Research progress on LT performance of sodium-ion battery electrolytes
Abstract
Sodium-ion batteries (SIBs) have gained attention for their low cost and abundant sodium resources. However, at low temperature (LT), their electrolytes suffer from reduced conductivity, higher viscosity, poor interfacial stability, and sluggish ion transport, leading to capacity loss and shortened cycle life. These problems significantly restrict the practical application of SIBs in harsh or LT environments, where performance degradation, capacity fading, or even complete failure can occur. Therefore, enhancing the LT performance of SIB electrolytes has become a key research focus. Improvements in electrolyte formulation-including solvent selection, sodium salt optimization, and functional additive engineering-play a vital role in addressing issues such as ion transport limitations and unstable electrode-electrolyte interfaces at LT. This review provides a comprehensive summary of the strategies developed to optimize various types of SIB electrolytes under LT conditions, including organic solvent systems, ionic liquids, solid-state electrolytes, and co-solvents. In addition, it discusses the latest research progress, highlights representative studies, and outlines potential directions for future development, with the aim of guiding the design of high-performance SIBs for LT.
Keywords
INTRODUCTION
Sodium-ion batteries (SIBs) have attracted increasing academic and industrial interest due to their low cost, abundant resources, and environmental compatibility[1]. Compared with lithium-ion batteries (LIBs), SIBs benefit from cheaper, more geographically balanced raw materials, with competitiveness rooted in sodium’s intrinsic physicochemical properties[2]. SIB electrolytes are largely analogous to lithium-ion systems in terms of solvent chemistry[3-5]. However, in the bill of materials for cell manufacturing, SIBs can often employ lower-cost aluminum current collectors on both the cathode and anode, whereas LIB anodes typically require copper foil[6-9]. Regarding salt chemistry, SIBs commonly use 1 M sodium hexafluorophosphate (NaPF6) in carbonate solvents as a benchmark formulation, which is considered readily implementable from an engineering standpoint[10,11]. Meanwhile, the widespread reliance of mainstream electrolytes on fluorinated components can impose additional burdens in terms of safety, environmental impact, and end-of-life compliance[12,13]. Therefore, if SIBs can more rapidly advance low-fluorine or fluorine-free salts and formulations, they may offer greater potential advantages for low-carbon commercialization and reduced Environment, Health and Safety-related costs[14-18].
Under extreme climates including LT environments, both SIBs and LIBs suffer from pronounced performance degradation, such as severe capacity loss, sluggish interfacial kinetics, and increased internal resistance, which significantly hinder their practical deployment in applications such as electric vehicles, military equipment, and polar communications[19-22]. The thermal motion and structural relaxation of solvent molecules slow markedly, as a result, electrolyte viscosity increases and ionic diffusion coefficients decrease, thereby suppressing bulk-phase transport that is largely governed by the vehicular motion of solvated ions[23-25]. Meanwhile, the reorganization of the solvation sheath becomes more difficult and desolvation kinetics become sluggish, which significantly increases the charge-transfer resistance for Na+ crossing the electrode/electrolyte interface[26,27]. In addition, the solid electrolyte interphase/cathode-electrolyte interphase (SEI/CEI) is more prone to thickening or becoming porous/heterogeneous under high polarization at LT, further elevating resistance of solid-electrolyte interphase and charge transfer resistance and inducing more severe polarization[28-30].
Organic, Inorganic, and Solid-State electrolytes require distinct, tailored optimization to improve battery performance at LT.
Organic electrolytes are widely used in both LIBs and SIBs; in sodium-ion systems, they offer a high electrochemical stability window, which enables the operation of SIBs across a wide voltage range[31,32]. Additionally, organic electrolytes with high ionic conductivity facilitate rapid ion transport, improving rate capability and overall electrochemical performance[33]. However, LT often leads to reduced ion mobility and sluggish interfacial reactions. To address these issues, here are the main strategies: (i) Optimization of sodium salts: Lowering salt concentration helps adjust the electrolyte’s freezing point and improves ion mobility. Combining different salts further enhances electrolyte stability and practicality under LT conditions[34-36]; (ii) Optimization of organic solvent. Low-freezing-point, low-viscosity organic solvents are employed to improve fluidity and desolvation[37-40]; (iii) Utilization of LT additives. Functional additives help stabilize the SEI at LT[41-43]; and (iv) The introduction of ionic liquid electrolytes based on Bis(fluorosulfonyl)imide anion (FSI-) or Bis(trifluoromethanesulfonyl)imide anion (TFSI-) anions[44,45].
Inorganic electrolytes naturally offer high ionic conductivity, low cost, and excellent safety[46]. However, they face severe limitations at LT, including water freezing and side reactions such as hydrogen evolution[47]. To improve their LT adaptability, several strategies have been proposed: (i) Developing “water-in-salt” electrolytes (WiSE) to suppress water activity, thereby expanding the electrochemical stability window and lowering the freezing point[48]; and (ii) Adding antifreeze agents (e.g., ethanol, ethylene glycol) or constructing hydrogel-based aqueous electrolytes to improve LT operability[49,50].
Solid-state electrolytes include inorganic solid electrolytes (such as oxides and sulfides) and polymer-based electrolytes[51]. At LT, they generally suffer from a significant decline in ionic conductivity and increased interfacial resistance[52]. Optimization strategies mainly include: (i) Enhancing the ionic conductivity of inorganic solid electrolytes through doping to improve their LT performance[53-55]; and (ii) Employing flexible polymer electrolytes or composite electrolytes to improve interfacial wettability and facilitate ion transport[56-58].
Although research on SIBs is currently a hotspot, reports on their LT performance remain limited, especially regarding the LT performance of electrolytes. This article reviews the recent methods and strategies for improving the LT performance of SIB electrolytes.
ORGANIC LIQUID ELECTROLYTES
Organic electrolytes typically comprise organic solvents, sodium salts, and additives[59]. However, under LT conditions, they often suffer from reduced conductivity, higher viscosity, accelerated passivation-layer growth, and solvent crystallization/precipitation[33,60]. These factors impede Na+ transport, increase internal resistance, and degrade capacity, efficiency, and power output[61,62]. Key levers for improvement include salt identity/loading, solvent system design, and tailored additives.
Sodium salts
Common salts such as NaClO4 and NaPF6 offer high conductivity and good compatibility with organic solvents. LT performance is mainly improved by (i) lowering salt concentration to weaken Na+ solvation and promote ion mobility/interfacial transport; and (ii) optimizing salt chemistry (e.g., sodium bis (trifluoromethanesulfonyl) imide (NaTFSI)) to enhance conductivity and form stable inorganic-rich SEI at LT.
Low-concentration electrolytes
Low-concentration electrolytes, owing to their weaker solvent-separated ion pairs (SSIP) and lower viscosity, are considered beneficial for enhancing the LT ionic transport performance of SIBs. Compared with conventional 1 M systems, dilute electrolytes show potential advantages in mitigating concentration polarization and improving ionic conductivity at LT.
Feng et al.[63] proposed a low-concentration LT electrolyte (0.5 M NaPF6 in diethylene glycol dimethyl ether (DEGDME), “0.5M-D”) that remains unfrozen at -30 °C [Figure 1A]. Raman analysis at 25 °C shows a higher SSIP fraction in 0.5M-D (52.6%) than in 1M-D (41.8%) [Figure 1B]. At -30 °C, 0.5M-D delivers
Figure 1. (A) Optical photographs of 0.5M-D (right) and 1M-D (left) electrolytes at different temperatures. (B) The fitted Raman spectra of 0.5M-D and 1M-D at 25 °C. (A and B) Figures reprinted with permission from Ref.[63]. Copyright 2024 Wiley-VCH. (C) In situ monitoring of an AG||PTPAn full cell at -40 °C using a three-electrode setup. Reprinted with permission from Ref.[64]. Copyright 2021 Wiley-VCH. (D) Proposed NaFSI-derived film-formation mechanism in the NNFM111||HC full cell. (E) Rate performance of NNFM111||HC full cells with different electrolytes at -20 °C. (D and E) Figures reprinted with permission from Ref.[69]. Copyright 2024 Wiley-VCH.
Dual-salt
Single-salt electrolytes often show sluggish Na+ transport and interfacial instability at LT[66], which can be mitigated by dual-salt formulations. Sodium trifluoromethanesulfonate (NaOTF) improves LT ionic conductivity and reversibility, NaBF4 enhances ion transport and suppresses side reactions, and Sodium Bis(fluorosulfonyl)imide (NaFSI) lowers Na+ solvation energy while promoting a stable interphase.
Yu et al.[67] prepared a weakly solvated dual-salt electrolyte containing 0.25 M NaPF6 and 0.25 M NaOTF dissolved in DEGDME with 3% 1,1,2,2-tetrafluoroethyl-2,2,3,3-tetrafluoropropyl ether (TTE).
Low-concentration electrolytes weaken Na+ solvation, lowering the desolvation barrier and promoting charge transfer while forming thin, uniform, organic-rich SEI/CEI films, thereby improving LT kinetics and reducing side reactions and salt precipitation. Dual-salt formulations add synergy by maintaining high conductivity and generating dense, stable SEI/CEI layers that enhance ion transport and suppress parasitic reactions and dendrite growth at LT.
Organic solvent
Organic solvents can be divided into ester-based and ether-based solvents. Ester-based solvents include PC, EC, EMC, DEC, dimethyl carbonate (DMC), ethyl acetate (EA), methyl propionate (MP), and methyl acetate (MA). Ether-based solvents include dimethoxyethane (DME), DEGDME, tetrahydrofuran (THF), and 2-methyltetrahydrofuran (MeTHF). Organic-solvent properties largely determine the LT performance of SIB electrolytes. In particular, a low solvent melting point suppresses crystallization, preserving the liquid phase and continuous ion-transport pathways[59]. In addition, the viscosity of the solvent significantly affects ion diffusion and migration rates. Low-viscosity solvents can maintain relatively high ionic conductivity under LT conditions[37].
As shown in Figure 2A and B, various conventional organic solvents, including ester-based solvents such as PC, EC, EMC, DMC, DEC, EA, MP, and MA, as well as ether-based solvents such as DEGDME, DME, THF, and MeTHF, exhibit different melting points and viscosities, all of which directly influence the performance of sodium-ion batteries under LT conditions[70-73].
Figure 2. (A) Melting points of different organic solvents. (B) Viscosity of different organic solvents at RT.
Carbonate esters
Among carbonate-based esters, cyclic carbonates such as EC and PC are widely used for their high dielectric constants and excellent salt-dissolving abilities[39]. Linear carbonates (DMC, EMC, DEC) offer low viscosity, optimizing their combinations is an effective route to enhance the electrochemical performance and stability of SIB electrolytes[40].
Combining cyclic and linear carbonates has proven effective in balancing conductivity and viscosity, especially at LT. Lin et al.[74] prepared a 1 M NaClO4 in EC:PC (1:1, vol/vol) electrolyte to assemble a
Figure 3. (A) Conductivity of various 0.8 M NaPF6 electrolytes at different temperatures. (B) Visual observation of various 0.8 M NaPF6 electrolytes at LT. (C) Capacity retention of NaNi1/3Fe1/3Mn1/3O2||HC pouch cells at various temperatures. (A-C) Figures reprinted with permission from Ref.[76]. Copyright 2021 Elsevier.
Non-carbonate esters
Non-carbonate esters such as EA, MA, and MP have also been studied as solvents of the LT electrolyte for their low viscosity, high ionic conductivity, and improved interfacial compatibility with sodium metal or HC anodes. EA is considered a promising electrolyte solvent due to its low viscosity, cost-effectiveness, and low toxicity[77]. Jayakumar et al.[78] used 1 M NaPF6 in EA with 5 wt% FEC in Na0.97Ca0.03[Mn0.39Fe0.31Ni0.22Zn0.08]O2 (NCMFNZO)||HC pouch cells, achieving 180 mA h at 0 °C. Compared to EA-based electrolytes.
EA-, MA-, and MP-based electrolytes mainly enhance LT performance by lowering viscosity and improving ionic conductivity, but strong Na+-solvent coordination still hampers desolvation and increases interfacial polarization. To further optimize LT solvation and interfacial kinetics, Hu et al.[81] developed a carboxylate-based low-temperature electrolyte [FTEFT (NaFSI + NaTFA in EDFA/FEC + TMSPi)] using a weakly solvating fluorinated ester (EDFA) with a high-donor-number anion [Trifluoroacetate (TFA-)] and Tris(trimethylsilyl) phosphite (TMSPi). TFA- dominates the Na+ solvation sheath, lowering desolvation/interfacial impedance (EA 68.4 kJ mol-1) and promoting a NaF-rich SEI for improved LT Na+ transport and uniform Na deposition, while TMSPi scavenges hydrofluoric acid (HF) to stabilize the CEI and suppress side reactions/metal dissolution. The electrolyte maintains 3.0 × 10-4 S cm-1 at -60 °C, enabling NaCrO2||Na to deliver 121.22 and 92.10 mA h g-1 at -40 and -60 °C, respectively.
Ether solvent
Ether-based solvents, such as DME, DEGDME, THF, and MeTHF, possess low viscosity, good reductive stability, and low freezing points. Therefore, they also attract widespread attention in the study of LT electrolytes for SIBs.
Zheng et al.[82] reported that the charge transfer energy of a 0.5 M NaPF6 in DEGDME electrolyte was
Figure 4. (A) Schematic illustration of SIBs with conventional electrolytes (left) and the designed electrolyte (right) operating at LT. (B) In situ EIS profiles of HC||Na cells measured at -40 °C. (C) Rate performance of HC anodes with five electrolytes at -40 °C. (A-C) Figures reprinted with permission from Ref.[83]. Copyright 2024 Wiley-VCH. (D) Arrhenius plot of RCT with derived EA for NVPF in both electrolytes. Cross-sectional SEM images of Sn@NC anode with a mass loading of 3 mg cm-2 after (E and F) fully sodiated and (G and H) desodiated at the first cycle in (E and F) the DEGDEM and (E, H) the EC/PC-based electrolytes. (D-H) Figures reprinted with permission from Ref.[84]. Copyright 2021 Wiley-VCH.
THF and MeTHF possess extremely low freezing points (THF ~ -108 °C; MeTHF ~ -136 °C) and low viscosity [Figure 2], which enable good fluidity and ion mobility at LT. Their weak solvation also promotes rapid Na+ desolvation, accelerating charge-transfer kinetics at LT. Tang et al.[85] developed a weakly solvated electrolyte (WSE) based on 1 M NaPF6 in THF to improve the anode/electrolyte interface. MD simulations indicate weak Na+-THF coordination (CN = 3.3, binding energy -79.214 kJ mol-1), facilitating desolvation and ion transport. Accordingly, Na||HC delivers 181 mA h g-1 at -20 °C and 2 A g-1, retaining 95% capacity after 1,000 cycles.
Yang et al.[86] designed a temperature-adaptive NaPF6-diethylene glycol dimethyl ether (DIG)/THF (2:8 vol/vol) electrolyte. The ionic conductivities of this system at 25 and -40 °C are 9.57 × 10-3 S cm-1 and
LT electrolyte research and development is shifting from viscosity/liquid-phase control to solvation-structure and interfacial-kinetics engineering. Since the key LT bottleneck is Na+ desolvation and interfacial transport, a core strategy is to build weakly solvated systems with tailored solvation structures. Accordingly, design is moving from empirical optimization to solvation-engineering approaches that combine weakly solvating solvents, mixed-solvent systems, and interfacial film regulation to boost LT ion transport and interfacial stability.
Additives
SIBs often suffer from increased electrolyte viscosity, sluggish ion transport, and elevated interfacial resistance under LT conditions, which severely impair their performance and cycle life. To address these challenges, introducing functional electrolyte additives has proven to be an effective strategy[43,89,90].
Cai et al.[91] introduced 1 wt% sodium difluorophosphate (NaDFP) into a 0.8 M NaPF6-EC/EMC (3:7, vol) electrolyte. The introduction of NaDFP reduces the Na+ desolvation energy from 9.16 kJ mol-1 in the baseline electrolyte to 8.24 kJ mol-1. He et al.[92] incorporated 0.5 wt% sodium difluoro(oxalato)borate (NaDFOB) and 0.5 wt% succinonitrile (SN) into an EC/PC/EP (25:25:50 vol/vol) solvent system, where NaDFOB preferentially decomposes to form NaF-rich inorganic SEI/CEI and SN participates in Na+ solvation while introducing -C≡N components, synergistically constructing stable inorganic interphases. The electrolyte exhibits an ionic conductivity of 1.7 × 10-4 S cm-1 at -60 °C. At -25 °C (2C), NVP||Na delivers 105 mA h g-1 with 95.1% retention after 500 cycles. Song et al.[93] added 3% adiponitrile (ADN) to 1.0 M NaPF6 in EC/DEC/PC. Na0.76Ni0.3Fe0.4Mn0.3O2||HC cells show ~8% and ~13% capacity gains at -10 and -20 °C.
Figure 5. (A) HOMO and LUMO levels. In situ XRD of NNFMO cathodes in FPPN electrolyte: (B) Charge/discharge profiles and (C) in situ XRD patterns during cycling. (A-C) Figures reprinted with permission from Ref.[94]. Copyright 2024 Wiley-VCH. (D) Cycling performance of MN cathode in TPP and THF electrolytes at -40 °C. Reprinted with permission from Ref.[95]. Copyright 2023 The American Chemical Society. In situ sodium metal deposition in (E) BLTE and (F) ES6-BLTE electrolytes at -40 °C. Reprinted with permission from Ref.[96]. Copyright 2023 Wiley-VCH. (G) Binding energy for various Na+-solvent (PC, FEC, and TMSPi) complexes. Reprinted with permission from Ref.[97]. Copyright 2024 The American Chemical Society.
Yin et al.[95] developed a THF-based TPP [main solvent (THF) and partial co-solvent (PC and ethoxy(pentafluoro) cyclotriphosphazene (PFPN))] electrolyte (0.8 M NaPF6 + trace PC/PFPN), leveraging THF's low viscosity and freezing point, and PC/PFPN's higher oxidative stability (HOMO: -8.08/-8.86 eV). PFPN forms a dense, inorganic-rich CEI, mitigating parasitic reactions. Conductivity is 6.5 × 10-3 S cm-1 at
Liang et al.[97] proposed a phosphorus/silicon synergistic strategy using NPFT (1 M NaClO4-PC + 10 vol% FEC + 1 vol% TMSPi), with NPF as the reference. The low Na+-TMSPi binding energy promotes preferential interfacial reactions [Figure 5G], enabling NVPF||HC to retain 94% capacity after 500 cycles at -25 °C and
Additives such as NaDFP stabilize interfaces and boost LT capacity, while NaDFOB and SN sustain stable interphases and long-term cycling at extreme LT. Polar additives (e.g., AND) enhance ionic conductivity and solvation, whereas fluorinated species (e.g., FPPN, ES6-BLTE) lower viscosity and reinforce the SEI for improved interfacial compatibility. FEC-TMSPi further promotes a stable SEI and reduces interfacial resistance. Collectively, these multifunctional additives improve efficiency and cycling stability and mitigate solvent crystallization, enabling more stable electrolytes and better LT performance.
Ionic liquid electrolytes
Common IL systems use imidazolium, pyrrolidinium, or ammonium cations with weakly coordinating anions such as TFSI- and FSI-, maintaining ionic conductivities of 10-3-10-4 S cm-1 at LT. Incorporating sodium salts such as sodium bis(fluorosulfonyl)amide (Na[FSA]) or NaTFSI forms dense, inorganic-rich interfacial layers (e.g., NaF, S/N species), enhancing interfacial stability and suppressing side reactions. IL-ether or IL-carbonate composites further lower viscosity and improve Na+ transport, boosting SIB performance at LT.
Yamaguchi et al.[98] developed a pyrrolidinium IL/NaFSA electrolyte with 1-7 wt% zwitterionic TEP3s (IL/Na/ZI-x): conductivity at 25 °C increased from 4.03 × 10-3 to 5.17 × 10-3 S cm-1, while IL/Na/ZI-3 (3 wt%) delivered 1.85 × 10-3 (0 °C) and 1.08 × 10-3 S cm-1 (-10 °C). Ding et al.[99] reported Na||HC capacities of
Figure 6. Isothermal TGA trace of 0.1NaTFSI-0.9IL sodium electrolytes: (A) FSI-based electrolytes. (B) TFSI- and IM14-based electrolytes. (C) Ionic conductivity vs. temperature dependence of 0.1NaTFSI-0.9IL sodium electrolytes. (A-C) Figures reprinted with permission from Ref.[101]. Copyright 2020 MDPI.
Composition of organic liquid electrolytes and their key LT electrochemical properties
| Composites | Cathode||Anode | Ionic conductivity (S cm-1)/ temperature (°C) | End of capacity (mA h g-1)/ capacity retention (%)/ current density/ cycle number measuring temperature (°C) | Reference |
| 0.5M NaPF6-DEGDME | LMNM`T||Na | 1.7 × 10-3/-10 | 91.5/90.1/1C/900/-30 | [63] |
| Na||HC | 330/97.1/0.2C/100/-30 | |||
| LMNM`T||HC | 86.2/83.2%/0.2C/100/-30 | |||
| 0.5 M NaPF6-DEGDME | - | 1.0 × 10-3/-20 3.0 × 10-4/-40 1.7 × 10-4/-60 | - | [64] |
| 0.3 M NaClO4 in EC:PC = 1:1 (vol/vol) 5% FEC | NVPF||Na | - | -/93.4/1C/1,000/-25 | [65] |
| NVPF||HC | - | ~100/91/0.2C/300 h/-25 | ||
| 0.25 M NaPF6-0.25M NaOTF/DEGDME/3%TTE | Na||Al@C | - | ~100/99/0.5 mA cm-2/100/-20 | [67] |
| CuS||Na | ~315/99%/0.5C/100/-20 | |||
| 0.8 M NaOTF and 0.2 M NaBF4 in DEGDME | NVP||Na | ~70/99%/0.5 mA h cm-2/80/-40 | [68] | |
| 0.5 M NaFSI + 0.5 M NaPF6 in EC/PC/EMC/DEC 1:2:6:1 | NNFM111||HC | - | /85.8/0.5C/-/-20 | [69] |
| 0.8 M NaPF6 PC/EMC (1:1, vol/vol)2 wt% FEC | NFM||HC | 2.2 × 10-3/-40 | -/45.8/-/-/-40 | [76] |
| 20%MA + Control (1 M NaPF6 in EC-PC-DMC-MA 1:1:2:1 by vol% | 7.2 × 10-3/-10 | [79] | ||
| 2 M NaPF6 in MP/FEC | NVPOF||Na | - | ~77/96/0.1C/100/-40 | [80] |
| 0.90 mol L-1 NaFSI + 0.10 mol L-1 NaTFA in EDFA/FEC (V:V = 9:1) + 1 wt% TMSPi | NaCrO2||Na | - | ~92/~72/0.1 A g-1/300/-40 | [81] |
| NFM||Na | ~80/~99/0.05 A g-1/50/-40 | |||
| 0.5 M DEGDME | NVPF||Na | ~90/95.58/10C/1,000/-20 | [82] | |
| NVPF||NTP | ~90/69.8/5C/10,000/-20 | |||
| 1 M NaPF6 DEGDME/DME | Na||HC | 1.387 × 10-2/-40 | 282.11/99.50/0.3 A g-1/1,000/ -40 | [83] |
| 1 M NaPF6 in DEGDME | NVP/C||Sn@NC | ~60/~98/0.1 A g-1/250/-20 | [84] | |
| 1.0 M NaPF6 in THF | Na||HC | - | ~225/90/2 A g-1/1,000/-5 ~200/90/5 A g-1/1,000/-25 | [85] |
| 1 M NaPF6-DIG/THF | Na||HC | - | -/90.6/0.2C/400/-40 | [86] |
| 0.8 M NaPF6-MeTHF/DME (6:1 vol/vol) | Na||Bi | 1.78 × 10-3/-20 | 306.2/96.34/0.4 A g-1/300/-20 -/-/0.5C/50/-20 | [87] |
| NVP||Bi | ||||
| 1.0 M NaPF6 in THF/MeTHF = 5:5 (vol/vol) | Na||HC | 8.7 × 10-4/-60 4.1 × 10-4/-80 | -/100/50 mA g-1/250/-40 | [88] |
| NVP||HC | -/99.9/50 mA g-1/100/-40 | |||
| 0.8 M NaPF6 in EC: EMC = 3:7 (vol/vol) 1 wt% NaDFP | NFM||HC | - | ~825-/90/100/-10 | [91] |
| Na TFSI in EC/PC/EP (25:25:50)/Na DFOB (0.5 wt%)/SN (0.5 wt%) | NVP||Na | 1.7 × 10-5/-60 | 64/~90/0.1C/130/-45 | [92] |
| 1 M NaPF6-EC/PC/DEC/5% FEC/2 wt% FPPN | 2.3 × 10-3/-40 | -/94.1/0.2C/100/-40 | [94] | |
| 0.8 M NaPF6 in THF/PC/PFPN | NMNO||Na | 2.3 × 10-3/-40 | -/94.1/0.2C/100/-40 | [95] |
| 1 M NaFSI in EC/PC/DEC (1:1:4, vol/vol) 6% ES6-BLTE | NVP||Na | 1.13 × 10-3/-40 | /88.2/0.1C/200/-40 | [96] |
| 1 M NaClO4 PC/10 vol % FEC/1 vol % TMSPi | NVPOF||Na | - | 102/83/0.5C/100/-25 | [97] |
| IL/Na/ZI-x | - | 5.17 × 10-3/RT 1.85 × 10-3/0 1.08 × 10-3/-10 | - | [98] |
| 1 M Na[FSA]-[C3C1pyrr][FSA] | NVPF-C||Na | - | 50/~53.8/0.1C/50/-20 | [100] |
| EMIFSI | - | 1.1 ± 0.1 × 10-3/20 | - | [101] |
| EMITFSI | - | 3.8 ± 0.2 × 10-5/20 | - | |
| 1 M NaPF6 in DEGDME/ [C4C1im][BF4] (4:1 vol/vol) | NVP||Na | 4.2 × 10-2/-20 | ~100/~90/3C/~450/20 | [102] |
Ionic liquids, with low glass transition temperatures and special molecular structures, maintain high ionic conductivity and resist crystallization at LT, ensuring electrolyte and battery stability. They excel in extreme LT environments where traditional organic electrolytes lose conductivity.
Improving LT performance in SIBs focuses on optimizing sodium salts, ester- and ether-based electrolytes, additives, and ionic liquids. Salt type and concentration affect conductivity and stability. ester electrolytes require co-solvents to lower viscosity, while ether-based ones offer higher conductivity. Additives help form stable SEI layers, and ionic liquids with wide electrochemical windows further enhance LT performance. Integrating these strategies significantly improves SIB efficiency across wide temperature ranges.
Solid-state electrolytes
Compared with organic electrolytes, solid electrolytes offer superior safety and interfacial stability, but their LT ionic conductivity remains limited. Solid electrolytes include inorganic, polymer, and composite systems[103,104]. Inorganics suffer from high interfacial resistance due to rigidity, polymers are constrained by segmental freezing, and composites integrate both advantages but still face LT challenges such as poor filler dispersion and weak interfacial bonding[52,58,105].
Inorganic solid electrolyte
Inorganic electrolytes are mainly oxides[106] (e.g., NASICON (Sodium Super Ionic Conductor)-type) and sulfides (e.g., Na3SbS4, Na11Sn2PS12). Oxides offer high stability and wide electrochemical windows but suffer from high interfacial impedance and ion migration barriers at LT[53,107]. Sulfides exhibit higher LT ionic conductivity due to flexible lattices and low interfacial resistance, but they have poorer stability[104]. Strategies such as elemental doping and structural regulation reduce migration barriers, improve interfacial contact, and enhance LT ion transport.
Despite these improvements achieved by compositional engineering of oxide-based electrolytes, their LT ionic conductivity remains fundamentally constrained, motivating increasing attention toward sulfide-based solid electrolytes. Zhang et al.[111] prepared a sulfide-based sodium-ion solid electrolyte, Na11Sn2PS12. AIMD simulations indicate that it is an approximately isotropic three-dimensional (3D) Na+ conductor, with an ionic conductivity of about 2.5 × 10-4 S cm-1 at -20 °C. The EA determined from measurements down to -20 °C is 0.25 eV. Zhang et al.[112] synthesized a Na11Sn2PS12 sulfide solid electrolyte with an ionic conductivity of
Compared with oxide solid electrolytes, sulfide counterparts generally offer higher intrinsic ionic conductivity and thus better LT ion transport. LT performance can be further improved via elemental doping (e.g., Sc, Zn, Mg) and structural/microstructural optimization to enhance pathway continuity and reduce grain-boundary resistance. Accordingly, sulfides such as Na11Sn2PS12 and Na3SbS4 typically outperform conventional oxides in LT conductivity, making them promising electrolytes for sodium-ion batteries LT.
Solid-state polymer electrolyte
Polymer electrolytes typically consist of a polymer matrix, sodium salts, and plasticizers or fillers[115-118]. Matrices such as Poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP) and Polyacrylonitrile (PAN) provide the mechanical framework and ion-transport pathways. Plasticizers (e.g., PC) reduce the glass-transition temperature and enhance LT segmental mobility, while inorganic fillers or crosslinkers tune porosity and improve interfacial stability[119].
Yang et al.[120] incorporated Sn(OTf)2 and FEC into a PVDF-HFP/PC quasi-solid polymer electrolyte. Sn(OTf)2 (where Sn(OTf) = Tin(II) trifluoromethanesulfonate) catalyzes PC ring-opening, reducing the EA from 0.272 to 0.198 eV [Figure 7A], and generates metallic Sn to improve interfacial contact and suppress dendrites. FEC decomposition forms a NaF-rich SEI/CEI to stabilize the interface. Accordingly, NVP||Na delivers 87.2/74.8/69.5/52.4 mA h g-1 at 0/-10/-15/-20 °C, respectively. Lonchakova et al.[121] reported a NaClO4/PC gel-polymer electrolyte (PAN:PC = 1:5.25) with crystalline domains for mechanical support and solvent-rich amorphous regions for ion transport, achieving ~10-3 S cm-1 at -20 °C (EA 12-15 kJ mol-1) and
Figure 7. (A) Arrhenius plot of the ionic conductivity. Reprinted with permission from Ref.[120]. Copyright 2024 Wiley-VCH. (B) Schematic diagram of the ionic-exchange process. (C) In situ Raman spectrum of SSIB and (D) Electrochemical performance. (B-D) Figures reprinted with permission from Ref.[122]. Copyright 2019 Wiley-VCH.
Du et al.[122] prepared a Perfluorosulfonic acid (PFSA)-Na polymer electrolyte via ion exchange using PFSA-Li as the precursor [Figure 7B]. It delivered ionic conductivities of 4.88 × 10-5 S cm-1 at -15 °C and
Krupina et al.[123] used EC/Tetramethylene sulfone (SL) (0.5:0.5, mass) as a binary plasticizer for a Na-Nafion electrolyte, exploiting its low eutectic point (-16 °C) to suppress crystallization and maintain Na+ transport, giving 110-4 S cm-1 at 30 °C and 10-6 S cm-1 at -60 °C. Voropaeva et al.[124] methanol-treated Nafion-117 to increase pore size and incorporated a PC-DMA (N,N-Dimethylacetamide)-THF solvent system to improve uptake and suppress LT phase transitions, achieving 4.0 × 10-3 S cm-1at 30 °C and 1.0 × 10-3 S cm-1 at -20 °C. These strategies provide new approaches to improving the LT performance of polymer electrolytes.
By introducing functional additives (such as Sn(OTf)2, FEC, etc.), employing crosslinking techniques, and optimizing the electrolyte structure, the conductivity and cycling stability of polymer electrolytes at LT have been successfully improved. These improvements effectively enhance ion migration pathways, strengthen the mechanical properties, and stabilize the interface of the electrolyte, significantly reducing battery performance degradation under LT conditions.
Composite solid electrolytes
Composite solid-state electrolytes are a key strategy to mitigate poor LT performance, interfacial instability, and dendrite growth in sodium-ion batteries. By integrating inorganic and polymer components, they leverage the advantages of both material classes[125,126], enhancing Na+ conductivity and LT stability to improve overall cell performance.
Xie et al.[127] added 5 wt% Al2O3 nanoparticles into a PVDF-HFP matrix, where Al2O3-polymer hydrogen bonding reduced crystallinity and created a uniform porous network. The resulting expanded graphite||Sn dual-ion battery maintained~10-3 S cm-1 at -20 °C and delivered 44.2 mA h g-1 at 5C, nearly twice that of a conventional liquid electrolyte.
Chen et al.[128] introduced NZSP ceramic fillers into a PVDF-HFP/PVDF framework to construct a composite gel polymer electrolyte [PVDF-HFP/PVDF/NZSP GPE (PHPNG)]. The NZSP ceramic fillers provide NASICON-type fast ion-conduction channels, reducing the Na+ migration barrier from 0.27 to 0.21 eV. In-situ XRD to track the NVP phase transformation. The NVP cathode using PHPNG was able to maintain a reversible phase transition. In contrast, the (211) and (116) crystal plane peaks of the NVP cathode using PHPG underwent irreversible changes, leading to irreversible capacity loss [Figure 8A and B]. The Na||PHPNG||NVP cell delivered capacities of approximately 116, 107, 93, and 61 mA h g-1 at 25, 0, -10, and -20 °C, respectively [Figure 8C].
Figure 8. The in‐situ XRD to track the NVP phase transformation of (A) Na//PHPNG//NVP and (B) Na//PHPG//NVP. (C) The capacity is linked to the change of temperatures from 25 to -20 °C at 1C. (A-C) Figures reprinted with permission from Ref.[128]. Copyright 2024 Wiley-VCH. (D) Schematic of the PSAMCE-AlF3 fabrication process. (E) Ionic conductivities of the symmetric coin cells at -30 °C. (F) Rate performance of the Na||PSAMCE-AlF3||NVP cell. (D-F) Figures reprinted with permission from Ref.[129]. Copyright 2022 Wiley-VCH.
Wu et al.[130] fabricated a 3D SiO2 nanofiber membrane (SNF) and, via surface modification/anion exchange, obtained TFSI--loaded SNF immobilized anion (SNF@IA). NaTFSI in an allyl acetoacetate (AAEM) matrix (with Poly(ethylene glycol) diacrylate (PEGDA) and 2-Hydroxy-2-methylpropiophenone (HMPP)) was infiltrated and UV (UV curing method)-cured to form PAAEM (the polymer electrolyte matrix derived from AAEM after UV-induced polymerization)-SNF@IA, which maintains 0.75 × 10-4 S cm-1 at -40 °C with tNa+ (Na+ transference number) = 0.72. Consequently, NVP||Na retains 96.1 mA h g-1 after 100 cycles at -40 °C and 0.05C.
In another study, Wu et al.[129] prepared a SNF via electrospinning and high-temperature calcination, followed by mixing with AAEM, N,N'-Methylenebisacrylamide, and PVDF-HFP, UV-curing, and magnetron sputtering of an ultrathin AlF3 layer (PSAMCE-AlF3) [Figure 8D]. The electrolyte improves Na+ transference number (0.63) and ensures conductivity and flexibility at LT due to the low glass transition temperature (~-44.7 °C) of the PVDF-HFP matrix. The ionic conductivities of the electrolyte at -30 °C are 1.53 × 10-4 S cm-1 [Figure 8E] with an EA below 0.22 eV. NVP||Na cell delivered ~92 mA h g-1 at 0 °C with ~96% capacity retention, and stable operation at -30 °C and 0.05C [Figure 8F]. The detailed information of the examples cited in the above three sections is shown in Table 2.
Composition of solid-state electrolytes and their key LT electrochemical properties
| composites | Cathode||Anode | Ionic conductivity (S cm-1)/ temperature (°C) | End of capacity (mA h g-1)/ capacity retention (%)/ current density/ cycle number measuring temperature (°C) | Reference |
| Na3Zr2Si2PO12 | - | 2.3 × 10-5/-20 | - | [108] |
| Na3.4Zr1.6Sc0.4Si2PO12 | NVP||Na | 9.1 × 10-4/0 6.6 × 10-4/-10 4.0 × 10-4/-20 | 90/90/0.1C/100/-10 | [109] |
| Na3.65Zr1.675Zn0.2Mg0.125Si2PO12 | - | 5.5 × 10-3/-15 | - | [110] |
| Na11Sn2PS12 | - | 2.5 × 10-5/-20 | - | [111] |
| Na11Sn2PS12 | - | 3.2 × 10-4/0 1.9 × 10-4/-10 1.1 × 10-4/-20 | - | [112] |
| Na3SbS4 | 2 × 10-4/-20 | - | [113] | |
| Na2.7W0.3Sb0.7S4 | 5.5 × 10-3/-15 | - | [114] | |
| PVDF/HFP-PC-FEC-Sn | NVP||Na | ~50/100/0.5C/60/-20 | [120] | |
| 0.41 M NaClO4 in PAN/PC 1:4 (vol/vol) | NVP||Na | ~70/99/0.5C/300/-20 | [121] | |
| Nafion®115 in EC/SL(w(EC) = 0.5) | [61] | |||
| Nafion-therm in PC/DMA/THF 1:1:1 (vol/vol) | - | 1.0 × 10-3/-20 | - | [122] |
| MTP92 | NVP||Na | ~1.99 × 10-3/0 > 1.01 × 10-3/-20 | -/~97.62/2C/1.200/-10 | [123] |
| PFSA-Na | Prussian blue cathode||Na | 4.88 × 10-5/ -15 | - | [124] |
| PVDF-HFP with 5 wt% Al2O3 | - | 1.3 × 10-3/RT | 65.6/99/5C/10/-10 | [127] |
| NVP||PHPNG||Na | NVP||Na | - | ~69/~92/1C/350/-20 | [128] |
| PAAEM-SNF@IA | NVP||Na | 0.75 × 10-4/-40 | ~110/83.2/0.02C/60/-40 | [130] |
| PSAMCE-AlF3 | - | 1.53 × 10-4/-30 | - | [129] |
At LT, inorganic fillers create fast-ion pathways and lower Na+ migration barriers, while the polymer matrix preserves flexibility and mitigates conductivity loss. Nanoparticles, ceramic frameworks, and nanofibers further enhance transport and suppress dendrites, and surface modification improves interfacial stability. Together, these approaches strengthen SIB performance in cold environments.
SIB solid-state electrolytes improve safety and interfacial stability; however, their LT performance is hindered by slow ion transport and increased interfacial resistance. Inorganics are stable but often less conductive, sulfides are more conductive yet less stable, and polymers are hindered by segmental freezing/crystallization (partly alleviated by additives, crosslinking, or solvent engineering). Composites combine inorganic rigidity with polymer flexibility to improve ion pathways and interface robustness. Future efforts should focus on doping, structural/microstructural optimization, and interfacial engineering to enhance LT SIB performance.
Inorganic liquid electrolytes
Inorganic electrolyte systems have attracted growing interest for SIBs due to their safety, environmental friendliness, and high ionic conductivity[46]. Compared to organic electrolytes, they are non-flammable, easier to prepare, and support faster ion transport[131]. Strategies to improve cold-condition performance include functional additives, salt-composition tuning, water-in-salt electrolyte (WiSE) designs, and physicochemical-property optimization, which collectively enhance electrochemical performance and interfacial stability[49,50,132,133].
Aqueous electrolytes
Aqueous SIBs use water-based electrolytes; however, water freezing at LT reduces ion mobility, increases interfacial resistance, and affects cycling stability. To address this, strategies such as combining sodium salts, using high-concentration water-in-salt electrolytes, adding additives to disrupt the hydrogen-bond network, and constructing gel networks have been explored to lower freezing points and improve stability[133-135].
Salt
Common aqueous sodium salts such as NaClO4 and NaCl often suffer from crystallization and hindered ion transport at LT, limiting their applicability. To improve LT performance, strategies such as employing deep eutectic sodium salt systems to lower the freezing point or introducing inorganic salts such as manganese chloride (MnCl2) to disrupt the hydrogen-bond network have been explored.
Nian et al.[136] reported a 2 M NaClO4 aqueous electrolyte enabling a Ni(OH)2 (NNH)||NTP@C full cell with excellent LT durability. At -20 °C, the cell retained ~85% capacity after 10,000 cycles at 10C with a low fading rate [Figure 9A]. Post-cycling analyses confirmed that the NTP (NaTi2(PO4)3)@C anode preserved stable morphology, composition, and crystal structure after 10,000 cycles at -20 °C [Figure 9B].
Figure 9. (A) 10,000 cycles of the battery at -20 °C with a rate of 10C. (B) SEM image, EDS elemental mappings, and TEM image of NTP@C electrode at pristine, 500th cycle, and 10,000th cycle. (A and B) Figures reprinted with permission from Ref.[136]. Copyright 2019 The American Chemical Society. (C) 1H NMR results for various electrolytes. (D) The ionic conductivity of 3.5 M Mg(ClO4)2 + 0.5 M NaClO4 electrolyte at different temperatures. (C and D) Figures reprinted with permission from Ref.[137]. Copyright 2022 Wiley-VCH.
Zhu et al.[137] developed a hybrid aqueous electrolyte by introducing Mg(ClO4)2 (3.5 M) at a deep eutectic concentration, combined with 0.5 M NaClO4. Proton nuclear magnetic resonance spectroscopy (1H NMR) showed that the strong polarization of Mg2+ disrupts the hydrogen-bond network, suppressing ice formation [Figure 9C]. Differential scanning calorimetry (DSC) results revealed the electrolyte freezing point is below -80 °C, with an ionic conductivity of 4.6 × 10-3 S cm-1 at -60 °C [Figure 9D]. Zhao et al.[138] introduced 4.0 M MnCl2·4H2O into a 0.5 M NaCl solution. Density functional theory/MD simulations show that adding a Mn salt reduces hydrogen bonding: strongly H-bonded water decreases from 48.1% to 26.7%, indicating an anti-freezing effect. The electrolyte exhibited an ionic conductivity of
Single-salt aqueous Na electrolytes typically show poor LT performance. In contrast, deep-eutectic salt systems or mixed-salt additives can enhance LT ionic conductivity, widen the electrochemical stability window, and sustain long-term cycling under extreme conditions.
Water-in-salt
WiSE, with typical concentrations of 10-17 M in SIBs (e.g., NaClO4, NaTFSI, or mixed-salt systems), can effectively suppress water crystallization, reduce the amount of free water, and enhance interfacial stability[139-141].
Wang et al.[142] used a highly concentrated 17 M NaClO4 WiSE, effectively reducing free water molecules and disrupting the hydrogen-bond network, lowering the freezing point to -50 °C. At -40 °C, it maintained
Zhu et al.[143] added formamide (FA) to 17 M NaClO4 aqueous electrolyte. Similar H2O-FA and H2O-H2O interaction energies favor H2O-FA clustering, disrupting the H-bond network and suppressing freezing; 1H NMR/DSC confirm a freezing point below -50 °C and 1.75 × 10-3 S cm-1 at -50 °C. Reber et al.[144] partially replaced NaFSI with an asymmetric anion (NaFTFSI) to enhance supercooling and inhibit crystallization [Figure 10A]. A 25 M NaFSI + 10 M NaFTFSI electrolyte shows no crystallization to -120 °C, with glass transition = -69 °C and a reduced liquidus temperature (~38 °C) [Figure 10B], accordingly, NVPOF||NTP retains ~74% capacity after 100 cycles at -10 °C [Figure 10C].
Figure 10. (A) Structural formulas of the symmetric and asymmetric anions. (B) DSC curves of 35 M NaFSI and mixed FSI/FTFSI electrolytes. (C) Discharge capacity of cells containing 25 M NaFSI + 10 M Na FTFSI operated at 30, 10, and -10 °C. (A-C) Figures reprinted with permission from Ref.[144]. Copyright 2019 The American Chemical Society.
Water-in-salt electrolytes (WiSE) reduce free water and disrupt hydrogen bonding, depressing the freezing point and widening the electrochemical stability window. Polar co-solvents further modify the H-bond network to suppress crystallization and enhance ion transport, while functionalized or asymmetric anions promote supercooling, inhibit ice formation, and preserve interfacial stability and reversibility at LT.
Additives
Suitable additives can markedly improve LT electrolyte performance. Organic molecules (e.g., Dimethyl sulfoxide (DMSO), glycerol) disrupt the water hydrogen-bond network, lowering the freezing point and widening the electrochemical stability window. Small molecules (e.g., methanol), combined with frameworks such as SiO2, suppress salt crystallization while preserving conductivity. Low-cost, non-toxic inorganic salts (e.g., CaCl2) are also attracting interest for enhancing ionic conductivity at LT.
Nian et al.[145] adding DMSO (mole fraction 0.3) to 2 M NaClO4 aqueous solution disrupts water’s H-bond network, suppresses ice formation, and lowers the freezing point to below -130 °C. The electrolyte still shows 1.1 × 10-4 S cm-1 at -50 °C. An NTP@C||AC full cell delivers 68 mA h g-1 at -50 °C with stable cycling, while the DMSO-free electrolyte cannot operate at this temperature. Sun et al.[146] added glycerol to a 1 M NaNO3 aqueous solution, where the hydroxyl groups of glycerol form strong hydrogen bonds with water molecules, weakening the water-water hydrogen bond interactions and lowering the freezing point to below -80 °C. At -10 °C (1C), Ni2ZnHCF (bimetallic Prussian blue analogue)||PTCDI (perylenetetracarboxylic diimide) delivers 40 mA h g-1 with negligible decay over 800 cycles.
Compared with flammable, toxic organic antifreeze additives, using common, low-cost inorganic inert additives to enhance LT performance is a promising research direction. Zhu et al.[148] used 3.86 M CaCl2 as an additive to the NaClO4 electrolyte. DSC shows that the freezing point temperature is the lowest when the solution concentration is 3.86 M [Figure 11A]. At -50 °C, the electrolyte ion conductivity with 3.86 M CaCl2 added was as high as 7.13 × 10-3 S cm-1. Na2CoFe(CN)6||AC (activated carbon) full cells deliver a capacity of 74.5 mA h g-1 at -30 °C and 1C. After three complex cycle tests on the battery [Figure 11B], the battery capacity can still be restored to its original level.
Figure 11. (A) Phase diagram of aqueous calcium chloride (3.86 M CaCl2 + 1 M NaClO4). (B) Long cycle life tests at a rate of 10C under
In summary, additives provide an effective approach to enhance the LT performance of aqueous SIBs. Organic molecules regulate the hydrogen-bond environment, inorganic frameworks with antifreeze agents suppress crystallization, and inorganic salts improve conductivity and stability at low cost. These studies highlight that rational additive design is key to achieving LT adaptability.
Other inorganic liquid electrolytes
Although aqueous electrolytes have advantages in enhancing the safety and environmental friendliness of sodium batteries, challenges still exist in their conductivity and stability under extremely LT conditions.
SO2-based electrolytes, such as NaAlCl4·2SO2, exhibit excellent LT performance and non-flammability.
Figure 12. (A) Conductivity of NaAlCl4·2SO2 at various temperatures. (B) In situ XRD patterns of the carbon cathode in a Na-SO2 cell during the first cycle. (C) Discharge voltage profiles at various LT. (A-C) Figures reprinted with permission from Ref.[149]. Copyright 2015 Nature.
The detailed information of the examples cited in the above three sections is shown in Table 3.
Composition of inorganic liquid electrolytes and their key LT electrochemical properties
| Composites | Cathode||Anode | Ionic conductivity (S cm-1)/ temperature (°C) | End of capacity (mA h g-1)/ capacity retention (%)/ current density/ cycle number measuring temperature (°C) | Reference |
| 2 M NaClO4 aqueous (aq.) | Ni(OH)2||NaTi2(PO4)3@C | - | ~70/100/10C/500/-20 | [136] |
| 3.5 M Mg(ClO4)2 + 0.5 M NaClO4 | AC||polyimide | 2.049 × 10-2/0 1.469 × 10-2/-10 8.26 × 10-3/-20 4.84 × 10-3/-30 3.10 × 10-3/-40 1.75 × 10-3/-50 | 85/99/1C/2,000/-50 68/99/1C/400/-50 (pouch battery) | [137] |
| 0.5 M NaCl + 4.0 M MnCl2·4H2O | Na2CoFe(CN)6||AC | 4.238 × 10-2/0 2.878 × 10-2/-10 1.926 × 10-2/-20 1.110 × 10-2/-30 5.62 × 10-3/-40 2.44 × 10-3/-50 | 54/94.4/1C/1,000/-40 28/~99/10C/10,000/-40 (pouch battery) | [138] |
| 17 M NaClO4 | NVP||NVP | 2.2 × 10-2/0 1.4 × 10-2/-10 9 × 10-3/-20 4 × 10-3/-30 3 × 10-3/-40 | ~16 mA h cm-3/91/10C/600/-40 | [142] |
| 17 M NaClO4 in H2O/FA = 3:7 (vol/vol) | AC||polyimide | 2.049 × 10-2/0 1.469 × 10-2/-10 8.26 × 10-3/-20 4.84 × 10-3/-30 3.10 × 10-3/-40 1.75 × 10-3/-50 | 85/99/1C/2000/-50 68/99/1C/400/-50 (pouch battery) | [143] |
| 25 M NaFSI + 10 M NaFTFS | NVOPF||NTP | - | 48/89/0.2C/500/-10 | [144] |
| 2M NaClO4 aqueous + 0.3M DMSO | AC||NTP@C | 1.1 × 10-4/-50 | 68/~90/0.5C/100/-50 | [145] |
| 1M NaNO3 in 2:1 Gly-Di | Ni2Zn[Fe(CN)6||PTCDI | ~30/80/0.1 A g-1/300/-10 | [146] | |
| Na2SO4-SiO2 in 50 mL methyl alcohol | AC||NTP@C | 7.0 × 10-5/RT | -/83.05/0.13 A g-1/500/-20 -/65.05/0.13 A g-1/500/-30 | [147] |
| 1 m NaClO4 aqueous 3.86 M CaCl2 | Na2CoFe(CN)6||AC | 1.518 × 10-2/0 5.083 × 10-2/-10 2.342 × 10-2/-20 1.523 × 10-2/-30 1.263 × 10-2/-40 7.13 × 10-3/-50 | 74.5/86.7/1C/1000/-30 | [148] |
| NaAlCl4·2SO2 | AC||Na | 9.0 × 10-5/-20 | 125.2/0.2C/400/-20 | [149] |
Inorganic electrolytes in SIBs face LT challenges such as freezing, slow ion transport, and interfacial instability. Introducing various sodium salts and WiSE systems reduces free water and disrupts hydrogen bonds, lowering the freezing point and expanding the electrochemical window. Adding organic molecules (e.g., DMSO, glycerol), inorganic frameworks (e.g., SiO2), or salts (e.g., CaCl2) helps maintain conductivity and stability. SO2-based electrolytes also show excellent LT adaptability due to high fluidity and conductivity, greatly improving LT electrochemical performance.
Co-solvent liquid electrolytes
Co-solvents can enhance ionic transport and improve the formation of stable SEI under LT conditions[150]. Certain co-solvents, such as ethers, fluorinated solvents, and nitriles, also help suppress crystallization and facilitate LT charge transfer kinetics[151]. Thus, co-solvent engineering offers a promising approach to extending the operational temperature window of SIBs.
Mogensen et al.[152] formulated 1 M sodium bis(oxalato)borate (NaBOB) in an NMP (N-Methyl-2-pyrrolidone)/TMP (Trimethyl phosphate) mixed solvent. Although TMP and NMP freeze at -46 and -25 °C, respectively. DSC shows that mixtures with ≥ 70% TMP depress the freezing point to below -60 °C. The electrolyte delivered ~1.0 × 10-3 S cm-1 at -30 °C and remained > 1.0 × 10-4 S cm-1 at -60 °C.
Liu et al.[153] designed 1 M NaClO4 SN/DEC/FEC electrolyte (47.5:47.5:5 v/v) balances high-voltage stability and LT performance. SN improves oxidative stability and promotes an inorganic CEI (Na3N/NaF). DEC reduces free SN to limit CEI dissolution and enhance LT Na+ diffusion, and FEC further stabilizes the interface. It forms a uniform 9-12 nm [Figure 13A-C]. SEI outperforms EC-PC and SN-FEC. An Na3V2O2(PO4)2F (NVOPF) cell delivers 115 mA h g-1 at -25 °C and 0.1C, while an NVOPF||HC full cell cycles stably for 100 cycles at
Figure 13. (A-C) HR-TEM images showing the CEI layer formed in EC-PC (A), SN-FEC (B), and DEC-SN-FEC (C) electrolytes. (A-C) Figures reprinted with permission from Ref.[153]. Copyright 2024 Wiley-VCH. (D) Discharge capacity of NVP/NTP using AWE in LT region. Reprinted with permission from Ref.[155]. Copyright 2024 Wiley-VCH. (E) Electrochemical performances of the Na0.44MnO2 plates in different electrolytes at different temperatures. Reprinted with permission from Ref.[156]. Copyright 2023 The American Chemical Society.
Zheng et al.[154] developed a weakly solvating ternary electrolyte (0.8 M NaPF6 in FEC/EMC/HFE = 3:3:4 v/v): FEC forms a fluoride-rich SEI, EMC lowers viscosity/melting point, and weakly coordinating HFE reduces solvation strength, decreasing the Na+ coordination number from 5.1 to 3.7. It remains liquid at -30 °C
Chua et al.[156] developed an ethanol/water hybrid electrolyte, where the optimal 5:1 ratio enables ethanol (freezing point -114 °C) to form cross H-bonds with water and disrupt its H-bond network, improving interactions and wettability and widening the electrochemical window from ~1.8 to 2.5 V [Figure 13E]. At
Composition of co-solvent liquid electrolytes and their key LT electrochemical properties
| Composites | Cathode||Anode | Ionic conductivity (S cm-1)/ temperature (°C) | End of capacity (mA h g-1)/ capacity retention (%)/ current density/ cycle number measuring temperature (°C) | Reference |
| 1 M NaBOB in NMP/TMP (> 70%) | - | 1.0 × 10-3/-30 > 1.0 × 10-4/-60 | 115/92/0.1C/120/-25 | [152] |
| 1 M NaClO4/DEC:SN:FEC (vol/vol 45:47:5) | NVOPF||Na | - | 115/92/0.1C/120/-25 | [153] |
| 0.8 M NaPF6 in FEC/EMC/HFE (3:3:4 by vol) | NVPOF||Na | - | ~89/88.7/0.5C/600/-20 | [154] |
| NaTFSI:H2O:ADN = 1:1:2.5 | NVP||NTP | - | ~88/~98/1C/30/-10 | [155] |
| 1M Na0.44MnO2 ethanol/ethanol = 5:1(vol/vol) | Na0.44MnO2||Zn | - | 47.34/94/50 mA g-1/50/0 | [156] |
Mixed-solvent designs that pair solvents of different polarity can depress the freezing point and optimize Na+ solvation. Tuning solvent ratios also reduces reactive free species, improving LT electrolyte stability, interfacial robustness, and ion-transport pathways, thereby enhancing LT cycling.
CONCLUSIONS AND PERSPECTIVES
This review summarizes the major classes and recent progress of LT electrolytes for SIBs. Although multiple strategies have improved LT performance, key limitations persist and substantial challenges remain.
Organic liquid electrolytes remain the most mature option for LT SIBs; however, their performance is limited by higher viscosity, lower salt solubility, and increased Na+ desolvation barriers at LTs. Molecular and formulation engineering can reduce desolvation energy and improve interfacial charge-transfer kinetics, thereby enhancing rate capability and cycling stability. Ester-based electrolytes offer good oxidative stability and electrode compatibility, yet their viscosity typically rises sharply at LT, impeding ion transport and requiring further molecular/design optimization to boost ionic conductivity. Ether-based electrolytes, in contrast, feature lower viscosity and better LT ion mobility, facilitating subzero transport and suppressing salt crystallization. Functional additives can further mitigate freezing, stabilize interphases, and maintain fluidity. Ionic-liquid electrolytes can also preserve relatively high conductivity and stable solvation at LT, making them promising for extremely cold operation.
Inorganic liquid electrolytes, especially aqueous systems, offer a high-safety, high-conductivity route for LT SIBs. They are nonflammable, low-cost, and environmentally benign, and typically deliver room-temperature ionic conductivity about two orders of magnitude higher than organic electrolytes, supporting LT and high-rate operation. However, aqueous electrolytes readily freeze at LTs, impeding ion transport, increasing interfacial resistance, and compromising electrochemical stability. To mitigate this, approaches such as high-concentration Wise, antifreeze co-solvents, and hydrogel electrolytes are used to reduce free-water activity, disrupt hydrogen-bond networks, and broaden the LT stability window. In addition, nonflammable inorganic systems such as SO2-based electrolytes can retain relatively high LT conductivity, offering promise for extreme-environment energy storage.
Co-solvent engineering improves LT electrolytes by combining solvents to tune viscosity and solvation, thereby enhancing ion transport, stabilizing interfaces, suppressing salt crystallization, and reducing charge-transfer resistance. Approaches such as carbonate-non-carbonate coupling, multi-solvent blending, and weakly solvating fluorinated ethers can achieve low melting points/viscosity while optimizing Na+ solvation-desolvation and interfacial stability, ultimately broadening the LT window of SIBs.
Solid-state and quasi-solid electrolytes offer high safety and structural stability for LT SIBs, but are still constrained by low LT ionic conductivity and high interfacial resistance. Inorganic solids have high intrinsic conductivity and thermal stability, yet their rigidity causes poor solid-solid contact at LT. Polymer electrolytes are flexible and processable, but segmental freezing severely limits ion transport. Gel electrolytes retain higher conductivity via liquid components while improving interfacial compatibility, and composite solid electrolytes leverage organic-inorganic synergy to strengthen mechanics, enhance transport, and partially suppress dendrites, though LT interfacial stability and kinetics still need further optimization.
SIBs have significant potential for future energy storage, especially in high-safety LT applications. The material composition, costs, and manufacturing of electrolytes must be carefully considered for practical use. Research on SIB electrolytes under LT conditions will drive the development of high-safety SIBs.
We compare the advantages and disadvantages of the different types of electrolytes in Table 5.
Pros and cons of the different types of electrolytes
| Type | Key characteristics | Pros and cons | Application scenarios and design considerations |
| Organic liquid electrolyte | Mature Wide operating voltage window Melting point Viscosity Solvation energy Desolvation energy | Pros: Suppress salt crystallization Low viscosity LT conductivity Cons: Interfacial kinetics slowdown SEI/CEI degradation Polarization exacerbation | Wide-temperature Reduce desolvation energy Functional additives Tune solvation structure Tune interfacial chemistry |
| Inorganic liquid electrolyte | Nonflammable Safety Environmental friendliness Higher ionic conductivity | Pros: Improved safety Favorable cost Favorable processing conditions Cons: Prone to freezing at LT Suppress water/solvent crystallization Polarization | High-safety applications Grid storage LT outdoor use Suppress crystallization Hydrogen-bond-disrupting co-solvents/additives Enhance anti-freezing |
| Co-solvent liquid electrolytes | Co-solvent systems Synergistically enhance | Pros: Conductivity Interfacial stability Reduce LT polarization Cons: Narrow formulation window High-voltage stability Trade-offs | Wide-temperature operation Carbonate-noncarbonate blends Weakly solvating fluorinated ethers |
| Solid-state electrolytes | High safety Inorganic solid Polymers gels/composites Suppress dendrites | Pros: High safety Stable interfaces Cons: Bulk ionic conductivity Interfacial contact bottleneck at LT | High-safety applications Extreme environments Material composites Enhance LT ion transport |
Moreover, to enable practical, low-carbon deployment of LT electrolytes, sodium-salt selection should prioritize scalability and sustainability, guided by solubility, thermal stability, and dissociation in weakly solvating/low-dielectric media. LT solubility governs the attainable concentration, precipitation risk, charge-carrier density, and conductivity retention, while Na+ desolvation/interfacial transport - linked to ion pairing/aggregation - can be assessed via conductivity-concentration behavior, ionicity, and tNa+. Thus, next-generation commercial salts should offer sufficient LT solubility, stable anions with minimal safety/regulatory concerns, and high dissociation to suppress aggregation and interfacial polarization.
Meanwhile, dry electrode technology has emerged as an attractive alternative to conventional wet slurry coating. Dry-electrode processing eliminates slurry solvents (e.g., NMP/water), reducing residual solvent/moisture, electrolyte consumption, HF formation, and LT interfacial side reactions. It also enables stronger conductive/binder networks and optimized porosity/tortuosity to lower charge-transfer resistance and transport polarization. For sodium metal, improved current uniformity helps achieve uniform deposition and suppress dendrites. Overall, integrating dry electrodes with LT-optimized electrolytes via co-design can extend the LT operating window and support greener, scalable manufacturing.
DECLARATIONS
Authors’ contributions
Data sourcing, collection, and original draft writing: Li, S.; Zhao, Y.
Data analysis and interpretation: Dang, Q.; Qin, J.; Hu, Q.
Editing and supervision: Hu, Q.
All authors participated in preparing the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This research was supported by fund project of the Science and Technology Project of Guangxi, China under Grant No. GK AD23026120, fund Project of the Guangxi Key R&D Programme Projects (No. AB24010346), fund Project of the Guangxi Key Laboratory of Automobile Components and Vehicle Technology under Grant No. 2023GKLACVTZZ10, Doctoral foundation of Guangxi University of Science and Technology under Grant No. XKB 22Z11 and No. XKB 22Z18.
Conflicts of interest
Hu, Q. is affiliated with Zhejiang Kaili New Materials Co., Ltd., China. There were no potential conflicts of interest during the study, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Hwang, J. Y.; Myung, S. T.; Sun, Y. K. Sodium-ion batteries: present and future. Chem. Soc. Rev. 2017, 46, 3529-614.
2. Yang, S.; Cheng, K.; Cao, Z. Status and strategies of electrolyte engineering for low-temperature sodium-ion batteries. J. Mater. Chem. A. 2024, 12, 13059-80.
3. Monti, D.; Jónsson, E.; Boschin, A.; Palacín, M. R.; Ponrouch, A.; Johansson, P. Towards standard electrolytes for sodium-ion batteries: physical properties, ion solvation and ion-pairing in alkyl carbonate solvents. Phys. Chem. Chem. Phys. 2020, 22, 22768-77.
4. Ould, D. M. C.; Oswald, S.; Smith, H. E.; et al. Properties of NaPF6 electrolytes and effect of electrolyte concentration on performance in sodium-ion batteries. Chem. Commun. 2025, 61, 8516-9.
5. Song, Z.; Xing, Z.; Yang, J.; et al. Electrolyte chemistry development for sodium-based batteries: a blueprint from lithium or a step toward originality? Angew. Chem. Int. Ed. 2025, 64, e202424543.
6. Zhu, P.; Gastol, D.; Marshall, J.; Sommerville, R.; Goodship, V.; Kendrick, E. A review of current collectors for lithium-ion batteries. J. Power. Sources. 2021, 485, 229321.
7. Fan, T.; Xie, H. Sb2S3-rGO for high-performance sodium-ion battery anodes on Al and Cu foil current collector. J. Alloys. Compd. 2019, 775, 549-53.
8. He, X. X.; Li, L.; Wu, X.; Chou, S. L. Sustainable hard carbon for sodium-ion batteries: precursor design and scalable production roadmaps. Adv. Mater. 2025, 37, e2506066.
9. Lege, N.; He, X.; Wang, Y.; et al. Reappraisal of hard carbon anodes for practical lithium/sodium-ion batteries from the perspective of full-cell matters. Energy. Environ. Sci. 2023, 16, 5688-720.
10. Chayambuka, K.; Cardinaels, R.; Gering, K. L.; et al. An experimental and modeling study of sodium-ion battery electrolytes. J. Power. Sources. 2021, 516, 230658.
11. Morales, D.; Chagas, L. G.; Paterno, D.; Greenbaum, S.; Passerini, S.; Suarez, S. Transport studies of NaPF6 carbonate solvents-based sodium ion electrolytes. Electrochim. Acta. 2021, 377, 138062.
12. Wang, P.; Qing, H.; Zhang, R.; Li, W. Fluorine-free electrolytes for sustainable lithium batteries: a review. NPJ. Mater. Sustain. 2025, 3, 32.
13. Tawalbeh, T.; Rangarajan, S.; Liu, H.; Wang, Y. Fluorinated substances in lithium-ion batteries and solid state batteries: Recycling challenges and environmental impacts. J. Power. Sources. 2025, 652, 237635.
14. Vinay, B.; Nikodimos, Y.; Agnihotri, T.; et al. Fluorine-free electrolytes in batteries: principles, strategies, and advances. Energy. Environ. Sci. 2025, 18, 7326-72.
15. Zhang, N.; Wang, B.; Jin, F.; et al. Modified cathode-electrolyte interphase toward high-performance batteries. Cell. Rep. Phys. Sci. 2022, 3, 101197.
16. Aslam, M. N.; Lu, Y.; Zhang, S.; et al. Development and characterization of laboratory-scale sodium-ion battery full cell containing low fluorinated electrolyte. J. Power. Sources. 2025, 660, 238548.
17. Fan, W.; Wang, A.; Cai, J.; Shi, L.; Wang, W.; Nan, J. Electrolyte salts for large-scale application of sodium-ion batteries: NaPF6 and emerging alternatives. J. Power. Sources. 2025, 650, 237519.
18. Wu, C.; Yang, Y.; Zhang, Y.; et al. Industrial-scale hard carbon designed to regulate electrochemical polarization for fast sodium storage. Angew. Chem. Int. Ed. 2024, 63, e202406889.
19. Li, P.; Hu, N.; Wang, J.; Wang, S.; Deng, W. Recent progress and perspective: Na ion batteries used at low temperatures. Nanomaterials 2022, 12, 3529.
20. Belgibayeva, A.; Rakhmetova, A.; Rakhatkyzy, M.; et al. Lithium-ion batteries for low-temperature applications: limiting factors and solutions. J. Power. Sources. 2023, 557, 232550.
21. Zhang, X.; Zhou, S.; Chen, X.; et al. Organic liquid electrolyte for low-temperature sodium-ion batteries. Chin. Chem. Lett. 2025, 111533.
22. Sai Bhargava Reddy, M.; Jeong, D.; Aich, S.; Pol, V. G. Low-temperature sodium-ion batteries: challenges, engineering strategies, safety considerations, and future directions. EES. Batteries. 2025, 1, 1444-80.
23. Bai, H.; Zhu, X.; Ao, H.; He, G.; Xiao, H.; Chen, Y. Advances in sodium-ion batteries at low-temperature: Challenges and strategies. J. Energy. Chem. 2024, 90, 518-39.
24. Qiu, X.; Chen, Y.; Sun, Y.; et al. Research on low-temperature sodium-ion batteries: challenges, strategies and prospect. Energy. Storage. Mater. 2024, 72, 103760.
25. Yang, Y.; Yang, W.; Yang, H.; Zhou, H. Electrolyte design principles for low-temperature lithium-ion batteries. eScience 2023, 3, 100170.
26. Glushenkov, A. M. Recent commentaries on the expected performance, advantages and applications of sodium-ion batteries. Energy. Mater. 2023, 3, 300010.
27. Su, Q.; Hu, C.; Li, S.; et al. Sodium-ion batteries at low temperature: Storage mechanism and modification strategies. Chin. Chem. Lett. 2025, 36, 111267.
28. Hao, M.; Weng, S.; Zhong, C.; Li, Y.; Wang, X. Structure and evolution of solid electrolyte interphase (SEI) at the electrode-electrolyte interface. Mater. Today. Energy. 2025, 53, 101998.
29. Singh, H.; Damron, J. T.; Shahriar, M.; et al. In-situ polymerized and crosslinked electrolytes with interchangeable Li/Na transport for battery applications. Energy. Mater. 2025, 5, 500149.
30. Yoo, D. J.; Liu, Q.; Cohen, O.; Kim, M.; Persson, K. A.; Zhang, Z. Understanding the role of SEI layer in low-temperature performance of lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2022, 14, 11910-8.
31. Zhang, J.; Li, J.; Wang, H.; Wang, M. Research progress of organic liquid electrolyte for sodium ion battery. Front. Chem. 2023, 11, 1253959.
32. Yang, M.; Luo, J.; Guo, X.; Chen, J.; Cao, Y.; Chen, W. Aqueous rechargeable sodium-ion batteries: from liquid to hydrogel. Batteries 2022, 8, 180.
33. Lin, Y.; Peng, Q.; Chen, L.; et al. Organic liquid electrolytes in sodium-based batteries: actualities and perspectives. Energy. Storage. Mater. 2024, 67, 103211.
34. Wang, Z.; Guo, X.; Dong, Y.; et al. Low-concentration electrolyte engineering for rechargeable batteries. Adv. Mater. 2025, 37, e2501215.
35. Zhang, L.; Sun, Y.; Zhou, Y.; et al. Investigation of the synergetic effects of LiBF4 and LiODFB as wide-temperature electrolyte salts in lithium-ion batteries. Ionics 2018, 24, 2995-3004.
36. Pham, T. D.; Bin, Faheem. A.; Kim, J.; Kwak, K.; Lee, K. K. Unlocking the potential of lithium metal batteries with a sulfite-based electrolyte. Adv. Funct. Mater. 2023, 33, 2305284.
37. Yang, J.; Wang, M.; Ruan, J.; et al. Research progress in non-aqueous low-temperature electrolytes for sodium-based batteries. Sci. China. Chem. 2024, 67, 4063-84.
38. Yi, Y.; Qi, J.; Yang, X.; Yang, H.; Zeng, G. Research progress on ester-based low-temperature electrolytes for sodium-ion batteries. Mater. Sci. Eng. B. 2025, 320, 118443.
39. Vidal-Abarca, C.; Lavela, P.; Tirado, J.; Chadwick, A.; Alfredsson, M.; Kelder, E. Improving the cyclability of sodium-ion cathodes by selection of electrolyte solvent. J. Power. Sources. 2012, 197, 314-8.
40. Ponrouch, A.; Goñi, A.; Palacín, M. R. High capacity hard carbon anodes for sodium ion batteries in additive free electrolyte. Electrochem. Commun. 2013, 27, 85-8.
41. Ren, X.; Chen, S.; Lee, H.; et al. Localized high-concentration sulfone electrolytes for high-efficiency lithium-metal batteries. Chem 2018, 4, 1877-92.
42. Lin, S.; Yang, Z.; Chen, J.; Qiao, Y.; Li, L.; Chou, S. Functional electrolyte additives for sodium-ion and sodium-metal batteries: progress and perspectives. Adv. Funct. Mater. 2024, 34, 2400731.
43. Sun, T.; Liu, C.; Wang, J.; et al. A phenazine anode for high-performance aqueous rechargeable batteries in a wide temperature range. Nano. Res. 2020, 13, 676-83.
44. Pal, U.; Girard, G. M. A.; O’dell, L. A.; et al. Improved Li-ion transport by DME chelation in a novel ionic liquid-based hybrid electrolyte for Li-S battery application. J. Phys. Chem. C. 2018, 122, 14373-82.
45. Aguilera, L.; Scheers, J.; Matic, A. Enhanced low-temperature ionic conductivity via different Li+ solvated clusters in organic solvent/ionic liquid mixed electrolytes. Phys. Chem. Chem. Phys. 2016, 18, 25458-64.
46. Nakamoto, K.; Tanaka, M.; Sakamoto, R.; Ito, M.; Okada, S. Kinetic effect of local pH on high-voltage aqueous sodium-ion batteries. Electrochemistry 2024, 92, 017003.
47. Xu, Z.; Ma, R.; Wang, X. Ultrafast, long-life, high-loading, and wide-temperature zinc ion supercapacitors. Energy. Storage. Mater. 2022, 46, 233-42.
48. Xing, Z.; Zhao, W.; Yu, B.; et al. Electrolyte design strategies for aqueous sodium-ion batteries: progress and prospects. Small 2024, 20, e2405442.
49. Wang, C. Y.; Zhang, G.; Ge, S.; et al. Lithium-ion battery structure that self-heats at low temperatures. Nature 2016, 529, 515-8.
50. Zhang, G.; Ge, S.; Xu, T.; Yang, X.; Tian, H.; Wang, C. Rapid self-heating and internal temperature sensing of lithium-ion batteries at low temperatures. Electrochim. Acta. 2016, 218, 149-55.
51. Ruan, Y.; Song, S.; Liu, J.; et al. Improved structural stability and ionic conductivity of Na3Zr2Si2PO12 solid electrolyte by rare earth metal substitutions. Ceram. Int. 2017, 43, 7810-5.
52. Wang, S.; Xiao, S.; Li, S.; et al. Organic Cationic-coordinated perfluoropolymer electrolytes with strong Li+-solvent interaction for solid state Li-metal batteries. Angew. Chem. Int. Ed. 2024, 63, e202412434.
53. Li, X. S.; Liang, J.; Cao, X.; et al. Research progress of inorganic solid electrolyte materials for all-solid-state sodium-ion batteries. Rare. Metals. 2025, 44, 2871-99.
54. Wu, M.; Liu, H.; Qi, X.; et al. Structure designing, interface engineering, and application prospects for sodium-ion inorganic solid electrolytes. InfoMat 2024, 6, e12606.
55. Zhao, S.; Che, H.; Chen, S.; et al. Research progress on the solid electrolyte of solid-state sodium-ion batteries. Electrochem. Energy. Rev. 2024, 7, 3.
56. Zhou, Q.; Ma, J.; Dong, S.; Li, X.; Cui, G. Intermolecular chemistry in solid polymer electrolytes for high-energy-density lithium batteries. Adv. Mater. 2019, 31, e1902029.
57. Gebert, F.; Knott, J.; Gorkin, R.; Chou, S.; Dou, S. Polymer electrolytes for sodium-ion batteries. Energy. Storage. Mater. 2021, 36, 10-30.
58. Nguyen, A. G.; Verma, R.; Song, G. C.; Kim, J.; Park, C. J. In situ polymerization on a 3D ceramic framework of composite solid electrolytes for room-temperature solid-state batteries. Adv. Sci. 2023, 10, e2207744.
59. Landesfeind, J.; Hosaka, T.; Graf, M.; Kubota, K.; Komaba, S.; Gasteiger, H. A. Comparison of ionic transport properties of non-aqueous lithium and sodium hexafluorophosphate electrolytes. J. Electrochem. Soc. 2021, 168, 040538.
60. Kulova, T. L.; Skundin, A. M. Electrode/Electrolyte interphases of sodium-ion batteries. Energies 2022, 15, 8615.
61. Zheng, J.; Zhang, J.; Li, W.; Ge, J.; Chen, W. Phosphate-based gel polymer electrolyte enabling remarkably long cycling stable sodium storage in a wide-operating-temperature. Chem. Eng. J. 2023, 465, 142796.
62. Lei, Z. Q.; Guo, Y. J.; Wang, E. H.; et al. Layered oxide cathode-electrolyte interface towards na-ion batteries: advances and perspectives. Chem. Asian. J. 2022, 17, e202200213.
63. Feng, Y. H.; Liu, M.; Wu, J.; et al. Monolithic interphase enables fast kinetics for high-performance sodium-ion batteries at subzero temperature. Angew. Chem. Int. Ed. 2024, 63, e202403585.
64. Chen, J.; Peng, Y.; Yin, Y.; et al. A Desolvation-free sodium dual-ion chemistry for high power density and extremely low temperature. Angew. Chem. Int. Ed. 2021, 60, 23858-62.
65. Deng, L.; Goh, K.; Yu, F.; et al. Self-optimizing weak solvation effects achieving faster low-temperature charge transfer kinetics for high-voltage Na3V2(PO4)2F3 cathode. Energy. Storage. Mater. 2022, 44, 82-92.
66. Hu, Y.; Lu, Y. The mystery of electrolyte concentration: from superhigh to ultralow. ACS. Energy. Lett. 2020, 5, 3633-6.
67. Yu, D.; Wang, Z.; Yang, J.; et al. Low-temperature and fast-charge sodium metal batteries. Small 2024, 20, e2311810.
68. Thenuwara, A. C.; Shetty, P. P.; Kondekar, N.; Wang, C.; Li, W.; Mcdowell, M. T. Enabling highly reversible sodium metal cycling across a wide temperature range with dual-salt electrolytes. J. Mater. Chem. A. 2021, 9, 10992-1000.
69. Fan, W.; Wang, W.; Xie, Q.; et al. A sodium bis(fluorosulfonyl)imide (NaFSI)-based multifunctional electrolyte stabilizes the performance of NaNi1/3Fe1/3Mn1/3O2/hard carbon sodium-ion batteries. Chemistry 2024, 30, e202401321.
70. Zhang, J.; Wang, D. W.; Lv, W.; et al. Ethers illume sodium-based battery chemistry: uniqueness, surprise, and challenges. Adv. Energy. Mater. 2018, 8, 1801361.
71. Li, Q.; Liu, G.; Cheng, H.; Sun, Q.; Zhang, J.; Ming, J. Low-temperature electrolyte design for lithium-ion batteries: prospect and challenges. Chemistry 2021, 27, 15842-65.
72. Sun, N.; Li, R.; Zhao, Y.; et al. Anionic coordination manipulation of multilayer solvation structure electrolyte for high-rate and low-temperature lithium metal battery. Adv. Energy. Mater. 2022, 12, 2200621.
73. Yun, S.; Liang, X.; Xi, J.; et al. Electrolytes for high-safety lithium-ion batteries at low temperature: a review. Polymers 2024, 16, 2661.
74. Lin, X.; Du, X.; Tsui, P. S.; Huang, J.; Tan, H.; Zhang, B. Exploring room- and low-temperature performance of hard carbon material in half and full Na-ion batteries. Electrochim. Acta. 2019, 316, 60-8.
75. Ma, X.; Wei, Y.; Wu, Y.; et al. High crystalline Na2Ni[Fe(CN)6] particles for a high-stability and low-temperature sodium-ion batteries cathode. Electrochim. Acta. 2019, 297, 392-7.
76. Che, H.; Yang, X.; Yu, Y.; et al. Engineering optimization approach of nonaqueous electrolyte for sodium ion battery with long cycle life and safety. Green. Energy. Environ. 2021, 6, 212-9.
77. Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 2014, 114, 11503-618.
78. Jayakumar, R.; Pollard, T. P.; Borodin, O.; et al. Weakly solvating ester electrolyte for high voltage sodium-ion batteries. Nano. Energy. 2024, 128, 109969.
79. Desai, P.; Abou-rjeily, J.; Tarascon, J.; Mariyappan, S. Practicality of methyl acetate as a co-solvent for fast charging Na-ion battery electrolytes. Electrochim. Acta. 2022, 416, 140217.
80. Liu, Y.; Liang, H.; Du, M.; et al. Ester-based anti-freezing electrolyte achieving ultra-low temperature cycling for sodium-ion batteries. J. Mater. Sci. Technol. 2024, 182, 111-8.
81. Hu, C.; Dai, L.; Huang, F.; et al. Carboxylate-based electrolyte with bilateral functions enable working sodium-metal batteries at -60°C. Angew. Chem. Int. Ed. 2025, 64, e202508584.
82. Zheng, Y.; Sun, M.; Yu, F.; et al. Utilizing weakly-solvated diglyme-based electrolyte to achieve a 10,000-cycles durable Na3V2(PO4)2F3 cathode endured at -20 °C. Nano. Energy. 2022, 102, 107693.
83. Wang, Y.; Lan, H.; Dong, S.; et al. A high-power rechargeable sodium-ion full battery operating at -40 °C. Adv. Funct. Mater. 2024, 34, 2315498.
84. Yang, J.; Guo, X.; Gao, H.; et al. A high-performance alloy-based anode enabled by surface and interface engineering for wide-temperature sodium-ion batteries. Adv. Energy. Mater. 2023, 13, 2300351.
85. Tang, Z.; Wang, H.; Wu, P. F.; et al. Electrode-electrolyte interfacial chemistry modulation for ultra-high rate sodium-ion batteries. Angew. Chem. Int. Ed. 2022, 61, e202200475.
86. Yang, C.; Liu, X.; Lin, Y.; Yin, L.; Lu, J.; You, Y. Entropy-driven solvation toward low-temperature sodium-ion batteries with temperature-adaptive feature. Adv. Mater. 2023, 35, e2301817.
87. Zhou, J.; Ding, Y.; Wang, Y.; et al. Bulk bismuth anodes for wide-temperature sodium-ion batteries enabled by electrolyte chemistry modulation. J. Colloid. Interface. Sci. 2024, 657, 502-10.
88. Fang, H.; Huang, Y.; Hu, W.; et al. Regulating ion-dipole interactions in weakly solvating electrolyte towards ultra-low temperature sodium-ion batteries. Angew. Chem. Int. Ed. 2024, 63, e202400539.
89. Abraham, K. M. How comparable are sodium-ion batteries to lithium-ion counterparts? ACS. Energy. Lett. 2020, 5, 3544-7.
90. Chen, J.; Peng, Y.; Yin, Y.; et al. High energy density Na-metal batteries enabled by a tailored carbonate-based electrolyte. Energy. Environ. Sci. 2022, 15, 3360-8.
91. Cai, J.; Fan, W.; Li, X.; et al. A dual-functional electrolyte additive for stabilizing the solid electrolyte interphase and solvation structure to enable pouch sodium ion batteries with high performance at a wide temperature range from -30 °C to 60 °C. Chem. Eng. J. 2024, 491, 151949.
92. He, M.; Zhu, L.; Ye, G.; et al. Tuning the electrolyte and interphasial chemistry for all-climate sodium-ion batteries. Angew. Chem. Int. Ed. 2024, 63, e202401051.
93. Song, X.; Meng, T.; Deng, Y.; et al. The effects of the functional electrolyte additive on the cathode material Na0.76Ni0.3Fe0.4Mn0.3O2 for sodium-ion batteries. Electrochim. Acta. 2018, 281, 370-7.
94. Liao, Y.; Yuan, L.; Han, Y.; et al. Pentafluoro(phenoxy)cyclotriphosphazene stabilizes electrode/electrolyte interfaces for sodium-ion pouch cells of 145 Wh Kg-1. Adv. Mater. 2024, 36, e2312287.
95. Yin, L.; Wang, M.; Xie, C.; Yang, C.; Han, J.; You, Y. High-voltage cyclic ether-based electrolytes for low-temperature sodium-ion batteries. ACS. Appl. Mater. Interfaces. 2023, 15, 9517-23.
96. Zhong, S.; Yu, Y.; Yang, Y.; et al. Molecular engineering on solvation structure of carbonate electrolyte toward durable sodium metal battery at -40 °C. Angew. Chem. Int. Ed. 2023, 62, e202301169.
97. Liang, H. J.; Liu, H. H.; Zhao, X. X.; et al. Electrolyte chemistry toward ultrawide-temperature (-25 to 75 °C) sodium-ion batteries achieved by phosphorus/silicon-synergistic interphase manipulation. J. Am. Chem. Soc. 2024, 146, 7295-304.
98. Yamaguchi, M.; Sato, M.; Takeoka, Y.; Rikuakwa, M.; Yoshizawa-Fujita, M. Zwitterion-enhanced performance of pyrrolidinium-based ionic liquid electrolytes as sodium-ion conductors. Electrochim. Acta. 2025, 527, 146269.
99. Ding, C.; Nohira, T.; Hagiwara, R.; Fukunaga, A.; Sakai, S.; Nitta, K. Electrochemical performance of hard carbon negative electrodes for ionic liquid-based sodium ion batteries over a wide temperature range. Electrochim. Acta. 2015, 176, 344-9.
100. Hwang, J.; Matsumoto, K.; Hagiwara, R. Electrolytes toward high-voltage Na3V2(PO4)2F3 positive electrode durable against temperature variation. Adv. Energy. Mater. 2020, 10, 2001880.
101. Bellusci, M.; Simonetti, E.; De, Francesco. M.; Appetecchi, G. B. Ionic liquid electrolytes for safer and more reliable sodium battery systems. Appl. Sci. 2020, 10, 6323.
102. Hu, X.; Matios, E.; Zhang, Y.; Wang, C.; Luo, J.; Li, W. Deeply cycled sodium metal anodes at low temperature and in lean electrolyte conditions. Angew. Chem. Int. Ed. 2021, 60, 5978-83.
103. Hayashi, A.; Noi, K.; Sakuda, A.; Tatsumisago, M. Superionic glass-ceramic electrolytes for room-temperature rechargeable sodium batteries. Nat. Commun. 2012, 3, 856.
104. Wu, J. F.; Zhang, R.; Fu, Q. F.; et al. Inorganic solid electrolytes for all-solid-state sodium batteries: fundamentals and strategies for battery optimization. Adv. Funct. Mater. 2020, 31, 2008165.
105. Wang, S.; Sun, Q.; Zhang, Q.; et al. Li-ion transfer mechanism of ambient-temperature solid polymer electrolyte toward lithium metal battery. Adv. Energy. Mater. 2023, 13, 2204036.
106. Wang, X.; Fan, Y.; Li, J.; et al. Comprehensive understanding of the NaxZr2SixPxO12 solid-state electrolyte in advanced sodium metal batteries: a critical review. Energy. Environ. Sc. 2025, 18, 1096-129.
107. Singh, K.; Chakraborty, A.; Thirupathi, R.; Omar, S. Recent advances in NASICON-type oxide electrolytes for solid-state sodium-ion rechargeable batteries. Ionics 2022, 28, 5289-319.
108. Tiliakos, A.; Iordache, M.; Marinoiu, A. Ionic conductivity and dielectric relaxation of NASICON superionic conductors at the near-cryogenic regime. Appl. Sci. 2021, 11, 8432.
109. Wang, Q.; Yu, C.; Li, L.; et al. Sc-doping in Na3Zr2Si2PO12 electrolytes enables preeminent performance of solid-state sodium batteries in a wide temperature range. Energy. Storage. Mater. 2023, 54, 135-45.
110. Jaschin, P. W.; Tang, C. R.; Wachsman, E. D. Fast charging and low temperature capabilities of sodium solid-state batteries enabled by thin NASICON bilayer architecture. ACS. Energy. Lett. 2025, 10, 2610-6.
111. Zhang, Z.; Ramos, E.; Lalère, F.; et al. Na11Sn2PS12: a new solid state sodium superionic conductor. Energy. Environ. Sci. 2018, 11, 87-93.
112. Zhang, X.; Huang, Y.; Hu, X.; et al. Seamless stitching of redox windows to enable high-voltage resilient solid sodium ion batteries. Energy. Environ. Mater. 2023, 6, e12477.
113. Wang, H.; Chen, Y.; Hood, Z. D.; et al. Revealing the structural stability and Na-Ion mobility of 3D superionic conductor Na3SbS4 at extremely low temperatures. ACS. Appl. Energy. Mater. 2018, 1, 7028-34.
114. Feng, X.; Fang, H.; Liu, P.; et al. Heavily tungsten-doped sodium thioantimonate solid-state electrolytes with exceptionally low activation energy for ionic diffusion. Angew. Chem. Int. Ed. 2021, 60, 26158-66.
115. Li, Z. Y.; Li, Z.; Fu, J. L.; Guo, X. Sodium-ion conducting polymer electrolytes. Rare. Metals. 2022, 42, 1-16.
116. Zheng, J.; Li, W.; Liu, X.; Zhang, J.; Feng, X.; Chen, W. Progress in Gel Polymer electrolytes for sodium-ion batteries. Energy. Environ. Mater. 2022, 6, e12422.
117. Ahmad, F.; Shahzad, A.; Sarwar, S.; et al. Gel polymer electrolyte composites in sodium-ion batteries: synthesis methods, electrolyte formulations, and performance analysis. J. Power. Sources. 2024, 619, 235221.
118. Aruchamy, K.; Ramasundaram, S.; Divya, S.; Chandran, M.; Yun, K.; Oh, T. H. Gel polymer electrolytes: advancing solid-state batteries for high-performance applications. Gels 2023, 9, 585.
119. He, Y.; Yang, S.; Liu, C.; et al. Composite polymer solid electrolytes for all-solid-state sodium batteries. Small. Methods. 2025, e2402220.
120. Yang, Z.; Jiang, H.; Li, X.; et al. Fabricating wide-temperature-range quasi-solid sodium batteries with fast ion transport via tin additives. Adv. Funct. Mater. 2024, 34, 2407713.
121. Lonchakova, O.; Semenikhin, O.; Zakharkin, M.; Karpushkin, E.; Sergeyev, V.; Antipov, E. Efficient gel-polymer electrolyte for sodium-ion batteries based on poly(acrylonitrile-co-methyl acrylate). Electrochim. Acta. 2020, 334, 135512.
122. Du, G.; Tao, M.; Li, J.; et al. Low-operating temperature, high-rate and durable solid-state sodium-ion battery based on polymer electrolyte and prussian blue cathode. Adv. Energy. Mater. 2019, 10, 1903351.
123. Krupina, A. A.; Kayumov, R. R.; Nechaev, G. V.; Lapshin, A. N.; Shmygleva, L. V. Polymer electrolytes based on Na-nafion plasticized by binary mixture of ethylene carbonate and sulfolane. Membranes 2022, 12, 840.
124. Voropaeva, D.; Novikova, S.; Kulova, T.; Yaroslavtsev, A. Solvation and sodium conductivity of nonaqueous polymer electrolytes based on Nafion-117 membranes and polar aprotic solvents. Solid. State. Ion. 2018, 324, 28-32.
125. Hou, J.; Zhu, T.; Wang, G.; et al. Composite electrolytes and interface designs for progressive solid-state sodium batteries. Carbon. Energy. 2024, 6, e628.
126. Helaley, A.; Liang, X. Solid-state sodium-ion batteries with composite polymer electrolytes and ALD-modified Na0.7MnO2 cathodes. Chem. Eng. J. 2025, 514, 163173.
127. Xie, D.; Zhang, M.; Wu, Y.; Xiang, L.; Tang, Y. A flexible dual-ion battery based on sodium-ion quasi-solid-state electrolyte with long cycling life. Adv. Funct. Mater. 2019, 30, 1906770.
128. Chen, C.; Li, Y.; Wang, C.; He, H.; Liu, M.; He, Y. B. A composite gel polymer electrolyte for sodium metal battery at a wide temperature range. Battery. Energy. 2024, 3, 20230048.
129. Wu, S.; Yu, Z.; Nie, X.; Wang, Z.; Huang, F.; Wei, Q. Fast ion conduction nanofiber matrix composite electrolyte for dendrite-free solid-state sodium-ion batteries with wide temperature operation. Adv. Energy. Mater. 2022, 12, 2202930.
130. Wu, S.; Ji, C.; Tang, F.; et al. Nanofiber matrix composite electrolyte for regulating ion distribution in fast kinetic sodium-ion batteries operating at wide temperatures. Energy. Storage. Mater. 2024, 67, 103308.
131. Zhu, K.; Li, Z.; Jin, T.; Jiao, L. Low defects potassium cobalt hexacyanoferrate as a superior cathode for aqueous potassium ion batteries. J. Mater. Chem. A. 2020, 8, 21103-9.
132. Dong, X.; Guo, Z.; Guo, Z.; Wang, Y.; Xia, Y. Organic batteries operated at -70 °C. Joule 2018, 2, 902-13.
133. Guo, Z.; Ma, Y.; Dong, X.; Huang, J.; Wang, Y.; Xia, Y. An environmentally friendly and flexible aqueous zinc battery using an organic cathode. Angew. Chem. Int. Ed. 2018, 57, 11737-41.
134. Fecko, C. J.; Loparo, J. J.; Roberts, S. T.; Tokmakoff, A. Local hydrogen bonding dynamics and collective reorganization in water: ultrafast infrared spectroscopy of HOD/D2O. J. Chem. Phys. 2005, 122, 54506.
135. Mohsen-Nia, M.; Amiri, H.; Jazi, B. Dielectric constants of water, methanol, ethanol, butanol and acetone: measurement and computational study. J. Solution. Chem. 2010, 39, 701-8.
136. Nian, Q.; Liu, S.; Liu, J.; et al. All-climate aqueous dual-ion hybrid battery with ultrahigh rate and ultralong life performance. ACS. Appl. Energy. Mater. 2019, 2, 4370-8.
137. Zhu, K.; Sun, Z.; Jin, T.; et al. Tailoring pure inorganic electrolyte for aqueous sodium-ion batteries operating at -60 °C. Batteries. Supercaps. 2022, 5, e202200308.
138. Zhao, B.; Hao, H.; Lei, H.; et al. An anti-freezing pure inorganic electrolyte for long cycle life aqueous sodium-ion batteries at -40 °C. Energy. Storage. Mater. 2024, 71, 103562.
139. Mishra, R. N.; Madikere, Raghunatha. Reddy. A. K.; Goulet, M.; Zaghib, K. Water-in-salt electrolytes: advances and chemistry for sustainable aqueous monovalent-metal-ion batteries. Batteries 2025, 11, 120.
140. Wang, Y.; Meng, X.; Sun, J.; Liu, Y.; Hou, L. Recent Progress in "water-in-salt" electrolytes toward non-lithium based rechargeable batteries. Front. Chem. 2020, 8, 595.
141. Sakamoto, R.; Yamashita, M.; Nakamoto, K.; et al. Local structure of a highly concentrated NaClO4 aqueous solution-type electrolyte for sodium ion batteries. Phys. Chem. Chem. Phys. 2020, 22, 26452-8.
142. Wang, X.; Huang, H.; Zhou, F.; et al. High-voltage aqueous planar symmetric sodium ion micro-batteries with superior performance at low-temperature of -40 ºC. Nano. Energy. 2021, 82, 105688.
143. Zhu, K.; Sun, Z.; Li, Z.; Liu, P.; Chen, X.; Jiao, L. Aqueous sodium ion hybrid batteries with ultra-long cycle life at -50 °C. Energy. Storage. Mater. 2022, 53, 523-31.
144. Reber, D.; Kühnel, R.; Battaglia, C. Suppressing crystallization of water-in-salt electrolytes by asymmetric anions enables low-temperature operation of high-voltage aqueous batteries. ACS. Mater. Lett. 2019, 1, 44-51.
145. Nian, Q.; Wang, J.; Liu, S.; et al. Aqueous batteries operated at -50 °C. Angew. Chem. Int. Ed. 2019, 58, 16994-9.
146. Sun, Y.; Zhang, Y.; Xu, Z.; et al. Dilute hybrid electrolyte for low-temperature aqueous sodium-ion batteries. ChemSusChem 2022, 15, e202201362.
147. Cheng, Y.; Chi, X.; Yang, J.; Liu, Y. Cost attractive hydrogel electrolyte for low temperature aqueous sodium ion batteries. J. Energy. Storage. 2021, 40, 102701.
148. Zhu, K.; Li, Z.; Sun, Z.; et al. Inorganic electrolyte for low-temperature aqueous sodium ion batteries. Small 2022, 18, e2107662.
149. Jeong, G.; Kim, H.; Lee, H. S.; et al. A room-temperature sodium rechargeable battery using an SO2-based nonflammable inorganic liquid catholyte. Sci. Rep. 2015, 5, 12827.
150. Zheng, C.; Yao, Q.; Sun, Y.; et al. Enhancing low-temperature performance of sodium-ion batteries via anion-solvent interactions. Adv. Funct. Mater. 2025, 2501303.
151. Miao, Y.; Yu, P.; Yang, F.; Zhao, Y.; Xu, H.; Wang, J. Effects of cosolvent and nonsolvating solvent on the structural dynamics of organic electrolytes in sodium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 1064-76.
152. Mogensen, R.; Buckel, A.; Colbin, S.; Younesi, R. A wide-temperature-range, low-cost, fluorine-free battery electrolyte based on sodium bis(oxalate)borate. Chem. Mater. 2021, 33, 1130-9.
153. Liu, Y.; Zhu, L.; Wang, E.; et al. Electrolyte engineering with tamed electrode interphases for high-voltage sodium-ion batteries. Adv. Mater. 2024, 36, e2310051.
154. Zheng, X.; Gu, Z.; Fu, J.; et al. Knocking down the kinetic barriers towards fast-charging and low-temperature sodium metal batteries. Energy. Environ. Sci. 2021, 14, 4936-47.
155. Wang, H.; Liu, T.; Du, X.; et al. Hybrid electrolytes enabling in-situ interphase protection and suppressed electrode dissolution for aqueous sodium-ion batteries. Batteries. Supercaps. 2022, 5, e202200246.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data







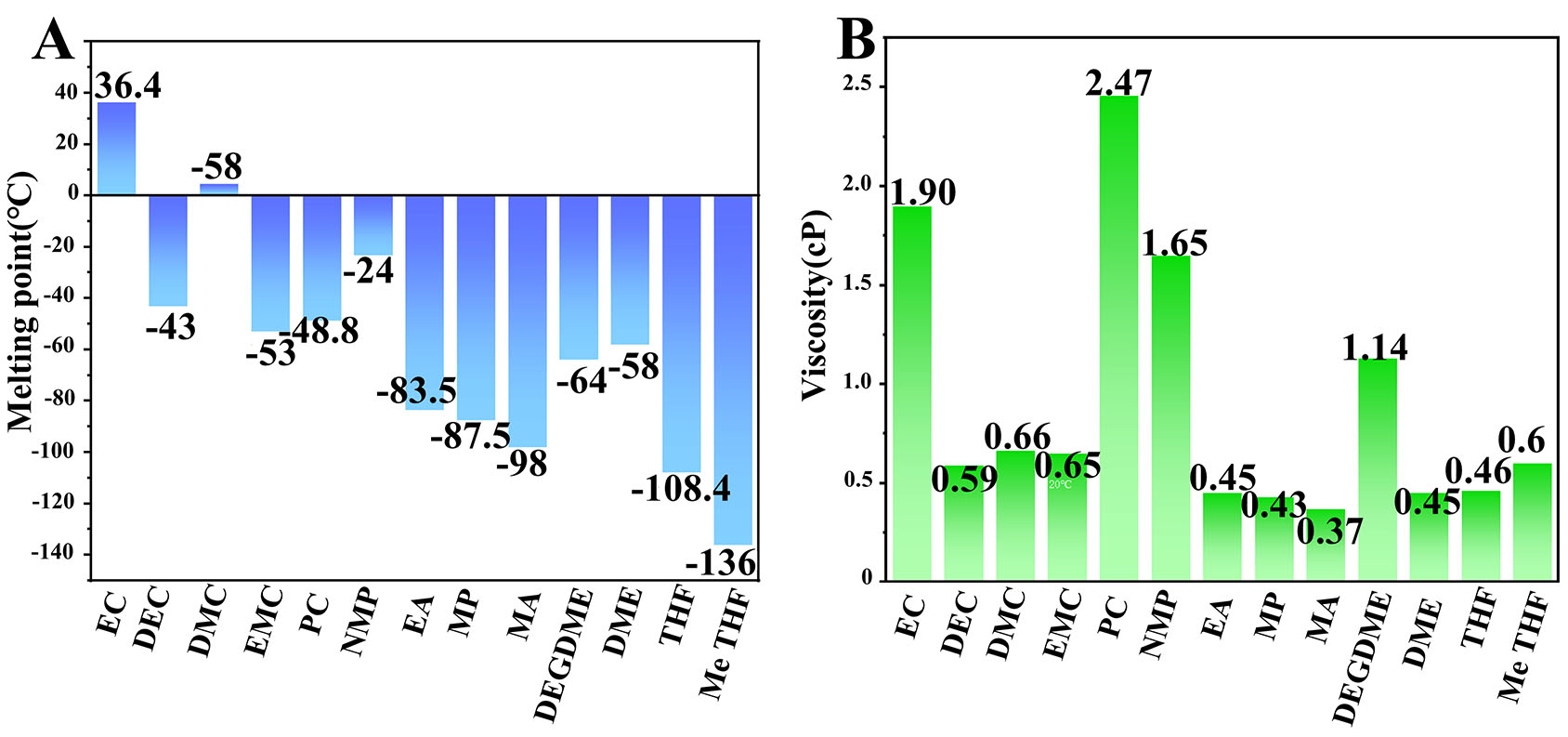
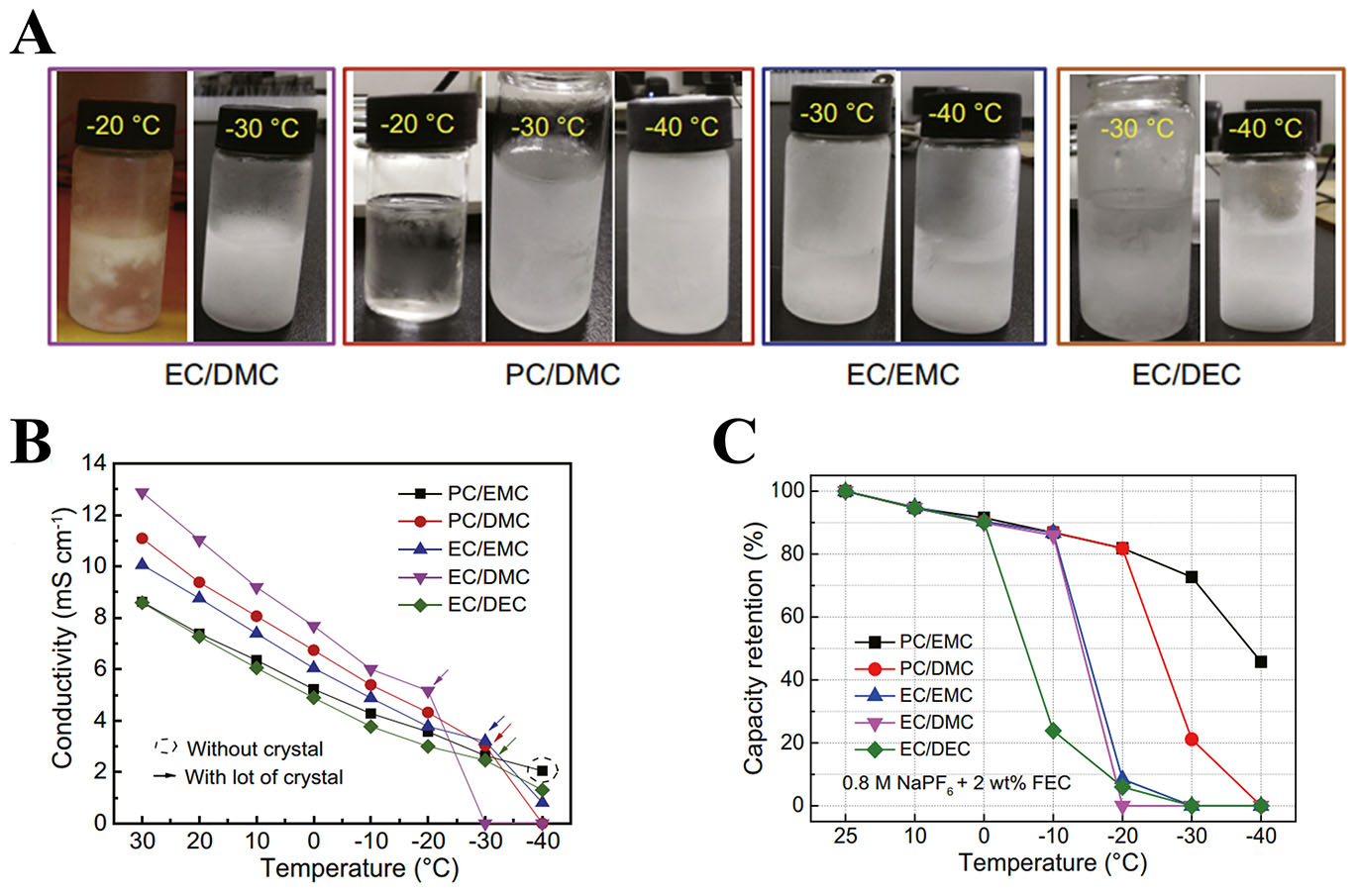
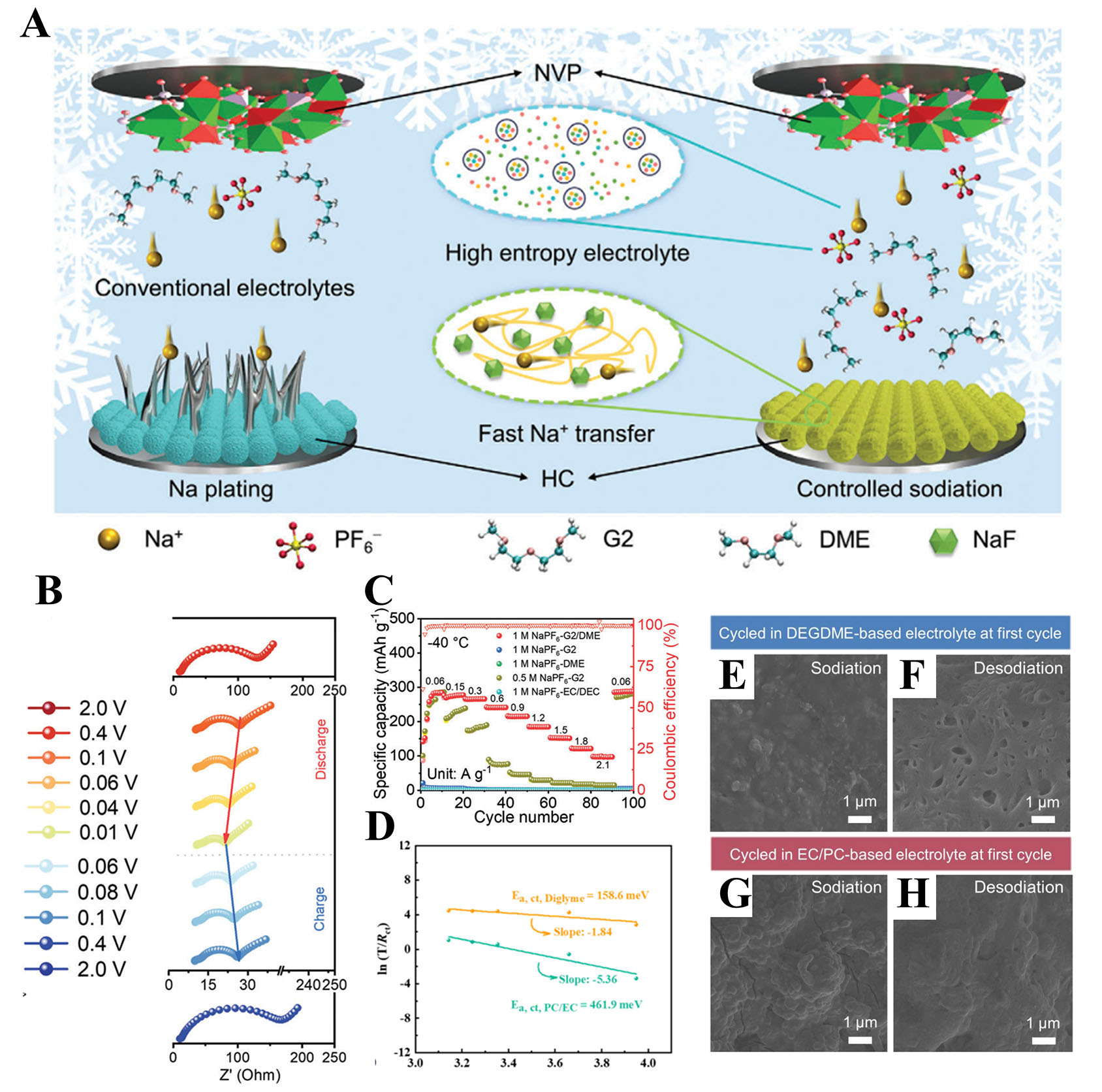





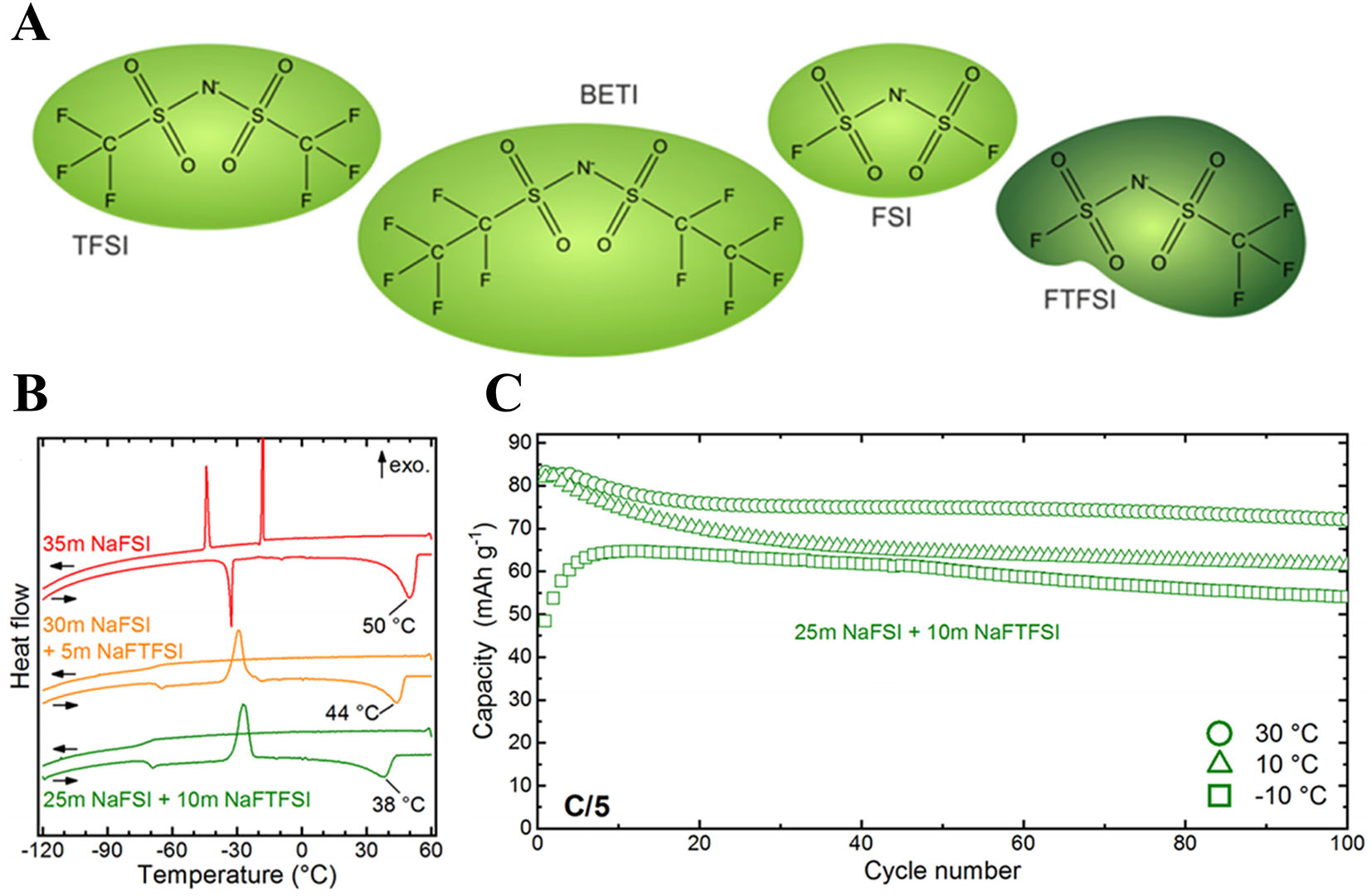

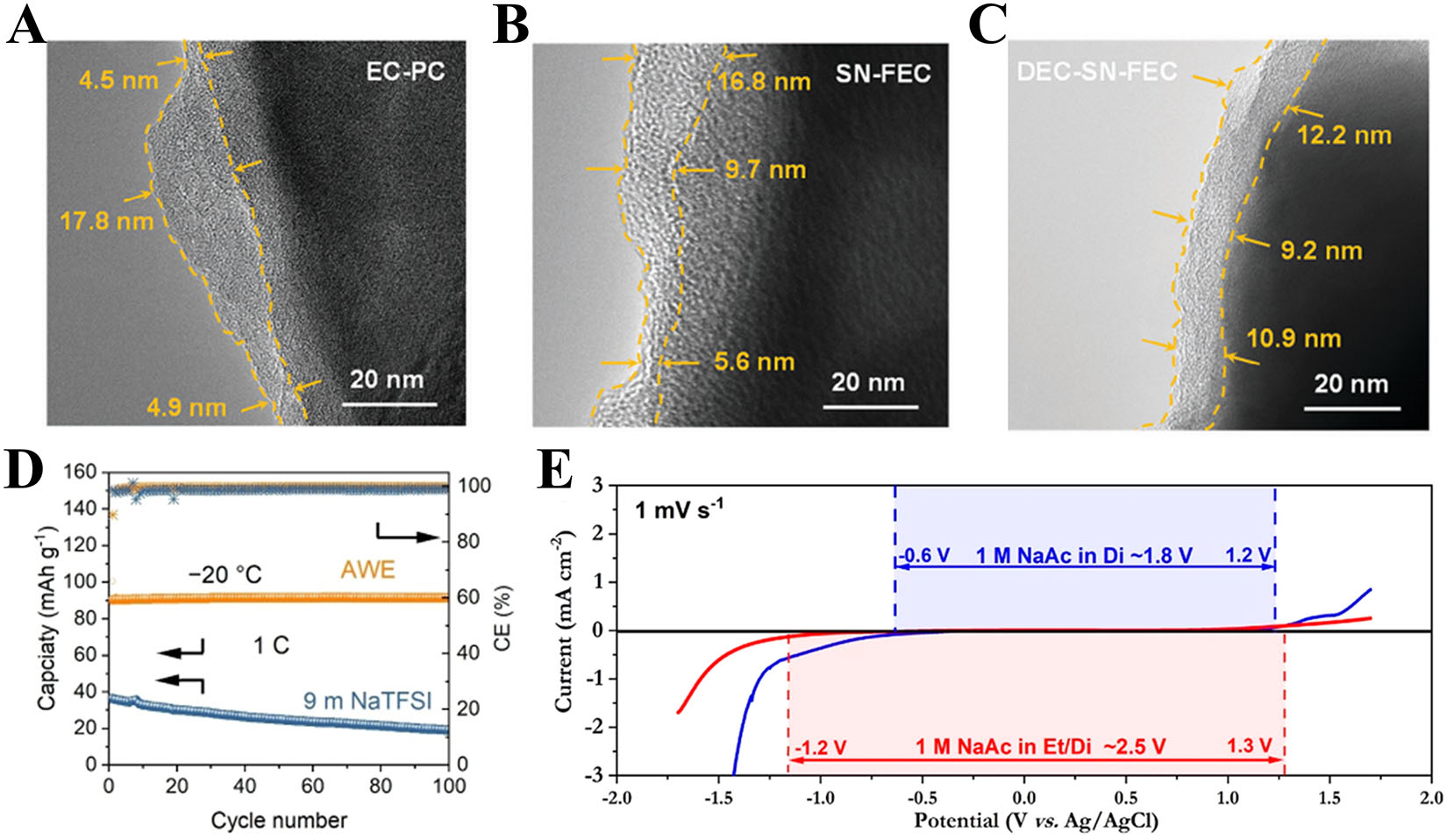












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].