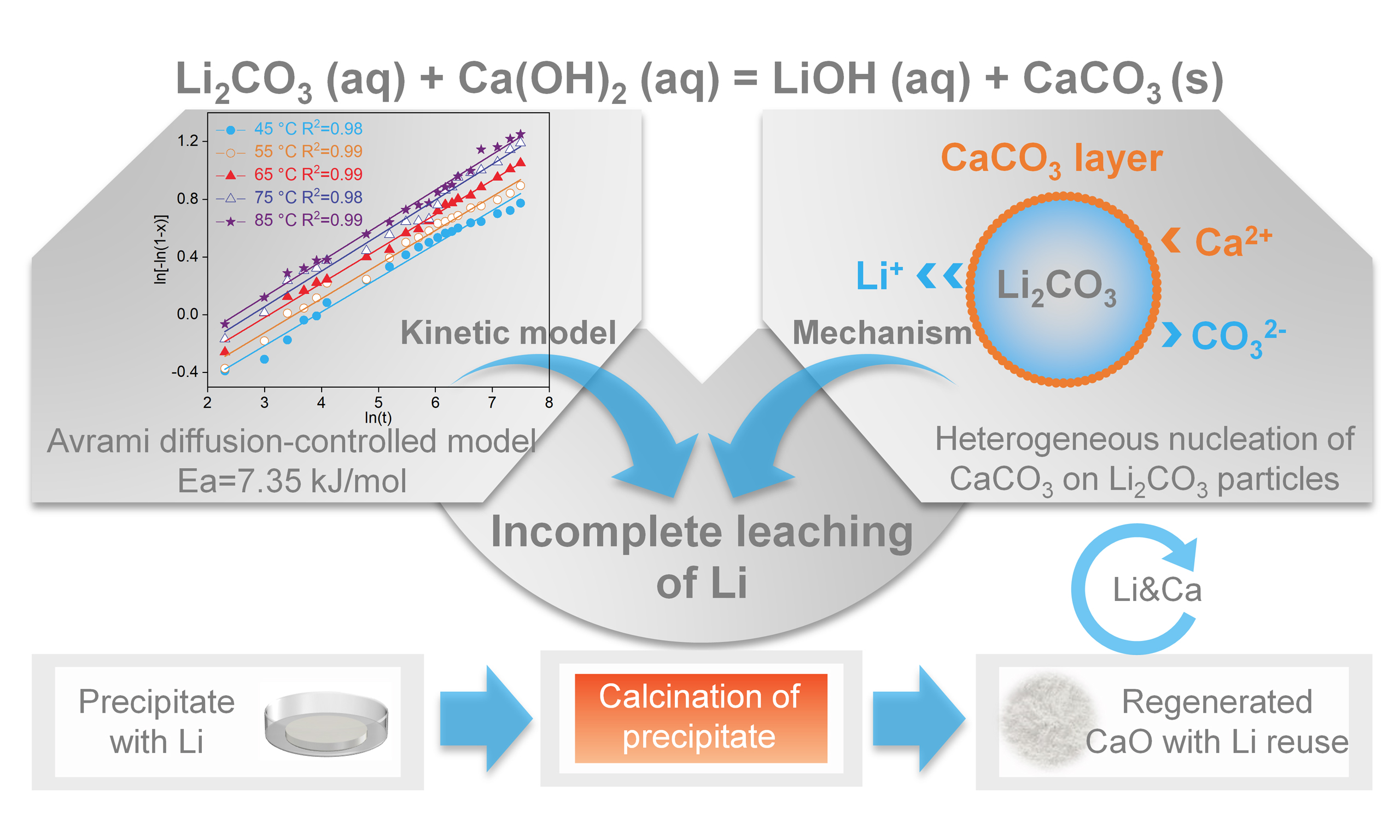
Optimizing waste management to improve the process for producing battery-grade LiOH·H2O from industrial-grade Li2CO3
Abstract
Battery-grade lithium hydroxide monohydrate (LiOH·H2O) is critical for lithium-ion batteries, yet its conventional causticization production faces sustainability challenges regarding waste management. This work systematically investigated the leaching kinetics and controlling mechanisms of lithium (Li) from lithium carbonate (Li2CO3) during causticization. The leaching behavior was found to follow the Avrami diffusion-controlled model, exhibiting a fast initial reaction rate with an apparent activation energy of
Keywords
INTRODUCTION
Lithium (Li) is one of the key elements driving the new round of scientific and technological revolution worldwide[1-4]. The rapid development of lithium-ion batteries (LIBs) has become the main driving force behind the increasing global lithium consumption. Therefore, cost-effective and environmentally sustainable synthesis of lithium compounds (e.g., lithium hydroxide and lithium carbonate) has attracted widespread attention from researchers. In recent years, LiOH and its monohydrate (LiOH·H2O) have become important raw materials for cathode production in Li-ion batteries. Studies have shown that the use of LiOH enhances the electrochemical performance of the cathode material compared with Li2CO3 or other Li sources[5-8].
Due to the complexity of global lithium resources, apart from high-grade spodumene in Australia[9-11] and some brine with high content of Li in South America (Argentina, Bolivia and Chile)[12-14], most minerals are difficult to directly use in the preparation of battery-grade lithium compounds. However, the synthesis of cathode materials and lithium hexafluorophosphate electrolyte for LIBs requires high-purity battery-grade lithium compounds[15-17]. Therefore, the development of technologies to further purify industrial-grade Li2CO3 and produce battery-grade lithium compounds can facilitate the commercialization of products from resources.
Currently, the majority of LiOH is produced from industrial-grade Li2CO3, with production technologies divided into the electrodialysis with bipolar membranes process (EBMP) and the causticization process (CP)[18]. EBMP involves first converting Li2CO3 into a Li-containing solution using sulfuric acid, then producing a LiOH solution via the cathodic reaction in membrane electrolysis, and finally crystallizing the solution to obtain LiOH·H2O. Under alkaline conditions in the cathodic chamber, impurities such as Mg2+, Fe3+, and Al3+ in industrial-grade Li2CO3 are converted into precipitates and removed[12,13,19]. The CP uses the reaction of Li2CO3 and calcium hydroxide in water to produce LiOH and calcium carbonate. The reaction formula is Equation (1).
The LiOH product is then obtained by crystallization of the LiOH solution. CP is more straightforward than EBMP, but it presents several challenges that must be addressed. First, CP suffers from low reaction conversion efficiency due to the low solubility of the raw materials. Second, large quantities of Li-containing CaCO3 waste residues are generated, leading to high waste disposal costs and substantial Li loss.
Previous studies have indicated that before the optimization of electrolysis efficiency and membrane costs, CP holds an advantage over EBMP in scaling up production more cost-effectively[18]. However, there is a research gap in the analysis of the complete life cycle and waste management of CP. Researchers[20] reported the use of CP in the laboratory to recover spent Li2CO3 from breathing apparatus and investigated the impact of causticization and crystallization operating conditions on the yield and purity of LiOH. However, there is a lack of mechanistic explanations and waste recycling methods suitable for large-scale production. When CP is used for large-scale production in factories, water circulation and equipment selection affect energy consumption, the yield of Li, and the purity of the product[21]. In particular, the conversion efficiency of the causticization reaction greatly affects the recovery efficiency of Li. Some researchers[22] have reported that the caustic conversion of Li2CO3 involves thermodynamic equilibrium, where CaCO3 dissolves when the concentration of LiOH in the solution reaches a certain level. However, because industrial processes operate within limited timeframes in which full equilibrium is seldom achieved, the reaction is primarily kinetically controlled. Therefore, it is necessary to investigate the kinetics of the causticization reaction. Some researchers[23] have reported the effects of temperature on causticization conversion efficiency and explained the decrease in Li leaching efficiency based on the principle of atomic exchange. However, the leaching efficiency of Li reached only 68% after 10 h, which poses challenges for industrial application.
In this study, the optimization pathways for leaching efficiency of Li from Li2CO3 were explored through a systematic investigation of the leaching kinetics and controlling mechanisms during the causticization process. Results of crystal structure, morphology, and elemental distribution of the causticization precipitate (CAC) under different reaction times validated the rationality of the selected kinetic model and elucidated the mechanistic link between Li leaching and CAC crystallization. Then, we proposed a strategy involving CAC calcination to regenerate calcium oxide, achieving closed-loop recycling of Li and calcium (Ca). Finally, we conducted a detailed economic and environmental evaluation of CP with closed-loop recycling of Li and Ca (CP-R), and compared it with conventional CP and the reported EBMP[24]. This work investigated the reaction mechanism of causticization and utilized calcination to achieve closed-loop recovery of Li and Ca, along with a significant reduction in solid waste. Consequently, this study provides an actionable strategy for waste management and lifecycle optimization of CP.
EXPERIMENTAL
Materials
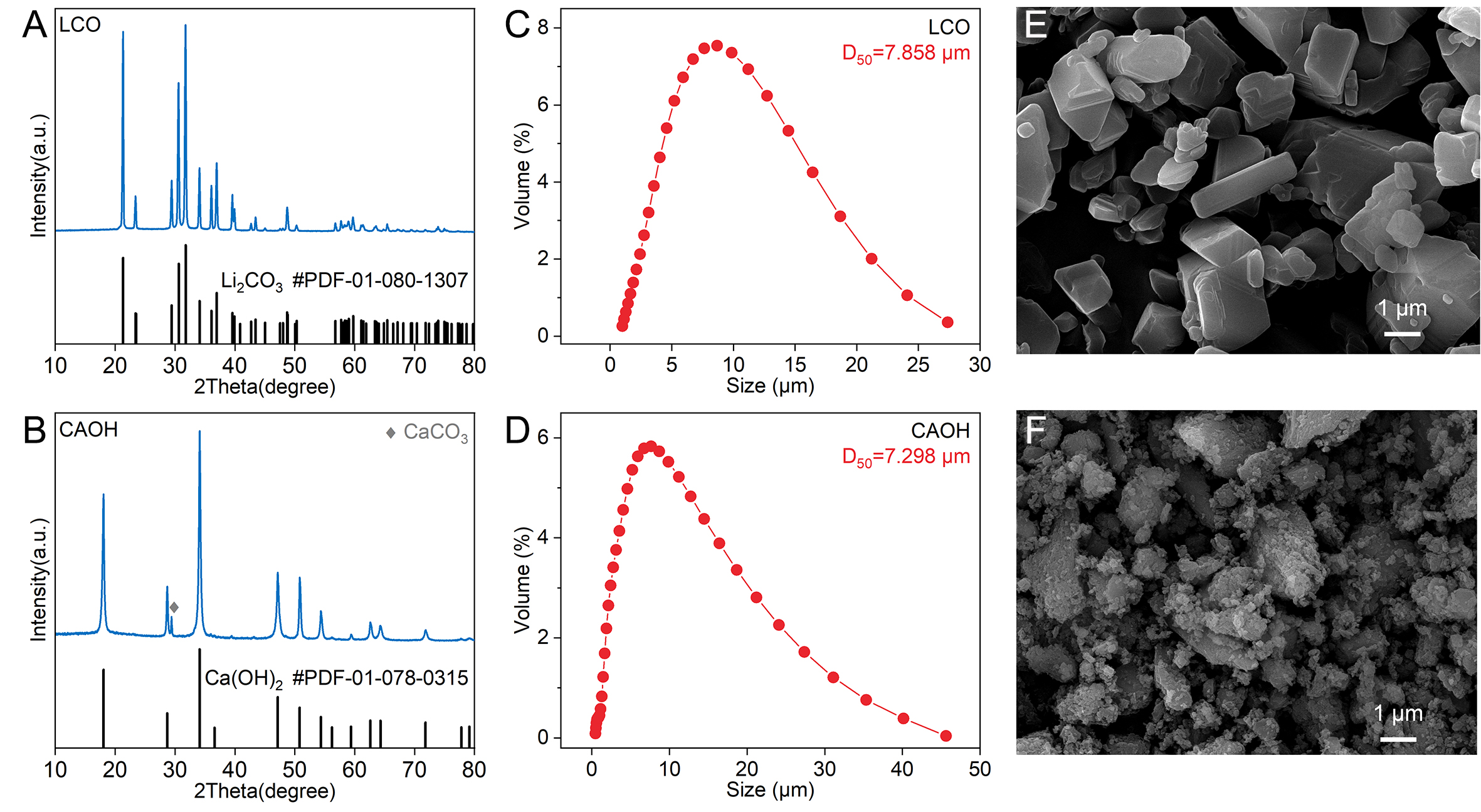
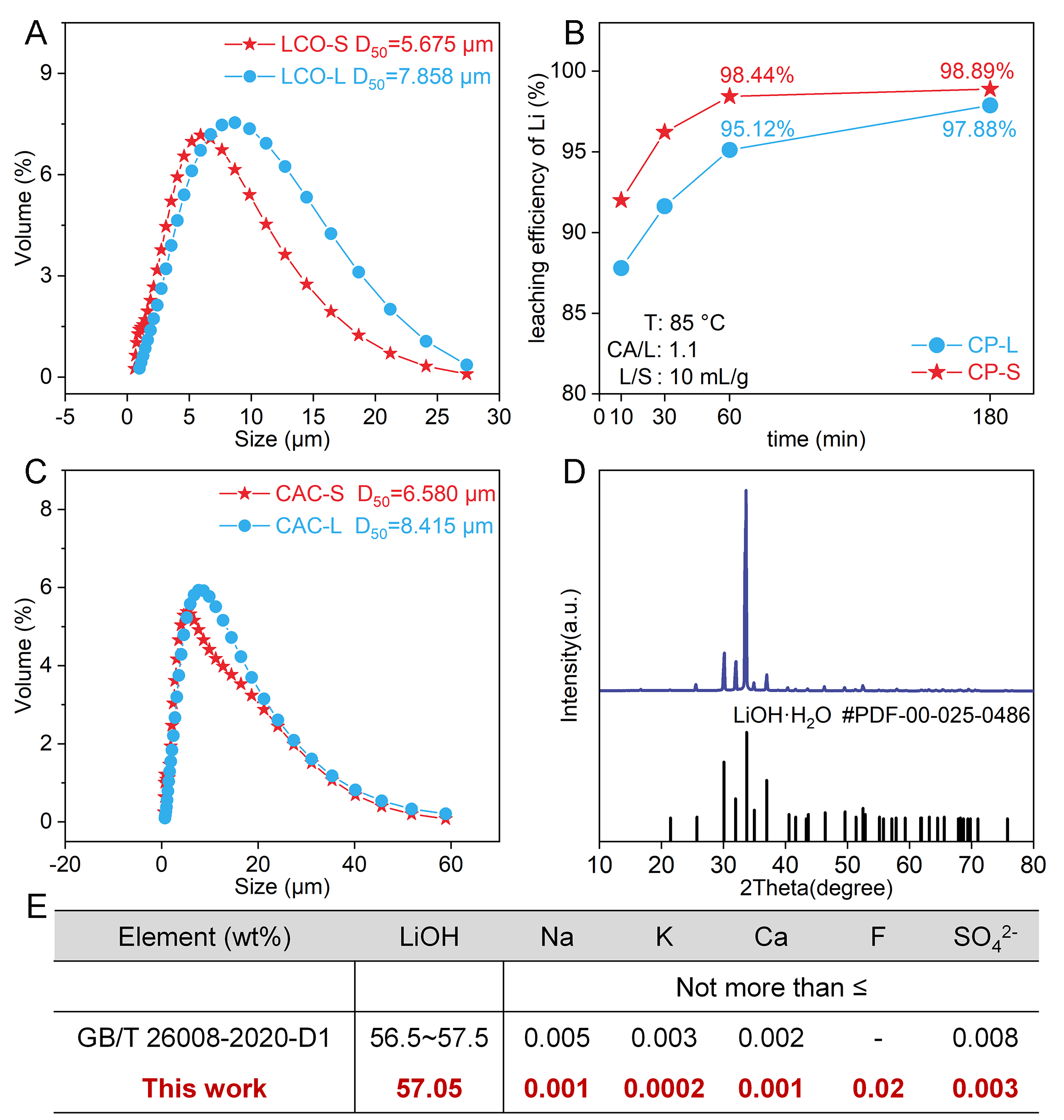
The reactants, industrial-grade lithium carbonate (referred to as LCO) and slaked lime (referred to as CAOH) used in this study were provided by Jiangxi Yunwei New Materials Co., Ltd., Yichun, China. The chemical compositions of LCO and CAOH are listed in Supplementary Table 1. The main diffraction peaks of LCO and CAOH correspond to the standard patterns of Li2CO3 and Ca(OH)2, respectively, with CAOH containing a small amount of CaCO3 formed by carbonation during storage in air [Figure 1A and B]. The D50 values of LCO and CAOH are close [Figure 1C and D], yet there is a distinct difference in their microscopic morphology. Specifically, the primary particles of LCO are blocky [Figure 1E], whereas CAOH is mostly composed of secondary particles formed by the agglomeration of small-sized primary particles [Figure 1F]. All other chemical reagents used in this work were of analytical grade and were purchased from Sinopharm Chemical Reagent Co., Ltd. All solutions were prepared with deionized water.
Methods
Causticization experiment
The experimental procedure is illustrated in Supplementary Figure 1. The pulping experiment of CAOH in deionized water was conducted in a reactor equipped with a condenser. The pulping time was 0.5 h, the stirring speed was 500 rpm, and the slurry was preheated to ensure that the desired temperature was reached at the beginning of causticization. The amount of deionized water used for pulping was the total quantity required for causticization, and no additional water was added during the process. The LCO powder and CAOH slurry were rapidly mixed and causticized at constant temperatures (45, 55, 65, 75, and 85 °C) for preset time intervals. After the reaction, the slurry was separated by suction filtration and the precipitate was dried in an oven at 150 °C for 3 h. The filtered solution was referred to as the Li-leachate. This leachate was then evaporated and crystallized using a rotary evaporator to obtain crystalline solids.
In the kinetic experiments, at regular intervals, 2 mL of the slurry was withdrawn and filtered to analyzing the elemental concentrations. The liquid-solid separation method was the same as described above. Sample extraction and liquid-solid separation were both performed rapidly to ensure accurate determination of leaching efficiencies corresponding to specific reaction times. The CAC samples extracted at different times were named CAC-time (e.g., CAC-1 represents the CAC sample withdrawn at 1 min of causticization).
In comparative experiments using reactants with different particle sizes, the experimental group employed mechanically ground Li2CO3 as the reactant (denoted as LCO-S, with the corresponding causticization process and precipitate denoted as CP-S and CAC-S, respectively), whereas the control group uses Li2CO3 prior to grinding (denoted as LCO-L, with CP-L and CAC-L). The causticization procedure was consistent with that used in the kinetic experiment.
Calcination of CAC
The CAC was placed in a tubular furnace and heated to the desired temperatures (920, 940, 960, 980, and 1,000 °C) at a controlled heating rate of 5 °C/min, held for 1 h, and then cooled naturally to room temperature, yielding regenerated calcium oxide (CAO).
Characterization
The crystalline phases of the samples were identified by X-ray diffraction (XRD, PANalytical, Empyrean 2, Netherlands) using CuKα radiation. Scanning electron microscopy (SEM, JEOL, JSM-IT700HR, Japan) coupled with energy dispersive X-ray spectroscopy (EDS, Oxford, Xplore 30, United Kingdom) was employed to analyze the microstructures and element distribution of the samples. The concentrations of metal ions in the samples were conducted by inductively coupled plasma (ICP, Thermo Fisher Scientific, ICAP7400radial, United States). The CaO content in CAO was determined by complexometric titration. The particle size of solid samples was characterized by a laser particle size analyzer (Malvern PANalytical, Mastersizer 3000, United Kingdom).
Analytical methods
Leaching efficiency of Li. Thermodynamic analysis data were obtained from the HSC software database. The reaction conversion was quantified by the Li leaching efficiency to directly reflect the recovery of the target product and eliminate interference from excess reagents. The leaching efficiency of Li was calculated according to Equation (2), where x is the leaching efficiency of Li (%), V is the volume of solution (L), cLi,i is theconcentration of Li in sample i (g/L), mLCO is the initial mass of the Li2CO3 used as reactant (g, dried solid), and Li is the content of Li in the Li2CO3 used as reactant (mass fraction, %, dried solid).
Life cycle assessment. OpenLCA software (version 2.4.0; GreenDelta GmbH) and the Ecoinvent database (version 3.9) were used to conduct the life cycle assessment (LCA) for analyzing the overall environmental performance of the entire CAL process. The cut-off allocation method was applied to account for the recyclability of condensate water and certain by-products within the system. Global warming potential (GWP) is a commonly used indicator for measuring the environmental impact and carbon footprint of processes[25-27]. The ecoinvent - ReCiPe 2016 v1.03, midpoint (H) impact assessment method was used to measure the GWP values of the processes (CP, CP-R and EBMP) to assess their respective environmental impacts.
RESULTS AND DISCUSSION
Optimization of causticization conditions
The orthogonal experimental design was employed to investigate the interactive effects of temperature (factor A), the molar ratio of Ca(OH)2 to Li2CO3 (CA/L, factor B), time (factor C), and liquid-solid ratio (L/S, factor D) for comprehensive optimization of the leaching parameters. Minitab software facilitated the experimental design and analysis. A total of 16 leaching trials measured variations in leaching efficiency of Li [Supplementary Tables 2 and 3].
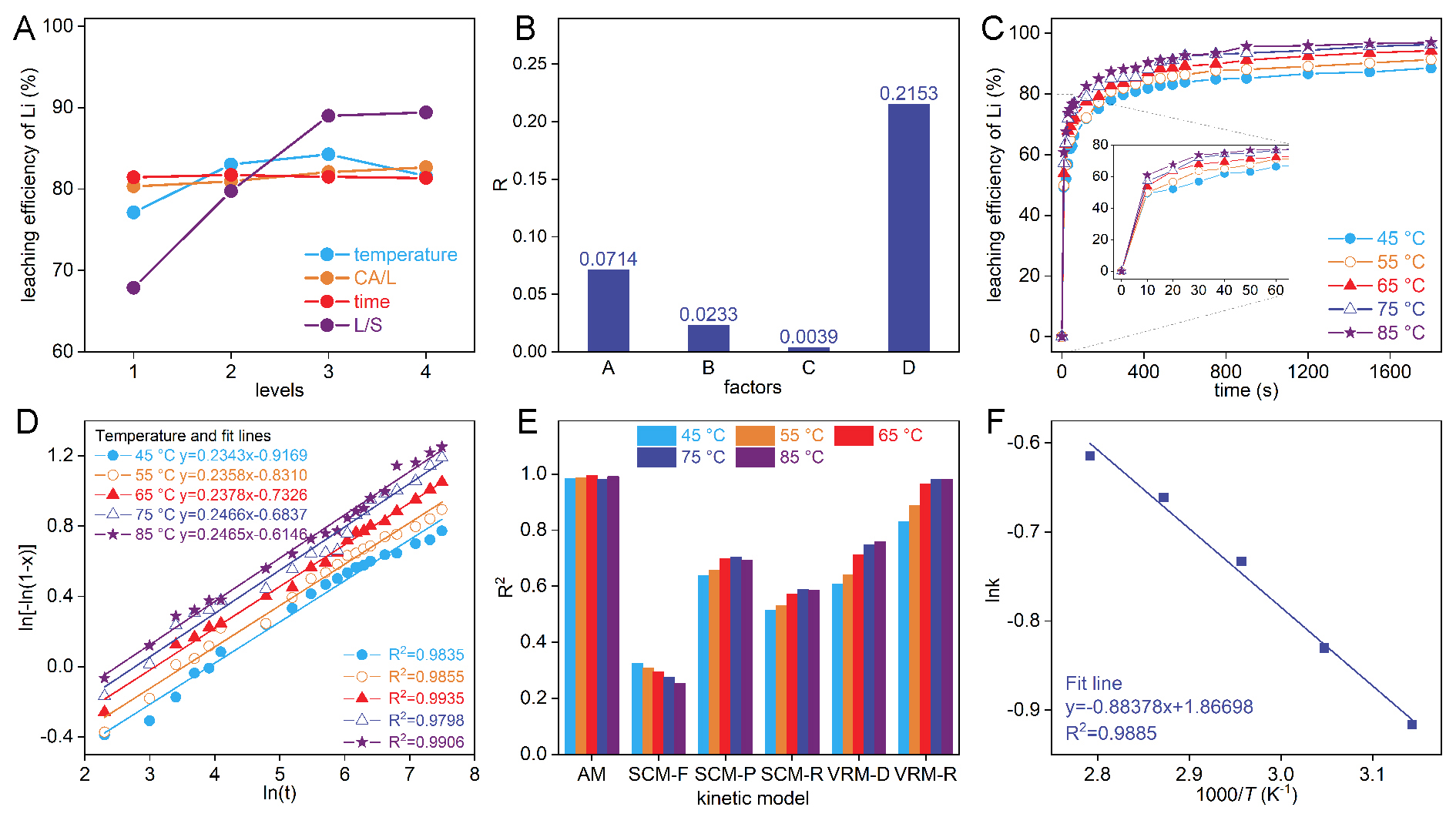
As shown in Figure 2A, the leaching efficiency of Li initially increases and then decreases with rising temperature. This is because rising temperature facilitates the crystallization of CaCO3[28,29], thereby promoting the conversion efficiency of the causticization reaction. However, an increasing in temperature reduces the solubility of Li2CO3 and Ca(OH)2[30]. When the temperature is excessively high, the reduced solubility of Li2CO3 and Ca(OH)2 results in a decrease in free calcium ions and carbonate ions in the slurry[23], thereby inhibiting the precipitation of CaCO3 and the leaching of Li. Therefore, 85 °C was selected as the optimized temperature. In addition, an increase in the CA/L is beneficial for Li leaching, as excess Ca(OH)2 as a reactant ensures the progression of the causticization reaction for Li2CO3. However, Ca(OH)2 in excess of stoichiometric ratio increases the mass of the precipitate and the inclusion loss of Li. Thus, the optimized CA/L ratio was 1.1. During the selected reaction period in the experiment, the Li leaching efficiency showed almost no fluctuation, indicating that the leaching of lithium approached equilibrium at 30 min. Therefore, the reaction time selected for the kinetic experiments was 30 min. Furthermore, the results in Figure 2A indicate that the Li leaching efficiency substantially increases with increasing L/S, and the rate of increase slows down when L/S exceeds 10 mL/g. This is because an increase in L/S usually indicates a reduction in the viscosity of the slurry, which enhances the diffusion rate[10]. However, adding solids and water in actual production increases the cost of the filtration and evaporation processes. Therefore, the optimized L/S selected for the kinetic experiments was 10 mL/g. The extreme difference analysis indicates that the order of the factors according to influence is D > A > B > C [Figure 2B]. The influence of L/S on Li leaching efficiency is substantially higher than that of other factors. This indicates that Li leaching is highly dependent on the dissolution amount of Li2CO3 and Ca(OH)2 in water.
Figure 2. (A) The mean values of factors at various levels and (B) the extreme difference analysis of the mean values in orthogonal experimental design. (C) The leaching efficiency of Li changes with time at different temperatures (CA/L: 1.1, L/S: 10 mL/g). (D) Plots of ln[-ln(1-x)] vs. ln(t) at different temperatures during 0-1,800 s (30 min). (E) The correlation coefficient (R2) between the kinetic models of different control mechanisms and the leaching of Li. (F) Arrhenius plot for Li leaching.
Kinetic analysis of Li leaching
To formulate a strategy for the industrial-scale production of LiOH through the causticization of Li2CO3 and Ca(OH)2, it is essential to conduct a detailed study on the kinetics of the causticization process. The results in Supplementary Figure 2 indicate that the Li leaching efficiency is no longer substantially affected by the stirring intensity once it exceeds 500 rpm, suggesting that the influence of liquid film diffusion can be considered eliminated at a stirring rate of 500 rpm. The changes in Li leaching efficiency with time under different temperatures were analyzed (see Section "Causticization experiment" for more details). As shown in Figure 2C, Li leaching is characterized by an initial transient reaction and a gradually decreasing rate.
In the causticization reaction, the reaction of Li2CO3, Ca(OH)2, and water is a non-catalytic heterogeneous reaction. Therefore, the shrinking core model (SCM) can be used to describe the leaching kinetics of Li[31,32]. The SCM includes three main mechanisms[33]: liquid film diffusion (SCM-F) (Equation 3), solid product layer diffusion (SCM-P) (Equation 4), and chemical reaction (SCM-R) (Equation 5). The relatively slow step is considered the rate-controlling step of the causticization process, and the corresponding equation is used to describe the kinetics of the causticization reaction[33,34].
where t is the real causticization time, x is the leaching efficiency of Li at t, and τF, τP, and τR are the time constants of completion of causticization in liquid film diffusion, solid product layer diffusion and chemical reaction control, respectively. The correlation coefficient (R2) is used to evaluate the goodness of fit between the Li leaching efficiency and the kinetic equations (Equations 3-5), with the results shown in
In addition, the volume reaction model (VRM) has been adopted to describe the kinetics of leaching reactions[33,35]. VRM includes the diffusion-controlled type (VRM-D, Equation 6) and the chemical reaction-controlled type (VRM-R, Equation 7).
Where t, x are the same as those in Equations 3-5, kd and kr are apparent reaction constants. The plots of -ln(1-x) vs. t and [1/(1-x)]-1 vs. t are shown in Supplementary Figure 4. For the Li leaching efficiency at different temperatures, the linear fit (R2) with the VRM-R equation is below 0.9 in all cases.
As shown in Figure 2C, Li leaching exhibits characteristics of an initial instantaneous reaction, with leaching efficiencies reaching 49.2% (45 °C), 49.8% (55 °C), 53.8% (65 °C), 57.1% (75 °C), and 60.8% (85 °C) at 10 s, respectively. The leaching efficiency slightly increased with temperature, indicating that Li leaching was endothermic, which corresponds to the endothermic nature of CaCO3 during crystallization. Specifically, the crystallization process is also characterized by an initially high rate that gradually slows down, meaning that under specific conditions (usually a higher degree of supersaturation), there is explosive nucleation followed by slow crystal growth and fusion[38]. Avrami proposed the Avrami-Erofeev equation based on the concepts of nucleation and growth of atomic nuclei[39]. This equation has also been employed to describe the leaching kinetics of solid-liquid heterogeneous reactions[34,40], and can be converted to a linear expression as follows:
where t, x are the same as those in Equations 3-7, k is the rate constant for the reaction, n is an Avrami exponent that depending on the reaction mechanism and remains constant under different leaching conditions[35,40]. This study analyzed Li leaching during the causticization of Li2CO3 using Equation 8. The linear results and the corresponding parameters are listed in Figure 2D and Table 1. The R2 values at different temperatures are all close to 1 and higher than those of SCM and VRM [Figure 2E], and the n values are nearly equal [Table 1], indicating that the Li leaching process conforms to the Avrami model (AM). The following analyses are all based on AM. The arithmetic mean of n at different temperatures is 0.2402. It has been reported that when this value is less than 0.5, it indicates that the reaction is diffusion-controlled in the Avrami model[36,40], suggesting a higher initial reaction rate, which is consistent with the variation pattern of lithium leaching efficiency over time [Figure 2C]. Additionally, the axis intercepts of all straight lines in Figure 2D were used to determine the apparent activation energy (Ea) of Li leaching based on the Arrhenius equation (Equation 9).
Values of n and lnk at different temperatures
| T (°C) | n | lnk | R2 |
| 45 | 0.2343 | -0.9169 | 0.9835 |
| 55 | 0.2358 | -0.8310 | 0.9855 |
| 65 | 0.2378 | -0.7326 | 0.9935 |
| 75 | 0.2466 | -0.6837 | 0.9798 |
| 85 | 0.2465 | -0.6146 | 0.9906 |
where A represents the pre-exponential factor, R is the ideal gas constant (8.314 J·mol-1·K-1), and T denotes the absolute temperature in Kelvin (K). As illustrated in Figure 2F, the Ea calculated from the slope of the line is 7.35 kJ·mol-1 within the temperature range of 45-85 °C. In kinetic studies[33,41,42], values of Ea below
Mechanism of the causticization for Li2CO3
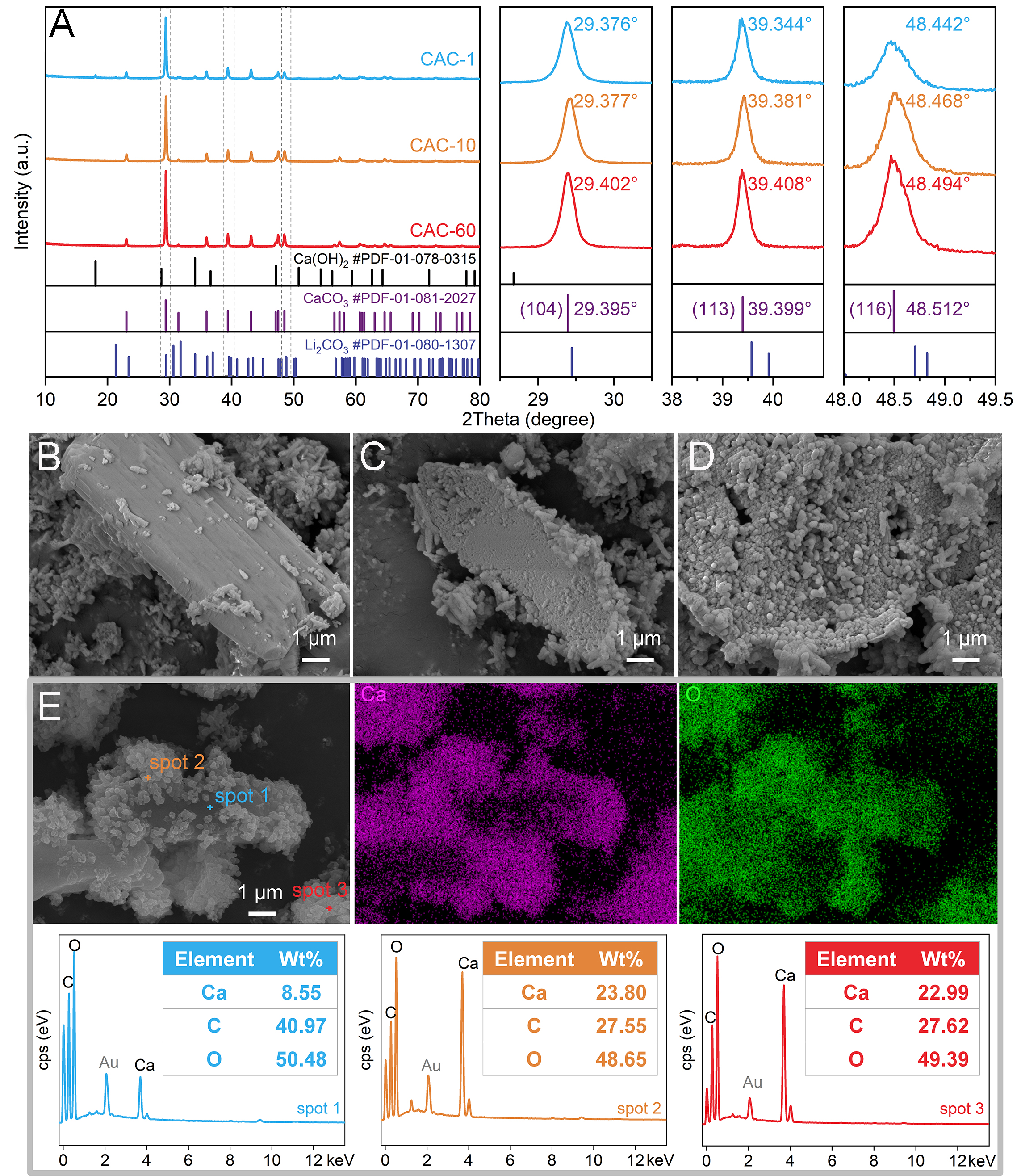
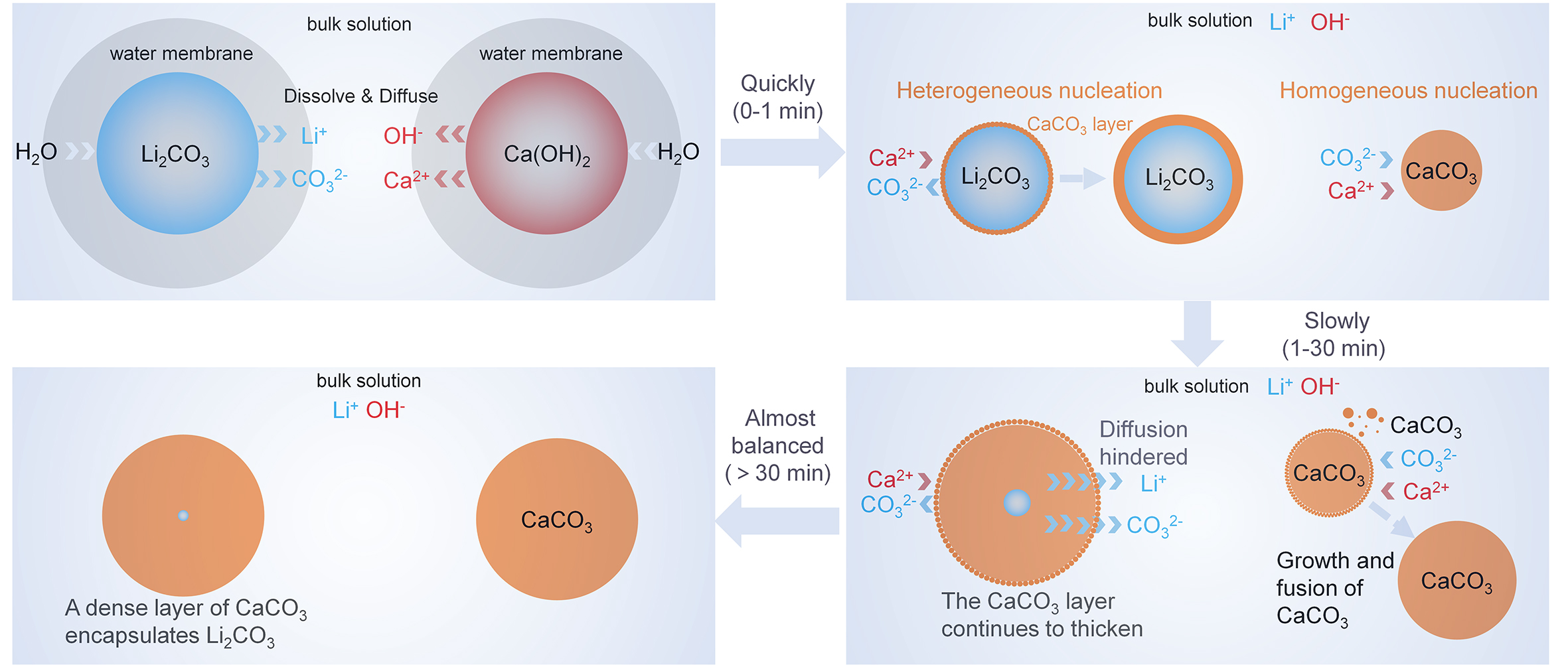
To analyze the causticization process, the crystal structure and microscopic morphology of the residual precipitate at different stages were characterized using the Avrami model. Specifically, reaction times of 1, 10, and 60 min were selected to represent the initial, intermediate, and equilibrium stages of causticization, respectively (temperature: 85 °C, CA/L: 1.1, L/S: 10 mL/g). The XRD patterns of CAC-1, CAC-10 and CAC-60 obtained from different causticization stages are shown in Figure 3A. Compared with the standard calcite CaCO3 (#PDF-01-081-2027), the diffraction peaks of the CACs at the (104), (113), and (116) planes are shifted due to the superposition of Li2CO3 diffraction peaks (#PDF-01-080-1307)[23,43,44]. Specifically, the diffraction peaks of CAC-60 are closer to the standard spectrum of calcite CaCO3 (#PDF-01-081-2027) than those of CAC-1 and CAC-10. This indicates the causticization reaction is characterized by the gradual disappearance of Li2CO3 and the progressive perfection of CaCO3 crystallization. Additionally, the XRD patterns [Figure 3A] confirm that the causticization precipitates prepared under the investigated conditions exhibit the calcite crystal structure. This is attributed to the lower solubility of calcite compared with aragonite and vaterite[22], making calcite the more stable phase during causticization.
Figure 3. (A) The XRD pattern of CAC-1, CAC-10 and CAC-60, the SEM images of (B) the CAC-1, (C) the CAC-10, and (D) the CAC-60, (E) the SEM images, EDS elemental mapping and point scanning results of CAC-1 (temperature: 85 °C, CA/L: 1.1, L/S: 10 mL/g).
The SEM images in Figure 3B-D show the morphology of CAC-1, CAC-10 and CAC-60, respectively. CAC-1 contains primary particles with a considerable disparity in size, and some of the smaller primary particles precipitate on the surface of the larger particles [Figure 3B]. In CAC-10, the large primary particles gradually corrode, and the small primary particles precipitated on their surface increase in number and grow
Based on the aforementioned results regarding the Li leaching kinetics and the changes in the crystal structure and morphology of CAC over time, the crystallization process of CaCO3 is used to explain the causticization of Li2CO3 [Figure 4]. During the pulping of Ca(OH)2 and the initial stage of the causticization reaction after mixing Li2CO3 with the slurry (within 1 min), Ca2+, Li+, CO32-, and OH- dissolve into the solution. Due to the low solubility of CaCO3[45], the supersaturation of Ca2+ and CO32- in the solution is high, promoting the homogeneous nucleation of CaCO3. However, due to the lower energy required for heterogeneous nucleation[46], some calcium carbonate still undergoes heterogeneous nucleation using Li2CO3 and Ca(OH)2 as nuclei [Figure 3E and Supplementary Figure 5]. Research shows[23] that high temperature is conducive to forming more isolated CaCO3 precipitates. An increase in temperature within a certain range helps to reduce the coating of Li2CO3 by CaCO3, thereby promoting Li leaching [Figure 2A]. Furthermore, based on the kinetic study conclusion that diffusion through the solid product layer is the rate-controlling step for Li leaching [Figure 2D-F], temperature influences the formation of the CaCO3 solid product layer on the surface of Li2CO3 by affecting the homogeneous and heterogeneous nucleation of CaCO3, thereby influencing the leaching of Li from Li2CO3 during causticization. In the subsequent causticization reaction (1-30 min), the CaCO3 layer coating the surface of Li2CO3 continues to thicken, hindering Li diffusion and resulting in a slower leaching rate of Li [Figure 2C]. The homogeneously nucleated calcium carbonate gradually grows during grain fusion and growth. When the causticization reaction approaches equilibrium
Effect of reactant particle size on Li leaching
While thermodynamic equilibrium sets the upper bound for conversion based on LiOH concentration (related to the L/S)[22], the observed Li leaching rates indicate that kinetics is the controlling factor in the causticization reaction. Consequently, enhancing kinetic performance is critical to bridging the gap between the actual Li yield and the thermodynamic limit within industrially relevant timescales. The kinetic characteristics of the causticization process indicate that increasing the temperature substantially enhances the crystallization rate of CaCO3 only in the initial stage, thereby promoting Li leaching. In contrast, the reduction in the particle size of the reactants is more conducive to minimizing the impact of CaCO3 coating on diffusion, thereby enhancing the Li leaching efficiency during the formation and thickening stages of the CaCO3 solid product layer. Figure 5A compares the two reactants, showing that the particle size of LCO-S is smaller than that of LCO-L. Since the amount of Ca(OH)2 added is in excess, even if a small portion of Ca(OH)2 is coated and unable to participate in the reaction, this has a negligible impact on Li leaching. Therefore, the same CAOH was used in both sets of experiments. To comprehensively compare the effects of Li2CO3 particle size on the maximum leaching efficiency and the time required to reach equilibrium, the reaction duration was extended to 180 min to ensure that the reactions in both experimental groups approached equilibrium as closely as possible. All other conditions were kept identical (temperature: 85 °C, CA/L: 1.1, L/S: 10 mL/g, as detailed in Section "Causticization experiment"). The comparison of Li leaching efficiency indicates that reducing particle size contributes to enhancing Li leaching. Furthermore, 60 min was identified as a characteristic time point to evaluate the reaction progress across different particle sizes. The leaching efficiency of Li in CP-S reaches 98.44%, which is higher than that in CP-L [Figure 5B]. Meanwhile, after
Figure 5. (A) Particle size distribution of LCO-S and LCO-L. (B) Leaching efficiency of Li in CP-S and CP-L at 10-180 min (temperature:
The XRD characteristic peaks of the crystalline solids obtained from the evaporation and crystallization of the Li-leachate [Supplementary Table 4] from CP-S conform to the standard pattern of
Economic and environmental assessment of the causticization process
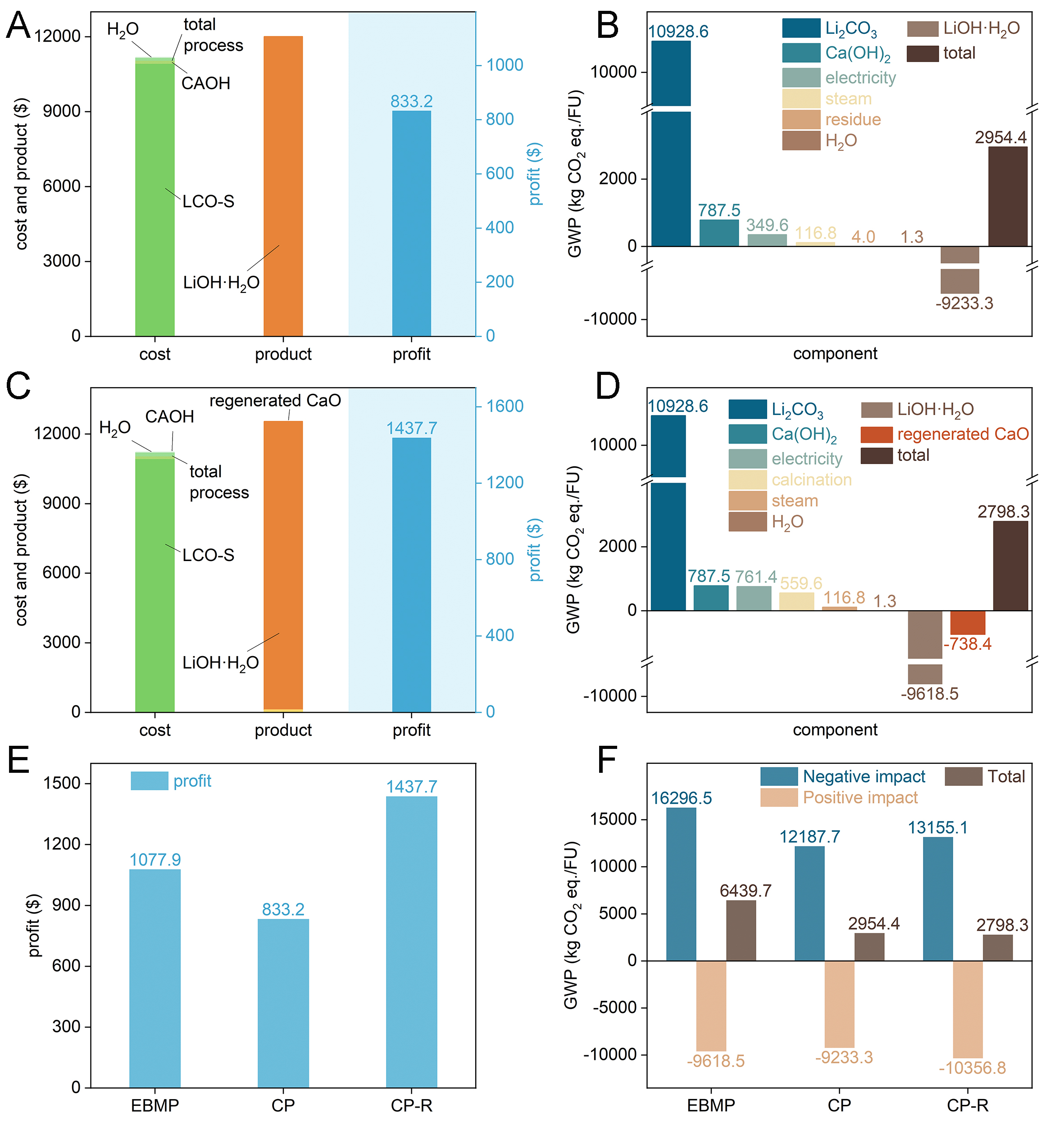
First, since the crystallization mother liquor can be recycled[21], this study assumes no lithium loss during the crystallization process and a Li leaching efficiency of 98.44% for the evaluate the economics. Moreover, the calculation assumes that the evaporated condensed water can be recycled. Supplementary Figure 7 presents the lithium flow in the causticization process (CP), including the masses of intermediate products and battery-grade LiOH·H2O produced per tonne of treated LCO-S, along with relevant process parameters. Based on these, an economic assessment of CP was performed to evaluate its costs and benefits. The valuation of products, raw materials, and processing expenses was based on current market prices for energy, water, and auxiliary materials [Supplementary Table 5], as well as operational data such as the power consumption of industrial-scale equipment [Supplementary Table 6]. The resulting cost estimate for processing one tonne of LCO-S via CP is summarized in Supplementary Table 7. As shown in Figure 6A, when the price per tonne of battery-grade LiOH·H2O exceeds that of industrial LCO-S by $281, the profit from CP treatment per tonne of LCO-S is $833.2. Since LCO-S accounts for a high proportion of the cost, this indicates that the price difference between LCO-S and battery-grade LiOH·H2O is a key factor affecting the economics of CP. Furthermore, a greater quantity of product implies a larger profit margin, indicating that Li recovery efficiency directly impacts the economic benefits of CP.
Figure 6. Economic estimation of the (A) CP and (C) CP-R for producing battery-grade LiOH·H2O from each tonne of LCO-S. The GWP values of single component to environmental impacts for (B) CP and (D) CP-R through LCA. (E) Economic estimation and (F) the GWP values of environmental impacts for EBMP, CP, and CP-R.
LCA was conducted on the causticization process (CP), involving its decomposition into individual units for environmental impact assessment [Supplementary Figure 8, Supplementary Table 8]. The findings
Regenerated calcium oxide enables a closed loop for Li
The economic and environmental assessment results indicate that the profitability and environmental impact of CP are highly dependent on Li recycling efficiency. Therefore, factories producing battery-grade LiOH·H2O from industrial-grade Li2CO3 aim to recover as much Li as possible[21]. Based on the above research findings, Li loss primarily stems from the presence of Li in the filtered CAC cake, mainly originating from unreacted Li2CO3 and entrained Li-leachate [Supplementary Table 10]. Repeated washing of the CAC filter cake can yield low-concentration lithium-containing wash water to recover entrained Li-leachate [Supplementary Table 4]. However, Li2CO3 cannot be effectively recovered through washing, as it is coated by a dense layer of CaCO3 [Figure 4]. Meanwhile, the heterogeneous nucleation of CaCO3 cannot be completely avoided[23], which means that a small portion of Li2CO3 inevitably fails to participate in the reaction.
CAC consists of CaCO3, unreacted Li2CO3, and Ca(OH)2. Therefore, the CaCO3 and Ca(OH)2 in CAC can be regenerated into CaO through calcination for reuse in the causticization reaction[48], thereby recovering Li into the system. The data in Supplementary Figure 9 show that the decomposition reactions of CaCO3 and Ca(OH)2 can proceed above 900 and 500 °C, respectively. Supplementary Figure 10 shows that the CaO content in CAO after CAC was calcined at 920, 940, 960, 980, and 1,000 °C, respectively (see Section "Calcination of CAC" for more details). The results indicate that increasing the calcination temperature contributes to enhancing the CaO content in CAO. Incompletely decomposed CAO contains impurities such as Ca(OH)2 and CaCO3 [Supplementary Figure 11].
The wash water was used to prepare the slurry for causticization, and the Li-containing CAC was regenerated into Li-containing CaO at high temperature and reused as a causticization reactant [Supplementary Table 10], realizing a closed loop for Li. Supplementary Figure 12 illustrates the Li and Ca flows within CP for recycling CAO (CP-R), detailing the mass of products obtained per tonne of LCO-S processed and the associated process parameters. Notably, although the 96.29% purity of the regenerated CaO slightly reduces the active calcium content, the optimized CA/L of 1.1 ensures a sufficient Ca2+ excess to drive Li leaching. Furthermore, to counteract for water consumption from CaO hydration and maintain the optimized L/S, additional water is added in the CP-R loop, as detailed in Supplementary Figure 12. The profit margin of CP-R is $1,437.7 [Figure 6C]. Compared with CP, the profit increase of CP-R is mainly attributed to the increased production of battery-grade LiOH·H2O and the reduced usage of CAOH enabled by the recycled CaO. Compared with CP, CP-R produces more products and reduces the usage of CAOH while consuming the same amount of LCO-S [Supplementary Figure 12], thereby lowering the carbon footprint [Figure 6D and Supplementary Figure 13, Supplementary Table 9]. However, the electricity-driven calcination of CAC inherently generates process CO2 and incurs additional energy costs, both of which are explicitly quantified in Supplementary Table 6-9. While these factors contribute to the overall footprint, CP-R still demonstrates a slightly lower total carbon footprint compared with CP. Future integration with green electricity offers a clear pathway to further minimize these indirect emissions[49].
Electrodialysis with bipolar membranes process (EBMP) is another internationally mainstream process for producing LiOH[18]. This study evaluates the economic benefits and environmental impacts
CONCLUSION
This work systematically investigated the leaching kinetics and controlling mechanisms of Li in the causticization process of Li2CO3. The leaching of Li conformed to the diffusion-controlled type of the Avrami model, with the leaching efficiency critically governed by the crystallization behavior of CaCO3. Specifically, the heterogeneous nucleation of CaCO3 on Li2CO3 particles reduced leaching efficiency of Li, and the method of reducing the initial particle size of Li2CO3 promoted Li leaching. Based on these insights, the proposed CP-R strategy successfully establishes a closed-loop cycle for both Li and Ca by regenerating high-purity quicklime from calcined residues. Economic evaluation and LCA demonstrate that CP-R achieves a higher profit margin and lower environmental impact than conventional CP and EBMP. In summary, CP-R offers a compelling alternative for producing battery-grade LiOH·H2O by simultaneously addressing solid waste disposal and achieving closed-loop Li recycling, which are challenges inherent to the conventional causticization process.
DECLARATIONS
Authors’ contributions
Conceived the research: Liu, S.; Wang, J.
Methodology, data analysis, and writing of the manuscript: Liu, S.
Supervision, writing - review and editing: Wang, J.
Data acquisition: Song, X.; Hu, Q.
Technical support and funding acquisition: Li, X.; Wang, Z.; Guo, H.; Yan, G.; Xiong, X.; Zhou, X.
All authors discussed the results and commented on the manuscript.
Availability of data and materials
Some results of supporting the study are presented in the Supplementary Materials. Other raw data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was financially supported by the National Key Research & Development Program of China (2022YF2906200), the Science and Technology Innovation Program of Hunan Province (2022RC3048), the Key Research and Development Program Project of Yichun City (2023ZDYFJH04) and the Science and Technology Innovation Program of Chenzhou City in Hunan Province (2023sfq61). Jiangxi Yunwei New Materials Co., Ltd. provided resource and financial support for this work.
Conflicts of interest
Song, X.; Hu, Q, Wang, J. are are affiliated with Jiangxi Yunwei New Materials Co., Ltd., their participation complies with the company’s internal policies, and there are no issues regarding affiliation or potential conflicts of interes, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Frith, J. T.; Lacey, M. J.; Ulissi, U. A non-academic perspective on the future of lithium-based batteries. Nat. Commun. 2023, 14, 420.
2. Cheng, A. L.; Fuchs, E. R. H.; Karplus, V. J.; Michalek, J. J. Electric vehicle battery chemistry affects supply chain disruption vulnerabilities. Nat. Commun. 2024, 15, 2143.
3. Nitou, M. V. M.; Fang, X.; Wang, J.; et al. The integration of LiNi0.8Co0.1Mn0.1O2 coatings on separators for elevated battery performance. Energy. Mater. 2025, 5, 500018.
4. Yang, X.; Huang, Y.; Li, J.; et al. Understanding of working mechanism of lithium difluoro(oxalato) borate in Li||NCM85 battery with enhanced cyclic stability. Energy. Mater. 2023, 3, 300029.
5. Zhang, B.; Ou, X.; Zheng, J.; et al. Electrochemical properties of Li2FeP2O7 cathode material synthesized by using different lithium sources. Electrochim. Acta. 2014, 133, 1-7.
6. Cao, K.; Shen, T.; Wang, K.; Chen, D.; Wang, W. Influence of different lithium sources on the morphology, structure and electrochemical performances of lithium-rich layered oxides. Ceram. Int. 2017, 43, 8694-702.
7. Anansuksawat, N.; Sangsanit, T.; Prempluem, S.; Homlamai, K.; Tejangkura, W.; Sawangphruk, M. How uniform particle size of NMC90 boosts lithium ion mobility for faster charging and discharging in a cylindrical lithium ion battery cell. Chem. Sci. 2024, 15, 2026-36.
8. Xiao, J.; Cao, X.; Gridley, B.; et al. From mining to manufacturing: scientific challenges and opportunities behind battery production. Chem. Rev. 2025, 125, 6397-431.
9. Rioyo, J.; Tuset, S.; Grau, R. Lithium extraction from spodumene by the traditional sulfuric acid process: a review. Min. Process. Extract. Metall. Rev. 2020, 43, 97-106.
10. Zhai, Q.; Liu, R.; Song, Y.; Mao, Z.; Sun, W. A novel technology for lithium extraction through low-temperature synergistic roasting of α-spodumene with lepidolite. Sep. Purif. Technol. 2025, 355, 129667.
11. Zhou, Q.; Ma, X.; Xiong, X. Extraction of lithium and phosphorus from amblygonite using calcium sulfate roasting and water leaching. Hydrometallurgy 2024, 225, 106282.
12. Ying, J.; Lin, Y.; Zhang, Y.; Yu, J. Developmental progress of electrodialysis technologies and membrane materials for extraction of lithium from salt lake brines. ACS. EST. Water. 2023, 3, 1720-39.
13. Niu, Y.; Yuan, Y.; Wang, Y.; et al. RETRACTED: unlocking industrial-scale lithium extraction from salt lake brines: a comprehensive sustainability, life cycle, and techno-economic comparative analysis. J. Clean. Prod. 2025, 525, 146535.
14. Guo, Z.; Ji, Z.; Wang, J.; Guo, X.; Liang, J. Electrochemical lithium extraction based on “rocking-chair” electrode system with high energy-efficient: the driving mode of constant current-constant voltage. Desalination 2022, 533, 115767.
15. Yu, Z.; Qu, X.; Wan, T.; et al. Synthesis and mechanism of high structural stability of nickel-rich cathode materials by adjusting Li-excess. ACS. Appl. Mater. Interfaces. 2020, 12, 40393-403.
16. Rosso, L.; Alcaraz, L.; Rodríguez-Largo, O.; López, F. A. Purification of Li2CO3 obtained through pyrometallurgical treatment of NMC black mass from electric vehicle batteries. ChemSusChem 2025, 18, e202401722.
17. Suárez, A.; Jara, A.; Castillo, R.; Gallardo, K. Analysis of trace impurities in lithium carbonate. ACS. Omega. 2024, 9, 20129-34.
18. Dahlkamp, J. M.; Quintero, C.; Videla, A.; Rojas, R. Production processes for LiOH - A review. Hydrometallurgy 2024, 223, 106217.
19. Yang, S.; Wang, Y.; Pan, H.; He, P.; Zhou, H. Lithium extraction from low-quality brines. Nature 2024, 636, 309-21.
20. Kim, K. J. Recovery of lithium hydroxide from spent lithium carbonate using crystallizations. Sep. Sci. Technol. 2008, 43, 420-30.
21. Grágeda, M.; González, A.; Alavia, W.; Ushak, S. Development and optimization of a modified process for producing the battery grade LiOH: optimization of energy and water consumption. Energy 2015, 89, 667-77.
22. Török, P.; Halasiné-Varga, I.; Duvivier, L.; et al. Lithium carbonate conversion to lithium hydroxide using calcium hydroxide: equilibrium is governed by vaterite formation. Inorg. Chem. 2025, 64, 23575-85.
23. Yuan, B.; Wang, J.; Cai, W.; Yang, Y.; Yi, M.; Xiang, L. Effects of temperature on conversion of Li2CO3 to LiOH in Ca(OH)2 suspension. Particuology 2017, 34, 97-102.
24. Chen, X.; Ruan, X.; Kentish, S. E.; Li, G.; Xu, T.; Chen, G. Q. Production of lithium hydroxide by electrodialysis with bipolar membranes. Sep. Purif. Technol. 2021, 274, 119026.
25. Miyan, N.; Omur, T.; Amed, B.; Özkan, H.; Aydın, R.; Kabay, N. Recycled waste concrete and metakaolin based alkali-activated paste: characterization, optimization, and life cycle assessment. Constr. Build. Mater. 2024, 416, 135233.
26. Cespi, D. A proposal of twelve principles for LCA of chemicals. Green. Chem. 2025, 27, 12107-14.
27. Liu, M.; Wang, F.; Zhang, S.; et al. Carbon footprint of battery-grade lithium chemicals in China. ACS. Sustain. Chem. Eng. 2025, 13, 3930-8.
28. Han, J. S.; Kang, D. S.; Seo, Y. B. Application of in situ calcium carbonate process for producing papermaking fillers from lime mud. ACS. Omega. 2021, 6, 3884-90.
29. Kingsbury, R. A guide to ion separations for the global energy transition. Joule 2025, 9, 102134.
30. Zhang, Y.; Hu, Y.; Wang, L.; Sun, W. Systematic review of lithium extraction from salt-lake brines via precipitation approaches. Miner. Eng. 2019, 139, 105868.
31. Liu, J.; Yin, Z.; Li, X.; Hu, Q.; Liu, W. Recovery of valuable metals from lepidolite by atmosphere leaching and kinetics on dissolution of lithium. Trans. Nonferrous. Met. Soc. China. 2019, 29, 641-9.
32. Zhang, D.; Sun, F.; Zhao, Z.; et al. Kinetics study on cobalt leaching from cobalt-bearing ternary sulfide in sulfuric acid solution under atmospheric pressure. Trans. Nonferrous. Met. Soc. China. 2024, 34, 1669-80.
33. Faraji, F.; Alizadeh, A.; Rashchi, F.; Mostoufi, N. Kinetics of leaching: a review. Rev. Chem. Eng. 2022, 38, 113-48.
34. Brahim J, Ait Hak S, Achiou B, Boulif R, Beniazza R, Benhida R. Kinetics and mechanisms of leaching of rare earth elements from secondary resources. Miner. Eng. 2022, 177, 107351.
35. Gu, K.; Li, W.; Han, J.; Liu, W.; Qin, W.; Cai, L. Arsenic removal from lead-zinc smelter ash by NaOH-H2O2 leaching. Sep. Purif. Technol. 2019, 209, 128-35.
36. Lin, M.; Liu, Y.; Lei, S.; Ye, Z.; Pei, Z.; Li, B. High-efficiency extraction of Al from coal-series kaolinite and its kinetics by calcination and pressure acid leaching. Appl. Clay. Sci. 2018, 161, 215-24.
37. Xu, Z.; Xu, L.; Wei, Q.; Shen, S.; Liu, J.; Zhu, Y. Microwave hydrothermal sulfuric acid leaching of spent cathode carbon from aluminum electrolysis for high efficiency removal of insoluble calcium fluoride. Waste. Manag. 2024, 179, 110-9.
39. Avrami, M. Granulation, phase change, and microstructure kinetics of phase change. III. J. Chem. Phys. 1941, 9, 177-84.
40. Hao, J.; Wang, X.; Wang, Y.; Wu, Y.; Guo, F. Optimizing the leaching parameters and studying the kinetics of copper recovery from waste printed circuit boards. ACS. Omega. 2022, 7, 3689-99.
41. Tan, Q.; Deng, C.; Li, J. Effects of mechanical activation on the kinetics of terbium leaching from waste phosphors using hydrochloric acid. J. Rare. Earths. 2017, 35, 398-405.
42. Lampinen, M.; Seisko, S.; Forsström, O.; et al. Mechanism and kinetics of gold leaching by cupric chloride. Hydrometallurgy 2017, 169, 103-11.
43. Hou, S.; Wang, J.; Wang, X.; Chen, H.; Xiang, L. Effect of Mg2+ on hydrothermal formation of α-CaSO4·0.5H2O whiskers with high aspect ratios. Langmuir 2014, 30, 9804-10.
44. Madejová, J.; Pálková, H.; Komadel, P. Behaviour of Li+ and Cu2+ in heated montmorillonite: evidence from far-, mid-, and near-IR regions. Vib. Spectrosc. 2006, 40, 80-8.
45. Meng, G.; Xu, J.; Cheng, R.; et al. Controllable synthesis and characterization of high purity calcium carbonate whisker-like fibers by electrochemical cathodic reduction method. J. Clean. Prod. 2022, 342, 130923.
46. Lin, R.; Zhang, J.; Bai, Y. Mass transfer of reactive crystallization in synthesizing calcite nanocrystal. Chem. Eng. Sci. 2006, 61, 7019-28.
47. Li, Z.; Wang, C.; Chen, J. Supply and demand of lithium in China based on dynamic material flow analysis. Renew. Sustain. Energy. Rev. 2024, 203, 114786.
48. Jo, Y.; Jeong, W.; Park, J.; Baek, K. Partially calcined CaCO3 for remediating multi-heavy metals-contaminated groundwater. Chem. Eng. J. 2023, 471, 144652.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].