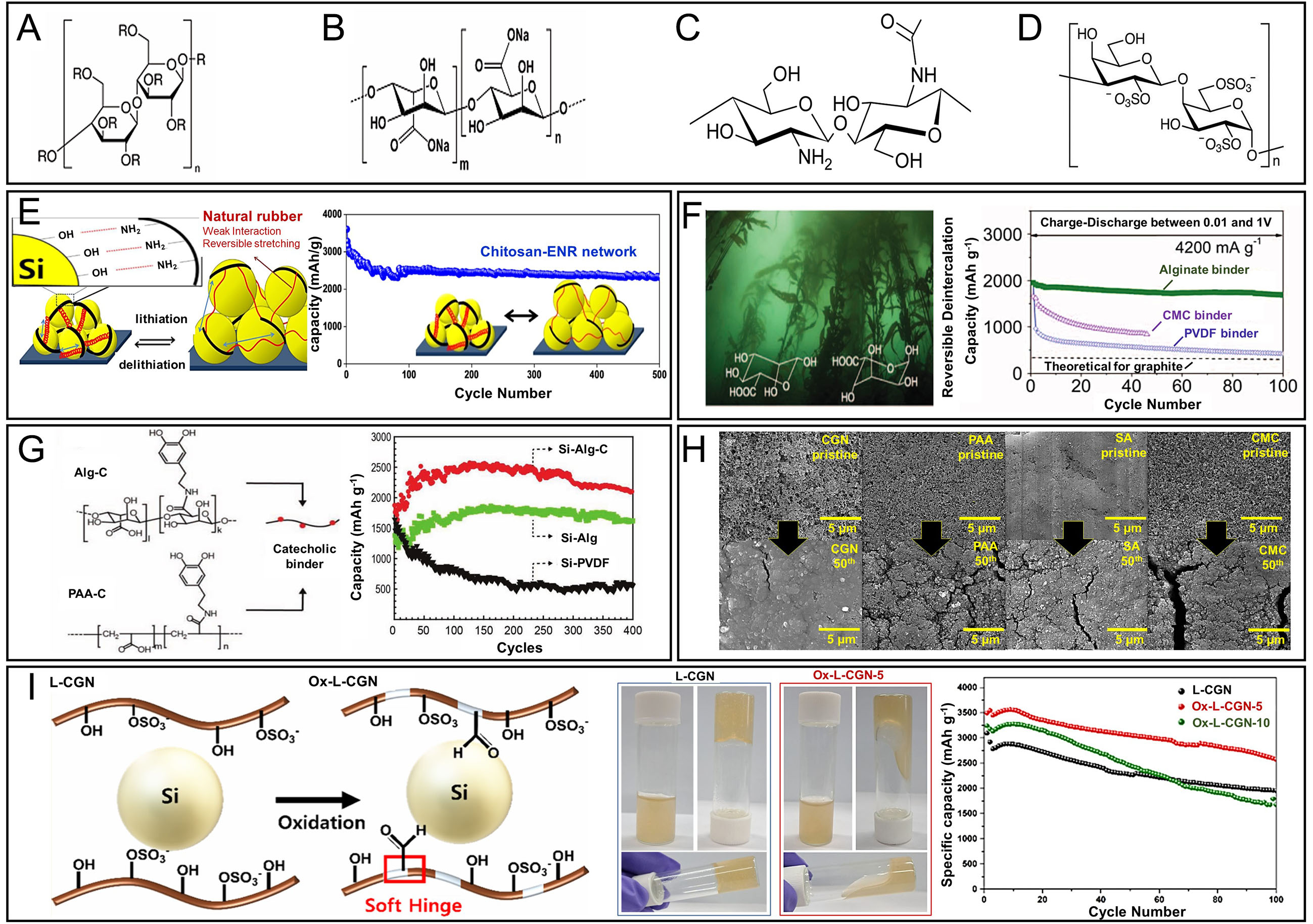
fig4
Figure 4. Chemical structures of polymer binders containing hydrophilic functional groups and their effects on Si anode performance. (A) CMC; (B) SA; (C) CS; (D) L-CGN; (E) Design of the binder material (left) and cycling stability (right) of a Si anode using a natural rubber/chitosan network binder. Adapted with permission from ref.[72]; (F) Chemical structures of the mannuronic and guluronic acid repeat units (left); cycling performance of nano-Si anodes with alginate (SA), CMC, and PVDF binders (right; 0.01-1.0 V vs. Li/Li+). Adapted from ref.[64]; (G) Catechol-conjugated polymer binders and cycling performance of Si electrodes using Alg-C, Alg, and PVDF binders. Adapted from ref.[64]; (H) Cycling performance of Si electrodes using different natural polymer binders (CGN, PAA, SA, CMC). The Si@CGN electrode demonstrated the highest capacity retention over 100 cycles, underscoring the superior binding and structural stabilizing effect of lambda carrageenan. Adapted with permission from ref.[71]; (I) Schematic illustration of the synthesis of Ox-L-CGN, comparison of its solubility in distilled water with that of L-CGN, and cycling performance of Si electrodes using different polymer binders. The optimized Ox L-CGN 5 binder demonstrated improved solubility and enhanced electrochemical performance, attributed to its optimized viscosity and formation of a stable polymer network. Adapted with permission from ref.[70].