Lipids at the helm: a metabolic playbook for liver regeneration
Abstract
Ancient mythology alluded to the liver’s regenerative capabilities, a phenomenon now validated by scientific research: the organ can regenerate up to 75% of its mass following partial resection, provided sufficient tissue remains. This commentary synthesizes and contextualizes a recent review article that emphasizes lipid metabolism as a key component of liver regeneration. Effective regeneration depends on the viability of hepatocytes, contributions from supporting cells, metabolic adaptability, and appropriate management of infections and hemodynamics. The regenerative process unfolds in distinct phases: cytokine priming, cellular proliferation, and termination. In cases of acute injury, hepatocytes are primarily responsible for regeneration, whereas in chronic disease states, progenitor-like cells also contribute. Recovery is influenced by hormonal status, nutritional state, age, and signals from non-parenchymal cells, with zone 2 hepatocytes playing a pivotal role. Lipid metabolism is critical; moderate lipid accumulation supports regeneration, whereas excessive fat deposition hinders it. Lipophagy provides essential energy and mitigates toxicity, while endoplasmic reticulum (ER) stress must be properly regulated. Therapeutic strategies focus on optimizing β-oxidation, enhancing lipophagy, and managing ER stress, guided by lipid profiling and fat quantification. Personalized interventions tailored to phase-specific lipid metabolic needs may optimize liver regenerative outcomes.
Keywords
FROM ANCIENT MYTH TO MODERN SCIENCE
Although the extent to which ancient Greeks might have been fully aware of the liver’s regenerative capacities is uncertain, few examples are more clearly illustrative than the myths of Tityos and Prometheus [Table 1].
Contemporary scientific research demonstrates that a healthy liver is capable of regenerating as much as 75% of its mass after partial hepatectomy, in a manner that is both efficient and strictly regulated[3].
THE CLINICAL CONTEXT
The most dramatic example of the importance of a preserved liver mass for survival is non-recovering post-resection liver failure (nrPRLF), which occurs in up to 30% of cases, and is burdened with high morbidity and mortality owing to jaundice, coagulopathy, and hepatic encephalopathy after liver resection[4]. Successful liver regeneration depends on adequate hepatocytes, non-parenchymal cells, functional capacity, and hemodynamic adaptation. The prevention of post-resection liver failure focuses on limiting parenchymal damage, reducing ischemia-reperfusion injury, promptly addressing infection or sepsis, and avoiding small-for-size syndrome[5]. Liver regeneration has high energy demands, which are fueled by mobilization of peripheral fat deposits[6].
Metabolic inflexibility supports mitochondrial health across populations by limiting the proliferation of cells with electron transport chain dysfunction, thereby serving as a mechanism for cellular quality control or selective pressure[7,8].
In the context of protein downregulation being closely associated with lipid metabolism, cellular transport, very low-density lipoprotein (VLDL) secretion, and hepatocyte proliferation, transient hepatic steatosis after major liver resection may serve as a useful early marker for liver regeneration and nrPRLF[9].
A study involving 112 individuals who underwent surgery for right lobe liver donation confirmed that moderate hepatic steatosis does not significantly compromise the safety of living liver donors[10].
A comprehensive understanding of the mechanisms underlying liver regeneration has the potential to benefit patients undergoing major hepatic resections or transplantation and may also contribute valuable insights to the advancement of regenerative medicine[11].
REGENERATION AS A PHASED, METABOLISM-DRIVEN PROGRAM
The liver is remarkable for its ability to regenerate mass and function after injury or surgical resection, making it unique among adult mammalian organs[12]. Classic rodent partial hepatectomy models have shown that up to two-thirds of the liver can be removed, yet the organ regrows within days through coordinated hypertrophy and cell-cycle re-entry of mature hepatocytes, supported by the proliferation of cholangiocytes and non-parenchymal cells[13,14]. Regeneration occurs in three distinct phases: (i) a rapid priming phase driven by cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) that activate nuclear factor-κB (NF-κB) and signal transducer and activator of transcription (STAT3), (ii) a proliferative phase orchestrated by mitogens including hepatocyte growth factor (HGF)/c-Met and epidermal growth factor (EGF) and its receptor (EGFR) along with Wingless (Wnt)/β-catenin and Hippo/Yes-associated protein (YAP) signaling, and (iii) a termination phase influenced by transforming growth factor-β (TGF-β), metabolic checkpoints, and extracellular matrix remodeling[15-18]. Experimental models may also involve the use of organoids and personalized biomarkers to translate metabolic discoveries into precise therapies for liver regeneration[19].
However, the context is important. In acute injury, lineage-traced hepatocytes are the main source of new parenchyma, while in chronic or cholestatic disease, ductular reactions and facultative progenitor-like cells contribute through transdifferentiation[20]. Non-parenchymal cells such as Kupffer cells, hepatic stellate cells, and sinusoidal endothelial cells play an essential role in providing inflammatory, fibrogenic, and angiogenic signals, while portal hemodynamics, bile acids such as farnesoid X receptor (FXR)/Takeda G protein-coupled receptor 5 (TGR5), hormones, nutrition, age, sex, and circadian rhythms affect the regenerative process. Specifically, zone 2 is an important source of new hepatocytes during homeostasis and regeneration[21,22].
LIPID ORCHESTRATION, LIPOPHAGY-ENDOPLASMIC RETICULUM STRESS AXIS, AND TRANSLATIONAL LEVERS
In the review by Duan et al., the authors argue that lipid metabolism plays a crucial role in orchestrating liver regeneration through stage-specific programs[23]. They highlight an early, regulated increase in hepatocellular triglycerides and cholesterol esters that fuel histone acetylation and membrane raft signaling followed by a transition to carnitine palmitoyltransferase 1A (CPT1A)-dependent β-oxidation. This process supplies more than two-thirds of the proliferative ATP before decreasing during the termination phase. The authors specifically focus on the balance between lipolysis and oxidative stress pointing to peroxisome proliferation-activated receptor-α (PPARα) as a central hub that coordinates mitochondrial, peroxisomal, and microsomal fatty acid oxidation with antioxidant defenses. This coordination is essential for maintaining redox homeostasis and promoting cell-cycle progression. They also discuss the dual impacts of lipid loading, demonstrating that moderate storage supports biosynthesis and energy production, while chronic overload leads to lipid peroxidation, shifts death programs towards necroptosis/pyroptosis, and impairs regeneration. Additionally, they note that diets high in unsaturated fats tend to have more favorable outcomes than those high in saturated fat[23].
A major focus is lipophagy, which is the selective autophagic degradation of lipid droplets via autophagy-related (ATG) machinery, Rab7/Rab10-directed trafficking, and chaperone-mediated autophagy (CMA) of perilipins. This process is activated early after hepatectomy to liberate fatty acids for β-oxidation and limit lipotoxic reactive oxygen species (ROS). Impaired lipophagy couples with endoplasmic reticulum (ER) stress and delays liver mass recovery. Mechanistically, an ER stress-lipogenesis-regeneration axis is triggered, in which transient inositol-requiring enzyme 1 (IRE1α)/X-box binding protein 1 (XBP1) and PKR-like endoplasmic reticulum kinase (PERK)/eukaryotic initiation factor 2α (eIF2α) activation is adaptive. However, chronic activation induces SREBP-1c-driven lipogenesis, triglyceride accumulation, and G1/S arrest. Sirtuin 1 (SIRT1) emerges as a modulator that alleviates stress and restores proliferation. This integrative framework extends to phospholipid metabolism [PC synthesis via Kennedy, Lands, and phosphatidylethanolamine N-methyltransferase (PEMT) pathways; circulating phosphatidylcholine containing polyunsaturated fatty acids (PUFA-PC) and high-density lipoprotein (HDL)/VLDL dynamics as regeneration correlates], the gut-liver-microbiota axis [short-chain fatty acids (SCFAs) driving lipogenesis for membrane biogenesis, GLP-1 restrains and Glucagon-like peptide-2 (GLP-2) promotes lipid provision, and fibroblast growth factor (FGF)15/19-FGFR4 accelerating proliferation while limiting early lipid overload], and immunometabolic control by macrophages[23]. Proliferation-activated receptor-γ (PPARγ)/fatty acid synthase (FASN)-dependent lipid networks activate STAT3 to coordinate growth factor release and ER expansion while reparative cells rely on fatty acid oxidation (FAO)/oxidative phosphorylation (OXPHOS).
Translationally, the review highlights therapeutic strategies within this network: PPARα agonism and L-carnitine to enhance β-oxidation; AMP-activated protein kinase (AMPK)/UNC-51-like kinase 1 (ULK1) activation and compounds such as rapamycin or FGF21 to improve lipophagy; interventions targeting the ER stress-lipophagy axis including ubiquitin-fold modifier 1-mediated protein conjugation (UFMylation) modulation, liver-specific SIRT1-AMPK activation with resveratrol nanoparticles, IRE1α inhibition, and ω-3 PUFAs; and immuno-metabolic targets such as monoacylglycerol lipase (MAGL) and human umbilical cord-derived mesenchymal stem cell (hUC-MSC) exosomes to restore balance in macrophage functions[23].
It is important to note that differences between species, such as inherent weaker PPARα activation in humans[24], stricter YAP regulation[25], potential differences in mouse and human endothelial WNT2-FZD5 signaling[26], and unique bile acid/SCFA hierarchies[27], require lipidomics-guided, human-centered approaches[23]. These experimental models may involve the use of organoids and personalized biomarkers to translate metabolic discoveries into precise therapies for liver regeneration[19].
CLINICAL IMPLICATIONS AND RESEARCH AGENDA
Clinically, the review supports rigorous demands for preoperative risk stratification that quantifies hepatic fat burden and composition[28]. Steatosis, present in over 20% of resection candidates, is associated with higher rates of post-hepatectomy complications, heightened vulnerability to ischemia-reperfusion injury, impaired regeneration, and inferior transplant outcomes[29,30]. This prompts more conservative resections and is reflected in meta-analytic evidence showing a doubled complication risk; it also explains the long-known fact of up to threefold higher mortality in severe steatosis[31].
Perioperative metabolic management should recognize that early, transient regenerative‑associated steatosis (TRAS) supports regrowth[23]. Suppressing lipid influx or providing excess glucose can blunt the regenerative program. Clinical protocols should be aligned with these pathobiological features. Lipidomics and phospholipid profiling, notably circulating PUFA-phosphatidylcholine and sphingomyelin, show promise for predicting delayed regeneration and guiding nutritional or pharmacologic strategies to restore phosphatidylcholine homeostasis[23]. This is important given the centrality of PEMT, Kennedy, and Lands pathways to membrane biogenesis.
Therapeutically, the authors outline ways to enhance β-oxidation (liver-targeted PPARα agonism, L-carnitine), boost lipophagy, and temper maladaptive ER stress. Strategies include AMPK-ULK1 activation, rapamycin or FGF21, SIRT1-AMPK strategies such as resveratrol nanoparticles, IRE1α modulation, ω-3 PUFAs, and UFMylation. These approaches, in preclinical models, normalize lipid handling, redox balance, and hepatocyte proliferation[23].
From an evidence standpoint, repurposing clinically available metabolic modulators with favorable hepatic safety profiles (such as PPARα agonists, ω-3 PUFAs, GLP-1 receptor agonists, cilostazol, or L-carnitine) is currently supported mainly by human observational and early-phase interventional data in metabolic dysfunction-associated steatotic liver disease (MASLD) and related metabolic conditions. Most other approaches we discuss (including AMPK-ULK1 activation, FGF21-based interventions, resveratrol nanoparticles, direct IRE1α/UFMylation modulation, MAGL inhibition, hUC-MSC exosomes, or MKK4 inhibition) remain confined to animal models or ex vivo systems and cannot yet be recommended outside clinical trials.
Among these candidates, a particularly actionable near-term research direction is a biomarker-enriched perioperative trial that uses lipidomics and imaging-based fat quantification to test whether modulating the TRAS-to-β-oxidation transition with a pragmatic intervention (for example, tailored macronutrient support combined with a PPARα agonist or ω-3 PUFA supplementation) can reduce delayed regeneration and non-recovering post‑resection liver failure.
Studies have shown that diets high in saturated or unsaturated fats impair liver regeneration after partial hepatectomy in mice, leading to metabolic dysfunction-associated steatohepatitis (MASH). However, unsaturated fat diets cause less inflammation and better regeneration than saturated fat diets[32]. Additionally, pharmacological approaches may also modulate the regenerative capacity of the liver. Cilostazol, a phosphodiesterase-3 (PDE-3) inhibitor, has been associated with decreased hepatic steatosis and inflammation in a rat model of steatosis[33]. Holländer et al.[34] have recently reported that proliferation of hepatocytes and liver regeneration after resection of 70% of the liver were significantly increased in a steatotic rat model treated with cilostazol.
Mitogen-activated protein kinase 4 (MKK4) functions as an important negative regulator of hepatocyte proliferation, particularly in response to stressors such as partial hepatectomy, chemically induced liver injury, and steatohepatitis[35]. HRX215 is the first selective small-molecule MKK4 inhibitor, developed using a structure-guided, NMR-based approach. It shows hepatoprotective, pro-regenerative, antifibrotic, and antisteatotic effects, indicating its potential for treating various liver diseases. HRX215 may prevent post-hepatectomy liver failure or reduce small-for-size syndrome after partial liver transplantation[35].
Modulating gut-liver crosstalk and hormones may be leveraged carefully. Ileal FGF15/19-hepatic FGF receptor 4 (FGFR4) activation accelerates proliferation while limiting early lipid overload. GLP-2 appears pro-regenerative by facilitating lipid supply, whereas glucagon-like peptide 1 (GLP‑1) can dampen early regrowth by curbing lipogenesis. Immunometabolic targeting of macrophages, by avoiding MAGL-driven interferon activation and promoting reparative PPARγ/STAT3 and PPARα pathways, represents another translational lever to improve the regenerative milieu[22]. Because of differences between rodents and humans in PPARα responsiveness, YAP constraints, bile acid versus SCFA hierarchies, and human-specific endothelial WNT2-FZD5 signaling, direct extrapolation is limited. Prospective, human-focused trials that integrate lipidomics stratification, organoid/perfusion models, and phase-specific interventions are required to safely translate these insights.
Accordingly, our conclusions regarding avoiding excessive early glucose supplementation, maintaining the transition from TRAS-to-β-oxidation, using lipidomics to guide nutritional or pharmacologic support, or anticipating a potential reduction in early regrowth due to GLP-1 agonism should be viewed as mechanistic, preliminary signals rather than immediate practice-changing recommendations. A specific, testable framework could involve a prospective perioperative study that integrates ongoing assessment of TRAS and lipid flux [e.g., magnetic resonance imaging proton density fat fraction (MRI-PDFF) or computed tomography (CT)-based liver fat measurements in conjunction with targeted lipidomic panels focusing on VLDL-triglycerides, PUFA-phosphatidylcholine, and sphingomyelin species] from preoperative baseline through postoperative days 1-7. This study could also include standardized evaluations of regenerative capacity (e.g., indocyanine green clearance, dynamic liver function tests, volumetric hypertrophy) and clinically significant outcomes such as delayed hypertrophy, nrPRLF, and major postoperative complications. These studies should be carefully designed to account for and control key variables that could influence lipid processing and regenerative outcome, such as baseline steatosis or MASLD, visceral adiposity and insulin resistance, and judicious perioperative blood sugar management.
CONCLUSION
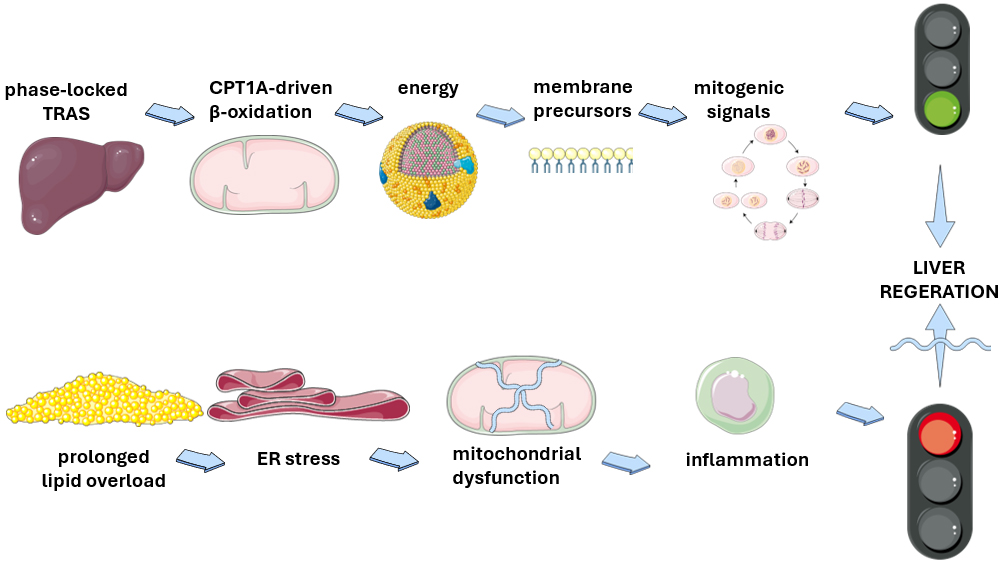
Viewed through a metabolic-centric perspective, lipid flux represents a crucial factor in liver repair. Specifically, phase-locked TRAS during priming and a subsequent CPT1A-driven β-oxidation surge provide energy, membrane precursors, and mitogenic signals necessary for regeneration. However, prolonged lipid overload can lead to ER stress, mitochondrial dysfunction, and inflammation, eventually hindering the regenerative process [Figure 1].
Figure 1. Lipid flux as a key player in liver regeneration. Schematic illustration of the Janus-faced actions of lipid fluxes on liver regeneration. Phase-locked transient steatosis may eventually facilitate liver regeneration by providing energy, substrates and signals. Conversely, prolonged lipid overload will impair liver regeneration by promoting endoplasmic reticulum stress, mitochondrial dysfunction and inflammation. This original illustration was created using Servier Medical ART (SMART) and is licensed under the Creative Commons Attribution 4.0 International License (CC BY 4.0). ER: endoplasmic reticulum; CPT1A: Carnitine Palmitoyltransferase 1A; TRAS: transient regenerative-associated steatosis.
Lipophagy and PPARα-centered redox/oxidation programs are identified as important control points, while interactions between the gut-liver and immune system (involving SCFAs, GLP-1/GLP-2, FGF15/19-FGFR4, and macrophage polarization) play a role in setting the regenerative tone. Therefore, targeted interventions that boost β-oxidation and lipophagy, while addressing maladaptive ER stress in a way that considers human-specific pathway limitations, present a viable approach to improving hepatic repair outcomes.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception, design and writing of the study; performed data analysis and interpretation: Lonardo A, Weiskirchen R
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool “Edit my English” was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Availability of data and materials
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Lonardo A is the Editor-in-Chief and Founding Editor of Metabolism and Target Organ Damage. Weiskirchen R is an Associate Editor of the journal. Neither of them was involved in any steps of the editorial process, including reviewers’ selection, manuscript handling, or decision-making.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Tiniakos DG, Kandilis A, Geller SA. Tityus: a forgotten myth of liver regeneration. J Hepatol. 2010;53:357-61.
2. Kattner AA. Greek gods and the double-edged sword of liver regeneration. Biomed J. 2021;44:515-20.
3. Michalopoulos GK, Bhushan B. Liver regeneration: biological and pathological mechanisms and implications. Nat Rev Gastroenterol Hepatol. 2021;18:40-55.
5. Colle I, Verhelst X, Vanlander A, et al. Pathophysiology and management of post resection liver failure. Acta Chir Belg. 2013;113:155-61.
6. Engelmann C. Peripheral fat mobilization and mitochondrial fat metabolism: Fueling the energy demands of liver regeneration. J Hepatol. 2025;82:942-4.
7. Smith RL, Soeters MR, Wüst RCI, Houtkooper RH. Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocr Rev. 2018;39:489-517.
8. Wang X, Menezes CJ, Jia Y, et al. Metabolic inflexibility promotes mitochondrial health during liver regeneration. Science. 2024;384:eadj4301.
9. Lund A, Thomsen MT, Kirkegård J, et al. Role of steatosis in preventing post-hepatectomy liver failure after major resection: findings from an animal study. J Clin Exp Hepatol. 2025;15:102453.
10. Peltec A, Hotineanu A, Popescu I, Braşoveanu V. The impact of liver steatosis on the postoperative evolution after right lobe living-donor hepatectomy. Med Pharm Rep. 2021;94 Suppl:S43-50.
11. Gilgenkrantz H, Collin de l’Hortet A. Understanding liver regeneration: from mechanisms to regenerative medicine. Am J Pathol. 2018;188:1316-27.
12. Hora S, Wuestefeld T. Liver injury and regeneration: current understanding, new approaches, and future perspectives. Cells. 2023;12:2129.
13. Boyce S, Harrison D. A detailed methodology of partial hepatectomy in the mouse. Lab Anim. 2008;37:529-32.
14. Zafarnia S, Mrugalla A, Rix A, et al. Non-invasive imaging and modeling of liver regeneration after partial hepatectomy. Front Physiol. 2019;10:904.
15. Xiaoguang Chen. A comprehensive review on liver regeneration termination: a non-neglectable phase. Cytol. Genet. 2024;58:343-61.
17. Zhao Y, Ye W, Wang YD, Chen WD. HGF/c-met: a key promoter in liver regeneration. Front Pharmacol. 2022;13:808855.
18. Théret N, Feret J, Hodgkinson A, Boutillier P, Vignet P, Radulescu O. Integrative models for TGF-β signaling and extracellular matrix. In: Ricard-Blum S, Editors. Extracellular matrix omics. Cham: Springer International Publishing; 2020. pp. 209-25.
19. Sljukic A, Green Jenkinson J, Niksic A, Prior N, Huch M. Advances in liver and pancreas organoids: how far we have come and where we go next. Nat Rev Gastroenterol Hepatol. 2026;23:44-64.
20. Malato Y, Naqvi S, Schürmann N, et al. Fate tracing of mature hepatocytes in mouse liver homeostasis and regeneration. J Clin Invest. 2011;121:4850-60.
21. Wei Y, Wang YG, Jia Y, et al. Liver homeostasis is maintained by midlobular zone 2 hepatocytes. Science. 2021;371.
22. He L, Pu W, Liu X, et al. Proliferation tracing reveals regional hepatocyte generation in liver homeostasis and repair. Science. 2021;371:eabc4346.
23. Duan L, Chang Y, Dai J, et al. Lipid metabolism orchestrates liver regeneration: an integrated metabolic network. J Transl Med. 2025;23:1115.
24. Yang Q, Nagano T, Shah Y, Cheung C, Ito S, Gonzalez FJ. The PPAR alpha-humanized mouse: a model to investigate species differences in liver toxicity mediated by PPAR alpha. Toxicol Sci. 2008;101:132-9.
25. Shizu R, Makida N, Sobe K, et al. Interaction with YAP underlies the species differences between humans and rodents in CAR-dependent hepatocyte proliferation. Toxicol Sci. 2024;198:101-12.
26. Koch PS, Sandorski K, Heil J, et al. Imbalanced activation of Wnt-/β-catenin-signaling in liver endothelium alters normal sinusoidal differentiation. Front Physiol. 2021;12:722394.
27. Visekruna A, Luu M. The role of short-chain fatty acids and bile acids in intestinal and liver function, inflammation, and carcinogenesis. Front Cell Dev Biol. 2021;9:703218.
28. Bhalla S, Mcquillen B, Cay E, Reau N. Preoperative risk evaluation and optimization for patients with liver disease. Gastroenterol Rep (Oxf). 2024;12:goae071.
29. Peloso A, Tihy M, Moeckli B, Rubbia-Brandt L, Toso C. Clearing steatosis prior to liver surgery for colorectal metastasis: a narrative review and case illustration. Nutrients. 2022;14:5340.
30. Ahmed EA, El-Badry AM, Mocchegiani F, et al. Impact of graft steatosis on postoperative complications after liver transplantation. Surg J (N Y). 2018;4:e188-96.
31. de Meijer VE, Kalish BT, Puder M, Ijzermans JN. Systematic review and meta-analysis of steatosis as a risk factor in major hepatic resection. Br J Surg. 2010;97:1331-9.
32. Islam SMT, Palanisamy AP, Chedister GR, Schmidt MG, Lewin DNB, Chavin KD. Unsaturated or saturated dietary fat-mediated steatosis impairs hepatic regeneration following partial hepatectomy in mice. PLoS ONE. 2023;18:e0284428.
33. Fujita K, Nozaki Y, Wada K, et al. Effectiveness of antiplatelet drugs against experimental non-alcoholic fatty liver disease. Gut. 2008;57:1583-91.
34. Holländer S, von Heesen M, Gäbelein G, et al. Perioperative treatment with cilostazol reverses steatosis and improves liver regeneration after major hepatectomy in a steatotic rat model. Sci Rep. 2025;15:2753.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].