Recent development of lithium niobate-based luminescent materials for photonics and optoelectronics
Abstract
Lithium niobate (LiNbO3) is a key material in photonics and optoelectronics, valued for its ferroelectric, electro-optic, and nonlinear optical properties. Despite its high Curie temperature and significant spontaneous polarization, which are advantageous for light modulation and frequency conversion, undoped LiNbO3 suffers from intrinsically weak luminescence, thereby restricting its application in light-emitting devices. Doping with rare earth ions such as erbium (Er3+), neodymium (Nd3+), and praseodymium (Pr3+) significantly enhances its luminescent properties by introducing efficient photon-emitting energy levels. This review provides a comprehensive overview of the luminescent properties of pure and doped LiNbO3, with a particular focus on doping and co-doping strategies using rare earth and transition-metal ions to enhance their photoluminescence efficiency, thermal stability, and spectral tunability. The roles of dopant site occupancy, defect engineering, and charge compensation mechanisms are discussed in detail. Co-doping approaches are highlighted as promising routes to synergistically tailor emission characteristics and mitigate concentration quenching. Furthermore, the review explores recent advances in LiNbO3-based luminescent devices, including waveguide-integrated photonic components, resonators, and thin films. Finally, future challenges and perspectives are outlined for the rational design of high-performance LiNbO3-based luminescent materials in next-generation photonic technologies.
Keywords
INTRODUCTION
Lithium niobate (LiNbO3) is a multifunctional ferroelectric material renowned for its exceptional electro-optic, acousto-optic, piezoelectric, and nonlinear optical properties. Owing to its wide bandgap, high optical transparency [covering ultraviolet (UV) to infrared], and excellent chemical and thermal stability, LiNbO3 has been extensively employed in a broad spectrum of photonic and optoelectronic applications, including electro-optic modulators[1,2], frequency converters[3,4], waveguides[5,6], and optical memory devices[7-9]. These foundational properties and applications have been extensively reviewed in earlier literature and constitute well-established knowledge in the field.
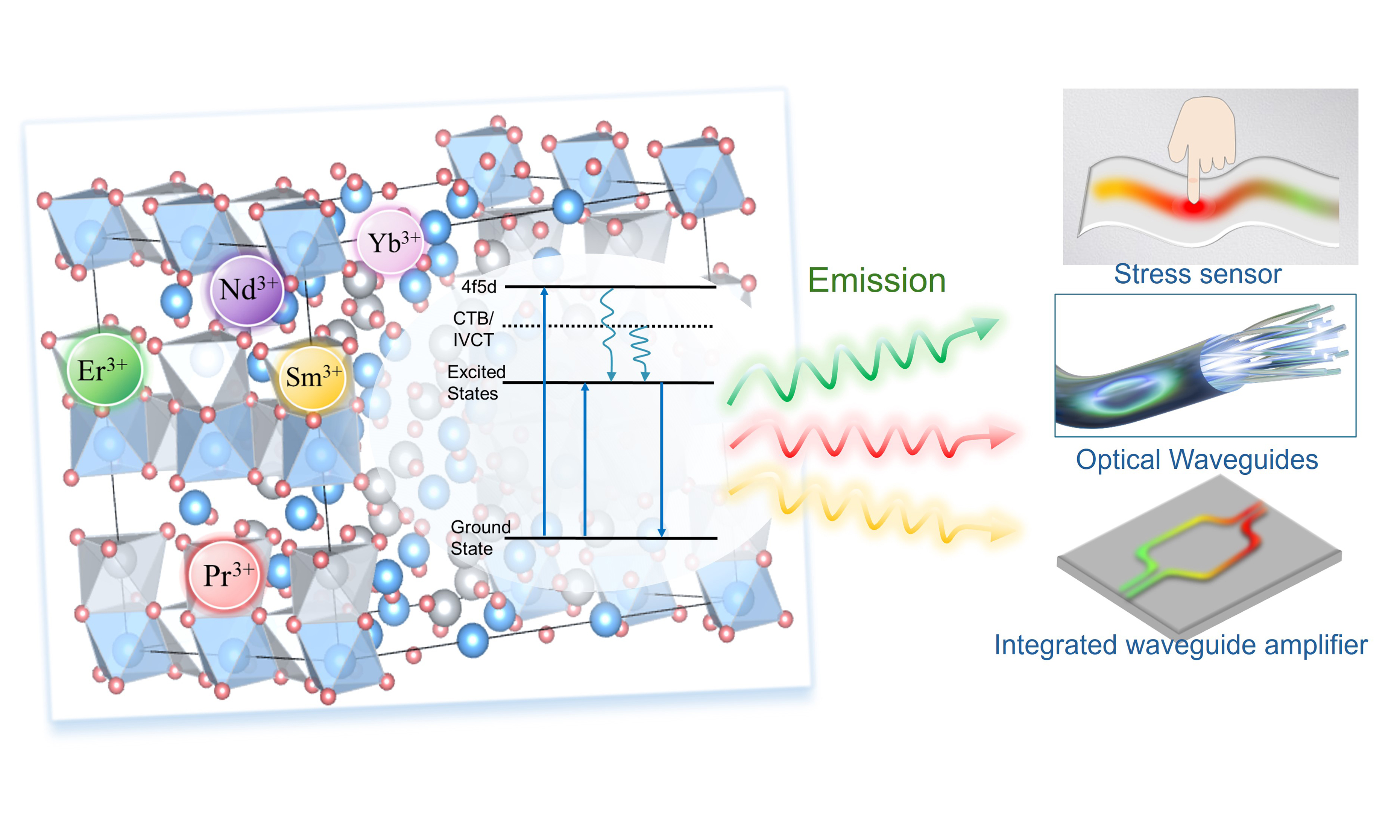
Beyond these established functionalities, the luminescent behavior of LiNbO3 has attracted increasing attention over the past decade, particularly its distinctive role in mechanoluminescence (ML)[10-12]. Growing efforts have focused on exploiting the coupling between ferroelectric polarization, piezoelectric fields, and defect engineering to activate and modulate luminescence in LiNbO3-based materials. This coupling provides a platform for probing piezoelectric-driven luminescence mechanisms and supports emerging applications in stress sensing, structural health monitoring, biomechanical imaging, and anti-counterfeiting technologies[13-16]. In general, intrinsic luminescence of pure LiNbO3 remains weak, while concentration quenching, complex defect regulation, and limited emission stability under thermal and mechanical perturbations restrict device performance. Significant progress has been achieved through the introduction of activator ions such as praseodymium (Pr3+), erbium (Er3+), Tm3+, Eu3+, and Mn2+, enabling efficient photoluminescence (PL) and upconversion luminescence (UCL) via f-f or d-d transitions[17-24]. Co-doping with sensitizer ions such as Yb3+[17,25] or charge compensators including Mg2+ and Zn2+ further enhances luminescence intensity, spectral tunability, and structural stability[26-31]. In parallel, dopants such as Mo or Fe improve photorefractive properties[32-36], while Mg, Zn, or Zr doping enhances resistance to optical damage[28,30,37].
Despite these advances, challenges including limited spectral tunability, low emission intensity, and inadequate environmental stability remain. To address these limitations, recent studies have investigated various enhancement strategies, including multi-ion synergy doping[26,38-41], more precise defect regulation[21,42,43], external stimulus responses (such as electric, force and thermal fields)[44,45], and heterostructure[46-48] enhancement effects. Compared with single ion-doped LiNbO3, multi-ion co-doping enables multiple emission channels within one host lattice, allowing controllable color modulation and enhanced emission. For instance, LiNbO3:Er3+/Pr3+ microcrystals show tunable green-to-orange emission through excitation and concentration control with good stability[39]. Bi3+-assisted trap engineering in Sm3+- or Pr3+-doped systems further enables reversible PL modulation and stepwise thermally stimulated luminescence[26,40]. In addition, NaNbO3-LiNbO3 heterostructures promote carrier migration and optimize trap distributions, leading to enhanced low-temperature persistent luminescence (PersL)[46]. Collectively, these developments reflect a transition from simple dopant activation toward integrated luminescence engineering that couples defect chemistry, internal electric fields, and external stimuli, motivating a focused review of recent advances and future directions in LiNbO3-based luminescent materials.
Taken together, the rapid advances achieved over the past decade underscore both the opportunities and the complexity of LiNbO3-based luminescent systems, highlighting the need for a timely and systematic synthesis of recent progress, challenges, and future directions. In this review, luminescence is highlighted as an emerging, stimulus-coupled functionality of LiNbO3. The underlying emission mechanisms are systematically examined, together with dopant and co-doping strategies and defect- and field-mediated approaches for regulating luminescent behavior. Representative device-level implementations, including integrated photonic components and stress-responsive systems, are also discussed, along with current challenges and future directions for the rational design of high-performance LiNbO3-based luminescent materials.
PURE LITHIUM NIOBATE
Crystalline structure and electro-optic properties of pure LiNbO3
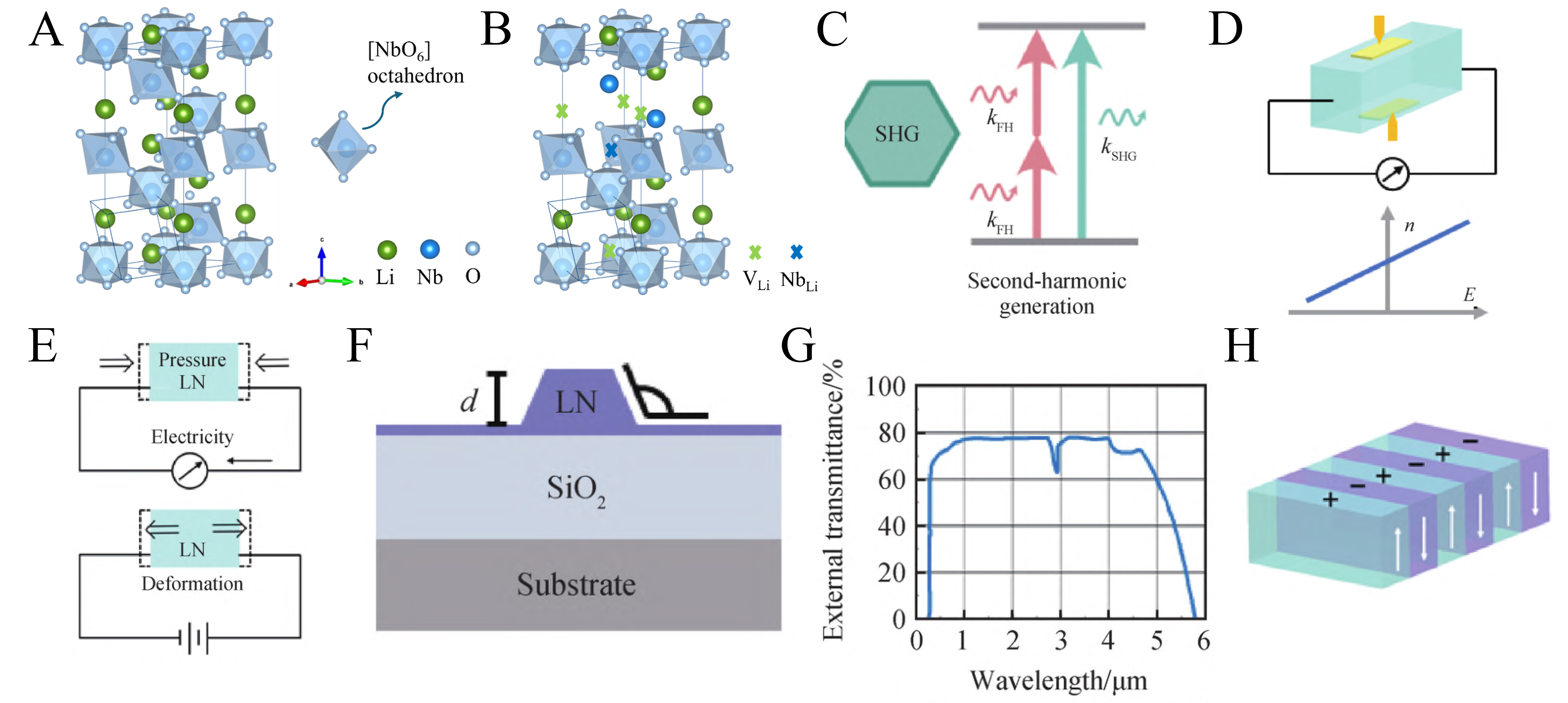
LiNbO3 is a prototypical ferroelectric oxide that integrates strong piezoelectricity, spontaneous polarization, and pronounced electro-optic response within a non-centrosymmetric lattice[49-51], rendering it particularly suitable for field-coupled luminescence regulation. At room temperature, LiNbO3 crystallizes in the ferroelectric rhombohedral R3c phase [Figure 1A][51,52], where off-center displacement of Li and Nb ions within oxygen octahedra generates a spontaneous polarization along the c-axis[53]. Together with large piezoelectric coefficients and a high Curie temperature of about 1,210 °C, this lattice configuration enables efficient coupling between mechanical, electrical, and optical energy domains[54]. Under mechanical stimulation, the large piezoelectric response facilitates stress-induced internal electric field generation, which promotes carrier de-trapping and recombination, thereby favoring efficient ML. Moreover, LiNbO3 crystals inherently contain a high density of intrinsic point defects, dominated by Nb antisites (
Figure 1. (A) Crystal structures of stoichiometric LiNbO3; (B) Congruent LiNbO3 with
Optically, LiNbO3 is characterized by a high refractive index and significant birefringence, making it ideal for phase matching in nonlinear optical processes such as second-harmonic generation (SHG)[55,56]. Its broad transparency range of approximately 400 to 5,000 nm facilitates applications in various optical devices, including waveguides and modulators. The high nonlinear optical coefficients of LiNbO3 are particularly advantageous for frequency conversion in laser systems, establishing its role in advanced photonic applications [Figure 1C-H][53,57].
Luminescence characteristics and application limitations of pure LiNbO3
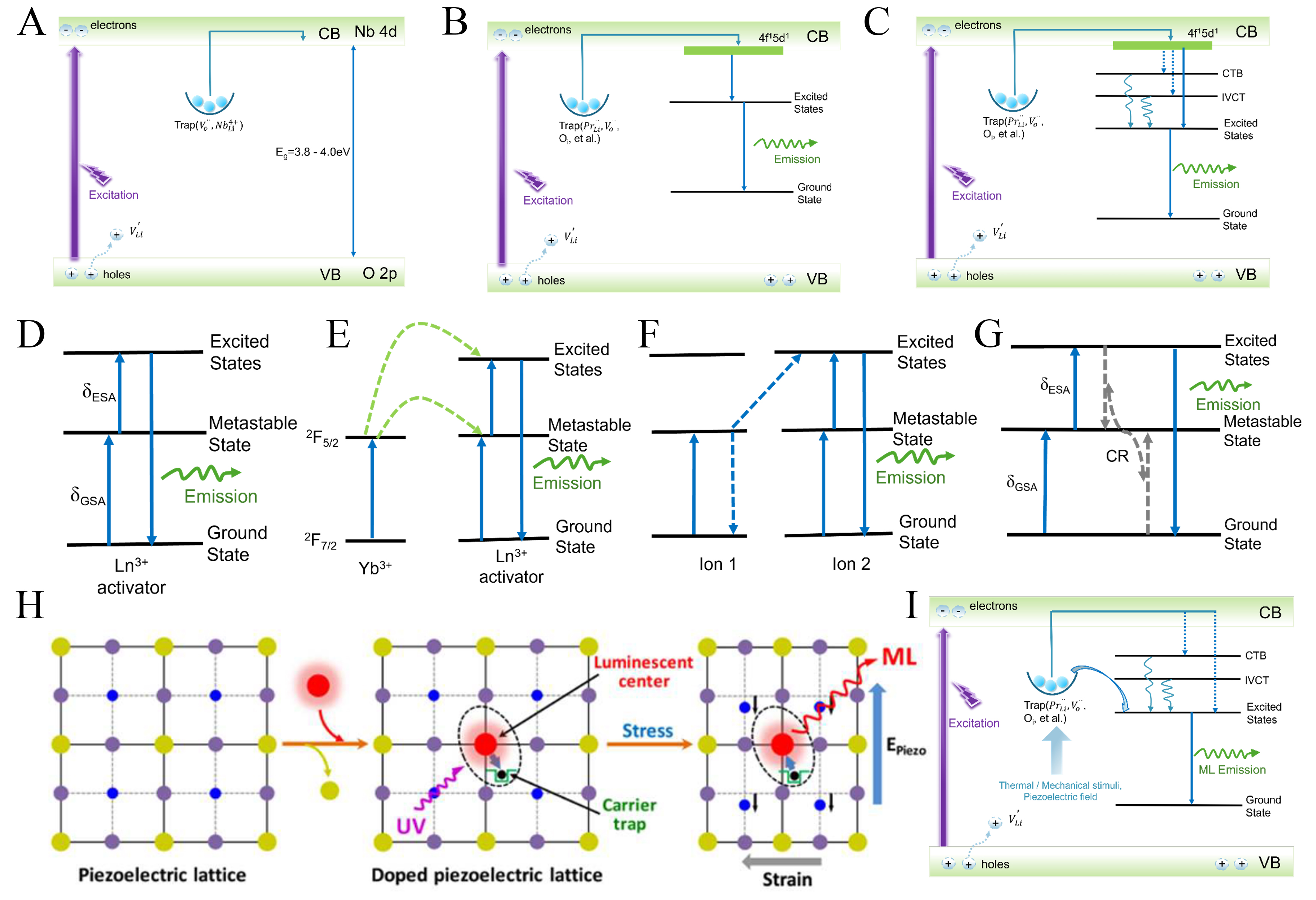
The luminescent performance of pure LiNbO3 is intrinsically limited by its electronic structure. LiNbO3 exhibits weak intrinsic emission that originates from O 2p-Nb 4d charge-transfer transitions and defect-mediated recombination involving oxygen vacancies, Nb antisites, and Li vacancies [Figure 2A]. Energy transfer from the host lattice to luminescent centers is inefficient, and the resulting emission is highly sensitive to crystal purity and structural quality[58,59], rendering defect- or impurity-related luminescence in undoped LiNbO3 generally weak[60-63]. These limitations preclude the direct use of pure LiNbO3 in applications requiring strong and stable emission, even though activator doping can partially enhance luminescence[50]. In addition, the pronounced photorefractive effect in LiNbO3 can further undermine emission stability through optically induced refractive index modulation[32].
Figure 2. (A) Schematic of the intrinsic luminescence mechanism in LiNbO3; (B and C) Luminescence mechanisms in rare earth-doped LiNbO3. Reproduced from Ref.[12] with permission from Elsevier and Ref.[68] with permission from American Chemical Society Publications; (D) Excited state absorption UCL mechanism; (E) Energy transfer UCL mechanism; (F) Cross-relaxation UCL mechanism; (G) Photon avalanche UCL mechanism. (D-G) Reproduced from Ref.[69] with permission from Elsevier; (H) Schematic illustration showing the design strategy for creating recoverable ML in piezoelectrics. Reproduced from Ref.[70] with permission from the American Chemical Society; (I) ML mechanism in LiNbO3:Pr3+. CB: Conduction band; VB: valence band; ESA: excited state absorption; GSA: ground state absorption; CR: cross relaxation; UV: ultraviolet; CTB: charge-transfer band; IVCT: intervalence charge-transfer; ML: mechanoluminescence; UCL: upconversion luminescence; LiNbO3: lithium niobate; Pr: praseodymium.
Due to these limitations, the application of pure LiNbO3 in luminescent devices is generally restricted to cases in which its other properties, such as large nonlinear optical coefficients and strong electro-optic effects, outweigh the requirement for high luminescence efficiency. In most cases, LiNbO3 serves as an excellent host matrix for luminescent centers, exhibiting remarkable solid solubility for various dopant ions. Its structural and optoelectronic properties can be systematically tuned through controlled variation of dopant type and concentration (typically 0.1-5 mol%), which introduce well-defined luminescent properties suitable for a range of photonic applications[11,40,64,65].
DOPING-MODIFIED LITHIUM NIOBATE
Non-rare earth ion doping modification and its mechanisms
Numerous studies have demonstrated that non-rare earth (RE) dopants can effectively regulate the luminescence behavior of LiNbO3 by modifying its intrinsic defect landscape. Tokko et al. demonstrated that the near-infrared (NIR) PL of LiNbO3:Zn2+/Mg2+ is predominantly governed by defect chemistry rather than dopant concentration alone[66]. Specifically, intrinsic defects such as niobium vacancies (VNb) and niobium at empty octahedral sites (Nb□oct) act as major nonradiative quenching centers, whereas homogeneous Zn2+/Mg2+ co-doping effectively suppresses these defects and introduces ZnLi-related centers that activate NIR emission. At higher Zn2+ concentrations, partial substitution of Li+ by Zn2+ induces pronounced modification of the local crystal field, leading to the emergence of a strong violet emission band centered at 418 nm[67]. This emission is attributed to the crystal field perturbation of niobate groups induced by Zn2+ incorporation, which becomes increasingly prominent as the Li+ content decreases with increasing Zn2+ concentration.
Although both Mg2+ and Zn2+ dopants have been shown to enhance luminescence in LiNbO3, they operate through fundamentally different mechanisms. Mg2+ co-doping improves emission efficiency by regulating intrinsic defect populations and stabilizing the carrier recombination pathway. In contrast, Zn2+ plays a more active role in reshaping the local lattice environment and crystal field symmetry, enabling the activation of new intrinsic emission bands and enhanced spectral tunability. However, this increased spectral tunability is often accompanied by greater lattice distortion at higher Zn2+ concentrations, indicating a clear trade-off between emission activation and structural stability. This behavior contrasts with the more defect-stabilizing role of Mg2+.
Rare earth-doped LiNbO3: luminescence mechanisms
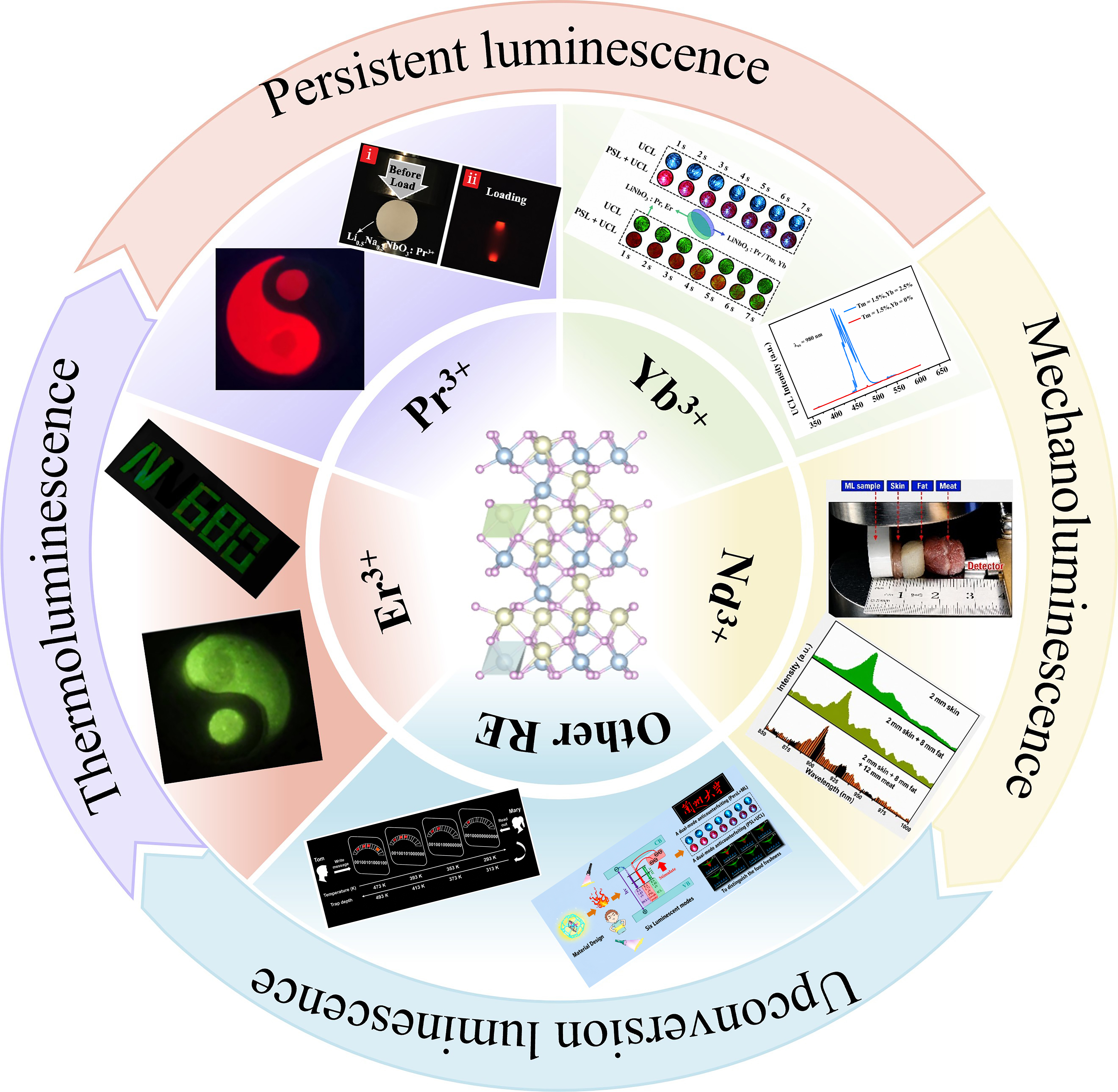
Compared with non-RE dopants, rare earth ions (RE3+) provide well-defined and optically efficient luminescent centers in LiNbO3 owing to their shielded 4f electronic configurations. Rare earth doping markedly modifies the luminescence behavior of LiNbO3[12,20,27,64]. The intrinsic 4f and 5d energy levels of RE3+ introduce stable radiative transition channels and enable characteristic narrow band PL [Figure 2B-G][12,68,69]. More importantly, RE doping not only directly introduces luminescent centers but also alters the defect distribution and electronic structure[70]. Figure 3 illustrates the multimodal luminescence behavior of LiNbO3-doped with different RE3+[11,39-41,44,47,68,71-74]. Notably, RE site occupancy in LiNbO3 serves as a key link between stoichiometry and defect chemistry. Li-deficient compositions tend to promote defect clustering and nonradiative recombination, which are detrimental to luminescence. By contrast, near-stoichiometric or slightly Li-rich conditions favor more effective RE incorporation and suppress excessive Nb antisites, leading to enhanced emission efficiency. Meanwhile, RE doping introduces and stabilizes charge-transfer band (CTB) and intervalence charge-transfer (IVCT) states [Figure 2C], which broaden the excitation window and facilitate carrier storage and migration. Together, the synergistic regulation of site occupancy, Li/Nb ratio (RLN), and defect-mediated charge compensation underpins the enhanced and multifunctional luminescence observed in PL, PersL, UCL, and ML.
Figure 3. Schematic illustration of the multimodal luminescence behavior of rare earth-doped LiNbO3[11,39-41,44,47,68,71-74]. Data for Pr3+ are summarized from Refs.[39,47,68,73,74], Er3+ from Refs.[39,68,72-74], Yb3+ from Refs.[68,74], Nd3+ from Refs.[11,44,68], other rare earth ions from Refs.[40,41,68,71,72,74]. Reproduced from Ref.[11] with permission from the Royal Society of Chemistry. Reproduced from Ref.[40] with permission from the Elsevier. Reproduced from Ref.[47] with permission from the John Wiley and Sons. Reproduced from Ref.[73,74] with permission from the American Chemical Society. Note: The Chinese characters appearing in the "Other RE" section represent "Lanzhou University". RE: Rare earth; Pr3+: praseodymium; Er: erbium; Nd3+: neodymium; Yb: ytterbium; PersL: persistent luminescence; PSL: photostimulated luminescence; UCL: upconversion luminescence.
UCL is a nonlinear optical process in which sequential absorption of low energy photons leads to higher energy emission[75,76]. In RE-doped LiNbO3, upconversion proceeds mainly through multiphoton pathways involving ground state absorption, excited state absorption, energy transfer upconversion, cross relaxation, and photon avalanche processes [Figure 2D-G]. Compared with low phonon energy hosts such as fluorides, LiNbO3 exhibits reduced upconversion efficiency due to enhanced multiphonon relaxation. Nevertheless, its dense oxide lattice and short interionic distances promote energy migration and Yb3+-sensitized energy transfer [Figure 2E]. In addition, the non-centrosymmetric ferroelectric lattice introduces internal electric fields that modulate local crystal fields and defect states, enabling field-responsive upconversion behavior. As a result, LiNbO3 combines moderate upconversion efficiency with unique field- and stress-coupled luminescence modulation. For ML in piezoelectric materials, luminescent lanthanide ions are commonly introduced as substitutional dopants to provide emission centers and carrier traps simultaneously [Figure 2H][70]. Under mechanical stimulation, the piezoelectric field generated in LiNbO3 promotes carrier release from trap states and subsequent recombination at luminescent centers, leading to light emission. In this process, piezoelectricity governs carrier activation, while defects control energy storage and mediation. As shown in Figure 2I, LiNbO3:Pr3+ exhibits pronounced ML performance under mechanical or piezoelectric excitation.
Table 1 summarizes key electronic configurations and emission characteristics of typical RE luminescent ions. Representative RE dopants are discussed according to their dominant luminescence modes, progressing from upconversion and energy transfer processes to NIR emission and stress stimulation luminescence.
Basic properties and luminescence characteristics of common rare earth elements
| Rare earth elements | Ground-state electron configuration | Primary emission wavelength | Transition energy level | Ground state | Excited-state features | Refs. |
| Cerium (Ce) | [Xe]5d16s2 | 358 nm 378 nm | 5d1→2F5/2 5d1→2F7/2 | 2F5/2 | 5d1 | [68,77] |
| Praseodymium (Pr) | [Xe]4f36s2 | 483 nm 540 nm 610 nm 643 nm 725 nm | 3P0 → 3H4 3P0 → 3H5 3P0 → 3H6,1D2 → 3H4 3P0 → 3F2 3P0 → 3F3,4 | 3H4 | 3P0, 1D2, 1G4 | [78,79] |
| Neodymium (Nd) | [Xe]4f46s2 | 413 nm 523 nm 588 nm 640 nm 661 nm 869-900 nm 1,060 nm 1,330 nm | 2D5/2, 2P1/2 → 4I 9/2 4G7/2, 2K13/2, 4G9/2 → 4I9/2 2G7/2, 2G5/2 → 4I9/2 2H11/2 → 4I9/2 4F9/2 → 4I9/2 4F3/2 → 4I9/2 4F3/2 → 4I11/2 4F3/2 → 4I13/2 | 4I9/2 | 2P1/2, 4G7/2 | [80,81] |
| Samarium (Sm) | [Xe]4f66s2 | 566 nm 602 nm 649 nm 728 nm | 4G5/2 → 6H5/2 4G5/2 → 6H7/2 4G5/2 → 6H9/2 4G5/2 → 6H11/2 | 6H15/2 | 4G5/2 | [82-84] |
| Europium (Eu) | [Xe]4f76s2 | 510 nm 536 nm 555 nm 584 nm 591 nm 615 nm 645 nm 696 nm | 5D2 → 7F3 5D1 → 7F1 5D1 → 7F2 5D1 → 7F3, 5D0 → 7F0 5D0 → 7F1 5D0 → 7F2 5D0 → 7F3 5D0 → 7F4 | 7F0 | 5D0, 5D1, 5D2, 5D3 | [85,86] |
| Gadolinium (Gd) | [Xe]4f75d16s2 | 246 nm 253 nm 270-281 nm 305 nm 311 nm | 6DJ → 8S7/2 (J = 1/2, 3/2, 5/2, 7/2) 6D9/2 → 8S7/2 6IJ → 8S7/2 (J = 7/2-15/2) 6P5/2 → 8S7/2 6P7/2 → 8S7/2 | 8S7/2 | 6P7/2 | [87,88] |
| Terbium (Tb) | [Xe]4f96s2 | 380 nm 414 nm 435 nm 486 nm 541 nm 587 nm 622 nm | 5D3 → 7F6 5D3 → 7F5 5D3 → 7F4 5D4 → 7F6 5D4 → 7F5 5D4 → 7F4 5D4 → 7F3 | 7F6 | 5D3, 5D4 | [89,90] |
| Dysprosium (Dy) | [Xe]4f106s2 | 480 nm 570 nm 673 nm 756 nm | 4F9/2 → 6H15/2 4F9/2 → 6H13/2 4F9/2 → 6H11/2 4F9/2 → 6H9/2 | 6H15/2 | I→F energy-level transition | [83,91] |
| Holmium (Ho) | [Xe]4f116s2 | 525 nm 650 nm 1,150 nm | 5F4 → 5I8 5F5 → 5I8 5I6 → 5I8 | 5I8 | 5F4, 5F5, 5I6 | [69,92] |
| Erbium (Er) | [Xe]4f126s2 | 410 nm 525 nm 546 nm 660 nm 1,550 nm | 2H9/2→ 4I15/2 2H11/2 → 4I15/2 4S3/2 → 4I15/2 4F9/2 → 4I15/2 4I13/2 → 4I15/2 | 4I15/2 | 2H9/2, 2H11/2, 4S3/2, 4F9/2 | [84,87,92] |
| Thulium (Tm) | [Xe]4f136s2 | 359 nm 450 nm 477 nm 650 nm 695 nm 800 nm | 1D2 → 3H4 1D2 → 3F4 1G4 → 3H6 1G4 → 3F4 3F2 → 3H6 3H4 → 3H6 | 3H6 | 1D2, 1G4 | [84,88] |
| Ytterbium (Yb) | [Xe]4f146s2 | 984nm | 2F5/2 → 2F7/2 | 2F7/2 | 2F5/2 | [68,93,94] |
Upconversion luminescence: Er3+-activated systems
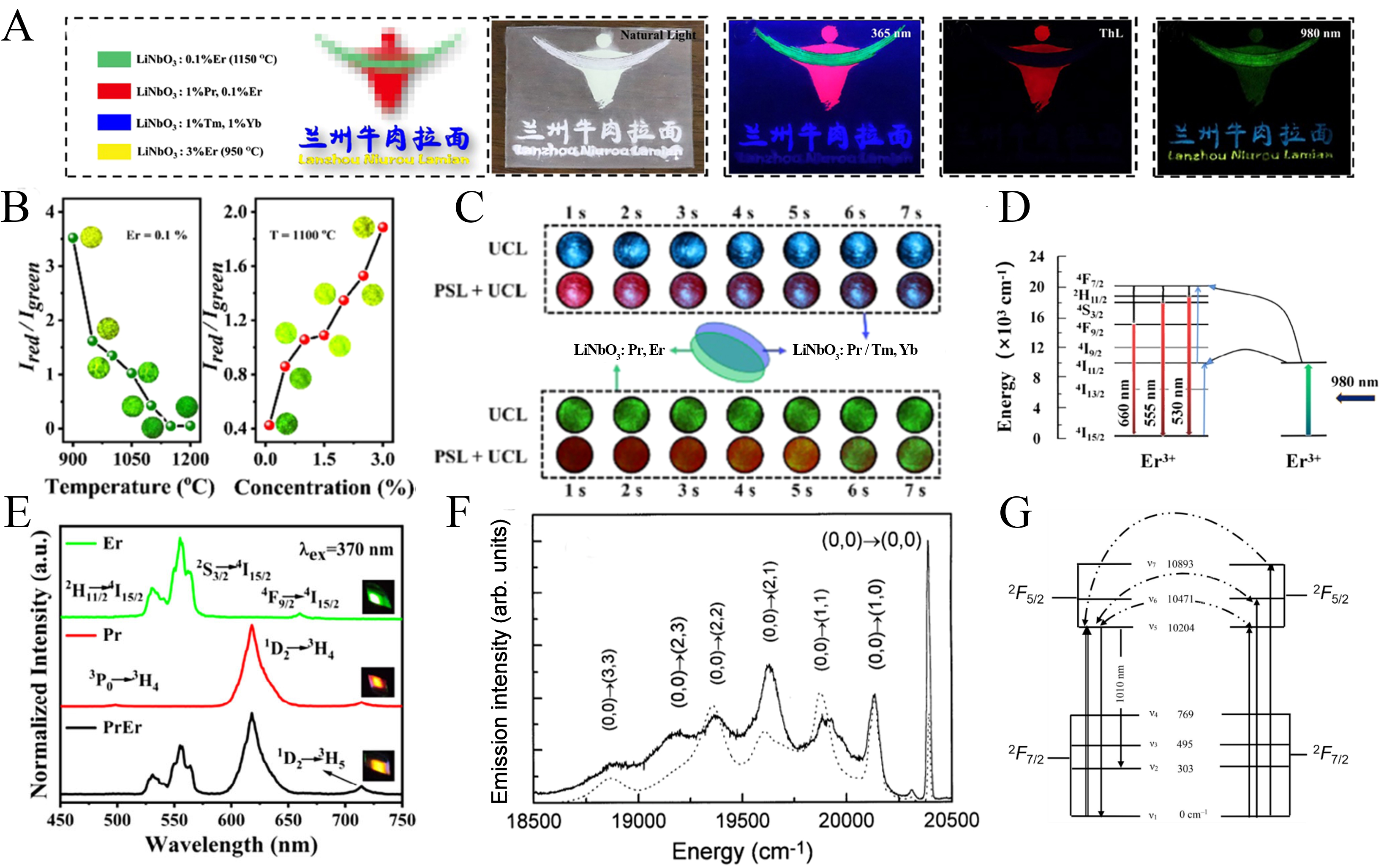
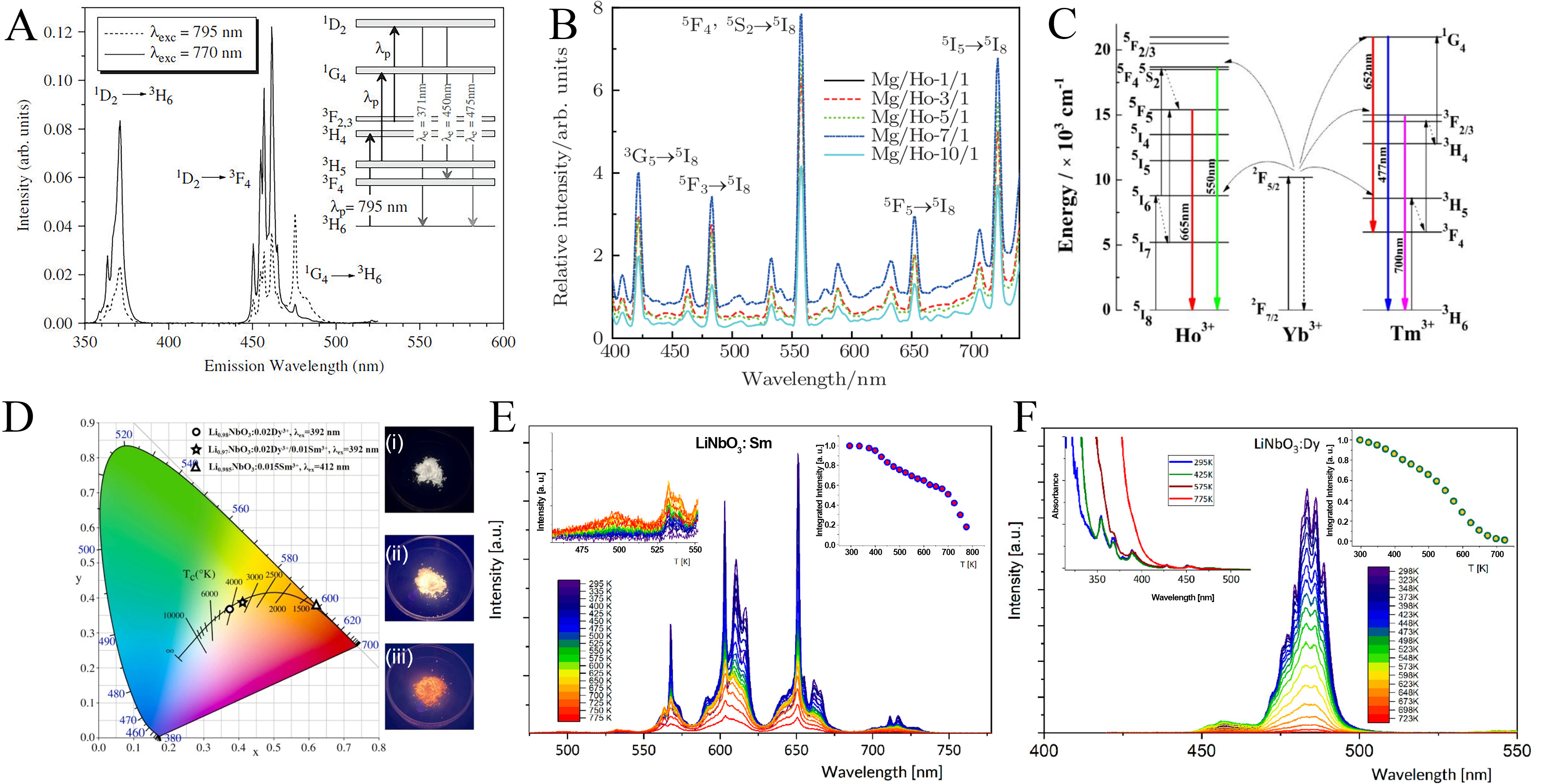
Er3+-doped LiNbO3 has been most extensively studied as an UCL system, in which NIR excitation is converted into visible emission through multiphoton processes. Wang et al. first reported that LiNbO3:Er3+ exhibits efficient NIR excited UCL with tunable color evolution[74]. By integrating downconversion luminescence (DCL), thermoluminescence (TL), PL, and UCL, a multimode static dynamic luminescence platform was further demonstrated for anti-counterfeiting applications [Figure 4A-C]. Under 980 nm excitation, LiNbO3:Er3+ follows a typical two-photon upconversion process, yielding green and red emissions [Figure 4D and E]. The temperature-dependent UCL response delivers a maximum relative sensitivity of 0.889% K-1, highlighting its potential for optical thermometry[39]. In addition, LiNbO3:Er3+ exhibits strong 1.54 μm emission under 980 or 1,480 nm excitation, observable in both bulk and waveguide configurations; the latter benefits from enhanced optical confinement. PL intensity is governed by Er3+ concentration, thermal treatment, crystal stoichiometry, and co-dopant or sensitizer engineering.
Figure 4. (A) Optical photographs of the as-fabricated four-mode anticounterfeiting film based on LiNbO3:RE3+ (RE3+ = Pr3+, Tm3+, Er3+, Yb3+), recorded under daylight, 365 nm excitation, 980 nm excitation, and after heat treatment[74]; (B) UCL spectra of LiNbO3:0.1%Er3+ samples synthesized at various reaction temperatures[74]; (C) Ired/Igreen variation in the UCL of LiNbO3:0.1%Er3+ dependent on the reaction temperature and doping concentration[74]; (D) Static-dynamic luminescence evolution of the fabricated anticounterfeiting device based on UCL and PSL[74]; (E) UCL spectra of LiNbO3:1.5%Tm3+ and LiNbO3:1.5%Tm3+, 2.5%Yb3+[74]. Reproduced from Ref.[74] with permission from the American Chemical Society; (F and G) Emission spectra of LiNbO3:4%Yb3+ at 12 K[93]. Reproduced from Ref.[93] with permission from the Elsevier. LiNbO3: Lithium niobate; RE3+: rare earth ions; UCL: upconversion luminescence; PSL: photostimulated luminescence; Tm: thulium; Yb: ytterbium.
The luminescence behavior of Er3+ in LiNbO3 is strongly governed by its local coordination environment and defect-mediated charge compensation. Owing to ionic size compatibility, Er3+ preferentially substitutes for Li+ sites, which induces local charge imbalance and the formation of compensating defects such as lithium vacancies, niobium antisites, or oxygen vacancies. These defects modify the local crystal field and trap distribution around Er3+ centers, thereby regulating radiative recombination and nonradiative relaxation. At low Er3+ concentrations, radiative emission dominates, whereas higher doping levels promote energy migration and cross-relaxation, reducing efficiency, leading to concentration quenching and reduced emission lifetime. To mitigate these effects, co-doping strategies have been widely employed. In particular, Mg2+ co-doping effectively suppresses Nb antisite-related quenching centers and enhances Er3+ emission by up to an order of magnitude[95]. Yb3+ sensitization further regulates population dynamics through RE energy exchange, enabling efficient energy transfer and controlled excitation processes in LiNbO3[25,96-98].
Sensitizer-assisted upconversion and energy transfer processes: Yb3+-based systems
In LiNbO3-based upconversion systems, Yb3+ primarily serves as a sensitizer that efficiently absorbs NIR light and transfers energy to activator ions such as Er3+ and Tm3+ [Figure 4E][74]. The luminescence of Yb3+ in LiNbO3 is governed by its simple two-manifold electronic structure, resulting in sharp NIR emission around 1.03 μm under excitation near 980 nm and enabling applications in solid-state lasers and infrared optical sources[93]. Owing to this simplified energy structure, Yb3+ exhibits reduced concentration quenching compared with other RE3+. Nevertheless, the emission efficiency and lifetime of Yb3+ in LiNbO3 remain sensitive to the local lattice environment, including site occupancy and nearby defects, which influence radiative and nonradiative processes[97,99].
To further enhance the luminescent performance of LiNbO3:Yb3+ systems, various strategies have been explored to improve efficiency, suppress nonradiative losses, and enhance stability. Montoya et al. reported green upconversion emission in LiNbO3:Yb3+ under NIR excitation [Figure 4F and G][93]. This emission was attributed to cooperative luminescence between neighboring Yb3+ ions, providing early evidence for short-range pairing in this nonlinear host. Based on cooperative luminescence, Demirkhanyan et al. developed an optical thermometry approach using LiNbO3:Yb3+ crystals[94]. Effective emission around 1,010 nm was observed over a temperature range of 200-500 K, with an average sensitivity of 0.12%K-1 [Figure 4G]. In addition, Ramírez et al. reported excitation-power-dependent hysteresis in the Yb3+ luminescence of LiNbO3 at low temperatures[100-102]. This behavior was attributed to laser-induced heating, which alters the distribution of excited states. The resulting intensity optical bistability highlights the potential of LiNbO3:Mg2+/Yb3+ crystals for optical switching applications.
Near-infrared luminescence and laser-relevant transitions: Nd3+-doped LiNbO3
In contrast to upconversion-dominated systems, neodymium (Nd3+)-doped LiNbO3 exhibits a characteristic NIR emission dominated by a strong band around 1,060 nm, accompanied by weaker emissions at longer and shorter wavelengths, which supports broadband laser and optical amplifier applications. Efficient excitation near 808 nm followed by nonradiative relaxation enables intense NIR emission[69,81]. The emission efficiency is further governed by host phonon coupling and the local crystal field, which regulate nonradiative decay processes.
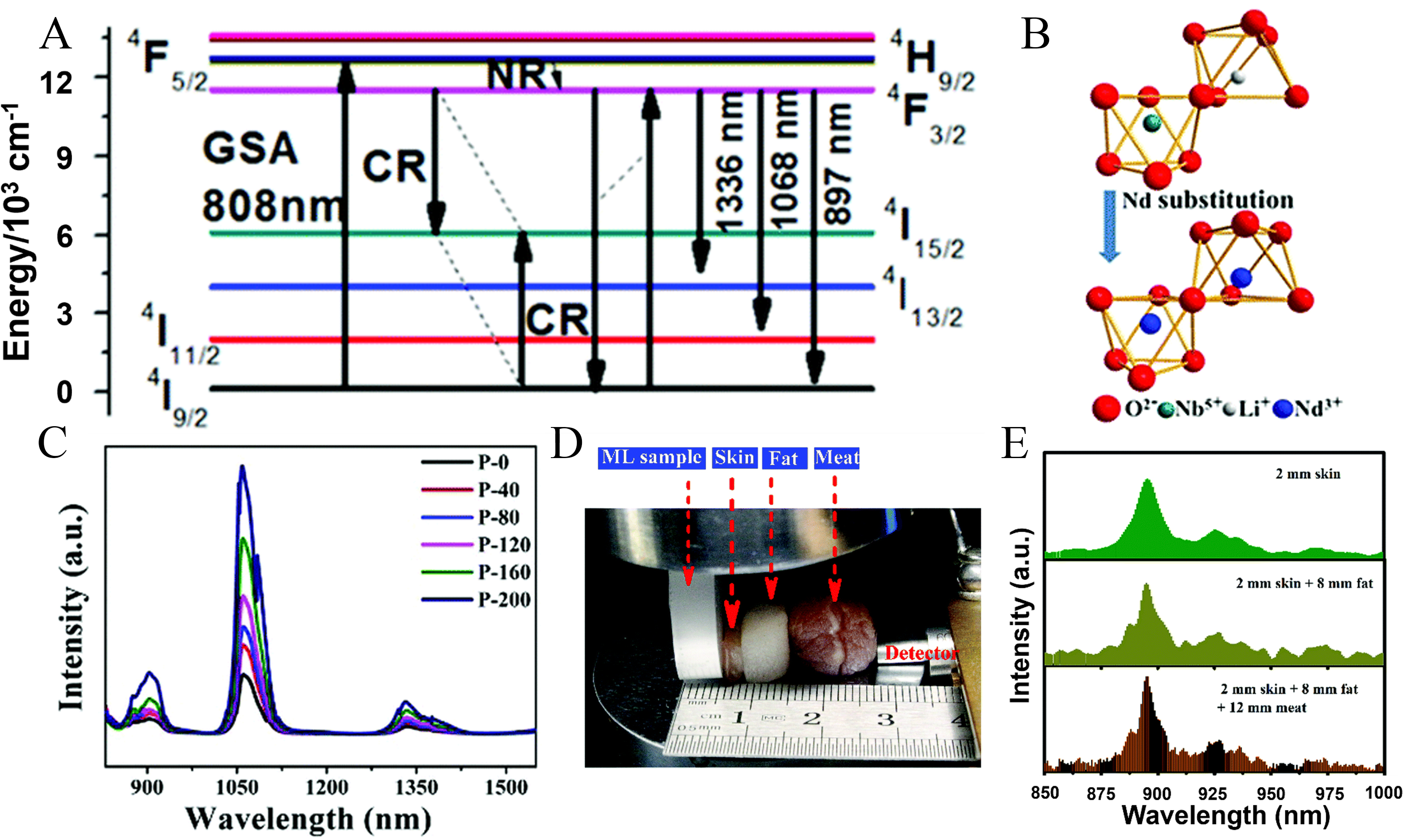
LiNbO3:Nd3+ is widely exploited in waveguide lasers, where Ti diffusion, proton exchange, or femtosecond laser inscription enables optical confinement, enhanced light matter interaction, and reduced lasing thresholds[103,104]. Pressure-dependent studies further reveal that increasing pressure causes a linear red shift and a decrease in Nd3+ emission intensity, reflecting modifications of the crystal field. The disappearance of multicenter emission above ~ 20 GPa indicates a pressure-driven evolution toward a more uniform, lower-symmetry local environment[105]. The associated energy transfer processes and structural evolution are summarized in Figure 5A and B. Pan et al. showed that ferroelectric LiNbO3 can actively modulate RE luminescence by constructing Nd3+-doped LiNbO3 nanocomposites[44]. The NIR emission was enhanced by up to 4.6 times [Figure 5C] and could be reversibly tuned under an external electric field. Polarization-induced internal fields and electric-field-driven symmetry modulation reshape the local crystal field and control carrier separation and recombination at Nd3+ centers, enabling electrically programmable luminescence for optical switching and information storage. Xiong et al. reported a novel Nd3+-doped LiNbO3 phosphor that exhibits intense and elastic NIR ML under compressive forces[11]. The ML intensity increases with applied force from 500 to 2,500 N and stabilizes under higher forces due to resin failure. Notably, both the onset and release of mechanical stress trigger ML emission, reflecting the transient evolution of internal piezoelectric fields during loading-unloading cycles. The efficient ML in LiNbO3:Nd3+ arises from the effective coupling between stress-induced piezoelectric field generation and Nd3+-induced local lattice distortion. Substitution of Nd3+ at Li+ sites modifies the local crystal field and defect environment, enhancing the efficiency of trapped carriers to piezoelectric field-driven de-trapping and radiative recombination. Elastic ML emission is observed at ~ 895 nm, within the first biological transparency window (650-950 nm), enabling tissue penetration depths exceeding 20 mm [Figure 5D and E].
Figure 5. (A) Energy-transfer mechanism of Nd3+ ions[44]; (B) Crystal structure of LiNbO3:Nd3+[44]; (C) Enhanced luminescence spectra under different electric fields[44]. Reproduced from Ref.[44] with permission from the Royal Society of Chemistry; Biological tissue penetration experiments: (D) The thickest tissue used in this test[11]; (E) ML spectra under 1,000 N after passing through different tissue samples:2 mm skin; 2 mm skin +8 mm fat; and 2 mm skin +8 mm fat +12 mm meat[11]. Reproduced from Ref.[11] with permission from the Royal Society of Chemistry. ML: Mechanoluminescence; GSA: ground state absorption; CR: cross relaxation; NR: non-radiative transition; Nd: neodymium.
Defect-coupled photoluminescence and mechanoluminescence: Pr3+-activated systems
Pr3+-doped LiNbO3 represents a distinct luminescent system characterized by intense red emission and defect-coupled stress-induced luminescence, arising from the interplay between Pr3+ electronic states and lattice defects. Pr3+ in LiNbO3 exhibits multicolor emission spanning blue (~ 483 nm), orange (~ 610 nm), and red (~ 643 nm) bands[46,68,106]. Excitation typically proceeds through UV or blue absorption followed by rapid nonradiative relaxation[10,68]. Emission efficiency is strongly governed by host phonon energy. The high-phonon-energy LiNbO3 lattice (~ 700 cm-1) suppresses multiphonon relaxation more effectively than oxide or phosphate glasses. Thermal quenching remains a challenge due to the relatively small energy gaps (~ 4,000-5,000 cm-1) between excited and lower-lying Pr3+ levels, but the high thermal and chemical stability of LiNbO3 partially mitigates this limitation[106].
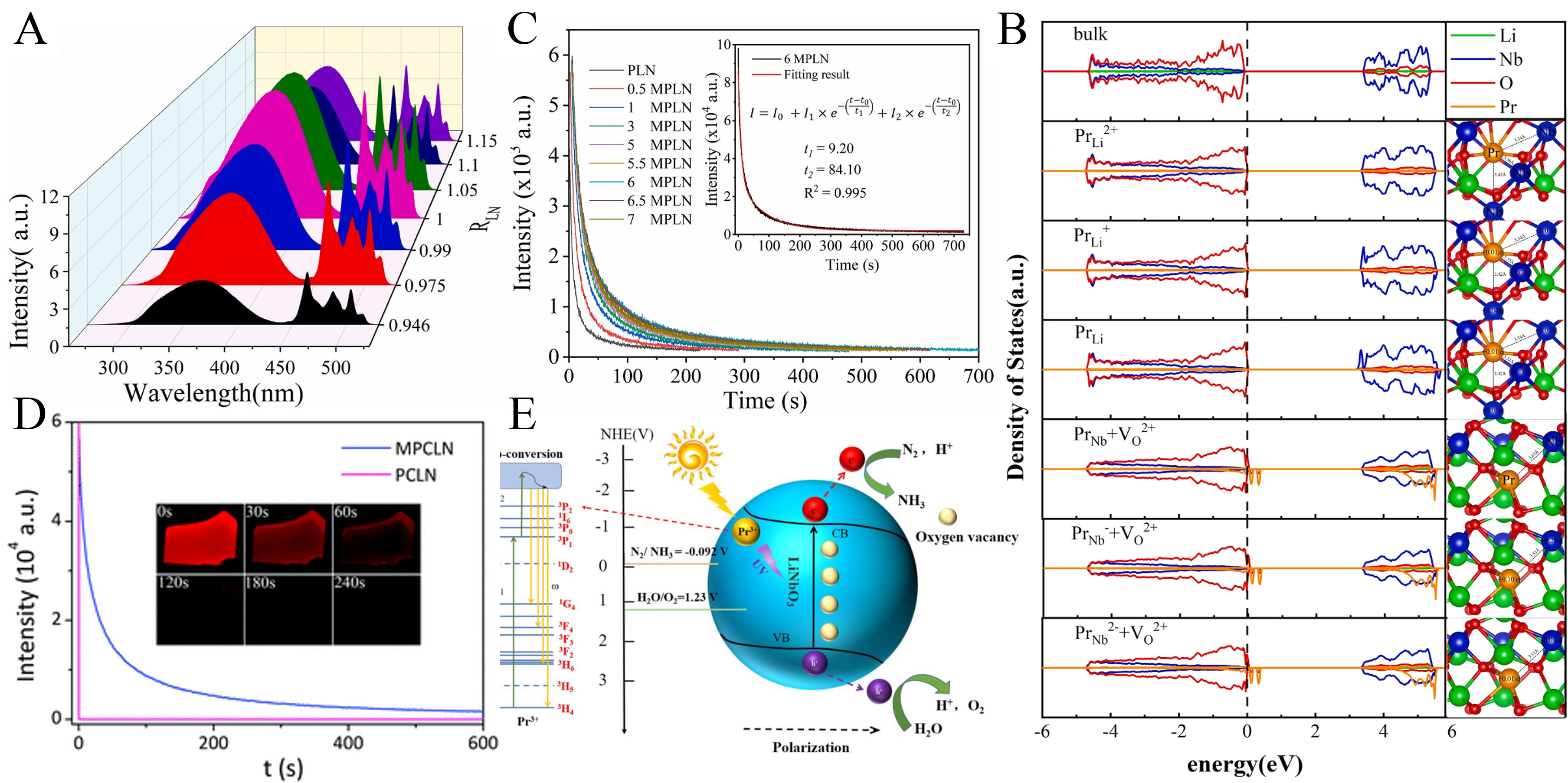
Xiong et al. demonstrated that LiNbO3:Pr3+[64], which initially shows no PersL, can exhibit pronounced PersL upon modulation of the RLN. Under 365 nm excitation, both PL intensity and afterglow duration first increase and then decrease with increasing RLN, reflecting RLN-induced changes in defect type and concentration. The formation of oxygen vacancies at elevated RLN was identified as a key factor enabling PersL [Figure 6A]. Structural, spectroscopic, and radioluminescence analyses further confirmed that RLN tuning effectively regulates trap depth and number, thereby modulating both PL and radioluminescence in the LiNbO3 matrix [Figure 6B][107]. To further enhance PersL, Mg2+ co-doping has proven effective. Hao et al. systematically investigated Mg2+ concentration effects in LiNbO3:Pr3+/Mg2+ polycrystals and proposed a microscopic charge transfer mechanism supported by combined experimental and computational analysis[29]. A semi-empirical dynamic model based on the Dorenbos framework was developed to rationalize the dependence of emission intensity and afterglow duration on dopant concentration [Figure 6C]. Consistently, Lin et al. reported room-temperature red afterglow in LiNbO3:Pr3+/Mg2+ single crystals[27], attributing PersL to charge relaxation via gap defect states associated with Mg2+ ions [Figure 6D]. Beyond luminescence, Sun et al. further demonstrated that LiNbO3:Pr3+ functions as an efficient photocatalyst for nitrogen fixation, where Pr3+ upconversion and intrinsic polarization synergistically enhance charge separation and enable a high
Figure 6. (A) Photoluminescence of the LixNbO3:1mol%Pr3+ sample[64]. Reproduced from Ref.[64] with permission from the Elsevier; (B) Spin-polarized partial density of states and Bader charges for bulk LiNbO3 and the sample doped with
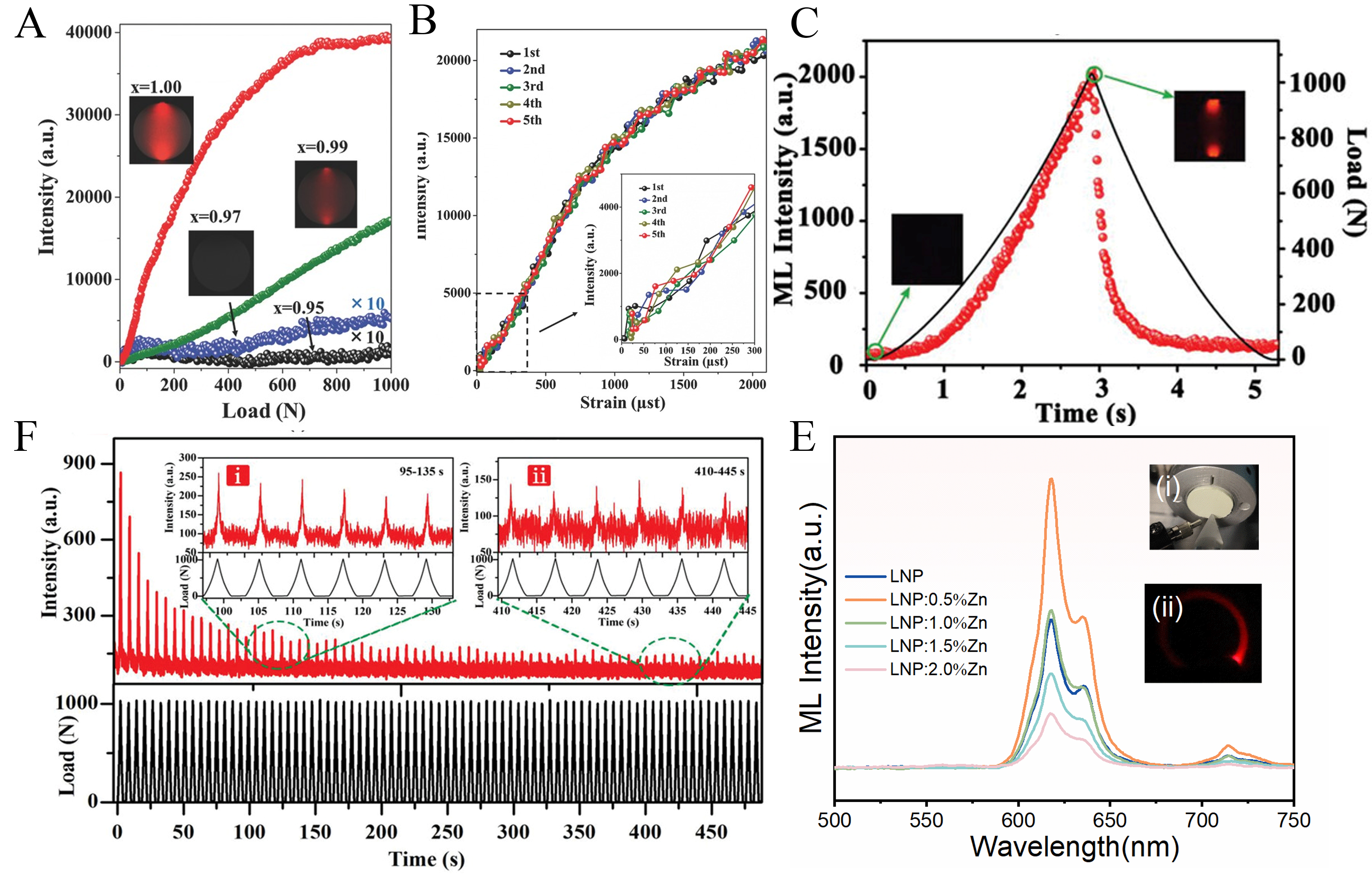
LiNbO3:Pr3+ exhibits distinctive ML, whereas similar behavior is rarely observed for other RE dopants. In 2017, Tu et al. first reported high-performance elastic ML in LiNbO3:Pr3+[10]. In this system, Pr3+ preferentially substitutes for Li+ sites, introducing charge-compensating defects that optimize trap distributions. Regulation of RLN effectively controls the number of defects and lattice distortion associated with Pr3+ incorporation, thereby enhancing carrier trapping and stress-induced release. As a result, nonstoichiometric LiNbO3:Pr3+ exhibits markedly enhanced ML and enables thresholdless stress sensing. Further enhancement is achieved through compositional and structural modulation. Yang et al.[47] and Hua et al.[109] developed Li1-xNaxNbO3:Pr3+ heterostructures that couple trap architecture with piezoelectric properties, enabling highly repeatable ML without pre-irradiation [Figure 7A-D]. Aliovalent co-doping with high field strength ions such as Gd3+ or divalent metal ions including Mg2+ and Zn2+ further tailors the local crystal field and defect chemistry[12,65]. Zn2+ incorporation specifically enhances the piezoelectric response and lowers the CTB energy, which facilitates charge transfer and carrier de-trapping. Consequently, LiNbO3:1%Pr3+/0.5%Zn2+ maintains strong ML even after 30 days of dark storage [Figure 7E][12], yielding high brightness, repeatability, and long-term stability. Similarly, Pr3+/Gd3+ co-doping increases the total trap concentration with minimal change in trap depth, resulting in a 177% enhancement in ML intensity compared to singly doped LiNbO3[65].
Figure 7. (A) The image of the Li0.5Na0.5NbO3:Pr3+ pellet i) before load and ii) loading[47]; (B) Comparison of experimental and simulated stress distribution along Y’OY of the Li0.5Na0.5NbO3:Pr3+ pellet under a compressive load of 1,000 N. Inset: schematic of the sample[47]; (C) ML response of Li0.5Na0.5NbO3:Pr3+ pellet under loading applied in a triangular wave mode[47]; (D) Stability and repeatability test of Li0.5Na0.5NbO3:Pr3+ pellet more than 50 load cycles: i) Real-time ML intensity during 95-135 s; ii) Real-time ML intensity during 410-445 s[47]. Reproduced from Ref.[47] with permission from the John Wiley and Sons; (E) ML spectra of LiNbO3:Pr3+/xZn2+: (i) before the test; (ii) under mechanical loading[12]. Reproduced from Ref.[12] with permission from the Elsevier. Pr: Praseodymium; ML: mechanoluminescence; LNP: LiNbO3:Pr3+; LiNbO3: lithium niobate.
Overall, the ML enhancement strategies discussed above originate from the coordinated coupling of piezoelectric field generation, defect engineering, and energy level modulation in LiNbO3. The pronounced anisotropy observed in Pr3+- and Nd3+-activated LiNbO3 reflects the combined influence of lattice distortion, optimized trap distributions, and crystal field. Within this framework, RLN stoichiometry tuning controls intrinsic defect populations, Zn2+ co-doping strengthens the piezoelectric response while increasing trap number and lowering the CTB energy, Gd3+ co-doping raises trap concentration, and heterostructure design couples trap architectures with piezoelectric properties to enhance carrier transport. Despite differing strategies, all these approaches converge on a common requirement for high-performance ML: rationally distributed traps that are effectively coupled to stress-induced piezoelectric fields. Although ML shares defect-regulated processes with PL, UCL, PersL, and TL, its distinctive feature is the stress-induced piezoelectric field in LiNbO3, which directly triggers carrier de-trapping without relying entirely on optical or thermal excitation.
Other rare earth dopants and emerging multimodal luminescence behaviors
Beyond the commonly investigated Er3+, Yb3+, Nd3+, and Pr3+ ions, other RE elements such as Sm3+, Ho3+, Tm3+, Eu3+, Tb3+, and Dy3+ have been explored for their unique emission characteristics, offering additional spectral diversity and multifunctionality for photonic device applications.
Beyond Er3+, Ho3+ and Tm3+ are also effective upconversion activators owing to their ladder-like energy level structures that favor sequential photon absorption. Because these level spacings are well matched with the Yb3+ excited-state transition, efficient sensitized upconversion is readily achieved in Er3+-Yb3+, Ho3+-Yb3+, and Tm3+-Yb3+ co-doped LiNbO3 systems, where Yb3+ acts as an efficient sensitizer[20,110]. LiNbO3:Tm 3+ exhibits broadband emission from the UV to the NIR. Cantelar et al. showed that the dominant emission pathway under excitation near 800 nm strongly depends on Tm3+ concentration[111], reflecting concentration-controlled upconversion dynamics [Figure 8A]. LiNbO3:Ho3+ exhibits multicolor upconversion emission under NIR excitation, with enhanced performance reported in Mg2+ co-doped single crystals grown by the Czochralski method [Figure 8B and C][19]. White upconversion with power-dependent color tuning was further achieved in Ho3+, Yb3+, and Tm3+ tri-doped LiNbO3 single crystals under 980 nm excitation, where multiple multiphoton processes coexist[18]. Similar multiband upconversion behavior has also been observed in LiNbO3:Hf3+/Dy3+ crystals, in which sequential excitation under NIR pumping produces broadband emission. In these systems, increased RLN enhance upconversion intensity by modifying local crystal fields around RE ions through Li+ incorporation[112].
Figure 8. (A) Upconversion emission of LiNbO3:Tm3+ under two excitation wavelengths. The inset shows the relevant energy levels and optical transitions[111]. Reproduced from Ref.[111] with permission from the Elsevier; (B) Upconversion emission spectra of LiNbO3:Mg2+/Ho3+ with varying Mg concentrations under 808 nm excitation[19]; (C) Energy levels of Ho3+ ion as well as the proposed upconversion mechanism in LiNbO3:Mg2+,Ho3+[19]. Reproduced from Ref.[19] with permission from the Chinese Physics B; (D) CIE coordinates of LiNbO3:Dy3+/Sm3+ phosphors under near-UV excitation, with inset photographs taken under 365 nm[41]. Reproduced from Ref.[41] with permission from the Elsevier; (E) Temperature-dependent luminescence spectra of LiNbO3:Sm3+ under 402 nm excitation[114]; (F) Temperature-dependent luminescence spectra of LiNbO3:Dy3+ under 356 nm excitation[114]. Adapted from Lisiecki et al.[114] under the Creative Commons Attribution 4.0 International (CC BY 4.0) license. LiNbO3: Lithium niobate; CIE: Commission Internationale de l’Éclairage; UV: ultraviolet; Tm: thulium; Ho: holmium; Dy: dysprosium; Sm: samarium.
Sm3+ ions exhibit characteristic orange-to-red emission spanning 560-650 nm. In LiNbO3, they incorporate well and display stable luminescence under UV or blue excitation, with sharp emission lines suitable for spectral conversion and optical and thermal sensing. Additionally, Sm3+ serves as an effective probe of lattice site occupation and symmetry, owing to its sensitivity to the local crystal field. Min et al. prepared single-phase LiNbO3:Sm3+ and LiNbO3:Sm3+,Dy3+ phosphors by solid-state reaction[41]. Under 412 nm excitation, LiNbO3:Sm3+ exhibits orange-red emission, while 392 nm excitation of LiNbO3:Sm3+/Dy3+ enables tunable PL from cool to warm white [Figure 8D]. Notably, Li0.97NbO3:0.02Dy3+/0.01Sm3+ displays high thermal stability with an activation energy of 0.15 eV, supporting its potential for warm white light-emitting diode (LED) applications. Li et al. reported multi-wavelength upconversion from LiNbO3:Tb3+ crystals under 800 nm femtosecond excitation, yielding blue, green, yellow, and red emissions[113]. Under continuous irradiation, the emission intensity decreases markedly due to laser-induced heating and the strong temperature sensitivity of Tb3+ luminescence. Lisiecki et al. further showed that LiNbO3:Sm3+/Tb3+/Dy3+ crystals exhibit pronounced temperature-dependent luminescence lifetimes[114], with abrupt quenching above ~ 700 K, ~ 600 K, and ~ 150 K, respectively, driven by thermally activated charge transfer processes. These behaviors define distinct sensing windows: LiNbO3:Sm3+ is suited for 500-750 K sensing, and LiNbO3:Dy3+ offers high sensitivity in the 300-400 K range [Figure 8E and F].
Incorporating RE3+ as luminescent centers endows LiNbO3 with diverse emission modes, including UCL, ML, PersL and TL. Upconversion originates from stepwise multiphoton absorption, exemplified by Yb3+-Er3+ systems where Yb3+ sensitization enables visible emission under 980 nm excitation. ML is driven by stress-induced internal electric fields that release trapped carriers, with emission influenced by stress level and defect distribution. PersL and TL depend on carrier exchange between shallow or deep traps and RE centers, enabling energy storage and temperature sensing. Collectively, RE doping substantially expands the optical functionality of LiNbO3. The preparation routes, excitation and emission wavelengths, and luminescence modes of RE doped LiNbO3 phosphors are summarized in Table 2.
Effective luminescent dopants, primary emission peaks and corresponding luminescence modes of LiNbO3-based materials
| Dopants | Preparation method | ex (nm) | em (nm) | Mode (PL/PersL/UCL/DCL/ML/TL) | Refs. |
| Pr3+ | Solid-state method | 254, 365 | ~ 620 | PL, DCL, TL, ML, PersL | [10,39,64,74,107] |
| Pr3+ | Sol-gel method | 550 | 283.9 | UCL | [108] |
| Pr3+,Er3+ | Solid-state method | 370, 980 | 520-560, 618 | PL, UCL, ML | [39] |
| Pr3+,Mg2+ | Solid-state method | 365 | 619 | PL, PersL | [27,29] |
| Pr3+,Zn2+ | Solid-state method | 334 | 616 | PL, ML | [12] |
| Pr3+,Gd3+ | Solid-state method | 280 | 618 | PL, ML | [65] |
| Nd3+ | Melting-quenching method | 808 | 897, 1,068, 1336 | PL | [44] |
| Nd3+ | Solid-state method | / | 895, 926, 936 | ML | [11] |
| Er3+ | Solid-state method | 980 365 | ~550 | UCL, DCL | [74] |
| Er3+,Mg2+ | Czochralski method | 300 | ~550 | PL | [95] |
| Er3+,Yb3+ | Czochralski method, Co-precipitation method | 808, 980 | ~525, ~550, ~670 | PL, UCL | [25,38,96-99] |
| Er3+,Ho3+,Tm3+ | Czochralski method | 800 | 475, 582, 653, 749 | PL, UCL | [112] |
| Tm3+ | Solid-state method/ Czochralski method | 365, 795 | 440-500 | PL, DCL | [74,111] |
| Tm3+, Yb3+, Ho3+, Mg2+ | Solid-state method /Czochralski method | 980 | 477, 549, 652, 665 | PL, UCL | [18,20,74,110] |
| Yb3+ | Czochralski method | 920 | 981 | UCL | [93] |
| Ho3+, Mg2+ | Czochralski method | 808 | 425, 561, 725 | PL, UCL | [19] |
| Dy3+ | Solid-state method /Czochralski method | 392, 800 | 488, 585 | PL | [22,41,114] |
| Sm3+ | Solid-state method /Czochralski method | 412 | 613, 650 | PL | [26,41,114] |
| Tb3+ | Czochralski method | 800 | 487, 544, 582, 623 | UCL | [113] |
LINBO3-BASED COMPOSITES: LUMINESCENT PROPERTIES AND APPLICATIONS
To further enhance the optical properties and functionalities of LiNbO3, researchers have developed a variety of LiNbO3-based composite materials by incorporating luminescent nanoparticles, RE oxides, quantum dots, or plasmonic nanostructures into the LiNbO3 matrix[47,115,116]. These composites are commonly fabricated using various material processing methods, including sol-gel synthesis, ion implantation, solid-state reaction, and thin-film deposition techniques [Table 3].
LiNbO3-based composites: preparation methods and optical properties
| Composition | Classification | Preparation method | Optical performance | Refs. |
| LiNbO3:Nd3+ | Nanocomposites | Melt crystallization method | > 4.6 times NIR luminescence enhancement | [44] |
| LiNbO3:Ni2+ | Nano materials | Melt-quenching technique | Broadband emission increased by up to 35%. | [115] |
| LiNbO3:Yb3+/Er3+ | Nano materials | Hydrothermal method | Two orders of magnitude UCL enhancement | [117] |
| LiNbO3 | Single crystals | Ion implantation (Kr+, Ag+, and Er3+) | Emergence of ML in LiNbO3 (absent in single crystals) | [116] |
| LiNbO3:Pr3+/Nd3+ and epoxy resin | Composites | High temperature solid-state method | Emergence of ML in composites (absent in pure powder) | [10,12,47] |
| LiNbO3:Er3+ | Single crystal films | E-beam evaporation technique | Green, red, and near-infrared emissions are enhanced | [118] |
| LiNbO3:Er3+ | Thin films | Lithium-rich vapor transport equilibrium | ~ 10 times enhancement in visible & NIR emission | [119] |
| LiNbO3:Er3+ | On-chip ridge waveguides | Damond blade dicing technique | Dual-color UCL | [120] |
| LiNbO3:Er3+/Yb3+ | Planar waveguides | Sol-gel method | The infrared emission is enhanced | [121] |
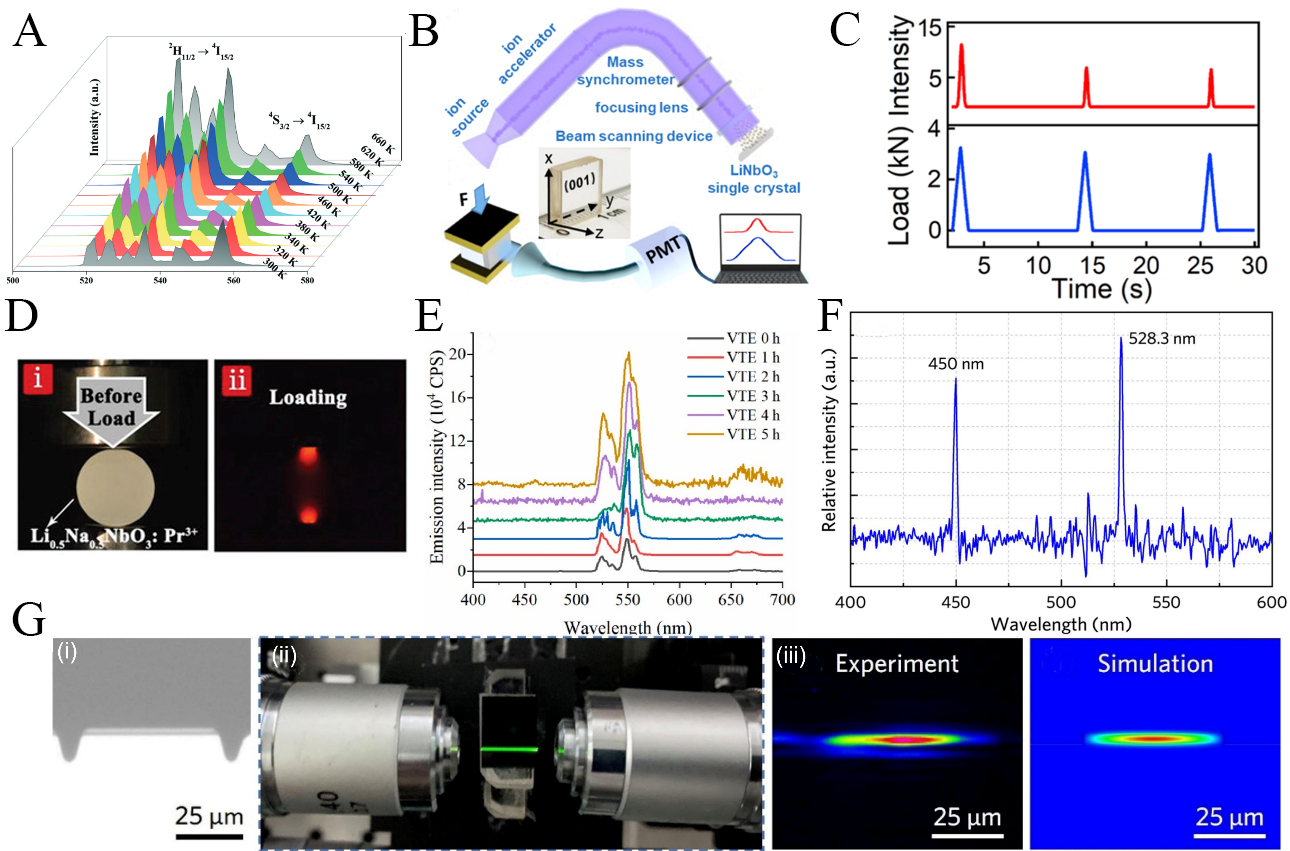
Pan et al. synthesized Nd3+-doped LiNbO3 ferroelectric nanocomposites via melt crystallization and achieved over a 4.6 times enhancement of NIR emission at room temperature[44]. These results demonstrate that embedding LiNbO3 nanocrystals in a glass matrix effectively amplifies optical performance by coupling ferroelectric functionality with nanoscale composite architectures. Using melt quenching, the research team introduced Ni2+ luminescent centers into transparent LiNbO3-based nano thermoelectric photonic composites[115], producing ultrabroadband NIR emission whose intensity increased by up to 35% under an applied electric field. The tunability was attributed to reversible field-induced nanoscale structural changes that regulate both emission intensity and wavelength. Tong et al. further reported LiNbO3:Yb3+/Er3+ nanocubes synthesized hydrothermally[117], where increased annealing temperature enhanced green and red upconversion by nearly two orders of magnitude due to reduced surface quenching and improved dopant incorporation. These nanocubes also exhibited effective optical thermometry, achieving a maximum sensitivity of 0.0094 K-1 at 340 K [Figure 9A].
Figure 9. (A) Temperature-dependent UC emission spectra of LiNbO3-1050 sample at 300-660 K under 980 nm excitation[117]. Reproduced from Ref.[117] with permission from the Royal Society of Chemistry; (B) Schematic of the experimental setups for ion irradiation and ML measurement[116]; (C) ML property of a Kr+-irradiated LiNbO3 single crystal under three loading/unloading cycles[116]; Reproduced from Ref.[116] with permission from the American Chemical Society; (D) Photographs of Li0.5Na0.5NbO3:Pr3+ pellet before loading and under compressive stress[47]. Reproduced from Ref.[47] with permission from the John Wiley and Sons; (E) Visible fluorescence spectra of VTE 0-5 h samples measured at 30 ℃[119]; (F) The room-temperature upconversion fluorescence spectra from LiNbO3:Er3+ on-chip ridge waveguides[119]. Reproduced from Ref.[119] with permission from the Elsevier; (G) Optical characterization of Er-doped LNOI ridge waveguides, including a microscopic image of the waveguide facet, green upconversion emission under 980 nm excitation, and experimental and simulated mode profiles at 1,550 nm[120]. Reproduced from Ref.[120] with permission from the Elsevier. LiNbO3: Lithium niobate; Pr: praseodymium; VTE: vapor transport equilibration; ML: mechanoluminescence; Er: Erbium; LNOI: lithium-niobate-on-insulator.
Chen et al. introduced oxygen vacancies into LiNbO3 single crystals through ion irradiation using Kr+, Ag+, and Er3+ ions[116], thereby endowing the crystals with ML [Figure 9B and C]. Studies on ion-irradiated LiNbO3 single crystals unambiguously demonstrate that ML is driven by piezoelectricity. Under mechanical stress, piezopotentials tilt the band structure and promote electron de-trapping from oxygen vacancy-related states, followed by radiative recombination with valence-band holes. In this process, piezoelectric fields function as the primary trigger for light emission, while defects serve as carrier storage and mediation centers. In addition, LiNbO3:Pr3+/Nd3+ exhibits distinct ML behavior [Figure 9D][10,12,47]. However, pristine LiNbO3:Pr3+/Nd3+ phosphors do not show detectable ML signals. Observable stress-induced emission emerges only after incorporation into an optical epoxy matrix to form mechanically responsive composites, typically as pellets under applied load, underscoring the essential role of composite architecture in enabling ML performance.
Light confinement in thin films enhances nonlinear optical responses and favors applications in modulators, switches, and sensors, while RE doping further improves luminescent performance in LiNbO3 films through controlled dopant incorporation and defect regulation. Xue et al. prepared LiNbO3:Er3+ single-crystal films on LiTaO3 substrates and observed enhanced visible and NIR emission with increasing Er3+ concentration[118]. Near-stoichiometric LiNbO3:Er3+ thin films obtained by lithium-rich vapor transport equilibrium treatment showed further emission enhancement, which increased with treatment duration and was attributed to suppression of Nb-related defects [Figure 9E and F][119]. Importantly, Jia et al. demonstrated on-chip upconversion from Er3+-doped LiNbO3 ridge waveguides with blue and green emission under 980 nm excitation, confirming high optical quality and integration potential [Figure 9G][120]. However, Planar LiNbO3:Er3+/Yb3+ waveguides also exhibited enhanced infrared emission, although excessive crystallization reduced transparency and waveguiding due to changes in film structure and local crystal fields[121]. These studies collectively indicate that emission efficiency in LiNbO3 thin film photonic devices is governed by the interplay between RE activation, defect suppression, and waveguide integrity, rather than dopant concentration alone.
CURRENT CHALLENGES AND POSSIBLE FUTURE DEVELOPING DIRECTIONS
LiNbO3 is a representative nonlinear optical and ferroelectric material that has attracted increasing interest for UCL, ML, NIR emission, and integrated optoelectronic devices, owing to its high optical transparency, wide bandgap, and compatibility with RE doping. Although undoped LiNbO3 exhibits weak intrinsic luminescence, broadband and tunable emission can be achieved through RE incorporation or co-doping with modulation ions such as Yb3+, Zn2+, and Mg2+. Despite this progress, LiNbO3-based luminescent materials still face several critical challenges [Figure 10]. Luminescence efficiency remains limited by the difficulty of precisely controlling intrinsic and extrinsic defects. At high dopant concentrations, emission efficiency in LiNbO3 is limited by concentration quenching and defect-induced nonradiative recombination, which jointly reduce energy transfer efficiency, shorten excited state lifetimes, and compromise optical performance.
Figure 10. Schematic illustration of the key challenges in LiNbO3-based luminescent materials. LiNbO3: Lithium niobate.
In addition, the mechanisms governing multi-ion-doped and stress-responsive luminescence are not yet fully clarified due to the complex coupling among energy transfer, defect trapping, and piezoelectric field-driven carrier dynamics. In LiNbO3 and related piezoelectric hosts, limited understanding of carrier trapping and release processes hinders the establishment of a unified mechanistic framework, highlighting the need for high quality crystals with minimized structural disorder. Furthermore, environmental and operational stability remains insufficient for practical application. Emission quenching and structural degradation occur under high optical pumping, elevated temperatures, or cyclic mechanical loading, while the effects of humidity, thermal cycling, and mechanical fatigue on ML and PersL have not been systematically evaluated. Finally, large-scale integration into on-chip photonic architectures is restricted by lattice mismatch, limited compatibility with silicon-based or flexible platforms, and the prohibitive cost and low scalability of single-crystal fabrication.
Future efforts should focus on defect engineering that combines precise stoichiometric control with trap state modulation to improve luminescence efficiency and reproducibility in LiNbO3. At the same time, advances in high-quality crystal growth and low-temperature thin-film fabrication with controllable defects will be essential for clarifying mechanical-optical-piezoelectric coupling mechanisms and enabling scalable, integrated LiNbO3 photonic systems.
CONCLUSION
In summary, LiNbO3-based luminescent materials demonstrate extraordinary potential across photonics, sensing, and stress-responsive applications. However, addressing challenges in luminescence efficiency, defect regulation, integration strategies, stability, scalability, and mechanistic understanding will be essential to fully realize this potential. Future advances in rational material design, advanced in situ characterization, defect and interface engineering, and scalable fabrication are expected to drive the development of next-generation LiNbO3-based optoelectronic and mechanoluminescent devices with high performance, stability, and multifunctionality.
DECLARATIONS
Authors’ contributions
Made substantial contributions to literature collection, conceptual framework design, and manuscript drafting: Yao, X.; Mu, X.
Provided scientific supervision, critical revision, and intellectual input throughout the writing process: Jiang, M.; Zhao, J.
All authors contributed to the discussion and approved the final version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (52172001), Guangxi Natural Science Foundation (2022GXNSFDA035082), the Central Government Guided Local Science and Technology Development Fund Project (ZY22096009), and the Innovation Project of GUET Graduate Education (2024YCXB03).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Wang, M.; Xu, Y.; Fang, Z.; et al. On-chip electro-optic tuning of a lithium niobate microresonator with integrated in-plane microelectrodes. Opt. Express. 2017, 25, 124-9.
2. Presti, D.; Guarepi, V.; Videla, F.; Fasciszewski, A.; Torchia, G. Intensity modulator fabricated in LiNbO3 by femtosecond laser writing. Opt. Lasers. Eng. 2018, 111, 222-6.
3. Chauvet, M.; Henrot, F.; Bassignot, F.; et al. High efficiency frequency doubling in fully diced LiNbO3ridge waveguides on silicon. J. Opt. 2016, 18, 085503.
4. Nguyen, K.; Chulukhadze, V.; Stolt, E.; et al. Near spurious-free thickness shear mode lithium niobate resonator for piezoelectric power conversion. IEEE. Trans. Ultrason. Ferroelectr. Freq. Control. 2023, 70, 1536-43.
5. Chen, H.; Chen, X.; Zhang, Y.; Xia, Y. Polarization-dependent coupling in a 1 × 4 waveguide array in lithium niobate fabricated by femtosecond pulses. Appl. Phys. B. 2007, 89, 25-8.
6. Saglamyurek, E.; Sinclair, N.; Jin, J.; et al. Broadband waveguide quantum memory for entangled photons. Nature 2011, 469, 512-5.
7. Ródenas, A.; Zhou, G.; Jaque, D.; Gu, M. Rare-earth spontaneous emission control in three‐dimensional lithium niobate photonic crystals. Adv. Mater. 2009, 21, 3526-30.
8. Chen, K.; Zhu, Y.; Liu, Z.; Xue, D. State of the art in crystallization of LiNbO3 and their applications. Molecules 2021, 26, 7044.
9. Huang, Z.; Tu, C.; Zhang, S.; et al. Femtosecond second-harmonic generation in periodically poled lithium niobate waveguides written by femtosecond laser pulses. Opt. Lett. 2010, 35, 877-9.
10. Tu, D.; Xu, C. N.; Yoshida, A.; Fujihala, M.; Hirotsu, J.; Zheng, X. G. LiNbO3:Pr3+: a multipiezo material with simultaneous piezoelectricity and sensitive piezoluminescence. Adv. Mater. 2017, 29, 1606914.
11. Xiong, P.; Peng, M. Near infrared mechanoluminescence from the Nd3+ doped perovskite LiNbO3:Nd3+ for stress sensors. J. Mater. Chem. C. 2019, 7, 6301-7.
12. Yao, X.; Li, L.; Liu, W.; et al. Effective long-term storage and repeatable mechanoluminescence of piezoelectrics LiNbO3:1%Pr3+, 0.5%Zn2+. J. Materiomics. 2025, 11, 101072.
13. Cordero-Edwards, K.; Domingo, N.; Abdollahi, A.; Sort, J.; Catalan, G. Ferroelectrics as smart mechanical materials. Adv. Mater. 2017, 29, 1702210.
14. Kong, Y.; Bo, F.; Wang, W.; et al. Recent progress in lithium niobate: optical damage, defect simulation, and on-chip devices. Adv. Mater. 2020, 32, e1806452.
15. Sun, W.; Zhang, Z.; Sun, H.; et al. Crystalline phase, profile characteristics and spectroscopic properties of Er3+/Tm3+ -diffusion-codoped LiNbO3 crystal. J. Lumin. 2017, 184, 191-8.
16. Xing, L.; Xu, Y.; Wang, R.; Xu, W.; Zhang, Z. Highly sensitive optical thermometry based on upconversion emissions in Tm3+/Yb3+ codoped LiNbO3 single crystal. Opt. Lett. 2014, 39, 454-7.
17. Bredillet, K.; Riporto, F.; Guo, T.; et al. Dual second harmonic generation and up-conversion photoluminescence emission in highly-optimized LiNbO3 nanocrystals doped and co-doped with Er3+ and Yb3. Nanoscale 2024, 16, 6739-47.
18. Xing, L.; Wu, X.; Wang, R.; Xu, W.; Qian, Y. Upconversion white-light emission in Ho3+/Yb3+/Tm3+ tridoped LiNbO3 single crystal. Opt. Lett. 2012, 37, 3537-9.
19. Dai, L.; Xu, C.; Zhang, Y.; Li, D.; Xu, Y. Influences of Mg2+ ion on dopant occupancy and upconversion luminescence of Ho3+ion in LiNbO3 crystal. Chinese. Phys. B. 2013, 22, 094201.
20. Xing, L.; Yang, W.; Lin, J.; Huang, M.; Xue, Y. Enhanced and stable upconverted white-light emission in Ho3+/Yb3+/Tm3+-doped LiNbO3 single crystal via Mg2+ ion doping. Sci. Rep. 2017, 7, 14725.
21. Sun, L.; Wang, B.; Xing, G.; Liang, C.; Ma, W.; Yang, S. Bi-induced photochromism and photo-stimulated luminescence with fast photochromic response for multi-mode dynamic anti-counterfeiting and optical information storage. Chem. Eng. J. 2023, 455, 140752.
22. Yang, S.; Dai, L.; Yuan, Y.; Yuan, H. Up-conversion luminescence and defect structures in Sc: Dy: LiNbO3 crystals affected by Sc3+ concentration. Opt. Mater. 2023, 143, 114196.
23. Li, A.; Lü, Q.; Zheng, Z.; et al. Yellow-green upconversion luminescence of Dy3+ ion in LiNbO3 crystal heavily codoped with ZnO. J. Appl. Phys. 2007, 102, 113102.
24. Xing, L.; Yang, W.; Ma, D.; Xue, Y.; Wang, B. White-light manipulation in Ho3+/Yb3+/Tm3+-doped LiNbO3 single crystals through transition metal Mn2+ ion doping. J. Alloys. Compd. 2017, 714, 1-5.
25. Quintanilla, M.; Cantelar, E.; Cussó, F.; Villegas, M.; Caballero, A. C. Temperature sensing with up-converting submicron-sized LiNbO3:Er3+/Yb3+ articles. Appl. Phys. Express. 2011, 4, 022601.
26. Lee, D.; Lee, Y. Bi3+-driven defect formation and related optical multimode in luminescent ferroelectrics Sm3+-doped LiNbO3. J. Lumin. 2024, 276, 120879.
27. Lin, S.; Xiong, C.; Ma, D.; Li, H.; Long, S.; Wang, B. Persistent luminescence found in Mg2+ and Pr3+ co-doped LiNbO3 single crystal. J. Mater. Chem. C. 2018, 6, 10067-72.
28. Nakamura, M.; Higuchi, S.; Takekawa, S.; Terabe, K.; Furukawa, Y.; Kitamura, K. Optical damage resistance and refractive indices in near-stoichiometric MgO-doped LiNbO3. Jpn. J. Appl. Phys. 2002, 41, L49-51.
29. Hao, R.; Xiong, C.; Li, H.; et al. Optimization of persistent luminescence via dopant concentration in LiNbO3. J. Lumin. 2022, 244, 118753.
30. Volk, T. R.; Pryalkin, V. I.; Rubinina, N. M. Optical-damage-resistant LiNbO3:Zn crystal. Opt. Lett. 1990, 15, 996-8.
31. Palatnikov, M.; Sidorov, N.; Panasjuk, S.; Teplyakova, N.; Makarova, O. Radiation modification of optical characteristics of LiNbO3:Zn and LiNbO3:Mg crystals. Crystals 2022, 12, 600.
32. Peithmann, K.; Wiebrock, A.; Buse, K. Photorefractive properties of highly-doped lithium niobate crystals in the visible and near-infrared. Appl. Phys. B:. Lasers. Opt. 1999, 68, 777-84.
33. Tian, T.; Kong, Y.; Liu, S.; et al. Photorefraction of molybdenum-doped lithium niobate crystals. Opt. Lett. 2012, 37, 2679-81.
34. Kong, Y.; Liu, S.; Xu, J. Recent Advances in the photorefraction of doped lithium niobate crystals. Materials 2012, 5, 1954-71.
35. Xu, L.; Chen, G. Optimization of blue photorefractive properties and exponential gain of photorefraction in Sc-doped Ru:Fe:LiNbO3 crystals. Crystals 2022, 12, 1059.
36. Li, L.; Li, Y.; Zhao, X.; Cheng, X.; Fan, Y. Abnormal lattice occupation of Mo and related polarons in LiNbO3: hybrid density functional theory investigation. J. Materiomics. 2020, 6, 183-91.
37. Kong, Y.; Liu, S.; Zhao, Y.; Liu, H.; Chen, S.; Xu, J. Highly optical damage resistant crystal: zirconium-oxide-doped lithium niobate. Appl. Phys. Lett. 2007, 91, 081908.
38. Tian, X.; Qi, Q.; Hou, B.; Qian, Y. Hydrothermal synthesis and upconversion luminescence of cubic-shaped LiNbO3:Yb3+/Er3+ nanocrystals. Inorg. Chem. Commun. 2023, 157, 111389.
39. Lin, N.; Wang, J.; Xiao, Z.; et al. Stimuli-responsive lanthanide activated piezoelectric LiNbO3 microcrystals for multimode luminescence and optical sensing applications. Laser. Photonics. Rev. 2024, 18, 2301352.
40. Hu, Y.; Gao, D.; Zhang, X.; Yun, S. Controllable thermo-stimulated luminescence in niobate persistent phosphor by constructing the photovoltaic/electrolytic cell for remote intelligent anti-counterfeiting. Chem. Eng. J. 2025, 509, 161181.
41. Min, Z.; Zeng, Q.; Chen, S.; Qin, Y.; Yao, C. Tunable photoluminescence of LiNbO3: RE3+ (RE3+ = Dy3+, Sm3+, Dy3+/Sm3+) single-phase phosphors for warm white LEDs. J. Alloys. Compd. 2022, 924, 166497.
42. Xu, L.; Xu, Y.; Chen, G. Defect Structure and upconversion luminescence properties of LiNbO3 highly doped congruent in:Yb:Ho:LiNbO3 crystals. Crystals 2022, 12, 710.
43. Runowski, M.; Woźny, P.; Martín, I. R.; et al. Multimodal optically nonlinear nanoparticles exhibiting simultaneous higher harmonics generation and upconversion luminescence for anticounterfeiting and 8-bit optical coding. Adv. Funct. Mater. 2023, 34, 2307791.
44. Pan, E.; Bai, G.; Lei, L.; Zhang, J.; Xu, S. The electrical enhancement and reversible manipulation of near-infrared luminescence in Nd doped ferroelectric nanocomposites for optical switches. J. Mater. Chem. C. 2019, 7, 4320-5.
45. Liu, Z.; Ma, D.; Zhu, Y.; Lin, S.; Xiong, C.; Wang, B. Building a highly concentration responsive optical thermometer via tunable electron transfer pathways supported by intervalence charge transfer states. Spectrochim. Acta. A. Mol. Biomol. Spectrosc. 2025, 324, 124973.
46. Liu, Q.; Luo, Y.; Li, D.; et al. Heterostructure-induced enhanced persistent luminescence for low temperature applications. Chem. Eng. J. 2024, 498, 155346.
47. Yang, X.; Liu, R.; Xu, X.; et al. Effective Repeatable Mechanoluminescence in Heterostructured Li1-xNaxNbO3:Pr3+. Small 2021, 17, e2103441.
48. Uchiyama, T.; Atsumi, T.; Otonari, K.; Fujio, Y.; Zheng, X.; Xu, C. Direct recording and reading of mechanical force by afterglow evaluation of multi-piezo mechanoluminescent material Li0.12Na0.88NbO3 on well-designed morphotropic phase boundary. Appl. Phys. Lett. 2024, 124, 171105.
49. Zhang, B.; Wang, L.; Chen, F. Recent advances in femtosecond laser processing of LiNbO3 crystals for photonic applications. Laser. Photonics. Rev. 2020, 14, 1900407.
50. Xie, Z.; Bo, F.; Lin, J.; et al. Recent development in integrated Lithium niobate photonics. Adv. Phys-X. 2024, 9, 2322739.
51. Weis, R. S.; Gaylord, T. K. Lithium niobate: summary of physical properties and crystal structure. Appl. Phys. A. 1985, 37, 191-203.
52. Huang, B.; Sun, M.; Peng, D. Intrinsic energy conversions for photon-generation in piezo-phototronic materials: a case study on alkaline niobates. Nano. Energy. 2018, 47, 150-71.
53. Xie, H. R.; Yang, T. F. Wei,, Y. M.; Guan H. Y.; Lu, H. H. Recent research progress of thin film lithium niobate photodetector. J. Synth. Cryst. 2024, 533, 410-24.
54. Chakkoria, J. J.; Dubey, A.; Mitchell, A.; Boes, A. Ferroelectric domain engineering of Lithium niobate. Opto-Electron. Adv. 2025, 8, 240139.
55. Peterson, G. E.; Ballman, A. A.; Lenzo, P. V.; Bridenbaugh, P. M. Electro-optic properties of LiNbO3. Appl. Phys. Lett. 1964, 5, 62-4.
56. Smith, R. G.; Nassau, K.; Galvin, M. F. Efficient continuous optical second-harmonic generation. Appl. Phys. Lett. 1965, 7, 256-8.
57. Boes, A.; Chang, L.; Langrock, C.; et al. Lithium niobate photonics: unlocking the electromagnetic spectrum. Science 2023, 379, eabj4396.
58. Wiegel, M.; Blasse, G.; Navrotsky, A.; Mehta, A.; Kumada, N.; Kinomura, N. Luminescence of the ilmenite phase of LiNbO3. J. Solid. State. Chem. 1994, 109, 413-5.
59. Pankratov, V.; Grigorjeva, L.; Millers, D.; Corradi, G.; Polgàr, K. Luminescence of ferroelectric crystals: LiNbO3 and KNbO3. Ferroelectrics 2000, 239, 241-50.
60. Li, Y.; Schmidt, W. G.; Sanna, S. Intrinsic LiNbO3 point defects from hybrid density functional calculations. Phys. Rev. B. 2014, 89, 094111.
61. Uda, S.; Koyama, C. The population and activity of oxygen in the diffusion boundary layer within a congruent LiNbO3 melt. J. Cryst. Growth. 2020, 548, 125837.
62. Schmidt, F.; Kozub, A. L.; Gerstmann, U.; Schmidt, W. G.; Schindlmayr, A. Electron polarons in lithium niobate: charge localization, lattice deformation, and optical response. Crystals 2021, 11, 542.
63. Schirmer, O.; Thiemann, O.; Wöhlecke, M. Defects in LiNbO3 - I. experimental aspects. J. Phys. Chem. Solids. 1991, 52, 185-200.
64. Xiong, C.; Hao, R.; Tao, K.; Li, H.; Lin, S.; Ma, D. Tunable effect on persistent luminescence via lithium-to-niobium ratio in LiNbO3: Pr polycrystals. J. Lumin. 2024, 274, 120695.
65. Qiu, G.; Fang, H.; Wang, X.; Li, Y. Largely enhanced mechanoluminescence properties in Pr3+/Gd3+ co-doped LiNbO3 phosphors. Ceram. Int. 2018, 44, 15411-7.
66. Tokko, O. V.; Kadetova, A. V.; Prusskii, A. I.; Smirnov, M. V.; Palatnikov, M. N.; Sidorov, N. V. Structure and luminescent properties of double-doped LiNbO3:Zn:Mg crystals. Phys. Status. Solidi. A. 2024, 221, 2300796.
67. Murillo, J.; Herrera, G.; Vega-Rios, A.; Flores-Gallardo, S.; Duarte-Moller, A.; Castillo-Torres, J. Effect of Zn doping on the photoluminescence properties of LiNbO3 single crystals. Opt. Mater. 2016, 62, 639-45.
68. Zheng, B.; Fan, J.; Chen, B.; et al. Rare-earth doping in nanostructured inorganic materials. Chem. Rev. 2022, 122, 5519-603.
69. Dai, L.; Liu, C.; Han, X.; Wang, L.; Shao, Y.; Xu, Y. Effect of Ho3+ and Tm3+ concentration on UV-VIS-NIR and upconversion luminescence in Ho:Tm:LiNbO3 crystals. J. Alloys. Compd. 2019, 771, 960-3.
70. Zhang, J.; Long, Y.; Yan, X.; Wang, X.; Wang, F. Creating recoverable mechanoluminescence in piezoelectric calcium niobates through Pr3+ doping. Chem. Mater. 2016, 28, 4052-7.
71. Liang, Z.; Qin, F.; Zheng, Y.; Zhang, Z.; Cao, W. Noncontact thermometry based on downconversion luminescence from Eu3+ doped LiNbO3 single crystal. Sensor. Actuat. A-Phys. 2016, 238, 215-9.
72. Zhang, Z.; Tang, X.; Qian, Y.; Zhang, H.; Wang, W.; Wang, R. Multicolor tunable yellow-red emission in Eu/Er:LiNbO 3 under ultraviolet and blue excitation. Opt. Laser. Technol. 2017, 97, 111-5.
73. Ahmad, S.; Chang, S.; Peng, D.; et al. Quadra-mode luminescent phosphors for force/thermo-encoded information storage and anticounterfeiting applications. ACS. Appl. Mater. Interfaces. 2025, 17, 29901-9.
74. Sang, J.; Zhou, J.; Zhang, J.; et al. Multilevel static-dynamic anticounterfeiting based on stimuli-responsive luminescence in a niobate structure. ACS. Appl. Mater. Interfaces. 2019, 11, 20150-6.
75. Dong, H.; Sun, L. D.; Yan, C. H. Energy transfer in lanthanide upconversion studies for extended optical applications. Chem. Soc. Rev. 2015, 44, 1608-34.
76. Huang, K.; Qiu, H.; Zhang, X.; et al. Orthogonal trichromatic upconversion with high color purity in core-shell nanoparticles for a full-color display. Angew. Chem. Int. Ed. Engl. 2023, 62, e202218491.
77. Riwotzki, K.; Meyssamy, H.; Schnablegger, H.; Kornowski, A.; Haase, M. Liquid-phase synthesis of colloids and redispersible powders of strongly luminescing LaPO4:Ce,Tb nanocrystals. Angew. Chem. Int. Ed. 2001, 40, 573-6.
78. Lemański, K.; Dereń, P. Luminescent properties of LaAlO3 nanocrystals, doped with Pr3+ and Yb3+ ions. J. Lumin. 2014, 146, 239-42.
79. Pan, Y.; Su, Q.; Xu, H.; et al. Synthesis and red luminescence of Pr3+-doped CaTiO3 nanophosphor from polymer precursor. J. Solid. State. Chem. 2003, 174, 69-73.
80. Chen, G.; Ohulchanskyy, T. Y.; Liu, S.; et al. Core/shell NaGdF4:Nd3+/NaGdF4 nanocrystals with efficient near-infrared to near-infrared downconversion photoluminescence for bioimaging applications. ACS. Nano. 2012, 6, 2969-77.
81. Yu, X. F.; Chen, L. D.; Li, M.; et al. Highly efficient fluorescence of NdF3/SiO2 core/shell nanoparticles and the applications for in vivo NIR detection. Adv. Mater. 2008, 20, 4118-23.
82. Chang, M.; Song, Y.; Chen, J.; et al. Multisite luminescence and photocatalytic properties of TiO2:Sm3+ and TiO2:Sm3+@TiO2/TiO2:Sm3+@SiO2 luminescent enhancement materials. J. Alloys. Compd. 2017, 725, 724-38.
83. Wang, H.; Yu, M.; Lin, C. K.; Lin, J. Core-shell structured SiO2@YVO4:Dy3+/Sm3+ phosphor particles: sol-gel preparation and characterization. J. Colloid. Interface. Sci. 2006, 300, 176-82.
84. Nakano, H.; Suehiro, S.; Furuya, S.; Hayashi, H.; Fujihara, S. Synthesis of new RE3+ doped Li1+xTa1-xTixO3 (RE: Eu, Sm, Er, Tm, and Dy) phosphors with various emission colors. Materials. (Basel). 2013, 6, 2768-76.
85. Back, M.; Marin, R.; Franceschin, M.; et al. Energy transfer in color-tunable water-dispersible Tb-Eu codoped CaF2 nanocrystals. J. Mater. Chem. C. 2016, 4, 1906-13.
86. Liu, Y.; Tu, D.; Zhu, H.; Li, R.; Luo, W.; Chen, X. A strategy to achieve efficient dual-mode luminescence of Eu3+ in lanthanides doped multifunctional NaGdF4 nanocrystals. Adv. Mater. 2010, 22, 3266-71.
87. Zheng, K.; Zhao, D.; Zhang, D.; Liu, N.; Shi, F.; Qin, W. Sensitized high-order ultraviolet upconversion emissions of Gd3+ by Er3+ in NaYF4 microcrystals. J. Alloys. Compd. 2011, 509, 5848-52.
88. Cao, C.; Qin, W.; Zhang, J.; et al. Ultraviolet upconversion emissions of Gd3+. Opt. Lett. 2008, 33, 857-9.
89. Wang, W.; Wang, S.; Gu, Y.; Zhou, J.; Zhang, J. Contact-separation-induced self-recoverable mechanoluminescence of CaF2:Tb3+/PDMS elastomer. Nat. Commun. 2024, 15, 2014.
90. Xue, M.; Zhu, X.; Qiu, X.; Gu, Y.; Feng, W.; Li, F. Highly enhanced cooperative upconversion luminescence through energy transfer optimization and quenching protection. ACS. Appl. Mater. Interfaces. 2016, 8, 17894-901.
91. Li, D.; Ma, Q.; Song, Y.; et al. NaGdF4:Dy3+ nanofibers and nanobelts: facile construction technique, structure and bifunctionality of luminescence and enhanced paramagnetic performances. Phys. Chem. Chem. Phys. 2016, 18, 27536-44.
92. Kamimura, M.; Matsumoto, T.; Suyari, S.; Umezawa, M.; Soga, K. Ratiometric near-infrared fluorescence nanothermometry in the OTN-NIR (NIR II/III) biological window based on rare-earth doped β-NaYF4 nanoparticles. J. Mater. Chem. B. 2017, 5, 1917-25.
93. Montoya, E.; Espeso, O.; Bausá, L. Cooperative luminescence in Yb3+:LiNbO3. J. Lumin. 2000, 87-89, 1036-8.
94. Demirkhanyan, H. G.; Demirkhanyan, G. G.; Kokanyan, E. P. LiNbO3-Yb3+ crystal as a material for optical temperature sensor. J. Contemp. Phys. 2017, 52, 381-6.
95. Zhao, L.-J.; Yang, J.; Xu, J.-J.; Huang, H.; Zhang, G.-Y. Enhancement of Er3+ luminescence in LiNbO3:Mg crystals. Chin. Phys. Lett. 2001, 18, 1205-7.
96. Babajanyan, V. G.; Kostanyan, R. B.; Muzhikyan, P. H.; Sargsyan, R. V. Dependences of up-conversional luminescence of LiNbO3:Yb, Er crystals on pump intensity. In International Conference on Laser Physics 2010, Ashtarak, Armenia; Papoyan, A. V., Eds.; pp 799806.
97. Babajanyan, V. G.; Kostanyan, R. B.; Muzhikyan, P. H. Luminescence kinetics of LiNbO3:Yb3+-Er3+, LiNbO3:Er3+, and LiNbO3:Yb3+ crystals under selective excitations in the impurity subsystem. J. Contemp. Phys. 2012, 47, 17-22.
98. Stroganova, E. V.; Nalbantov, N. N.; Galutsky, V. V.; Yakovenko, N. A. A study of the quantum efficiency of multichannel relaxation in LiNbO3:Yb, Er crystals. Opt. Spectrosc. 2016, 121, 856-61.
99. Li, J.; Xie, Y.; Sun, R.; Zhou, J.; Sun, L. Exploring luminescence quenching on lanthanide-doped nanoparticles through changing the spatial distribution of sensitizer and activator. Nano. Res. 2023, 17, 4517-24.
100. Ramírez, M.; Bausá, L. Hysteretic behaviour in the fluorescence of Yb3+ in LiNbO3:MgO crystals. J. Lumin. 2003, 102-103, 206-10.
101. Dai, L.; Jiao, S.; Yan, Z.; Dai, P.; Lui, G.; Xu, Y. Influence of magnesium concentration on the optical properties of ytterbium and holmium co-doped lithium niobate crystal. Mod. Phys. Lett. B. 2016, 30, 1550260.
102. Brenier, A.; Madej, C.; Pédrini, C.; Boulon, G. Luminescence of ytterbium doped LiNbO3:MgO under UV excitation. Radiat. Eff. Defects. Solids. 2006, 135, 77-80.
103. Fujimura, M.; Khan, M.; Tsugawa, H.; et al. Thermally Nd-diffused Z-propagation Ti:LiNbO3 waveguide laser pumped by laser diode. Electron. Lett. 1996, 32, 2003-4.
104. Rüter, C. E.; Hasse, K.; Chen, F.; Kip, D. Optical characterization of a neodymium-doped lithium-niobate-on-insulator (LNOI). Opt. Mater. Express. 2021, 11, 4007.
105. Muñoz Santiuste, J.; Lavín, V.; Rodríguez-Mendoza, U.; Tardio, M.; Ramírez-Jiménez, R. Pressure-induced effects on the spectroscopic properties of Nd3+ in MgO:LiNbO3 single crystal. A crystal field approach. J. Lumin. 2017, 184, 293-303.
106. Zhou, X.; Deng, Y.; Jiang, S.; et al. Investigation of energy transfer in Pr3+, Yb3+ co-doped phosphate phosphor: the role of 3P0 and 1D2. J. Lumin. 2019, 209, 45-51.
107. Xiong, C.; Hao, R.; Li, H.; Lin, S.; Ma, D. Tunability of radioluminescence in LiNbO3:Pr polycrystals via lithium-to-niobium ratio. Ceram. Int. 2024, 50, 30093-9.
108. Sun, L.; Chu, X.; He, C.; Zuo, S.; Li, X.; Yao, C. Efficient photocatalytic nitrogen fixation over up-conversion nonlinear optical material Pr3+:LiNbO3 under visible light irradiation. Appl. Phys. A. 2021, 127, 35.
109. Hua, Y.; Jing, Z.; Ge, P. Pr3+-doped lithium niobate and sodium niobate with persistent luminescence and mechano-luminescence properties. Appl. Sci. 2024, 14, 2947.
110. Xing, L.; Xu, Y.; Wang, R.; Xu, W. Influence of temperature on upconversion multicolor luminescence in Ho3+/Yb3+/Tm3+-doped LiNbO3 single crystal. Opt. Lett. 2013, 38, 2535-7.
111. Cantelar, E.; Torchia, G.; Cussó, F. Characterization of up-conversion processes in Tm3+-doped LiNbO3. J. Lumin. 2007, 122-123, 459-62.
112. Dai, L.; Yang, S.; Chen, R.; Liu, C.; Han, X.; Shao, Y. Effect of [Li]/[Nb] ratios on UV-VIS-NIR spectroscopy and up-conversion luminescence in Hf:Dy:LiNbO3 crystals. J. Lumin. 2020, 217, 116773.
113. Li, A. H.; Zheng, Z. R.; Lü, Q.; et al. Two-photon-excited luminescence in a Tb3+ -doped lithium niobate crystal pumped by a near-infrared femtosecond laser. Opt. Lett. 2008, 33, 1014-6.
114. Lisiecki, R.; Macalik, B.; Kowalski, R.; Komar, J.; Ryba-romanowski, W. Effect of temperature on luminescence of LiNbO3 crystals single-doped with Sm3+, Tb3+, or Dy3+ ions. Crystals 2020, 10, 1034.
115. Pan E, ; Bai G, ; Cai M, ; Hua Y, ; Chen L, ; Xu S,. Reversible modification of ultra-broadband luminescence in transparent photonic materials through field-induced nanoscale structural transformation. Nanoscale 2020, 12, 22002-8.
116. Chen, F.; Jiang, T.; Zhai, B.; et al. Mechanoluminescence from an ion-irradiated single crystal of lithium niobium oxide. J. Phys. Chem. Lett. 2022, 13, 5394-8.
117. Tong, X.; Zhou, X.; Tang, X.; et al. Upconversion luminescence and optical temperature-sensing properties of LiNbO3:Yb3+/Er3+ nanoparticles. CrystEngComm 2022, 24, 1407-12.
118. Xue, S.; Zhang, D. Structure and spectroscopic properties of Er3+ doped LiNbO3 thin film grown by e-beam evaporation. Mater. Today. Commun. 2023, 36, 106522.
119. Xue, S.; Zhang, D. Near-stoichiometric Er3+ doped LiNbO3 thin films prepared by vapor transport equilibration treatment. Opt. Mater. 2023, 136, 113476.
120. Jia, Y.; Yao, Y.; Wang, S.; Ren, Y.; Zhao, X.; Chen, F. Dual-color upconversion luminescence emission from Er:LiNbO3 on-chip ridge waveguides. Results. Phys. 2021, 27, 104526.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].