Recent advances in ionogels for biomedical applications: wound healing, drug delivery, tissue engineering and bioadhesion
Abstract
Ionogels are a class of soft materials comprising a three-dimensional network that immobilizes ionic liquids (ILs). They have drawn considerable attention due to a suite of exceptional and tunable physicochemical properties, such as nonvolatility, excellent thermal and electrochemical stability, adjustable mechanical strength and high ionic conductivity. Building upon these inherent properties and by leveraging the functions of selected ILs, research interest is increasingly shifting toward their biocompatible and biofunctional aspects. This review investigates the recent advances of ionogels in the biomedical field, particularly in the past five years, focusing on antibacterial agents, wound healing, drug delivery, tissue engineering, tumor therapy and bioadhesion. As a cutting-edge interdisciplinary field, ionogels present significant opportunities that require close collaboration among scientists to accelerate the transition from the laboratory to industrialization and bring revolutionary advances to human health.
Keywords
INTRODUCTION
At the intersection of materials science and biology, gel biomaterials serve as dynamic scaffolds[1-4]. They can support soft biological structures ranging from individual cells and skins to entire tissues and organs, while also delivering therapeutic molecules[5-7]. An ideal gel biomaterial must not only exhibit excellent biocompatibility and tunable mechanical properties but also actively interact with biological systems to perform complex functions, such as combating infection[8-12], promoting tissue regeneration[13-15], and enabling controlled drug release[16,17]. While established materials such as hydrogels (which use water as a dispersion medium) and organogels (which use organic solvents) have been widely developed, their application in critical areas such as antibacterial therapy, wound healing, drug delivery, and tissue engineering remains limited[17-22]. These limitations stem from inherent disadvantages, including the tendency of hydrogels to dehydrate and the potential cytotoxicity of the organic solvents in organogels.
Ionogels are another kind of soft gel that confines ionic liquids (ILs) within a three-dimensional (3D) organic or inorganic network. In 2005, the concept of ionogels was first introduced by Susan et al.[23]. Since then, widespread attention worldwide has been drawn to the unique properties inherited from both ILs and 3D networks[24]. The ionogel exhibits a unique combination of properties of excellent ionic conductivity, remarkable thermal and electrochemical stability, strong solubility, non-volatility and loading capacity[25]. Moreover, with a wide selection of available ILs (almost 1018 kinds) and the designability of 3D networks, the physicochemical properties of ionogels can be precisely programmed with different preparation methods[26]. Specifically, the mechanical strength, viscoelasticity, hydrophilicity/hydrophobicity, and even specific functions of the gel (e.g., self-healing, adhesion, antibacterial, and biocompatibility) can be precisely designed and customized for various biomedical applications[27-32].
Given these unique properties of ionogels, they exhibit unprecedented application potential in the biomedical field, particularly in combating infection and promoting tissue repair[33,34]. This review aims to systematically summarize and comment on the latest research progress of ionogel in several closely interconnected areas: antibacterial agents, wound healing, drug delivery, tissue engineering, tumor therapy, and bioadhesion, excluding applications in biosensing and bioelectronics, which have been detailed in other reviews[18,35-37]. In this review, the preparation methods and exceptional properties of ionogels were introduced first [Figure 1]. Then, the latest advances in biomedical applications, particularly in wound healing, drug delivery, tissue engineering, and bioadhesion, are systematically investigated and discussed.
Figure 1. Preparation methods, unique properties, and recent advances of ionogels in biomedical fields[29,38-43]. Copyright 2025, Elsevier B.V; Copyright 2025, Elsevier B.V; Copyright 2025, Wiley-VCH GmbH; Copyright 2023, American Chemical Society; Copyright 2025, Springer Nature Limited; Copyright 2024, American Chemical Society; Copyright 2024, John Wiley & Sons. ES: Electrical stimulation; PTT: photothermal therapy; PES: photothermal electrical stimulation; DES: deep eutectic solvent; TPU: thermoplastic polyurethane.
PREPARATION OF IONOGELS
The fabrication of ionogels is fundamentally based on the formation of a crosslinked polymer network that can effectively confine and disperse an IL. The crosslinking strategy, which defines the network’s architecture, plays a critical role in determining the final properties of the ionogels[20,25,44,45]. Based on the crosslinking mechanisms, ionogels are broadly classified into two categories: physically crosslinked and chemically crosslinked. Physically crosslinked ionogels form through reversible, non-covalent interactions such as hydrogen bonding, electrostatic interactions, and van der Waals forces[5,19]. In contrast, chemically crosslinked ionogels possess permanent networks and superior mechanical strength formed through covalent bonds, which are typically established via chemical reactions or polymerization[46-48]. The selection of an appropriate synthesis method is essential for achieving the desired network structure and functionality. Furthermore, the interactions between the ILs and the polymer matrix are also critical, as ILs can actively participate in and modulate the gelation process through various mechanisms[27,45]. To introduce these interactions, several techniques have been developed, the most common of which are direct mixing, in-situ polymerization/gelation, and solvent exchange[45,49].
Direct mixing
The direct mixing method is one of the most straightforward and versatile strategies for preparing ionogels. This approach involves physically blending ILs with polymeric or inorganic matrices, often facilitated by a co-solvent to ensure homogeneity before its removal[50,51]. The resulting ionogels are typically formed through various non-covalent interactions, including electrostatic forces, hydrogen bonding and physical entanglements, which confine the ILs within a solid network[19,20,52].
This method’s versatility is demonstrated by its application across diverse ionogel types. For polymeric ionogels, a copolymer network was synthesized from behenyl methacrylate and ethyl acrylate via ultraviolet (UV)-induced polymerization [Figure 2A][53]. The semicrystalline dynamic ionogels (SDIGs) were prepared by directly immersing the resulting copolymer film in the IL 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide ([EMIM][TFSI]) for 72 h, allowing the ILs to swell the polymer network, thereby forming a robust ionogel with shape memory behavior. Similarly, Zhu et al. created an adhesive ionogel by simply blending an imidazolium-based IL, 1-methyl-3-butylimidazolium ibuprofenate ([BMIM][Ibu]), with a charged copolymer under the assistance of ethanol[56]. This homogeneous ionogel was formed via electrostatic interactions between the IL and the charged copolymer during mixing, allowing the viscoelastic properties to be finely tuned by adjusting the copolymer composition and the copolymer-to-IL mass ratio. This tuning was crucial for balancing adhesive and cohesive strength to maximize bonding performance across various substrates.
Figure 2. Preparation methods of ionogels. (A) The SDIGs were prepared through direct mixing of the polymer matrix with IL[53]. Copyright 2022, American Chemical Society; (B) The PTIG was synthesized by in situ polymerization under UV light[54]. Copyright 2020, Wiley-VCH GmbH; (C) The click-ionogels were fabricated via solvent exchange under vacuum at 80 °C[55]. Copyright 2019, The American Association for the Advancement of Science. SDIGs: Semicrystalline dynamic ionogels; PTIG: phase-transformable ionogel; UV: ultraviolet; EA: ethyl acrylate; EMIM: 1-ethyl-3-methylimidazolium; TFSI: bis(trifluoromethylsulfonyl)imide; MMITFSI: 1,3-dimethyl imidazolium bis(trifluorosufonylmethane imide); BA: butyl acrylate; FBA: fluorinated butyl acrylate; HDDA: 1,6-hexandiol diacrylate; IL-BF4: 1-propyl-3-methylimidazolium tetrafluoroborate.
In the realm of inorganic ionogels, a remarkably simple physical blending process was demonstrated by mixing fumed silica nanopowder with [EMIM][TFSI], which gelled spontaneously without any chemical reaction[57]. This gelation is primarily driven by hydrogen bonding and other weak intermolecular forces between the silica’s hydroxyl groups and the IL, resulting in an ionogel with outstanding thermal stability[57].
The direct mixing strategy is also applicable to organic-inorganic hybrid and multifunctional systems. Viau et al. developed a novel one-step sol-gel method to fabricate a hybrid ionogel for drug delivery[58]. They used the pharmaceutically active IL [BMIm][Ibu] as a key component during the sol-gel process of silica precursors [i.e., tetramethoxysilane (TMOS) and TMOS/methyltrimethoxysilane]. The gelation formed a silica network that physically confined the IL within its mesopores, achieving a high drug loading of up to 0.80 g of ibuprofen per gram of dried silica, where the release kinetics were governed by the properties of the silica wall. Furthermore, Ye et al. prepared an organic ionogel with unique surface properties via a solution-casting method[59]. They directly mixed binary IL mixtures with solutions of poly(vinylidene fluoride-co-hexafluoropropylene) in acetone. A bicontinuous network was formed through phase separation during solvent evaporation. This structure endowed the ionogel surface with autonomous self-replenishment and durable antibiofouling properties, effectively resisting microbial contamination[59].
The direct mixing method offers significant advantages, such as operational simplicity and wide applicability, as discussed above. However, its effectiveness is often limited by the compatibility and affinity between the IL and the host matrix. Poor compatibility may necessitate a co-solvent and can result in ionogels with limited mechanical strength because they rely on physical interactions.
In situ polymerization/gelation
The in situ polymerization/gelation method represents a pivotal strategy for fabricating ionogels, wherein monomers or gelators are directly polymerized or self-assembled within an IL medium[60]. This approach typically involves either free radical polymerization (thermally or photo-initiated) or the use of non-covalent interactions (e.g., hydrogen bonding, electrostatic forces) and dynamic covalent bonds for network formation[18,48]. A key advantage of this method is the creation of homogeneous polymer-IL composites with finely tuned nanostructures, such as microphase-separated domains or dual networks. These structures are crucial for enhancing mechanical robustness, ionic conductivity, adhesiveness and stimulus responsiveness[61].
This versatility is demonstrated by its application in developing diverse ionogel types with tailored properties. For instance, an ultra-strong adhesive ionogel was fabricated via one-pot photocopolymerization-induced in situ microphase separation[62]. By copolymerizing methyl methacrylate (MMA) and acrylic acid (AA) directly in [EMIM][TFSI], a nanostructure with polymer-rich and IL-rich domains was constructed. This unique architecture, reinforced by hydrogen bonds and ion-dipole interactions, enabled an exceptional lap shear strength above 13 MPa on glass[62]. Alternatively, Wang et al. developed another ultra-robust ionogel by employing an in situ UV-initiated copolymerization of N,N-dimethylacrylamide (DMA) and acrylamide (AM) in IL 1-ethyl-3-methylimidazolium ethyl sulfate, forming soft and rigid segments due to the different binding abilities between[63]. This approach yielded ionogel with remarkable transparency (> 90%), high toughness (> 40 MJ·m-3), and excellent elongation
Besides, poly(ionic liquid) (PIL) is a unique ionogel that is polymerized from IL monomers. PIL-based bioactive microneedles (MNs) were prepared through photocrosslinking of a vinyl-imidazolium IL monomer with hydroxyethyl methacrylate (HEMA), followed by anion exchange to load salicylic acid (SA)[10]. This design resulted in a mechanically robust framework with intrinsic antibacterial properties and sustained drug release capability for treating acne infection. A similar method can also be extended to fabricate organic-inorganic hybrid ionogels. As exemplified by Xu et al., a zinc ion-coordinated PIL membrane was formed via in situ UV copolymerization of IL monomers and subsequent coordination with Zn2+[64]. The resulting dual crosslinking strategy, combining both covalent bonds and metal-ligand coordination, exhibited enhanced antibacterial efficacy against various pathogens, including methicillin-resistant Staphylococcus aureus (MRSA)[64].
Compared to the direct mixing method[53], which primarily relies on physical blending and is often limited by the compatibility between pre-formed networks and ILs, the in situ polymerization/gelation technique offers superior control over the network architecture at the molecular level. This enables the creation of more homogeneous and mechanically robust ionogels with designed functionalities. The in situ polymerization/gelation offers key advantages, including the formation of stable, intricate nanostructures, enhanced mechanical strength, and precise incorporation of functional groups or inorganic components during synthesis. However, it can be more complex and may face challenges, such as potential inhibition of polymerization by certain ILs or the need for careful selection of initiators and reaction conditions.
Solvent exchange
The solvent exchange method is a versatile strategy for preparing ionogels, particularly when monomers are insoluble in ILs or when gelation reactions are incompatible with ILs[65]. This approach generally involves a two-step process: the initial formation of a polymeric network within a compatible solvent to form a hydrogel or organogel, followed by its immersion in a target IL. During this immersion, the solvent is progressively replaced by the IL via diffusion, ultimately yielding the ionogel[45]. This method effectively decouples the network formation from the incorporation of ILs, offering significant flexibility in material design.
The solvent exchange method facilitates a wide spectrum of ionogel designs, from simple polymer networks to systems with sophisticated molecular interactions. A poly(vinyl alcohol) (PVA) organogel was prepared in a mixture of dimethylformamide (DMF) and dimethyl sulfoxide (DMSO), using PVA crystallites as physical crosslinkers[66]. Subsequent solvent exchange with 1-ethyl-3-methylimidazolium dicyanamide ([EMIM][DCA]) IL produced tough, conductive ionogels. Similarly, Ren et al. formed a covalent polymer network via thiol-ene click chemistry in methanol[56]. The methanol was then replaced with 1-propyl-3-methylimidazolium tetrafluoroborate (IL-BF4) IL under vacuum at elevated temperature to fabricate a transparent click-ionogel [Figure 2C].
Moreover, researchers have adopted solvent exchange to introduce specific physicochemical interactions within the gel matrix, thereby enhancing specific properties[67]. For example, Ding et al. utilized the solvent exchange process to lock an IL into a charged poly(2-acrylamido-2-methyl-1-propanesulfonic acid) (PAMPS) double network. By first swelling the PAMPS polymer in water and then replacing water with [EMIM][DCA], the electrostatic interactions are established between the polymer’s sulfonates and the IL cations, successfully immobilizing the IL within a robust matrix while acquiring high conductivity[32]. Besides, solvent exchange can be employed as a trigger for supramolecular assembly. PVA hydrogels were immersed in halometallate ILs. This process replaced the water within the hydrogel, which not only changed the medium but also induced the formation of a robust supramolecular network through coordination and hydrogen bonding, resulting in ultratough ionogels with exceptional mechanical properties[68]. Furthermore, the solvent exchange strategy has been used to fabricate ionogels with good biocompatibility and environmental stability. A notable example is to combine freeze-thaw cycling with solvent exchange, using a biocompatible choline-glycine ([Ch][Gly]) IL. This IL served a dual role as the dispersion medium for cellulose (Cel) and the exchange solvent to reinforce the PVA-based network. This method enhanced polymer cross-linking via a salting-out effect and hydrogen bonding, producing ionogels with anti-swelling, anti-freezing, and biocompatible properties for potential use in flexible biosensors[67].
This progression of studies underscores how solvent exchange has matured from a simple preparation technique into a powerful tool for precise structural, property and functional control in ionogel design. However, despite its utility in processing incompatible polymers, the solvent exchange method is often time-consuming and can cause structural damage during the diffusion process. Furthermore, incomplete solvent removal can compromise the ionogel’s long-term stability.
PROPERTIES OF IONOGELS
Ionogels have emerged as a highly promising material platform in biomedicine, primarily due to their tailorable and multifunctional properties derived from the synergistic combination of a solid network and ILs [Figure 3][5,45]. This unique architecture provides essential mechanical properties for tissue interfacing and durability, as well as advantageous ionic conductivity and stability[1,2,71]. Critically, the IL component can directly impart or enhance key bioactive functionalities, including potent antibacterial activity, intrinsic biocompatibility, and dynamic self-healing capabilities for sustained performance[6,72,73]. Furthermore, engineered adhesive properties enable strong interfacial bonding with biological tissues[1,74]. Together, this combination of mechanical property, self-healing, ionic conductivity, adhesion, biocompatibility and antibacterial property makes ionogels uniquely suited for advanced biomedical applications.
Figure 3. Schematic illustration of the unique properties of ionogels, such as (A) mechanical properties[29]. Copyright 2024, American Chemical Society; (B) self-healing[69]. Copyright 2023, John Wiley & Sons; (C) ionic conductivity[32]. Copyright 2017, WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim; (D) adhesion[29]. Copyright 2024, American Chemical Society; (E) antibacterial[70]. Copyright 2025, Elsevier B.V; (F) biocompatibility[29]. Copyright 2024, American Chemical Society.
Mechanical properties
The mechanical properties of ionogels, such as fracture strength, fracture strain, Young’s modulus, fatigue resistance, and crack propagation resistance, are essential for their biomedical applications (e.g., wound healing[33], tissue engineering[75], bioadhesion[29]). These properties enable ionogels to adapt to soft tissue structure, protect wounds from mechanical impacts, and maintain durability under cyclic loading[6,45,76,77]. These mechanical characteristics are primarily governed by ILs and the polymer matrix. Specifically, ILs modulate the intermolecular interactions within the polymer network[52,78,79], while the matrix determines the structural frameworks. For example, PILs can form hydrogen bonds for stabilization, and metal-organic frameworks (MOFs) can serve as stress-transfer centers[19,46,80,81].
To address the demand for high stretchability and toughness in biomedical scenarios involving cyclic loading (e.g., tissue engineering scaffolds), Xia et al. developed a highly stretchable ionogel by coordinating DMA polymer chains with UiO-66, a zirconium-based MOF, in 1-(2-carboxyethyl)-3-methylimidazolium bis(trifluoromethanesulfonyl)imide ([CMMIM][TFSI]) IL[46]. The key molecular design relies on reversible cross-links formed between the amide groups of DMA chains and zirconium metal sites of UiO-66; these cross-links, together with hydrogen bonds between the IL and polymer matrix, enable efficient energy dissipation during deformation. This dynamic network structure endowed the ionogel with exceptional mechanical properties (an ultrahigh fracture strain of 11,000%, a Young’s modulus of 58 MPa, and a toughness of 25 MJ·m-3). Notably, the UiO-66 MOF nanoparticles not only reinforced the network but also significantly enhanced fracture energy (125 kJ·m-2) and fatigue resistance, allowing the ionogel to sustain over 20,000 compression cycles without obvious performance degradation. Such robust mechanical performance, coupled with recyclability, makes this ionogel a promising candidate for durable biomedical devices requiring long-term resistance to cyclic mechanical stress[46].
Apart from the abovementioned nanocomposite strategy, bio-based materials were adopted to develop ionogels that balance mechanical strength, biodegradability, and temperature stability, which are key requirements for transient biomedical applications (e.g., biodegradable wound dressings)[82]. This ionogel was composed of Cel and soybean protein (SP) dissolved in 1-butyl-3-methylimidazolium chloride ([BMIM]Cl) IL. The preparation process involved thermal dissolution of Cel and SP in [BMIM]Cl to disrupt their native hydrogen bonds, followed by acetone stimulation. During this step, a reinforced Cel-protein gel with an entangled supramolecular network was formed, in which SP molecules wound around Cel chains via reconstituted hydrogen bonds. The [BMIM]Cl IL played a critical role in facilitating molecular dispersion of Cel and SP, while the entangled supramolecular network impeded chain relaxation and dissociation, directly enhancing mechanical strength and durability. The resulting ionogel exhibited exceptional mechanical properties: a tensile strength of 30.7 MPa, an elongation at break over 160%, and an elastic modulus of
For applications requiring ultrathin structures, such as transdermal drug delivery systems (TDDS) that need to conform to skin surfaces, Zhang et al. developed an ultrathin ionogel by integrating a thermoplastic polyurethane (TPU) nanonet with a polymerized network of 2,2,3,4,4,4-hexafluorobutyl acrylate and 2-hydroxyethyl acrylate (HFBA-HEA) in the presence of the hydrophobic IL [EMIM][TFSI][83]. The fabrication process involved electrostatic spinning of the TPU nanonet (serving as a reinforcing framework), followed by drop-coating of the HFBA-HEA precursor solution and UV-induced polymerization. Despite a remarkable thickness of only ≈ 16.7 µm, the TPU nanonet-supported ionogel exhibited excellent mechanical performance, including a high tensile strength of 2.78 MPa, an elongation at break of 375%, and an outstanding toughness of 5.51 MJ·m-3. The synergistic effect between the robust TPU nanonet (providing structural integrity) and dynamic non-covalent interactions within the ionogel matrix (e.g., ionic-dipolar interactions between [EMIM]+ cations and -CF3 groups of the fluoropolymer) contributed to excellent fatigue resistance, enabling the ionogel to endure over 3,000 cycles at 100% strain without significant degradation. Importantly, this ultrathin ionogel demonstrated promising biomedical applicability as a carrier in TDDS for the sustained release of propranolol. It exhibited good biosafety and skin cytocompatibility, thus bridging its superior mechanical performance with practical biofunctionalities required for biomedical translation[83].
Self-healing
The self-healing capability of ionogels is critically important for enhancing the durability, reliability, and lifespan of biomedical devices such as wound dressings, drug delivery systems (DDS), and tissue engineering scaffolds[6,24,84]. This property allows ionogels to autonomously repair damage, maintain structural integrity during use, and prevent secondary injuries, thereby ensuring prolonged and effective performance in dynamic biological environments[85,86]. The self-healing behavior primarily originates from the reversible interactions within the ionogel’s network, which can be tailored through its composition. These include dynamic covalent bonds (e.g., disulfide bonds, imine bonds) and non-covalent interactions (e.g., hydrogen bonds, ion–dipole interactions), often facilitated by the unique properties of ILs that can act as plasticizers or mediate reversible interactions to promote chain mobility and network reformation[18].
A typical example of self-healing ionogels based on dynamic covalent and noncovalent synergy is the fluorescent double-network (DN) ionogel developed by Zhao et al.[87]. They fabricated this ionogel via one-pot photopolymerization, which integrates a covalently cross-linked polyacrylamide (PAM) network and a dynamic network composed of amino-modified agarose (AG-NH2) and 1,3,5-benzenetricarboxaldehyde (BTC) in the IL 1-ethyl-3-methylimidazolium chloride ([EMIM]Cl)[87]. The self-healability of this ionogel originates from dynamic imine bonds (formed between AG-NH2 and BTC) and abundant hydrogen bonds; notably, [EMIM]Cl acts as a dispersion medium to facilitate the formation of a homogeneous precursor solution, laying a foundation for uniform cross-link reformation during healing. This design enables the ionogel to achieve autonomous self-healing within 10 min at room temperature and rapidly restore ionic conductivity after repair. Additionally, the DN structure endows the ionogel with good mechanical properties, along with high transparency (> 95%), wide temperature tolerance (-96 to 260 °C), and fluorescence, which collectively support its potential in biomedical scenarios requiring stable performance under varied conditions[87].
Beyond DN design, the regulation of noncovalent interactions and IL functions also contributes to high-performance self-healing ionogels, as demonstrated by the spider-silk-inspired ionogel reported by Sun et al.[73]. They prepared this ionogel through one-pot photopolymerization of N-isopropylacrylamide (NIPAM) and DMA in the ionic liquid [EMIM][TFSI] without chemical cross-linkers[73]. Its self-healing capability is enabled by multilevel dynamic hydrogen bonding interactions (between poly(NIPAM) (PNIPAM) chains, poly(DMA) (PDMA) chains, and [EMIM][TFSI]). Here, [EMIM][TFSI] plays a dual role: as a plasticizer, it enhances the mobility of polymer chains, thereby facilitating rapid bond recombination across damaged interfaces. This ionogel achieves autonomous self-healing at room temperature within 24 h, recovering over 90% of its original tensile strength and toughness, while its electrical conductivity restores 99% within just 10 s. Moreover, it exhibits high transparency (> 90%), mechanical robustness (0.59 MPa tensile strength, 916% elongation), and processability via melt-spinning and 3D printing, which are features that are particularly valuable for fabricating customized biomedical devices (e.g., personalized wound dressings or tissue scaffolds) with complex structures[73].
Furthermore, the extension of self-healing design from single ionogels to composite bionic systems broadens their biomedical application potential, as shown in the bionic artificial skin reported by Zhang et al.[88]. They designed this system comprising a 3D-printed rigid skeleton (composed of disulfide-containing polyurethane acrylate and methacrylate monomers) and a soft ionogel matrix (based on polyurethane acrylate and the ionic liquid [EMIM][TFSI]), integrated via direct ink writing and co-curing[88]. The self-healability of the composite originates from dynamic disulfide bond exchange in both the skeleton and the ionogel matrix: the ionogel matrix heals at 80 °C for 6 h (tensile strength efficiency: 87.7% ± 5.0%), the skeleton heals at 100 °C for 24 h (tensile strength efficiency: 88.7% ± 3.0%), and the entire composite shows a 69.8% strength healing efficiency. To ensure stability in biomedical use, the IL-phobic skeleton prevents IL swelling, while the co-cured interface enhances the durability of the composite. Additionally, the system exhibits skin-like mechanical anisotropy, which is an essential property for mimicking biological tissues in applications such as artificial skin for wound coverage or tissue repair[88].
Ionic conductivity
Ionic conductivity is a pivotal property of ionogels that significantly influences their efficacy in various biomedical applications, such as electrically stimulated drug delivery and wound healing[5,18,45,89]. This conductivity primarily originates from the mobile ions provided by the ILs encapsulated within the polymer network. The intrinsic ionic conductivity of an IL depends on its chemical structure, including the size and viscosity of its constituent ions. Generally, ILs composed of smaller anions (e.g., tetrafluoroborate [BF4]- and dicyanamide [DCA]-) and cations with shorter alkyl chains exhibit lower viscosity and higher ionic conductivity[27,32,90,91]. The polymer matrix also plays a critical role, as its interactions with the ILs, the pore size and volume of its cross-linked network, and its own chemical nature collectively govern ion transport[92,93]. However, a critical balance must be struck, as excessively high IL content, while boosting conductivity, can lead to IL exudation and compromised mechanical strength[94-97].
Ding et al. developed a transparent ionogel based on [EMIM][DCA] and PAMPS-based DN[32]. Fabricated by exchanging water with [EMIM][DCA] after PAMPS DN synthesis, its conductivity enhancement stems from high intrinsic conductivity of [EMIM][DCA] (2.8 S·m-1 at 25 °C) and free ionic transport, combined with electrostatic locking and tunable micropores [via 2-acrylamido-2-methylpropanesulfonic acid (AMPS) concentration] of the DN. The conductivity of the ionogel is up to 1.9 S·m-1 at 25 °C, increasing with temperature and stable for 30 days. It exhibits excellent transparency, good mechanical strength, and harsh-condition stability, suitable for flexible skin sensors to monitor human motions[32].
Based on the strategy of integrating IL intrinsic conductivity with matrix network regulation, Su et al. further optimized ionogels for biomedical adaptability by employing biocompatible components[98]. They developed composite ionogels (BC/PILs) based on choline-amino acid polyionic liquids ([Cho][AA] PILs) and bacterial cellulose (BC) via ex situ method. Conductivity enhancement relies on ionic transport from Cho cations and AA anions in PILs, with higher vinyl Cho-AA ionic liquid monomer (VIL) concentration (up to 1.6 mol·L-1) and hydrophilic/ionizable groups in PILs facilitating ion mobility. The ionogels exhibit a conductivity range of 0.093 to 1.31 S·m-1, which aligns with that of human skin and tissues. More importantly, they possess excellent antimicrobial activity and high biocompatibility. These key merits for biomedical applications collectively make them promising candidates for biomedical wound dressings[98].
Taking a step further toward multifunctional integration for biomedical scenarios, Zhou et al. developed sustainable and antibacterial ionogels that balance ionic conductivity with mechanical robustness and biological activity[74]. They integrated cellulose acetate (CA) into a poly(acrylic acid) (PAA) network containing [BMIM]Cl IL via free-radical polymerization. The core mechanism for ionic conductivity enhancement relies on CA-[BMIM]Cl interactions: CA’s hydroxyl groups form ordered hydrogen-bonded networks with Cl- to provide ion transport channels, CA competes with PAA for hydrogen bonds to weaken PAA-Cl- binding (releasing free Cl-), and [BMIM]+ approaches CA chains via electrostatic interactions to further assist Cl- migration. With CA addition, its ionic conductivity increased by 373% from 0.76 to
Adhesion
Adhesive capability is crucial for ionogels in biomedical applications such as wound dressings and tissue adhesives, enabling reliable attachment to biological tissues. This property stems from the ability of IL cations and anions to form multiple noncovalent bonds (e.g., hydrogen bonds, ion-dipole interactions) with substrates[29,79,99]. Effective adhesion depends not only on interfacial interactions but also on the ionogel’s viscoelasticity for stress distribution. Consequently, improving adhesion performance primarily involves optimizing interfacial interactions and designing dynamic networks[65,100,101].
Rational design strategies have been successfully illustrated to enable robust adhesion in ionogels. Cheng
Advancing from strategies that utilize multiple interfacial interactions, researchers have introduced dynamic bonds to improve environmental adaptability. Pan et al. developed eco-friendly ionogels by crosslinking sodium alginate (SA)/PVA networks with Mg2+ and boron in a deep eutectic solvent (DES) system[103]. The adhesion enhancement primarily stems from boron complexing with polyhydroxyl groups in SA and PVA to form dynamic “cis-diol” structures, which intensify interfacial hydrogen bonding and van der Waals interactions with substrates. Replacing traditional Ca2+ with Mg2+ further reduced gel brittleness and improved flexibility. The resulting Mg/B-crosslinked sodium alginate/polyvinyl alcohol (Mg/B-SA/PVA) ionogel achieved an ultra-high adhesion strength of 548.25 kPa on glass and strong bonding to diverse substrates such as rubber, fabric, and skin. The DES solvent also contributed to conductivity (0.82 S·m-1) and mechanical toughness (513% elongation, 0.60 MPa strength), highlighting its potential in wearable and biomedical applications such as skin-adherent biosensors.
Furthermore, achieving extreme adhesion requires a critical balance between internal cohesion and interfacial adhesion. Xiong et al. developed a supramolecular ionogel by in situ photo-cross-linking of DMA in a biocompatible IL composed of citric acid and L-(-)-carnitine (CAC)[29]. CAC’s carboxyl/hydroxyl groups form strong H-bonds with substrates, balancing cohesive/interfacial energies to boost adhesion (24.4 MPa on glass; 191 J·m-2 on wet tissues). It exhibits high mechanical strength, can be detached by saline treatment, and is promising for wound adhesion and organ transplantation. This progression illustrates a strategic shift: initially harnessing multiple interfacial interactions, then incorporating dynamic coordination bonds for environmental adaptability, and finally achieving an optimal balance between internal cohesion and interfacial adhesion for advanced biomedical applications.
Antibacterial property
In the field of biomedical applications, bacterial infections pose a severe threat to wound healing, tissue repair, and medical device safety. Consequently, developing materials with efficient and biocompatible antibacterial properties has become a critical research focus[36,104-106]. As a class of advanced soft materials, ionogels consist of ILs immobilized in a polymeric matrix. Their outstanding antibacterial capabilities are directly linked to their unique composition, specifically the type of ILs (including cationic, anionic, or PILs) and the structure of the polymeric network[5,24,107,108]. The core antibacterial mechanism of ionogels is primarily attributed to two synergistic effects: (i) the electrostatic interaction between the positively charged moieties (e.g., imidazolium, pyridinium, pyrrolidinium, or quaternary ammonium cations) of ILs and the negatively charged phosphate groups on bacterial cell membranes, which increases membrane permeability and causes leakage of cytoplasmic contents[109]; and (ii) the insertion of hydrophobic alkyl chains of ILs into the lipid bilayer of bacterial membranes, leading to membrane perforation, disorganization, and ultimately bacterial cell death[110-113]. Additionally, the antibacterial activity of ionogels can be modulated by several factors, such as the alkyl chain length of ILs (where longer alkyl chains generally enhance surface activity and membrane disruption), the cation charge density, and the presence of synergistic components (e.g., antibiotics or antibacterial nanoparticles). Crucially, favorable biocompatibility toward normal eukaryotic cells is a key prerequisite for their biomedical application[24,112,114-117].
The antibacterial performance of ionogels is first rooted in the intrinsic regulation of their IL-based components, and optimizing the interaction between cations and anions has become a core strategy to enhance efficacy. Guo et al. fabricated free-standing PIL membranes via in-situ photo-cross-linking of an IL monomer followed by anion exchange with amino acids[115]. The antibacterial mechanism involves synergistic effects where imidazolium cations electrostatically interact with bacterial cell walls while hydrophobic segments [e.g., from exchanged L-tryptophan (Trp)- anions] penetrate lipid membranes, with PIL-Trp membranes showing superior efficacy against both Escherichia coli (E. coli) and Staphylococcus aureus
Based on the intrinsic antibacterial mechanism of ILs, researchers have further explored the integration of ionogels with other functional components to achieve enhanced and diversified antibacterial effects. Singh
Building upon optimized antibacterial mechanisms and enhanced functional integration, antibacterial ionogels have been expanded to meet the demands of different biomedical scenarios. For example, Zheng
Beyond direct medical applications such as wound dressings and medical devices, antibacterial ionogels have shown promise in related fields such as food safety, leveraging their efficient and safe antibacterial properties. An antibacterial ionogel film was developed by incorporating gelatin/carboxymethyl cellulose (CMC) based polymeric film with a synthesized imidazolium-based IL[28]. The concentration-dependent antibacterial activity primarily stemmed from the IL’s imidazolium cations that electrostatically interacted with bacterial cell walls, and the long alkyl chains disrupted the cell membrane integrity. The film with 20% IL content exhibited potent efficacy against E. coli and S. aureus with low minimum inhibitory concentration (MIC) values and significantly prolonged the shelf life of red apples, demonstrating its potential as an active food packaging material.
Biocompatibility
Biocompatibility is fundamental for ionogels in biomedical applications such as wound dressings, implantable devices, and DDS, where direct contact with biological tissues or skin is involved. This property is critically influenced by the chemical structure of the constituent ILs[72,89,120]. Generally, cations with longer alkyl chains are more cytotoxic due to their increased lipophilicity, which can disrupt lipid bilayers. In contrast, cations such as cholinium, derived from natural sources, often demonstrate enhanced biocompatibility. Therefore, a key strategy for developing biosafe ionogels is to select ILs composed of biocompatible ions, such as cholinium cations paired with amino acid or carboxylate anions[5,121,122].
Several studies have exemplified how the rational design of ionogel composition can achieve outstanding biocompatibility. Le et al. prepared a chloride-based ionogel via evaporation-induced polymerization of thioctic acid (TA) and the ionic liquid [BMIM]Cl in ethanol[31]. The biocompatibility of the resulting ionogel-coated fabric was demonstrated, as cell proliferation and viability tests with human dermal fibroblast cells showed performance comparable to the control sample and even better than a standard reference gel electrolyte material over 24 h. The inherent biocompatibility of the [BMIM]Cl ionic liquid, which has been utilized as a stabilizer for proteins and enzymes in biopharmaceuticals, likely contributes to this favorable cellular response. This excellent biocompatibility is supported by the dynamic supramolecular polymer network formed through disulfide bonds and hydrogen bonds.
Building upon this foundation of utilizing ILs with inherent biological safety, Shen et al. developed a metal-phenolic DN ionogel (MP-DN ionogel) via a facile one-pot method using Fe3+-tannic acid and H2O2 as a dual self-catalysis system to copolymerize hydrophilic ionic liquid monomer 1-butyl-3-vinylimidazolium chloride ([BVIM]Cl) and hydrophobic glycidyl methacrylate (GMA)[123]. The ionogel demonstrated good biocompatibility, as evidenced by a cell viability exceeding 80% at concentrations up to 7.5 mg·mL-1 and a significant enhancement in HaCaT cell migration, which is crucial for wound healing and was attributed to the unique properties of the polyphenol components. Histological evaluation of rat heart, liver, spleen, lung, kidney, and skin tissues, along with hemolysis ratios below 5%, confirmed excellent tissue and blood compatibility. The multi-layer wrinkle structure resulting from the phase separation between the hydrophobic poly(glycidyl methacrylate) (polyGMA) and hydrophilic polymeric IL networks likely contributed to a favorable microenvironment for cell interactions.
Researchers have engineered ionogels where high biocompatibility is seamlessly integrated with other advanced functionalities, such as tunable biological activity. Zhang et al. synthesized a series of ferrocene-containing imidazolium-based PIL membranes via esterification and quaternization reactions[124]. These membranes exhibited excellent biocompatibility, demonstrating extremely low hemolytic activity
APPLICATION OF IONOGELS
Ionogels as antibacterial agents
The escalating global health challenge of antimicrobial resistance (AMR) necessitates the development of novel non-antibiotic strategies. Ionogels, which incorporate ILs into a polymer network, have emerged as promising antimicrobial agents for biomedical applications such as wound dressings and antibacterial coatings[24,125,126]. Their inherent antibacterial properties primarily stem from the cationic moieties and hydrophobic alkyl chains of the ILs, which can disrupt bacterial membranes[112,116], as discussed in Section “Antibacterial property”. However, a major challenge is to mitigate the potential cytotoxicity of ILs while maintaining their antimicrobial efficacy. The composition of ionogels, particularly the choice of polymer matrix and IL, is therefore important. This composition directly influences mechanical properties, biocompatibility, and antimicrobial performance, making ionogels a versatile platform for designing effective and biosafe antimicrobial materials[1,6,24,127].
The constant skin contact of wearables, especially these ionogel sensors and flexible bioelectronics, creates an ideal environment for microbial growth. By incorporating advanced antibacterial properties, these wearable ionogels now combat this hidden threat, making user protection as important as data collection[38,74,119]. The aforementioned PIL-based multilayer nanofiber membrane with antibacterial properties made from a spinning process[119], was used as an electronic skin to collect the bioelectrical signals such as the electrocardiography and electromyography. Zhou et al. developed sustainable ionogels through UV-induced copolymerization of CA, AA, and ionic liquid [BMIM]Cl [Figure 4A][74]. The resulting ionogels demonstrated integrated mechanical robustness (tensile strength of 168.9 kPa, elongation of 1,250.7%) and high ionic conductivity (3.62 mS·cm-1). Notably, these materials exhibited effective antibacterial properties against both Gram-positive S. aureus and Gram-negative E. coli. This activity is attributed to the [BMIM]+ cations, which disrupt bacterial membranes through electrostatic interactions, leading to the leakage of intracellular components. Confocal laser scanning microscopy confirmed markedly reduced viability of both bacterial strains after contact with the ionogels. Furthermore, the materials maintained good cytocompatibility with cell viability exceeding 70%, making them suitable candidates for wearable sensors. Jiang et al. developed transparent and flexible ionogels by incorporating the ionic liquid [EMIM][TFSI] into a TPU matrix [Figure 4B(i)][38]. These ionogels exhibited significant antibacterial activity against both
Figure 4. The ionogels were used as antibacterial agents. (A) Wearable ionogels with antibacterial effects. (i) Molecular structure of raw materials. (ii) Schematic illustration of interwoven networks of different samples[74]. Copyright 2025, Elsevier B.V; (B) Transparent antibacterial TPU@IL ionogels. (i) Optical transparency and schematic structure. (ii) Photos and measured diameters of the inhibition zones of TPU@IL ionogels with different mass ratios for E. coli and S. aureus[38]. Copyright 2021, Elsevier B.V; (C) A fully bio-based bionogel. (i) The formulation of bionogels reinforced with chitosan. (ii) Diameters of inhibition obtained from the antibacterial tests for S. aureus and E. coli[128]. Copyright 2019, Elsevier Ltd; (D) pH-responsive charge conversion feature of HPIL-Glu with high antimicrobial activity. (i) Schematic illustration of the responsiveness. (ii) Concentration-dependent bactericidal effect of HPIL-Br, HPIL-Glu, and erythromycin against planktonic MRSA bacteria. (iii) Concentration-dependent MRSA biofilm eradication effect of HPIL-Br, HPIL-Glu, and erythromycin[129]. Copyright 2024, American Chemical Society. TPU: Thermoplastic polyurethane; IL: ionic liquid; E. coli: Escherichia coli; S. aureus: Staphylococcus aureus; HPIL-Glu: Glu-decorated hyperbranched poly(ionic liquid); HPIL-Br: quaternary ammonium bromide-tethered HPIL; MRSA: methicillin-resistant Staphylococcus aureus; PAA: poly(acrylic acid); CA: cellulose acetate; MBAA: N, N′-methylenebis (acrylamide); PAA-BM: ionogels made by acrylic acid and [BMIM]Cl ionic liquid; PAA-BM-CA: ionogels made by acrylic acid, [BMIM]Cl ionic liquid and cellulose acetate; DD: degree of deacetylation; RT: room temperature.
To improve the biocompatibility while maintaining the antimicrobial efficacy of ionogels, biomass polymers or naturally derived ILs with inherent antibacterial properties are increasingly attracting attention. A fully bio-based bionogel was prepared by using the bio-ionic liquid cholinium lysinate and Cel as the matrix, which was reinforced with different loadings of chitosan (0.25-1 wt%)[128]. The resulting materials were physically weak gels crosslinked via hydrogen bonds, exhibiting enhanced viscoelastic properties with increasing chitosan content [Figure 4C(i)]. Due to the inherent antibacterial property of chitosan, primarily attributed to its amino groups, the bionogels showed significant inhibitory effects against both S. aureus and E. coli bacteria, with a notably higher efficiency against E. coli (minimum inhibition zones of 4 mm for
The use of natural polymers in bio-based ionogels improves biocompatibility, but their antibacterial efficacy against acidic and robust biofilms remains a significant challenge. Lin et al. developed a biocompatible Glu-decorated hyperbranched poly(ionic liquid) (HPIL-Glu) nanoassembly, synthesized from a hyperbranched polyurea core modified with charge-convertible ammonium-based ILs and gluconate (Glu) counteranions, forming homogeneous spherical nanostructures in water [Figure 4D(i)][129]. This material exhibits pH-responsive charge conversion, where Glu shields cationic moieties under neutral conditions to minimize toxicity, but protonates to expose cationic groups for antimicrobial action in acidic environments (e.g., within biofilms). HPIL-Glu demonstrated effective concentration-dependent bactericidal activity against planktonic bacteria such as E. coli, S. aureus and MRSA [Figure 4D(ii)] with a notable reduction in bacterial viability observed at concentrations above 32 μg/mL. HPIL-Glu also achieved antibiofilm efficacy comparable to HPIL-Br, effectively eradicating mature biofilms at similar concentrations [Figure 4D(iii)]. This activity is attributed to cationic disruption of bacterial membranes. In vivo, HPIL-Glu promoted the healing of bacteria-infected chronic wounds without inducing AMR. Thus, HPIL-Glu represents a promising resistance-free antibacterial agent for medical applications such as wound dressings.
Beyond the threats posed by conventional and drug-resistant bacteria, fungi constitute another critical class of pathogens requiring effective antimicrobial strategies. Su et al. developed a series of choline-amino acid-based PIL and ionogels composed of BC and PIL via an ex situ method, where vinyl-functionalized IL monomers were polymerized within the BC network using ammonium persulfate (APS) as an initiator[98]. The resulting ionogels exhibited broad-spectrum antimicrobial activity against Gram-negative E. coli, Gram-positive S. aureus, and fungi Cryptococcus neoformans (C. neoformans), primarily attributed to the hydrophobic interactions between the amino acid anions of the PILs and microbial cell membranes, leading to membrane disruption. Notably, D-phenylalanine-based PIL demonstrated the strongest efficacy, with MIC and minimum bactericidal concentration (MBC) values as low as 32 ± 1.6 and 64 ± 3.2 mmol·L-1, achieving the fastest bacterial elimination within 2 h. Ionogels with higher PIL content showed enhanced antimicrobial performance, forming inhibition zones up to 40 ± 2.0 mm against E. coli, while maintaining excellent biocompatibility (hemolysis rate < 5% and cell viability > 80%), highlighting their potential as advanced wound dressings[98].
Application of ionogels in wound healing
Ionogels have emerged as promising biomaterials for wound healing, effectively addressing critical challenges such as bacterial infection, persistent inflammation, and low healing efficiency[2,5,127,130-132]. Unlike traditional dressings, which often suffer from poor mechanical properties, limited antibacterial efficacy, and an inability to modulate the wound microenvironment, ionogels uniquely combine the antimicrobial activity, biocompatibility, and stimuli-responsive properties of ILs with the mechanical robustness of the polymer networks, thereby offering tailored solutions for advanced wound management[2,77,127,133].
Ionogel dressings for wound healing
Electrospun nanofibrous membranes serve as an excellent platform for developing ionogel-based wound dressings. Luo et al. developed electrospun PIL/cerium (PIL-Ce) nanofibrous membranes, which demonstrated remarkable efficacy in treating MRSA-infected wounds [Figure 5A(i)][33]. This membrane functions through a synergistic antibacterial mechanism: the imidazolium cations directly disrupt bacterial membranes, while Ce4+ ions catalyze the degradation of antibiotic resistance genes (ARGs). These membranes completely eradicated MRSA within 4 h in mouse wound models while effectively blocking the drug-resistance dissemination by degrading the mecA ARGs [Figure 5A(ii) and (iii)]. By achieving rapid wound disinfection and preventing the spread of resistance genes, these membranes show significant potential as advanced dressings for managing drug-resistant bacterial infections[33].
Figure 5. The application of ionogel in wound healing. (A) PIL-Ce -based nanofibrous membranes for treating MRSA-infected wounds. (i) Schematic illustration of the synthesis and its application for bacterial killing and ARGs decomposition. (ii) Bacterial viabilities of MRSA after contacting with PIL and PIL-Ce membranes for 4 h. (iii) Representative bioluminescent images taken at 0 and 4 h after bioluminescent MRSA inoculation and treatment with PIL-Ce[33]. Copyright 2021, Wiley-VCH GmbH; (B) DES based ionogel for electrical stimulation dressing. (i) Preparation of DES ionogel. (ii) Wound healing in rats treated with DES ionogel and electrical stimulation. (iii) Representative wound images at days 0 and 14[39]. Copyright 2025, Elsevier B.V; (C) Mxene-based ionogel patch with PES effect. (i) Schematic diagram of the design and PES effect. (ii) Wound model established and wound healing process after treatments within 9 days. (iii) The statistics of wound area under different treatments on different days. (iv) Wound contraction degrees of PES group during the treatment[8]. Copyright 2023, Elsevier B.V; (D) The PILMN-Chl for diabetic wound healing. (i) Schematic of the synergistic wound healing process, where Chlorella supplies oxygen to the wound bed under light and PIL provides sterilization. (ii) SEM image of PILMN-Chl, optical photos of a dye labeled PILMN and corresponding pinholes left inside the skin after inserting the dyed MNs. (iii) Wound closure images of PILMN-Chl over 9 days (scale bar: 3 mm). (iv) Oxygen produced by PILMN-Chl in PBS under light (5500 lux)[134]. Copyright 2024, Wiley-VCH GmbH. PIL-Ce: Poly(ionic liquid)/cerium; MRSA: methicillin-resistant Staphylococcus aureus; ARGs: antibiotic resistance genes; DES: deep eutectic solvent; PES: photothermal electrical stimulation; PILMN-Chl: Chlorella-loaded PIL-based microneedles; SEM: scanning electron microscopy; MNs: microneedles; PBS: phosphate buffered saline; AA: acrylic acid; AAM: acrylamide; S. aureus: Staphylococcus aureus; EMIES: 1-ethyl-3-methylimidazole; ATP: adenosine triphosphate; FGF-2: fibroblast growth factor-2; MMP: mitochondrial transmembrane potential; PTS: photothermal stimulation; UV: ultraviolet; E.coli: Escherichia coli.
Smart ionogel dressings with integrated multifunctionality are garnering increasing attention for chronic wound management. Yang et al. developed an all-in-one wound dressing based on a water-free ionogel made from a DES[39]. This ionogel, synthesized via UV polymerization of N-Hydroxyethyl acrylamide (HEAA) in a choline chloride (ChCl)/ethylene glycol (EG) DES and incorporated with Zn2+ ions, exhibited robust antibacterial efficacy against both E. coli and S. aureus [Figure 5B(ii)]. Furthermore, its inherent conductivity enabled localized electrical stimulation (ES), which significantly accelerated in vivo wound healing in a rat model [Figure 5B(ii) and (iii)]. The treatment enhanced wound closure and remarkably promoted epidermal regeneration (2.16-fold) and neo-epithelium formation (2.03-fold) compared to controls. Additionally, the integration of battery-free near-field communication technology allowed for real-time wound status monitoring via resistance-based sensing, showcasing its potential as a promising platform for chronic wound management[39].
Wearable ionogel patches with synergistic therapeutic effects are another innovative direction in wound care. Ju et al. developed a wearable composite ionogel patch by polymerizing AA and AM in 1-ethyl-3-methylimidazole solution doped with MXene Ti3C2Tx (T = F, O, or OH), followed by UV-crosslinking[8]. The patch exhibited remarkable electrical conductivity and photothermal properties under near-infrared (NIR) light, enabling synergistic photothermal electrical stimulation (PES) effect [Figure 5C(i)]. In vitro antibacterial assays revealed that PES treatment significantly suppressed bacterial growth of
pH-sensitive ionogel dressings that simultaneously monitor wound status and promote healing have also been developed in smart wound management. Zhi et al. developed a green, pH-sensitive ionogel using pectin, an ionic liquid (1-hydroxyethyl-3-methylimidazolium chloride), AM, and anthocyanidin[135]. The ionogel functioned as a smart wound dressing by dynamically monitoring the wound status via pH sensing. It accurately detected the wound transition from the acidic inflammation stage (pH ~6.14) to the near-neutral physiological phase (pH ~7.53) within the first three days of healing. This pH sensitivity, combined with the antibacterial effect of the IL and the antioxidant capacity of anthocyanidin, facilitated excellent re-epithelialization and significantly promoted wound healing over 14 days.
Janus fibrous membrane ionogels, known for their directional liquid transport and stimuli-responsive properties, offer a unique strategy for managing infected burn wounds. A PIL/carbon nitride-based Janus fibrous membrane (PIL-C3N4) was composed of a hydrophilic layer and a hydrophobic layer incorporating L-arginine-functionalized C3N4 (C3N4-arg) as a light-responsive nitric oxide (NO) donor[136]. When applied to Pseudomonas aeruginosa (P. aeruginosa)-infected burn wounds, the dressing provided two key functions under light irradiation: controlled NO release and unidirectional exudate transport. This synergistic effect resulted in potent antibacterial activity. In vivo experiments demonstrated that PIL-C3N4 with light exposure reduced wound area to 8.3% by day 11, decreased bacterial viability to 1%, and lowered interleukin-1β (IL-1β) and interleukin-6 (IL-6) levels to 79.7 and 84.9 pg·mL-1. Furthermore, the therapy enhanced collagen deposition to 86.9%, promoted angiogenesis and alleviated inflammation, thereby accelerating wound healing without significant toxicity.
Ionogel microneedles for wound healing
For treating deep-seated or inaccessible infected wounds, MNs offer superior advantages over traditional dressings, including minimally invasive drug delivery, enhanced skin penetration, and targeted therapeutic effects. Living Chlorella-loaded PIL-based microneedles (PILMN-Chl) were prepared by UV-crosslinking a mixture of 3-(2-hydroxyethyl)-1-vinylimidazolium bromide ([HEIM][Br]), N-(2-amino-2-oxoethyl)-2-propenamide (NAGA) and poly(ethylene glycol) diacrylate (PEGDA) [Figure 5D(i)][134]. These MNs exhibited excellent mechanical strength, successfully piercing the isolated skin under an applied force of 8 N for 10 s [Figure 5D(ii)]. In a diabetic mouse model with MRSA-infected wounds, PILMN-Chl treatment reduced the wound area to just 2.26% of its original size within 9 days [Figure 5D(iii)], with a sustained oxygen production for over 30 h under light [Figure 5D(iv)]. The accelerated healing was attributed to a synergistic combination of potent antibacterial effects, derived from the electrostatic and hydrophobic interactions between bacteria and MNs, and significant anti-inflammatory activity. This anti-inflammatory effect, possibly due to the oxygen supply of Chlorella, was evidenced by the suppression of IL-6 and IL-1β[134]. Moreover, ionogel MNs have shown promise in managing specific infected skin diseases, such as acne. SA-loaded PIL-MNs (SA-PIL-MNs) were used to treat acne[10]. These MNs demonstrated synergistic antibacterial efficacy, achieving near-complete eradication of Propionibacterium acnes (P. acnes) in vitro. In a murine acne infection model, SA-PIL-MN treatment significantly reduced ear thickness and achieved 83% inhibition rate of acne lesions within 72 h by downregulating inflammatory factors (TNF-α, IL-8), showing significant potential for treating infected inflammatory skin diseases.
Applications of ionogels in DDS
Ionogels have emerged as promising DDS due to their unique properties, such as high biocompatibility, tunable release profiles, and the ability to enhance drug solubility and bioavailability. They address key challenges in conventional DDS, including poor drug permeability, low absorption, and uncontrolled pharmacokinetics. The composition of ionogels, combining ILs with polymeric networks, allows for stimuli-responsive behavior and improved therapeutic efficacy, making them suitable for various administration routes such as oral, transdermal, and parenteral delivery.
Ionogels for oral drug delivery
Oral delivery is preferred for patient compliance but suffers from first-pass metabolism and drug instability. Raptis et al. developed a choline decanoate 1:2 molar ratio (chC10 1:2) ionogel with an additional mole of decanoic acid for oral insulin delivery [Figure 6A(i)][40]. This viscous, hydrophobic ionogel displayed sustained release properties, dissolving over 70 min in vitro via surface erosion and facilitating the co-release of insulin and the permeation enhancer decanoate. Upon intrajejunal administration in rats, (chC10 1:2) ionogel achieved a remarkable bioavailability of 6.5%, which was 7-fold and 13-fold higher than that of the choline geranate (CAGE) IL and sodium decanoate, respectively. The ionogel mediated a prolonged absorption profile [Figure 6A(ii)] and a significant blood glucose-lowering effect. Histology confirmed that the (chC10 1:2) ionogel caused only transient epithelial erosion, with fully recovered intestinal mucosa within 5 h, demonstrating its potential for an effective and relatively safe oral peptide delivery.
Figure 6. The applications of ionogels in drug delivery systems. (A) Choline decanoate based ionogel for oral drug delivery. (i) The chC10 1:1 was an IL prepared by salt metathesis reaction between choline bicarbonate and decanoic acid. An extra mole of decanoic acid was added to prepare gel-like sample (chC10 1:2). (ii) PK profile of insulin after intrajejunal administration in rats[40]. Copyright 2025, Wiley-VCH GmbH; (B) CAGE/CPVA gel patch for buccal drug delivery. (i) The design and sandwich structure of patch. (ii) Cumulative penetration of Insulin in the buccal tissue with CAGE/CPVA patch and CPVA patch as delivery vehicle. (iii) The CAGE/CPVA patch showed superior in vivo pharmacodynamic efficacy compared to other tested formulations[137]. Copyright 2020, Elsevier B.V; (C) CAGE-Pluronic ionogel for transdermal drug delivery. (i) Preparation of CAGE - Pluronic ionogel loaded with vancomycin hydrochloride and the transdermal delivery performance comparison between the intact skin and tape-stripped skin of rats. (ii) Ex vivo skin permeation profile (left) and retention (right) of vancomycin across and within the intact skin and tape-stripped skin[138]. Copyright 2025, American Chemical Society; (D) The Cu2+ loaded gel for local drug delivery. (i) The preparation of the gel and its capability in bacterial killing and tissue regeneration. (ii) The release curve of Cu2+ from F-Gel, F-IL-Gel and D-IL-Gel. (iii) The cumulative percentage of Cu2+ permeated across the skin after incubation with F-Gel and D-IL-Gel[139]. Copyright 2025, Royal Society of Chemistry. Statistical significance as stated by *P < 0.05, **P < 0.01, ***P < 0.001 or ****P < 0.0001. chC10 1:1: Choline decanoate 1:1 molar ratio; IL: ionic liquid; PK: pharmacokinetic; CAGE: choline geranate; CPVA: chitosan-modified poly(vinyl alcohol); F-Gel: Gels made by freeze-thaw method without CGLY (IL); F-IL-Gel: Gels made by freeze-thaw method with CGLY (IL); D-IL-Gel: Gels made by drying method with CGLY (IL); CGLY: choline–glycolate; chCl: choline chloride; PVA: poly(vinyl alcohol); DA: dopamine; PDA: polydopamine.
pH-responsive oral ionogels offer targeted delivery to acidic tumor environments. Mokhtarpour et al. developed an ion-gel for delivering sunitinib malate (SUM), an anti-cancer drug, by polymerizing HEMA in a therapeutic DES composed of ChCl and ascorbic acid[140]. The ion-gel maintained stability for 6 months without degradation and exhibited low cytotoxicity. Drug release was pH-dependent, with 89% of SUM released at pH = 1.2 over 96 h, compared to 54% at pH = 6.8 and slower release at pH = 7.4. This trend is attributed to protonation of SUM’s amino groups, which increases its aqueous solubility in acidic environments. This system enables targeted oral delivery for cancer treatment by leveraging acidic tumor environments.
Ionogels for buccal drug delivery
Buccal delivery avoids first-pass metabolism but requires effective mucoadhesion and permeation. A CAGE-based IL/DES was mixed with insulin to form a CAGE gel[137]. This CAGE gel was then sandwiched between two poly-(vinyl alcohol) patches to create a buccal DDS [CAGE/chitosan-modified poly(vinyl alcohol) (CPVA) patch] [Figure 6B(i)]. This CAGE gel exhibited solid-like viscoelastic behavior, dominated by its elastic modulus. The buccal DDS demonstrated efficient drug release, with over 27% of the loaded insulin released within 15 min in vitro. Ex vivo studies on porcine buccal tissue revealed that CAGE acted as a potent permeation enhancer, inducing a 7-fold increase in cumulative insulin transport (~26% of the loaded dose) compared to the control without CAGE [Figure 6B(ii)]. In vivo studies in rats demonstrated that the CAGE/CPVA patch mediated a sustained and dose-dependent hypoglycemic effect, lowering blood glucose levels by up to 50% at 6 h [Figure 6B(iii)]. Importantly, histological evaluation indicated no obvious tissue damage, highlighting the system’s potential for the buccal delivery of therapeutics such as insulin.
Ionogels for transdermal drug delivery
While transdermal delivery is a promising non-invasive route that bypasses gastrointestinal degradation and first-pass metabolism, its efficacy is limited by the skin’s natural barrier, the stratum corneum. Strategies to enhance skin permeability are therefore key to achieving effective sustained release. Datta et al. formulated a transdermal ionogel by incorporating CAGE IL with Pluronic F-127 (PF-127) for delivering vancomycin hydrochloride (VH) [Figure 6C(i)][138]. This ionogel significantly enhanced the skin permeation of VH, achieving a permeation of 7,543 ± 585 μg·cm-2 across tape-stripped skin [Figure 6C(ii)]. The drug release followed sustained, diffusion-controlled kinetics. The permeation enhancement is attributed to the transdermal ionogel’s ability to modify the skin barrier. The formulation demonstrated a reversible impact on the skin, supporting its potential for localized VH administration[138].
Transdermal ionogels are also effective for chronic diseases such as rheumatoid arthritis (RA). Bhamble et al. developed an etodolac (ETO)-loaded ionogel for RA management by incorporating ETO into a CAGE IL and gelling it with Carbopol®934P[141]. This ionogel significantly enhanced the solubility of ETO by approximately 19,600-fold and provided a sustained drug release over 48 h. Ex vivo permeation studies demonstrated a 1.3-fold higher flux compared to conventional ETO gel, facilitating improved skin penetration. In the rat model of collagen-induced arthritis (CIA), the ETO-CAGE ionogel effectively alleviated arthritic symptoms, reducing clinical scores by approximately 7.7-fold and paw thickness by 2.16-fold compared to the free ETO gel, showcasing its potential for transdermal delivery in RA treatment.
Ionogels for local drug delivery
Local delivery targets specific sites (e.g., wounds, tumors) to minimize systemic side effects. Gao et al. developed a copper ion (Cu2+)-loaded ionogel for wound management by incorporating the IL choline–glycolate (CGLY) into a PVA and polydopamine (PDA) network[139]. The CGLY facilitated gel formation via intermolecular hydrogen bonds, avoiding toxic chemical crosslinkers [Figure 6D(i)]. This IL-based gel demonstrated an 8.8-fold enhancement in Cu2+ transdermal delivery compared to the IL-free gel, attributed to the ability of CGLY to temporarily open the tight junctions of the stratum corneum. The released Cu2+ [21.2% from F-IL-Gel and 12.5% from D-IL-Gel, Figure 6D(ii)] permeated the skin [Figure 6D(iii)] to achieve enhanced sterilization through a dual mechanism: direct bactericidal activity and the generation of hydroxyl radicals (•OH) via a Fenton-like reaction (chemodynamic therapy, CDT) with endogenous hydrogen peroxide (H2O2) at the wound site. In vivo studies in mice with infected wounds confirmed that the D-IL-Gel, especially when combined with a low dose of H2O2, significantly reduced bacterial colonies (to 7%) and promoted tissue remodeling with minimal inflammation, showcasing its dual function as an antibacterial and wound-healing promoting dressing.
For localized chemotherapy, injectable ionogels offer tunable release. Hermida-Merino et al. developed ionogels by blending shark gelatin with a fluorinated ionic liquid (FIL, 1-ethyl-3-methylpyridinium perfluorobutanesulfonate) to enhance drug solubility for local administration[142]. These ionogels significantly improved the loading and sustained release of antitumor drugs doxorubicin and mithramycin. Doxorubicin exhibited a continuous release profile over 120 h, while mithramycin showed a triphasic release pattern with formulation-dependent kinetics: it was completely released within 80 h from GE/10IL but achieved only 40% release after 330 h from GE/25IL. The ionogels maintained injectable viscoelastic properties through a
Ionogels for nose-to-brain drug delivery
Nose-to-brain delivery bypasses the blood-brain barrier for neurological disorders. Jadhav et al. developed a thermoreversible ionogel for nose-to-brain delivery of Tetrabenazine hydrochloride (TBZ) by incorporating TBZ-loaded choline oleate ([Cho][Ole]) micelles into a PF-127 matrix[143]. The optimized ionogel (particle size: 164.98 nm) demonstrated a 1.37-fold and 1.42-fold enhancement in in vitro drug release and ex vivo permeation, respectively, compared to a plain gel. Both processes followed zero-order kinetics over 12 h. This system significantly improved the solubility and nasal permeability of the biopharmaceutics classification system (BCS) Class IV drug, facilitating direct drug delivery to the brain for managing neurological disorders such as Huntington’s disease, thereby showcasing the potential of ionogels in overcoming biological barriers for enhanced central nervous system therapy[143].
Ionogels demonstrate versatility across administration routes, offering solutions to drug delivery challenges through enhanced solubility, permeation, and controlled release. Their tunable composition and stimuli-responsive properties pave the way for personalized medicine applications.
Ionogels in tissue engineering applications
Ionogel-based materials have become promising in tissue engineering due to their biocompatibility, tunable mechanical properties, and ability to support cell proliferation and tissue regeneration[18,41,132]. Despite progress in muscle, bone, and neural tissue engineering, challenges such as scalable fabrication and long-term in vivo stability optimization remain[18,41]. Ionogel composition, especially IL types and polymer matrices, greatly affects ionic conductivity, biodegradability, and bioactivity, thus influencing the applications in tissue engineering[2,132,144-146].
In neural tissue engineering, a conformable neural interface was prepared based on a multicross-linked membrane–ionogel assembly (McMiA), which integrated a genipin-cross-linked biopolymeric ionogel (X-BI) conductor with a dopamine-cross-linked graphene oxide (X-GO) membrane [Figure 7A][41]. The X-BI comprised chitosan biopolymers and choline malate ([Ch]+[MA]-) ILs, forming a biocompatible ionic conductive network [Figure 7B]. The McMiA exhibited excellent biocompatibility (86% neuronal cell viability) and served as a stable, non-faradaic neurostimulator that prevented adverse ion exchange and charge transfer reactions. When applied to sciatic nerve stimulation for treating overactive bladder syndrome, the McMiA-based interface significantly suppressed urination, increasing the urination interval by 35% and urine volume per void by 75%, as validated by in vivo bladder pressure and urine output monitoring [Figure 7C]. This approach demonstrates the potential of ionogels in enabling safe, long-term neuromodulation for treating neurological disorders.
Figure 7. McMiA-based flexible neural interface for managing overactive bladder syndrome. (A) Schematics of this implantable neural interface, with images demonstrating its flexibility; (B) Material design of the McMiA-based neural interface, employing a X-GO layer serving as an ion diffusion barrier and a X-BI layer as a conductor to enable bioelectronics for a stable and conformable contact without undesirable electrochemical reactions; (C) In vivo evaluation demonstrated that McMiA-mediated neurostimulation modulates both urine volume and urination interval in an overactive bladder model[41]. Copyright 2023, American Chemical Society. Statistical significance as stated by **P < 0.01 or ***P < 0.001. McMiA: Multicross-linked membrane–ionogel assembly; X-GO: dopamine-cross-linked graphene oxide; X-BI: genipin-cross-linked biopolymeric ionogel; ECM: extracellular matrix.
Beyond neural interfaces, ionogels have also shown great potential in developing artificial muscles, a key component in tissue engineering for prosthetics and soft robotic assistive devices. Ananieva et al. developed bio-friendly artificial muscles by coating commercial coiled carbon nanotube (CNT) yarns with DES-derived eutectogel composites[34]. The gels were synthesized via free-radical copolymerization in a DES electrolyte [Figure 8A]. The DES was composed of ChCl and glycerol, and the ionic monomers, such as sulfopropyl acrylate or 2-acryloxyethyltrimethylammonium chloride (AETA), were copolymerized with hydroxyethyl acrylate (HEA) and cross-linked using PEGDA. The yarns coated with oppositely charged polyelectrolyte were used to obtain the unipolar stroke. This design yielded solid-state actuators with both yarns contracted under an applied voltage [Figure 8B], achieving a maximum strain of 2.25% under a 90 mN load and a volumetric work capacity of 120 kJ·m-3, which significantly surpasses that of natural skeletal muscles
Figure 8. Ionogels for the application of artificial muscles. (A) Schematic of a solid-state textile actuator using oppositely charged polyelectrolyte-coated yarns for unipolar actuation; (B) Strain and potential profiles of the textile actuator under chronoamperometric stimulation at a 90 mN load[34]. Copyright 2025, Wiley-VCH GmbH. SPA: 3-Sulfopropyl acrylate potassium salt; AETA: 2-acryloxyethyltrimethylammonium chloride; WE: working electrode; CE: counter electrode.
Other polymer-IL composite systems have also been explored to enhance the electromechanical properties of ionogel-based artificial muscles for tissue engineering. Ali et al. prepared polyvinyl chloride (PVC)/IL gels by blending PVC, dibutyl adipate (DBA), and ILs 1-pentyl-3-methylimidazolium hexafluorophosphate ([PF6]) via a solvent evaporation method[147]. The gels demonstrated high flexibility with an 373% elongation at break and strong actuation performance, achieving a displacement of 0.16 mm and 0.2 s response time under
Natural polymer-based ionogels have gained attention for tissue engineering due to their inherent biocompatibility, with Cel-based systems being a notable example. Nevstrueva et al. prepared natural Cel ionogels by dissolving Cel in 1-ethyl-3-methylimidazolium acetate ([EMIM][OAc]) and fabricating multilayer actuators via phase inversion[148]. These ionogel actuators demonstrated reversible bending at low voltages (±500 mV) with a maximum strain difference of 0.6% at 2 V, attributed to ion diffusion-induced actuation. Exhibiting high ionic conductivity (15.8 mS·cm-1) and biocompatibility, these ionogels are suitable for soft artificial muscles in biomedical applications such as prosthetic devices or surgical tools, providing precise, noiseless actuation for tissue engineering and rehabilitative therapies[148].
To better achieve natural muscle function, recent progress in bio-friendly ionogels has concentrated on optimizing their flexibility and work capacity. A biofriendly ionogel was prepared by UV-polymerizing HEA and PEGDA in choline acetate (ChAc) IL, which exhibited an ionic conductivity of 1.9 mS·cm-1 and mechanical flexibility (75% elongation at break)[75]. Artificial muscle actuators made from CNT yarns coated with this biofriendly ionogel achieved a 1.7% contractile strain and 35 mN force via electrochemical double-layer charging. This biocompatible system, with a work capacity of 3.37 mJ·g-1 that matches natural muscle, shows promise for skin-contact applications in prosthetic devices and rehabilitative assistive technologies[75].
Applications of ionogels in tumor therapy and biological adhesion
Ionogels are significant important for biomedical applications, especially for tumor therapy and bioadhesives. In cancer therapy, they address limitations of conventional nanocarriers, such as poor stability and low bioavailability. As bioadhesives, they provide superior wet-surface adhesion and enhanced flexibility compared to commercial products[6,24,149]. However, balancing performance and biosafety remains challenging. The type and concentration of ILs in ionogels greatly influence their antibacterial efficacy, drug delivery performance, and adhesive properties[132,146,149].
Ionogels for tumor therapy
Ju et al. developed an electrostimulation-augmented photothermal patch (eT-patch) doped with MXene for melanoma treatment[42]. The patch was fabricated by copolymerizing AM and AA in the IL 1-ethyl-3-methylimidazolium, incorporating MXene as a photothermal agent [Figure 9A(i)]. Under combined photothermal therapy (PTT) and electrical stimulation (ES), termed PES, the eT-patch achieved effective tumor suppression [Figure 9A(ii) and (iii)], resulting in a significant inhibition of tumor weight and volume. The anti-tumor mechanism involved synergistic apoptosis and pyroptosis activation, which was associated with reactive oxygen species (ROS) generation and mitochondrial dysfunction. Importantly, the treatment caused no significant body weight loss or organ damage, demonstrating its high biosafety. This study highlights the potential of ionogel-based patches as a safe and efficient platform for synergistic tumor therapy.
Figure 9. The application of ionogel in tumor treatment and bioadhesive agent. (A) a wearable MXene-enhanced ionogel patch (eT-patch) for skin tumor treatment. (i) Design and mechanism of eT-patch with PES effect to treat melanoma. (ii) In vivo therapeutic protocol and representative photographs of excised tumors from B16F10 tumor-bearing mice after 15 days with different treatments (Control, Laser, PTT, ES, and PES). (iii) Quantitative analysis of tumor weight and relative volume after 15-day therapy across different treatment groups[42]. Copyright 2025, Springer Nature Limited. (B) Supramolecular ionogels for soft tissue adhesion. (i) Design of tough ionogel bioadhesive. (ii) Ex vivo demonstration of strong, reversible, and biocompatible tissue adhesion on porcine organs (lung and heart), highlighting its potential for surgical applications[29]. Copyright 2024, American Chemical Society. (C) Adhesion and repair for hard tissues based on an IGBI. (i) Fabrication and adhesion mechanism of IGBI for hard tissue integration. (ii) Adhesion performance of IGBI on various hard tissues (calvarials and teeth) and biomaterials (tantalum metal and zirconia ceramics). (iii) Application of IGBI for the repair of critical-sized calvarial bone defects. (iv) Schematic of a canine tooth reimplantation model utilizing the IGBI[43]. Copyright 2024, Wiley-VCH GmbH. PES: Photothermal electrical stimulation; PTT: photothermal therapy; ES: electrical stimulation; IGBI: ionogel bioadhesive interface; MBAA: N, N′-methylenebis (acrylamide); HF: mixed solution of lithium fluoride and hydrochloric acid; EMIES: 1-ethyl-3-methylimidazole; LDH: lactate dehydrogenase; GSDME: the gene expressions of markers from pyroptosis; ROS: reactive oxygen species; ATP: adenosine triphosphate; MMP: mitochondrial transmembrane potential; DMAA: N,N-dimethylacrylamide; PDMAA: poly(N,N-dimethylacrylamide); CAC: citric acid and L-(-)-carnitine; PET: polyethylene terephthalate; SF: silk fibroin.
Ionogels as bioadhesives
Bioadhesives play a crucial role in wound management, tissue repair, and organ transplantation[150,151]. Ionogels, with tunable IL components and polymer networks, exhibit enhanced adhesion through multiple interactions such as hydrogen bonding, electrostatic forces, and van der Waals interactions, making them promising alternatives to commercial adhesives[149]. They can be categorized based on the target biological tissues: soft tissues and hard tissues.
Ionogels for soft tissue adhesion
A biocompatible ionogel with reversible supramolecular adhesion was fabricated through the in situ photo-cross-linking of DMA, using CAC as the solvent [Figure 9B(i)][29]. The resulting ionogel exhibited high toughness (115.2 MJ·m-3) and achieved a remarkable adhesion strength of 24.4 MPa on glass, facilitated by supramolecular hydrogen bonding between its abundant carboxyl/hydroxyl groups and substrates. Critically for biomedical applications, the ionogel formed a tough, mechanically matched adhesion on wet biological tissues, demonstrating an interfacial toughness of 191.1 J·m-2 on porcine skin, which is more than twice that of commercial tissue adhesives. This strong adhesion enabled the sealing of porcine lung incisions and the gentle lifting of a porcine heart during simulated transplantation [Figure 9B(ii)]. The adhesion is reversible, allowing the ionogel to be benignly detached on-demand using saline solution without tissue damage, highlighting its significant potential as a reversible bioadhesive for wound management and organ transplantation.
Ge et al. developed a biocompatible ionogel by photopolymerizing HEAA and zwitterionic N-(3-sulfopropyl)-N-(methacryloxyethyl)-N, N-dimethylammonium betaine (DMAPS) in a ChCl/glycerol DES[152]. The multiple hydrogen bonds and electrostatic interactions enabled ultrafast gelation (14-36 s) and instant, pressure-free adhesion, achieving a remarkable burst pressure of 41.77 kPa on rabbit intestine tissue. This ionogel effectively sealed punctures in stomach and intestine tissues, preventing liquid leakage, while demonstrating excellent biocompatibility with HaCaT cells. Its strong adhesion in wet physiological environments highlights significant potential as a surgical sealant for gastrointestinal repairs and wound closure.
Ionogels for hard tissue adhesion
Jiang et al. developed an ionogel bioadhesive interface (IGBI) using silk fibroin and calcium ions dissolved in a formic acid/CaCl2 system [Figure 9C(i)][43]. The IGBI demonstrated exceptional adhesion to a range of important hard tissues (such as rat calvarial and human teeth) and hard biomaterials (such as tantalum metal and zirconia ceramics) [Figure 9C(ii)], achieving interfacial toughness > 180 J·m-2 on dog tooth and shear strength of 240 kPa, which is 14-55 times higher than commercial adhesives. This strong adhesion originated from synergistic hydrogen bonding and electrostatic interactions between the ionogel components and tissue surfaces. Remarkably, the IGBI exhibited mechanical self-reinforcement under physiological conditions, increasing its strength by 2.6-fold after implantation. IGBI adhered well upon bone defect sight of SD rats and significantly enhanced bone regeneration [Figure 9C(iii)]. In canine tooth reimplantation experiment, roots wrapped with IGBI were implanted to facilitate the generation of periodontal ligament-like tissue between the root surface and alveolar bone [Figure 9C(iv)]. These in vivo experiments show the potential of IGBI in hard tissue adhesion and repair. Moreover, by integrating bioelectronic devices, the IGBI platform enabled wireless monitoring of tissue regeneration, highlighting its dual functionality in both hard tissue repair and healing assessment.
Ionogels for adhesion in harsh environments
A hydrophobic ionogel was prepared by UV-polymerizing lauryl acrylate (LA) and tetrahydrofurfuryl acrylate (THFA) in the presence of lauroyl chloride-modified Cel within the [EMIM][TFSI] ionic liquids[153]. The Cel-mediated energy dissipation through interfacial friction within the mitigated sea–island structure endowed the ionogel with ultrahigh adhesion. It achieved an interfacial toughness of 1.06 kJ·m-2 on stainless steel and retained 0.68 kJ·m-2 underwater. This ionogel is a promising robust bioadhesive, capable of adhering to diverse substrates in harsh environments due to its excellent acid/alkali resistance and reusability. This makes it suitable for challenging underwater applications, including motion sensing and vital sign monitoring[153].
CONCLUSION AND OUTLOOK
An ionogel is a soft gel material that immobilizes ILs within a 3D network. It consists of two primary components: a solid 3D network (typically a polymer matrix) that serves as the structural framework, and a liquid phase of swollen or dispersed ILs. The advantages of ionogels include high ionic conductivity, remarkable thermal and electrochemical stability, and near-zero volatility. Furthermore, their physicochemical properties are highly tunable due to the inherent designability of both the solid matrix and the dispersed ILs. Specifically, by selecting and combining different polymer matrices with functionalized ILs, properties such as mechanical strength, viscoelasticity, hydrophilicity/hydrophobicity, and specific functions (e.g., self-healing, adhesion, and antibacterial activity) can be precisely customized. These unique characteristics establish a solid foundation for the application of ionogels in numerous cutting-edge biomedical fields, particularly in wound healing, drug delivery, biological adhesion, and tissue engineering.
Despite its promising future, ionogels face a series of significant challenges in transitioning from an excellent laboratory concept to biomedical practice.
1. While several studies have confirmed the short-term biocompatibility of some ionogels, their long-term in vivo safety profiles remain largely uncharacterized. On one side, the metabolic pathways, accumulation effects, potential genotoxicity, and immunogenicity of ILs in vivo have not been systematically and thoroughly studied. Their degradation products may cause unknown interference with normal cell function. On the other side, a lack of unified biosafety evaluation standards hindered cross-sectional data comparisons. As we can find, significant attention is being directed toward biomass polymers and naturally derived ILs with inherent antibacterial properties/biofunctions, as they offer a promising route to enhance ionogel biocompatibility.
2. The stability of ionogel properties and functions, such as adhesion, antibacterial activity, and drug release as mentioned above, under complex in vivo conditions (e.g., enzymes, stress, pH changes) for the duration of a treatment cycle requires extensive study.
3. A significant technological gap exists between gram-level preparation in the laboratory and large-scale production. Several key challenges must be overcome to enable the industrialization of ionogels. These include balancing cost with performance and developing standardized, low-cost, and environmentally friendly large-scale production methods, particularly for high-purity ILs. A further critical requirement is ensuring high batch-to-batch consistency in their mechanical, chemical, and biological properties.
4. The critical challenge is whether traditional sterilization methods (e.g., steam sterilization, plasma irradiation, ethylene oxide) compromise the intricate chemical structures (such as dynamic bonds) and functions of ionogels. Furthermore, designing appropriate packaging and storage solutions to maintain their long-term activity and stability is crucial.
5. A universal material design cannot meet the varied demands of different patients and disease stages. Current ionogels, while promising, lack personalization capabilities. A major future challenge is the rapid customization of their mechanical properties, drug combinations, and degradation rates to match a patient’s specific wound condition, tumor type, or tissue defect shape. This relies on a two-way feedback loop: application data informs performance optimization, and performance outcomes, in turn, refine the application model. This loop facilitates the collection, analysis, establishment and predictive use of relevant big data, which is essential for effective personalization.
Overall, close collaboration between chemists, materials scientists, biologists, and clinicians will enable ionogels to bridge the gap from the laboratory to the biomedical practice. Ultimately, ionogels are believed to play an increasingly important role in cutting-edge biomedical fields such as drug delivery, wound healing, tissue engineering and cancer therapy.
DECLARATIONS
Authors’ contributions
Conceptualization: Nie, Q.; He, W.; Ming, X.
Writing: Nie, Q.; He, W.
Validation, resources: Dong, W.; Chen, F.; Li, H.; Li, K.; Tian, L.
Review and editing, supervision, project administration: He, W.; Ming, X.; Ren, S.
Funding acquisition: Ming, X.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant No. 22205190), the Shaanxi Provincial Joint Recruitment and Shared Utilization Program (SXXZGY202405), the “Young Talent Support Plan” of Xi’an Jiaotong University (HX6J020), and the Shaanxi Provincial “Sanqin Talents” Program (2024SYJ14). He, W. and Nie, Q. thank the Scientific Research Startup Foundation of Guang’an Institute of Technology.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Gao, Y.; Cao, J.; Shu, Y.; Wang, J. Research progress of ionic liquids-based gels in energy storage, sensors and antibacterial. Green. Chem. Eng. 2021, 2, 368-83.
2. Datta, D.; Colaco, V.; Bandi, S. P.; et al. Stimuli-responsive self-healing ionic gels: a promising approach for dermal and tissue engineering applications. ACS. Biomater. Sci. Eng. 2025, 11, 1338-72.
3. He, W.; Liu, P.; Zhang, J.; Yao, X. Emerging applications of bioinspired slippery surfaces in biomedical fields. Chemistry 2018, 24, 14864-77.
4. Bertsch, P.; Diba, M.; Mooney, D. J.; Leeuwenburgh, S. C. G. Self-healing injectable hydrogels for tissue regeneration. Chem. Rev. 2023, 123, 834-73.
5. Pedro, A. Q.; Castro, L. S.; Coutinho, J. A.; Freire, M. G. Ionogels as advanced materials for overcoming challenges in wound healing and drug delivery. Nano. Mater. Sci. 2025, 7, 599-626.
6. Kuddushi, M.; Xu, B. B.; Malek, N.; Zhang, X. Review of ionic liquid and ionogel-based biomaterials for advanced drug delivery. Adv. Colloid. Interface. Sci. 2024, 331, 103244.
7. Karamikamkar, S.; Yalcintas, E. P.; Haghniaz, R.; et al. Aerogel-based biomaterials for biomedical applications: from fabrication methods to disease-targeting applications. Adv. Sci. 2023, 10, e2204681.
8. Ju, X.; Kong, J.; Qi, G.; et al. Photoelectric-driven conductive composite ionogel patch for effective wound healing. eScience 2024, 4, 100223.
9. Zhang, Z.; Zhang, Q.; Gao, S.; Xu, H.; Guo, J.; Yan, F. Antibacterial, anti-inflammatory and wet-adhesive poly(ionic liquid)-based oral patch for the treatment of oral ulcers with bacterial infection. Acta. Biomater. 2023, 166, 254-65.
10. Zhang, T.; Sun, B.; Guo, J.; et al. Active pharmaceutical ingredient poly(ionic liquid)-based microneedles for the treatment of skin acne infection. Acta. Biomater. 2020, 115, 136-47.
11. He, W.; Wang, Z.; Hou, C.; et al. Mucus-inspired supramolecular adhesives with oil-regulated molecular configurations and long-lasting antibacterial properties. ACS. Appl. Mater. Interfaces. 2020, 12, 16877-86.
12. Chen, Y.; Jiang, C.; Zhang, C.; He, W.; Hou, C. Antibacterial micro/nanorobots: innovations, challenges, and future perspectives in combatting infection. Nano. Select. 2025, e70084.
13. Noro, J.; Vilaça-Faria, H.; Reis, R. L.; Pirraco, R. P. Extracellular matrix-derived materials for tissue engineering and regenerative medicine: a journey from isolation to characterization and application. Bioact. Mater. 2024, 34, 494-519.
14. He, W.; Kong, S.; Lin, R.; et al. Machine learning assists in the design and application of microneedles. Biomimetics 2024, 9, 469.
15. Wang, F.; He, W.; Dai, B.; Zhang, X.; Wen, Y. Recent advances in asymmetric wettability dressings for wound exudate management. Research 2025, 8, 0591.
16. Zielińska, A.; Karczewski, J.; Eder, P.; et al. Scaffolds for drug delivery and tissue engineering: the role of genetics. J. Control. Release. 2023, 359, 207-23.
17. Boruah, M.; Sarma, A. Dry gels: concept, current trends, and new avenues in drug delivery and biomedical application. Adv. Healthc. Mater. 2025, 14, e00863.
18. Fan, X.; Liu, S.; Jia, Z.; et al. Ionogels: recent advances in design, material properties and emerging biomedical applications. Chem. Soc. Rev. 2023, 52, 2497-527.
19. Cao, Z.; Liu, H.; Jiang, L. Transparent, mechanically robust, and ultrastable ionogels enabled by hydrogen bonding between elastomers and ionic liquids. Mater. Horiz. 2020, 7, 912-8.
20. Marr, P. C.; Marr, A. C. Ionic liquid gel materials: applications in green and sustainable chemistry. Green. Chem. 2016, 18, 105-28.
21. Lu, G.; Chen, C.; Wang, Z.; et al. High-performance supramolecular organogel adhesives for antimicrobial applications in diverse conditions. ACS. Appl. Mater. Interfaces. 2023, 15, 44194-204.
22. Wang, Z.; Yi, B.; Wu, M.; et al. Bioinspired supramolecular slippery organogels for controlling pathogen spread by respiratory droplets. Adv. Funct. Mater. 2021, 31, 2102888.
23. Susan, M. A. B. H.; Nada, A.; Watanabe, M. Ion gels prepared by in situ radical polymerization of vinyl monomers in room temperature ionic liquids as novel highly conductive polymer electrolytes. J. Am. Chem. Soc. 2005, 127, 4976-83.
24. Gao, Y.; Zhang, W.; Li, L.; Wang, Z.; Shu, Y.; Wang, J. Ionic liquid-based gels for biomedical applications. Chem. Eng. J. 2023, 452, 139248.
25. Chen, N.; Zhang, H.; Li, L.; Chen, R.; Guo, S. Ionogel electrolytes for high-performance lithium batteries: a review. Adv. Energy. Mater. 2018, 8, 1702675.
26. Ahangaran, F. Functionalized ionogels with self-healing performance: material design, chemistry aspects, applications, and future prospects. Adv. Colloid. Interface. Sci. 2025, 345, 103633.
27. Luo, Z.; Li, W.; Yan, J.; Sun, J. Roles of ionic liquids in adjusting nature of ionogels: a mini review. Adv. Funct. Mater. 2022, 32, 2203988.
28. Singh, A.; Singh, J.; Singh, N.; Kaur, N. Development and characterization of gelatin/carboxymethyl cellulose based polymeric film with inclusion of ionic liquid to enhance the shelf life of food. Food. Packag. Shelf. Life. 2024, 42, 101251.
29. Xiong, J.; Duan, M.; Zou, X.; et al. Biocompatible tough ionogels with reversible supramolecular adhesion. J. Am. Chem. Soc. 2024, 146, 13903-13.
30. Sharma, A.; Rawat, K.; Solanki, P. R.; Bohidar, H. B. Self-healing gelatin ionogels. Int. J. Biol. Macromol. 2017, 95, 603-7.
31. Le, K.; Sun, X.; Chen, J.; et al. Stretchable, self-healing, biocompatible, and durable ionogel for continuous wearable strain and physiological signal monitoring. Chem. Eng. J. 2023, 471, 144675.
32. Ding, Y.; Zhang, J.; Chang, L.; Zhang, X.; Liu, H.; Jiang, L. Preparation of high-performance ionogels with excellent transparency, good mechanical strength, and high conductivity. Adv. Mater. 2017, 29, 1704253.
33. Luo, Z.; Cui, H.; Guo, J.; et al. Poly(ionic liquid)/Ce-based antimicrobial nanofibrous membrane for blocking drug-resistance dissemination from MRSA-infected wounds. Adv. Funct. Mater. 2021, 31, 2100336.
34. Ananieva, G.; Vancaeyzeele, C.; Nguyen, G. T.; et al. Bio-friendly artificial muscles based on carbon nanotube yarns and eutectogel derivatives. Adv. Funct. Mater. 2025, 36, e15458.
35. He, W.; Lin, R.; Kong, S.; et al. Intelligent sensing: the emerging integration of machine learning and soft sensors based on hydrogels and ionogels. Adv. Sci. 2025, 12, e17851.
36. Liu, C.; Raza, F.; Qian, H.; Tian, X. Recent advances in poly(ionic liquid)s for biomedical application. Biomater. Sci. 2022, 10, 2524-39.
37. Niu, H.; Yin, F.; Kim, E. S.; et al. Advances in flexible sensors for intelligent perception system enhanced by artificial intelligence. InfoMat 2023, 5, e12412.
38. Jiang, N.; Chang, X.; Hu, D.; et al. Flexible, transparent, and antibacterial ionogels toward highly sensitive strain and temperature sensors. Chem. Eng. J. 2021, 424, 130418.
39. Yang, J.; Tsai, Y.; Cheng, T.; et al. All-in-one and on-demand wound dressing based on a deep eutectic solvent ionic gel. Chem. Eng. J. 2025, 515, 163849.
40. Raptis, K.; Heade, J.; Cunha, C.; et al. Permeation enhancer-based ionogel shows remarkable potential for oral insulin delivery. Adv. Healthc. Mater. 2025, 14, e2500946.
41. Kim, J. S.; Kim, J.; Lim, J. W.; et al. Implantable multi-cross-linked membrane-ionogel assembly for reversible non-faradaic neurostimulation. ACS. Nano. 2023, 17, 14706-17.
42. Ju, X.; Kong, J.; Qi, G.; et al. A wearable electrostimulation-augmented ionic-gel photothermal patch doped with MXene for skin tumor treatment. Nat. Commun. 2024, 15, 762.
43. Jiang, C.; Fu, J.; Zhang, H.; et al. Self-reinforcing ionogel bioadhesive interface for robust integration and monitoring of bioelectronic devices with hard tissues. Adv. Mater. 2025, 37, e2413028.
44. Wang, H.; Wang, Z.; Yang, J.; Xu, C.; Zhang, Q.; Peng, Z. Ionic gels and their applications in stretchable electronics. Macromol. Rapid. Commun. 2018, e1800246.
45. Yan, C. C.; Li, W.; Liu, Z.; et al. Ionogels: preparation, properties and applications. Adv. Funct. Mater. 2023, 34, 2314408.
46. Xia, Q.; Li, W.; Zou, X.; et al. Metal-organic framework (MOF) facilitated highly stretchable and fatigue-resistant ionogels for recyclable sensors. Mater. Horiz. 2022, 9, 2881-92.
47. Ueki, T.; Watanabe, M. Macromolecules in ionic liquids: progress, challenges, and opportunities. Macromolecules 2008, 41, 3739-49.
48. Le Bideau, J.; Viau, L.; Vioux, A. Ionogels, ionic liquid based hybrid materials. Chem. Soc. Rev. 2011, 40, 907-25.
49. Ye, Y.; Rick, J.; Hwang, B. Ionic liquid polymer electrolytes. J. Mater. Chem. A. 2013, 1, 2719-43.
50. Dai, S.; Ju, Y. H.; Gao, H. J.; Lin, J. S.; Pennycook, S. J.; Barnes, C. E. Preparation of silica aerogel using ionic liquids as solvents. Chem. Commun. 2000, 243-4.
51. Wu, S.; Wu, X.; Xing, W.; et al. Backbone structure effect on the thermoelectric properties of IDT-based p-type conjugated polymers. Macromol. Rapid. Commun. 2020, 41, e1900322.
52. Wang, M.; Hu, J.; Dickey, M. D. Tough ionogels: synthesis, toughening mechanisms, and mechanical properties - a perspective. JACS. Au. 2022, 2, 2645-57.
53. He, W.; Ming, X.; Xiang, Y.; et al. Bioinspired semicrystalline dynamic ionogels with adaptive mechanics and tactile sensing. ACS. Appl. Mater. Interfaces. 2022, 14, 20132-8.
54. Ming, X.; Shi, L.; Zhu, H.; Zhang, Q. Stretchable, phase-transformable ionogels with reversible ionic conductor–insulator transition. Adv. Funct. Mater. 2020, 30, 2005079.
55. Ren, Y.; Guo, J.; Liu, Z.; et al. Ionic liquid-based click-ionogels. Sci. Adv. 2019, 5, eaax0648.
56. Zhu, J.; Lu, X.; Zhang, W.; Liu, X. Substrate-independent, reversible, and easy-release ionogel adhesives with high bonding strength. Macromol. Rapid. Commun. 2020, 41, e2000098.
57. Wang, S.; Hsia, B.; Alper, J. P.; Carraro, C.; Wang, Z.; Maboudian, R. Comparative studies on electrochemical cycling behavior of two different silica-based ionogels. J. Power. Sources. 2016, 301, 299-305.
58. Viau, L.; Tourné-Péteilh, C.; Devoisselle, J. M.; Vioux, A. Ionogels as drug delivery system: one-step sol-gel synthesis using imidazolium ibuprofenate ionic liquid. Chem. Commun. 2010, 46, 228-30.
59. Ye, L.; Chen, F.; Liu, J.; et al. Responsive ionogel surface with renewable antibiofouling properties. Macromol. Rapid. Commun. 2019, 40, e1900395.
60. Suen, J. W.; Elumalai, N. K.; Debnath, S.; Mubarak, N. M.; Lim, C. I.; Reddy, M. M. The role of interfaces in ionic liquid-based hybrid materials (ionogels) for sensing and energy applications. Adv. Mater. Inter. 2022, 9, 2201405.
61. Bielejewski, M. Effect of self-assembly aggregation on physical properties of non-aqueous ionogels based on LMWG. J. Sol. Gel. Sci. Technol. 2018, 88, 671-83.
62. Huang, Y.; Ming, X.; Tang, Z.; et al. Ultra-strong ionogel adhesives via in situ microphase separation. Adv. Funct. Mater. 2024, 35, 2417011.
63. Wang, J.; Zheng, Y.; Cui, T.; et al. Bioinspired ultra-robust ionogels constructed with soft-rigid confinement space for multimodal monitoring electronics. Adv. Funct. Mater. 2023, 34, 2312383.
64. Xu, Q.; Zheng, Z.; Wang, B.; Mao, H.; Yan, F. Zinc ion coordinated poly(ionic liquid) antimicrobial membranes for wound healing. ACS. Appl. Mater. Interfaces. 2017, 9, 14656-64.
65. Jiang, H.; Cheng, Y.; Zhang, X.; et al. Progress of ionogels in flexible pressure sensors: a mini-review. Polymers 2025, 17, 1093.
66. Kim, M. S.; Kim, J. H.; Yoo, H.; et al. Ultrastretchable, tough, and highly conductive ionogels for multipurpose motion monitoring. ACS. Mater. Lett. 2024, 6, 4658-66.
67. Pan, H.; Huang, J.; Huang, X.; et al. Biosafety, anti-swelling and anti-freezing amino acid-based ionogels for flexible strain sensors. Chem. Eng. J. 2025, 520, 166097.
68. Li, W.; Li, L.; Liu, Z.; Zheng, S.; Li, Q.; Yan, F. Supramolecular ionogels tougher than metals. Adv. Mater. 2023, 35, e2301383.
69. Sun, L.; Huang, H.; Zhang, L.; et al. Spider-silk-inspired tough, self-healing, and melt-spinnable ionogels. Adv. Sci. 2024, 11, e2305697.
70. Zhou, Y.; Ma, D.; Luo, Y.; et al. Simultaneous enhancement of mechanical properties and ionic conductivity of sustainable and antibacterial Ionogels by cellulose acetate for versatile flexible sensors. Chem. Eng. J. 2025, 524, 169307.
71. Correia, D. M.; Fernandes, L. C.; Martins, P. M.; et al. Ionic liquid–polymer composites: a new platform for multifunctional applications. Adv. Funct. Mater. 2020, 30, 1909736.
72. Hu, Y.; Xing, Y.; Yue, H.; et al. Ionic liquids revolutionizing biomedicine: recent advances and emerging opportunities. Chem. Soc. Rev. 2023, 52, 7262-93.
73. Liu, G.; Zhong, R.; Hu, R.; Zhang, F. Applications of ionic liquids in biomedicine. Biophys. Rev. Lett. 2013, 07, 121-34.
74. Patel, V.; Das, E.; Bhargava, A.; Deshmukh, S.; Modi, A.; Srivastava, R. Ionogels for flexible conductive substrates and their application in biosensing. Int. J. Biol. Macromol. 2024, 254, 127736.
75. Ni, B.; Gelas, L.; Ananieva, G.; et al. Artificial muscle based on coiled CNT yarns and biofriendly ionogels. Sens. Actuators. B. Chem. 2024, 403, 135227.
76. Huo, X.; Wang, J.; Cong, Z.; et al. Strong and tough eutectogels with broad-range tunable mechanical properties via the hydrogen bond network-specific effect. Adv. Funct. Mater. 2025, 35, 2422464.
77. Zhang, Y.; Yuan, B.; Zhang, Y.; et al. Biomimetic lignin/poly(ionic liquids) composite hydrogel dressing with excellent mechanical strength, self-healing properties, and reusability. Chem. Eng. J. 2020, 400, 125984.
78. Jiang, J.; Gao, D.; Li, Z.; Su, G. Gel polymer electrolytes prepared by in situ polymerization of vinyl monomers in room-temperature ionic liquids. React. Funct. Polym. 2006, 66, 1141-8.
79. Wang, M.; Hu, J.; Dickey, M. D. Emerging applications of tough ionogels. NPG. Asia. Mater. 2023, 15, 66.
80. Sun, J.; Lu, G.; Zhou, J.; Yuan, Y.; Zhu, X.; Nie, J. Robust physically linked double-network ionogel as a flexible bimodal sensor. ACS. Appl. Mater. Interfaces. 2020, 12, 14272-9.
81. Zhao, X. Multi-scale multi-mechanism design of tough hydrogels: building dissipation into stretchy networks. Soft. Matter. 2014, 10, 672-87.
82. Li, X.; Jiang, H.; Zhang, Y.; et al. Stimulation-reinforced cellulose–protein ionogels with superior mechanical strength and temperature resistance. Adv. Funct. Mater. 2024, 34, 2408160.
83. Zhang, J.; Ma, Z.; Li, M.; Lou, M.; Wang, H.; Jia, L. Development of ultrathin, breathable, waterproof, and durable nanonet-supported ionogel sensors for electrophysiological monitoring. Adv. Funct. Mater. 2024, 35, 2415694.
84. Kuddushi, M.; Patel, N. K.; Rajput, S.; El Seoud, O. A.; Mata, J. P.; Malek, N. I. Temperature-responsive low molecular weight ionic liquid based gelator: an approach to fabricate an anti-cancer drug-loaded hybrid ionogel. ChemSystemsChem 2020, 2, e1900053.
85. Kuddushi, M.; Ray, D.; Aswal, V.; Hoskins, C.; Malek, N. Poly(vinyl alcohol) and functionalized ionic liquid-based smart hydrogels for doxorubicin release. ACS. Appl. Bio. Mater. 2020, 3, 4883-94.
86. Kuddushi, M.; Pandey, D. K.; Singh, D. K.; Mata, J.; Malek, N. An ionic hydrogel with stimuli-responsive, self-healable and injectable characteristics for the targeted and sustained delivery of doxorubicin in the treatment of breast cancer. Mater. Adv. 2022, 3, 632-46.
87. Zhao, X.; Xu, J.; Zhang, J.; et al. Fluorescent double network ionogels with fast self-healability and high resilience for reliable human motion detection. Mater. Horiz. 2023, 10, 646-56.
88. Zhang, M.; Zhao, L.; Tian, F.; et al. Bionic artificial skin based on self-healable ionogel composites with tailored mechanics and robust interfaces. Adv. Mater. 2024, 36, e2405776.
89. Adawiyah, N.; Moniruzzaman, M.; Hawatulaila, S.; Goto, M. Ionic liquids as a potential tool for drug delivery systems. Med. Chem. Commun. 2016, 7, 1881-97.
90. Lewandowski, A.; Świderska-Mocek, A. Properties of the lithium and graphite–lithium anodes in N-methyl-N-propylpyrrolidinium bis(trifluoromethanesulfonyl)imide. J. Power. Sources. 2009, 194, 502-7.
91. Lewandowski, A.; Świderska-mocek, A. Ionic liquids as electrolytes for Li-ion batteries - an overview of electrochemical studies. J. Power. Sources. 2009, 194, 601-9.
92. Ma, L.; Wang, J.; He, J.; et al. Ultra-sensitive, durable and stretchable ionic skins with biomimetic micronanostructures for multi-signal detection, high-precision motion monitoring, and underwater sensing. J. Mater. Chem. A. 2021, 9, 26949-62.
93. Han, J.; Choi, Y.; Lee, J.; Pyo, S.; Jo, S.; Yoo, J. UV curable ionogel for all-solid-state supercapacitor. Chem. Eng. J. 2021, 416, 129089.
94. Qian, W.; Texter, J.; Yan, F. Frontiers in poly(ionic liquid)s: syntheses and applications. Chem. Soc. Rev. 2017, 46, 1124-59.
95. Chen, H.; Choi, J.; Salas-de La Cru, D.; Winey, K. I.; Elabd, Y. A. Polymerized ionic liquids: the effect of random copolymer composition on ion conduction. Macromolecules 2009, 42, 4809-16.
96. Zhang, S. Y.; Zhuang, Q.; Zhang, M.; et al. Poly(ionic liquid) composites. Chem. Soc. Rev. 2020, 49, 1726-55.
97. Guo, J.; Sun, Z.; Zhou, Y.; Yan, F. Poly(ionic liquid)-based energy and electronic devices. Chin. J. Chem. 2022, 40, 1099-108.
98. Su, Q.; He, X.; Wang, S.; Song, H.; Dong, J. Choline-amino acid-based polyionic liquids and ionogels for antimicrobial infections. Mater. Today. Commun. 2024, 40, 109593.
99. Lei, B.; Cao, L.; Qu, X.; et al. Thermal-sensitive ionogel with NIR-light controlled adhesion for ultrasoft strain sensor. Nano. Res. 2023, 16, 5464-72.
100. Sun, Y.; Wang, C.; Guo, B.; Zhang, S.; Niu, W. Mussel-inspired tough ionogel with robust adhesion and mechanical adaptivity for impact resistance. Chem. Eng. J. 2024, 498, 155059.
101. Wang, S.; Li, J.; Li, S.; et al. Self-contained underwater adhesion and informational labeling enabled by arene-functionalized polymeric ionogels. Adv. Funct. Mater. 2023, 33, 2306814.
102. Cheng, Y.; Gao, Z.; Zhang, J.; et al. Tough, stretchable, and adhesive ionogels for impact resistance and wearable ionotronics. Chem. Eng. J. 2025, 522, 167762.
103. Pan, T.; Wang, X.; Mao, L.; Wang, H. Highly adhesive and conductive eco-friendly Mg/B-SA/PVA ionogels. Chem. Eng. J. 2025, 522, 167816.
104. Laxminarayan, R. Antibiotic effectiveness: balancing conservation against innovation. Science 2014, 345, 1299-301.
105. Ng, V. W. L.; Tan, J. P. K.; Leong, J.; Voo, Z. X.; Hedrick, J. L.; Yang, Y. Y. Antimicrobial polycarbonates: investigating the impact of nitrogen-containing heterocycles as quaternizing agents. Macromolecules 2014, 47, 1285-91.
106. Liu, P.; Jin, K.; Wong, W.; et al. Ionic liquid functionalized non-releasing antibacterial hydrogel dressing coupled with electrical stimulation for the promotion of diabetic wound healing. Chem. Eng. J. 2021, 415, 129025.
107. Lu, J.; Yan, F.; Texter, J. Advanced applications of ionic liquids in polymer science. Prog. Polym. Sci. 2009, 34, 431-48.
108. Torimoto, T.; Tsuda, T.; Okazaki, K.; Kuwabata, S. New frontiers in materials science opened by ionic liquids. Adv. Mater. 2010, 22, 1196-221.
109. Demberelnyamba, D.; Kim, K. S.; Choi, S.; et al. Synthesis and antimicrobial properties of imidazolium and pyrrolidinonium salts. Bioorg. Med. Chem. 2004, 12, 853-7.
110. Hajebi, S.; Rabiee, N.; Bagherzadeh, M.; et al. Stimulus-responsive polymeric nanogels as smart drug delivery systems. Acta. Biomater. 2019, 92, 1-18.
111. Carmona-Ribeiro, A. M.; de Melo Carrasco, L. D. Cationic antimicrobial polymers and their assemblies. Int. J. Mol. Sci. 2013, 14, 9906-46.
112. Borkowski, A.; Ławniczak, Ł.; Cłapa, T.; et al. Different antibacterial activity of novel theophylline-based ionic liquids - growth kinetic and cytotoxicity studies. Ecotoxicol. Environ. Saf. 2016, 130, 54-64.
113. Zheng, Z.; Guo, J.; Mao, H.; Xu, Q.; Qin, J.; Yan, F. Metal-containing poly(ionic liquid) membranes for antibacterial applications. ACS. Biomater. Sci. Eng. 2017, 3, 922-8.
114. Zheng, Z.; Xu, Q.; Guo, J.; et al. Structure-antibacterial activity relationships of imidazolium-type ionic liquid monomers, poly(ionic liquids) and poly(ionic liquid) membranes: effect of alkyl chain length and cations. ACS. Appl. Mater. Interfaces. 2016, 8, 12684-92.
115. Guo, J.; Xu, Q.; Zheng, Z.; et al. Intrinsically antibacterial poly(ionic liquid) membranes: the synergistic effect of anions. ACS. Macro. Lett. 2015, 4, 1094-8.
116. Qin, J.; Guo, J.; Xu, Q.; Zheng, Z.; Mao, H.; Yan, F. Synthesis of pyrrolidinium-type poly(ionic liquid) membranes for antibacterial applications. ACS. Appl. Mater. Interfaces. 2017, 9, 10504-11.
117. Fernandes, M. M.; Carvalho, E. O.; Correia, D. M.; et al. Ionic liquids as biocompatible antibacterial agents: a case study on structure-related bioactivity on Escherichia coli. ACS. Appl. Bio. Mater. 2022, 5, 5181-9.
118. Singh, G.; Singh, G.; Damarla, K.; Sharma, P. K.; Kumar, A.; Kang, T. S. Gelatin-based highly stretchable, self-healing, conducting, multiadhesive, and antimicrobial ionogels embedded with Ag2O nanoparticles. ACS. Sustainable. Chem. Eng. 2017, 5, 6568-77.
119. Zheng, S.; Li, W.; Ren, Y.; et al. Moisture-wicking, breathable, and intrinsically antibacterial electronic skin based on dual-gradient poly(ionic liquid) nanofiber membranes. Adv. Mater. 2022, 34, e2106570.
120. Krishnadoss, V.; Melillo, A.; Kanjilal, B.; et al. Bioionic liquid conjugation as universal approach to engineer hemostatic bioadhesives. ACS. Appl. Mater. Interfaces. 2019, 11, 38373-84.
121. Md Moshikur, R.; Chowdhury, M. R.; Moniruzzaman, M.; Goto, M. Biocompatible ionic liquids and their applications in pharmaceutics. Green. Chem. 2020, 22, 8116-39.
122. Wheelis, S. E.; Biguetti, C. C.; Natarajan, S.; et al. Investigation of the early healing response to dicationic imidazolium-based ionic liquids: a biocompatible coating for titanium implants. ACS. Biomater. Sci. Eng. 2020, 6, 984-94.
123. Shen, L.; Kong, T.; Yu, J.; et al. Self-polymerized metal-phenolic ionogel with multifunctional properties towards theranostic wearable electronics. Acta. Biomater. 2025, 199, 154-65.
124. Zhang, T.; Guo, J.; Ding, Y.; Mao, H.; Yan, F. Redox-responsive ferrocene-containing poly(ionic liquid)s for antibacterial applications. Sci. China. Chem. 2018, 62, 95-104.
125. Sun, D.; Pang, X.; Cheng, Y.; et al. Ultrasound-switchable nanozyme augments sonodynamic therapy against multidrug-resistant bacterial infection. ACS. Nano. 2020, 14, 2063-76.
127. Gao, Y.; Hao, Y.; Zhang, W.; Wei, Y.; Shu, Y.; Wang, J. Microwave-triggered ionic liquid-based hydrogel dressing with excellent hyperthermia and transdermal drug delivery performance. Chem. Eng. J. 2022, 429, 131590.
128. Villar-Chavero, M. M.; Domínguez, J. C.; Alonso, M. V.; Oliet, M.; Rodriguez, F. Chitosan-reinforced cellulosic bionogels: viscoelastic and antibacterial properties. Carbohydr. Polym. 2020, 229, 115569.
129. Lin, B.; Luo, Y.; Xie, D.; Ren, Y.; Zhao, P.; Yue, J. pH-Responsive charge convertible hyperbranched poly(ionic liquid) nanoassembly with high biocompatibility for resistance-free antimicrobial applications. Nano. Lett. 2024, 24, 7408-16.
130. Duflo, S.; Thibeault, S. L.; Li, W.; Shu, X. Z.; Prestwich, G. D. Vocal fold tissue repair in vivo using a synthetic extracellular matrix. Tissue. Eng. 2006, 12, 2171-80.
131. Chen, K.; Wu, Z.; Liu, Y.; Yuan, Y.; Liu, C. Injectable double-crosslinked adhesive hydrogels with high mechanical resilience and effective energy dissipation for joint wound treatment. Adv. Funct. Mater. 2021, 32, 2109687.
132. Nikfarjam, N.; Ghomi, M.; Agarwal, T.; et al. Antimicrobial ionic liquid-based materials for biomedical applications. Adv. Funct. Mater. 2021, 31, 2104148.
133. Bhar, B.; Singh, R.; Ramesh, V. K.; et al. Wearable e-Bandage with antimicrobial ionogel as an integrated electroceutical device for accelerated wound healing. ACS. Mater. Lett. 2024, 6, 3453-61.
134. Gao, S.; Rao, Y.; Wang, X.; et al. Chlorella-loaded antibacterial microneedles for microacupuncture oxygen therapy of diabetic bacterial infected wounds. Adv. Mater. 2024, 36, e2307585.
135. Zhi, H.; Wang, F.; Zhang, X.; et al. Green, pH-sensitive, highly stretchable, and hydrogen bond-dominated ionogel for wound healing activity. ACS. Appl. Bio. Mater. 2024, 7, 498-507.
136. Wang, Y.; Zhang, Q.; Liu, Z.; et al. Janus fibrous dressing with controllable nitric oxide-releasing and unidirectional exudate transport activities for bacteria-infected burn wound therapy. Adv. Funct. Mater. 2025, 35, 2503992.
137. Vaidya, A.; Mitragotri, S. Ionic liquid-mediated delivery of insulin to buccal mucosa. J. Control. Release. 2020, 327, 26-34.
138. Datta, D.; Bandi, S. P.; Venuganti, V. V. K. Influence of choline-based ionogel on transdermal delivery of vancomycin hydrochloride. Mol. Pharm. 2025, 22, 3331-50.
139. Gao, Y. R.; Zhang, W. X.; Wei, Y. N.; et al. Ionic liquids enable the preparation of a copper-loaded gel with transdermal delivery function for wound dressings. Biomater. Sci. 2022, 10, 1041-52.
140. Mokhtarpour, M.; Shekaari, H.; Shayanfar, A. Design and characterization of ascorbic acid based therapeutic deep eutectic solvent as a new ion-gel for delivery of sunitinib malate. J. Drug. Deliv. Sci. Technol. 2020, 56, 101512.
141. Bhamble, S.; Dighe, S.; Katari, O.; Yadav, V.; Ansari, M. M.; Jain, S. Ionic liquid-based ionogel: a novel strategy to improve dermal delivery and in vivo efficacy of etodolac in rheumatoid arthritis management. J. Mol. Liq. 2024, 412, 125841.
142. Hermida-Merino, C.; Cabaleiro, D.; Gracia-Fernández, C.; et al. Ionogels derived from fluorinated ionic liquids to enhance aqueous drug solubility for local drug administration. Gels 2022, 8, 594.
143. Jadhav, N.; Chavan, M.; Patil, P.; Takale, P.; Mane, M.; Mohite, S. Thermoreversible ionogel for enhanced nose-to-brain delivery of tetrabenazine hydrochloride: optimization by central composite design and in vitro ex vivo evaluation. AAPS. PharmSciTech. 2025, 26, 210.
144. Gupta, M. K.; Khokhar, S. K.; Phillips, D. M.; et al. Patterned silk films cast from ionic liquid solubilized fibroin as scaffolds for cell growth. Langmuir 2007, 23, 1315-9.
145. Li, X.; Fan, D. Smart collagen hydrogels based on 1-ethyl-3-methylimidazolium acetate and microbial transglutaminase for potential applications in tissue engineering and cancer therapy. ACS. Biomater. Sci. Eng. 2019, 5, 3523-36.
146. Correia, D. M.; Fernandes, L. C.; Fernandes, M. M.; et al. Ionic liquid-based materials for biomedical applications. Nanomaterials 2021, 11, 2401.
147. Ali, I.; Latif, A.; Hussain, K.; et al. Ionic liquids enhanced performance of PVC gels actuator. J. Appl. Polym. Sci. 2021, 138, 50710.
148. Nevstrueva, D.; Murashko, K.; Vunder, V.; et al. Natural cellulose ionogels for soft artificial muscles. Colloids. Surf. B. Biointerfaces. 2018, 161, 244-51.
149. Huang, Y.; Zhu, H.; Zhang, Q.; Zhu, S. Ionogel adhesives: from structural design to emerging applications. Macromol. Rapid. Commun. 2025, 46, e2400973.
150. Hou, C.; He, W.; Yao, X. Mucus-inspired supramolecular adhesives: exploring the synergy between dynamic networks and functional liquids. ACS. Nano. 2025, 19, 14540-56.
151. Hou, C.; He, W.; Wang, Z.; et al. Particulate-aggregated adhesives with exudate-sensitive properties and sustained bacteria disinfection to facilitate wound healing. ACS. Appl. Mater. Interfaces. 2020, 12, 31090-8.
152. Ge, G.; Mandal, K.; Haghniaz, R.; et al. Deep eutectic solvents-based ionogels with ultrafast gelation and high adhesion in harsh environments. Adv. Funct. Mater. 2023, 33, 2207388.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






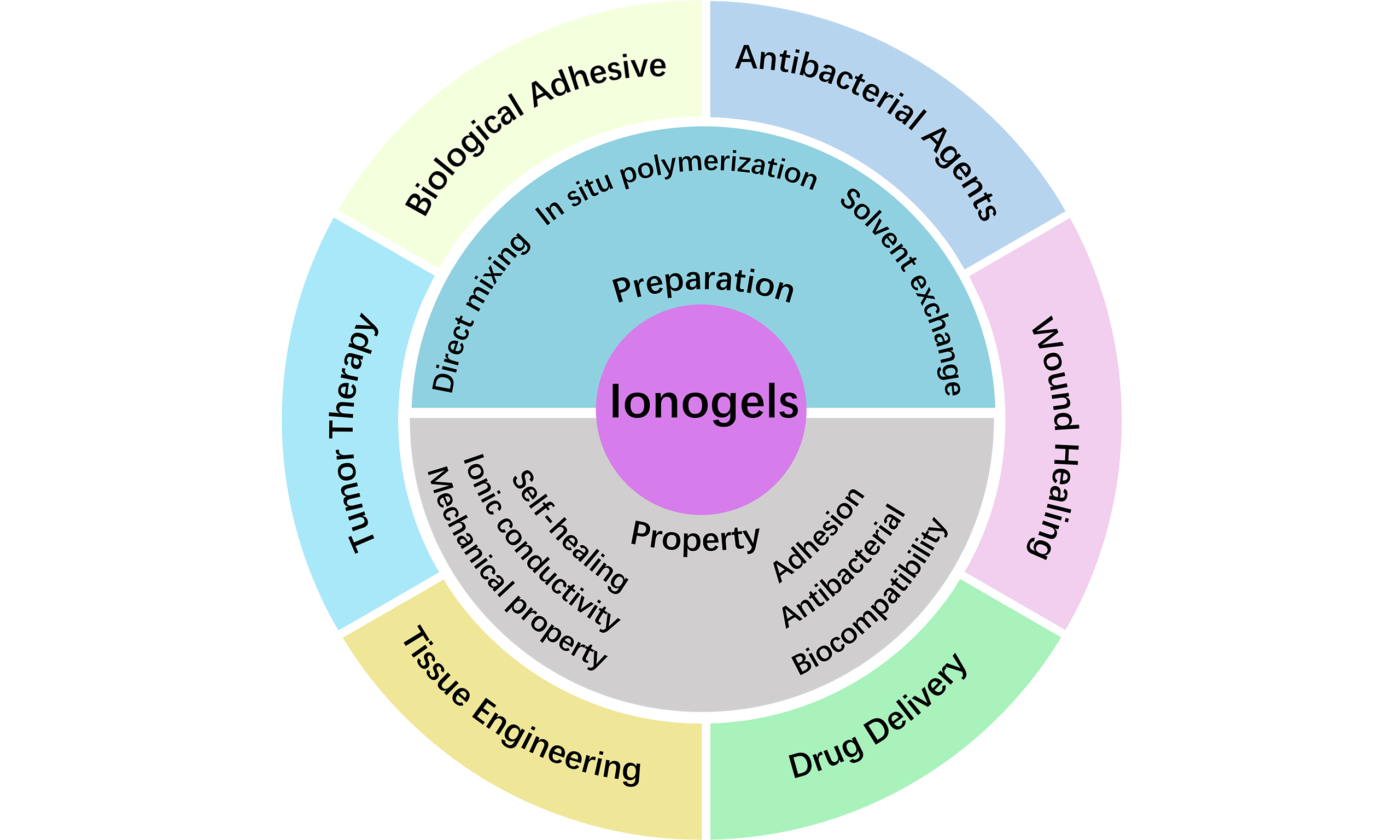
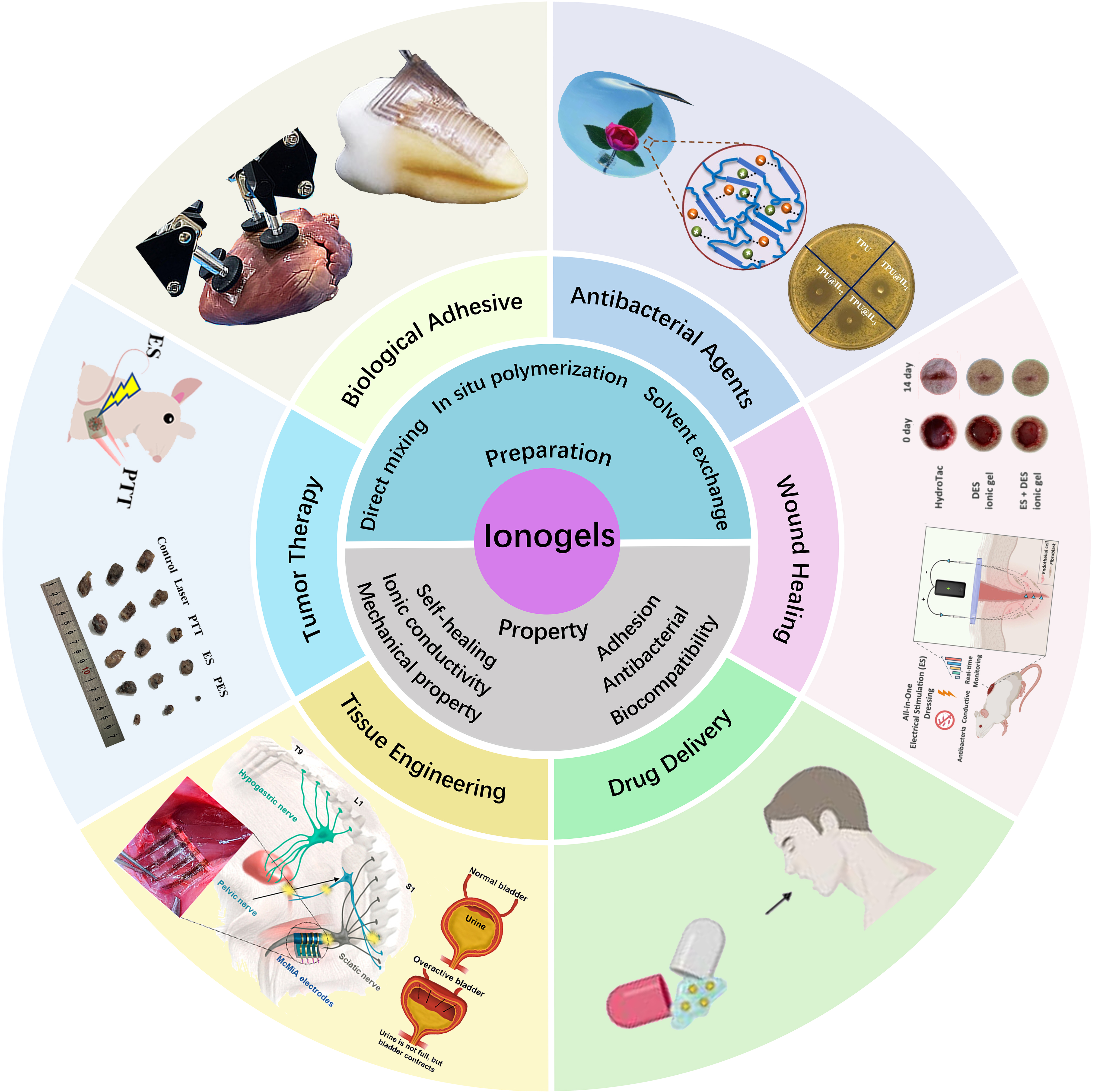
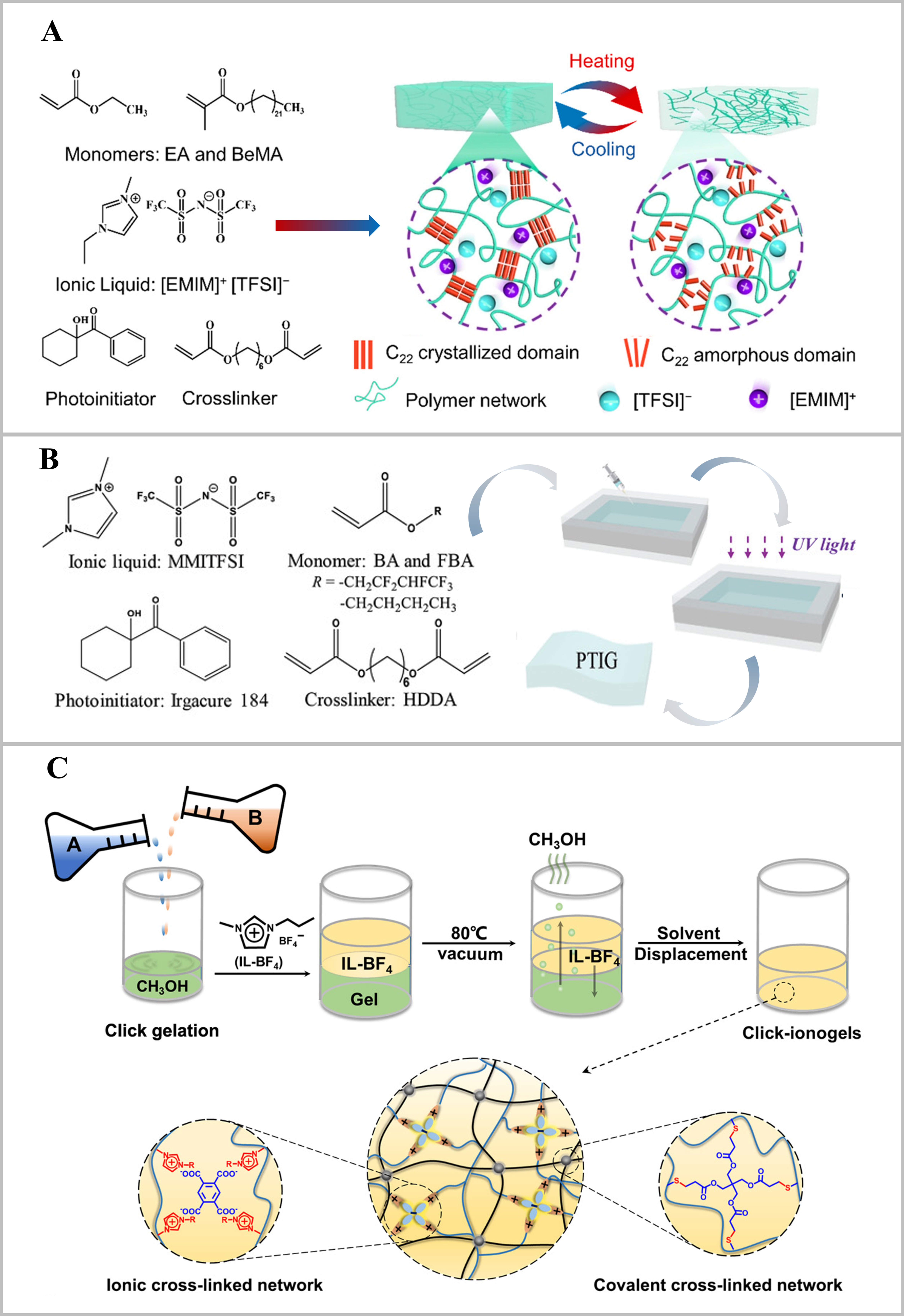
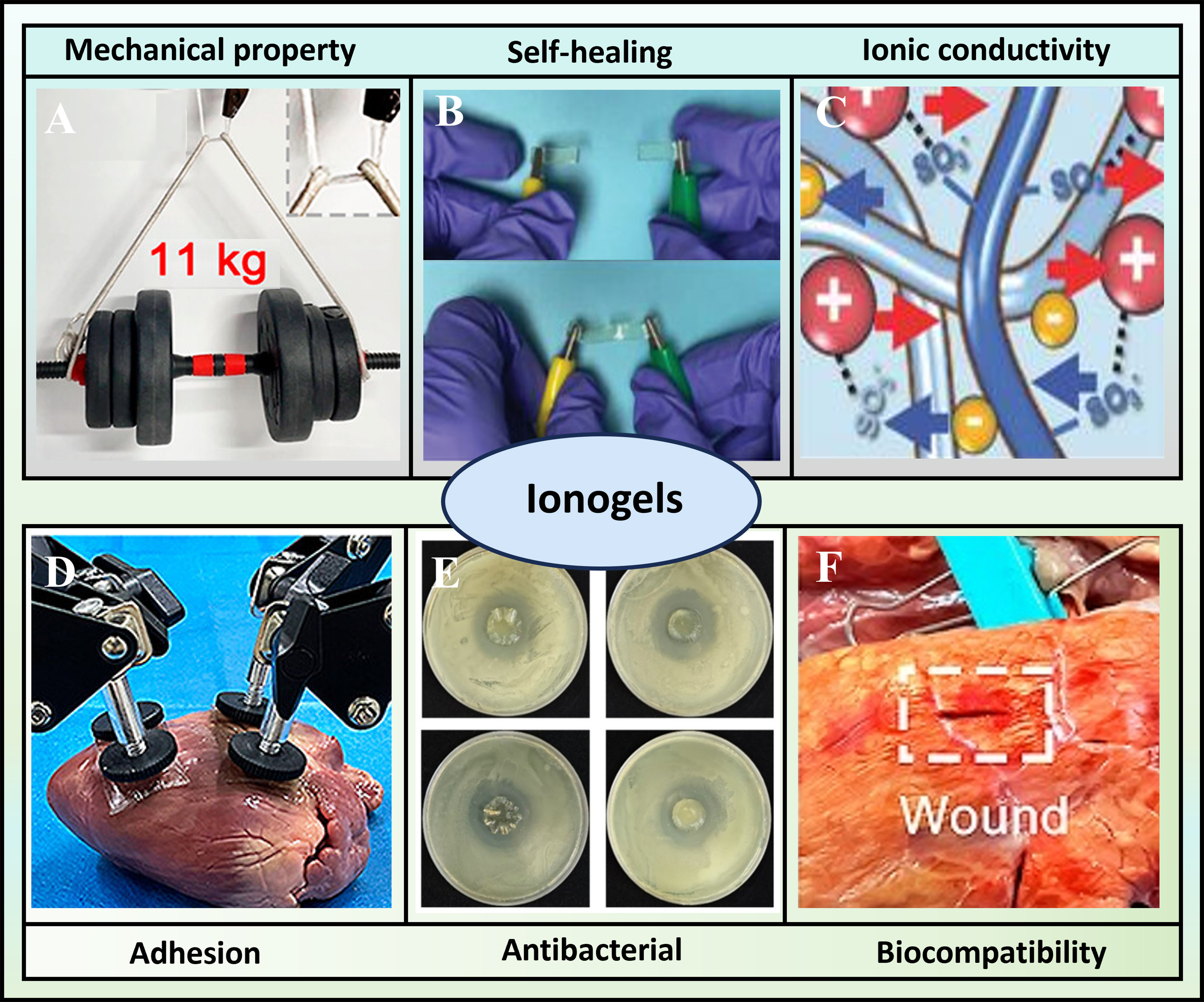
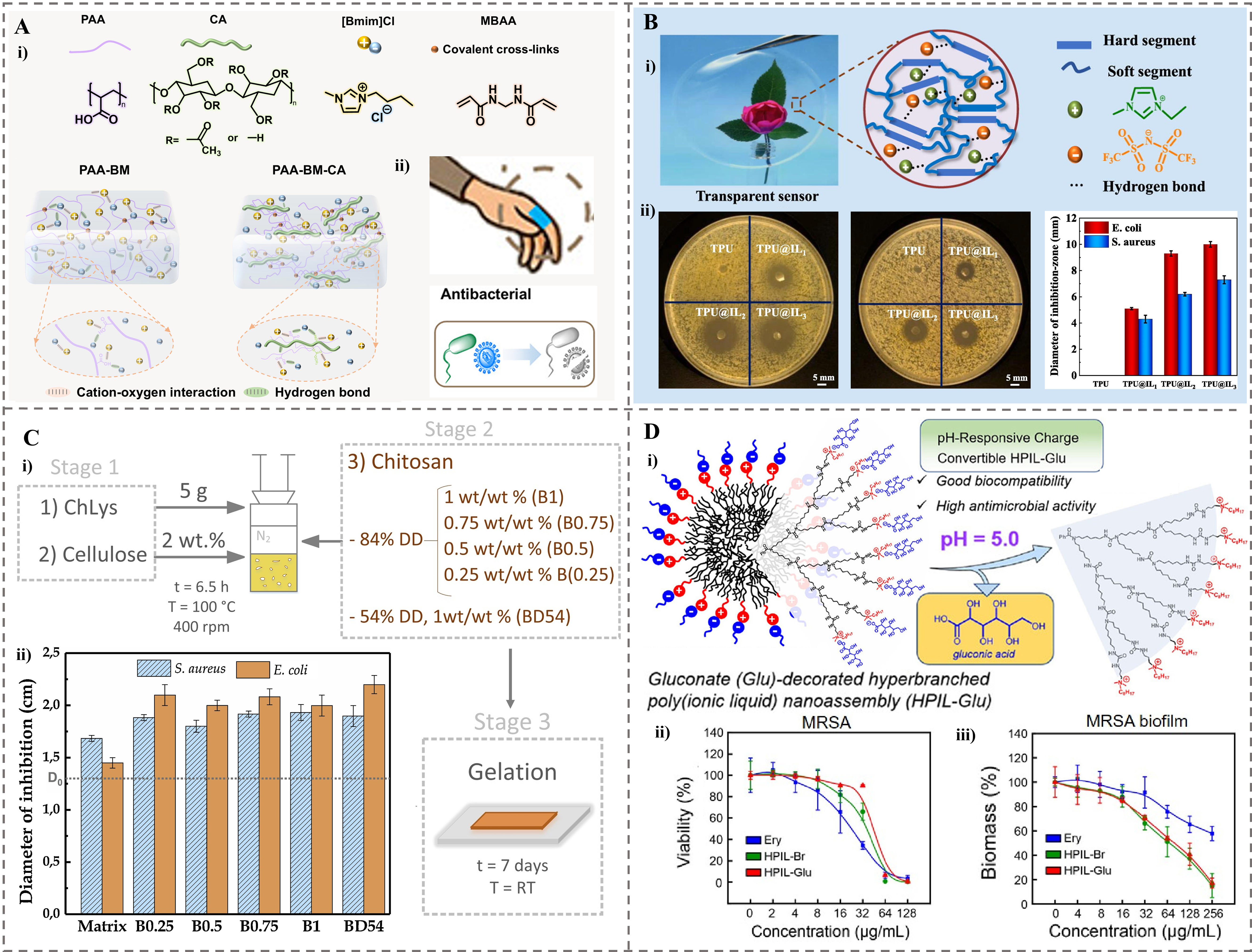
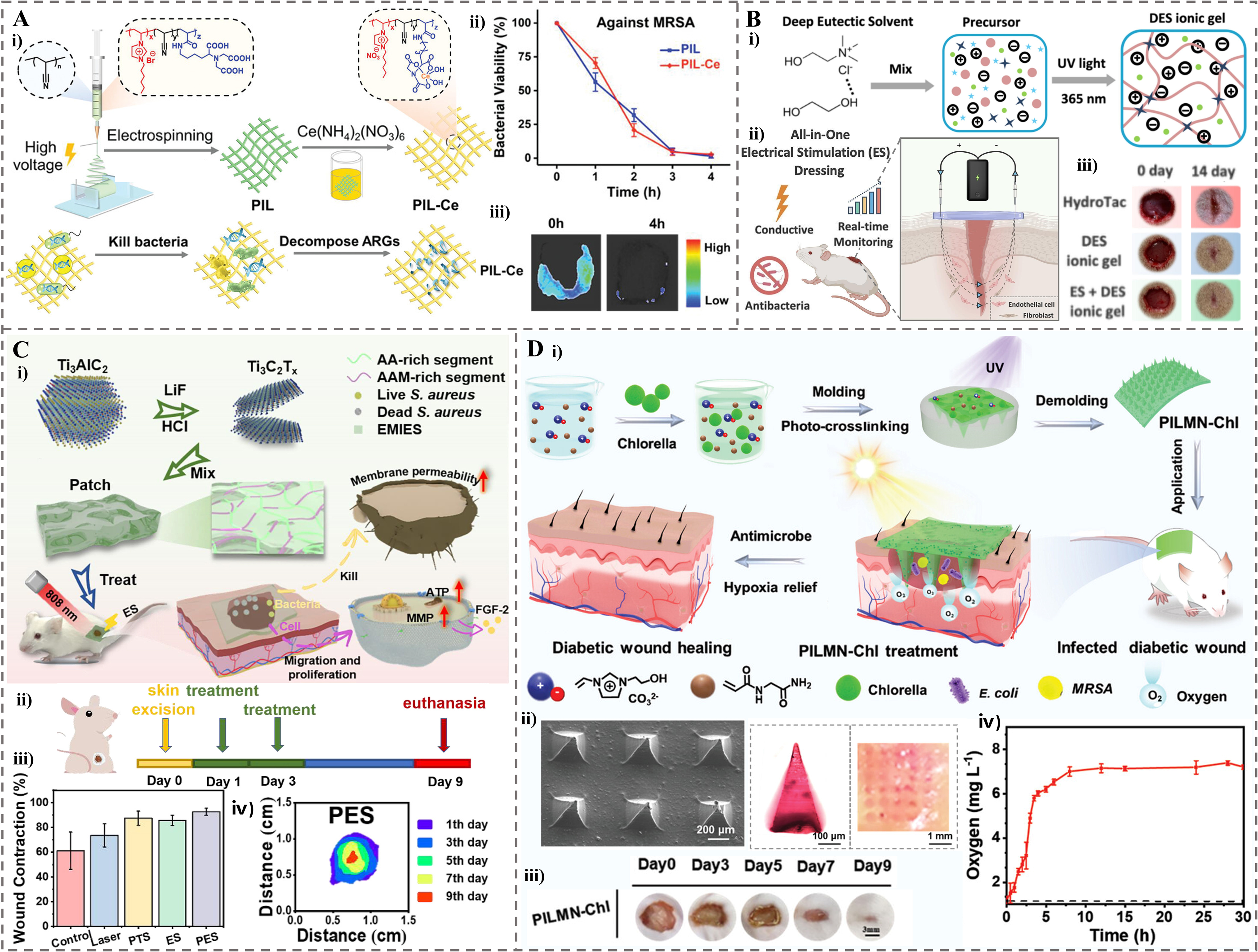
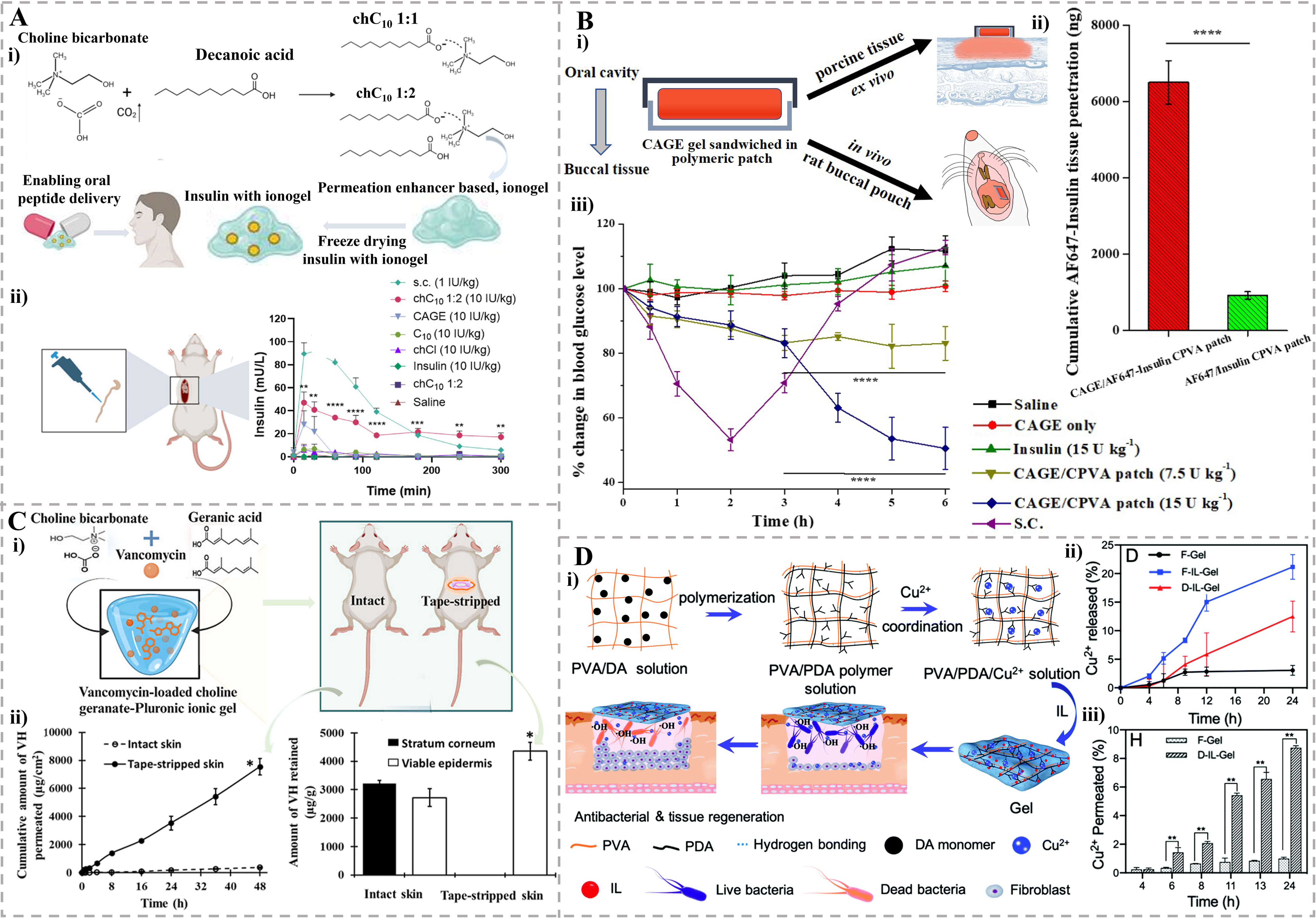
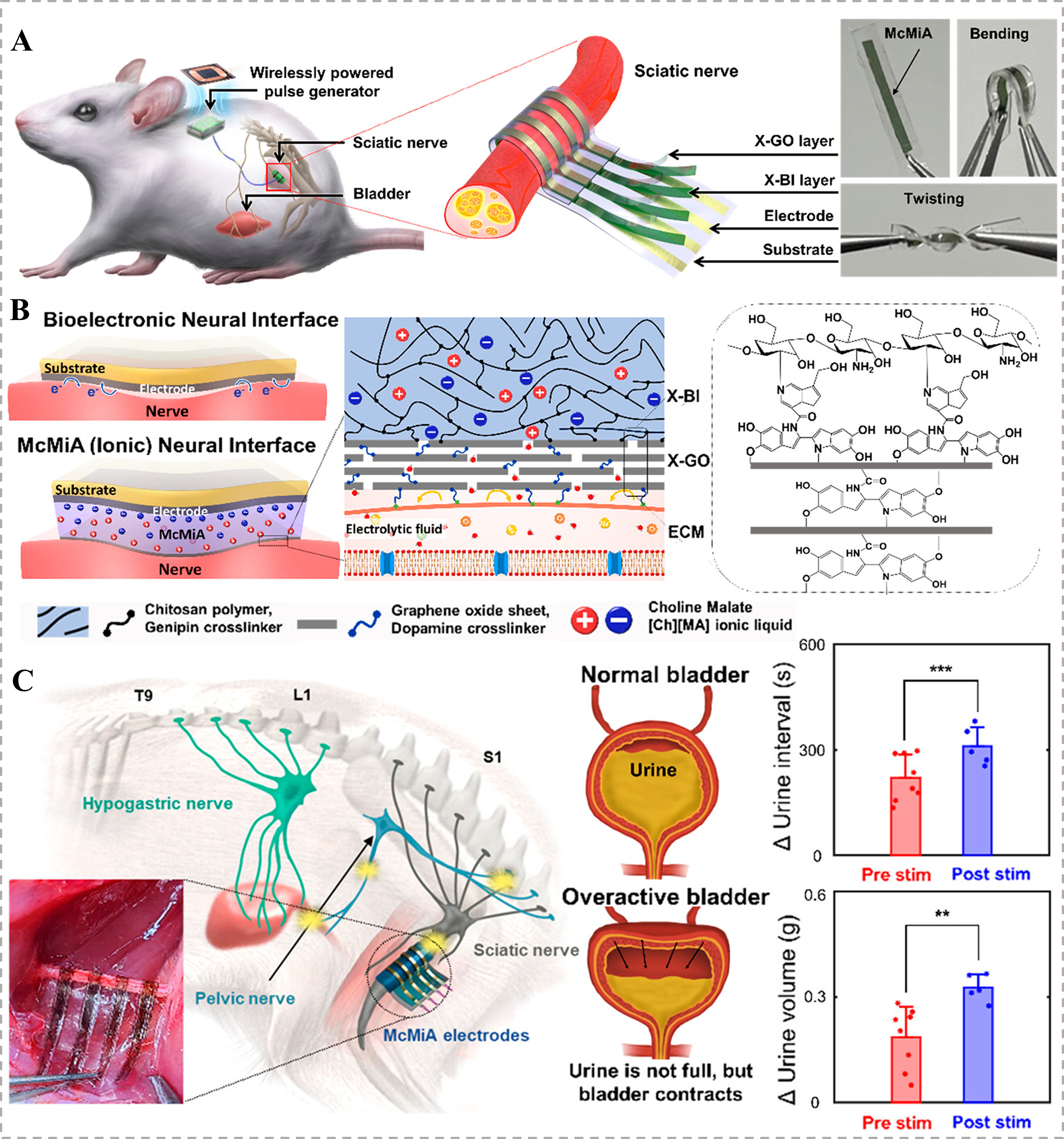
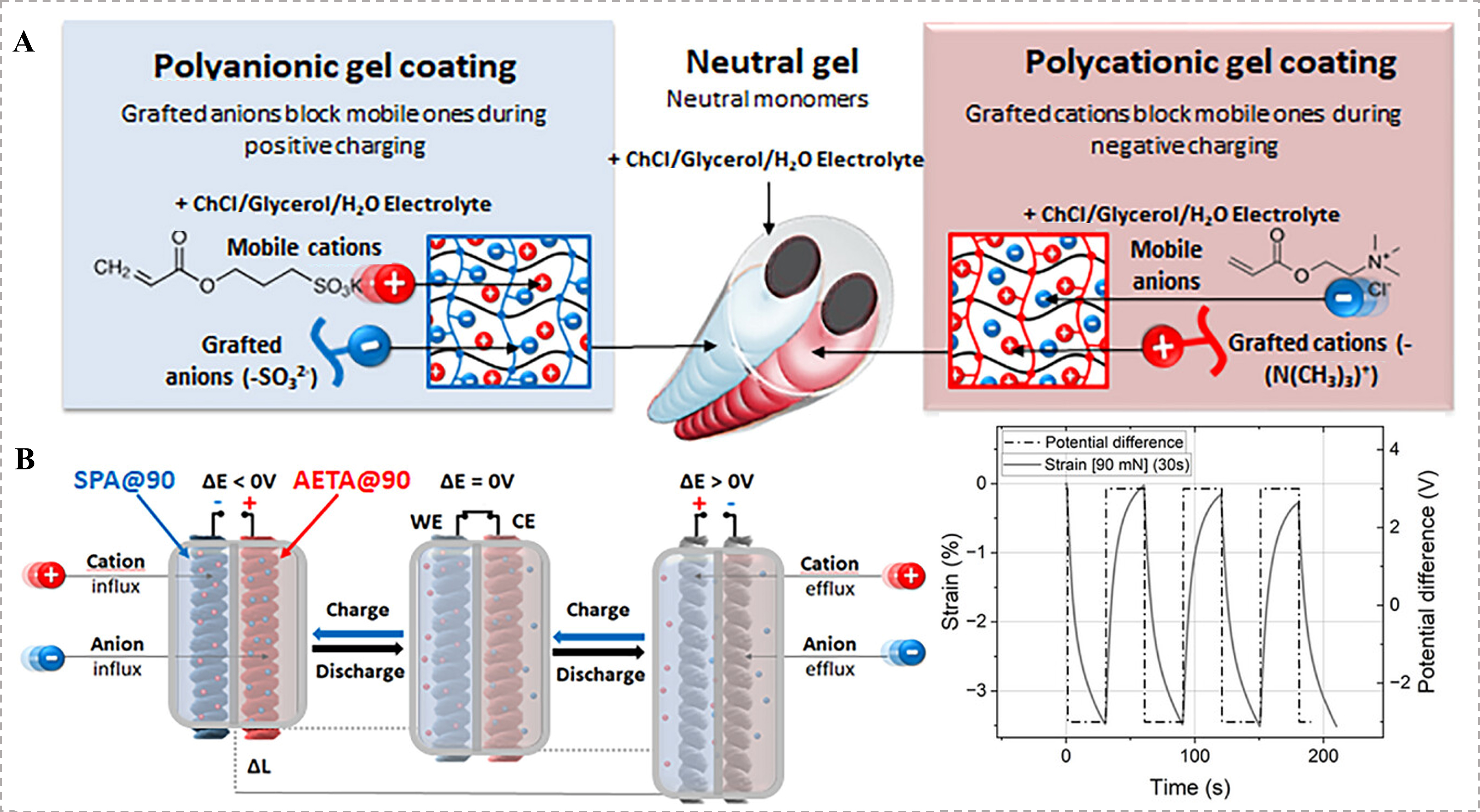
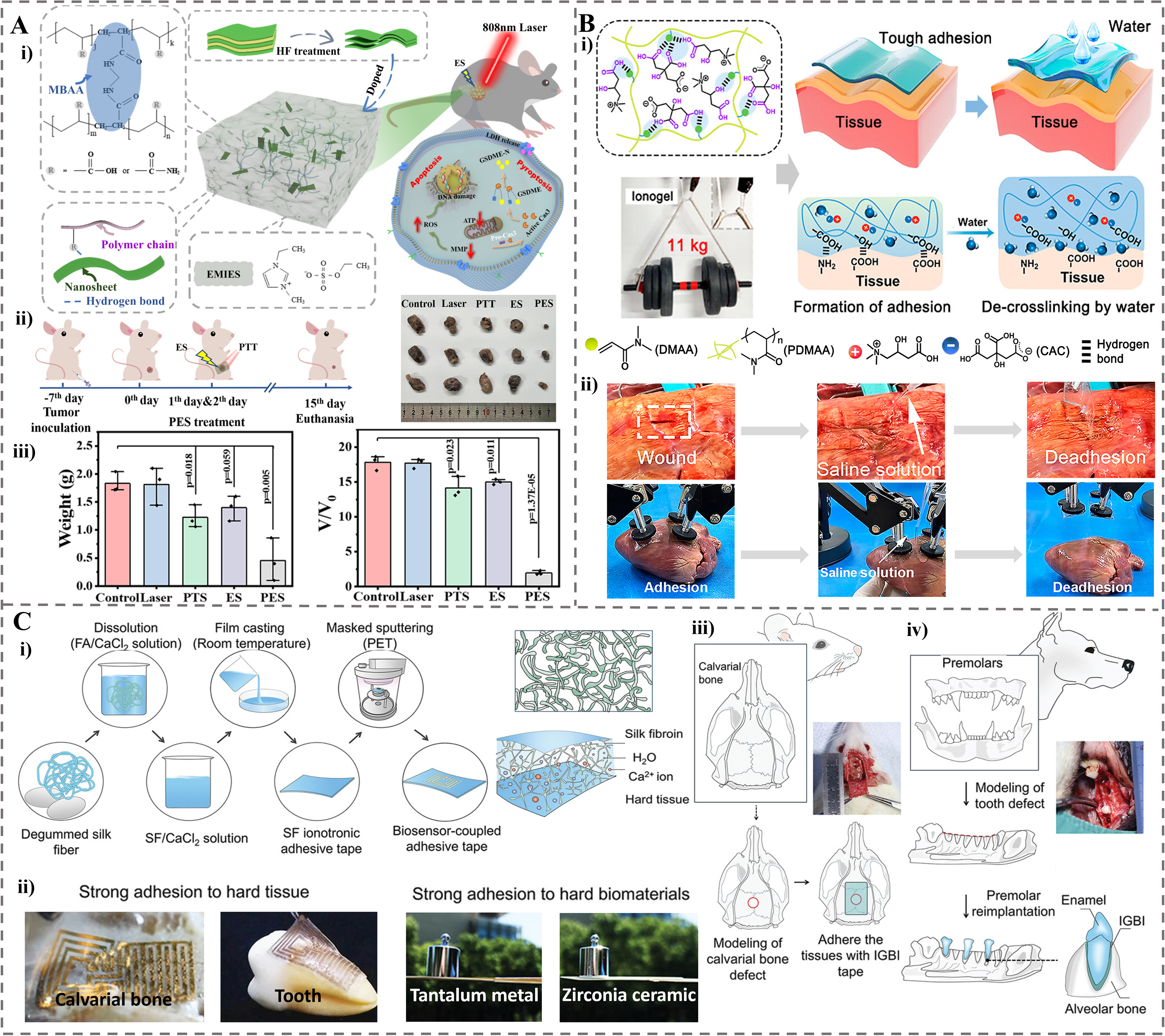












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].