Mechanism-guided high-throughput design of Al-Cr-Ti-Si-N coatings achieving synergistic thermal stability and oxidation resistance
Abstract
Transition metal nitride coatings often face a trade-off between thermal stability and oxidation resistance at high temperatures. Here, we address this challenge using a mechanism-guided high-throughput combinatorial strategy, implemented through multi-target co-deposition, to rapidly probe the Al-Cr-Ti-Si-N compositional space. An optimal composition window was identified (Al 13.3-24.1 at.%, Cr 7.6-14.6 at.%,
Keywords
INTRODUCTION
Advances in high-end manufacturing increasingly expose tools and molds to harsher service environments, imposing more stringent performance demands on protective coatings. Transition metal nitride coatings deposited by physical vapor deposition (PVD) are indispensable in critical applications, such as high-speed dry cutting and die-casting molds, owing to their high hardness and excellent wear resistance[1-3]. However, under such service conditions, the combined effects of high temperature, mechanical wear, and aggressive oxidation alter the mechanisms by which coatings fail. Mechanical failure is no longer the sole dominant mode. Instead, high-temperature microstructural instabilities (e.g., phase transformations, atomic diffusion, grain coarsening) and detrimental chemical reactions with the environment, particularly oxidation, govern coating degradation[4-6]. Consequently, high-temperature stability and oxidation resistance have become decisive performance metrics, critically determining coating service life and limiting the development of advanced manufacturing technologies.
To address these challenges, coating systems have evolved from simple binary nitrides to ternary and quaternary systems, such as AlCrN[6,7], TiSiN[8], and AlCrSiN[9]. Multi-element alloying improves performance via solid solution strengthening, grain refinement, and the promotion of protective oxide formation, thereby enhancing mechanical and chemical durability[10]. Nonetheless, a fundamental trade-off between thermal stability and oxidation resistance persists. For instance, while a high Al content promotes the formation of a dense Al2O3 scale that improves oxidation resistance, it also predisposes the coating to precipitate brittle hexagonal close-packed (hcp) AlN and triggers the decomposition of the CrN-rich matrix into Cr2N/Cr, degrading toughness and thermal stability[10,11]. Introducing silicon offers a partial solution: silicon exceeds its solubility limits in the nitride matrix, promoting the formation of an amorphous silicon nitride (a-Si3N4) phase along grain boundaries. This results in a nanocomposite structure consisting of face-centered cubic (fcc) MeN (Me = Cr, Al) nanocrystallites embedded within an a-Si3N4 matrix[12,13], which concurrently enhances oxidation resistance and thermal stability to some extent[10,11]. However, even these advanced coatings undergo mechanical degradation above 900 °C due to fcc→hcp/body-centered cubic (bcc) phase transformations and grain coarsening[14]. These findings underscore that precise control over the concentrations of key elements, such as Al, Cr, Ti, and Si, is essential for overcoming this performance trade-off and achieving coatings with superior high-temperature stability and oxidation resistance.
In our previous work, we developed an Al11.3Cr8.1Ti26.9Si3.6N50.1 coating featuring a single-phase fcc solid solution structure. This coating exhibited exceptional thermal stability, maintaining its structure at 1,000 °C, and remarkable oxidation resistance via the formation of a dense protective oxide scale[15]. Its performance is markedly superior to that of an earlier Al39.9Cr4.9Ti8.1Si3.1N coating, which had a nanocomposite architecture of fcc nanocrystallites embedded in an amorphous a-SixNγ matrix[16]. Although this "nanocrystalline + amorphous" structure initially provided good thermal resistance, it underwent detrimental fcc→hcp transformation upon annealing above 900 °C, leading to substantial mechanical degradation[16]. These results highlight the exquisite sensitivity of multicomponent coatings' high-temperature performance to their precise composition and stoichiometry. The Al-Cr-Ti-Si-N system encompasses a vast combinatorial space, with millions of potential compositions. Given this complexity, traditional trial-and-error approaches are highly inefficient and inadequate for establishing systematic composition-structure-property relationships, thereby hindering the rational design and optimization of such coatings.
To overcome these limitations, this study introduces a mechanism-guided high-throughput combinatorial design strategy. A library of AlCrTiSiN coatings with continuous composition gradients was synthesized via multi-target co-deposition. High-throughput screening of the compositional space was achieved through nanoindentation, multi-scale structural characterization, and elevated-temperature performance testing. This process identified an optimal compositional window that delivers a unique combination of ultra-high hardness (> 32 GPa), excellent thermal stability (no fcc-phase decomposition at 1,000 °C), and outstanding oxidation resistance (oxide scale thickness < 0.5 μm after oxidation at 1,000 °C). The enhanced performance originates from multi-scale synergistic mechanisms. At the nanoscale, spinodal decomposition creates Ti-rich and Al/Cr-rich domains, complemented by high-density dislocations and lattice distortion, together conferring exceptional mechanical and thermal properties. Furthermore, during oxidation, an in situ "sandwich" oxide structure forms, comprising an inner amorphous SiO2 layer and an intermediate dense (Al,Cr)2O3 layer. This structure synergistically blocks both inward oxygen diffusion and outward cation migration, drastically improving oxidation resistance. Machining tests under high-speed dry cutting conditions validated its superior engineering applicability. This work not only overcomes the performance trade-off between thermal stability and oxidation resistance, providing a new material solution for extreme conditions, but also establishes an effective pathway from "empirical exploration" to "rational design", offering significant methodological insights and theoretical support for developing high-performance multicomponent coatings.
MATERIALS AND METHODS
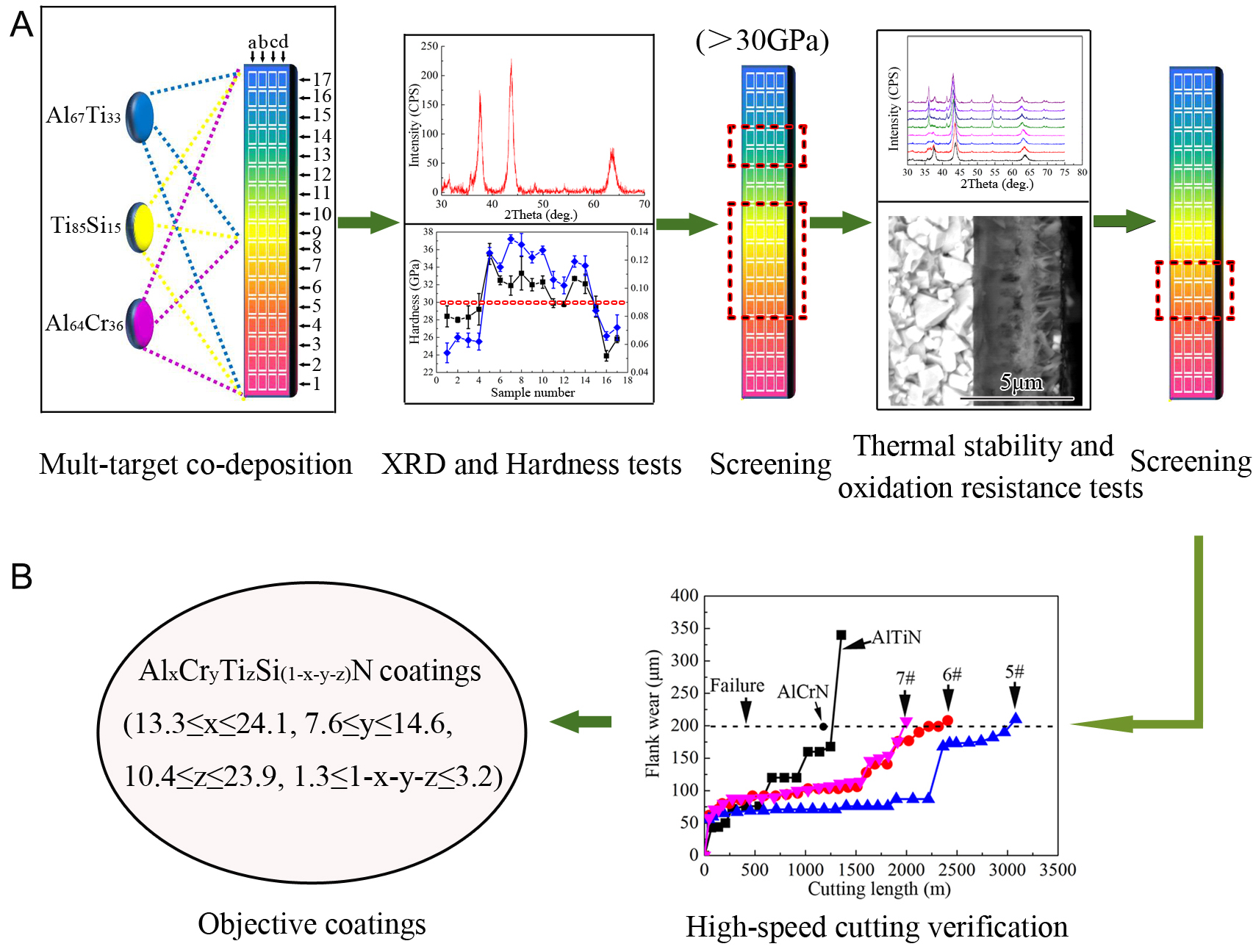
Commercial YG8 cemented carbides (WC-8Co, 10 mm × 15 mm × 5 mm) with an average surface roughness (Ra) of 0.05 μm were mounted on a custom-designed sample holder (500 mm × 69 mm × 69 mm). The holder configuration comprised four vertical columns [Figure 1A], each accommodating seventeen substrate plates at 20 mm intervals. The assembly was loaded into a Hauzer Flexicat 850 cathodic vacuum arc system equipped with three columnar target holders, each containing three 100 mm-diameter alloy targets. Among them, one column holder was installed by Al64Cr36 to produce a buffer layer, while the remaining two holders contained three alloy targets with different compositions to deposit AlxCryTizSi(1-x-y-z)N coatings.
Figure 1. Schematic diagrams of the deposition and characterization workflow. (A) Preparation and screening of coatings. Coatings with varying compositions were first deposited via multi-target co-deposition, followed by phase and hardness screening based on XRD and hardness tests to select coatings with an FCC solid solution and hardness above 30 GPa. The selected coatings were then further evaluated for thermal stability and oxidation resistance through annealing and oxidation tests at elevated temperatures. (B) Performance verification and optimization of coatings. The heat- and oxidation-resistant coatings were further assessed by high-speed cutting tests in comparison with conventional AlCrN- and AlTiN-coated tools, and the optimal coatings were finally identified based on the overall performance.
Prior to coating deposition, the chamber was evacuated to a base pressure of 5.0 × 10-4 Pa while the substrates were radiantly heated to 450 °C. The workpiece turntable rotated at 3 rpm throughout the process. To enhance the adhesion strength of AlxCryTizSi(1-x-y-z)N coatings, an AlCrN buffer layer was deposited using the Al64Cr36 target column. Multilayered AlxCryTizSi(1-x-y-z)N coatings were synthesized through multi-target co-deposition using the three distinct alloy targets: Al64Cr36, Ti85Si15, and Al67Ti33. Deposition parameters are shown in Table 1. For clarity, samples in column x were labeled as No. x (e.g., the first column was labeled No. 1). Then, selected as-deposited coatings underwent isothermal annealing at 900 °C for 2 h in an argon-protected environment, while others were subjected to isothermal oxidation at 1,000 °C for 2 h under ambient atmospheric conditions.
Deposition parameters for AlxCryTizSi(1-x-y-z)N coatings
| Procedure | Target | Target current (A) | Substrate bias voltage (V) | Deposition temperature (°C) | Gas pressure (Pa) | Deposition time (min) |
| 1 | Al64Cr36 | 150 | -40 | 450 | 3 | 10 |
| 2 | Al64Cr36 | 150 | -60 | 110 | ||
| Ti85Si15 | 140 | |||||
| Al67Ti33 | 130 |
Nanohardness (H) and elastic modulus were measured by an MTS XP nanoindenter with a Berkovich diamond tip. Phase structure was identified via X-ray diffraction (Bruker D8 Advance, Cu Kα radiation,
Based on hardness, thermal stability, and oxidation resistance results, coating compositions were efficiently screened to develop target AlxCryTizSi(1-x-y-z)N coatings that simultaneously achieve high hardness (> 30 GPa), exceptional thermal stability (retaining a fcc structure at elevated temperatures), and superior oxidation resistance (formation of a thin and dense oxide layer at high temperature), as illustrated in Figure 1A. The optimized coatings were subsequently applied to cutting tools, which were tested in high-speed dry cutting experiments on martensitic precipitation hardening stainless steel (average hardness 47 HRC) under aggressive machining conditions: cutting speed 180 m/min, feed rate 0.2 mm/r, and cut depth 2.0 mm. A flank wear threshold of 0.2 mm was used to define tool lifetime. The high-speed cutting verification results are shown in Figure 1B.
RESULTS
Elemental composition
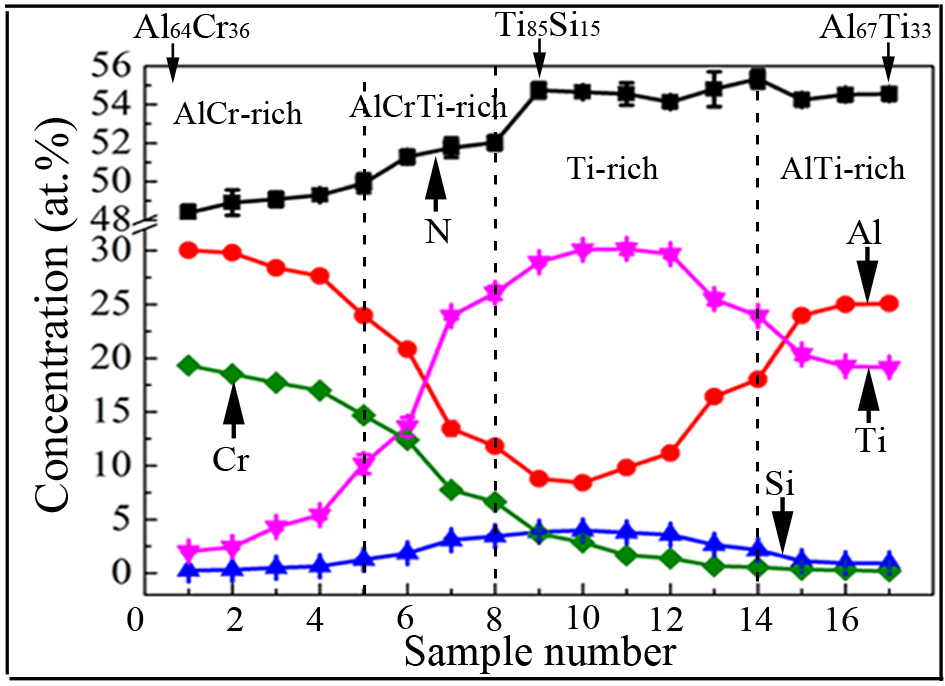
The elemental profiles of the AlxCryTizSi(1-x-y-z)N coatings were analyzed by EDS point analysis. The results show continuous compositional gradients from sample No. 1 to No. 17, confirming the successful synthesis of compositionally graded coatings via the synchronized co-deposition of Al64Cr36, Ti85Si15, and Al67Ti33 targets. Such precise modulation demonstrates effective control over the multi-arc cathode configuration, which facilitates atomic-scale inter-doping during deposition. The compositional profiles [Figure 2] reveal four distinct compositional zones in the AlxCryTizSi(1-x-y-z)N coatings: (i) an AlCr-dominant zone (Nos. 1-4) exhibiting progressive decreases in Al/Cr concentrations accompanied by gradual Ti enrichment; (ii) an AlCrTi transitional zone (Nos. 5-8) characterized by sharp decreases in Al/Cr content and rapid Ti accumulation; (iii) a Ti-saturated zone (Nos. 9-14) maintaining consistently elevated Ti levels; (iv) an AlTi-enriched terminal zone (Nos. 15-17) showing co-dominance of Ti and Al with stable concentrations.
Phase structure analysis
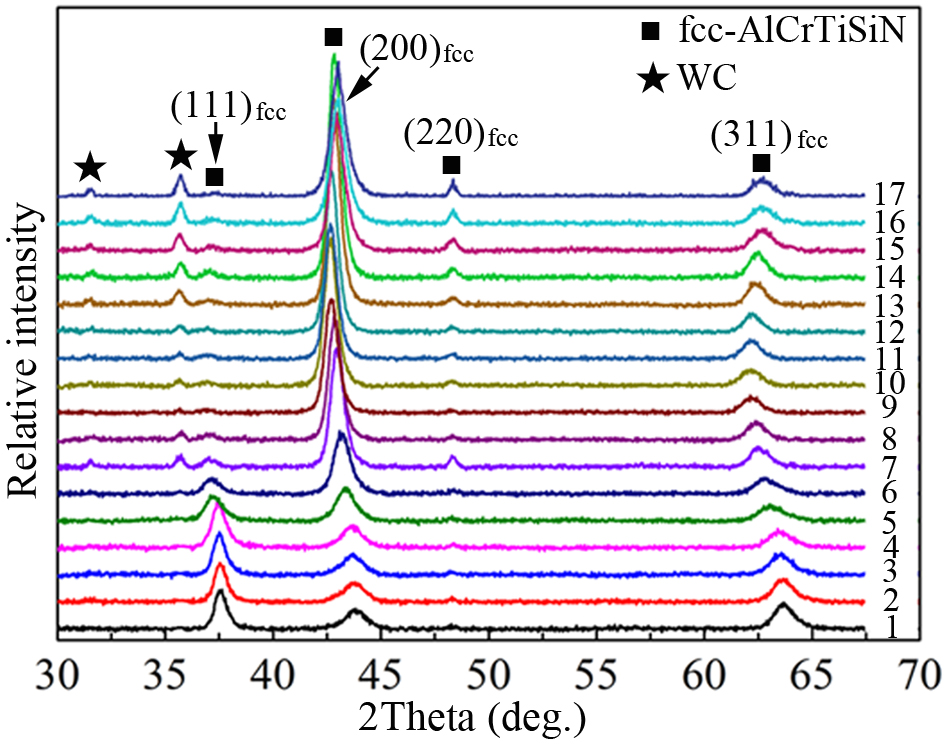
The XRD patterns in Figure 3 reveal the critical structural features of the as-deposited coatings (Nos. 1-17). All coatings exhibit a single-phase fcc-AlCrTiSiN solid solution structure, with no detectable hcp-AlN phase. The persistent diffraction peaks from the WC substrate indicate that the X-ray beam penetrated through the coating thickness during measurement. The coating series exhibits a clear composition-dependent crystallographic evolution. From sample No. 1 to No. 17, the intensity of the (111)fcc peak decreases, while that of the (200)fcc peak increases. This systematic variation indicates a texture transition from predominant (111)fcc to dominant (200)fcc orientation along the growth direction. Furthermore, the diffraction peaks of the fcc-AlCrTiSiN solid solution gradually shift toward lower angles from sample No. 1 to No. 11, followed by a shift toward higher angles. This trend indicates that the lattice parameter of the fcc-AlCrTiSiN solid solution initially increases and subsequently decreases. Previous studies have reported that the lattice parameter of TiN is larger than that of CrN[17]. Therefore, the observed variation in lattice parameter is attributed to changes in Ti concentration within the fcc-AlCrTiSiN solid solution.
Microstructural analysis by TEM (HRTEM)
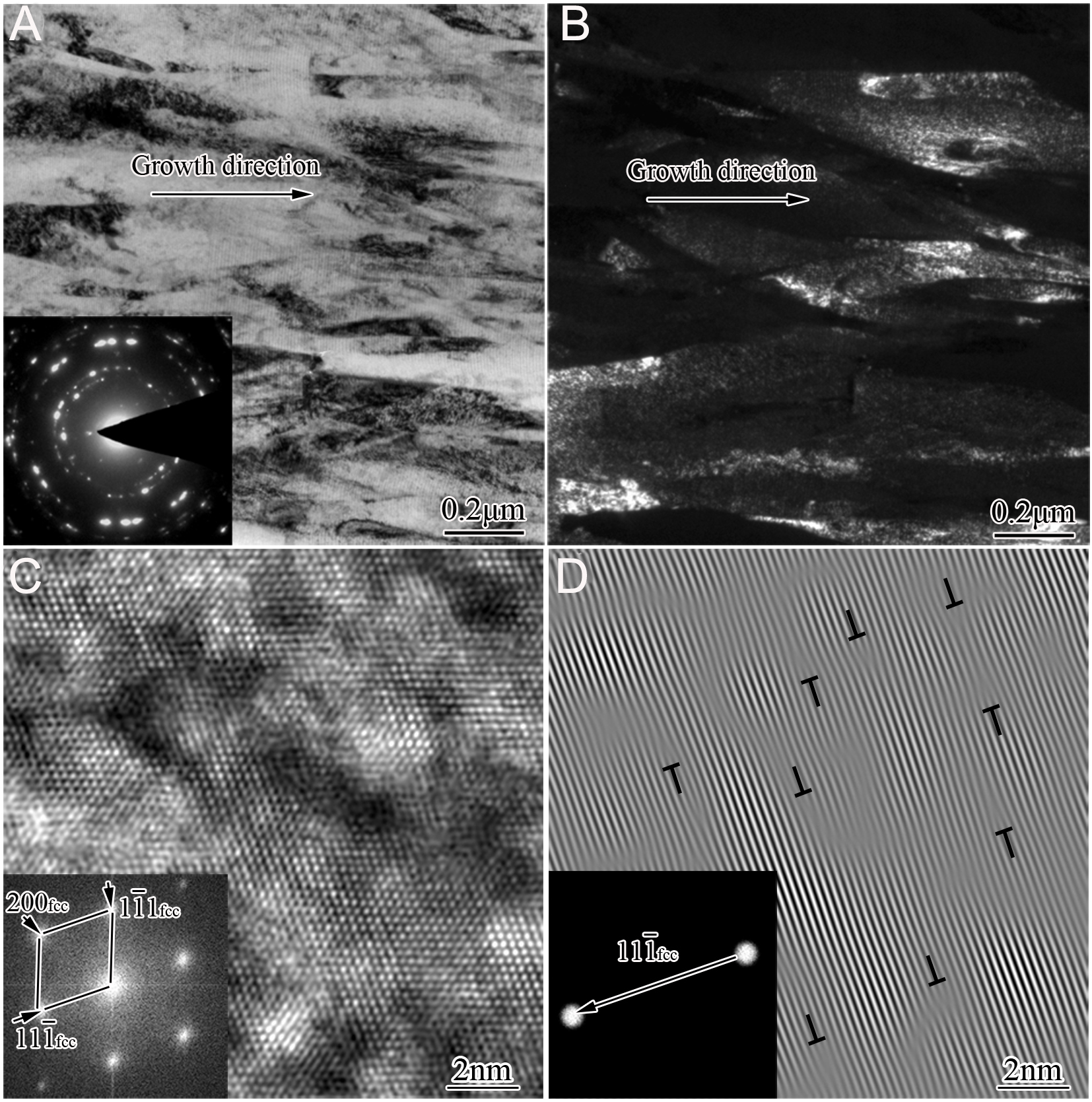
Figure 4A presents a cross-sectional TEM overview of the as-deposited No. 3 coating. Coarse columnar grains extend throughout the entire coating thickness. The corresponding selected area electron diffraction pattern (SAEDP) exhibits discontinuous polycrystalline rings with randomly distributed diffraction spots, confirming that the columnar grains possess a fcc-AlCrTiSiN solid solution structure. The corresponding centered dark-field (CDF) image [Figure 4B] resolves individual columnar grains, with widths ranging from 50 to 300 nm and lengths extending up to several micrometers. Further high-resolution TEM analysis [Figure 4C] verifies a single-phase fcc-AlCrTiSiN structure, as evidenced by continuous lattice fringes with minimal distortion. The inverse fast Fourier transform (IFFT) reconstruction in Figure 4D, acquired along the [011]fcc zone axis, reveals discrete lattice dislocations preferentially aligned along the (111)fcc crystallographic planes.
Figure 4. Cross-sectional TEM and HRTEM images of the as-deposited No. 3 sample: (A) Bright-field image with corresponding SAEDP; (B) CDF image viewed along the g = 200 reflection; (C) HRTEM image with corresponding FFT; (D) IFFT image viewed along the g = [111] direction.
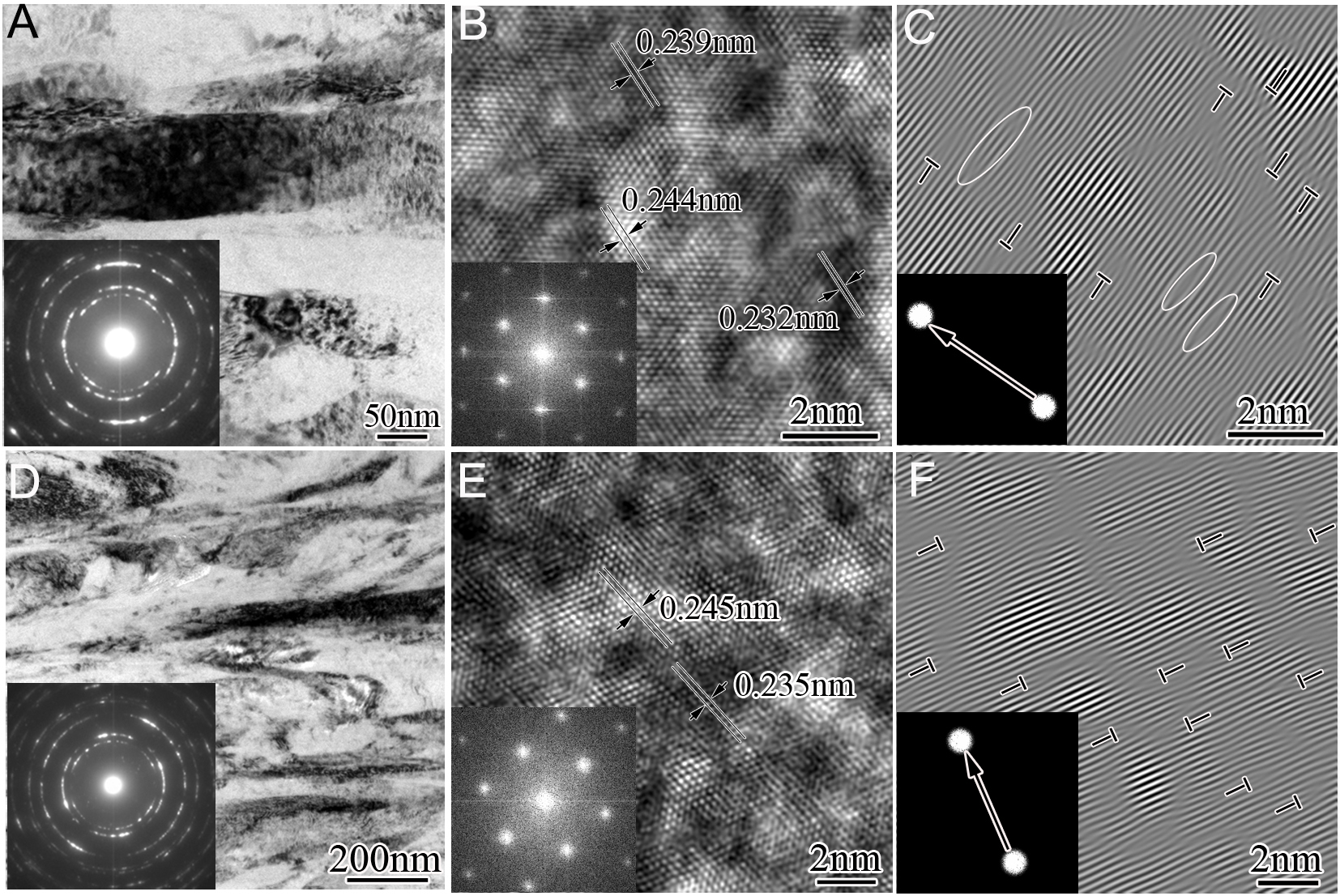
Figure 5A presents a cross-sectional CDF overview of the as-deposited coating No. 5 coating. Fine, less-developed columnar grains are observed, with diameters ranging from 20 to 150 nm and lengths of several micrometers. The corresponding SAEDP (inset in Figure 5A) displays only continuous polycrystalline diffraction rings, confirming that these fine columns possess a fcc-AlCrTiSiN solid solution structure. Comparative analysis demonstrates grain refinement in sample No. 5 relative to sample No. 3 under equivalent sampling areas. EDS-elemental mapping [Figure 5B] confirms a homogeneous micrometer-scale distribution of the constituent elements (Al, Cr, Ti, and Si). Atomic-scale HRTEM observations [Figure 5C] reveal bimodal contrast variations featuring 1-3 nm nanodomains. Crystallographic analysis identifies distinct d-spacings: bright regions exhibit expanded (111)fcc planes (0.242 nm) compared to darker areas (0.234-0.237 nm), suggesting Ti-enriched and AlCr-rich fcc solid solution phases, respectively. This significant lattice mismatch (Δa = 1.7%-3.4%) between these phases induces high-density dislocation networks [Figure 5D] and pronounced lattice distortions, potentially amplified by elevated Ti concentrations. Further geometric phase analysis (GPA) images [Figure 5E and F] illustrate pronounced atomic-scale coherency strain at the Ti- and AlCr-rich domains, strongly supporting a spinodal mechanism. Extended TEM investigations of samples No. 9 and No. 14 [Figure 6], employing similar analytical methods, confirm a preserved columnar architecture [Figure 6A and D] and retained fcc structure (SAED insets in Figure 6B and E). Atomic-resolution imaging [Figure 6C and F] reveals persistent compositional partitioning into Ti/AlCr-enriched nanodomains, accompanied by severe lattice strain, demonstrating structural consistency across the coating series. However, comparison of the SAEDPs in Figure 4A, Figure 5A,
Figure 5. Typical cross-sectional TEM and HRTEM images of sample No. 5: (A) CDF image viewed along the g = 200 reflection with indexed SAEDP inset; (B) EDS mapping of Al, Cr, Ti, and Si within the region in (A); (C) HRTEM image of an fcc grain in (A) with FFT inset; (D) IFFT image of (C) viewed along the g = (11
Nano-mechanical test
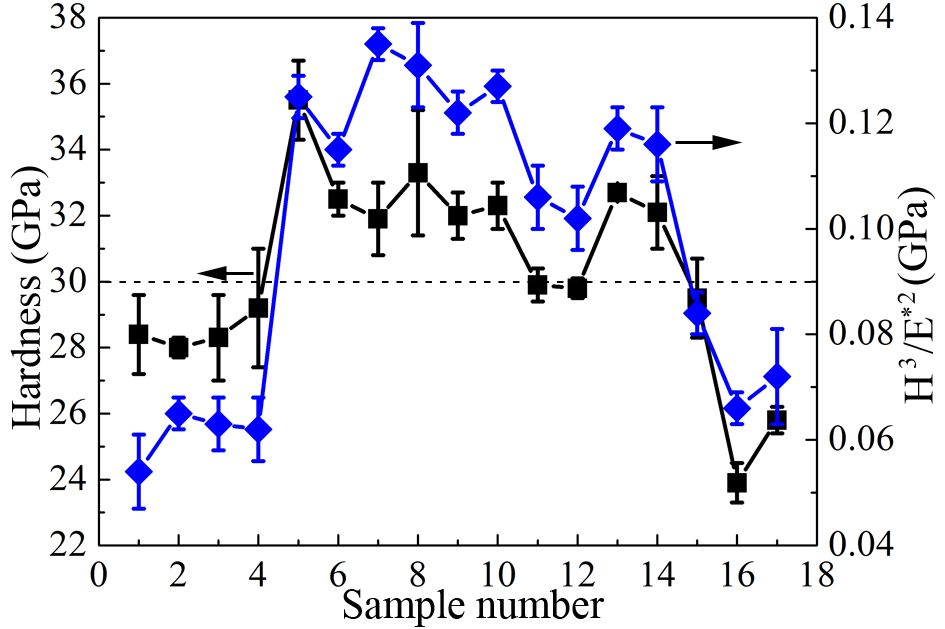
Nano-hardness characterization based on load-displacement curves reveals significant variations in hardness (H) and plasticity indices (H3/E*2) across the coating series [Figure 7]. These variations correlate well with the observed microstructural evolution. The elevated H values indicate improved abrasive wear resistance, while the increased H3/E*2 ratios suggest enhanced resistance to plastic deformation[18]. Atomic-scale HRTEM analysis confirms the solid solution strengthening effect of Si incorporated into the fcc lattice. This effect acts synergistically with the solute elements of Al, Cr, and Ti. Coatings Nos. 1-4, classified as Ti/Si-modified AlCrN systems, exhibit hardness values (22-28 GPa) comparable to those of conventional AlCrN references[19,20]. Although densely packed (111)fcc planes are theoretically expected to provide superior plastic resistance, the H3/E*2 values for these samples are relatively low [Figure 8]. This is predominantly attributed to residual compressive stresses induced by the relatively thick coatings
The mechanical screening protocol identified coatings Nos. 5-10 and 13-14 as exceeding the critical hardness threshold (> 30 GPa), thereby qualifying these systems for comprehensive evaluation of elevated-temperature performance.
High-temperature properties
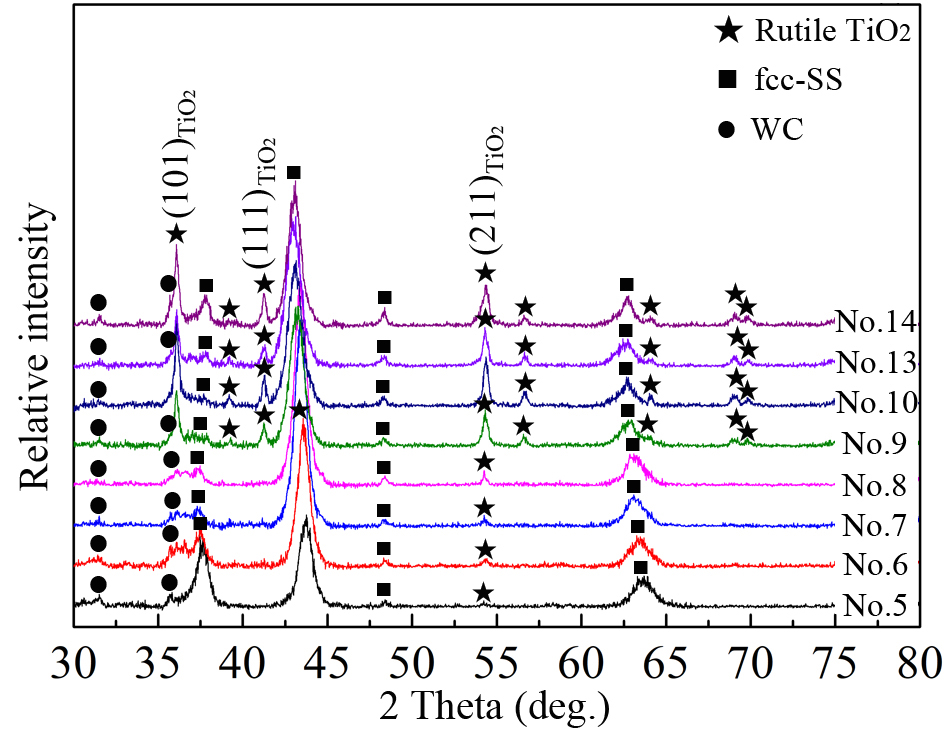
Figure 8 presents the phase evolutions of selected coatings (Nos. 5-10 and Nos. 13-14) after isothermal annealing at 900 °C for 2 h. Samples Nos. 5-8 retain a predominantly intact fcc-AlCrTiSiN solid solution structure. The presence of only a trace amount of rutile TiO2 is evidenced by weak (211)TiO2 reflections, which originate from residual oxygen in the annealing environment. In contrast, samples Nos. 9-10 and 13-14 exhibit a marked intensification of the TiO2 phase, as indicated by the pronounced growth of the (211)TiO2 and (111)TiO2 peaks. Notably, all annealed systems demonstrate complete suppression of hcp-AlN, hcp-Cr2N, and bcc-Cr phase decomposition products. This behavior contrasts sharply with conventional AlCrN systems, which degrade at 700 and 800 °C[21,22]. The observed improvement in thermal stability, exceeding 100-200 °C, demonstrates the superior high-temperature performance of the developed coatings.
To characterize the structural evolution within the oxidation layers, grazing incidence X-ray diffraction (GIXRD) measurements were performed at an incident angle of 2°. According to calculations by Gullikson[23], the probing depth at this angle is approximately 0.26 μm, which is smaller than the total thickness of the oxide layer. Therefore, the GIXRD signals originate primarily from the oxidation layer, ensuring that the obtained structural information is representative of this region.
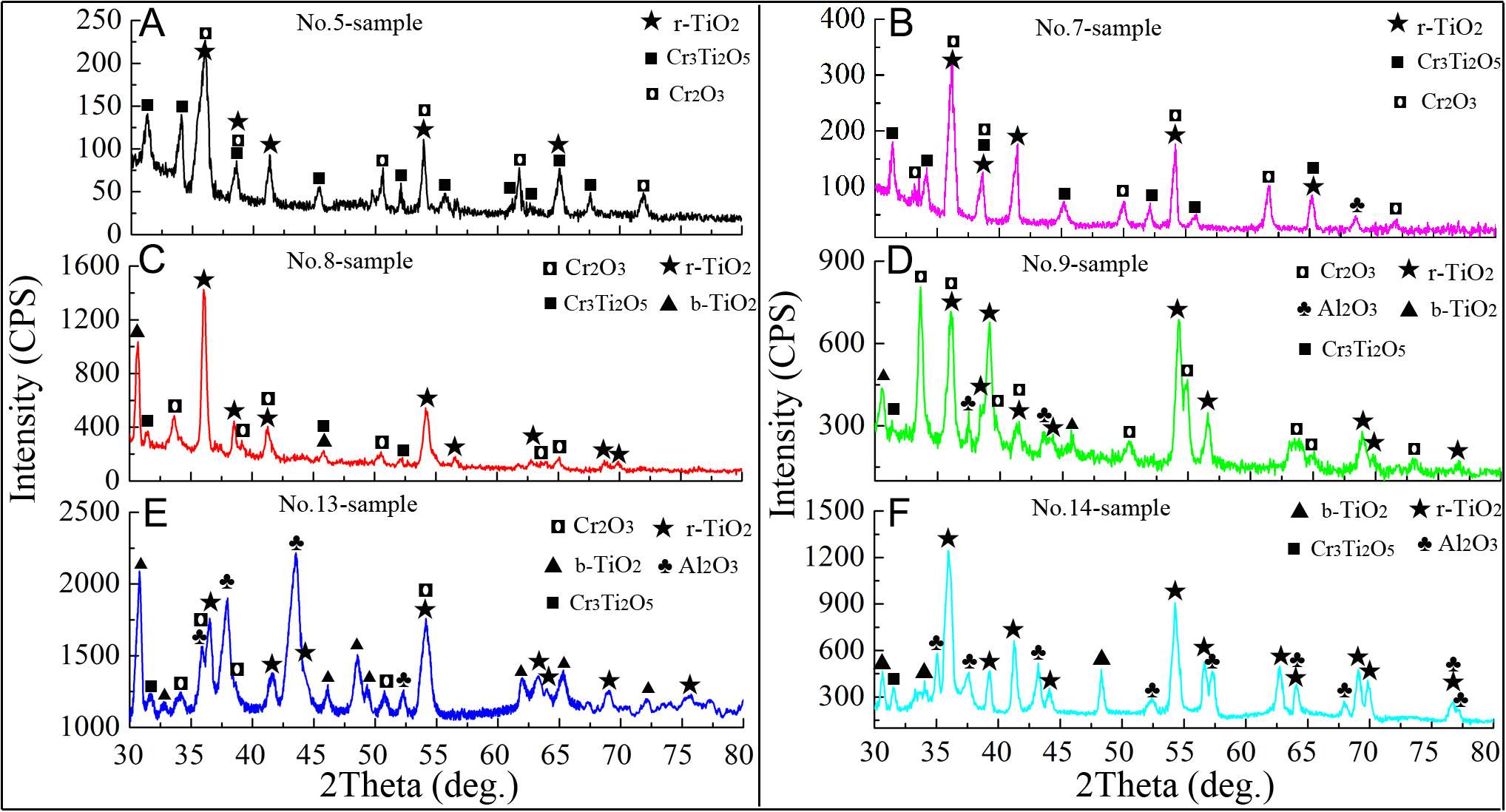
Figure 9 presents the phase structures of the experimental samples after isothermal oxidation at 1,000 °C for 2 h. The results indicate that the coating composition strongly influences phase formation within the oxidation layers. According to GIXRD analysis, the oxide scales on AlCrTi-rich coatings (Nos. 5-7) are mainly composed of rutile TiO2 (r-TiO2), Cr3Ti2O5, and Cr2O3. This phase assemblage differs from that of the Ti0.32Al0.38Cr0.30N coating oxidized at 1,000 °C, which contains r-TiO2, Al2O3, and Cr2Ti7O17, as well as from the Ti0.17Al0.19Cr0.64N coating, in which only TiO2 and Cr2O3 phases were detected[24]. As the TiSi concentration increases and the AlCr concentration decreases, significant changes occur in the oxide phases of Ti-rich coatings (Nos. 8-9). These oxides primarily consist of r-TiO2, brookite TiO2 (b-TiO2), and Cr2O3, along with minor amounts of Cr3Ti2O5. In the AlTi-rich coatings, the oxide layer of sample No. 13 is dominated by r-TiO2, b-TiO2, and Al2O3, with additional Cr2O3 and Cr3Ti2O5. A similar phase composition is observed in sample No. 14, though no Cr2O3 is detected. These findings indicate that under conditions of high Ti/Cr concentrations, Cr atoms combine with Ti/O atoms to form Cr3Ti2O5, retarding the formation of loose TiO2, and eventually enhancing the densification of the oxide layer.
Figure 9. Typical XRD spectra of selected samples oxidized at 1000 °C for 2 h: (A) No. 5-sample; (B) No. 7-sample; (C) No. 8-sample; (D) No. 9-sample; (E) No. 13-sample; (F) No. 14-sample.
To further elucidate the oxidation behavior, the cross-sectional microstructures of the oxide layers formed on the samples were examined by SEM combined with EDS [Figure 10]. Coatings Nos. 5-7 develop exceptionally dense and continuous oxide scale with thicknesses below 0.5 μm, as precisely measured from cross-sectional SEM images [Figure 10A and B]. O-specific elemental mapping [Figure 10C] further confirms the continuity and compactness of the oxide layers, showing no detectable porosity or interfacial delamination. The oxide thicknesses of these coatings are significantly smaller than those of conventional AlCrN (1.5 μm)[11], AlTiN (5 μm)[11], Ti0.32Al0.38Cr0.30N (2.5 μm)[24], and Ti0.17Al0.19Cr0.64N (1 μm)[24] coatings oxidized at 1,000 °C for 2 h, as well as nano-multilayered AlTiN/TiSiN (2 μm)[25] and AlCrN/TiSiN
Figure 10. Typical cross-sectional SEM/BSE images and EDS elemental maps of oxide layers in selected oxidized coatings: (A)
In contrast, the Ti-rich coatings (Nos. 8-9) develop thicker oxide layers ranging from 2 to 3.5 μm
A similar “sandwich” structure is also observed in sample No. 13 [Figure 10G and H], although each sublayer is thicker and contains a high density of voids, particularly within the Ti-rich layer and at the interface between the coating and the Si-rich layer. In sample No. 14, these voids coalesce and develop into cracks [Figure 10I]. This failure mode is likely attributable to the mismatch in the coefficients of thermal expansion between the oxide scale and the underlying nitride coating. Similar interfacial cracks have been reported in AlTiN/TiSiN coatings oxidized at 900 °C for 2 h[26].
Combined XRD and EDS results indicate that samples Nos. 5-7 possess significantly better oxidation resistance than samples Nos. 8-9. Our previous work has further demonstrated that their outstanding oxidation resistance is due to the “sandwich” structure consisting of an inner amorphous SiO2 layer and an intermediate dense (Al,Cr)2O3 layer, which effectively prevent the inward diffusion of O and outward diffusion of Ti[15]. In addition, a synergistic effect between Ti and Cr promotes the formation of Cr3Ti2O5, which retards the formation of a porous TiO2 layer, thereby contributing to improved oxidation resistance. However, the mechanisms behind the inferior oxidation resistance of samples Nos. 8-9 remain unclear. To elucidate this, sample No. 9 was selected for detailed TEM (and HRTEM) analysis.
Figure 11A shows a cross-sectional TEM image of the oxidation layer formed on sample No. 9 after oxidation at 1,000 °C for 2 h. The oxidized coating is clearly divided into three distinct sublayers: an inner “A” sublayer, an intermediate “B” sublayer, and an outermost “C” sublayer. The indexed SAEDP from region “A” (inset in Figure 11A) shows polycrystalline diffraction rings with randomly distributed spots, confirming that this innermost region retains an fcc-AlCrTiSiN solid solution structure despite oxidation, thereby demonstrating exceptional thermal stability. Furthermore, the lattice fringes appear significantly straighter than those in the as-deposited coating, which can be attributed to defect annihilation and stress relaxation at high temperatures[27,28]. EDS mapping analysis also reveals the presence of Cr-rich domains at the interface between the A and B sublayers [Figure 11B].
Figure 11. (A) Cross-sectional TEM image of sample No. 9 oxidized at 1,000 °C, with the inset showing the SAEDP obtained from region “A”; (B) EDS mapping of Al, Cr, Ti, Si, N, and O in the region outlined in (A); (C) HRTEM image of a representative zone within the “A” sublayer; (D) High-magnification TEM image of a region in the “B” sublayer; (E) HRTEM image of a Cr-rich domain at the “A/B” interface, with the corresponding FFT pattern shown in the inset; (F-G) HRTEM images of Cr2N, fcc-AlCrTiSiN, Ti2N, and TiO2 grains within the “B” sublayer; (H) TEM image of coarse TiO2 grains, with indexed SAEDP and EDS point analysis shown in the insets.
Closer examination shows that the “B” sublayer is enriched in Si, while the “C” sublayer is rich in Al but depleted in Cr, exhibiting a dendritic morphology indicative of dendritic (Al,Cr)2O3 grains. High-magnification TEM imaging reveals that the “B” sublayer consists of ultrafine equiaxed grains
Verification experiment of high-speed dry cutting
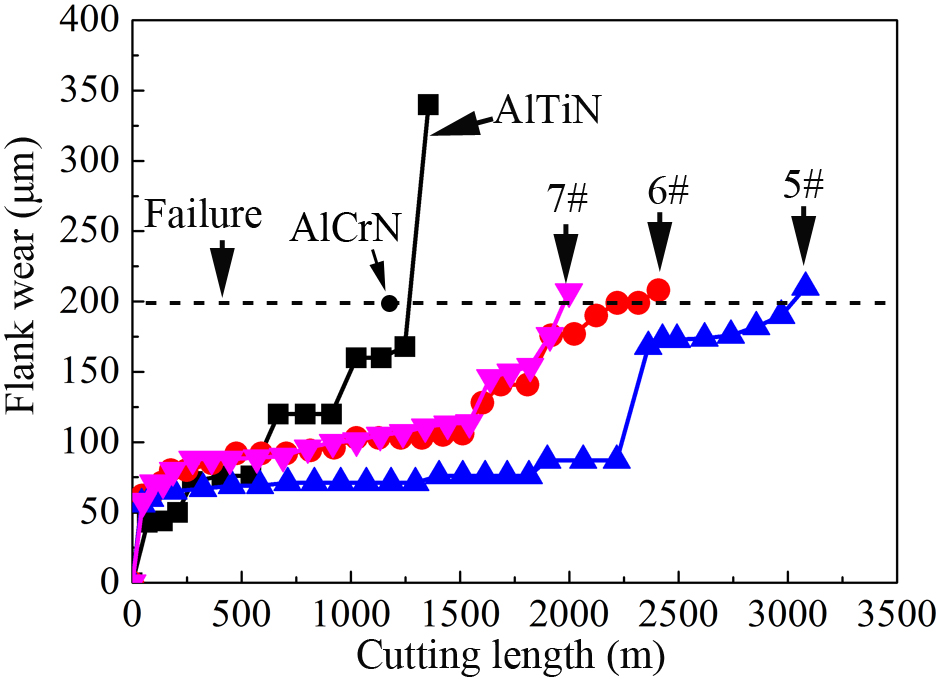
High-speed dry machining of martensitic precipitation-hardened stainless steel (PH17-4) involves extreme service conditions, with operational temperatures reaching 900-1,000 °C[29]. These conditions necessitate coatings that possess both high hardness and outstanding durability at elevated temperatures. Based on these requirements, coatings Nos. 5-7 were selected for machining performance evaluation, as they demonstrate superior hardness, thermal stability, and high-temperature oxidation resistance. As illustrated in Figure 12, these engineered coatings achieved remarkable tool life extensions, with cutting lengths of 3,081, 2,409, and
Figure 12. Tool life comparison under high-speed dry cutting conditions. The four curves represent the evolution of flank wear as a function of cutting length for the optimized AlCrTiSiN coatings (No. 5, No. 6, and No. 7) and a commercial AlTiN coating. The data points (circles, squares, and triangles) correspond to individual wear measurements for each coating type.
DISCUSSION
Thermal stability analysis
The current understanding of decomposition mechanisms in PVD coatings identifies two predominant pathways: spinodal decomposition and recrystallization-driven phase transformation[26]. These degradation processes are compositionally dependent, with critical Al concentration thresholds (0.08 ≤ x ≤ 0.92) dictating the dominant mechanism through variations in Gibbs free energy[26]. The established thermal degradation sequence typically progresses as follows. First, spinodal phase separation occurs, forming Al-rich and metal-enriched fcc domains, which results in age hardening. Subsequently, a recrystallization-mediated transformation converts the metastable fcc phase into stable hcp-AlN, accompanied by a catastrophic loss of hardness[27,30]. Concurrent nitrogen depletion initiates decomposition in Cr-containing systems, transforming the Cr-rich fcc phase into bcc-Cr via an intermediate hcp-Cr2N phase[31,32]. This multi-step transformation fundamentally alters the microstructure, changing it from a columnar to an equiaxed grain morphology. Monitoring these crystallographic transitions (fcc→hcp/bcc) and morphological evolutions (columnar→equiaxed) provides critical indicators for assessing coating thermal stability.
Multi-scale characterization reveals exceptional thermal stability in the developed AlxCryTizSi(1-x-y-z)N coatings. As-deposited coatings exhibit microscale homogeneity, characterized by columnar fcc-AlCrTiSiN grains with uniform elemental distributions, as well as nanoscale spinodal decomposition, reflected by self-organized Ti-rich and Al(Cr)-rich fcc domains (1-3 nm) after cooling in the PVD chamber, accompanied by numerous interfacial dislocations. It has been reported that metastable fcc-AlxCr1-xN could decompose into nanosized Cr-rich and Al-rich fcc domains with a small molar volume mismatch[26], indicating the formation of interfacial dislocations at neighboring domains, in which is consistent with our findings. Spinodal decomposition-induced phase separation has also been observed in Cr-Si-N[33] and Ti-Si-N[34] systems. Based on the XRD results after annealing at 900 °C, the AlxCryTizSi(1-x-y-z)N coatings (samples Nos. 5-9 and Nos. 13-14) remain a fcc solid solution, showing a columnar morphology, without hcp-AlN, bcc-Cr2N, or bcc-Cr phases. Furthermore, regions of the coatings far from oxidation zones remain composed of a single-phase fcc-AlCrTiSiN solid solution even after oxidation at 1,000 °C (equivalent to annealing at 1,000 °C). This demonstrates that the coatings can withstand annealing at1,000 °C. In contrast, the fcc→hcp phase transformation has been observed in traditional AlCrN coatings annealed at 800 °C[35], CrN/CrTiSiN coatings annealed at 600 °C[36], and nanocomposite AlCrTiSiN coatings annealed at 900 °C[27], all accompanied by a transition from columnar to equiaxed morphologies. These comparisons indicate that the optimized AlxCryTizSi(1-x-y-z)N coatings reveal excellent thermal stability as compared to the traditional transition metal nitride coatings.
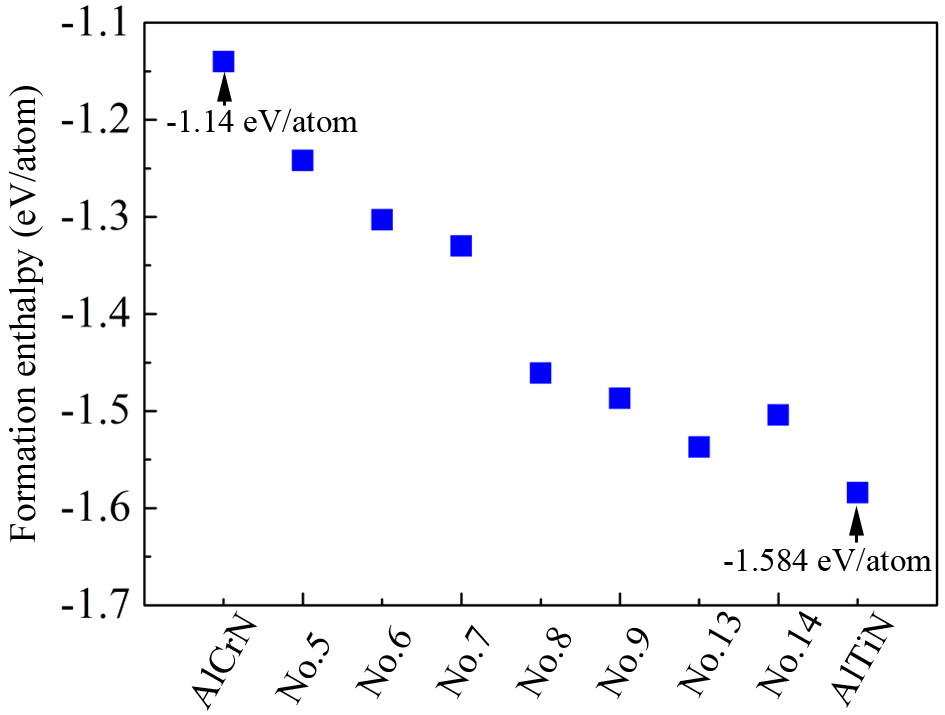
The AlxCryTizSi(1-x-y-z)N coatings are categorized as multicomponent transition metal nitrides[37,38]. Their thermal stability is analyzed from the following perspectives. First, according to the Open Quantum Materials Database (OQMD)[39-41], the formation energies of coatings Nos. 5-9 and Nos. 13-14 at 0 K were calculated and compared with those of traditional AlCrN and AlTiN coatings. The corresponding results are presented in Figure 13. The formation energies of samples Nos. 5-9, 13, and 14 are lower than that of the commercial Al29.7Cr21.1N49.2 coating (-1.14 eV/atom), but higher than that of the commercial Al30.1Ti18.6N51.3 coating (-1.584 eV/atom). This indicates that, from a thermodynamic perspective, the optimized coatings are more stable than Al29.7Cr21.1N49.2, but less stable than Al30.1Ti18.6N51.3. This result is consistent with previous findings[42], which report that AlTiN coatings can endure annealing temperatures up to 1,100 °C.
Figure 13. Formation energies of experimental samples and commercial Al29.7Cr21.1N49.2 and Al30.1Ti18.6N51.3 coatings at 0 K.
Secondly, according to existing literature, the thermal stability of AlxCr1-xN coating systems is associated with the maximum solubility of fcc-AlN within the coatings[34]. The maximum solubility (x) of fcc-AlN in AlxCr1-xN coating systems at 1,000 °C exceeds 0.62; beyond this critical value, the fcc→hcp transformation occurs via a two-step mechanism[34]. In the present coatings (Nos. 5-9 and Nos. 13-14), the effective Al content (analogous to x) ranges from 0.176 to 0.48, which is below the critical value of 0.62 at 1,000 °C. This illustrates that the superior thermal stability of the optimized coatings is associated with their relatively low Al concentration.
Third, Kainz et al.[27] reported that Ti addition retards the nucleation of hcp-AlN and further stabilizes the fcc structure. It has been proposed that Ti addition suppresses N loss due to the higher binding energy of the Ti-N bond (475.0 kJ/mol) compared to that of the Cr-N bond (377.8 kJ/mol)[43]. Therefore, a high Ti concentration contributes to the thermal stability of the optimized coatings. Furthermore, the improved thermal stability is also related to severe lattice distortion within the coatings. This distortion hinders the long-range diffusion of metal atoms, thereby preventing the nucleation and growth of hcp-AlN.
The results from thermodynamic calculations and previous findings both indicate that the outstanding thermal stability of the optimized AlxCryTizSi(1-x-y-z)N coatings can be attributed to low formation energy, low Al concentration, Ti addition, and severe lattice distortion.
Oxidation analysis at elevated temperatures
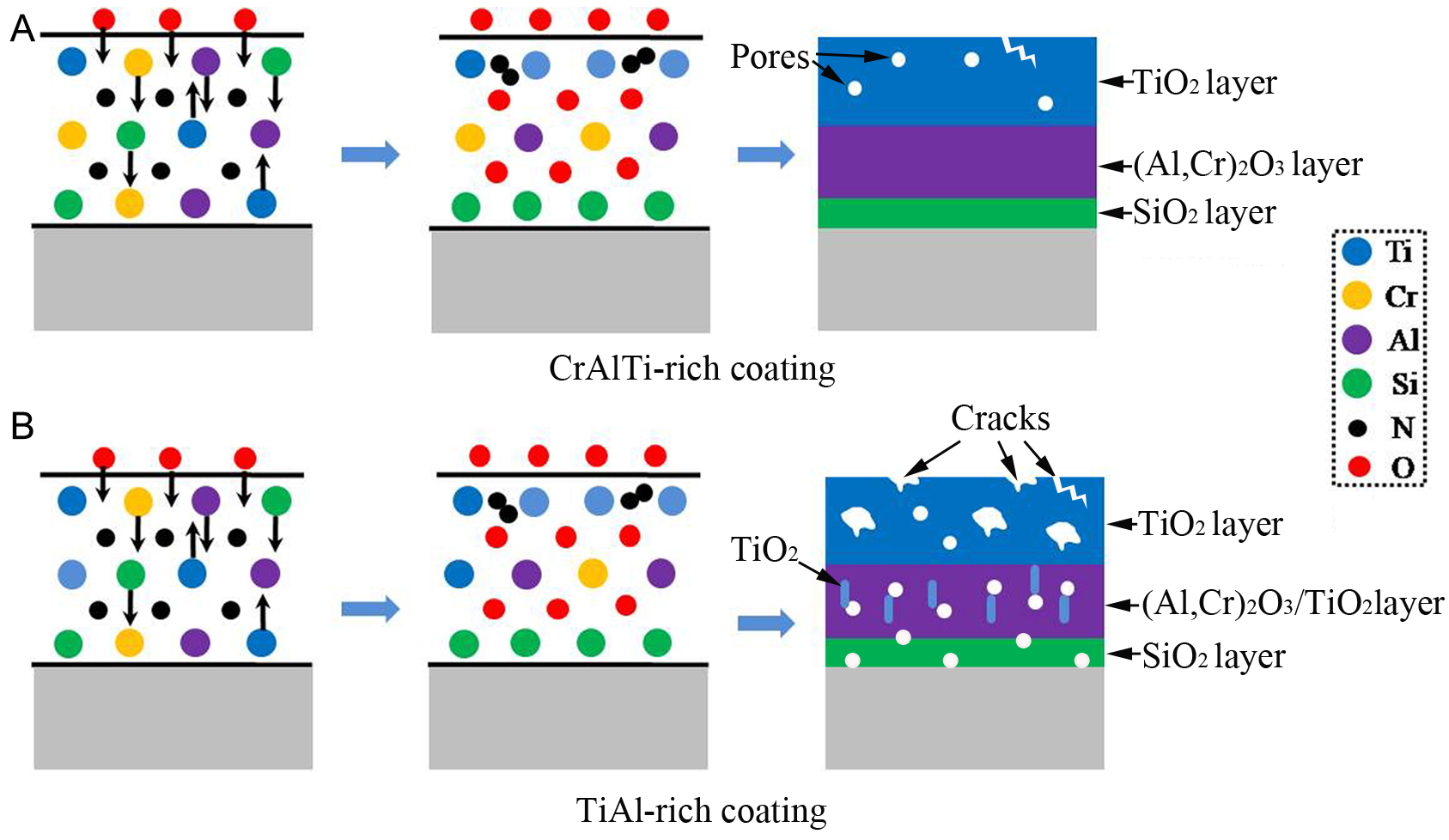
Based on the results of high-temperature oxidation experiments characterized by SEM [Figure 10] and HRTEM [Figure 11], specific compositional ranges were obtained in the AlxCryTizSi(1-x-y-z)N coating systems, where 13.3 ≤ x ≤ 24.1, 7.6 ≤ y ≤ 14.6, 10.4 ≤ z ≤ 23.9, and 1.3 ≤ 1-x-y-z ≤ 3.2 (corresponding to samples
Figure 14. Schematic illustration of the oxidation mechanisms of AlxCryTizSi(1-x-y-z)N coatings oxidized at 1,000 °C: (A) CrAlTi-rich; (B) TiAl-rich.
“Sandwich” composite structures were also observed by TEM [Figure 12] in sample No. 9. However, the oxidation layer becomes thicker and looser compared with that in samples Nos. 5-7 [Figure 14B]. This is primarily attributed to the higher Ti concentration and lower concentrations of Cr and Al. The formation of r-TiO2 is accompanied by cracking and void formation[46,47], and these defects provide fast diffusion pathways for inward O and outward Ti diffusion, thereby accelerating oxidation in the coating subsurface. Consequently, coatings with higher Ti content generally exhibit poorer oxidation resistance. Moreover, the lower Cr concentration impedes the formation of the beneficial Cr3Ti2O5 phase and instead promotes the formation of brookite-TiO2 (b-TiO2), as confirmed by XRD analysis [Figure 10], which further reduces oxidation resistance. Cr atoms can be replaced by Al atoms in the Cr2O3 lattice to form (Cr,Al)2O3, resulting in increased densification because Al atoms have a larger atomic radius than Cr atoms. This indicates that the (Cr,Al)2O3 oxides in samples Nos. 8-9 have lower densification than those in samples Nos. 5-7 due to their lower Al concentration. As a result of the existence of TiO2 and the reduced densification of (Cr,Al)2O3 oxides, a high density of voids forms within the “B-sublayer” of sample No. 9, which accelerates inward O diffusion and leads to the formation of a thick SiO2 sublayer. Therefore, samples with high Ti/Al concentration and low Cr concentrations (samples Nos. 8-9 and Nos. 13-14) exhibit poor oxidation resistance.
CONCLUSIONS
This work addresses the persistent challenge of achieving both high thermal stability and superior oxidation resistance in nitride ceramic coatings at elevated temperatures. By adopting a mechanism-guided, high-throughput combinatorial design strategy, we constructed a compositionally graded library of Al-Cr-Ti-Si-N coatings and systematically clarified the intrinsic composition-structure-property relationships. An optimal compositional window (Al: 13.3-24.1 at.%, Cr: 7.6-14.6 at.%, Ti: 10.4-23.9 at.%,
(1) Coatings within this window exhibit superior integrated properties, including ultrahigh hardness
(2) The superior properties originate from nanoscale synergistic mechanisms. Spinodal decomposition generates Ti-rich and Al/Cr-rich domains, which, coupled with dense dislocation networks and pronounced lattice distortion, collectively enhance mechanical strength and thermal stability.
(3) Upon high-temperature oxidation, an in situ “sandwich” oxide architecture, consisting of an inner amorphous SiO2 layer and an intermediate dense (Al,Cr)2O3 layer, effectively suppresses oxygen ingress and cation outward diffusion. Concurrently, appropriate Cr concentrations promote the formation of Cr3Ti2O5, which suppresses the growth of porous TiO2 and further enhances oxidation resistance.
(4) High-speed dry cutting tests demonstrate that the optimized coatings extend tool life by up to 240% compared to commercial AlTiN, confirming their robustness for extreme service environments. The integrated strategy of mechanism-guided design combined with high-throughput screening established in this work offers a transferable paradigm for the rational development of advanced multicomponent coatings.
DECLARATIONS
Authors’ contributions
Design: Chen, W.; Xiao, H.
Experiments: Huang, J.; Hu, T.; Meng, X.; Li, B.
Data curation: Chen, W.; Huang, J.; Wu, F.
Manuscript writing: Chen, W.; Hu, T.; Xiao, H.
Manuscript revision and supervision: Chen, W.; Huang, J.; Xiao, H.
Availability of data and materials
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China [No. 52001065], the Guangdong Basic and Applied Basic Research Foundation (2025A1515010944, 2022A1515010210, 2024A1515030147, 2023A1515140190), the Hunan Provincial Natural Science Foundation of China [No. 2023JJ30157], and the Scientific Research Project of the Department of Education of Guangdong Province [2023ZDZX3031].
Conflicts of Interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Fan, G.; Zhang, J.; Chen, H.; et al. Analysis of tool wear of TiAlN coated tool, machined surface morphology and chip during titanium alloy milling. Tribol. Int. 2024, 197, 109751.
2. Lin, X.; Fan, D.; Li, F.; et al. Characterization of pulsed plasma nitriding and physical vapor deposition duplex-treated CrAlN coatings. Thin. Solid. Films. 2025, 825, 140726.
3. Gronostajski, Z.; Kaszuba, M.; Widomski, P.; Smolik, J.; Ziemba, J.; Hawryluk, M. Analysis of wear mechanisms of hot forging tools protected with hybrid layers performed by nitriding and PVD coatings deposition. Wear 2019, 420-1, 269-80.
4. Birol, Y. Sliding wear of CrN, AlCrN and AlTiN coated AISI H13 hot work tool steels in aluminium extrusion. Tribol. Int. 2013, 57, 101-6.
5. Zhang, J.; Yan, F.; Yang, H.; Yue, X.; Qing, Z.; Chen, S. Tool wear mechanism of machining refractory high-entropy alloys. Int. J. . Refract. Metals. Hard. Mater. 2026, 134, 107461.
6. Vončina, M.; Nagode, A.; Medved, J.; Balaško, T. Interaction kinetics between molten aluminium alloy Al99.7 and H11 tool steel with and without an AlCrN protective coating. Appl. Surf. Sci. Adv. 2023, 18, 100474.
7. Campos, Neto. N. D. D.; Brune, R.; Luo, A. A.; et al. Towards lube-free aluminum high pressure die casting using duplex AlCrN physical vapor deposition coatings. J. Mater. Res. Technol. 2023, 24, 7409-26.
8. Liu, Z.; Zapolsky, H.; Tang, S.; et al. Atomic scale formation mechanism of the Amorphous-Nanocrystalline biphase structure in TiSiN Coating: phase field crystal simulation and experimental characterization. Appl. Surf. Sci. 2024, 654, 159458.
9. Chang, Y.; Yang, M.; Chang, C.; Chang, B. Cyclic thermal shock resistance and tribological properties of AlCrSiN, AlTiSiN and AlCrSiN/AlTiSiN multilayer hard coatings. Surf. Coat. Technol. 2025, 496, 131650.
10. Chen, L.; Yan, C.; Mei, F.; Yu, Y.; Gao, J.; Yuan, T. A comparative investigation of the thermal stability and oxidation resistance of AlCrSiWN and AlCrSiN/AlCrWN coatings. Ceram. Int. 2024, 50, 55766-78.
11. Xie, J.; Wang, J.; Zeng, F.; et al. Oxidation resistance of TiSiN and AlCrN hard-coatings: Ab initio calculations and experiments. Appl. Surf. Sci. 2023, 616, 156459.
12. Wang, Y.; Su, C.; Wang, S.; et al. Cr incorporation and hybrid bond control in amorphous carbon films for superior thermal radiation efficiency. Appl. Surf. Sci. 2025, 697, 163063.
13. Cedeño-Vente, M.; Manríquez, J.; Mondragón-Rodríguez, G.; et al. Application of a transmission line model to evaluate the influence of structural defects on the corrosion behavior of arc-PVD CrN coatings. Ceram. Int. 2021, 47, 20885-99.
14. Castaldi, L.; Kurapov, D.; Reiter, A.; Shklover, V.; Schwaller, P.; Patscheider, J. High temperature phase changes and oxidation behavior of Cr-Si-N coatings. Surf. Coat. Technol. 2007, 202, 781-5.
15. Chen, W.; Wang, W.; Hu, T.; Zhang, D.; Li, B.; Xiao, H. Enhancing the high-temperature oxidation performance of AlCrTiSiN coating by in-situ generation of multi-layer antioxidant composite structures. Corros. Sci. 2024, 233, 112097.
16. Chen, W.; Wu, W.; Huang, Y.; et al. Influence of annealing on microstructures and mechanical properties of arc-deposited AlCrTiSiN coating. Surf. Coat. Technol. 2021, 421, 127470.
17. International Centre for Diffraction Data. JCPDS database, No. 11-0065(CrN), No. 38-1420 (TiN). https://www.icdd.com (accessed 2026-03-30).
18. Musil, J.; Kunc, F.; Zeman, H.; Poláková, H. Relationships between hardness, Young's modulus and elastic recovery in hard nanocomposite coatings. Surf. Coat. Technol. 2002, 154, 304-13.
19. Aktarer, S. M.; Sert, Y.; Küçükömeroğlu, T. Investigation of structural, hardness, adhesion, and tribological properties of CrN and AlCrN coatings deposited on cylinder liner. Mater. Des. 2025, 253, 113972.
20. Vereschaka, A.; Milovich, F.; Migranov, M.; et al. Investigation of the tribological and operational properties of (Mex,Moy,Al1-(x+y))N (Me -Ti, Zr or Cr) coatings. Tribol. Int. 2022, 165, 107305.
21. Willmann, H.; Mayrhofer, P. H.; Hultman, L.; Mitterer, C. Hardness evolution of Al-Cr-N coatings under thermal load. J. Mater. Res. 2011, 23, 2880-5.
22. Jie, S.; An, S.; Pengjiao, Z.; Junzhou, L. Microstructure, mechanical performance, thermal stability, and oxidation resistance of AlCrN/AlCrBN nano-multilayer coating. Surf. Coat. Technol. 2024, 493, 131301.
23. Henke, B.; Gullikson, E.; Davis, J. X-ray interactions: photoabsorption, scattering, transmission, and reflection at E = 50-30,000 eV,
24. Sui, X.; Li, G.; Zhou, H.; et al. Evolution behavior of oxide scales of TiAlCrN coatings at high temperature. Surf. Coat. Technol. 2019, 360, 133-9.
25. Xiao, B.; Li, H.; Mei, H.; et al. A study of oxidation behavior of AlTiN-and AlCrN-based multilayer coatings. Surf. Coat. Technol. 2018, 333, 229-37.
26. Zhang, R.; Veprek, S. Phase stabilities and spinodal decomposition in the Cr1-xAlxN system studied by ab initio LDA and thermodynamic modeling: comparison with the Ti1-xAlxN and TiN/Si3N4 systems. Acta. Mater. 2007, 55, 4615-24.
27. Kainz, C.; Schalk, N.; Tkadletz, M.; Winkler, M.; Czettl, C. Microstructure, mechanical and thermo-physical properties of CVD TiCxN1-x coatings on cemented carbide substrates grown with C2H6 as C feeding precursor. Surf. Coat. Technol. 2020, 394, 125868.
28. Fan, Q.; Chen, H.; Zhang, S.; et al. Microstructure evolution and degradation process of AlCrTiSiN coatings with different Si contents at high temperatures. Surf. Coat. Technol. 2024, 494, 131505.
29. Kagnaya, T.; Boher, C.; Lambert, L.; Lazard, M.; Cutard, T. Microstructural analysis of wear micromechanisms of WC-6Co cutting tools during high speed dry machining. Int. J. Refract. Metals. Hard. Mater. 2014, 42, 151-62.
30. Sheng, S.; Zhang, R.; Vepřek, S. Decomposition mechanism of Al1-xSixNy solid solution and possible mechanism of the formation of covalent nanocrystalline AlN/Si3N4 nanocomposites. Acta. Mater. 2013, 61, 4226-36.
31. Feng, Y.; Zhang, L.; Ke, R.; Wan, Q.; Wang, Z.; Lu, Z. Thermal stability and oxidation behavior of AlTiN, AlCrN and AlCrSiWN coatings. Int. J. Refract. Metals. Hard. Mater. 2014, 43, 241-9.
32. Yan, C.; Yu, Y.; Mei, F.; Gao, J.; Lin, X.; Lin, J. A Si-Nb co-alloyed AlCrN coating with good mechanical properties, excellent thermal stability and resistance to high temperature oxidation. Surf. Coat. Technol. 2024, 484, 130825.
33. Wang, Q. M.; Kim, K. H. Microstructural control of Cr-Si-N films by a hybrid arc ion plating and magnetron sputtering process. Acta. Mater. 2009, 57, 4974-87.
34. Zhang, R.; Veprek, S. On the spinodal nature of the phase segregation and formation of stable nanostructure in the Ti-Si-N system. Mater. Sci. Eng. A. 2006, 424, 128-37.
35. Mei, F.; Zhang, Z.; Yu, Y.; et al. Microstructure, mechanical, tribological, and oxidizing properties of AlCrSiN/AlCrVN/AlCrNbN multilayer coatings with different modulated thicknesses. Ceram. Int. 2022, 48, 32973-85.
36. Sohrabizadeh, M.; Elmkhah, H.; Lin, N.; Nouri, M. Enhancing mechanical and tribological performance of CrN/CrTiSiN coating through annealing treatment. Int. J. Refract. Metals. Hard. Mater. 2024, 119, 106551.
37. Zhang, Y.; Zuo, T. T.; Tang, Z.; et al. Microstructures and properties of high-entropy alloys. Prog. Mater. Sci. 2014, 61, 1-93.
38. Li, W.; Liu, P.; Liaw, P. K. Microstructures and properties of high-entropy alloy films and coatings: a review. Mater. Res. Lett. 2018, 6, 199-229.
39. Saal, J. E.; Kirklin, S.; Aykol, M.; Meredig, B.; Wolverton, C. Materials design and discovery with high-throughput density functional theory: the Open Quantum Materials Database (OQMD). JOM 2013, 65, 1501-9.
40. Kirklin, S.; Saal, J. E.; Meredig, B.; et al. The Open Quantum Materials Database (OQMD): assessing the accuracy of DFT formation energies. NPJ. Comput. Mater. 2015, 1, 15010.
41. The Open Quantum Materials Database. https://oqmd.org/materials/composition/ (accessed 2026-03-30).
42. Alhafian, M.; Valle, N.; Chemin, J.; et al. Influence of Si addition on the phase structure and oxidation behavior of PVD AlTiN and AlTiCrN coatings using high-resolution characterization techniques. J. Alloys. Compd. 2023, 968, 171800.
43. Zhang, M.; Li, R.; Zhang, Y.; et al. Comparison of microstructure stability and oxidation resistance of Ti/TiN, Ti/TiAlN, and Cr/TiAlN multilayer coatings on TC4 titanium alloy under thermal cycling. J. Alloys. Compd. 2025, 1019, 179310.
44. Barin, I. Thermochemical Data of Pure Substances. VCH VerlagsgesellschaftmbH: Weinheim, 1995. https://onlinelibrary.wiley.com/doi/book/10.1002/9783527619825 (accessed 2026-03-30).
45. Shugurov, A.; Kuzminov, E.; Panin, A. The effect of substrate bias on the structure, mechanical and tribological properties of DC magnetron sputtered Ti-Al-Ta-Si-N coatings. Surf. Coat. Technol. 2025, 517, 132858.
46. Lim, H. P.; Jiang, Z.; Melvin, G. J. H.; et al. A systematic investigation of the tribological behaviour of oxides formed on AlSiTiN, CrAlTiN, and CrAlSiTiN coatings. Wear 2023, 512-3, 204552.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data





















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].