Enhanced electrocatalytic Activity of NiCoP/Ni5P4@NiFe-LDH catalysts via Ir doping and oxygen vacancy engineering
Abstract
Alkaline water electrolysis offers a promising route for large-scale hydrogen production, but its efficiency is limited by the sluggish kinetics of both the hydrogen evolution
Keywords
INTRODUCTION
Driven by the twin pressures of global energy scarcity and environmental degradation, the development of clean, renewable energy technologies has emerged as a critical imperative[1,2]. Among the promising candidates, hydrogen stands out as a carbon-neutral energy carrier with exceptionally high energy density[3,4]. Electrocatalytic water splitting offers a sustainable and efficient pathway for hydrogen production that can be seamlessly integrated with renewable electricity sources[5]. While noble-metal catalysts - particularly Pt for the hydrogen evolution reaction (HER) and RuO2/IrO2 for the oxygen evolution reaction (OER) - demonstrate benchmark performance, their prohibitive cost and scarcity severely constrain widespread commercial deployment[6]. These challenges underscore the urgent need to develop earth-abundant, cost-effective electrocatalysts that combine high catalytic activity with robust long-term stability[7,8].
Transition metal phosphides (TMPs) have attracted considerable interest as electrocatalysts owing to their high activity, stability, and low cost[9]. Filled d-orbitals flexible redox behavior and high electron-transfer capability, lowering activation barriers and thereby enhancing hydrogen production efficiency[10]. Numerous synthetic routes have been reported, including direct reactions between red phosphorus and transition metals, electrolysis in molten metal salts, reactions of metal halides with PH3 or Na3P, decomposition of organometallic precursors, and reduction of metal phosphates[11-15]. For example, Liu et al. prepared the NiCoP/CoxP catalyst on nickel foam (NF) via electrodeposition followed by chemical vapor deposition (CVD), achieving an HER overpotential of 290 mV at 10 mA cm-2 and operating stably for 500 h at 10 mA cm-2 in seawater with excellent corrosion resistance[16]. Wu et al. produced a core-shell CoPx@FeOOH/NF catalyst using a three-step hydrothermal-phosphidation-electrodeposition method, which necessitated 190 and 248 mV to achieve 100 and 500 mA cm-2, respectively, while demonstrating exceptional durability for over 80 h in 1.0 M KOH seawater[17]. Despite these advances, pristine TMPs suffer from two critical limitations that hinder their practical application: (i) severe surface oxidation and phosphorus leaching under OER conditions, leading to gradual deactivation, and (ii) insufficient active site density and poor mass transport due to inevitable nanoparticle agglomeration. To overcome these challenges, constructing hierarchical composite architectures by integrating TMPs with layered double hydroxides (LDHs) has emerged as a promising strategy. LDHs play multifunctional roles: their 2D layered structure serves as a physical barrier against phosphorus leaching, tunable interlayer spacing facilitates ion diffusion, and abundant surface hydroxyl groups provide additional active sites for OER via a lattice-oxygen-mediated mechanism. Recent studies have demonstrated that strategic compositional engineering of LDH-based composites can significantly enhance OER performance[18]. For instance, Li et al. reported that atomically dispersed Ru doped into iron-nickel LDH forms an asymmetric Ru-Fe dipole that creates electron-deficient active sites, substantially accelerating OER kinetics and achieving an overpotential of only 230 mV at
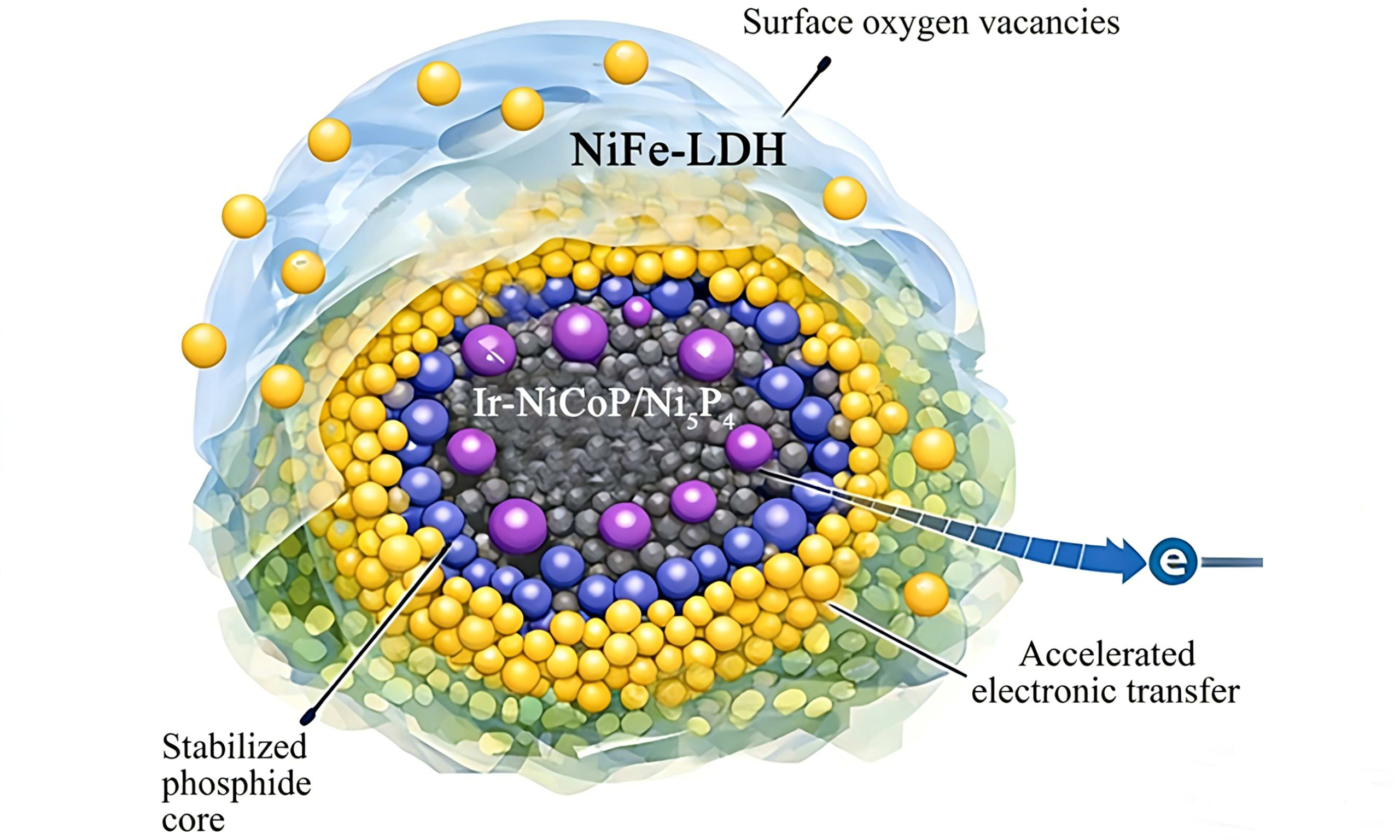
In this work, core-shell structured NiCoP/Ni5P4 composites were subsequently coated with NiFe-LDH. Ir incorporation and NaBH4-induced oxygen vacancy formation were further introduced to modulate the electronic structure and increase the density of exposed active sites. The material exhibited outstanding HER and OER performance in 1 M KOH solution, as well as excellent ammonia oxidation reaction (AOR) performance in 1 M KOH + NH3·H2O solution, demonstrating remarkable advantages including low overpotentials, low Tafel slopes, large electrochemical capacitance, and exceptional long-term cycling stability. Furthermore, it displayed superior electrocatalytic performance for efficient overall water splitting, achieving excellent long-term durability.
EXPERIMENTAL
Synthesis of the samples
All chemicals used in this experiment were purchased from Aladdin Reagent (Shanghai) Co., Ltd., with a purity of ≥ 96% and were used directly without further purification. They include nickel(II) nitrate hexahydrate [Ni(NO3·6H2O], cobalt(II) nitrate hexahydrate [Co(NO3)2·6H2O], iron(III) nitrate nonahydrate [Fe(NO3)·9H2O], urea, ammonium fluoride (NH4F), sodium hypophosphite monohydrate (NaH2PO2·H2O), iridium(III) chloride (IrCl3), sodium hydroxide (NaOH), sodium borohydride (NaBH4), and hydrochloric acid (HCl). All reagents met the required purity standards, ensuring reaction stability and reproducibility. NF (4 cm × 4 cm) was sonicated sequentially in hydrochloric acid and ultrapure water (30 min each) to remove surface oxides. A precursor solution was prepared by dissolving Ni(NO3)2·6H2O (1 mM), Co(NO3)2·6H2O (1 mM), urea (6 mM), and NH4F (2.5 mM) in 60 mL of deionized water. After 30 min of stirring, the solution and NF were transferred to a 100 mL stainless steel autoclave and hydrothermally treated at 120 °C for 6 h. The product was dried at 60 °C for 6 h. For phosphidation, the dried sample and NaH2PO2·H2O (5 g) were placed downstream and upstream, respectively, in a tube furnace and annealed at 350 °C for 2 h under Ar flow.
An aqueous precursor was prepared by dissolving Ni(NO3)2·6H2O (1.5 mM), Fe(NO3)3·9H2O (0.5 mM), urea (5 mM), and NH4F (5 mM) in 60 mL of deionized water. The solution and the substrate obtained in step 1 were transferred to a 100 mL stainless steel autoclave and hydrothermally treated at 120 °C for 4 h, then allowed to cool and dried to yield the product. The procedure was repeated with the Fe(NO3)3·9H2O concentration increased to 1.0 mM while keeping the other reagent concentrations unchanged, producing a second sample under identical conditions. Finally, the Fe(NO3)3·9H2O concentration was increased to
Six milligrams of IrCl3 were dissolved in 60 mL of 0.01 M NaOH and stirred for 20 min. The resulting solution and two samples prepared with 1.0 mM Fe(NO3)3·9H2O were transferred to a 100 mL stainless steel autoclave and hydrothermally treated at 120 °C for 2 h. The product was then washed and dried. The as-prepared material was subsequently immersed in 100 mL of 1 mM NaBH4 for 15 min, removed, thoroughly rinsed, and dried. Based on the Fe(NO3)3·9H2O content, the samples were designated as NiCoP/Ni5P4, NiCoP/Ni5P4@NiFe-LDH-0.5, NiCoP/Ni5P4@NiFe-LDH-1, NiCoP/Ni5P4@NiFe-LDH-1.5, Ir-NiCoP/Ni5P4@NiFe-LDH-1, and Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV.
Structure characterization
Morphology and crystal structure were characterized by X-ray diffraction (XRD, Shimadzu XRD-7000, Cu Kα), X-ray photoelectron spectroscopy (XPS, Thermo ESCALAB 250, Al Kα), scanning electron microscopy (SEM, Zeiss Gemini 300-71-31), and transmission electron microscopy (TEM, JEOL JEM-2100 Plus).
RESULTS AND DISCUSSION
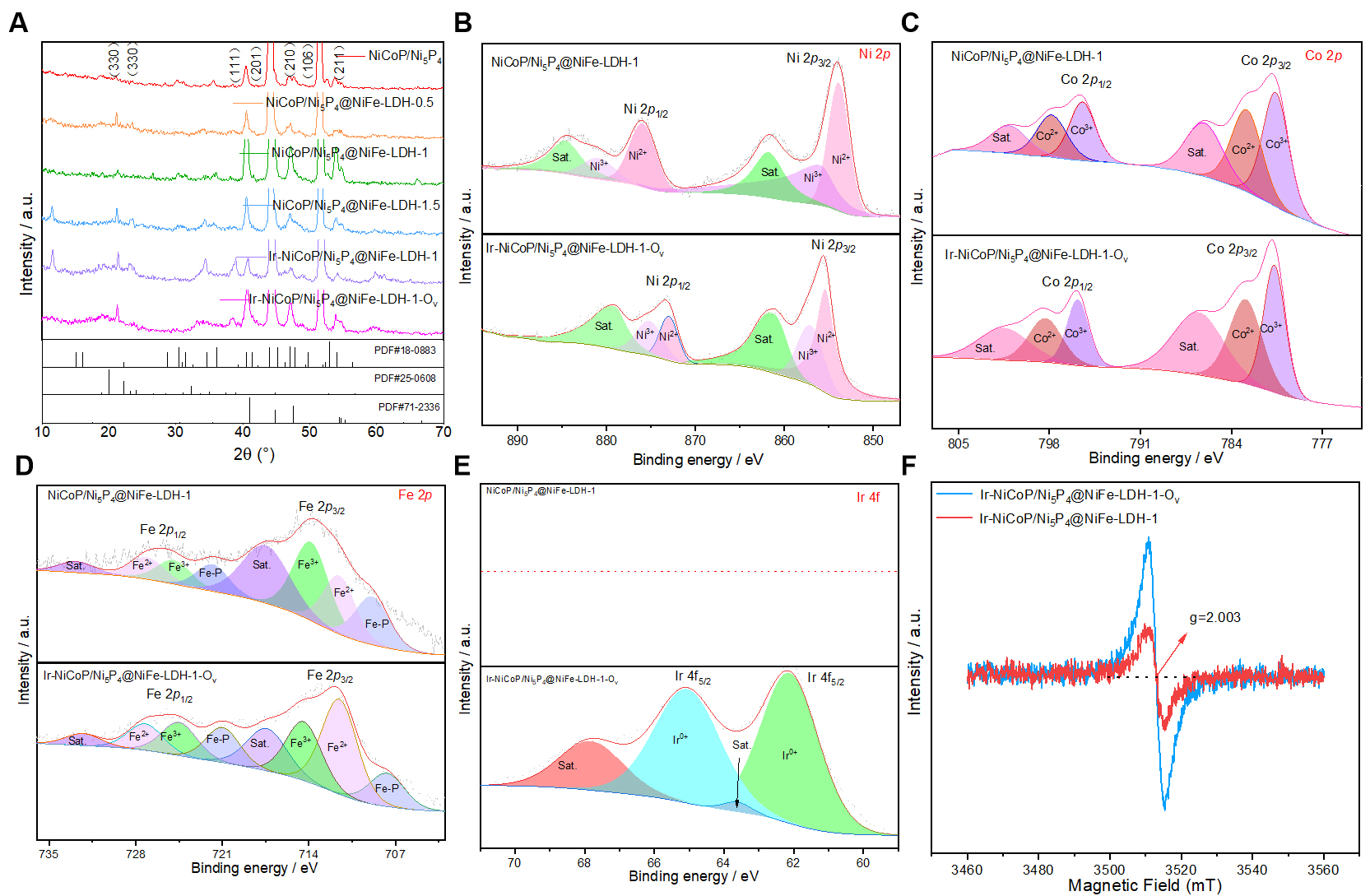
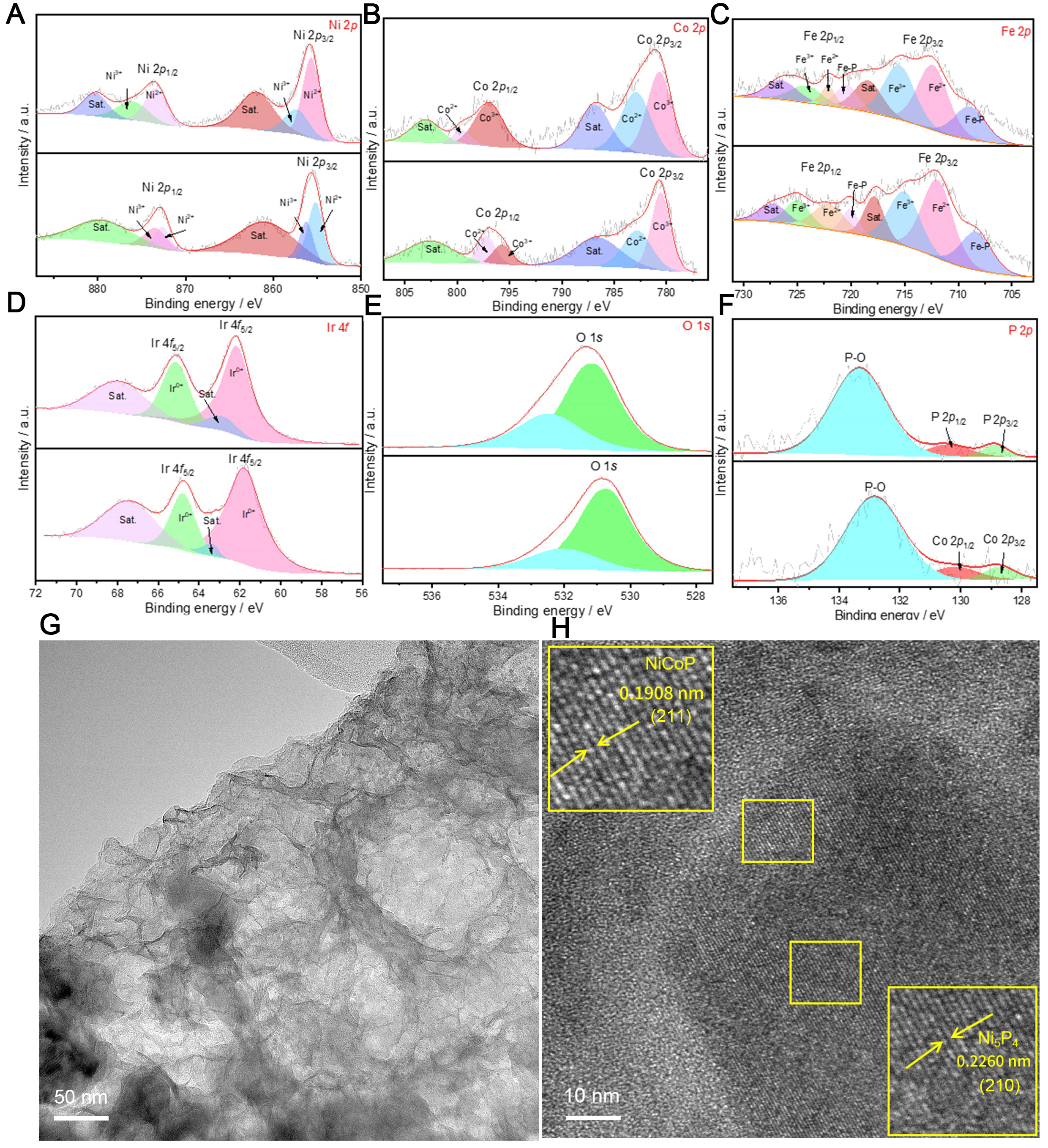
Figure 1A presents the XRD pattern of the synthesized material. The characteristic peaks observed at 2θ values of 40.78°, 44.72°, 47.66°, and 55.29° are attributed to the (111), (201), (210), and (211) planes of hexagonal NiCoP/Ni5P4, respectively (JCPDS PDF No.71-2336). Additionally, the peaks at 2θ = 31.55°, 41.49°, 49.34°, and 52.26° correspond well to the (201), (211), (302), and (106) planes (JCPDS PDF No.18-0336). The peaks at 2θ = 19.98° and 22.21° are consistent with the (330) and (332) planes (JCPDS PDF No. 25-0608), respectively[21,22]. XPS was further performed to investigate the composition and elemental states of NiCoP/Ni5P4@NiFe-LDH-1 and Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV. Supplementary Figure 1 presents a comparison of the survey spectra for both materials, which clearly reveals the characteristic peaks of Ir, confirming the successful incorporation of Ir into the material. Supplementary Figure 2 shows the elemental composition of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV, revealing the presence of Ni, Co, P, Ir, O, and C as the main elements, with contents of 11.47%, 3.03%, 3.2%, 3.12%, 47.77%, and 25.81%, respectively. Figure 1B-E illustrates the XPS spectra of the NiCoP/Ni5P4@NiFe-LDH-1 and Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV samples. Characterization of the NiCoP/Ni5P4@NiFe-LDH-1 material was shown, revealing that the Ni 2p spectrum consists of peaks at approximately 856.3 and 873.5 eV, corresponding to the Ni2+ 2p3/2 and 2p1/2 states, respectively [Figure 1B]. The peaks for Ni3+ 2p3/2 and 2p1/2 appear at binding energies of 858.2 and 875.5 eV, respectively, accompanied by two satellite peaks centered at 861.8 and 880 eV[23]. Furthermore, Figure 1C shows the Co 2p spectrum, where peaks at 782.98 and 797.88 eV are associated with Co2+ 2p3/2 and 2p1/2, while those at 780.1 and 795.78 eV correspond to Co3+ 2p3/2and 2p1/2. The peaks at 786.1 and 803.2 eV are indexed as satellite peaks[24]. In Figure 1D, two characteristic peaks at 714.82 and 725.84 eV, with a spin energy separation of 13.02 eV, are ascribed to Fe 2p3/2 and Fe 2p1/2, characteristic of Fe3+. The peaks at binding energies of 717.16 and 728.30 eV are satellite peaks of Fe 2p3/2 and Fe 2p1/2, while those at 712.30 and
Figure 1. Structural characterization of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV. (A) XRD. (B) XPS of Ni 2p; (C) Co 2p; (D) Fe 2p; (E) Ir 4f; (F) EPR of the samples. EPR: Electron paramagnetic resonance; XRD: X-ray diffraction; LDH: layered double hydroxide; XPS: X-ray photoelectron spectroscopy.
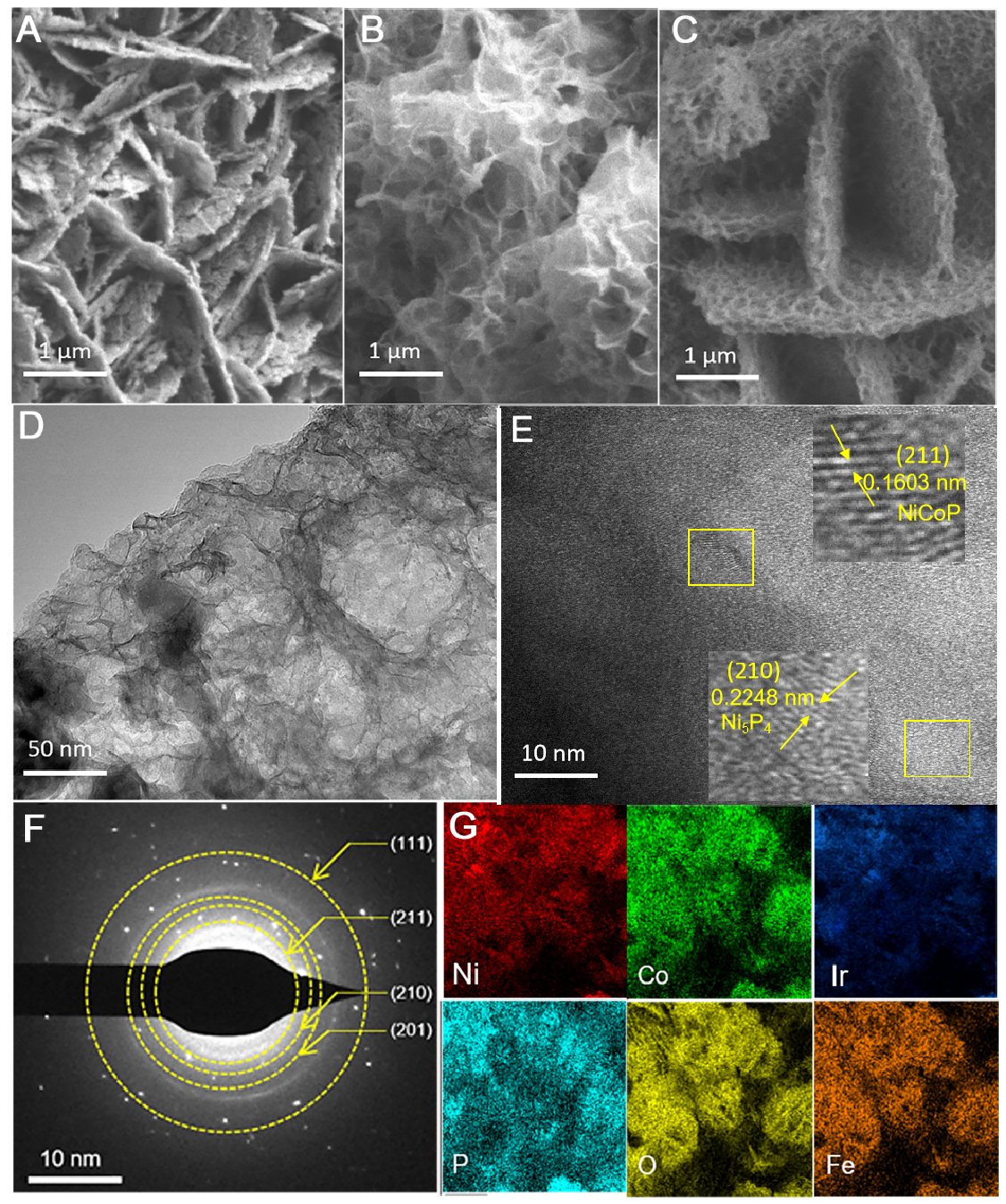
SEM was utilized to clarify the microstructural properties of the materials. Figure 2A illustrates that the SEM images of NiCoP/Ni5P4@NiFe-LDH-1 exhibit a sheet-like shape on the material’s surface. Following Ir incorporation, the modified NiCoP/Ni5P4@NiFe-LDH-1 displays a unique porous structure, as demonstrated by the SEM images in Figure 2B. This porous structure is anticipated to reveal numerous active sites during catalytic reactions, therefore improving catalytic efficacy. SEM images of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV [Figure 2C] demonstrate that NaBH4 treatment causes a significant morphological alteration, leading to a hierarchical spheroidal structure formed by interconnected nanosheets. This shape augments the density of available active sites, hence improving catalytic performance while maintaining intrinsic activity throughout extended cycling. The microstructural characteristics support the enhanced catalytic efficacy of
Figure 2. Morphology of the prepared samples. (A) SEM image of NiCoP/Ni5P4@NiFe-LDH-1; (B) SEM image of Ir-NiCoP/Ni5P4@NiFe-LDH-1; (C) SEM image of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV; (D) TEM image of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV; (E) HRTEM image; (F) SAED pattern of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV; (G) Elemental mapping of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV. SEM: Scanning electron microscopy; LDH: layered double hydroxide; TEM: transmission electron microscopy; HRTEM: high-resolution TEM; SAED: selected area electron diffraction.
Supplementary Figure 4, Figure 2D and E show a low-magnification TEM image and high-resolution TEM (HRTEM) images of the as-prepared NiCoP/Ni5P4@NiFe-LDH-1 and Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV. For NiCoP/Ni5P4@NiFe-LDH-1 [Supplementary Figure 4], lattice spacings of 0.2268 nm and 0.1658 nm are indexed to the Ni5P4 (210) and NiCoP (211) planes, respectively, indicating the presence of both phases and high sample purity[30]. In the Ir-doped, NaBH4-treated sample [Figure 2D and E], the corresponding spacings are 0.2248 nm for Ni5P4 (210) and 0.1603 nm for NiCoP (211), representing changes of 0.0020 and
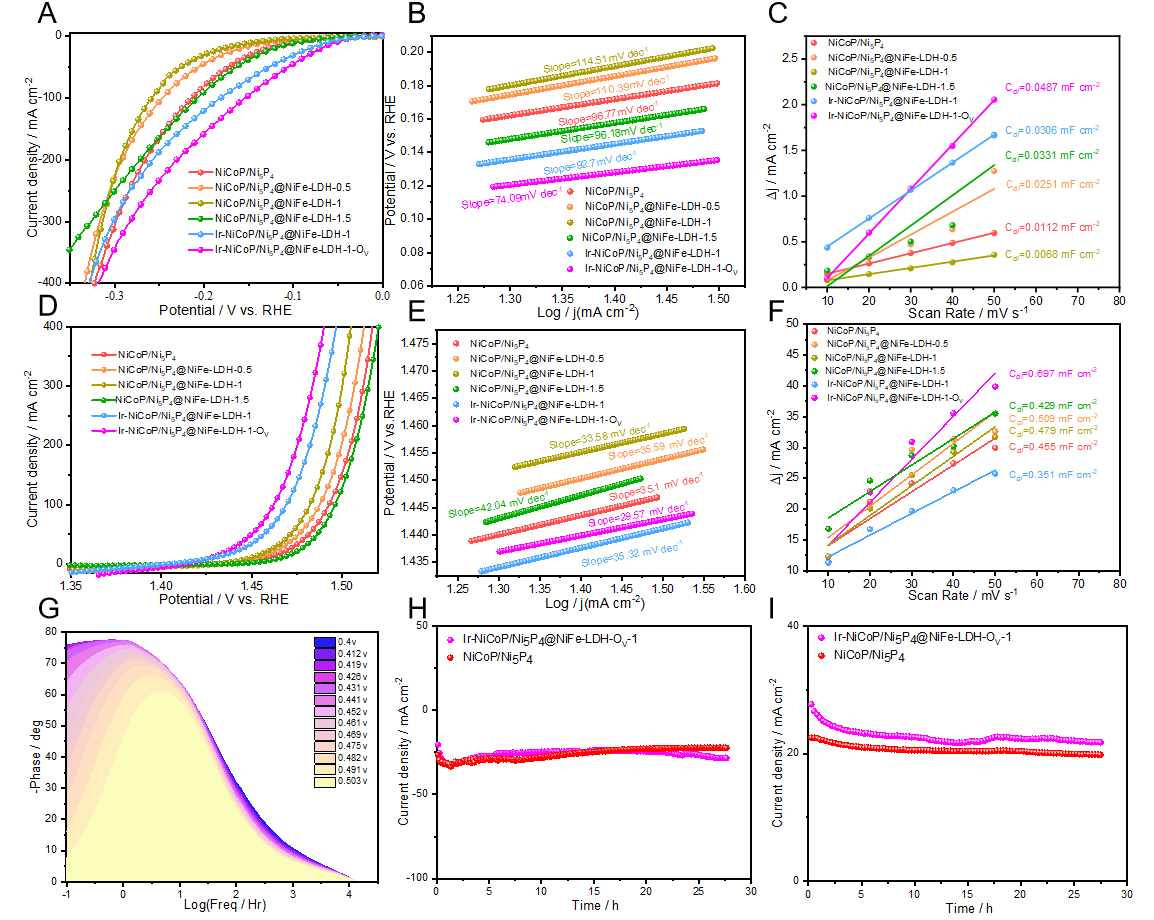
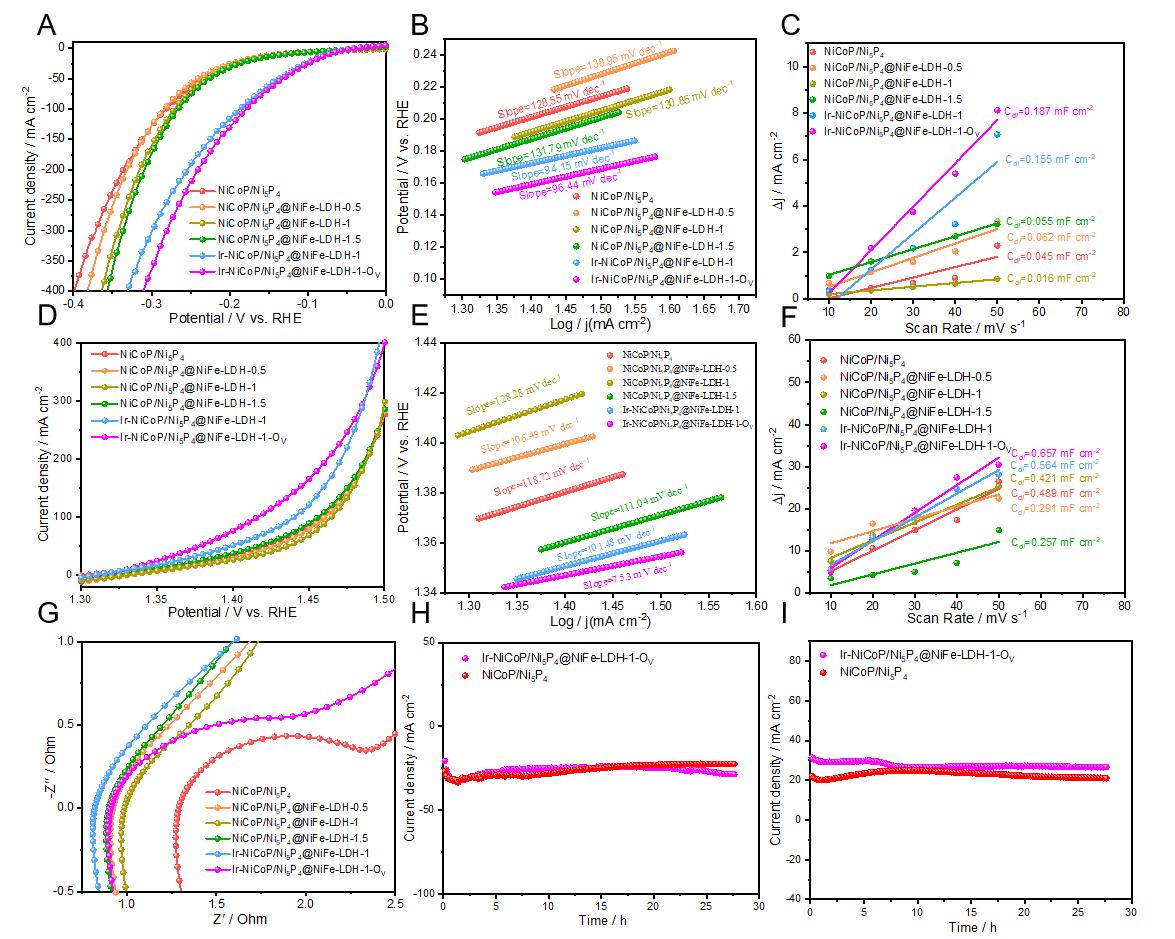
The electrocatalytic performance of all synthesized samples was evaluated in a three-electrode configuration using an alkaline electrolyte (1 M KOH). Figure 3A presents the linear sweep voltammetry (LSV) curves for HER at a scan rate of 5 mV s-1. The results demonstrate that Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV samples exhibit a significantly lower overpotential compared to all other catalysts, indicating superior HER activity. A systematic comparison of overpotentials at the various current densities (-10, -50, and -100 mA cm-2) is presented in Supplementary Figure 5. Among all catalysts, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV exhibits superior performance with the lowest overpotentials of 52.7, 106.7, and 154.7 mV, respectively. This represents a notable enhancement compared to Ir-NiCoP/Ni5P4@NiFe-LDH-1 (62.7, 127.7, and 182.7 mV) and substantially surpasses all reference materials. The marked improvement following NaBH4 reduction validates the efficacy of oxygen vacancy introduction as a strategy for enhancing electrocatalytic performance[31]. Tafel slopes were extracted from the LSV curves [Figure 3B]. The values are 96.77, 110.39, 96.18, 92.70, and 74.09 mV dec-1. Among these, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV shows the smallest slope, indicating faster HER kinetics[32]. Double-layer capacitances (Cdl) were extracted from the linear dependence of capacitive current on scan rate [Figure 3C], yielding values of 0.0112, 0.0251, 0.0068, 0.0331, 0.0306, and 0.0487 mF cm-2 for the above catalysts. Notably, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV exhibits the highest Cdl, indicating a substantially enlarged electrochemically active surface area (ECSA). This enhanced ECSA is attributed to the synergistic effects of Ir doping and oxygen vacancy engineering via NaBH4 treatment, which collectively increase both the density and accessibility of catalytically active sites for HER.
Figure 3. HER and OER performance of the samples in 1 M KOH. (A) LSV curves in HER; (B) Tafel plots in HER; (C) Cdl value; (D) LSV curves in OER; (E) Tafel plots in OER; (F) Cdl value; (G) Bode diagram; (H) Chronoamperometric stability tests (HER); (I) Chronoamperometric stability tests (OER). LSV: Linear sweep voltammetry.
The OER activity of the as-synthesized materials was evaluated in a three-electrode electrochemical cell. The electrocatalytic performance was investigated in 1 M KOH electrolyte at a scan rate of 2 mV s-1. Figure 3D presents LSV curves of the materials. Among all the materials examined, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV exhibited the lowest overpotential, demonstrating superior OER activity compared to the other catalysts. To provide a more intuitive comparison, Supplementary Figure 6 displays the overpotentials required to achieve current densities of 10, 50, and 100 mA cm-2. Among all tested materials, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV exhibited the lowest overpotentials of 197.3, 220.3, and 232.3 mV at 10, 50, and 100 mA cm-2, respectively. These values are substantially lower than those of the other five materials, confirming its superior OER catalytic performance. Tafel analysis [Figure 3E] yields slopes of 35.1, 35.59, 33.58, 42.04, 35.32, and
To elucidate the surface evolution during catalysis, XPS spectra were acquired for Ir-NiCoP/Ni5P4@NiFe-LDH-OV-1 before and after prolonged OER operation in an alkaline electrolyte. The spectral changes are consistent with OER-induced surface reconstruction, revealing substantial alterations in the near-surface chemical environment and the in situ generation of catalytically active sites that are absent in the as-prepared material[33,34]. As shown in Figure 4A, NaBH4 pretreatment has a negligible effect on the intrinsic electronic states: the binding energies of the constituent elements remain essentially unchanged, indicating that the composition and stability are preserved. Following OER cycling, significant binding energy shifts are observed across multiple metal centers. The Ni3+ 2p1/2 peak shifts by 2.1 eV toward lower binding energy[34], while the Co2+ 2p1/2 peak exhibits a more pronounced 2.8 eV negative shift [Figure 4B]. In contrast, Fe displays negligible variation [Figure 4C], suggesting relative chemical stability under OER conditions[35]. Notably, the Ir 4f7/2 component undergoes a 0.30 eV downshift [Figure 4D]. These systematic negative binding energy shifts for Ni, Co, and Ir provide compelling evidence of extensive surface reconstruction accompanied by charge redistribution during electrochemical activation[36]. The distinct electronic perturbations, particularly involving Ir, underscore its pivotal role in modulating the local electronic structure and thereby enhancing the electrode’s electrocatalytic performance. As shown in Figure 4E and F, the O 1s and P 2p spectra exhibit no discernible changes, indicating that the oxygen- and phosphorus-related chemical environments remain largely unchanged during the process. The binding energies of metal centers (Ni, Co, Ir) exhibit pronounced negative shifts, whereas the O 1s and P 2p peak positions remain virtually unchanged. The negative shift of metal 2p peaks is attributed to the decreased oxidation states of metal centers during the OER process. The invariant O 1s peak position arises from the dynamic equilibrium established among lattice oxygen, surface hydroxyl groups, and adsorbed oxygen species. More significantly, the stability of P 2p peaks substantiates the effective protective role of the outer NiFe-LDH layer in shielding the phosphide core from electrochemical oxidation. From the TEM images, it is clear that after electrochemical cycling [Figure 4G], the Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV sample retains its nanosheet morphology, indicating high structural stability of the catalyst. As shown in the HRTEM images [Figure 4H], lattice fringes with spacings of 0.1908 and 0.2260 nm can be indexed to the (211) plane of NiCoP and the (210) plane of Ni5P4, respectively, revealing a pronounced increase in lattice spacing after cycling.
Figure 4. Structural characterization of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV before and after electrochemical cycling. (A) XPS of Ni 2p; (B) Co 2p; (C) Fe 2p; (D) Ir 4f; (E) O 1s; (F) P 2p; (G) TEM image of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV after electrochemical cycling; (H) HRTEM image. LDH: Layered double hydroxide; XPS: X-ray photoelectron spectroscopy; TEM: transmission electron microscopy; HRTEM: high-resolution TEM.
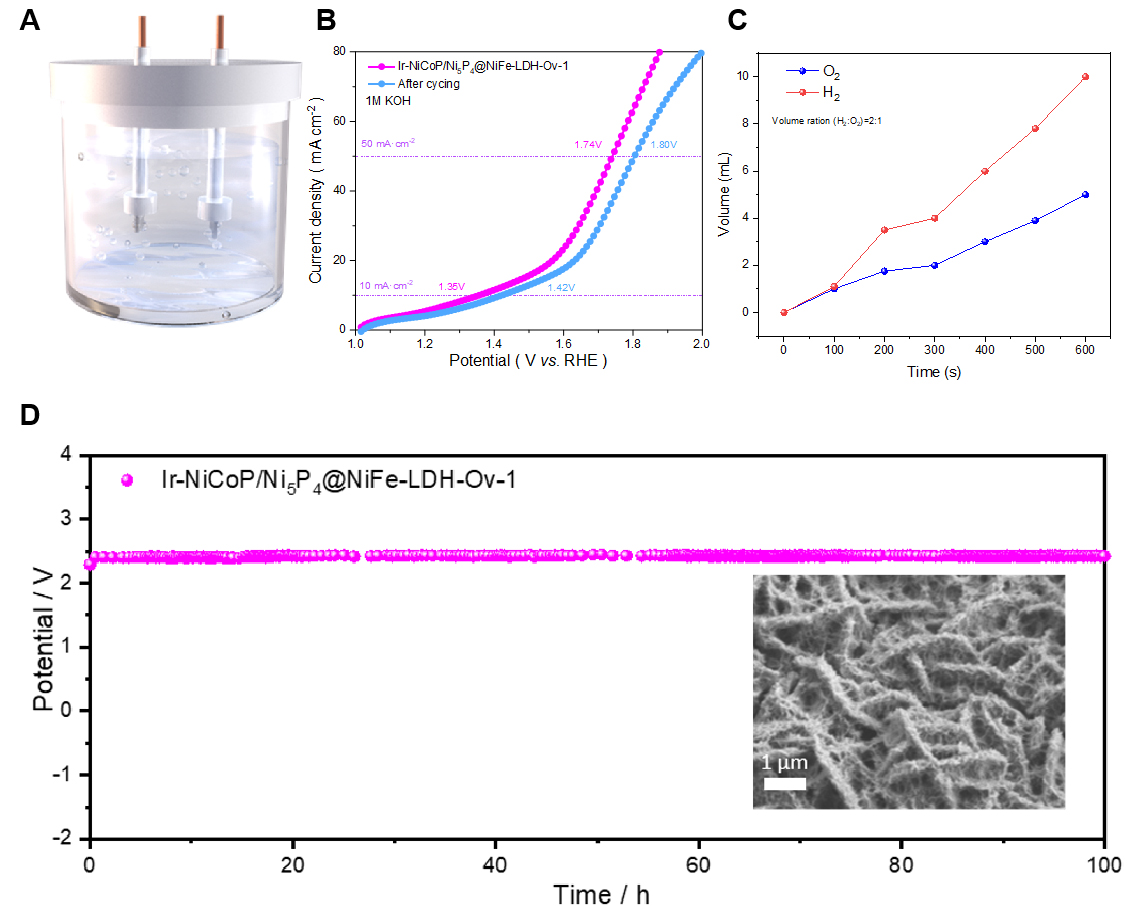
To evaluate the performance of the synthesized catalyst, a two-electrode overall water splitting (OWS) cell was assembled, using the as-prepared material as both anode and cathode [Figure 5A]. The LSV curves [Figure 5B] show that current densities of 10 and 50 mA cm-2 are achieved at cell voltages of 1.35 and 1.74 V, respectively. After cycling, the required voltages increased only slightly to 1.42 and 1.80 V, indicating minimal performance decay. The overall cell architecture remains intact and the OWS activity is largely preserved. Faradaic efficiency (FE) was quantified by collecting H2 and O2 generated during electrolysis in a symmetric Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV. Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV cell using the water-displacement method. The gas volume-time plots [Figure 5C] show an H2:O2 volume ratio of ~2:1, in excellent agreement with water splitting stoichiometry, indicating an FE approaching 100%. Continuous electrolysis for 100 h further demonstrates the durability of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV [Figure 5D]. The post-cycling SEM inset reveals slight surface roughening, consistent with reconstruction during long-term electrolysis.
Figure 5. Overall water splitting performance of samples and Faraday efficiency test. (A) Schematic of the overall water splitting device; (B) LSV curves; (C) Faradaic efficiency curve; (D) Chronoamperometric stability tests. LSV: Linear sweep voltammetry; LDH: layered double hydroxide.
To explore this potential, the performance of Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV was evaluated in an ammoniacal alkaline electrolyte (1 M KOH + NH3·H2O). This medium, which combines KOH with aqueous ammonia, provides a favorable environment for AOR while maintaining high ionic conductivity. Regarding selectivity, under the operating conditions employed in this work, ammonia molecules are preferentially oxidized at the catalyst surface in the alkaline ammonia solution, making AOR the dominant anodic reaction rather than OER. Furthermore, the theoretical potential of AOR is only -0.06 V vs. RHE (under alkaline conditions), which is significantly lower than that of OER (1.23 V vs. RHE), thereby substantially reducing the overall cell voltage and improving energy efficiency[36,37]. Electrochemical testing in this electrolyte enables a comprehensive assessment of catalytic activity, stability, and selectivity, generating essential data to guide optimization of ammonia-enabled hydrogen production. In summary, the combination of low thermodynamic energy demand, environmental compatibility, and high hydrogen-storage capacity makes ammonia electrolysis a compelling research direction. With continued advances in catalyst design and electrolyte engineering, ammonia electrolysis could assume a pivotal role in the hydrogen value chain, accelerating the hydrogen economy and offering practical pathways to address hydrogen storage and transport.
For HER evaluation, Figure 6A presents the LSV curves of all materials. The catalysts (Ir-NiCoP/Ni5P4@NiFe-LDH-1 and Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV) exhibit significantly lower overpotentials compared to the other materials. Among these, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV demonstrates superior performance compared with Ir-NiCoP/Ni5P4@NiFe-LDH-1, consistent with results obtained from alkaline water electrolysis, further confirming the beneficial effect of NaBH4 treatment on enhancing electrocatalytic activity. Supplementary Figure 9 provides a quantitative comparison of overpotentials at current densities of -10, -50, and -100 mA cm-2. The baseline materials show relatively high overpotentials. In contrast, Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV achieves remarkably low overpotentials of 72.7, 132.7, and 188.7 mV at -10, -50, and -100 mA cm-2, respectively. These values represent further improvements compared to Ir-NiCoP/Ni5P4@NiFe-LDH-1 (76.7, 138.7, and 188.7 mV), unambiguously demonstrating that NaBH4 treatment substantially enhances the catalytic performance. To further probe reaction kinetics, Tafel slopes were derived from Tafel plots constructed from the LSV data [Figure 6B]. Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV shows a Tafel slope of 96.44 mV dec-1, smaller than those of NiCoP/Ni5P4 (128.55 mV dec-1), NiCoP/Ni5P4@NiFe-LDH-0.5 (138.95 mV dec-1), NiCoP/Ni5P4@NiFe-LDH-1
Figure 6. HER and AOR performance of the samples in 1 M KOH + NH3·H2O. (A) LSV curves, (B) Tafel plots, and (C) Cdl value of HER; (D) LSV curves, (E) Tafel plots, (F) Cdl value, and (G) EIS curves of AOR; (H) Chronoamperometric stability tests of HER; (I) Chronoamperometric stability tests of AOR. HER: Hydrogen evolution reaction; AOR: ammonia oxidation reaction; LSV: linear sweep voltammetry; EIS: electrochemical impedance spectroscopy.
Further analysis shows that Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV exhibits excellent activity for water electrolysis. Its engineered architecture and optimally distributed active sites accelerate charge-transfer kinetics while lowering the overpotential. Consequently, water splitting proceeds efficiently with reduced energy input, and the catalyst maintains stability during prolonged operation. Together, these results establish Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV as a high-performance water-electrolysis catalyst with broad application potential. The combination of high energy conversion efficiency and durability provides a robust technological basis for hydrogen production by water splitting in the clean energy sector. This work not only provides an experimental foundation for developing efficient, low-cost electrocatalysts, but also suggests new avenues for the sustainable growth of the hydrogen energy value chain.
CONCLUSION
In summary, we successfully prepared Ir-doped NiCoP/Ni5P4@NiFe-LDH composite materials through a facile two-step hydrothermal method. Following NaBH4 treatment, the Ir-NiCoP/Ni5P4@NiFe-LDH-1-OV material achieved 10 mA cm-2 at an overpotential of 52.7 mV for HER in 1 M KOH electrolyte. The catalyst retained its activity after 28 h of continuous cycling, indicating robust durability. In the two-electrode electrolyzer configuration, the catalyst achieved a remarkably low cell voltage for overall water splitting and maintained stable operation for 100 h without performance degradation. Furthermore, in NH3·H2O + 1 M KOH electrolyte, the catalyst exhibited an exceptionally low overpotential of only 72.7 mV at a current density of 10 mA cm-2. This work provides valuable insights into the rational design of advanced multifunctional electrocatalysts.
DECLARATIONS
Authors’ contributions
Conceptualization, data curation, writing - original draft: Zhao, Z.; Sui, L.
Data curation, figure design: Wang, Z.; Song, S.; Wang, J.; Li, Y.
Writing - review and editing, funding acquisition, supervision: Zhao, D.; Li, G.; Miao, L.
Availability of data and materials
The raw data supporting the conclusions of this article will be made available by the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This project was supported by the College Student Innovation and Entrepreneurship Project (S202410164017).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Pu, T.; Ding, J.; Zhang, F.; et al. Dual atom catalysts for energy and environmental applications. Angew. Chem. Int. Ed. 2023, 62, e202305964.
2. Cao, P.; Liu, Y.; Zhang, Q.; Guo, C.; Li, J.; Zeng, Q. MXene/MOF composites: synergistic platforms for advanced Energy, catalysis, and environmental applications. Coord. Chem. Rev. 2025, 545, 217018.
3. Zhou, H.; Xu, S.; Zhang, B.; et al. In situ reconstructed amorphous MOOH-enhanced NiCoP@NiFe-LDH bifunctional electrocatalyst for long durable seawater electrolysis. Inorg. Chem. Front. 2026, 13, 666-79.
4. Johnson, N.; Liebreich, M.; Kammen, D. M.; Ekins, P.; Mckenna, R.; Staffell, I. Realistic roles for hydrogen in the future energy transition. Nat. Rev. Clean. Technol. 2025, 1, 351-71.
5. Zhang, L.; Qi, F.; Ren, R.; et al. Recent advances in green hydrogen production by electrolyzing water with anion-exchange membrane. Research 2025, 8, 0677.
6. Mao, J.; Liang, J.; Li, Y.; et al. Electrochemical lithiation regulates the active hydrogen supply on Ru-Sn nanowires for hydrogen evolution toward the high-performing anion exchange membrane water electrolyzer. J. Am. Chem. Soc. 2025, 147, 7711-20.
7. Zhang, X.; Cao, C.; Ling, T.; Ye, C.; Lu, J.; Shan, J. Developing practical catalysts for high‐current‐density water electrolysis. Adv. Energy. Mater. 2024, 14, 2402633.
8. Gao, X.; Chen, Y.; Wang, Y.; et al. Next-generation green hydrogen: progress and perspective from electricity, catalyst to electrolyte in electrocatalytic water splitting. Nano. Micro. Lett. 2024, 16, 237.
9. Cao, L.; Zhang, J.; Ding, L.; Du, Z.; He, C. Metal-organic frameworks derived transition metal phosphides for electrocatalytic water splitting. J. Energy. Chem. 2022, 68, 494-520.
10. Geng, P.; Ji, B.; Guo, Y.; Xiao, C.; Wan, J.; Li, C. M. MOF-derived transition metal phosphides for rechargeable batteries. Chem. Eng. J. 2025, 516, 164033.
11. Zhou, Z.; Shi, X.; Yin, J.; Kong, W.; Sun, J.; Wen, Z. Symbiotic growth of cobalt phosphides facilitates multiphase synergic function on long-cycling stability. Chem. Phys. Lett. 2020, 749, 137403.
12. Gao, Z.; Gao, Q.; Liu, Z.; et al. High-efficiency hydrogen evolution reaction catalyzed by iron phosphide nanocrystals. RSC. Adv. 2016, 6, 114430-5.
13. Song, L.; Zhang, S. A versatile route to synthesizing bulk and supported nickel phosphides by thermal treatment of a mechanical mixing of nickel chloride and sodium hypophosphite. Powder. Technol. 2011, 208, 713-6.
14. Tang, D.; Li, T.; Li, C. M. Metal and phosphonium-based ionic liquid: a new Co and P dual-source for synthesis of cobalt phosphide toward hydrogen evolution reaction. Int. J. Hydrogen. Energy. 2019, 44, 1720-6.
15. Galindo-ortega, Y.; Infantes-molina, A.; Huirache-acuña, R.; et al. Active ruthenium phosphide as selective sulfur removal catalyst of gasoline model compounds. Fuel. Process. Technol. 2020, 208, 106507.
16. Liu, D.; Ai, H.; Chen, M.; et al. Multi-phase heterostructure of CoNiP/CoxP for enhanced hydrogen evolution under alkaline and seawater conditions by promoting H2O dissociation. Small 2021, 17, e2007557.
17. Wu, L.; Yu, L.; Mcelhenny, B.; et al. Rational design of core-shell-structured CoPx@FeOOH for efficient seawater electrolysis. Appl. Catal. B. Environ. 2021, 294, 120256.
18. Liu, Y.; Hu, H.; Wang, K.; Huang, J.; Wang, D. Advancing seawater electrolysis: NiFe‐LDH‐based electrocatalysts for the oxygen evolution reaction. Adv. Energy. Mater. 2025, 16, e04101.
19. Li, J.; Naren, G.; Tang, C.; et al. Asymmetric ruthenium-iron dipole enabling fast alkaline water splitting on ruthenium-doped iron-nickel layered double hydroxides. Mater. Rep. Energy. 2025, 5, 100359.
20. Abdelghafar, F.; Xu, X.; Guan, D.; et al. New nanocomposites derived from cation-nonstoichiometric Bax(Co, Fe, Zr, Y)O3-δ as efficient electrocatalysts for water oxidation in alkaline solution. ACS. Mater. Lett. 2024, 6, 2985-94.
21. Fan, M.; Cui, J.; Wu, J.; Vajtai, R.; Sun, D.; Ajayan, P. M. Improving the catalytic activity of carbon-supported single atom catalysts by polynary metal or heteroatom doping. Small 2020, 16, e1906782.
22. Wang, S.; Chen, Z.; Sun, L.; et al. Synergistic mechanism of disparate surface hydroxyls and oxygen vacancies towards peroxymonosulfate activation for durable water decontamination. Chem. Eng. J. 2025, 503, 158259.
23. Xue, N.; Lin, Z.; Li, P.; Diao, P.; Zhang, Q. Sulfur-doped CoSe2 porous nanosheets as efficient electrocatalysts for the hydrogen evolution reaction. ACS. Appl. Mater. Interfaces. 2020, 12, 28288-97.
24. Yang, Y.; Hao, J.; Lyu, C.; et al. Electron islands-induced interface engineering in FeP@NiCoP/Mo4P3 for efficient hydrogen evolution catalysis. Adv. Funct. Mater. 2025, 35, 2507225.
25. Sial, M. A. Z. G.; Liu, S.; Zou, J.; et al. NiCoFe alloy multishell hollow spheres with lattice distortion to trigger efficient hydrogen evolution in acidic medium. Sustain. Energy. Fuels. 2019, 3, 3310-7.
26. Li, X.; Wu, T.; Li, N.; et al. Vertically staggered porous Ni2P/Fe2P nanosheets with trace Ru doping as bifunctional electrocatalyst for alkaline seawater splitting. Adv. Funct. Mater. 2024, 34, 2400734.
27. Gao, L.; Yang, Z.; Sun, T.; et al. Autocatalytic surface reduction‐assisted synthesis of PtW ultrathin alloy nanowires for highly efficient hydrogen evolution reaction. Adv. Energy. Mater. 2022, 12, 2103943.
28. Su, P.; Huang, W.; Zhang, J.; et al. Fe atoms anchored on defective nitrogen doped hollow carbon spheres as efficient electrocatalysts for oxygen reduction reaction. Nano. Res. 2020, 14, 1069-77.
29. Ma, F.; Chen, H.; Wu, H.; et al. Phase transition during sintering of layered transition metal oxide sodium cathodes. Nano. Lett. 2025, 25, 9293-301.
30. Luo, J.; Deng, J.; Wang, H.; et al. High-performance FeCoNiMo bifunctional catalysts with hierarchical porosity for water splitting. Tungsten 2025, 8, 242-54.
31. Zhang, P.; Wu, Q.; Wang, H.; et al. Z-scheme heterojunction Zn3(OH)2(V2O7)(H2O)2/V-Zn(O,S) for enhanced visible-light photocatalytic N2 fixation via synergistic heterovalent vanadium states and oxygen vacancy defects. Chin. J. Catal. 2025, 74, 279-93.
32. Ma, W.; Zhang, Y.; Wang, B.; et al. Significantly enhanced OER and HER performance of NiCo-LDH and NiCoP under industrial water splitting conditions through Ru and Mn bimetallic co-doping strategy. Chem. Eng. J. 2024, 494, 153212.
33. Feng, J.; Wang, X.; Pan, H. In-situ reconstruction of catalyst in electrocatalysis. Adv. Mater. 2024, 36, e2411688.
34. Wang, M.; Tan, H.; Liu, X.; Yan, X.; Liang, J. Energy and resource generation via electrochemical ammonia oxidation: from materials design to system integration. Adv. Energy. Mater. 2025, 15, e03846.
35. Yu, T.; Tan, K.; Wu, J.; Zou, Y.; Yin, S. Bifunctional interstitial phosphorous doping strategy boosts platinum-zinc alloy for efficient ammonia oxidation reaction and hydrogen evolution reaction. Nano. Res. 2023, 17, 1182-9.
36. Huan, Y.; He, Y.; Liu, S.; et al. Electrochemical ammonia oxidation reaction on nickel‐based non‐noble metal electrocatalysts: from mechanistic understanding to practical applications. Adv. Energy. Mater. 2025, 15, e03815.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].