Exploring the failure mechanisms of quasi/all solid-state Li-ion batteries with Si-based electrodes
Abstract
Lithium-ion batteries are widely applied in the field of energy storage due to their high energy density and long cycle life. However, traditional liquid electrolytes have safety hazards such as leakage and thermal runaway. The quasi-solid-state battery (QSSB) and all-solid-state battery (ASSB) have emerged as promising alternatives with higher safety and stability. In addition, Si-based electrodes are attractive due to their high theoretical capacity. Currently, researchers apply Si-based electrodes in QSSB and ASSB, but the failure mechanisms within them are not fully summarized and organized. Herein, this work systematically studies the failure mechanisms of QSSB and ASSB with Si-based electrodes, including particle fracture, solid electrolyte interphase breakdown, pore evolution, and electrical contact loss. The influence of rigid solid electrolytes on ASSB is discussed, as well as the limitations of quasi-solid electrolytes, such as low ionic conductivity and side reactions. The strategies for alleviating these problems are also reviewed, including the structural design of Si electrodes, electrolyte optimization, and interface engineering. This article aims to summarize the key failure mechanisms and provide guidance and technological development directions for the subsequent development of high-energy density and long-life batteries.
Keywords
INTRODUCTION
Lithium-ion batteries (LIBs), owing to their advantages of high energy density, high power density, long cycle life, and low self-discharge rate, have been widely used in portable electronic products, electric vehicles, and grid energy storage devices[1-4]. Electrolytes are a very important part of LIBs, and liquid electrolytes (LEs) are mainly used. LEs not only have high ionic conductivity, but also excellent electrode material wettability. It provides fast charge transfer capability and good interface contact for lithium-ion batteries, thereby maintaining high power output and long cycle stability of the battery. With the continuous improvement of battery performance requirements, liquid electrolyte systems are also constantly developing. Liu et al.[5] systematically optimized the composition of the electrolyte by developing high-concentration electrolytes and introducing key additives such as fluorinated vinyl carbonate. These designs effectively suppress side reactions at the interface between the electrode and electrolyte, and significantly enhance the cycling stability and thermal safety of the battery under high voltage and high nickel positive electrode systems. However, they can cause serious safety issues, such as electrolyte combustion, internal short circuits, and thermal runaway caused by excessive heat released from chemical reactions between battery components[6]. Therefore, there is an increasing demand for electrolyte technologies that combine high performance and safety[7].
Both solid-state electrolytes (SSEs) and quasi-solid-state electrolytes (QSEs) are promising alternatives to LEs. SSEs have advantages such as high mechanical strength, good electrochemical stability, and good thermal stability. QSEs have also become a hot topic because of their unique properties and broad application prospects. When it works with the LE, the framework of the quasi-solid electrolyte is fully infiltrated and solidified so that a composite system with a gel-like microstructure is constructed. This continuous gel matrix not only gives the material excellent mechanical integrity but also plays an optimization role in the efficient ion transport process. It can significantly promote the rapid migration of ions at the electrode electrolyte interface by reducing the interface impedance and providing a continuous conduction path, ultimately enabling batteries to achieve more efficient charge transfer and energy storage[8]. In terms of battery stability, methods such as multi-layer active oxygen protection, multifunctional separator, and electrode multi-stage protection have been proposed at the level of QSEs, greatly improving the safety[9]. However, the solid-state system still has problems such as poor interface contact and difficulty in maintaining the integrity of electrode structure during cycling[10]. People have explored many advanced technologies to address these issues, such as dry electrode machining technology[11]. It prepares electrodes through dry powder mixing and mechanical compaction, thereby achieving a denser electrode structure and interface contact, improving interface adhesion and ion transport pathways in solid and semi-solid systems[12].
Lithium metal electrodes are widely used in electrochemical energy storage materials due to their high theoretical capacity and lowest electrochemical potential[13]. However, in practical applications, the cycling stability of lithium metal batteries is poor. And due to the uncontrollable growth of lithium dendrites and the accumulation of dead lithium, it is easy to cause safety hazards such as internal short circuits, thermal runaway, and even fire and explosion[14]. In addition to lithium metal, other negative electrode materials such as TiO2 and Zn2GeO4 have also received research attention due to their structural stability and good ion transport performance. For example, the mechanism of “local electron enhanced ion transport” proposed by Chen et al.[15]. They introduced localized electrons into TiO2 through defect engineering, significantly reducing the ion migration barrier. Silicon (Si) has a higher theoretical capacity than lithium metal and has attracted widespread attention as an electrode material in LIBs[16]. In addition, Si resources are abundant, environmentally friendly, and inexpensive, making them suitable for scalable and sustainable battery production[17,18]. However, its significant volume change (approximately 300%) during lithium-ion insertion and extraction can seriously damage the electrode structure and force the continuous reconstruction of the solid-electrolyte interphase film, ultimately leading to rapid capacity decay[19,20]. When this issue shifts from liquid-state batteries to quasi-solid-state battery (QSSB) and all-solid-state battery (ASSB), and the interphase stability and mechanical failure problems become more complex and important. Exploring the aging mechanisms of Si-based electrodes under specific environments, including the evolution of interface side reactions during cycling, stress accumulation and release, and the resulting core issues, such as contact failure, capacity decay, and impedance growth, is the key to overcoming the bottleneck of their long cycle life.
This article introduces the characteristics of quasi/all solid-state LIBs, focusing on the key failure mechanisms using Si-based electrodes in practical applications. The various failure mechanisms of quasi/all solid‐state LIBs, including electrode structure damage, interface side reactions, electrolyte mechanical fatigue, and ion transport limitations, are summarized and analyzed. Various strategies to alleviate battery failure are discussed, such as negative electrode structure design, electrolyte modification, and improving interface stability. It is hoped that this study can provide theoretical references and technical directions for promoting the engineering application of high-performance LIBs.
FUNDAMENTALS OF QUASI/ALL SOLID-STATE BATTERIES
All solid-state batteries
All solid-state batteries, as the name suggests, are characterized mainly by their solid electrolytes, which are specifically divided into solid polymer electrolytes (SPE), inorganic solid electrolytes (ISE), and solid composite electrolytes (SCE)[21]. Figure 1 presents the comparison of performance among LE, SPE, ISE, and SCE. SPEs enable lithium ions to conduct through the polymer chain matrix. They solve the leakage and flammability issues associated with LEs, and meanwhile maintaining stable interface contact, processability, and cost-effectiveness[22,23]. However, their ionic conductivity remains low under room temperature conditions, which restricts the scope of the application[24]. Compared with liquid batteries, although solid-state batteries (SSBs) have improved safety performance, there are still some safety issues that have not been fully resolved[25]. To address the problems of high temperature as well as flammability, Cui et al.[26] developed a fire-resistant and lightweight polymer composite solid electrolyte. This electrolyte consists of a porous polyimide skeleton, the flame retardant decabromodiphenyl ethane, and the ion-conducting system of polyethylene oxide/lithium bis(trifluoromethanesulfonyl)imide (PEO/LiTFSI). It demonstrates an ultrathin structure while exhibiting high mechanical strength and flame-retardant properties. LIBs fabricated with this electrolyte show enhanced rate performance and maintain extended cycle life at 60 °C. Notably, pouch-type batteries utilizing this electrolyte continue to operate normally during flame burning tests, demonstrating significantly improved safety performance. SPE based on polymers such as PEO has advantages such as flexibility, ease of processing, and good interface contact with electrodes. However, when combined with high-capacity electrodes such as Si, their relatively low mechanical strength and limited long-term interface stability under repeated volume changes pose significant challenges. To address these challenges, the development of composite polymer electrolyte (CPE) has become a promising direction. Liu et al.[27] designed a CPE that utilizes hydrogen bonding interactions to enhance the mechanical robustness, thermal stability, and interfacial compatibility of the electrolyte. This provides a feasible strategy for improving the overall performance of SPE in Si-based solid-state batteries.
Figure 1. Performance comparison of LEs, SPE, ISE, and SCE. The blue-green materials represent SPE, while purple particles represent inorganic fillers. Reproduced with permission from Li et al., Advanced Science (2020). Copyright © Wiley-VCH[33]. LEs: Liquid electrolytes; SPE: solid polymer electrolytes; ISE: inorganic solid electrolytes; SCE: solid composite electrolytes.
Compared with SPEs, the use of an ISE is considered an effective method to improve the safety, as the flammable properties of an ISE can reduce the risk of fire and explosion[28], and it also results in high ion conductivity and environmental tolerance[29]. The ISEs are mainly divided into three types: sulfides, oxides, and halides. Sulfide electrolytes include silver mercury sulfide, lithium phosphate sulfide, and Lithium germanium phosphorus sulfur (LGPS) sulfide. The silver mercury sulfide electrolyte has good room-temperature ionic conductivity, but there are issues with air sensitivity and interface instability, which need to be improved through coating and structural modification. Lithium phosphate sulfide can further enhance its electrochemical performance by doping Si. Although LGPS type sulfides have good conductivity, their application in batteries is limited due to the high cost of Ge and the induced side reactions. Oxide electrolytes include garnet type, Na super ionic conductor (NASICON) type, perovskite type, and Li super ionic conductor (LISICON) type. Garnet type (such as Li6.4La3Zr1.4Ta0.6O12, LLZO) can stabilize the cubic phase and achieve good room temperature ion conductivity by doping with elements such as Ta, Al, Ga, etc.[30]. However, they still face challenges such as high sintering temperatures and interfacial side reactions. NASICON types [such as Li1.3Al0.3Ti1.7(PO4)3, LATP and Li1.5Al0.5Ge1.5(PO4)3, LAGP] have high ion conductivity and good air stability, but LAGP is limited by the high cost of Ge, while LATP faces the problem of Ti4+ reduction at low potentials. Perovskite-type Li0.3La0.557TiO3 (LLTO) has good conductivity but many side reactions, and is often used in composite electrolytes or hybrid battery systems. LISICON type (such as lithium phosphorus oxynitride) is mainly used in the field of thin film microbatteries due to its good film-forming properties and interface stability[31]. Halide electrolytes, such as Li3YCl6 and Li3InCl6, have gained significant attention for their high room-temperature ionic conductivity, wide electrochemical stability window, and excellent compatibility with high-voltage oxide cathode materials. However, their high hygroscopicity and interfacial instability remain critical challenges, especially when coupled with Si-based electrodes where significant volume changes exacerbate contact loss and promote side reactions. Fluoride-based halides (for example, Li3AlF6) exhibit superior air stability and the widest electrochemical window. However, the low ionic conductivity of fluoride-based halides at room temperature is on the order of 10-5 S cm-1. This is much lower than chloride/bromide (about 10-3 S cm-1), which limits its applicability in high-speed Si-based systems[32].
SCE comprises a flexible polymer body, dissolved lithium salts, as well as rigid inorganic fillers. SCEs take the advantages of high ionic conductivity from SPEs and inorganic fillers. They also possess good flexibility, which allows for closer contact with electrodes[33]. However, the phase boundary between polymer and ceramic fillers may raise the migration barrier during lithium-ion transport. This can lead to high interface impedance, along with poor electrochemical performance in the battery. Structural construction, together with surface modification of SCEs, is required to tackle these issues[34]. Wang et al.[35] have developed a composite polymer solid electrolyte that features a three-dimensional interpenetrating structure. This electrolyte is based on polyethylene oxide and is embedded with a self-supporting three-dimensional LLTO ceramic nano-skeleton network, which serves as a reinforcing phase. This structure notably enhances the mechanical strength of the electrolyte, along with its thermal stability. It enables Li symmetric batteries to cycle in a stable manner for more than 800 hours under a current density of 0.1 mA/cm2.
Quasi-solid-state batteries
To address the limitations of both liquid and solid-state systems, researchers have developed a solid-liquid hybrid electrolyte, commonly referred to as a QSE, as an intermediate strategy to bridge the gap between conventional liquid and all-solid-state LIBs[36,37]. Among them, gel polymer electrolyte (GPE) is the most widely used QSE. When the electrolyte is combined with the LE, it contains an injected polymer framework, resulting in a gel-like structure. This electrolyte helps the battery achieve more efficient ion transfer, which facilitates effective charge transfer[8]. Compared with commonly used separators in liquid batteries, it can significantly reduce the possibility of electrolyte leakage. GPEs can be divided into heterogeneous and homogeneous gels[38]. The heterogeneous GPE is the most common and has a polymer framework filled with an interconnected electrolyte pore system[39]. The main process for manufacturing heterogeneous GPEs at present is in situ polymerization, which involves injecting precursor fluids that contain monomers, lithium salts, and initiators into the battery. It then solidifies these into a film under specific conditions (such as heat, light, or electrochemistry). This notably improves the interface compatibility between electrolyte and electrode. For example, Liu et al.[40] developed a self-healing QSE through in situ polymerization of a dynamic cross-linked polymer network, which effectively repairs electrode interface microcracks and maintains stable contact, enabling high-load Si-based batteries [Figure 2]. However, the QSE is a multicomponent, the individual and synergistic effects of these components on the safety performance of QSSBs remain insufficiently understood, which hinders the rational design of highly safe QSEs. Furthermore, when QSSBs are exposed to elevated temperatures, solvent evaporation occurs while the polymer skeleton remains on the electrode surface, adversely affecting the interfacial stability and electrochemical performance of the battery[41].
Figure 2. Schematic diagram of self-healing polymer aggregation process. Reproduced with permission from Liu et al., Energy Storage Materials (2025). Copyright © Elsevier[40]. PPO: Propylene glycol; DTDA: dithiodianiline; HEA: hydroxyethyl acry late; ETPTA: ethoxylated trimethylolpropane triacrylate.
FAILURE MECHANISM OF SI-BASED QUASI/ALL SOLID-STATE BATTERIES
The failure mechanism of Si-based quasi/all solid-state batteries mainly comes from three aspects: failure caused by Si electrodes, interface failure between electrodes and quasi/all solid-state electrolytes, and failure of electrolytes themselves. These three aspects interact with each other to determine the cycle life and performance degradation of the battery. This chapter will conduct a systematic analysis of these three aspects to reveal the failure paths faced by Si-based systems in quasi/all solid-state systems.
Si-based electrodes
Macroscopic manifestations of Si electrode failure
The core issue of Si lies in the significant volume changes during the lithiation and delithiation cycles[42]. Especially after complete lithiation, the volume expansion can reach a very high value, resulting in significant mechanical stress and leading to the rupture of Si particles[43]. This crushing disrupts the electrical contact between the active material and the conductive material, significantly reducing the effective surface area available for lithium-ion intercalation, resulting in capacity loss and reduced cycle stability[44]. During the first lithiation process, a solid electrolyte interphase (SEI) rapidly forms on the Si surface, which irreversibly consumes a large amount of lithium-ions, resulting in a first coulombic efficiency typically only 60%-90%[45]. In the actual configuration of a full battery, the low Coulombic efficiency of the first cycle means that excess lithium must be replenished from the positive electrode to compensate for irreversible losses on the negative electrode side, which increases the complexity of full battery design and management. Figure 3A shows the in situ transmission electron microscopy (TEM) image of the battery with the Si/Li21Si5 electrode during lithiation and delithiation processes, from which lithium-ion is alloyed with Si through Li21Si5, accompanied by significant volume expansion of Si and completion of lithiation. Interface fusion has already taken place in the contact area between Li21Si5 and Si. During delithiation, lithium-ions are extracted from the lithiated phase (Li15Si4), resulting in substantial particle shrinkage upon complete lithium-ion removal. After the battery undergoes continuous discharge for 420 s, Li21Si5 still maintains good structural stability. This result indicates that Li21Si5 can act as an effective conductive channel for ions and electrons, as well as a lithium source for Si particles. The cross-sectional scanning electron microscopy (SEM) image of the pure Si electrode following lithiation is presented in Figure 3B1, from which cracks are formed in the electrode itself, reducing the cycling stability. On the contrary, no obvious cracks were found on the Si/Li21Si5 electrodes after lithiation [Figure 3B2], and the electrode structure was relatively complete[46].
Figure 3. Macroscopic behaviors of Si-based electrodes. (A1-A6) In situ TEM analysis of the Si/Li21Si5 electrode during lithiation and delithiation processes; (B1 and B2) SEM images of the cross-sections of Si and Si/Li21Si5 electrodes before and after lithiation. Reproduced with permission from Zhang et al., Energy & Environmental Science (2024). Copyright © Royal Society of Chemistry[46]; (C) Porosity analysis of mercury intrusion into the Si electrode; Original, after 10 cycles, and after 100 cycles; (D) Porosity values calculated based on focused ion beam (FIB) cutting observations and mercury intrusion analysis, mass and thickness of 3 Si electrodes (original electrode, after 10 cycles, and after 100 cycles). Radvanyi et al., Physical Chemistry Chemical Physics (2014). Copyright © Royal Society of Chemistry[49]; Simulation of the pore structure at the interface between the PE diaphragm and Si electrode under pressures of 0 MPa (E) and 6 MPa (F). Seo et al., Nature Communications (2024). Copyright © Springer Nature[50]. TEM: Transmission electron microscopy; SEM: scanning electron microscopy; PE: polyethylene.
Furthermore, significant changes in volume during the cycling process lead to continuous rupture of the SEI, resulting in the continuous exposure of the fresh Si surface to the electrolyte[47]. This process promotes further decomposition of the electrolyte and continuous growth of the SEI, irreversibly consuming the electrolyte and lithium-ions, thereby increasing the impedance of the entire battery, ultimately leading to a decrease in the Coulombic efficiency of the battery and accelerated capacity decay[48]. Moreover, repeated expansion and contraction of Si particles can cause microcracks, forming microscale pores and subhundred nanometer pores, thereby increasing the overall porosity of the electrode. Radvanyi et al.[49] measured the evolution of porosity through ex situ mercury porosity analysis and electrode cross-section observation. From the mercury pore analysis results in Figure 3C and the quantitative data in Figure 3D, the volume expansion of Si particles in the early stage of the cycle (10 cycles) induces microcracks, causing the pore structure to change dramatically from the initial single peak distribution to a double peak distribution. The porosity increases from 59% to 74%, and the electrode thickness increases synchronously by 36%. With cycle progression, fracture of the Si surface promotes continuous and irreversible SEI reformation, and the resulting decomposition products gradually fill the micrometer-sized pores. This pore blockage will significantly deteriorate the ion transport pathway after multiple cycles, and this process may lead to uneven lithiation gradients. Due to more facile electrolyte infiltration, particles near the electrode surface experience excessive lithiation, while those in the electrode interior suffer from insufficient lithiation because of hindered ion transport.
The mechanical closing effect of the diaphragm also acts as an important factor that causes the failure of Si electrodes. The significant volume expansion of Si materials in cycling processes can produce high-pressure stress in local regions of the battery. When this stress acts on the battery separator, it can result in the collapse of pores. To investigate this phenomenon, Seo et al.[50] employed advanced 3D digital twin models for simulation studies. The key parameters on which these simulations are based are derived from in situ measurements of the internal force evolution of Si full cells during cycling. Under the condition of applying an external stack pressure of 100 kPa, the measurement recorded in real-time the dynamic internal forces generated by the Si negative electrode during lithium insertion/extraction through the load unit. The results shown in Figure 3E and F prove that in Si-based full battery systems, mechanical fracture occurs when the polyethylene (PE) separator is subjected to a pressure of approximately 6 MPa, and this fracture is particularly evident in the contact interface area between the separator and Si particles (as shown by the red dashed line in the figure). In contrast, the part of the diaphragm that contacts the electrode pores (as marked by the blue dashed line) stays basically unaffected. The simulation results show that the internal stress produced by the volume expansion of Si becomes highly concentrated at the interface between the diaphragm and the electrode particles. This stress concentration effect directly causes local collapse of the membrane pores in the area close to the interface. The serious consequence of this situation is that it greatly hinders the normal transport of lithium ions within the membrane, and further exacerbates the non-uniformity of oxidation-reduction reactions inside the electrode, ultimately leading to the breakage of Si materials and the failure of the battery.
Microscopic origin of Si electrode failure
The macroscopic failure behaviors of Si electrodes during cycling, including particle fracture, persistent SEI growth, pore structure evolution, and limited ion/electron transport, primarily originate from the substantial volume changes that occur during charge and discharge processes. Apart from these external manifestations, the inherent physical and chemical properties of Si are the origin of these failure behaviors.
Si has two phases of crystalline (c-Si) and amorphous (a-Si)[51]. During high-temperature treatment, a-Si undergoes recrystallization towards c-Si, which can be verified by the enhancement of peak intensity and narrowing of peak width in X-ray diffraction (XRD)[52]. Recrystallization not only changes the initial structure of the material, but more importantly, transforms the expansion mode during lithiation from isotropic [Figure 4A] to anisotropic [Figure 4B], thereby causing local stress concentration and significantly increasing the risk of fracture[53,54]. There are also significant anisotropic differences in the lithiation mechanism. Although both c-Si and a-Si eventually form Li15Si4 crystalline phases, the phase boundary movement of c-Si during lithiation is significantly affected by crystal orientation, resulting in highly anisotropic uneven expansion, which is more likely to cause particle rupture. In contrast, due to the lack of long-range ordered grain boundary constraints, the lithium insertion process of a-Si is more uniform and exhibits isotropic expansion, resulting in relatively better mechanical stability[55,56].
Figure 4. Microscopic origin of Si-based electrode failure. (A) Anisotropic expansion of c-Si. Reproduced with permission from Lee et al., Nano Letters (2011). Copyright © American Chemical Society[53]; (B) Isotropic expansion of a-Si. McDowell et al., Nano Letters (2013). Copyright © American Chemical Society[54]; (C) After one cycle, pure μ Si particles are transformed into μ Si thin films. Tan et al., Science (2021). Copyright © American Association for the Advancement of Science[60].
Electrochemical sintering is a special behavior of material structure evolution, which can be understood as the activation of nanoparticles to agglomerate into larger sizes, accompanied by the generation of stress. This behavior will damage the integrity and activity of the electrode, and is a key cause of performance deterioration[57]. During the cycling process of Si-based batteries, Si particles produce mechanical pressure because of repeated volume changes, which facilitates the breaking and reformation of Si-Si bonds, leading to the merging of adjacent particles via new chemical bonds[58,59]. This process increases the size of the active material particles and diminishes nanoscale effects, which in turn aggravates local strain and crack propagation. Figure 4C shows that micro Si (μ Si) particles develop a continuous thin film through electrochemical sintering after one cycle, and larger-scale cracks appear; this situation fully proves the destructive effect of this mechanism on the electrode structure[60].
Si has low intrinsic electron conductivity, which also limits electron transport in composite electrodes[61]. To improve the electronic conductivity of the electrode, conductive additives must be added so that they can connect the electrode with the active material so as to keep the electron permeation pathway. However, conductive carbon additives are likely to cause the decomposition of solid electrolytes like sulfides under early cycling voltages[60]. In liquid-based LIBs, LEs can give great help to ion transport between particles. In SSB systems, most SSEs are mixed with electrode materials so that they can build an effective ion transport channel network around the active material particles[62]. However, when Si particles mix with the SSE, the irregular shape and random distribution of these particles greatly prevent the formation of tight and continuous interface contacts among the active particles and the ion transport medium[63]. The looseness and porosity of this internal structure not only compresses the effective phase volume fraction available for ion transport but also leads to a more bizarre and complex geometric shape of the SSE phase. This significantly lengthens the ion migration pathway, ultimately degrading battery performance[64].
Other reasons for the failure of Si electrodes
The manufacturing process of battery materials is also a critical factor in determining electrochemical performance. The Si composite materials and various SSEs in SSB electrodes are typically composed of multiple components, and the production of these components requires complex slurry engineering. The electrode slurry is prepared by mixing a liquid solvent, polymer binder, active material (AM) powder, and conductive agent (CA). Parameters including the preparation sequence, AM/CA particle size, and dispersant type influence both the electrode structure and the resulting battery performance. For example, the uniformity of slurry preparation directly determines the stability of the ion/electron conduction network in the electrode and profoundly affects its mechanical integrity. If the slurry is unevenly dispersed, the conductive agent cannot form a uniform three-dimensional network, resulting in local differences in conductivity. During the cycling process, significant volume changes in Si particles can rapidly deteriorate battery performance, leading to local current overload, electrical contact failure, and separation of active substances. Meanwhile, uneven distribution of binders can hinder ion transport or fail to effectively buffer stress, accelerating electrode structure damage. In addition, improper drying processes can lead to the migration of solid components, resulting in uneven distribution of electrode pores and components, further exacerbating polarization and stress concentration during charge and discharge processes[65].
All solid-state electrolytes
The design of composite Si electrodes in SSBs usually incorporates a relatively high proportion of solid electrolytes and conductive additives so as to guarantee higher ion and electron conductivity rates. However, compared to liquid electrolyte batteries, the high content of SSE and conductive additives in this design renders it hard to release effectively the stress that forms inside the electrode. The hysteresis of stress diffusion may lead to structural collapse, and in turn disturbs the ion and electron transport channels, and this explains why the active material content in composite Si electrodes is usually set to relatively low levels[66].
The rigid physical contact between the electrode and SSE interface typically results in a discrete point-contact mode. This contact method significantly reduces the effective interaction area and also limits the ion transport path. Especially for SSE types that have insufficient mechanical strength, such as solid polymer electrolytes, along with solid sulfide electrolytes, these SSE types are hard to effectively constrain the significant volume changes that take place in Si electrodes during cycling[67]. Nelson et al.[68], found that the chemical mechanical coupling between Si and SSE leads to different fracture modes through operating X-ray tomography. Namely, the biaxial tensile stress induced by delithiation promotes the formation of mud cracks through electrodes [Figure 5A and B], while local reaction competition between adjacent Si domains of different sizes drives the initiation of interface cracks at the Si/electrolyte interface [Figure 5C]. Once microcracks are formed, lithium ions will propagate along grain boundaries or through the electrolyte matrix [Figure 5D][69]. The diminished interface integrity, which rigid SSEs cannot rapidly dissipate unlike LEs, results in highly localized strain concentration. This induces complex non-local strain effects that cause chemical-mechanical failures, such as interface delamination and crack formation. These failures manifest macroscopically as a sharp drop in contact area, ultimately leading to sudden failure[70].
Figure 5. Failure behavior at the Si/solid electrolyte interface. (A) Planar view of the delithiated Si electrode; (B) 3D crack network; (C) Lithium-ions propagated along grain boundaries, forming vertical channel cracks and interface fractures caused by lithium removal. Reproduced with permission from Nelson et al., ACS Energy Letters (2024). Copyright © American Chemical Society[68]; (D) Lithium-ion propagation along grain boundaries. Kalnaus et al., Science (2023). Copyright © American Association for the Advancement of Science[69]. LPSC: Lithium phosphorus sulfur composite.
The failure mechanism of solid electrolytes comes from not just the interface between electrolyte and electrode, but also the electrolyte itself. Taking ISE as an example, its inherent electrochemical stability window is limited by its elemental composition, crystal structure, and microstructure. Its relatively narrow electrochemical stability window (ESW), together with its strong chemical, as well as electrochemical reactivity with electrode materials, can bring about significant structural degradation during electrode lithiation[71]. If the operating voltage of the battery goes beyond the intrinsic ESW of the ISE, its structure itself will undergo irreversible decomposition or breakdown. Besides, even when the operating voltage remains within the stability window, continuous side reactions may take place at the interface[72]. The occurrence of side reactions during battery operation can make inert components stick to the interface between ISE and electrodes, increasing the interface impedance and bringing about a rapid drop in battery capacity[73]. Additionally, some ISEs are sensitive to air. Exposure to the ambient environment due to battery damage can accelerate their structural degradation[74]. Besides these electrochemical and chemical instability issues, the ISE is also susceptible to mechanical crushing. This failure mode is primarily induced by the internal volume changes of the active material during (de)lithiation, a process that exerts repeated mechanical stress on the rigid electrolyte. Furthermore, the degradation of the electrolyte can exacerbate the volume changes within the electrode, potentially establishing persistent side reactions during charge and discharge[75,76].
Quasi solid-state electrolytes
QSEs have turned into a viable alternative because of their hybrid properties, which offers improved interfacial compliance and strain adaptability. One key purpose of developing QSEs in Si-based batteries is to reduce the impact of electrode expansion on the battery, but the intrinsic factors of QSEs may also form the failure mechanism.
Insufficient ionic conductivity stands as a key limitation of QSEs, particularly when combined with Si-based electrodes that go through repeated volume changes. The continuous expansion and contraction of the electrode can disrupt the ion transport pathways within the gel network, increasing interfacial resistance and leading to uneven current distribution. Even though the impedance of gel electrolyte (GE) can be lowered by optimizing the interface, their intrinsic ion mobility remains a bottleneck. The ionic conductivity of the GE (up to 6.42 × 10-3 S cm-1) is significantly lower than that of the LE (approximately 17.90 × 10-3 S cm-1), which is determined by the higher activation energy of the GE (Ea = 0.31 eV), which indicates that lithium-ion has greater migration resistance in the gel network[77]. This issue is especially critical in batteries with Si, since effective ion transport plays a crucial role in adapting to spatial lithiation heterogeneity along with reducing local stress accumulation.
Insufficient resistance to interface side reactions and poor chemical stability are also significant drawbacks of QSEs, particularly when they are used together with Si-based electrodes. Under high-temperature operating conditions, QSE experiences interface side reactions during cycling processes, like the decomposition of electrolytes and the repeated reconstruction of SEI. The mechanical stress produced by the expansion of Si further worsens these side reactions, and in the end leads to the accelerated degradation of battery capacity. While QSEs show significantly enhanced high-temperature stability over baseline LEs, their performance in Si-based systems is still hampered by critical issues. When paired with a Si-graphite (Si-Gr) electrode, a QSSB still exhibits a 19.3% capacity loss after 60 cycles, highlighting persistent interface side reactions and SEI instability aggravated by si's volume changes[78]. In addition, the quasi-solid polymer electrolyte begins to thermally decompose at 80 °C. Although this temperature is higher than that of the LE, it remains insufficient for high-temperature applications (> 120 °C)[79]. Besides, in most QSSBs, the presence of residual LE components during the solidification process will cause continuous side reactions with the Si electrode during operation, leading to gradual deterioration of the interface and ultimately resulting in battery failure. Kido et al.[80] separated negative and positive electrodes with solid electrolyte sheets and mixed two different types of near-saturated electrolyte solutions to prepare a QSE Si|NCM811 battery. When the electrolyte solution comes into contact with the Li6La3ZrTaO12 (LLZT) solid electrolyte, it decomposes to form products containing fluorine precipitate. The acidic decomposition substances containing fluorine are prone to attack the positive electrode structure, causing the transition metal to dissolve and thus have a significant impact on the performance of the battery.
The mechanical performance defects of QSEs also serve as important causes for the failure. The buffering capacity of QSEs also has a certain upper limit. Taking the GE as an example, the polymer framework of the GE can alleviate the significant volume expansion of Si-based materials during cycling to a certain extent. However, its mechanical properties are still not enough to fully prevent the formation of microcracks. After the cycling process, a small number of tiny cracks can still be observed on the electrode surface. This observation shows that GE cannot fully avoid the risk of structural failure in Si particles. The SEM comparison results from Figure 6A and B further show that the electrode with a LE exhibits extensive cracking. In contrast, the electrode with GE only produces localized and limited microcracks, indicating that the cracking problem has not been fully resolved. Therefore, the main role of GE is to reduce the severity of electrode cracking rather than completely preventing it from occurring[77]. In addition, traditional QSE does not have the ability for dynamic repair, and the microcracks formed during cycling cannot be reversed. These microcracks cannot adapt to changes in electrode volume, which accelerates interface failure[40]. When nonelastic polymers (such as polyacrylic acid and polyvinyl alcohol) are used as the electrolyte and also as the buffer layer, the cycling stability of Si-based electrodes is poor because these polymers cannot buffer stress and instead accelerate the structural damage of the battery [Figure 6C][81].
Figure 6. Polydopamine (PD)@Si morphology evolution and interface chemistry of the electrode after cycling in LE and GE. (A and B) SEM images of the PD@Si electrode surface after cycling in LE and GE. Reproduced with permission from Suebsing et al., Sustainable Energy & Fuels (2025). Copyright © Royal Society of Chemistry[77]; (C) Long-term cycling performance and discharge capacity of the SiO electrode with various polymers. Huang et al., Nature Communications (2019). Copyright © Springer Nature[81]; microscopy images of the punched surface of the original PE (D) and PEAE-0 (E) separators. Xue et al., Colloids and Surfaces A: Physicochemical and Engineering Aspects (2022). Copyright © Elsevier[82]. GPE: Gel polymer electrolyte; LE: liquid electrolyte; GE: gel electrolyte; SEM: scanning electron microscopy; PE: polyethylene; PEAE-0: pure ceramic-coated separators; PAA: polyvinyl alcohol; PVA: polyacrylic acid; PVdF-HFP: polyvinylidene fluoride-hexafluoropropylene.
The breakage of the microporous structure in quasi solid-state separators can also lead to battery failure mechanisms. Xue et al.[82] have reported that puncture events during cycling can make the microporous branches of uncoated PE separators [Figure 6D] and pure ceramic-coated separators (PEAE-0, Figure 6E) in QSSBs break and lead to an increase in pore size. The active substance then migrates to the counter electrode, a process that causes a local soft short circuit and further results in a significant increase in the steady-state current. The PEAE-0 membrane, which contains only Al2O3, undergoes coating peeling during puncture. Meanwhile, 30 nm Al2O3 particles (smaller than the PE pore size ranging from 100 nm to 200 nm) block the micropores [Figure 6E], and this blocking leads to a sudden drop in ion conductivity and also a decrease in the high-rate discharge capability.
Besides the aforementioned failure mechanisms, infiltration also plays a crucial role. In situ polymerization demands that the liquid precursor fully infiltrate the pores of the electrode. If infiltration is insufficient, incomplete contact at the interface takes place after polymerization, which hinders local ion transport and it may lead to side reactions at the interface. This problem can be alleviated through the extension of infiltration time yet this issue cannot be fully eliminated[83].
In summary, both ASSB and QSSB using Si-based electrodes face the common challenge of Si volume expansion. But due to the different electrolyte characteristics of the two, their main failure mechanisms are also different. The main problem with ASSB lies in the rigid solid-solid interface, which is mechanically mismatched. This can easily cause sudden contact failure, interface delamination, and ultimately lead to complete battery damage. In contrast, the failure process of QSSB is smoother. The problem mainly comes from the chemical and electrochemical performance limitations of gel electrolyte, such as insufficient ion conductivity and continuous occurrence of side reactions. In addition, the mechanical fatigue of gel polymer framework slowly appears under the repeated stress of charge discharge cycle, which eventually leads to battery failure.
STRATEGIES FOR MITIGATING FAILURE
Structural design of Si-based electrodes
In SSE/QSE battery systems, the significant volume expansion of Si-based electrodes during cycling still stands as one of the primary causes behind electrode structure damage, interface contact failure, and accelerated capacity decay. Unlike LEs, SSEs and QSEs cannot adaptively fill the gaps generated by Si volume changes through flow due to their solid-solid interfacial contact nature. This leads to a greater tendency for contact loss and local stress concentration, which in turn exacerbates interface delamination and the disruption of ion transport channels. To address this challenge, current research pursues a dual strategy: optimizing the electrode structure to alleviate internal stress and maintain integrity, while synchronously designing SSE/QSE systems with robust mechanical adaptability, high ionic conductivity, and excellent interface compatibility, thereby synergistically suppressing aging and failure[84]. These strategies are considered as the core solutions to address the issues of particle fracture, electrical contact loss, and SEI film breakdown caused by significant volume expansion of Si electrodes. Cangaz et al.[85] put forward an innovative columnar Si electrode (col Si) structure design, which employs physical vapor deposition technology for the preparation of Si thin films with vertically arranged columnar structures and adopts dendritic substrates to guarantee excellent adhesion of the Si film onto the current collector. Under this structure, the SSE maintains 2D contact with the Si electrode, a contact mode that can preserve the mechanical integrity of the SEI layer and accommodate the 1D growth of Si pillars [Figure 7A-C]. This change notably cuts down the reaction area between Si and the electrolyte, and it can also inhibit the continuous growth of the SEI along with electrolyte consumption throughout cycling. Another unique advantage of this structure lies in its one-dimensional breathing mechanism. During the charge-discharge process, the Si pillar mainly expands or contracts in the direction perpendicular to the current collector while deformation in the horizontal direction gets suppressed [Figure 7D]. This situation then effectively eases the aging and failure of the battery and helps attain better battery performance.
Figure 7. Design of the columnar Si electrode structure. (A) Reversible volume change of the columnar Si electrode system during (de) lithiation in the ASSB battery configuration; SEM images of col Si (B) top view and (C) cross-section; (D) Morphology and phase transition diagram of col Si during the first lithiation process. Reproduced with permission from Cangaz et al., Advanced Energy Materials (2020). Copyright © Wiley-VCH[85]. SEM: Scanning electron microscopy; ASSB: all-solid-state battery; SE: soild electrolyte.
The challenges brought about by Si volume expansion get significantly amplified in ASSB and QSSB configurations. The inherent rigidity of many SSEs together with the point-contact nature of solid-solid interfaces makes them particularly vulnerable to contact loss, delamination as well as high impedance caused by electrode dimensional changes. For this reason, the structural design of Si electrodes applied in SSE/QSE systems not only needs to limit internal stress but also needs to maintain tight interface contact as well as effective ion transport pathways in the confined solid electrolyte environment.
Dunlap et al.[86] found that the carbon matrix obtained by thermal decomposition of coal tar pitch (CTP) can be used as an integrated mixed conductive adhesive without additional conductive additives. This unique characteristic enables the direct transmission of ions and electrons through carbon-coated Si particles, while its conformal coating maintains structural integrity by partially adapting to volume changes. In the course of the pyrolysis process, CTP is converted into an amorphous soft carbon matrix that integrates ion/electron mixed conductivity and electrochemical activity. The SEM images presented in Figure 8A and B show that the dense carbon coating layer imposes physical constraints on the volume expansion of Si particles, thereby preventing their pulverization and electrochemical failure. The cyclic performance of the electrode in the ASSB and the voltage curves of some cycles are shown in Figures 8C and D.The optimal 6:4 (Si/C:SSE) ratio enablesthe SSE to function as an ion-conductive binder, which mitigates contact loss by securing interfacial integrity loss by securing interfacial integrity and ion transport [Figure 8E-G]. Besides this, Gao et al.[87] developed hollow Si nanospheres via an aluminum-thermal reduction process in molten salt. This structure effectively accommodates volumetric changes during cycling and enhances the integrity of the electrode, which is particularly crucial for maintaining stable interfacial contact in solid-state battery systems. As a result, the negative electrode delivered a high specific capacity of 1,774 mAh g-1 after 200 cycles at 1 A g-1, and retained 80% of its initial capacity even after 1100 cycles at a high current density of 8 A g-1, demonstrating the efficacy of hollow engineering in achieving long-term cycling stability.
Figure 8. Performance of Si-carbon composite particles obtained from CTP in SSBs; (A) Low-magnification SEM image of heat-treated Si/C composite particles containing 325 mesh-like Si particles; (B) SEM image of FIB cross-sectional particles showing EDS point scanning positions; The selected cycle voltage distribution (C) and dQ/dV (D) of the entire SSB; FIB cross-sectional view of the Si/C: SSE composite electrode after 100 charge-discharge cycles; (E) 6:4 nSi/C: SSE composite electrode; (F) 7:3 nSi/C: SSE composite electrode; (G) 7:3 Si/c: SSE composite electrode, containing 1~3 μm Si particles. Reproduced with permission from Dunlap et al., Solid State Ionics (2018). Copyright © Elsevier[86]. CTP: Coal tar pitch; SEM: scanning electron microscopy; SSE: solid-state electrolyte; EDS: energy dispersive spectrometer; SSBs: solid-state battery; FIB: focused ion beam.
In terms of Si-based electrode design under QSSB systems, Pandey et al.[88] put forward an innovative design of a three-dimensional core-shell structure. They sputtered a Si layer onto carbon nanofiber arrays, which were directly attached to a copper foil current collector. The open porous structure between the nanofibers allowed the GPE to thoroughly penetrate and encapsulate each core-shell fiber. After solidification, the resulting flexible interface effectively accommodated cyclic strain induced by Si volume changes and ensured complete penetration of the GPE to achieve stable interfacial contact. SEM results confirmed that even after hundreds of cycles, the vertical Si-coated arrays remained intact with no signs of electrode fracture or macroscopic cracks. The integrity of this structure ensures that it can achieve a reversible capacity of up to 3,450 mAh g-1, and stable circulation for 100 weeks at a high magnification of 2.6 A g-1, which indicates that the volume change of the Si-based electrode is effectively limited in the micro nano space composed of carbon fibers and gel.
The core goal of electrode structure design is to adapt to or constrain the volume expansion of Si, thereby maintaining the integrity of electrical contacts and ion pathways. The electrode structure design strategy often brings new problems while improving cycling stability. For example, the preparation process of columnar Si electrodes is complex, costly, and usually reduces the volumetric energy density of the electrode. The conductive network of the carbon matrix in Si/C composite materials may gradually fail due to the repeated expansion of Si. These inherent limitations mean that electrode structure design must coordinate battery manufacturing processes and overall performance requirements[89].
Design and optimization of electrolytes
The design and optimization of electrolytes are crucial for improving electrochemical performance. Low ion conductivity, poor interface stability, insufficient mechanical buffering capacity, and parasitic side reactions can directly damage battery performance. Therefore, in the design and optimization of electrolytes, people pay more attention to the ion conductivity, mechanical modulus, elasticity, and electrochemical stability of the electrolyte itself. The current research focus is on developing ASSB and QSSB electrolyte systems that are compatible with the large volume expansion of Si-based electrodes, thereby achieving good compatibility with Si electrodes and promoting effective ion transport[90].
For QSE, an effective optimization strategy is to regulate their cross-linked network structure to achieve constraints on the encapsulation and volume changes of Si particles. Zhao et al.[78] designed a novel in situ polymerized QSE for full batteries [Figure 9A], which uses 1,3,5-trioxane (TXE) as the monomer, a dual salt system [LiTFSI/ LiBF2(C2O4), LiDFOB] as the lithium-ion source, and fluoroethylene carbonate (FEC)/ dimethyl carbonate (DMC) as the solvent. The Lewis acid produced through LiDFOB decomposition induces TXE ring-opening polymerization, thus forming a three-dimensional network structure of polyformaldehyde [Figure 9B] that wraps around Si/graphite electrode particles. LiDFOB acts as a film-forming additive and works synergistically with FEC to form an inorganic-organic composite SEI layer rich in LiF. This enhances interfacial stability and reduces the consumption caused by electrolyte side reactions. Besides this, LiTFSI is capable of providing high ionic conductivity while LiDFOB is able to promote lithium-ion dissociation. The synergistic effect of the dual-salt system results in a quasi-solid-state electrolyte (QSSE) with a high ionic conductivity of 2.16 mS cm-1 at 25 °C and an notably high lithium-ion transference number of 0.61. These superior transport properties significantly enhance the rate capability of QSSBs[78].
Figure 9. Constructing a quasi solid electrolyte network through in situ polymerization. (A) Schematic diagram of the preparation of a full battery using an in situ-generated quasi solid electrolyte; (B) The mechanism of TXE polymerization to form a polymer skeleton. Reproduced with permission from Zhao et al., Chemical Engineering Journal (2023). Copyright © Elsevier[78]. TXE: 1,3,5-trioxane.
Another optimization method for GEs is to develop more elastic polymers to alleviate the expansion stress generated by Si particles. Zhao et al.[91] developed and fabricated a super-elastic GPE, which was synthesized through a stepwise reaction process. Initially, the hard ring domain and the soft ether domain were reacted at a temperature of 80 °C to form a prepolymer, and subsequently, ethylene diamine was added to enable crosslinking and curing at a temperature of 70 °C. The critical optimization measure in this step resides in the setting of a hard-to-soft ratio of 2:1, which ensures high elasticity of the electrolyte while enabling it to effectively absorb LE. The GE fills the electrode in an even manner, and it functions like a spring to buffer the expansion stress of Si particles during the charge-discharge process, thus greatly reducing the change in electrode thickness and the displacement of particles [Figure 10A]. This GPE strongly adheres to Si particles and acts as a spring during charging and discharging, buffering the expansion stress of Si particles and greatly reducing electrode thickness changes and particle displacement. It also inhibits the separation of active substances and helps to form a stable SEI layer rich in organic components. From Figure 10B-E, compared with the control SiO electrode, the generation of cracks alongside the increase in electrode impedance were effectively suppressed. This elastic GPE enables a stable capacity of 1,068 mAh g-1 over 250 cycles and 70.0% capacity retention after 350 cycles, which is attributed to its exceptional ability to limit electrode expansion from 94% to 53% and accommodate strain with over 2300% elongation.
Figure 10. The effect of highly elastic GE on the stability of electrode structure. (A) Thickness evolution of the SiO electrode and SiO electrode with elastic GPE during the initial three lithiation/delithiation cycles; SEM images of the top view (B) and cross-section (C) of the SiO electrode after 10 cycles were compared; SEM images of the top view (D) and cross-section (E) of the SiO electrode with elastic GPE after 10 cycles. Reproduced with permission from Zhao et al., Nature Communications (2019). Copyright © Springer Nature[91]. GE: Gel electrolyte; GPE: gel polymer electrolyte; SEM: scanning electron microscopy.
For all-solid-state electrolytes (ASSEs), the optimization focus must balance ionic conductivity and mechanical properties, while also enhancing the elastic modulus to a level comparable to QSEs to withstand the volume changes of Si electrodes. The inherent trade-off between modulus, stability, and conductivity of single solid electrolytes hinders their application. CPE provide a promising alternative by combining conductive fillers with flexible matrices. However, their performance depends on meticulous structural optimization to balance ion conduction and mechanical compliance[92-95]. For example, the 3D porous composite polymer electrolyte developed by Zheng et al.[96] not only utilizes a three-dimensional porous skeleton to promote ion transport but also utilizes in situ electrochemical polymerization of propylene carbonate to further improve ion conductivity and mechanical strength. Meanwhile, a LiF-rich interface layer is formed in situ at the electrolyte/electrode interface, which effectively inhibits dendrite growth and interface failure. This structure notably enhances the mechanical strength and thermal stability of the electrolyte, and this enhancement is crucial for enduring the stress caused by Si electrodes expansion and preventing interface failure. The micro-Si battery equipped with this electrolyte achieved a capacity retention of 98.5% after 100 cycles at 0.2C and maintained good performance under high-rate conditions.
The core of electrolyte optimization lies in the synergistic balance between ion conductivity, mechanical strength, and chemical/electrochemical stability. Although electrolyte optimization has significantly improved interface contact and stress buffering, its application still has significant limitations. The long-term electrochemical stability of highly elastic GE needs to be further verified, especially the tolerance under high voltage and high temperature conditions. The interface impedance problem between fillers and polymer matrix in ASSBs and CPE has not yet been thoroughly overcome. In addition, most electrolyte optimization strategies often need to make trade-offs in at least one dimension, such as ionic conductivity, electrochemical stability window, or cost, while improving material mechanical properties or interface characteristics[97].
Optimization of the electrolyte-electrode interface
In QSSB and ASSB, the instability of the electrolyte electrode interface severely restricts the practical application of Si-based electrodes, which will lead to contact loss, increased impedance, and increased side reactions. Therefore, interface optimization has become a key research direction for improving battery performance[98]. This section introduces two main methods: optimizing the SEI formed in situ during battery operation and constructing an artificial interface protective layer.
For the first method, researchers bring about the formation of high-performance in situ SEI films by adjusting the electrolyte composition, specifically by adding electrolyte additives like fluoroethylene carbonate[99] or vinylene carbonate[100] to modify the composition of the SEI and electrode/electrolyte interface, thereby obtaining a durable interface. Furthermore, Zhang et al.[101] created a thermoplastic polyurethane (TPU)-succinonitrile (SN) reference solid electrolyte which is based on the reinforcement of high-concentration double salt [LiTFSI/LiPF2(C2O4)2, LiDFBOP]. The matrix composed of TPU and SN improves battery performance through adjusting the electrolyte structure and the electrode interface. In addition to providing ionic conductivity, LiDFBOP can also guide the formation of LIF rich SEI layer on the surface of Si electrode, which has low interface energy and high mechanical strength. This interface layer can effectively boost the uniform deposition and desorption of lithium-ions and accommodate the volume changes of Si materials during cycling, thus notably enhancing the interfacial redox kinetics and the cycling stability.
In addition to mixing lithium salts into electrolytes for interface optimization, another innovative approach is to design biomimetic interface structures inspired by natural systems. Liu et al.[102] used a biomimetic interface design to construct a three-dimensional desolvation interface and a stable SEI film derived from anions. In this design, lithium-ion solvation is analogous to impurities in water, and the role of the 3D interface is similar to that of aquatic plants, which can accelerate Li desolvation. The polydioxolane (PDOL) GE was formed through in situ ring-opening polymerization in a 1,3-dioxolane (DOL) solvent. The seaweed-like fiber structure of PDOL is able to increase the contact area with the electrode it can also form a biomimetic three-dimensional interface and this structure significantly lowers the desolvation energy barrier of lithium-ions. The weak coordination ability of DOL/PDOL enables more anions to enter the solvation sheath, a process that forms contact ion pairs and aggregates it also induces the formation of a homogeneous SEI film rich in inorganic substances and this in turn raises the lithium-ion migration rate.
In addition to enhancing the SEI film formed during battery operation to improve battery performance, the construction of artificial interface coatings stands as another effective strategy, one that is particularly suitable for Si-based electrodes with significant volume changes. Zeng et al.[103] designed an artificial interface layer that is based on the garnet-structured solid electrolyte Li6.4La3Zr1.4Ta0.6O12 (LLZTO). This interface layer forms stable Si-O chemical bonds via a crosslinking reaction between polyvinyl alcohol (PVA) and polyacrylic acid (PAA), which is a binder commonly used in Si electrodes, and this reaction allows the LLZTO coating to maintain tight contact with the Si electrode even as the Si undergoes volume expansion [Figure 11A-D]. Compared with conventional Si electrodes, the high ionic conductivity of LLZTO itself offers fast lithium-ion transport channels, while its high mechanical modulus further restricts the expansion of Si. The combined effect of these two factors maintains the interface structure and electrochemical stability. The initial Coulombic efficiency of ASSB equipped with this coating reached 90.4%, much higher than the 81.10% of uncoated Si electrodes. And it can still maintain extremely high capacity and nearly 100% Coulombic efficiency after 1000 cycles, which fully proves the positive effect of artificial interface layer on extending battery life. Similarly, Abdollahifar et al.[104] developed a multifunctional polymer-based artificial SEI layer (A-SEI) that is based on sulfonated chitosan (SCS) crosslinked with glutaraldehyde (GA). This A-SEI layer attains a stable electrode/electrolyte interface via the synergistic effects of mechanical reinforcement, ion-selective conduction, and chemical passivation. Its high mechanical strength and toughness can effectively buffer the large volume changes of Si-based electrodes during charge and discharge processes, reduce the loss of active material, avoid continuous exposure of fresh Si surfaces, and thus suppress electrolyte side reactions and repeated SEI reformation. Schematic diagram of battery A-SEI layer and corresponding cycle performance and stress-strain curve are shown in Figure 11E and F. The A-SEI layer has cation selectivity, which can preferentially guide lithium-ions to rapidly transport through a jumping mechanism, thereby achieving the goal of reducing the interface impedance and electrolyte salt decomposition and LiF deposition.
Figure 11. Effect of A-SEI on structural integrity of the Si-based electrode after cycling (A) Si and (B) Si@LLZTO SEM images of the surface of the electrode before and after 100 cycles. Before and after 100 cycles, (C) Si and (D) Si@LLZTO SEM images of the cross-section of the electrode; (E) Schematic diagram of the A-SEI layer and corresponding cycling performance of the battery. Reproduced with permission from Zeng et al., Surfaces and Interfaces (2024). Copyright © Elsevier[103]; (F) Stress-strain curve of the artificial SCS-GA membrane at
Interface engineering is a key bridge connecting active electrodes with quasi solid/solid electrolytes. Although current strategies can significantly improve initial loop performance, they have limitations in terms of generality, long-term dynamic stability, and process compatibility. The chemical and mechanical stability of the A-SEI layer under extreme potential, long-term cycling, and high volume deformation still needs to be verified. The mechanism of action of biomimetic or dynamic repair interfaces is highly dependent on specific conditions such as temperature, and its effect under actual working conditions still has uncertainty. In addition, most high-performance interface layer preparation processes are intricate and complex, making it difficult to achieve large-scale production and be compatible with existing battery manufacturing processes[105,106].
CONCLUSION AND OUTLOOK
This article conducts a systematic study on the failure mechanisms of QSSBs and ASSBs based on Si-based electrodes, and proposes effective optimization strategies to address these failure causes [Figure 12].
Figure 12. Failure mechanisms and optimization methods of QSSBs and ASSBs with Si-based electrodes. QSSB: Quasi-solid-state battery; ASSB: all-solid-state battery; SSE: solid-state electrolyte; QSE: quasi-solid-state electrolyte; CPE: composite polymer electrolyte.
The failure of Si-based electrode is fundamentally due to its microscopic characteristics, such as anisotropic lithiation of crystalline Si, electrochemical sintering effect and intrinsic low conductivity, which are the internal causes of stress concentration and insufficient transmission power in the battery system. At the macro level, these micro mechanisms triggered a series of chain reactions, such as particle breakage, continuous rupture and reconstruction of SEI film, evolution of electrode pores, loss of electrical contact and blocking of ion transport. Under the combined action of these factors, the capacity of the battery decays rapidly and the impedance increases.In SSB systems, the inherent rigidity of solid electrolytes exacerbates these issues. Solid/solid interfaces are typically characterized by limited point contact and are particularly vulnerable to mechanical stress caused by Si expansion. This results in contact loss, crack propagation, and interface delamination, ultimately leading to battery failure. In addition, the inherent limitations of various solid electrolytes can also lead to the failure of SSB, such as the narrow electrochemical stability window of ISEs, the low room-temperature ionic conductivity of SPEs, and the parasitic side reactions caused by the air sensitivity of sulfide-based electrolytes. These factors further intensify structural degradation and the increase in impedance at the interface.
Compared with ASSBs, QSSBs exhibit better interfacial compliance and strain accommodation ability. However, they also bring their own challenges. The intrinsic ionic conductivity of QSE is generally lower than that of LEs, which limits its rate-conducting capacity and thereby exacerbates battery polarization. The residual liquid components from the QSE curing process will undergo continuous side reactions with the Si electrode, leading to gradual deterioration of the interface, which can also cause battery failure. Although the mechanical buffering capacity of QSE has been significantly enhanced compared to SSE, it is often insufficient to completely prevent microcrack formation under the extreme strain of Si cycling. In addition, the thermal instability of the polymer matrix and the decomposition of residual solvents at high temperatures also bring significant safety and service life issues.
To alleviate the failure mechanisms of ASSB and QSSB using Si-based electrodes, many optimization strategies have been proposed, which can be broadly categorized into three types: Si electrode design, electrolyte optimization, and interface engineering. Columnar Si arrays, core-shell structures, and Si-carbon composite materials, among others, have been developed in the design of Si-based electrode structures to accommodate the directional volume expansion that occurs in Si-based batteries and to maintain the pathways for the transfer of ions and electrons. In terms of electrolyte optimization, for QSEs, the main approach is to construct a network covering Si particles through in situ polymerization to achieve the purpose of suppressing volume expansion. Additionally, high-elastic GEs can be used to enhance stress buffering capacity, while stable SEI layers can be formed by incorporating dual salts and functional additives. For SSEs, CPEs can be developed to balance the high ionic conductivity of ceramic fillers and the high flexibility of polymers, and to enhance the mechanical strength, thermal stability and ion transport performance of the electrolyte through a three-dimensional interpenetrating network. In the field of interface engineering, the focus is on optimizing the mechanical and electrical properties of in situ SEI through additives. Additionally, bionic three-dimensional structures can be designed to promote lithium-ion migration. By constructing artificial interface layers with high modulus and passivation functions, the stability of the interface during long-term cycling can be enhanced.
The current bottleneck in the development of Si-based ASSBs and QSSBs is the fundamental contradiction between the volume change of Si and the intrinsic rigidity of quasi/all solid-state systems. Specifically, (1) the interface inevitably experiences contact loss and mechanical failure during cycling; (2) the existing electrolyte materials are difficult to synergistically achieve high ionic conductivity, mechanical adaptability, and chemical/electrochemical stability to Si; (3) Lack of electrode structure construction methods that can actively adapt to volume changes and maintain continuous ion transport. To promote the subsequent development of Si-based QSSB and ASSB, future research needs to build a multi-layer collaborative solution. At the material level, a Si carbon composite electrode with pre-strain structure and a rigid flexible composite electrolyte system should be developed to actively offset the expansion stress brought by Si through the built-in stress field. At the electrode level, it is necessary to break through the traditional slurry coating process, adopt advanced manufacturing technologies such as template-assisted or 3D printing, and accurately build a 3D interpenetrating network of active substances, solid electrolytes and conductive agents, so that the battery system can maintain the smooth ion/electron transmission path under repeated volume changes. At the interface level, a multi-functional A-SEI with high ionic conductivity, mechanical strength and self repairing function should be designed to realize the transformation from passive stability to active adaptation. Finally, at the level of system integration, engineering challenges such as solid-solid interface stress management under stacking pressure, stability of residual liquid components and compatibility with electrodes must be solved to promote the transformation of QSSBs and ASSBs of Si-based system from laboratory prototype to industrial application. The collaborative promotion of these four levels will establish a cross-scale correlation mechanism of material structure performance, promote the breakthrough of Si-based QSSB and ASSB on the practical road, and finally realize the next generation battery system with high energy density and long cycle stability.
DECLARATIONS
Authors’ contribution
Conceptualization, investigation: Kang, Z.
Investigation: Zhou, H.; Zheng, S.; Yang, D.; Wang, A.
Conceptualization, funding acquisition, project administration, supervision, writing-review and editing:
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors thank the funding support from National Natural Science Foundation of China (52561145238) and Gotion High-Tech. Co., Ltd. (EF2090130053).
Conflicts of interest
Tan, P. is the Guest Editor of the Special Issue “Quasi/All-Solid-State Electrolytes for Advanced Batteries” in Energy Materials; however, he was not involved in any stage of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. Wang, A. and Yang D. are affiliated with Gotion High-Tech Co., Ltd. All other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Pomerantseva, E.; Bonaccorso, F.; Feng, X.; Cui, Y.; Gogotsi, Y. Energy storage: the future enabled by nanomaterials. Science 2019, 366.
2. Kwon, T. W.; Choi, J. W.; Coskun, A. The emerging era of supramolecular polymeric binders in silicon anodes. Chem. Soc. Rev. 2018, 47, 2145-64.
3. Goodenough, J. B.; Park, K. S. The Li-ion rechargeable battery: a perspective. J. Am. Chem. Soc. 2013, 135, 1167-76.
4. Yang, Z.; Zhang, J.; Kintner-Meyer, M. C.; et al. Electrochemical energy storage for green grid. Chem. Rev. 2011, 111, 3577-613.
5. Liu, L.; Li, M.; Chu, L.; et al. Layered ternary metal oxides: performance degradation mechanisms as cathodes, and design strategies for high-performance batteries. Prog. Mater. Sci. 2020, 111, 100655.
6. Wang, Q.; Mao, B.; Stoliarov, S. I.; Sun, J. A review of lithium ion battery failure mechanisms and fire prevention strategies. Prog. Energy. Combust. Sci. 2019, 73, 95-131.
7. Cao, C.; Zhong, Y.; Shao, Z. Electrolyte engineering for safer lithium‐ion batteries: a review. Chin. J. Chem. 2023, 41, 1119-41.
8. Ahmed, M. S.; Islam, M.; Raut, B.; Yun, S.; Kim, H. Y.; Nam, K. W. A Comprehensive review of functional gel polymer electrolytes and applications in lithium-ion Battery. Gels 2024, 10, 563.
9. Gan, L.; Chen, R.; Yu, X.; Li, H. Understanding the battery safety improvement enabled by a quasi-solid-state battery design. Chinese. Phys. B. 2022, 31, 118202.
10. Pervez, S. A.; Cambaz, M. A.; Thangadurai, V.; Fichtner, M. Interface in solid-state lithium battery: challenges, progress, and outlook. ACS. Appl. Mater. Interfaces. 2019, 11, 22029-50.
11. Schumm, B.; Dupuy, A.; Lux, M.; et al. Dry Battery electrode technology: from early concepts to industrial applications. Adv. Energy. Mater. 2025, 15, 2406011.
12. Yu, J.; Kim, B.; Yoo, Y. Dry Electrode processing for lithium‐ion battery cathodes and anodes: materials, fabrication strategies, and future outlook. Adv. Mater. Technol. 2025, 10, e01420.
13. Sun, Y.; Liu, N.; Cui, Y. Promises and challenges of nanomaterials for lithium-based rechargeable batteries. Nat. Energy. 2016, 1, 16071.
14. Zhang, R.; Li, N. W.; Cheng, X. B.; Yin, Y. X.; Zhang, Q.; Guo, Y. G. Advanced micro/nanostructures for lithium metal anodes. Adv. Sci. 2017, 4, 1600445.
15. Chen, J.; Luo, B.; Chen, Q.; et al. Localized electrons enhanced ion transport for ultrafast electrochemical energy storage. Adv. Mater. 2020, 32, e1905578.
16. Obrovac, M. N.; Krause, L. J. Reversible cycling of crystalline silicon powder. J. Electrochem. Soc. 2007, 154, A103.
17. Chang, H.; Wu, Y.; Han, X.; Yi, T. Recent developments in advanced anode materials for lithium-ion batteries. Energy. Mater. 2022, 1, 100003.
18. Zuo, X.; Zhu, J.; Müller-buschbaum, P.; Cheng, Y. Silicon based lithium-ion battery anodes: a chronicle perspective review. Nano. Energy. 2017, 31, 113-43.
19. Song, S.; Gao, W.; Yang, G.; et al. Hybrid poly-ether/carbonate ester electrolyte engineering enables high oxidative stability for quasi-solid-state lithium metal batteries. Mater. Today. Energy. 2022, 23, 100893.
20. Xu, Z.; Yang, J.; Li, H.; Nuli, Y.; Wang, J. Electrolytes for advanced lithium ion batteries using silicon-based anodes. J. Mater. Chem. A. 2019, 7, 9432-46.
21. Guo, Y.; Wu, S.; He, Y.; et al. Solid-state lithium batteries: safety and prospects. eScience 2022, 2, 138-63.
22. Zhang, H.; Liu, G.; Li, H.; et al. Recent progress of multilayer polymer electrolytes for lithium batteries. Energy. Mater. 2023, 3, 300030.
23. Zhang, Q.; Liu, K.; Ding, F.; Liu, X. Recent advances in solid polymer electrolytes for lithium batteries. Nano. Res. 2017, 10, 4139-74.
24. Castillo, J.; Qiao, L.; Santiago, A.; et al. Perspective of polymer-based solid-state Li-S batteries. Energy. Mater. 2022, 2, 200021.
25. Su, H.; Jiang, Z.; Liu, Y.; et al. Recent progress of sulfide electrolytes for all-solid-state lithium batteries. Energy. Mater. 2022, 2, 200013.
26. Cui, Y.; Wan, J.; Ye, Y.; Liu, K.; Chou, L. Y.; Cui, Y. A fireproof, lightweight, polymer-polymer solid-state electrolyte for safe lithium batteries. Nano. Lett. 2020, 20, 1686-92.
27. Liu, L.; Mo, J.; Li, J.; et al. Comprehensively-modified polymer electrolyte membranes with multifunctional PMIA for highly-stable all-solid-state lithium-ion batteries. J. Energy. Chem. 2020, 48, 334-43.
28. Zhu, X.; Jiang, W.; Zhao, S.; et al. Exploring the concordant solid-state electrolytes for all-solid-state lithium-sulfur batteries. Nano. Energy. 2022, 96, 107093.
29. Zhu, X.; Wu, J.; Lu, J. Insight into Inorganic solid‐state electrolytes: ionic transport and failure mechanisms. Adv. Funct. Mater. 2024, 34, 2409547.
30. Jiang, P.; Du, G.; Cao, J.; et al. Solid‐state Li ion batteries with oxide solid electrolytes: progress and perspective. Energy. Tech. 2023, 11, 2201288.
31. Reddy, M. V.; Julien, C. M.; Mauger, A.; Zaghib, K. Sulfide and oxide inorganic solid electrolytes for all-solid-state Li batteries: a review. Nanomaterials. 2020, 10, 1066.
32. Nie, X.; Hu, J.; Li, C. Halide‐based solid electrolytes: the history, progress, and challenges. Interdiscip. Mater. 2023, 2, 365-89.
33. Li, S.; Zhang, S. Q.; Shen, L.; et al. Progress and perspective of ceramic/polymer composite solid electrolytes for lithium batteries. Adv. Sci. 2020, 7, 1903088.
34. Sun, C.; Liu, J.; Gong, Y.; Wilkinson, D. P.; Zhang, J. Recent advances in all-solid-state rechargeable lithium batteries. Nano. Energy. 2017, 33, 363-86.
35. Wang, X.; Zhang, Y.; Zhang, X.; et al. Lithium-salt-rich PEO/Li0.3La0.557TiO3 interpenetrating composite electrolyte with three-dimensional ceramic nano-backbone for all-solid-state lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2018, 10, 24791-8.
36. Park, R. J.; Eschler, C. M.; Fincher, C. D.; et al. Semi-solid alkali metal electrodes enabling high critical current densities in solid electrolyte batteries. Nat. Energy. 2021, 6, 314-22.
37. Lahiri, A.; Pulletikurthi, G.; Shapouri Ghazvini, M.; Höfft, O.; Li, G.; Endres, F. Ionic liquid-organic solvent mixture-based polymer gel electrolyte with high lithium concentration for Li-ion batteries. J. Phys. Chem. C. 2018, 122, 24788-800.
38. Mazzapioda, L.; Tsurumaki, A.; Di Donato, G.; Adenusi, H.; Navarra, M. A.; Passerini, S. Quasi-solid-state electrolytes - strategy towards stabilising Li|inorganic solid electrolyte interfaces in solid-state Li metal batteries. Energy. Mater. 2023, 3, 300036.
39. Zhu, M.; Wu, J.; Wang, Y.; et al. Recent advances in gel polymer electrolyte for high-performance lithium batteries. J. Energy. Chem. 2019, 37, 126-42.
40. Liu, H.; Luo, S.; Yang, Y.; et al. In-situ polymerization formed self-healing quasi-solid electrolyte for high-loading lithium batteries. Energy. Storage. Mater. 2025, 78, 104250.
41. Rui, X.; Hua, R.; Ren, D.; et al. In situ polymerization facilitating practical high-safety quasi-solid-state batteries. Adv. Mater. 2024, 36, e2402401.
42. He, Z.; Zhang, C.; Zhu, Z.; Yu, Y.; Zheng, C.; Wei, F. Advances in carbon nanotubes and carbon coatings as conductive networks in silicon‐based anodes. Adv. Funct. Mater. 2024, 34, 2408285.
43. Feng, K.; Li, M.; Liu, W.; et al. Silicon-based anodes for lithium-ion batteries: from fundamentals to practical applications. Small 2018, 14, 1702737.
44. Li, S.; Liu, Y.; Zhang, Y.; et al. A review of rational design and investigation of binders applied in silicon-based anodes for lithium-ion batteries. J. Power. Sources. 2021, 485, 229331.
45. Lee, W.; Byeon, Y. S.; Lee, S.; Kong, S.; Park, M. S.; Yoon, W. S. Over‐ and hyper‐lithiated oxides as sacrificial cathodes for lithium‐ion batteries. Adv. Energy. Mater. 2024, 15, 2304533.
46. Zhang, Z.; Sun, Z.; Han, X.; et al. An all-electrochem-active silicon anode enabled by spontaneous Li-Si alloying for ultra-high performance solid-state batteries. Energy. Environ. Sci. 2024, 17, 1061-72.
47. Kumar, R.; Lu, P.; Xiao, X.; Huang, Z.; Sheldon, B. W. Strain-induced lithium losses in the solid electrolyte interphase on silicon electrodes. ACS. Appl. Mater. Interfaces. 2017, 9, 28406-17.
48. Xu, K. Electrolytes and interphases in Li-ion batteries and beyond. Chem. Rev. 2014, 114, 11503-618.
49. Radvanyi, E.; Porcher, W.; De Vito, E.; Montani, A.; Franger, S.; Jouanneau Si Larbi, S. Failure mechanisms of nano-silicon anodes upon cycling: an electrode porosity evolution model. Phys. Chem. Chem. Phys. 2014, 16, 17142-53.
50. Seo, J. Y.; Kim, S.; Kim, J. H.; et al. Mechanical shutdown of battery separators: silicon anode failure. Nat. Commun. 2024, 15, 10134.
51. Zhao, M.; Zhang, J.; Wang, W.; Zhang, Q. The preparation of mass producible, highly-cycling stable Si/C anode materials with nano-sized silicon crystals embedded in highly amorphous silicon matrix. Nanotechnology 2021, 32, 485404.
52. Chon, M. J.; Sethuraman, V. A.; McCormick, A.; Srinivasan, V.; Guduru, P. R. Real-time measurement of stress and damage evolution during initial lithiation of crystalline silicon. Phys. Rev. Lett. 2011, 107, 045503.
53. Lee, S. W.; McDowell, M. T.; Choi, J. W.; Cui, Y. Anomalous shape changes of silicon nanopillars by electrochemical lithiation. Nano. Lett. 2011, 11, 3034-9.
54. McDowell, M. T.; Lee, S. W.; Harris, J. T.; et al. In situ TEM of two-phase lithiation of amorphous silicon nanospheres. Nano. Lett. 2013, 13, 758-64.
55. Hatchard, T. D.; Dahn, J. R. In situ XRD and electrochemical study of the reaction of lithium with amorphous silicon. J. Electrochem. Soc. 2004, 151, A838.
56. Wang, J. W.; He, Y.; Fan, F.; et al. Two-phase electrochemical lithiation in amorphous silicon. Nano. Lett. 2013, 13, 709-15.
57. Zhang, D.; Lu, J.; Pei, C.; Ni, S. Electrochemical activation, sintering, and reconstruction in energy‐storage technologies: origin, development, and prospects. Adv. Energy. Mater. 2022, 12, 2103689.
58. Piper, D. M.; Yersak, T. A.; Lee, S. Effect of compressive stress on electrochemical performance of silicon anodes. J. Electrochem. Soc. 2012, 160, A77-81.
59. Whiteley, J. M.; Kim, J. W.; Piper, D. M.; Lee, S. High-capacity and highly reversible silicon-tin hybrid anode for solid-state lithium-ion batteries. J. Electrochem. Soc. 2015, 163, A251-4.
60. Tan, D. H. S.; Chen, Y. T.; Yang, H.; et al. Carbon-free high-loading silicon anodes enabled by sulfide solid electrolytes. Science 2021, 373, 1494-9.
61. Shen, D.; Huang, C.; Gan, L.; Liu, J.; Gong, Z.; Long, M. Rational design of Si@SiO2/C composites using sustainable cellulose as a carbon resource for anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2018, 10, 7946-54.
62. Kato, Y.; Shiotani, S.; Morita, K.; Suzuki, K.; Hirayama, M.; Kanno, R. All-solid-state batteries with thick electrode configurations. J. Phys. Chem. Lett. 2018, 9, 607-13.
63. Chen, C.; Xu, S.; Kuang, Y.; et al. Nature‐inspired tri‐pathway design enabling high‐performance flexible Li-O2 batteries. Adv. Energy. Mater. 2019, 9, 1802964.
64. Hlushkou, D.; Reising, A. E.; Kaiser, N.; et al. The influence of void space on ion transport in a composite cathode for all-solid-state batteries. J. Power. Sources. 2018, 396, 363-70.
65. Kraytsberg, A.; Ein‐eli, Y. Conveying advanced Li‐ion battery materials into practice the impact of electrode slurry preparation skills. Adv. Energy. Mater. 2016, 6, 1600655.
66. Koerver, R.; Zhang, W.; De Biasi, L.; et al. Chemo-mechanical expansion of lithium electrode materials - on the route to mechanically optimized all-solid-state batteries. Energy. Environ. Sci. 2018, 11, 2142-58.
67. Song, A.; Zhang, W.; Guo, H.; et al. A review on the features and progress of silicon anodes‐based solid‐state batteries. Adv. Energy. Mater. 2023, 13, 2301464.
68. Nelson, D. L.; Sandoval, S. E.; Pyo, J.; et al. Fracture dynamics in silicon anode solid-state batteries. ACS. Energy. Lett. 2024, 9, 6085-95.
69. Kalnaus, S.; Dudney, N. J.; Westover, A. S.; Herbert, E.; Hackney, S. Solid-state batteries: the critical role of mechanics. Science 2023, 381, eabg5998.
70. Koerver, R.; Aygün, I.; Leichtweiß, T.; et al. Capacity fade in solid-state batteries: interphase formation and chemomechanical processes in nickel-rich layered oxide cathodes and lithium thiophosphate solid electrolytes. Chem. Mater. 2017, 29, 5574-82.
71. Schwietert, T. K.; Arszelewska, V. A.; Wang, C.; et al. Clarifying the relationship between redox activity and electrochemical stability in solid electrolytes. Nat. Mater. 2020, 19, 428-35.
72. Zhang, C.; Zhuang, H.; Qi, Z.; Liu, X.; Ren, Y. The effect of defects for the ion transport of Li3ScCl6 and Li3InCl6 with the interface of lithium metal anode: a first-principles study. Mater. Today. Commun. 2023, 35, 105764.
73. Wang, H.; Tan, R.; Yang, Z.; Feng, Y.; Duan, X.; Ma, J. Stabilization perspective on metal anodes for aqueous batteries. Adv. Energy. Mater. 2020, 11, 2000962.
74. Zhu, Y.; Mo, Y. Materials design principles for air-stable lithium/sodium solid electrolytes. Angew. Chem. Int. Ed. 2020, 59, 17472-6.
75. De Biasi, L.; Kondrakov, A. O.; Geßwein, H.; Brezesinski, T.; Hartmann, P.; Janek, J. Between scylla and charybdis: balancing among structural stability and energy density of layered NCM cathode materials for advanced lithium-ion batteries. J. Phys. Chem. C. 2017, 121, 26163-71.
76. Ryu, H.; Park, K.; Yoon, C. S.; Sun, Y. Capacity fading of Ni-rich Li[NixCoyMn1-x-y]O2 (0.6 ≤ x ≤ 0.95) cathodes for high-energy-density lithium-ion batteries: bulk or surface degradation? Chem. Mater. 2018, 30, 1155-63.
77. Suebsing, N.; Jaumaux, P.; Safaei, J.; et al. Integrating surface coating and quasi-solid deep eutectic electrolytes for enhanced cycling of micro-Si anodes in Li-ion batteries. Sustain. Energy. Fuels. 2025, 9, 1480-5.
78. Zhao, E.; Luo, S.; Hu, A.; et al. Rational design of an in-build quasi-solid-state electrolyte for high-performance lithium-ion batteries with the silicon-based anode. Chem. Eng. J. 2023, 463, 142306.
79. Yu, M.; Ma, J.; Dong, L.; et al. Stabilizing the SiOx anode by a highly elastic quasi-solid polyether-based electrolyte via an in situ fabrication. ACS. Appl. Mater. Interfaces. 2025, 17, 14097-108.
80. Kido, R.; Horikawa, T.; Sano, A.; et al. Highly safe quasi-solid-state lithium ion batteries with two kinds of nearly saturated and non-flammable electrolyte solutions. J. Energy. Storage. 2024, 102, 114115.
81. Huang, Q.; Song, J.; Gao, Y.; et al. Supremely elastic gel polymer electrolyte enables a reliable electrode structure for silicon-based anodes. Nat. Commun. 2019, 10, 5586.
82. Xue, N.; Yuan, Z.; Wang, W.; Xu, R.; Xie, J.; Lei, C. Influence of flexible gelatinous coating layer on electrochemical performance and fatigue failure of lithium battery separator. Colloids. Surf. A. Physicochem. Eng. Asp. 2022, 633, 127875.
83. Liu, T.; Zhang, J.; Han, W.; et al. Review-in situ polymerization for integration and interfacial protection towards solid state lithium batteries. J. Electrochem. Soc. 2020, 167, 070527.
84. Zhan, X.; Li, M.; Li, S.; et al. Challenges and opportunities towards silicon-based all-solid-state batteries. Energy. Storage. Mater. 2023, 61, 102875.
85. Cangaz, S.; Hippauf, F.; Reuter, F. S.; et al. Enabling high‐energy solid‐state batteries with stable anode interphase by the use of columnar silicon anodes. Adv. Energy. Mater. 2020, 10, 2001320.
86. Dunlap, N. A.; Kim, S.; Jeong, J. J.; Oh, K. H.; Lee, S. Simple and inexpensive coal-tar-pitch derived Si-C anode composite for all-solid-state Li-ion batteries. Solid. State. Ionics. 2018, 324, 207-17.
87. Gao, P.; Huang, X.; Zhao, Y.; et al. Formation of Si hollow structures as promising anode materials through reduction of silica in AlCl3-NaCl molten salt. ACS. Nano. 2018, 12, 11481-90.
88. Pandey, G. P.; Klankowski, S. A.; Li, Y.; et al. Effective infiltration of gel polymer electrolyte into silicon-coated vertically aligned carbon nanofibers as anodes for solid-state lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2015, 7, 20909-18.
89. Li, B.; Zhang, K.; Yu, B.; et al. Achieving cycling stability of silicon-based anodes for lithium-ion batteries: From lithium storage/failure mechanism to structure optimization. J. Energy. Storage. 2025, 136, 118395.
90. Li, J.; Daniel, C.; Wood, D. Materials processing for lithium-ion batteries. J. Power. Sources. 2011, 196, 2452-60.
91. Zhao, M.; Zhang, J.; Costa, C. M.; Lanceros-Méndez, S.; Zhang, Q.; Wang, W. Unveiling challenges and opportunities in silicon-based all-solid-state batteries: thin-film bonding with mismatch strain. Adv. Mater. 2024, 36, e2308590.
92. Bachman, J. C.; Muy, S.; Grimaud, A.; et al. Inorganic solid-state electrolytes for lithium batteries: mechanisms and properties governing ion conduction. Chem. Rev. 2016, 116, 140-62.
93. Jung, W. D.; Jeon, M.; Shin, S. S.; et al. Functionalized sulfide solid electrolyte with air-stable and chemical-resistant oxysulfide nanolayer for all-solid-state batteries. ACS. Omega. 2020, 5, 26015-22.
94. Zhang, F.; Huang, Q.; Tang, Z.; et al. A review of mechanics-related material damages in all-solid-state batteries: Mechanisms, performance impacts and mitigation strategies. Nano. Energy. 2020, 70, 104545.
95. Xu, Z.; Tang, X.; Wang, S.; et al. Overview of research on composite electrolytes for solid-state batteries. Energy. Storage. Sci. Technol. 2021, 10, 1737-51.
96. Zheng, Y.; Yao, Y.; Ou, J.; et al. A review of composite solid-state electrolytes for lithium batteries: fundamentals, key materials and advanced structures. Chem. Soc. Rev. 2020, 49, 8790-839.
97. Cao, Z.; Zheng, X.; Qu, Q.; Huang, Y.; Zheng, H. Electrolyte design enabling a high-safety and high-performance Si anode with a tailored electrode-electrolyte interphase. Adv. Mater. 2021, 33, e2103178.
98. Liu, G.; Yuan, Z.; Deng, Y.; et al. A review on “growth mechanisms and optimization strategies for the interface state of solid‐state lithium‐ion batteries”. Int. J. Appl. Ceram. Technol. 2023, 21, 5-43.
99. Etacheri, V.; Haik, O.; Goffer, Y.; et al. Effect of fluoroethylene carbonate (FEC) on the performance and surface chemistry of Si-nanowire Li-ion battery anodes. Langmuir 2012, 28, 965-76.
100. Aurbach, D.; Gamolsky, K.; Markovsky, B.; Gofer, Y.; Schmidt, M.; Heider, U. On the use of vinylene carbonate (VC) as an additive to electrolyte solutions for Li-ion batteries. Electrochim. Acta. 2002, 47, 1423-39.
101. Zhang, Y.; Zhang, L.; Zhao, Z.; et al. Lithiophilic Li-Si alloy-solid electrolyte interface enabled by high-concentration dual salt-reinforced quasi-solid-state electrolyte. J. Energy. Chem. 2024, 95, 216-30.
102. Liu, X.; Wang, D.; Zhang, Z.; et al. Gel polymer electrolyte enables low-temperature and high-rate lithium-ion batteries via bionic interface design. Small 2024, 20, e2404879.
103. Zeng, B.; Gu, Q.; Zhang, Y.; et al. Engineering electrode-electrolyte interface for ultrastable Si-based solid-state batteries. Surf. Interfaces. 2024, 44, 103687.
104. Abdollahifar, M.; Vinograd, A.; Lu, C. Y.; et al. Enabling long-cycling life of Si-on-graphite composite anodes via fabrication of a multifunctional polymeric artificial solid-electrolyte interphase protective layer. ACS. Appl. Mater. Interfaces. 2022, 14, 38824-34.
105. Luo, W.; Chen, X.; Xia, Y.; et al. Surface and interface engineering of silicon‐based anode materials for lithium‐ion batteries. Adv. Energy. Mater. 2017, 7, 1701083.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






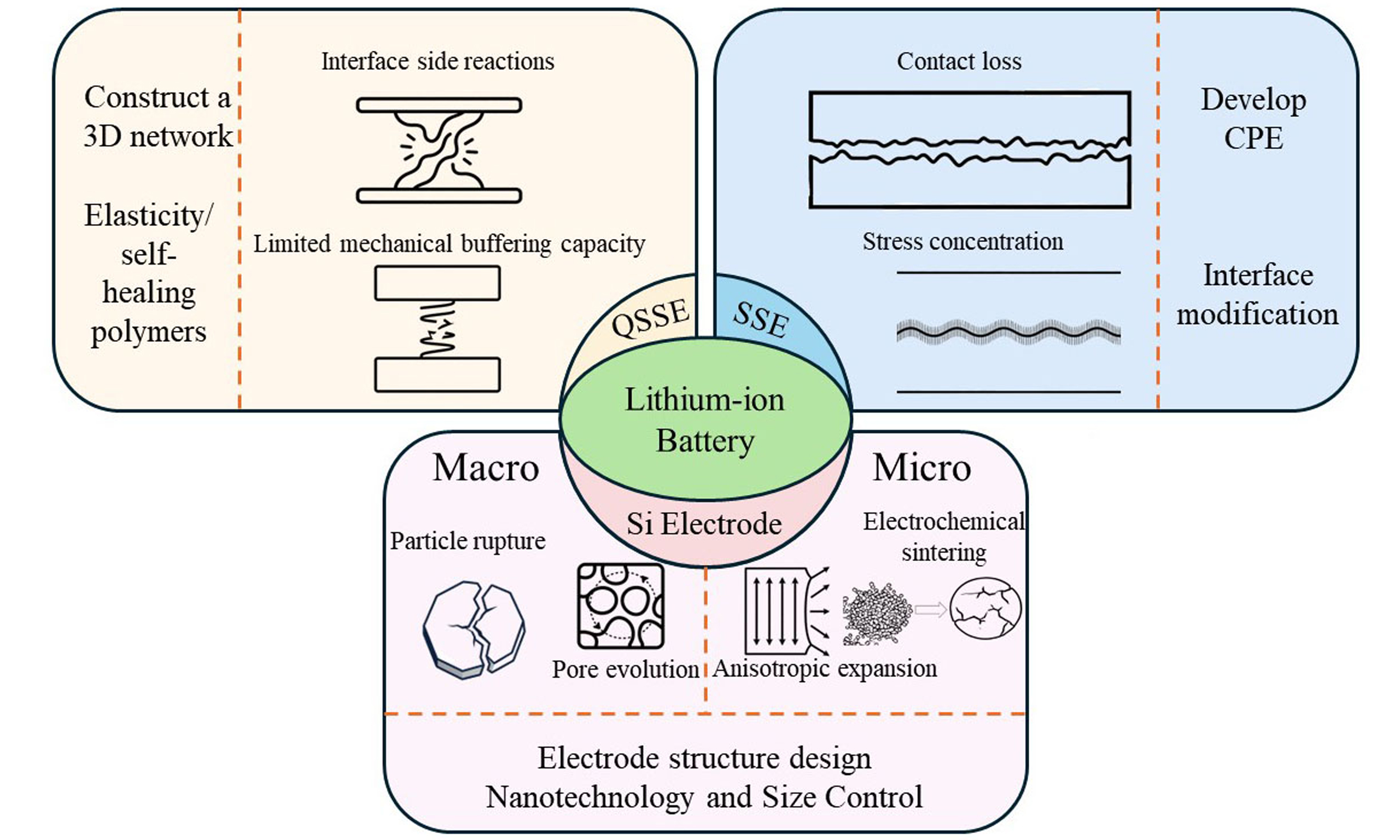
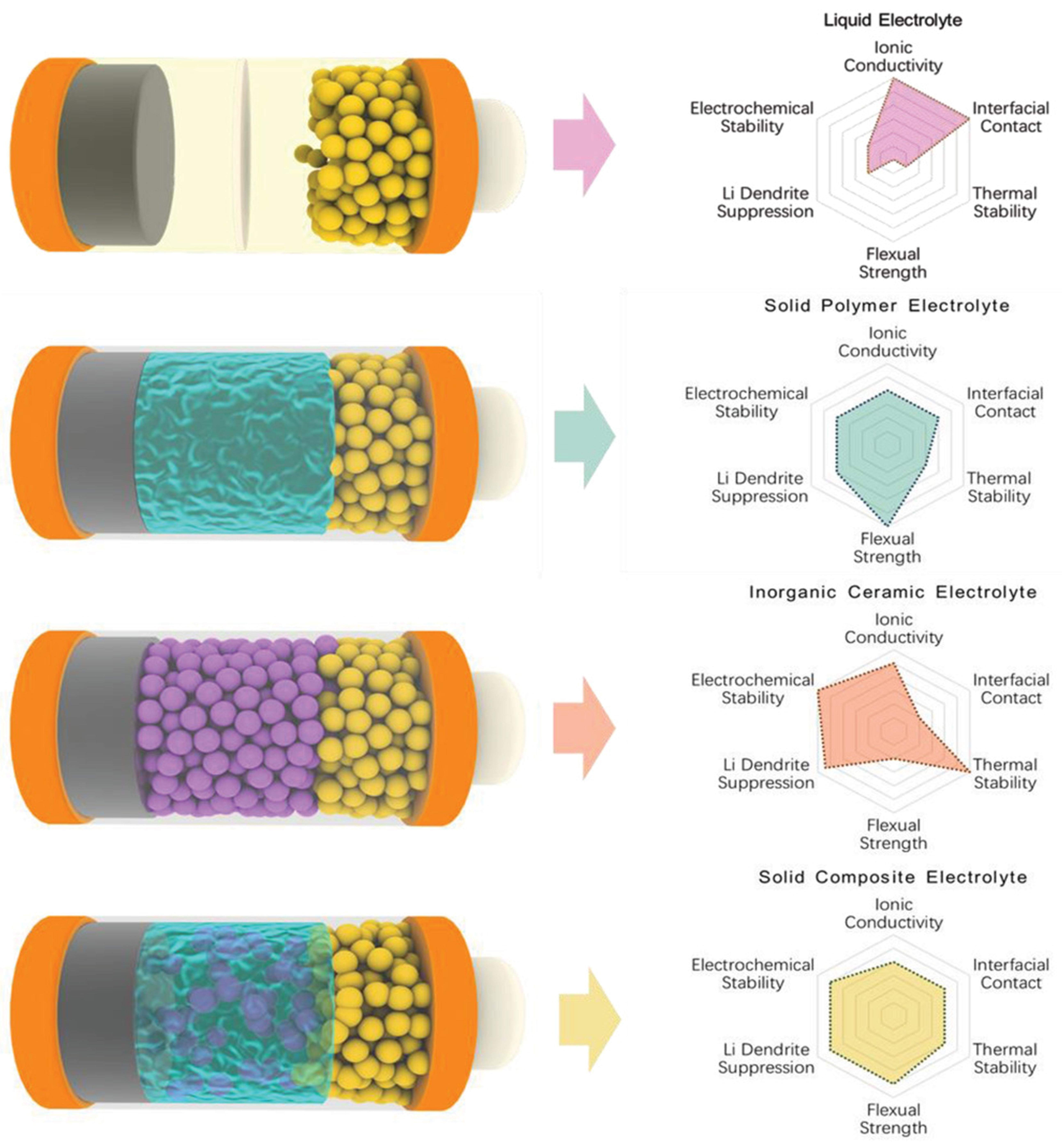
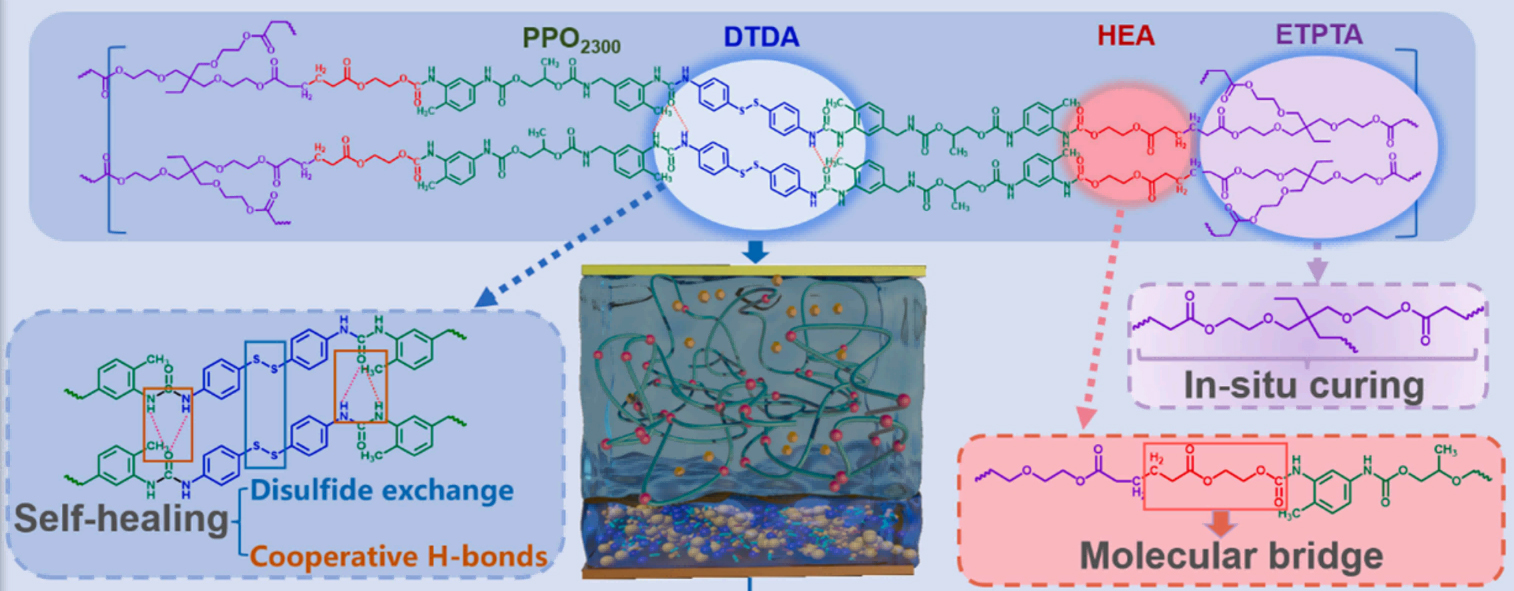
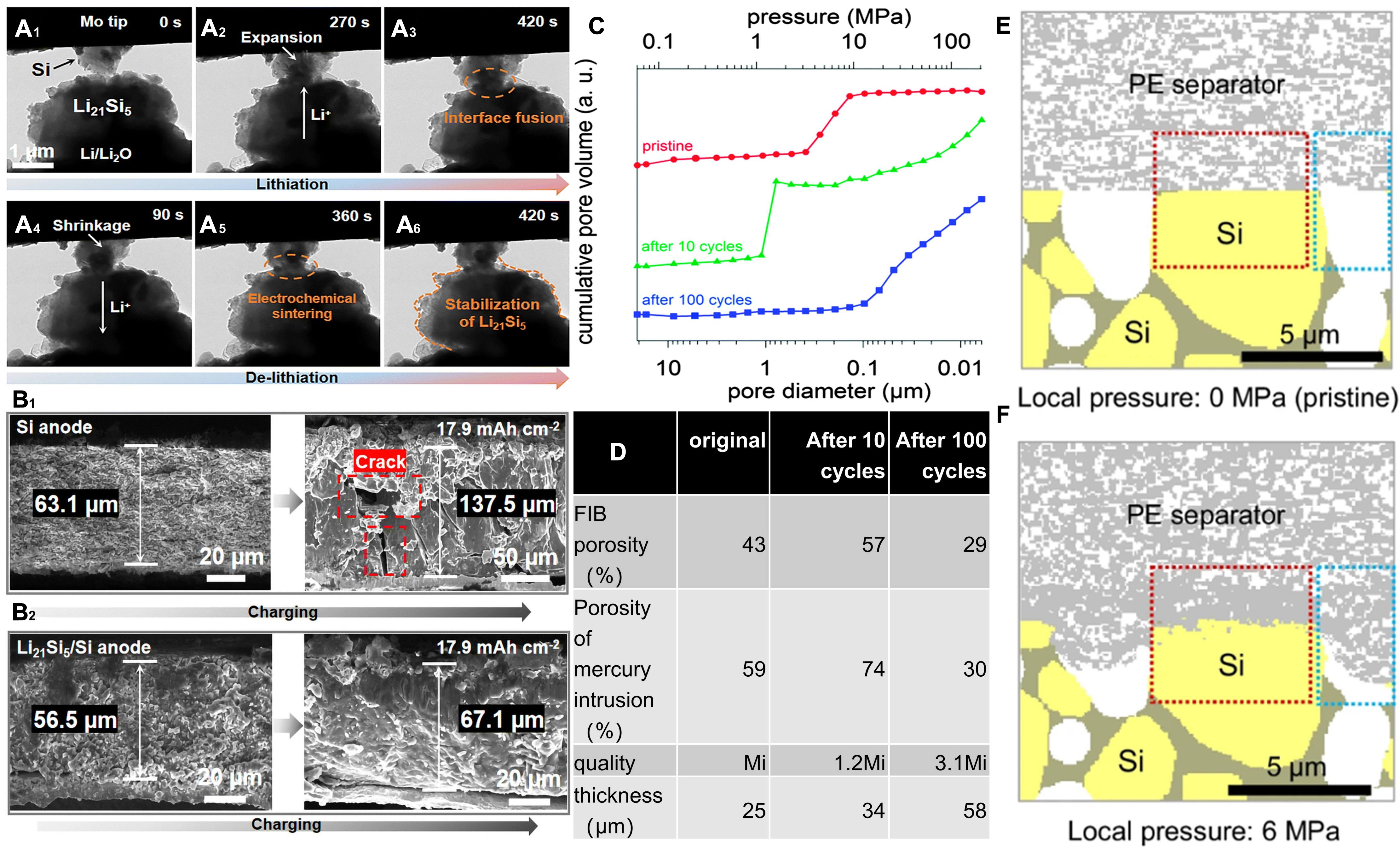

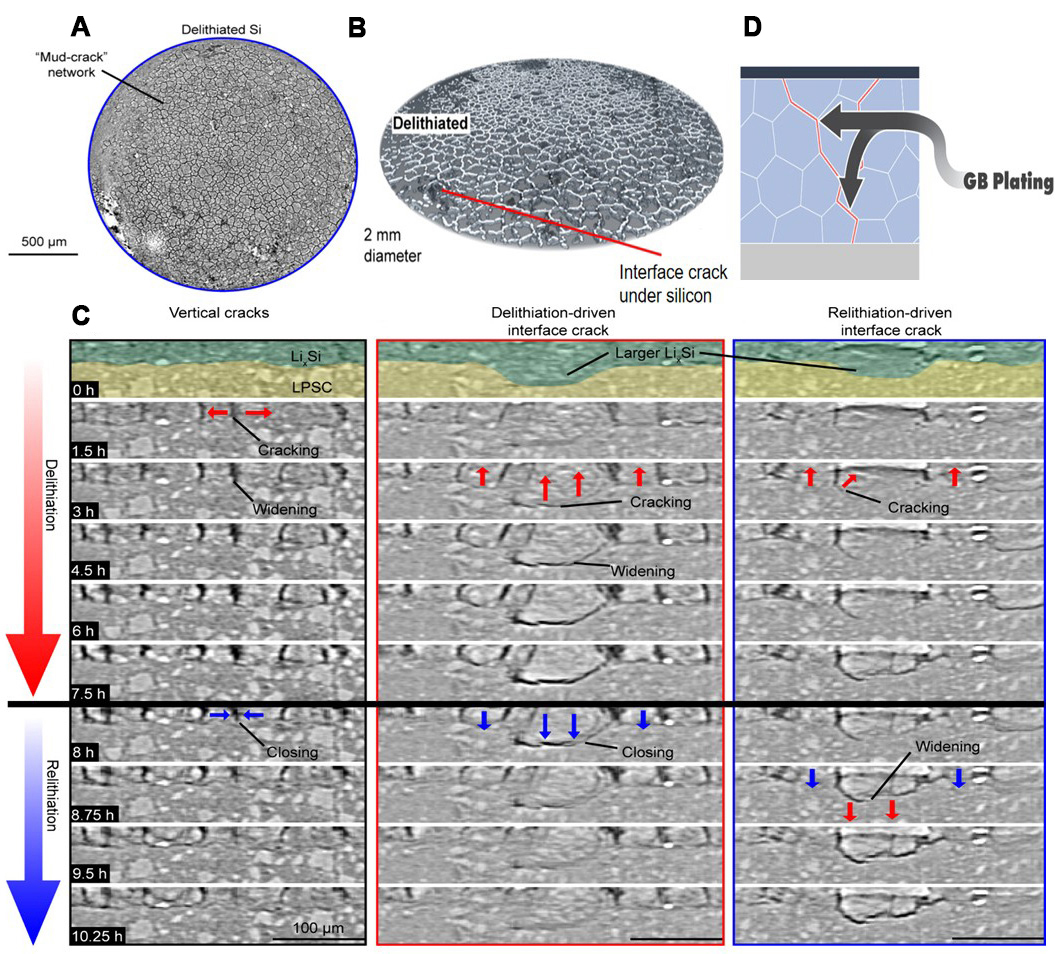
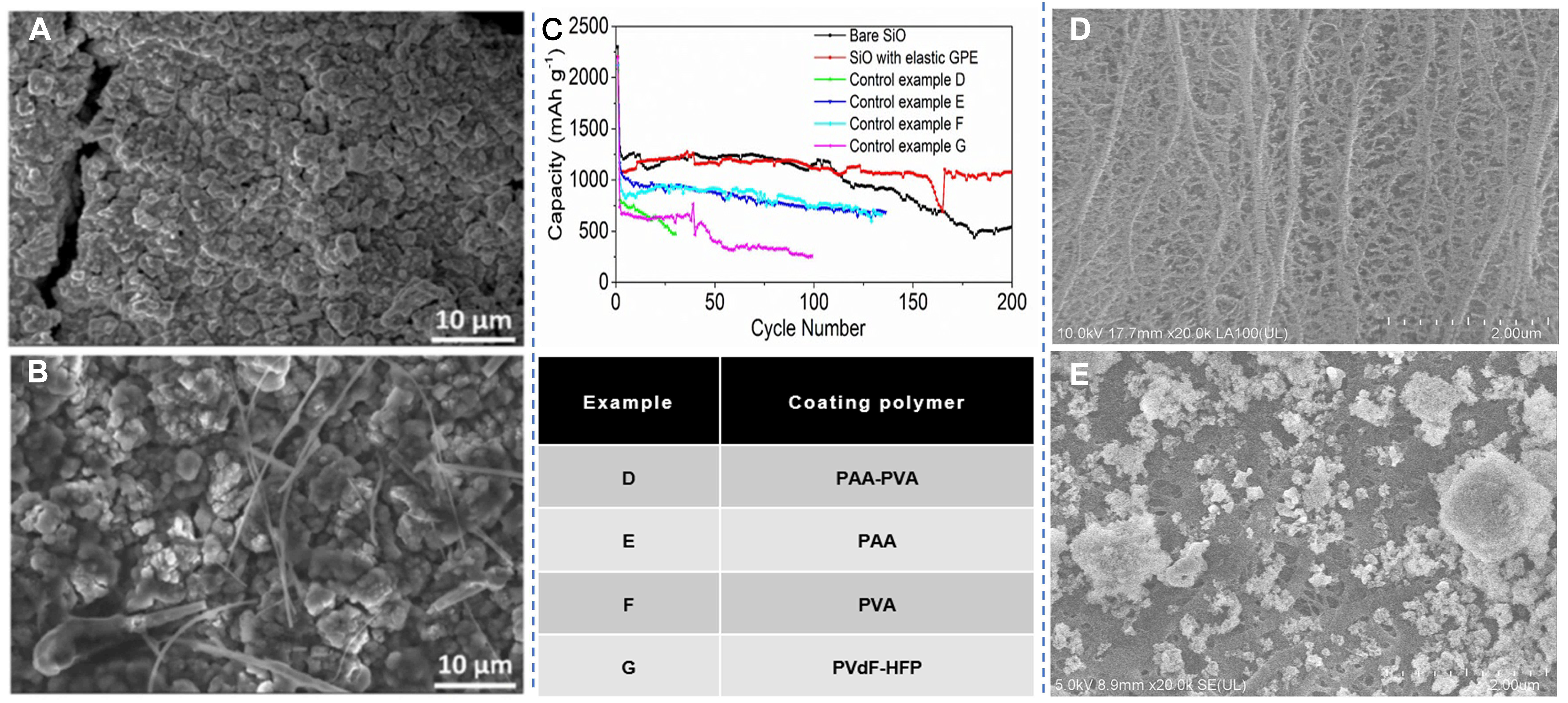
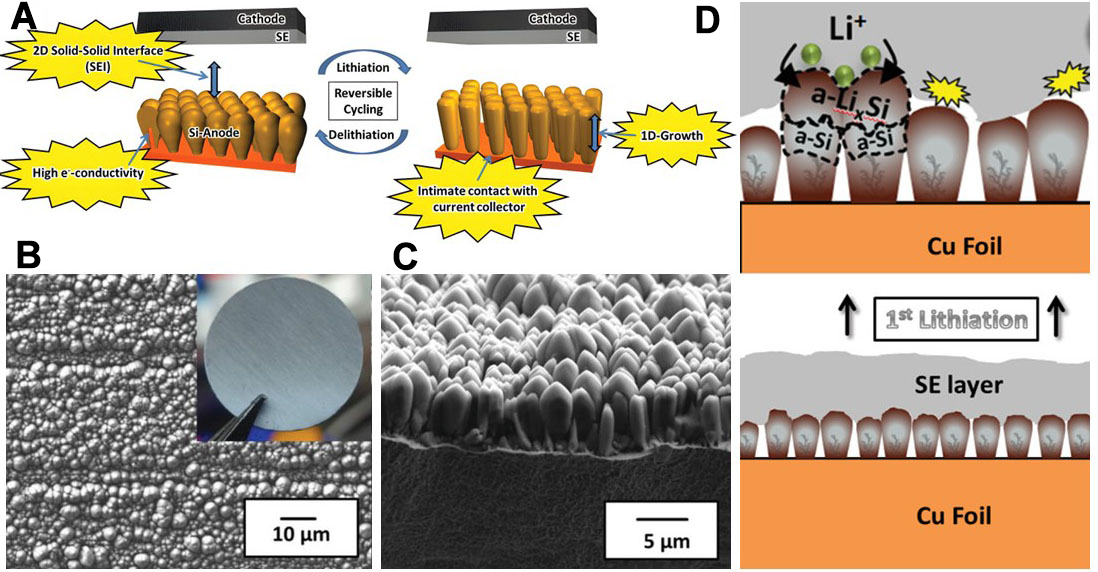
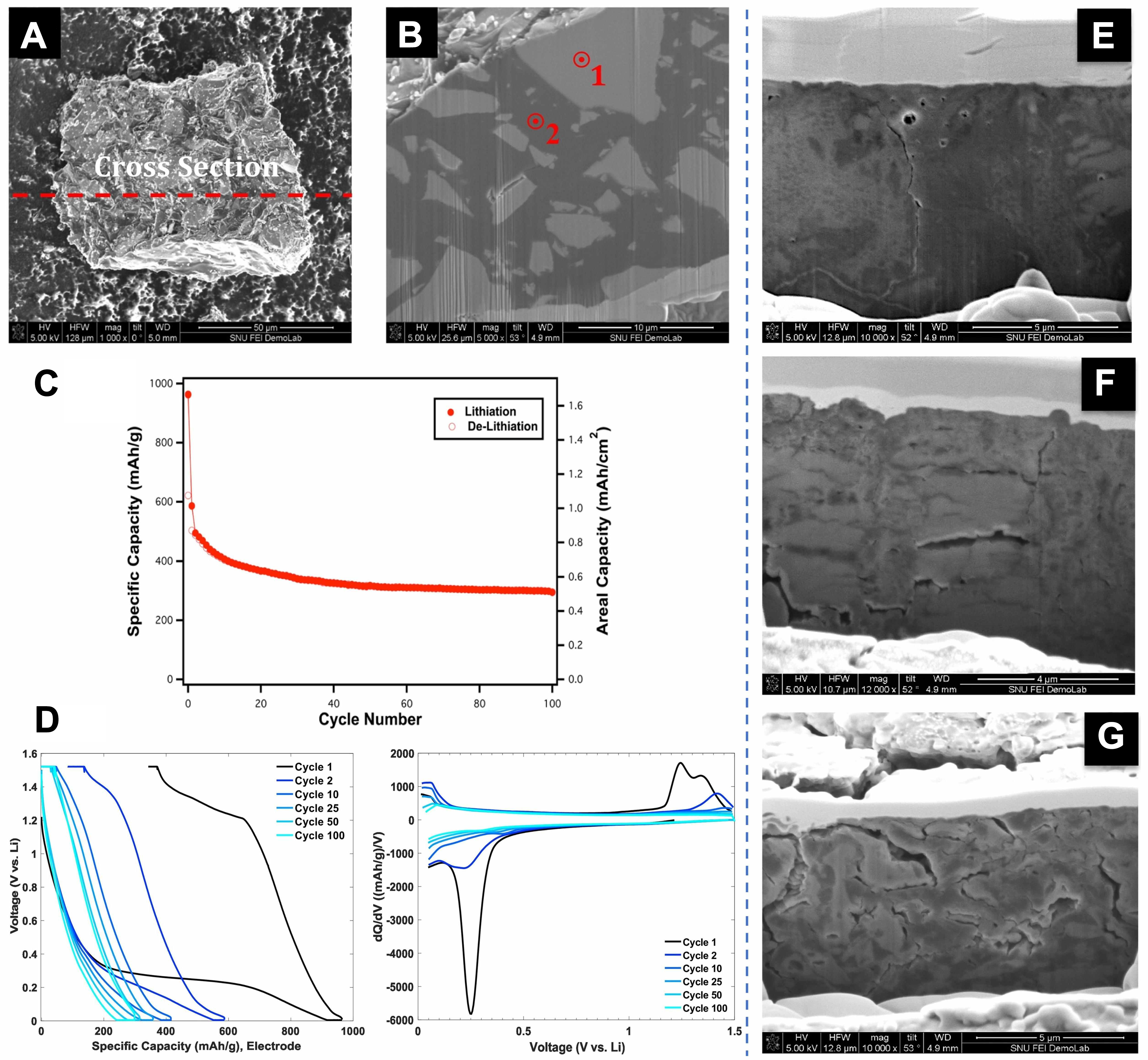
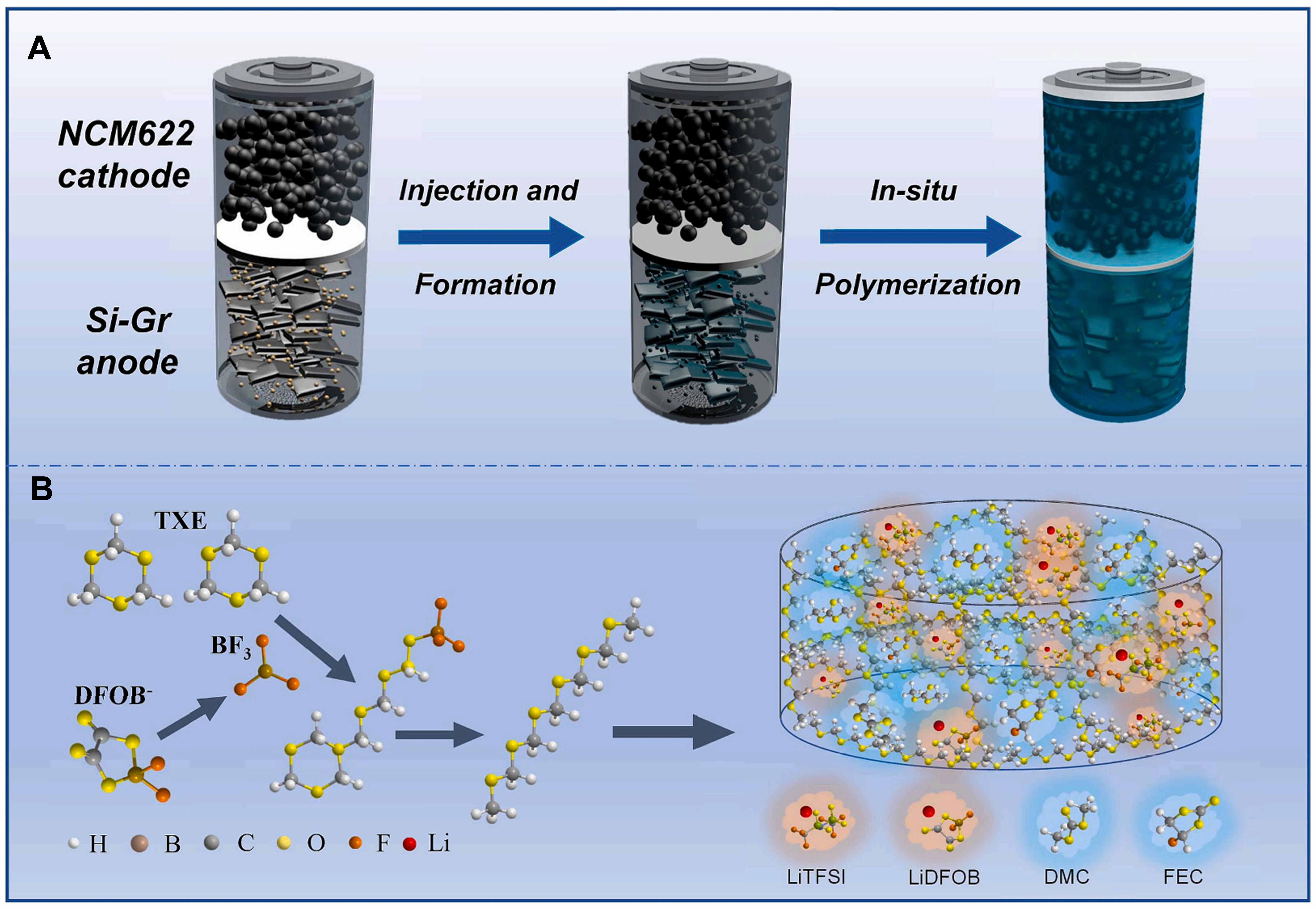
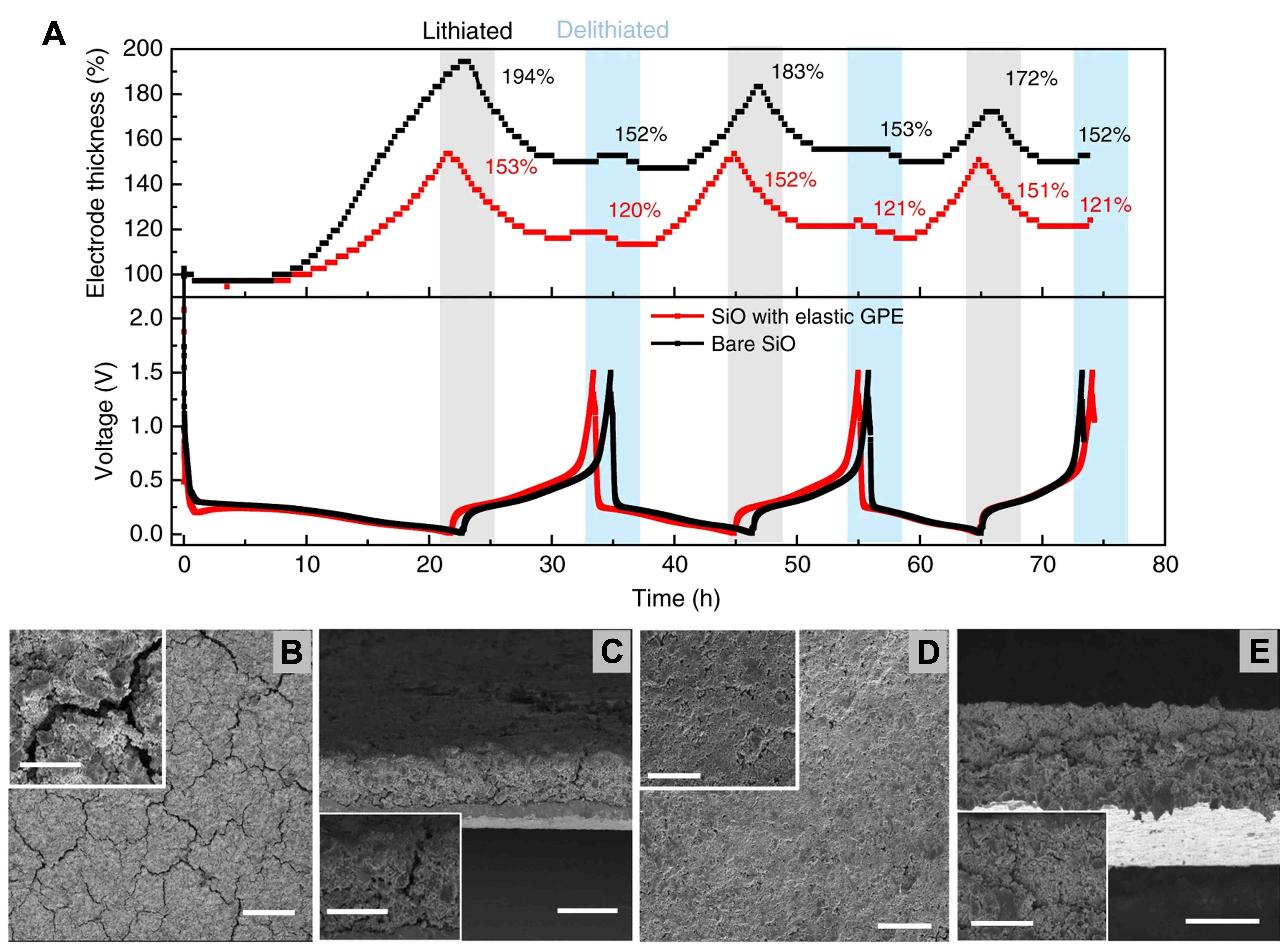
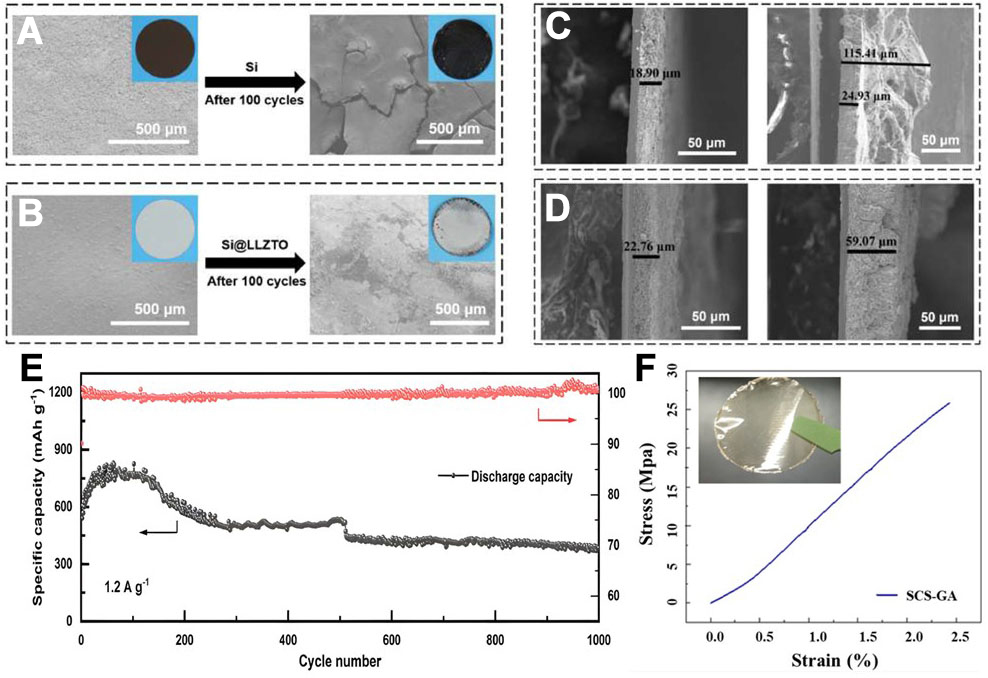
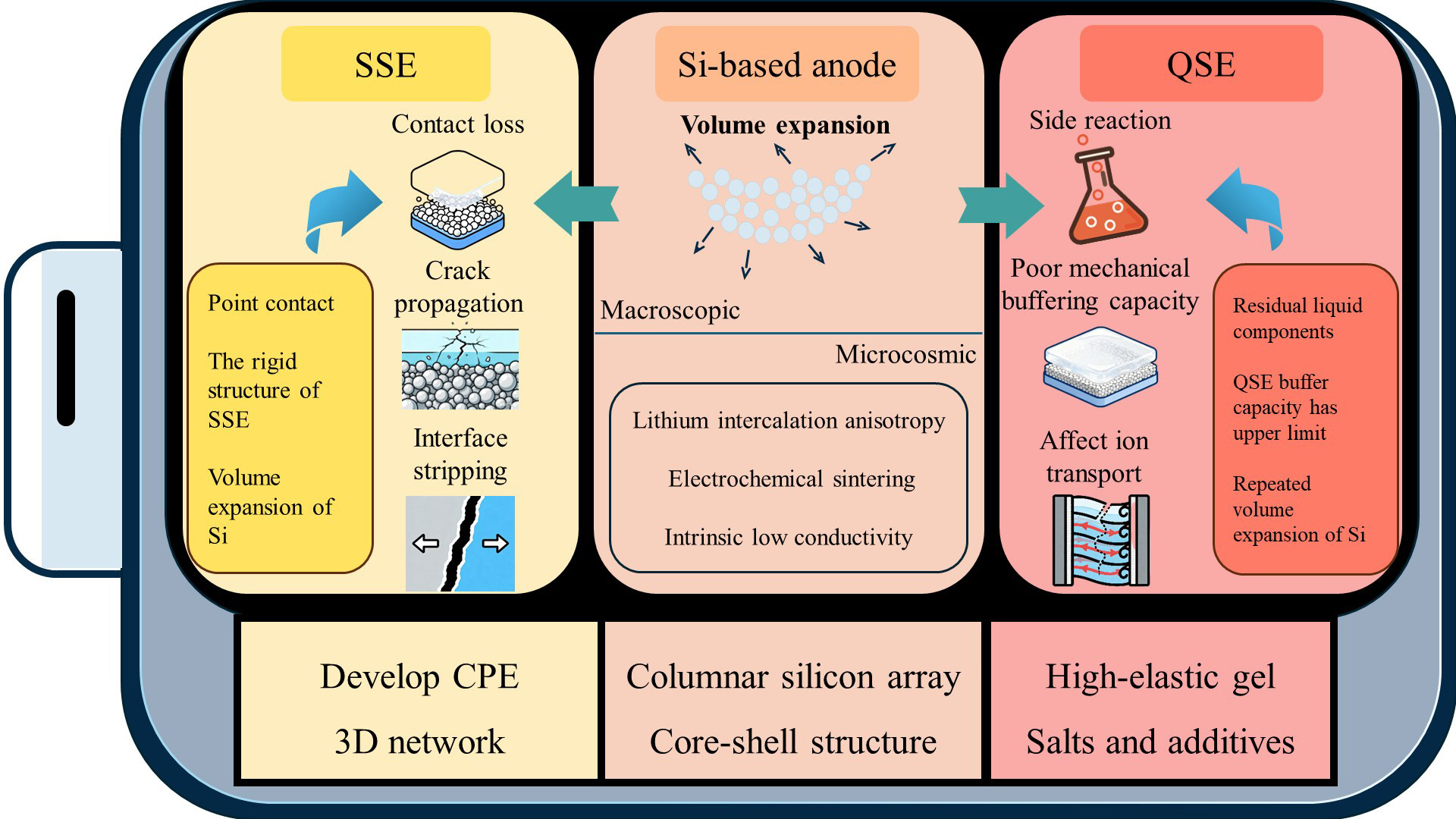












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].