The interplay between lifestyle and cardiovascular aging: mechanisms and therapeutic opportunities
Abstract
With the global trend of population aging, age-related diseases are increasing annually. The vascular system exhibits high sensitivity to aging, characterized by myocardial cell apoptosis, endothelial damage, oxidative stress, impaired autophagy, and chronic low-grade inflammatory responses. These factors significantly elevate the incidence of atherosclerosis, hypertension, myocardial infarction, and stroke, which are major cardiovascular diseases. Previous research has predominantly focused on the isolated impact of single lifestyle factors on cardiovascular aging, lacking a comprehensive framework that integrates multiple exposomes and their complex interactions. This review aims to reassess how the exposome factors, such as smoking, sedentary behavior, and the natural environment, interact and dysregulate core mechanisms such as oxidative stress and autophagy. These interactions collaboratively drive the aging of blood vessels, myocardium, and valves. We also propose targeted intervention strategies, providing insights for researchers investigating cardiovascular aging.
Keywords
INTRODUCTION
According to the global burden of disease data, cardiovascular diseases (CVDs) are significant causes of mortality and permanent disability among individuals aged 65 and above[1]. Aging induces degeneration in the cardiac and vascular systems, including changes in cardiac size and structure - such as left ventricular and left atrial dilation, reduced left ventricular diastolic function, and decreased left ventricular ejection fraction[2] - as well as increased arterial thickness and stiffness and impaired vascular endothelial function. These processes may involve complex pathophysiological mechanisms, including mitochondrial dysfunction, oxidative stress, myocardial cell death, and chronic inflammation. These mechanisms lead to remodeling and structural changes in the cardiovascular system, resulting in conditions such as atherosclerosis, atrial fibrillation, heart failure, and aortic valve stenosis[3].
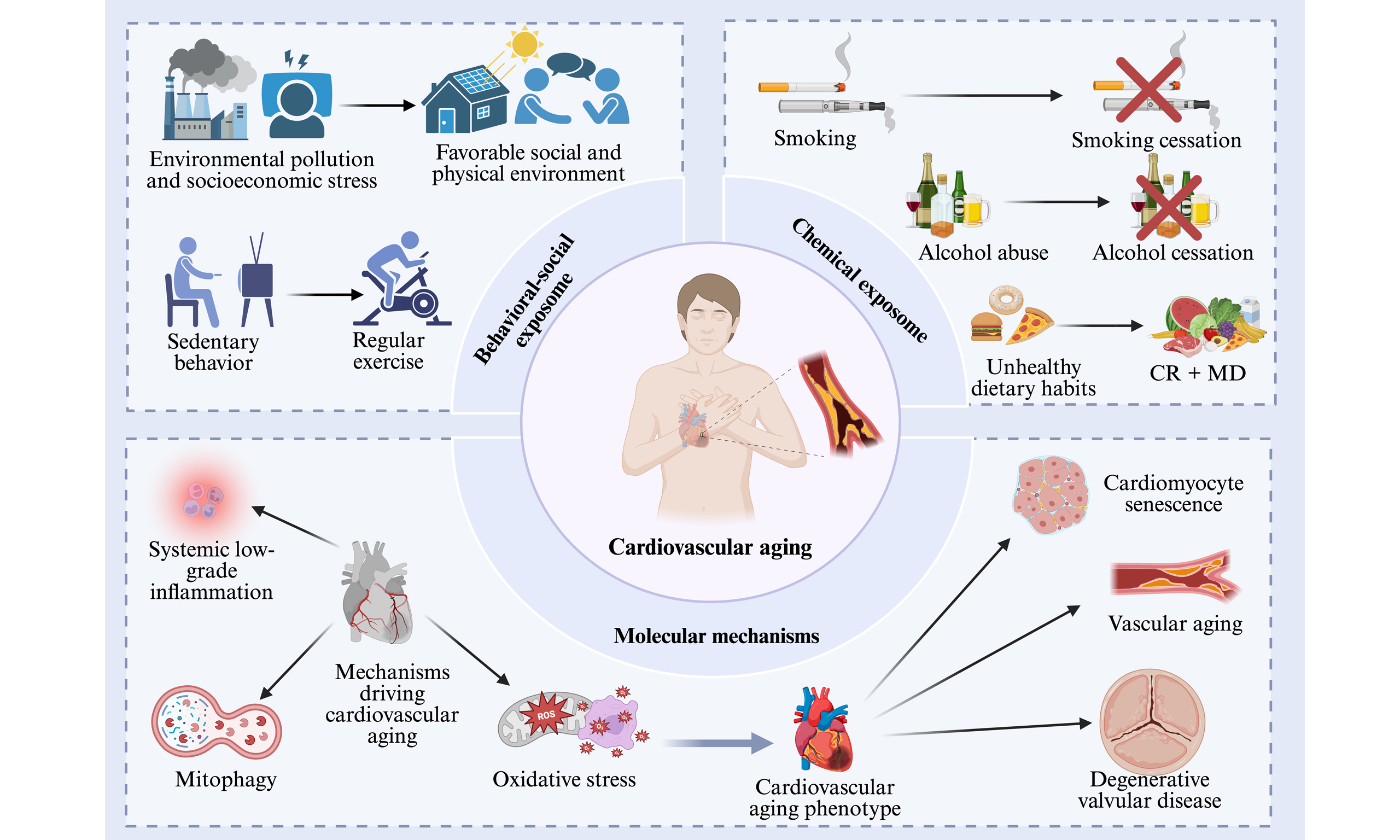
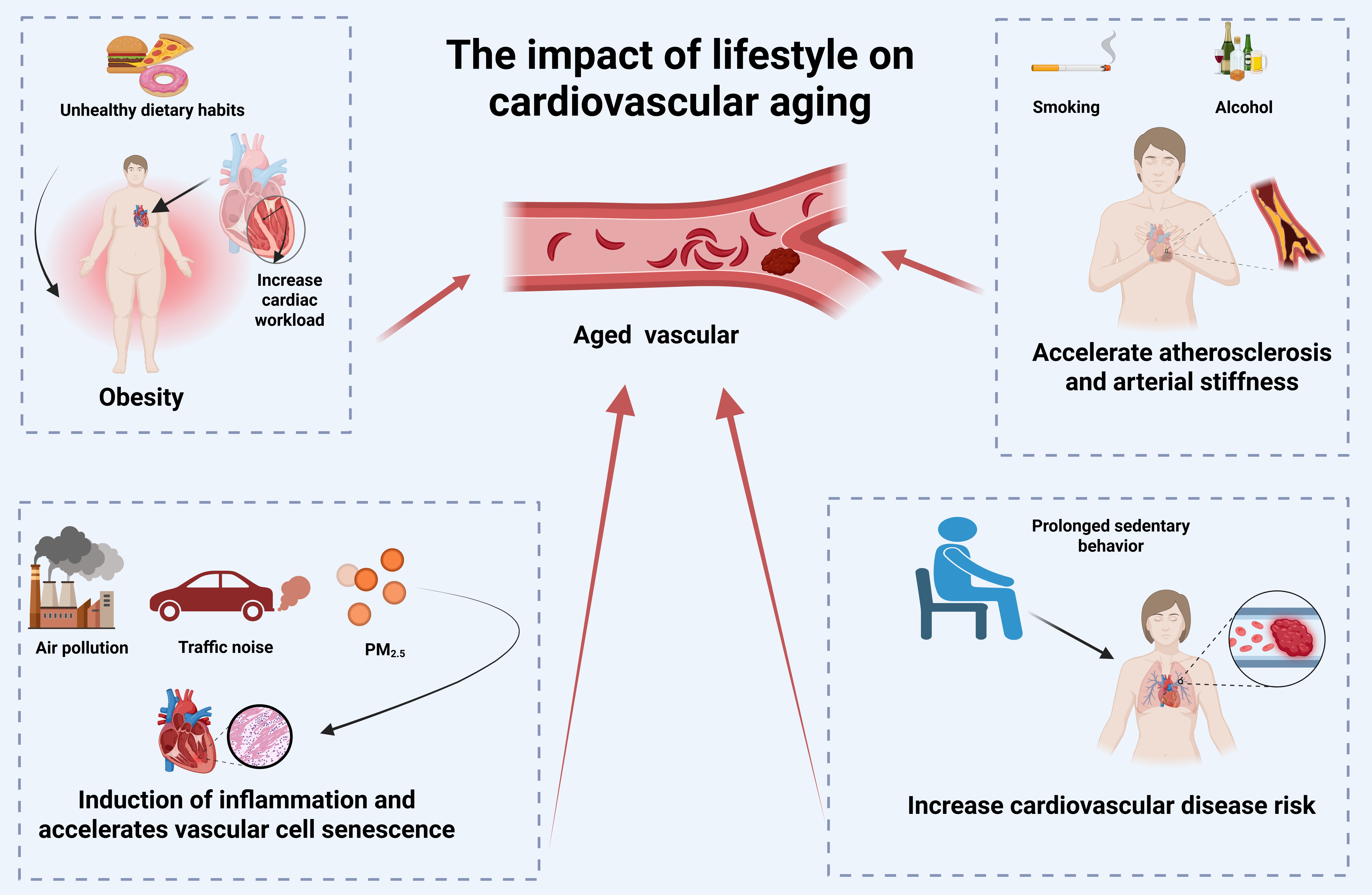
In recent years, an increasing body of research has highlighted the impact of lifestyle on aging and CVDs. Sedentary behavior[4], smoking[5], obesity[6], insufficient sleep[7], and social and psychological stress are unhealthy lifestyle factors that may serve as risk factors for cardiovascular aging, promoting the occurrence of CVDs and accelerating the aging process[8] [Figure 1]. Conversely, a healthy lifestyle, such as aerobic exercise[9] and the consumption of fruits, vegetables, and legumes[10], plays a crucial role in enhancing cardiorespiratory function and maintaining cardiovascular health. However, current research predominantly focuses on the isolated impact of single lifestyle factors on cardiovascular aging, lacking a systematic framework that integrates multiple exposomes and their complex interactions. This paper aims to revisit the cumulative damage and accelerated biological aging of the cardiovascular system resulting from shared exposures to smoking, sedentary behavior, and the natural environment, using an exposome-based approach. Based on this, targeted intervention strategies are proposed, providing insights for researchers investigating cardiovascular aging.
Figure 1. The impact of lifestyle on cardiovascular disease. Unhealthy lifestyle factors - such as sedentary behavior, smoking, alcohol consumption, obesity, and social or psychological stress - can contribute to cardiovascular aging, promote the development of cardiovascular disease, and accelerate aging. Created in BioRender Xu C (2025) https://BioRender.com/g1q8bpb.
COMPOSITION OF THE CARDIOVASCULAR EXPOSOME
Behavioral-social exposome
Sedentary behavior
Prolonged sedentary behavior is increasingly becoming one of the prevalent adverse lifestyles in modern society. In most adults, prolonged sitting occupies a large portion of daily time, and this behavior has become more prevalent due to the coronavirus disease 2019 (COVID-19) pandemic, emerging as a novel determinant of CVD risk[11]. Existing research has demonstrated a dose-response relationship between prolonged sitting time and increased CVD risk in elderly females, suggesting that extended periods of sedentary behavior may exacerbate the burden of CVDs[12], and elevate the incidence of heart failure and atrial fibrillation[13,14]. More alarmingly, a sedentary lifestyle can reduce testosterone levels, subsequently leading to depression, fatigue, decreased vitality, and weakened skeletal and muscular strength[15].
Fortunately, regular physical exercise, as a unique form of physiological stress, can trigger cardiovascular autophagy activity and exert a protective effect on the cardiovascular system. Studies have shown that exercise stimulates increased Parkin recruitment to mitochondria, thereby promoting mitochondrial autophagy. However, in Parkin knockout models, autophagy is absent, indicating that Parkin is essential for mitochondrial ubiquitination[16]. An early 10-year longitudinal cohort study confirmed that moderate physical activity can delay age-related decline in physical function[17]. It also promotes physiological left ventricular remodeling, enhances arterial function and aerobic capacity[18], improves blood pressure and maximal metabolic capacity[19], and reduces the risk of depression[20]. More recent research has provided a clearer elucidation of the benefits of exercise, with findings indicating that accumulating 9,000-10,500 steps of walking per day can significantly reduce the CVD risk associated with prolonged sitting[21], and this benefit is more pronounced in females[22]. Appropriate intensity of aerobic exercise aids in the production of reactive oxygen species (ROS), activates interleukin (IL)-6 and its receptors, as well as glycoprotein 130 and other anti-inflammatory cytokines, promoting antioxidant processes in the vascular system[23,24]. With the development of artificial intelligence, its application in cardiovascular aging-related diseases has garnered increasing attention. Limited evidence suggests that interventions using wearable technology positively influence the daily activity patterns and blood pressure changes in the elderly, potentially improving CVD risk factors[25]. In the future, encouraging moderate physical activity should be a focal point in addressing the aging trend to alleviate the health burden associated with aging.
Environmental pollution and socio-economic pressures
Air pollution is a complex mixture of particulate matter (PM) and gaseous pollutants [e.g., ozone, nitrogen oxides (NOx), and sulfur dioxide], and it is an important external factor contributing to cardiovascular aging. Major anthropogenic sources of air pollution include vehicular emissions and industrial discharges[26]. Currently, global health organizations such as the American Heart Association (AHA) and the European Society of Cardiology (ESC) have identified PM2.5 as a significant risk factor for cardiovascular morbidity and mortality[27,28]. A study examining the relationship between urban traffic design and health risks demonstrated that cities with higher levels of public transportation show greater resilience to infectious diseases and cardiovascular threats. Lower private vehicle use was also associated with reduced all-cause mortality and a lower incidence of ischemic heart disease[29].
It is noteworthy that the living environment is closely associated with the incidence and mortality of CVDs. Higher traffic density is linked to an increased risk of coronary heart disease (CHD), whereas access to parks and green spaces is associated with a reduced incidence of CHD[30]. These associations are partly explained by environmental exposures, as poorer living conditions are often characterized by higher levels of air pollution and noise, both of which are closely linked to the development of CVDs[31]. More direct evidence shows that living within 150 meters of major urban roads, compared with distances greater than 300 meters, is associated with a 6.67% increase in carotid intima-media thickness[32]. Epigenetic mechanisms, particularly DNA methylation, may further underlie these associations. DNA methylation patterns are influenced by traffic noise, air pollution, and green space exposure, and are enriched in pathways related to inflammation, cellular development, and immune responses[2,33].
External social stressors, including work stress[34] and low socioeconomic status[35], are significant risk factors for CVDs. These factors can trigger psychological stress and lead to physiological and behavioral responses, including anxiety[36], depression[37], and social inhibition[38]. Psychological stress promotes immune activation and inflammation, as well as endothelial dysfunction[39], thereby increasing the risk of adverse cardiovascular events such as CHD and heart failure. Together, these findings highlight the importance of both the living environment and the social context in shaping cardiovascular health.
Chemical exposome
Smoking
Smoking is one of the most significant risk factors for CVDs, with tobacco-derived smoke containing highly toxic compounds: nitrosamines and polycyclic aromatic hydrocarbons (PAHs). These compounds are known carcinogens and are associated with various complications, including CVDs[40,41]. Evidence indicates that smoking increases the incidence and mortality of CVDs, which is linked to smoking-induced endothelial dysfunction, reduced high-density lipoprotein (HDL) levels, and exacerbated inflammatory states[42]. Studies have shown that prolonged exposure to cigarette smoke in mice leads to macrophage infiltration, inducing vascular endothelial damage, increased nicotinamide adenine dinucleotide phosphate (NADPH) oxidase levels, and ROS production in the vasculature, thereby triggering a vicious cycle of oxidative stress and endothelial damage[43]. The harmful effects of secondhand smoke are also significant, and it has become an independent risk factor for CVDs. Research has found that exposure to secondhand smoke induces myocardial cell dysfunction, disrupts intracellular Ca2+ homeostasis, promotes cellular apoptosis, and impairs autophagy, with adiponectin deficiency exacerbating these effects[44].
There is a widespread misconception regarding the addictive potential and adverse health effects of waterpipe smoke, with many people believing that waterpipe smoking is less harmful and less addictive than cigarette smoking. However, the opposite is true; many harmful substances found in tobacco smoke are also present in waterpipe smoke, often at higher concentrations[45]. Notably, the AHA has stated that the acute cardiopulmonary toxicity of a single waterpipe smoking session is more severe than that of smoking a single cigarette, due to significantly higher levels of cardiopulmonary toxicants such as heavy metals and PM[46]. Research has shown that waterpipe smoking is associated with significantly lower levels of vascular endothelial growth factor (VEGF) in adolescents[47]. Additionally, long-term waterpipe smoking significantly increases the severity of coronary artery atherosclerosis, with similar cardiovascular changes being more pronounced in physically inactive individuals[48], indicating a critical role for physical activity in delaying cardiovascular aging.
This situation is similarly concerning for e-cigarettes. When e-cigarette liquids are heated, toxic aldehydes and ketones - notably formaldehyde and acrolein - are produced. These compounds can form toxic polymers with proteins and DNA, significantly elevating serum endothelin-1 (ET-1) and IL-8 levels, thereby exacerbating oxidative stress, mitochondrial dysfunction, and inflammation. This process intensifies with prolonged exposure to e-cigarettes[49]. Another study indicates that phagocytic NADPH oxidase (NOX-2) is one of the mechanisms by which e-cigarettes exert their effects. E-cigarette vapor exposure increases oxidative stress in the vasculature, brain, and lungs through NOX-2-dependent mechanisms, significantly impairing endothelial function and increasing arterial stiffness[50]. Therefore, smoking cessation is crucial; it can lower circulating ET-1 and Tumour Necrosis Factor alpha (TNF-α) concentrations, reduce systemic inflammation, and decrease the risk of CVDs[51].
Alcohol consumption
Alcohol is another potential factor that endangers cardiovascular health. Genetic studies indicate that alcohol consumption may shorten lifespan[52]. Chronic heavy alcohol consumption can lead to alcoholic cardiomyopathy, characterized by left ventricular dilation, reduced myocardial contractility, and myocardial fibrosis, ultimately resulting in heart failure and arrhythmias[53]. Pulse wave velocity (PWV) is an independent indicator for assessing arterial stiffness and predicting CVD; studies have shown that light-to-moderate alcohol consumption is associated with higher PWV, thereby increasing arterial stiffness, while high alcohol intake accelerates arterial aging[54].
Ethanol is primarily enzymatically converted to acetaldehyde by alcohol dehydrogenase (ADH), which is then metabolized to acetate by aldehyde dehydrogenase. Acetaldehyde is subsequently transported to the mitochondria, where it is further converted to acetyl coenzyme A (acetyl-CoA). Excessive alcohol consumption can damage mitochondrial function, disrupt cholesterol and triglyceride metabolism, promote oxidative stress and inflammatory responses, and accelerate myocardial cell death and aging, leading to impaired cardiac diastolic function and reduced cardiovascular reserve capacity[55]. Peroxides can react with nitric oxide (NO) in a diffusion-limited manner to form peroxynitrite, a reactive nitrogen species (RNS). Both RNS and ROS can damage contractile proteins, enzymes, and mitochondria through oxidative and nitrosative processes, intensifying stress signaling, exacerbating cellular dysfunction, and ultimately leading to myocardial and endothelial cell (EC) death[56]. These findings suggest that alcohol intake is an independent risk factor for CVDs, and chronic heavy drinking impairs cardiovascular function, increasing the risk of CVDs. Reducing alcohol intake could help ameliorate the process of cardiovascular aging.
Dietary patterns
Caloric restriction is a dietary regimen that has been shown to benefit vascular health. Evidence indicates that animals subjected to caloric restriction exhibit signs of vascular rejuvenation, which may be associated with reduced extracellular matrix (ECM) and collagen deposition in the vascular wall, activation of endothelial nitric oxide synthase (eNOS), and increased NO levels, thereby mitigating oxidative stress and inflammation[57,58]. Nuclear factor erythroid-2-related factor 2 (Nrf2) confers protective effects against cellular oxidative stress; however, this effect diminishes with age. Further research has shown that caloric restriction reduces apoptosis in microvascular ECs of the brain in rats, significantly lowers the expression of miR-144, and upregulates NRF2, thereby restoring the physiological function of vascular ECs[59]. Similar phenomena occur at the cellular level, as the use of serum from calorically restricted monkeys to culture vascular ECs results in upregulated VEGF expression[60], suggesting that moderate caloric restriction may be a potential mechanism for improving cardiovascular function.
The Mediterranean diet (MD) is characterized by a high intake of vegetables, legumes, fruits, fish, nuts, and olive oil, with a reduced consumption of red meat and processed dairy products. It is currently considered one of the most healthful dietary patterns, with significant antioxidant and anti-inflammatory effects[61]. A strict adherence to the MD can significantly reduce the probability of vascular aging[62]. The Mediterranean-DASH (Dietary Approaches to Stop Hypertension) Intervention for Neurodegenerative Delay (MIND) diet is a novel dietary approach that combines elements of the Mediterranean and DASH diets, emphasizing the importance of berries and leafy greens[63]. These foods, rich in vitamins E, folate, polyphenols, flavonoids, monounsaturated fatty acids (MUFAs), and omega-3 fatty acids, can inhibit the deposition of amyloid-beta (Aβ). Incorporating them into a healthy diet can serve as a lifestyle strategy to reduce the risk of CVDs and Alzheimer’s disease (AD)[64]. Interestingly, a study has shown that moderate coffee consumption (1 to 5 cups per day) is associated with a reduced risk of heart failure, type 2 diabetes (T2D), and chronic kidney disease (CKD)[65]. However, further understanding of its underlying mechanisms is necessary to determine its benefits for cardiovascular health. Nevertheless, it is undoubtedly true that quality dietary habits confer significant benefits on the cardiovascular system.
MOLECULAR MECHANISMS OF CARDIOVASCULAR AGING INDUCED BY EXPOSOME
Oxidative stress
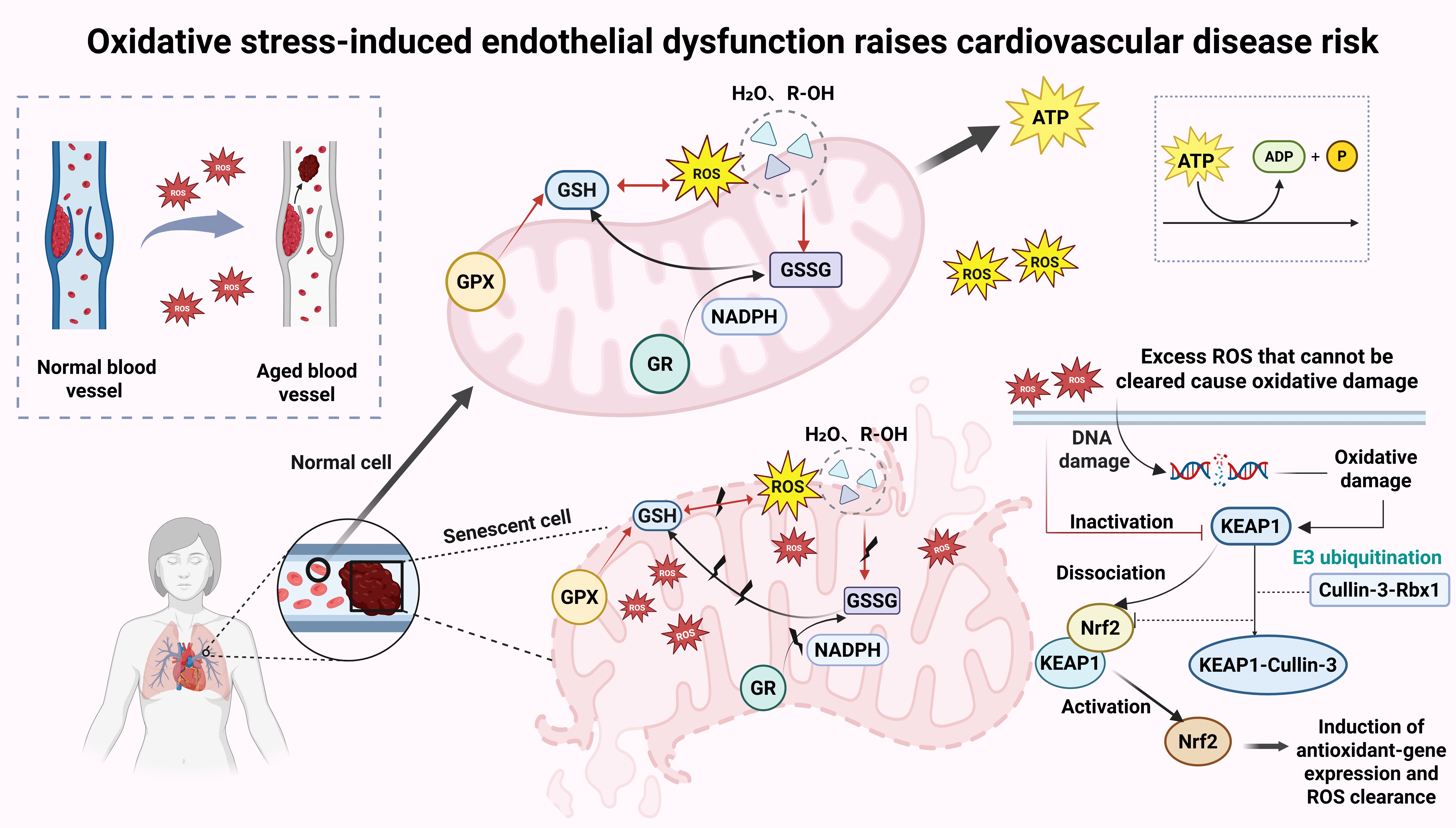
Oxidative stress-induced endothelial dysfunction is a critical mechanism for the increased risk of CVDs. With age, ROS gradually increase, while the capacity for endogenous antioxidant defense decreases, leading to reduced bioavailability of NO and impaired vascular dilation[66]. Under normal conditions, cells exert antioxidant functions through the production of glutathione peroxidase (GPX), glutathione reductase, catalase, NADPH-quinone oxidoreductase-1 (NQO1), heme oxygenase (HO-1), thioredoxin (Trx), and sulfiredoxin (Srx)[67]. In this process, cardiomyocytes maintain their function through high aerobic metabolism, with mitochondria accounting for 45% of cardiac cell volume; thus, mitochondrial oxidative phosphorylation is the primary source of adenosine triphosphate (ATP) in cardiomyocytes. Previous studies have highlighted that mitochondrial oxidative stress can lead to mutations in mitochondrial DNA (mtDNA) and increased permeability, thereby impairing mitochondrial and overall cardiomyocyte functions and playing a crucial role in the antioxidant process[68]. Additionally, it has been shown that transcription factor NRF2 is a key regulator of cellular redox homeostasis. Under conditions of oxidative damage, Kelch-like ECH-associated protein 1 (KEAP1) can dissociate from the NRF2-KEAP1 complex, thereby activating NRF2 to exert antioxidant functions[69] [Figure 2]. However, the detrimental effects of oxidative stress are not irreversible. Accumulating evidence suggests that a healthy lifestyle, characterized by moderate physical activity and balanced nutrition, can activate antioxidant mechanisms, thus achieving the goal of delaying aging[70].
Figure 2. Oxidative stress-induced endothelial dysfunction increases cardiovascular disease risk. Oxidative stress elevates ROS levels, causes endothelial dysfunction, damages mitochondrial oxidative phosphorylation, gradually reduces endogenous antioxidant defenses, and impairs vasodilator function. Created in BioRender Xu C (2025) https://BioRender.com/pad4sgo. ROS: Reactive oxygen species; GPX: glutathione peroxidase; ATP: adenosine triphosphate; ADP: adenosine diphosphate; GR: glutathione reductase; NADPH: nicotinamide adenine dinucleotide phosphate; GSSG: glutathione disulfide; GSH: glutathione.
Systemic low-grade inflammation
Inflammation is one of the key factors in vascular aging and plays a central role in age-related chronic diseases. Frasca et al. coined the term “inflammaging” to describe the chronic, low-grade, and systemic inflammation that occurs with age[71]. Studies have shown that chronic inflammation elevates the expression of biomarkers for heart failure, such as high-sensitivity cardiac troponin T (hs-cTnT), and exacerbates frailty in patients with CVDs[72]. Currently, pro-inflammatory molecules identified to be involved in the inflammatory process include IL-1, IL-6, IL-8, IL-13, IL-18, C-reactive protein (CRP), nucleotide-binding oligomerization domain-like receptor protein 3 (NLRP3) inflammasome, and TNF-α and its receptors[73,74], which are strongly associated with age-related diseases such as cancer[75], atherosclerosis[76], and AD[77]. Interestingly, some perspectives posit that certain inflammation may be beneficial; regular exposure to inflammatory stimuli can train the immune system to fine-tune its cellular processes, potentially leading to better outcomes in the face of life-threatening diseases[78].
Unhealthy lifestyle habits accelerate both vascular aging and cognitive decline. A study investigating the effects of a high-fat diet on systemic inflammation in aged mice demonstrated that a high-fat diet induces both systemic and localized neuroinflammation, alters exploratory behavior of both male and female mice, and impairs learning and memory abilities of male mice[79]. In contrast, a healthy lifestyle can help improve the inflammatory environment. For example, oats can reduce the expression of Eotaxin-1 protein, thereby alleviating systemic chronic inflammation[80], and increasing the intake of foods rich in omega-3, such as those found in the MD, is recommended[81]. Regular moderate exercise can also prevent premature vascular aging and reduce the risk of all-cause mortality[82]. Notably, the living environment is also a significant exacerbating factor in cardiovascular inflammation aging; for instance, long-term exposure to micro- and nanoplastics can promote oxidative stress, impair mitochondrial function, damage autophagy, and activate DNA damage responses, thereby increasing the production of pro-inflammatory factors[83]. Moreover, unhealthy social relationships, such as increased feelings of loneliness and social stress, can lead to glucocorticoid resistance, up-regulated expression of pro-inflammatory genes, and oxidative stress[84,85]. Considering these factors comprehensively is crucial for reducing inflammatory responses and promoting cardiovascular health.
Impaired autophagy
Cellular homeostasis is a prerequisite for optimal cardiovascular function. Autophagy, by removing accumulated proteins and damaged organelles, balances biosynthesis and catabolism of macromolecules, thereby maintaining cellular homeostasis and protecting organisms from CVDs such as myocardial infarction and atherosclerosis[86]. However, cardiac autophagy is impaired with age, leading to the accumulation of dysfunctional mitochondria and other metabolic waste in aging hearts. Activation of autophagy can help ameliorate cardiovascular damage caused by aging. Studies have confirmed that PTEN-induced putative kinase 1 (PINK)/Parkin[87], BCL2 Interacting Protein 3-Like/NIP3-like protein X (BNIP3L/NIX)[88], and FUN14 Domain Containing 1 (FUNDC1)[89] are critical pathways mediating mitophagy, which alleviates damage from myocardial infarction and heart failure. Consequently, autophagy has emerged as a potential therapeutic target for age-related cardiac dysfunction[90]. Current research suggests that mechanistic target of rapamycin (mTOR), AMP-activated protein kinase (AMPK), Autophagy-related gene (ATG) proteins, as well as insulin/Insulin-like Growth Factor 1 (IGF1), Target of Rapamycin (TOR), Protein Kinase A (PKA), and AKR mouse strain T/Protein Kinase B (AKT/PKB), show age-related changes and may regulate or interact with autophagy. These molecules are therefore considered promising targets for autophagy regulation[91]. DBI/ACBP (dibenzylindole/acyl-CoA binding protein) is a phylogenetically conserved paracrine inhibitor that promotes aging by inhibiting macroautophagy/autophagy, while the knockdown of Acyl-CoA-binding protein (ACB1) induces autophagy and extends lifespan in an autophagy-dependent manner[92].
Previous studies have shown that trehalose, spermidine, and polyphenols are natural autophagy activators. They enhance myocardial relaxation function and reduce cardiac hypertrophy by activating transcription factor EB[93], improve myocardial diastolic function and reduce cardiac hypertrophy[94], thereby positively affecting redox and autophagy and promoting the repair of CVDs[95]. Spermidine, a natural polyamine present in durian, mushrooms, fresh green peppers, amaranth, broccoli, and soy products[96], delays the onset of cardiovascular and neurodegenerative diseases by stimulating protective autophagy, making it a candidate for aging research[97]. Recent studies have shown that lifestyle changes, such as caloric restriction and intermittent fasting, can induce enhanced autophagy, significantly reducing the incidence of CVDs and extending lifespan[98]. Additionally, physical exercise has emerged as an important intervention for regulating autophagy. Moderate-intensity physical activity upregulates proteins such as BECLIN1, microtubule-associated protein 1 light chain 3 (LC3), and autophagy-related protein 12 (ATG12), thereby enhancing autophagy, reducing ROS production, inhibiting NLRP3 inflammasome activation, and lowering IL-1β and IL-18 levels, ultimately restoring cellular homeostasis and reducing age-related inflammation[99]. This plays a positive role in mitigating the effects of aging and preventing age-related diseases.
AGING PHENOTYPES CAUSED BY DYSREGULATION OF MOLECULAR MECHANISMS
Cardiac remodeling
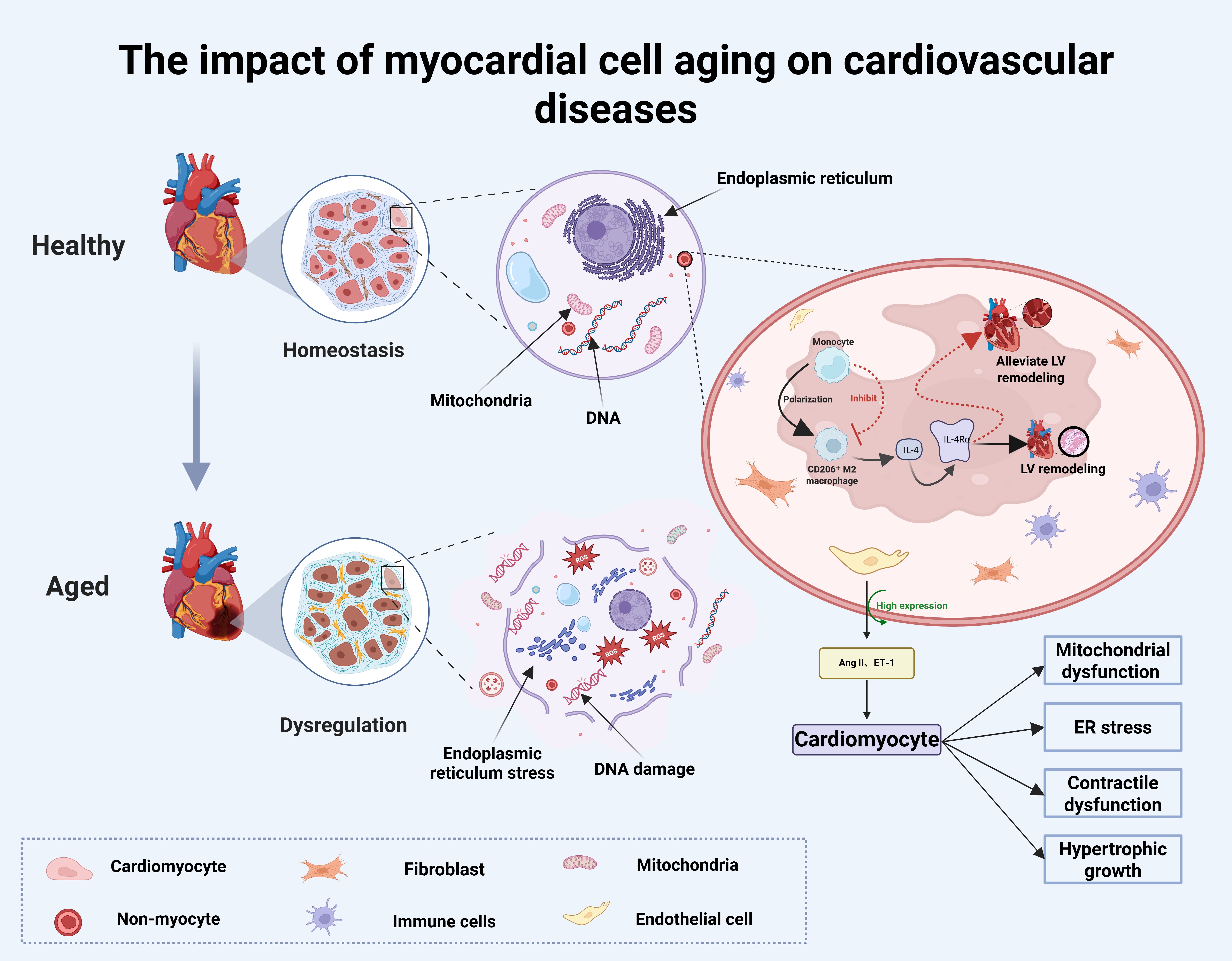
Cardiomyocytes are crucial for maintaining cardiac function. Given that the heart lacks endogenous cardiomyocyte regeneration capacity, the impact of cardiomyocyte death cannot be naturally reversed and is a primary cause of reduced cardiac function in CVDs[100,101]. The aging of cardiomyocytes is a result of various functional impairments. In aged cardiomyocytes, double-strand breaks (DSBs) activate cytoplasmic DNA sensing proteins (CDSPs), which, together with the DNA damage response pathway (DDR), induce the senescence-associated secretory phenotype (SASP)[102]. Furthermore, the accumulation of mitochondrial ROS[103]induces DNA damage characterized by telomere shortening[104], which subsequently triggers programmed cell death mechanisms such as apoptosis, autophagy, pyroptosis, and ferroptosis[105]. These processes lead to a reduction in the absolute number of cardiomyocytes, thereby promoting the development of CVDs, including myocardial infarction, ischemia-reperfusion injury, and heart failure.
In fact, another crucial component of cardiac tissue - non-myocytes, including ECs, fibroblasts, and immune cells[106] - is equally essential for the function of myocytes. Functional impairment of fibroblasts induced by aging and mechanical stress exacerbates the functional impairment and aging of myocytes[107]. ECs, through their high expression and release of Angiotensin II (AngII) and ET-1, can stimulate mitochondrial dysfunction, endoplasmic reticulum stress, contractile dysfunction, and hypertrophic growth in myocytes[108]. Macrophages are key immune cells in myocardial pathological remodeling and fibrosis, and inhibiting the CD206 macrophage IL-4Rα signaling pathway can reduce left ventricular remodeling in ischemic cardiomyopathy[109], thereby delaying the progression of heart disease [Figure 3]. However, encouragingly, research has already highlighted the benefits of the ECM on myocytes, which can promote myocyte proliferation, induce re-entry into the cell cycle, and protect against stress responses in senescent cells[110]. Further studies have revealed that the absence of Sirtuin 2 (SIRT2) in myocytes is a key feature of human cardiac aging, and the SIRT2-STAT3-Cyclin Dependent Kinase Inhibitor 2B (CDKN2B) regulatory axis is a potential therapeutic target for CVDs[111]. Emerging clinical evidence suggests that increasing cellular Nicotinamide adenine dinucleotide (NAD) content may be a promising therapeutic approach[112]. Nicotinamide riboside (NR) as a dietary supplement can be converted to NAD in cells to support mitochondrial energy metabolism. Mice treated with NR after ischemia/reperfusion (I/R) exhibited higher ejection fractions and smaller infarct areas[113]. Additionally, exercise is an important measure to delay myocyte aging, and moderate exercise training can improve myocyte function and delay age-related pathological changes in the heart[114]. This indicates that lifestyle changes can significantly enhance the vitality of myocytes.
Figure 3. The impact of myocardial cell aging on cardiovascular diseases. Cardiovascular aging results from dysfunction in both cardiomyocytes and non-myocytes, including DNA damage responses, endoplasmic reticulum stress, mitochondrial dysfunction, SASP release, and other cellular changes. Created in BioRender Xu C (2025) https://BioRender.com/url7kgt. SASP: Senescence-associated secretory phenotype; ER: endoplasmic reticulum.
Vascular aging
Vascular aging is a gradual process. Large longitudinal studies have shown that vascular endothelial dysfunction, a specific phenotype of vascular aging, is a core factor in the increased incidence and mortality of CVDs. Vascular ECs play an important physiological role in vascular homeostasis, maintaining blood
Evidence has shown that vascular aging is positively correlated with sedentary time and negatively correlated with physical activity[129]. Regular and adequate aerobic exercise can significantly slow vascular aging, reduce age-related arterial stiffness, and lower the incidence of CVDs[130]. Excessive salt intake is another lifestyle that exacerbates vascular aging. Reducing salt intake helps restore aortic elasticity and reduces the risk of CVD by reducing the production of the pro-fibrotic factor Marinobufagenin (MBG)[131]. Surprisingly, vascular aging can occur in childhood[132] and progresses with increased psychosocial stress and unhealthy habits, such as exposure to air pollution and smoking[133]. It can be seen that early lifestyle changes and living environment adjustments have a profound impact on vascular aging.
Degenerative changes in valves
The mitral valve is a complex structure made up of anterior and posterior leaflets, chordae tendineae, and Papillary muscle. Its main function is to allow blood to flow from the left atrium to the left ventricle during diastole. It also prevents blood from flowing backward during systole, playing a crucial role in normal heart function[134]. With age, the compliance of cardiac valves to mechanical stress decreases, making them susceptible to degeneration, which is typically manifested as mitral valve calcification. The prevalence of this condition rises from 10% in people aged 60-69 to 30% in those aged 70-79, and reaches 60% in individuals over 80. It has become an independent risk factor for all-cause and cardiovascular mortality[135]. Mitral valve calcification can lead to mitral stenosis and mitral regurgitation[136,137]. Functional mitral regurgitation can result in left atrial dilation and reduced left atrial compliance, leading to adverse outcomes such as heart failure[138]. The aging process of the mitral valve is influenced by multiple molecular pathways. On the one hand, the renin-angiotensin system (RAS) induces inflammation by stimulating the release of inflammatory cytokines through angiotensin II, increasing the production of ROS[139]; On the other hand, the TGF-β signaling pathway enhances collagen accumulation, promoting the differentiation of fibroblasts into myofibroblasts, thus leading to valve fibrosis[140].
Recent evidence highlights the involvement of mitochondria in the development and progression of aortic valve stenosis. A key function of mitochondria is their involvement in the unfolded protein response (UPR). In this process, mitochondria reduce the accumulation of misfolded proteins, thereby lowering cell death and fibrosis, and decreasing plasma levels of cardiac injury biomarkers, such as high-sensitivity troponin T[141]. Additionally, Rogers et al. discovered that dynamin-related protein 1 (DRP1) is a critical regulator of mitochondrial fission in calcified human aortic valves. RNA-mediated interference in Human Aortic Valve Interstitial Cells (HAVICs) reduces calcium deposition by increasing SRY-box transcription factor 9 (SOX9) expression, thereby providing protective effects on cardiac tissue[142].
A 10-year absolute risk chart for aortic valve stenosis shows that factors such as high body mass index, poor adherence to dietary guidelines, smoking, and excessive alcohol use can raise the 10-year risk from less than 1% to 19%. This suggests that lifestyle changes can help reduce the risk of valvular heart disease[143]. Notably, long-term exposure to severe air pollution is a long-neglected factor contributing to valve calcification, with left-sided valvular calcification serving as a marker for CVD risk, directly related to atherosclerosis[144]. Therefore, a good living environment and a healthy lifestyle are beneficial factors in delaying valvular heart disease and maintaining cardiovascular health.
CONCLUSION
Aging of the cardiovascular system is a complex and inevitable physiological process, but it is strongly influenced by intrinsic biological factors, lifestyle choices, and the surrounding environment.
At the biological level, degeneration of cardiac and vascular structure and function serves as the pathological basis for cardiovascular aging. Vascular endothelial dysfunction, calcification of heart valves, and apoptosis of myocardial cells directly impair cardiac pumping efficiency. At the molecular level, chronic inflammation, impaired autophagy, mitochondrial dysfunction, and oxidative stress act as catalysts for aging, activating a series of age-related signaling pathways that accelerate cellular senescence and apoptosis.
However, these intrinsic aging processes are not independent; their rate and severity are significantly amplified by adverse lifestyle and environmental factors. Smoking and excessive alcohol consumption are well-recognized exogenous aging triggers. Harmful substances in tobacco directly induce oxidative stress and inflammation, damaging the vascular endothelium; alcohol may elevate blood pressure and exacerbate cardiac burden. Sedentary lifestyles contribute to energy metabolic imbalance, promoting obesity, insulin resistance, and dyslipidemia - each of which is an independent risk factor for CVD. Additionally, air pollution, an often-overlooked factor, has become a significant threat to cardiovascular health in modern society. These fine particulates can enter the bloodstream via the alveoli, inducing systemic oxidative stress and inflammation, and directly attacking the cardiovascular system.
In summary, cardiovascular aging is a result of the continuous interaction between intrinsic biological decline and extrinsic environmental factors such as lifestyle. Understanding this intertwined network not only elucidates the underlying mechanisms of aging but also points the way forward for interventions. By adopting positive lifestyle interventions, such as smoking cessation, moderate alcohol consumption, regular exercise, and improving the living environment, intrinsic biological aging can be delayed. On the other hand, through bioinformatics and artificial intelligence methods, integrating large global datasets with genomics, transcriptomics, and proteomics techniques, key biological targets in the process of cardiovascular aging can be identified. This will facilitate a comprehensive system from precise molecular targeted therapy to social care and lifestyle improvement, with the ultimate goal of achieving healthy cardiovascular aging.
DECLARATIONS
Acknowledgments
The Graphical Abstract was created with BioRender.com (Created in BioRender Xu C (2026) https://BioRender.com/ry3q6if).
Authors’ contributions
Made contributions to this article equally: Zhang, S.; Xu, C.
Made substantial contributions to conception and design of the study: Zhang, X.; Song, Y.
Performed data acquisition and provided technical and material support: Liu, S.; Li, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020;396:1204-22.
2. Xu R, Li S, Li S, et al. Residential surrounding greenness and DNA methylation: an epigenome-wide association study. Environ Int. 2021;154:106556.
3. Zhang W, Song M, Qu J, Liu GH. Epigenetic modifications in cardiovascular aging and diseases. Circ Res. 2018;123:773-86.
4. Lavie CJ, Ozemek C, Carbone S, Katzmarzyk PT, Blair SN. Sedentary behavior, exercise, and cardiovascular health. Circ Res. 2019;124:799-815.
5. Teo KK, Ounpuu S, Hawken S, et al.; INTERHEART Study Investigators. Tobacco use and risk of myocardial infarction in 52 countries in the INTERHEART study: a case-control study. Lancet. 2006;368:647-58.
6. Carbone S, Del Buono MG, Ozemek C, Lavie CJ. Obesity, risk of diabetes and role of physical activity, exercise training and cardiorespiratory fitness. Prog Cardiovasc Dis. 2019;62:327-33.
7. Zhao B, Meng Y, Jin X, et al. Association of objective and self-reported sleep duration with all-cause and cardiovascular disease mortality: a community-based study. J Am Heart Assoc. 2023;12:e027832.
8. Kaminsky LA, German C, Imboden M, Ozemek C, Peterman JE, Brubaker PH. The importance of healthy lifestyle behaviors in the prevention of cardiovascular disease. Prog Cardiovasc Dis. 2022;70:8-15.
9. Mehta A, Kondamudi N, Laukkanen JA, et al. Running away from cardiovascular disease at the right speed: the impact of aerobic physical activity and cardiorespiratory fitness on cardiovascular disease risk and associated subclinical phenotypes. Prog Cardiovasc Dis. 2020;63:762-74.
10. Miller V, Mente A, Dehghan M, et al.; Prospective Urban Rural Epidemiology (PURE) study investigators. Fruit, vegetable, and legume intake, and cardiovascular disease and deaths in 18 countries (PURE): a prospective cohort study. Lancet. 2017;390:2037-49.
11. Dunstan DW, Dogra S, Carter SE, Owen N. Sit less and move more for cardiovascular health: emerging insights and opportunities. Nat Rev Cardiol. 2021;18:637-48.
12. Bellettiere J, LaMonte MJ, Evenson KR, et al. Sedentary behavior and cardiovascular disease in older women: the OPACH study. Circulation. 2019;139:1036-46.
13. Boursiquot BC, Bellettiere J, LaMonte MJ, LaCroix AZ, Perez MV. Sedentary behavior and atrial fibrillation in older women: the OPACH study. J Am Heart Assoc. 2022;11:e023833.
14. LaMonte MJ, LaCroix AZ, Nguyen S, et al. Accelerometer-measured physical activity, sedentary time, and heart failure risk in women aged 63 to 99 years. JAMA Cardiol. 2024;9:336-45.
15. Abdel-Sater KA. The role of anti-aging approaches in managing hypogonadism in sedentary older males. Front Aging. 2024;5:1514438.
16. Chen CCW, Erlich AT, Crilly MJ, Hood DA. Parkin is required for exercise-induced mitophagy in muscle: impact of aging. Am J Physiol Endocrinol Metab. 2018;315:E404-15.
17. Moreno-Agostino D, Daskalopoulou C, Wu YT, et al. The impact of physical activity on healthy ageing trajectories: evidence from eight cohort studies. Int J Behav Nutr Phys Act. 2020;17:92.
18. Fujimoto N, Prasad A, Hastings JL, et al. Cardiovascular effects of 1 year of progressive and vigorous exercise training in previously sedentary individuals older than 65 years of age. Circulation. 2010;122:1797-805.
19. Grace F, Herbert P, Elliott AD, Richards J, Beaumont A, Sculthorpe NF. High intensity interval training (HIIT) improves resting blood pressure, metabolic (MET) capacity and heart rate reserve without compromising cardiac function in sedentary aging men. Exp Gerontol. 2018;109:75-81.
20. Yang Y, Wang Y, Yang L. Association between physical activity and sedentary behavior and depression in US adults with cardiovascular disease: NHANES 2007-2016. J Affect Disord. 2024;367:342-9.
21. Ahmadi MN, Rezende LFM, Ferrari G, Del Pozo Cruz B, Lee IM, Stamatakis E. Do the associations of daily steps with mortality and incident cardiovascular disease differ by sedentary time levels? A device-based cohort study. Br J Sports Med. 2024;58:261-8.
22. Ji H, Gulati M, Huang TY, et al. Sex differences in association of physical activity with all-cause and cardiovascular mortality. J Am Coll Cardiol. 2024;83:783-93.
23. Pedersen BK. Anti-inflammatory effects of exercise: role in diabetes and cardiovascular disease. Eur J Clin Investig. 2017;47:600-11.
24. Villar-Fincheira P, Sanhueza-Olivares F, Norambuena-Soto I, et al. Role of interleukin-6 in vascular health and disease. Front Mol Biosci. 2021;8:641734.
25. Roberts LM, Jaeger BC, Baptista LC, et al. Wearable technology to reduce sedentary behavior and CVD risk in older adults: a pilot randomized clinical trial. Clin Interv Aging. 2019;14:1817-28.
26. Al-Kindi SG, Brook RD, Biswal S, Rajagopalan S. Environmental determinants of cardiovascular disease: lessons learned from air pollution. Nat Rev Cardiol. 2020;17:656-72.
27. Brook RD, Rajagopalan S, Pope CA 3rd, et al.; American Heart Association Council on Epidemiology and Prevention, Council on the Kidney in Cardiovascular Disease, and Council on Nutrition, Physical Activity and Metabolism. Particulate matter air pollution and cardiovascular disease: an update to the scientific statement from the American Heart Association. Circulation. 2010;121:2331-78.
28. Brauer M, Casadei B, Harrington RA, Kovacs R, Sliwa K.; WHF Air Pollution Expert Group. Taking a stand against air pollution - the impact on cardiovascular disease: a joint opinion from the world heart federation, american college of cardiology, american heart association, and the european society of cardiology. Eur Heart J. 2021;42:1460-3.
29. Nice KA, Thompson J, Zhao H, et al. Effects of city design on transport mode choice and exposure to health risks during and after a crisis: a retrospective observational analysis. Lancet Planet Health. 2025;9:e467-79.
30. Chen H, Burnett RT, Bai L, et al. Residential greenness and cardiovascular disease incidence, readmission, and mortality. Environ Health Perspect. 2020;128:87005.
31. Zhang K, Brook RD, Li Y, Rajagopalan S, Kim JB. Air pollution, built environment, and early cardiovascular disease. Circ Res. 2023;132:1707-24.
32. Wang Y, Wellenius GA, Hickson DA, Gjelsvik A, Eaton CB, Wyatt SB. Residential proximity to traffic-related pollution and atherosclerosis in 4 vascular beds among African-American adults: results from the Jackson heart study. Am J Epidemiol. 2016;184:732-43.
33. Eze IC, Jeong A, Schaffner E, et al. Genome-wide DNA methylation in peripheral blood and long-term exposure to source-specific transportation noise and air pollution: the SAPALDIA study. Environ Health Perspect. 2020;128:67003.
34. Niedhammer I, Bertrais S, Witt K. Psychosocial work exposures and health outcomes: a meta-review of 72 literature reviews with meta-analysis. Scand J Work Environ Health. 2021;47:489-508.
35. Potter EL, Hopper I, Sen J, Salim A, Marwick TH. Impact of socioeconomic status on incident heart failure and left ventricular dysfunction: systematic review and meta-analysis. Eur Heart J Qual Care Clin Outcomes. 2019;5:169-79.
36. Li J, Ji F, Song J, et al. Anxiety and clinical outcomes of patients with acute coronary syndrome: a meta-analysis. BMJ Open. 2020;10:e034135.
37. Cao H, Zhao H, Shen L. Depression increased risk of coronary heart disease: a meta-analysis of prospective cohort studies. Front Cardiovasc Med. 2022;9:913888.
38. Wang Y, Huang B, Sun M, Yu B, Lin P. Type D personality as a risk factor for 3-year cardiovascular events in patients with coronary artery disease and their spouse: a prospective cohort study. Eur J Prev Cardiol. 2025;32:430-40.
39. Wirtz PH, von Känel R. Psychological stress, inflammation, and coronary heart disease. Curr Cardiol Rep. 2017;19:111.
40. Konstantinou E, Fotopoulou F, Drosos A, et al. Tobacco-specific nitrosamines: a literature review. Food Chem Toxicol. 2018;118:198-203.
41. Kim KH, Jahan SA, Kabir E, Brown RJ. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ Int. 2013;60:71-80.
42. Messner B, Bernhard D. Smoking and cardiovascular disease: mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler, Thromb, Vasc Biol. 2014;34:509-15.
43. El-Mahdy MA, Abdelghany TM, Hemann C, et al. Chronic cigarette smoke exposure triggers a vicious cycle of leukocyte and endothelial-mediated oxidant stress that results in vascular dysfunction. Am J Physiol Heart Circ Physiol. 2020;319:H51-65.
44. Hu N, Yang L, Dong M, Ren J, Zhang Y. Deficiency in adiponectin exaggerates cigarette smoking exposure-induced cardiac contractile dysfunction: role of autophagy. Pharmacol Res. 2015;100:175-89.
45. Qasim H, Alarabi AB, Alzoubi KH, Karim ZA, Alshbool FZ, Khasawneh FT. The effects of hookah/waterpipe smoking on general health and the cardiovascular system. Environ Health Prev Med. 2019;24:58.
46. Bhatnagar A, Maziak W, Eissenberg T, et al. Water pipe (hookah) smoking and cardiovascular disease risk: a scientific statement from the American Heart Association. Circulation. 2019;139:e917-36.
47. Alomari MA, Al-Sheyab NA, Khabour OF, Alzoubi KH. Serum VEGF level is different in adolescents smoking waterpipe versus cigarettes: the irbid TRY. Biomolecules. 2018;8:102.
48. Alomari MA, Khabour OF, Alzoubi KH, Shqair DM, Stoner L. Acute vascular effects of waterpipe smoking: importance of physical activity and fitness status. Atherosclerosis. 2015;240:472-6.
49. El-Mahdy MA, Ewees MG, Eid MS, Mahgoup EM, Khaleel SA, Zweier JL. Electronic cigarette exposure causes vascular endothelial dysfunction due to NADPH oxidase activation and eNOS uncoupling. Am J Physiol Heart Circ Physiol. 2022;322:H549-67.
50. Kuntic M, Oelze M, Steven S, et al. Short-term e-cigarette vapour exposure causes vascular oxidative stress and dysfunction: evidence for a close connection to brain damage and a key role of the phagocytic NADPH oxidase (NOX-2). Eur Heart J. 2020;41:2472-83.
51. Derella CC, Tingen MS, Blanks A, et al. Smoking cessation reduces systemic inflammation and circulating endothelin-1. Sci Rep. 2021;11:24122.
52. Wang M, Li Y, Lai M, et al. Alcohol consumption and epigenetic age acceleration across human adulthood. Aging. 2023;15:10938-71.
53. Fernández-Solà J. The effects of ethanol on the heart: alcoholic cardiomyopathy. Nutrients. 2020;12:572.
54. Del Giorno R, Maddalena A, Bassetti S, Gabutti L. Association between alcohol intake and arterial stiffness in healthy adults: a systematic review. Nutrients. 2022;14:1207.
55. Mukhopadhyay P, Yokus B, Paes-Leme B, et al. Chronic alcohol consumption accelerates cardiovascular aging and decreases cardiovascular reserve capacity. Geroscience. 2025;47:5881-901.
56. Piacenza L, Zeida A, Trujillo M, Radi R. The superoxide radical switch in the biology of nitric oxide and peroxynitrite. Physiol Rev. 2022;102:1881-906.
57. Ungvari Z, Tarantini S, Donato AJ, Galvan V, Csiszar A. Mechanisms of vascular aging. Circ Res. 2018;123:849-67.
58. Al Attar AA, Fahed GI, Hoballah MM, et al. Mechanisms underlying the effects of caloric restriction on hypertension. Biochem Pharmacol. 2022;200:115035.
59. Csiszar A, Gautam T, Sosnowska D, et al. Caloric restriction confers persistent anti-oxidative, pro-angiogenic, and anti-inflammatory effects and promotes anti-aging miRNA expression profile in cerebromicrovascular endothelial cells of aged rats. Am J Physiol Heart Circ Physiol. 2014;307:H292-306.
60. Csiszar A, Sosnowska D, Tucsek Z, et al. Circulating factors induced by caloric restriction in the nonhuman primate Macaca mulatta activate angiogenic processes in endothelial cells. J Gerontol A Biol Sci Med Sci. 2013;68:235-49.
61. Trichopoulou A, Martínez-González MA, Tong TY, et al. Definitions and potential health benefits of the Mediterranean diet: views from experts around the world. BMC Med. 2014;12:112.
62. GómezSánchez M, Gómez Sánchez L, Patino-Alonso MC, et al. Adherence to the mediterranean diet in Spanish population and its relationship with early vascular aging according to sex and age: EVA study. Nutrients. 2020;12:1205.
63. Li J, Capuano AW, Agarwal P, et al. The MIND diet, brain transcriptomic alterations, and dementia. Alzheimers Dement. 2024;20:5996-6007.
64. Akbar Z, Fituri S, Ouagueni A, et al. Associations of the MIND diet with cardiometabolic diseases and their risk factors: a systematic review. Diabetes Metab Syndr Obes. 2023;16:3353-71.
65. Ungvari Z, Kunutsor SK. Coffee consumption and cardiometabolic health: a comprehensive review of the evidence. Geroscience. 2024;46:6473-510.
66. Scioli MG, Storti G, D'Amico F, et al. Oxidative stress and new pathogenetic mechanisms in endothelial dysfunction: potential diagnostic biomarkers and therapeutic targets. J Clin Med. 2020;9:1995.
67. N Kolodkin A, Sharma RP, Colangelo AM, et al. ROS networks: designs, aging, Parkinson’s disease and precision therapies. NPJ Syst Biol Appl. 2020;6:34.
68. Wang Y, Li Y, He C, Gou B, Song M. Mitochondrial regulation of cardiac aging. Biochim Biophys Acta Mol Basis Dis. 2019;1865:1853-64.
69. Schmidlin CJ, Dodson MB, Madhavan L, Zhang DD. Redox regulation by NRF2 in aging and disease. Free Radical Biol Med. 2019;134:702-7.
70. Dato S, Crocco P, D'Aquila P, et al. Exploring the role of genetic variability and lifestyle in oxidative stress response for healthy aging and longevity. Int J Mol Sci. 2013;14:16443-72.
71. Frasca D, Blomberg BB. Inflammaging decreases adaptive and innate immune responses in mice and humans. Biogerontology. 2016;17:7-19.
72. McKechnie DGJ, Papacosta AO, Lennon LT, Ramsay SE, Whincup PH, Wannamethee SG. Associations between inflammation, cardiovascular biomarkers and incident frailty: the British Regional Heart Study. Age Ageing. 2021;50:1979-87.
73. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15:505-22.
74. Youm YH, Grant RW, McCabe LR, et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 2013;18:519-32.
76. Ramji DP, Davies TS. Cytokines in atherosclerosis: key players in all stages of disease and promising therapeutic targets. Cytokine Growth Factor Rev. 2015;26:673-85.
77. Feng YS, Tan ZX, Wu LY, Dong F, Zhang F. The involvement of NLRP3 inflammasome in the treatment of Alzheimer’s disease. Ageing Res Rev. 2020;64:101192.
78. Rea IM, Gibson DS, McGilligan V, McNerlan SE, Alexander HD, Ross OA. Age and age-related diseases: role of inflammation triggers and cytokines. Front Immunol. 2018;9:586.
79. Evans AK, Saw NL, Woods CE, et al. Impact of high-fat diet on cognitive behavior and central and systemic inflammation with aging and sex differences in mice. Brain Behav Immun. 2024;118:334-54.
80. Dioum EHM, Schneider KL, Vigerust DJ, et al. Oats lower age-related systemic chronic inflammation (iAge) in adults at risk for cardiovascular disease. Nutrients. 2022;14:4471.
81. Estruch R, Ros E, Martínez-González MA. Mediterranean diet for primary prevention of cardiovascular disease. N Engl J Med. 2013;369:676-7.
82. Hamer M, Lavoie KL, Bacon SL. Taking up physical activity in later life and healthy ageing: the English longitudinal study of ageing. Br J Sports Med. 2014;48:239-43.
83. Mahmud F, Sarker DB, Jocelyn JA, Sang QA. Molecular and cellular effects of microplastics and nanoplastics: focus on inflammation and senescence. Cells. 2024;13:1788.
84. Li H, Xia N. The role of oxidative stress in cardiovascular disease caused by social isolation and loneliness. Redox Biol. 2020;37:101585.
85. Xia N, Li H. Loneliness, social isolation, and cardiovascular health. Antioxid Redox Signaling. 2018;28:837-51.
86. Bravo-San Pedro JM, Kroemer G, Galluzzi L. Autophagy and mitophagy in cardiovascular disease. Circ Res. 2017;120:1812-24.
87. Nguyen TN, Padman BS, Lazarou M. Deciphering the molecular signals of PINK1/Parkin mitophagy. Trends Cell Biol. 2016;26:733-44.
88. Marinković M, Šprung M, Novak I. Dimerization of mitophagy receptor BNIP3L/NIX is essential for recruitment of autophagic machinery. Autophagy. 2021;17:1232-43.
89. Zhang W, Siraj S, Zhang R, Chen Q. Mitophagy receptor FUNDC1 regulates mitochondrial homeostasis and protects the heart from I/R injury. Autophagy. 2017;13:1080-1.
91. Wu N, Zheng W, Zhou Y, et al. Autophagy in aging-related diseases and cancer: principles, regulatory mechanisms and therapeutic potential. Ageing Res Rev. 2024;100:102428.
92. Montégut L, Joseph A, Chen H, et al. DBI/ACBP is a targetable autophagy checkpoint involved in aging and cardiovascular disease. Autophagy. 2023;19:2166-9.
93. Sciarretta S, Yee D, Nagarajan N, et al. Trehalose-induced activation of autophagy improves cardiac remodeling after myocardial infarction. J Am Coll Cardiol. 2018;71:1999-2010.
94. Eisenberg T, Abdellatif M, Schroeder S, et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat Med. 2016;22:1428-38.
95. D'Amico A, Fossati C, Pigozzi F, et al. Natural activators of autophagy reduce oxidative stress and muscle injury biomarkers in endurance athletes: a pilot study. Nutrients. 2023;15-459.
96. Kiechl S, Pechlaner R, Willeit P, et al. Higher spermidine intake is linked to lower mortality: a prospective population-based study. Am J Clin Nutr. 2018;108:371-80.
97. Hofer SJ, Simon AK, Bergmann M, Eisenberg T, Kroemer G, Madeo F. Mechanisms of spermidine-induced autophagy and geroprotection. Nat Aging. 2022;2:1112-29.
98. Hofer SJ, Daskalaki I, Bergmann M, et al. Spermidine is essential for fasting-mediated autophagy and longevity. Nat Cell Biol. 2024;26:1571-84.
99. Ramos ELP, Sousa Neto IV, Pinto AP, et al. Mechanisms underlying the interplay between autophagy and the inflammasome in age-related diseases: implications for exercise immunology. Ageing Res Rev. 2025;110:102821.
100. Hassink RJ, Pasumarthi KB, Nakajima H, et al. Cardiomyocyte cell cycle activation improves cardiac function after myocardial infarction. Cardiovasc Res. 2008;78:18-25.
101. Del Re DP, Amgalan D, Linkermann A, Liu Q, Kitsis RN. Fundamental mechanisms of regulated cell death and implications for heart disease. Physiol Rev. 2019;99:1765-817.
102. Marian AJ. Causes and consequences of DNA double-stranded breaks in cardiovascular disease. Mol Cell Biochem. 2025;480:2043-64.
103. Lozhkin A, Vendrov AE, Ramos-Mondragón R, et al. Mitochondrial oxidative stress contributes to diastolic dysfunction through impaired mitochondrial dynamics. Redox Biol. 2022;57:102474.
104. Mir SM, Samavarchi Tehrani S, Goodarzi G, et al. Shelterin complex at telomeres: implications in ageing. Clin Interv Aging. 2020;15:827-39.
105. Cai K, Jiang H, Zou Y, et al. Programmed death of cardiomyocytes in cardiovascular disease and new therapeutic approaches. Pharmacol Res. 2024;206:107281.
106. Kamo T, Akazawa H, Komuro I. Cardiac nonmyocytes in the hub of cardiac hypertrophy. Circ Res. 2015;117:89-98.
107. Saucerman JJ, Tan PM, Buchholz KS, McCulloch AD, Omens JH. Mechanical regulation of gene expression in cardiac myocytes and fibroblasts. Nat Rev Cardiol. 2019;16:361-78.
108. Adao DMT, Ching C, Fish JE, Simmons CA, Billia F. Endothelial cell-cardiomyocyte cross-talk: understanding bidirectional paracrine signaling in cardiovascular homeostasis and disease. Clin Sci. 2024;138:1395-419.
109. Wang Q, Ismahil MA, Zhu Y, et al. CD206+IL-4Rα+ macrophages are drivers of adverse cardiac remodeling in ischemic cardiomyopathy. Circulation. 2025;152:257-73.
110. Ozcebe SG, Bahcecioglu G, Yue XS, Zorlutuna P. Effect of cellular and ECM aging on human iPSC-derived cardiomyocyte performance, maturity and senescence. Biomaterials. 2021;268:120554.
111. Ye Y, Yang K, Liu H, et al. SIRT2 counteracts primate cardiac aging via deacetylation of STAT3 that silences CDKN2B. Nat Aging. 2023;3:1269-87.
112. Okabe K, Yaku K, Uchida Y, et al. Oral administration of nicotinamide mononucleotide is safe and efficiently increases blood nicotinamide adenine dinucleotide levels in healthy subjects. Front Nutr. 2022;9:868640.
113. Nie H, Zhang Y, Yu H, Xiao H, Li T, Yang Q. Oral delivery of carrier-free dual-drug nanocrystal self-assembled microspheres improved NAD+ bioavailability and attenuated cardiac ischemia/reperfusion injury in mice. Drug Delivery. 2021;28:433-44.
114. Hastings MH, Castro C, Freeman R, et al. Intrinsic and extrinsic contributors to the cardiac benefits of exercise. JACC Basic Transl Sci. 2024;9:535-52.
115. Monk BA, George SJ. The effect of ageing on vascular smooth muscle cell behaviour-a mini-review. Gerontology. 2015;61:416-26.
116. Akinmolayan A, Papacosta AO, Lennon LT, et al. Carotid intima-media thickness, carotid distensibility, and incident heart failure in older men: the British regional heart study. J Am Heart Assoc. 2025;14:e037167.
117. Toda N. Age-related changes in endothelial function and blood flow regulation. Pharmacol Ther. 2012;133:159-76.
118. Burnier M, Damianaki A. Hypertension as cardiovascular risk factor in chronic kidney disease. Circ Res. 2023;132:1050-63.
120. Hwang HJ, Kim N, Herman AB, Gorospe M, Lee JS. Factors and pathways modulating endothelial cell senescence in vascular aging. Int J Mol Sci. 2022;23:10135.
121. Jia G, Aroor AR, Jia C, Sowers JR. Endothelial cell senescence in aging-related vascular dysfunction. Biochim Biophys Acta Mol Basis Dis. 2019;1865:1802-9.
122. Donato AJ, Machin DR, Lesniewski LA. Mechanisms of dysfunction in the aging vasculature and role in age-related disease. Circ Res. 2018;123:825-48.
123. Nicoll R, Henein MY. The predictive value of arterial and valvular calcification for mortality and cardiovascular events. Int J Cardiol Heart Vessel. 2014;3:1-5.
124. Ceneri N, Zhao L, Young BD, et al. Rac2 modulates atherosclerotic calcification by regulating macrophage interleukin-1β production. Arterioscler, Thromb, Vasc Biol. 2017;37:328-40.
125. Van Campenhout A, Golledge J. Osteoprotegerin, vascular calcification and atherosclerosis. Atherosclerosis. 2009;204:321-9.
126. De Maré A, Opdebeeck B, Neven E, D'Haese PC, Verhulst A. Sclerostin protects against vascular calcification development in mice. J Bone Miner Res. 2022;37:687-99.
127. Climie RE, Alastruey J, Mayer CC, et al. Vascular ageing: moving from bench towards bedside. Eur J Prev Cardiol. 2023;30:1101-17.
128. Olive M, Harten I, Mitchell R, et al. Cardiovascular pathology in Hutchinson-Gilford progeria: correlation with the vascular pathology of aging. Arterioscler, Thromb, Vasc Biol. 2010;30:2301-9.
129. Gomez-Sanchez M, Gomez-Sanchez L, Patino-Alonso MC, et al.; EVA Investigators. Vascular aging and its relationship with lifestyles and other risk factors in the general Spanish population: early vascular ageing study. J Hypertens. 2020;38:1110-22.
130. Sugawara J, Tomoto T, Noda N, et al. Effects of endothelin-related gene polymorphisms and aerobic exercise habit on age-related arterial stiffening: a 10-yr longitudinal study. J Appl Physiol. 2018;124:312-20.
131. Grigorova YN, Wei W, Petrashevskaya N, et al. Dietary sodium restriction reduces arterial stiffness, vascular TGF-β-dependent fibrosis and marinobufagenin in young normotensive rats. Int J Mol Sci. 2018;19:3168.
132. Cavalcante JL, Lima JA, Redheuil A, Al-Mallah MH. Aortic stiffness: current understanding and future directions. J Am Coll Cardiol. 2011;57:1511-22.
133. Herzog MJ, Müller P, Lechner K, et al. Arterial stiffness and vascular aging: mechanisms, prevention, and therapy. Signal Transduct Target Ther. 2025;10:282.
134. Hinton RB, Yutzey KE. Heart valve structure and function in development and disease. Annu Rev Physiol. 2011;73:29-46.
135. Massera D, Kizer JR, Dweck MR. Mechanisms of mitral annular calcification. Trends Cardiovasc Med. 2020;30:289-95.
136. Sengupta SP, Mohan JC. Calcific mitral stenosis: echoes of aging. J Am Coll Cardiol. 2020;75:3058-60.
137. Zoghbi WA, Levine RA, Flachskampf F, et al. Atrial functional mitral regurgitation: a JACC: cardiovascular imaging expert panel viewpoint. JACC Cardiovasc Imaging. 2022;15:1870-82.
138. Pio SM, Medvedofsky D, Delgado V, et al.; COAPT Trial Investigators. Left atrial improvement in patients with secondary mitral regurgitation and heart failure: the COAPT trial. JACC Cardiovasc Imaging. 2024;17:1015-27.
139. Forrester SJ, Booz GW, Sigmund CD, et al. Angiotensin II signal transduction: an update on mechanisms of physiology and pathophysiology. Physiol Rev. 2018;98:1627-738.
140. Ren LL, Miao H, Wang YN, Liu F, Li P, Zhao YY. TGF-β as a master regulator of aging-associated tissue fibrosis. Aging Dis. 2023;14:1633-50.
141. Smyrnias I, Gray SP, Okonko DO, et al. Cardioprotective effect of the mitochondrial unfolded protein response during chronic pressure overload. J Am Coll Cardiol. 2019;73:1795-806.
142. Rogers MA, Maldonado N, Hutcheson JD, et al. Dynamin-related protein 1 inhibition attenuates cardiovascular calcification in the presence of oxidative stress. Circ Res. 2017;121:220-33.
143. Kjeldsen EW, Thomassen JQ, Rasmussen KL, Nordestgaard BG, Tybjærg-Hansen A, Frikke-Schmidt R. Cardiovascular risk factors and aortic valve stenosis: towards 10-year absolute risk charts for primary prevention. Eur J Prev Cardiol. 2025;32:1684-93.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].