properties for impaired wound healing. J. Nanobiotechnol. 2024, 22, 126.
111. Che, J.; Huang, D.; Wang, Y.; Gao, G.; Zhao, Y. Natural multi-active nanoparticles integrated biological hydrogel microcarriers for wound healing. J. Nanobiotechnol. 2025, 23, 582.
112. Yan, J.; Zhao, Y.; Cui, C.; et al. Dynamic multistage nanozyme hydrogel reprograms diabetic wound microenvironment: synergistic oxidative stress alleviation and mitochondrial restoration. Mater. Today. Bio. 2025, 32, 101780.
113. Yang, W.; Liu, L.; Cheng, X. Research progress on scar-free healing. Ann. Med. 2025, 57, 2575109.
114. Cavallo, I.; Sivori, F.; Mastrofrancesco, A.; et al. Bacterial biofilm in chronic wounds and possible therapeutic approaches. Biology 2024, 13.
115. Das, T.; Das, M.; Sultana, S.; Swain, R. Stimuli-responsive nanogels in wound care: a comprehensive review. Next. Nanotechnol. 2025, 8, 100260.
116. Guo, J.; Cao, Y.; Wu, Q. Y.; Zhou, Y. M.; Cao, Y. H.; Cen, L. S. Implications of pH and ionic environment in chronic diabetic wounds: an overlooked perspective. Clin. Cosmet. Investig. Dermatol. 2024, 17, 2669-86.
117. Li, Y.; Wang, D.; Wen, J.; et al. Chemically grafted nanozyme composite cryogels to enhance antibacterial and biocompatible performance for bioliquid regulation and adaptive bacteria trapping. ACS. Nano. 2021, 15, 19672-83.
118. Yang, Y.; Wang, J.; Huang, S.; et al. Bacteria-responsive programmed self-activating antibacterial hydrogel to remodel regeneration microenvironment for infected wound healing. Natl. Sci. Rev. 2024, 11, nwae044.
119. Wang, X.; Shan, M.; Zhang, S.; et al. Stimuli-responsive antibacterial materials: molecular structures, design principles, and biomedical applications. Adv. Sci. 2022, 9, e2104843.
120. Yang, Y.; Fang, Q.; Wang, J.; et al. Glucose-activated programmed hydrogel with self-switchable enzyme-like activity for infected diabetic wound self-adaptive treatment. Adv. Mater. 2025, 37, e2419158.
121. Jiang, S.; Xie, D.; Hu, Z.; et al. Enhanced diabetic wound healing with injectable hydrogel containing self-assembling nanozymes. J. Control. Release. 2024, 372, 265-80.
122. Li, L.; Wang, Y.; Hu, S.; et al. Peroxidase-like copper-doped carbon-dots embedded in hydrogels for stimuli-responsive bacterial biofilm elimination and wound healing. Acta. Biomater. 2025, 195, 467-78.
123. He, L.; Di, D.; Chu, X.; et al. Photothermal antibacterial materials to promote wound healing. J. Control. Release. 2023, 363, 180-200.
124. Cui, Y.; Niu, Q.; Hu, Z.; et al. Multifunctional nanozyme-hydrogel system in bacterial-infected wounds. Cell. Biomater. 2025, 1, 100074.
125. Feng, Y.; Su, L.; Zhang, Z.; et al. pH-responsive wound dressing based on biodegradable CuP nanozymes for treating infected and diabetic wounds. ACS. Appl. Mater. Interfaces. 2024, 16, 95-110.
126. Wang, Q.; Jiang, J.; Gao, L. Catalytic antimicrobial therapy using nanozymes. Wiley. Interdiscip. Rev. Nanomed. Nanobiotechnol. 2022, 14, e1769.
127. Ren, X.; Wang, X.; Yang, J.; et al. Multi-enzyme-based superabsorbent hydrogel for self-enhanced NIR-II photothermal-catalytic antibacterial therapy. Adv. Healthc. Mater. 2024, 13, e2303537.
128. Liang, Z.; Sang, Z.; Li, Y.; Chen, Z.; Mei, X.; Ren, X. Enhancing the performance of injectable self-activating PVA-alginate hydrogel by Ag@MXene nanozyme as NIR responsive and photoenhanced antibacterial platform for wound healing. Carbohydr. Polym. 2025, 357, 123434.
129. Bibi, F.; Hanan, A.; Soomro, I. A.; Numan, A.; Khalid, M. Double transition metal MXenes for enhanced electrochemical applications: challenges and opportunities. EcoMat 2024, 6, e12485.
130. Zhuang, Z. M.; Wang, Y.; Xu, F. W.; et al. Programmed nanozyme hydrogel enabling spatiotemporal modulation of wound healing achieves skin regeneration after biofilm infection. J. Nanobiotechnol. 2025, 23, 694.
131. Li, Y.; Fu, R.; Duan, Z.; Zhu, C.; Fan, D. Construction of multifunctional hydrogel based on the tannic acid-metal coating decorated MoS2 dual nanozyme for bacteria-infected wound healing. Bioact. Mater. 2022, 9, 461-74.
132. Quan, Z.; Ye, F.; Yu, H.; Li, H.; Sun, S.; Xu, Y. Bioinspired glycocalyx-mimetic hydrogel enabling photothermal-amplified therapeutic cascade for chronic wound healing. J. Colloid. Interface. Sci. 2025, 694, 137669.
133. Chen, Y. Z.; Qiu, X. O.; Wang, L.; et al. Novel ultrasound capsule endoscopy for gastrointestinal scanning: an in vivo animal study. Endosc. Ultrasound. 2024, 13, 253-8.
134. Bai, Y.; Qin, X.; Ao, X.; Ran, T.; Zhou, C.; Zou, D. The role of EUS in the diagnosis of early chronic pancreatitis. Endosc. Ultrasound. 2024, 13, 232-8.
135. Zhang, D.; Wu, C.; Yang, Z.; et al. The application of artificial intelligence in EUS. Endosc. Ultrasound. 2024, 13, 65-75.
136. Sun, X.; Chen, X.; Wang, S.; et al. Oxygenous and biofilm-targeted nanosonosensitizer anchored with Pt nanozyme and antimicrobial peptide in the gelatin/sodium alginate hydrogel for infected diabetic wound healing. Int. J. Biol. Macromol. 2025, 293, 139356.
137. Zhang, F.; Kang, Y.; Feng, L.; et al. Infected wound repair with an ultrasound-enhanced nanozyme hydrogel scaffold. Mater. Horiz. 2023, 10, 5474-83.
138. Jiang, Y.; Li, T.; Yang, J.; et al. Sustained intra-articular reactive oxygen species scavenging and alleviation of osteoarthritis by biocompatible amino-modified tantalum nanoparticles. Front. Bioeng. Biotechnol. 2023, 11, 1118850.
139. Li, Z.; Jia, G.; Su, Z.; Zhu, C. Nanozyme-based strategies against bone infection. Research 2025, 8, 0605.
140. Xi, L.; Yang, L. R.; Liu, X.; Shen, C. Nanozymes in bone regeneration: mechanistic insights and therapeutic applications. ACS. Appl. Mater. Interfaces. 2025, 17, 50103-21.
141. Wang, W.; Duan, J.; Ma, W.; et al. Trimanganese tetroxide nanozyme protects cartilage against degeneration by reducing oxidative stress in osteoarthritis. Adv. Sci. 2023, 10, e2205859.
142. Wang, F.; Han, Y.; Zhou, Q.; et al. Polymer-modified DNA hydrogels for living mitochondria and nanozyme delivery in the treatment of rheumatoid arthritis. Bioact. Mater. 2025, 47, 448-59.
143. Zhao, Y.; Song, S.; Wang, D.; et al. Nanozyme-reinforced hydrogel as a H2O2-driven oxygenerator for enhancing prosthetic interface osseointegration in rheumatoid arthritis therapy. Nat. Commun. 2022, 13, 6758.
144. He, H.; Zhang, Q.; Zhang, Y.; et al. Injectable bioadhesive and lubricating hydrogel with polyphenol mediated single atom nanozyme for rheumatoid arthritis therapy. Nat. Commun. 2025, 16, 2768.
145. Huang, W.; Hsu, J. C.; Zhou, J.; et al. Stimuli-responsive nanomaterials for targeted drug delivery in inflammatory bowel disease: Advances and emerging directions. Matter 2025, 8.
146. Sun, W.; Li, Z.; Zhang, X.; Luo, Q.; Wei, L.; Xiao, C. Biomaterial-based therapeutic strategies for inflammatory bowel disease. Biomaterials 2026, 324, 123462.
147. Gan, R.; Ni, E.; Li, G.; Chen, W. A new paradigm for precision drug delivery in inflammatory bowel disease: effective transfer, enhanced retention, and pathology-targeting treatment via biomaterials and engineered platforms. MedComm. -. Biomater. Appl. 2025, 4, e70022.
148. Yang, Z.; Zhou, X.; Wang, L.; et al. Mn3O4 nanozyme loaded thermosensitive PDLLA-PEG-PDLLA hydrogels for the treatment of inflammatory bowel disease. ACS. Appl. Mater. Interfaces. 2023, 15, 33273-87.
149. Zhang, D.; Chen, X.; Feng, Y.; Li, L.; Liu, M. Adhesive thermosensitive polydopamine hydrogel containing Mn3O4 anchored halloysite clay for treatment of ulcerative colitis. J. Colloid. Interface. Sci. 2025, 683, 147-59.
150. Li, L.; He, S.; Liao, B.; et al. Orally administrated hydrogel harnessing intratumoral microbiome and microbiota-related immune responses for potentiated colorectal cancer treatment. Research 2024, 7, 0364.
151. Zhou, P.; Sun, Q.; Huang, L.; et al. Dual-targeting Mn@CeO2 nanozyme-modified probiotic hydrogel microspheres reshape gut homeostasis in inflammatory bowel disease. ACS. Nano. 2025, 19, 31619-42.
152. Luo, L.; Liu, Q.; Zhang, Y.; et al. Precisely edited gut microbiota by tungsten-doped Prussian blue nanoparticles for the treatment of inflammatory bowel disease. J. Control. Release. 2025, 382, 113755.
153. Cao, Y.; Cheng, K.; Yang, M.; et al. Orally administration of cerium oxide nanozyme for computed tomography imaging and anti-inflammatory/anti-fibrotic therapy of inflammatory bowel disease. J. Nanobiotechnol. 2023, 21, 21.
154. Kan, L.; Li, T.; Zhang, W.; et al. Orally administered ZnxCeyO2/Se hydrogel with effective antioxidant activity for treating inflammatory bowel disease by inhibiting ferroptosis. Adv. Healthc. Mater. 2025, 14, e2500088.
155. Wu, S.; Hu, Y.; Tao, Z.; et al. Comprehensive management of ulcerative colitis and its associated intra-extra intestinal complications with a multifunctional inulin hydrogel complex. Small 2025, 21, e2500726.
156. Huang, Y.; Ding, Z.; Li, Y.; Xi, F.; Liu, J. Magnetic nanozyme based on loading nitrogen-doped carbon dots on mesoporous Fe3O4 nanoparticles for the colorimetric detection of glucose. Molecules 2023, 28.
157. Chen, M.; Zhou, H.; Liu, X.; et al. Single iron site nanozyme for ultrasensitive glucose detection. Small 2020, 16, e2002343.
158. Peng, J.; Cao, J.; Wang, L.; Guo, Z.; Hou, X. A portable hydrogel kit based on Au@GM88A/I combined with mobile phone for polychromatic semi-quantitative and quantitative sensing analysis. Biosens. Bioelectron. 2024, 266, 116682.
159. Yang, X.; Wu, F.; Huang, H.; et al. Au nanoparticles anchored carbonized ZIF-8 for enabling real-time and noninvasive glucose monitoring in sweat. Biosens. Bioelectron. 2025, 272, 117138.
160. Fei, J.; Yang, W.; Dai, Y.; et al. A biosensor based on Fe3O4@MXene-Au nanocomposites with high peroxidase-like activity for colorimetric and smartphone-based detection of glucose. Mikrochim. Acta. 2023, 190, 336.
161. Wang, Q.; Zhang, X.; Huang, L.; Zhang, Z.; Dong, S. One-pot synthesis of Fe3O4 nanoparticle loaded 3D porous graphene nanocomposites with enhanced nanozyme activity for glucose detection. ACS. Appl. Mater. Interfaces. 2017, 9, 7465-71.
162. Yin, J.; Li, Y.; Sun, X.; Qin, Z. Tough gelatin-based biogels for wearable sensors. Soft. Sci. 2025, 5, 30.
163. Cheng, W.; Zheng, Z.; Li, X.; et al. A general synthesis method for patterning PEDOT toward wearable electronics and bioelectronics. Research 2024, 7, 0383.
164. Reina, G.; Panáček, D.; Rathammer, K.; et al. Light irradiation of N-doped graphene acid: metal-free strategy toward antibacterial and antiviral coatings with dual modes of action. EcoMat 2025, 7, e70009.
165. Mahajan, S.; Siyu, S.; Bhutani, M. S. What can artificial intelligence do for EUS? Endosc. Ultrasound. 2025, 14, 1-3.
166. Zhang, C.; Tan, W.; Li, X.; et al. Risk stratification system of gastrointestinal stromal tumors under EUS elastography based on artificial intelligence. Endosc. Ultrasound. 2025, 14, 220-7.
167. Qu, H.; Yang, F.; Chen, T.; Cui, X.; Sun, S. Enhancing gastrointestinal stromal tumor risk stratification: a novel deep learning approach applied to EUS imaging. Endosc. Ultrasound 2025, 14, 120-8.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






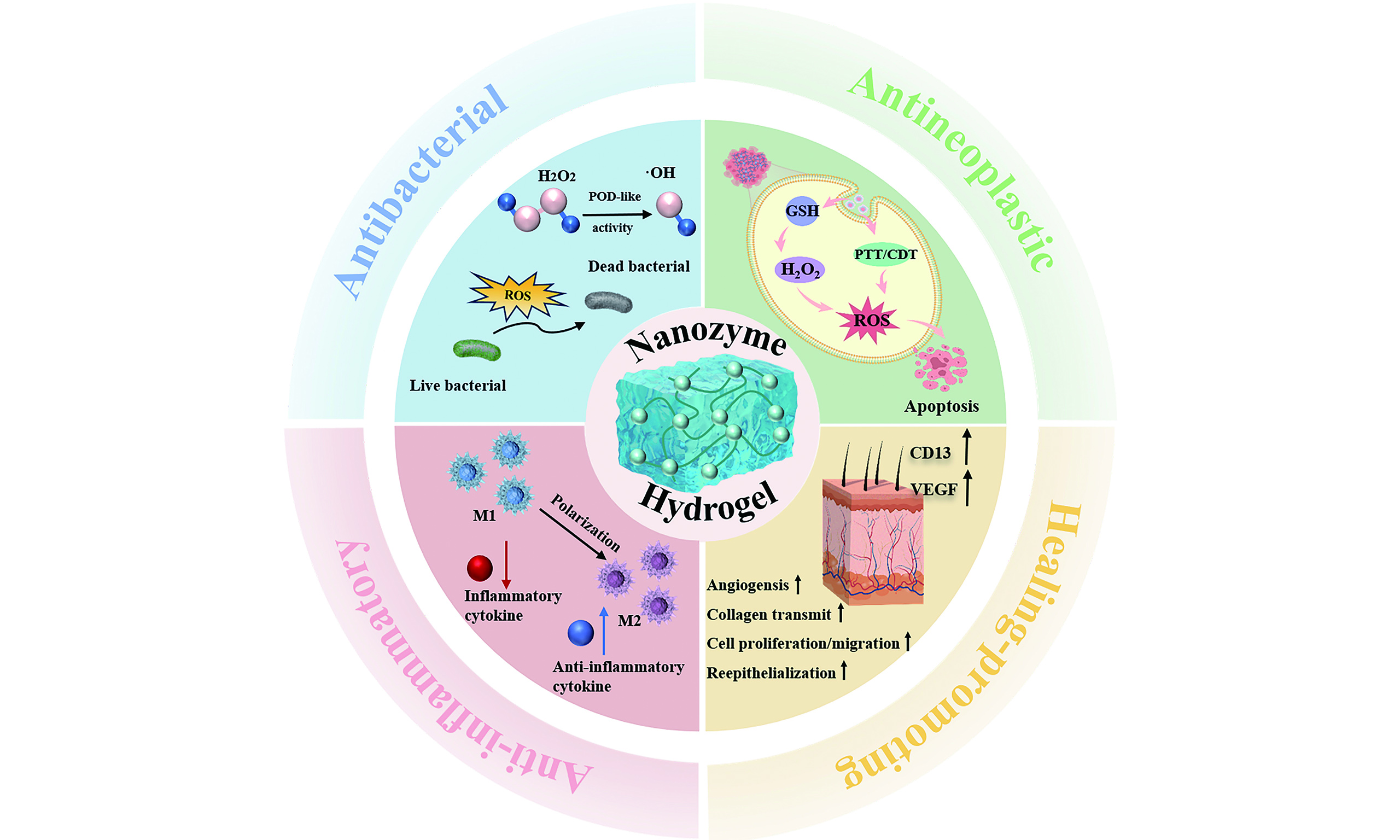
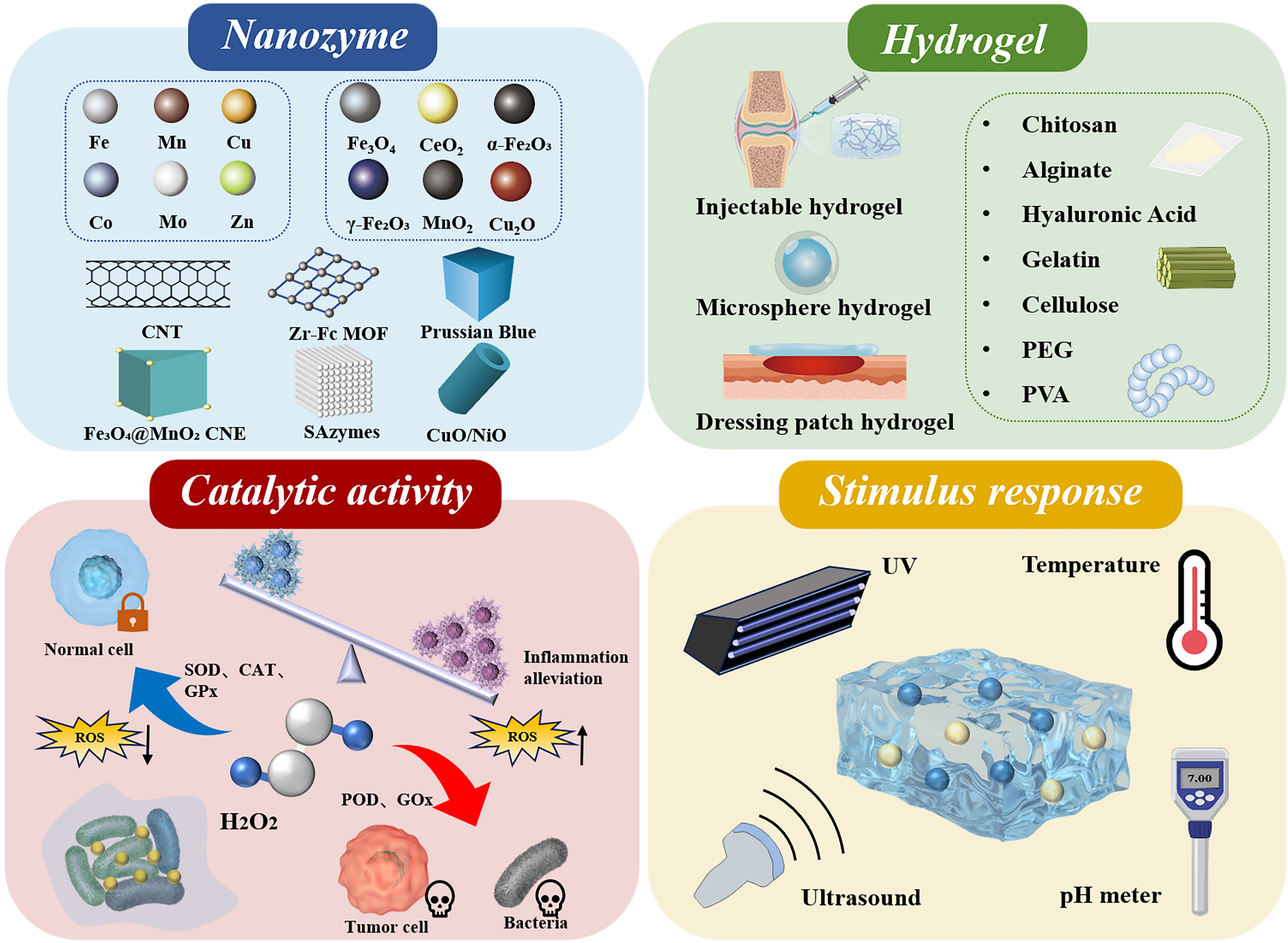
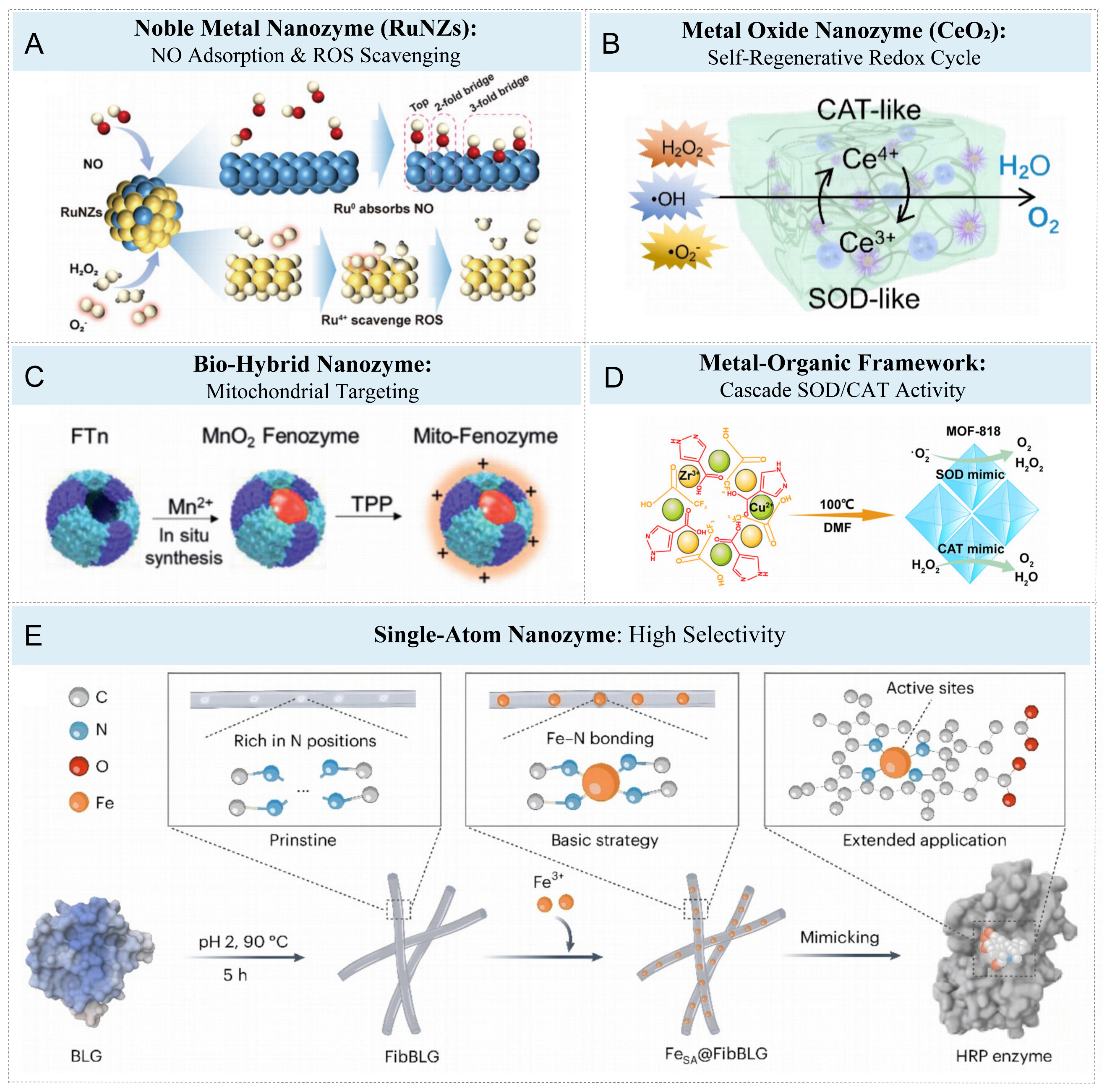
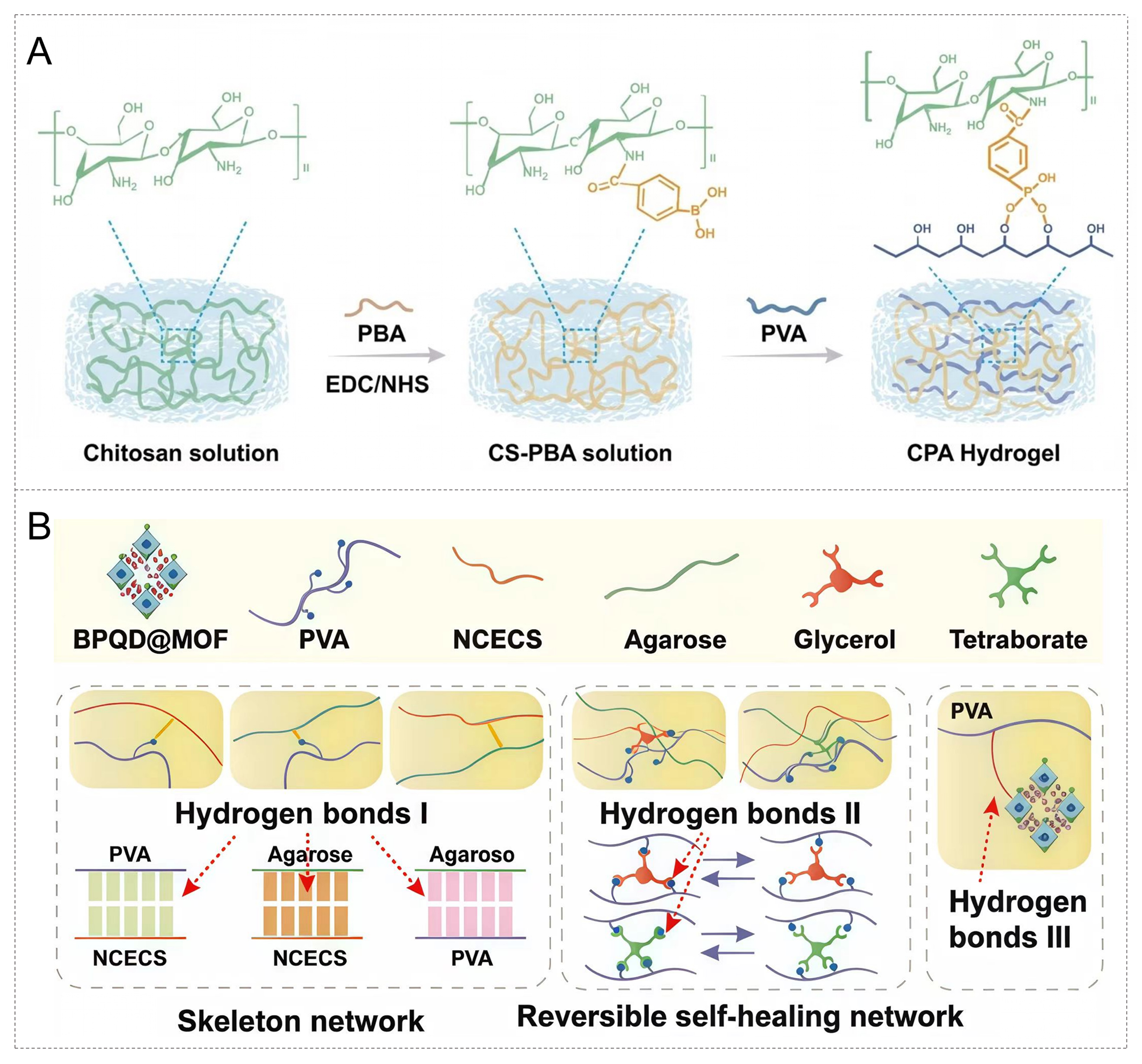
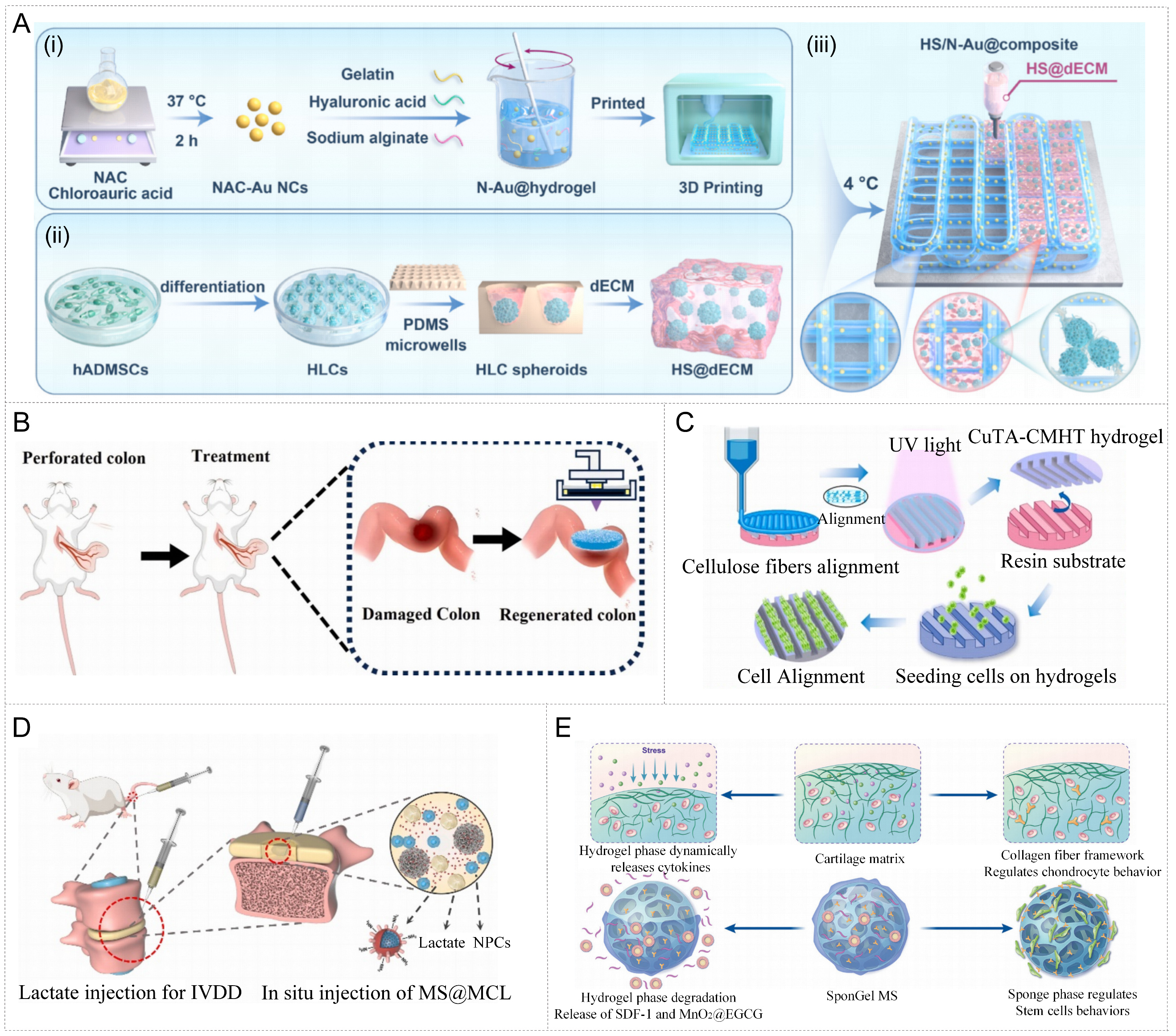
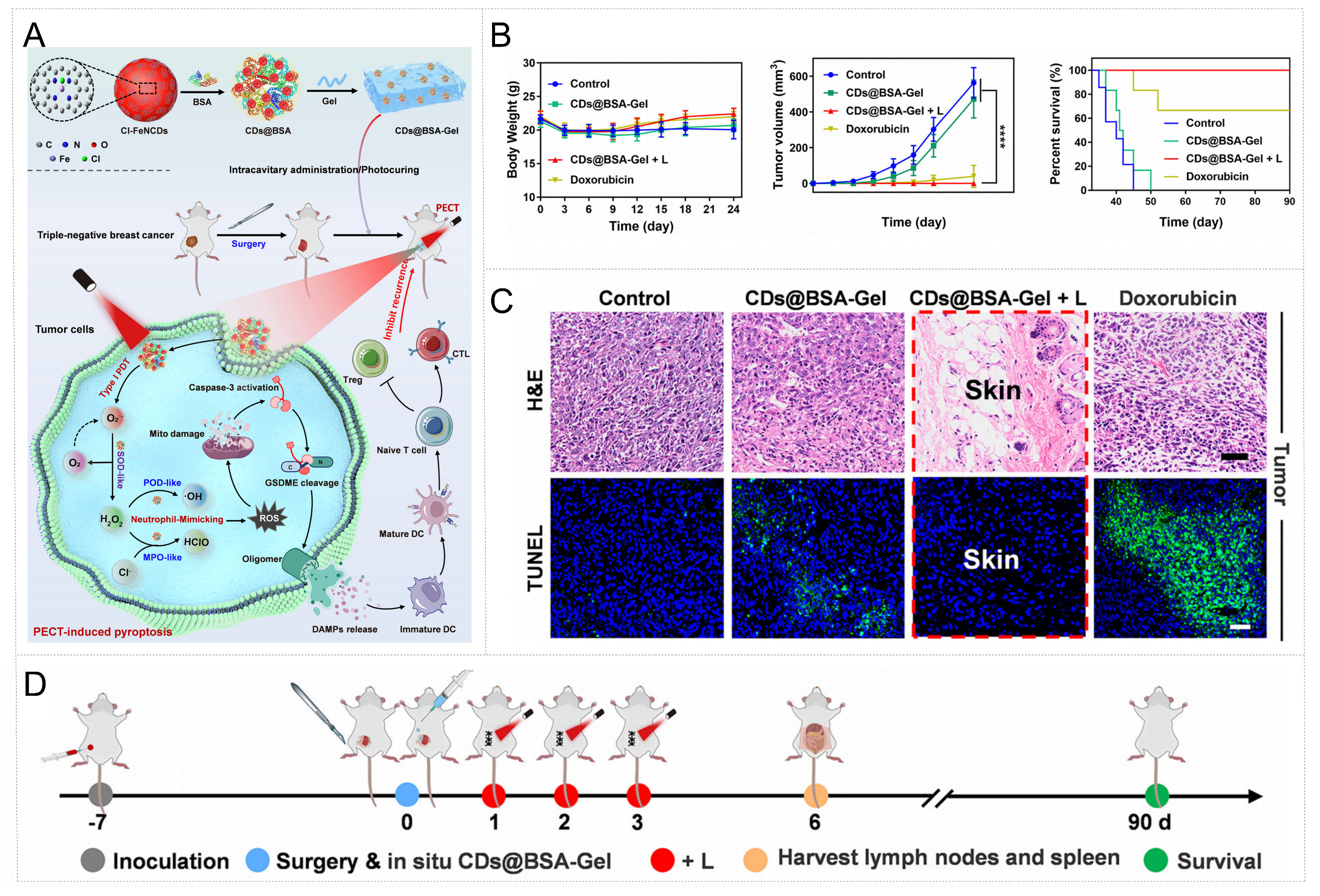
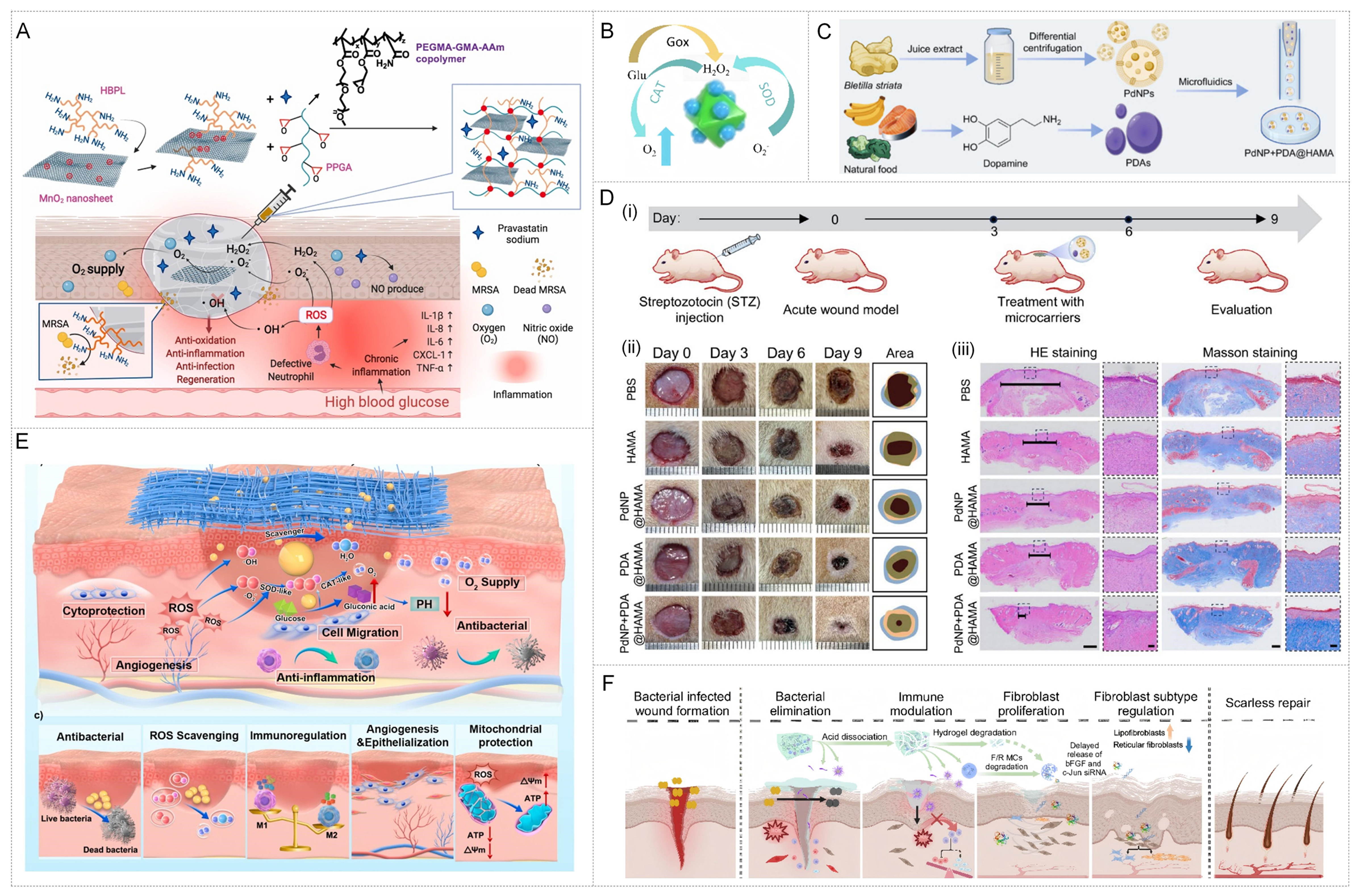
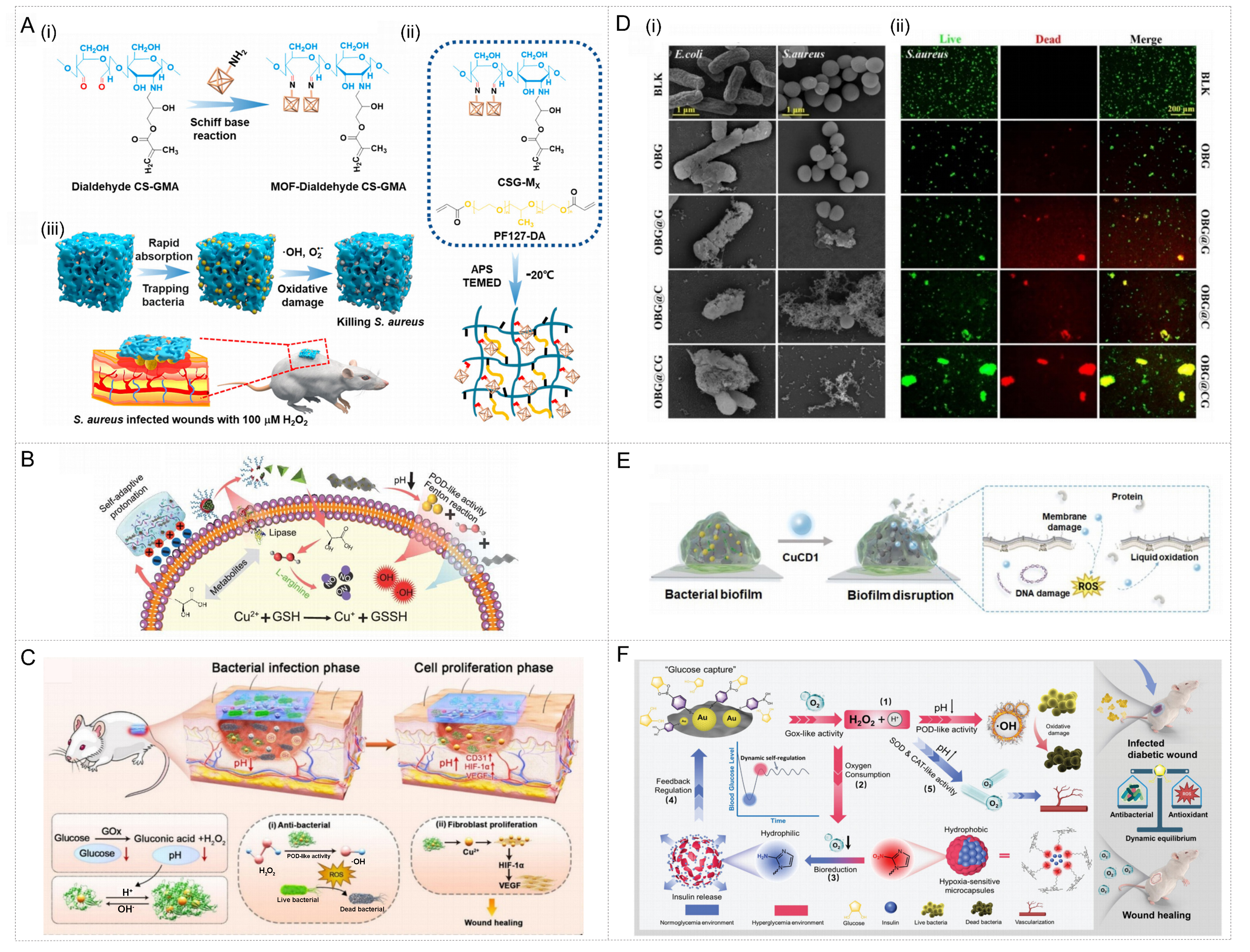
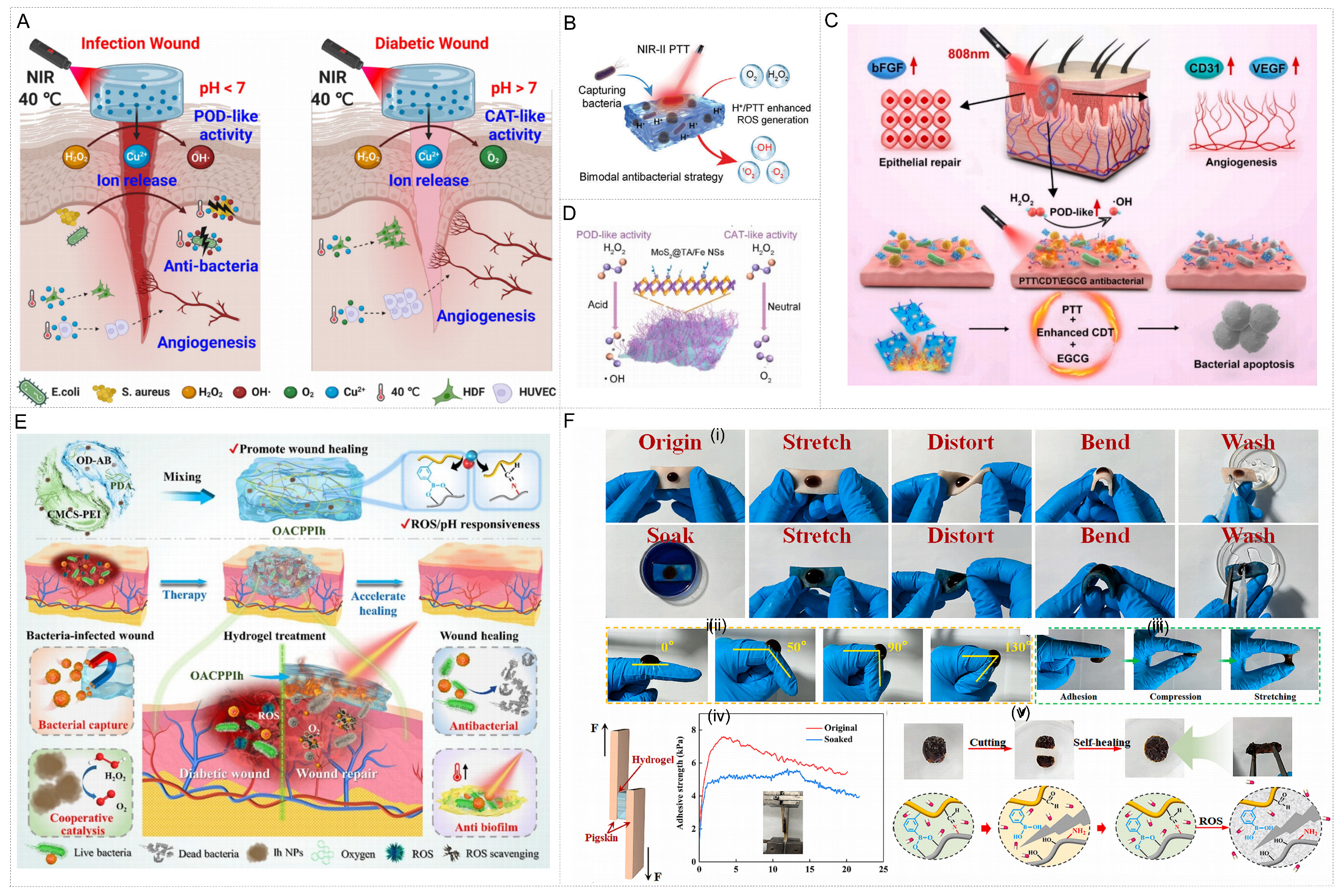
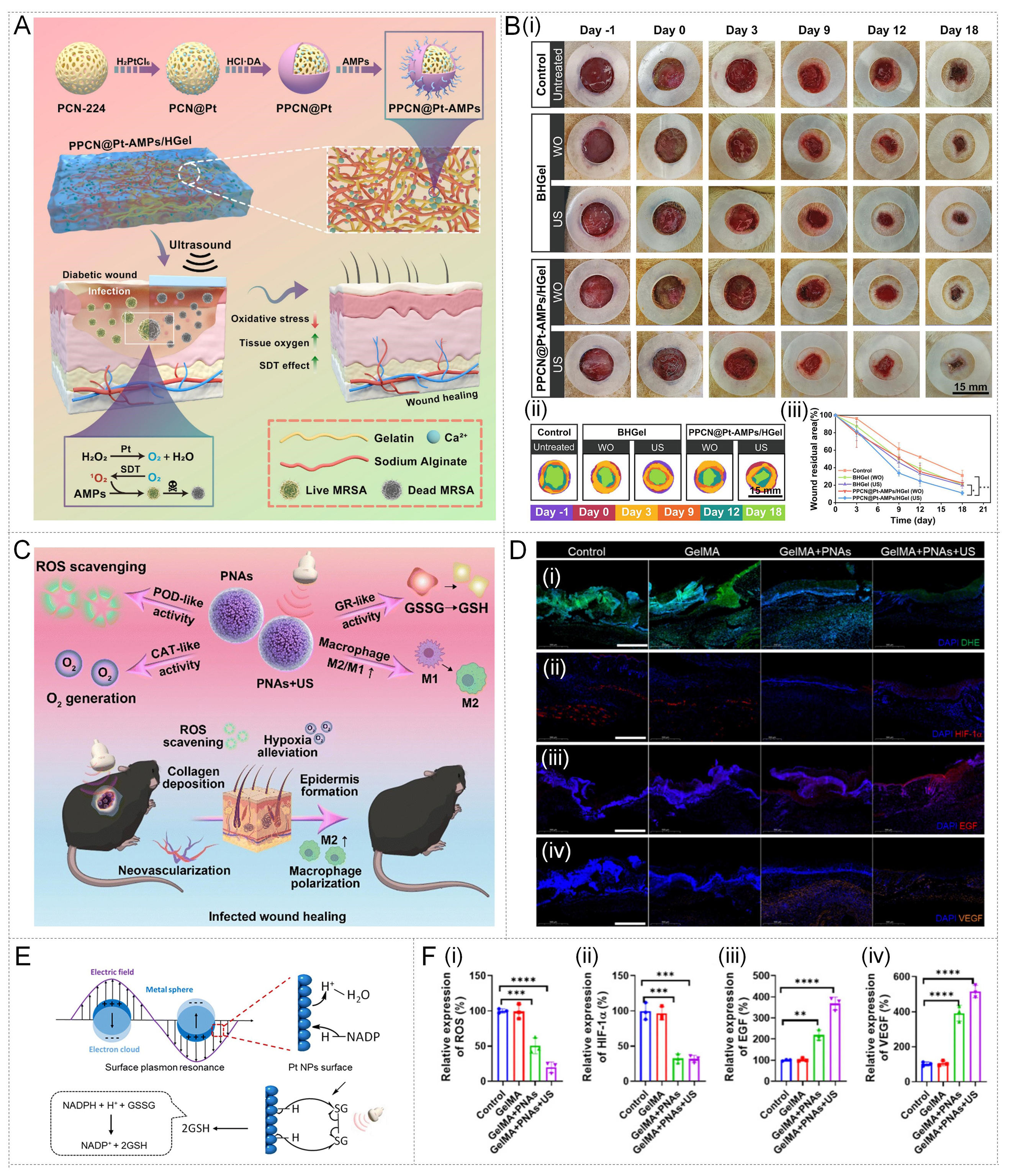












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].