Arginine metabolism disruption mediates 6-PPD quinone-induced mitochondrial toxicity at environmentally relevant concentrations in Caenorhabditis elegans
Abstract
The 6-PPD quinone (6-PPDQ) is frequently detected in environment. However, the possible effect of 6-PPDQ on amino acid metabolism and corresponding mechanisms remain unclear. In Caenorhabditis elegans, we examined effect of 6-PPDQ exposure on the absorption and catabolism of arginine. In nematodes, 6-PPDQ exposure reduced arginine content, and decreased expression of amino acid transporter 1 (aat-1) and C50D2.2 encoding intestinal transporters. Intestinal RNA interference (RNAi) of aat-1 and C50D2.2 reduced arginine content. Additionally, 6-PPDQ increased the expression of slc-25A29, which governs arginine import into the mitochondria, and argn-1, which governs mitochondrial arginine catabolism. Arginine content was increased by slc-25A29 and argn-1 RNAi. 6-PPDQ-induced mitochondrial dysfunction was strengthened by aat-1 and C50D2.2 RNAi and suppressed by slc-25A29 and argn-1 RNAi. The expression of slc-25A29 and argn-1 was further increased by RNAi of aat-1 and C50D2.2, and in the mitochondria, gas-1, mev-1, sod-3, and hsp-6 were identified as targets of argn-1 for controlling 6-PPDQ toxicity. Therefore, exposure risk of 6-PPDQ in disrupting arginine absorption and catabolism was suggested, which was associated with 6-PPDQ-induced mitochondrial dysfunction.
Keywords
INTRODUCTION
As an antioxidant, N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine (6-PPD) is supplied to tires to reduce oxidation[1]. The compound can be converted into 6-PPD quinone (6-PPDQ)[2]. In various environments, including rivers, dust, and soil, it has been found at a high detection rate[3-7]. The 6-PPDQ in aquatic environment has been detected ranging in ng/L or tens of μg/L[8-13]. Initially, this pollutant received attention for its acute toxicity in inducing fish lethality[13], and this has also been observed in other organisms, such as Sciaenops ocellatus[14-17]. After chronic exposure, 6-PPDQ toxicity in various aspects has also been observed in organisms[18-20]. In mice, 6-PPDQ accumulated in several organs and further caused damage to these organs[21-24]. 6-PPDQ detection in human samples implied potential health risk following environmental exposure [25-27]. In addition, 6-PPDQ exposure has also affected biochemical metabolism, such as energy metabolism[28,29]. Additionally, exposure to 6-PPDQ caused mitochondrial dysfunction[30].
Increasing evidence has indicated the value of Caenorhabditis elegans (C. elegans) in assessing pollutant toxicity[31-35]. It is considered that C. elegans is sensitive to assess pollutant toxicity[36,37]. After 6-PPDQ exposure, some aspects of toxicity were observed[38-40]. In cells, 6-PPDQ exposure caused damage to mitochondria, inducing mitochondrial dysfunction by affecting mitochondrial complexes[41,42] and suppressing mitochondrial unfolded protein response (UPR) and mitophagy[43,44]. Considering the important value of C. elegans in elucidating the molecular basis of metabolism[45], metabolic alterations were further examined, and 6-PPDQ disrupted the metabolism of the citric acid cycle, amino acids, and vitamin D3[46-48]. Certain altered metabolic processes (such as lipid accumulation) by 6-PPDQ could even be found in the offspring[49].
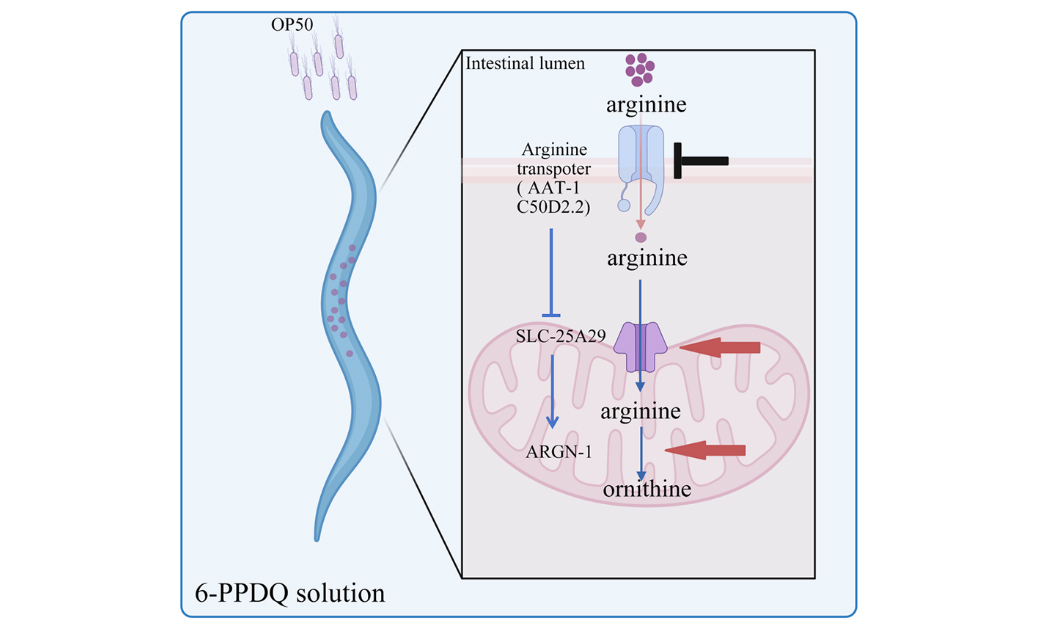
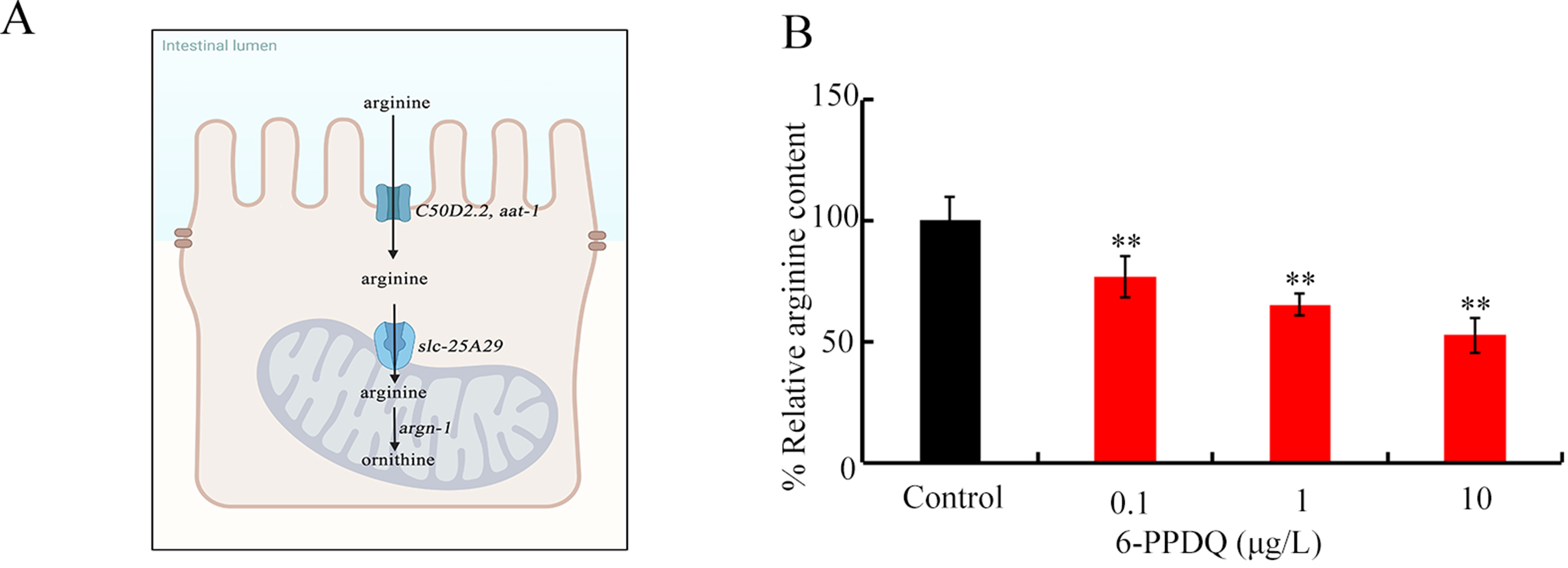
As an essential amino acid, arginine modulates some physiological events[50]. Supplementation with arginine has been used to suppress high blood pressure and viral infections[51,52], suggesting its beneficial effect. However, the possible toxicity of 6-PPDQ on the absorption and catabolism of arginine and the underlying mechanism remain unclear. We thus assumed that the absorption and catabolism of arginine might be disrupted by 6-PPDQ exposure. In humans, y+L amino acid transporter (y+LAT) and cationic amino acid transporter 1 (CAT1) are solute carriers (SLCs) that facilitate transmembrane transport of arginine[53,54], and AAT-1 (Amino Acid Transporter 1) and C50D2.2 are their homologs expressed in the C. elegans intestine (Figure 1A; see https://wormbase.org). After intestinal absorption, arginine is transported into the mitochondrial matrix via the mitochondrial membrane transporter SLC-25A29 (Solute Carrier Family 25 Member 29), and arginine catabolism is catalyzed by arginase ARGN-1 to generate ornithine [Figure 1A][55]. We first examined possible effect of 6-PPDQ on the disruption of the absorption and catabolism of arginine in nematodes. Moreover, considering that arginine treatment has been used for mitochondrial therapy[56,57], we further examined the association between disruption of the absorption and catabolism of arginine and damage to mitochondrial function caused by 6-PPDQ. A risk of 6-PPDQ exposure in decreasing arginine content by disrupting its absorption and catabolism was suggested, which in turn causes mitochondrial dysfunction in organisms.
Figure 1. Exposure to 6-PPDQ reduces arginine content. (A) Diagram showing arginine absorption and catabolism in nematodes; (B) Effect of 6-PPDQ on arginine content. **P < 0.01 vs. control. 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; slc-25a29: solute carrier family 25 member 29.
EXPERIMENTAL
Maintenance of C. elegans
Strains used are wild-type strain and two transgenic strains: CF1553 and SJ4100 [Supplementary Table 1]. Nematodes were cultured as described[58]. Gravid hermaphrodites were treated with lysis buffer[59], and collected embryos were cultured overnight to prepare L1-larval population.
Exposure
Exposure doses of 6-PPDQ (Toronto Research Chemicals; CAS no. 2795-39-3; purity > 98%) were selected at 0.1-10 μg/L, which correspond to those in actual aquatic environments[2,13]. L1-larvae were exposed in 6-PPDQ for 6.5 days[60]. Exposure solutions added with E. coli OP50 as the food were refreshed daily.
Arginine content
This was assessed using assay kit (Wuhan Mosher Biotechnology Co., Ltd.) utilizing an enzyme-linked immunosorbent competitive method. For sample preparation, 20,000 nematodes per group were collected after 6-PPDQ exposure and homogenized. Microplate wells were coated with a purified anti-arginine monoclonal antibody. Arginine and a horseradish peroxidase (HRP)-conjugated arginine antigen were added. After washing, the 3,3A,5,5A-tetramethylbenzidine (TMB) substrate was added, and supernatant was measured at 450 nm. Experiments were conducted in triplicate.
Mitochondrial function
Oxygen consumption and adenosine triphosphate (ATP) content were measured as described[41]. Totally 20,000 animals were collected. Nematodes were homogenized on using pre-cooled lysis buffer, and mitochondria were isolated. Fluorescence was measured every 5 min over a 30-min period at 468 nm for assessing oxygen consumption. ATP levels were measured at 340 nm. Experiments were conducted in triplicate.
Toxicity assessment
For reactive oxygen species (ROS) assay, animals were treated by 1 mM CM-H2DCFDA [5-(and-6)-chloromethyl-2’,7’-dichlorodihydrofluorescein diacetate] for 2.5 h in the dark[60]. After that, ROS fluorescence signals were analyzed under excitation/emission wavelengths of 488/510 nm using an Olympus fluorescence microscope (model BX53). Fifty C. elegans were tested.
Body bending and head thrashing reflect locomotion. Body bending and head thrashing in 50 nematodes per treatment were examined as described[61]. Fifty C. elegans were tested.
For brood size assay, nematodes were transferred individually to fresh NGM plates daily, and total number of offspring was counted[62]. Thirty nematodes were tested.
Transcriptional expression
After exposure to 6-PPDQ, approximately 5,000 nematodes were lysed by TRIzol. Chloroform was added, followed by vigorous shaking and centrifugation. RNA in the supernatant was precipitated using isopropanol. For complementary DNA (cDNA) synthesis, RNA from each group was reverse-transcribed using M-MuLV reverse transcriptase. Expression of genes was assessed by quantitative real-time polymerase chain reaction (qRT-PCR), with tba-1 (tubulin) as reference gene[63]. Primers are listed in Supplementary Table 2.
RNA interference
L1 larvae were seeded with double-stranded RNA-expressing E. coli HT115[64]. VP303 was used for intestinal gene RNA interference (RNAi). The empty vector L4440 served as the control[64]. RNAi efficiency is given in Supplementary Figures 1 and 2.
Pharmacological treatment
Arginine (Sangon Biotech Co., Ltd.) was added to NGM agar during plate preparation to achieve a final concentration of 10 mM. Following 6-PPDQ exposure (10 μg/L), nematodes were treated by 10 mM arginine for 24 h[65].
Data analysis
Statistical analysis was performed by SPSS Statistics 25.0. One-way or two-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test was determined for significant difference between groups. Two-way ANOVA was applied for multi-factor comparisons. Statistical significance threshold was set at P < 0.01.
RESULTS AND DISCUSSION
6-PPDQ reduced arginine content
The 6-PPDQ reduced arginine content [Figure 1B]. After exposure to 0.1-10 μg/L 6-PPDQ, arginine content decreased by 23.6%, 37.5%, and 48.2%, respectively [Figure 1B].
6-PPDQ disrupted intestinal absorption of arginine
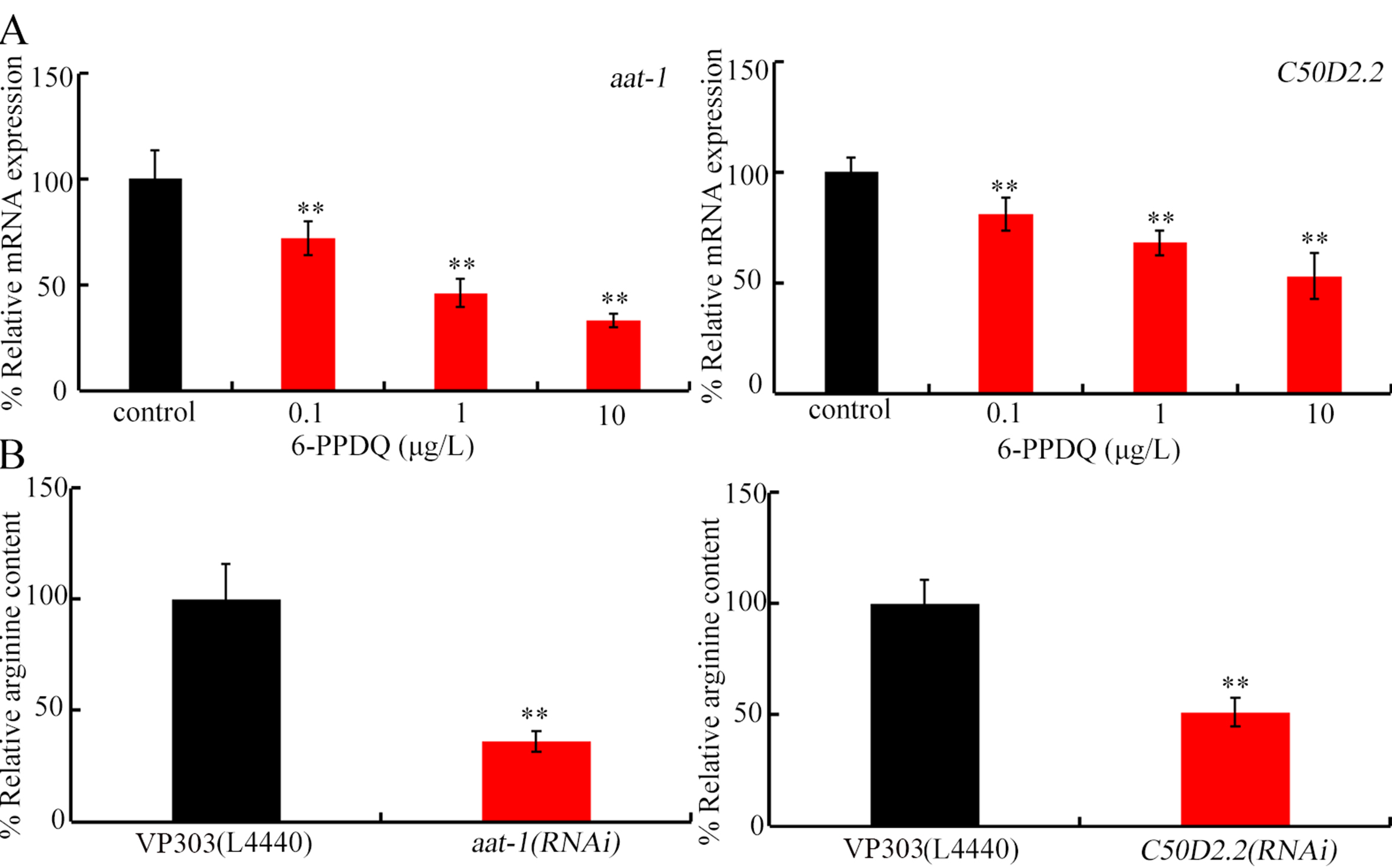
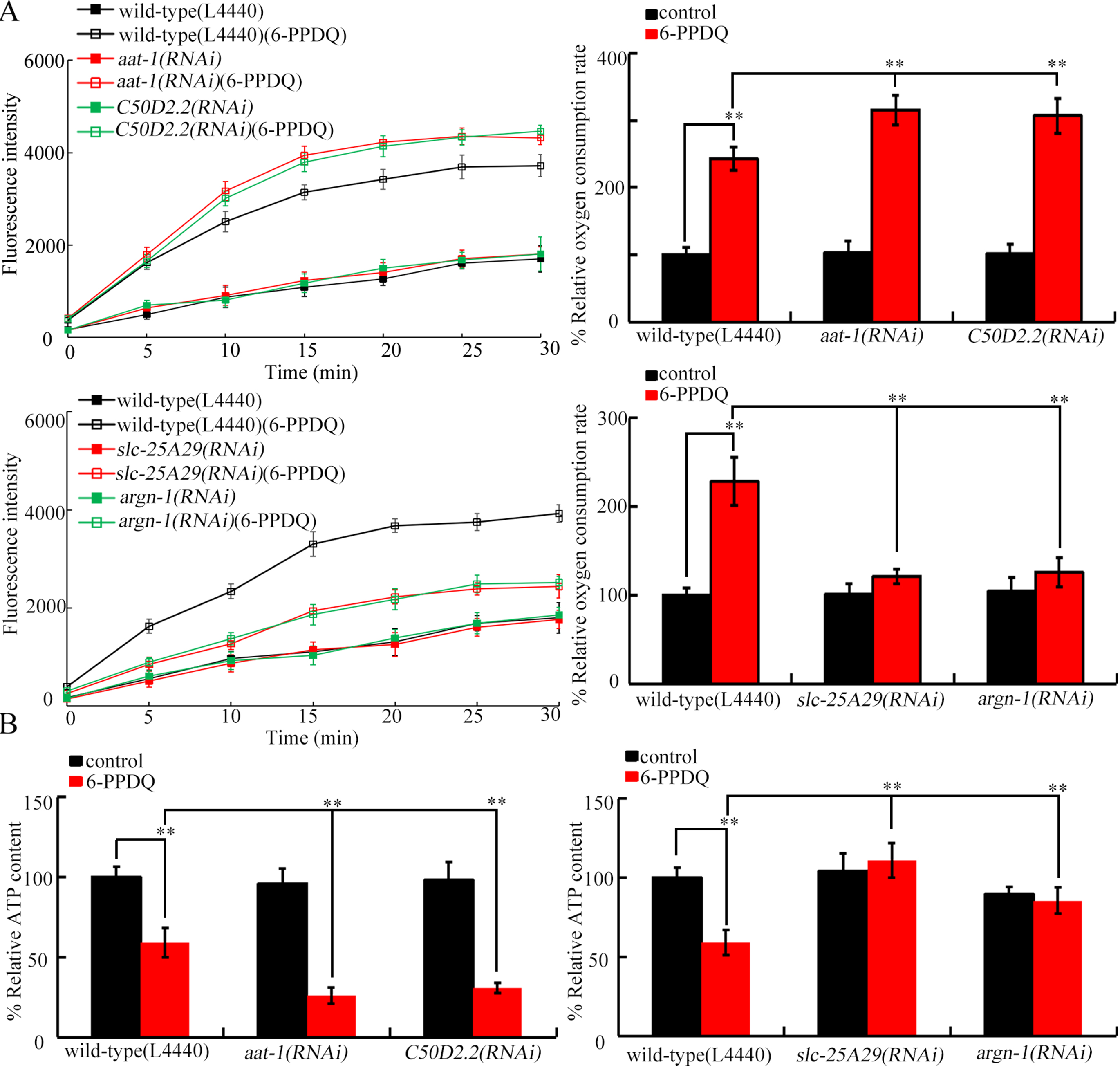
Among candidate intestinal transporter genes involved in arginine absorption, intestinal amino acid transporter 1 (aat-1) and C50D2.2 expressions were decreased by 6-PPDQ [Figure 2A]. After exposure to 0.1-10 μg/L 6-PPDQ, aat-1 expression decreased by 28.3%, 53.7%, and 66.2%, respectively, and C50D2.2 expression decreased by 18.3%, 31.6%, and 47.1%, respectively [Figure 2A]. Moreover, arginine content was further reduced by intestinal RNAi of aat-1 and C50D2.2 [Figure 2B].
Figure 2. Effect of 6-PPDQ exposure on arginine absorption. (A) Effect of 6-PPDQ exposure on expression of intestinal aat-1 and C50D2.2. Thirty intact intestines were isolated for qRT-PCR analysis. **P < 0.01 vs. control. (B) Effect of intestinal RNAi of aat-1 and C50D2.2 on arginine content in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01 vs. VP303(L44440). 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; aat-1: amino acid transporter 1; qRT-PCR: quantitative real-time polymerase chain reaction.
6-PPDQ accelerated arginine catabolism
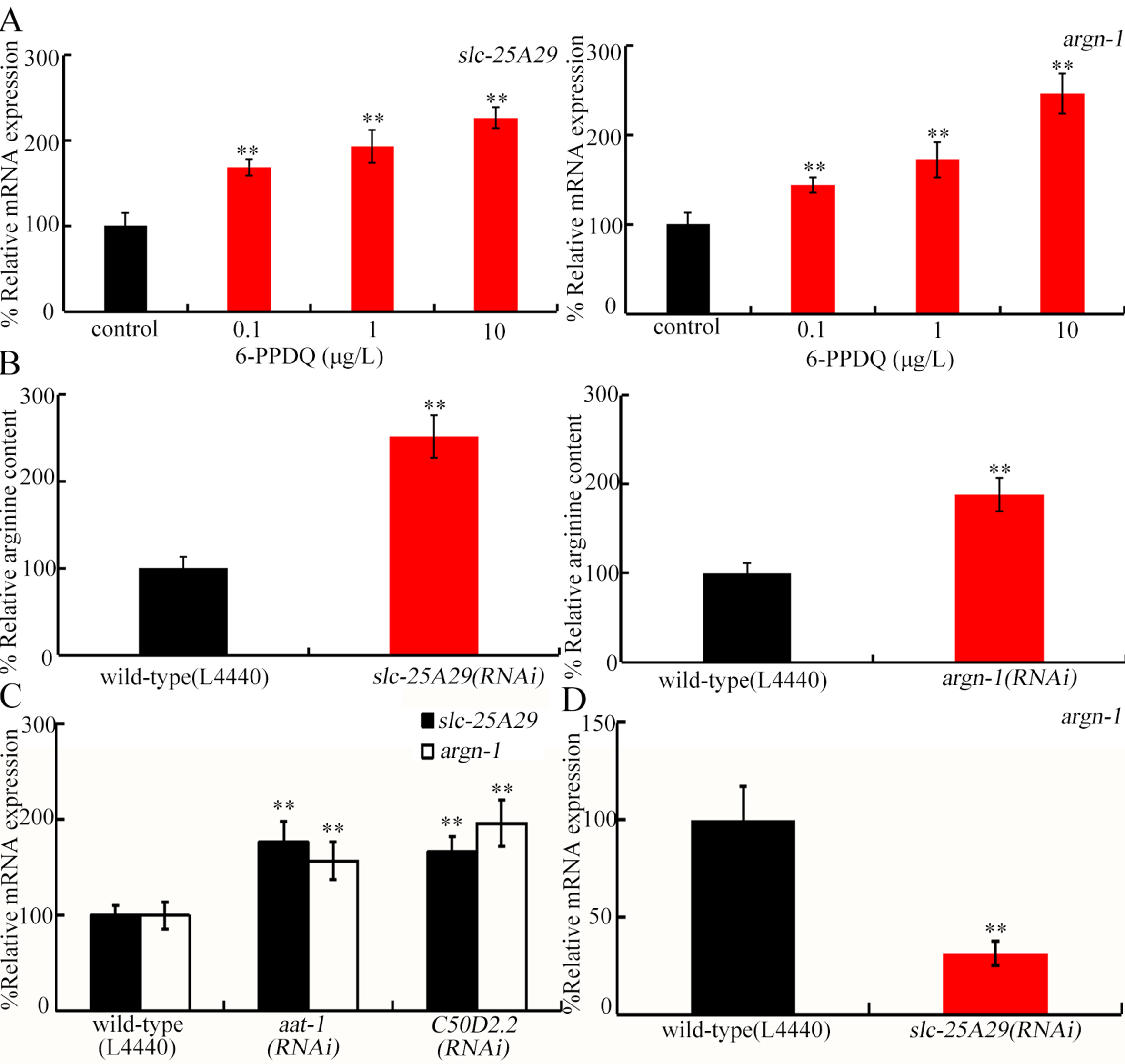
C. elegans slc-25A29 is required for the import of arginine into mitochondria[55], and argn-1 governs arginine catabolism in mitochondria[55]. Expressions of both slc-25A29 and argn-1 were increased by 6-PPDQ [Figure 3A]. After exposure to 0.1-10 μg/L 6-PPDQ, slc-25A29 expression increased by 69.3%, 93.6% and 127.1%, respectively, and argn-1 expression increased by 44.6%, 72.3%, and 146.6%, respectively [Figure 3A]. Additionally, arginine content was increased by slc-25A29 and argn-1 RNAi [Figure 3B]. Expression of these two genes was further increased by aat-1 and C50D2.2 RNAi [Figure 3C], and argn-1 expression was further reduced by slc-25A29 RNAi [Figure 3D].
Figure 3. Effect of 6-PPDQ exposure on arginine catabolism. (A) Effect of 6-PPDQ exposure on expression of slc-25A29 and argn-1. **P < 0.01 vs. control; (B) Effect of RNAi of slc-25A29 and argn-1 on arginine content in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01 vs. wild-type(L44440); (C) Effect of RNAi of aat-1 and C50D2.2 on expression of slc-25A29 and argn-1 in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01 vs. wild-type(L44440). (D) Effect of RNAi of slc-25A29 on expression of argn-1 in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01 vs. wild-type(L44440). 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; aat-1: amino acid transporter 1; RNAi: RNA interference; slc-25a29: solute carrier family 25 member 29.
RNAi of aat-1, C50D2.2, slc-25A29, and argn-1 influenced 6-PPDQ-induced mitochondrial dysfunction
In nematodes, 6-PPDQ caused mitochondrial dysfunction[41]. Considering that arginine catabolism occurs in mitochondria, we investigated the effects of related genes RNAi on 6-PPDQ-induced mitochondrial dysfunction. Elevation in oxygen consumption and reduction in ATP content by 6-PPDQ were enhanced by aat-1 and C50D2.2 RNAi, and attenuated by scl-25A29 and argn-1 RNAi [Figure 4A and B].
Figure 4. Effect of RNAi of aat-1, C50D2.2, slc-25A29, and argn-1 on oxygen consumption rate (A) and ATP content (B) in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01. 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; aat-1: amino acid transporter 1; RNAi: RNA interference; slc-25a29: solute carrier family 25 member 29.
RNAi of aat-1, C50D2.2, slc-25A29, and argn-1 affected 6-PPDQ-induced other aspects of toxicity
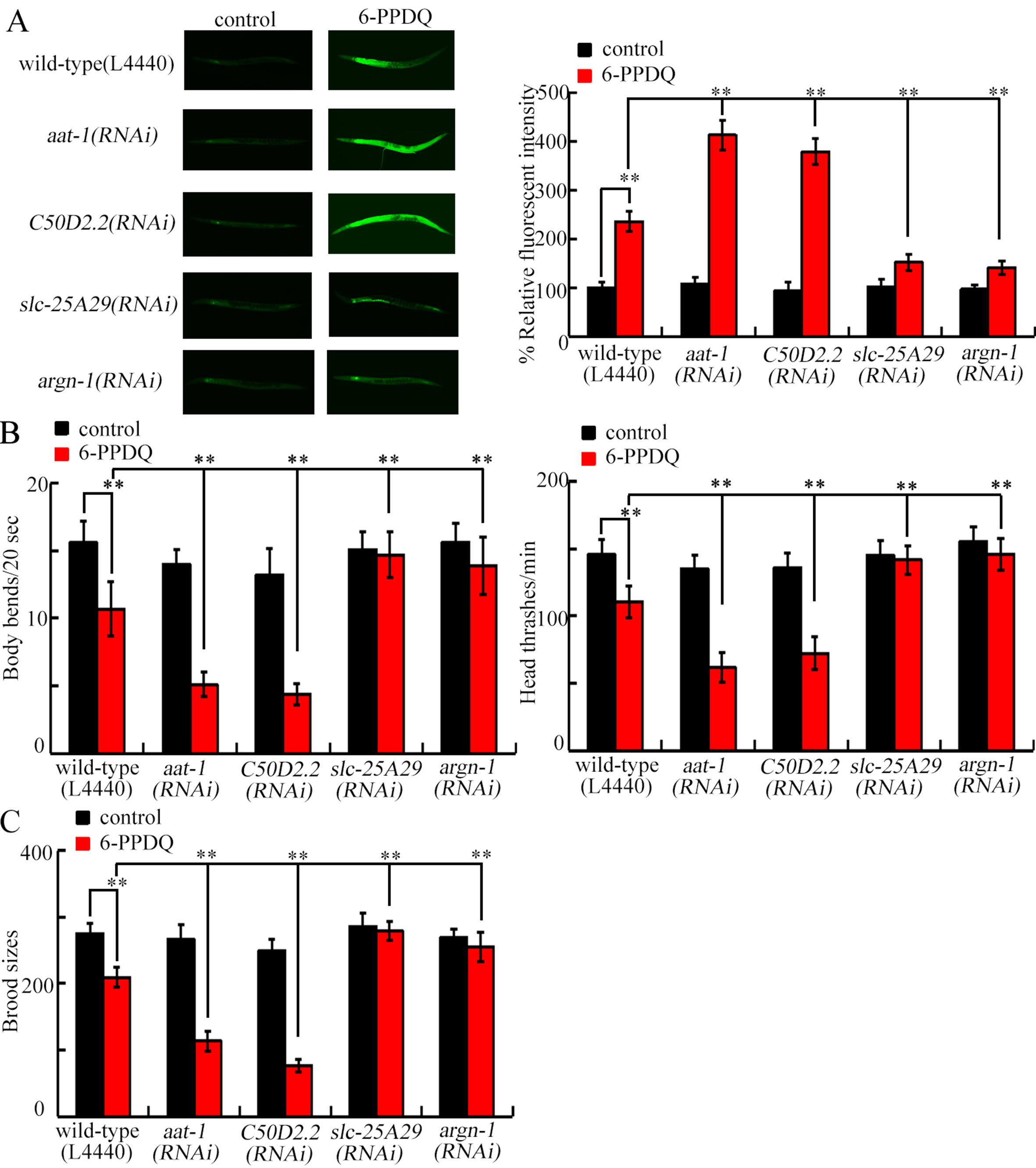
Induction of ROS indicates intestinal 6-PPDQ toxicity[60]. ROS generation induced by 6-PPDQ was enhanced by aat-1 and C50D2.2 RNAi and suppressed by slc-25A29 and argn-1 RNAi [Figure 5A]. Inhibited locomotion reflects 6-PPDQ neurotoxicity[66]. Locomotion inhibition induced by 6-PPDQ was enhanced by aat-1 and C50D2.2 RNAi and attenuated by slc-25A29 and argn-1 RNAi [Figure 5B]. Reduced brood size indicates 6-PPDQ reproductive toxicity[67]. Reduction in brood size by 6-PPDQ was enhanced by aat-1 and C50D2.2 RNAi and mitigated by slc-25A29 and argn-1 RNAi [Figure 5C].
Figure 5. Effect of RNAi of aat-1, C50D2.2, slc-25A29, and argn-1 on 6-PPDQ toxicity in causing intestinal ROS generation (A), decreasing locomotion (B), and reducing brood size (C). Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01. 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; aat-1: amino acid transporter 1; RNAi: RNA interference; slc-25a29: solute carrier family 25 member 29; ROS: reactive oxygen species.
Identification of targets of mitochondrial argn-1
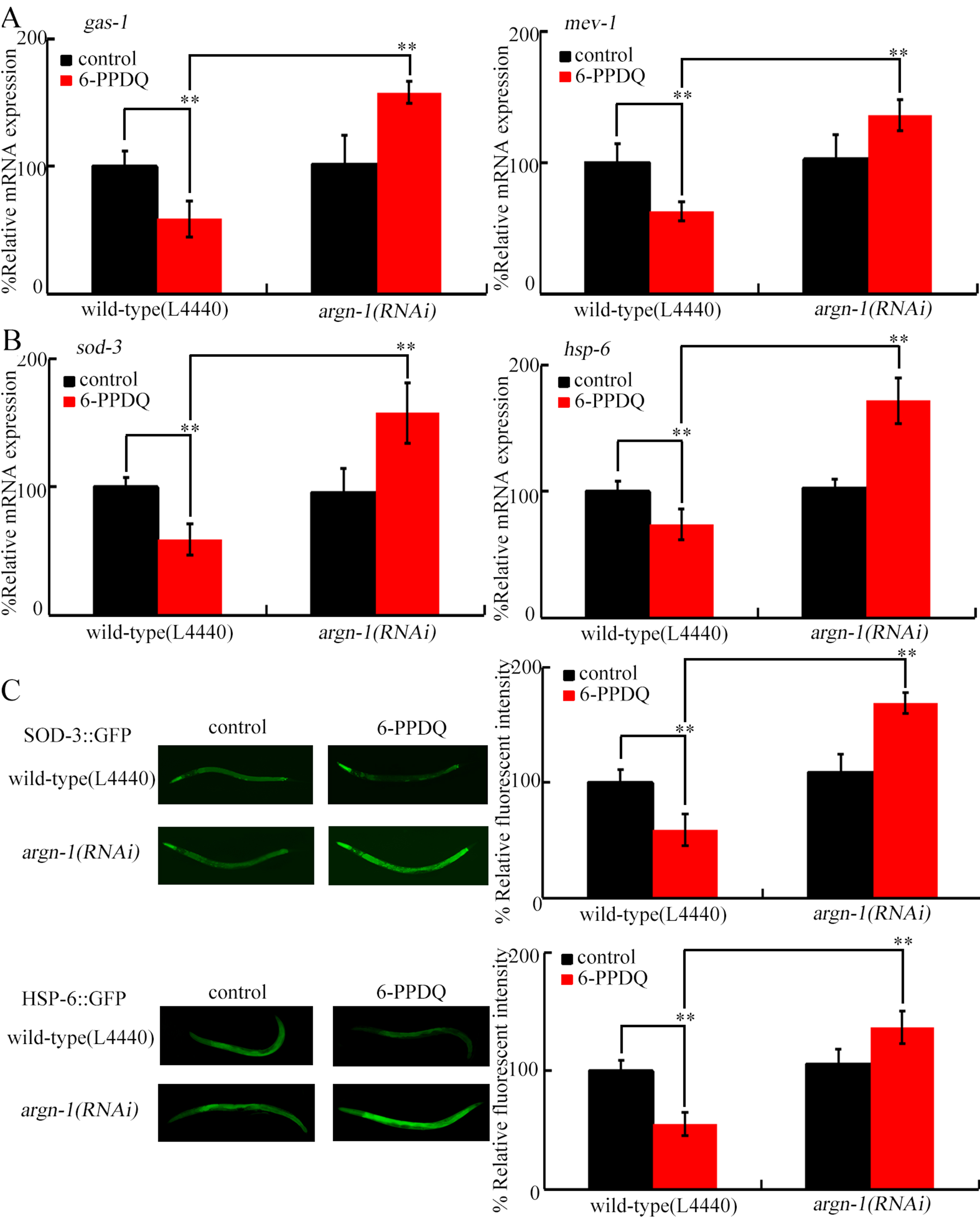
Recently, we identified that gas-1, encoding a complex I component, and mev-1, encoding a complex II component, are required for controlling 6-PPDQ-caused mitochondrial dysfunction[68]. The decrease in gas-1 and mev-1 expressions by 6-PPDQ was suppressed by argn-1 RNAi [Figure 6A].
Figure 6. Effect of RNAi of argn-1 on expressions of gas-1, mev-1, sod-3, and hsp-6 in 6-PPDQ-exposed nematodes. (A) Effect of RNAi of argn-1 on expressions of gas-1 and mev-1 in 6-PPDQ-exposed nematodes; (B) Effect of RNAi of argn-1 on expressions of sod-3 and hsp-6 in 6-PPDQ-exposed nematodes; (C) Effect of RNAi of argn-1 on expressions of SOD-3::GFP and HSP-6::GFP in 6-PPDQ-exposed nematodes. Exposure concentration of 6-PPDQ was 10 μg/L. **P < 0.01. gas-1: General Anaesthetic Sensitivity abnormal; mev-1: abnormal MEthyl Viologen sensitivity; sod-3: Superoxide Dismutase 3; hsp-6: Heat Shock Protein 6; 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; RNAi: RNA interference; GFP: green fluorescent protein.
Suppression of SOD-3/Mn-SOD and HSP-6 (Heat Shock Protein 6, a mitochondrial UPR marker)[69] mediated mitochondrial damage by 6-PPDQ[43,70]. Decreases in expressions of sod-3, hsp-6, SOD-3::GFP, and HSP-6::GFP by 6-PPDQ were suppressed by argn-1 RNAi [Figure 6B and C].
Benefit of arginine administration against 6-PPDQ toxicity on mitochondrial function
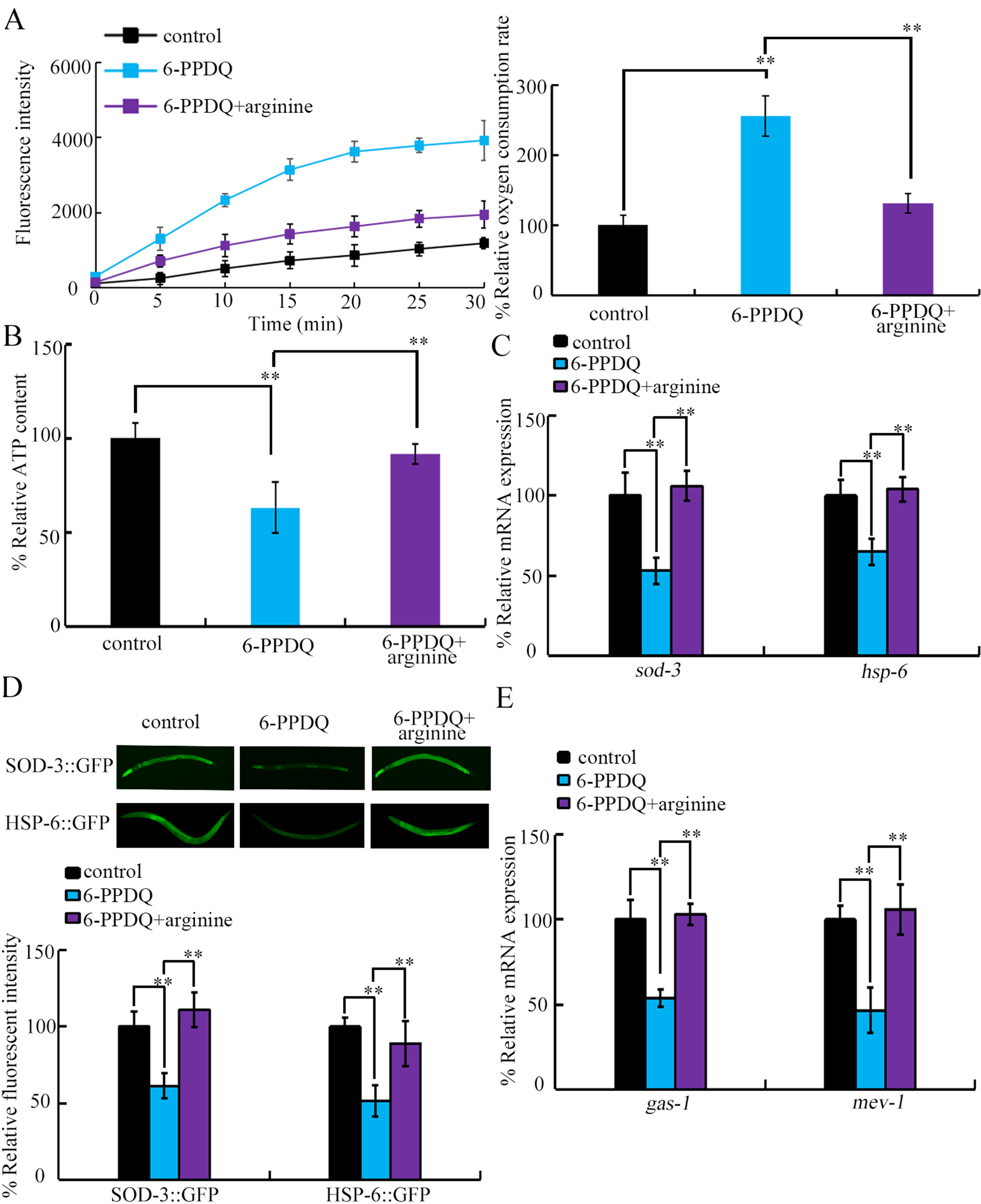
First, 6-PPDQ-caused mitochondrial dysfunction was suppressed by arginine administration [Figure 7A and B]. Second, 6-PPDQ-caused suppression of mitochondrial Mn-SOD and the mitochondrial UPR was alleviated by arginine treatment [Figure 7C and D]. In nematodes, 6-PPDQ induces mitochondrial dysfunction by inhibiting gas-1 and mev-1[68]. Decreases in gas-1 and mev-1 expression by 6-PPDQ were suppressed by arginine administration [Figure 7E].
Figure 7. Effect of administration with 10 mM arginine on toxicity of 10 μg/L 6-PPDQ on mitochondrial function. (A) Effect of administration with arginine on 6-PPDQ toxicity in increasing oxygen consumption rate; (B) Effect of administration with arginine on 6-PPDQ toxicity in decreasing ATP content; (C) Effect of administration with arginine on 6-PPDQ toxicity in decreasing expression of sod-3 and hsp-6; (D) Effect of administration with arginine on 6-PPDQ toxicity in decreasing expression of SOD-3::GFP and HSP-6::GFP; (E) Effect of administration with arginine on 6-PPDQ toxicity in decreasing expression of gas-1 and mev-1. **P < 0.01. 6-PPDQ: N-(1,3-dimethylbutyl)-N′-phenyl-p-phenylenediamine quinone; SOD-3: Superoxide Dismutase 3; GFP: green fluorescent protein; HSP-6: Heat Shock Protein 6; gas-1: General Anaesthetic Sensitivity abnormal; mev-1: abnormal MEthyl Viologen sensitivity.
Benefit of arginine administration against other aspects of 6-PPDQ toxicity
ROS generation caused by 6-PPDQ was inhibited by 10 mM arginine [Supplementary Figure 3A]. Damage to locomotion and brood size caused by 6-PPDQ was also alleviated by arginine [Supplementary Figure 3B and C].
In nematodes, 6-PPDQ disrupted multiple metabolic pathways[46-49]. Arginine content was decreased by 6-PPDQ [Figure 1B], indicating that 6-PPDQ can significantly affect arginine levels. One major reason for this reduction was inhibition of intestinal arginine absorption. Expression of intestinal aat-1 and C50D2.2 was decreased by 6-PPDQ [Figure 2A]. Furthermore, RNAi of aat-1 and C50D2.2 RNAi reduced arginine content [Figure 2B], demonstrating that AAT-1 and C50D2.2 acted in the intestine to control arginine absorption. Thus, suppression of aat-1 and C50D2.2 contributes to 6-PPDQ-induced reduction in arginine content. Role of AAT-1 in facilitating amino acid absorption has been confirmed in Xenopus oocytes[71]. In addition, our study highlights the important function of C50D2.2 in regulating intestinal arginine absorption.
Another important reason for the reduced arginine content following 6-PPDQ exposure was accelerated arginine catabolism. scl-25A29 and argn-1 expressions were increased by 6-PPDQ [Figure 3A]. Furthermore, arginine content was increased by slc-25A29 and argn-1 RNAi [Figure 3B]. SLC-25A29 is a mitochondrial amino acid transporter, and ARGN-1 is localized in mitochondria[55]. Mutation of argn-1 resulted in increased arginine content under normal conditions[55]. These results suggest that upregulation of slc-25A29 and argn-1 contributed to reduction in arginine content by enhancing mitochondrial arginine catabolism.
For the role of genes governing arginine absorption and catabolism, we found that they have opposite functions. RNAi of slc-25A29 and argn-1 inhibited 6-PPDQ-induced mitochondrial dysfunction [Figure 4], indicating that upregulation of slc-25A29 and argn-1 in mitochondria potentially mediated 6-PPDQ toxicity. In contrast, aat-1 and C50D2.2 RNAi enhanced 6-PPDQ-induced mitochondrial dysfunction [Figure 4]. Considering that aat-1 and C50D2.2 RNAi could increase slc-25A29 and argn-1 expressions [Figure 3C], suppression of aat-1 and C50D2.2 could contribute to mitochondrial dysfunction by promoting upregulation of slc-25A29 and argn-1. Furthermore, 6-PPDQ-induced intestinal, neuronal, and reproductive toxicities were enhanced by aat-1 and C50D2.2 RNAi and suppressed by slc-25A29 and argn-1 RNAi [Figure 5], suggesting that decreased expression of aat-1 and C50D2.2, together with increased expression of slc-25A29 and argn-1, was possibly associated with multiple aspects of 6-PPDQ toxicity. Additionally, argn-1 expression was decreased by slc-25A29 RNAi [Figure 3D], indicating that 6-PPDQ exposure could induce damage via the aat-1/C50D2.2-slc-25A29-argn-1 signaling axis.
For the molecular basis of mitochondrial ARGN-1 function, we propose that ARGN-1 regulates 6-PPDQ toxicity in mitochondria by modulating the expression and activity of GAS-1, MEV-1, SOD-3, and HSP-6 [Figure 6A]. GAS-1 and MEV-1 are essential for maintaining mitochondrial function[72,73]. Decreases in gas-1 and mev-1 expressions were linked to 6-PPDQ-induced mitochondrial dysfunction[68]. Additionally, sod-3 and hsp-6 expressions were increased by argn-1 RNAi [Figure 6B and C]. SOD-3 protects nematodes from oxidative damage[74], with its activation providing defense against 6-PPDQ toxicity[70]. HSP-6 is mitochondrial UPR marker in response to stress[75] or pollutants[69], and its activation protects nematodes from 6-PPDQ-induced mitochondrial dysfunction, whereas decreased hsp-6 mediated mitochondrial damage[43]. Therefore, in mitochondria, ARGN-1 regulated 6-PPDQ toxicity by modulating both mitochondrial complex components and SOD-3/HSP-6-mediated protective responses.
The protective effect of arginine administration against 6-PPDQ toxicity was further confirmed. Mitochondrial dysfunction induced by 6-PPDQ was decreased by arginine treatment [Figure 7A and B]. Arginine supplementation has been used as a mitochondrial therapy in certain diseases, such as cardiomyopathy[55,76]. Additionally, other 6-PPDQ-induced toxicities were also suppressed by arginine administration [Supplementary Figure 3]. These observations further support the association between disrupted arginine absorption and catabolism and multiple aspects of 6-PPDQ toxicity. At the molecular level, arginine administration inhibited 6-PPDQ toxicity by activating gas-1 and mev-1 [Figure 7E] and by increasing SOD-3 and HSP-6 expressions [Figure 7C and D]. Inhibition in gas-1 and mev-1 was linked to toxicity from di(2-ethylhexyl) phthalate and 2,4,6-trinitrotoluene[77,78]. Therefore, the beneficial effect of arginine administration is not limited to 6-PPDQ, and it may also help mitigate the toxicity of other pollutants.
We further propose that inhibition of arginine absorption and enhancement of arginine catabolism contribute to reduction in arginine content by 6-PPDQ. Nevertheless, as an essential amino acid, arginine metabolism in E. coli OP50 may also influence arginine levels in the host C. elegans, which requires further investigation. In addition, the proposed molecular mechanisms underlying reduction in arginine content by 6-PPDQ and the observed protective effect of arginine against its toxicity still need to be validated in mammalian models.
CONCLUSIONS
Overall, arginine content was reduced by 0.1-10 μg/L 6-PPDQ. This reduction resulted from both inhibition of arginine absorption and acceleration of arginine catabolism. Following 6-PPDQ exposure, alterations in expression of genes governing arginine absorption (aat-1 and C50D2.2) and mitochondrial arginine catabolism (slc-25A29 and argn-1) were associated with induction of mitochondrial dysfunction. gas-1, mev-1, sod-3, and hsp-6 were identified as targets of argn-1 in mediating 6-PPDQ toxicity. Pharmacological analysis further indicated that arginine administration could serve as intervention strategy against 6-PPDQ toxicity. These findings highlight the potential risk of 6-PPDQ in disrupting arginine absorption and catabolism, which in turn contributes to mitochondrial dysfunction.
DECLARATIONS
Authors’ contributions
Investigation and data analysis: Wang, Y.
Experimental design and supervision: Wang, D.
All authors have read and approved the final manuscript.
Availability of data and materials
The data generated or analyzed during the current study are included in the Supporting Information.
Financial support and sponsorship
This study was supported by the Postgraduate Research & Practice Innovation Program of Jiangsu Province (Grant No. KYCX25_0537).
Conflicts of interest
Wang, D. is the Guest Editor of the Special Topic “Environmental Exposure Modelling and Risk Analysis” in the Journal of Environmental Exposure Assessment. He had no involvement in the review or editorial process of this manuscript, including reviewer selection, evaluation, or the final decision. The other author has declared no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Li, X.; Chen, X.; Chen, B.; Zhang, W.; Zhu, Z.; Zhang, B. Tire additives: evaluation of joint toxicity, design of new derivatives and mechanism analysis of free radical oxidation. J. Hazard. Mater. 2024, 465, 133220.
2. Hua, X.; Wang, D. Tire-rubber related pollutant 6-PPD quinone: A review of its transformation, environmental distribution, bioavailability, and toxicity. J. Hazard. Mater. 2023, 459, 132265.
3. Kazmi, S. S. U. H.; Xu, Q.; Tayyab, M.; et al. Navigating the environmental dynamics, toxicity to aquatic organisms and human associated risks of an emerging tire wear contaminant 6PPD-quinone. Environ. Pollut. 2024, 356, 124313.
4. Li, Y.; Zeng, J.; Liang, Y.; et al. A review of N-(1,3-dimethylbutyl)-N'-phenyl-p-phenylenediamine (6PPD) and its derivative 6PPD-quinone in the environment. Toxics 2024, 12, 394.
5. Chen, X.; He, T.; Yang, X.; et al. Analysis, environmental occurrence, fate and potential toxicity of tire wear compounds 6PPD and 6PPD-quinone. J. Hazard. Mater. 2023, 452, 131245.
6. Yi, J.; Ruan, J.; Yu, H.; et al. Environmental fate, toxicity, and mitigation of 6PPD and 6PPD-Quinone: Current understanding and future directions. Environ. Pollut. 2025, 375, 126352.
7. Yin, T.; Liang, Y.; Liu, Y.; Liu, J.; Wang, X. Environmental occurrence, influencing factors, and toxic effects of 6PPD-Q. Toxics 2025, 13, 906.
8. Xu, F.; Su, M.; Tang, S. Spatiotemporal distribution of 6PPD-Q in China revealed by a national-scale quantification framework. Environ. Sci. Technol. 2026, 60, 1253-62.
9. Zhang, Y.; Yan, L.; Wang, L.; Zhang, H.; Chen, J.; Geng, N. A nation-wide study for the occurrence of PPD antioxidants and 6PPD-quinone in road dusts of China. Sci. Total. Environ. 2024, 922, 171393.
10. Zhang, H.; Li, L.; Jiang, J.; Zhou, Z.; Chen, Q.; Long, T. Developing water quality criteria and assessing ecological risks for 6PPD and 6PPD-Q in freshwater ecosystems. Environ. Pollut. 2025, 387, 127303.
11. Cao, G.; Wang, W.; Zhang, J.; et al. New evidence of rubber-derived quinones in water, air, and soil. Environ. Sci. Technol. 2022, 56, 4142-50.
12. Zhu, J.; Guo, R.; Ren, F.; Jiang, S.; Jin, H. Occurrence and partitioning of p-phenylenediamine antioxidants and their quinone derivatives in water and sediment. Sci. Total. Environ. 2024, 914, 170046.
13. Tian, Z.; Zhao, H.; Peter, K. T.; et al. A ubiquitous tire rubber-derived chemical induces acute mortality in coho salmon. Science 2021, 371, 185-9.
14. Ackerly, K. L.; Roark, K. J.; Lu, K.; Esbaugh, A. J.; Liu, Z.; Nielsen, K. M. Acute toxicity testing of 6PPD-quinone on the estuarine-dependent sport fish, Sciaenops ocellatus. Ecotoxicology 2024, 33, 582-9.
15. Jin, R.; Venier, M.; Chen, Q.; Yang, J.; Liu, M.; Wu, Y. Amino antioxidants: a review of their environmental behavior, human exposure, and aquatic toxicity. Chemosphere 2023, 317, 137913.
16. Roberts, C.; Kohlman, E.; Jain, N.; et al. Subchronic and acute toxicity of 6PPD-quinone to early life stage rainbow trout (oncorhynchus mykiss). Environ. Sci. Technol. 2025, 59, 6771-7.
17. Baker, J. A.; Cronshaw, I.; Monaghan, J.; Jaeger, A.; Bailey, H. C.; Krogh, E. T. Toxicity identification evaluation techniques isolate zinc and 6PPD-Q as causes of acute lethality to rainbow trout in road runoff. Environ. Toxicol. Chem. 2026, 45, 184-94.
18. Jiang, Y.; Wang, C.; Ma, L.; Gao, T.; Wāng, Y. Environmental profiles, hazard identification, and toxicological hallmarks of emerging tire rubber-related contaminants 6PPD and 6PPD-quinone. Environ. Int. 2024, 187, 108677.
19. Liang, Y.; Zhu, F.; Li, J.; et al. P-phenylenediamine antioxidants and their quinone derivatives: A review of their environmental occurrence, accessibility, potential toxicity, and human exposure. Sci. Total. Environ. 2024, 948, 174449.
20. Li, X.; Zhou, S.; Zhang, T.; et al. Occurrence and environmental fate/behaviors of tire wear particles and their human and ecological health: an emerging global issue. Arch. Toxicol. 2025, 99, 4353-66.
21. Zhang, J.; Cao, G.; Wang, W.; et al. Stable isotope-assisted mass spectrometry reveals in vivo distribution, metabolism, and excretion of tire rubber-derived 6PPD-quinone in mice. Sci. Total. Environ. 2024, 912, 169291.
22. Fang, L.; Fang, C.; Di, S.; et al. Oral exposure to tire rubber-derived contaminant 6PPD and 6PPD-quinone induce hepatotoxicity in mice. Sci. Total. Environ. 2023, 869, 161836.
23. Li, X.; Wu, C.; Yang, P.; et al. Environmental factors ultraviolet a and ozone exacerbate the repeated inhalation toxicity of 6PPD in mice via accelerating the aging reaction. J. Hazard. Mater. 2025, 486, 137000.
24. Wang, L.; Tang, W.; Sun, N.; et al. Low-dose tire wear chemical 6PPD-Q exposure elicit fatty liver via promoting fatty acid biosynthesis in ICR mice. J. Hazard. Mater. 2025, 489, 137574.
25. Shi, R.; Zhang, Z.; Zeb, A.; et al. Environmental occurrence, fate, human exposure, and human health risks of p-phenylenediamines and their quinones. Sci. Total. Environ. 2024, 957, 177742.
26. Zhao, H. N.; Thomas, S. P.; Zylka, M. J.; Dorrestein, P. C.; Hu, W. Urine excretion, organ distribution, and placental transfer of 6PPD and 6PPD-quinone in mice and potential developmental toxicity through nuclear receptor pathways. Environ. Sci. Technol. 2023, 57, 13429-38.
27. Chen, H.; Jin, H.; Ren, F.; Guo, R.; Zhu, J.; Huang, K. Enantioselectivity in human urinary excretion of N-(1,3-dimethylbutyl)-N'-phenyl-1,4-benzenediamine (6PPD) and 6PPD-quinone. Environ. Pollut. 2025, 378, 126489.
28. Jia, K.; Sun, J.; Du, Q.; et al. Mass Spectrometry imaging unveils the metabolic effect of 6PPD-quinone in exposed mice. Environ. Sci. Technol. 2025, 59, 4282-91.
29. Qin, Z.; Li, Y.; Qin, Y.; et al. Correlation between 6PPD-Q and immune along with metabolic dysregulation induced liver lesions in outdoor workers. Environ. Int. 2025, 199, 109455.
30. Fang, J.; Wang, X.; Cao, G.; et al. 6PPD-quinone exposure induces neuronal mitochondrial dysfunction to exacerbate Lewy neurites formation induced by α-synuclein preformed fibrils seeding. J. Hazard. Mater. 2024, 465, 133312.
31. Chen, H.; Wang, C.; Li, H.; et al. A review of toxicity induced by persistent organic pollutants (POPs) and endocrine-disrupting chemicals (EDCs) in the nematode Caenorhabditis elegans. J. Environ. Manage. 2019, 237, 519-25.
32. Tao, R.; Sun, M.; Ma, J.; et al. Advancing the understanding of PFAS-induced reproductive toxicity in key model species. Environ. Sci. Process. Impacts. 2025, 27, 3050-75.
33. Yang, B.; Aschner, M.; Lu, R. Toxicological insights into hydrogen sulfide biology in Caenorhabditis elegans: detection, metabolism, and functional outcomes. Crit. Rev. Toxicol. 2025, 55, 735-50.
34. Wang D-Y. Exposure toxicology in Caenorhabditis elegans. Springer Nature Singapore Pte Ltd, 2020.
35. Wu, J.; Shao, Y.; Hua, X.; Li, Y.; Wang, D. Photo-aged polylactic acid microplastics causes severe transgenerational decline in reproductive capacity in C. elegans: insight into activation of DNA damage checkpoints affected by multiple germline histone methyltransferases. Environ. Pollut. 2025, 382, 126697.
36. Wang D-Y. Toxicology at environmentally relevant concentrations in Caenorhabditis elegans. Springer Nature Singapore Pte Ltd, 2022.
37. Wang, W.; Hu, G.; Li, Y.; Wang, D. 6-PPD quinone inhibits ammonia excretion to cause multiple aspects of toxicity in Caenorhabditis elegans by activating dual oxidase complex-SKN-1 axis. Environ. Pollut. 2026, 390, 127528.
38. Song, M.; Ruan, Q.; Wang, D. Paeoniflorin alleviates toxicity and accumulation of 6-PPD quinone by activating ACS-22 in Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 2024, 286, 117226.
39. Liu, Z.; Bian, Q.; Wang, D. 6-PPD quinone induces response of nuclear hormone receptors in the germline associated with formation of reproductive toxicity in Caenorhabditis elegans. J. Hazard. Mater. 2025, 495, 138815.
40. Hu, D.; Wang, Y.; Hu, G.; Liu, R.; Wang, D. 6-PPD quinone-inhibited retinoic acid synthesis mediates toxicity through feedback loop between ALH-3/DHS-19-SEX-1 axis and intestinal signals in Caenorhabditis elegans. J. Environ. Expo. Assess. 2025, 4, 40.
41. Hua, X.; Wang, D. 6-PPD quinone causes alteration in ubiquinone-mediated complex III associated with toxicity on mitochondrial function and longevity in Caenorhabditis elegans. J. Environ. Chem. Eng. 2025, 13, 116571.
42. Wang, Y.; Hu, G.; Wang, D. 6-PPD quinone reduces lifespan by activating a feedback loop between cholesterol transformation related signal and insulin signaling in C. elegans. J. Environ. Sci. 2025, S1001074225006990.
43. Hua, X.; Wang, D. 6-PPD quinone at environmentally relevant concentrations induced damage on longevity in C. elegans: mechanistic insight from inhibition in mitochondrial UPR response. Sci. Total. Environ. 2024, 954, 176275.
44. Hua, X.; Wang, D. An environmentally relevant concentration of 6-PPD quinone inhibits two types of mitophagy to cause mitochondrial dysfunction and lifespan reduction in Caenorhabditis elegans. Environ. Sci. Process. Impacts. 2025, 27, 1928-40.
45. An, L.; Fu, X.; Chen, J.; Ma, J. Application of Caenorhabditis elegans in lipid metabolism research. Int. J. Mol. Sci. 2023, 24, 1173.
46. Wan, X.; Liang, G.; Wang, D. 6-PPD quinone at environmentally relevant concentrations disrupts citric acid cycle in Caenorhabditis elegans: role of reduction in acetyl CoA and pyruvate contents. Environ. Chem. Ecotoxicol. 2025, 7, 1119-29.
47. Wang, W.; Li, Y.; Wang, D. Long-term exposure to 6-PPD quinone inhibits glutamate synthesis and glutamate receptor function associated with its toxicity induction in Caenorhabditis elegans. Toxics 2025, 13, 434.
48. Wu, J.; Li, L.; Hu, D.; Liu, R.; Bian, Q.; Wang, D. Environmentally relevant concentrations of 6-PPDQ disrupt vitamin D3 adsorption and receptor function in Caenorhabditis elegans. Environ. Sci. Process. Impacts. 2025, 27, 2798-808.
49. Wang, Y.; Wu, J.; Wang, D. 6-PPD quinone causes lipid accumulation across multiple generations differentially affected by metabolic sensors and components of COMPASS complex in Caenorhabditis elegans. Environ. Pollut. 2025, 366, 125539.
50. Flynn NE, Meininger CJ, Haynes TE, Wu G. The metabolic basis of arginine nutrition and pharmacotherapy. Biomed. Pharmacother. 2002, 56, 427-38.
51. Khalaf, D.; Krüger, M.; Wehland, M.; Infanger, M.; Grimm, D. The effects of oral l-arginine and l-citrulline supplementation on blood pressure. Nutrients 2019, 11, 1679.
52. Santulli, G.; Trimarco, V.; Trimarco, B.; Izzo, R. Beneficial effects of Vitamin C and l-arginine in the treatment of post-acute sequelae of COVID-19. Pharmacol. Res. 2022, 185, 106479.
53. Rotoli, B. M.; Barilli, A.; Visigalli, R.; Ferrari, F.; Dall’Asta, V. y+LAT1 and y+LAT2 contribution to arginine uptake in different human cell models: implications in the pathophysiology of lysinuric protein intolerance. J. Cell. Mol. Med. 2020, 24, 921-9.
54. Díaz-Pérez, F.; Radojkovic, C.; Aguilera, V.; et al. L-arginine transport and nitric oxide synthesis in human endothelial progenitor cells. J. Cardiovasc. Pharmacol. 2012, 60, 439-49.
55. Tang, R.; Wang, X.; Zhou, J.; et al. Defective arginine metabolism impairs mitochondrial homeostasis in Caenorhabditis elegans. J. Genet. Genomics. 2020, 47, 145-56.
56. Fiordelisi, A.; Cerasuolo, F. A.; Avvisato, R.; et al. L-arginine supplementation as mitochondrial therapy in diabetic cardiomyopathy. Cardiovasc. Diabetol. 2024, 23, 450.
57. Wang, Z.; Yang, N.; Hou, Y.; et al. L-arginine-loaded gold nanocages ameliorate myocardial ischemia/reperfusion injury by promoting nitric oxide production and maintaining mitochondrial function. Adv. Sci. (Weinh). 2023, 10, e2302123.
59. Wang, Y.; Wang, D. Effect of disruption in the intestinal barrier function during the transgenerational process on nanoplastic toxicity induction in Caenorhabditis elegans. Environ. Sci.:. Nano. 2025, 12, 2741-9.
60. Wang, W.; Li, Y.; Wang, D. Exposure to 6-PPD quinone disrupts adsorption and catabolism of leucine and causes mitochondrial dysfunction in Caenorhabditis elegans. Toxics 2025, 13, 544.
61. Wu, T.; He, J.; Ye, Z.; et al. Aged biodegradable nanoplastics enhance body accumulation associated with worse neuronal damage in Caenorhabditis elegans. Environ. Sci. Technol. 2025, 59, 4352-63.
62. Hua, X.; Wang, D. Transgenerational response of germline histone acetyltransferases and deacetylases to nanoplastics at predicted environmental doses in Caenorhabditis elegans. Sci. Total. Environ. 2024, 952, 175903.
63. Wang, Y.; Hu, G.; Wang, D. Increased S-adenosyl methionine strengthens the suppression in mitochondrial unfolded protein response induced by 6-PPD quinone at environmentally relevant concentrations in Caenorhabditis elegans. Environ. Pollut. 2025, 386, 127231.
64. Hua, X.; Wang, D. 6-PPD quinone at environmentally relevant concentrations activates feedback response of electron transport chain to mediate damage on mitochondrial function and longevity in Caenorhabditis elegans. Environ. Chem. Ecotoxicol. 2025, 7, 2356-65.
65. Edwards, C.; Canfield, J.; Copes, N.; et al. Mechanisms of amino acid-mediated lifespan extension in Caenorhabditis elegans. BMC. Genet. 2015, 16, 8.
66. Wu, J.; Shen, S.; Wang, D. 6-PPD quinone at environmentally relevant concentrations induces immunosenescenece by causing immunosuppression during the aging process. Chemosphere 2024, 368, 143719.
67. Liu, Z.; Li, Y.; Wang, D. Transgenerational response of glucose metabolism in Caenorhabditis elegans exposed to 6-PPD quinone. Chemosphere 2024, 367, 143653.
68. Hua, X.; Liang, G.; Chao, J.; Wang, D. Exposure to 6-PPD quinone causes damage on mitochondrial complex I/II associated with lifespan reduction in Caenorhabditis elegans. J. Hazard. Mater. 2024, 472, 134598.
69. Wang D-Y. Molecular toxicology in Caenorhabditis elegans. In: Nematodes as Model Organisms, Springer Nature Singapore Pte Ltd, 2019; pp. 244-75.[DOI: 10.1079/9781789248814.0010].
70. Hua, X.; Wang, D. Exposure to 6-PPD quinone at environmentally relevant concentrations inhibits both lifespan and healthspan in C. elegans. Environ. Sci. Technol. 2023, 57, 19295-303.
71. Veljkovic, E.; Stasiuk, S.; Skelly, P. J.; Shoemaker, C. B.; Verrey, F. Functional characterization of Caenorhabditis elegans heteromeric amino acid transporters. J. Biol. Chem. 2004, 279, 7655-62.
72. Ishii, N.; Fujii, M.; Hartman, P. S.; et al. A mutation in succinate dehydrogenase cytochrome b causes oxidative stress and ageing in nematodes. Nature 1998, 394, 694-7.
73. Kayser, E. B.; Morgan, P. G.; Hoppel, C. L.; Sedensky, M. M. Mitochondrial expression and function of GAS-1 in Caenorhabditis elegans. J. Biol. Chem. 2001, 276, 20551-8.
74. Suthammarak, W.; Somerlot, B. H.; Opheim, E.; Sedensky, M.; Morgan, P. G. Novel interactions between mitochondrial superoxide dismutases and the electron transport chain. Aging. Cell. 2013, 12, 1132-40.
75. Anderson, N. S.; Haynes, C. M. Folding the Mitochondrial UPR into the Integrated Stress Response. Trends. Cell. Biol. 2020, 30, 428-39.
76. Rashid, J.; Kumar, S. S.; Job, K. M.; Liu, X.; Fike, C. D.; Sherwin, C. M. T. Therapeutic potential of citrulline as an arginine supplement: a clinical pharmacology review. Paediatr. Drugs. 2020, 22, 279-93.
77. Yin, J.; Liu, R.; Jian, Z.; et al. Di (2-ethylhexyl) phthalate-induced reproductive toxicity involved in dna damage-dependent oocyte apoptosis and oxidative stress in Caenorhabditis elegans. Ecotoxicol. Environ. Saf. 2018, 163, 298-306.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].