Regenerative and surgical strategies for medication-related osteonecrosis of the jaw (MRONJ): a systematic review
Abstract
Aim: Medication-related osteonecrosis of the jaw (MRONJ) challenges clinicians with its complex pathology and high risk of complications.
Methods: We searched seven databases - PubMed, Scopus, Web of Science, Cochrane Library, Embase, CINAHL, and Google Scholar - and adapted the search strings for each database to optimize retrieval of relevant studies. We then systematically reviewed the included studies to identify the most effective treatments for promoting MRONJ healing.
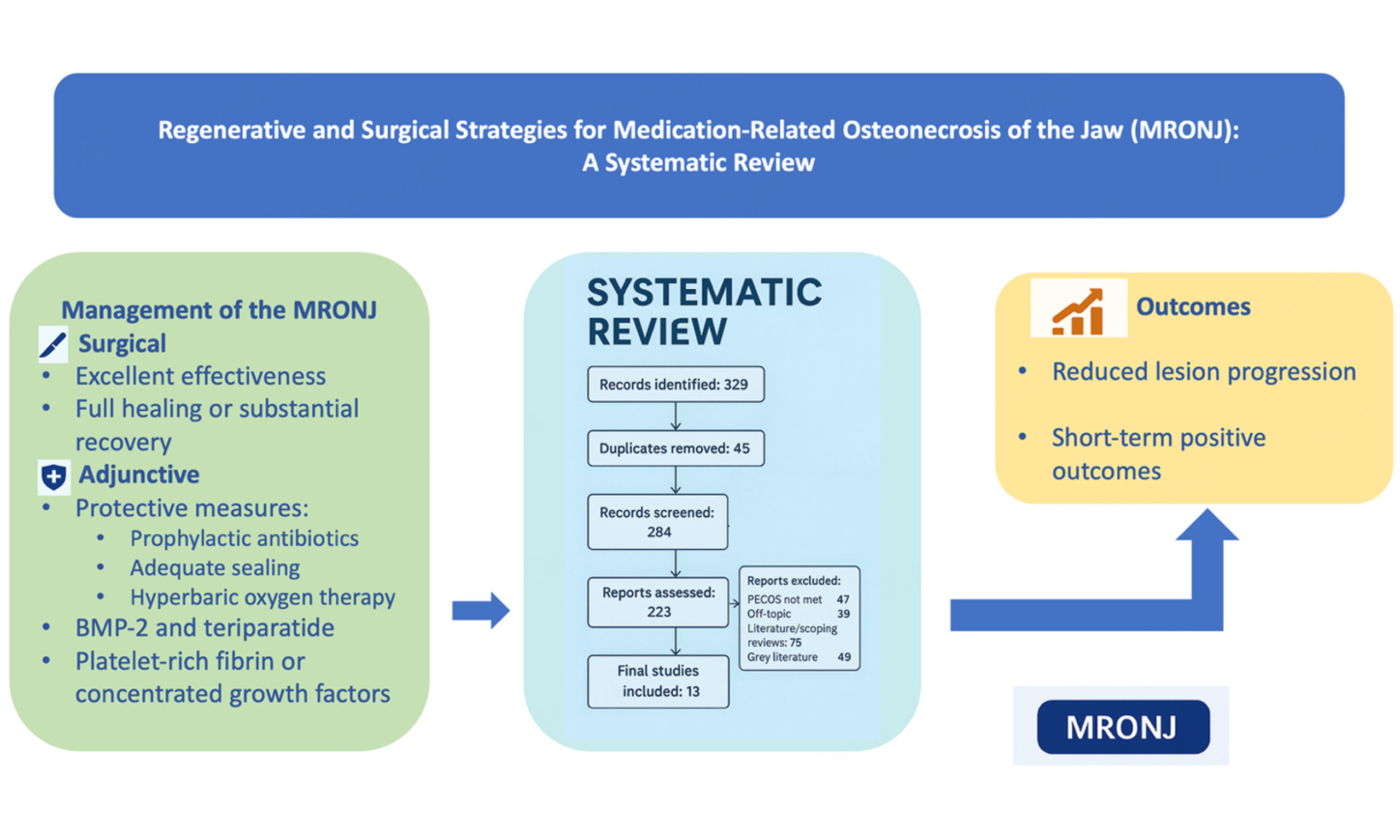
Results: A total of 329 records were identified through database searches across seven electronic databases. After removal of 45 duplicates, 284 records were screened. Following title and abstract screening and full-text assessment, 13 studies met the eligibility criteria and were included in the qualitative synthesis. The included studies consisted primarily of randomized controlled trials and retrospective cohort studies investigating surgical, pharmacological, and adjunctive therapeutic strategies for MRONJ management. Risk-of-bias assessment using the RoB 2.0 and ROBINS-I (Risk of Bias in Non-randomised Studies - of Interventions) tools showed that most studies presented low to moderate risk of bias, although some methodological concerns were identified. Surgical interventions were commonly associated with improved clinical healing, while adjunctive therapies such as bone morphogenetic protein-2 and teriparatide showed promising outcomes in selected cases. Follow-up periods ranged from 1 month to 2 years, and outcome measures included clinical, radiologicalal, and histologicalal evaluations.
Conclusion: MRONJ treatment is diverse and multifaceted. Such inferences were made to varying degrees in previous investigations, illustrating the limitations for clinical use, revealing the need for further studies to clarify both the efficacy and optimal use of the interventions in diverse clinical settings.
Keywords
INTRODUCTION
Medication-related osteonecrosis of the jaw (MRONJ) has recently captured interest due to its increasing prevalence as well as its poorly defined etiology. It is an extremely disabling disorder that causes severe pain and is associated with impaired oral function in affected individuals[1,2]. It is commonly characterized as the exposed or necrotic jawbone, often due to long-term treatment by bisphosphonates, other antiresorptive agents, and anti-angiogenic drugs that are usually used for osteoporosis, malignancy, or rheumatoid arthritis[3]. MRONJ pathogenesis is multifactorial and mediated through a variety of molecular, cellular, and biomechanical mechanisms that disrupt the homeostatic integrity of bone and favor the formation of osteonecrotic lesions[4,5].
The mandible is highly sensitive to the pharmacodynamic actions of these medications as bone resorption is suppressed. Nevertheless, the precise etiology of MRONJ pathways continues to be
Certain anatomical and physiological features make the jaw particularly prone to the negative impact of these drugs. This propensity is associated with the high vascularity and frequent bone remodeling of the alveolar process and periodontium, due to the mechanical stress of chewing[2,7]. MRONJ is thought to result from disturbances in bone remodeling, impaired angiogenesis, microtrauma, impaired immune function, or the presence of inflammation or infection[9].
The American Society for Bone and Mineral Research and the American Association of Oral and Maxillofacial Surgeons define MRONJ based on the following criteria: (i) current or prior treatment with antiresorptive or antiangiogenic agents; (ii) exposed or necrotic bone in the maxillofacial region persisting for more than eight weeks; (iii) no history of radiation therapy to the jaws; and (iv) absence of metastatic disease in the jawbones[2,10]. Treatment modalities should be individualized according to the stage of MRONJ, since early lesions may be managed conservatively, whereas surgery may be required for advanced lesions[11]. The duration of drug treatment and temporary interruptions (“drug holidays”) play an essential role in lesion management and healing[12]. MRONJ has four stages based on clinical features. Stage 0 includes non-exposed, prodromal disease, while stages 1 through 3 involve exposed bone, with or without infection, and increasing severity, including pathological fractures, fistulae, and osteolysis reaching the mandibular border or sinus floor[11-13]. Despite the lack of research into the pathogenesis, there are relatively few available evidence-based therapy indications for MRONJ[11,14]. The present systematic review presents the most recent evidence-based medical treatment approaches for MRONJ, focusing on the effectiveness of strategies aimed at regenerating bone and new tissue. Data from randomized controlled trials and observational studies were compiled to assess the effectiveness of interventions for promoting MRONJ healing using the Population, Exposure, Comparison, Outcome, Study Design (PECOS) framework.
METHODS
Eligibility criteria
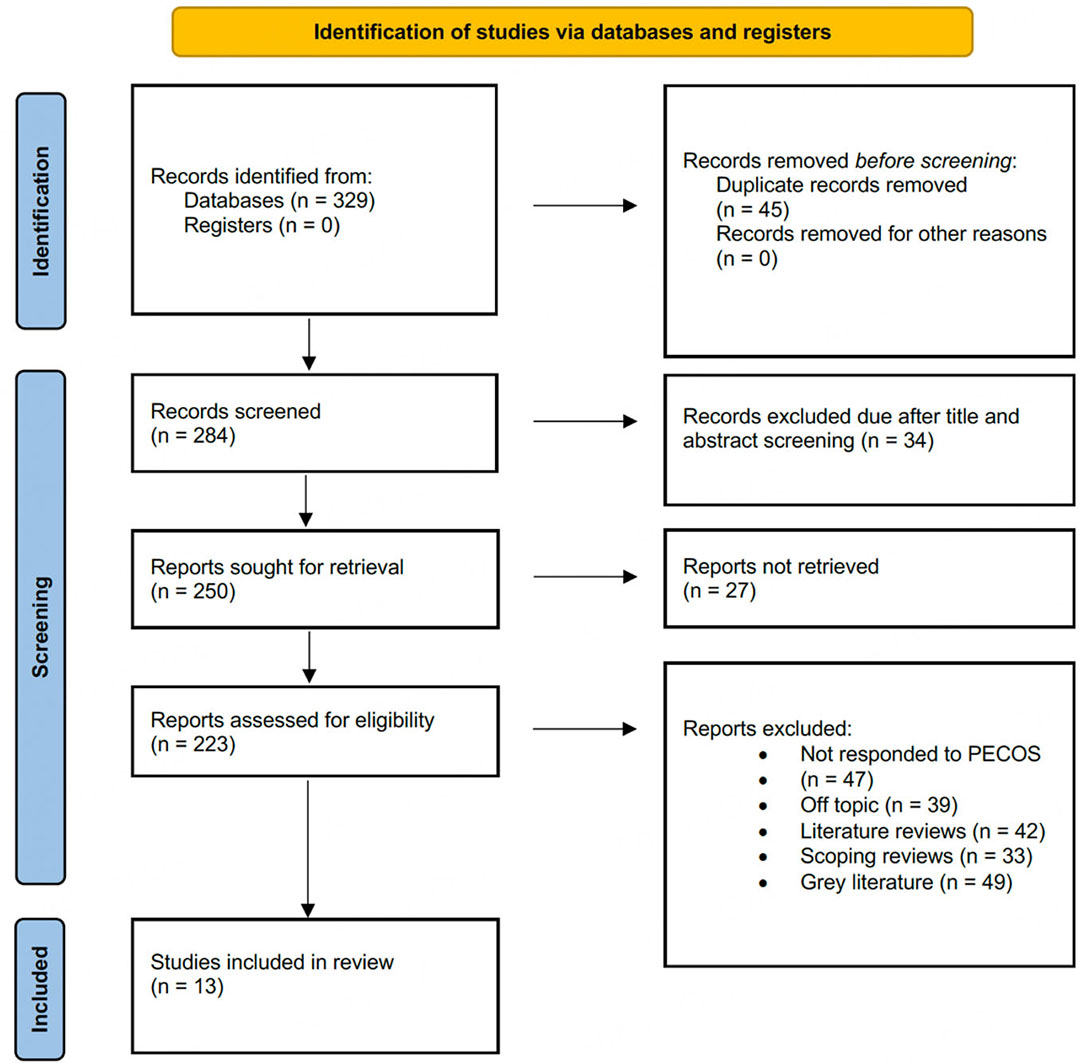
The PECOS framework was developed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 reporting guidelines [Figure 1][15]:
• Population (P): Patients diagnosed with MRONJ at any stage (early, moderate and advanced).
• Exposure (E): Therapeutic interventions - surgical, pharmacological, or adjunctive - intended to promote bone healing and soft tissue regeneration in MRONJ.
• Comparator (C): Standard care, placebo, or alternative interventions.
• Outcome (O): Evidence of bone and soft tissue healing assessed through clinical, radiologicalal, and histologicalal outcomes, as well as measures of pain reduction and recurrence.
• Study design (S): Randomized controlled trials, non-randomized clinical trials, cohort studies, and case series with a minimum of ten patients.
Figure 1. Description of the different stages of article selection process for the review (PRISMA FLOWCHART).
A summary of the inclusion and exclusion criteria is presented in Table 1.
Inclusion and exclusion criteria for the review
| Inclusion criteria | Exclusion criteria |
| Studies investigating therapeutic interventions for MRONJ healing | Title and abstrct Studies not focused on MRONJ healing |
| Studies involving human participants | Animal studies, in vitro studies, or reviews |
| Studies published in English language | Studies published in languages other than English |
| RCTs, retrospective/cohort studies | Case reports, literature reviews, grey literature |
| Studies with a clear description of the therapeutic intervention and outcome measures | Studies with incomplete or unclear descriptions of the therapeutic intervention or outcome measures |
| Studies published within the last 20 years (2005-2025) | |
Database search plan
A thorough systematic search was undertaken on seven databases: PubMed, Scopus, Web of Science, Cochrane Library, Embase, CINAHL, and Google Scholar. Google Scholar was included to retrieve grey literature and studies not indexed in the other databases, despite its limitations regarding the quality of literature. The search strategies employed MeSH terms [Table 2]. The search was limited to English-language publications published within the last 20 years (January 2005-January 2025) to ensure contemporary relevance and feasibility of analysis. The final literature search was completed on January 5, 2025.
Search strings/phrases used across the different databases
| Database | Search string |
| PubMed | (("osteonecrosis of the jaw"[MeSH]) OR ("medication-related osteonecrosis of the jaw"[All Fields])) AND (("healing"[MeSH]) OR ("bone regeneration"[MeSH])) AND (("therapy"[MeSH]) OR ("intervention"[MeSH])) |
| Scopus | ("osteonecrosis of the jaw" OR "medication-related osteonecrosis of the jaw") AND ("healing" OR "bone regeneration") AND ("therapy" OR "intervention") |
| Web of Science | (("osteonecrosis of the jaw" OR "medication-related osteonecrosis of the jaw") AND ("healing" OR "bone regeneration")) AND ("therapy" OR "intervention") |
| Cochrane Library | ("osteonecrosis of the jaw" OR "medication-related osteonecrosis of the jaw") AND ("healing" OR "bone regeneration") AND ("therapy" OR "intervention") |
| Embase | (("osteonecrosis of the jaw"/exp) OR ("medication-related osteonecrosis of the jaw"/exp)) AND (("healing"/exp) OR ("bone regeneration"/exp)) AND (("therapy"/exp) OR ("intervention"/exp)) |
| CINAHL | (("osteonecrosis of the jaw" OR "medication-related osteonecrosis of the jaw") AND ("healing" OR "bone regeneration")) AND ("therapy" OR "intervention") |
| Google Scholar | ("osteonecrosis of the jaw" OR "medication-related osteonecrosis of the jaw") AND ("healing" OR "bone regeneration") AND ("therapy" OR "intervention") |
Although grey literature was retrieved during the search process, it was excluded during the screening phase according to the predefined exclusion criteria.
Data extraction protocol
Both authors independently extracted data using a structured extraction form. Variables included study details (author, year, geographic location), participant information (sample size, age, gender), treatment modalities, MRONJ severity, treatment and follow-up period, evaluation methods (clinical, radiological, histological), and primary outcomes. Any discrepancies were resolved through discussion or referral to a third person. Cohen’s kappa was calculated to assess inter-rater reliability for study inclusion in this systematic review.
Bias analysis strategy
Risk of bias in non-randomized studies was assessed using the ROBINS-I (Risk Of Bias In Non-randomised Studies - of Interventions) tool[16], while the Cochrane's Risk of Bias 2 Tool (RoB 2) 2.0[17] was applied to randomized controlled trials. Two reviewers independently evaluated the risk of bias; disagreements were resolved through consensus or third-party adjudication. Inter-reviewer agreement, assessed using Cohen’s kappa, was 0.87, indicating strong concordance.
RESULTS
Trial inclusion process
In total 329 records were identified from database searches, and no records from the trial registries were found. After the elimination of 45 duplicate entries, 284 records remained for screening. After title and abstract screening, 250 articles were considered eligible for full-text assessment. However, 27 full-text articles could not be retrieved, leading to 223 studies considered eligible. The primary reasons for exclusion were studies not meeting PECOS criteria (n = 47), off-topic articles (n = 39), literature reviews (n = 42), scoping reviews (n = 33), and grey literature (n = 49). After applying all exclusion criteria, 13 studies[18-30] met the eligibility requirements and were included in the final analysis.
Assessed levels of bias
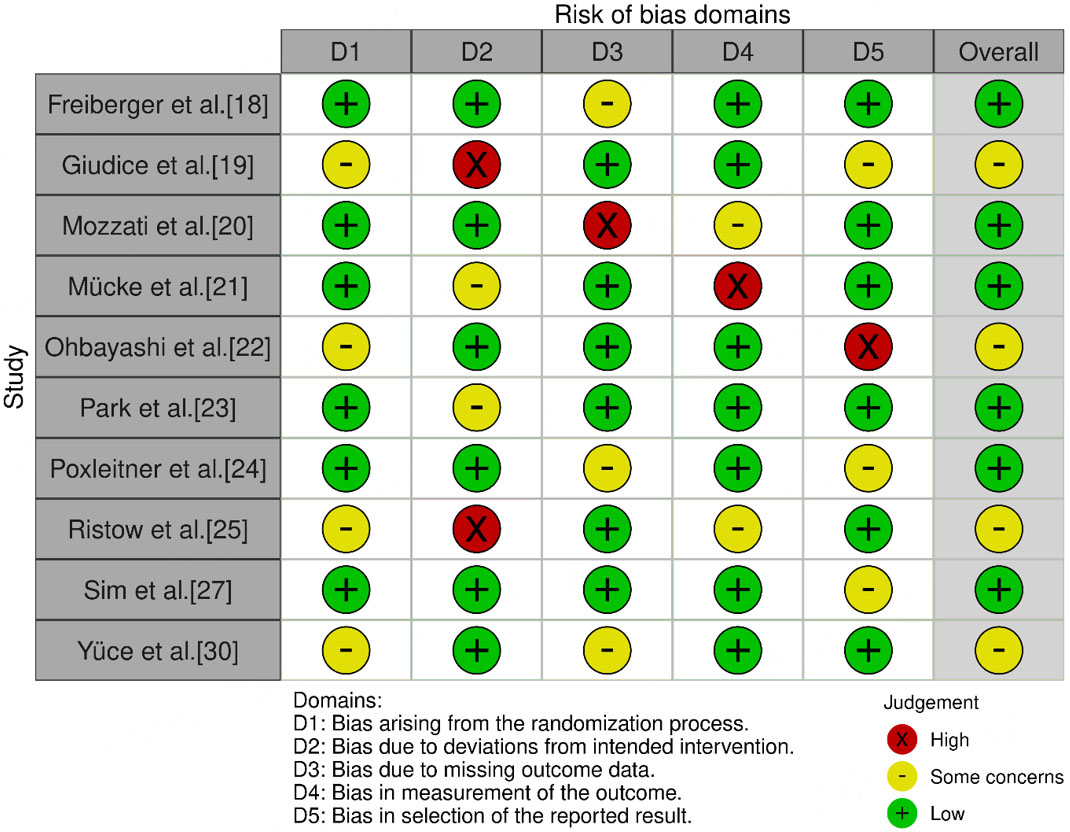
Risk of bias was classified as low, moderate, or high, based on the domain-specific criteria outlined in the Cochrane RoB 2.0 tool (for randomized trials) and the ROBINS-I tool (for non-randomized studies). Each domain - such as confounding (D1) and participant selection (D2) - was independently assessed by two reviewers, with the overall risk of bias determined by the highest level of concern in any domain.
Most studies[18,20,23,24,26] were rated as having a low overall risk of bias [Figure 2], while some[19,25,30] presented moderate concerns, and one study[28] was classified as having moderate risk of bias [Figure 3]. In detail, the studies by Freiberger et al.[18], Mozzati et al.[20], Park et al.[23], Poxleitner et al.[24], and Sahin et al.[26] showed a low risk of bias across all domains. Giudice et al.[19], Ristow et al.[25], and Yüce et al.[30] demonstrated concerns in multiple domains. Soutome et al.[28] had a moderate overall risk of bias, while Mücke et al.[21] exhibited a high risk specifically in domain D4, and Ohbayashi et al.[22] showed some concern in domain D5. Sim et al.[27] and Varoni et al.[29] demonstrated low risk in most domains, although minor concerns were noted in domains D5 and D7, respectively.
Baseline characteristics observed
Table 3 presents the trials included in this review[18-30] and their respective outcome assessments. The trials by Freiberger et al.[18] and Giudice et al.[19] had a slight majority of female participants, with 44%:56% and 42%:58%, respectively. In direct contrast, Mozzati et al.[20] reported marked imbalance, at 4%:96% and 2%:98% male:female (M:F) ratios for Protocols A and B, respectively. This marked gender imbalance could be due to the specific population sampled in that protocol (e.g., postmenopausal women with osteoporosis). Among the available data, Mücke et al.[21] included only male participants, and Ohbayashi et al.[22] included only female participants. Park et al.[23] reported an almost equal distribution: 57%:43%, and 43%:57% M:F in the Leukocyte- and Platelet-Rich Fibrin (L-PRF) and L-PRF + rhBMP-2 (recombinant human bone morphogenetic protein-2) groups, respectively. Poxleitner et al.[24] found a majority of females: 0%:100% and 3%:97% M:F in Groups A and B, respectively. The other studies presented various M:F ratios, ranging between 7:14 (reported by Sahin et al.[26]) and 53:47 (reported by Sim et al.[27]). Sample sizes differed between 13 participants in the study of Ohbayashi et al.[22] and 700 participants in the study of Mozzati et al.[20]. Most of the sample sizes included between 30 and 100 subjects.
Studies included in the review and their observed assessments
| Study | Year | Design/Sample | Intervention | Follow-up | Primary outcome | Key findings |
| Surgical interventions | ||||||
| Freiberger et al.[18] | 2012 | RCT, 49 | HBO vs. standard care | 2 years | Lesion size, pain | HBO improved lesions (68%) and faster pain relief |
| Mozzati et al.[20] | 2013 | RCT, 700 | Protocol A vs. Protocol B | 12-72 month | Post-op MRONJ incidence | Both protocols effective; Protocol B preferred (less trauma) |
| Mücke et al.[21] | 2016 | RCT, 253 | Standard care vs. prophylactic antibiotics + tension-free closure | 2-4 years | MRONJ incidence/year | Group B reduced MRONJ incidence by 82% |
| Sahin et al.[26] | 2022 | Retrospective, 21 | Ultrasonic surgery + L-PRF + Nd:YAG laser | 1-12 month | Mucosal healing | 100% success; 2 delayed healing cases |
| Soutome et al.[28] | 2021 | Retrospective, 38 | Segmental/marginal mandibulectomy | 1-3+ month | Healing | 55.3% complete healing; PR type influences outcome |
| Varoni et al.[29] | 2021 | Retrospective, 35 | Antibiotics, pentoxifylline, tocopherol + sequestrectomy | 24 month | Healing, recurrence | 92% long-term healing; 30% recurrence |
| Adjunctive/pharmacological interventions | ||||||
| Giudice et al.[19] | 2018 | RCT, 47 | PRF vs. standard care | 1 year | Mucosal integrity | PRF improved short-term mucosal integrity (T1 only) |
| Ohbayashi et al.[22] | 2020 | RCT, 13 | Weekly vs. daily TPTD | 6 month | MRONJ stage | Daily TPTD showed better improvement |
| Park et al.[23] | 2017 | RCT, 55 | L-PRF vs. L-PRF + rhBMP-2 | 6 month | Healing | BMP-2 addition improved resolution (75% vs. 48%) |
| Poxleitner et al.[24] | 2020 | RCT, 77 | Post-extraction primary closure vs. PRF clot | 90 days | Mucosal coverage | No significant difference between groups |
| Ristow et al.[25] | 2020 | RCT, 160 | Epiperiosteal vs. subperiosteal closure | ≥ 6 month | MRONJ prevention | Comparable success rates |
| Sim et al.[27] | 2020 | RCT, 34 | Teriparatide vs. placebo | 1 year | MRONJ staging | Teriparatide improved lesion resolution |
| Yüce et al.[30] | 2021 | RCT, 28 | CGF vs. control | 6 month | Soft tissue healing | No significant benefit of CGF |
Interventions and groups assessed
The study objectives differed: some focused on the prevention of MRONJ, including those conducted by Freiberger et al.[18], Mozzati et al.[20], Park et al.[23], Poxleitner et al.[24], and Sahin et al.[26]; whereas others - Giudice et al.[19], Mücke et al.[21], Ohbayashi et al.[22], Ristow et al.[25], Sim et al.[27], Soutome et al.[28],
Interventions for MRONJ varied across studies. Hyperbaric oxygen (HBO) therapy was investigated in the study by Freiberger et al.[18], while platelet-rich fibrin (PRF) was evaluated in studies by Mozzati et al.[20],
Results observed
The follow-up periods ranged from 1 month[28] to 2 years[18], with varying intervals in between. The outcome measures also differed across studies, reflecting the variations in objectives and interventions.
Freiberger et al.[18] reported a statistically significant improvement in the HBO group following treatment; 68% of patients improved, with a mean time to improvement of 39.7 weeks. Giudice et al.[19] found that the PRF group showed significant variations in mucosal integrity, absence of infection, and pain only at T1. Mozzati et al.[20] reported 100% success in both groups. Conversely, Mücke et al.[21] concluded that Group B showed a highly significant reduction in MRONJ incidence, with an observed risk of 82%.
Ohbayashi et al.[22] reported significant improvement in MRONJ stage for the daily teriparatide (TPTD) group. Park et al.[23] observed a significantly higher complete resolution rate in the L-PRF + rhBMP-2 group compared to the L-PRF group (75.2% vs. 48%, P = 0.028). Poxleitner et al.[24] reported no statistically significant between-group differences. Ristow et al.[25] reported successful healing in 100% of cases; however, two patients showed delayed healing.
Sahin et al.[26] demonstrated that a combined approach of surgical debridement and adjunctive teriparatide significantly improved bone healing in patients with MRONJ. Sim et al.[27] reported a complete healing rate of 55.3%, with poor outcomes influenced by platelet reactivity (PR) patterns. Soutome et al.[28] reported a long-term healing rate of 92%, though the recurrence rate was 30.4%. Varoni et al.[29] found no significant differences in postoperative healing between groups. For Yüce et al.[30], specific outcome measures were not clearly extractable.
DISCUSSION
Converging and diverging findings on MRONJ treatment strategies were observed during comparison with the included studies. As we see, surgical procedures are effective therapeutic approaches to MRONJ in numerous studies. Mozzati et al.[20] and Ristow et al.[25] presented consistent results, with
Another group of studies supports the use of adjunctive therapies to enhance MRONJ treatment outcomes. Park et al.[23] demonstrated an enhanced healing in the presence of bone morphogenetic protein 2 (BMP-2) and Sim et al.[27] also proposed that teriparatide, possibly used as an effective therapeutic agent, may be potentially useful in MRONJ treatment. These findings imply that adjunctive therapy with adjuvant therapies such as BMP-2 and teriparatide can also be effective in increasing success rates in the treatment of MRONJ. However, Yüce et al.[30] identified no benefit from CGF, thus, the effect of adjunctive treatments may be based on patient characteristics, disease stage or treatment protocol.
In contrast, in numerous studies, outcomes suggested findings that varied from most others.
Notably, Yüce et al.[30] found that CGF did not significantly improve soft tissue repair in MRONJ, a result that contradicts the findings from other adjunctive therapies. These results may have limited generalizability, as they are specific to the patient population and clinical setting of the study. Ohbayashi et al.[22] and
Studies by Mozzati et al.[20], Ristow et al.[25], and Sahin et al.[26] share a common focus on evaluating the effectiveness of surgical interventions in treating MRONJ. Similarly, Park et al.[23] and Sim et al.[27] both investigated the role of adjuvant therapies - BMP-2 and teriparatide, respectively - in MRONJ treatment. The remaining studies varied considerably in design and focus, examining diverse methods for both prevention and treatment of MRONJ.
In cases of early-stage MRONJ, patients are often asymptomatic with minimal bone exposure and conservative/noninvasive treatment protocols have been proposed as a reasonable alternative[2]. The problem is that these patients are often poor candidates for long-term antibiotic use due to advanced age, previous prolonged chemotherapy regimens, or metastatic bone disease. While local or systemic antibiotic therapy may alleviate symptoms, this relief is often short-lived; recurrences occur on average every three weeks[31], leading researchers to seek alternative treatment modalities. Conservative management might alleviate the symptoms and infections in the short run, but one must not forget that osteonecrosis is not going to be cured by these methods[32]. However, if there is exposed necrotic bone, then conservative management fails because the bone tissue is dead and therefore can never be restored to its former state.
The decision to discontinue medications prior to surgical intervention for MRONJ remains a topic of debate[33]. A recent systematic review and meta-analysis found that patients with multiple myeloma or osteoporosis who underwent a "drug holiday" from alendronate, ibandronate, or zoledronate exhibited a higher prevalence of complete recovery[34]. On the other hand, some researchers argue that withdrawal of these drugs is unnecessary due to the long half-life of bisphosphonates (approximately 11 years), which are sequestered in bone hydroxyapatite crystals. There is also a concern that drug discontinuation may exacerbate underlying conditions, such as bone pain or skeletal weakness.
Similar to our findings, Beth-Tasdogan et al.[35] conducted a systematic review to assess interventions for the prophylaxis and treatment of MRONJ. Including only RCTs, however, they made the conclusion that the certainty of existing evidence was low or very low. Our review demonstrated this and found that evidence for MRONJ interventions remains limited and of low quality. Similarly, Kawahara et al.[36] described a systematic literature review to produce updated recommendations for dental treatment which would be directed to avoid the onset of MRONJ. Their findings emphasized the importance of producing clinical guidelines from scientific evidence; they mirrored our own findings that high-quality evidence is a requisite for clinical practice.
Kuroshima et al.[37] conducted this review to summarize the current knowledge and theories on MRONJ from clinical perspective and basic science perspective. The study included MRONJ definition, epidemiology, causative drugs, histopathology, and pathophysiology. Likewise, our work explored MRONJ epidemiology, pathophysiology and risk factors. Similarly, AlRowis et al.[38] performed a review of the literature to identify diagnostic criteria and risk factors of MRONJ. They also discussed its pathophysiology, preventive methods, and treatments. Their review had covered the commonality across epidemiology, diagnostic criteria, risk factors, pathogenesis and therapeutic management. As there is high heterogeneity in interventions and outcome measures as well as follow-up periods across the included studies, a meta-analysis was not possible and a qualitative assessment was thus conducted solely.
Limitations
Our results should be interpreted in the context of several limitations. Variability in study design and population may have contributed to heterogeneity in the findings, limiting the ability to perform a meta-analysis. Moreover, only a few studies were available investigating specific interventions such as add-on therapies, which could limit the extent to which the findings can be generalized. Publication bias is also a consideration as this review is based entirely on published literature. Similarly, there were limitations with respect to differences in follow-up periods and outcome measures, but both were included to prevent a significant loss of the body of available literature.
Clinical recommendations
Based on our findings, several recommendations were made for managing MRONJ. Oral surgeons, oncologists and other health professionals must take a comprehensive multidisciplinary approach to ensure optimal treatment outcomes. For the more advanced stages of MRONJ, surgical procedures such as debridement and resection are indicated. Prophylactic measures should be properly defined, with tension-free wound closure. Adjuvants are shown to possess promising therapeutic potential, especially bone morphogenetic protein-2 and teriparatide. Care must be tailored to patients' individual disease stage and history. Finally, future research will need to determine more definitively the degree to which these interventions are both effective and best implemented depending on the clinical case and also to formulate evidence-based guidelines in managing MRONJ.
CONCLUSION
Based on this review, surgical interventions appear most successful for advanced-stage MRONJ, while BMP-2 and teriparatide have shown efficacy in promoting healing in some cases. Prophylactic measures, such as antibiotics and tension-free wound closure, may reduce the risk of MRONJ in high-risk populations. However, well-designed, multicenter studies are needed to establish optimal treatment protocols for the various stages of MRONJ.
DECLARATIONS
Acknowledgments
The authors would like to acknowledge Prof. Luis Eduardo Almeida, a native English speaker and Associate Professor at the Department of Surgical Sciences, School of Dentistry, Marquette University, Milwaukee, Wisconsin, USA, for reviewing and editing the manuscript to ensure proper scientific English.
Authors’ contributions
Conceptualized and designed the study: Veeraraghavan VP, Minervini G
Drafted the manuscript: Veeraraghavan VP, Piccolo S
Data analysis: Fiorito N, Minervini G, Russo D
Conducted the literature search and data collection: Uzuncibuk H, Fiorito N
Prepared the figures and tables: Uzuncibuk H, Fiorito N, Marrapodi MM, Russo D
Contributed to literature selection, quality evaluation, and manuscript revision: Cervino G, Minervini G
All authors reviewed and approved the final version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the authors used DeepL Translate for language checking/grammar correction. After its use, the authors thoroughly reviewed, verified, and revised all content to ensure accuracy and originality. The authors take full responsibility for the integrity and final content of the published article.
Financial support and sponsorship
None.
Conflicts of interest
Minervini G and Marrapodi MM serve as Junior Editorial Board Members of the Journal of Cancer Metastasis and Treatment. They had no involvement in the editorial process of this manuscript, including reviewer selection, handling, or decision-making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Ferneini EM. Medication-related osteonecrosis of the jaw (MRONJ). J Oral Maxillofac Surg. 2021;79:1801-2.
2. Ruggiero SL, Dodson TB, Aghaloo T, Carlson ER, Ward BB, Kademani D. American association of oral and maxillofacial surgeons' position paper on medication-related osteonecrosis of the jaws-2022 update. J Oral Maxillofac Surg. 2022;80:920-43.
3. Aguirre JI, Castillo EJ, Kimmel DB. Preclinical models of medication-related osteonecrosis of the jaw (MRONJ). Bone. 2021;153:116184.
4. Beth-Tasdogan NH, Mayer B, Hussein H, Zolk O. Interventions for managing medication-related osteonecrosis of the jaw. Cochrane Database Syst Rev. 2017;10:CD012432.
5. Ali IE, Sumita Y. Medication-related osteonecrosis of the jaw: prosthodontic considerations. Jpn Dent Sci Rev. 2022;58:9-12.
6. Bansal H. Medication-related osteonecrosis of the jaw: an update. Natl J Maxillofac Surg. 2022;13:5-10.
7. Boston B, Ipe D, Capitanescu B, et al. Medication-related osteonecrosis of the jaw: a disease of significant importance for older patients. J Am Geriatr Soc. 2023;71:2640-52.
8. Bedogni A, Mauceri R, Fusco V, et al. Italian position paper (SIPMO-SICMF) on medication-related osteonecrosis of the jaw (MRONJ). Oral Dis. 2024;30:3679-709.
9. Mohd Yunus SS, Soh HY, Abdul Rahman M, Peng X, Guo C, Ramli R. MicroRNA in medication related osteonecrosis of the jaw: a review. Front Physiol. 2023;14:1021429.
10. Rosenberg SA, Migliorati C, Romanos GE. Is medication-related osteonecrosis of the jaw associated with tumor necrosis factor-α inhibition? Oral Surg Oral Med Oral Pathol Oral Radiol. 2021;131:422-7.
11. Roato I, Mauceri R, Notaro V, Genova T, Fusco V, Mussano F. Immune dysfunction in medication-related osteonecrosis of the jaw. Int J Mol Sci. 2023;24:7948.
12. Kün-Darbois JD, Fauvel F. Medication-related osteonecrosis and osteoradionecrosis of the jaws: update and current management. Morphologie. 2021;105:170-87.
13. Byrne H, O'Reilly S, Weadick CS, Brady P, Ríordáin RN. How we manage medication-related osteonecrosis of the jaw. Eur J Med Res. 2024;29:402.
14. Migliorati CA. Oral complications in cancer patients-medication-related osteonecrosis of the jaw (MRONJ). Front Oral Health. 2022;3:866871.
15. Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
16. Igelström E, Campbell M, Craig P, Katikireddi SV. Cochrane's risk of bias tool for non-randomized studies (ROBINS-I) is frequently misapplied: a methodological systematic review. J Clin Epidemiol. 2021;140:22-32.
17. Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898.
18. Freiberger JJ, Padilla-Burgos R, McGraw T, et al. What is the role of hyperbaric oxygen in the management of bisphosphonate-related osteonecrosis of the jaw: a randomized controlled trial of hyperbaric oxygen as an adjunct to surgery and antibiotics. J Oral Maxillofac Surg. 2012;70:1573-83.
19. Giudice A, Barone S, Giudice C, Bennardo F, Fortunato L. Can platelet-rich fibrin improve healing after surgical treatment of medication-related osteonecrosis of the jaw? A pilot study. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;126:390-403.
20. Mozzati M, Arata V, Gallesio G. Tooth extraction in osteoporotic patients taking oral bisphosphonates. Osteoporos Int. 2013;24:1707-12.
21. Mücke T, Deppe H, Hein J, et al. Prevention of bisphosphonate-related osteonecrosis of the jaws in patients with prostate cancer treated with zoledronic acid - A prospective study over 6 years. J Craniomaxillofac Surg. 2016;44:1689-93.
22. Ohbayashi Y, Iwasaki A, Nakai F, Mashiba T, Miyake M. A comparative effectiveness pilot study of teriparatide for medication-related osteonecrosis of the jaw: daily versus weekly administration. Osteoporos Int. 2020;31:577-85.
23. Park JH, Kim JW, Kim SJ. Does the addition of bone morphogenetic protein 2 to platelet-rich fibrin improve healing after treatment for medication-related osteonecrosis of the jaw? J Oral Maxillofac Surg. 2017;75:1176-84.
24. Poxleitner P, Steybe D, Kroneberg P, et al. Tooth extractions in patients under antiresorptive therapy for osteoporosis: primary closure of the extraction socket with a mucoperiosteal flap versus application of platelet-rich fibrin for the prevention of antiresorptive agent-related osteonecrosis of the jaw. J Craniomaxillofac Surg. 2020;48:444-51.
25. Ristow O, Rückschloß T, Moratin J, et al. Wound closure and alveoplasty after preventive tooth extractions in patients with antiresorptive intake-A randomized pilot trial. Oral Dis. 2021;27:532-46.
26. Sahin O, Akan E, Tatar B, et al. Combined approach to treatment of advanced stages of medication-related osteonecrosis of the jaw patients. Braz J Otorhinolaryngol. 2022;88:613-20.
27. Sim IW, Borromeo GL, Tsao C, et al. Teriparatide promotes bone healing in medication-related osteonecrosis of the jaw: a placebo-controlled, randomized trial. J Clin Oncol. 2020;38:2971-80.
28. Soutome S, Yanamoto S, Sumi M, et al. Effect of periosteal reaction in medication-related osteonecrosis of the jaw on treatment outcome after surgery. J Bone Miner Metab. 2021;39:302-10.
29. Varoni EM, Lombardi N, Villa G, Pispero A, Sardella A, Lodi G. Conservative management of medication-related osteonecrosis of the jaws (MRONJ): a retrospective cohort study. Antibiotics. 2021;10:195.
30. Yüce MO, Adalı E, ISık G. The effect of concentrated growth factor (CGF) in the surgical treatment of medication-related osteonecrosis of the jaw (MRONJ) in osteoporosis patients: a randomized controlled study. Clin Oral Investig. 2021;25:4529-41.
31. Nisi M, La Ferla F, Karapetsa D, et al. Conservative surgical management of patients with bisphosphonate-related osteonecrosis of the jaws: a series of 120 patients. Br J Oral Maxillofac Surg. 2016;54:930-5.
32. Coropciuc RG, Grisar K, Aerden T, Schol M, Schoenaers J, Politis C. Medication-related osteonecrosis of the jaw in oncological patients with skeletal metastases: conservative treatment is effective up to stage 2. Br J Oral Maxillofac Surg. 2017;55:787-92.
33. Reich W, Bilkenroth U, Schubert J, Wickenhauser C, Eckert AW. Surgical treatment of bisphosphonate-associated osteonecrosis: prognostic score and long-term results. J Craniomaxillofac Surg. 2015;43:1809-22.
34. Hayashida S, Yanamoto S, Fujita S, et al. Drug holiday clinical relevance verification for antiresorptive agents in medication-related osteonecrosis cases of the jaw. J Bone Miner Metab. 2020;38:126-34.
35. Beth-Tasdogan NH, Mayer B, Hussein H, Zolk O, Peter JU. Interventions for managing medication-related osteonecrosis of the jaw. Cochrane Database Syst Rev. 2022;7:CD012432.
36. Kawahara M, Kuroshima S, Sawase T. Clinical considerations for medication-related osteonecrosis of the jaw: a comprehensive literature review. Int J Implant Dent. 2021;7:47.
37. Kuroshima S, Al-Omari FA, Sasaki M, Sawase T. Medication-related osteonecrosis of the jaw: a literature review and update. Genesis. 2022;60:e23500.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].