Definition, prevalence, and healthcare burden of MetALD - a comprehensive assessment of epidemiology from the east and west
Abstract
Steatotic liver disease (SLD) encompasses a spectrum of liver diseases that span from predominantly metabolic dysfunction-associated SLD, a combination of both metabolic and alcohol-related SLD (MetALD), to those driven predominantly by excessive alcohol use (e.g., alcohol-associated liver disease). The epidemiology of MetALD is not well described and reflects challenges in accurate alcohol consumption reporting and inconsistencies in diagnostic criteria. Existing data estimate the global prevalence of MetALD to range from 2% to 8%, though it is likely underestimated due to variability in alcohol consumption patterns and metabolic risk factors across regions. MetALD is associated with more severe clinical outcomes, including advanced liver fibrosis and increased cardiovascular risk, necessitating early identification and intervention. However, further research is needed that focuses on long-term, population-based studies including objective biomarkers of alcohol consumption, such as phosphatidylethanol, and assessing disease progression using non-invasive methods to accurately categorize disease severity in MetALD. In this review, we provide an update on the epidemiology and healthcare burden of MetALD across Western populations and the Asia-Pacific region.
Keywords
INTRODUCTION
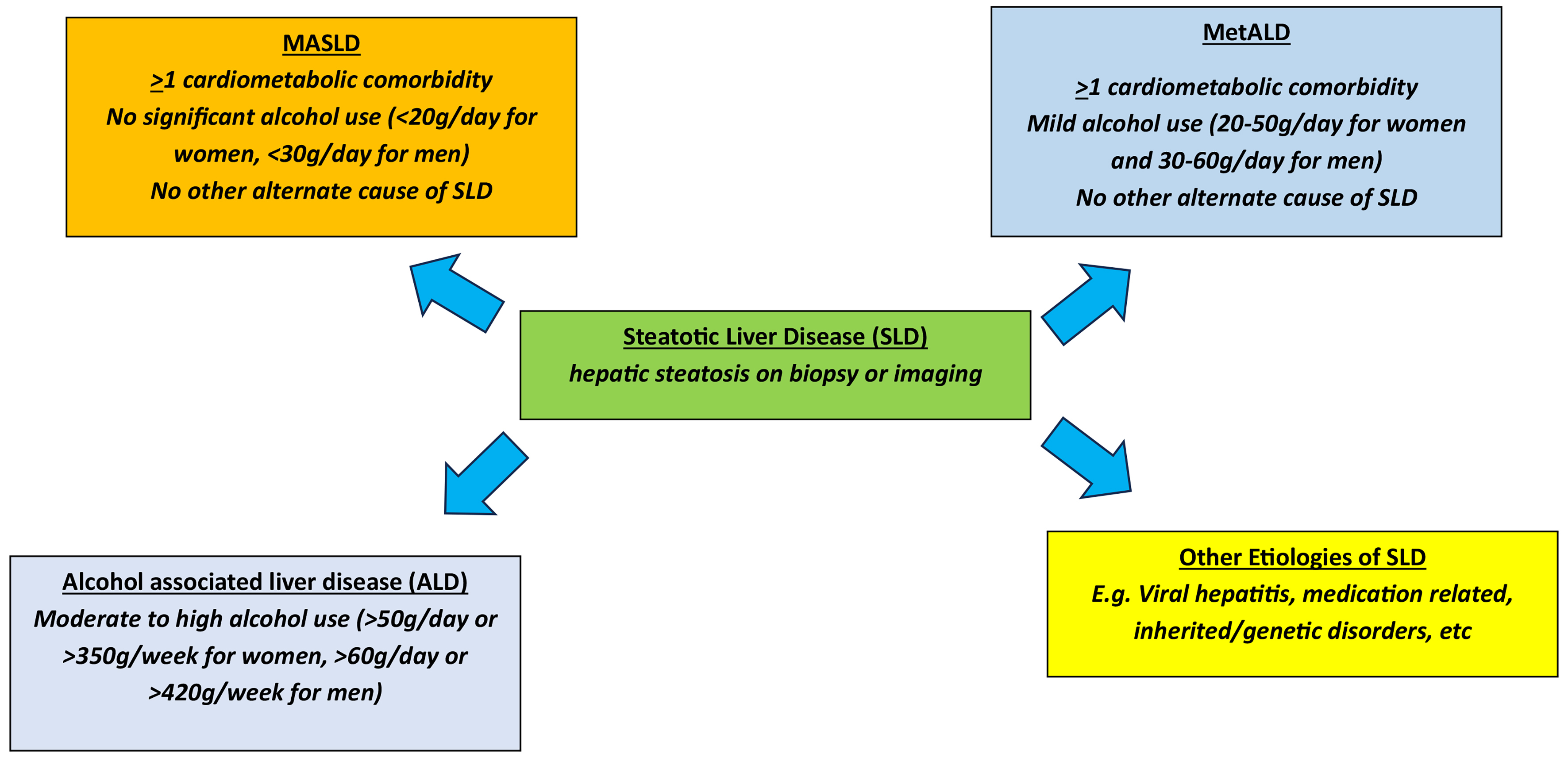
Steatotic liver disease (SLD) is a leading cause of chronic liver disease worldwide[1-4]. SLD encompasses a spectrum of liver diseases, ranging from conditions predominantly driven by metabolic comorbidities to those driven predominantly by excessive alcohol use. These span from metabolic dysfunction-associated steatotic liver disease (MASLD) to alcohol-associated liver disease (ALD). SLD also includes other miscellaneous etiologies of steatosis (e.g., hepatitis C virus infection, human immunodeficiency virus infection, celiac disease, Wilson disease, lysosomal acid lipase deficiency)[5,6]. Many patients have risk factors for both MASLD and ALD. Reflecting this, updated SLD nomenclature categorizes individuals who have both metabolic and alcohol-related risk factors as metabolic and alcohol-associated liver disease (MetALD) [Figure 1][5-7]. Despite this classification, the epidemiology of MetALD is not well described, largely due to challenges in accurate alcohol consumption reporting and inconsistencies in diagnostic criteria. In this review, we provide an updated assessment of the existing literature describing the epidemiology of MetALD, with a focus on Western populations and the Asia-Pacific region.
DEFINITION OF METALD
MetALD is defined by presence of hepatic steatosis, > 1 cardiometabolic risk factor (CMRF) and mild levels of alcohol use. CMRF include the presence of overweight or obesity [defined as body mass index (BMI)
EPIDEMIOLOGY OF METALD IN THE ASIA-PACIFIC REGION
MetALD is emerging as a distinct clinical entity with significant implications for both liver and cardiovascular health in the Asia-Pacific region. While the prevalence of MetALD in various cohorts ranges from 2% to 7.9%, the true extent of the disease is likely underestimated due to limited data and inconsistencies in reporting alcohol use among individuals with SLD[12-14]. Most cases of SLD in the region are attributed to MASLD; however, emerging data suggest that MetALD may be more prevalent than previously understood, with key studies shedding light on its epidemiology, clinical characteristics, and outcomes. In a Chinese cohort of 361 biopsy-confirmed SLD, 7.5% were diagnosed with MetALD[12]. These patients were predominantly male and exhibited more severe hepatic and renal dysfunction, evidenced by significantly elevated levels of γ-glutamyl transferase (γ-GT), creatinine, and blood urea nitrogen, compared to other forms of SLD. Similarly, in a population-based survey of 7,388 adults in Guangzhou, China, 7.3% were diagnosed with MetALD, while MASLD accounted for 30.3%[13]. MetALD patients exhibited higher metabolic risk and more pronounced hepatic injury compared with cryptogenic cases or patients meeting the MASLD definition but not the metabolic dysfunction-associated fatty liver disease (MAFLD) definition. In a Japanese health checkup cohort of 694 adults, MASLD accounted for 26% of participants, while MetALD represented 2%. These patients exhibited higher γ-GT levels, lower platelet counts, and higher FIB-4 scores compared with those with MASLD, indicating more advanced hepatic fibrosis[14]. Furthermore, a larger Japanese cohort of 15,788 adults undergoing annual health check-ups found MetALD to represent 5% of cases, with MASLD at 26.8%[15]. Another study from Korea included a health check-up cohort of 844 adults assessed by Magnetic Resonance Imaging-Proton Density Fat Fraction (MRI-PDFF) and found that 3.7% had MetALD, with these patients being predominantly male, younger, and presenting with higher serum aspartate aminotransferase (AST), γ-GT, and triglyceride levels compared to MASLD patients[16]. In a Korean primary care cohort of 2,535 adults, it was further confirmed that MetALD patients had a higher hepatic fibrosis burden, assessed via magnetic resonance elastography, compared to those with MASLD or ALD[17]. A recent study in Taiwan evaluated 332,175 individuals who participated in health screening programs between 1997 and 2013[11]. After a median follow-up of 16 years, the authors noted distinct risk profiles with respect to long-term risks of cirrhosis or HCC, which were lowest among MASLD and highest among ALD. For example, the incidence of cirrhosis was 95.4, 145.6, and 282.0 per 100,000 person-years in MASLD, MetALD, and ALD, respectively. Among non-cirrhotic populations, the incidence of HCC was 30.7, 52.7, and 69.0 per 100,000 person-years in MASLD, MetALD, and ALD, respectively[11].
Despite its lower prevalence, MetALD is associated with worse outcomes in terms of both liver-related and systemic complications. In the Ansung-Ansan Korean cohort (n = 9,497), 38.3% had MASLD and 4.5% had MetALD[18]. When evaluating long-term outcomes over a median follow-up of 17.5 years, MetALD patients exhibited a higher risk of advanced fibrosis [hazards ratio (HR): 1.75, 95% confidence interval (CI): 1.38-2.23] compared with MASLD patients. A nationwide Japanese cohort of 761,400 patients from the Japan Medical Data Center claims database found that MetALD patients had intermediate risks of liver-related events (HR: 1.42, 95%CI: 1.1-1.8) and all-cause mortality (HR: 1.13, 95%CI: 0.98-1.3) compared to those with MASLD or ALD[19]. Additionally, in a Taiwanese cohort of 332,175 adults with a median follow-up of
In addition to liver damage, MetALD is also associated with increased cardiovascular risk. A longitudinal cohort study in South Korea of 13,811 adults showed that the annual progression rate of coronary artery calcium (CAC) was higher in MetALD patients (7% increase) compared to those with MASLD (3% increase)[20]. In a nationwide Korean cohort of 9.78 million adults, MetALD accounted for 4.4% of participants with an increased risk of cardiovascular disease (HR: 1.28, 95%CI: 1.26-1.30) compared to those without MASLD or related SLD[21]. Patients with MetALD in the Ansung-Ansan Korean cohort also exhibited a higher risk of cardiovascular disease (HR: 1.88, 95%CI: 1.33-2.65) compared with those with MASLD[18]. Interestingly, the cardiovascular outcomes of MetALD appear to differ in Japan. In the same nationwide Japanese cohort (n = 761,400) mentioned earlier, MetALD patients exhibited a lower incidence of major adverse cardiovascular events compared to those with MASLD (HR: 0.68, 95%CI: 0.63-0.73)[19]. This may be due to factors such as moderate alcohol consumption, a diet rich in fish and vegetables, and Japan’s advanced healthcare system, which ensures early screening and effective management of cardiovascular risk factors.
MetALD, while increasingly recognized as a distinct clinical entity in the Asia-Pacific region, faces significant challenges in both its evaluation and research. One major difficulty lies in the wide variability in alcohol consumption patterns across countries. Different nations in the region consume a diverse range of alcoholic beverages, each with varying alcohol content. For example, China commonly consumes high-proof spirits such as baijiu and beer. In contrast, in Korea, people drink a mix of soju, beer, and other alcoholic beverages, often consumed together. Japan, on the other hand, favors beverages such as sake and shochu. These differences in alcohol types and strengths complicate the assessment of alcohol intake, making it difficult to standardize measurements, which may lead to inconsistencies in reporting. This hampers the ability to accurately determine alcohol’s role in MetALD across populations. In addition, there are differences in diagnostic criteria, healthcare infrastructure, and lifestyle habits, all of which further complicate the region’s ability to accurately assess the prevalence and impact of MetALD. While countries such as China, Korea, and Japan have extensive studies, there is a notable lack of data from other regions of the Asia-Pacific, including Southeast Asia, South Asia and Oceania countries. These regions, with their unique genetic, cultural, and healthcare landscapes, are underrepresented in current research, limiting the development of region-specific strategies for MetALD management and prevention. To effectively address the growing burden of MetALD, it is essential to overcome these challenges and bridge existing research gaps. This requires a more unified approach to alcohol consumption assessment, greater regional collaboration, and expanded studies in underrepresented populations. Additionally, a comprehensive understanding of the disease’s impact on both hepatic and extrahepatic systems, particularly cardiovascular health and cancer risks, is needed to improve clinical outcomes and inform public health strategies in the Asia-Pacific region.
EPIDEMIOLOGY OF METALD AMONG WESTERN POPULATIONS
SLD remains a leading cause of chronic liver disease among Western populations[2-4,17,22-24]. While the majority of individuals with SLD are due to MASLD, there are emerging data that suggests that MetALD and ALD prevalence may be more prevalent than previously understood[3,4,22,24]. The true prevalence of MetALD is likely underestimated due to limited data and under-reporting on alcohol use among individuals with SLD. Two recent studies in the U.S. utilized 2017-2020 data from the National Health and Nutrition Examination Survey to understand the prevalence of MetALD[3,4]. Overall, the prevalence of SLD was 37.87%, the vast majority of which was due to MASLD (32.45% MASLD, 2.56% MetALD, 1.17% ALD, and 1.46% other etiologies)[3,4]. Another similar study that spanned more recent time periods from 2017 to 2023, defining steatosis with controlled attenuation parameter measurements of > 280 dB/m, reported similar findings. Age-standardized prevalence of MASLD was 32.42%, of MetALD 2.20%, and of ALD 1.29%[24]. However, both studies utilized a similar database. While data on alcohol use were available from survey questionnaires, heterogeneity in the surveys across study periods, lack of granularity, and potential reporting bias (e.g., social desirability bias) may have contributed to the underestimation of alcohol use and, consequently, the prevalence of MetALD or ALD. Under-reporting of alcohol is a major limitation to accurate classification and estimation of MetALD or ALD prevalence. The drivers of alcohol under-reporting are multi-factorial and may reflect psychological, social, religious, and cultural factors that influence an individual’s motivation to disclose alcohol use patterns[25]. This further emphasizes the need for wider adoption of objective measures of alcohol use such as phosphatidylethanol (PEth) testing.
Among Western populations, some differences have been observed between American and European populations. For example, aforementioned studies in the U.S. based on data from National Health and Nutrition Examination Survey (NHANES) have reported MetALD prevalence from 2.20%-2.56%, representing approximately 6%-7% of the SLD population[3,4]. However, studies among European populations have reported the proportion of SLD represented by MetALD ranging from 2% to 24%[26-28]. While these discrepancies may reflect geographical differences in prevalence of metabolic comorbidities as well as drinking patterns, these differences may also reflect the limited availability of data to accurately assess MetALD epidemiology, especially as it relates to accurate assessments of alcohol use.
A recent study among U.S. Veterans reported much higher prevalence of MetALD than aforementioned NHANES studies. Wong et al. evaluated longitudinal data on all adult Veterans receiving care at Veterans Affairs healthcare facilities in the U.S. Presence of hepatic steatosis was identified using the hepatic steatosis index > 36[22]. Longitudinal alcohol assessment based on the Alcohol Use Disorders Identification Test Concise (AUDIT-C) scores was available for inclusion in the analyses. MASLD was defined as the presence of hepatic steatosis and > 1 concurrent CMRF in patients with no alcohol use. MetALD was defined as the presence of hepatic steatosis and > 1 CMRF in patients with low-risk alcohol use (AUDIT-C = 1-2 for women and 1-3 for men). ALD was defined as presence of hepatic steatosis and high-risk alcohol use (AUDIT-C > 3 for women and > 4 for men)[6,22]. Among 6,192,643 Veterans, the study identified 534,494 (8.6%) with MASLD, 356,619 (5.8%) with MetALD, and 119,422 (1.9%) with ALD, corresponding to a prevalence of 8,631 per 100,000 persons for MASLD, 5,759 per 100,000 persons for MetALD, and 1,928 per 100,000 persons for ALD. Among this study of Veterans with SLD, 35% were categorized as MetALD[22]. Understanding differences in SLD subtypes is important given existing data demonstrating differences in long-term risks of cirrhosis or HCC[11,29].
Another important factor contributing to heterogeneity in assessing prevalence of MetALD is the dynamic nature of SLD subtypes. Across the SLD spectrum, patients often experience changes in longitudinal alcohol use over time (both increases and decreases) and may also have variations in the optimal management of metabolic comorbidities. Particularly for alcohol use, sustained changes in baseline level of alcohol use can lead to transitioning from different subtypes of SLD depending on the amount and pattern of alcohol use. Alcohol use data are challenging to collect. In studies that evaluate alcohol use, data are often limited or missing, which can lead to inaccurate classification or reliance on surrogate questions or markers that do not allow for more granular assessment, such as average daily or weekly consumption. A recent study by Israelsen et al. prospectively evaluated individuals in the general population at risk for SLD and followed them longitudinally for 2 years to assess the robustness of SLD diagnosis[28]. Among 1,042 participants, 595 (57%) had SLD. Of these, 371 (62%) met the criteria for MASLD, 140 (24%) for MetALD, and 83 (14%) for ALD. Over a median follow-up of 25 months, 38% of the initial 1,042 changed their SLD classification[28]. For example, among participants who did not have SLD at baseline, 25% developed SLD during follow-up. In contrast, 33% of participants classified as having SLD at baseline no longer met the criteria for SLD during follow-up. Among those with MASLD, 32% were reclassified during follow-up, with 4.0% transitioning to MetALD. Among those with MetALD at baseline, only 37.9% remained in the MetALD state. Among those with baseline ALD, 16.9% transitioned to MetALD during follow-up[28]. These data and others emphasize the importance of continued monitoring of metabolic comorbidities and alcohol use longitudinally to improve accurate assessment of classification of SLD subtypes.
The rationale for accurately classifying individuals with MetALD is the hypothesis that long-term outcomes differ compared with those of MASLD or ALD populations. Data from a national cohort of Veterans recently demonstrated no significant difference in risks of cirrhosis between MASLD and MetALD. Among 682,274 adults with MASLD and 517,464 with MetALD over a median follow-up of 7.2 years, incidence of cirrhosis was 0.43 per 100 person-years among MASLD, 0.39 per 100 person-years in MetALD, and 0.66 per 100 person-years in ALD[29]. Sripongpun et al. utilized data from the NHANES III dataset and observed similar 20-year survival probabilities between MASLD (69.7%) and MetALD (69.1%), both of which were significantly greater than observed among individuals with ALD (64.9%)[30]. More long-term studies are needed to better understand the epidemiology of MetALD in light of the aforementioned limitations of existing studies.
SEX DIFFERENCES IN METALD EPIDEMIOLOGY
Given the aforementioned limited data describing overall MetALD epidemiology, an accurate understanding of how MetALD prevalence may differ by sex has not been well described. A recent systematic review and meta-analysis evaluated 32 studies that provided data to estimate MetALD prevalence globally[31]. Overall, among the SLD population, the authors estimated a pooled prevalence of 10% for MetALD (95%CI: 7-13). When specifically evaluating differences in MetALD prevalence by sex, they reported a significantly higher pooled prevalence in men (86%, 95%CI: 79-91) compared with women (14%, 95%CI: 9-21), P < 0.01[31]. However, the majority of studies included in the analyses were from the Asia-Pacific regions.
EXISTING GAPS IN METALD
Despite the availability of epidemiological data thus far, important gaps remain in our understanding of the burden of MetALD. Future studies that incorporate longitudinal measures of alcohol use and employ objective biomarkers (e.g., PEth) will be able to more accurately assess MetALD burden while capturing real-world disease state transitions that occur across SLD subtypes. While not the focus of the current review, disease severity assessment with non-invasive markers (e.g., elastography, blood-based biomarkers) is becoming increasingly important and utilized in the standard care of patients with SLD. However, the utilization and interpretation of existing non-invasive measurements of disease severity have been mostly validated in MASLD populations, and whether similar thresholds for categorizing disease severity in MASLD can be uniformly applied to MetALD populations remains unclear.
LIMITATIONS AND FUTURE DIRECTIONS
Many unmet needs and evidence gaps remain with respect to MetALD [Table 1]. First and foremost, more accurate data on alcohol use, particularly with objective measures such as PEth testing, are needed to better understand the true epidemiology of MetALD. Reliance on surrogate measures such as self-reported alcohol use may underestimate the true burden and contribute to misclassification bias. This also leads to an inability to accurately understand differences in MetALD incidence and prevalence by geographical regions, sociodemographics, and socioeconomics. Secondly, more data is needed to understand how the synergy of metabolic comorbidities with alcohol use affects long-term outcomes and whether this varies by populations with different underlying genetics and risk factors. In addition, further research is needed to determine how treatment approaches for patients with MetALD should be optimized, and whether treatment responses to therapies used for MASLD/metabolic dysfunction-associated steatohepatitis (MASH) differ in the MetALD population. For example, the class for Glucagon-Like Peptide-1 agonist (GLP-1a) therapies is effective for treatment of metabolic comorbidities such as obesity and diabetes[32,33], and semaglutide has now been approved for MASH with stage F2/F3 fibrosis in the U.S.[34]. There are also emerging data that utilization of GLP-1a therapies has been associated with a reduction in alcohol use[35,36]. Hence, the class of GLP-1a-based therapies may be particularly useful in patients with MetALD, and more research is needed to understand how utilization of GLP-1a therapies may be optimized in MetALD populations.
Future directions for MetALD research
| · Larger, diverse population-based research to understand epidemiology and natural history of MetALD |
| · Routine assessment of objective alcohol biomarkers (e.g., PEth) to allow more accurate and consistent assessment of alcohol use and identification of MetALD |
| · Better understanding how synergy of metabolic comorbidities and alcohol use patterns affects long-term patient outcomes and whether this synergy differs depending on varying genetics and co-morbid risk factors |
| · Research to understand whether existing therapies used for MASH have equal effectiveness in MetALD populations and what factors influence response to treatment |
| · Exploring the role of GLP-1a-based therapies specifically for MetALD and impact on long-term outcomes, given emerging data on GLP-1a therapies may have a beneficial impact in reducing alcohol use in addition to its validated benefits on optimizing metabolic comorbidities and treatment of MASH |
CONCLUSION
MetALD, characterized by the coexistence of metabolic dysfunction and moderate alcohol consumption, is increasingly recognized as a distinct clinical entity with significant implications for both liver and systemic health. While several studies from East Asia and Western populations provide valuable insights, the true global burden of MetALD is likely underestimated due to challenges in accurate alcohol consumption reporting and inconsistencies in diagnostic criteria. MetALD patients often exhibit more severe liver damage, as well as increased risks of liver-related events and cardiovascular diseases compared to those with MASLD. The shared clinical features of MetALD highlight its systemic nature, which extends beyond liver-related complications. Future research should focus on long-term, population-based studies that include objective biomarkers of alcohol consumption, such as PEth, and assess disease progression using non-invasive methods to accurately categorize disease severity in MetALD. Moreover, there is an urgent need for more robust studies in underrepresented regions to better inform region-specific prevention, management strategies, and public health policies. Addressing these gaps will be essential for improving clinical outcomes and mitigating the growing burden of MetALD worldwide.
TAKE HOME POINTS
1. The global prevalence of MetALD ranges from 2% to 8%, though it is likely underestimated due to variability in alcohol consumption patterns and metabolic risk factors across regions.
2. MetALD represents a distinct clinical phenotype that combines metabolic dysfunction and mild alcohol use, requiring tailored approaches for management.
3. MetALD is associated with more severe clinical outcomes, including advanced liver fibrosis and increased cardiovascular risk, necessitating early identification and intervention.
4. Challenges in MetALD research include limited and inconsistent data, difficulty in accurately measuring alcohol use, and complexities in classifying disease subtypes across diverse populations.
DECLARATIONS
Authors’ contributions
Conception and design of the manuscript: Zhou XD, Zheng MH, Wong RJ
Analysis and interpretation of data: Zhou XD, Zheng MH, Wong RJ
Drafted the manuscript: Zhou XD, Zheng MH, Wong RJ
Revision of the manuscript for final submission: Zhou XD, Zheng MH, Wong RJ
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interests
Zheng MH is an Editorial Board Member of Metabolism and Target Organ Damage. Wong RJ is also an Editorial Board Member of Metabolism and Target Organ Damage. Zheng MH serves as a speaker for AstraZeneca, Hisky Medical Technologies, and Novo Nordisk; as a consultant for Boehringer Ingelheim and Eieling Technology; and has received consulting fees from Boehringer Ingelheim. Wong RJ reports receiving research grants (to his institution) from Gilead Sciences, Madrigal Pharmaceuticals, Exact Sciences, Theratechnologies, and Durect Corporation unrelated to this work. He also serves as a consultant (without compensation) to Gilead Sciences, Salix Pharmaceuticals, GSK, and Mallinckrodt Pharmaceuticals. Neither had any involvement in the review or editorial process of this manuscript, including reviewer selection, evaluation, or the final decision. Zhou XD has declared no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Ho GJK, Tan FXN, Sasikumar NA, et al. High global prevalence of steatotic liver disease and associated subtypes: a meta-analysis. Clin Gastroenterol Hepatol. 2025;23:2423-32.e1.
2. Younossi ZM, Golabi P, Price JK, et al. The global epidemiology of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among patients with type 2 diabetes. Clin Gastroenterol Hepatol. 2024;22:1999-2010.e8.
3. Lee BP, Dodge JL, Terrault NA. National prevalence estimates for steatotic liver disease and subclassifications using consensus nomenclature. Hepatology. 2024;79:666-73.
4. Kalligeros M, Vassilopoulos A, Vassilopoulos S, Victor DW, Mylonakis E, Noureddin M. Prevalence of steatotic liver disease (MASLD, MetALD, and ALD) in the United States: NHANES 2017-2020. Clin Gastroenterol Hepatol. 2024;22:1330-2.e4.
5. Rinella ME, Neuschwander-Tetri BA, Siddiqui MS, et al. AASLD Practice Guidance on the clinical assessment and management of nonalcoholic fatty liver disease. Hepatology. 2023;77:1797-835.
6. Rinella ME, Lazarus JV, Ratziu V, et al.; NAFLD Nomenclature consensus group. A multisociety Delphi consensus statement on new fatty liver disease nomenclature. Hepatology. 2023;78:1966-86.
7. Gao B, Arab JP, Liangpunsakul S, et al. Metabolic dysfunction and alcohol-associated liver disease (MetALD). eGastroenterology. 2025;3:e100319.
8. Yeoh A, Yang Z, Cheung R, Do A, Ahmed A, Wong RJ. Incidence of cirrhosis and hepatocellular carcinoma among veterans with noncirrhotic metabolic dysfunction-associated fatty liver disease. J Clin Gastroenterol. 2024;58:718-25.
9. Wong RJ, Yang Z, Cheung R, et al. Impact of longitudinal alcohol use patterns on long-term risk of cirrhosis among US veterans with steatotic liver disease. Gastroenterology. 2024;166:1156-65.e4.
10. Rivera-Esteban J, Muñoz-Martínez S, Higuera M, et al. Phenotypes of metabolic dysfunction-associated steatotic liver disease-associated hepatocellular carcinoma. Clin Gastroenterol Hepatol. 2024;22:1774-89.e8.
11. Chen YT, Chen TI, Yang TH, et al. Long-term risks of cirrhosis and hepatocellular carcinoma across steatotic liver disease subtypes. Am J Gastroenterol. 2024;119:2241-50.
12. Chen L, Tao X, Zeng M, Mi Y, Xu L. Clinical and histological features under different nomenclatures of fatty liver disease: NAFLD, MAFLD, MASLD and MetALD. J Hepatol. 2024;80:e64-6.
13. Wu T, Ye J, Mo S, et al. Impact of nomenclature as metabolic associated steatotic liver disease on steatotic liver disease prevalence and screening: a prospective population survey in Asians. J Gastroenterol Hepatol. 2024;39:1636-47.
14. Miwa T, Tajirika S, Imamura N, et al. Prevalence of steatotic liver disease based on a new nomenclature in the japanese population: a health checkup-based cross-sectional study. J Clin Med. 2024;13:1158.
15. Mori K, Akiyama Y, Tanaka M, et al. Deciphering metabolic dysfunction-associated steatotic liver disease: insights from predictive modeling and clustering analysis. J Gastroenterol Hepatol. 2024;39:1382-93.
16. Park HJ, Lee S, Lee JS. Differences in the prevalence of NAFLD, MAFLD, and MASLD according to changes in the nomenclature in a health check-up using MRI-derived proton density fat fraction. Abdom Radiol. 2024;49:3036-44.
17. Lee CM, Yoon EL, Kim M, et al. Prevalence, distribution, and hepatic fibrosis burden of the different subtypes of steatotic liver disease in primary care settings. Hepatology. 2024;79:1393-400.
18. Choe HJ, Moon JH, Kim W, Koo BK, Cho NH. Steatotic liver disease predicts cardiovascular disease and advanced liver fibrosis: a community-dwelling cohort study with 20-year follow-up. Metabolism. 2024;153:155800.
19. Tamaki N, Kimura T, Wakabayashi SI, et al. Long-term clinical outcomes in steatotic liver disease and incidence of liver-related events, cardiovascular events and all-cause mortality. Aliment Pharmacol Ther. 2024;60:61-9.
20. Kim A, Kang D, Choi SC, Cho J, Sinn DH, Gwak GY. Steatotic liver disease and its newly proposed sub-classifications correlate with progression of the coronary artery calcium score. PLoS ONE. 2024;19:e0301126.
21. Lee HH, Lee HA, Kim EJ, et al. Metabolic dysfunction-associated steatotic liver disease and risk of cardiovascular disease. Gut. 2024;73:533-40.
22. Wong RJ, Yang Z, Sedki M, Zhang W, Singal AK, Cheung R. Estimating steatotic liver disease prevalence among US veterans. Gastro Hep Adv. 2025;4:100804.
23. Younossi ZM, Golabi P, Paik JM, Henry A, Van Dongen C, Henry L. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology. 2023;77:1335-47.
24. Paik JM, Hobbs K, Gupta A, Alkalbani RJ, Reyes MA, Younossi ZM. Prevalence of MASLD, Met-ALD, and ALD and associated fibrosis among US adults: insights from NHANES 2017 to 2023. J Clin Gastroenterol. 2025;Epub ahead of print.
25. Krag A, Torp N, Younossi ZM, Israelsen M. Reporting discrepancy of alcohol intake affecting estimated prevalence of MetALD and ALD. Lancet Gastroenterol Hepatol. 2025;10:282-4.
26. Marti-Aguado D, Calleja JL, Vilar-Gomez E, et al. Low-to-moderate alcohol consumption is associated with increased fibrosis in individuals with metabolic dysfunction-associated steatotic liver disease. J Hepatol. 2024;81:930-40.
27. Männistö V, Salomaa V, Jula A, et al. ALT levels, alcohol use, and metabolic risk factors have prognostic relevance for liver-related outcomes in the general population. JHEP Rep. 2024;6:101172.
28. Israelsen M, Thorhauge KH, Andersen P, et al. Steatotic liver disease classification is dynamic, affecting clinical trial eligibility and subclass-specific treatments. Clin Gastroenterol Hepatol. 2025;23:2509-18.
29. Sedki M, Yang Z, Singal AK, et al. Longitudinal risk of cirrhosis by steatotic liver disease subtype among 1.5 million individuals in the US. JHEP Rep. ;8:101680.
30. Sripongpun P, Kaewdech A, Udompap P, Kim WR. Characteristics and long-term mortality of individuals with MASLD, MetALD, and ALD, and the utility of SAFE score. JHEP Rep. 2024;6:101127.
31. Tampaki M, Tsochatzis E, Lekakis V, Cholongitas E. Prevalence, characteristics and outcomes of patients with metabolic and alcohol related/associated liver disease (MetALD): a systematic review and meta-analysis. Metabolism. 2025;163:156101.
32. Celletti F, Farrar J, De Regil L. World health organization guideline on the use and indications of glucagon-like peptide-1 therapies for the treatment of obesity in adults. JAMA. 2026;335:434-8.
33. Alfaris N, Waldrop S, Johnson V, Boaventura B, Kendrick K, Stanford FC. GLP-1 single, dual, and triple receptor agonists for treating type 2 diabetes and obesity: a narrative review. EClinicalMedicine. 2024;75:102782.
34. Sanyal AJ, Newsome PN, Kliers I, et al.; ESSENCE Study Group. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N Engl J Med. 2025;392:2089-99.
35. Hendershot CS, Bremmer MP, Paladino MB, et al. Once-weekly semaglutide in adults with alcohol use disorder: a randomized clinical trial. JAMA Psychiatry. 2025;82:395-405.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].