Should we shift from delaying diabetes to pursuing pre-diabetes remission for cardiovascular disease prevention?
Abstract
The global epidemic of dysglycemia, spanning from pre-diabetes to overt type 2 diabetes (T2D), constitutes a primary driver of cardiovascular morbidity and mortality worldwide. While managing established T2D remains crucial, accumulating evidence underscores that the pre-diabetic state itself represents a period of active, subclinical cardiovascular injury. This commentary argues for a fundamental strategic shift: from merely delaying progression to T2D toward actively pursuing remission of pre-diabetes - defined as the sustained return to normoglycemia - as a potent and likely essential target for curbing cardiovascular disease. This approach moves beyond traditional risk factor management by targeting a reversible pathophysiological nexus. The rationale is robust, implicating insulin resistance, hyperinsulinemia, β-cell stress, chronic inflammation, endothelial dysfunction, and a pro-atherogenic lipid profile. Notably, long-term follow-up data from landmark prevention trials now provide compelling evidence that achieving remission is associated with a striking reduction of approximately 50% in the risk of cardiovascular death and heart failure hospitalization. Although intensive lifestyle intervention remains the cornerstone, emerging therapeutic options - including potent glucagon-like peptide-1 (GLP-1) receptor agonists, glucose-dependent insulinotropic polypeptide (GIP)/GLP-1 dual agonists, and metabolic surgery - offer new pathways to induce remission. Nevertheless, significant gaps persist, primarily the lack of a consensus definition for pre-diabetes remission and the absence of prospective trials with remission as a primary cardiovascular endpoint. Addressing these gaps through coordinated research, consensus-building, and integration into clinical guidelines is imperative to translate this paradigm from a compelling hypothesis into a standard of care capable of altering the cardiovascular trajectory of hundreds of millions at risk.
Keywords
INTRODUCTION
Cardiovascular disease (CVD) remains the leading cause of death and disability in individuals with dysglycemia, imposing an unsustainable burden on global health systems. This special issue on “Emerging Mechanisms and Therapeutic Strategies in Diabetic Cardiovascular Disease” rightly focuses on innovative approaches within the diabetic population. However, a compelling case can be made that the most impactful window for intervention lies further upstream, in the vast and often overlooked population with pre-diabetes. In this commentary, we explore the pathophysiological foundations, review the clinical evidence mosaic, discuss the expanding therapeutic toolkit, highlight the urgent need for a standardized definition, and outline a roadmap for future research and clinical integration. Characterized by impaired fasting glucose (IFG), impaired glucose tolerance (IGT), or elevated glycosylated hemoglobin A1c (HbA1c) (5.7%-6.4%), pre-diabetes affects over one-third of adults in some populations, representing more than a billion individuals globally[1,2]. This is not a benign or “pre-” condition; it is a state of heightened cardiometabolic risk where endothelial damage and atherogenesis are already underway[3]. Emerging evidence suggests that an even earlier marker - 1-h postprandial hyperglycemia - may identify individuals at a highly reversible stage of metabolic dysfunction, preceding the conventional diagnosis of pre-diabetes, thus offering a potential earlier window for interception[4]. Recently, a Commentary in The Lancet Diabetes & Endocrinology proposed defining pre-diabetes remission as a distinct prevention endpoint. While that publication established the definitional framework, our commentary argues for the clinical necessity of this target specifically for cardiovascular protection, extending the concept from diabetes prevention to direct CVD risk modification[5].
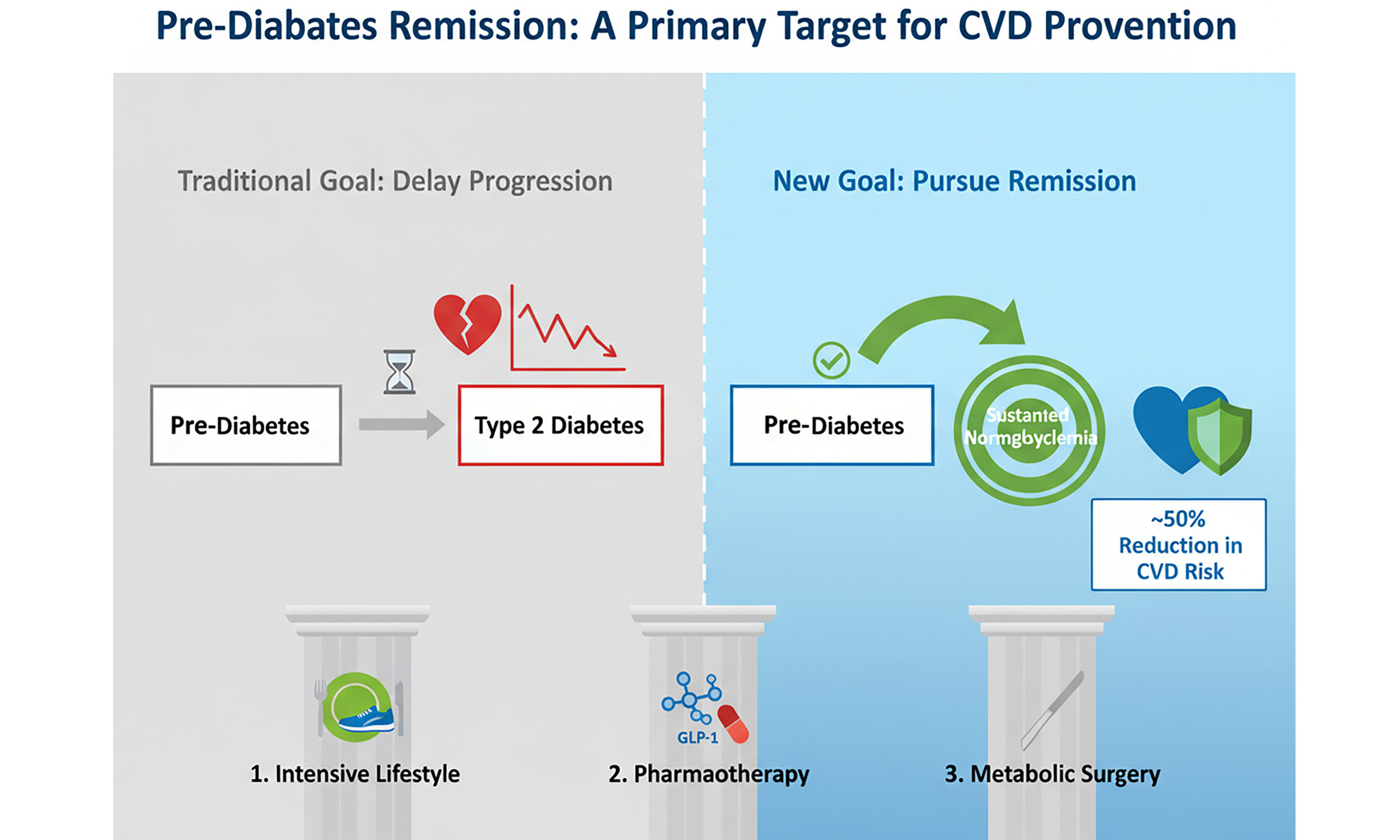
Historically, the clinical and research focus in pre-diabetes has been narrowly centered on preventing progression to type 2 diabetes (T2D). Seminal trials such as the Diabetes Prevention Program (DPP) and the Da Qing study unequivocally demonstrated that intensive lifestyle changes or metformin could reduce diabetes incidence by 31%-58%[6-9]. This “delay strategy” has justifiably formed the bedrock of prevention guidelines. However, a deeper interrogation reveals a critical nuance: a significant proportion of participants in these studies, particularly in the lifestyle arms, did not just delay diabetes but actually reversed their dysglycemia, reverting to normal glucose levels[10]. This phenomenon, which we term pre-diabetes remission, was often a secondary observation. We posit that remission is not merely a synonym for non-progression but represents a qualitatively superior metabolic state with distinct and potent cardiovascular protective effects[11]. In this commentary, we propose adapting the principles from T2D remission, defined as the achievement of an HbA1c level < 6.5%, sustained for at least 3 months after the cessation of glucose-lowering pharmacotherapy[12]. It signifies a fundamental reset of underlying metabolic derangements rather than a temporary pause in disease progression. The recent landmark harmonized analysis of the DPP Outcomes Study (DPPOS) and the Da Qing Diabetes Prevention Outcome Study provides the most powerful evidence to date, showing that remission is associated with a halving of cardiovascular mortality and heart failure risk over decades of follow-up[13]. This commentary synthesizes this emerging evidence, contending that the active pursuit of pre-diabetes remission must become a central, explicit goal in cardiovascular prevention.
THE PATHOPHYSIOLOGICAL IMPERATIVE: WHY PRE-DIABETES ACTIVELY DRIVES CARDIOVASCULAR DAMAGE
The elevated CVD risk in pre-diabetes is not passive but the direct result of a concerted pathogenic cascade. Remission can disrupt this cascade at its source, providing rationale for its cardioprotective effects.
At its epicenter lies insulin resistance in skeletal muscle, liver, and adipose tissue[14]. The resulting compensatory hyperinsulinemia is not merely a biomarker but a direct driver of cardiovascular injury[15]. It stimulates vascular smooth muscle cell proliferation and migration into the arterial intima, promotes renal sodium reabsorption (exacerbating hypertension), and, crucially, induces endothelial dysfunction by impairing the insulin-stimulated phosphoinositide 3-kinase-protein kinase B (PI3K-Akt) pathway, which reduces nitric oxide (NO) bioavailability[16]. This creates a pro-inflammatory, vasoconstrictive, and pro-atherogenic endothelial phenotype[17,18].
Even in pre-diabetes, pancreatic β-cells are under duress, exhibiting loss of first-phase insulin response and disrupted secretory pulsatility[19]. Concurrently, the incretin effect - the augmented insulin secretion after oral glucose intake - is blunted due to reduced sensitivity to glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP)[20,21]. This defect not only hampers glucose control but also deprives the cardiovascular system of the direct protective benefits of these hormones, such as GLP-1-mediated vasodilation and anti-inflammatory actions[22].
Concurrently, dysfunctional visceral adipose tissue acts as a potent endocrine organ. It secretes excessive pro-inflammatory cytokines [e.g., tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6)] and alters adipokine balance, reducing protective adiponectin. This ectopic fat deposition, particularly in the liver and around blood vessels, further exacerbates systemic insulin resistance and releases free fatty acids that contribute to hepatic gluconeogenesis and dyslipidemia. This state of chronic low-grade inflammation and associated oxidative stress further damages the endothelium, promotes the oxidation of low-density lipoprotein cholesterol (LDL-C), and fosters a prothrombotic environment. The resultant atherogenic dyslipidemia - elevated triglycerides, low high-density lipoprotein cholesterol (HDL-C), and a predominance of small, dense LDL particles - accelerates the formation of atherosclerotic plaque[23].
This pathogenic triad of insulin resistance/hyperinsulinemia, β-cell stress, and inflammation/dyslipidemia culminates in endothelial dysfunction, the seminal event in atherogenesis. In pre-diabetes, this is often clinically silent but detectable via increased carotid intima-media thickness or impaired flow-mediated dilation[24]. Achieving remission, particularly through mechanisms that significantly reduce ectopic and visceral fat mass, directly and simultaneously ameliorates each component of this triad, thereby restoring vascular homeostasis and halting the progression of subclinical atherosclerosis. Central to this restoration is the remarkable plasticity of pancreatic β-cell function. Research indicates that even in established T2D, a significant proportion of dysfunctional β-cells can recover their glucose-responsive insulin secretion under appropriate metabolic correction, underscoring that pre-diabetes and early diabetes represent a critical window for reversing β-cell stress through interventions that alleviate metabolic demand[25]. Normoglycemia restoration marks broader metabolic repair, extending beyond glucose to improve blood pressure, lipids, and inflammation - collectively reducing cardiovascular risk.
CLINICAL EVIDENCE MOSAIC: FROM SURROGATE MARKERS TO HARD OUTCOMES
While definitive, prospective trials with remission as a primary endpoint are awaited, a converging body of evidence strongly supports the cardiovascular benefits of achieving normoglycemia.
The most transformative evidence comes from a 2025 joint analysis of two landmark prevention trials. This study harmonized data from the U.S. DPPOS and the Chinese Da Qing Outcome Study, following participants for up to 30 years. Critically, the assessment methods and timing were robust: in the Da Qing study, glucose status was evaluated via a 75 g oral glucose tolerance test (OGTT) every two years during the active intervention, while the DPPOS utilized annual assessments of fasting plasma glucose and HbA1c, with OGTTs used to confirm diabetes status. The findings are unequivocal: individuals who achieved remission from pre-diabetes (reverting to normal glucose levels within the first 1-6 years of intervention) experienced a dramatic reduction in hard cardiovascular endpoints. Specifically, the adjusted hazard ratio for the composite of cardiovascular death or heart failure hospitalization was 0.41 in DPPOS and 0.49 in the Da Qing study, translating to an approximate 50%-60% risk reduction. This effect was sustained over decades, demonstrating a profound “legacy” or “metabolic memory” benefit. As lead author Andreas Birkenfeld noted, pre-diabetes remission could become “a fourth major primary prevention tool” alongside blood pressure control, cholesterol reduction, and smoking cessation[13].
Large observational cohorts consistently reinforce that the cardiovascular risk of pre-diabetes is malleable. Studies like the Whitehall II cohort show that individuals who regress from pre-diabetes to normoglycemia have a subsequent CVD risk profile equivalent to those who never had dysglycemia, effectively “resetting” their risk trajectory[26]. This risk normalization underscores the reversibility of early cardiometabolic damage. Moreover, this effect appears to be independent of subsequent weight regain, suggesting that the metabolic reset achieved during remission confers durable vascular protection. Furthermore, intervention studies using surrogate endpoints confirm the mechanistic plausibility. Intensive lifestyle programs leading to weight loss in pre-diabetes consistently demonstrate benefits such as slowed progression of carotid intima-media thickness, improved endothelial function measured by flow-mediated dilation, and reduced arterial stiffness[27]. Recent mechanistic evidence further elucidates these findings: among individuals achieving ≥ 5% weight loss, those who return to normoglycemia exhibit greater improvements in insulin sensitivity and more pronounced reductions in visceral adipose tissue compared to weight-matched individuals who remain dysglycemic - differences that translate to a substantially lower risk of progressing to T2D[28]. These improvements in insulin sensitivity and visceral adiposity are key determinants of microvascular health, including urinary albumin-to-creatinine ratio and small vessel integrity. Collectively, these improvements in vascular health provide a direct biological link between the process of achieving remission and the reduction of atherogenic burden.
THE THERAPEUTIC ARSENAL: A TIERED STRATEGY FOR INDUCING REMISSION
Inducing sustained remission requires interventions of sufficient potency to overcome insulin resistance and allow for metabolic recovery. The contemporary clinical toolkit now enables a personalized, stepped-care model.
The DPP protocol (≥ 7% weight loss via calorie reduction and ≥ 150 min/week of moderate activity) remains foundational, achieving remission in roughly 30% of participants. Recent studies from China have further validated the efficacy of low-calorie diets (around 815-835 kcal/day) and intermittent calorie restriction (e.g., 5 days of very low energy intake followed by 10 days of regular diet) in inducing T2D remission, with remission rates comparable to international data and significant improvements in medication reduction[29,30]. While these data originate from T2D populations, they support the metabolic reversibility principle relevant to pre-diabetes. The 2025 American College of Lifestyle Medicine guideline formalizes a holistic “Six Pillars” approach: nutrition, physical activity, stress management, sleep optimization, positive social connection, and avoidance of harmful substances[31]. It emphasizes that the magnitude of weight loss is a key predictor; remission rates correlate strongly with losing 5%-15% or more of body weight. This weight loss threshold is critical for mobilizing ectopic fat stores in the liver and pancreas, a biological “reset” that alleviates lipotoxicity and facilitates the restoration of β-cell function. Such findings suggest that remission represents a distinct physiological recovery - potentially reversing β-cell dedifferentiation - rather than merely being a proxy for weight reduction[32,33]. Dietary patterns emphasizing whole foods, low glycemic load, and healthy fats show particular promise in improving insulin sensitivity and promoting remission. Innovative, less-intensive models are also being explored, such as the 6-week “Project Health T2D” program, which uses cognitive dissonance and behavioral techniques to encourage sustainable habit change[34].
For many, lifestyle alone is insufficient. Newer agents with cardiovascular benefits are paradigm-shifting candidates, aligning with the European Association for the Study of Obesity (EASO) framework endorsing pharmacotherapy to achieve normoglycaemia in obesity and pre-diabetes[35]: (1) GLP-1 Receptor Agonists (GLP-1 RAs): Drugs such as semaglutide and liraglutide cause profound weight loss and glycemic improvement. A 2025 dynamic systematic review reaffirmed their cardiovascular benefits in T2D, including reduced mortality, myocardial infarction, stroke, and heart failure hospitalization[36]. Although this evidence base is primarily derived from T2D trials, their efficacy in achieving normoglycemia in obese individuals with pre-diabetes makes them a powerful remission-induction tool; (2) Sodium-Dependent Glucose Transporters 2 (SGLT2) Inhibitors: These agents offer glycemic control, weight loss, and unique benefits on heart failure hospitalization and renal outcomes[36]. Their cardioprotective role is established in T2D and heart failure populations, offering a rationale for consideration in pre-diabetic individuals with high heart failure risk; (3) Tirzepatide: This dual GIP/GLP-1 receptor agonist has demonstrated superior efficacy for both glycemic control and weight loss in T2D, suggesting it may offer the highest pharmacological remission rates for pre-diabetes to date[37]. Early SURMOUNT data show potential to induce normoglycemia in obesity with pre-diabetes[21]. These findings, while promising, largely extrapolate from T2D and obesity trials. The choice among these agents should be individualized, considering comorbidities, cost, and patient preferences. Combination strategies are also emerging. A multicenter study from China demonstrated that adding dapagliflozin to calorie restriction significantly improved remission rates and metabolic parameters (body fat, blood pressure, lipids) compared to placebo with calorie restriction alone, with a good safety profile[38].
For individuals with pre-diabetes and severe obesity [body mass index (BMI) ≥ 35 kg/m2] who have not achieved remission through intensive lifestyle or pharmacological interventions, metabolic surgery is the most effective remission-inducing therapy. While much of the existing evidence is derived from established T2D cohorts, recent data specifically involving individuals with pre-diabetes have demonstrated that metabolic surgery significantly reduces the long-term risk of developing T2D and major adverse cardiovascular events (MACE)[39]. Large registries show high remission rates for diabetes and its comorbidities, often preceding maximal weight loss due to profound hormonal changes[40]. Long-term data demonstrate sustained remission and significant reductions in cardiovascular mortality. Furthermore, its application as a primary prevention strategy must be weighed against surgical risks, high procedural costs, and challenges in patient access. However, real-world data, such as from the UK National Bariatric Surgery Registry, indicate it is often offered too late in the disease course, reducing its potential impact[41]. This highlights the need for timely referral as primary prevention in eligible individuals. A multidisciplinary team approach is essential.
DEFINING THE GOAL AND CHARTING THE FUTURE: AN AGENDA FOR RESEARCH AND IMPLEMENTATION
A major impediment to research and clinical integration is the lack of a consensus definition for pre-diabetes remission. It is important to note that definitions of pre-diabetes vary globally, and a unified consensus - similar to that achieved for T2D remission - is urgently needed. However, the definition has limitations in clinical practice. For instance, a real-world observational cohort study of 14,141 Italian T2D patients followed for four years showed that remission rates varied significantly depending on the stringency of the definition, with more relaxed criteria associated with higher remission rates and longer remission duration. Notably, patients in the intermediate definition group showed more significant cardiovascular risk improvement, suggesting the need to refine the traditional remission criteria[42]. A formal consensus statement from leading professional societies is urgently needed, alongside definitive prospective trials. The paramount question is: Does a strategy explicitly targeting sustained pre-diabetes remission reduce MACE? We advocate for: (1) comparative trials of multi-modal “remission-induction” vs. standard care in high-risk pre-diabetes, with co-primary endpoints of sustained remission and MACE; (2) omics and advanced imaging studies to define remission's “molecular signature” and its effects on vascular inflammation; (3) development of scalable, cost-effective care models ensuring equitable access across diverse populations; (4) research to identify biomarkers predicting individual likelihood of achieving remission with specific interventions. Implementation science must also address barriers to adoption in routine practice, including reimbursement policies, clinician education, and patient engagement strategies.
CONCLUSION AND FUTURE DIRECTIONS
The pursuit of pre-diabetes remission signifies a paradigm shift from defensive risk delay to active disease interception. The pathophysiological rationale is robust, therapeutic modalities are increasingly potent, and indirect clinical evidence is powerfully suggestive. By decisively intervening to restore normal glucose metabolism before irreversible vascular damage, we hold a critical opportunity to alter cardiovascular trajectories globally. Moving forward, focus must expand from diagnosing pre-diabetes to staging metabolic severity and implementing targeted remission strategies. Importantly, this approach complements - rather than replaces - established cardiovascular risk management. For the community dedicated to mitigating diabetic CVD, rigorously validating the remission paradigm may represent our most impactful path forward, promising to reduce individual suffering and alleviate the economic burden of cardiovascular complications. To enable scalability, digital health tools powered by artificial intelligence offer promise for sustaining behavioral change. The era of passive observation must yield to active metabolic restoration.
DECLARATIONS
Authors’ contributions
Conceptualization: Li Z, Li H
Methodology: Li Z, Gong H
Validation: Li Z, Li X
Investigation: Li X
Resources: Zhao A
Data curation: Wang W, Yan Y
Writing - original draft preparation: Li Z, Li H
Writing - review and editing: Yang H, Gao L
Supervision: Liu S, Li H
Project administration: Liu S, Li H
Funding acquisition: Liu S
All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by Collaborative Traditional Chinese and Modern Medicine for Chronic Disease Management Research Project (No. CXZH2024085); Research Topics in Chronic Disease Management for 2025 (No. GWJJMB202510024123); Shanxi Province Metabolic Disease (Type 1 Diabetes) Clinical Medical Research Center (No. 20240410501001); Shanxi Province Science and Technology Achievements Transformation Guidance Special Fund (No. 202304021301066); Shanxi Province Science and Technology Innovation Talent Team Special Plan (No. 202204051002029); Shanxi Province Research Funding for Returned Overseas Scholars (No. 2024-143); Shanxi Province Basic Research Program (No. 202303021212330); Shanxi Province Key Laboratory of Endocrine and Metabolic Diseases (No. 202404010920011).
Conflicts of interest
Liu S is the Guest Editor of the Special Issue entitled “Emerging Mechanisms and Therapeutic Strategies in Diabetic Cardiovascular Disease” in the journal Metabolism and Target Organ Damage. Liu S was not involved in any steps of editorial processing, notably including reviewers’ selection, manuscript handling and decision making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Rooney MR, He JH, Salpea P, et al. Global and regional prediabetes prevalence: updates for 2024 and projections for 2050. Diabetes Care. 2025;48:e142-4.
3. Cai X, Zhang Y, Li M, et al. Association between prediabetes and risk of all cause mortality and cardiovascular disease: updated meta-analysis. BMJ. 2020;370:m2297.
4. Wang Y, Sandforth A, Jumprtz-von Schwartzenberg R, et al. Lifestyle intervention is more effective in high 1-hour post-load glucose than in prediabetes for restoring β-cell function, reducing ectopic fat, and preventing type 2 diabetes. Metabolism. 2026;174:156430.
5. Birkenfeld AL, Perreault L, Schmidt MI, et al. Defining prediabetes remission as a distinct prevention endpoint. Lancet Diabetes Endocrinol. 2026;14:100-2.
6. Aroda VR, Knowler WC, Crandall JP, et al.; Diabetes Prevention Program Research Group. Metformin for diabetes prevention: insights gained from the Diabetes Prevention Program/Diabetes Prevention Program Outcomes Study. Diabetologia. 2017;60:1601-11.
7. Diabetes Prevention Program Research Group. Long-term effects of lifestyle intervention or metformin on diabetes development and microvascular complications over 15-year follow-up: the Diabetes Prevention Program Outcomes Study. Lancet Diabetes Endocrinol. 2015;3:866-75.
8. Lindström J, Peltonen M, Eriksson JG, et al.; Finnish Diabetes Prevention Study (DPS). Improved lifestyle and decreased diabetes risk over 13 years: long-term follow-up of the randomised Finnish Diabetes Prevention Study (DPS). Diabetologia. 2013;56:284-93.
9. Tuomilehto J, Lindström J, Eriksson JG, et al.; Finnish Diabetes Prevention Study Group. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N Engl J Med. 2001;344:1343-50.
10. Lindström J, Louheranta A, Mannelin M, et al.; Finnish Diabetes Prevention Study Group. The Finnish Diabetes Prevention Study (DPS): lifestyle intervention and 3-year results on diet and physical activity. Diabetes Care. 2003;26:3230-6.
11. Chen Y, Xu L, Cheng Z, et al. Progression from different blood glucose states to cardiovascular diseases: a prospective study based on multi-state model. Eur J Prev Cardiol. 2023;30:1482-91.
12. Khunti K, Papamargaritis D, Aroda VR, Anjana RM, Kashyap SR. Re-evaluating the concept of remission in type 2 diabetes: a call for patient-centric approaches. Lancet Diabetes Endocrinol. 2025;13:615-34.
13. Vazquez Arreola E, Gong Q, Hanson RL, et al. Prediabetes remission and cardiovascular morbidity and mortality: post-hoc analyses from the Diabetes Prevention Program Outcome study and the DaQing Diabetes Prevention Outcome study. Lancet Diabetes Endocrinol. 2026;14:137-48.
14. Suh YH, Kim Y, Bang JH, et al. Analysis of gene expression profiles in insulin-sensitive tissues from pre-diabetic and diabetic Zucker diabetic fatty rats. J Mol Endocrinol. 2005;34:299-315.
15. Ormazabal V, Nair S, Elfeky O, Aguayo C, Salomon C, Zuñiga FA. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc Diabetol. 2018;17:122.
16. Yu C, Wang Z, Han Y, et al. Dopamine D4 receptors inhibit proliferation and migration of vascular smooth muscle cells induced by insulin via down-regulation of insulin receptor expression. Cardiovasc Diabetol. 2014;13:97.
17. Limberg JK, Smith JA, Soares RN, et al. Sympathetically mediated increases in cardiac output, not restraint of peripheral vasodilation, contribute to blood pressure maintenance during hyperinsulinemia. Am J Physiol Heart Circ Physiol. 2020;319:H162-70.
18. Muniyappa R, Chen H, Montagnani M, Sherman A, Quon MJ. Endothelial dysfunction due to selective insulin resistance in vascular endothelium: insights from mechanistic modeling. Am J Physiol Endocrinol Metab. 2020;319:E629-46.
19. Liu Y, Zeng Y, Miao Y, et al. Relationships among pancreatic beta cell function, the Nrf2 pathway, and IRS2: a cross-sectional study. Postgrad Med. 2020;132:720-6.
20. Cariou B. Harnessing the incretin system beyond glucose control: potential cardiovascular benefits of GLP-1 receptor agonists in type 2 diabetes. Diabetes Metab. 2012;38:298-308.
21. Mari A, Stefanski A, van Raalte DH, et al. Tirzepatide treatment and associated changes in β-cell function and insulin sensitivity in people with obesity or overweight with prediabetes or normoglycemia: a post hoc analysis from the SURMOUNT-1 trial. Diabetes Care. 2025;48:1622-7.
22. Alharbi SH. Anti-inflammatory role of glucagon-like peptide 1 receptor agonists and its clinical implications. Ther Adv Endocrinol Metab. 2024;15:20420188231222367.
23. Zieleniewska N, Jamiołkowski J, Kondraciuk M, et al. Preclinical atherosclerosis and prediabetes: a cross-sectional metabolic assessment in apparently healthy population. Cardiovasc Diabetol. 2025;24:280.
24. Zhang J, Song X, Zhang G, Chen S, Zhao X, Zhang Q. Glycemic control and asymptomatic carotid plaque and carotid intima-media thickness. BMJ Open Diabetes Res Care. 2025;13:e004786.
25. Suleiman M, Sawatani T, Tesi M, et al. Functional recovery of islet β cells in human type 2 diabetes: transcriptome signatures unveil therapeutic approaches. Sci Adv. 2025;11:eads2905.
26. Vistisen D, Kivimäki M, Perreault L, et al. Reversion from prediabetes to normoglycaemia and risk of cardiovascular disease and mortality: the Whitehall II cohort study. Diabetologia. 2019;62:1385-90.
28. Sandforth A, von Schwartzenberg RJ, Arreola EV, et al. Mechanisms of weight loss-induced remission in people with prediabetes: a post-hoc analysis of the randomised, controlled, multicentre Prediabetes Lifestyle Intervention Study (PLIS). Lancet Diabetes Endocrinol. 2023;11:798-810.
29. Liu Z, Feng N, Wang S, et al. Low-calorie diets and remission of type 2 diabetes in Chinese: phenotypic changes and individual variability. Nutr J. 2025;24:42.
30. Xiao Z, Yin X, Lai X, et al. Real-world effect of intermittent calorie-restricted diet on type 2 diabetes remission: a dual-cohort retrospective study. Front Nutr. 2025;12:1648314.
31. Rosenfeld RM, Grega ML, Karlsen MC, et al. Lifestyle interventions for treatment and remission of type 2 diabetes and prediabetes in adults: A Clinical Practice Guideline From the American College of Lifestyle Medicine. Am J Lifestyle Med. 2025;19:964-96.
32. Adapa S, Fahey P, Piya M, Birhanu F, Atlantis E. Effect of clinically significant weight loss on remission of metabolic complications: a rapid review and meta-analysis of comparative controlled studies. Obes Rev. ;2026:e70091.
33. Taylor R. Calorie restriction for long-term remission of type 2 diabetes. Clin Med. 2019;19:37-42.
34. Stice E, Rohde P, Desjardins CD, Gee K, Shaw H, Kim S. A preliminary randomized trial of a brief dissonance-based type 2 diabetes prevention programme for adults with pre-diabetes. Diabetes Obes Metab. 2025;27:1572-9.
35. McGowan B, Ciudin A, Baker JL, et al. Framework for the pharmacological treatment of obesity and its complications from the European Association for the Study of Obesity (EASO). Nat Med. 2025;31:3229-32.
36. Nong K, Jeppesen BT, Shi Q, et al. Medications for adults with type 2 diabetes: a living systematic review and network meta-analysis. BMJ. 2025;390:e083039.
37. Nauck MA, D’Alessio DA. Tirzepatide, a dual GIP/GLP-1 receptor co-agonist for the treatment of type 2 diabetes with unmatched effectiveness regrading glycaemic control and body weight reduction. Cardiovasc Diabetol. 2022;21:169.
38. Liu Y, Chen Y, Ma J, et al. Dapagliflozin plus calorie restriction for remission of type 2 diabetes: multicentre, double blind, randomised, placebo controlled trial. BMJ. 2025;388:e081820.
39. Sandforth L, Raverdy V, Sandforth A, et al. Subphenotype-dependent benefits of bariatric surgery for individuals at risk for type 2 diabetes. Diabetes Care. 2025;48:996-1006.
40. Grieco A, Huffman KM, Cohen ME, Hall BL, Morton JM, Ko CY. Metabolic and bariatric surgery accreditation and quality improvement program: bariatric surgical risk/benefit calculator: 1-year comorbidity remission. Surg Obes Relat Dis. 2024;20:275-82.
41. Currie A, Bolckmans R, Askari A, et al. Bariatric-metabolic surgery for NHS patients with type 2 diabetes in the United Kingdom National Bariatric Surgery Registry. Diabet Med. 2023;40:e15041.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].