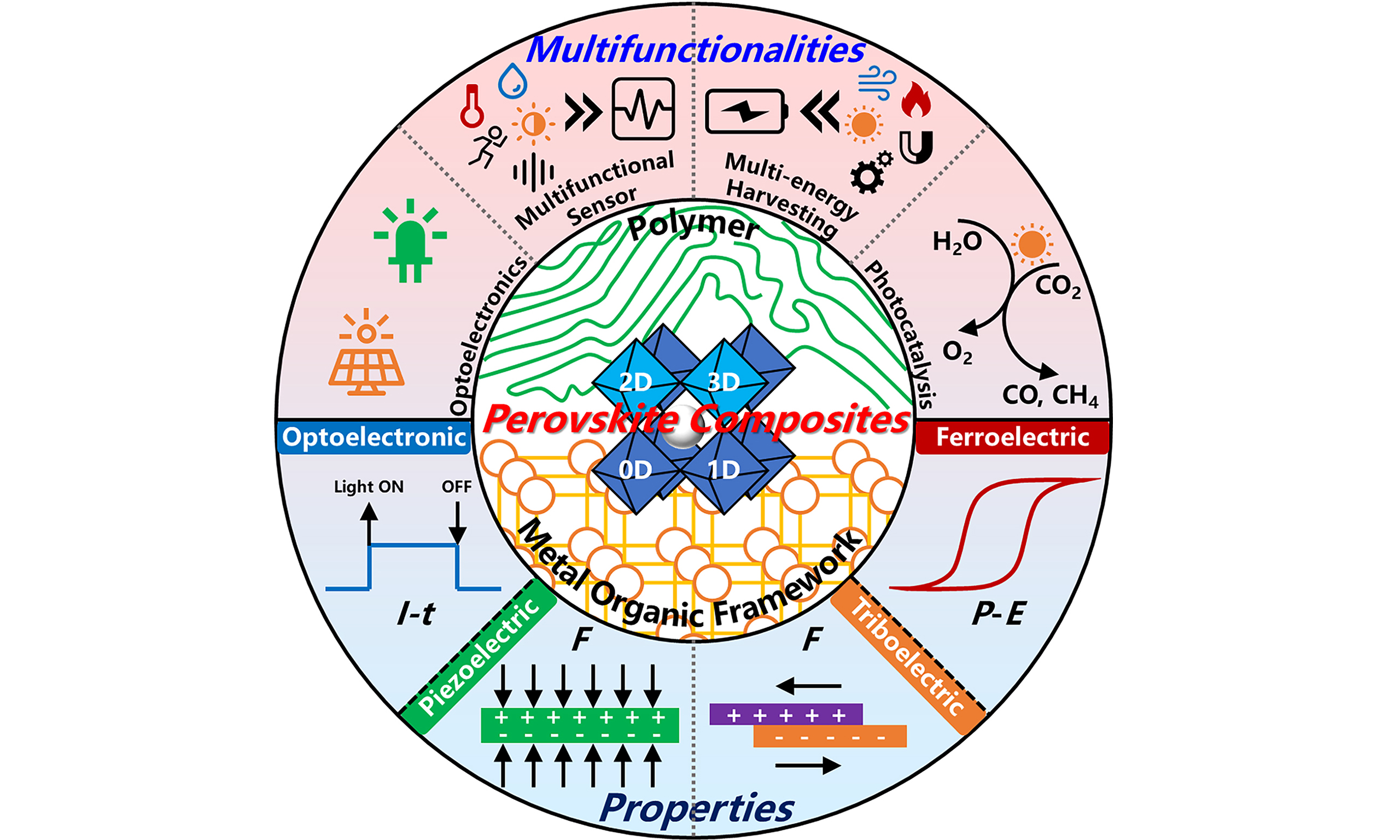
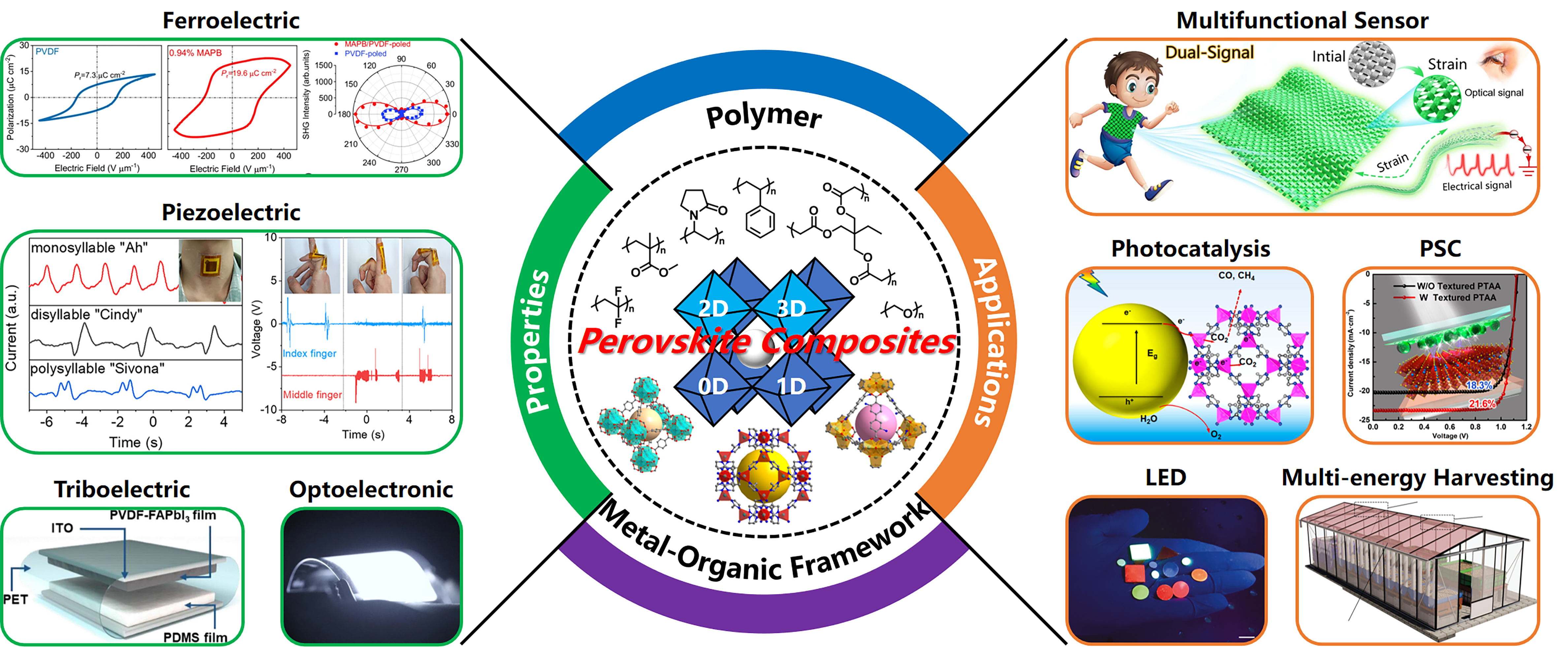
Compositing halide perovskites for multifunctionalities beyond optoelectronics
Abstract
Metal halide perovskites (MHPs) have emerged as highly promising optoelectronic materials due to their high absorption coefficients, tunable bandgaps, long carrier diffusion lengths, and low exciton binding energies. In addition, their unique polar structures enable electric polarization-related properties, such as ferroelectricity, opening new avenues for integrating light-responsive and ferroelectric functionalities in next-generation electronic devices. Despite the exciting progress achieved in MHPs-based optoelectronic devices, compositing MHPs with polymers or metal-organic frameworks not only improves their mechanical flexibility, stability, and optoelectronic performance, but also extends their functions to multi-energy harvesting and multifunctional sensing. Herein, we highlight the structural diversity, tunability, and rich physics of MHPs, and focus on strategies for structural design, optimization of optical and ferroelectric properties, as well as functional implementation across diverse applications of MHPs-based composites in sensors, energy harvesters, solar cells, and photocatalysts. Finally, we conclude with perspectives on the prospects of MHPs-based composites, emphasizing their significant potential to advance the development of highly stable, efficient, low-cost, and multifunctional flexible optoelectronic devices.
Keywords
INTRODUCTION
Metal halide perovskites (MHPs) have received enormous attention in the past decade due to their remarkable optoelectronic properties, including high absorption coefficients, tunable bandgaps, long carrier diffusion lengths, and low exciton binding energies, making them ideal candidates for a wide range of applications, particularly in solar cells, light-emitting diodes (LEDs), and photodetectors[1-4]. The rapid rise in the efficiency of perovskite solar cells (PSCs), surpassing 27% in just over a decade, highlights their potential to revolutionize the photovoltaic landscape[5-7]. Beyond photovoltaics, MHPs are also gaining attention in piezoelectric and ferroelectric applications due to their unique intrinsic polar properties, offering opportunities to exploit multiple energy conversion mechanisms, such as piezoelectricity, pyroelectricity, electrostriction, and electrical energy storage[8]. While traditional perovskite oxides such as BaTiO3 have long been used in ferroelectric applications, MHPs - with their natural advantages of structural softness and lightweight - are opening a new era in ferroelectric research[9,10].
Despite their impressive properties and promising future, perovskite materials still face various significant challenges. One of the primary drawbacks is their inherent instability, particularly when exposed to moisture, oxygen, or elevated temperatures, leading to rapid degradation of material performance[11-14]. The susceptibility to environmental factors has raised concerns about the long-term durability of perovskite-based devices. One promising strategy is to develop MHP composites by combining perovskites with other materials, such as polymers[15], metal-organic frameworks (MOFs)[16], or metal oxides. In this way, the advantages of both components can be retained, and new effects may emerge. The composite approach can generally help improve environmental stability, enhance mechanical properties, and optimize electronic or ionic transport pathways of the MHPs[17-19]. For instance, encapsulating perovskites within protective matrices or combining them with more stable materials can significantly improve their environmental resistance[20-22]. Additionally, such composite systems may enable better control over the crystallization process of perovskites, leading to improved phase purity and defect passivation ability[23,24].
On the other hand, incorporating perovskites into composite materials offers new opportunities for additional functionalities. For example, although MHPs have the potential to exhibit ferroelectricity, their narrow bandgaps impede effective screening of polarization charges. Therefore, by compositing with polymers, the resulting nanocomposites can exhibit unprecedentedly large electromechanical coupling, making them suitable for next-generation sensors, actuators, and energy-harvesting devices[25-27]. Meanwhile, the interplay between perovskites and other phases within the composites may result in enhanced light-matter interactions, thereby benefiting optoelectronic applications such as more efficient solar cells, brighter LEDs, and faster photodetectors.
Herein, we aim to provide a brief review of MHPs from the perspective of MHPs-based composites, in light of their great potential for multifunctional flexible electronics beyond solar cells. We begin with an introduction to the structures of various MHPs-based composites and then elaborate in detail on the advantages of these composites. Next, we focus on discussing the physical properties of the above-mentioned composites, including ferroelectricity, piezoelectricity, triboelectricity, and photoelectricity, and their applications in optoelectronics, multifunctional sensors, multifunctional generators, and photocatalysis are illustrated. We finally conclude with prospects of MHPs-based composites as ferroelectric semiconductors exhibiting both ferroelectricity and optoelectronic properties, with great prospects in light-controlled electronics. The content of this review is schematically illustrated in Figure 1.
STRUCTURES OF MHPs-BASED COMPOSITES
Diverse structures of metal halide perovskites
The history of perovskites begins with the discovery of the prototypical calcium titanate (CaTiO3) mineral in 1839, and has evolved from a naturally occurring mineral to a synthetically designed material with a ABX3-type structure, where A and B are cations of different atomic radii that occupy the corner and center of the unit cell, respectively, and X is an anion located at the face of the unit cell coordinated with B to form BX6 octahedra[36]. With regard to MHPs, "A" represents an organic cation or an alkaline earth metal [such as CH(NH2)2+ (FA+), CH3NH3+ (MA+), or Cs+]. “B” is typically a transition metal such as Pb2+ or Sn2+, and X is the halide ion (Cl-, Br-, or I-). The stability of three-dimensional (3D) perovskite crystal structure could be evaluated by the tolerance factor t[37]:
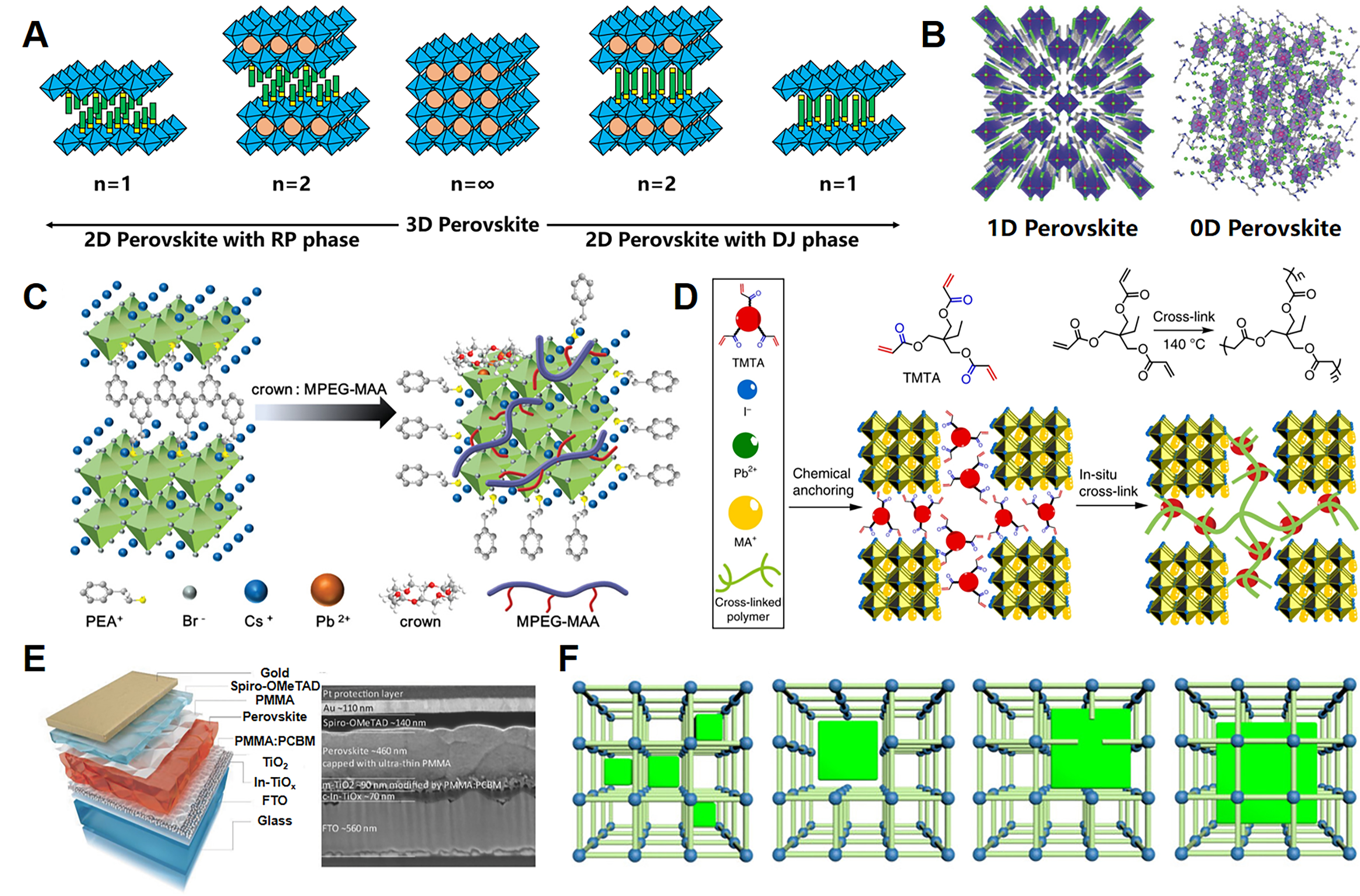
where RA, RB and RX are the ionic radii of the corresponding site elements. Stable perovskite structures typically exhibit tolerance factors in the range of 0.76-1.13. When t falls between 0.87 and 1, the perovskite adopts an ideal cubic structure characterized by high symmetry and optimal lattice packing, and it transforms into a less symmetric tetragonal or orthorhombic phase when t lies between 0.76 and 0.87. When t is below 0.76 or above 1, the structure deviates significantly from the traditional 3D perovskite and may transform into two-dimensional (2D), one-dimensional (1D), or zero-dimensional (0D) structures, as shown in Figure 2A and B[38,39].
Figure 2. (A) Schematic diagram of crystal structures transformation from a 3D perovskite to 2D perovskites with RP and DJ phase, respectively. (B) Schematic illustrations of 1D and 0D perovskite structures[39]. (C) Schematic illustration of crystal structure change and defect passivation by crown and MPEG-MAA[48]. (D) Schematic diagram of in situ cross-linked organic/perovskite films[49]. (E) Schematic illustration of the device structure and corresponding SEM cross-sectional image[50]. (F) Schematic diagrams of ultrasmall perovskite quantum dots within micropore structure of MOFs, perovskite nanocrystals within mesopores or flexible pores of MOFs, perovskite nanocrystals throughout the pores of MOFs, and perovskite nanocrystals coated with MOF shells[16].
Compared with 3D perovskites, 2D MHPs, composed of alternating organic and inorganic layers, offer tunable optoelectronic properties, as the thickness of the inorganic layers and the nature of the organic interlayers can be adjusted to modify the bandgap, photoluminescence (PL), and charge-transport characteristics of the materials[40]. Free from the strict constraints of the tolerance factor, 2D perovskites offer greater flexibility in composition design, resulting in diverse structures. Moreover, 2D MHPs are more stable than 3D perovskites due to stronger van der Waals interactions between the capping organic molecules and the [PbI6] units, as well as the higher hydrophobicity of the bulkier organic components with longer alkyl chains compared to smaller ions such as MA+[41]. As the dimension of MHPs reduces to 1D and 0D, more exotic properties have emerged due to their highly confined structures. In 1D perovskites, the BX6 octahedra form chains or ribbons[42], while in 0D perovskites, the octahedra are isolated, with no connectivity between them[43]. These lower-dimensional structures exhibit stronger quantum confinement effects, leading to unique optical and electronic behaviors. Similar to 2D perovskites, 1D or 0D perovskites have stronger environmental stability in PSCs applications, with the ability to alleviate internal stress in 3D perovskite layers and passivate interface defects[44-46]. However, due to the absence of an inorganic octahedral dimension, 0D-3D or 1D-3D PSCs may experience a significant reduction in carrier mobility and carrier separation capability, encountering a more pronounced challenge concerning charge-transport obstruction.
Structures of MHPs-based composites
MHPs are exceptional optoelectronic materials themselves; when compositing with another component, additional functions shall be endowed with, and more importantly, their intrinsic environmental instability could also be overcome. Herein, two major types of MHPs-based composites are discussed: MHPs/polymer and MHPs/MOF composites.
MHPs/polymer composites
For MHPs/polymer composite structure, three types are typically employed to ensure optimal functional integration: core-shell structures, mixtures, and layered configurations. Specifically, in the core-shell configuration, MHP crystals are encapsulated by a protective polymer shell, creating a robust barrier that effectively isolates the perovskite core from adverse environmental factors, such as moisture and oxygen, thereby significantly boosting its stability[21,25,47]. For instance, Liu et al. adopted a synergistic dual-additive strategy, using 18-crown-6 and poly(ethylene glycol) methyl ether acrylate (MPEG-MAA) as additives, to form a quasi-core/shell structure as shown in Figure 2C[48]. The dual additives containing C-O-C bonds not only effectively reduce perovskite defects but also disrupt the self-aggregation of organic ligands, enhancing the electroluminescence performance of the perovskite light-emitting diodes (PeLEDs)[48]. Secondly, the mixture involves polymers acting as modifiers that are fully dispersed within the MHP matrix to form a homogeneous composite. As shown in Figure 2D, Li et al. employed an in situ cross-linking strategy to facilitate the polymerization of the small organic molecule additive trimethylolpropane triacrylate (TMTA) directly within the 3D MHP films (MAPbI3), resulting in the formation of a continuous polymeric network that was chemically anchored to the perovskite grain boundaries[49]. Lastly, the layered configuration comprises alternating layers of MHPs and polymer, which optimizes charge separation and transport across the interfaces, minimizes charge recombination stemming from defects within the MHPs, and ultimately enhances the overall performance of the device. As presented in Figure 2E, the double-sided passivated PSCs by incorporating ultrathin polymethyl methacrylate (PMMA) films at the interfaces between the MHPs and both the electron transport layer (ETL) and hole transport layer (HTL), effectively minimized non-radiative recombination, thereby an open-circuit voltage (VOC) of approximately 1.22 V and a power conversion efficiency (PCE) of 20.8% have been obtained[50].
MHPs/MOF composites
MOFs, composed of metal ions or clusters connected by organic ligands, possess a unique framework with tunable structure, pore size and large surface area. The abundance of pore structures within MOF materials facilitates the confined growth of perovskite nanoparticles, enabling effective size control of perovskite nanocrystals through MOF pore dimensions[32,51,52]. Furthermore, the flexibility of the organic framework allows the MOF matrix to accommodate larger perovskite particles without incurring noticeable macroscopic cracks[53]. Similar to MHPs/polymer composite structures, MHPs/MOF composites can be classified into four types according to the size and position of the MHP grains within the MOF framework, as shown in Figure 2F: (1) ultra-small perovskite quantum dots (QDs) confined within the micropores of MOFs; (2) perovskite nanocrystals nestled within mesopores or flexible pores; (3) perovskite nanocrystals distributed throughout the pores of MOFs; and (4) perovskite nanocrystals encapsulated by MOF shells[16]. The variations in location, crystal size, and combination of perovskite particles within MOFs greatly influence the electrical and optical properties of MHPs/MOF composites, such as bandgap, emission wavelength, and photoluminescence quantum yield (PLQY)[16,54,55].
ELECTRICAL PROPERTIES OF MHPs-BASED COMPOSITES
By combining MHPs with polymers or MOFs, the toughness, ductility, and processability of perovskites can be enhanced, allowing for more uniform distribution of internal stress, reducing crack formation, and extending the lifespan of perovskite materials[26,56,57]. Meanwhile, the polymers or MOFs can effectively passivate the surface defects of perovskite nanocrystals, suppress ion migration, and reduce structural degradation caused by direct exposure to the external environment, thereby lowering defect density and enhancing the stability of perovskite nanocrystals[15]. More importantly, the selected secondary component can further improve charge-transport properties without compromising the light-absorption capabilities of MHPs, achieving a synergistic enhancement effect greater than the sum of the individual components[58,59].
MHPs-based composites with boosted photoelectric performances
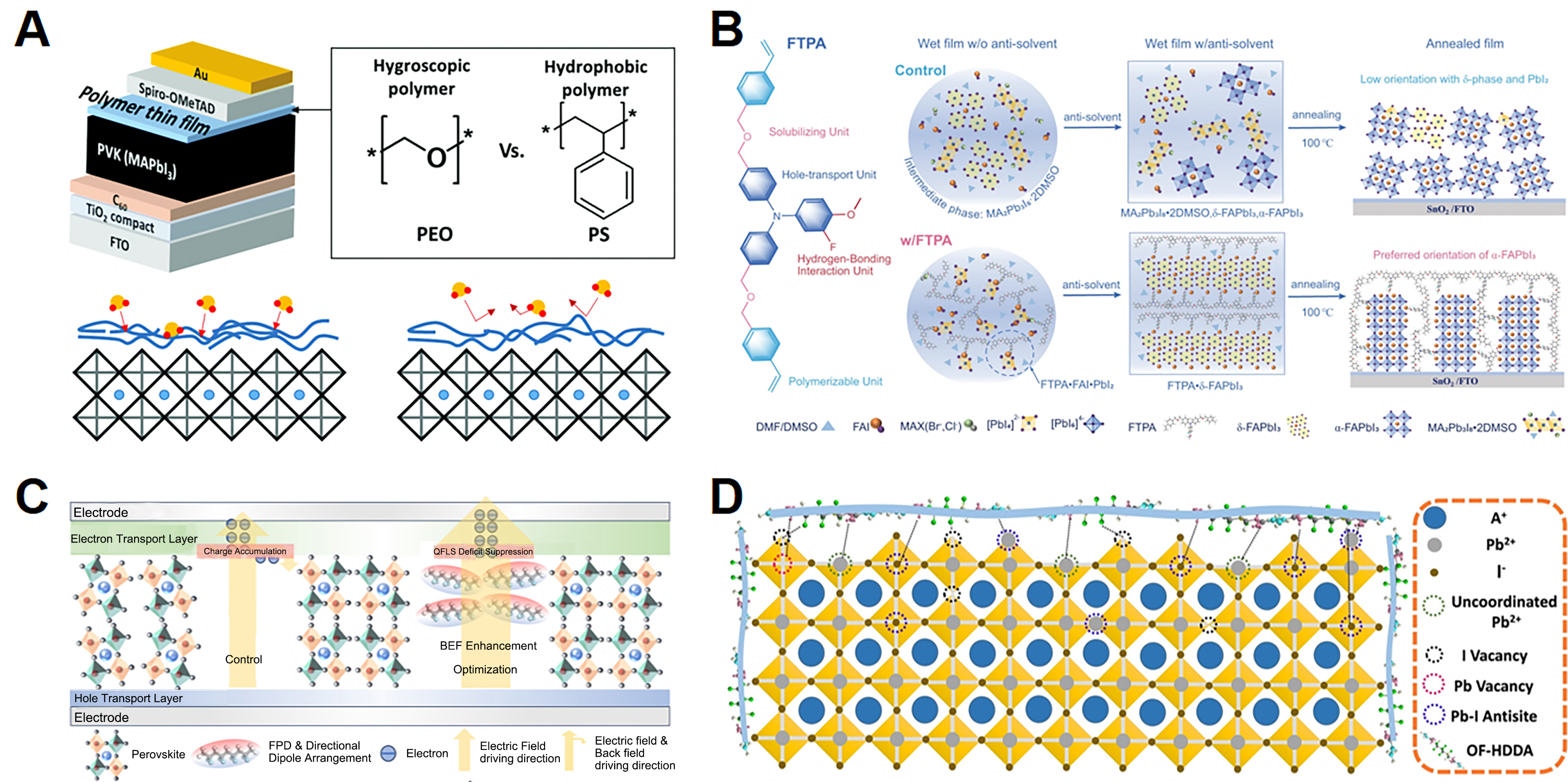
As shown in Figure 3A, studies from Jeong et al.[60] and Kim et al.[61] have revealed that hygroscopic poly(ethylene oxide) (PEO) polymers with oxygen-containing functional groups can not only efficiently mitigate the hydration process of the perovskite layer by absorbing moisture before it reaches the MHPs, but also passivate undercoordinated defect sites through the interaction between PEO and MHPs. These two effects together improve the operational device stability under exposure to high humidity, light, and polarization in two beneficial ways. Similarly, the porous and chemically tunable nature of MOFs provides an ideal platform for encapsulating MHPs, mitigating issues related to ion migration, phase instability and moisture sensitivity, protecting them from environmental degradation, while preserving or even enhancing their optoelectronic performance[16].
Figure 3. (A) Structural schematic diagram of perovskite planar heterojunction solar cells with polymer thin films as a passive layer[61]. (B) Schematic diagram of the possible phase evolution of FA-based mixed anion perovskites [FA0.95MA0.05Pb(I0.95Br0.05)3] during the film formation with or without FTPA (3-fluoro-4-methoxy-4’,4”c-bis((4-vinyl benzyl ether) methyl) triphenylamine)[64]. (C) Schematic diagram of suppression mechanism on quasi-Fermi level splitting deficiency in PSCs[68]. (D) Schematic diagram of perovskite defects distribution and the mechanism of defects passivation by POF-HDDA (octafluoro-1,6-hexanediol diacrylate polymer)[74].
In addition to the effects of reducing defect density and enhancing stability, the free volumes in polymers also provide a natural confined environment for the crystallization of perovskites, enabling the control of the nucleation and crystallization processes, avoiding excessive growth of perovskite crystals and enhancing the grain quality[56,62,63]. Li et al. designed a multifaceted fluorinated additive 3-fluoro-4-methoxy-4′,4″c-bis((4-vinyl benzyl ether) methyl) triphenylamine (FTPA), which undergoes in situ polymerization during perovskite film formation through a hydrogen-bonding network[64]. This process effectively suppresses the formation of undesirable intermediate phases of MHPs and significantly promotes the oriented growth of α-formamidinium lead iodide (α-FAPbI3). Therefore, with the simultaneous improvement in balanced charge transport, low defect density and gradient energy level alignment, planar PSCs incorporating FTPA exhibited a PCE of up to 24.1% [Figure 3B][64].
Doping ferroelectric polymers into the solar cell layer or inserting them between the cell and charge-transport layers has emerged as a straightforward and effective strategy to boost PSC performance. This is attributed to the inherent polarization characteristics of ferroelectric polymers, which can strengthen the built-in electric field, facilitate charge separation, and suppress non-radiative recombination, thereby improving device efficiency, reducing current hysteresis, and extending lifespan simultaneously[65-67].
Additionally, since toxic lead leakage caused by ion migration is another important factor limiting the application of MHPs[69,70], previous studies have demonstrated that polymers can not only stabilize the perovskite structure but also act as a natural barrier to inhibit lead leakage, providing long-term environmental protection[71-73]. Zhang et al. utilized an in situ thermal crosslinking strategy to obtain an octafluoro-1,6-hexanediol diacrylate polymer (POF-HDDA) that bound uncoordinated Pb2+ and inhibited up to 85% of lead leakage, as shown in Figure 3D[74]. Through coordination and hydrogen-bond interactions, the polymer passivates various defects related to Pb and I, regulates the crystallization of MHPs with reduced trap density, relieves lattice strain, and promotes carrier transport and extraction. The optimal efficiencies of polymer-treated PSCs can reach 24.76%[74].
Ferroelectricity
Ferroelectricity is a characteristic of non-centrosymmetric crystals, exhibiting spontaneous polarization that can be reversibly switched by an external electric field. Perovskite oxides, such as BaTiO3 and Pb(TixZr1-x)O3, as major ferroelectric materials, have been extensively studied and widely applied as transducers, memories, sensors, and actuators due to their large polarization and high Curie temperature (Tc)[75-78]. In contrast, MHP ferroelectrics, with the natural advantages of structural softness, light weight, diverse chemical composition, and structural tunability, are receiving increasing interest and are being actively investigated[9,10,79].
The controversy on ferroelectricity in MHPs
Breaking inversion symmetry via octahedral distortion or cation displacement is crucial for MHPs to induce ferroelectricity, particularly when such octahedral tilts and polar cation displacements align along specific crystallographic directions rather than canceling out[80]. MAPbI3 is a typical 3D MHP, whose ferroelectricity is controversial because the fluctuating dipole moments induced by the dynamic disorder of organic cations are coupled with hysteresis phenomena in current-voltage characteristics caused by ion migration, together posing significant challenges in accurately interpreting evidence of ferroelectricity. To date, it is widely accepted that MAPbI3 is inherently ferroelastic rather than truly ferroelectric under operating conditions[81-83]. MHPs with 2D or lower-symmetry structures, featuring asymmetric organic cations and layered architectures, can break inversion symmetry and achieve robust ferroelectric order[42,84,85]. However, the strategy of simply introducing bulky spacer cations and creating 2D perovskite layers seldom produces genuine ferroelectric materials, since mobile ions, dynamic molecular reorientation, and residual inversion symmetry can prevent the emergence of long-range ferroelectric order[86,87]. To address this, structural strategies based on ferroelectrochemistry theory - such as quasi-spherical theory, incorporation of homochiral organic cations, and H/F substitution - have been proposed and widely applied to induce macroscopic polarization more efficiently in MHPs, greatly enriching the family of high-performance MHPs-based ferroelectric devices[79,88].
Enhanced ferroelectricity of MHPs-based composites
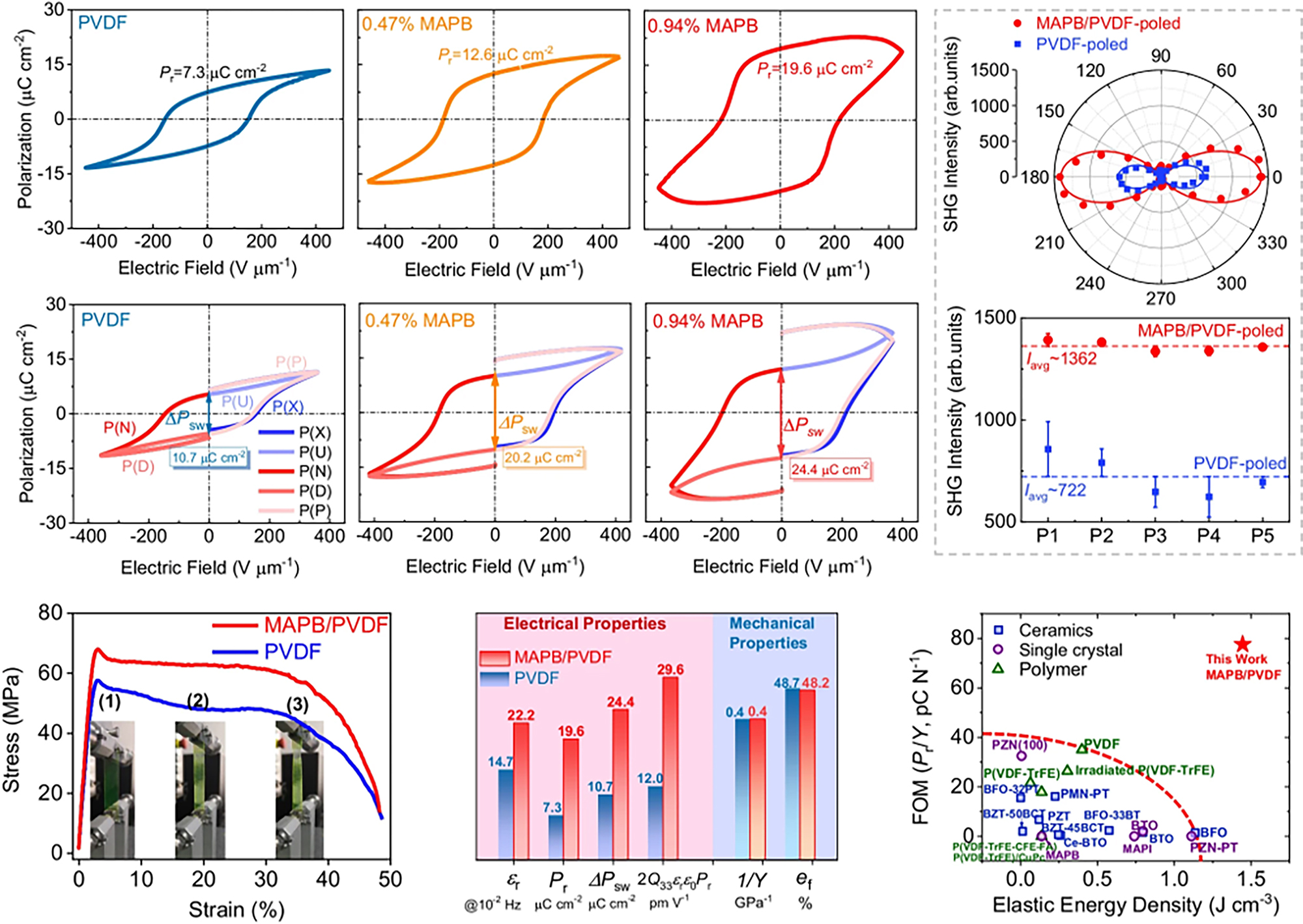
Compared with inorganic ferroelectric ceramics, ferroelectric polymers are easy to process, lightweight, flexible, and low-cost. Nevertheless, their relatively low polarization values restrict their applications in ferroelectric-related devices, making the development of ferroelectric polymers with high polarization strength urgently needed. Generally, there are two primary approaches to boost the sensing performance of PVDF-based ferroelectric materials. One approach is to optimize the manufacturing process, such as stretching, annealing, and poling, or structural design to increase the polar β-phase content; the other is to incorporate ferroelectric nanofillers with large polarization into ferroelectric polymers[27,89]. Despite these efforts, only limited success has been achieved, leaving the competing demands for both strong polarization and large mechanical compliance yet to be satisfied. Our recent work demonstrated that a PVDF nanocomposite containing only 0.94% self-nucleated MAPbBr3 nanocrystals exhibits anomalously large polarization (~19.6 μC cm-2) while retaining superior stretchability and PL, resulting in unprecedented electromechanical figures of merit among developed ferroelectrics, as shown in Figure 4[25]. The anomalous polarization enhancement in the MAPbBr3/PVDF nanocomposite is accomplished via delicate defect engineering, with field-induced Frenkel pairs in the halide perovskite stabilized by the poled ferroelectric polymer through interfacial coupling[25]. Specifically, the poling process induces the generation of Frenkel pairs V•Br-I’Br in MAPbBr3 nanocrystals[90], which strengthens the interfacial coupling between MAPbBr3 and PVDF via interactions between positively charged Br vacancies and strongly electronegative F atoms. This favors an ordered arrangement of V•Br-I’Br dipoles along the poling direction, contributing to the large enhancement in overall remnant polarization. This strategy is also general, indicating that MHP/polymer composites provide an ideal platform for investigating the interactions between optical, mechanical, and electrical properties[25].
Figure 4. Ferroelectric polarization of CH3NH3PbBr3 (MAPB)/PVDF nanocomposites compared with PVDF film[25].
Piezoelectricity
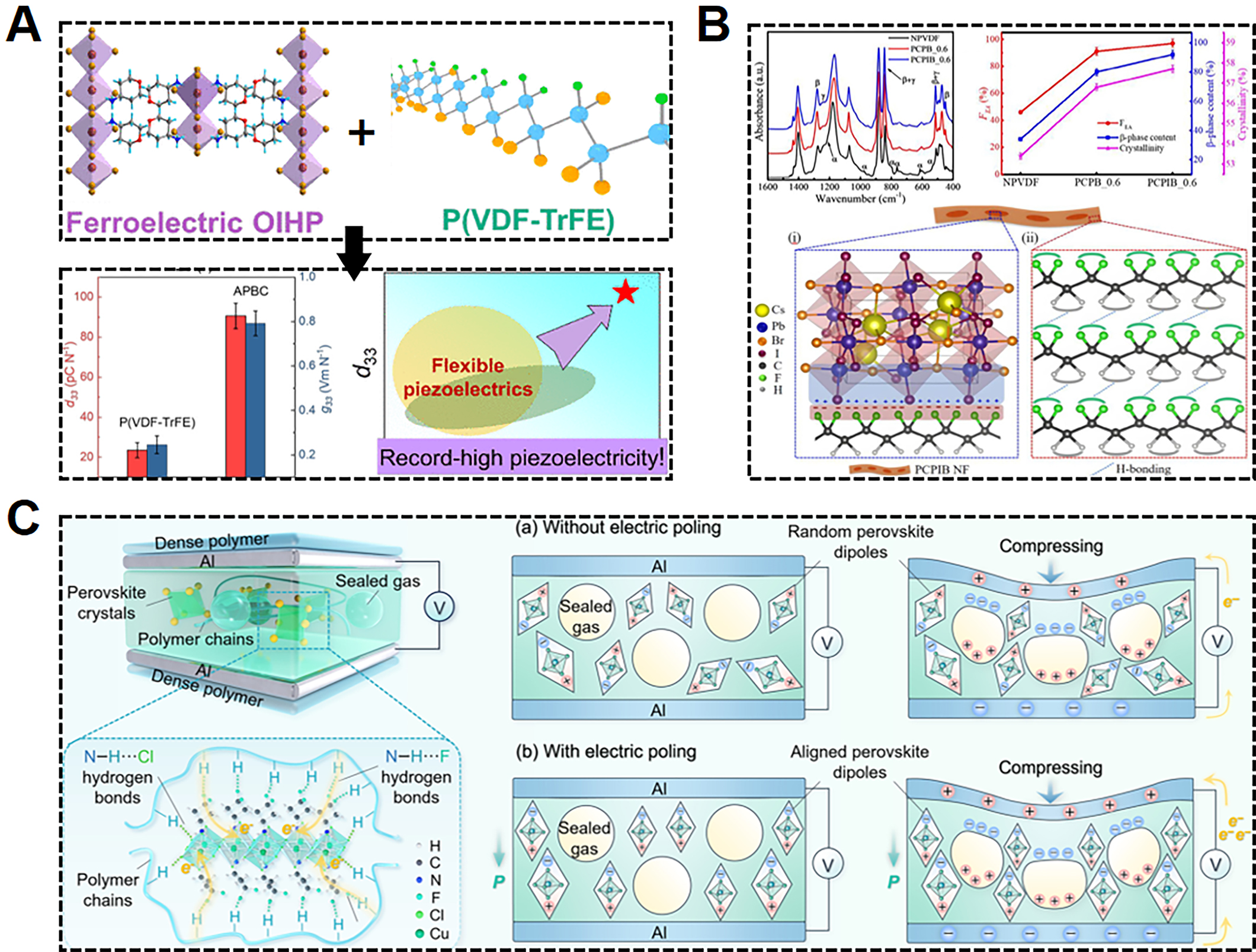
Piezoelectricity is the ability of non-centrosymmetric materials to generate electric charges in response to mechanical stress, or conversely, to undergo mechanical deformation when subjected to an electric field, corresponding to the direct piezoelectric effect and the converse piezoelectric effect, respectively. The integration of piezoelectric materials with high piezoelectric coefficients, such as lead zirconate titanate (PZT)[91,92] and barium titanate (BT)[93,94], into PVDF-based piezoelectric polymers can substantially enhance output performance; however, this often comes at the cost of degraded mechanical properties and reduced durability of the polymers. In contrast to the inorganic counterparts, highly polar MHPs, which have a much lower modulus[95], are more compatible with polymers, suggesting their potential to enhance the piezoelectricity of polymers[96-98]. Chai et al. incorporated the hybrid perovskite (4-aminotetrahydropyran)2PbBr2Cl2 (APBC), with a high piezoelectric coefficient (d33) of 90.6 pC N-1, into P(VDF-TrFE) to enhance the piezoelectricity of the APBC/P(VDF-TrFE) core-sheath nanofibers to 78.1 pC N-1, as illustrated in
Figure 5. (A) Schematic diagram of integrating (4-aminotetrahydropyran)2PbBr2Cl2 (APBC) nanorods with P(VDF-TrFE) to achieve both high piezoelectric and pyroelectric performances[99]. (B) Schematic illustration on interfacial interaction between MHP and PVDF for enhanced polarization[102]. (C) Illustration of double-electrode gas-solid triboelectric nanogenerators (GS-TENGs) device based on porous poly(siloxane-diphenylglyoxime-urethane)-perovskite (PSDU-PK) composite and its power generation mechanism[112].
Triboelectricity
The triboelectric effect describes the phenomenon in which electrons move due to variations in electron affinity between two different materials upon intimate contact, and the magnitude of charge transfer depends on factors such as surface roughness, contact pressure, environmental humidity, and the inherent properties of the materials[104]. By utilizing the triboelectric effect and electrostatic induction, mechanical energy can be converted into electrical energy, enabling energy harvesting from a wide range of environmental sources, including wind, ocean, and human movements. Since MHPs naturally possess various point defects, they can function as efficient charge acceptors, storing and attracting more charges, which contributes to the enhancement of the triboelectric effect[105-107]. For example, Wang et al. employed PVDF and inorganic CPB perovskite as a triboelectric pair and systematically optimized the electron binding energy, dielectric properties, and surface potential of CPB by doping Ba2+[108]. This increased space charge polarization and work function, thereby significantly enhancing the output performance[108]. In addition, incorporating MHPs into polymer matrices such as PVDF can significantly expand the surface charge potential difference between triboelectric layers, which is another widely employed approach to boost triboelectric output[109-111]. As shown in Figure 5C, Jiang et al. developed a porous TENG by integrating an adhesive poly(siloxane-diphenylglyoxime-urethane) (PSDU) elastomer with ferroelectric (3,3-difluorocyclobutylammonium)2CuCl4 [(DF-CBA)2CuCl4] functional fillers, leveraging the formation of a hydrogen-bonding network to enhance the charge transfer process[112]. The dipoles of the MHP are aligned along the electric field direction when electric poling is applied to the composite, which not only contributes to surface charge accumulation on the elastomer but also facilitates charge separation and redistribution within the voids, leading to a substantial enhancement in power generation[112].
APPLICATIONS
Since Kojima et al. first used organic-inorganic hybrid perovskites MAPbI3 and MAPbBr3 as novel photosensitizers to fabricate PSCs in 2009[113], extensive research efforts have been dedicated to understanding the photophysical properties of MHPs and improving the photovoltaic performance of PSCs, and numerous excellent reviews on this topic are available[36,114-117]. With the rapid and extensive exploration of the structures and properties of the MHP family, great opportunities beyond optoelectronic applications have also emerged. Table 1 summarizes representative perovskite-based composites with various functions and performances.
Perovskite-based composites with various functions and performances
| Perovskites | Composite component | Stability enhancement | Crystallization | Charge separation | Prevent Pb leakage | Flexibility | Applications | Performances | Ref. |
| MAPbI3 | PEOXA | √ | √ | LED | EQE: 5.4%, EM: 760 nm | [118] | |||
| BA2Csn-1PbnI3n+1 | PEO | √ | √ | LED | EQE: 6.23%, EM: 680 nm | [119] | |||
| MAPbBr3 | Pb-MOF | √ | √ | LED | EQE: 15.96%, PLQY: 70.48%, PEM: 520 nm | [120] | |||
| CsPbI3 | agZIF-62 | √ | √ | √ | LED | PLQY: 50.6%, EM: 674 nm | [34] | ||
| PEABr:CsPbBr3 | MPEG-MAA | √ | √ | LED | EQE 28.1%, PLQY: 78%, EM: 514 nm | [48] | |||
| MAPbBr3 | PVDF | √ | √ | √ | LED | PLQY: 94.6%, EM: 525 nm | [56] | ||
| FA0.7MA0.2Cs0.1Sn0.5Pb0.5I3 | β-PV2F | √ | √ | √ | PSC | PCE: 23.44%, FF: 80.9% | [68] | ||
| (FAPbI3)1-x(MAPbBr3)x | HDA-HBPs | √ | √ | √ | √ | PSC | PCE: 23.86%, FF: 81% (Flexible) PCE: 25.05%, FF: 84% (Solid) | [58] | |
| Cs0.05(FA0.98MA0.02) 0.95Pb(I0.98Br0.02)3 | β-PV2F | √ | √ | √ | PSC | PCE: 24.6%, FF: 84.3% | [121] | ||
| (FAPbI3)0.93(MAPbBr3)0.04(CsPbI3)0.03 | MOF-808@TpPa-1-COF | √ | √ | √ | PSC | PCE: 23.61%, FF: 78% | [122] | ||
| MAPbI3 | P(VDF-TrFE) | √ | √ | √ | PSC | PCE: 21.38%, FF: 78% | [66] | ||
| Cs0.05(MA0.15FA 0.85)0.95Pb(I0.85Br 0.15)3 | PVB | √ | √ | √ | PSC | PCE: 22.47%, FF: 83.8% | [71] | ||
| Cs0.03FA0.95MA0.02Pb(I0.975Br0.025)3 | POF-HDDA | √ | √ | √ | √ | PSC | PCE: 24.76%, FF: 80.13% | [74] | |
| (FAPbI3)1-x(MAPbBr3)x | HPDA | √ | √ | √ | √ | PSC | PCE: 24.43%, FF: 82% (Flexible) PCE: 25.92%, FF: 85% (Solid) | [22] | |
| FAPbI3 | PPETA | √ | √ | √ | √ | PSC | PCE: 24.9%, FF: 83.9% | [123] | |
| MAPbI3 | PCN-221(Fex) | √ | √ | Photocatalysis | Rate: 112 μmol g-1 h-1 | [124] | |||
| CsPbI3 | PCN-222 | √ | √ | Photocatalysis | Rate: 81.7% (630 nm, 7 mW cm-2, 4 h) | [125] | |||
| CsPbBr3 | UiO-67@PTZ | √ | √ | Photocatalysis | Rate: 82.2% (100 mW cm-2, 5 h) | [126] | |||
| MAPbI3 | P(VDF-TrFE) | √ | √ | Optical Sensor | D: 1.4 × 1013 Jones; R: 20 mA W-1 | [127] | |||
| MAPbI3 | ZIF-11 | √ | √ | √ | Optical Sensor | D: 4.22 × 1012 Jones; R: 185 mA W-1 | [128] | ||
| BA2CsPb2Br7 | PVDF | √ | √ | √ | PENG | d33: 63.3 pC N-1, VOC: 135 V, ISC: 355 nA, Pmax: 15.7 μW | [26] | ||
| CsPbBr3 | PVDF | √ | √ | √ | √ | PENG | VOC: 13 V, ISC: 1.3 μA, | [28] | |
| CsPb1-xBaxBr3 (x = 0.01-0.13) | PVDF | √ | TENG | VOC: 220 V, JSC: 22.8 mA m-2, | [108] | ||||
| CsPbBr3 | PAN, PANI | √ | √ | √ | TENG | VOC: 324 V, JSC: 12 μA cm-2, Pmax: 1,224 mW m-2 | [129] | ||
| (DF-CBA)2CuCl4 | PSDU | √ | √ | √ | TENG | VOC: 5.1 V, JSC: 44.4 μA cm-2, Pmax: 141.6 mW m-2 | [112] | ||
| FAPbI3 | PVDF | √ | √ | √ | TENG | VOC: 392 V, ISC: 61 μA, Pmax: 2,587 μW cm-2 | [29] | ||
| MAPbI3 | PVDF | √ | √ | √ | P-PENG | d33: 19.7 pC N-1, VOC: 220 mV, ISC: 4 nA, Pmax: 0.8 mW m-2 p: 44 pC m-2 K-1, VOC: 41.78 mV, | [96] | ||
| (ATHP)2PbBr2Cl2 | P(VDF-TrFE) | √ | √ | P-PENG | d33: 78.1 pC N-1, VOC: 192 V, p: 58.2 pC m-2 K-1 | [99] | |||
| Cs3Bi2Br9 | P(VDF-HFP), SEBS | √ | √ | √ | T-PENG | VOC: 400 V, JSC: 1.63 μA cm-2, | [97] | ||
| CsPbBr3 | PDMS-MWCNTs | √ | √ | PSCs, TENG | PCE: 1.80%, FF: 58.1% JSC: 0.735 μA cm-2, Pmax: 0.06 W cm-2 (Light OFF) JSC: 0.72 mA cm-2, Pmax: 5.98 W cm-2 (Light ON@100 mW cm-2) | [130] | |||
| MAPbBr3 | PVDF | √ | √ | √ | LED, PENG | PLQY: 32.6%, EM: 533 nm VOC: 5 V, ISC: 60 nA | [131] | ||
| FAMAPbI3 | PSU | √ | √ | √ | Optical sensor, TENG | R: 14.26 mA W-1, 119.67 V W-1 VOC: 285 V, ISC: 20 μA, | [132] | ||
| MAPbI3 | P(VDF-TrFE)/PEDOT:PSS | √ | √ | √ | Optical sensor, T-PENG | VOC: 228 V, JSC: 118 μA cm-2, Pmax: 4.5 mW m-2 (Light OFF) VOC: 208 V, JSC: 254 μA cm-2, Pmax: 12.5 mW m-2 (Light ON@40 mW cm-2) | [133] | ||
| MAPbI3 | PVA, PTFE | √ | √ | √ | Optical-mechanical sensor | D: 1.85 × 1010 Jones; R: 64 mA W-1 | [134] | ||
| CsPb0.25Zn0.75I3 | PVDF, CNC | √ | √ | √ | Photocatalysis, P-PENG | VOC: 20.3 V, JSC: 1.2 μA cm-2, | [135] |
Optoelectronics
For MHPs-based composites, when applied in optoelectronics, the additional component slows down the nucleation rate and reduces the nucleation density of perovskite, resulting in the growth of larger grains and effective defect passivation. Therefore, PSC devices are correspondingly designed in mixture or layered configurations to maximize photoelectric performance and stability[58,136-138]. As demonstrated in Figure 6A, in polymer-assisted PSC fabrication, perovskite crystallization is guided by long-chain polymers, causing the MHP film to crystallize into greatly enlarged grains with high crystallinity due to increased activation energy for nucleation and diffusion of precursor molecules. Furthermore, the residual polymeric Lewis base effectively passivates high-energy defect sites at the grain boundaries, thereby significantly improving both environmental and operational stability[137].
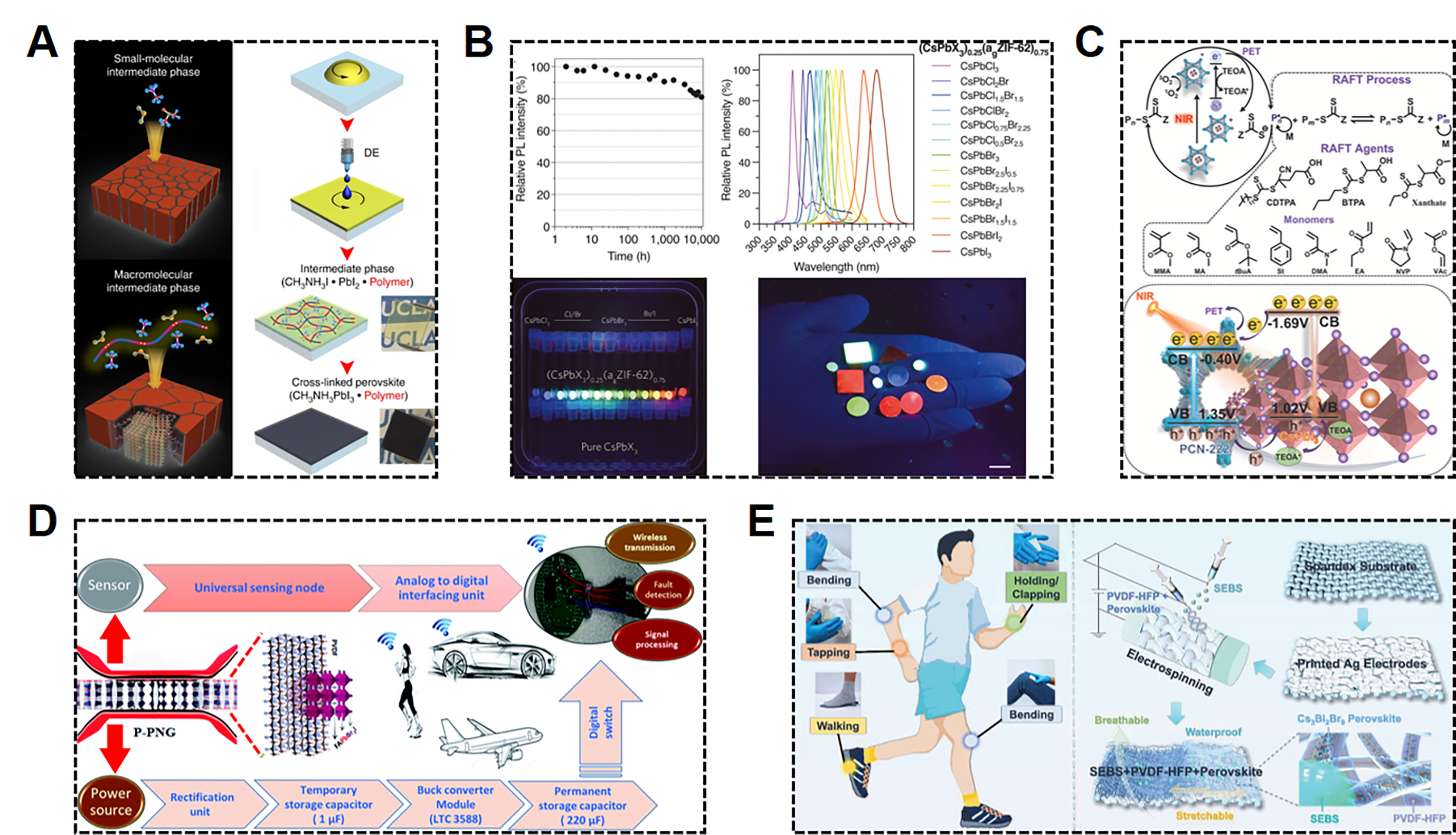
Figure 6. (A) Schematic illustrations of the perovskite film formation and fabrication steps[137]. (B) Stability and optical performance of the (CsPbI3)0.25(agZIF-62)0.75 composites[34]. (C) CsPbI3@PCN-222 mediated PET-RAFT polymerization process under NIR light and monomer structures investigated in this work, and schematic of energy-band structure and NIR-light induced charge separation and transfer over CsPbI3@PCN-222 photocatalyst[125]. (D) Framework of the self-powered integrated wireless electronics node by simultaneously using perovskite/polymer PENG as a power source and a sensor[154]. (E) Conceptual demonstrations of lead-free perovskite/P(VDF-HFP) [poly(vinylidene fluoride-co-hexafluoropropylene)] and PPS nanofiber composite for energy harvesting from various human motions[97].
For mass production of PSCs, mechanical robustness becomes a critical issue in device design. Therefore, incorporating MHPs into polymers, either as additives or as buffer layers, also provides advantages in mechanical flexibility and interfacial adhesion[22,58,123]. Li et al. developed highly adhesive polyamide-amine-based hyperbranched polymers to reinforce the interface of flexible PSCs, increasing the interfacial fracture energy from 1.08 to 2.13 J m-2[58]. The abundant carboxyl and amine groups present in the intramolecular cavities of the hyperbranched polymers exhibit a strong capacity for binding Pb2+ ions, enabling a remarkable lead sequestration efficiency of 98%[58]. In the work by Yuan et al., the mechanism of PCE enhancement via insertion of an ultra-thin ferroelectric P(VDF-TrFE) film at the interface between the electrode and the semiconductor layer was clarified[67,139,140]. The enhancement originates from significantly improved dissociation of charge-transfer excitons through the induced electric field within the ferroelectric polymer, rather than the previously suggested electrochemical reactions between the metal electrode and P(VDF-TrFE) under an applied electric field[67,139,140].
MHPs emitters and LEDs are promising candidates for next-generation high-colour-purity displays and lighting applications due to their advantageous optoelectronic properties[141]. MHPs-based composites with additional components, such as MOFs, provide an ideal template for the confined growth of ultra-small perovskite QDs with strong quantum confinement for high external quantum efficiency (EQE) luminescence and uniform size distribution. This approach offers a promising route to overcome bottlenecks such as insufficient color reproducibility and low operational stability[16,55]. In the study reported by Hou et al., a composite of MHP nanocrystals embedded in a subfamily of MOFs, namely zeolitic imidazolate frameworks (ZIFs) glass matrix, exhibited a two-orders-of-magnitude enhancement in PL performance compared to single perovskite. Notably, as shown in Figure 6B, the composite maintained 80% of its initial PL intensity after 10,000 h of immersion in water, storage under ambient conditions for 650 days, mild heating, and continuous laser excitation (~57 mW cm-2) for over 500 s, demonstrating its exceptional stability[34].
Photocatalysis
The unique nanoporous structures of MHPs composited with MOFs possess unprecedented chemical and structural tunability, which may unlock new catalytic functions, such as photocatalytic CO2 reduction[32,142,143], by leveraging the light-harvesting capabilities of perovskites together with the catalytic activity of MOF materials. This provides a promising platform for photocatalytic applications. As demonstrated in the study, a series of composite photocatalysts composed of MAPbI3 QDs embedded in the pores of Fe-porphyrin-based MOF PCN-221(Fex) were fabricated, with the optimized composition MAPbI3@PCN-221(Fe0.2) exhibiting a 38-fold higher yield than PCN-221(Fe0.2) in CO2 reduction, due to efficient charge separation between perovskite QDs and MOFs[124]. Moreover, MHP@MOF composite can also be used to catalyze photo-induced polymerization reactions. As shown in Figure 6C, a binary hybridization strategy for designing a highly efficient and stable photocatalyst for photoinduced electron transfer-reversible addition-fragmentation chain transfer (PET-RAFT) polymerization has been demonstrated by introducing CsPbI3 into a porphyrinic Zr-MOF (PCN-222)[125]. The composite exhibits enhanced absorption in the red to near-infrared (NIR) spectrum, enabling efficient PET-RAFT polymerization of various monomers, including both conjugated (e.g., styrene) and less-activated unconjugated monomers (e.g., vinyl acetate), under visible or NIR light illumination, achieving high monomer conversion, narrow molecular weight distribution, and excellent chain-end fidelity[125].
Multifunctional sensor
With the boom of the Internet of Things (IoTs), there is a growing demand for multifunctional sensors that integrate more than one sensing capability and meet the needs for system miniaturization and multi-functionality[144-148]. Current technologies face challenges related to device flexibility, energy consumption, and the complexity of decoupling fused information when realizing multifunctional sensors[149,150]. Therefore, attention has been focused on developing multifunctional material-integrated devices where sensing signals can be readily extracted, decoupled, and processed. MHPs-based composites, possessing multiple functions, are simultaneously responsive to light, force, temperature, electric fields, and more, making them promising candidates for multifunctional sensors[107,151,152].
For example, the integration of a photodetector based on CPB QD films with a pressure sensor made from PEDOT:PSS (poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate))-coated cellulose paper produces a flexible bifunctional sensor capable of detecting both light and pressure stimuli[153]. Similarly, a dual-mode self-powered optoelectronic device, constructed from a photo-conductive layer of MAPbI3/polyvinyl alcohol (PVA) composite and a triboelectric layer of polytetrafluoroethylene (PTFE), can convert mechanical and illumination signals into pulsed electrical signals and continuous resistance variations, respectively, enabling concurrent monitoring of human motion and environmental illumination conditions[134]. As shown in
Multi-energy harvesting
MHPs-based composites possess both optical and ferroelectric-related properties, enabling them to convert various forms of energy - such as solar, wind, ocean, and thermal energy - into electricity via photoelectric, piezoelectric, triboelectric, and pyroelectric effects, respectively, highlighting their potential in distributed self-powered systems[96,100,135,155]. For example, a multi-energy harvesting device was designed using a stretchable, breathable, and stable nanofiber composite of Cs3Bi2Br9 incorporated into P(VDF-HFP) and styrene-ethylene-butylene-styrene (SEBS), exhibiting both triboelectric and piezoelectric functions[97]. As shown in Figure 6E, efficient energy conversion from various human movements into electrical power enables the device to drive electronics such as LEDs and capacitors. Additionally, these devices demonstrate robust resistance to extreme mechanical deformations, including washing, folding, and crumpling, while maintaining stable electrical performance for up to five months, indicating great potential as self-powered wearable devices[97]. Beyond mechanical energy, light energy harvesting has also emerged for perovskite-based energy harvesters by integrating the photoelectric effect with piezoelectric or triboelectric effects, enabling synergistic enhancement of device power output. A photo-responsive wearable hybrid nanogenerator (HNG) was designed using a polyacrylonitrile (PAN)/CPB/polyaniline (PANI) composite as the electropositive layer, integrating both triboelectric and photovoltaic energy conversion. Under illumination, CPB generates photo-induced carriers, while PANI facilitates charge transport, resulting in enhanced output power density[129].
CONCLUSION AND OUTLOOK
MHPs have been actively investigated as excellent light-sensitive materials ever since their first application in solar cells. Beyond this, they also exhibit other functionalities, such as ferroelectricity, opening opportunities for next-generation multifunctional electronic devices. MHPs-based composites not only offer improved mechanical flexibility, stability, and enhanced optoelectronic properties of MHPs, but also provide multifunctionality suitable for multi-energy harvesting and multifunctional sensor applications. Despite the exciting and ongoing progress achieved in MHPs-based composites, they remain at an early stage, and several major challenges still need to be addressed.
Delicate composition and structure design
When compositing MHPs with another component to enable additional functions or improve stability, maintaining the exceptional optoelectronic properties of MHPs is a primary requirement. However, the coexistence of photoelectricity and ferroelectricity can be partially mutually exclusive: photovoltaic performance requires high carrier mobility, while polarization relies on bound charges. Therefore, careful selection of components, precise control of composition and microstructure - including the distribution of MHPs and the quality of interfaces - is vital to ensure coexistence of functions or even synergistic enhancement beyond the sum of individual properties.
Enhancing thermal stability
Although compositing MHPs with polymers and MOFs aims to overcome the environmental instability of MHPs, the thermal stability of both polymers and MHPs themselves remains limited, as they lose long-range structural order at elevated temperatures. Consequently, the practical operating temperature range is still insufficient. Strategies to develop stable MHP composites with ferroelectricity at higher temperatures are needed, such as using more thermally stable components - e.g., inorganic perovskites and high-temperature-resistant MOFs - or employing high-temperature-resistant polymers such as polyimide and its derivatives.
Multi-field coupling mechanism
MHPs-based composites offer a promising platform for integrating light-related properties with ferroelectricity, which could enable both large output voltage and high efficiency in light energy harvesting. Achieving this requires elucidation of the coupling mechanisms involving light, charge, and polarization, which demands significant effort. In particular, in situ multi-field characterization methods at atomic to mesoscopic scales, in both spatial and temporal dimensions with high resolution, remain underdeveloped. In situ ultrafast spectroscopy with high spatial resolution may offer a promising approach for investigating these mechanisms.
DECLARATIONS
Authors’ contributions
Wrote the original draft: Zheng, Y.
Investigation: Zheng, Y.; Zhang, L.
Supervised, reviewed, and revised the manuscript: Wang, Y.
Availability of data and materials
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant Nos. 52472118, 92466201).
Conflict of interest
Wang, Y. is the Guest Editor of the Special Issue “Polymer-based Nanocomposites for Energy Storage and Conversion” of the journal. Wang, Y. was not involved in any steps of the editorial process, notably including reviewers' selection, manuscript handling, or decision-making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Han, Y.; Yue, S.; Cui, B. B. Low-dimensional metal halide perovskite crystal materials: structure strategies and luminescence applications. Adv. Sci. 2021, 8, e2004805.
2. Dong, H.; Zhang, C.; Liu, X.; Yao, J.; Zhao, Y. S. Materials chemistry and engineering in metal halide perovskite lasers. Chem. Soc. Rev. 2020, 49, 951-82.
3. Fu, Y.; Zhu, H.; Chen, J.; Hautzinger, M. P.; Zhu, X. Y.; Jin, S. Metal halide perovskite nanostructures for optoelectronic applications and the study of physical properties. Nat. Rev. Mater. 2019, 4, 169-88.
4. Filip, M. R.; Eperon, G. E.; Snaith, H. J.; Giustino, F. Steric engineering of metal-halide perovskites with tunable optical band gaps. Nat. Commun. 2014, 5, 5757.
5. Xiong, Z.; Zhang, Q.; Cai, K.; et al. Homogenized chlorine distribution for >27% power conversion efficiency in perovskite solar cells. Science 2025, 390, 638-42.
6. Zhou, Q.; Huang, G.; Wang, J.; et al. Aromatic interaction-driven out-of-plane orientation for inverted perovskite solar cells with improved efficiency. Nat. Energy. 2025, 10, 1371-81.
7. He, D.; Ma, D.; Zhang, J.; et al. Universal ion migration suppression strategy based on supramolecular host-guest interaction for high-performance perovskite solar cells. Adv. Mater. 2025, 37, e2505115.
8. Pandey, R.; Vats, G.; Yun, J.; et al. Mutual insight on ferroelectrics and hybrid halide perovskites: a platform for future multifunctional energy conversion. Adv. Mater. 2019, 31, e1807376.
9. Zhang, T.; Xu, K.; Li, J.; et al. Ferroelectric hybrid organic-inorganic perovskites and their structural and functional diversity. Natl. Sci. Rev. 2023, 10, nwac240.
10. Zheng, W.; Wang, X.; Zhang, X.; et al. Emerging halide perovskite ferroelectrics. Adv. Mater. 2023, 35, e2205410.
11. Li, X.; Zhang, W.; Guo, X.; Lu, C.; Wei, J.; Fang, J. Constructing heterojunctions by surface sulfidation for efficient inverted perovskite solar cells. Science 2022, 375, 434-7.
12. Bai, Y.; Dong, Q.; Shao, Y.; et al. Enhancing stability and efficiency of perovskite solar cells with crosslinkable silane-functionalized and doped fullerene. Nat. Commun. 2016, 7, 12806.
13. Wang, R.; Xue, J.; Meng, L.; et al. Caffeine improves the performance and thermal stability of perovskite solar cells. Joule 2019, 3, 1464-77.
14. Wang, Y.; He, J.; Chen, H.; et al. Ultrastable, highly luminescent organic-inorganic perovskite-polymer composite films. Adv. Mater. 2016, 28, 10710-7.
15. Qin, J.; Chen, Y.; Guo, X.; et al. A polymer strategy toward high-performance multifunctional perovskite optoelectronics: from polymer matrix to device applications. Adv. Opt. Mater. 2023, 11, 2202809.
16. Zhang, C.; Li, W.; Li, L. Metal halide perovskite nanocrystals in metal-organic framework host: not merely enhanced stability. Angew. Chem. Int. Ed. 2021, 60, 7488-501.
17. Khare, S.; Sundar, S. K.; Gohel, J. V. Advanced materials to overcome the challenges in the fabrication of stable and efficient perovskite solar cells by additive engineering: a review. J. Mater. Sci. 2023, 58, 16565-90.
18. He, Y.; Liang, Y.; Liang, S.; et al. Dual-protected metal halide perovskite nanosheets with an enhanced set of stabilities. Angew. Chem. Int. Ed. 2021, 60, 7259-66.
19. Zhang, Z.; Qiao, L.; Meng, K.; Long, R.; Chen, G.; Gao, P. Rationalization of passivation strategies toward high-performance perovskite solar cells. Chem. Soc. Rev. 2023, 52, 163-95.
20. Wu, S.; Li, Z.; Li, M. Q.; et al. 2D metal-organic framework for stable perovskite solar cells with minimized lead leakage. Nat. Nanotechnol. 2020, 15, 934-40.
21. Liu, Y.; Knaus, T.; Wei, Z.; et al. Confined flash printing and synthesis of stable perovskite nanofilms under ambient conditions. Adv. Mater. 2024, 36, e2409592.
22. Li, Z.; Jia, C.; Wan, Z.; et al. Boosting mechanical durability under high humidity by bioinspired multisite polymer for high-efficiency flexible perovskite solar cells. Nat. Commun. 2025, 16, 1771.
23. Zhou, Y.; Fei, C.; Uddin, M. A.; Zhao, L.; Ni, Z.; Huang, J. Self-powered perovskite photon-counting detectors. Nature 2023, 616, 712-8.
24. Yang, X.; Luo, D.; Xiang, Y.; et al. Buried interfaces in halide perovskite photovoltaics. Adv. Mater. 2021, 33, e2006435.
25. Wang, Y.; Huang, C.; Cheng, Z.; et al. Halide perovskite inducing anomalous nonvolatile polarization in poly(vinylidene fluoride)-based flexible nanocomposites. Nat. Commun. 2024, 15, 3943.
26. Chen, Z.; Zhang, M.; Hu, Y.; Wang, S.; Gu, H.; Xiong, J. Ultrahigh energy harvesting ability of PVDF incorporated with 2D halide perovskite nanosheets via interface effect. Chem. Eng. J. 2024, 497, 154558.
27. Park, S. Y.; Jang, M.; Kim, J.; et al. 2D perovskite nanosheet-driven polymeric nanocomposites as gate dielectrics for flexible negative-capacitance applications. Adv. Funct. Mater. 2024, 34, 2404466.
28. Sun, X.; Zhang, F.; Zhang, L.; et al. Enhanced electromechanical conversion via in situ grown CsPbBr3 nanoparticle/poly(vinylidene fluoride) fiber composites for physiological signal monitoring. Soft. Sci. 2022, 2, 1.
29. Zain, Karimy. N. H.; Li, H.; Lee, H. B.; et al. Highly efficient 3D-printed PVDF-based triboelectric nanogenerators featuring polymorphic perovskite nanofillers. Adv. Funct. Mater. 2025, 35, 2424271.
30. Zhao, X.; Tan, Z. K. Large-area near-infrared perovskite light-emitting diodes. Nat. Photonics. 2020, 14, 215-18.
31. Li, X.; Chen, L.; Yuan, S.; et al. Stretchable luminescent perovskite-polymer hydrogels for visual-digital wearable strain sensor textiles. Adv. Fiber. Mater. 2023, 5, 1671-84.
32. Kong, Z. C.; Liao, J. F.; Dong, Y. J.; et al. Core@shell CsPbBr3@zeolitic imidazolate framework nanocomposite for efficient photocatalytic CO2 reduction. ACS. Energy. Lett. 2018, 3, 2656-62.
33. Xu, C. Y.; Hu, W.; Wang, G.; et al. Coordinated optical matching of a texture interface made from demixing blended polymers for high-performance inverted perovskite solar cells. ACS. Nano. 2020, 14, 196-203.
34. Hou, J.; Chen, P.; Shukla, A.; et al. Liquid-phase sintering of lead halide perovskites and metal-organic framework glasses. Science 2021, 374, 621-5.
35. Xia, P.; Sun, H.; Guo, H.; et al. Luminescent solar concentrator with advanced structure for reabsorption loss suppression and synergistic energy harvesting. Adv. Funct. Mater. 2024, 34, 2401121.
36. Green, M. A.; Ho-Baillie, A.; Snaith, H. J. The emergence of perovskite solar cells. Nat. Photonics. 2014, 8, 506-14.
38. Liu, Y.; Yuan, S.; Zheng, H.; et al. Structurally dimensional engineering in perovskite photovoltaics. Adv. Energy. Mater. 2023, 13, 2300188.
39. Zhou, C.; Tian, Y.; Wang, M.; et al. Low-dimensional organic tin bromide perovskites and their photoinduced structural transformation. Angew. Chem. Int. Ed. 2017, 56, 9018-22.
40. Mao, L.; Stoumpos, C. C.; Kanatzidis, M. G. Two-dimensional hybrid halide perovskites: principles and promises. J. Am. Chem. Soc. 2019, 141, 1171-90.
41. Quan, L. N.; Yuan, M.; Comin, R.; et al. Ligand-stabilized reduced-dimensionality perovskites. J. Am. Chem. Soc. 2016, 138, 2649-55.
42. You, Y. M.; Liao, W. Q.; Zhao, D.; et al. An organic-inorganic perovskite ferroelectric with large piezoelectric response. Science 2017, 357, 306-9.
43. Sun, S.; Lu, M.; Gao, X.; et al. 0D perovskites: unique properties, synthesis, and their applications. Adv. Sci. 2021, 8, e2102689.
44. Zhao, X.; Liu, T.; Loo, Y. L. Advancing 2D perovskites for efficient and stable solar cells: challenges and opportunities. Adv. Mater. 2022, 34, e2105849.
45. Yu, S.; Liu, H.; Wang, S.; Zhu, H.; Dong, X.; Li, X. Hydrazinium cation mixed FAPbI3-based perovskite with 1D/3D hybrid dimension structure for efficient and stable solar cells. Chem. Eng. J. 2021, 403, 125724.
46. Kong, T.; Xie, H.; Zhang, Y.; et al. Perovskitoid-templated formation of a 1D@3D perovskite structure toward highly efficient and stable perovskite solar cells. Adv. Energy. Mater. 2021, 11, 2101018.
47. Tian, T.; Yang, M.; Fang, Y.; et al. Large-area waterproof and durable perovskite luminescent textiles. Nat. Commun. 2023, 14, 234.
48. Liu, Z.; Qiu, W.; Peng, X.; et al. Perovskite light-emitting diodes with EQE exceeding 28% through a synergetic dual-additive strategy for defect passivation and nanostructure regulation. Adv. Mater. 2021, 33, e2103268.
49. Li, X.; Zhang, W.; Wang, Y. C.; Zhang, W.; Wang, H. Q.; Fang, J. In-situ cross-linking strategy for efficient and operationally stable methylammoniun lead iodide solar cells. Nat. Commun. 2018, 9, 3806.
50. Peng, J.; Khan, J. I.; Liu, W.; et al. A universal double-side passivation for high open-circuit voltage in perovskite solar cells: role of carbonyl groups in poly(methyl methacrylate). Adv. Energy. Mater. 2018, 8, 1801208.
51. Qiao, G. Y.; Guan, D.; Yuan, S.; et al. Perovskite quantum dots encapsulated in a mesoporous metal-organic framework as synergistic photocathode materials. J. Am. Chem. Soc. 2021, 143, 14253-60.
52. Liu, S.; Fang, H.; Su, Y.; et al. Optimizing printed quasi-2D luminescent perovskite films via delaminated metal-organic framework modulation. Adv. Mater. 2025, 37, e2501939.
53. He, H.; Cui, Y.; Li, B.; et al. Confinement of perovskite-QDs within a single MOF crystal for significantly enhanced multiphoton excited luminescence. Adv. Mater. 2019, 31, e1806897.
54. Hou, J.; Wang, Z.; Chen, P.; Chen, V.; Cheetham, A. K.; Wang, L. Intermarriage of halide perovskites and metal-organic framework crystals. Angew. Chem. Int. Ed. 2020, 59, 19434-49.
55. Fang, X.; Xie, Z.; Sharshir, S.; et al. Lead halide perovskite nanocrystals in metal-organic frameworks: synthesis, properties, and applications. Adv. Sci. 2025, 12, e05407.
56. Zhou, Q.; Bai, Z.; Lu, W. G.; Wang, Y.; Zou, B.; Zhong, H. In situ fabrication of halide perovskite nanocrystal-embedded polymer composite films with enhanced photoluminescence for display backlights. Adv. Mater. 2016, 28, 9163-8.
57. Hu, X.; Huang, Z.; Li, F.; et al. Nacre-inspired crystallization and elastic “brick-and-mortar” structure for a wearable perovskite solar module. Energy. Environ. Sci. 2019, 12, 979-87.
58. Li, Z.; Jia, C.; Wan, Z.; et al. Hyperbranched polymer functionalized flexible perovskite solar cells with mechanical robustness and reduced lead leakage. Nat. Commun. 2023, 14, 6451.
59. Li, M.; Yang, Y. G.; Wang, Z. K.; et al. Perovskite grains embraced in a soft fullerene network make highly efficient flexible solar cells with superior mechanical stability. Adv. Mater. 2019, 31, e1901519.
60. Jeong, B.; Han, H.; Choi, Y. J.; et al. All-inorganic CsPbI3 perovskite phase-stabilized by poly(ethylene oxide) for red-light-emitting diodes. Adv. Funct. Mater. 2018, 28, 1706401.
61. Kim, M.; Motti, S. G.; Sorrentino, R.; Petrozza, A. Enhanced solar cell stability by hygroscopic polymer passivation of metal halide perovskite thin film. Energy. Environ. Sci. 2018, 11, 2609-19.
62. Cao, Q.; Li, Y.; Zhang, H.; et al. Efficient and stable inverted perovskite solar cells with very high fill factors via incorporation of star-shaped polymer. Sci. Adv. 2021, 7, eabg0633.
63. Wang, S.; Gong, X. Y.; Li, M. X.; Li, M. H.; Hu, J. S. Polymers for perovskite solar cells. JACS. Au. 2024, 4, 3400-12.
64. Li, M.; Sun, R.; Chang, J.; et al. Orientated crystallization of FA-based perovskite via hydrogen-bonded polymer network for efficient and stable solar cells. Nat. Commun. 2023, 14, 573.
65. Yuan, Y.; Xiao, Z.; Yang, B.; Huang, J. Arising applications of ferroelectric materials in photovoltaic devices. J. Mater. Chem. A. 2014, 2, 6027-41.
66. Zhang, C. C.; Wang, Z. K.; Yuan, S.; et al. Polarized ferroelectric polymers for high-performance perovskite solar cells. Adv. Mater. 2019, 31, e1902222.
67. Yuan, Y.; Reece, T. J.; Sharma, P.; et al. Efficiency enhancement in organic solar cells with ferroelectric polymers. Nat. Mater. 2011, 10, 296-302.
68. Cheng, J.; Cao, H.; Zhang, S.; et al. Enhanced electric field minimizing quasi-fermi level splitting deficit for high-performance tin-lead perovskite solar cells. Adv. Mater. 2024, 36, e2410298.
69. Li, Z.; Wu, X.; Wu, S.; et al. An effective and economical encapsulation method for trapping lead leakage in rigid and flexible perovskite photovoltaics. Nano. Energy. 2022, 93, 106853.
70. Chen, B.; Fei, C.; Chen, S.; Gu, H.; Xiao, X.; Huang, J. Recycling lead and transparent conductors from perovskite solar modules. Nat. Commun. 2021, 12, 5859.
71. Dong, H.; Shen, G.; Fang, H.; et al. Internal encapsulation strategy using a polymer enables efficient, stable, and lead-safe inverted perovskite solar cells. Adv. Funct. Mater. 2024, 34, 2402394.
72. Xu, Y.; Guo, X.; Lin, Z.; et al. Perovskite films regulation via hydrogen-bonded polymer network for efficient and stable perovskite solar cells. Angew. Chem. Int. Ed. 2023, 62, e202306229.
73. Jiang, Y.; Qiu, L.; Juarez-Perez, E. J.; et al. Reduction of lead leakage from damaged lead halide perovskite solar modules using self-healing polymer-based encapsulation. Nat. Energy. 2019, 4, 585-93.
74. Zhang, J.; Li, Z.; Guo, F.; et al. Thermally crosslinked F-rich polymer to inhibit lead leakage for sustainable perovskite solar cells and modules. Angew. Chem. Int. Ed. 2023, 62, e202305221.
75. Fu, H.; Cohen, R. E. Polarization rotation mechanism for ultrahigh electromechanical response in single-crystal piezoelectrics. Nature 2000, 403, 281-3.
76. Choi, K. J.; Biegalski, M.; Li, Y. L.; et al. Enhancement of ferroelectricity in strained BaTiO3 thin films. Science 2004, 306, 1005-9.
77. Kingon, A. I.; Srinivasan, S. Lead zirconate titanate thin films directly on copper electrodes for ferroelectric, dielectric and piezoelectric applications. Nat. Mater. 2005, 4, 233-37.
78. Huangfu, G.; Zeng, K.; Wang, B.; et al. Giant electric field-induced strain in lead-free piezoceramics. Science 2022, 378, 1125-30.
79. Pan, Q.; Gu, Z. X.; Zhou, R. J.; et al. The past 10 years of molecular ferroelectrics: structures, design, and properties. Chem. Soc. Rev. 2024, 53, 5781-861.
80. Guo, Z.; Lin, J.; Mao, L. Dynamic control of halide perovskite structures for tailored ferroelectric and second-order nonlinear optical functionalities. Chem. Soc. Rev. 2025, 54, 8845-87.
81. Quarti, C.; Mosconi, E.; De, Angelis. F. Interplay of orientational order and electronic structure in methylammonium lead iodide: implications for solar cell operation. Chem. Mater. 2014, 26, 6557-69.
82. Wilson, J. N.; Frost, J. M.; Wallace, S. K.; Walsh, A. Dielectric and ferroic properties of metal halide perovskites. APL. Mater. 2019, 7, 010901.
83. Ali, W.; Qin, W.; Tian, H.; Guo, J.; Feng, Z.; Li, C. Tuning lattice structure of ferroelastic twin-domains achieving efficient perovskite solar cells. ACS. Energy. Lett. 2023, 8, 5070-78.
84. Li, P. F.; Liao, W. Q.; Tang, Y. Y.; Ye, H. Y.; Zhang, Y.; Xiong, R. G. Unprecedented ferroelectric-antiferroelectric-paraelectric phase transitions discovered in an organic-inorganic hybrid perovskite. J. Am. Chem. Soc. 2017, 139, 8752-7.
85. Yang, C. K.; Chen, W. N.; Ding, Y. T.; et al. The first 2D homochiral lead iodide perovskite ferroelectrics: [R- and S-1-(4-chlorophenyl)ethylammonium]2PbI4. Adv. Mater. 2019, 31, e1808088.
86. Leguy, A. M.; Frost, J. M.; McMahon, A. P.; et al. The dynamics of methylammonium ions in hybrid organic-inorganic perovskite solar cells. Nat. Commun. 2015, 6, 7124.
87. Rossi, D.; Pecchia, A.; der Maur, M. A.; et al. On the importance of ferroelectric domains for the performance of perovskite solar cells. Nano. Energy. 2018, 48, 20-6.
88. Liu, H. Y.; Zhang, H. Y.; Chen, X. G.; Xiong, R. G. Molecular design principles for ferroelectrics: ferroelectrochemistry. J. Am. Chem. Soc. 2020, 142, 15205-18.
89. Shi, K.; Chai, B.; Zou, H.; et al. Interface induced performance enhancement in flexible BaTiO3/PVDF-TrFE based piezoelectric nanogenerators. Nano. Energy. 2021, 80, 105515.
90. Chen, B.; Li, T.; Dong, Q.; et al. Large electrostrictive response in lead halide perovskites. Nat. Mater. 2018, 17, 1020-6.
91. He, W.; Guo, Y.; Zhao, Y. B.; et al. Self-supporting smart air filters based on PZT/PVDF electrospun nanofiber composite membrane. Chem. Eng. J. 2021, 423, 130247.
92. Tian, G.; Deng, W.; Gao, Y.; et al. Rich lamellar crystal baklava-structured PZT/PVDF piezoelectric sensor toward individual table tennis training. Nano. Energy. 2019, 59, 574-81.
93. Shi, K.; Sun, B.; Huang, X.; Jiang, P. Synergistic effect of graphene nanosheet and BaTiO3 nanoparticles on performance enhancement of electrospun PVDF nanofiber mat for flexible piezoelectric nanogenerators. Nano. Energy. 2018, 52, 153-62.
94. Luo, S.; Yu, J.; Yu, S.; et al. Significantly enhanced electrostatic energy storage performance of flexible polymer composites by introducing highly insulating-ferroelectric microhybrids as fillers. Adv. Energy. Mater. 2019, 9, 1803204.
95. Dunlap-Shohl, W. A.; Zhou, Y.; Padture, N. P.; Mitzi, D. B. Synthetic approaches for halide perovskite thin films. Chem. Rev. 2019, 119, 3193-295.
96. Sultana, A.; Ghosh, S. K.; Alam, M. M.; et al. Methylammonium lead iodide incorporated poly(vinylidene fluoride) nanofibers for flexible piezoelectric-pyroelectric nanogenerator. ACS. Appl. Mater. Interfaces. 2019, 11, 27279-87.
97. Jiang, F.; Zhou, X.; Lv, J.; et al. Stretchable, breathable, and stable lead-free perovskite/polymer nanofiber composite for hybrid triboelectric and piezoelectric energy harvesting. Adv. Mater. 2022, 34, e2200042.
98. Kumar, P.; Paul, T.; Sahoo, A.; et al. Harnessing self-powered and photoresponsive biomechanical activity sensors by exploring the piezo-phototronic effect in lead-free layered halide perovskite/PVDF composites. J. Mater. Chem. A. 2025, 13, 18420-35.
99. Chai, B.; Shi, K.; Wang, Y.; et al. Integrated piezoelectric/pyroelectric sensing from organic-inorganic perovskite nanocomposites. ACS. Nano. 2024, 18, 25216-25.
100. Salesh, M.; Sharma, S. K.; Mridha, N.; Nannapaneni, C. M.; Mural, P. K. S.; Yella, A. Multifunctional lead-free halide perovskite-based nanogenerator for enhanced energy harvesting and information-encrypted transmission. ACS. Appl. Mater. Interfaces. 2025, 17, 38481-92.
101. Wu, Y.; Pan, X.; Jin, X.; et al. High-performance flexible molecular-based piezoelectric composites for wireless microelectronics. Chem. Eng. J. 2025, 515, 163755.
102. Garain, S.; He, D.; Monluc, H.; Dammak, H.; Bai, J. Halide-tunable bond engineering for high-performance multi-responsive piezoelectric sensors via enhanced electrostatic polarization in in situ perovskite-embedded PVDF nanofibers. Small 2025, 21, e2504787.
103. Wu, S.; Zabihi, F.; Yeap, R. Y.; et al. Cesium lead halide perovskite decorated polyvinylidene fluoride nanofibers for wearable piezoelectric nanogenerator yarns. ACS. Nano. 2023, 17, 1022-35.
104. Zhao, Z.; Zhou, L.; Li, S.; et al. Selection rules of triboelectric materials for direct-current triboelectric nanogenerator. Nat. Commun. 2021, 12, 4686.
105. Musiienko, A.; Pipek, J.; Praus, P.; et al. Deciphering the effect of traps on electronic charge transport properties of methylammonium lead tribromide perovskite. Sci. Adv. 2020, 6, eabb6393.
106. Chen, Y.; Zhou, H. Defects chemistry in high-efficiency and stable perovskite solar cells. J. Appl. Phys. 2020, 128, 060903.
107. Su, L.; Zhao, Z. X.; Li, H. Y.; et al. High-performance organolead halide perovskite-based self-powered triboelectric photodetector. ACS. Nano. 2015, 9, 11310-6.
108. Wang, Y.; Duan, J.; Yang, X.; Liu, L.; Zhao, L.; Tang, Q. The unique dielectricity of inorganic perovskites toward high-performance triboelectric nanogenerators. Nano. Energy. 2020, 69, 104418.
109. Yu, A.; Zhu, Y.; Wang, W.; Zhai, J. Progress in triboelectric materials: toward high performance and widespread applications. Adv. Funct. Mater. 2019, 29, 1900098.
110. He, Y.; Wang, H.; Sha, Z.; Boyer, C.; Wang, C. H.; Zhang, J. Enhancing output performance of PVDF-HFP fiber-based nanogenerator by hybridizing silver nanowires and perovskite oxide nanocrystals. Nano. Energy. 2022, 98, 107343.
111. Cheon, S.; Kang, H.; Kim, H.; et al. High-performance triboelectric nanogenerators based on electrospun polyvinylidene fluoride-silver nanowire composite nanofibers. Adv. Funct. Mater. 2018, 28, 1703778.
112. Jiang, F.; Thangavel, G.; Lee, J. P.; et al. Self-healable and stretchable perovskite-elastomer gas-solid triboelectric nanogenerator for gesture recognition and gripper sensing. Sci. Adv. 2024, 10, eadq5778.
113. Kojima, A.; Teshima, K.; Shirai, Y.; Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 2009, 131, 6050-1.
114. Luo, J.; Li, J.; Grater, L.; et al. Vapour-deposited perovskite light-emitting diodes. Nat. Rev. Mater. 2024, 9, 282-94.
115. Wen, J.; Hu, H.; Chen, C.; et al. Present status of and future opportunities for all-perovskite tandem photovoltaics. Nat. Energy. 2025, 10, 681-96.
116. Prince, K. J.; Mirletz, H. M.; Gaulding, E. A.; et al. Sustainability pathways for perovskite photovoltaics. Nat. Mater. 2025, 24, 22-33.
117. Han, J.; Park, K.; Tan, S.; et al. Perovskite solar cells. Nat. Rev. Methods. Primers. 2025, 5, 3.
118. Chen, Z.; Li, Z.; Zhang, C.; et al. Recombination dynamics study on nanostructured perovskite light-emitting devices. Adv. Mater. 2018, 30, e1801370.
119. Tian, Y.; Zhou, C.; Worku, M.; et al. Highly efficient spectrally stable red perovskite light-emitting diodes. Adv. Mater. 2018, 30, e1707093.
120. Tsai, H.; Shrestha, S.; Vilá, R. A.; et al. Bright and stable light-emitting diodes made with perovskite nanocrystals stabilized in metal-organic frameworks. Nat. Photonics. 2021, 15, 843-49.
121. Li, G.; Su, Z.; Canil, L.; et al. Highly efficient p-i-n perovskite solar cells that endure temperature variations. Science 2023, 379, 399-403.
122. Dong, Y.; Zhang, J.; Zhang, H.; et al. Multifunctional MOF@COF nanoparticles mediated perovskite films management toward sustainable perovskite solar cells. Nanomicro. Lett. 2024, 16, 171.
123. Wu, Y.; Xu, G.; Shen, Y.; et al. Stereoscopic polymer network for developing mechanically robust flexible perovskite solar cells with an efficiency approaching 25. Adv. Mater. 2024, 36, e2403531.
124. Wu, L. Y.; Mu, Y. F.; Guo, X. X.; et al. Encapsulating perovskite quantum dots in iron-based metal-organic frameworks (MOFs) for efficient photocatalytic CO2 reduction. Angew. Chem. Int. Ed. 2019, 58, 9491-5.
125. Xia, Z.; Shi, B.; Zhu, W.; Xiao, Y.; Lü, C. Binary hybridization strategy toward stable porphyrinic Zr-MOF encapsulated perovskites as high-performance heterogeneous photocatalysts for red to NIR light-induced PET-RAFT polymerization. Adv. Funct. Mater. 2022, 32, 2207655.
126. Yin, Z.; Liu, X.; Liang, G.; Cheng, H.; Zhao, C. Facile construction of a double-heterojunction perovskite quantum dot system for efficient photocatalytic Cr6+ reduction. ACS. Appl. Mater. Interfaces. 2024, 16, 39506-16.
127. Cao, F.; Tian, W.; Meng, L.; Wang, M.; Li, L. Ultrahigh-performance flexible and self-powered photodetectors with ferroelectric P(VDF-TrFE)/perovskite bulk heterojunction. Adv. Funct. Mater. 2019, 29, 1808415.
128. Guan, S.; Cheng, C.; Ning, Y.; Zhang, B.; Qin, B.; Huang, B. Effect of metal-organic frameworks with different ligand structures (ZIF-11 & ZIF-23) on the optoelectronic performance of perovskite photodetectors. ACS. Appl. Mater. Interfaces. 2024, 16, 41341-50.
129. Yang, Y.; Yin, X.; Liu, X.; et al. Flexible photo-responsive nanofiber composites coupled triboelectric-photovoltaic effect for energy harvesting. Compos. Part. B. 2025, 307, 112879.
130. Guo, Q.; Yang, X.; Wang, Y.; Xu, W.; Duan, J.; Tang, Q. Dielectric hole collector toward boosting charge transfer of CsPbBr3 hybrid nanogenerator by coupling triboelectric and photovoltaic effects. Adv. Funct. Mater. 2021, 31, 2101348.
131. Sultana, A.; Alam, M. M.; Sadhukhan, P.; et al. Organo-lead halide perovskite regulated green light emitting poly(vinylidene fluoride) electrospun nanofiber mat and its potential utility for ambient mechanical energy harvesting application. Nano. Energy. 2018, 49, 380-92.
132. Lin, H.; Xia, Z.; Yao, H.; et al. Robustly stable perovskite-based triboelectric nanogenerators via grain-boundary manipulation for light-assisted energy harvesting and wireless control of smart windows. Nano. Energy. 2025, 144, 111355.
133. Kim, M.; Lee, S.; Cao, V. A.; Kim, M. C.; Nah, J. Performance enhancement of triboelectric nanogenerators via photo-generated carriers using a polymer-perovskite composite. Nano. Energy. 2023, 112, 108474.
134. Wu, W.; Xiao, Y.; Li, M.; Wei, Z.; Long, H.; Shen, G. A flexible bimodal self-powered optoelectronic skin for comprehensive perception of multiplexed sensoring signals. Nano. Energy. 2024, 125, 109593.
135. Venkatesan, M.; Chen, W. C.; Cho, C. J.; et al. Enhanced piezoelectric and photocatalytic performance of flexible energy harvester based on CsZn0.75Pb0.25I3/CNC-PVDF composite nanofibers. Chem. Eng. J. 2022, 433, 133620.
136. Zuo, L.; Guo, H.; deQuilettes, D. W.; et al. Polymer-modified halide perovskite films for efficient and stable planar heterojunction solar cells. Sci. Adv. 2017, 3, e1700106.
137. Han, T. H.; Lee, J. W.; Choi, C.; et al. Perovskite-polymer composite cross-linker approach for highly-stable and efficient perovskite solar cells. Nat. Commun. 2019, 10, 520.
138. Ma, L.; Yan, Z.; Zhou, X.; et al. A polymer controlled nucleation route towards the generalized growth of organic-inorganic perovskite single crystals. Nat. Commun. 2021, 12, 2023.
139. Yuan, Y.; Bi, Y.; Huang, J. Achieving high efficiency laminated polymer solar cell with interfacial modified metallic electrode and pressure induced crystallization. Appl. Phys. Lett. 2011, 98, 063306.
140. Yuan, Y.; Sharma, P.; Xiao, Z.; et al. Understanding the effect of ferroelectric polarization on power conversion efficiency of organic photovoltaic devices. Energy. Environ. Sci. 2012, 5, 8558-63.
141. Han, T. H.; Jang, K. Y.; Dong, Y.; Friend, R. H.; Sargent, E. H.; Lee, T. W. A roadmap for the commercialization of perovskite light emitters. Nat. Rev. Mater. 2022, 7, 757-77.
142. Xi, Y.; Zhang, X.; Shen, Y. et al. Aspect ratio dependent photocatalytic enhancement of CsPbBr3 in CO2 reduction with two-dimensional metal organic framework as a cocatalyst. Appl. Catal. B. 2021, 297, 120411.
143. Wan, S.; Ou, M.; Zhong, Q.; Wang, X. Perovskite-type CsPbBr3 quantum dots/UiO-66(NH2) nanojunction as efficient visible-light-driven photocatalyst for CO2 reduction. Chem. Eng. J. 2019, 358, 1287-95.
144. Yang, T.; Xie, D.; Li, Z.; Zhu, H. Recent advances in wearable tactile sensors: materials, sensing mechanisms, and device performance. Mater. Sci. Eng. R. 2017, 115, 1-37.
145. Wu, C.; Wang, A. C.; Ding, W.; Guo, H.; Wang, Z. L. Triboelectric nanogenerator: a foundation of the energy for the new era. Adv. Energy. Mater. 2019, 9, 1802906.
146. Xia, P.; Sun, H.; Xu, S.; et al. Luminescent solar concentrators with dual functions of photovoltaic and piezoelectric properties for wireless self-powered speed measurement. Adv. Energy. Mater. 2023, 13, 2301332.
147. Cao, X.; Xiong, Y.; Sun, J.; Zhu, X.; Sun, Q.; Wang, Z. L. Piezoelectric nanogenerators derived self-powered sensors for multifunctional applications and artificial intelligence. Adv. Funct. Mater. 2021, 31, 2102983.
148. Ma, X.; Wang, C.; Wei, R.; et al. Bimodal tactile sensor without signal fusion for user-interactive applications. ACS. Nano. 2022, 16, 2789-97.
149. Son, D.; Lee, J.; Qiao, S.; et al. Multifunctional wearable devices for diagnosis and therapy of movement disorders. Nat. Nanotechnol. 2014, 9, 397-404.
150. Wang, W.; Jiang, Y.; Zhong, D.; et al. Neuromorphic sensorimotor loop embodied by monolithically integrated, low-voltage, soft e-skin. Science 2023, 380, 735-42.
151. Peng, W.; Yu, R.; Wang, X.; et al. Temperature dependence of pyro-phototronic effect on self-powered ZnO/perovskite heterostructured photodetectors. Nano. Res. 2016, 9, 3695-704.
152. Chen, T. W.; Ramachandran, R.; Chen, S. M.; Anushya, G.; Ramachandran, K. Graphene and perovskite-based nanocomposite for both electrochemical and gas sensor applications: an overview. Sensors 2020, 20, 6755.
153. Li, W.; Jia, J.; Sun, X.; Hao, S.; Ye, T. A light/pressure bifunctional electronic skin based on a bilayer structure of PEDOT:PSS-coated cellulose paper/CsPbBr3 QDs film. Polymers 2023, 15, 2136.
154. Khan, A. A.; Rana, M. M.; Huang, G.; et al. Maximizing piezoelectricity by self-assembled highly porous perovskite-polymer composite films to enable the internet of things. J. Mater. Chem. A. 2020, 8, 13619-29.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].