Exploring Ru/CeO2 catalysts supported by ceria MOF-derived materials for improved ammonia synthesis and decomposition efficiency at mild conditions
Abstract
Ammonia (NH3) plays a crucial role in global agriculture and the development of emerging hydrogen energy systems. However, developing highly efficient catalysts for NH3 synthesis under mild conditions remains a significant challenge due to kinetic and thermodynamic limitations. In this study, we report a Ru/CeO2 catalyst synthesized using a metal-organic framework (MOF)-derived strategy, which allows for simultaneous control over the morphology of CeO2 (nanorod shaped), the concentration of oxygen vacancies, and the dispersion of Ru. The optimized catalyst containing 0.5 wt.% Ru delivered an impressive NH3 synthesis rate of 4,665 μmol gcat-1 h-1 at 400 °C and 50 bar, while maintaining excellent stability for 45 h of continuous operation. In addition, it achieved a high NH3 conversion of 93% at 550 °C and 1 bar. Notably, its performance surpasses that of conventional Ru-based catalysts by nearly twofold when normalized to the Ru loading. Comprehensive characterizations, including in situ X-ray photoelectron spectroscopy, Raman spectroscopy, and scanning transmission electron microscopy, reveal the formation of abundant oxygen vacancies, sub-nanometer Ru clusters, and strong metal support interaction. These factors collectively enhance the activation of N2 and its hydrogenation. This study highlights the effectiveness of MOF-templated defect engineering in developing robust Ru/CeO2 catalysts and provides valuable insights into structure-performance relationships. The dual functionality in both NH3 synthesis and decomposition highlights the potential of this approach for energy-efficient NH3-based energy systems.
Keywords
INTRODUCTION
Ammonia (NH3) is a vital component in fertilizers and various chemicals[1]. Its potential as a renewable energy carrier, thanks to its substantial hydrogen storage capacity (17.6 wt.%) and ease of storage and transportation[2], has recently been recognized. However, the current NH3 synthesis process, which requires high pressure and temperature (400-500 °C, 20-30 MPa)[3], consumes a significant portion (1%-2%) of global energy annually[4,5]. The high associated costs underscore the need for technological advancements and scale-up of synthesis processes to enhance the NH3-based economy[5-7]. Therefore, it is imperative to develop a sustainable process for NH3 production, with the primary challenge being the development of highly active and stable catalysts at mild conditions.
Ceria-based catalysts have demonstrated remarkable efficacy, specificity, and enduring stability across numerous catalytic reactions, marking significant advancements in ceria catalysis technology[8]. Recently, CeO2 has emerged as a prime candidate for supporting Ru catalysts in NH3 synthesis, owing to its distinct oxygen storage capacity, Ce4+/Ce3+ redox properties, electronic characteristics, and thermal resilience[9-12]. Variations in ceria's morphology, size and heteroatom doping typically impact the formation energy (EOv) of oxygen vacancies, thus influencing catalytic activity. Additionally, the electronic environment of ceria plays a pivotal role in catalytic performance by altering the metal-support interaction effects[13]. Previous studies[14-18] have shown that the morphology of CeO2 strongly influences Ru dispersion, oxygen vacancy concentration, and the nature of Ru-Ce interactions, which in turn determine NH3 synthesis activity. For instance, Ru supported on CeO2 nanorods exhibited higher activity than nanocubes due to the formation of more oxygen vacancies and smaller Ru species, whereas larger Ru particles on nanocubes led to reduced efficiency[19]. Similarly, morphology-dependent variations in exposed facets and electronic structure of CeO2 have been linked to significant differences in catalytic performance. Despite these advances, the optimal configuration of Ru species on CeO2 surfaces for efficient NH3 synthesis remains unresolved. A major challenge lies in the structural sensitivity of Ru, where only specific active sites (e.g., B5-type step edge sites) are effective for N2 activation[20]. Conventional preparation methods often produce broad Ru particle size distributions and weak metal-support interactions, limiting atom utilization and stability. Although atomically dispersed Ru or sub-nanoclusters on oxide supports have shown promise[22]. Metal-organic frameworks (MOFs), which consist of metal ions or clusters coordinated by organic ligands, offer a promising solution due to their tunable attributes, such as pore size and geometry, metal composition, breathable framework[23,24]. MOF-derived supports have recently emerged as a powerful approach for designing advanced oxide-supported catalysts with precise control over structural and electronic properties[25]. During the thermal transformation of Ce-based MOFs, the ordered framework decomposes into highly crystalline CeO2 with preferentially exposed facets (e.g., {111}), while the release of organic linkers simultaneously generates abundant oxygen vacancies. These defects not only enhance the redox capability of CeO2 but also provide anchoring sites that stabilize sub-nanometer Ru species, thereby strengthening the Ru-Ce interfacial interactions. Moreover, the homogeneous distribution of metal precursors within the MOF lattice enables the formation of well-dispersed Ru nanoparticles with finely tuned size, preventing aggregation and maximizing metal utilization. Such synergistic regulation of morphology, oxygen vacancy concentration, and Ru dispersion is critical for optimizing N2 activation and hydrogenation pathways, thereby offering a promising route to high-performance catalysts for ammonia synthesis under mild conditions[26,27].
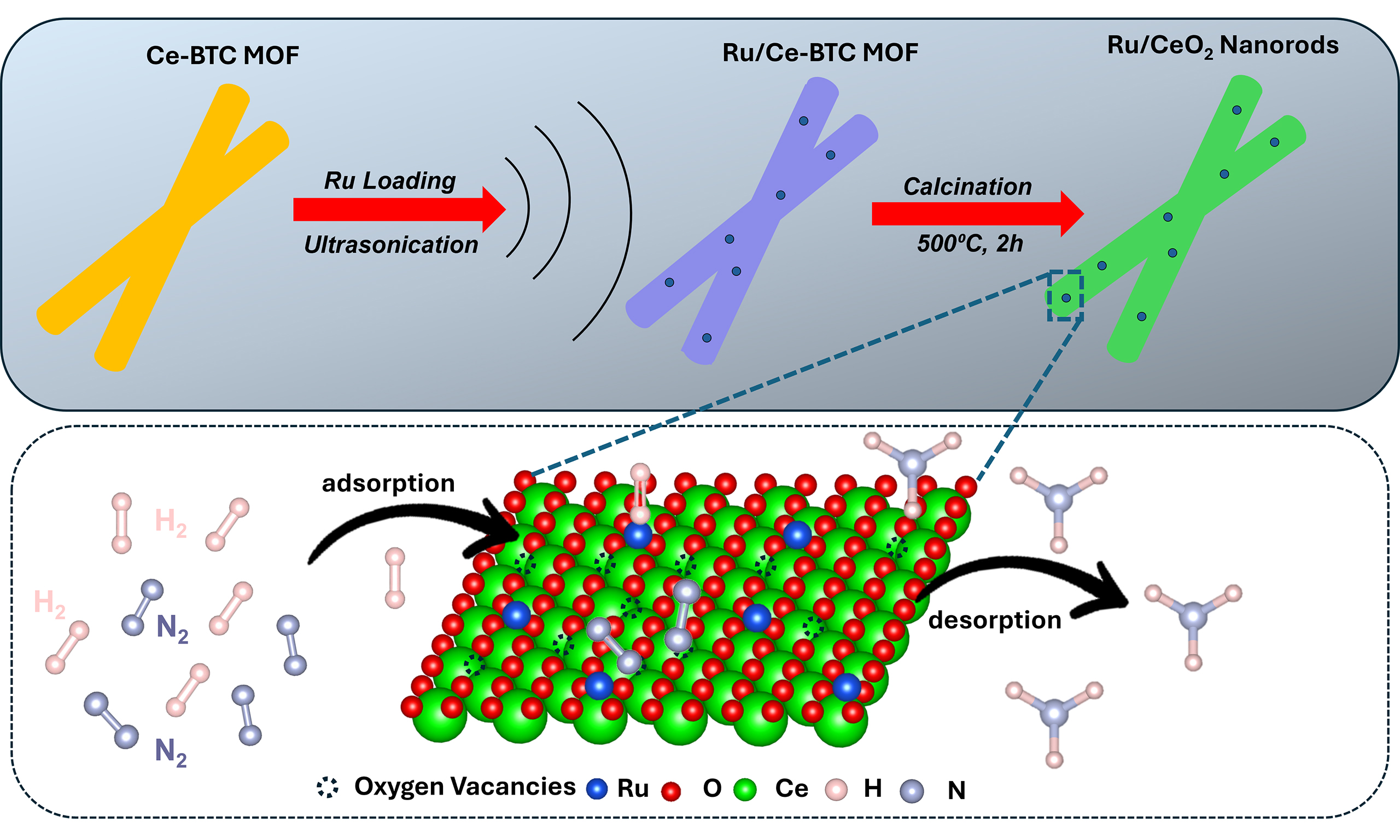
In this study, catalysts consisting of MOF-derived CeO2 as the support with varying loadings of Ru metal were developed. Initially, Ceria-MOF and Ru-MOF were prepared and subsequently calcined to yield the Ru/CeO2 support. The aim was to induce enhanced metal-support interactions. The MOF-derived strategy employed in this work achieves several key effects that have not been realized in previous studies. The complete physicochemical properties of the developed catalysts were explored using various in-situ and ex-situ characterization techniques, including Inductively Coupled Plasma-Optical Emission Spectroscopy (ICP-OES), X-ray Diffraction (XRD), Raman Spectroscopy, Scanning Electron Microscopy (SEM), N2-adsorption, High-Resolution Transmission Electron Microscopy (HR-TEM), H2 Temperature-programmed Reduction (TPR), H2-Temperature-programmed Desorption (TPD), X-ray photoelectron spectroscopy (XPS). The results of this study demonstrated that the catalysts had an optimal loading of 0.5 wt.% Ru for maximum NH3 formation rate (4,665 µmol g-1 h-1 at 400 °C and 50 bar) was achieved at 10,000 mL g-1h-1 weight hourly space velocity (WHSV) along with a NH3 conversation of 93% at 550 °C, 1 bar.
EXPERIMENTAL
Materials
(Ce(NO3)3·6H2O > 99%), ruthenium (III) chloride hydrate (RuCl3·3H2O, > 99%), 1,3,5-benzenetricarboxylate (BTC), and ethanol (> 99%), were procured from Sigma-Aldrich and used without further purification; Deionized (DI) water.
Catalyst preparation
Ce-MOF was synthesized following a reported procedure[28] and subsequently calcined at 500 °C to obtain CeO2. Ru-MOF was prepared via a solvothermal method using RuCl3 and BTC. The XRD
Characterization
Trace elemental analysis was performed using a Thermo Scientific™ iCAP™ 7600 ICP-OES. Powder XRD was recorded on a X’Pert PRO (Cu Kα, 1.5406 Å, 45 kV, 40 mA) over 2θ = 5°-80° (0.02° step, 10 s/step) using a zero-background holder. Raman spectra were obtained using Witec Alpha 300 RA (532 nm). Surface area and pore distribution were measured on a 3Flex Micromeritics system at 77 K after 8 h degassing at 120 °C (BET and BJH methods). TEM, high-angle annular dark-field (HAADF)-scanning transmission electron microscopy (STEM), and elemental mapping were performed on an FEI TITAN Cs-corrected ChemiSTEM; post-catalysis HR-TEM was acquired on Titan 80-300 ST (300 kV). H2-TPR and H2-TPD were conducted on 70 mg of catalyst in a U-tube microreactor (AutoChem 2920). Ex-situ XPS was performed on ESCALAB Theta Probe (Al Kα, 1,486.6 eV), calibrated to C 1s (284.8 eV). In-situ XRD was carried out on an INEL EQUINOX 3000 with XRK900 reactor under H2/N2 (3:1) with stepwise heating to 200-700 °C (1 h per step) and cooling to room temperature (RT). In-situ high-resolution XPS was conducted on ULVAC-PHI Genesis 900 under H2/N2 (3:1) at RT, 700 °C (2 h), and 400 °C. Full experimental details and characterization data are provided in the Supplementary Materials.
Evaluation of catalytic performance
NH3 synthesis and decomposition assessment
NH3 synthesis was evaluated in a fixed-bed quartz tube reactor using catalysts diluted with SiC. Reactions were conducted at 400 °C, pressures of 10-50 bar, and a WHSV of 10,000 mL g-1 h-1 with an N2/H2 (1:3) feed. Catalysts were pre-reduced under H2/N2 at 800 °C. Helium was used as an internal standard, and outlet NH3 was quantified online using GC-TCD. NH3 decomposition was performed in the same reactor after reduction at 700 °C under H2/N2. Measurements were carried out at a WHSV of 24,000 mL g-1 h-1 over a temperature range of 350-550 °C, with NH3 as the feed gas and He as an internal standard. Each experiment was repeated thrice.
Stability of NH3 synthesis activity test
Catalyst stability was assessed under continuous NH3 synthesis conditions at 400 °C and 50 bar for 45 h following reduction at 800 °C. NH3 concentration in the outlet stream was continuously monitored to evaluate long-term performance. Each experiment was repeated thrice. Full experimental procedures and operating details are provided in the Supplementary Materials.
RESULTS AND DISCUSSION
Elemental composition and morphology of the prepared catalysts
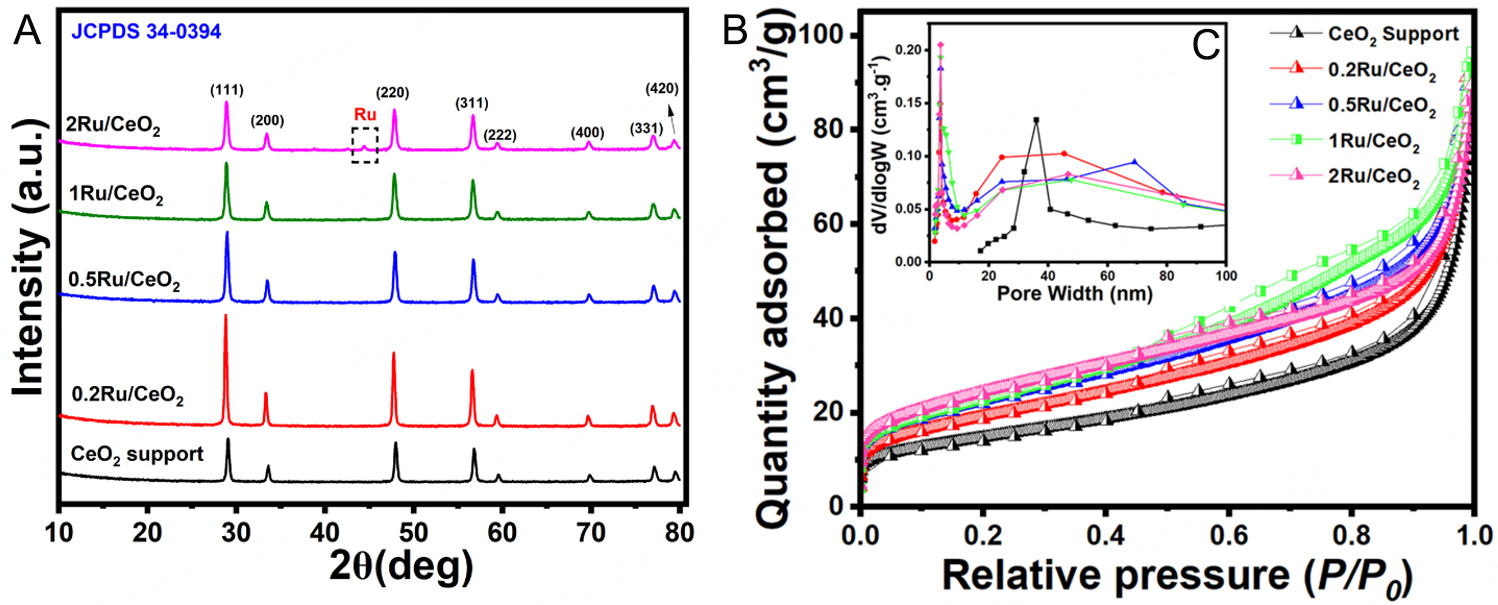
The Ru content in the as-synthesized catalysts, prepared via the MOF-derived method described in Section "Materials", was quantified using ICP-OES. The actual Ru compositions are provided in Table 1. Across all samples, the measured Ru content was slightly lower than the nominal loading, which is consistent with known losses due to the volatility of ruthenium oxides during thermal treatment[29]. Previous studies[28-30] have reported that CeO2 can interact strongly with RuOx species, effectively stabilizing them and minimizing their volatilization compared to other oxide supports. In line with this, the ICP-OES data reveal minimal Ru loss for the 0.2Ru/CeO2 and 0.5Ru/CeO2 catalysts, especially at lower loadings. This suggests that stronger metal-support interactions may occur at reduced Ru content, enhancing the retention of Ru during synthesis. SEM analysis [Supplementary Figure 3A-C] revealed that the Ce-MOF-derived CeO2 support consists of interwoven nanorods (100-200 nm in diameter, 1-2 µm in length) forming a fibrillar network[31]. After calcination at 500 °C, the rod-like morphology is largely preserved, indicating strong shape inheritance from the Ce-MOF precursor. Ru deposition (0.2-1 wt.%) does not alter the primary ceria morphology; the nanorods remain cross-linked with open interparticle voids. At higher Ru loadings (0.5-1 wt.%), slight bundle thickening and local necking are observed, but no Ru agglomerates are detected, suggesting high Ru dispersion. The preserved open rod-based framework provides short diffusion paths, mechanical stability, and high external surface area, which are favorable for catalysis and consistent with previous reports[30-32].
Physicochemical properties of nRu/CeO2 catalysts (n = 0.2, 0.5, 1, and 2)
| Catalyst before reaction | Ru (wt.%) experimental | Rua (wt.%) ICP-OES | 2θ (111) | d-spacing (Å)b | Lattice constant a (nm) | Crystallite size (nm)c | Surface area (m2/g) | Pore volume (cm3/g) | Pore diameter (nm) | Intensity ratio (I(598)/I464+1168)d |
| CeO2 support | - | - | 29.07 | 3.069 | 0.5316 | 23.9 | 48 | 0.13 | 11 | 1.02 |
| 0.2Ru/CeO2 | 0.2 | 0.22 | 28.81 | 3.096 | 0.5363 | 28.0 | 61 | 0.14 | 9 | 1.35 |
| 0.5Ru/CeO2 | 0.5 | 0.35 | 28.97 | 3.080 | 0.5334 | 21.8 | 72 | 0.14 | 8 | 1.64 |
| 1Ru/CeO2 | 1 | 0.41 | 28.9 | 3.087 | 0.5347 | 21.0 | 74 | 0.15 | 8 | 1.75 |
| 2Ru/CeO2 | 2 | 0.79 | 28.8 | 3.097 | 0.5365 | 20.5 | 78 | 0.13 | 7 | 1.84 |
Structural and textural study of Ce MOF-derived supported catalysts
Supplementary Figure 4 displays the XRD patterns of the as-prepared CeO2 supports and their corresponding nRu/CeO2 catalysts. Figure 1A shows the XRD patterns of the ex-situ reduced (under H2/Ar at 700 °C) catalysts, revealing the characteristic planes of CeO2 as observed for the fresh catalysts with enhanced crystallinity across the board. The diffraction peaks observed at 28.7°, 33.3°,47.7°, 56.5°, 59.4°, 69.7°, 76.9°, and 79.3° correspond to the CeO2 (111), (200), (220), (311), (222), (331), (420), and (422) planes, respectively. However, when compared, it was noted that crystallinity decreases as the Ru loading increases, along with slight peak broadening. The decrease in crystallinity with increased Ru loading is often due to the introduction of structural disorder (defects), formation of amorphous phases, or interference with the host material's crystal lattice[33,34]. These effects are typically more pronounced at higher loadings, where the influence of Ru on the material's structure becomes more significant, suggesting that the doping of Ru species could suppress the crystallinity of CeO2, probably due to the substitution of Ce4+ (0.97 Å) in CeO2 by Ru ions with a smaller radius (< 0.7 Å)[35]. The crystallite size of fluorite CeO2 support and nRu/CeO2 catalysts were in the range of 21-28 nm. The calculated lattice parameters for CeO2 support and the Ru-loaded CeO2 catalysts were 5.316 Å (CeO2), 5.3363 Å (0.2Ru/CeO2), 5.334 Å (0.5Ru/CeO2), 5.347 Å (1Ru/CeO2), and
In-situ XRD (structural properties)
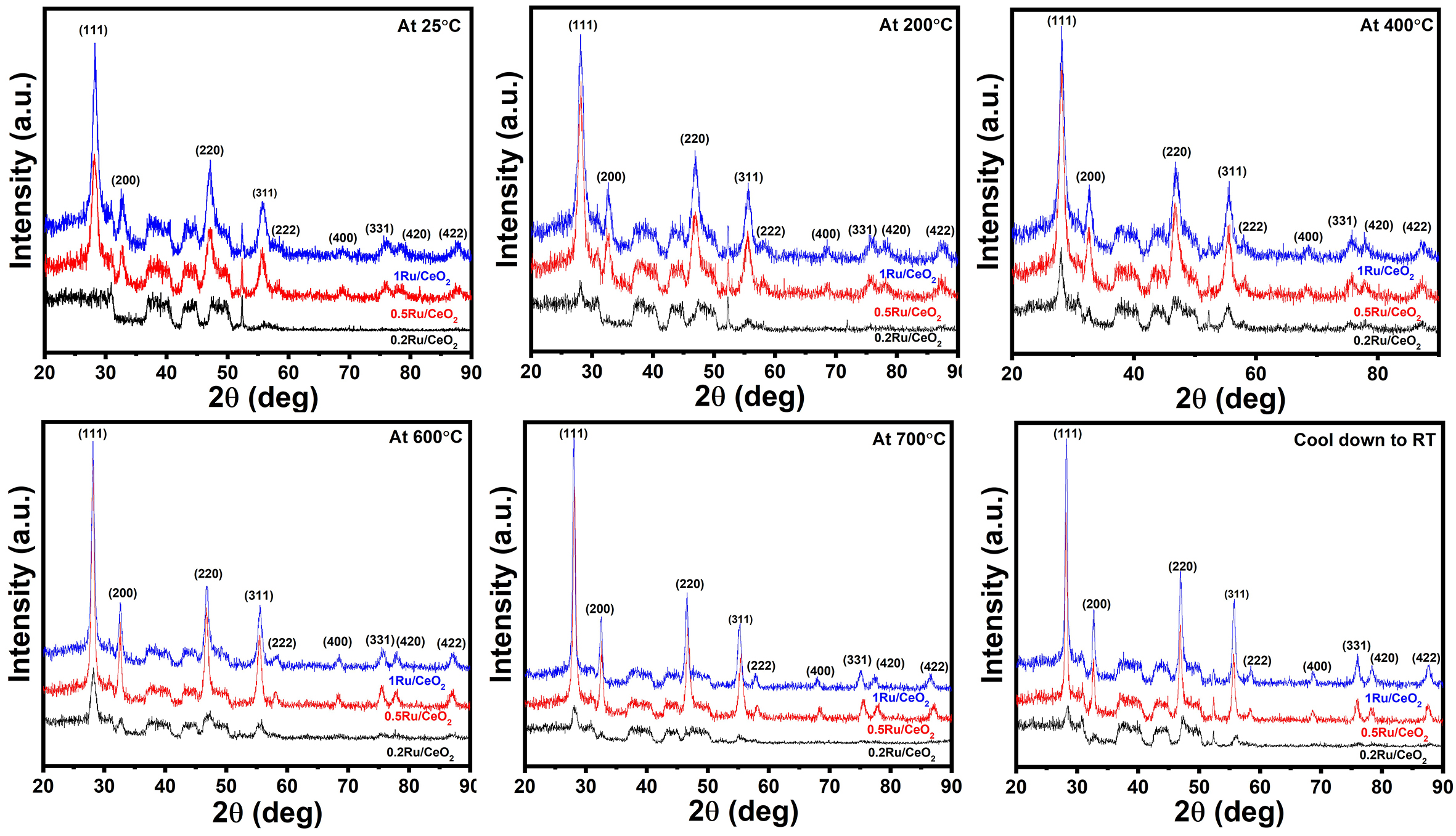
For the nRu/CeO2 catalysts, XRD analysis was conducted under controlled reduction conditions to monitor structural changes in the 200-800 °C temperature range. The initial XRD measurement was performed at RT. The catalyst was then heated up to 200 °C and held for 1 h under a reducing gas mixture atmosphere (H2/N2 = 3:1), followed by the acquisition of the XRD measurement. This process was repeated sequentially at 400, 600, and 700 °C, with each temperature maintained for 1 h before collecting the XRD measurements. After the final step at 700 °C, the catalyst was cooled down to RT under the same reducing atmosphere, and a final XRD analysis was performed to assess any post-reduction structural changes. As observed from Figure 2, the XRD peaks of the catalysts exhibit no changes in the characteristic CeO2 diffraction peaks under atmospheric conditions or with increasing temperatures during reduction atmosphere, apart from an improvement in crystallinity. Notably, no Ru-related peaks are observed in the in-situ XRD patterns. This absence is attributed to the exceptionally small Ru particle size, determined to be less than 1.5 nm by HR-TEM, which falls below the detection limit of XRD analysis. Additionally, HR-TEM analysis (discussed below) confirmed the uniform dispersion of Ru nanoparticles across the catalyst surface, further explaining the absence of distinct Ru peaks in the in-situ XRD results.
Raman studies
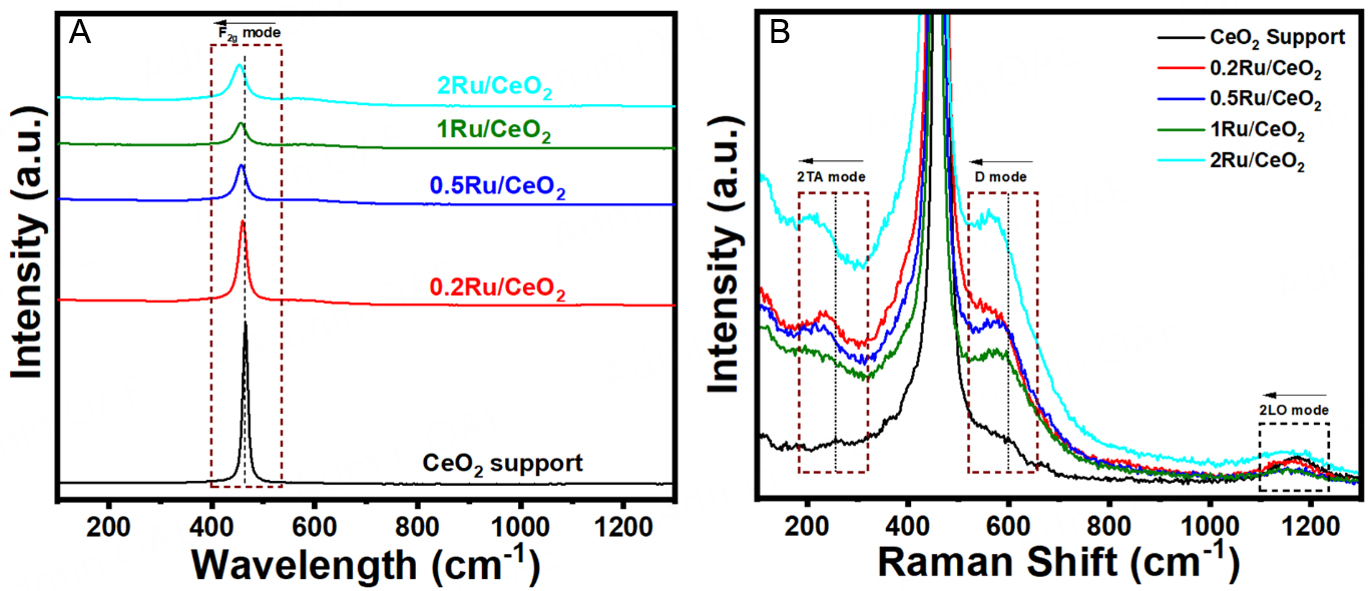
Oxygen vacancies (OVs) are widely recognized as crucial descriptor for catalytic activity, as they not only lower the activation energy barrier for N2 adsorption but also serve as active reaction sites. To investigate these defect sites, Raman spectroscopy was employed on ex-situ reduced (under H2/Ar at 700 °C) catalysts, as illustrated in Figure 3. The Raman spectra of CeO2 primarily feature a prominent F2g band at 465.3 cm-1, characteristic of the fluorite phase. Additionally, weaker bands are observed at 257, 598, and 1,172 cm-1, which are attributed to the second-order transverse acoustic (2TA) mode, defect-induced (D) mode, and second-order longitudinal optical (2LO) mode, respectively[47-49]. A sharp and symmetric F2g band at
Reduction behavior
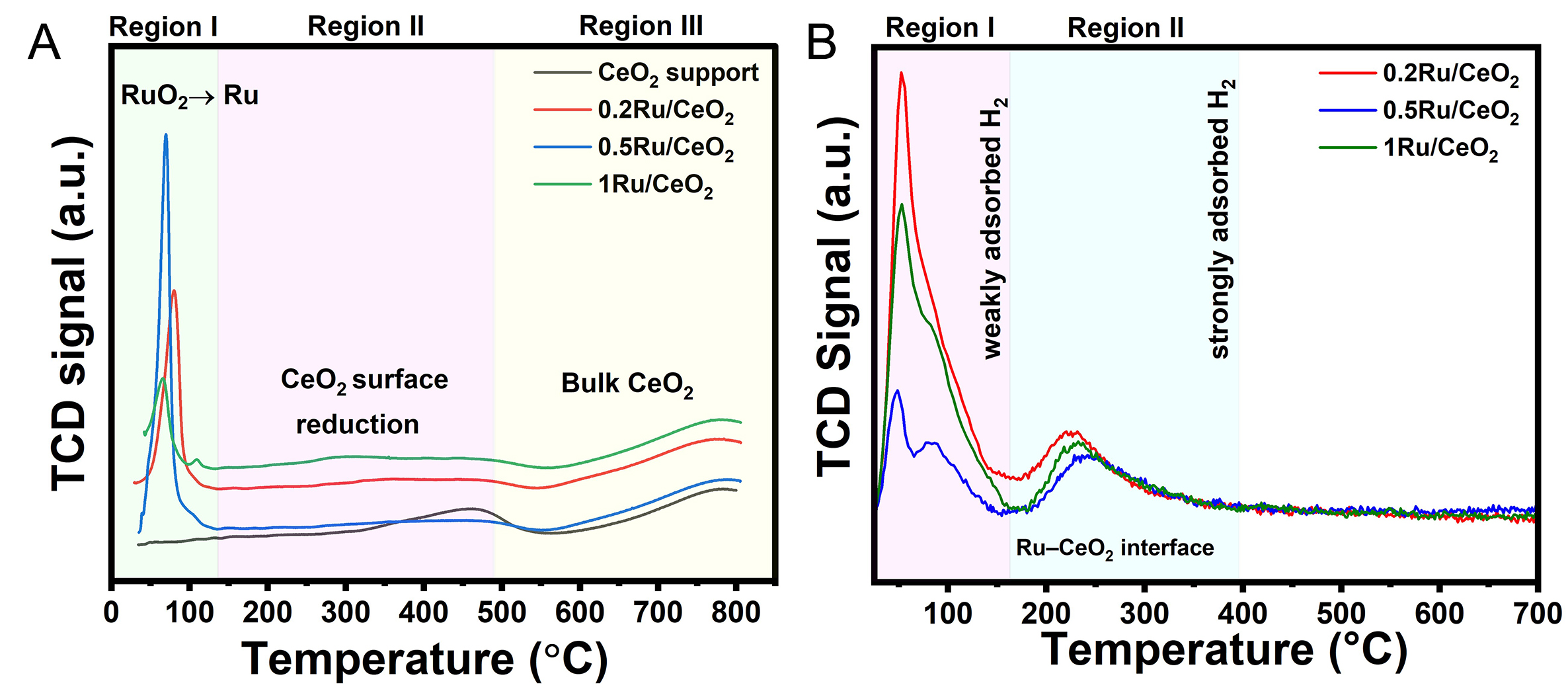
To gain deeper insight into the metal-support interactions and redox characteristics of the synthesized catalysts, H2-TPR was conducted over the 35-800 °C temperature range of [Figure 4A]. H2-TPR profiles of nRu/CeO2 catalysts exhibit a prominent low-temperature reduction peak in Region I
Metal dispersion
The H2-TPD analysis of the nRu/CeO2 catalysts shown in Figure 4B revealed two distinct regions. Region I corresponds to the weakly adsorbed H-species on highly dispersed Ru nanoclusters with two different H adsorption sites observed: one on Ru metallic sites of varying sizes. Region II corresponds to the strongly adsorbed dissociative H-species that are taken up by the Ru-CeO2 metal-support interface, which is closely associated with oxygen vacancies on the CeO2 support. In the higher temperature range (150-350 °C), the peak associated with strongly adsorbed hydrogen on Ru-CeO2 interfaces or O vacancies showed a slight increase in intensity with higher Ru loading. This suggests the increase in O vacancies along with enhanced hydrogen spillover from Ru to CeO2 with increased Ru loading, leading to more hydrogen being stored and subsequently desorbed from these interfacial sites. The crystallite sizes [Supplementary Table 1] obtained from the H2-TPD analysis are provided in Supplementary Materials.
Ex-situ XPS for surface analysis
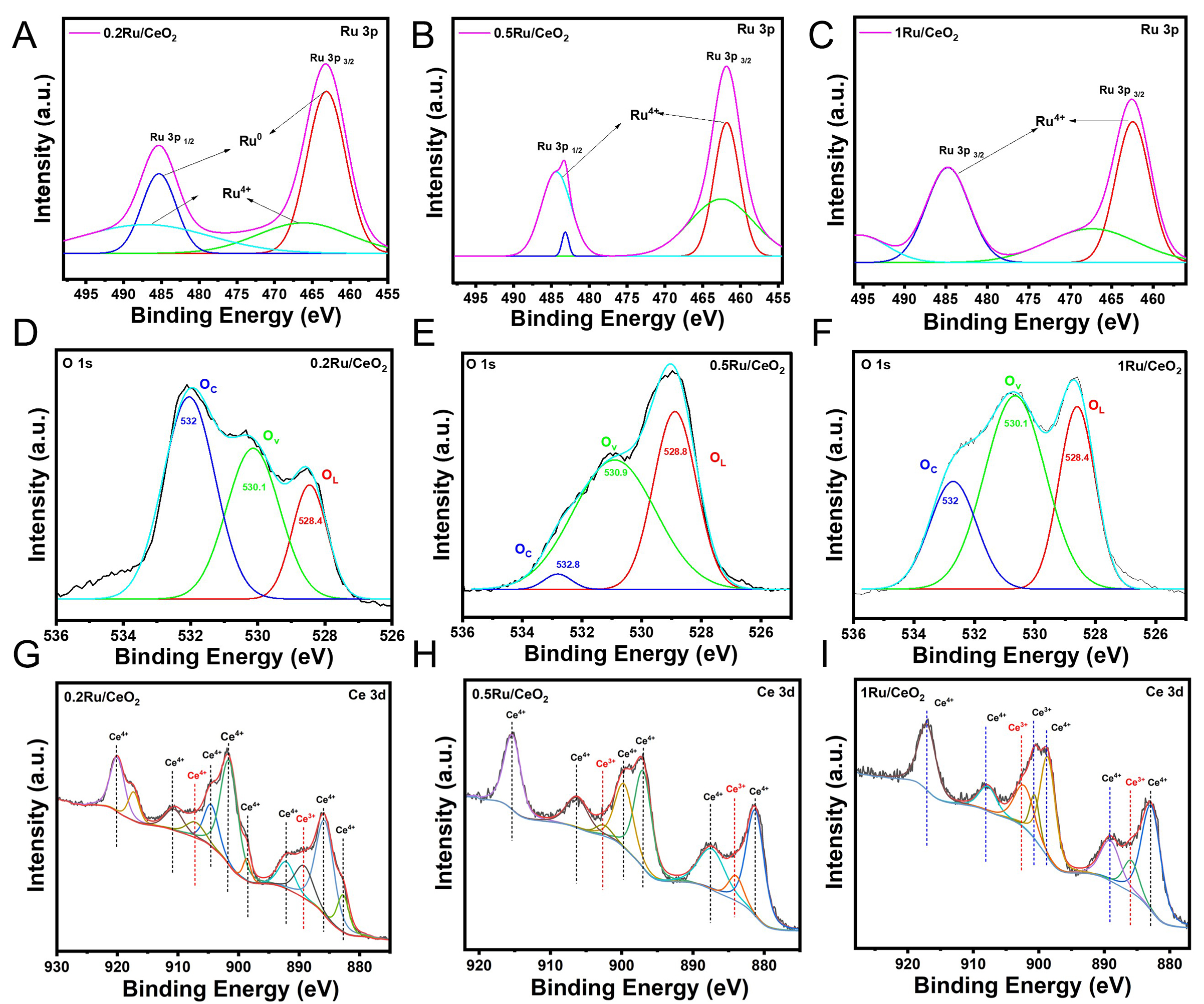
To gain a clearer understanding of the oxidation states and local environment of Ru species, XPS was performed on the catalysts following ex-situ reduction in H2/Ar at 700 °C. This analysis primarily aimed to evaluate the extent of Ru reduction and the interaction between Ru and the CeO2 support. The Ru 3p peaks at 461.9 and 484.1 eV are indicative of metallic Ru, whereas those at 463.7 and 485.9 eV correspond to oxidized Ru in the form of RuO2, as previously reported by Wang et al.[70]. Deconvolution of Ru 3p signals was carried out using Shirley background correction, revealing distinct electronic environments for the different loadings, and is shown in Figure 5A-C. The 0.5Ru/CeO2 catalyst predominantly exhibited metallic Ru, with characteristic peaks at 461.8 and 484.3 eV. In contrast, 0.2Ru/CeO2 and 1Ru/CeO2 catalysts showed a noticeable shift toward higher binding energies (463.1/485.3 eV and 462.4/484.7 eV, respectively), suggesting a greater proportion of oxidized Ru species (Ru4+). This shift likely arises from variations in the chemical surroundings or electron transfer between Ru and the CeO2 support, thus driving the Ru oxidation state and of the SMSI phenomena between Ru and ceria support.
Figure 5. Deconvoluted XPS spectra of (A-C) Ru 3p, (D-F) O 1s, and (G-I) Ce 3d XPS core-level spectra of ex-situ reduced (under H2/Ar at 700 °C) nRu/CeO2 catalysts (n = 0.2, 0.5, and 1).
The O 1s XPS spectra [Figure 5D-F] revealed three distinct oxygen environments in all three catalysts under study. The peak at ~528.8 eV corresponds to lattice oxygen (OL), while those around ~530 and
The Ce 3d XPS spectra shown in Figure 5G-I display a complex pattern, characteristic of cerium oxide, and have been deconvoluted based on established literature assignments[72-74]. The spectral features at ~915.4, 906.4, 899.7, 897.1, 887.4, 881.3 eV correspond to the presence of Ce4+ oxidation states. In contrast, the peaks centered around 902.6 and 884.1 eV are attributed to the presence of Ce3+ species, indicating the partial reduction of cerium within the oxide lattice.
In-situ XPS (surface atomic%)
An in-situ XPS study was performed to examine the surface composition of the catalysts under different conditions. XPS spectra were collected for fresh samples at RT, after reduction in N2/H2 (1:3) at 700 °C for
The reduction process performed under the N2/H2 atmosphere significantly impacts the surface composition trends of the nRu/CeO2 catalysts as presented in Table 2. Initially, at 25 °C, the catalysts exhibit a lower atomic% of Ru and varying amounts of Ce and O, reflective of the as-synthesized state of catalysts before reduction. As the temperature increases to 700 °C (reduction condition), the Ru atomic percentage rises notably, indicating that the reducing environment promotes Ru migration toward the surface and enhances its dispersion on the ceria support. This trend is further corroborated by XPS analysis, which reveals a temperature-dependent increase in the surface Ru/Ce ratio accompanied by a decrease in the O/Ce ratio, evidencing Ru surface enrichment and partial reduction of CeO2 to CeO2-x[75]. The resulting Ru-rich, oxygen-deficient surface provides an optimal population of Ru-CeO2-x interfacial sites. The elevated temperature likely enhances the reduction of both Ru and CeO2, facilitating an interaction between metal and support that stabilizes Ru in a metallic form, which is often more catalytically active. After this step, when the temperature is reduced back to 400 °C (reaction condition), the atomic% of Ru remains almost similar with a slight reduction. This suggests that the stability of Ru on the surface is maintained after the reduction.
Surface compositions of nRu/CeO2 catalysts (n = 0.2, 0.5, and 1)
| Catalysts | Temperature ( °C) | Ru (atomic%) | Ce (atomic%) | O (atomic%) | O/Ce | Ru/Ce |
| 0.2Ru/CeO2 | 25 | 0.24 | 31.33 | 68.98 | 2.22 | 0.0076 |
| 700 | 0.30 | 34.39 | 65.31 | 1.89 | 0.0087 | |
| 400 | 0.30 | 34.44 | 65.26 | 1.89 | 0.0087 | |
| 0.5Ru/CeO2 | 25 | 0.35 | 34.27 | 65.39 | 1.90 | 0.0102 |
| 700 | 0.64 | 35.74 | 63.62 | 1.78 | 0.0179 | |
| 400 | 0.57 | 36.87 | 62.56 | 1.77 | 0.0154 | |
| 1Ru/CeO2 | 25 | 0.49 | 33.68 | 65.84 | 1.95 | 0.0145 |
| 700 | 1.09 | 36.38 | 62.53 | 1.72 | 0.0299 | |
| 400 | 0.82 | 35.49 | 63.68 | 1.79 | 0.0231 |
Moreover, oxygen vacancies (Ov)[76-78] are prevalent anionic point defects commonly observed in transition and f-block metal oxides. These vacancies typically arise in materials containing cations that can alternate between multiple oxidation states, such as Ce3+/Ce4+[77,79,80] and Ti3+/Ti4+[81]. The formation of Ov is often induced by high-temperature treatments[82] or under reducing environments[83], where lattice or surface O2- ions are removed alongside the reduction of metal cations, all while preserving the overall crystal framework. Such defect sites are particularly significant in the context of heterogeneous catalysis[77,79,80]. In the case of CeO2, which adopts a cubic fluorite structure (space group Fm
At ambient conditions (25 °C), the surface environment of the catalyst shows an O/Ce ratio close to 2, indicating a well-oxidized surface (where Ov population is expected to be low)[85]. However, under reducing conditions, the O/Ce ratio decreases to ~1.7 signifying the removal of O atoms from the lattice, thus leading to the formation of O vacancies (CeO2-x). In the case of 0.5Ru/CeO2, most Ov (O/Ce = 1.77) was observed at the reaction condition (400 °C) as compared to the rest of the catalysts. These vacancies play a critical role in enhancing catalytic performance by serving as active sites and improving the mobility of lattice oxygen for redox reactions[86]. Additionally, the removal of oxygen reduces Ce4+ to Ce3+, altering the electronic environment and influencing the adsorption and activation of reactants[87]. The formation of O vacancies also induces slight lattice distortions, which can enhance interactions with other species, such as Ru in the nRu/CeO2 catalysts[88]. This demonstrates the importance of the reducible nature of CeO2 in enabling its superior catalytic activity under reaction conditions as discussed below.
HR-TEM microstructural studies
HR-TEM was carried out on the reduced catalysts to examine their microstructure and elemental distribution under reaction conditions. The HRTEM images shown in Supplementary Figure 8 reveal that all three catalysts exhibit a rod-like structure with elongated particles. This suggests a distinct morphology of the nRu/CeO2 catalyst system, likely influenced by the MOF-derived preparation method.
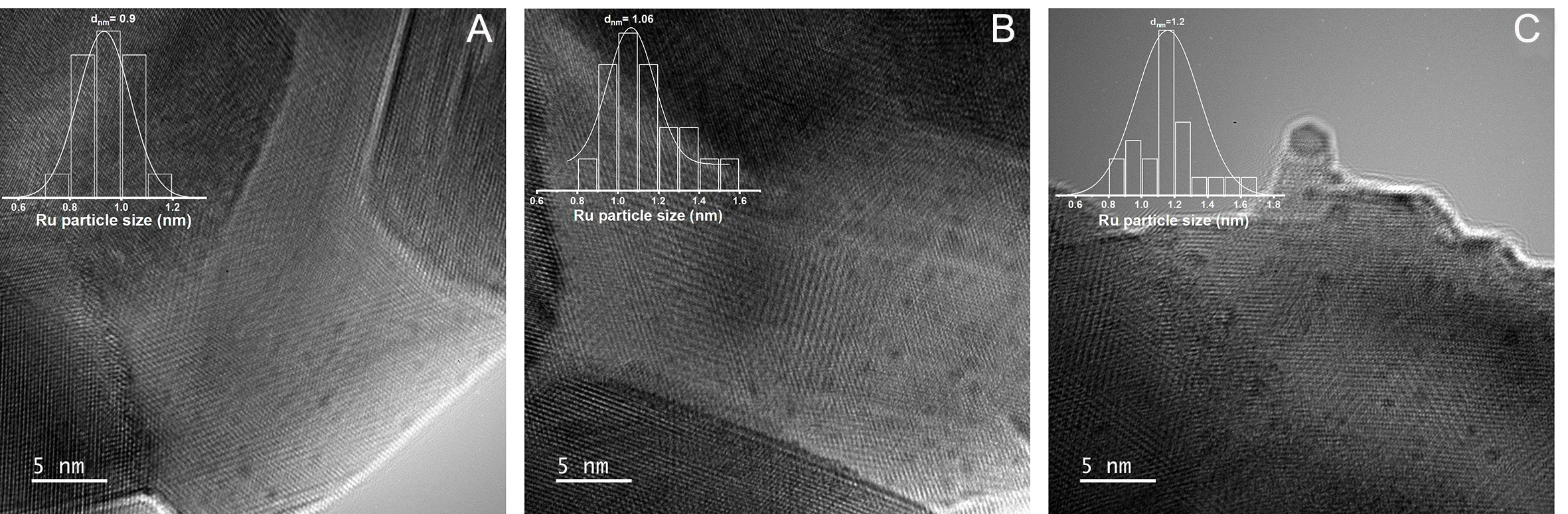
Figure 6A-C present the HRTEM images of the nRu/CeO2 catalysts. The inset in each image shows the Ru particle size distribution, which indicates an increase in particle size across the three catalysts. Specifically, the particle sizes range from 0.9 to 1.06 to 1.2 nm as the n values increase from 0.2 to 0.5, and then to 1, respectively. This indicates a clear relationship between Ru loading and particle size growth, as anticipated. Additionally, good dispersion of Ru was observed for all three nRu/CeO2 catalysts. However, at a Ru loading of 1 wt.%, slight agglomeration of Ru was observed, particularly at the edges of the surface. This agglomeration can be attributed to the high surface energy of Ru (~2.5-3.0 J/m2) at the edges, which promotes particle growth as a way to minimize surface energy (J/m2)[89]. Moreover, HR-TEM images of the reduced 0.5Ru/CeO2 catalyst [Supplementary Figure 9A-C] show lattice fringes indexed to the CeO2 (220) and (111) planes, with d-spacings of ~0.19 and ~0.32 nm, respectively. The corresponding FFT patterns (insets) are consistent with a retained CeO2 fluorite structure after reduction.
Figure 6. HR-TEM micrographs of (A) 0.2Ru/CeO2, (B) 0.5Ru/CeO2, and (C) 1Ru/CeO2. The inset contains particle size distribution.
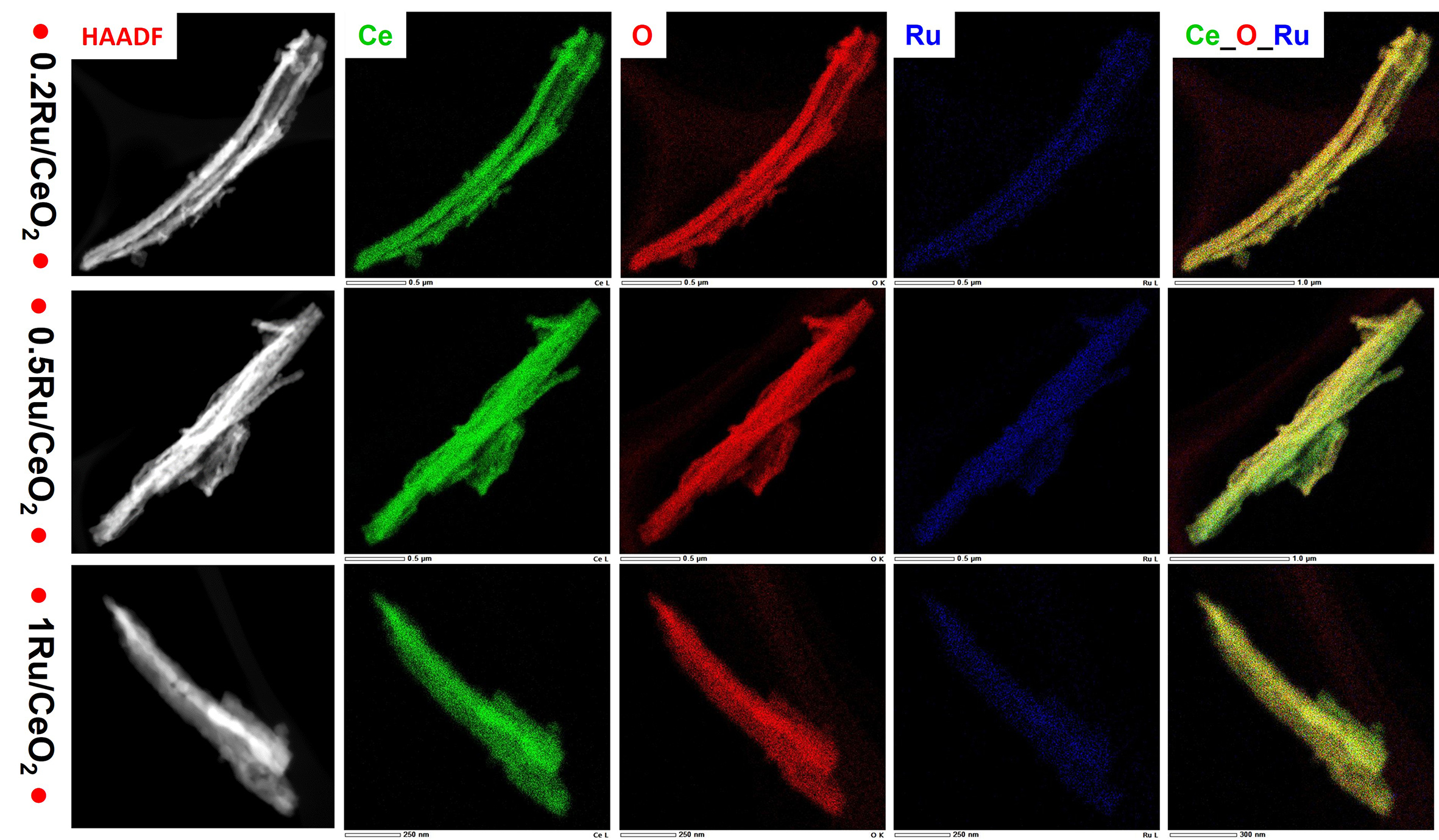
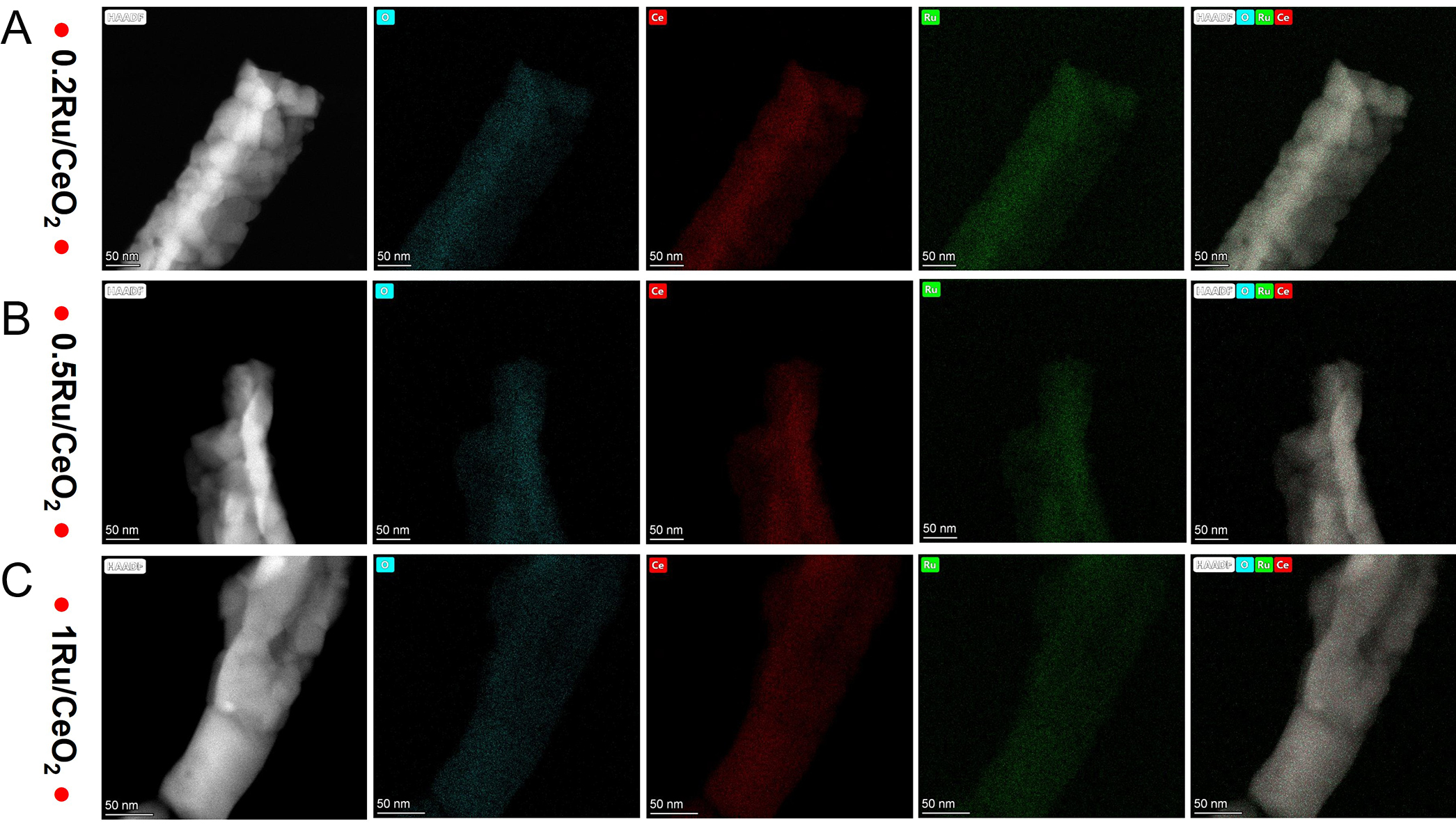
Elemental mapping [Figure 7] further confirmed the distribution of elements (Ce, Ru, and O) across the nRu/CeO2 catalysts. The mapping data showed that Ru is homogeneously dispersed throughout the catalyst, providing evidence of uniform distribution even at varying Ru loadings in the range studied. These findings highlight the interplay between Ru loading, particle size, and dispersion characteristics, which are critical for catalytic interface and performance afterall.
Figure 7. STEM-HAADF image and elemental mapping of the selected area of nRu/CeO2, n = 0.2, 0.5, and 1wt.%. Ce (Green), O (Red), Ru (Blue), Ce-O-Ru (GRB).
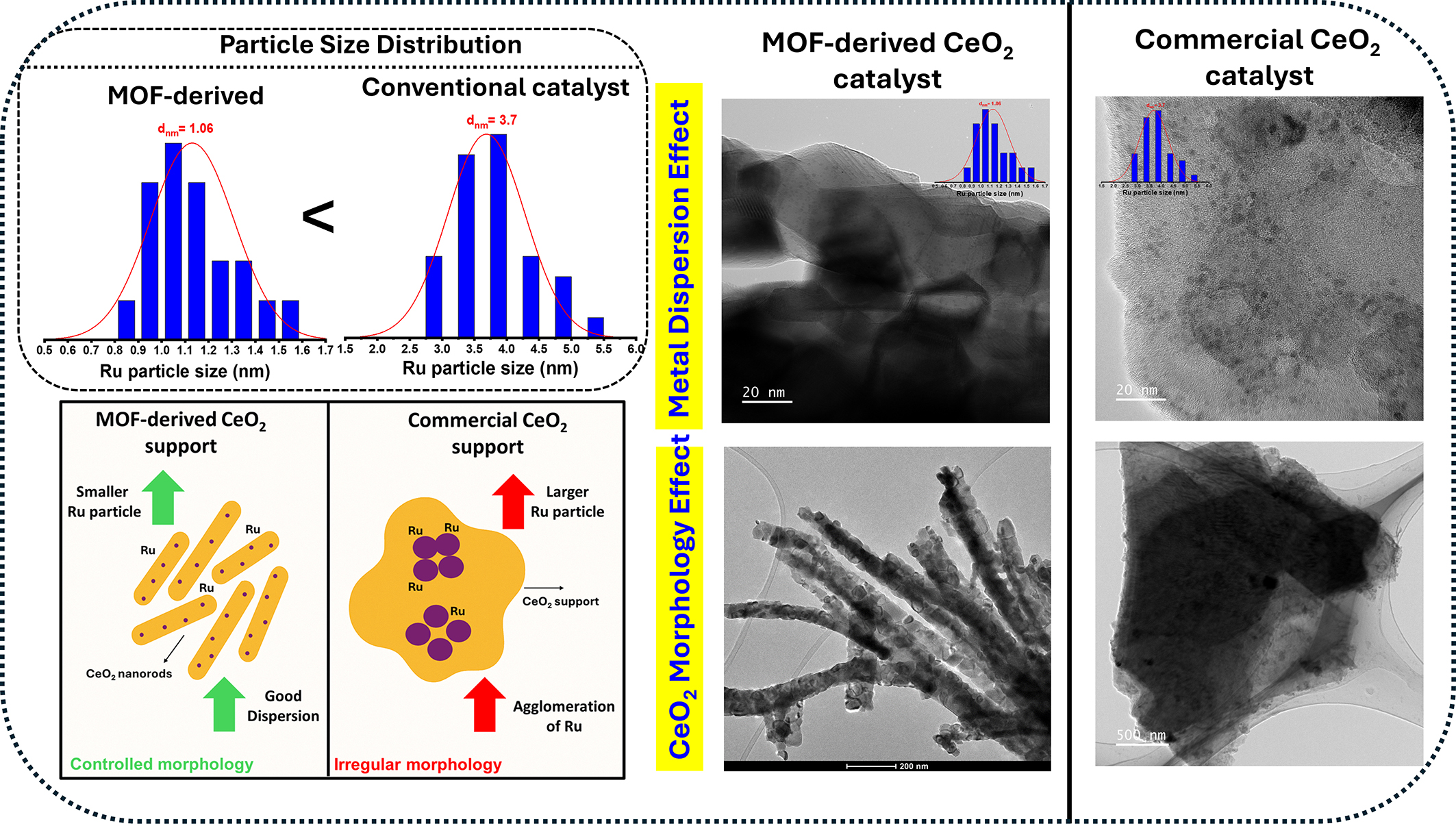
To critically evaluate the MOF-derived catalyst, Figure 8 compares HR-TEM images and Ru particle size distributions of 0.5Ru/CeO2 catalysts prepared using MOF-derived CeO2 and commercial CeO2 supports. The MOF-derived catalyst exhibits well-defined nanorod morphology and uniformly dispersed Ru nanoparticles with a smaller average size (davg ≈ 1.06 nm), whereas the commercial CeO2-based catalyst shows no distinct morphology and significantly larger, less uniform Ru particles (davg ≈ 3.7 nm). The nanorod structure formed via MOF templating enhances Ru dispersion due to higher surface area, abundant oxygen vacancies, and exposure of reactive {111} and {100} facets, which promote strong metal-support interactions and suppress Ru sintering. These features provide a higher density of active sites for N2 activation, favoring NH3 synthesis, consistent with previous reports highlighting the superior performance of Ru/CeO2 nanorods over other morphologies[90,91].
Catalysts’ assessment towards NH3 synthesis
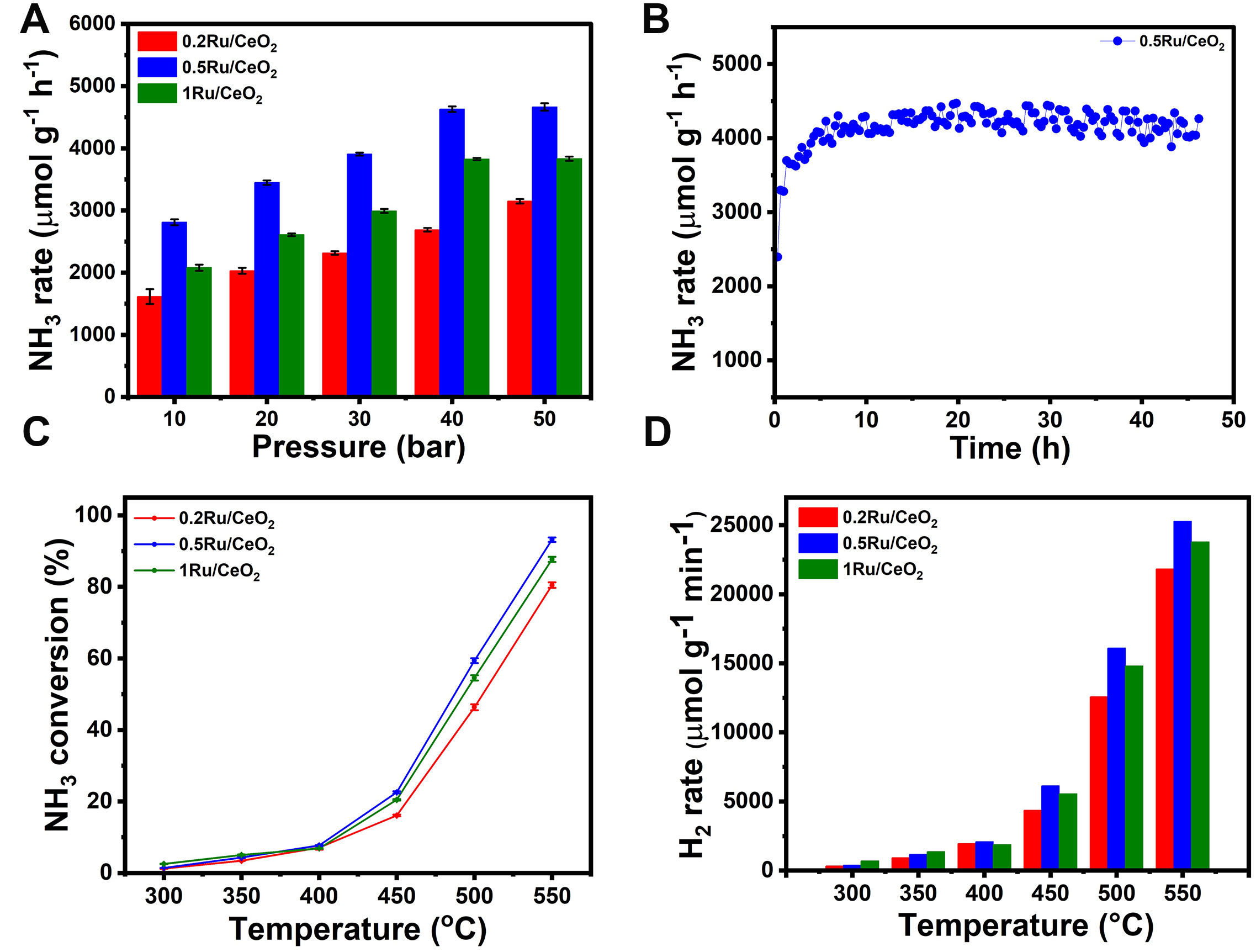
CeO2 is widely used in catalysis for its Ce4+/Ce3+ redox behavior, oxygen storage capacity, and electronic structure, which promote NH3 synthesis by creating oxygen vacancies (Ov) and modulating Ru species[19,92,93]. Beyond reduction behavior, CeO2 morphology, Ru loading, and dispersion strongly influence catalytic performance. Nanorods and polyhedrons enhance activity by providing high surface area, abundant Ov, and exposed {111}/{100} facets, which stabilize small, low-crystallinity Ru species with higher surface Ru4+, whereas nanocubes form larger, poorly dispersed Ru particles with weaker metal-support interactions[19,90,94]. Ru-O-Ce linkages further generate Ov via partial Ce4+ reduction, facilitating H2 and N2 adsorption and N2 dissociation, a rate-limiting step in NH3 formation[95,96]. Similar effects of Ov on N2 activation have been observed in photocatalytic systems such as BiOBr[97], TiO2[98], and TiO2/Au nanorods[99], highlighting the critical role of oxygen-deficient sites. The NH3 synthesis of nRu/CeO2 catalysts (n = 0.2, 0.5, 1) was evaluated using a 25% N2-75% H2 feed. Figure 9 and Supplementary Table 2 illustrate the NH3 synthesis rates at 400 °C across a pressure range of 10-50 bar for the CeO2 support and nRu/CeO2 catalysts.
Figure 9. (A) The NH3 synthesis rate of nRu/CeO2 catalysts (n = 0.2, 0.5, and 1) at 400 °C, 10,000 mL g-1h-1 WHSV and different pressure ranges (10-50 bar), (B) Stability test of 0.5Ru/CeO2 catalyst at 50 bar, 400 °C, and 10,000 mL g-1h-1, (C) NH3 decomposition conversion graph with respect to temperature, at P = 1 atm, T = 350-550 °C, and (D) H2 production rate (mmol g-1 min-1) with respect to temperature, at P = 1 atm, T = 350-550 °C. (A and C) represent the standard error of the mean (SEM).
CeO2 morphology effect
The MOF-derived CeO2 support was evaluated for NH3 synthesis across varying pressures. It remained largely inactive at 10-20 bar but showed noticeable activity at higher pressures, achieving an NH3 yield of
Ru loading effect
To better understand how Ru particle size influences N2 dissociation, it's helpful to revisit the recent study by Yanliang Zhou et al.[100]. Their findings clearly indicate that smaller Ru nanoparticles, specifically in Ru/BaCeO3 systems, promote the generation of Ce3+ species and Ov. These vacancies enhance the electron transfer to Ru sites, thereby facilitating N2 dissociation. Additionally, the structure-sensitive nature of NH3 synthesis under mild conditions was emphasized by Peng et al.[101]. Their work shows that even subtle changes in Ru structure can significantly alter catalytic activity. Specifically, as Ru particle size decreases, the proportion of low-coordinated corner sites increases, while terrace site populations decline. This geometric evolution alters the electronic structure of Ru metal particles, effectively reducing the catalyst’s work function (ϕcat). This reduction enhances electron transfer from Ru d-orbitals to N2, promoting N2 activation and subsequent N-H bond formation. Furthermore, isotopic labeling experiments combined with DRIFTS can shed light on the pool of N-containing intermediates and the underlying reaction mechanism, whether associative or dissociative. This is particularly relevant, as the Ru particle size can critically influence the mechanism. Larger Ru particles (> 2 nm) tend to follow a dissociative pathway[102], whereas atomically dispersed Ru favors an associative mechanism[18,19]. Smaller Ru sizes, with higher dispersion, also support hydrogen spillover to the support or interface, leading to the formation of Ov-H species. These can trap hydrogen and help mitigate H-poisoning effects on Ru[103], which typically occur under reaction conditions. Previous research[104] has indicated that Ru dispersion diminishes within the range of 1%-5% Ru, with optimal loading observed around 3.5%. Additionally, another study by Li et al.[105] showed that catalytic activity has no significant improvement beyond 4% Ru loading. To further support the role of Ru particle size and its impact on catalytic performance, the Ru loading was systematically varied from 0.2 to 1 wt.% over CeO2. This variation allowed us to tune the Ru nanoparticle size from 0.9 to 1.06 and 1.2 nm, respectively. At 10 bar and 400 °C [Figure 9A], the NH3 synthesis activity of the Ru/CeO2 catalysts displayed a clear dependence on Ru loading. The 0.5Ru/CeO2 catalyst exhibited the highest NH3 production rate of 2,811 ± 47.5 µmol g-1 h-1, followed by 1Ru/CeO2 with 2,080 ± 48.1 µmol g-1 h-1, and 0.2Ru/CeO2 with 1,615 ± 118.8 µmol g-1 h-1. The CeO2 support alone showed negligible activity under the same conditions. This trend highlights the importance of optimizing Ru dispersion and particle size, with 0.5 wt% Ru offering the most favorable balance for efficient NH3 production at moderate pressures. The NH3 synthesis activity at 400 °C and 10 bar, showed a non-linear trend: 0.2Ru/CeO2 < 0.5Ru/CeO2 > 1Ru/CeO2, indicating an optimum size and dispersion effect of Ru nanoparticles. The catalyst with 0.5 wt% Ru loading, possessing an average Ru particle size of approximately 1.02 nm, exhibited the best activity. This directly aligns with literature findings
Effect of pressure
To investigate the effect of reaction pressure on the NH3 synthesis activity, a series of nRu/CeO2 catalysts with varying Ru loadings (0.2-1 wt%) were tested under pressures ranging from 10 to 50 bar and 400 °C and is shown in Figure 9A. According to Le Chatelier's principle[106], any change in system pressure will shift the equilibrium to counteract that change. For the ammonia synthesis reaction, N2(g) + 3H2(g) ⇌ 2NH3(g), an increase in pressure shifts the equilibrium toward the formation of ammonia, as the product side contains fewer gas molecules. This theoretical understanding is consistent with our experimental findings, where an increase in reaction pressure from 10 to 50 bar led to a significant enhancement in NH3 production rates across all nRu/CeO2 catalysts. The 0.5Ru/CeO2 catalyst exhibited the highest activity, with NH3 production reaching 4,665 µmol g-1 h-1 at 50 bar, highlighting its optimal balance of Ru loading and dispersion. The enhanced performance is not only attributed to the pressure-driven equilibrium shift but also to the catalyst’s finely tuned Ru nanoparticle size (~1.02 nm), which promotes effective N2 activation. On the other hand, catalysts with either lower (0.2 wt%) or higher (1-2 wt%) Ru loadings showed inferior performance due to insufficient active sites or excessive particle growth, respectively. These results emphasize that, while high pressure thermodynamically favors NH3 formation, the structural and electronic properties of the catalyst play an equally critical role in achieving high catalytic efficiency.
Prolong stream effect
The catalyst with the best performance (0.5Ru/CeO2) was selected for a prolonged stability study for 45 h time on stream. As shown in Figure 9B, the catalytic activity initially exhibited a slight increase during the early stages of the reaction. This behavior is likely attributed to the activation of the catalyst surface (exposure of some active sites) as it reaches its optimal working state under reaction conditions. Following this activation phase, the activity stabilized, indicating the catalyst maintained consistent performance throughout the prolonged study. This stability demonstrates that the catalyst is robust over extended periods and resistant to common deactivation pathways. Importantly, no signs of sintering (agglomeration of Ru particles) or hydrogen poisoning of the Ru active sites were observed. This confirms the catalyst's ability to retain its structural integrity and chemical functionality under prolonged reaction conditions, making it a promising candidate for long-term industrial applications. The catalyst maintained a stable NH3 synthesis activity at 50 bar, 400 °C, and 10,000 mL g-1h-1.
NH3 catalytic cracking performance
Given the high reversibility of NH3 synthesis, we also evaluated the applicability of the nRu/CeO2 catalysts in NH3 cracking at 350-550 °C and 1 bar. NH3 cracking (2NH3 → 3H2+N2, ∆H = 92.44 kJ/mol) involves the cleavage of N-H bonds followed by the recombination of nitrogen and hydrogen atoms to form N2 and H2 gases[112]. As an endothermic reaction, its catalytic efficiency is highly dependent on temperature. The influence of temperature on NH3 conversion% over the nRu/CeO2 catalysts is presented in Figure 9C. Within the range of 300 to 550 °C, NH3 conversion steadily increases with temperature across all nRu/CeO2 catalysts. Notably, the 0.5Ru/CeO2 catalyst exhibits the highest activity, achieving an impressive 93% NH3 conversion at 550 °C. At this temperature, the conversion follows the trend: 0.2Ru/CeO2 (80%) < 1Ru/CeO2 (87%) < 0.5Ru/CeO2 (93%). A similar trend is observed in the hydrogen production rate at 550 °C, with 0.5Ru/CeO2 reaching 25,282 µmol g-1 min-1, outperforming both 0.2Ru/CeO2 (21,832 µmol g-1 min-1) and 1Ru/CeO2 (23,784 µmol g-1 min-1) as shown in Figure 9D. Remarkably, the 0.5Ru/CeO2 catalyst demonstrates superior NH3 conversion and a faster decomposition rate compared to previously reported Ru/CeO2 catalysts with Ru nanoparticles. The enhanced catalytic performance of the nRu/CeO2 catalyst for NH3 cracking can be attributed to several key factors. First, the increased concentration of oxygen vacancies (Ovs) in the catalyst plays a critical role in improving catalytic activity[10,107,108]. These Ov act as active sites for the adsorption and dissociation of N2 and H2, which are essential steps in the NH3 cracking reaction. Raman and XPS analysis confirmed the higher Ov concentration, as indicated by the Raman spectroscopy, which correlates with improved catalytic efficiency. Additionally, the hydrogen spillover effect, observed through H2-TPR analysis, further facilitates the NH3 cracking process. The spillover effect allows for the migration of atomic hydrogen from the Ru nanoparticles to the CeO2 support, thereby enhancing the cleavage of N-H bonds and improving the overall hydrogen production rate[109]. Moreover, the fine dispersion of Ru on the MOF-derived CeO2 support, as confirmed by HR-TEM, ensures that a large surface area of Ru is available for the reaction, preventing agglomeration and maximizing the number of active sites[110]. These combined effects: higher oxygen vacancy concentration, effective hydrogen spillover, and enhanced Ru dispersion contribute to the superior catalytic performance of the nRu/CeO2 catalyst, which outperforms previously reported Ru/CeO2 catalysts in NH3 cracking.
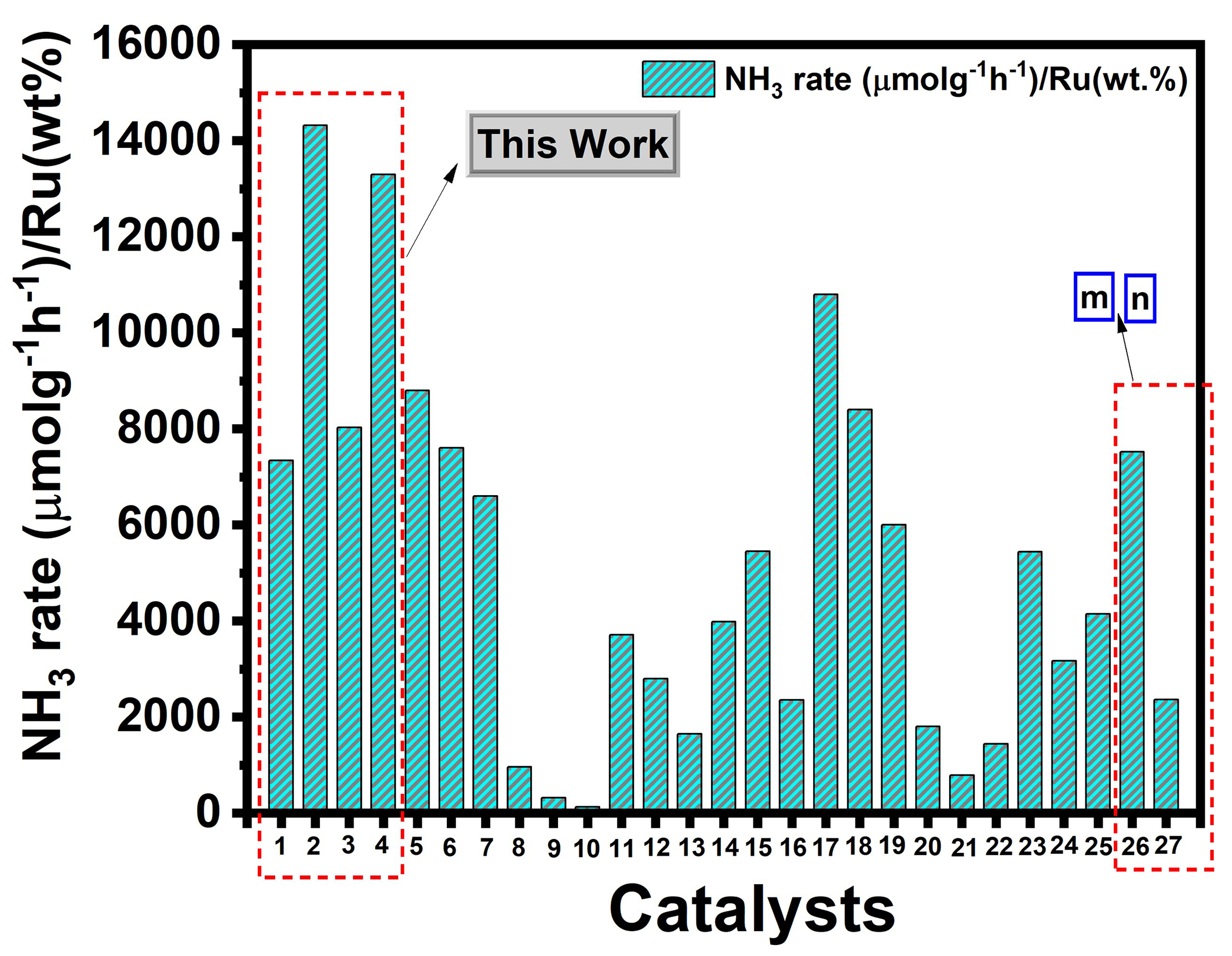
When benchmarked against other reported Ru/CeO2 systems with varying Ru weight loadings, our 0.5Ru/CeO2 catalyst [Supplementary Table 3, Figure 10] demonstrated superior performance, further underscoring the importance of precise control over Ru particle size and metal-support interactions. These findings validate the previously discussed mechanism, where Ru size not only alters site populations and electronic structure (thus affecting work function and N2 dissociation) but also determines the operative reaction pathway: associative or dissociative, especially near the critical ~1 nm (Ru particle size) threshold.
Post-catalytic study
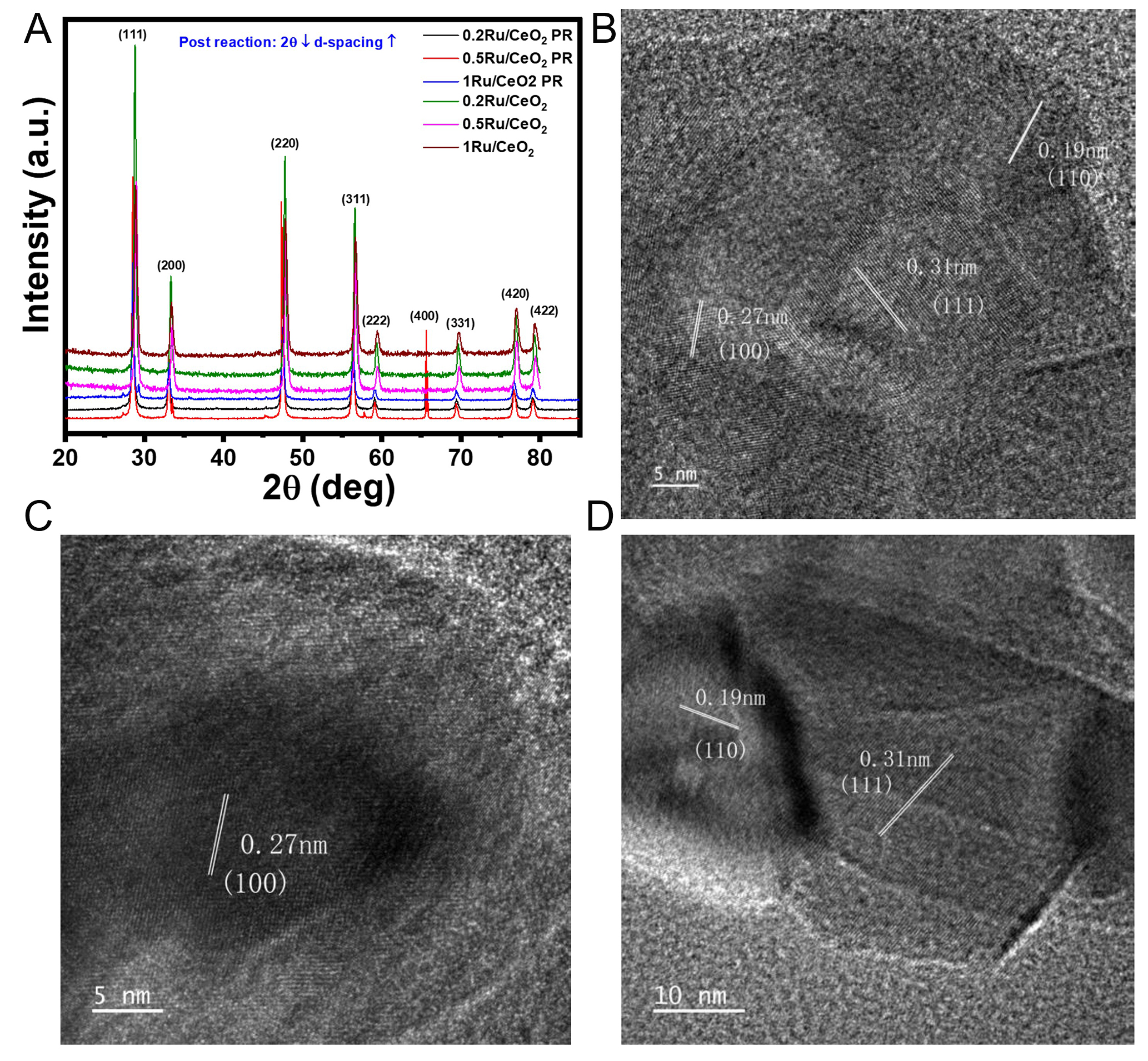
The post-catalytic surface and structure characterization was performed after conducting the following tests: NH3 synthesis, NH3 cracking (300-550 °C), and the NH3 synthesis stability test. The provided post-catalytic assessment results in the XRD [Figure 11A, Supplementary Table 4] and HRTEM images [Figure 11B-D] provide important insights into the structural changes and metal dispersion on nRu/CeO2 catalysts after the catalytic reaction for NH3 synthesis and cracking. The XRD patterns show the characteristic peaks of CeO2, indicating that the ceria support retains its fluorite structure after the catalytic reaction. The peaks observed at 28.4°, 33°, 47.4°, 56.3°, and 59°, 65.6°, 69.4°, 76.6°, and 79° correspond to the CeO2 (111), (200), (220),
Figure 11. (A) XRD of CeO2 support and nRu/CeO2 (n = 0.2, 0.5, and 1 wt%) before (reduced (under 10%H2/Ar, 800 °C, 1 h)) and after catalytic activity test, and HRTEM of (B) 0.2Ru/CeO2 (C), 0.5Ru/CeO2, and (D) 1Ru/CeO2 catalysts after catalytic activity test.
The post-catalytic HRTEM images [Supplementary Figure 10A-C] reveal that the nRu/CeO2 catalysts are rod-shaped, with widths ranging from 60 to 70 nm and lengths of approximately 1 μm. The lattice fringe spacings observed are 0.19, 0.27, and 0.32 nm, which correspond to the (220), (200), and (111) facets of CeO2[107]. Notably, no identifiable Ru nanoclusters were observed on the surfaces of the nRu/CeO2 catalysts, suggesting that the Ru species are likely dispersed at the subnanometer or even single-atom level[116].
The EDX mapping results [Figure 12A-C] further confirm the homogeneous distribution of Ru species across the entire nanostructure. High-magnification HAADF-STEM images reveal no clusters, indicating that Ru does not exist as clusters or particles in the observed regions[117].
CONCLUSIONS
This work demonstrates the rational design of MOF-derived Ru/CeO2 catalysts for efficient NH3 synthesis and decomposition. Catalytic performance strongly depends on Ru loading and particle size, with 0.5 wt.% Ru/CeO2 showing optimal activity, stability, and resistance to sintering. The enhanced performance arises from abundant oxygen vacancies and strong Ru-CeO2 interfacial interactions, which promote H2 activation and N2 dissociation. Raman and in-situ XPS confirm the key role of oxygen vacancies in facilitating N2 activation. These findings provide valuable structural insights for optimizing Ru-based catalysts toward sustainable energy applications.
DECLARATIONS
Authors’ contributions
Writing - original draft, writing - review & editing, methodology, investigation, formal analysis, data curation: Singh, S.
Writing - review & editing, investigation, data curation: Kim, S. J.
Writing - review & editing, supervision, resources, formal analysis: Yavuz, C. T.
Formal analysis, investigation: Komarala, E. P.
Writing -review & editing, investigation, resources: Tan, M.
Writing - review & editing, validation, supervision, resources, project administration, funding acquisition, conceptualization: Polychronopoulou, K.
Availability of data and materials
Some results of supporting the study are presented in the Supplementary Materials. Other raw data that supports the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors acknowledge the financial support from Khalifa University through the Center for Catalysis and Separations (CeCaS), grant RC2-2018-024.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. He, Y.; Guan, B.; Zhuang, Z.; et al. Advances in ammonia (NH3) adsorption and storage: materials, mechanisms, and applications. Adsorption 2025, 31, 48.
2. Hassan, Q.; Algburi, S.; Sameen, A. Z.; Jaszczur, M.; Salman, H. M. Hydrogen as an energy carrier: properties, storage methods, challenges, and future implications. Environ. Syst. Decis. 2023, 44, 327-50.
3. Marakatti, V. S.; Gaigneaux, E. M. Recent advances in heterogeneous catalysis for ammonia synthesis. ChemCatChem 2020, 12, 5838-57.
4. Lim, J.; Fernández, C. A.; Lee, S. W.; Hatzell, M. C. Ammonia and nitric acid demands for fertilizer use in 2050. ACS. Energy. Lett. 2021, 6, 3676-85.
5. Macfarlane, D. R.; Cherepanov, P. V.; Choi, J.; et al. A roadmap to the ammonia economy. Joule 2020, 4, 1186-205.
6. Ma, Y.; Lan, G.; Fu, W.; et al. Role of surface defects of carbon nanotubes on catalytic performance of barium promoted ruthenium catalyst for ammonia synthesis. J. Energy. Chem. 2020, 41, 79-86.
7. Singh, S.; Mohammed, A. K.; Alhammadi, A. A.; Shetty, D.; Polychronopoulou, K. Hypes and hopes on the materials development strategies to produce ammonia at mild conditions. Int. J. Hydrogen. Energy. 2023, 48, 34700-39.
8. Othman, A.; Gowda, A.; Andreescu, D.; et al. Two decades of ceria nanoparticle research: structure, properties and emerging applications. Mater. Horiz. 2024, 11, 3213-66.
9. Liu, H.; Zhang, R.; Liu, S.; Liu, G. CeO2/Ni inverse catalyst as a highly active and stable Ru-free catalyst for ammonia decomposition. ACS. Catal. 2024, 14, 9927-39.
10. Teng, B.; Ma, C.; Chen, J.; et al. Ru dispersed on oxygen-defect-rich CeO2 nanorods for ammonia decomposition. ACS. Appl. Nano. Mater. 2024, 7, 15012-24.
11. Zhu, X.; Liu, S.; Li, Y.; et al. Redox CeO2 substituted Na2WO4-Mn catalyst for high-throughput chemical looping oxidative coupling of methane. Appl. Catal. B. Environ. Energy. 2025, 367, 125108.
12. Liu, B.; Pan, Y.; Han, Z.; et al. Interplay of structural properties and redox behavior in CeO2 nanoparticles: impact on reactivity and bioavailability. Environ. Sci. Technol. 2025, 59, 4641-51.
13. Parastaev, A.; Muravev, V.; Huertas, Osta. E.; et al. Boosting CO2 hydrogenation via size-dependent metal-support interactions in cobalt/ceria-based catalysts. Nat. Catal. 2020, 3, 526-33.
14. Buhori, A.; Choi, J.; Lee, H.; et al. Effect of CeO2 morphology and Ru impregnation method on CH4 selectivity reduction in polyethylene waste conversion to liquid fuels and lubricants. Chem. Eng. J. 2024, 499, 156097.
15. Wei, J.; Sun, Y.; Ma, Y.; et al. Tailored metal-oxygen defect engineering to modulate catalytic interaction of ultra-low Ru supported morphology-oriented CeO2 for hydrogenolysis of lignin-derived phenols. Chem. Eng. J. 2025, 509, 161199.
16. Yamazaki, K.; Goto, Y.; Kikugawa, M.; et al. Effect of support morphology on the ammonia synthesis activity of Ru/CeO2-based catalysts. Int. J. Hydrogen. Energy. 2024, 94, 406-19.
17. Xie, B.; Wang, Z.; Zhang, X.; et al. Morphology effect of cerium dioxide on the catalytic performance of Ru/CeO2 catalyst for the oxidation of different CVOCs. Sep. Purif. Technol. 2024, 345, 127428.
18. Wang, J.; Zhang, H.; Shi, L.; et al. Morphological effects of cerium oxide on photothermal synergistic catalytic degradation of toluene. Sep. Purif. Technol. 2026, 380, 135271.
19. Ma, Z.; Zhao, S.; Pei, X.; Xiong, X.; Hu, B. New insights into the support morphology-dependent ammonia synthesis activity of Ru/CeO2 catalysts. Catal. Sci. Technol. 2017, 7, 191-9.
20. Li, W. Q.; Xu, M.; Chen, J. S.; Ye, T. N. Enabling sustainable ammonia synthesis: from nitrogen activation strategies to emerging materials. Adv. Mater. 2024, 36, 2408434.
21. Zhang, L.; Zhou, M.; Wang, A.; Zhang, T. Selective Hydrogenation over supported metal catalysts: from nanoparticles to single atoms. Chem. Rev. 2019, 120, 683-733.
22. Sarma, B. B.; Kim, J.; Amsler, J.; et al. One‐pot cooperation of single‐atom Rh and Ru Solid catalysts for a selective tandem olefin isomerization‐hydrosilylation process. Angew. Chem. Int. Ed. 2020, 59, 5806-15.
23. Ansari, M. Z.; Habib, F.; Gupta, J.; et al. Frontiers in metal-organic frameworks: innovative nanomaterials for next-generation supercapacitors. Adv. Compos. Hybrid. Mater. 2024, 7, 215.
24. Zulfiqar, A.; Miao, B.; Khan, F.; et al. Metal-organic framework (MOF)-based catalysts for sustainable energy technologies: a review. Langmuir 2025, 41, 24049-77.
25. Sousa, A.; Rendon, Patino. A.; Garzon, Tovar. L.; Mateo, D.; Gascon, J.; Bavykina, A. Ammonia decomposition via MOF‐derived photothermal catalysts. ChemSusChem 2024, 18, e202401896.
26. Wu, W.; Yao, W.; Liu, Y.; Xi, S.; Zhang, T. Efficient hydrogen production from ammonia using Ru nanoparticles on Ce-based metal-organic framework (MOF)-derived CeO2 with oxygen vacancies. Molecules 2025, 30, 2301.
27. Song, Q.; Yin, X.; Zhang, H. Ni‐MOF‐74 derived carbon‐based Ni catalysts for efficient catalytic ammonia synthesis via pulsed DBD plasma. Plasma. Processes. Polym. 2024, 22, 2400173.
28. Li, S.; Wang, N.; Yue, Y.; Wang, G.; Zu, Z.; Zhang, Y. Copper doped ceria porous nanostructures towards a highly efficient bifunctional catalyst for carbon monoxide and nitric oxide elimination. Chem. Sci. 2015, 6, 2495-500.
30. Porta, A.; Falbo, L.; Visconti, C. G.; Lietti, L.; Bassano, C.; Deiana, P. Synthesis of Ru-based catalysts for CO2 methanation and experimental assessment of intraporous transport limitations. Catal. Today. 2020, 343, 38-47.
31. Hussain, J.; Lin, K.; Chowdhury, S.; Hussain, A. Synthesis and characterization of hexagonal ceria-BTC microrods for methanol decomposition. J. Taiwan. Inst. Chem. Eng. 2024, 159, 105486.
32. Borges, Serra. A. R.; Castro, De. Sousa. G.; De, Carvalho. Gomes. V.; et al. Enhancing photocatalytic tetracycline degradation through the fabrication of high surface area CeO2 from a cerium-organic framework. RSC. Adv. 2024, 14, 17507-18.
33. Zhao, Z.; Jiang, Q.; Wang, Q.; et al. Effect of rutile content on the catalytic performance of Ru/TiO2 catalyst for low-temperature CO2 methanation. ACS. Sustain. Chem. Eng. 2021, 9, 14288-96.
34. Li, Y.; Zhang, L. A.; Qin, Y.; et al. Crystallinity dependence of ruthenium nanocatalyst toward hydrogen evolution reaction. ACS. Catal. 2018, 8, 5714-20.
35. Sivan, S. E.; Kang, K. H.; Han, S. J.; et al. Facile MOF-derived one-pot synthetic approach toward Ru single atoms, nanoclusters, and nanoparticles dispersed on CeO2 supports for enhanced ammonia synthesis. J. Catal. 2022, 408, 316-28.
36. Shannon, R. D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta. Cryst. A. 1976, 32, 751-67.
37. Sharma, S.; Hu, Z.; Zhang, P.; Mcfarland, E. W.; Metiu, H. CO2 methanation on Ru-doped ceria. J. Catal. 2011, 278, 297-309.
38. Singh, P.; Hegde, M. S. CexRuxO2-δ (x = 0.05, 0.10): a new high oxygen storage material and Pt, Pd-free three-way catalyst. Chem. Mater. 2009, 21, 3337-45.
39. Carrillo, A. J.; Navarrete, L.; Laqdiem, M.; Balaguer, M.; Serra, J. M. Boosting methane partial oxidation on ceria through exsolution of robust Ru nanoparticles. Mater. Adv. 2021, 2, 2924-34.
40. Pandit, N. A.; Alshehri, S. M.; Ahmad, T. CeO2/ZrO2 p-n heterojunction nanostructures for efficient NO2 gas sensing. J. Alloys. Compd. 2024, 1004, 175782.
41. Iqbal, M. W.; Yu, Y.; Simakov, D. S. Enhancing the surface area stability of the cerium oxide reverse water gas shift nanocatalyst via reverse microemulsion synthesis. Catal. Today. 2023, 407, 230-43.
42. Kang, W.; Ozgur, D. O.; Varma, A. Solution combustion synthesis of high surface area CeO2 nanopowders for catalytic applications: reaction mechanism and properties. ACS. Appl. Nano. Mater. 2018, 1, 675-85.
43. Zaidi, R.; Khan, S. U.; Farooqi, I. H.; et al. Performance, isotherm, kinetics and mechanism of simultaneous removal of Cr(VI), Cu(II) and F ions by CeO2-MgO binary oxide nanomaterials. Sci. Rep. 2025, 15, 1431.
44. López-Rodríguez, S.; Davó-Quiñonero, A.; Bailón-García, E.; Lozano-Castelló, D.; Bueno-López, A. Effect of Ru loading on Ru/CeO2 catalysts for CO2 methanation. Mol. Catal. 2021, 515, 111911.
45. Feng, Q.; Zou, J.; Wang, Y.; et al. Influence of surface oxygen vacancies and ruthenium valence state on the catalysis of pyrochlore oxides. ACS. Appl. Mater. Interfaces. 2020, 12, 4520-30.
46. Zhou, S.; Ban, T.; Li, T.; et al. Defect engineering in Ce-based metal-organic frameworks toward enhanced catalytic performance for hydrogenation of dicyclopentadiene. ACS. Appl. Mater. Interfaces. 2024, 16, 38177-87.
47. Giri, G.; Verploegen, E.; Mannsfeld, S. C. B.; et al. Tuning charge transport in solution-sheared organic semiconductors using lattice strain. Nature 2011, 480, 504-8.
48. Dave, M.; Dalela, S.; Kumar, S.; Alvi, P. A. Defect induced structural and Raman study of Nd-doped CeO2 nanomaterials. AIP. Conf. Proc. 2020, 2265, 030108.
49. Bezkrovnyi, O. S.; Kraszkiewicz, P.; Ptak, M.; Kepinski, L. Thermally induced reconstruction of ceria nanocubes into zigzag {111}-nanofacetted structures and its influence on catalytic activity in CO oxidation. Catal. Commun. 2018, 117, 94-8.
50. Angermann, M.; Jakopic, G.; Prietl, C.; Griesser, T.; Reichmann, K.; Deluca, M. Highly conductive RuO2 thin films from novel facile aqueous chemical solution deposition. J. Sol-Gel. Sci. Technol. 2023, 108, 575-87.
51. Yao, X.; Zhang, L.; Li, L.; et al. Investigation of the structure, acidity, and catalytic performance of CuO/Ti0.95Ce0.05O2 catalyst for the selective catalytic reduction of NO by NH3 at low temperature. Appl. Catal. B. Environ. 2014, 150-151, 315-29.
52. Wu, Z.; Li, M.; Howe, J.; Meyer, H. M.; Overbury, S. H. Probing defect sites on CeO2 nanocrystals with well-defined surface planes by Raman spectroscopy and O2 adsorption. Langmuir 2010, 26, 16595-606.
53. Dai, Q.; Huang, H.; Zhu, Y.; et al. Catalysis oxidation of 1,2-dichloroethane and ethyl acetate over ceria nanocrystals with well-defined crystal planes. Appl. Catal. B. Environ. 2012, 117-8, 360-8.
54. Chen, H. First-principles study of CO adsorption and oxidation on Ru-doped CeO2(111) surface. J. Phys. Chem. C. 2012, 116, 6239-46.
55. Huang, H.; Dai, Q.; Wang, X. Morphology effect of Ru/CeO2 catalysts for the catalytic combustion of chlorobenzene. Appl. Catal. B. Environ. 2014, 158-9, 96-105.
56. Zhang, X.; Zheng, J.; Boch, F. J.; Nickl, S.; Köhler, K. Decomposition of N2O by ruthenium catalysts - RuO2 as active phase on non‐reducible supports. ChemCatChem 2024, 16, e202400347.
57. Mi, R.; Li, D.; Hu, Z.; Yang, R. T. Morphology effects of CeO2 nanomaterials on the catalytic combustion of toluene: a combined kinetics and diffuse reflectance infrared fourier transform spectroscopy study. ACS. Catal. 2021, 11, 7876-89.
58. Costa Zonetti P, Landers R, Cobo AJG. Thermal treatment effects on the Ru/CeO2 catalysts performance for partial hydrogenation of benzene. Appl. Surf. Sci. 2008, 254, 6849-53.
59. Kim, H.; Yang, S.; Lim, Y. H.; Lee, J.; Ha, J.; Kim, D. H. Enhancement in the metal efficiency of Ru/TiO2 catalyst for guaiacol hydrogenation via hydrogen spillover in the liquid phase. J. Catal. 2022, 410, 93-102.
60. Li, W.; Liu, P.; Niu, R.; Li, J.; Wang, S. Influence of CeO2 supports prepared with different precipitants over Ru/CeO2 catalysts for ammonia synthesis. Solid. State. Sci. 2020, 99, 105983.
61. Sun, Y.; Xue, Z.; Liu, Q.; et al. Modulating electronic structure of metal-organic frameworks by introducing atomically dispersed Ru for efficient hydrogen evolution. Nat. Commun. 2021, 12, 1369.
62. Villarroel-rocha, J.; Gil, A. Modeling the temperature-programmed reduction of metal oxide catalysts by considering the particle-size distribution effect. Chem. Eng. J. 2024, 487, 150722.
63. Unutulmazsoy, Y.; Cancellieri, C.; Lin, L.; Jeurgens, L. P. Reduction of thermally grown single-phase CuO and Cu2O thin films by in-situ time-resolved XRD. Appl. Surf. Sci. 2022, 588, 152896.
64. Chen, J.; Ni, J.; Xu, H.; He, G.; Chen, H. Mechanism and research progress of hydrogen spillover in hydrogen evolution reaction. J. Alloys. Compd. 2024, 1004, 175883.
65. Lee, S.; Kim, H.; Ryoo, R.; Park, J. Y.; Choi, M. Hydrogen spillover in nonreducible oxides: mechanism and catalytic utilization. Nano. Res. 2022, 15, 10357-65.
66. Li, Y.; Li, L.; Xu, S.; et al. Hydrogen spillover mechanism at the metal-metal interface in electrocatalytic hydrogenation. Angew. Chem. Int. Ed. 2024, 63, e202407810.
67. Li, Y.; Chen, S.; Li, K.; et al. Plasma-assisted ammonia synthesis over La2O3 modified by RF discharge: effect of oxygen vacancies on N2 adsorption and associative mechanism. ACS. Sustain. Chem. Eng. 2024, 12, 11728-38.
68. Luo, S.; Liu, Y.; Guo, L.; et al. Plasma-derived hydrogen radical-mediated N2 activation for mild ammonia synthesis: insights into the importance of oxygen vacancies in the reaction mechanism. J. Mater. Chem. A. 2025, 13, 30546-53.
69. Zhu, H.; Wang, C.; He, Y.; et al. Oxygen vacancies engineering in electrocatalysts nitrogen reduction reaction. Front. Chem. 2022, 10, 1039738.
70. Wang, H.; Li, X.; Ruan, Q.; Tang, J. Ru and RuOx decorated carbon nitride for efficient ammonia photosynthesis. Nanoscale 2020, 12, 12329-35.
71. Polychronopoulou, K.; Alkhoori, S.; Albedwawi, S.; et al. Decoupling the chemical and mechanical strain effect on steering the CO2 activation over CeO2-based oxides: an experimental and DFT approach. ACS. Appl. Mater. Interfaces. 2022, 14, 33094-119.
72. Jiang, P.; Zhang, L.; Liu, X.; et al. Tuning oxidant and antioxidant activities of ceria by anchoring copper single-site for antibacterial application. Nat. Commun. 2024, 15, 1010.
73. Siakavelas, G.; Charisiou, N.; Alkhoori, S.; et al. Highly selective and stable nickel catalysts supported on ceria promoted with Sm2O3, Pr2O3 and MgO for the CO2 methanation reaction. Appl. Catal. B. Environ. 2021, 282, 119562.
74. Isaacs, M. A.; Drivas, C.; Lee, R.; Palgrave, R.; Parlett, C. M.; Morgan, D. J. XPS surface analysis of ceria-based materials: experimental methods and considerations. Appl. Surface. Sci. Adv. 2023, 18, 100469.
75. Elmutasim, O.; Hussien, A. G.; Sharan, A.; et al. Evolution of oxygen vacancy sites in ceria-based high-entropy oxides and their role in N2 activation. ACS. Appl. Mater. Interfaces. 2024, acsami.3c16521.
76. Xie, C.; Yan, D.; Li, H.; et al. Defect chemistry in heterogeneous catalysis: recognition, understanding, and utilization. ACS. Catal. 2020, 10, 11082-98.
77. Peera, S. G.; Kim, S. W. Rare earth Ce/CeO2 Electrocatalysts: role of high electronic spin state of Ce and Ce3+/Ce4+ redox couple on oxygen reduction reaction. Nanomaterials 2025, 15, 600.
78. Kumar, A.; Lee, J.; Kim, M. G.; et al. Efficient nitrate conversion to ammonia on f-block single-atom/metal oxide heterostructure via local electron-deficiency modulation. ACS. Nano. 2022, 16, 15297-309.
79. Zhang, Y.; Zhao, S.; Feng, J.; et al. Unraveling the physical chemistry and materials science of CeO2-based nanostructures. Chem 2021, 7, 2022-59.
80. Kang, L.; Wang, B.; Bing, Q.; et al. Adsorption and activation of molecular oxygen over atomic copper(I/II) site on ceria. Nat. Commun. 2020, 11, 4008.
81. Yang, X.; Han, R.; Shi, L.; et al. Dual-site engineering of La/Ti co-doped BiFeO3 for enhanced visible-light photocatalytic degradation: synergistic effects and high stability. J. Alloys. Compd. 2025, 1039, 183239.
82. Yu, W.; Wang, W.; Li, S.; et al. Construction of active site in a sintered copper-ceria nanorod catalyst. J. Am. Chem. Soc. 2019, 141, 17548-57.
83. Yan, H.; Yang, C.; Shao, W.; et al. Construction of stabilized bulk-nano interfaces for highly promoted inverse CeO2/Cu catalyst. Nat. Commun. 2019, 10, 3470.
84. Li, H.; Wang, H.; Gong, X.; et al. Multiple configurations of the two excess 4f electrons on defective CeO2(111): origin and implications. Phys. Rev. B. 2009, 79, 193401.
85. Holgado, J.; Munuera, G.; Espinós, J.; González-Elipe, A. XPS study of oxidation processes of CeOx defective layers. Appl. Surf. Sci. 2000, 158, 164-71.
86. Han, Z.; Liu, W.; Gao, Y. Advancing the understanding of oxygen vacancies in ceria: insights into their formation, behavior, and catalytic roles. JACS. Au. 2025, 5, 1549-69.
87. Feng, Z.; Guo, F.; Zhang, Y.; Ichikawa, T.; Zheng, J. Oxygen vacancies rich CeO2 supported Ru catalyst for efficient hydrogenation of N-ethylcarbazole at mild temperature. Appl. Catal. B. Environ. Energy. 2025, 366, 125059.
88. Lu, X.; Zhang, R.; Liu, Y.; et al. Oxygen vacancies and lattice distortion synergistically enhanced piezocatalysis of CaZn2(BO3)2 for nonantibiotic pharmaceutical degradation. ACS. Appl. Mater. Interfaces. 2024, 16, 63692-702.
89. Chou, M. Y.; Chelikowsky, J. R. Structural properties of the Ru(0001) surface. Phys. Rev. B. 1987, 35, 2124-7.
90. Lin, B.; Liu, Y.; Heng, L.; et al. Morphology effect of ceria on the catalytic performances of Ru/CeO2 catalysts for ammonia synthesis. Ind. Eng. Chem. Res. 2018, 57, 9127-35.
91. Xu, B.; Xia, L.; Zhou, F.; et al. Enhancing electrocatalytic N2 reduction to NH3 by CeO2 nanorod with oxygen vacancies. ACS. Sustain. Chem. Eng. 2019, 7, 2889-93.
92. Yang, X.; Tian, Y.; Mukherjee, S.; et al. Constructing oxygen vacancies via engineering heterostructured Fe3C/Fe3O4 catalysts for electrochemical ammonia synthesis. Angew. Chem. Int. Ed. 2023, 62, e202304797.
93. Zhang, L.; Li, R.; Cui, L.; et al. Boosting photocatalytic ammonia synthesis performance over OVs-rich Ru/W18O49: insights into the roles of oxygen vacancies in enhanced hydrogen spillover effect. Chem. Eng. J. 2023, 461, 141892.
94. Jiang, F.; Wang, S.; Liu, B.; et al. Insights into the influence of CeO2 crystal facet on CO2 hydrogenation to methanol over Pd/CeO2 catalysts. ACS. Catal. 2020, 10, 11493-509.
95. Aika, K.; Kumasaka, M.; Oma, T.; et al. Support and promoter effect of ruthenium catalyst. III. Kinetics of ammonia synthesis over various Ru catalysts. Appl. Catal. 1986, 28, 57-68.
96. Fuller, J.; An, Q.; Fortunelli, A.; Goddard, W. A. Reaction mechanisms, kinetics, and improved catalysts for ammonia synthesis from hierarchical high throughput catalyst design. Acc. Chem. Res. 2022, 55, 1124-34.
97. Li, H.; Shang, J.; Ai, Z.; Zhang, L. Efficient visible light nitrogen fixation with BiOBr nanosheets of oxygen vacancies on the exposed {001} facets. J. Am. Chem. Soc. 2015, 137, 6393-9.
98. Hirakawa, H.; Hashimoto, M.; Shiraishi, Y.; Hirai, T. Photocatalytic conversion of nitrogen to ammonia with water on surface oxygen vacancies of Titanium dioxide. J. Am. Chem. Soc. 2017, 139, 10929-36.
99. Lee, Y.; He, G.; Akey, A. J.; Si, R.; Flytzani-stephanopoulos, M.; Herman, I. P. Raman analysis of mode softening in nanoparticle CeO2-δ and Au-CeO2-δ during CO oxidation. J. Am. Chem. Soc. 2011, 133, 12952-5.
100. Zhou, Y.; Wang, J.; Liang, L.; et al. Unraveling the size-dependent effect of Ru-based catalysts on Ammonia synthesis at mild conditions. J. Catal. 2021, 404, 501-11.
101. Peng, X.; Chen, X.; Zhou, Y.; et al. Size-dependent activity of supported Ru catalysts for ammonia synthesis at mild conditions. J. Catal. 2022, 408, 98-108.
102. Hinrichsen, O.; Rosowski, F.; Hornung, A.; Muhler, M.; Ertl, G. The kinetics of ammonia synthesis over Ru-based catalysts. J. Catal. 1997, 165, 33-44.
103. Yao, Y.; Zhu, S.; Wang, H.; Li, H.; Shao, M. A spectroscopic study on the nitrogen electrochemical reduction reaction on gold and platinum surfaces. J. Am. Chem. Soc. 2018, 140, 1496-501.
104. Rossetti, I.; Forni, L. Effect of Ru loading and of Ru precursor in Ru/C catalysts for ammonia synthesis. Appl. Catal. A. Gen. 2005, 282, 315-20.
105. Li, Z. Ammonia synthesis on graphitic-nanofilament supported Ru catalysts. J. Mol. Catal. A. Chem. 2004, 211, 103-9.
106. Wang, H.; Wu, Q.; Xiao, F. Design of zeolite-based catalysts by Le Chatelier's principle. Appl. Catal. B. Environ. Energy. 2025, 379, 125671.
107. Shin, J.; Jung, U.; Kim, J.; et al. Elucidating the effect of Ce with abundant surface oxygen vacancies on MgAl2O4-supported Ru-based catalysts for ammonia decomposition. Appl. Catal. B. Environ. 2024, 340, 123234.
108. Huang, C.; Yu, Y.; Tang, X.; et al. Hydrogen generation by ammonia decomposition over Co/CeO2 catalyst: influence of support morphologies. Appl. Surf. Sci. 2020, 532, 147335.
109. Su, Q.; Gu, L.; Yao, Y.; et al. Layered double hydroxides derived Nix(MgyAlzOn) catalysts: enhanced ammonia decomposition by hydrogen spillover effect. Appl. Catal. B. Environ. 2017, 201, 451-60.
110. Zhang, B.; Yang, J.; Mu, Y.; et al. Fabrication of highly dispersed Ru catalysts on CeO2 for efficient C3H6 oxidation. Environ. Sci. Technol. 2024, 58, 19533-44.
111. Rocha, L. S. R.; Aparecido, Ciola. Amoresi. R.; Duarte, T. M.; et al. Experimental and theoretical interpretation of the order/disorder clusters in CeO2:La. Appl. Surf. Sci. 2020, 510, 145216.
112. Ranasinghe, K. S.; Singh, R.; Leshchev, D.; Vasquez, A.; Stavitski, E.; Foster, I. Synthesis of nanoceria with varied ratios of Ce3+/Ce4+ utilizing soluble borate glass. Nanomaterials 2022, 12, 2363.
113. Zheng, C.; Mao, D.; Xu, Z.; Zheng, S. Strong Ru-CeO2 interaction boosts catalytic activity and stability of Ru supported on CeO2 nanocube for soot oxidation. J. Catal. 2022, 411, 122-34.
114. Castleton, C. W. M.; Kullgren, J.; Hermansson, K. Tuning LDA+U for electron localization and structure at oxygen vacancies in ceria. J. Chem. Phys. 2007, 127, 244704.
115. Tomar, S.; Bhadoria, B. S.; Jeong, H.; Choi, J. H.; Lee, S.; Bhattacharjee, S. Single-atom Pd catalyst on a CeO2 (111) surface for methane oxidation: activation barriers and reaction pathways. J. Phys. Chem. C. 2024, 128, 8580-9.
116. Guo, Y.; Mei, S.; Yuan, K.; et al. Low-temperature CO2 methanation over CeO2-supported Ru single atoms, nanoclusters, and nanoparticles competitively tuned by strong metal-support interactions and H-spillover effect. ACS. Catal. 2018, 8, 6203-15.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].