Pseudoalteromonas spongiae-induced biomineralization on Q235B and 2507 steel for corrosion and biofouling resistance
Abstract
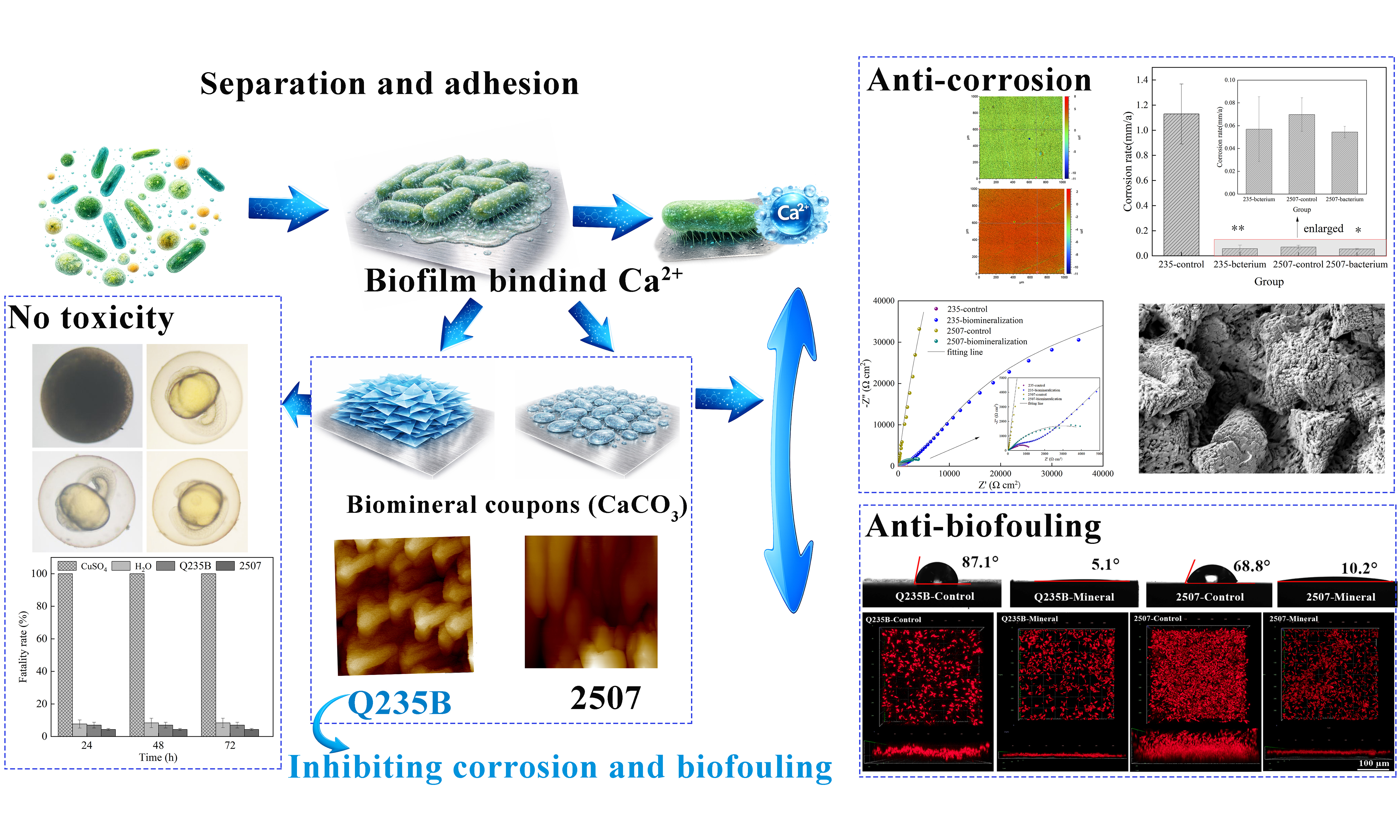
The biomineralization process is initiated when bacteria recruit dissolved mineral ions, leading to the spontaneous formation of biomineral layers on metal surfaces, which can substantially influence the service life of the metal. Herein, we investigated Pseudoalteromonas spongiae (P. spongiae)-induced biomineralization layer formation on Q235B carbon steel and 2507 duplex stainless-steel in bacterial suspension, and we evaluated the anti-corrosion and antifouling performance of mature biomineralized layers in artificial seawater (ASW). On Q235B, the higher surface reactivity promoted a uniform, dark-gray, superhydrophilic biomineralized layer with minor iron oxides. This layer increased interfacial (charge-transfer) resistance, suppressed corrosion, and reduced the attachment of Phaeodactylum tricornutum. On 2507, the native passive barrier limited nucleation, producing Ca–Mg carbonate deposits with locally exposed metal and faster charge transfer. Nonetheless, the hydrophilic protective layer still reduced biofouling in ASW. Overall, P. spongiae–mediated biomineralization was substrate-dependent, exhibiting dual anti-corrosion and antifouling capacity on carbon steel. On 2507, the heterogeneous biomineralized layer, while reducing biofouling, locally compromised the integrity of the passive film, resulting in pitting corrosion.
Keywords
INTRODUCTION
Metals used in offshore equipment operate continuously in high-salinity seawater and are prone to chemical and electrochemical attacks, which degrade performance and shorten service life. In particular, chloride ions (Cl-) promote localized corrosion that can perforate the metallic materials of ships, platforms, and subsea pipelines. Common marine alloys - such as copper, carbon steel, and high-manganese steels - are all susceptible to corrosion in seawater, resulting in major economic losses and safety risks[1-3]. Meanwhile, coastal eutrophication caused by industrial activities promotes bacterial and algal blooms, whose metabolites promote the formation of complex biofilms. Microorganisms, algae, and barnacles co-exist and stabilize these communities (marine biofouling)[4]. Biofilms establish oxygen gradients at the metal–water interface that accelerate electrochemical corrosion[5] and increase hydrodynamic drag (thereby raising fuel consumption and CO2 emissions)[6]. In addition, some organisms within biofilms can damage protective coatings via surface attacks or delamination[7]. Q235B carbon steel and 2507 duplex stainless steel are still widely used in marine applications, but both still corrode under harsh conditions[8,9], and microbial metabolites within biofilms can further initiate localized corrosion on Q235B and 2507 surfaces[10,11]. Historically, tributyltin (TBT) coatings provided strong biofouling resistance, but their ecological and human toxicity in marine environments were unacceptable, highlighting the urgent need for environmentally friendly strategies[12]. In recent years, some marine microorganisms have been shown to play dual roles in mitigating corrosion and biofouling by secreting extracellular polymeric substances (EPS) that chelate Ca2+/Mg2+ and promote protective biomineralization on metal surfaces. These biomineralized layers offered a promising green route to simultaneously inhibit corrosion and prevent biofouling.
Biomineralization proceeds via two principal pathways: biologically controlled mineralization (BCM) and biologically induced mineralization (BIM). BIM refers to the process of mineral formation driven by microbial alteration of local environmental conditions, such as pH, carbonate alkalinity, and redox[13]; it is the dominant pathway for forming biomineral layers on metal surfaces. Several microbes can produce uniform, adherent, and environmentally benign biomineral coatings that conform to complex surfaces and serve as protective barriers from corrosion. Previous studies support this conclusion: for example, Guo et al. showed that Pseudoalteromonas lipolytica (P. lipolytica) secreted bacterial cellulose polysaccharides that induced calcite precipitation and protected steel substrates[14], with the cellulose polysaccharides regulating the biomineralization process via biofilm mediation[15]. Guo et al. reported that molybdenum increased the adhesion of Bacillus subtilis (B. subtilis) to material surfaces and improved biomineralization[16], while earlier work indicated that biomineral layers induced by B. subtilis inhibited Vibrio natriegens-induced corrosion in seawater[17]. Similarly, methanogens-driven carbonate deposition on Dutch metal dikes produced dense biomineral layers that prevented corrosion for about 50 years[18]. Notably, Shewanella putrefaciens has been used to restore damaged epoxy coatings and to suppress corrosion on carbon steel[19]. Overall, extensive studies have confirmed that biomineral coatings can significantly mitigate metal corrosion[20,21]. The corrosion protection mainly arose from (i) microbial metabolic activity that consumes dissolved oxygen at the biofilm/metal interface, counteracting oxygen gradients that drive electrochemical corrosion; (ii) physical barriers that effectively limit direct contact between corrosive agents and substrates[22]. Conventional corrosion control and prevention strategies include sacrificial anodes, cathodic protection systems, and organic/inorganic chemical coatings[23-25]. However, these methods still present limitations, including finite service life[26], uneven potential control, and environmental concerns. Moreover, biofouling still remains a pressing challenge in marine environments and, in combination with bacteria, can accelerate metallic corrosion[27,28]. Therefore, it is particularly important to develop coatings that simultaneously inhibit corrosion and biofouling. Biomineralization offers a promising sustainable route to develop protective coatings using marine bacteria such as Alteromonas, Pseudoalteromonas, and Pseudomonas, which exhibit high mineralization capacity, making them strong candidates for green, durable anti-corrosion and antibiofouling strategies[29-31]. Most previous studies on bacteria-induced biomineralization have mainly focused on corrosion mitigation, whereas substrate-dependent film nucleation, growth, and coverage have received less attention. In particular, for carbon steel vs. duplex stainless steel, the roles of substrate reactivity and passive films in governing film continuity and protection remain understudied.
Pseudoalteromonas is a genus of Gram-negative bacteria widely distributed in seawater, with remarkable metabolic capabilities and strong environmental adaptability[32]. Species of Pseudoalteromonas have become model organisms in biofilm and biomineralization research because they secrete abundant EPS during growth[33,34]. Their metabolic byproducts can modulate the local environment, particularly pH and ion concentrations, thereby promoting the formation of continuous and dense biomineralized layers on metal surfaces[35,36]. In this study, Pseudoalteromonas spongiae (P. spongiae) was isolated from the coastal waters near Guishan Island in the South China Sea, at approximately 22 °C and 30 ‰ salinity. We then investigated P. spongiae-induced biomineralization on Q235B carbon steel and 2507 duplex stainless steel, with particular focus on the corrosion resistance and antifouling performance of the resulting biomineralized layers in artificial seawater (ASW). This work aims to clarify the substrate-dependent characteristics of biomineral layer nucleation and growth and to evaluate the potential of this biomineralization strategy for marine metal protection.
MATERIALS AND METHODS
Bacterial culture and steel
The bacterial strain used in this study was isolated from seawater collected near Guishan Island (South China Sea). After purification and taxonomic identification, 16S ribosomal RNA (rRNA) gene-based phylogenetic analysis revealed that the isolate shared the highest sequence similarity with P. spongiae UST010723-006. Cells were grown in 2216 E broth (Hopebio, Qingdao, China), prepared by dissolving 37.4 g·L-1 of dehydrated powder in deionized (DI) water and autoclaving at 121 °C for 20 min. After cooling to room temperature, sterile NaHCO3 [ultraviolet (UV)-sterilized] was added to achieve a final concentration of
The Q235B carbon steel used in this study had the following chemical composition (wt.%): 0.7 Mn, 0.35 Si, 0.045 P, 0.045 S, 0.22 C, with the balance being Fe. The chemical composition of the 2507 duplex stainless steel was (wt.%): 24.0-26.0 Cr, 6.0-8.0 Ni, 3.0-5.0 Mo, 0.2-0.3 N, 1.2 Mn, 0.8 Si, 0.035 P, 0.02 S, 0.03 C, with the balance being Fe. Both alloys were sourced from Shenzhen Dinglong Industrial Materials Co., Ltd. Both steels were cut into rectangular coupons (10 mm × 10 mm × 5 mm), sequentially ground with SiC papers (120-1,000 grit), ultrasonically cleaned in anhydrous ethanol to remove any surface contaminants, air-dried, and sterilized under UV irradiation for 30 min. UV-sterilized Q235B and 2507 coupons were immersed in either bacterial suspensions or sterile medium in subsequent experiments. The pH of bacterial suspensions was monitored daily for 8 days using a pH meter (Sartorius PB-10, Germany). Viable bacteria were enumerated by serial dilution and spot-plating on nutrient agar plates, with triplicate plates for each group.
Properties of the surface mineral layer on Q235B and 2507
During immersion of Q235B and 2507 coupons in bacterial suspensions, samples were retrieved after 3, 5, and 7 days, gently rinsed with DI water, air-dried, and photographed to document the morphology of the biomineralized layer. The phase composition of the samples immersed for 7 days was analyzed by X-ray diffraction (XRD; SmartLab, Japan) over a 2θ range of 10°-80° at a scanning rate of 5° min-1, and datasets were processed with Jade software. In a separate set of samples, coupons immersed for 7 days were rinsed three times with DI water, dried at 40 °C for 24 h, and then subjected to static water contact-angle measurements to evaluate the hydrophilicity of the biomineralized layer. Polished (1,000-grit SiC) Q235B and 2507 without immersion served as blank controls, and all measurements were performed in triplicate.
Microscopic morphology of both substrates
Samples immersed for 7 days were fixed in 2.5% glutaraldehyde for 15 min, then dehydrated in graded ethanol (30, 40, 50, 60, 70, 80, 90, and 100 vol%; 10 min per step). After air-drying at room temperature, coupons were sputter-coated with Pt using a vacuum coater (nanoPVD S10A, UK; 30 mA, 60 s). The microstructure of the biomineralized layer was examined by focused ion beam scanning electron microscopy (FIB–SEM; Crossbeam 350, Germany), and the cross-sectional morphologies of the biomineralized layer were obtained by FIB milling. Elemental composition and spatial distribution on coupons were analyzed by energy-dispersive X-ray spectroscopy (EDS; ZEISS SmartEDX, Germany).
Corrosion behavior during biomineralization formation
Samples were welded to copper wires and embedded in epoxy resin, leaving a defined working area exposed (1 cm2). Coupons were immersed in bacterial suspensions with the initial inoculum (1 mL, OD600 = 0.1), while sterile medium served as the control group. In situ electrochemical impedance spectroscopy (EIS) was performed using a three-electrode system, with the experimental sample as working electrode (WE), saturated calomel electrode (SCE) as the reference electrode (RE), and a platinum plate as the counter electrode (CE). Measurements were carried out on an electrochemical workstation (Parstat IM6e, Princeton Applied Research, USA). A 10 mV (rms) sinusoidal perturbation was applied at the open-circuit potential (OCP) after stabilization (300 s), over a frequency range of 10-2-105 Hz. EIS spectra were collected after 3 and 7 days of immersion. For the mature biomineralized layer, EIS was additionally measured after 7 days of immersion under ambient laboratory conditions. The EIS data was fitted and analyzed using ZSimpWin.
The corrosion rate of the samples was calculated using:
where Vcorr, t, ρ, A and Δm represented the corrosion rate (mm·a-1), t is the immersion time (h), ρ is the steel density (g·cm-3), A is the exposed area (cm2), and Δm is the mass loss (g), respectively. Steel samples were immersed in various bacterial suspensions for 7 days at 22 °C (initial inoculum as above). After immersion, samples were sequentially rinsed with concentrated hydrochloric acid, saturated NaHCO3 (China National Medicines Corporation Ltd.), and DI water, then air-dried. Surface topography was examined with a white-light interferometer (ContourGT, Bruker, Germany) to assess pitting corrosion morphology.
Biofouling influence of mature biomineralized layer
The mature biomineralized layer was obtained by immersing coupons in bacterial suspension for 7 days. Adhesion assays were performed in 24-well polypropylene plates (Corning, USA). Bacterial suspension
To evaluate the capacity of mature biomineralized layers to inhibit biofilm formation, Q235B and 2507 coupons with biomineralized layers were subsequently immersed in Vibrio chagasii (V. chagasii) suspension for 7 days. A staining solution was prepared by mixing 3 µL SYTO 9 Green fluorescent nucleic acid stain and 3 µL propidium iodide (PI) in 1 mL phosphate-buffered saline (PBS). Steel coupons were removed from the bacterial suspension, placed in a sterile Petri dish, and gently rinsed three times with sterile PBS to remove non-adherent bacteria. Subsequently, 200 µL of staining solution was added and samples were incubated at room temperature in the dark for 20 min. Stained samples were rinsed with sterile PBS to remove excess dye, air-dried in the dark, and imaged with a confocal laser scanning microscope (CLSM 900, ZEISS, Germany) to assess biofilm viability and structure.
Diatom attachment is a common initial step in marine biofouling development. In this work, “antibiofouling in marine” refers to inhibition of microfouling, mainly bacterial biofilm formation and early-stage diatom attachment. To simulate algal attachment under marine conditions, ASW (ASW composition in Table 1) was prepared, and supplemented with Phaeodactylum tricornutum (P. tricornutum, 4.1 × 106 cells/mL) and
Electrochemical impedance parameters fitted from the Nyquist plots [Figure 5]
| Steel | Groups | R s (Ω·cm2) | Y f (S.sn/cm2) | n | R f (Ω·cm2) | Y dl (S.sn/cm2) | n | R ct (Ω·cm2) |
| Q235B | Control, 3 days | 10.7 | 9.4 × 10-5 | 0.7 | 6.6 | 5.2 × 10-4 | 0.7 | 5,248 |
| P. spongiae, 3 days | 7.8 | 1.0 × 10-4 | 0.9 | 1.2 × 104 | 3.2 × 10-5 | 0.9 | 8,446 | |
| Control, 7 days | 9.3 | 1.3 × 10-1 | 0.7 | 9.4 | 2.1 × 10-3 | 0.8 | 1,601 | |
| P. spongiae, 7 days | 8.0 | 3.0 × 10-5 | 0.9 | 16.7 | 4.7 × 10-5 | 0.9 | 8.6 × 104 | |
| 2507 | Control, 3 days | 10.5 | / | / | / | 1.1 × 10-5 | 0.9 | 3.3 × 106 |
| P. spongiae, 3 days | 7.3 | / | / | / | 1.1 × 10-4 | 0.9 | 1,231 | |
| Control, 7 days | 6.9 | / | / | / | 1.9 × 10-5 | 0.9 | 1.9 × 107 | |
| P. spongiae, 7 days | 7.3 | / | / | / | 2.1 × 10-4 | 0.8 | 4.5 × 104 |
Corrosion resistance of mature biomineral layer
Q235B and 2507 coupons embedded in epoxy resin were immersed in bacterial suspensions for 7 days to form a mature biomineralized layer. Following retrieval, superficial biofilm was removed using 75% ethanol; samples were air-dried and then immersed for 3 days in ASW, which contained NaCl 23.50, MgCl2·6H2O 10.78, Na2SO4 3.92, CaCl2·2H2O 1.47, KCl 0.66, NaHCO3 0.20, H3BO3 0.10, SrCl2·6H2O 0.025, NaF 0.003. EIS was performed as in Section “Corrosion behavior during biomineralization formation”, except the electrolyte was replaced with ASW.
Zebrafish embryo toxicity of biomineral coupons.
Ten biomineralized Q235B coupons (10 mm × 10 mm × 5 mm) with adherent bacteria removed were gently rinsed with up to 500 mL of sterile DI water. In parallel, ten 2507 duplex stainless-steel coupons (10 mm ×
Statistical analysis
Data analysis was conducted using SPSS 19.0 (IBM SPSS Statistics). One-way analysis of variance (ANOVA) was applied to determine differences between experimental groups. Results are reported as the mean ± standard deviation (SD). Statistical significance thresholds were set at *P < 0.05, **P < 0.01, and ***P < 0.005.
RESULTS AND DISCUSSION
Growth and metabolic conditions of bacteria
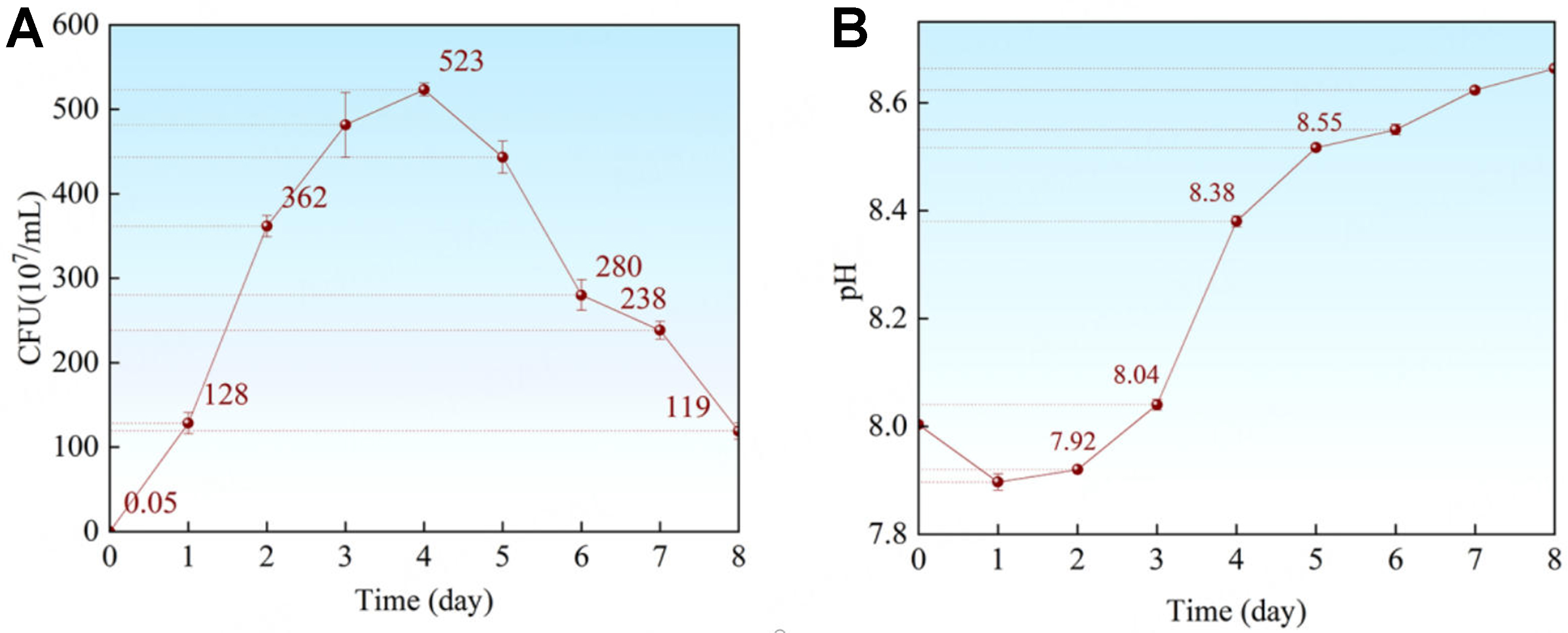
The 8-day growth trajectory of P. spongiae is shown in Figure 1A. During the first 48 h, the results exhibited an exponential phase, with bacterial concentration increasing rapidly from ~ 5.0 × 105 to ~ 2.5 × 109 cells/mL. The accompanying pH changes of the bacterial suspension during this period are presented in Figure 1B. As an aerobic bacterium, P. spongiae caused pH to decrease slightly during rapid proliferation, consistent with the production of acidic metabolites. In agreement with this result, Kim et al. reported that EPS secreted by P. elyakovii isolated from the Arctic Ocean was enriched in –COOH groups that ionize to release H+ and acidify the medium[37].
Figure 1. (A) Time dependence growth curve of bacteria and (B) the pH curve during 8 days. Error bars represent standard deviation (SD). CFU: Colony forming unit.
Additionally, Respiratory CO2 may further contribute to this modest pH decline. Subsequently, P. spongiae entered the stationary phase when bacterial proliferation slowed significantly (2‐4 days), accompanied by the accumulation of metabolic products. The pH rose from ~7.92 to ~8.55, representing the steepest pH ascent observed [Figure 1B]. After 4 days, cell death commenced gradually; nevertheless, bacterial concentrations remained above 109 cells/mL throughout the period from 2 to 7 days. Overall, the pH of bacterial suspension increased steadily, but the rate slowed markedly after 5 days [Figure 1B]. Taken together, these results indicated that at 22 °C in a 200 mL nutrient-rich medium, P. spongiae completed a full growth cycle within approximately one week; accordingly, the 7-day experimental period was adopted for this study.
The surface characteristics of the biomineralized layer
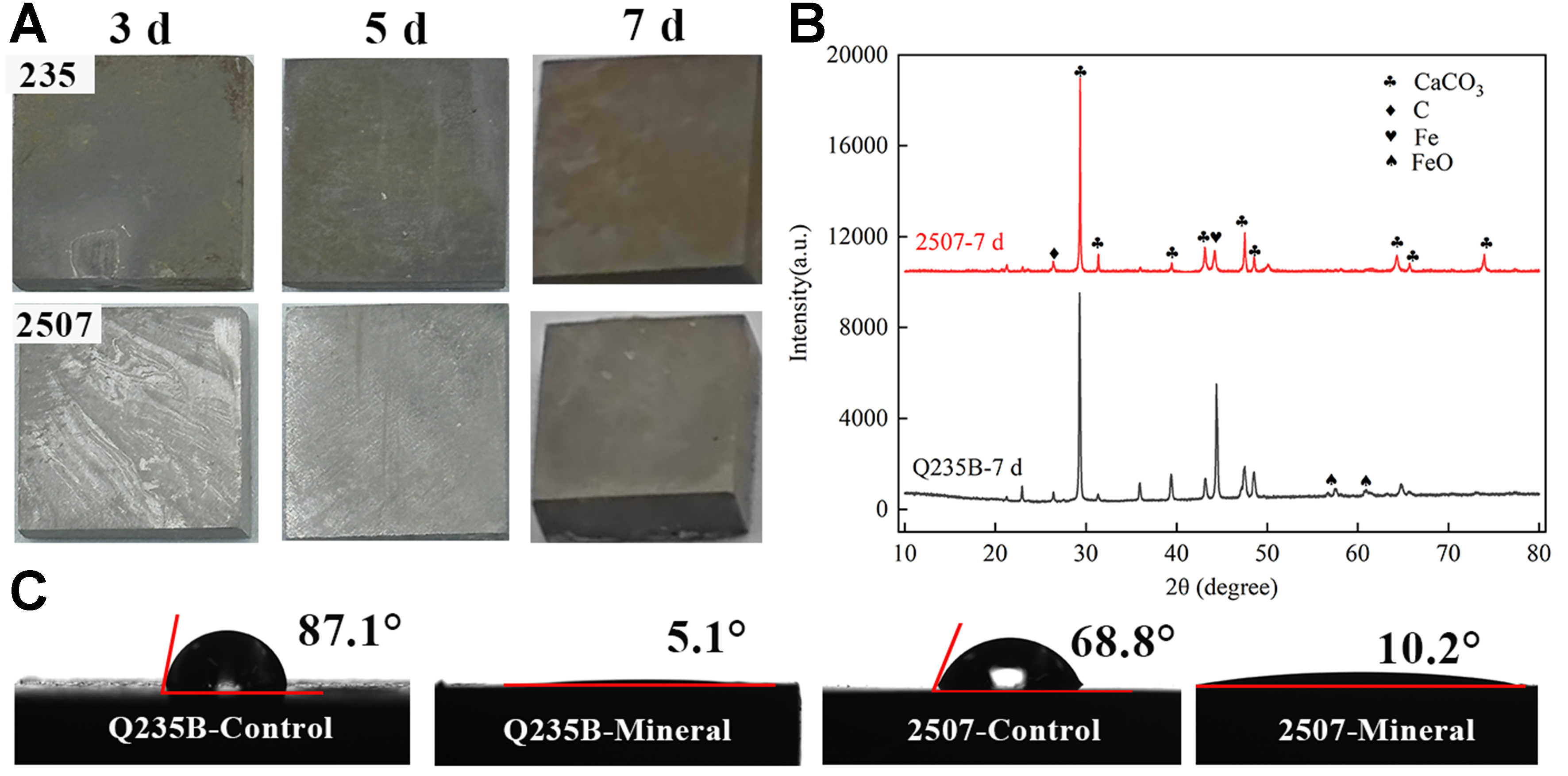
During biomineral layer formation, the metal surface progressively lost its metallic luster and developed a matte coating. The macroscopic appearance of coupons immersed in the bacterial suspension is shown in Figure 2A. P. spongiae produced biomineralized layers with distinct coloration on Q235B and 2507. A dark gray layer formed on Q235B, whereas a comparatively lighter layer formed on 2507, with the contrast most pronounced on day 5. Compositional analysis revealed that calcium carbonate was the predominant constituent of the biomineralized layers on both substrates [Figure 2B]. However, the coating on Q235B also contained minor amounts of iron oxides, indicating that partial dissolution and Fe2+/Fe3+ release from the carbon steel occurred during the biomineralization reaction, thereby imparting the darker product. Some studies have reported that mild substrate corrosion, by elevating Fe2+ concentrations, can enhance bacteria-induced carbonate deposition and promote biomineral layer formation, thus possibly providing nucleation sites of biomineralization[22].
Figure 2. (A) Macro images of Q235B and 2507 during 7 days; (B) the XRD image and (C) contact angle images of Q235B and 2507 after 7 days. XRD: X-ray diffraction.
Figure 2C shows the hydrophilicity of bare substrates and their biomineralized coatings. The static contact angles of bare Q235B and 2507 were ~87.1° and ~68.8°, respectively, consistent with moderately hydrophilic surfaces. In contrast, the contact angles of biomineralized layers decreased to ~5.1° and ~10.2°, imparting the coatings strong hydrophilicity. Unlike conventional corrosion-inhibiting strategies that emphasize superhydrophobicity to minimize the contact between water and substrate[38,39], a superhydrophilic interface could sustain a continuous water layer that impedes oxygen and other corrosive species and reduces biofouling in ASW. Notably, the biomineralized layer on Q235B exhibited stronger wetting ability than that on 2507, plausibly due to a more uniform hydrophilic mineral phase.
Structure of the biomineralized layer
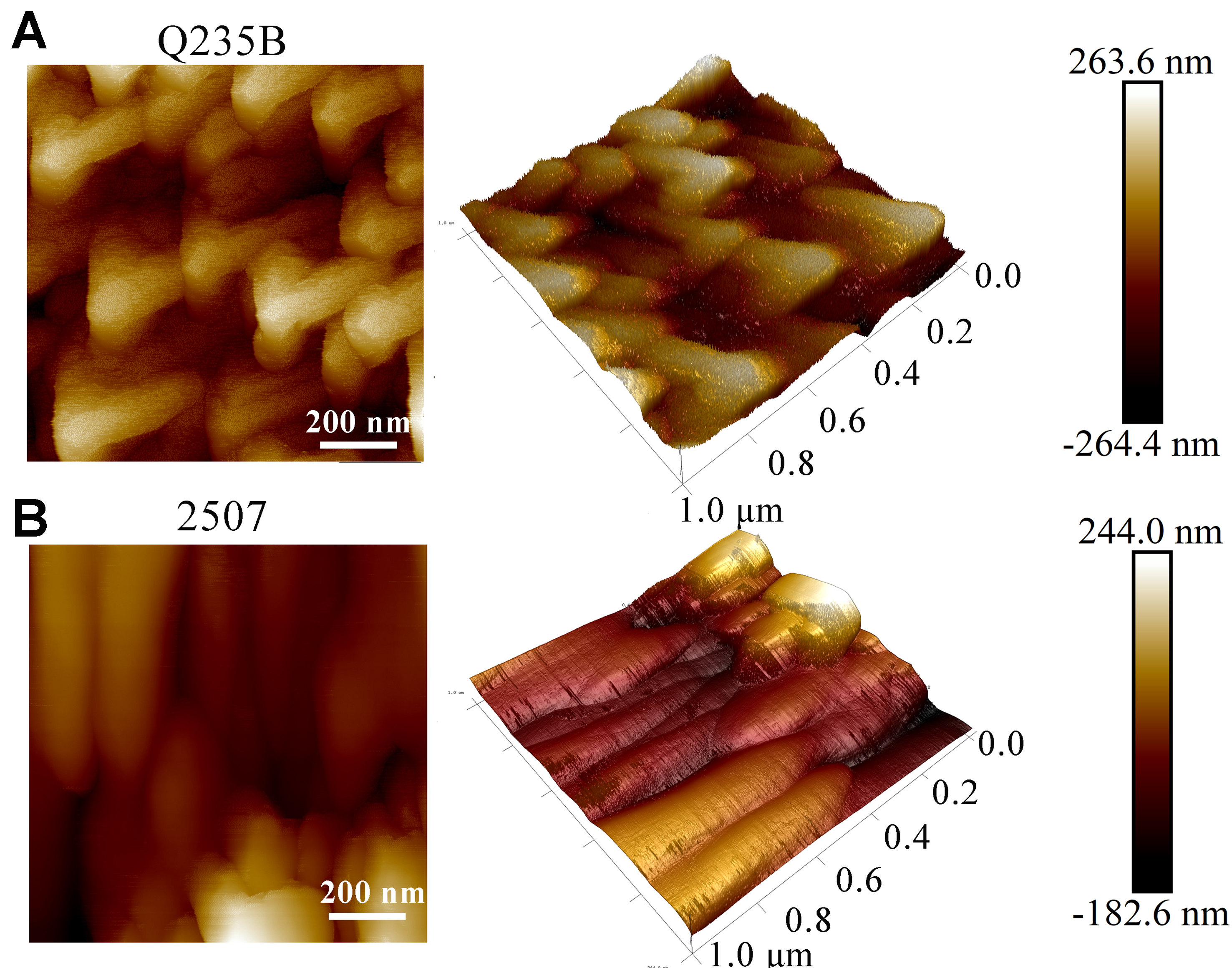
Figure 3A and B show SEM images of the microscopic morphology of biomineralized layers formed on Q235B and 2507 after 7 days of immersion in P. spongiae suspension, respectively. On Q235B, the deposit was dominated by clumps and other irregular accretions that formed a continuous blanket over the substrate. Higher-magnification micrographs resolved stacked mineral domains with no discernible exposure of the underlying steel. Surface EDS identified C, O, and Ca as the major elements, minor Fe uniformly distributed, consistent with the XRD results. Cross-sectional images further confirmed a relatively uniform, compact biomineralized coating that was strongly bonded to the substrate, with neither exposed metal nor internal voids [Figure 3C]. Atomic force microscopy (AFM) images reveal the biomineralized architectures formed on the two substrates at higher spatial resolution in Figure 4A. The surface layer on Q235B consisted of orderly triangular motifs ~350-400 nm in size arranged in a stacked pattern. The mean topographic relief was ~580.2 nm, exceeding that of the layer on the 2507 surface
Figure 3. The SEM images of (A) Q235B and (B) 2507 with corresponding EDS images immersed in bacterial solution after 7 days; The cross-sectional SEM images with calcium element distribution of (C) Q235B and (D) 2507 after immersion of 7 days. SEM: Scanning electron microscopy; EDS: energy-dispersive X-ray spectroscopy.
Figure 4. AFM images of (A) Q235B and (B) 2507 immersed in bacterial suspension after 7 days. AFM: Atomic force microscopy.
In contrast, the biomineralized layer on 2507 was spatially heterogeneous, leaving portions of the substrate uncovered. The prevalent morphology was rod-like, and at higher magnification, the coating comprised small triangular subunits and contained pores. EDS indicated a composition similar to that on Q235B (C, O, Ca) but with localized Fe enrichment, indicating that portions of the substrate matrix were directly exposed, which may have induced electrochemical corrosion. FIB–SEM cross-sections highlighted two salient features of the 2507 layer: (1) an externally discontinuous CaCO3 overlayer that exposes isolated regions of the steel; and (2) a highly porous interior with a honeycomb-like architecture [Figure 3D]. The nanoscale images indicated that the surface structure of 2507 was disorganized and lacked sufficient barrier efficacy toward corrosive environments [Figure 4B]. In conclusion, P. spongiae formed biomineralized layers of distinct structure on the two substrates, resulting in different levels of corrosion inhibition, consistent with findings on the corrosion inhibition behavior of X80 pipeline steel[40].
Corrosion behavior of the substrate
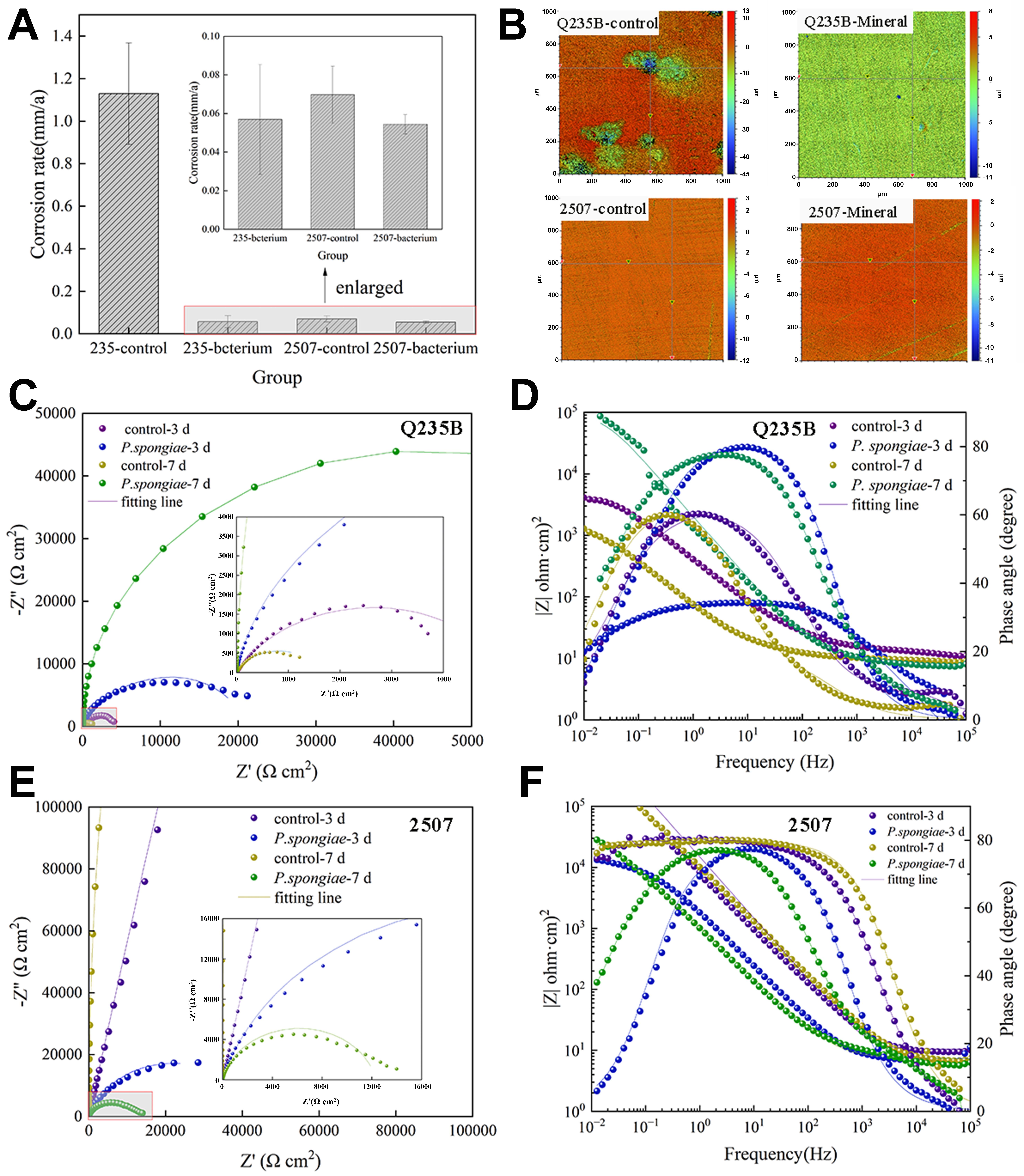
Carbon steel is one of the most widely used engineering materials, yet it is prone to corrosion in seawater and atmospheric environments[41,42]. The biomineralization formation process on Q235B provided immediate anticorrosion capacity. Figure 5A shows the uniform corrosion rates of Q235B and 2507 immersed in bacterial suspensions after 7 days. For Q235B, the biomineralized layer reduced the uniform corrosion rate from
Figure 5. (A) The uniform corrosion rate of 235 and 2507 steels. Error bars represent standard deviation (SD); (B) Heat map of the localized corrosion of Q235B and 2507 after 7 days immersion; (C) Nyquist plot and (D) Bode plot of Q235B immersed in bacterial suspension for 7 days; (E) Nyquist plot and (F) Bode plot of 2507 immersed in bacterial suspension during 7 days (error bars represent the variance of the three repeating groups. The statistical significance is *P < 0.05, **P < 0.01).
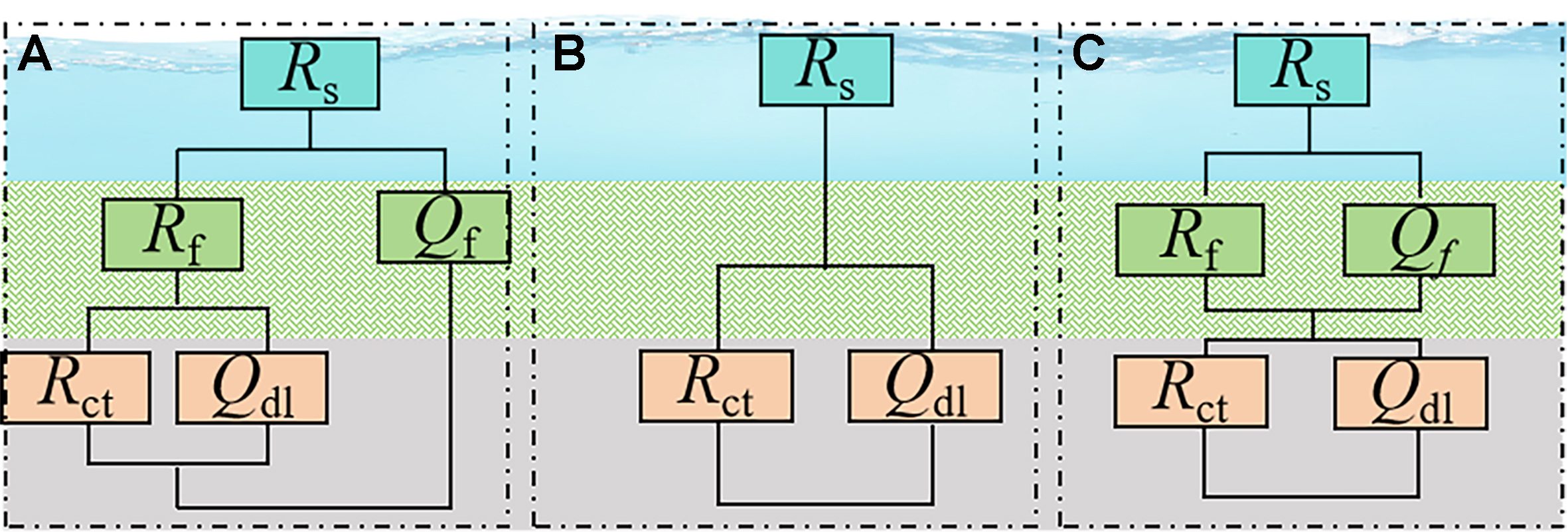
A relatively uniform biomineralized layer formed by P. spongiae on Q235B yielded an impedance response approaching ideal capacitive behavior, thereby inhibiting substrate corrosion. The Q235B and 2507 Nyquist and Bode plots [Figure 5C-F] were described by equivalent circuits of the form Rs(RfQf(RctQdl)) and Rs(RctQdl) [Figure 6A and B]. The fitting results showed a higher film capacitance than the sterile control, a higher mid-frequency phase angle during 3-7 days, and a substantial increase in film resistance (Rf). These features indicate progressive thickening of the biomineral layer and enhanced corrosion resistance of Q235B. By contrast, on 2507 duplex stainless steel, the biomineral layer locally degraded the passive film, lowering impedance and accelerating both charge transfer and corrosion. In the sterile control, the peak of the EIS curve extended to low frequencies. However, in the bacterial suspension, the phase angle collapsed into a single peak centered in the mid-frequency region after the formation of the biomineral layer [Figure 5E and F]. The peak narrowed and the low-frequency phase angle dropped sharply, indicating faster charge transfer kinetics and partial disruption of the passive film by a heterogeneous biomineral layer.
Corrosion and biofouling resistance of mature biomineralized layers
Biofouling not only imposes additional economic costs but it is also linked with adhesion-induced corrosion of materials[46,47]. To further evaluate the biofilm-inhibiting capacity of the biomineral layer induced by
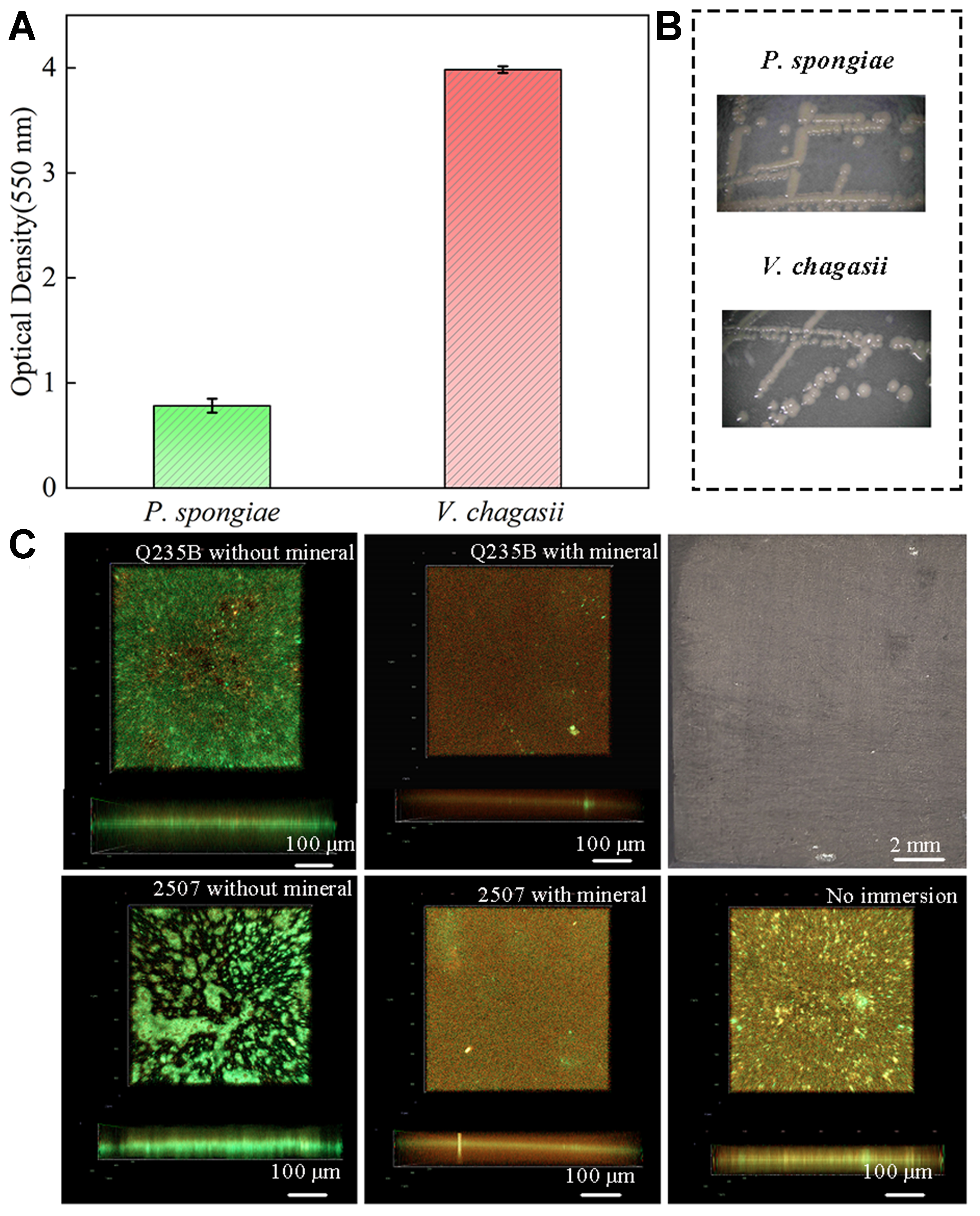
Figure 7. (A and B) The adhesive capacity and morphology of P. spongiae and V. chagasii. Error bars represent standard deviation (SD); (C) The biofilm of V. chagasii on Q235B, 2507 without and with biomineral after 7 days immersion, the macroscopic view of the biomineralized layer is in the upper right corner.
To more faithfully simulate marine biofouling, we employed V. chagasii and P. tricornutum as the model bacterium and alga, respectively, to assess the biofouling-inhibiting capacity of biomineralization in ASW alongside a sterile control. Figure 8A showed the macroscopic appearance of bare coupons and coupons covered with the mineral layer after immersion in ASW containing both organisms. Bare Q235B corroded severely and formed a yellow rust layer; nevertheless, Q235B with a dark gray biomineral coating showed strong corrosion-inhibiting capacity, and no visible corrosion products. CLSM images indicated that the biomineral layer on Q235B also reduced diatom attachment; the attached diatom film was thinner than that on bare steel [Figure 8B]. There were no obvious corrosion products on bare 2507, but it was covered by numerous diatoms. This result suggested that stainless steel is more vulnerable to biofouling than carbon steel under these conditions. This is likely because its surface does not release a large amount of heavy metal ions, allowing bacteria and diatoms to continue to grow. Thus, although stainless steel resists corrosion, it can experience heavier biofouling than carbon steel. The super hydrophilic mineral layer greatly protected 2507 against biofouling; diatom attachment was reduced in both thickness and area [Figure 8B]. The biomineral layer with a surface biofilm had a stronger inhibitory effect on diatom attachment, possibly due to interspecific biological competition, which inhibits the colonization of diatoms on the surface [Figure 8C]. Taken together, these results highlight the significant role of the superhydrophilic biomineral layer in metallic anti-corrosion and antibiofouling. It effectively limited corrosion and biofouling on carbon steel, and even a non-uniform layer reduced the attachment of biofouling organisms on stainless steel. However, the uneven biomineral layers can damage the corrosion resistance of the passivation film. Biomineralization still has the potential to become a green and economical method for corrosion and biofouling inhibition with further research.
Figure 8. (A) Macro and (B) micro images of bare Q235B, 2507 and its biomineralization; (C) Biomineral Q235B without and with biofilm immersed in ASW with V. chagasii and P. tricornutum after 3 days. ASW: Artificial seawater; V. chagasii: Vibrio chagasii; P. tricornutum: Phaeodactylum tricornutum.
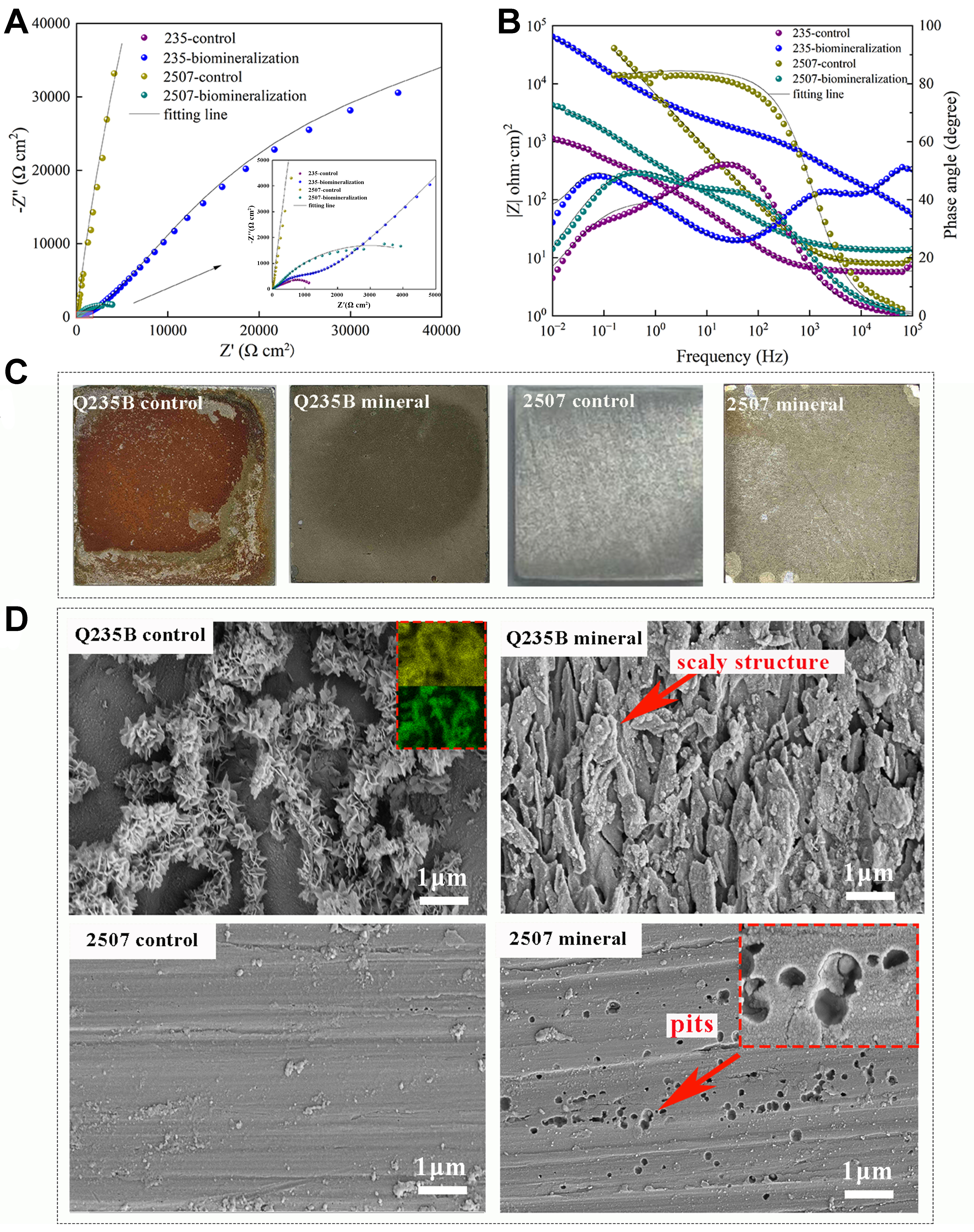
Figure 9 shows the Nyquist and Bode plots of bare steel and steel covered by the mature mineral layer after 3 days immersion in simulated seawater, which were fit using the equivalent circuit Rs(RfQf)(RctQdl) shown in Figure 6C. Q235B with the biomineral layer exhibited a larger capacitive loop and a clear diffusion feature. The film resistance (Rf) was approximately an order of magnitude higher than that of bare Q235B, and the surface charge transfer process was inhibited [Table 2]. The impedance modulus (|Z|) of biomineralized Q235B was also much higher than that of bare steel, indicating strongly suppressed electrochemical reactions. Bare Q235B showed one typical electrochemical process in the mid-frequency range, corresponding to corrosion between substrates and seawater, with an oxide layer formed.
Figure 9. (A) Nyquist plot and (B) Bode plot after 3-day immersion; (C) Macro images and (D) micro morphology of Q235B and 2507 immersed in ASW after 3 days with product removed. ASW: Artificial seawater.
Electrochemical impedance parameters fitted from the Nyquist plots [Figure 9]
| Groups | R s (Ω·cm2) | Y f (S.sn/cm2) | n | R f (Ω·cm2) | Y dl (S.sn/cm2) | n | R ct (Ω·cm2) |
| Q235B control | 5.8 | 2.7 × 10-2 | 0.7 | 1,134.0 | 9.1 × 10-4 | 0.8 | 162.2 |
| Q235B mineralization | 1.0 | 7.2 × 10-5 | 0.7 | 1.5 × 105 | 1.7 × 10-5 | 0.6 | 2,093.0 |
| 2507 control | 8.2 | 3.4 × 10-4 | 0.8 | 100.5 | 2.4 × 10-5 | 0.9 | 2.0 × 105 |
| 2507 mineralization | 13.2 | 7.5 × 10-4 | 0.8 | 5,737.0 | 7.1 × 10-4 | 0.8 | 81.4 |
Pitting corrosion is one of the main causes of duplex stainless-steel failure[51]. The EIS analysis for 2507 showed the opposite behavior: bare 2507 exhibited high impedance with a large capacitive loop and a phase angle of ~80°‐89°, behaving close to a capacitor and thus being less prone to corrosion. The outstanding corrosion resistance of bare 2507 comes from the highly stable Cr2O3 passive film on its surface; the Rct was ~2.0 × 105 Ω·cm2, indicating that the surface hardly reacted with external ions in high-salinity conditions [Table 2]. After the biomineral layer formed, the capacitive loop of 2507 became smaller, and both |Z| and the phase angle dropped sharply, showing the damage caused by locally aggregated minerals to the passive film. The mineral layer induced by P. spongiae on 2507 was thin and contained gaps; nonuniform regions could generate local stresses and allow Cl- to reach the substrate. Unmineralized regions likely acted as anodic sites (electron loss), leading to passive film breakdown.
Figure 9C showed the surface morphology of the bare steel and the protective layer after 3 days immersion in ASW. On the Q235B substrate, vigorous electrochemical activity was evident, leading to the formation of abundant yellow corrosion products that adhered tightly and extensively to the surface. The corrosion products exhibited a characteristic flower-like morphology. By contrast, the biomineral layer effectively suppressed corrosion of Q235B, and no corrosion products were detected. After removing the biomineral layer, the underlying substrate displayed a scaly structure, which was possibly related to early-stage bacterial activity [Figure 9D]. For 2507 stainless steel, the native passive film conferred remarkable corrosion resistance, allowing the substrate to retain metallic luster without visible degradation. Nevertheless, the biomineral layer formed by P. spongiae disrupted the stability of the passive film; the uneven biomineralized layer and accelerated electrochemical reaction resulted in pitting corrosion beneath the non-uniform biomineral coating. These pits were likely caused either by localized film rupture or by electron transfer effects during the biomineral layer formation. Overall, the biomineral layers induced by P. spongiae on different steel substrates exhibited varying efficiencies in mitigating high salinity corrosion, closely correlated with their structural characteristics.
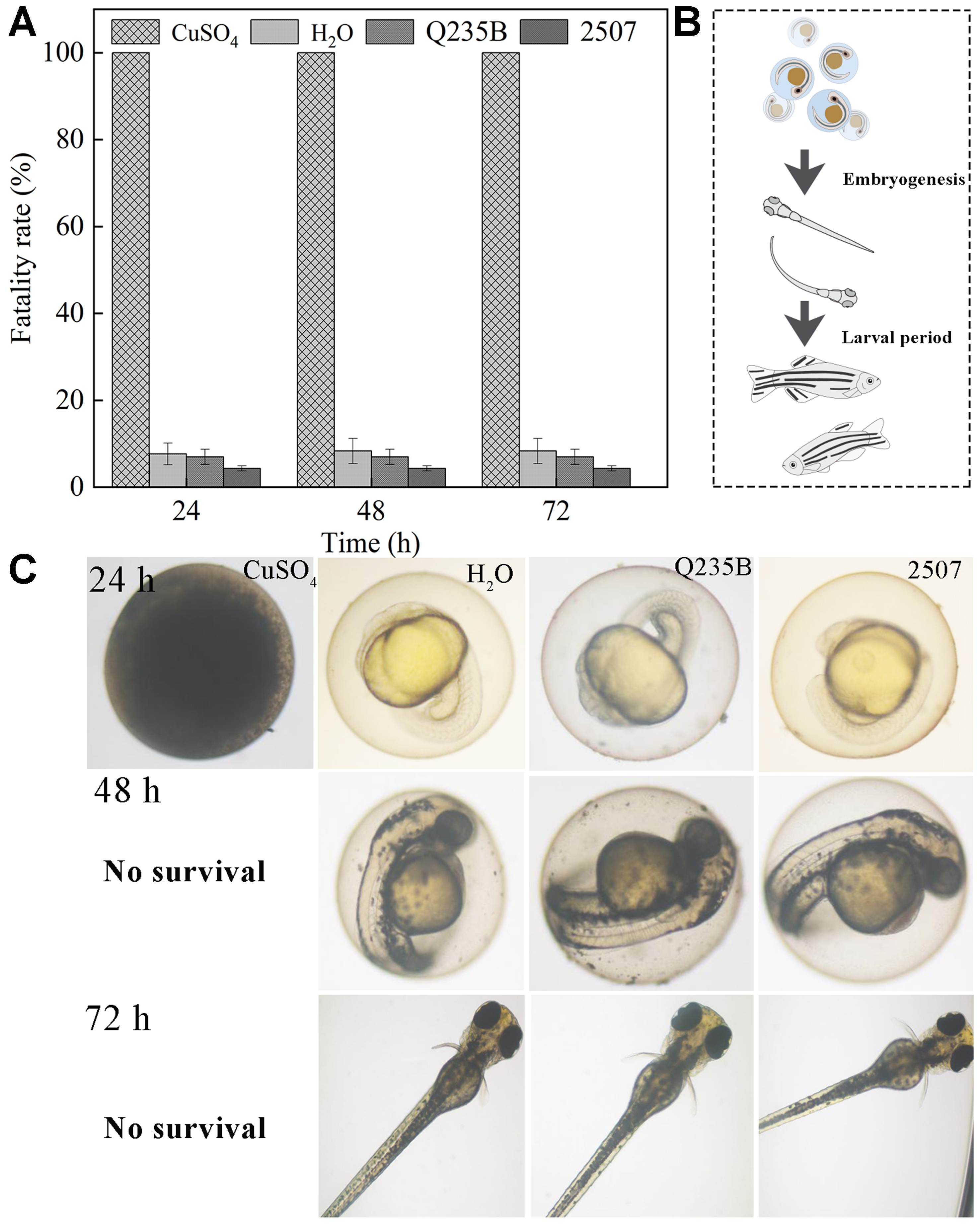
The vast majority of embryos in the blank control group developed and hatched normally, with no obvious mortality, confirming that the experimental conditions were appropriate [Figure 10]. In the positive control group, all embryos died within 24 h, consistent with acute toxicity induced by elevated Cu2+, and no viable larvae were obtained, thereby validating the sensitivity of the assay. By contrast, embryos exposed to biomineral leachates maintained high survival throughout the 3-day observation period, the survival rate at each time point remained at approximately 94%-97%, and most embryos successfully hatched into active larvae [Figure 10]. These results indicated that the biomineral coupons caused negligible adverse effects on early zebrafish embryos and demonstrated favorable ecological safety under the tested conditions. Collectively, the findings suggested that the biomineralized layer can reduce surface reactivity while maintaining environmental compatibility, supporting its potential application as an eco-friendly protective strategy for marine engineering.
Figure 10. (A) Zebrafish survival rate with biomineral coupons. Error bars represent standard deviation (SD); (B) Theoretical growth process of zebrafish; (C) Actual growth of zebrafish with biomineral coupons (The statistical significance is *P < 0.05, **P < 0.01).
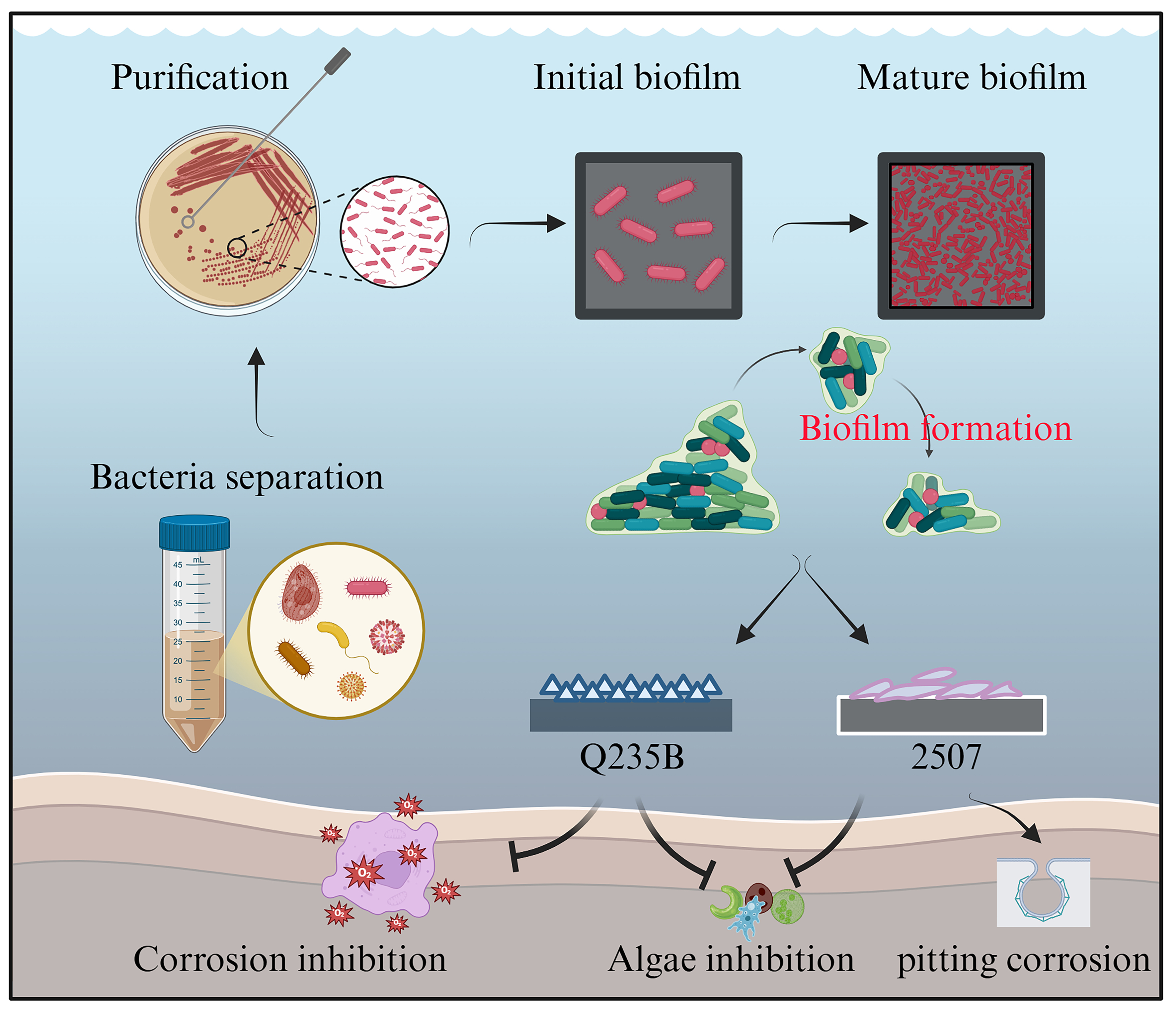
Figure 11 showed the formation process of biomineralization layers induced by P. spongiae. The biomineral layer on Q235B exhibited a dense and super-hydrophilic structure that effectively inhibited both corrosion and biofouling. Although the superhydrophilic barrier formed on 2507 also suppressed biofouling, it adversely affected the integrity of the passive film, leading to deterioration and pitting corrosion of stainless steel; this phenomenon deserved a comprehensive investigation.
Figure 11. Representation of the formation process of biomineralization layers and their application in inhibiting corrosion and biofouling. This figure was created in BioRender. Sun, Z. (2026) https://BioRender.com/zio0x73.
CONCLUSION
This study aimed to evaluate the formation and functional performance of P. spongiae-induced biomineralized layers on Q235B carbon steel and 2507 stainless steel in ASW. The results demonstrated that the biomineralization behavior and the protective performance of the formed layers were strongly dependent on the substrate. The biomineralized layer showed promising potential for suppressing corrosion and biofouling on carbon steel, while on stainless steel, it mainly exhibited antifouling capability with limited corrosion protection. Overall, this work provides a basis for the future design and optimization of environmentally friendly biomineralization-based surface protection strategies for marine metallic materials.
DECLARATIONS
Authors’ contributions
Conducted the experiments, analyzed the data, and wrote the manuscript: Zhang, Y.
Conceived the study, supervised the work, and revised the manuscript: Sun, Z.; Yin, Y.
Assisted with the experiments, data collection, and manuscript revision: Li, J.; Wan, X.; Zhou, G.; Lian, C.; Zhang, R.; Lu, J.
Provided funding support: Zhang, J.
All authors reviewed and approved the final manuscript.
Availability of data and materials
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version 5.3 instant) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the Guangdong Province Special Funding Project for the Development of Marine Economy (Grant No. GDNRC [2024] 41).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Wang, M.; Yang, L.; Wu, S.; et al. Research on marine atmospheric corrosion behavior of carbon steel during Western Pacific voyage. J Mater Res Technol 2025, 36, 9678-91.
2. Yan, X.; Yan, L.; Kang, S.; Qi, X.; Xu, M.; Zhang, P. Corrosion behavior and electrochemical corrosion of a high manganese steel in simulated marine splash zone. Mater Res Express 2021, 8, 126507.
3. Wang, A.; de Silva, K.; Jones, M.; Gao, W. Investigation of electrolysis corrosion on marine propellers. Mod Phys Lett B 2025, 39, 2442011.
4. de Mesquita, M. M. F.; Crapez, M. A. C.; Teixeira, V. L.; Cavalcanti, D. N. Potential interactions bacteria-brown algae. J Appl Phycol 2019, 31, 867-83.
5. Sun, J.; Zhao, X.; Rong, H.; et al. Effect of Ochrobactrum sp. on the corrosion behavior of 10MnNiCrCu steel in simulated marine environment. Int J Electrochem Sci 2020, 15, 2364-74.
6. Ariyanti, D.; Sugianto, D. N.; Purbasari, A.; Lesdantina, D.; Handayani, E. P. Synthesis of environmental-friendly fouling release coating, PDMS-X, to prevent the attachment of marine biofouling. J Indian Chem Soc 2025, 102, 101768.
7. Satasiya, G.; Kumar, M. A.; Ray, S. Biofouling dynamics and antifouling innovations: transitioning from traditional biocides to nanotechnological interventions. Environ Res 2025, 269, 120943.
8. Yang, D.; Huang, Y.; Li, J.; Gao, Y.; Zhao, X.; Xu, W. Corrosion behavior of Q235B carbon steel in simulated seawater pumped storage system under operational conditions. J Ocean Limnol 2020, 38, 1537-47.
9. Zhu, M.; Zhang, Q.; Yuan, Y.; Guo, S.; Huang, Y. Study on the correlation between passive film and AC corrosion behavior of 2507 super duplex stainless steel in simulated marine environment. J Electroanal Chem 2020, 864, 114072.
10. Chen, J.; Liu, J.; Zhou, C.; Zhu, F.; Chen, T.; Zhang, H. An automatic image enhancement method based on the improved HCTLS. In 2018 10 th IAPR workshop on pattern recognition in remote sensing (PRRS), Beijing, China. August 19-20, 2018; IEEE, 2018; p. 1-6.
11. Sun, Z.; Moradi, M.; Chen, Y.; et al. Simulation of the marine environment using bioreactor for investigation of 2507 duplex stainless steel corrosion in the presence of marine isolated Bacillus Vietnamensis bacterium. Mater Chem Phys 2018, 208, 149-56.
12. Qiu, H.; Feng, K.; Gapeeva, A.; et al. Functional polymer materials for modern marine biofouling control. Prog Polym Sci 2022, 127, 101516.
13. Päßler, J.; Jarochowska, E.; Bestmann, M.; Munnecke, A. Distinguishing biologically controlled calcareous biomineralization in fossil organisms using electron backscatter diffraction (EBSD). Front Earth Sci 2018, 6, 16.
14. Liu, T.; Guo, Z.; Zeng, Z.; et al. Marine bacteria provide lasting anticorrosion activity for steel via biofilm-induced mineralization. ACS Appl Mater Interfaces 2018, 10, 40317-27.
15. Guo, N.; Zhao, Q.; Hui, X.; et al. Enhanced corrosion protection action of biofilms based on endogenous and exogenous bacterial cellulose. Corros Sci 2022, 194, 109931.
16. Guo, Z.; Feng, Q.; Guo, N.; Yin, Y.; Liu, T. Positive effects of molybdenum on the biomineralization process on the surface of low-alloy steel catalyzed by Bacillus subtilis. Front Microbiol 2024, 15, 1428286.
17. Guo, Z.; Pan, S.; Liu, T.; et al. Bacillus subtilis inhibits vibrio natriegens-induced corrosion via biomineralization in seawater. Front Microbiol 2019, 10, 1111.
18. Kip, N.; Jansen, S.; Leite, M. F. A.; et al. Methanogens predominate in natural corrosion protective layers on metal sheet piles. Sci Rep 2017, 7, 11899.
19. Hao, X.; Bai, Y.; Ren, C.; et al. Self-healing effect of damaged coatings via biomineralization by Shewanella putrefaciens. Corros Sci 2022, 196, 110067.
20. Marques, M.; Jaume, J.; Mercier, D.; et al. The positive impact of biomineralization for marine corrosion protection of AA5083 alloy. Corros Sci 2024, 233, 112053.
21. Wang, H.; Sun, X.; Wang, Y.; Shi, W.; Wu, L.; Miao, L. Marine steel protection based on biomineralization for sustainable development of coastal cities. Bioresour Technol 2025, 428, 132404.
22. Lv, M.; Du, M.; Zhao, X.; Du, Y. Fundamental understanding of microbiologically influenced corrosion inhibition via biomineralization: a critical review. Crit Rev Environ Sci Technol 2025, 55, 928-50.
23. Guo, S.; Jiang, J.; Yang, D.; et al. Microstructure evolution of Al-Zn-In-Mg-Ti sacrificial anodes during multi-pass equal-channel angular pressing and its impact on corrosion protection performance. Mater Today Commun 2025, 48, 113384.
24. Liu, R.; Guan, Y.; Cui, Y.; Meng, F.; Wang, F.; Liu, L. The self-powered cathodic protection effect of flexible PDMS-based piezoelectric composites in a simulated pressure-alternating marine environment. Compos Commun 2025, 53, 102166.
25. Wu, Y.; Zhao, W.; Wang, L. State of the art and current trends on the metal corrosion and protection strategies in deep sea. J Mater Sci Technol 2025, 215, 192-213.
26. Zhou, Z.; He, J.; Deng, Y.; Shi, X. Improving the protective performance of discrete Zn-based sacrificial anode by increasing porosity and alkali concentration of its encapsulation matrix. Cem Concr Compos 2025, 163, 106200.
27. Vinagre, P. A.; Simas, T.; Cruz, E.; Pinori, E.; Svenson, J. Marine biofouling: a European database for the marine renewable energy sector. J Mar Sci Eng 2020, 8, 495.
28. Dong, Y.; Song, G.; Zhang, J.; Gao, Y.; Wang, Z. M.; Zheng, D. Biocorrosion induced by red-tide alga-bacterium symbiosis and the biofouling induced by dissolved iron for carbon steel in marine environment. J Mater Sci Technol 2022, 128, 107-17.
29. He, W.; Xue, H.; Liu, C.; Zhang, A. H.; Huang, J.; Zhang, D. Biomineralization of struvite induced by indigenous marine bacteria of the genus Alteromonas. Front Mar Sci 2023, 10, 1085345.
30. Guo, N.; Wang, Y.; Hui, X.; et al. Marine bacteria inhibit corrosion of steel via synergistic biomineralization. J Mater Sci Technol 2021, 66, 82-90.
31. Beurmann, S.; Ushijima, B.; Svoboda, C. M.; et al. Pseudoalteromonas piratica sp. nov., a budding, prosthecate bacterium from diseased Montipora capitata, and emended description of the genus Pseudoalteromonas. Int J Syst Evol Microbiol 2017, 67, 2683-8.
32. Zheng, K.; Dong, Y.; Liang, Y.; et al. Genomic diversity and ecological distribution of marine Pseudoalteromonas phages. Mar Life Sci Technol 2023, 5, 271-85.
33. Saravanan, P.; Jayachandran, S. Preliminary characterization of exopolysaccharides produced by a marine biofilm-forming bacterium Pseudoalteromonas ruthenica (SBT 033). Lett Appl Microbiol 2008, 46, 1-6.
34. Hao, L.; Liu, W.; Liu, K.; et al. Isolation, optimization of fermentation conditions, and characterization of an exopolysaccharide from Pseudoalteromonas agarivorans Hao 2018. Mar Drugs 2019, 17, 703.
35. Colin, B.; Vincent, J.; Koziorowszki, L.; et al. Calcareous deposit formation under cathodic polarization and marine biocalcifying bacterial activity. Bioelectrochemistry 2022, 148, 108271.
36. Torres-Crespo, N.; Martínez-Ruiz, F.; González-Muñoz, M. T.; Bedmar, E. J.; De Lange, G. J.; Jroundi, F. Role of bacteria in marine barite precipitation: a case study using Mediterranean seawater. Sci Total Environ 2015, 512-3, 562-71.
37. Kim, S. J.; Kim, B. G.; Park, H. J.; Yim, J. H. Cryoprotective properties and preliminary characterization of exopolysaccharide (P-Arcpo 15) produced by the Arctic bacterium Pseudoalteromonas elyakovii Arcpo 15. Prep Biochem Biotechnol 2016, 46, 261-6.
38. Moldabayeva, G. Z.; Kozlovskiy, A. L.; Kuldeyev, E. I.; Syzdykov, A. K.; Bakesheva, A. Study of the effectiveness of corrosion resistance growth by application of layered AlN–TiO2 coatings. Coatings 2024, 14, 373.
39. Zhang, Z.; Zhao, N.; Qi, F.; Zhang, B.; Liao, B.; Ouyang, X. Reinforced superhydrophobic anti-corrosion epoxy resin coating by fluorine–silicon–carbide composites. Coatings 2020, 10, 1244.
40. Liu, H.; Chen, W.; Tan, Y.; et al. Characterizations of the biomineralization film caused by marine Pseudomonas stutzeri and its mechanistic effects on X80 pipeline steel corrosion. J Mater Sci Technol 2022, 125, 15-28.
41. Li, M.; Wu, H.; Sun, Y. Corrosion performance of welded joints for E40 marine steel. Metals 2023, 13, 1528.
42. Alcántara, J.; Fuente, D.; Chico, B.; Simancas, J.; Díaz, I.; Morcillo, M. Marine atmospheric corrosion of carbon steel: a review. Materials 2017, 10, 406.
43. Wang, X.; Melchers, R. E. Corrosion of carbon steel in presence of mixed deposits under stagnant seawater conditions. J Loss Prev Process Ind 2017, 45, 29-42.
44. Singh, H.; Xiong, Y.; Rani, E.; et al. Unveiling nano-scaled chemical inhomogeneity impacts on corrosion of Ce-modified 2507 super-duplex stainless steels. npj Mater Degrad 2022, 6, 263.
45. Chen, H.; Cui, H.; Ma, G.; Zhang, H.; Zhu, Y.; Song, X. Mechanical and corrosion behavior of oscillating laser beam welded 2507 super duplex stainless steel: synergistic effects of acidic seawater and strain states. J Mater Sci Technol 2026, 251, 227-40.
46. Zhao, T.; He, L.; Qiu, Z.; Zhang, Z.; Lin, C. Synergistic effect between sulfate-reducing bacteria and Shewanella algae on corrosion behavior of 321 stainless steel. J Mater Res Technol 2023, 26, 4906-17.
47. Cui, T.; Qian, H.; Chang, W.; et al. Towards understanding Shewanella algae-induced degradation of passive film of stainless steel based on electrochemical, XPS and multi-mode AFM analyses. Corros Sci 2023, 218, 111174.
48. Wang, X.; Liu, J.; Zhao, W.; et al. Fine-scale structuring of planktonic Vibrio spp. in the Chinese marginal seas. Appl Environ Microbiol 2022, 88, e0126222.
49. Liu, S.; Dong, C.; Zhu, Y.; Wang, Z.; Li, Y.; Feng, G. Recent developments on biomineralization for erosion control. Appl Sci 2025, 15, 6591.
50. Reiprich, S.; Akova, E.; Aszódi, A.; Schönitzer, V. Hyaluronan synthases’ expression and activity are induced by fluid shear stress in bone marrow-derived mesenchymal stem cells. Int J Mol Sci 2021, 22, 3123.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data




















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].