Semaglutide and metformin improve menstrual cyclicity in overweight/obese women with polycystic ovary syndrome: a randomized trial
Abstract
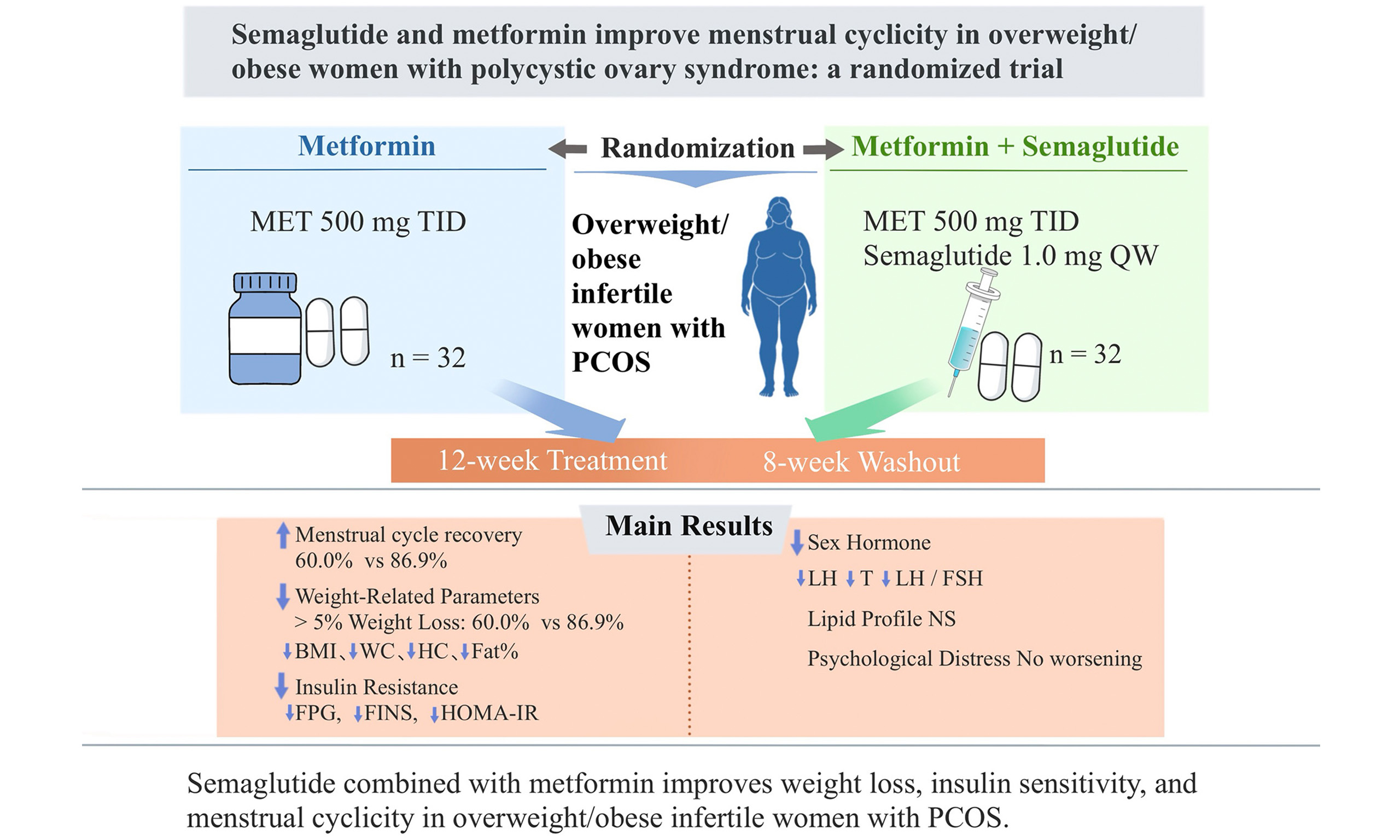
Aim: This study investigated the effects of semaglutide combined with metformin on body weight, glucose and lipid metabolism, sex hormone levels, menstrual cyclicity, and psychological parameters of women living with polycystic ovary syndrome (PCOS)-related infertility and overweight/obesity.
Methods: Sixty-four patients living with infertility and PCOS [body mass index (BMI) ≥
Results: The semaglutide-intensified treatment group showed a higher percentage of patients with restored menstrual cycles (86.87% vs. 60.00%, P = 0.038). Significant reductions in luteinizing hormone (LH), androgen levels, and the LH/follicle-stimulating hormone ratio were observed. Greater reductions were also noted in BMI, waist and hip circumference, absolute weight loss, body fat percentage, fasting plasma glucose, fasting insulin, and Homeostatic Model Assessment of Insulin Resistance (P < 0.05). No significant differences were observed in total cholesterol, triglyceride, and low-density lipoprotein cholesterol levels. Psychological distress was not exacerbated in the semaglutide-intensified treatment group.
Conclusion: In women living with PCOS-related infertility and overweight/obesity, semaglutide combined with metformin provided superior improvements in weight reduction, glucose metabolism, insulin sensitivity, and menstrual cyclicity compared with metformin monotherapy, without exacerbating psychological distress. These findings suggest that semaglutide represents a promising therapeutic option for the clinical management of this population.
Keywords
INTRODUCTION
Infertility refers to the inability to establish a clinical pregnancy after 12 months of regular unprotected sexual intercourse. Recently, infertility has become a major global clinical burden, and a cross-sectional study involving 15,162 participants in the UK reported that 12.5% of women and 10.1% of men were infertile[1]. A population-based study in China investigating 25,270 couples of reproductive age found that the prevalence of infertility was 25% in this population[2].
Overweight or obesity is a critical contributor to infertility. The overall prevalence of obesity among Chinese adults is 14.0%[3]. Being overweight or obese is associated with difficulty in pregnancy, both in women preparing for natural pregnancy and in those receiving assisted reproductive treatment[4,5]. Body mass index (BMI) ≥ 24 kg/m2 is positively correlated with impaired reproductive capacity, with fertility rates decreasing by 8% in women who are overweight and 18% in women living with obesity. A higher BMI is also associated with increased age at conception[6]. Obesity can impair ovarian function, leading to anovulation, menstrual disorders, and a decrease in spontaneous conception[7]. Furthermore, the probability of achieving pregnancy using assisted reproductive technology is lower in women who are overweight than in normal-weight women[8]. Additionally, obesity is an independent risk factor for miscarriage and adverse pregnancy outcomes[9,10]. Accordingly, optimizing the BMI is essential for women trying to conceive.
Polycystic ovary syndrome (PCOS) is the primary cause of anovulatory infertility[11,12]. The prevalence of overweight or obesity among patients with PCOS exceeds 40%[13]. Patients living with obesity and PCOS exhibit more pronounced disruptions in sex hormones and glucose-lipid metabolism, negatively impacting follicular development and endometrial receptivity, which leads to elevated rates of infertility and a higher risk of adverse pregnancy outcomes[14]. Achieving a weight loss of 5%-15% or more within six months significantly improves ovulatory function, regulates menstrual cycles, mitigates hirsutism, enhances insulin sensitivity, and reduces serum androgen levels in women who are overweight or obese with infertility[15].
The primary interventions for weight loss include lifestyle changes, such as diet, regular exercise, and cognitive-behavioral therapy. However, some individuals may have difficulty achieving an ideal weight loss of 5%-10% through lifestyle interventions alone[16] or adhering to long-term diets or exercise programs. Currently, pharmacotherapy remains the cornerstone treatment for patients who are overweight or obese with PCOS. Metformin can enhance glycemic control and ovarian function, lower androgen levels, and improve ovulation and pregnancy rates[17,18]. However, its efficacy in promoting weight loss is often suboptimal in certain patients, necessitating combination therapy.
Glucagon-like peptide-1 receptor agonist (GLP-1 RA) analogs are useful treatments for type 2 diabetes mellitus and obesity, and their mechanisms of action include reducing food intake and delaying gastric emptying[19,20]. Semaglutide is the latest long-acting GLP-1 RA, with remarkable effects on weight loss[21]. A double-blind clinical trial showed that after 68 weeks of semaglutide treatment, the mean change in body weight from baseline was -14.9%, and the proportion of participants achieving weight loss from baseline exceeding 15% was 50.5%[22]. The STEP 5 (Semaglutide Treatment Effect in People with Obesity 5) study showed that semaglutide achieved a substantial and sustained weight loss during the 2-year follow-up[23]. A meta-analysis showed that discontinuation of semaglutide resulted in significant weight rebound, characterized by a body weight gain of 8.21 kg[24]. Adhering to exercise and a healthy diet can effectively reduce weight rebound after discontinuing GLP-1 RA[25]. These results suggest that women living with infertility who are overweight or obese can achieve ideal weight loss via pharmacotherapy combined with lifestyle modification.
Recently, clinical studies have investigated the potential of GLP-1 RA in improving menstrual regularity in women who are overweight and obese during the preconception period[26-29]. A double-blind randomized trial demonstrated that liraglutide increased menstrual frequency and alleviated ovarian dysfunction in women with PCOS[29]. However, the potentially beneficial effects of semaglutide on ovarian function and reproductive outcomes in women living with infertility who are overweight and obese remain unclear. Therefore, a 20-week clinical trial was conducted to investigate the effects of semaglutide combined with metformin on body weight, glucose and lipid profiles, sex hormone levels, menstrual cyclicity, and psychological parameters in women living with infertility who are obese and diagnosed with PCOS. We hypothesized that semaglutide would improve ovarian function and reproductive outcomes in this population by improving weight loss.
METHODS
Study design, setting, and ethical considerations
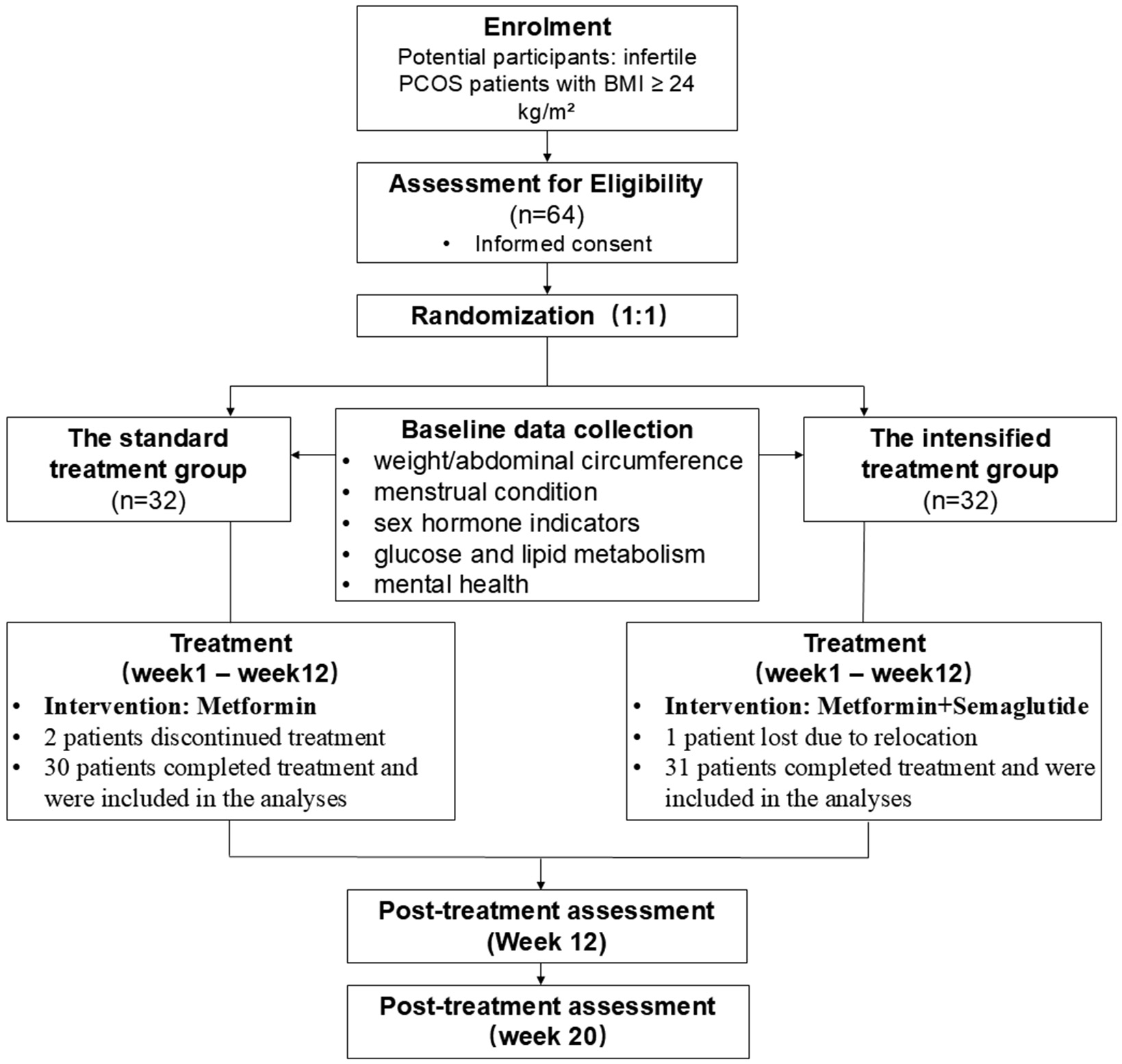
This was a 20-week, single-center, open-label, prospective, randomized clinical trial [Figure 1]. All examinations were performed at the Weight Management Clinic and Reproductive Clinic, The Ninth Medical Center of the PLA General Hospital (the former PLA Strategic Support Force Medical Center), Beijing, China. This study was retrospectively registered in the Chinese Trial Registry (registration number: ChiCTR2500099998) (04/01/2025). The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Ninth Medical Center of PLA General Hospital (Approval number: K2021-No.02). All the participants signed an informed consent form before the start of the study.
Participants and recruitment
A total of 64 participants were enrolled in this study. The participants were recruited from outpatient units, specifically the Weight Management and Reproductive Clinics at the Ninth Medical Center of the PLA General Hospital between June 2023 and June 2024. All eligible participants received written and oral information regarding the study. After the patients agreed to participate and signed an informed consent form, they underwent screening based on the inclusion and exclusion criteria.
Eligible patients were randomly assigned to either the standard treatment (n = 32) or semaglutide-intensified treatment group (n = 32). The allocation ratio was set at 1:1. Randomization was performed by an independent researcher with no involvement in the study. Random numbers were generated using computers, and the allocation codes were placed in sealed opaque envelopes. The envelopes were opened in the order of case enrollment, and participants were assigned according to the allocation codes within them.
Inclusion criteria
The inclusion criteria were as follows: women with clinically diagnosed infertility; BMI ≥ 24 kg/m2; and women with a diagnosis of PCOS based on the 2003 Rotterdam criteria. A diagnosis was established if at least two of the following conditions were met: (1) ovulatory dysfunction (oligo- or anovulation); (2) clinical and/or biochemical signs of hyperandrogenism; and (3) polycystic ovaries visualized using ultrasound. Women who planned to conceive and voluntarily participated in this trial provided informed oral and written consent.
Exclusion criteria
The exclusion criteria were as follows: infertility caused by organic factors, including bilateral obstruction of the fallopian tubes, or male factors (a total motile sperm count < 5 million on the most recent seminal analysis); use of weight-lowering medications or glucagon-like peptide (GLP-1) drugs within the preceding 3 months; use of medications that may lead to clinically significant weight gain within the preceding 3 months; use of medications that affect sex hormone levels within the preceding 3 months; allergy to any of the ingredients of the study medication, including semaglutide, disodium phosphate dihydrate, sodium chloride, hydrochloric acid, and sodium hydroxide, or other GLP-1 RA drugs; a family or a personal history of multiple endocrine neoplasia syndrome type 2; a family or a personal history of medullary thyroid carcinoma; a history of acute and chronic pancreatitis; psychiatric disease, such as depression, schizophrenia, or bipolar disorder; a history of carcinoma; severe cardiovascular disease, hepatic impairment, or renal impairment; type 1 or 2 diabetes; congenital adrenal hyperplasia, Cushing’s syndrome; unable to take effective contraceptive measures during the trial; unable to understand the experimental requirements; and any condition that the investigator feels would interfere with trial participation.
All the participants were advised to use barrier contraceptives throughout the study period. The participants were instructed to promptly notify the investigators and seek specialized medical consultations in the event of suspected pregnancy symptoms or a positive urine pregnancy test to ensure safety. Furthermore, a mandatory 8-week washout period was implemented following the treatment phase before the participants could attempt spontaneous or assisted conception.
Treatment protocol
Both the standard and semaglutide-intensified treatment groups received lifestyle modification interventions combined with pharmacological therapy. Lifestyle intervention involved the provision of weight loss education manuals to all participants, as well as guidance on a Calorie Restriction Diet and regular moderate-intensity physical activity, with consistent implementation criteria applied to both groups. The standard treatment group received metformin monotherapy [metformin 500 mg three times daily (TID)] for 12 weeks, whereas the semaglutide-intensified treatment group received metformin in combination with semaglutide (metformin 500 mg TID with semaglutide) for 12 weeks. Semaglutide was initiated at 0.25 mg once weekly for 4 weeks. The dose was increased at 4-week intervals until a dose of 1.0 mg was reached. The maintenance dose was 1.0 mg once a week. The titration procedure was prolonged when participants did not tolerate rapid up-titrations. Participants who could not tolerate the 1.0 mg dose were maintained on a lower dose under special circumstances.
Baseline data were collected after patients provided informed consent. The standard or semaglutide-intensified treatment groups received metformin or metformin combined with semaglutide for 12 weeks, respectively, followed by an 8-week washout period. The participants were followed up after the washout period. BMI, absolute weight loss, waist and hip circumference, body fat percentage, sex hormone profiles, Homeostatic Model Assessment of Insulin Resistance (HOMA-IR), glucose and lipid metabolism indices, and psychological assessments - the World Health Organization-Five Well-Being Index (WHO-5), Generalized Anxiety Disorder Scale (GAD-7), and Patient Health Questionnaire Depression Scale (PHQ-9) - were measured at baseline and at 12 and 20 weeks. One patient in the semaglutide-intensified treatment group was lost to follow-up owing to relocation, and two patients in the standard treatment group voluntarily discontinued treatment.
Observational indicators and measurement metrics
Anthropometric parameters: Body height, body weight, BMI, body fat percentage, and waist and hip circumference were recorded for both groups before treatment, as well as after 12 and 20 weeks of treatment. BMI was calculated as weight (kg) divided by height (m2). The waist-to-height ratio (WHtR) is calculated as waist circumference (cm) divided by body height (cm).
Sex hormone indicators: Blood samples were collected on days 2-5 of the menstrual cycle or upon bleeding cessation before and after 12 and 20 weeks of treatment. Serum luteinizing hormone (LH), follicle-stimulating hormone (FSH), testosterone, and estradiol levels were measured.
Glucose and lipid metabolism indicators and HOMA-IR: Fasting plasma glucose, fasting insulin, triglyceride, total cholesterol, and low-density lipoprotein cholesterol (LDL-C) levels were measured before and after 12 and 20 weeks of treatment. Insulin resistance was assessed using HOMA-IR, calculated as HOMA-IR = fasting insulin × fasting plasma glucose/22.5.
Menstrual cycle follow-up: Changes in menstrual cycle were tracked before treatment and at 12 and 20 weeks of treatment in both groups to assess any alterations in menstrual regularity.
Psychological status: Psychological well-being was assessed before treatment and at 12 and 20 weeks of treatment using the WHO-5, the Generalized Anxiety Disorder Scale (GAD-7), and the Patient Health Questionnaire Depression Scale (PHQ-9), with comparisons made between baseline and follow-up scores.
Statistical analyses
Statistical analyses were performed using SPSS software (version 23.0; IBM Corporation, New York, USA). For continuous data, results are presented as mean ± standard deviation (
RESULTS
Baseline characteristics
A total of 64 patients were recruited for this study. The semaglutide-intensified treatment group consisted of 32 patients, with a mean age of 32.77 ± 6.10 years and a mean body weight of 83.86 ± 11.55 kg. One patient was lost to follow-up owing to relocation, leaving 31 patients who completed the study. The standard treatment group included 32 patients with a mean age of 31.93 ± 4.81 years and a disease duration of 82.89 ± 9.92 months. Two patients voluntarily discontinued the treatment, and 30 completed the study. No significant differences were observed between the two groups in terms of age, body weight, waist circumference, BMI, or baseline sex hormone levels (all P > 0.05; Table 1), indicating that the groups were comparable.
Changes in body mass index, glucose and lipid metabolism, and sex hormones between the two groups before and after the treatment
| Parameters | Standard treatment group (n = 30) | Intensified treatment group (n = 31) | P baseline | P valuea | P valueb | ||||
| Baseline | Treatment after 12 weeks | Treatment after 20 weeks | Baseline | Treatment after 12 weeks | Treatment after 20 weeks | ||||
| Weight (kg) | 82.89 ± 9.92 | 80.05 ± 9.56# | 79.01 ± 9.29# | 83.86 ± 11.55 | 75.36 ± 11.2 | 74.04 ± 10.99 | 0.727 | 0.084 | 0.061 |
| BMI (kg/m2) | 31.40 ± 3.97 | 30.32 ± 3.78# | 29.92 ± 3.63# | 31.44 ± 4.30 | 28.26 ± 4.17* | 27.75 ± 4.06* | 0.971 | 0.048 | 0.032 |
| Absolute weight loss (kg) | - | 2.84 ± 1.60 | 3.88 ± 2.49 | - | 8.50 ± 2.74 | 9.82 ± 3.66 | - | 0.000 | 0.000 |
| WC (cm) | 96.82 ± 7.64 | 91.25 ± 9.10# | 93.53 ± 7.18# | 97.40 ± 9.45 | 90.08 ± 8.73 | 89.10 ± 8.66 | 0.972 | 0.032 | 0.033 |
| WHtR | 0.52 ± 0.06 | 0.49 ± 0.06# | 0.48 ± 0.05# | 0.52 ± 0.07 | 0.46 ± 0.07 | 0.45 ± 0.06 | 0.946 | 0.023 | 0.025 |
| Fat (%) | 37.02 ± 4.33 | 36.02 ± 4.43# | 35.41 ± 4.42# | 38.71 ± 4.47 | 33.14 ± 4.47 | 32.22 ± 4.42 | 0.140 | 0.014 | 0.007 |
| HC (cm) | 108.48 ± 6.84 | 106.53 ± 6.98# | 105.50 ± 6.68# | 108.75 ± 7.43 | 102.55 ± 8.08 | 100.62 ± 7.60 | 0.880 | 0.044 | 0.010 |
| TC (mmol/L) | 5.17 ± 0.70 | 4.87 ± 0.69# | 4.53 ± 0.70# | 5.35 ± 0.85 | 4.57 ± 0.57 | 4.36 ± 0.52 | 0.353 | 0.063 | 0.306 |
| TG (mmol/L) | 2.14 ± 0.83 | 1.97 ± 0.76 | 1.84 ± 0.66# | 2.23 ± 0.93 | 1.81 ± 0.68 | 1.63 ± 0.58 | 0.692 | 0.367 | 0.194 |
| LDL-C (mmol/L) | 3.09 ± 0.64 | 2.90 ± 0.54# | 2.72 ± 0.58# | 3.40 ± 0.61 | 2.81 ± 0.57 | 2.57 ± 0.45 | 0.057 | 0.528 | 0.264 |
| FPG (mmol/L) | 5.44 ± 0.60 | 5.15 ± 0.42# | 4.96 ± 0.42# | 5.26 ± 0.66 | 4.82 ± 0.47 | 4.65 ± 0.41 | 0.265 | 0.006 | 0.005 |
| FINS (mIU/L) | 24.67 ± 9.95 | 21.52 ± 8.63# | 19.52 ± 7.55# | 26.01 ± 10.06 | 17.63 ± 6.32 | 15.01 ± 4.97 | 0.603 | 0.048 | 0.011 |
| HOMA-IR | 5.99 ± 2.52 | 4.85 ± 1.89# | 4.27 ± 1.74# | 6.06 ± 2.42 | 3.76 ± 1.36 | 3.15 ± 1.14 | 0.907 | 0.011 | 0.005 |
| FSH (mIU/mL) | 5.84 ± 1.19 | 5.64 ± 1.13 | 5.35 ± 1.09# | 5.68 ± 1.15 | 5.35 ± 1.07 | 5.37 ± 1.20 | 0.594 | 0.308 | 0.959 |
| LH (mIU/mL) | 8.09 ± 2.40 | 6.59 ± 1.71# | 5.74 ± 1.67# | 8.33 ± 1.67 | 5.19 ± 1.05 | 4.62 ± 0.87 | 0.646 | 0.000 | 0.002 |
| E2 (pg/mL) | 44.56 ± 23.41 | 46.07 ± 23.02 | 42.75 ± 17.17 | 42.99 ± 17.60 | 37.07 ± 11.15 | 37.36 ± 12.76 | 0.768 | 0.056 | 0.168 |
| T (μg/L) | 0.75 ± 0.20 | 0.65 ± 0.14# | 0.60 ± 0.09# | 0.79 ± 0.17 | 0.59 ± 0.09 | 0.54 ± 0.11 | 0.406 | 0.049 | 0.014 |
| LH/FSH | 1.42 ± 0.47 | 1.18 ± 0.27# | 1.07 ± 0.24# | 1.52 ± 0.40 | 1.00 ± 0.25 | 0.89 ± 0.18 | 0.379 | 0.011 | 0.001 |
Comparison of therapeutic outcomes between the semaglutide-intensified and standard treatment groups
Comparison of weight-related parameters
In the semaglutide-intensified treatment group, significant reductions in BMI, absolute weight loss, waist, hip circumference, WHtR and body fat percentage were observed at 12 and 20 weeks compared with baseline (P < 0.05). Furthermore, at both 12 and 20 weeks post-treatment, BMI, waist and hip circumference, WHtR, absolute weight loss, and body fat percentage in the semaglutide-intensified treatment group were significantly lower than those in the standard treatment group (P < 0.05; Table 1).
After 12 weeks of treatment, 28 patients (90.32%) in the semaglutide-intensified treatment group achieved a weight reduction of > 5%, and 15 patients (48.39%) achieved a reduction of > 10%. At 20 weeks, 27 patients (87.10%) experienced weight loss exceeding 5%, and 17 patients (54.83%) lost > 10% of their body weight. The proportion of patients who achieved > 5% and > 10% weight reduction was significantly higher in the semaglutide-intensified treatment group than in the standard treatment group (P < 0.05; Table 2).
Changes in the percentage of weight loss between the two groups before and after treatment [cases (%)]
| Standard treatment group (n = 30) | Intensified treatment group (n = 31) | χ 2 | P value | |
| Weight loss ≥ 5% | ||||
| Treatment after 12 weeks | 7 (23.33) | 28 (90.32) | 17.281 | 0.000 |
| Treatment after 20 weeks | 14 (46.67) | 27 (87.10) | 12.170 | 0.000 |
| Weight loss ≥ 10% | ||||
| Treatment after 12 weeks | 0 (0.00) | 15 (48.39) | 19.250 | 0.000 |
| Treatment after 20 weeks | 0 (0.00) | 17 (54.83) | 16.497 | 0.000 |
Comparison of sex hormone indicators
In the semaglutide-intensified treatment group, significant reductions in LH and androgen levels and in the LH/FSH ratio were observed at 12 and 20 weeks of treatment, with significant differences compared to baseline (P < 0.05) [Table 1]. The semaglutide-intensified treatment group showed greater reductions in LH and androgen levels and the LH/FSH ratio than the standard treatment group, with significant intergroup differences (P < 0.05). No significant differences were observed in estradiol or total estrogen levels between the two groups (P > 0.05) [Table 1].
Comparison of biochemical indicators
At 12 and 20 weeks of treatment, the semaglutide-intensified treatment group demonstrated significant reductions in fasting plasma glucose, fasting insulin, and HOMA-IR compared to the standard treatment group, with significant differences (P < 0.05). Although the semaglutide-intensified group showed lower total cholesterol, triglyceride, and LDL-C levels than the standard treatment group, these differences were not significant (P > 0.05) [Table 1].
Comparison of established menstrual cycles
Although no participants had amenorrhea, oligomenorrhea was prevalent among certain participants at study enrollment. In the semaglutide-intensified treatment group, 24 patients (77.41%) established a regular menstrual cycle by week 12, increasing to 26 patients (86.87%) by week 20. These differences were significant compared with the baseline values (P < 0.05). Furthermore, at week 20, the number of patients with established menstrual cycles in the semaglutide-intensified treatment group was significantly higher than that in the standard treatment group (P = 0.038) [Table 3].
Comparison of menstrual cycle recovery after 20 weeks of treatment between the two groups [cases (%)]
| Standard treatment group (n = 30) | Intensified treatment group (n = 31) | χ 2 | P value | |
| Menstrual cycle | ||||
| Before treatment | 13 (43.33) | 12 (38.71) | 0.135 | 0.714 |
| Treatment after 12 weeks | 17 (56.67) | 24 (77.41) | 2.980 | 0.084 |
| Treatment after 20 weeks | 18 (60.00) | 26 (86.87) | 4.322 | 0.038 |
Comparison of mental and psychological scale levels
After 20 weeks of treatment, the semaglutide-intensified treatment group demonstrated a significant increase in the WHO-5 compared to baseline, whereas GAD-7 Anxiety Assessment and PHQ-9 Depression Scales scores were significantly reduced (P < 0.05). Although increased WHO-5 scores and decreased GAD-7 and PHQ-9 scores were observed in the semaglutide-intensified treatment group compared to those in the standard treatment group, these differences were not significant (P > 0.05) [Table 4].
Comparison of mental and psychological scale levels between the two groups before and after treatment (
| Standard treatment group (n = 30) | Intensified treatment group (n = 31) | t | P value | |
| WHO-5 | ||||
| Before treatment | 48.00 ± 12.99 | 49.61 ± 16.59 | -0.422 | 0.675 |
| Treatment after 12 weeks | 55.07 ± 12.50 | 55.81 ± 14.65 | -0.212 | 0.833 |
| Treatment after 20 weeks | 57.93 ± 15.83 | 60.26 ± 15.75* | -0.575 | 0.568 |
| GAD-7 | ||||
| Before treatment | 4.87 ± 1.93 | 5.19 ± 4.19 | -0.389 | 0.698 |
| Treatment after 12 weeks | 3.80 ± 1.73 | 3.90 ± 3.54* | -0.144 | 0.886 |
| Treatment after 20 weeks | 3.43 ± 2.31 | 2.74 ± 2.92* | 1.022 | 0.311 |
| PHQ-9 | ||||
| Before treatment | 7.13 ± 3.66 | 7.58 ± 3.93 | -0.460 | 0.647 |
| Treatment after 12 weeks | 5.43 ± 3.05 | 5.19 ± 3.17* | 0.301 | 0.764 |
| Treatment after 20 weeks | 4.60 ± 3.16 | 3.90 ± 2.89* | 0.899 | 0.372 |
Adverse events
No major medical complications were documented throughout the treatment phase, and all participants remained in the trial with no attrition associated with adverse drug reactions. Clinical manifestations were primarily confined to the digestive tract and characterized by mild-to-moderate episodes of nausea, vomiting, diarrhea, and abdominal distension. A small number of patients reported headache and general malaise. These symptoms were typically transient and largely resolved as the participants adapted to the intervention within the first month.
DISCUSSION
This study indicated that after 12 weeks of treatment, 90.32% of patients in the semaglutide-intensified treatment group experienced a weight reduction. At 20 weeks, 87.10% of the patients maintained a weight reduction > 5%, and 54.83% sustained a loss of over 10%. Consistently, the STEP (Semaglutide Treatment Effect in People with Obesity) trial[22] demonstrated that after 68 weeks of semaglutide therapy in individuals who are overweight or obese, the average weight loss achieved was 14.9%, with 86.4% of participants achieving a reduction of at least 5% from baseline, and 69.1% and 50.5% achieving reductions exceeding 10% and 15%, respectively.
Women with PCOS are frequently overweight or obese and exhibit a greater abdominal circumference than women with isolated obesity. Reductions in BMI, particularly visceral fat, may play a pivotal role in mitigating infertility risk factors and ameliorating hyperandrogenism and its associated clinical symptoms[30,31]. Although metformin is a pharmacological option for patients living with obesity and PCOS, it does not significantly reduce weight or modify fat distribution. Animal studies have suggested that metformin enhances the weight loss efficacy of GLP-1 RAs by stimulating GLP-1 receptor expression and augmenting insulin-mediated signaling pathways. In this study, the semaglutide-metformin combination showed that although no significant difference was observed in overall weight loss between the semaglutide-intensified and standard treatment groups, BMI, waist circumference, hip circumference, and body fat percentage were significantly lower in the semaglutide-intensified group. Additionally, absolute weight loss and the proportion of patients with ≥ 5% weight loss were significantly greater in the semaglutide-intensified treatment group than in the standard treatment group, confirming that semaglutide injection promotes weight reduction and reduces waist circumference in women living with obesity and PCOS-related infertility.
Both metformin and GLP-1 RAs can induce ovulation and restore the menstrual cycle[32-35]. A placebo-controlled trial reported that after 26 weeks of treatment with liraglutide, menstrual frequency increased and ovarian function improved[29]. Additionally, a pilot randomized trial reported that short-term combination treatment with beinaglutide and metformin was more effective than metformin monotherapy in lowering body weight, improving insulin sensitivity, and reducing androgen excess in women living with PCOS and obesity[33]. In this study, a significant increase was observed after 12 weeks of treatment with metformin combined with semaglutide injections in the number of patients living with overweight/obesity and PCOS-related infertility who established regular menstrual cycles, with a higher maintenance rate than in the metformin-only group. This finding suggests that semaglutide injections may be beneficial for establishing menstrual cycle regularity in this population. In addition, women’s menstrual cycles are related to ovarian reserve function, and improvements in the menstrual cycle are beneficial for birth and pregnancy outcomes after reproductive fertility[36,37]. The synergistic pharmacological mechanisms of GLP-1 RAs combined with metformin may modulate the expression of reproductive function-related hormonal indicators, thereby ameliorating endocrine disturbances and restoring normal menstrual cycles. Semaglutide injection may promote conception by regulating the menstrual cycle and improving ovulatory function in obese patients with PCOS who experience infertility. With an extended follow-up, potential improvements in pregnancy outcomes could be observed.
The pathophysiological mechanisms underlying PCOS are closely associated with the dysfunction of the hypothalamic-pituitary-ovarian axis, hyperandrogenemia, insulin resistance, and adrenal endocrine disorders. Hyperandrogenemia, characterized by an elevated LH/FSH ratio, represents a significant hormonal derangement in patients living with infertility and PCOS[38]. Furthermore, insulin resistance and hyperinsulinemia are critical contributors to the increased androgen secretion observed in this population[39].
GLP-1 RAs mitigate obesity, improve insulin sensitivity, and exert beneficial effects on sex hormone levels, glucose-lipid metabolism, and reproductive function in patients living with PCOS[28,40]. However, previous randomized controlled trials were generally characterized by small sample sizes, single-center designs, and limited follow-up durations. GLP-1 RAs enhance the regulatory mechanisms of the hypothalamic-pituitary-ovarian axis, affecting LH surges or reductions induced by estrogen aromatization in adipose tissue, and subsequently modulating LH and FSH levels that drive ovarian androgen synthesis[41]. In the present study, significant reductions in testosterone, LH, LH/FSH ratio, fasting insulin, and HOMA-IR were observed in the semaglutide-intensified treatment group compared to baseline measurements after 12 weeks of semaglutide treatment. These parameters continued to decline compared to those in the standard treatment group. Even after a 20-week washout period, the semaglutide-intensified treatment group maintained lower testosterone and LH levels, LH/FSH ratio, fasting insulin, and HOMA-IR, suggesting that semaglutide injections could improve insulin resistance and modulate androgen and sex hormone levels in patients living with obesity and infertility. A study in mice with PCOS found that semaglutide alleviated autophagy and ovarian oxidative stress via the PI3K/AKT/mTOR (phosphatidylinositol 3 kinase/protein kinase B/mammalian target of rapamycin) pathway[42], further establishing a theoretical framework for the clinical application of GLP-1 RAs in PCOS.
Patients living with obesity and PCOS often present significant visceral fat accumulation, resulting in elevated free fatty acid release that contributes to increased hepatic total cholesterol synthesis and dyslipidemia, manifesting as elevated triglyceride, total cholesterol, and LDL-C levels. GLP-1 RAs enhance insulin sensitivity and promote insulin secretion, thereby facilitating glucose homeostasis, and assist in lipid management by reducing caloric intake and improving glucose-lipid metabolism in patients with PCOS. In a study involving 30 patients living with obesity and PCOS, those treated with liraglutide, either alone or in combination with metformin, for 12 weeks demonstrated significant improvements in glycemic control and fasting insulin levels[43]. Consistent with these findings, the current study confirmed that the semaglutide-intensified treatment group exhibited reduced fasting plasma glucose, fasting insulin, total cholesterol, triglyceride, and LDL-C levels compared with baseline. This effect may be attributed to the appetite-suppressing and weight-reducing effects of GLP-1 RAs, which decrease postprandial lipoprotein secretion and mitigate systemic inflammation. However, no significant differences in lipid parameters were observed when compared to the standard treatment group, which may be attributed to the small sample size and considerable heterogeneity among the study participants.
Patients living with infertility and PCOS frequently exhibit varying degrees of metabolic dysregulation and reproductive dysfunction, along with potential psychological health risks stemming from long-term fertility challenges and concerns regarding body image. The prevalence of anxiety and depression among patients is 37% and 42%, respectively, with obese individuals being more susceptible to negative emotional states[44]. Insulin resistance is also associated with an increased risk of depression in patients with PCOS[45]. A recent meta-analysis evaluating the effects of GLP-1 RAs on depression found that these agents did not increase the risk of depression and may reduce depression scores[46]. This finding aligns with the results of this study, where the semaglutide-intensified treatment group demonstrated an increase in the WHO-5 and a decrease in anxiety and depression scores compared to baseline. These findings suggest that semaglutide injections can enhance the well-being of patients living with obesity and PCOS-related infertility and alleviate negative psychological emotions. The lack of significant differences in well-being indices and anxiety and depression scores between the two groups may be attributable to numerous confounding psychological factors, strong patient heterogeneity, small sample size, and short follow-up duration.
This study had several limitations. First, the single-center study design limits the extrapolation of the findings to diverse populations and may introduce selection bias. Second, the lack of characterized liver tests/ultrasonography in the patient population precludes the evaluation of obesity-related liver complications. Additionally, the study was constrained by a relatively short follow-up period, which prevented the assessment of long-term reproductive outcomes such as live birth rates. Although the short-term results are promising, post-treatment weight rebound and subsequent pregnancy outcomes are key factors that require validation through extended longitudinal studies.
In conclusion, the addition of semaglutide injections to lifestyle and metformin interventions can reduce BMI, waist and hip circumference, and body fat percentage, alleviate hyperandrogenemia, improve glucose metabolism and insulin resistance, and promote the restoration of normal menstrual cycles without exacerbating negative psychological emotions in individuals living with overweight/obesity and PCOS-related infertility. Therefore, semaglutide represents a novel intervention strategy for weight reduction and improved insulin resistance, providing new insights into the clinical management of patients living with obesity and PCOS. However, multicenter, large-sample studies are warranted to further explore the therapeutic effects of semaglutide on individuals living with obesity and PCOS-related infertility.
DECLARATIONS
Acknowledgements
The authors thank all participants and investigators involved in this study.
Authors’ contributions
Designed and conducted the study, performed data acquisition and analysis, and wrote the manuscript: Zhang W, Xue J
Collected and managed data: Wang J, Ni D, Zhou W
Performed data management: Wang B, Shi J
Conceived and designed the study, conducted it, interpreted the data, and revised the manuscript: Wang A
All the authors have read and approved the final version of this manuscript.
Availability of data and materials
The datasets generated and/or analyzed in the current study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Studies involving humans were approved by the Ethics Committee of the Ninth Medical Center of the PLA General Hospital, Beijing, China (Approval number: K2021-No.02). The studies were conducted in accordance with local legislation and institutional requirements. All the participants provided written informed consent to participate in this study.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Datta J, Palmer MJ, Tanton C, et al. Prevalence of infertility and help seeking among 15 000 women and men. Hum Reprod. 2016;31:2108-18.
2. Zhou Z, Zheng D, Wu H, et al. Epidemiology of infertility in China: a population-based study. BJOG. 2018;125:432-41.
3. Zhang X, Zhang M, Zhao Z, et al. Geographic variation in prevalence of adult obesity in china: results from the 2013-2014 national chronic disease and risk factor surveillance. Ann Intern Med. 2020;172:291-3.
4. Barbouni K, Jotautis V, Metallinou D, et al. When weight matters: how obesity impacts reproductive health and pregnancy - a systematic review. Curr Obes Rep. 2025;14:37.
5. Johnson AP, Vu TT, Simon J, Ryan GL. Special considerations in assisted reproductive technology for patients with obesity. Fertil Steril. 2025;124:212-8.
7. Wu M, Huang Y, Zhu Q, et al. Adipose tissue and ovarian aging: potential mechanism and protective strategies. Ageing Res Rev. 2022;80:101683.
8. Rafael F, Rodrigues MD, Bellver J, et al. The combined effect of BMI and age on ART outcomes. Hum Reprod. 2023;38:886-94.
9. Wang RQ, Deng ZM, Chen GT, Dai FF, Xia LB. Obesity and recurrent spontaneous abortion: the crucial role of weight management in pregnancy. Reprod Biol Endocrinol. 2025;23:10.
10. Cozzolino M, García-Velasco JA, Meseguer M, Pellicer A, Bellver J. Female obesity increases the risk of miscarriage of euploid embryos. Fertil Steril. 2021;115:1495-502.
11. Palomba S, Piltonen TT, Giudice LC. Endometrial function in women with polycystic ovary syndrome: a comprehensive review. Hum Reprod Update. 2021;27:584-618.
12. Balen AH, Morley LC, Misso M, et al. The management of anovulatory infertility in women with polycystic ovary syndrome: an analysis of the evidence to support the development of global WHO guidance. Hum Reprod Update. 2016;22:687-708.
13. Tay CT, Loxton D, Bahri Khomami M, Teede H, Harrison CL, Joham AE. High prevalence of medical conditions and unhealthy lifestyle behaviours in women with PCOS during preconception: findings from the Australian Longitudinal Study on Women’s Health. Hum Reprod. 2023;38:2267-76.
14. Zhang Y, Chen ZJ, Zhao H. Polycystic ovary syndrome: a metabolic disorder with therapeutic opportunities. Cell Metab. 2025;37:1932-49.
15. Experts Consensus Group on the Body Quality Management Approach and Process for Overweight/Obese Infertility Patients. Expert consensus on the weight management of overweight/obese infertility patients in China. Chin J Reprod Contracept. 2020;40:965-71. (in Chinese).
16. Ruiz-González D, Cavero-Redondo I, Hernández-Martínez A, et al. Comparative efficacy of exercise, diet and/or pharmacological interventions on BMI, ovulation, and hormonal profile in reproductive-aged women with overweight or obesity: a systematic review and network meta-analysis. Hum Reprod Update. 2024;30:472-87.
17. Saadati S, Mason T, Godini R, Vanky E, Teede H, Mousa A. Metformin use in women with polycystic ovary syndrome (PCOS): opportunities, benefits, and clinical challenges. Diabetes Obes Metab. 2025;27 Suppl 3:31-47.
18. Sharpe A, Morley LC, Tang T, Norman RJ, Balen AH. Metformin for ovulation induction (excluding gonadotrophins) in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2019;12:CD013505.
19. Abu-Hamdah R, Rabiee A, Meneilly GS, Shannon RP, Andersen DK, Elahi D. Clinical review: the extrapancreatic effects of glucagon-like peptide-1 and related peptides. J Clin Endocrinol Metab. 2009;94:1843-52.
20. Drucker DJ. Efficacy and safety of GLP-1 medicines for type 2 diabetes and obesity. Diabetes Care. 2024;47:1873-88.
21. Papamargaritis D, le Roux CW, Holst JJ, Davies MJ. New therapies for obesity. Cardiovasc Res. 2024;119:2825-42.
22. Wilding JPH, Calanna S, Kushner RF. Once-weekly semaglutide in adults with overweight or obesity. Reply. N Engl J Med. 2021;385:e4.
23. Garvey WT, Batterham RL, Bhatta M, et al.; STEP 5 Study Group. Two-year effects of semaglutide in adults with overweight or obesity: the STEP 5 trial. Nat Med. 2022;28:2083-91.
24. Tzang CC, Wu PH, Luo CA, et al. Metabolic rebound after GLP-1 receptor agonist discontinuation: a systematic review and meta-analysis. EClinicalMedicine. 2025;90:103680.
25. Jensen SBK, Blond MB, Sandsdal RM, et al. Healthy weight loss maintenance with exercise, GLP-1 receptor agonist, or both combined followed by one year without treatment: a post-treatment analysis of a randomised placebo-controlled trial. EClinicalMedicine. 2024;69:102475.
26. Jensterle M, Janez A, Fliers E, DeVries JH, Vrtacnik-Bokal E, Siegelaar SE. The role of glucagon-like peptide-1 in reproduction: from physiology to therapeutic perspective. Hum Reprod Update. 2019;25:504-17.
27. Salamun V, Jensterle M, Janez A, Vrtacnik Bokal E. Liraglutide increases IVF pregnancy rates in obese PCOS women with poor response to first-line reproductive treatments: a pilot randomized study. Eur J Endocrinol. 2018;179:1-11.
28. Liu X, Zhang Y, Zheng SY, et al. Efficacy of exenatide on weight loss, metabolic parameters and pregnancy in overweight/obese polycystic ovary syndrome. Clin Endocrinol. 2017;87:767-74.
29. Nylander M, Frøssing S, Clausen HV, Kistorp C, Faber J, Skouby SO. Effects of liraglutide on ovarian dysfunction in polycystic ovary syndrome: a randomized clinical trial. Reprod Biomed Online. 2017;35:121-7.
30. Siamashvili M, Davis SN. Update on the effects of GLP-1 receptor agonists for the treatment of polycystic ovary syndrome. Expert Rev Clin Pharmacol. 2021;14:1081-9.
31. Darıcı E, Pais F, Leemans L, et al. From body mass index to body composition in female infertility. Reprod Biomed Online. 2025;51:104941.
32. Ling J, Wang T, Huang W, et al. Combined liraglutide and metformin therapy in overweight or obese women with polycystic ovary syndrome: a systematic review and meta-analysis. Diabetes Obes Metab. 2025;27:6139-53.
33. Wen Q, Fang S, Liang Y, et al. Short-term effect of beinaglutide combined with metformin versus metformin alone on weight loss and metabolic profiles in obese patients with polycystic ovary syndrome: a pilot randomized trial. Front Endocrinol. 2023;14:1156521.
34. Huang R, He Y. The safety and efficacy of liraglutide combined with metformin in clinical treatment of polycystic ovary syndrome patients: a meta-analysis. BMC Womens Health. 2025;25:282.
35. Xing C, Zhao H, Zhang J, He B. Effect of metformin versus metformin plus liraglutide on gonadal and metabolic profiles in overweight patients with polycystic ovary syndrome. Front Endocrinol. 2022;13:945609.
36. Carmina E, Longo RA. Semaglutide treatment of excessive body weight in obese PCOS patients unresponsive to lifestyle programs. J Clin Med. 2023;12:5921.
37. Younis JS, Iskander R, Fauser BCJM, Izhaki I. Does an association exist between menstrual cycle length within the normal range and ovarian reserve biomarkers during the reproductive years? Hum Reprod Update. 2020;26:904-28.
38. Zeng X, Xie YJ, Liu YT, Long SL, Mo ZC. Polycystic ovarian syndrome: correlation between hyperandrogenism, insulin resistance and obesity. Clin Chim Acta. 2020;502:214-21.
39. Lazaridou S, Dinas K, Tziomalos K. Prevalence, pathogenesis and management of prediabetes and type 2 diabetes mellitus in patients with polycystic ovary syndrome. Hormones. 2017;16:373-80.
40. Roberts R, Markande A, Kasaven L, et al. Obesity and female reproductive health; is there a role for glucagon-like peptide-1 receptor agonists? Obes Rev. 2026;27:e70015.
41. Bednarz K, Kowalczyk K, Cwynar M, et al. The role of Glp-1 receptor agonists in insulin resistance with concomitant obesity treatment in polycystic ovary syndrome. Int J Mol Sci. 2022:23.
42. Guo S, Li X, Liu M, et al. Semaglutide alleviates ovarian oxidative stress and autophagy via the PI3K/AKT/mTOR pathway in mice with polycystic ovary syndrome. Drug Des Devel Ther. 2025;19:4297-310.
43. Jensterle M, Kravos NA, Goričar K, Janez A. Short-term effectiveness of low dose liraglutide in combination with metformin versus high dose liraglutide alone in treatment of obese PCOS: randomized trial. BMC Endocr Disord. 2017;17:5.
44. Wang Y, Ni Z, Li K. The prevalence of anxiety and depression of different severity in women with polycystic ovary syndrome: a meta-analysis. Gynecol Endocrinol. 2021;37:1072-8.
45. Greenwood EA, Pasch LA, Cedars MI, Legro RS, Eisenberg E, Huddleston HG; Eunice Kennedy Shriver National Institute of Child Health and Human Development Reproductive Medicine Network. Insulin resistance is associated with depression risk in polycystic ovary syndrome. Fertil Steril. 2018;110:27-34.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].