Fluorine-doped Bi2O3 derived from metal-organic framework for electrocatalytic CO2 reduction to formate at industrial current densities
Abstract
Achieving high activity and stability at high current densities is critical in the practical application of electrocatalytic CO2 reduction (ECO2R). In this study, a fluorine-doped Bi2O3 electrocatalyst with oxygen vacancies (denoted as F-Bi2O3-Ov) has been synthesized using a Bi-based metal-organic framework as the sacrificial template. Catalyst characterizations reveal that the fluorine doping induces results in lattice expansion and generates abundant oxygen vacancies. The F-Bi2O3-Ov catalyst delivers a formate Faradaic efficiency of 94% at a current density of 300 mA cm-2 and with stable performance for over 25 h in a flow cell. Electrochemical kinetics analysis and in situ attenuated total reflectance Fourier transform infrared spectroscopy establish a fluorine doping and oxygen vacancies synergy that facilitates interfacial charge transfer, lowering the energy barrier for the formation of the key *OCHO intermediate. The findings of this study offer an effective strategy for modulating the electronic and geometric characteristics of metal oxide catalysts for high-performance ECO2R.
Keywords
INTRODUCTION
Electrocatalytic CO2 reduction (ECO2R) powered by renewable electricity represents a sustainable process that mitigates greenhouse gas emissions in the production of value-added chemicals[1-4]. Formate, as a target reduction product, is recognized as a highly promising liquid hydrogen carrier and feedstock for direct formic acid fuel cells, offering high volumetric capacity and economic viability[5-7]. Consequently, the development of efficient electrocatalysts for the selective conversion of CO2 to formate at industrially relevant current densities is a critical research goal. Bismuth-based materials have emerged as promising candidates for formate production due to their non-toxicity, low cost and unique electronic structure that stabilizes the key intermediate (*OCHO)[8-10]. In particular, bismuth(III) oxide (Bi2O3) has demonstrated high intrinsic activity for formate generation[11,12]. However, bulk Bi2O3 catalysts are known to have a low electrical conductivity, limited active site exposure and poor structural stability under reductive potentials. These limitations result in low current densities and rapid decay in Faradaic efficiency (FE) during long-term operation, particularly at high reaction rates and severe competition from the hydrogen evolution reaction (HER)[13,14]. In order to overcome these obstacles, a simultaneous modulation of the electronic structure and morphological engineering is required to enhance both catalyst activity and durability[15,16].
Defect engineering and heteroatom doping are effective strategies to optimize the intrinsic catalytic properties of metal oxides[17-21]. Introducing oxygen vacancies (Ov) can generate unsaturated coordination sites that facilitate CO2 adsorption and charge transfer[22,23]. Furthermore, doping with high-electronegativity anions, such as fluorine, can induce strong charge polarization and lattice distortion[24]. In theory, the incorporation of fluorine modulates the p-orbital electron density of Bi centers and optimizes the intermediate binding energy while also promoting the formation of oxygen vacancies, creating a synergistic environment for CO2 activation[25,26]. Moreover, the fabrication of catalysts from metal-organic frameworks (MOFs) offers distinct structural advantages[27-29]. The inherent porosity and periodic metal centers that characterize MOFs enable the synthesis of derivative oxides, which maximize the exposure of active sites and facilitate mass transport at high current densities[30-32].
In this study, we report the synthesis of fluorine-doped Bi2O3 catalysts with abundant oxygen vacancies (denoted as F-Bi2O3-Ov) via the fluoride-assisted pyrolysis of a Bi-based MOF (denoted as Bi-MOF). The optimized fluorine-doped Bi2O3 catalyst for electrocatalytic CO2 reduction provides a remarkable formate FE of 94% at a current density of 300 mA cm-2 in a flow cell operation. Fluorine doping causes Bi2O3 lattice expansion, generating more oxygen vacancies and enhancing the stability of the Bi3+ active sites. A combined theoretical simulation and experimental testing has demonstrated a synergism associated with oxygen vacancies and fluorine doping that promotes the hydrogenation of the *OCHO intermediate with a consequent efficient conversion of CO2 to formate at industrial-grade current densities.
EXPERIMENTAL
Materials
Bismuth nitrate pentahydrate (Bi(NO3)3·5H2O, 99.0%) was purchased from Tianjin Fengchuan Chemical Reagent Technology Co., Ltd. 1,3,5-benzenetricarboxylic acid (H3BTC, 98%) was purchased from Shandong Kexuan Biochemical Co., Ltd. Methanol (CH3OH, 99.5%) was purchased from Tianjin Xintebote Chemical Co., Ltd. Sodium fluoride (NaF, 99.99%) was purchased from Shanghai McLyn Biochemical Technology Co., Ltd. Deuterium oxide (D2O, 99.9%) and dimethyl sulfoxide (DMSO, 99.9%) were purchased from Beijing Innochem Science & Technology Co., Ltd. Sodium fluoride (NaF, 99.99%) was purchased from Shanghai McLyn Biochemical Technology Co., Ltd. Ultrapure water was used in all the experiments with a resistivity of 18.2 MΩ·cm-1. The gas diffusion layer (GDL, Sigracet 38 BC) was purchased from Wuhan Gaoshi Ruilian Technology Co., Ltd. The anion exchange membrane (FAB-PK-130) was purchased from FuMA Technology Co., Ltd. A Nafion solution (5 wt%) was purchased from Suzhou Shengernuo Technology Co., Ltd. High-purity argon (Ar, 99.99%), high-purity nitrogen (N2, 99.999%) and high-purity carbon dioxide (CO2, 99.999%) were used in all experiments. All the chemicals were used as supplied without further purification.
Catalyst preparation
Synthesis of Bi-MOF
Bi(NO3)3·5H2O (150 mg) and H3BTC (750 mg) were dissolved in methanol (60 mL) and stirred for 30 min. The resulting solution was transferred into a 100 mL Teflon-lined autoclave and heated at 120 °C for 24 h. After the reaction, the product was collected by centrifugation and washed with methanol.
Synthesis of F-Bi2O3-Ov
A mixture of Bi-MOF (20 mg) and NaF (2 mg) was placed in a tube furnace, and heated to 450 °C for 6 h at
Synthesis of 0.5F-Bi2O3-Ov
The sample was synthesized using a similar procedure to that employed for F-Bi2O3-Ov, but utilizing 1 mg NaF.
Synthesis of 2F-Bi2O3-Ov
The sample was synthesized using a similar procedure to that employed for F-Bi2O3-Ov, but utilizing 4 mg NaF.
Synthesis of Bi2O3-Ov
The sample was synthesized using a similar procedure to that employed for F-Bi2O3-Ov, without the inclusion of NaF.
Synthesis of Bi2O3
Bi (NO3)3·5H2O (200 mg) was placed in a muffle furnace, and heated to 450 °C for 6 h at 5 °C·min-1 in air. The resulting product was cooled to room temperature to obtain Bi2O3.
Synthesis of F-Bi2O3
The sample was synthesized using a similar procedure to that employed for F-Bi2O3-Ov, substituting Bi-MOF with Bi2O3.
Characterization
X-ray diffraction (XRD) analyses were performed on a Bruker D8 ADVANCE X-ray diffractmeter with Cu Kα radiation at 40 kV and 40 mA. Scanning electron microscopy (SEM) analyses were conducted using a Hitachi S4800 microscope. Transmission electron microscope (TEM), high-resolution TEM (HRTEM) and energy-dispersive spectroscopy were performed on a FEI TECNAI G2 F20 microscope. X-ray photoelectron spectroscopy (XPS) analyses were carried out on a Thermo Scientific ESCALAB 250Xi unit equipped with an Al-Kα X-ray excitation source. Nitrogen adsorption-desorption isotherm measurements were carried out on a Micromeritics ASAP 2460 unit. Thermogravimetric analysis (TGA) was performed on a TA Q5000IR TGA thermal analyzer. The chemical composition was determined by inductively coupled plasma optical emission spectrometry (ICP-OES), using an Aglient 5110 unit. Electron paramagnetic resonance (EPR) spectra were performed on a Bruker EMXplus 10/12 unit equipped with an Oxford ESR910 Liquid Helium cryostat. Raman spectra were recorded on a Horiba LabRAM HR Evolution Raman microscope with a laser excitation source of 532 nm. In situ attenuated total reflectance Fourier transform infrared spectroscopy (ATR-FTIR) analyses were performed on a Nicolet iS 50 spectrometer (Thermo Fisher) equipped with a liquid nitrogen-cooled MCT detector. The liquid products were analyzed using a Bruker AVANCE NEO 600 MHZ 1H NMR spectrometer. The gaseous products were analyzed by gas chromatography (GC) using a GC-2014 chromatograph (Shimadzu Corporation, Japan). Electrochemical measurements were conducted using an electrochemical workstation (CHI 1140D, Shanghai Chenhua Instrument Co., Ltd., Shanghai, China).
RESULTS AND DISCUSSION
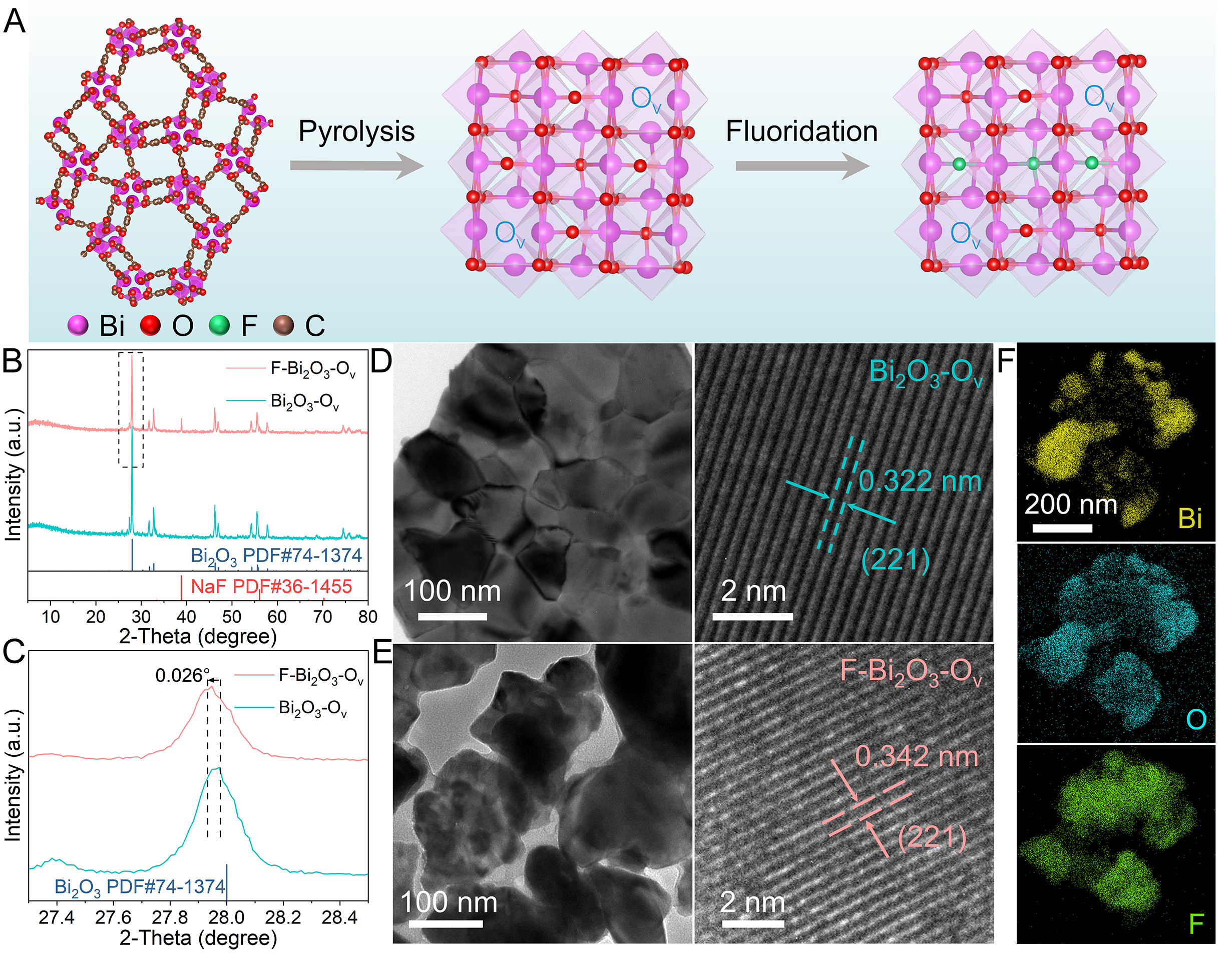
The preparation of the F-Bi2O3-Ov catalyst is illustrated in Figure 1A. The Bi-MOF precursor was synthesized under solvothermal conditions, as confirmed by the XRD, SEM and TEM analyses
Figure 1. (A) Schematic diagram of the catalyst synthesis. (B) XRD patterns of Bi2O3-Ov and F-Bi2O3-Ov and (C) the enlarged patterns. TEM and HRTEM images of (D) Bi2O3-Ov and (E) F-Bi2O3-Ov. (F) Elemental maps for F-Bi2O3-Ov.
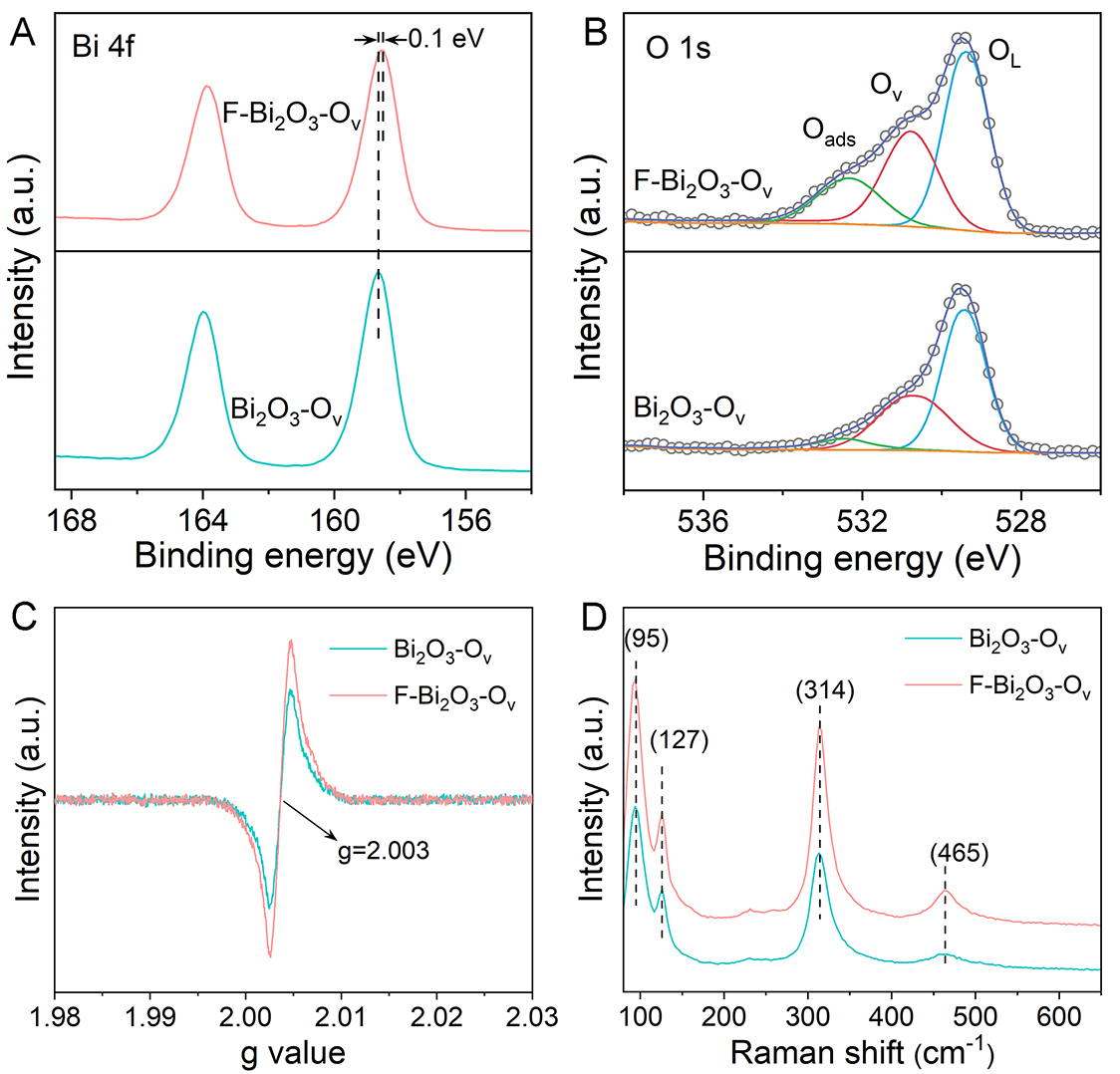
The XPS measurements were conducted to determine the elemental chemical states of elements in the as-synthesized catalysts. In the high-resolution Bi 4f spectra [Figure 2A and Supplementary Figure 15], the two peaks at 158.6 and 163.8 eV are assigned to Bi 4f7/2 and Bi 4f5/2 spin-orbit, respectively[34]. Following fluorine doping, the Bi peak is shifted by 0.1 eV to a lower binding energy, primarily due to the introduction of more oxygen vacancies by fluorine doping. This facilitates an efficient electron transfer to the metal cations. The F 1s spectra exhibit a dominant peak at 685.7 eV, attributed to the Bi-F bond. The peak at
Figure 2. XPS spectra of (A) Bi 4f and (B) O 1s for Bi2O3-Ov and F-Bi2O3-Ov. (C) EPR and (D) Raman spectra for Bi2O3-Ov and F-Bi2O3-Ov.
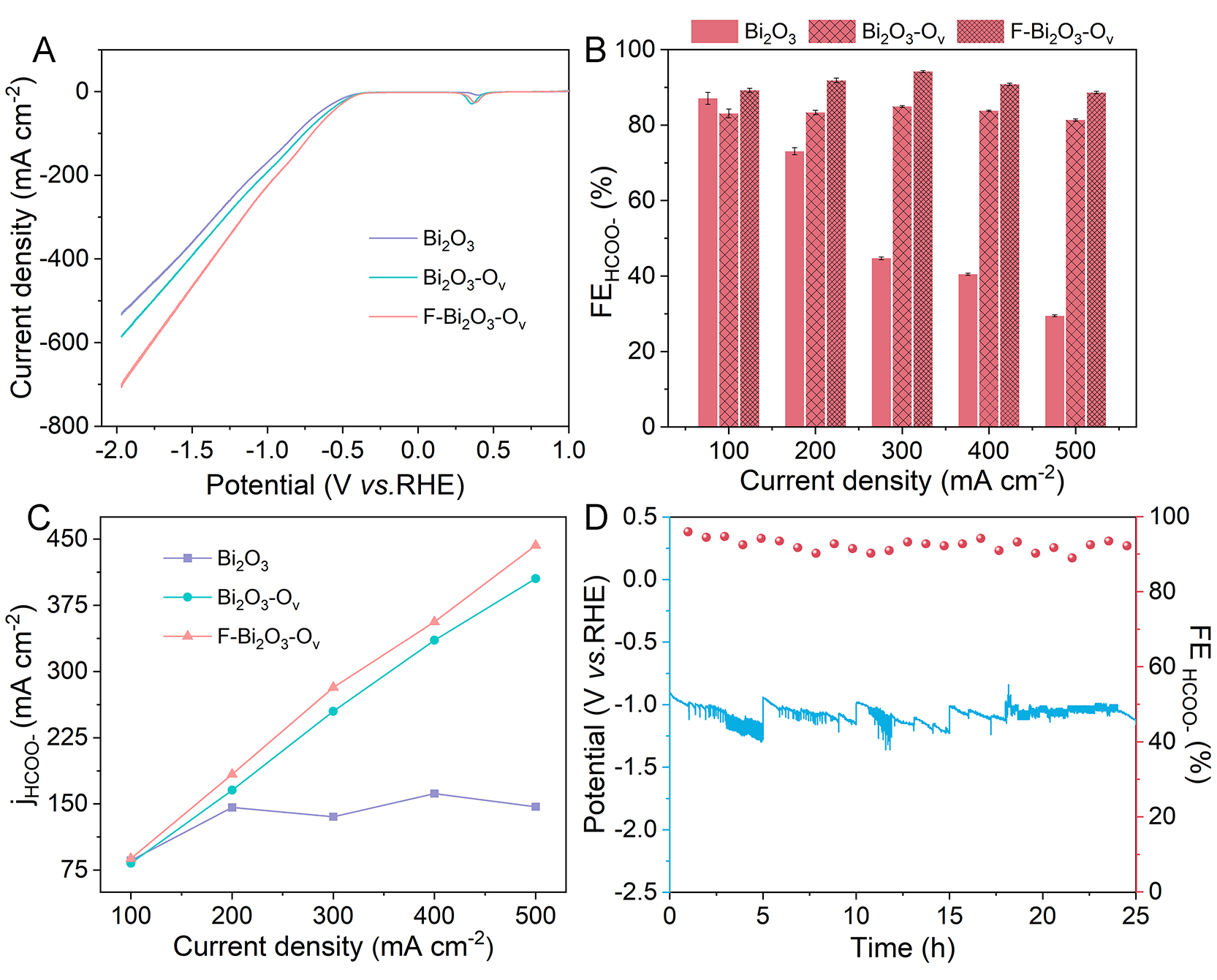
The ECO2R performance of the Bi2O3, Bi2O3-Ov and F-Bi2O3-Ov catalysts was evaluated in a flow cell. Linear sweep voltammetry (LSV) results reveal that all the catalysts have significantly higher current densities in a CO2-saturated electrolyte than observed in an Ar-saturated electrolyte at identical potentials
Figure 3. (A) LSV curves for Bi2O3, Bi2O3-Ov and F-Bi2O3-Ov in a CO2-saturated 1 M KOH electrolyte at a scan rate of 5 mV s-1. (B) Current-dependent formate FE for Bi2O3, Bi2O3-Ov and F-Bi2O3-Ov; the error bars represent the standard deviation of the mean of three independent replicates. (C) Partial current densities for Bi2O3, Bi2O3-Ov and F-Bi2O3-Ov under different current conditions. (D) Stability of F-Bi2O3-Ov at a current density of 300 mA cm-2.
The overpotentials at various current densities were systematically analyzed [Supplementary Figure 29] to evaluate the influence of the local environment in modulating the reaction energy barrier. At a current density of 100 mA cm-2, the F-Bi2O3-Ov catalyst requires a potential of -0.48 V (vs. RHE) to drive the CO2 reduction reaction toward formate production. In the high current density region
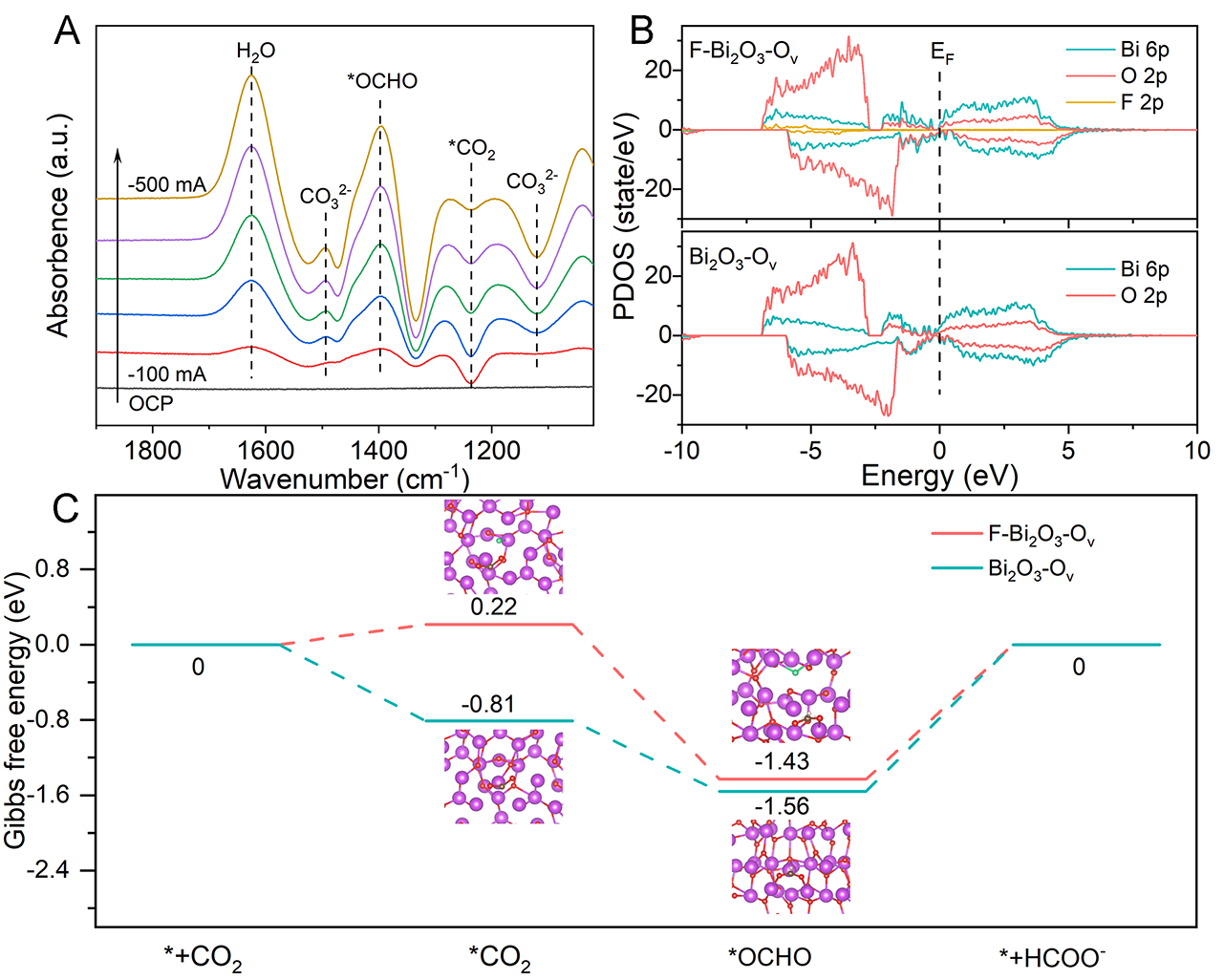
In situ ATR-FTIR analysis was employed to investigate the reaction intermediates associated with CO2 reduction over F-Bi2O3-Ov [Figure 4A]. The spectra collected at applied currents ranging from -100 to
Figure 4. (A) In situ ATR-FTIR spectra of F-Bi2O3-Ov at different current densities. (B) PDOS for the Bi and O atomic p-orbitals in Bi2O3-Ov and Bi, O and F atomic p-orbitals in F-Bi2O3-Ov. (C) Gibbs free energy diagrams of the CO2 to formate pathways on Bi2O3-Ov and F-Bi2O3-Ov with the adsorption configurations of the corresponding intermediates.
The Gibbs free energy profiles for CO2 reduction to formate and the corresponding intermediate adsorption configurations were constructed to evaluate the reaction thermodynamics [Figure 4C,
CONCLUSIONS
In conclusion, we have designed and synthesized fluorine-doped Bi2O3 catalysts with abundant oxygen vacancies via a fluoride-assisted pyrolysis of Bi-MOF. The catalysts deliver an efficient selective conversion of CO2 to formate at industrial current densities. The combination of DFT calculations and experimental measurements has established the role of oxygen vacancies in promoting CO2 activation. Fluorine doping optimizes the local electronic structure of Bi2O3, leading to a high-performance electrocatalytic generation of formate. The findings of this work can inform the design of doped metal-based catalysts. Moreover, the study offers new insights to the electrocatalytic reduction of CO2 into value-added products.
DECLARATIONS
Authors’ contributions
Conceptualization, methodology, investigation, formal analysis and writing (first draft): Qiao, Y.
Conceptualization, validation, writing-review and editing: Xu, Y.
Data provision, review and editing: Yang, J.
Data verification and manuscript revision: Ding, J.; Gu, X.
Availability of data and materials
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author(s).
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22162019, 22261040, 22462019), the Science and Technology Projects of Inner Mongolia Autonomous Region (2021GG0195) and the Natural Science Foundation of Inner Mongolia Autonomous Region (2021MS02017).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Gidden, M. J.; Joshi, S.; Armitage, J. J.; et al. A prudent planetary limit for geologic carbon storage. Nature 2025, 645, 124-32.
2. O’brien, C. P.; Miao, R. K.; Shayesteh Zeraati, A.; Lee, G.; Sargent, E. H.; Sinton, D. CO2 electrolyzers. Chem. Rev. 2024, 124, 3648-93.
3. Tyne, R. L.; Barry, P. H.; Lawson, M.; et al. Rapid microbial methanogenesis during CO2 storage in hydrocarbon reservoirs. Nature 2021, 600, 670-4.
4. Wang, H.; Kang, X.; Han, B. Optimizing CO2 electroreduction: theoretical insights for enhancing efficiency across elementary steps. Chem. Soc. Rev. 2025, 54, 10156-244.
5. Ye, N.; Wang, K.; Tan, Y.; et al. Industrial-level CO2 to formate conversion on Turing-structured electrocatalysts. Nat. Synth. 2025, 4, 799-807.
6. Khan, B.; Faheem, M. B.; Peramaiah, K.; et al. Photoelectrochemical CO2-to-formic acid conversions: advances in photoelectrode designs and scale-up strategies. Adv. Energy Mater. 2025, 16, e04018.
7. Huang, Q.; Qian, Z.; Ye, N.; et al. In situ reconstructed hydroxyl-rich atomic-thin Bi2O2CO3 enables ampere-scale synthesis of formate from CO2 with activated water dissociation. Adv. Mater. 2025, 37, 2415639.
8. Chu, Y.; Yang, Y.; Cao, D.; et al. Regulating the rate-determining step of bismuth electrocatalysts by directional facet reconstruction for efficient CO2 reduction. Adv. Funct. Mater. 2025, 35, 2508387.
9. Chen, Z.; Xiao, Y.; Qiao, X.; et al. Monitoring chalcogenide ions-guided in situ transform active sites of tailored bismuth electrocatalysts for CO2 reduction to formate. Proc. Natl. Acad. Sci. USA 2025, 122, e2420922122.
10. Guo, W.; Cao, X.; Tan, D.; Wulan, B.; Ma, J.; Zhang, J. Thermal-driven dispersion of bismuth nanoparticles among carbon matrix for efficient carbon dioxide reduction. Angew. Chem. Int. Ed. 2024, 63, e202401333.
11. Xiao, Y.; Liu, D.; Yang, J.; et al. Controllable reconstruction of β-Bi2O3/Bi2O2CO3 composite for highly efficient and durable electrochemical CO2 conversion. Nano Lett. 2025, 25, 6548-55.
12. Ren, X.; Liu, F.; Wu, H.; et al. Reconstructed bismuth oxide through in situ carbonation by carbonate-containing electrolyte for highly active electrocatalytic CO2 reduction to formate. Angew. Chem. Int. Ed. 2024, 63, e202316640.
13. Deng, P.; Wang, H.; Qi, R.; et al. Bismuth oxides with enhanced bismuth-oxygen structure for efficient electrochemical reduction of carbon dioxide to formate. ACS Catal. 2020, 10, 743-50.
14. Zhang, Y.; Zhang, G.; Song, Q.; et al. Spin-polarization in rigid/soft layered oxide catalyst regulates key intermediates for efficient CO2-to-formate conversion. Angew. Chem. Int. Ed. 2026, 65, e17481.
15. Chen, X.; Lu, R.; Li, C.; et al. Activating inert non-defect sites in Bi catalysts using tensile strain engineering for highly active CO2 electroreduction. Nat. Commun. 2025, 16, 1927.
16. Wang, S.; Yuan, X.; Zhou, S.; et al. Single-atomic-Ni electrocatalyst derived from phthalocyanine-modified MOF for convoying CO2 intelligent utilization. Energy Mater. 2024, 4, 400032.
17. Wang, Y.; Li, G.; Feng, J.; et al. Boosting electrochemical CO2 reduction to formate over La-doped SnO2 via pinning effect and water activation. J. Am. Chem. Soc. 2025, 147, 40126-35.
18. Feng, J.; Wu, L.; Liu, S.; et al. Improving CO2-to-C2+ product electroreduction efficiency via atomic lanthanide dopant-induced tensile-strained CuOx catalysts. J. Am. Chem. Soc. 2023, 145, 9857-66.
19. Yang, X.; Wu, Z.; Li, Y.; et al. Atomically dispersed cerium on copper tailors interfacial water structure for efficient CO-to-acetate electroreduction. Nat. Commun. 2025, 16, 2811.
20. Lei, H.; Yang, W.; Hu, S.; et al. Synergistic effect of boron and oxygen coordination on ruthenium clusters for industrial water splitting in alkaline medium. Angew. Chem. Int. Ed. 2025, 64, e202503871.
21. Wang, Q.; Dai, M.; Li, H.; et al. Asymmetric coordination induces electron localization at Ca sites for robust CO2 electroreduction to CO. Adv. Mater. 2023, 35, 2300695.
22. Li, Y.; Zou, J.; Sun, L.; et al. Strong electronic interactions of the abundant Cu/Ce interfaces stabilized Cu2O for efficient CO2 electroreduction to C2+ products under large current density. Adv. Funct. Mater. 2025, 35, 2509899.
23. Huang, F.; Chen, X.; Sun, H.; et al. Atmosphere induces tunable oxygen vacancies to stabilize single-atom copper in ceria for robust electrocatalytic CO2 reduction to CH4. Angew. Chem. Int. Ed. 2024, 64, e202415642.
24. Chawla, G.; Dutta, N.; Kediya, S.; et al. Enhancing C2+ product faradaic efficiency in CO2 reduction using fluorine-stabilized superhydrophobic copper (δ+). J. Am. Chem. Soc. 2025, 147, 38169-79.
25. Jia, C.; Tan, X.; Sun, Q.; et al. Fluorine doping-assisted reconstruction of isolated Cu sites for CO2 electroreduction toward multicarbon products. Adv. Mater. 2025, 37, 2417443.
26. An, X.; Li, S.; Yang, Z.; et al. F-doped In(OH)3 for electrochemical reduction of CO2 to formate. Chem. Eng. J. 2023, 455, 140720.
27. Yang, Y.; Gao, X.; Yang, S.; Shen, Y.; Xie, A. Synthesis and superior SERS performance of porous octahedron Cu2O with oxygen vacancy derived from MOFs. J. Mater. Sci. 2021, 56, 9702-11.
28. Zhang, C.; Lin, Z.; Jiao, L.; Jiang, H. L. Metal-organic frameworks for electrocatalytic CO2 reduction: from catalytic site design to microenvironment modulation. Angew. Chem. Int. Ed. 2024, 63, e202414506.
29. Ma, Y.; Zhang, G.; Yu, Q.; Lyu, S.; Duan, X.; Zhang, S. Heterogeneous nanoporous organic frameworks-based catalysts for electrochemical CO2 reduction reaction. Energy Mater. 2025, 5, 500053.
30. Qi, Q.; Zhang, C.; Guo, T.; et al. Spatiotemporal control of MOF reconstruction unlocks efficient oxygen evolution. Adv. Mater. 2026, 38, e15800.
31. Su, Y.; Otake, K.; Zheng, J. J.; et al. Diffusion-rate sieving of propylene and propane mixtures in a cooperatively dynamic porous crystal. Nat. Commun. 2024, 15, 2898.
32. Su, Y.; Otake, K.; Zheng, J. J.; Horike, S.; Kitagawa, S.; Gu, C. Separating water isotopologues using diffusion-regulatory porous materials. Nature 2022, 611, 289-94.
33. Liu, C.; Mei, B.; Shi, Z.; et al. Operando formation of highly efficient electrocatalysts induced by heteroatom leaching. Nat. Commun. 2024, 15, 242.
34. Wang, J.; Tang, W.; Zhu, Z.; et al. Stabilizing lattice oxygen of Bi2O3 by interstitial insertion of indium for efficient formic acid electrosynthesis. Angew. Chem. Int. Ed. 2025, 64, e202423658.
35. Liu, W.; Zhai, P.; Li, A.; et al. Electrochemical CO2 reduction to ethylene by ultrathin CuO nanoplate arrays. Nat. Commun. 2022, 13, 1877.
36. Xin, Y.; Zhu, Q.; Gao, T.; et al. Photocatalytic NO removal over defective Bi/BiOBr nanoflowers: the inhibition of toxic NO2 intermediate via high humidity. Appl. Catal. B Environ. 2023, 324, 122238.
37. Yang, C.; Wang, R.; Yu, C.; et al. Engineering stable Cu+-Cu0 sites and oxygen defects in boron-doped copper oxide for electrocatalytic reduction of CO2 to C2+ products. Chem. Eng. J. 2024, 484, 149710.
38. Deng, P.; Yang, F.; Wang, Z.; et al. Metal-organic framework-derived carbon nanorods encapsulating bismuth oxides for rapid and selective CO2 electroreduction to formate. Angew. Chem. Int. Ed. 2020, 59, 10807-13.
39. Wang, X.; Zhang, Y.; Wang, S.; et al. Steering geometric reconstruction of bismuth with accelerated dynamics for CO2 electroreduction. Angew. Chem. Int. Ed. 2024, 63, e202407665.
40. Jia, B.; Chen, Z.; Li, C.; et al. Indium cyanamide for industrial-grade CO2 electroreduction to formic acid. J. Am. Chem. Soc. 2023, 145, 14101-11.
41. Zheng, Z.; Zheng, X.; Wang, L.; et al. Harnessing electrocatalytic coupling of carbon dioxide and methanol for high-efficiency formic acid production. Angew. Chem. Int. Ed. 2025, 64, e202512078.
42. Zhu, S.; Jiang, B.; Cai, W.; Shao, M. Direct observation on reaction intermediates and the role of bicarbonate anions in CO2 electrochemical reduction reaction on Cu surfaces. J. Am. Chem. Soc. 2017, 139, 15664-7.
43. Delmo, E. P.; Wang, Y.; Song, Y.; et al. In situ infrared spectroscopic evidence of enhanced electrochemical CO2 reduction and C-C coupling on oxide-derived copper. J. Am. Chem. Soc. 2024, 146, 1935-45.
44. Ling, Z.; Yin, Y.; Ni, W.; et al. Efficient CO2-to-HCOOH conversion at industrial current density in strong acid via synergistic water activation. Adv. Funct. Mater. 2025, 36, e29166.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].