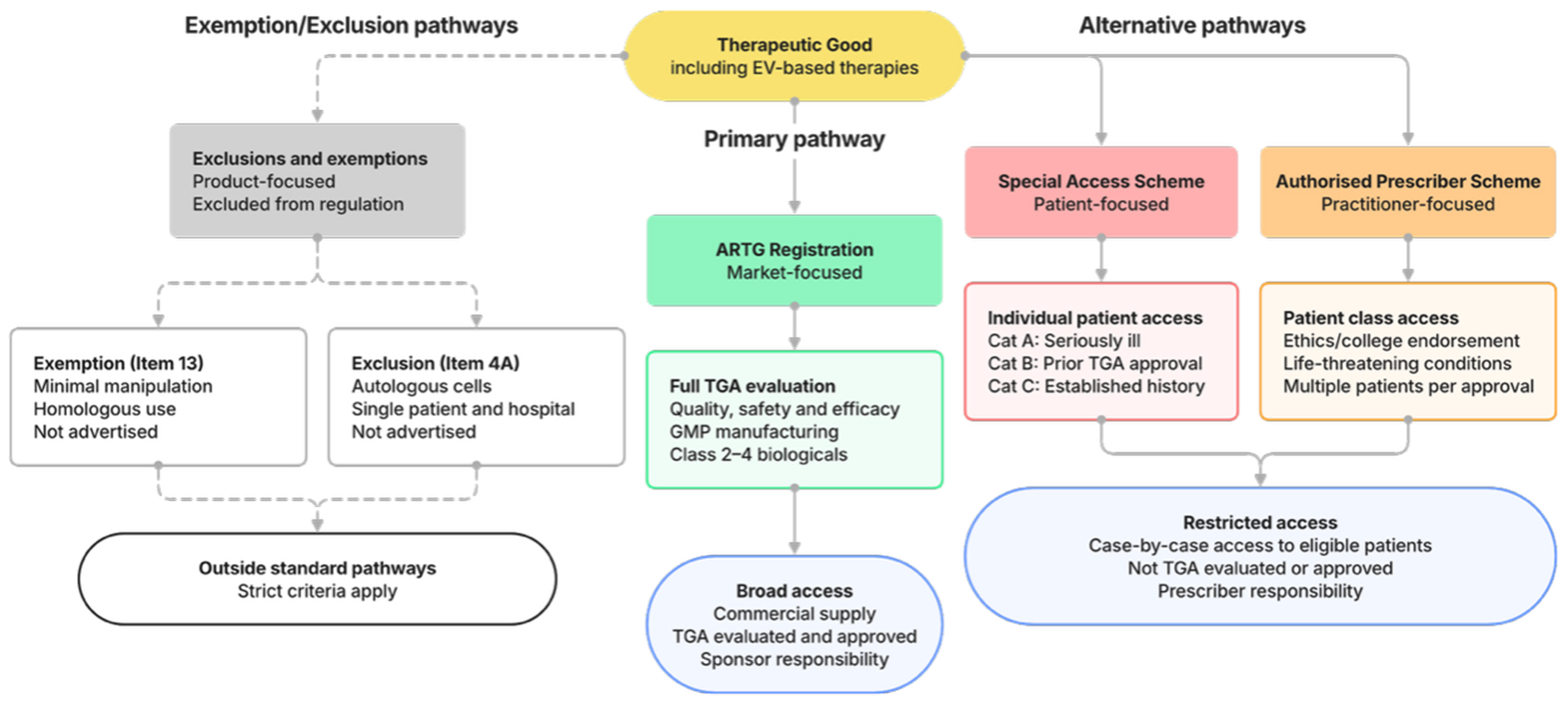
fig1
Figure 1. Regulatory pathways for therapeutic goods in Australia, including EV-based therapies. Products may access patients through ARTG registration (approved pathway) or the Special Access Scheme and Authorized Prescriber pathways (unapproved). Certain biologicals may also fall outside standard TGA oversight through exclusions (Item 4A) or exemptions (Item 13), subject to strict criteria. ARTG: Australian Register of Therapeutic Goods; EV: extracellular vesicle; TGA: Therapeutic Goods Administration.