Nanoplastics remodel the extracellular matrix mechanical microenvironment to activate hepatic stellate cells
Abstract
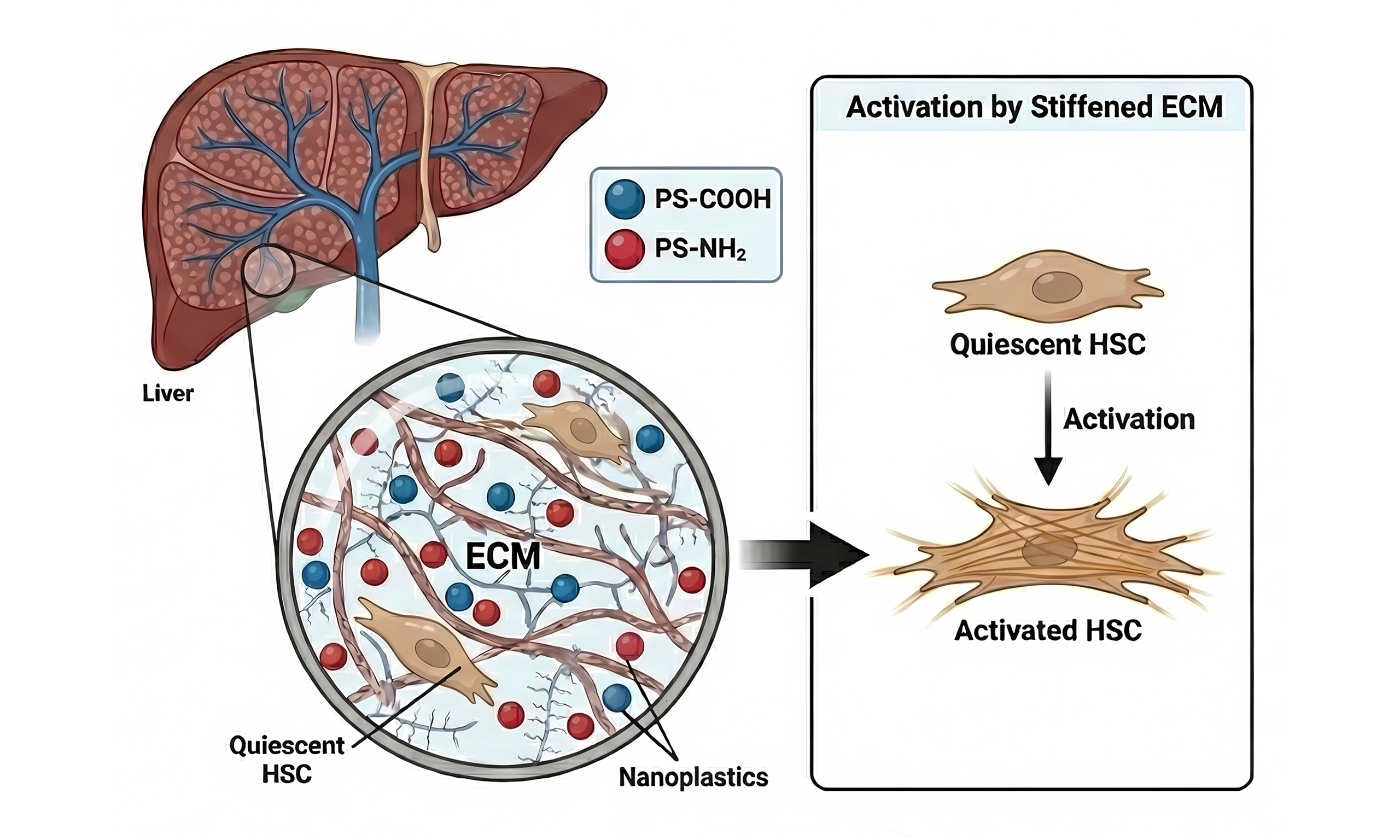
Nanoplastics, an emerging class of environmental contaminants, have become a growing concern due to their widespread distribution and potential to interfere with cellular and tissue homeostasis. The liver, as a primary site of xenobiotic metabolism and clearance, is particularly vulnerable to nanoplastic exposure. Hepatic stellate cells (HSCs) play a central role in maintaining liver extracellular matrix (ECM) homeostasis and in initiating fibrogenic responses; therefore, understanding how nanoplastics affect HSC behavior is critical for elucidating early mechanisms of nanoplastic-induced liver injury. To achieve physiologically relevant modeling, we employed HSC and collagen hydrolysate to construct a simplified matrix–cell–matrix mimicking the microenvironment of the hepatic space of Disse, we found that both aminated (PS-NH2) and carboxylated (PS-COOH) polystyrene nanoplastics were retained within collagen hydrolysate matrices and altered their viscoelastic properties, with more significantly negatively charged PS-COOH increasing matrix viscosity. HSCs cultured within PS-COOH–treated matrices exhibited enhanced proliferation and migration without apparent cytotoxicity, accompanied by intensified F-actin stress fiber formation and nuclear translocation of the mechanosensitive coactivator yes-associated protein (YAP). Together, these findings indicate that nanoplastics may indirectly activate quiescent HSCs by remodeling ECM mechanical properties, rather than through direct cellular uptake. This work provides a mechanobiological perspective linking environmental nanoplastic exposure to early hepatic fibrogenesis.
Keywords
INTRODUCTION
With the increasing severity of plastics pollution, micro/nanoplastics have been confirmed to accumulate in human tissues, including the brain, blood, intestines, and liver[1-4]. Micro/nanoplastics are primarily ingested through dietary intake and inhalation[5]. The nanoplastics can enter the liver via the hepatic portal vein, which is connected to the gastrointestinal tract, leading to the liver as a primary target organ for the toxicity of ingested nanoplastics[6].
During processing or degradation, micro/nanoplastics acquire various surface charges, contributing to distinct toxicological mechanisms. In mammalian models, amino-functionalized polystyrene (PS-NH2) exacerbates non-alcoholic fatty liver disease (NAFLD) in mice, closely associated with hepatocyte damage, insulin resistance, and disruption of immune homeostasis[7,8]. In vitro, positively charged microplastics are prone to bind to negatively charged cell membranes, facilitating cellular uptake, and inducing reactive oxygen species (ROS) accumulation and oxidative damage[9,10]. For example, PS-NH2 induces malondialdehyde (MDA) accumulation and glutathione (GSH) depletion in human hepatocellular carcinoma cell line (HepG2), leading to oxidative cellular damage[11]. Similarly, mouse experiments demonstrated that carboxyl-functionalized polystyrene (PS-COOH) inhibits fatty acid β-oxidation in the liver, impairing lipid catabolism and exacerbating NAFLD development[12]. Negatively charged microplastics disrupt G protein-coupled receptor (GPCR) signaling on the cell membrane and inhibit the peroxisome proliferator-activated receptor (PPAR) lipid metabolism pathway, leading to developmental abnormalities and hepatic lipid accumulation[13].
From a physical perspective, these findings suggest that surface charge mediates electrostatic interactions between micro/nanoplastics and the phospholipid bilayer of cell membranes[14]. Previous studies have shown that microplastics can increase erythrocyte membrane tension through physical adsorption, thereby exerting mechanical stress on cells[15]. Positively charged nanoplastics can mechanically perturb cell membranes through electrostatic adsorption[15]. Notably, negatively charged nanoplastics have also been shown to induce the aggregation of stress fibers directly associated with cytoskeletal mechanotransduction in neural cells[16], suggesting that both positive and negative surface charges are capable of disturbing cellular mechanical homeostasis. Such disruption of cellular mechanical homeostasis can directly lead to functional impairment or cell death, representing one of the potential mechanisms underlying micro-/nanoplastic-induced toxicity.
The mechanical microenvironment in liver tissue, involved in extracellular matrix (ECM), cell membrane, and cytoskeleton, encompasses NAFLD progression including abnormal liver tissue deformation, inflammation, lipid metabolism disorders, and fibrosis[17,18]. Consequently, the effect of the surface charge of micro- and nanoplastics on liver tissue mechanical homeostasis represents a critical scientific issue in elucidating the mechanisms of their hepatotoxicity. Hepatic stellate cells (HSCs) are key regulators of liver mechanical homeostasis. During NAFLD progression, HSCs become activated, leading to fibrosis and collagen hydrolysate accumulation in the ECM, which disrupts the liver’s mechanical microenvironment and exacerbates disease progression.
In this study, a three-dimensional (3D) ECM-HSC-ECM model was developed to simulate the physiological interaction of nanoplastics with HSCs under physiological conditions. The study investigates the impact of the surface charge of nanoplastics on HSCs and the mechanical microenvironment in liver tissue at human-relevant concentrations, elucidating novel mechanisms underlying the hepatotoxicity of nanoplastics driven by surface charge.
EXPERIMENTAL
Preparation of nanoplastics
PS-COOH was selected as the representative negatively charged nanoplastics, while PS-NH2 was chosen as the representative positively charged nanoplastics. Both types had an average particle size of 100 nm and were purchased from Jiangsu Zhichuan Technology Co. Ltd. (Nanjing, China). The stock suspensions had a concentration of 20 mg/mL. To prevent aggregation, 0.09% sodium azide surfactant was added, and the suspensions were stored at 4 °C. The working suspensions were prepared by diluting the nanoplastic stock solutions with ultrapure water followed by ultrasonication for 30 min. Ultrapure water was used as the solvent control (designated as Ctrl). Glass centrifuge tubes were used for the storage of nanoplastics suspensions.
Cell culture
Human hepatic stellate cell line (LX2) is an immortalized cell line derived from human HSCs through induction and was obtained from Wuhan Procell Life Science & Technology Co., Ltd. (Wuhan, China; Procell, CL-0506). LX2 cells were cultured in a complete growth medium under standard conditions of 5% CO2 at 37 °C, with medium renewal every other day. When cell confluence reached approximately 80%, subculturing was performed using trypsin (Gibco, Waltham, MA, USA, R001100) for enzymatic dissociation. The complete LX2 culture medium consisted of 80% high-glucose Dulbecco’s Modified Eagle Medium (DMEM; Gibco, 11965084), supplemented with 1% penicillin–streptomycin (Gibco, 15140122) and 20% fetal bovine serum (FBS; Sigma, St. Louis, MO, USA, F0193).
Immunofluorescence staining of cellular markers
Cells were fixed with 4% paraformaldehyde (Beyotime, Shanghai, China, P0099) at room temperature for
Quantitative real-time polymerase chain reaction
Total RNA was extracted from cells using TRIzol reagent (Thermo Fisher Scientific), and complementary DNA (cDNA) was synthesized using the High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific). Quantitative real-time polymerase chain reaction (qRT-PCR) was performed on an ABI PRISM 7900HT Fast Real-Time PCR System (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA, USA) with SYBR Green Master Mix (Thermo Fisher Scientific). Target gene expression levels were normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and relative expression was calculated using the 2-ΔΔCt method. Gene-specific primers were synthesized by Integrated DNA Technologies (Thermo Fisher Scientific), and their sequences are listed as follows: collagen type I alpha 1 chain (COL1A1), Forward: GATTCCCTGGACCTAAAGGTGC; Reverse: AGCCTCTCCATCTTTGCCAGCA; alpha-smooth muscle actin (ACTA2), Forward: CTATGCCTCTGGACGCACAAC; Reverse: CAGATCCAGACGCATGATGGCA.
Dynamic light scattering and Zeta potential
A mixture containing 32.5 µL PBS, 50 µL of 100 nm polystyrene (PS) nanoplastics, and 12.5 µL of 0.8% collagen hydrolysate (Sigma, 73865) solution was sonicated for 5 min. Samples were transferred into a Zeta potential cell and analyzed using a Malvern Zetasizer (Mastersizer 3000E, Malvern Instruments, UK). Each experiment was independently repeated three times, and the results are presented as mean ± standard deviation (SD).
Cytotoxicity assay
LX2 cells were seeded in 48-well plates at a density of 5 × 104 cells per well and then exposed to various concentrations of PS-NH2 or PS-COOH suspensions without medium replacement. After 24 and 48 h of exposure, cell viability was evaluated using the Cell Counting Kit-8 (CCK-8; Sigma, 96992) assay and measured with a microplate reader. Cell viability was calculated as (experimental group/control group) × 100%.
Real-time observation of fluorescent nanoplastics
A thin layer of gelatin methacryloyl (GelMA)/lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) hydrogel was added to confocal culture dishes and crosslinked under 405 nm light to allow gelation. LX2 cells were then seeded onto the hydrogel in medium and cultured for 24 h to promote cell attachment. Subsequently, a 20% gelatin (Sigma, 73865) layer was applied on top of the cell-laden GelMA hydrogel to mimic the ECM environment.
The fluorescent PS-NH2 and PS-COOH nanoparticles exhibited intrinsic fluorescence resulting from the incorporation of europium ions into the polymer matrix, rather than from chemical surface labeling. Therefore, the fluorescence did not interfere with the surface functional groups or alter the surface charge properties of the nanoparticles. PS-NH2 and PS-COOH nanoparticles were then added to the cultures, and real-time nanoparticle internalization was monitored at 1 and 6 h using a Nikon A1R confocal laser scanning microscope (Nikon, Japan) equipped with a live-cell imaging chamber (37 °C, 5% CO2). 3D (z-stack) images were acquired under identical acquisition parameters to ensure quantitative comparability. Fluorescence distribution and intensity were analyzed using Nikon Analysis software 5.42.01 (Nikon, Tokyo, Japan).
Rheological measurements
Rheological properties of the hydrogels were characterized using a rotational rheometer (MCR302e, Anton Paar, Austria) in oscillatory mode. The instrument was equipped with a 25 mm diameter cone plate (2° cone angle, 200 μm truncation gap). The platform temperature was maintained at 10 °C. After the temperature stabilized, 300 μL of 20% gelatin (Sigma, 73865) containing various concentrations of PS-NH2 or PS-COOH nanoparticles was placed at the center of the plate and thermally equilibrated for 5 min before measurement. Amplitude and frequency sweep tests were performed to monitor the dynamic changes in the storage modulus (G′) and loss modulus (G″) of the hydrogels in real time. Each measurement was repeated three times independently.
Transmission electron microscopy
Transmission electron microscopy (TEM) imaging was performed using a JEM-1400 Flash electron microscope (JEOL, Japan) at 80 kV. Sample preparation involved mixing 32.5 µL PBS, 50 µL 100 nm PS nanoplastics, and 12.5 µL of 0.8% collagen hydrolysate solution, followed by 5 min sonication. Ten microliters of the mixture were placed onto a 300-mesh carbon-coated copper grid (Cu300-HD, Pacific Grid Tech, San Francisco, CA, USA) and dried at 60 °C for 12 h. After insertion into the microscope chamber and vacuum stabilization, the filament emission was activated, and TEM images were collected.
F-actin and Nile Red staining
LX2 cells were seeded in 12-well plates and fresh culture medium was then supplemented with either positively charged PS-NH2 (10 µg/mL) or negatively charged PS-COOH
Scratch wound assay
LX2 cells were seeded in 12-well plates and a linear scratch was then made across the cell monolayer using a sterile pipette tip, followed by washing with PBS to remove detached cells. Fresh culture medium was then supplemented with either positively charged PS-NH2 (10 µg/mL) or negatively charged PS-COOH
Statistical analysis
Statistical analyses were performed using GraphPad Prism 10.1. Data were expressed as mean ± standard error of the mean (SEM). Comparisons between two groups were performed using Student’s t-test, and multiple group comparisons were conducted using one-way analysis of variance (ANOVA). A P-value < 0.05 was considered statistically significant.
RESULTS AND DISCUSSION
Differential retention and cellular uptake of positively and negatively charged nanoplastics in a 3D microenvironment
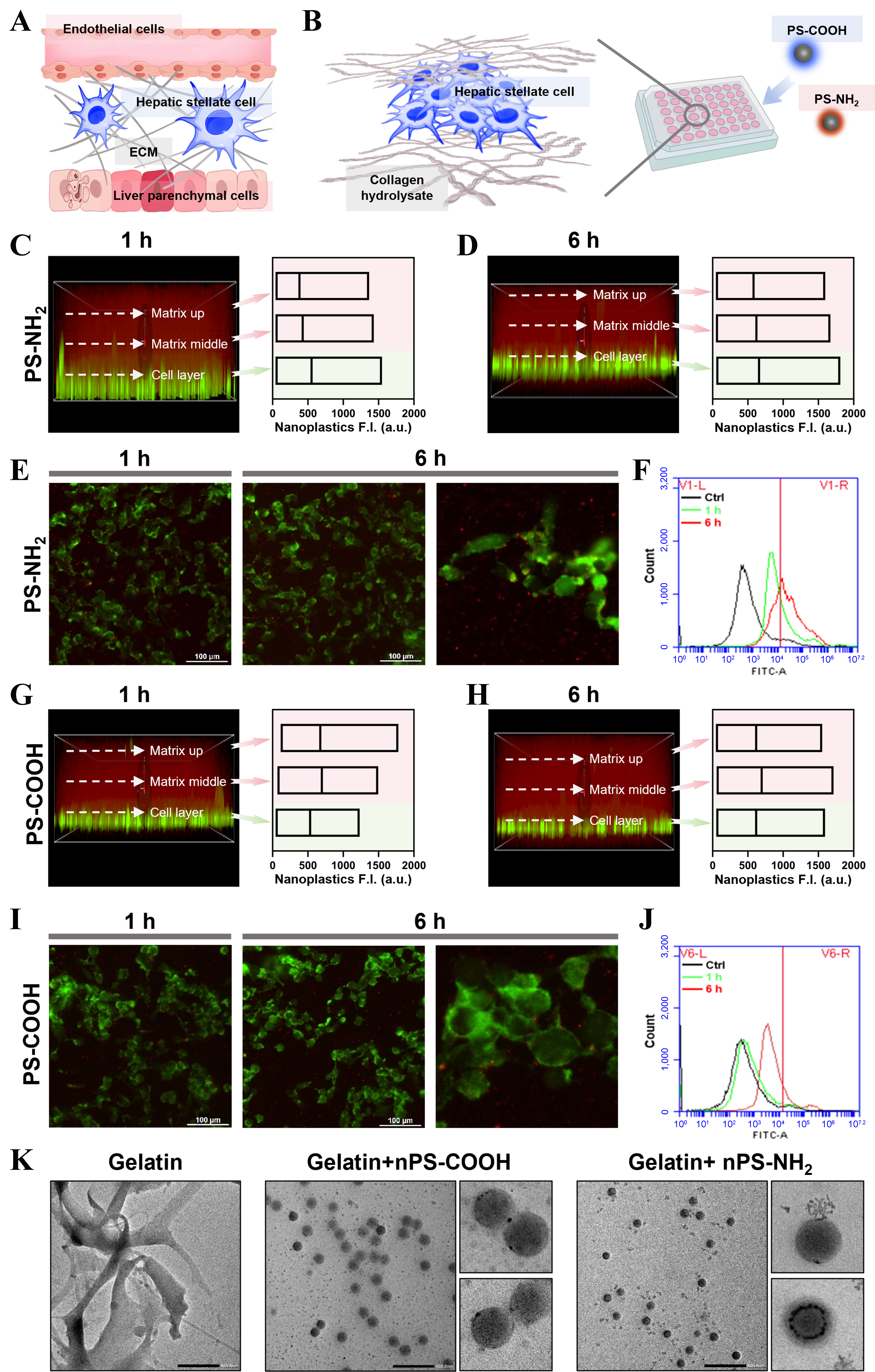
To recapitulate the spatial and mechanical microenvironment of HSCs within the hepatic space of Disse, a “collagen hydrolysate–HSC–collagen hydrolysate” sandwich model was established [Figure 1A and B]. Briefly, a layer of collagen hydrolysate gel was first cast onto the culture surface, followed by seeding LX2 on top. A second collagen hydrolysate layer was then overlaid to form a 3D construct. Using the “collagen hydrolysate–HSC–collagen hydrolysate” sandwich model, we employed confocal laser scanning microscopy (CLSM) to examine the spatiotemporal distribution of fluorescent PS-NH2 and PS-COOH nanoplastics within the 3D construct. After 1 h of incubation, PS-NH2 fluorescence was homogeneously distributed throughout the sandwich structure [Figure 1C], and the overall dispersion pattern remained similar after 6 h [Figure 1D], indicating rapid diffusion of PS-NH2 within the matrix. CLSM revealed a pronounced increase in intracellular fluorescence as early as 1 h, with maximal intensity observed at 6 h [Figure 1E], suggesting that PS-NH2 nanoparticles were efficiently internalized by HSCs within a short time frame. Flow cytometry further confirmed rapid PS-NH2 uptake within 1 h [Figure 1F].
Figure 1. Differential retention and cellular uptake of positively and negatively charged nanoplastics within the 3D hepatic model. (A) Schematic illustration of the HSC microenvironment; (B) Diagram of the 3D model of the HSC treated with nanoplastics; the labeled collagen hydrolysate is gelatin; (C) Confocal z-stack images and fluorescence intensity comparison of PS-NH2 in the 3D model after 1 h exposure; (D) Confocal z-stack images and fluorescence intensity comparison of PS-NH2 after 6 h exposure; (E) Magnified fluorescence images of the cellular layer after 1 and 6 h PS-NH2 treatment; (F) Flow cytometric analysis of PS-NH2 uptake efficiency by HSCs after 1 and 6 h exposure; (G) Confocal z-stack images and fluorescence intensity comparison of PS-COOH after 1 h exposure; (H) Confocal z-stack images and fluorescence intensity comparison of PS-COOH after 6 h exposure; (I) Magnified fluorescence images of the cellular layer after 1 and 6 h PS-COOH treatment; (J) Flow cytometric analysis of PS-COOH uptake efficiency by HSCs after 1 and 6 h exposure; (K) TEM images showing gelatin adsorption on nanoplastics with different surface charges. 3D: Three-dimensional; HSC: hepatic stellate cell; PS-NH2: amino-functionalized polystyrene; PS-COOH: carboxyl-functionalized polystyrene; TEM: transmission electron microscopy; ECM: extracellular matrix.
In contrast, after 1 h of incubation with PS-COOH, strong fluorescence signals were detected in the collagen hydrolysate matrix. The cell layer exhibited weak fluorescence [Figure 1G], indicating that negatively charged PS-COOH particles were predominantly retained within the collagen hydrolysate network at early time points. After 6 h, matrix fluorescence intensity decreased while that in the cell layer gradually increased, reflecting progressive diffusion of PS-COOH toward the cells [Figure 1H]. CLSM images showed only a few intracellular particles at 1 h, whereas intracellular particles increased markedly at 6 h [Figure 1I]. Flow cytometric analysis showed limited cellular uptake of PS-COOH at 1 h. This uptake increased modestly over time. However, it remained markedly lower than that of PS-NH2 [Figure 1J], suggesting that most PS-COOH particles were retained within the ECM [Supplementary Figure 1A and B]. TEM further showed extensive adsorption of collagen hydrolysate molecules on both PS-NH2 and PS-COOH surfaces [Figure 1K], supporting the hypothesis that nanoplastics alter ECM mechanics through direct nanoparticle–collagen hydrolysate interactions.
Collectively, these results demonstrate that within the matrix–cell–matrix 3D configuration, cells exposed to positively charged nanoplastics displayed stronger intracellular fluorescence than those treated with negatively charged particles, indicating enhanced internalization of PS-NH2. More importantly, negatively charged nanoplastics, though less readily internalized, tended to accumulate within the ECM, highlighting distinct interaction mechanisms between nanoplastics of different surface charges and HSCs.
Modulation of ECM mechanics by nanoplastics
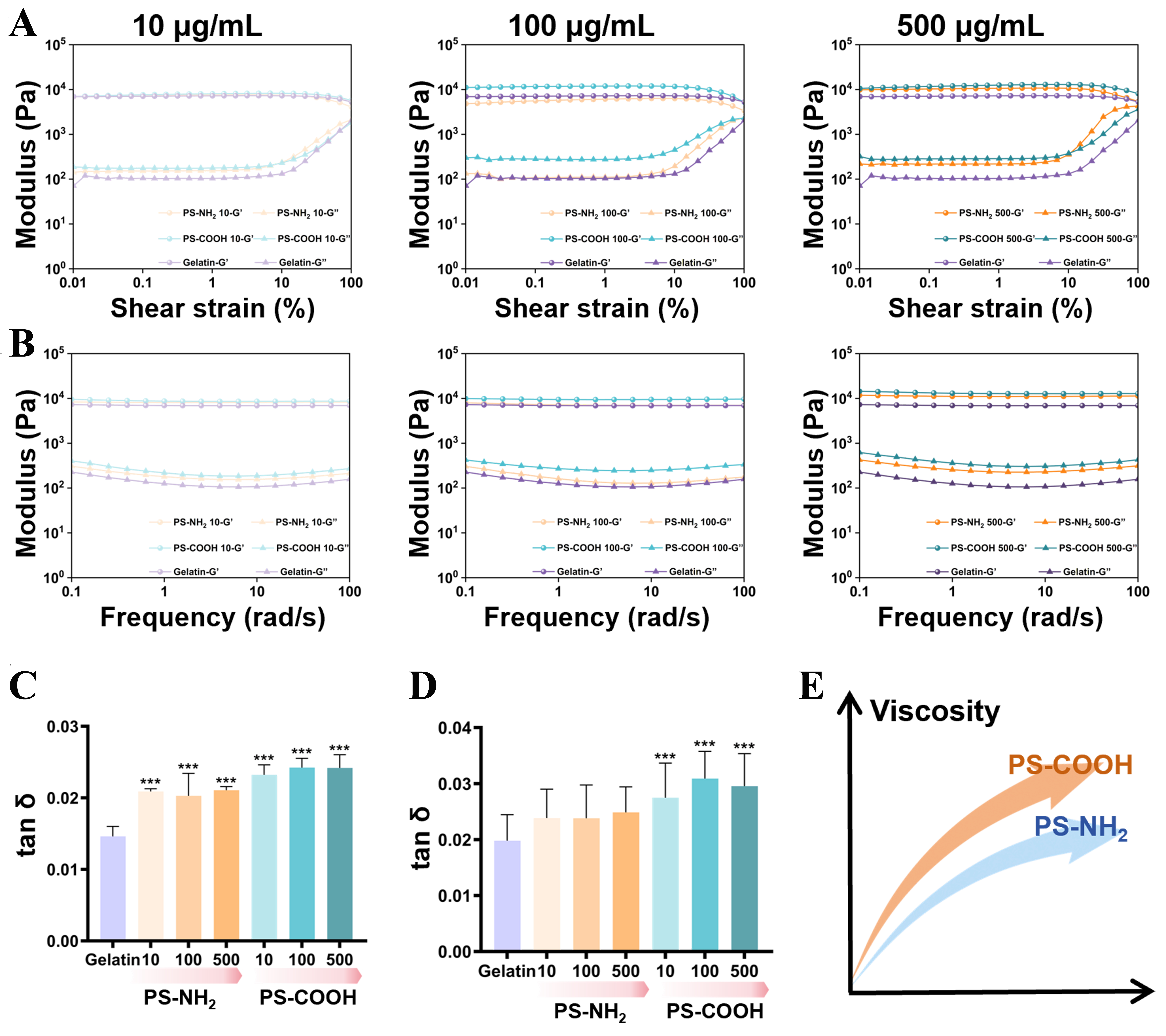
In the liver Disse space–mimicking model, numerous nanoplastic particles were retained within the collagen hydrolysate matrix, suggesting potential interactions with the ECM that could modify its mechanical properties. To assess these effects, rheological analyses were performed. Both PS-NH2 and PS-COOH increased the storage modulus (G′) of collagen hydrolysate hydrogels in a concentration-dependent manner [Supplementary Figure 2A-D], with PS-COOH exhibiting a more pronounced stiffening effect [Figure 2A and B].
Figure 2. Effects of nanoplastics on the mechanical properties of collagen hydrolysate-based matrices. (A) Changes in storage modulus (G′) of collagen hydrolysate matrices containing nanoplastics under shear deformation; (B) Frequency-dependent variation of storage modulus in collagen hydrolysate matrices with nanoplastic incorporation; (C) Comparison of loss factor (tan δ) under shear deformation in collagen hydrolysate matrices with nanoplastics; (D) Frequency-dependent variation of loss factor in nanoplastic-containing collagen hydrolysate matrices; (E) Schematic illustration of viscosity modulation by nanoplastics with different surface charges. Values represent the mean of three independent biological replicates. ***P ≤ 0.001 compared to the control group. PS-NH2: Amino-functionalized polystyrene; PS-COOH: carboxyl-functionalized polystyrene.
The loss factor (tan δ), defined as the ratio of the loss modulus to the storage modulus, represents the viscoelastic nature of ECM, where higher values indicate greater viscous behavior. Both PS-NH2 and PS-COOH significantly elevated tan δ at relatively low concentrations (10-100 μg/mL) [Figure 2C and D], indicating substantial impacts on the viscoelasticity of the ECM. These results indicate that nanoplastic incorporation increases viscosity and alters the mechanical microenvironment of the ECM [Figure 2E].
Cytotoxicity assessment of surface-charged polystyrene nanoplastics
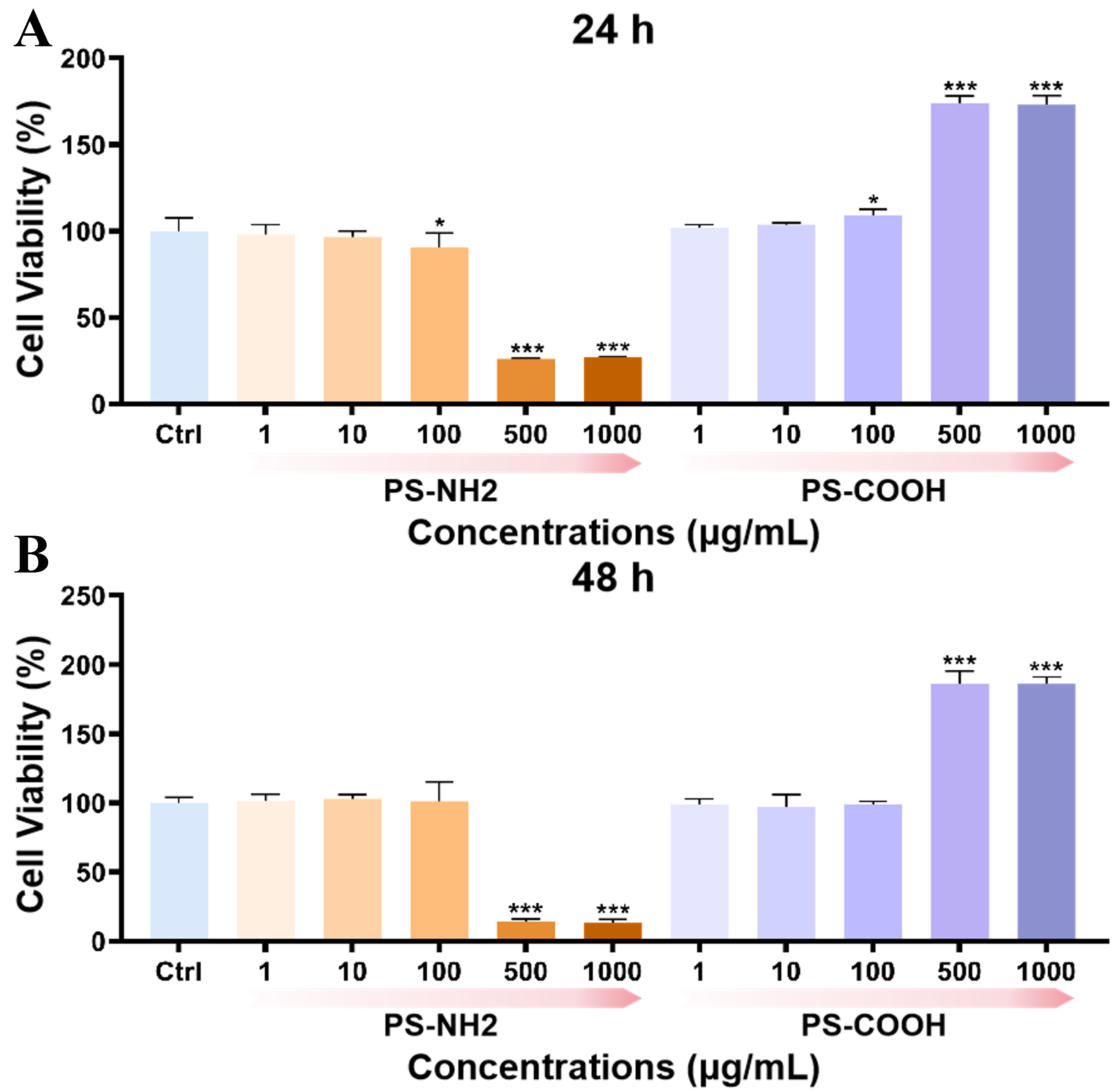
With the “collagen hydrolysate–HSC–collagen hydrolysate” sandwich model, LX2 cells were exposed to PS-NH2 or PS-COOH at concentrations ranging from 1 to 1,000 μg/mL covering environmental and human exposure concentrations for 24 and 48 h [Figure 3A and B]. Low concentrations (1-100 μg/mL) of PS-NH2 exhibited negligible cytotoxicity; however, cell viability decreased in a concentration-dependent manner at higher doses, reaching only about 10% survival at 500 μg/mL. In contrast, low concentrations (1-100 μg/mL) of PS-COOH caused no apparent cytotoxicity but promoted significant cell proliferation at higher concentrations (500-1,000 μg/mL). Overall, within environmentally and physiologically relevant exposure levels (1-100 μg/mL) over 48 h, both types of nanoplastics showed minimal effects on LX2 viability. At higher concentrations, however, positively and negatively charged nanoparticles exhibited opposite cytotoxicity trends.
Figure 3. Cytotoxicity assessment of nanoplastics. (A) Toxic effects on HSCs after 24 h continuous exposure to nanoplastic particles at five graded concentrations; (B) Toxic effects on HSCs after 48 h of continuous exposure under the same concentration gradient. Values represent the mean of three independent biological replicates. *P ≤ 0.05, ***P ≤ 0.001 compared to the control group. HSCs: Hepatic stellate cells; PS-NH2: amino-functionalized polystyrene; PS-COOH: carboxyl-functionalized polystyrene.
Our study is consistent with previous research in demonstrating that, at environmentally and physiologically relevant exposure levels (1-100 μg/mL), neither negatively nor positively charged polystyrene nanoplastics exhibited significant cytotoxicity toward the in vitro human colorectal adenocarcinoma cell line (Caco-2)/mucus-secreting human colorectal adenocarcinoma cell line (HT29-MTX-E12) co-culture model simulating the intestinal barrier[19], the human choriocarcinoma cell line BeWo (clone b30) cell model representing the placental barrier[20], or HepG2 cells[21]. However, the higher concentrations (> 100 μg/mL) of nanoplastics induced marked cytotoxicity in HepG2 cells[22]. Interestingly, this was inconsistent with our observation that PS-COOH promoted the proliferation of HSCs. Therefore, we speculate that PS-COOH may influence HSCs through mechanisms distinct from direct cytotoxicity, prompting further investigation into its potential microenvironment-mediated mechanisms.
Nanoplastics induce activation of quiescent HSCs via ECM mechanical modulation
Given that HSCs can transition from a quiescent to an activated fibrogenic state in response to the changes in their mechanical microenvironment, we hypothesized that nanoplastics may modulate HSC behavior by altering ECM mechanics. Based on the collagen hydrolysate sandwich model, we investigated the effects of nanoplastics on the quiescent state of HSCs.
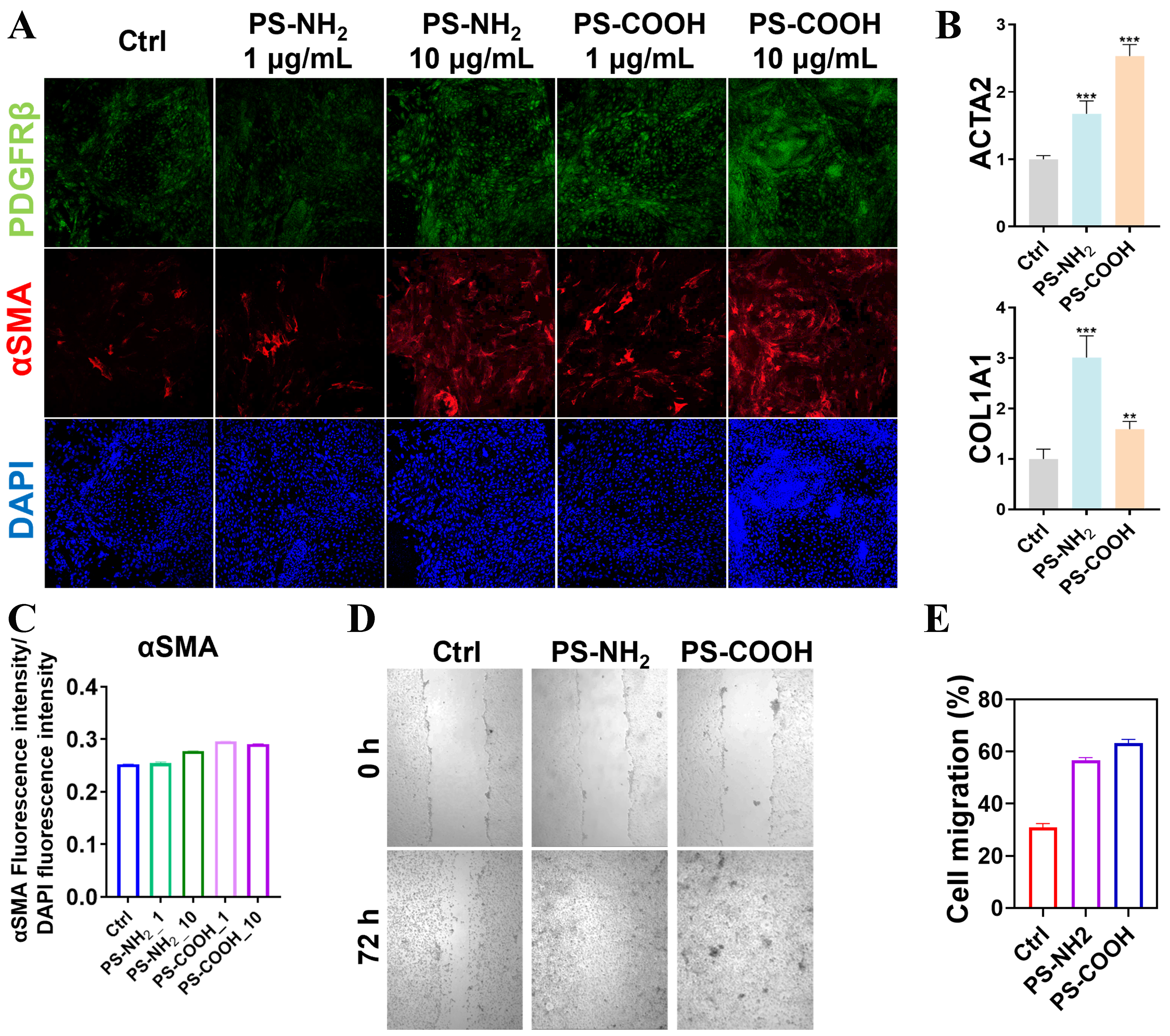
PS-NH2 or PS-COOH at 1 µg/mL caused no obvious change in the α-SMA protein level, a marker of HSC activation [Figure 4A]. However, at 10 µg/mL, both PS-NH2 and PS-COOH significantly increased α-SMA expression at both the protein and messenger RNA (mRNA) levels [Figure 4B and C], with PS-COOH producing a stronger activation effect. This trend corresponds with the observed changes in ECM mechanical properties. Furthermore, cell migration assays showed markedly enhanced migratory activity in the 10 µg/mL nanoplastic-treated groups compared with the control, with cells nearly covering the entire migration area [Figure 4D]. In addition, PS-COOH elicited a stronger pro-migratory effect than the other groups [Figure 4E]. These findings indicate that, although this concentration did not cause significant cytotoxicity, both types of nanoplastics effectively induced activation of quiescent HSCs.
Figure 4. Regulation of HSC behavior by nanoplastics. (A) Immunofluorescence staining of HSC protein markers following treatment with different nanoplastics (PDGFRβ, α-SMA, and DAPI); (B) Quantitative PCR analysis of mechanotransduction-related genes in HSCs after nanoplastic exposure; (C) Comparison of α-SMA fluorescence staining intensity under different nanoplastic treatments; (D) Images of
Previous studies, such as those by Bera et al.[23], published in Nature have proved that increased ECM viscoelasticity can enhance cell migration and tumor invasiveness. Consistent with our findings, nanoplastics promote hepatic fibrosis in vivo[24], and polystyrene nanoplastics exacerbate hepatic fibrosis in a high-fat diet model[25], further supporting the fibrotic potential of nanoplastics. Our observations extend them to an environmentally relevant toxicology context. Specifically, negatively charged PS-COOH nanoplastics increased the loss factor (tan δ) of collagen hydrolysate hydrogels and enhanced matrix viscosity. Within PS-COOH–treated matrices, HSCs exhibited higher proliferation rates [Figure 2] and enhanced migration [Figure 4D]. Collectively, these results indicate that HSCs are more likely to become activated in an environment with higher mechanical viscosity, displaying mesenchymal-like spreading and motility.
Nanoplastics disrupt mechanotransduction in HSCs
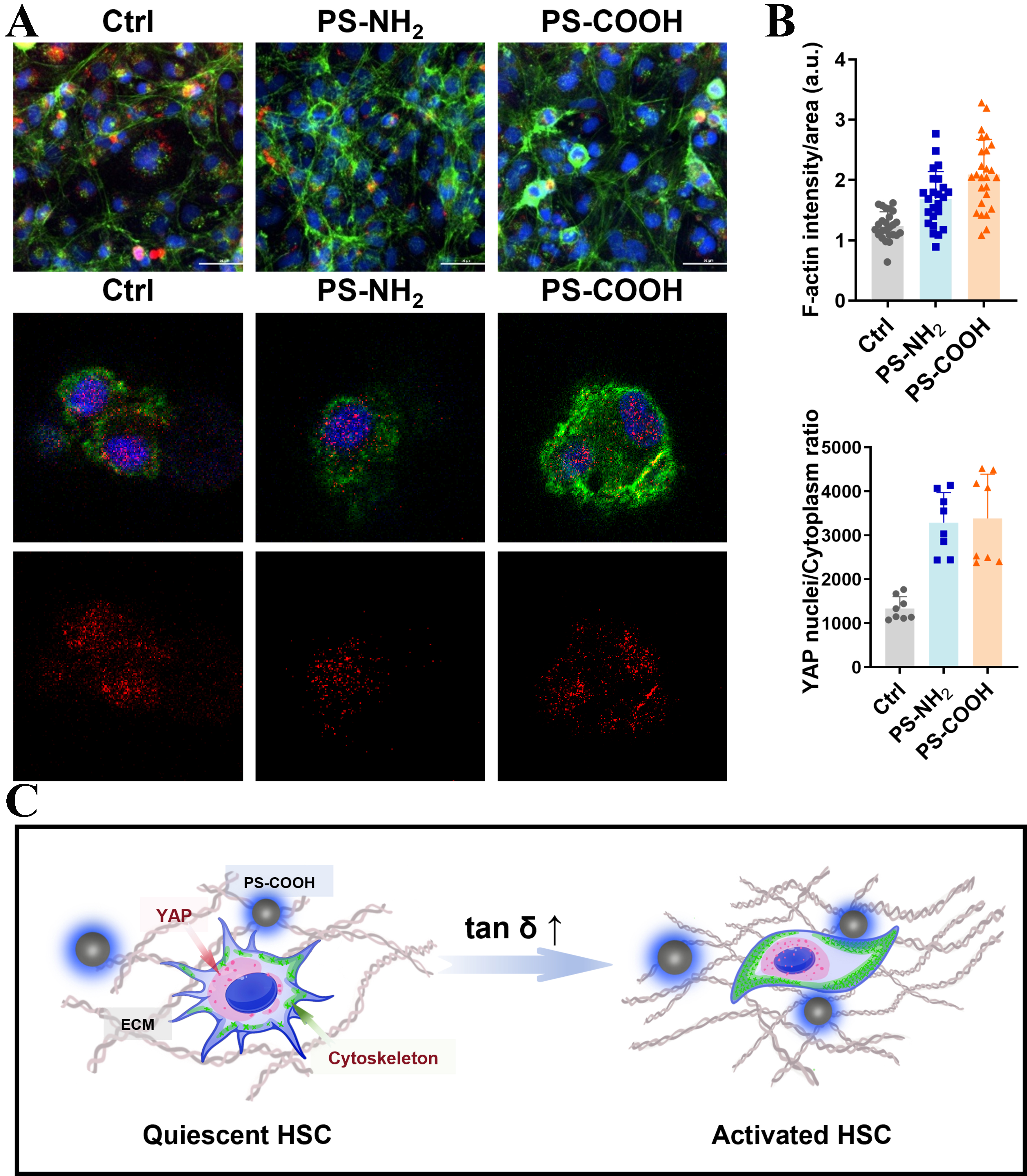
To further elucidate the molecular mechanisms linking the effects of PS-NH2 and PS-COOH on HSC activation and the alterations of the extracellular mechanical microenvironment, we examined changes in the cytoskeletal effector F-actin and the intracellular distribution of the mechanosensitive transcriptional regulator YAP. Immunofluorescence analysis revealed that exposure to 10 μg/mL PS-NH2 or PS-COOH markedly enhanced F-actin stress fiber formation and promoted nuclear localization of YAP [Figure 5A and B], indicating that both types of charged nanoplastics perturb HSC mechanotransduction signaling. Consistent with reports that nanoplastics can disrupt cytoskeletal tension and integrin-mediated adhesion, these changes likely weaken HSC sensing of matrix stiffness and reshape YAP/transcriptional co-activator with PDZ-binding motif (TAZ)-driven transcriptional outputs[17]. Moreover, microplastics have been implicated in promoting hepatic fibrogenesis via inflammation-related pathways such as cyclic GMP-AMP synthase (cGAS)–stimulator of interferon genes (STING) signaling pathway[26], raising the possibility that mechanical dysregulation and inflammatory signaling may act in concert to amplify pro-fibrotic responses.
Figure 5. Nanoplastic-mediated modulation of mechanotransduction and YAP signaling in HSCs. (A) Immunofluorescence staining of F-actin and YAP in HSCs treated with 10 μg/mL of positively or negatively charged nanoplastics; (B) Quantitative analysis of F-actin fluorescence intensity and YAP nuclear-to-cytoplasmic ratio; (C) Schematic illustration depicting the HSC cytoskeletal reorganization and mechanotransduction modulation induced by nanoplastics. YAP: Yes-associated protein; HSCs: hepatic stellate cells; PS-NH2: amino-functionalized polystyrene; PS-COOH: carboxyl-functionalized polystyrene; ECM: extracellular matrix.
Together with the rheological data and biochemical markers of HSC activation, these results suggest that PS-COOH adheres to collagen hydrolysate fibers and modifies the viscoelastic properties [Figure 2 and Figure 5C]. Through enhanced cell–matrix interactions, these mechanical alterations promote F-actin assembly, YAP nuclear translocation, and subsequent HSC activation, as evidenced by increased α-SMA expression [Figure 4C]. Our findings suggest that nanoplastics can act as regulators of ECM mechanical properties, thereby influencing HSC mechanotransduction, proliferation, and migration.
Limitations
Our study reveals the interference of nanoplastic charge properties with the mechanical microenvironment of the ECM, but there are some limitations. First, by disturbing the mechanical microenvironment of the liver ECM, the study primarily focuses on how microplastics activate HSCs. However, the regulatory role of nanoplastics in the development and progression of liver diseases still requires in vivo experiments for further verification from histological and pathological perspectives. Second, the mechanical toxicity effects of nanoplastics on other liver cell types, such as hepatocytes and endothelial cells, and their intercellular interactions remain unclear. Therefore, future research will combine animal models with in vitro biomimetic hepatic and pathological models to further explore the mechanisms of mechanical toxicity induced by nanoplastic charge effects.
CONCLUSION
Nanoplastics may influence HSC activation indirectly by modulating ECM mechanical properties rather than solely through direct cellular uptake. This work bridges environmental toxicology and cellular mechanobiology, suggesting that assessments of nanoplastic health risks should also consider their potential to alter the tissue mechanical microenvironment.
DECLARATIONS
Authors’ contributions
Conceptualization, project administration, visualization, original draft writing: Cao, Z.; Xiao, M.
Data curation: Yang, M.; Liu, Q.; Jing, H.
Methodology: Wang, Z.; Ding, X.; Wang, P.
Funding acquisition, supervision, manuscript review: Liang, S.
Availability of data and materials
Further data are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Tianjin Natural Science Foundation (Grant No. 23JCQNJC00790), the Interdisciplinary Research Program of Tianjin University of Technology - Toxicity Mechanisms and Prevention Strategies of Microparticles (Grant No. 01002503), and the project Evaluation of Microplastic Toxicity Using a Bio-3D-Printed Biomimetic Intrahepatic Bile Duct Chip (Grant No. 202510060095).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Nihart, A. J.; Garcia, M. A.; El Hayek, E.; et al. Bioaccumulation of microplastics in decedent human brains. Nat. Med. 2025, 31, 1114-9.
2. Leslie, H. A.; van Velzen, M. J. M.; Brandsma, S. H.; Vethaak, A. D.; Garcia-Vallejo, J. J.; Lamoree, M. H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199.
3. Zhang, Z.; Chen, W.; Chan, H.; et al. Polystyrene microplastics induce size-dependent multi-organ damage in mice: insights into gut microbiota and fecal metabolites. J. Hazard. Mater. 2024, 461, 132503.
4. Horvatits, T.; Tamminga, M.; Liu, B.; et al. Microplastics detected in cirrhotic liver tissue. EBioMedicine 2022, 82, 104147.
5. Cheng, W.; Li, X.; Zhou, Y.; et al. Polystyrene microplastics induce hepatotoxicity and disrupt lipid metabolism in the liver organoids. Sci. Total. Environ. 2022, 806, 150328.
6. Shi, C.; Han, X.; Guo, W.; et al. Disturbed gut-liver axis indicating oral exposure to polystyrene microplastic potentially increases the risk of insulin resistance. Environ. Int. 2022, 164, 107273.
7. Xu, D.; Ma, Y.; Han, X.; Chen, Y. Systematic toxicity evaluation of polystyrene nanoplastics on mice and molecular mechanism investigation about their internalization into Caco-2 cells. J. Hazard. Mater. 2021, 417, 126092.
8. Hou, Y.; Bian, D.; Xiao, Y.; et al. MRI-based microplastic tracking in vivo and targeted toxicity analysis. Sci. Total. Environ. 2024, 954, 176743.
9. Banerjee, A.; Shelver, W. L. Micro- and nanoplastic induced cellular toxicity in mammals: a review. Sci. Total. Environ. 2021, 755, 142518.
10. Sadeghinia, H.; Hanachi, P.; Ramezani, R.; Karbalaei, S. Toxic effects of polystyrene nanoplastics on MDA-MB-231 breast cancer and HFF-2 normal fibroblast cells: viability, cell death, cell cycle and antioxidant enzyme activity. Environ. Sci. Eur. 2025, 37, 1.
11. He, Y.; Li, J.; Chen, J.; et al. Cytotoxic effects of polystyrene nanoplastics with different surface functionalization on human HepG2 cells. Sci. Total. Environ. 2020, 723, 138180.
12. Shen, Q.; Liu, Y. J.; Qiu, T. T.; Loon K, S.; Zhou, D. Microplastic-induced NAFLD: hepatoprotective effects of nanosized selenium. Ecotoxicol. Environ. Saf. 2024, 272, 115850.
13. Teng, M.; Zhao, X.; Wu, F.; et al. Charge-specific adverse effects of polystyrene nanoplastics on zebrafish (Danio rerio) development and behavior. Environ. Int. 2022, 163, 107154.
14. Koelmans, A. A.; Redondo-Hasselerharm, P. E.; Nor, N. H. M.; De Ruijter, V. N.; Mintenig, S. M.; Kooi, M. Risk assessment of microplastic particles. Nat. Rev. Mater. 2022, 7, 138-52.
15. Fleury, J. B.; Baulin, V. A. Microplastics destabilize lipid membranes by mechanical stretching. Proc. Natl. Acad. Sci. U. S. A. 2021, 118, e2104610118.
16. Wei, S.; Wu, F.; Liu, J.; et al. Direct quantification of nanoplastics neurotoxicity by single-vesicle electrochemistry. Angew. Chem. Int. Ed. Engl. 2023, 62, e202315681.
17. Mitten, E. K.; Baffy, G. Mechanotransduction in the pathogenesis of non-alcoholic fatty liver disease. J. Hepatol. 2022, 77, 1642-56.
18. Long, Y.; Niu, Y.; Liang, K.; Du, Y. Mechanical communication in fibrosis progression. Trends. Cell. Biol. 2022, 32, 70-90.
19. Domenech, J.; Hernández, A.; Rubio, L.; Marcos, R.; Cortés, C. Interactions of polystyrene nanoplastics with in vitro models of the human intestinal barrier. Arch. Toxicol. 2020, 94, 2997-3012.
20. Hesler, M.; Aengenheister, L.; Ellinger, B.; et al. Multi-endpoint toxicological assessment of polystyrene nano- and microparticles in different biological models in vitro. Toxicol. In. Vitro. 2019, 61, 104610.
21. Ma, L.; Wu, Z.; Lu, Z.; et al. Differences in toxicity induced by the various polymer types of nanoplastics on HepG2 cells. Sci. Total. Environ. 2024, 918, 170664.
22. Wang, N.; Zhu, X.; Xu, Z.; et al. Photoaged polystyrene nanoplastics induce perturbation of glucose metabolism in HepG2 cells via oxidative stress. Environ. Pollut. 2025, 379, 126534.
23. Bera, K.; Kiepas, A.; Godet, I.; et al. Extracellular fluid viscosity enhances cell migration and cancer dissemination. Nature 2022, 611, 365-73.
24. Han, J.; Jeong, H.; Lee, C.; Sung, A.; Choi, Y. H.; Jung, Y. Chronic nanoplastic exposure promotes the development and progression of metabolic dysfunction-associated steatotic liver disease. Liver. Int. 2025, 45, e70224.
25. Li, L.; Xu, M.; He, C.; Wang, H.; Hu, Q. Polystyrene nanoplastics potentiate the development of hepatic fibrosis in high fat diet fed mice. Environ. Toxicol. 2022, 37, 362-72.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].