Metabolic biomarkers predict subclinical cardiac remodeling in obese individuals
Abstract
Aim: To explore the relationship between metabolic biomarkers and cardiac remodeling indices derived from cardiac magnetic resonance (CMR) in obese adults, focusing on the role of insulin (INS) resistance and lipotoxicity in early cardiac changes.
Methods: A total of 107 obese adults undergoing preoperative metabolic evaluation were included. CMR was used to assess left ventricular (LV) structure and function. Metabolic biomarkers, including INS, free fatty acids (FFAs), triglycerides (TG), and glycated hemoglobin (HbA1c), were measured. Correlation and multivariable regression analyses were performed to examine associations between biomarkers and CMR-derived cardiac parameters. Logistic regression with receiver operating characteristic (ROC) analysis identified predictors of left ventricular hypertrophy (LVH).
Results: In this cohort of obese adults without overt hyperglycemia and with preserved ejection fraction, higher body mass index (BMI) was associated with greater left ventricular mass (LVM), volumes, and cardiac output. In multivariable models, TG (adjusted for BMI) independently predicted LVM, while a combined INS + FFAs model was a strong predictor of LVH [area under the curve (AUC) = 0.757; sensitivity = 88.7%; specificity = 37.8%; P < 0.001]. Results remained consistent when LVM was indexed to height2.7.
Conclusions: Metabolic biomarkers reflecting INS resistance and lipotoxicity are associated with early cardiac remodeling in obesity. TG captured the continuous burden of LVM, and the INS + FFAs model offered a practical metabolic prescreen for LVH risk, supporting early cardiovascular risk stratification in obese population.
Keywords
INTRODUCTION
Obesity is a growing global health concern, increasingly prevalent among both adults and children[1]. It is closely linked to metabolic disorders such as type 2 diabetes, dyslipidemia, insulin (INS) resistance, and metabolic-associated fatty liver disease (MAFLD), and is a well-established risk factor for cardiovascular disease[2,3]. Beyond systemic metabolic dysregulation, obesity induces nutritional-metabolic imbalance, characterized by excess nutrient-derived lipid accumulation, impaired INS signaling, and chronic low-grade inflammation. These alterations contribute to cardiac remodeling manifested as left ventricular hypertrophy (LVH), chamber dilation, and increased myocardial mass, which may precede systolic or diastolic dysfunction[4]. The underlying mechanisms include both hemodynamic overload and direct metabolic insults related to lipotoxicity and nutrient-driven metabolic stress[5].
Obesity is metabolically heterogeneous, and metabolically healthy obesity may still lead to early structural cardiac changes[6,7]. Obesity-related cardiac remodeling is partially reversible, with bariatric surgery showing significant improvements in cardiac structure, including reductions in left ventricular mass (LVM) and end-diastolic volume (EDV)[8]. Prior studies have shown that obesity is associated with elevated high-sensitivity cardiac troponin T and echocardiographic remodeling indices[9]. However, echocardiography is less precise than cardiac magnetic resonance (CMR), the gold standard for quantifying LVM, volumes, and function[10]. In clinical practice, anti-obesity pharmacotherapy, particularly incretin-based agents, is gaining attention, and early identification of high-risk individuals is crucial for guiding prevention strategies[11].
Experimental human evidence suggests that increasing circulating free fatty acids (FFAs) can adversely affect myocardial energetics, supporting a potential lipotoxic pathway linking metabolic stress to early cardiac remodeling[12]. Concurrently, circulating metabolic biomarkers, such as fasting glucose (GLU), INS, and liver enzymes, potentially explain obesity-related cardiac changes[13,14]. Nevertheless, most prior studies have examined CMR phenotypes and metabolic profiles separately, leaving the mechanistic links between nutrition-related metabolic dysfunction, glycemic status, and subclinical cardiac remodeling incompletely understood[15]. To address this gap, we examined the associations between routinely available metabolic biomarkers and CMR-derived cardiac remodeling parameters in individuals with obesity and evaluated whether a simple combined metabolic index (INS + FFAs) could serve as a pragmatic prescreen to identify individuals at higher risk of cardiac remodeling.
METHODS
Patient selection
The study included 107 adults with obesity who underwent preoperative evaluation at the Center for Bariatric and Metabolic Disorders, Zhongnan Hospital of Wuhan University. All participants met the clinical criteria for metabolic bariatric surgery[16]. Exclusion criteria were: (1) severe cardiovascular diseases (e.g., uncontrolled hypertension, coronary artery disease, or heart failure); (2) advanced metabolic or hepatic disorders; and (3) conditions compromising image quality or surgical safety. Detailed baseline characteristics, including age and sex distribution, are provided in Table 1.
Baseline characteristics across obesity classes
| Parameter | Class I (n = 47) | Class II (n = 40) | Class III (n = 20) | Overall P value |
| Age years | 31.0 (25.0-38.0) | 28.5 (25.0-36.5) | 26.0 (23.0-30.8) | 0.113 |
| Sex (%) | 41 (87.2) | 31 (77.5) | 14 (70.0) | 0.226 |
| TC (mmol/L) | 4.76 (4.21-5.78) | 5.00 (4.40-5.69) | 4.71 (4.04-4.97) | 0.185 |
| TG (mmol/L) | 1.59 (1.27-2.03) | 2.09 (1.60-2.41) | 1.47 (0.96-2.20) | 0.057 |
| HDL (mmol/L) | 1.08 (0.95-1.27) | 1.01 (0.90-1.16) | 1.10 (0.98-1.23) | 0.784 |
| LDL (mmol/L) | 2.91 (2.59-3.80) | 3.31 (2.76-3.94) | 3.02 (2.63-3.49) | 0.288 |
| FFAs (μmol/L) | 505.40 (411.90-578.55) | 568.07 (451.85-649.90) | 538.50 (472.73-704.58) | < 0.001 |
| GLU (mmol/L) | 5.01 (4.74-5.75) | 5.51 (5.08-6.59) | 5.47 (4.75-6.07) | 0.013 |
| HbA1c (%) | 5.50 (5.15-6.00) | 5.80 (5.60-6.23) | 5.70 (5.50-5.93) | 0.104 |
| INS (μIU/mL) | 16.60 (11.90-23.30) | 23.55 (20.15-31.48) | 24.40 (19.88-30.43) | 0.028 |
| C-peptide (ng/mL) | 3.50 (2.71-4.24) | 4.90 (3.96-5.69) | 4.93 (3.60-5.97) | < 0.001 |
| HOMA-IR | 3.47 (2.74-6.31) | 5.92 (4.80-8.65) | 6.28 (4.50-7.59) | 0.010 |
| WBC (× 109/L) | 6.70 (5.65-7.99) | 7.55 (6.23-9.95) | 7.61 (6.88-8.12) | 0.009 |
| ALT (U/L) | 29.00 (19.50-59.00) | 37.00 (26.25-59.00) | 44.50 (39.75-53.00) | 0.085 |
| AST (U/L) | 24.00 (16.00-41.00) | 26.50 (17.25-33.00) | 24.50 (18.50-37.75) | 0.491 |
| GGT (U/L) | 31.00 (17.50-40.50) | 34.00 (26.50-52.00) | 29.50 (24.50-54.00) | 0.016 |
| LVEF (%) | 64.20 (60.42-67.46) | 65.43 (55.98-70.95) | 65.20 (59.63-68.39) | 0.434 |
| CO (L/min) | 6.19 (5.56-6.98) | 6.97 (5.77-8.52) | 6.99 (6.45-9.19) | 0.047 |
| CI (L/min/m2) | 3.20 (2.70-3.51) | 3.18 (2.83-4.03) | 3.20 (2.67-3.93) | 0.977 |
The study protocol was reviewed and approved by the Institutional Ethics Committee of Zhongnan Hospital of Wuhan University (Approval No. 2019021). Written informed consent was obtained from all participants prior to inclusion.
Clinical and laboratory assessment
Preoperative evaluations included demographic data (age, sex), anthropometric parameters [weight, height, and body mass index (BMI)], and cardiovascular assessments. According to the American Association of Clinical Endocrinology (AACE)/American College of Endocrinology (ACE) criteria[17], participants were categorized as Class I (BMI 30.0-34.9 kg/m2; n = 47), Class II (BMI 35.0-39.9 kg/m2; n = 40), and Class III (BMI ≥ 40.0 kg/m2; n = 20).
Fasting venous blood samples collected within three days before surgery were analyzed for the following parameters. All biochemical tests were conducted in the hospital’s central laboratory using standard automated clinical analyzers under routine quality control procedures. Specifically, the measured parameters included:
Glucose metabolism: GLU, INS, glycated hemoglobin (HbA1c), C-peptide, and homeostasis model assessment of insulin resistance (HOMA-IR);
Lipid profile: total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), and FFAs;
Liver and inflammatory markers: alanine aminotransferase (ALT), aspartate aminotransferase (AST), γ-glutamyl transferase (GGT), and white blood cell count (WBC).
Cardiac magnetic resonance imaging acquisition and parameters
All participants underwent CMR on a 1.5-T clinical magnetic resonance imaging (MRI) system. CMR data were analyzed by two trained radiologists using semi-automated software. The derived indices were classified as structural parameters - including LVM, EDV, end-systolic volume (ESV), and stroke volume (SV) - and functional parameters, comprising ejection fraction (EF), cardiac output (CO), and cardiac index (CI). Although SV represents a functional output, it was categorized under structural indices because it directly reflects volumetric remodeling.
LVH was predefined as a binary variable based on sex-specific cut-offs for CMR-derived LVM (> 115 g in men and > 95 g in women)[18].
Statistical analysis
Statistical analyses were performed using SPSS version 29.0, Python (scikit-learn, pymatch), and R (glmnet, ggplot2, pheatmap). Continuous variables were expressed as medians [interquartile ranges (IQRs)] and compared across groups using the Kruskal-Wallis H test, whereas categorical variables (n, %) were compared using Chi-square or Fisher’s exact tests. This analysis was exploratory and hypothesis-generating. All tests were two-sided, with P < 0.05 considered statistically significant. Regression estimates are presented with 95% confidence intervals (CIs). Structural indices (LVM, EDV, ESV, SV) were additionally indexed to height2.7 (g/m2.7 or mL/m2.7), as recommended for obese cohorts.
To account for baseline differences in age, sex, and BMI across metabolic syndrome (MetS) strata, 1:1 nearest-neighbor propensity score matching (PSM; caliper = 0.10; without replacement) was conducted, achieving covariate balance (standardized mean difference < 0.10; Supplementary Table 1). Primary analyses were performed in the full cohort with multivariable adjustment.
Metabolic predictors of cardiac parameters were first identified using least absolute shrinkage and selection operator (LASSO) regression, followed by multivariable linear modeling. Standardized regression coefficients (β), P values, and variance inflation factors (VIFs; acceptable if < 3) were reported. For sensitivity analysis, hierarchical linear regression models were also performed with LVM indexed to height2.7 as a continuous outcome.
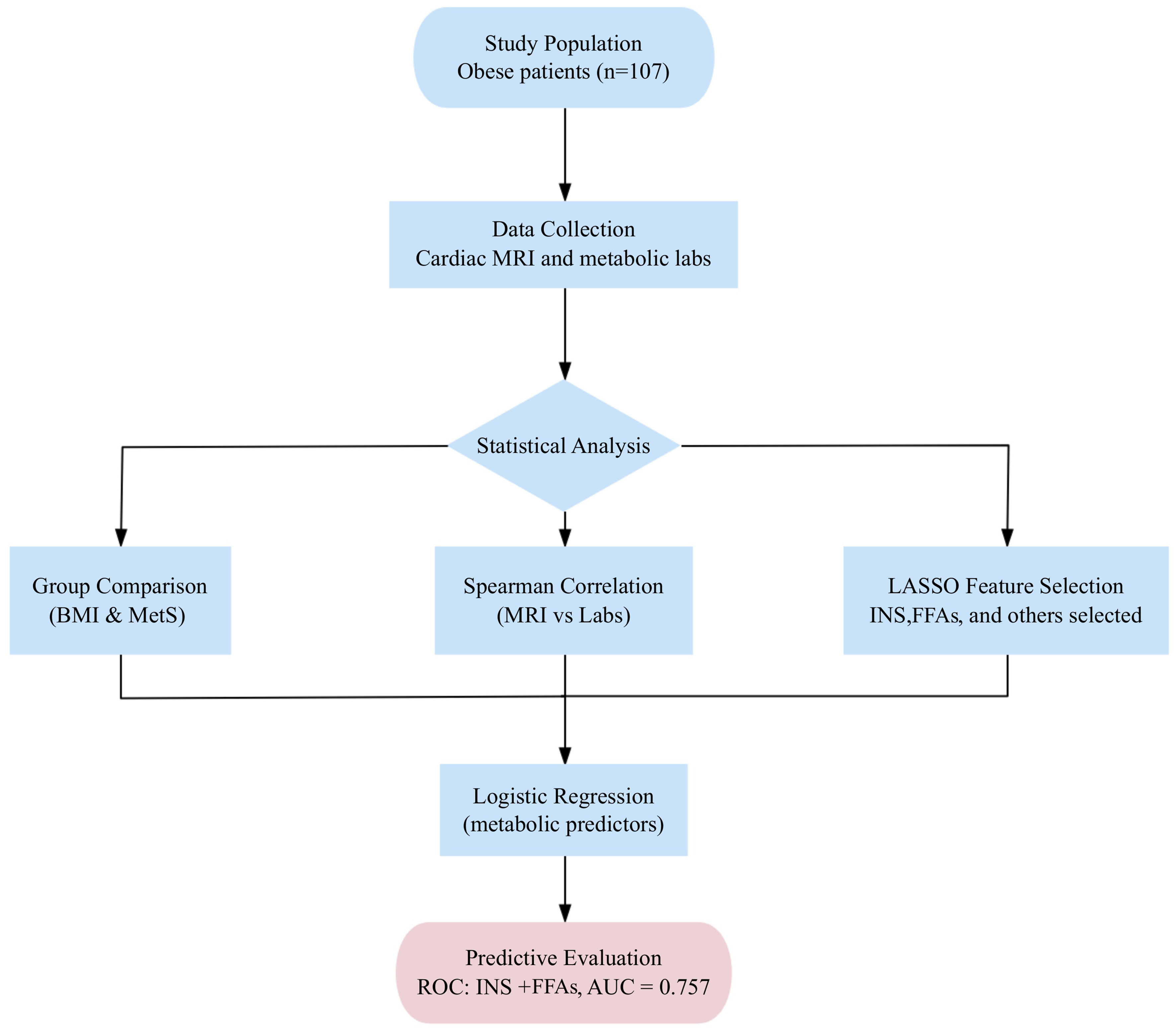
Logistic regression was applied for binary outcomes (e.g., LVH), and model performance was evaluated by receiver operating characteristic (ROC) analysis, including the area under the curve (AUC), optimal cut-off, sensitivity, and specificity. Figure 1 summarizes the analytical workflow.
Figure 1. Study flowchart. The study included 107 adults with obesity who underwent CMR and metabolic laboratory assessments. The analytical workflow comprised group comparisons (BMI and MetS strata), correlation analyses between CMR and laboratory indices, LASSO feature selection, and logistic regression for predictive evaluation of metabolic biomarkers associated with cardiac remodeling. MRI: Magnetic resonance imaging; CMR: cardiac magnetic resonance; BMI: body mass index; MetS: metabolic syndrome; INS: insulin; FFAs: free fatty acids; LASSO: least absolute shrinkage and selection operator; ROC: receiver operating characteristic; AUC: area under the curve.
RESULTS
Clinical and metabolic characteristics by obesity class
Patients were stratified into three BMI-based groups: Class I (30.0-34.9 kg/m2, n = 47), Class II (35.0-
Liver and inflammatory markers also differed across classes. GGT showed significant variation (P = 0.016), with the highest median value in Class II. WBC increased progressively and reached its peak in Class III (P = 0.009), consistent with obesity-related low-grade inflammation. Although TG tended to peak in Class II, the difference did not reach statistical significance (P = 0.057). Other biochemical parameters - including TC, HDL, LDL, ALT, and AST - did not differ significantly among groups (P > 0.05).
Cardiac MRI findings across obesity classes
Functional indices [Table 2] showed preserved left ventricular ejection fraction (LVEF) and CI across obesity classes (P = 0.434 and P = 0.977), whereas CO was higher in Class II-III (P = 0.047), consistent with increased circulatory demand. Structural measurements [Table 3] demonstrated stepwise increases in LVM (P = 0.001), EDV (P < 0.001), ESV (P = 0.007), and SV (P = 0.007), indicating concentric-eccentric remodeling with hypertrophy and chamber dilation. After adjustment for body surface area (BSA) [Supplementary Table 2], indexed values [left ventricular mass index (LVMi), end-diastolic volume index (EDVi), end-systolic volume index (ESVi), and stroke volume index (SVi)] showed no significant differences across obesity classes (all P > 0.3). When indices were normalized to height2.7, significant increases were observed in LVM/ht2.7, EDV/ht2.7 and ESV/ht2.7 in Class III obesity compared with lower classes, whereas SV/ht2.7 demonstrated only a nonsignificant upward trend [Supplementary Table 3].
Cardiac functional parameters across BMI categories
| Parameter | Class I (n = 47) | Class II (n = 40) | Class III (n = 20) | Overall P value |
| LVEF (%) | 64.20 (60.42-67.46) | 65.43 (55.98-70.95) | 65.20 (59.63-68.39) | 0.434 |
| CO (L/min) | 6.19 (5.56-6.98) | 6.97 (5.77-8.52) | 6.99 (6.45-9.19) | 0.047 |
| CI (L/min/m2) | 3.20 (2.70-3.51) | 3.18 (2.83-4.03) | 3.20 (2.67-3.93) | 0.977 |
Cardiac structural parameters (unindexed) across BMI categories
| Parameter | Class I (n = 47) | Class II (n = 40) | Class III (n = 20) | Overall P value |
| LVM (g) | 96.80 (81.43-120.09) | 107.03 (94.76-139.09) | 115.45 (101.20-124.85) | 0.001 |
| EDV (mL) | 139.68 (120.90-152.55) | 136.40 (128.63-165.88) | 168.31 (156.94-182.87) | < 0.001 |
| ESV (mL) | 49.97 (38.97-59.91) | 51.46 (40.94-62.33) | 61.57 (49.83-68.81) | 0.007 |
| SV (mL) | 89.56 (78.22-98.74) | 92.40 (79.40-101.32) | 109.35 (102.26-120.44) | 0.007 |
Association between MetS and cardiac remodeling
Patients were categorized into MetS- (n = 28) and MetS+ (n = 79) groups according to the revised Adult Treatment Panel III (ATP III) criteria[19], defined as the presence of three or more of the following components: central obesity, elevated blood pressure, hypertriglyceridemia, low HDL cholesterol, and elevated GLU.
Cardiac function
As summarized in Supplementary Table 4, individuals in the MetS+ group exhibited significantly lower CO and CI compared with those in the MetS- group (CO: 6.67 L/min vs. 7.27 L/min, P = 0.017; CI:
Cardiac structure
As shown in Supplementary Table 5, absolute LVM did not differ significantly between MetS- and MetS+ groups (99.18 g vs. 102.97 g, P = 0.662). However, when indexed to BSA, the LVMi tended to be higher in the MetS+ group compared with the MetS- group (48.75 g/m2 vs. 47.09 g/m2, P = 0.061). In addition, the EDV was significantly lower in participants with MetS (P = 0.021), while the ESV was similar between groups (P = 0.738). Consequently, the SV and SVi were both significantly reduced in the MetS+ group (SV: P < 0.001; SVi: P = 0.004).
Distribution trend
The prevalence of MetS increased progressively with obesity severity: 66.0% in Class I, 80.0% in Class II, and 85.0% in Class III. This linear trend (Z = 1.836, P = 0.044) showed a rising metabolic burden accompanying higher degrees of obesity [Supplementary Figure 1].
Correlation between metabolic indicators and cardiac MRI parameters
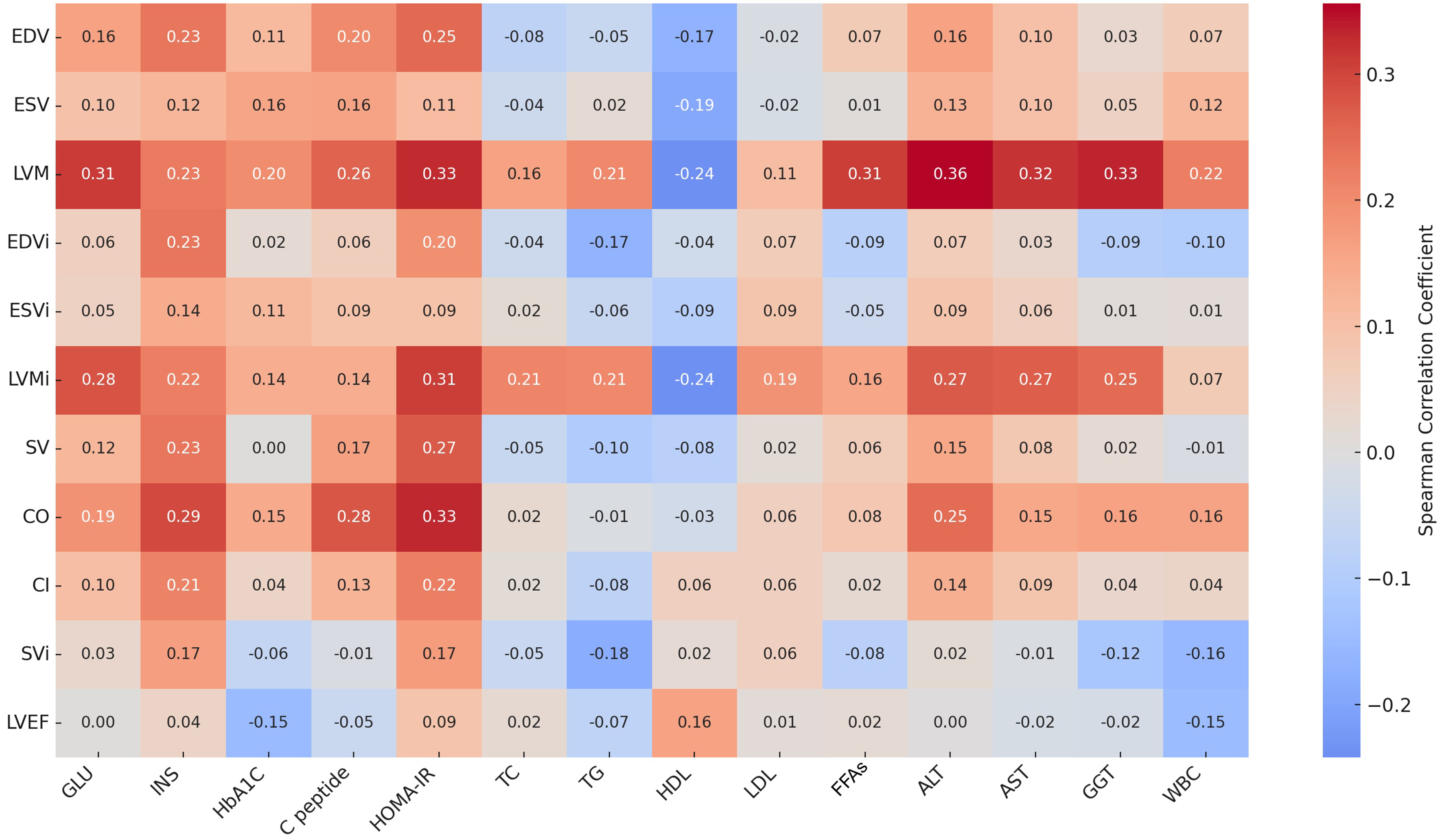
Spearman correlation analysis [Figure 2] demonstrated that LVM and LVMi were positively correlated with INS resistance markers (HOMA-IR: r = 0.31; C-peptide: r = 0.26; INS: r = 0.23), FFAs (r = 0.31), and liver enzymes (ALT: r = 0.36; GGT: r = 0.33). CO correlated with HOMA-IR (r = 0.33) and C-peptide (r = 0.28), while SV correlated with HOMA-IR (r = 0.27). LVEF showed only weak correlations, whereas volumetric indices such as EDV, ESV, EDVi, and SVi exhibited mild-to-moderate associations with metabolic markers. Although SVi showed weaker relationships with FFAs (r = 0.08) and HOMA-IR (r = 0.17), the overall correlation pattern remained consistent.
Figure 2. Spearman correlation heatmap between metabolic indicators (x-axis) and cardiac MRI parameters (y-axis). Color intensity represents correlation strength (red = positive, blue = negative). LVM and LVMi correlated positively with HOMA-IR, FFAs, ALT, and GGT, while other parameters showed mild-to-moderate associations. Detailed coefficients are listed in Supplementary Table 6. MRI: Magnetic resonance imaging; EDV: end-diastolic volume; ESV: end-systolic volume; LVM: left ventricular mass; EDVi: end-diastolic volume index; ESVi: end-systolic volume index; LVMi: left ventricular mass index; SV: stroke volume; CO: cardiac output; CI: cardiac index; SVi: stroke volume index; LVEF: left ventricular ejection fraction; GLU: fasting glucose; INS: insulin; HbA1c: hemoglobin A1c; C-peptide: connecting peptide; HOMA-IR: homeostasis model assessment of insulin resistance; TC: total cholesterol; TG: triglycerides; HDL: high-density lipoprotein; LDL: low-density lipoprotein; FFAs: free fatty acids; ALT: alanine aminotransferase; AST: aspartate aminotransferase; GGT: gamma-glutamyl transferase; WBC: white blood cell.
LASSO selection and multivariable regression analysis
Key metabolic predictors of cardiac remodeling were identified using LASSO regression followed by multivariable linear modeling [Supplementary Table 7]. TG remained a significant positive predictor of LVM, with BMI also showing a significant association, whereas the relationship with GGT was attenuated after indexation and adjustment. HDL exhibited a non-significant inverse trend. For CO, ALT was a significant predictor, and INS showed a near-significant association. Comparable results were observed for CI. The identified metabolic predictors are summarized in Supplementary Figure 2, and detailed regression results are presented in Supplementary Table 8.
Predictive performance of metabolic indicators for LVH
Logistic regression was used to examine associations between metabolic factors and LVH. In univariable analysis, INS was associated with LVH [odds ratio (OR) 1.053, 95%CI: 1.014-1.094; P = 0.007]. In the multivariable model including the selected metabolic variables, INS (OR 1.046, 95%CI: 1.002-1.092; P = 0.038) and FFAs (OR 1.003, 95%CI: 1.001-1.006; P = 0.018) remained associated with LVH, whereas other covariates were not statistically significant (GGT: OR 1.018, 95%CI: 0.995-1.042; P = 0.122; TG: OR 1.160, 95%CI: 0.734-1.835; P = 0.525; HDL: OR 1.329, 95%CI: 0.109-16.229; P = 0.824; GLU: OR 0.956, 95%CI: 0.759-1.204; P = 0.701). Full model outputs are provided in Supplementary Table 9. As a sensitivity analysis, LVM/height2.7 was modeled continuously; inclusion of metabolic variables improved explained variance (ΔR2 = 0.265, P < 0.001), with TG and BMI remaining significant [Supplementary Table 10].
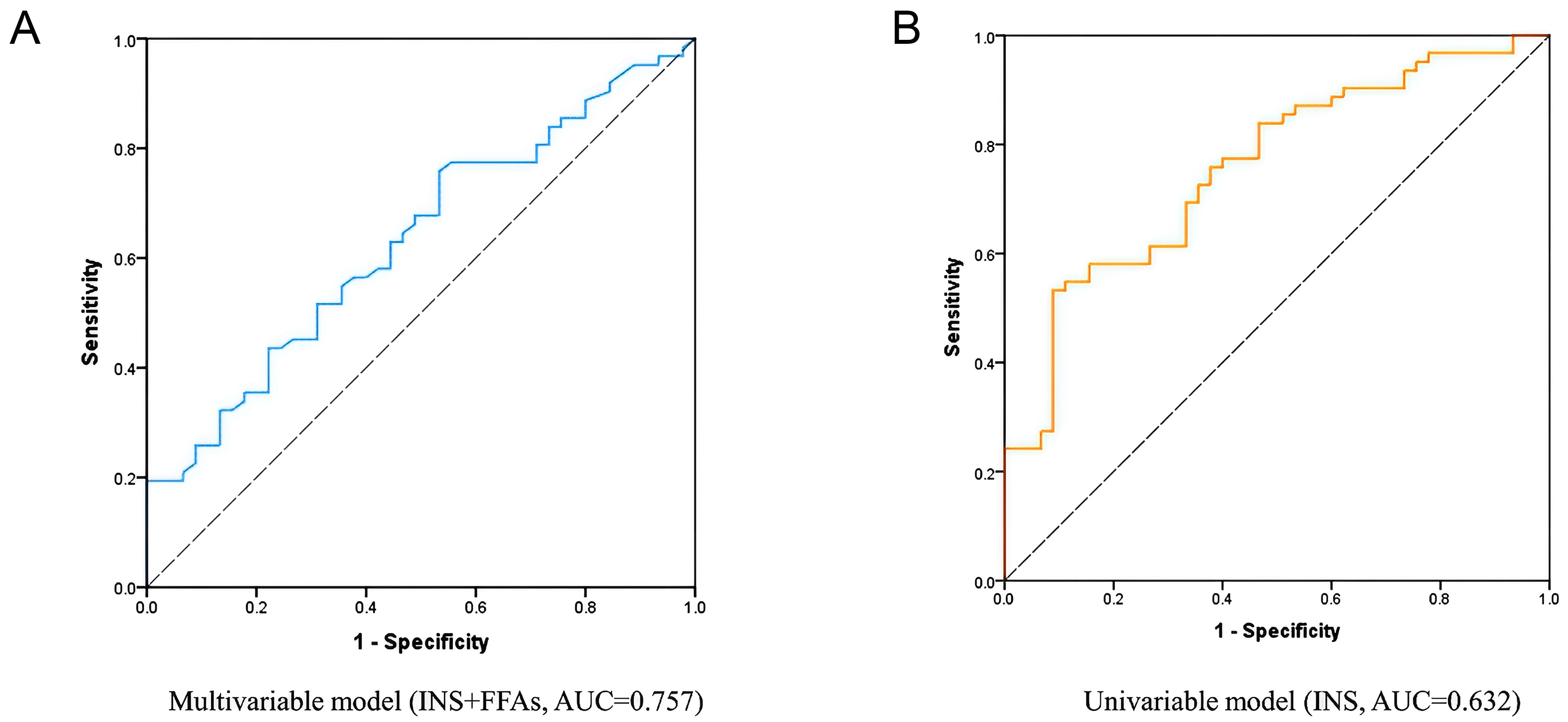
ROC analysis was performed to evaluate the discriminative performance of metabolic models for LVH. The INS univariable model yielded an AUC of 0.632 (95%CI: 0.525-0.739; P = 0.015) with an optimal cut-off of 0.513, sensitivity of 74.2%, and specificity of 46.7%. The combined INS + FFAs model showed improved discrimination with an AUC of 0.757 (95%CI: 0.667-0.848; P < 0.001) and an optimal cut-off of 0.41, achieving sensitivity of 88.7% and specificity of 37.8% [Figure 3 and Supplementary Table 11].
Figure 3. ROC curves for LVH prediction. (A) Combined model incorporating INS and FFAs; (B) INS-only model. The combined model yielded an AUC of 0.757 (95%CI: 0.667-0.848), compared with 0.632 (95%CI: 0.525-0.739) for the INS-only model. ROC: Receiver operating characteristic; LVH: left ventricular hypertrophy; INS: insulin; FFAs: free fatty acids; AUC: area under the curve; CI: confidence interval.
DISCUSSION
In this study, we integrated high-resolution cardiac MRI with comprehensive metabolic profiling to investigate the relationship between obesity-related myocardial remodeling and metabolic dysfunction. We found that, despite preserved LVEF, LVM and LVMi were significantly elevated in individuals with obesity - indicating early structural remodeling prior to overt systolic impairment.
These results are consistent with the concept of metabolic cardiomyopathy (McCM), in which obesity-related INS-lipid dysregulation and chronic low-grade inflammation contribute to myocardial hypertrophy and chamber dilation, whereas reductions in contractile function typically manifest at later stages of disease progression[20].
Across BMI strata, stepwise elevations in LVM, EDV, SV, and CO align with prior observations[15] and likely reflect combined effects of greater body size-related hemodynamic load[21] and worsening metabolic imbalance (elevated FFAs and INS resistance)[22,23]. Prior cohort studies have also reported a positive association between obesity and LVM[24]. Our CMR-based results extend this evidence by providing a detailed quantitative characterization of LV structure and output within a preoperative bariatric cohort, reinforcing the concept of a “cardiac obesity phenotype” that manifests before clinically apparent heart disease.
INS, FFAs, and TG emerged as key metabolic correlates of LV remodeling, consistent with evidence linking myocardial lipid accumulation to hypertrophy and functional impairment[25,26]. In this cohort, higher fasting INS and FFAs were associated with greater LVM and CO in correlation analyses, consistent with metabolic-lipid toxicity contributing to subclinical cardiomyopathy[27-29]. However, in multivariable linear models of continuous LVM (including height2.7 indexation in sensitivity analyses), TG remained the most robust independent predictor. BMI also showed a significant association, whereas INS and FFAs were not retained. By contrast, in logistic regression models for LVH, both INS and FFAs were significant predictors. Their combination achieved relatively high sensitivity (AUC = 0.757), supporting its potential utility for metabolic risk screening. Thus, TG reflects the continuous burden of hypertrophy, while the INS + FFAs combination discriminates the threshold transition to LVH.
Metabolic dysfunction-associated steatotic liver disease (MASLD) has been increasingly linked to early cardiac remodeling[30-34]. Recent studies show that MASLD is independently associated with cardiovascular disease and heart failure, including heart failure with preserved EF[35]. This is thought to be driven by mechanisms such as oxidative stress, systemic inflammation, and metabolic dysfunction, which contribute to the observed elevations in ALT and GGT - markers of liver injury and metabolic stress[36]. In our study, participants with MetS exhibited higher LVM and lower stroke volume (SV/SVi) with preserved EF, indicating concentric remodeling and reduced volumetric output. This pattern reflects hemodynamic adaptation under advanced metabolic stress, which aligns with the trend of higher CO associated with increased BMI.
The logistic model combining INS and FFAs showed superior discrimination for LVH compared with single predictors, consistent with a synergistic effect between INS resistance and lipotoxicity. Mechanistically, INS may contribute to LVH through sympathetic activation, extracellular volume expansion, and downstream Akt/mTOR (Akt/mammalian target of rapamycin) signaling[37-39]. Additionally, FFAs play a key role in cardiac structural remodeling, primarily via mitochondrial stress, oxidative injury, and activation of PPARα (peroxisome proliferator-activated receptor alpha), NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells), and TGF-β (transforming growth factor beta) pathways, all of which have been implicated in myocardial hypertrophy and fibrosis[40]. The interaction between INS and FFAs further exacerbates these effects, underscoring the critical role of metabolic dysfunction in the development of LVH. Elevated HOMA-IR levels have been shown to correlate with LVH, highlighting the intersection between INS resistance, metabolic dysregulation, and adverse cardiac remodeling in obese individuals[41,42].
Compared with electrocardiogram (ECG)-based artificial intelligence models or computed tomography (CT)-based tools that require ionizing radiation and complex algorithms, this non-invasive, laboratory-only method is practical and broadly accessible[43-45]. When implemented within standard metabolic test workflows, the INS+FFAs threshold could assist in triaging patients for cardiac imaging or closer follow-up. From a translational perspective, incretin-based therapies may specifically benefit the subgroup with high INS resistance and high FFAs. In particular, dual GIP (glucose-dependent insulinotropic polypeptide)/GLP-1 (glucagon-like peptide-1) receptor agonists, such as tirzepatide, could potentially improve both metabolic homeostasis and cardiac remodeling in these individuals[46,47].
This study has several limitations. First, the cross-sectional, single-center design precludes causal inference and may introduce selection bias. Second, restricting participants to bariatric surgery candidates with a marked female predominance limits broader applicability. Finally, this design precludes the assessment of sex hormone and menopausal effects, which may influence INS resistance, circulating FFAs, and myocardial remodeling[48]. Future studies with balanced sex representation and hormonal profiling are needed. Although CMR provided precise structural assessment, advanced tissue characterization (e.g., strain/mapping) and direct quantification of myocardial steatosis/ectopic fat were not performed; thus, lipid-deposition interpretations remain hypothesis-generating. No formal a priori sample size calculation was conducted because this was an exploratory analysis based on available CMR and laboratory data.
In conclusion, CMR integrated with metabolic profiling revealed that TG was independently associated with the continuous burden of LVM, whereas the combined INS and FFAs index effectively identified LVH. These findings suggest that simple metabolic biomarkers may provide a practical, laboratory-based prescreening tool for the early detection of subclinical cardiac remodeling in obesity, supporting targeted risk assessment in clinical practice. Despite the aforementioned limitations, this study emphasizes the importance of these tools for scalable risk stratification. Future multi-center longitudinal studies are needed to validate and calibrate the INS+FFAs model, exploring its broader clinical applications.
DECLARATIONS
Authors’ contributions
Conducted CMR data processing, clinical data collection, and statistical analysis: Hu M, Wang H
Participated in metabolic marker analysis and figure preparation: Mei Z
Contributed to data curation and patient recruitment: Gong C, Lan L, Ma Z
Supported the literature review and manuscript formatting: Zhang B, Fan H, Zhang Y
Assisted in manuscript revision and graphical abstract design: Wang H, Chun W
Supervised the study and provided critical revisions to the manuscript: Pan D, Zhou J, Li Z
All authors read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed in the current study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant No. 82270612) and the Science, Technology and Innovation Seed Fund of Zhongnan Hospital of Wuhan University (Grant No. CXPY2022087).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
This study was approved by the Medical Ethics Committee of Zhongnan Hospital of Wuhan University (Ethical Approval No. 2019021). Written informed consent was obtained from all participants prior to enrollment and data collection.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Risk Factor Collaboration (NCD-RisC). Worldwide trends in underweight and obesity from 1990 to 2022: a pooled analysis of 3663 population-representative studies with 222 million children, adolescents, and adults. Lancet. 2024;403:1027-50.
3. Powell-Wiley TM, Poirier P, Burke LE, et al; American Heart Association Council on Lifestyle and Cardiometabolic Health; Council on Cardiovascular and Stroke Nursing; Council on Clinical Cardiology; Council on Epidemiology and Prevention; and Stroke Council. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:e984-1010.
4. Szabo L, McCracken C, Cooper J, et al. The role of obesity-related cardiovascular remodelling in mediating incident cardiovascular outcomes: a population-based observational study. Eur Heart J Cardiovasc Imaging. 2023;24:921-9.
5. Zlobine I, Gopal K, Ussher JR. Lipotoxicity in obesity and diabetes-related cardiac dysfunction. Biochim Biophys Acta. 2016;1861:1555-68.
6. Mohammed AS, Ahmed H, Singh S, et al. Impact of metabolically healthy obesity on cardiovascular outcomes in older adults with HFpEF: insights from a nationwide sample. J Clin Med. 2025;14:5495.
7. Su Y, Li Y, Ding R, et al. Association between metabolically healthy obesity and risk of dementia: a systematic review and meta‐analysis. Med Adv. 2024;2:176-85.
8. Aggarwal R, Harling L, Efthimiou E, Darzi A, Athanasiou T, Ashrafian H. The effects of bariatric surgery on cardiac structure and function: a systematic review of cardiac imaging outcomes. Obes Surg. 2016;26:1030-40.
9. Huang J, Liu M, Su E, et al. Elevated circulating high-sensitivity cardiac troponin t and cardiac remodeling in obesity. BMC Cardiovasc Disord. 2021;21:620.
10. Pontone G, Guaricci AI, Andreini D, et al. Prognostic benefit of cardiac magnetic resonance over transthoracic echocardiography for the assessment of ischemic and nonischemic dilated cardiomyopathy patients referred for the evaluation of primary prevention implantable cardioverter-defibrillator therapy. Circ Cardiovasc Imaging. 2016;9:e004956.
11. Abid MA, Asad MB, Razzak MJ. Natural peptides versus semaglutide: a safer approach to obesity management. Med Adv. 2025;3:202-4.
12. Holloway CJ, Cochlin LE, Emmanuel Y, et al. A high-fat diet impairs cardiac high-energy phosphate metabolism and cognitive function in healthy human subjects. Am J Clin Nutr. 2011;93:748-55.
13. Larios G, Uribe S, Trincado C, et al. Myocardial function, mechanics and work by echocardiography in adolescents with severe obesity. Cardiovasc Endocrinol Metab. 2024;13:e0301.
14. Bakhtiyari M, Kazemian E, Kabir K, et al. Contribution of obesity and cardiometabolic risk factors in developing cardiovascular disease: a population-based cohort study. Sci Rep. 2022;12:1544.
15. Liu J, Li J, Yu J, et al. Regional fat distributions are associated with subclinical right ventricular dysfunction in adults with uncomplicated obesity. Front Cardiovasc Med. 2022;9:814505.
16. De Luca M, Shikora S, Eisenberg D, et al. Scientific evidence for the updated guidelines on indications for metabolic and bariatric surgery (IFSO/ASMBS). Obes Surg. 2024;34:3963-4096.
17. Garvey WT, Mechanick JI, Brett EM, et al; Reviewers of the AACE/ACE Obesity Clinical Practice Guidelines. American association of clinical endocrinologists and american college of endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract. 2016;22:1-203.
18. Ponikowski ADGDTP, Voors AA, Anker SD, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Rev Esp Cardiol. 2016;69:1167.e1-85.
19. Grundy SM, Cleeman JI, Daniels SR, et al; American Heart Association, National Heart, Lung, and Blood Institute. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112:2735-52.
21. Sha R, Baines O, Hayes A, et al. Impact of obesity on atrial fibrillation pathogenesis and treatment options. J Am Heart Assoc. 2024;13:e032277.
22. Tian Y, Yang J, Lan M, Zou T. Construction and analysis of a joint diagnosis model of random forest and artificial neural network for heart failure. Aging. 2020;12:26221-35.
23. Pinheiro LCHT, Rossi M, Dos Santos CAF, et al. Prevalence of associations among sarcopenia, obesity, and metabolic syndrome in Brazilian older adults. Front Med. 2023;10:1206545.
24. Lieb W, Gona P, Larson MG, et al. The natural history of left ventricular geometry in the community: clinical correlates and prognostic significance of change in LV geometric pattern. JACC Cardiovasc Imaging. 2014;7:870-8.
25. Gao Y, Ren Y, Guo YK, et al. Metabolic syndrome and myocardium steatosis in subclinical type 2 diabetes mellitus: a 1H-magnetic resonance spectroscopy study. Cardiovasc Diabetol. 2020;19:70.
26. Levelt E, Mahmod M, Piechnik SK, et al. Relationship between left ventricular structural and metabolic remodeling in type 2 diabetes. Diabetes. 2016;65:44-52.
27. Hirose K, Nakanishi K, Daimon M, et al. Impact of insulin resistance on subclinical left ventricular dysfunction in normal weight and overweight/obese japanese subjects in a general community. Cardiovasc Diabetol. 2021;20:22.
28. Deng J. Research progress on the molecular mechanism of coronary microvascular endothelial cell dysfunction. Int J Cardiol Heart Vasc. 2021;34:100777.
29. Bugger H, Byrne NJ, Abel ED. Animal models of dysregulated cardiac metabolism. Circ Res. 2022;130:1965-93.
30. Cao Y, Wang Y, Zhou Z, et al. Liver-heart cross-talk mediated by coagulation factor XI protects against heart failure. Science. 2022;377:1399-406.
31. Zhou XD, Targher G, Byrne CD, et al. An international multidisciplinary consensus statement on MAFLD and the risk of CVD. Hepatol Int. 2023;17:773-91.
32. Cheng PN, Chen WJ, Hou CJ, et al. Taiwan Association for the Study of the Liver-Taiwan Society of Cardiology Taiwan position statement for the management of metabolic dysfunction- associated fatty liver disease and cardiovascular diseases. Clin Mol Hepatol. 2024;30:16-36.
33. Zhou XD, Cai J, Targher G, et al; CHESS-MAFLD consortium. Metabolic dysfunction-associated fatty liver disease and implications for cardiovascular risk and disease prevention. Cardiovasc Diabetol. 2022;21:270.
34. Yang K, Song M. New insights into the pathogenesis of metabolic-associated fatty liver disease (MAFLD): gut-liver-heart crosstalk. Nutrients. 2023;15:3970.
35. Sonaglioni A, Cerini F, Fagiani V, et al. Effect of metabolic dysfunction-associated steatotic liver disease (MASLD) on left ventricular mechanics in patients without overt cardiac disease: a systematic review and meta-analysis. J Clin Med. 2025;14:2690.
36. Zheng H, Sechi LA, Navarese EP, Casu G, Vidili G. Metabolic dysfunction-associated steatotic liver disease and cardiovascular risk: a comprehensive review. Cardiovasc Diabetol. 2024;23:346.
37. Chen Y, Cheng X, Li S, Yin Y, Xing S, Guo Y. Insulin-like growth factor-1 levels are associated with interventricular septal thickening. Front Endocrinol. 2022;13:997023.
38. Zhao Z, Gao Y, Lin J, et al. Association of depression with long-term cardiovascular risks in older patients with obstructive sleep apnea. Nat Sci Sleep. 2023;15:1033-43.
39. Li Y, He Q, Chen S, et al. BI-7273, a BRD9 inhibitor, reduces lipid accumulation by downregulating the AKT/mTOR/SREBP1 signaling pathway. Biochem Pharmacol. 2024;226:116412.
40. Cui X, Wang J, Zhang Y, Wei J, Wang Y. Plin5, a new target in diabetic cardiomyopathy. Oxid Med Cell Longev. 2022;2022:2122856.
41. Kianu Phanzu B, Nkodila Natuhoyila A, Kintoki Vita E, M’Buyamba Kabangu JR, Longo-Mbenza B. Association between insulin resistance and left ventricular hypertrophy in asymptomatic, black, sub-Saharan African, hypertensive patients: a case-control study. BMC Cardiovasc Disord. 2021;21:1.
42. Catena C, Brosolo G, Da Porto A, et al. Association of non-alcoholic fatty liver disease with left ventricular changes in treatment-naive patients with uncomplicated hypertension. Front Cardiovasc Med. 2022;9:1030968.
43. Kay FU, Abbara S, Joshi PH, Garg S, Khera A, Peshock RM. Identification of high-risk left ventricular hypertrophy on calcium scoring cardiac computed tomography scans: validation in the DHS. Circ Cardiovasc Imaging. 2020;13:e009678.
44. Sparapani R, Dabbouseh NM, Gutterman D, et al. Detection of left ventricular hypertrophy using Bayesian additive regression trees: the MESA. J Am Heart Assoc. 2019;8:e009959.
45. Liu CM, Hsieh ME, Hu YF, et al. Artificial intelligence-enabled model for early detection of left ventricular hypertrophy and mortality prediction in young to middle-aged adults. Circ Cardiovasc Qual Outcomes. 2022;15:e008360.
46. Wadden TA, Chao AM, Machineni S, et al. Tirzepatide after intensive lifestyle intervention in adults with overweight or obesity: the SURMOUNT-3 phase 3 trial. Nat Med. 2023;29:2909-18.
47. Jastreboff AM, Aronne LJ, Ahmad NN, et al; SURMOUNT-1 Investigators. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205-16.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].