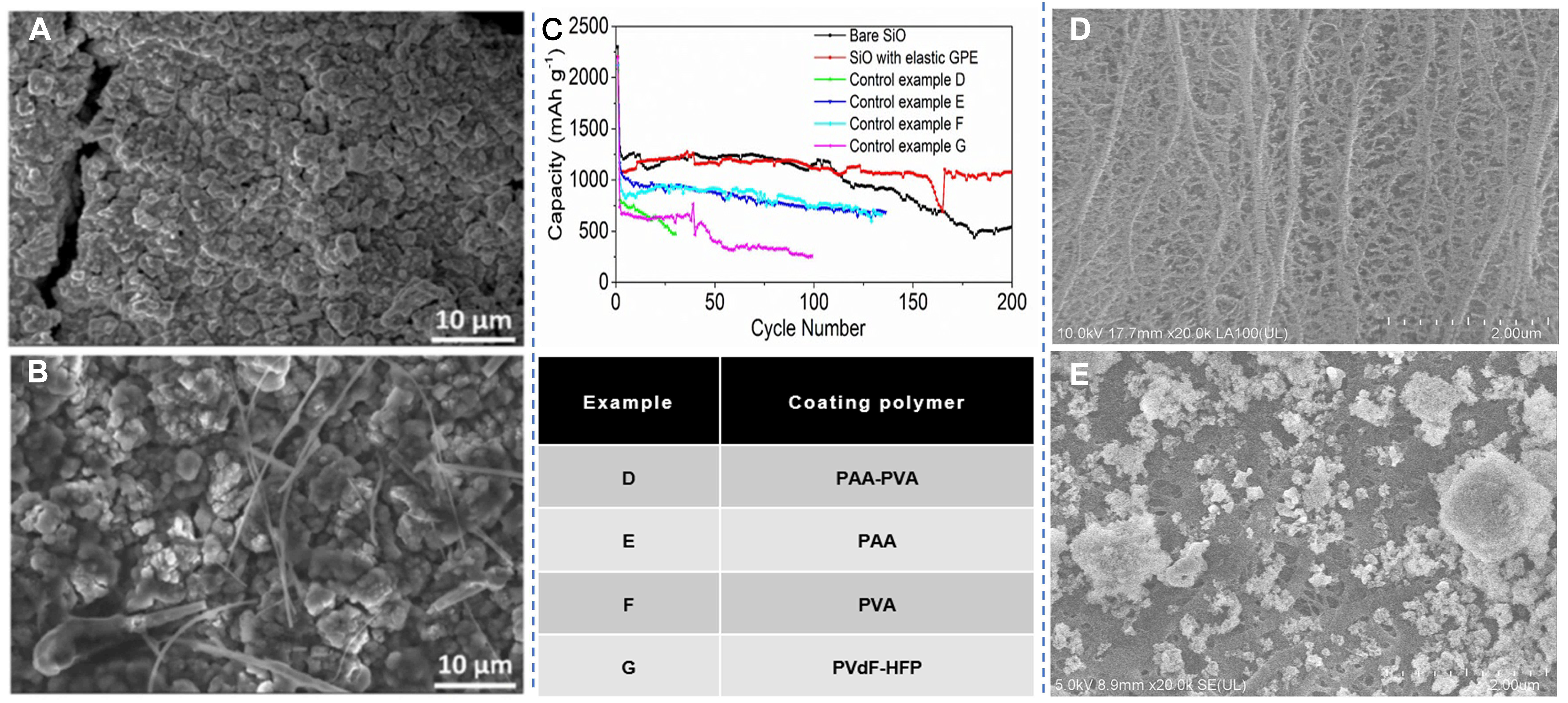
fig6
Figure 6. Polydopamine (PD)@Si morphology evolution and interface chemistry of the electrode after cycling in LE and GE. (A and B) SEM images of the PD@Si electrode surface after cycling in LE and GE. Reproduced with permission from Suebsing et al., Sustainable Energy & Fuels (2025). Copyright © Royal Society of Chemistry[77]; (C) Long-term cycling performance and discharge capacity of the SiO electrode with various polymers. Huang et al., Nature Communications (2019). Copyright © Springer Nature[81]; microscopy images of the punched surface of the original PE (D) and PEAE-0 (E) separators. Xue et al., Colloids and Surfaces A: Physicochemical and Engineering Aspects (2022). Copyright © Elsevier[82]. GPE: Gel polymer electrolyte; LE: liquid electrolyte; GE: gel electrolyte; SEM: scanning electron microscopy; PE: polyethylene; PEAE-0: pure ceramic-coated separators; PAA: polyvinyl alcohol; PVA: polyacrylic acid; PVdF-HFP: polyvinylidene fluoride-hexafluoropropylene.