Application of ceramic membranes for the removal of micro- and nanoplastics from water and wastewater: a review
Abstract
Micro- and nanoplastics (MPs/NPs) have become a global environmental challenge due to their persistence and widespread occurrence in aquatic systems. Ceramic membranes, composed of inorganic materials, inherently avoid secondary microplastic release and exhibit superior hydrophilicity, leading to weaker interactions with MPs/NPs and improved resistance to flux decline compared to polymeric membranes. This review systematically summarizes the application of ceramic membranes for MPs/NPs removal, with a focus on performance across different pore size regimes, where rejection efficiencies can exceed 95% under optimized conditions. The fouling mechanisms associated with MPs/NPs are critically analyzed, and emerging antifouling strategies, including piezoelectric ceramic membranes and membrane-advanced oxidation process coupling, are highlighted, achieving up to 99% MPs removal efficiency. Finally, key challenges and future research directions are outlined to support the practical implementation of ceramic membranes in MPs/NPs removal.
Keywords
INTRODUCTION
Plastic pollution has become a global environmental issue that has attracted widespread attention[1,2]. With the extensive use and improper disposal of plastic products, a significant amount of plastic waste gradually decomposes into smaller particles, collectively known as microplastics (MPs). MPs are typically defined as plastic particles with a diameter less than 5 mm[3,4], and as plastics continue to degrade, they can form even smaller particles, known as nanoplastics (NPs), which typically have a size smaller than 1 μm[5,6]. The distribution of MPs and NPs has now permeated aquatic systems worldwide. These plastic particles not only accumulate in water bodies but also, due to their tiny size, can easily enter the food chain, affecting the growth and reproduction of aquatic organisms and, ultimately, posing severe risks to human health. Recent studies have reported the presence of MPs and NPs in human tissues, including the lungs, liver, and kidneys[7-9]. The persistence and bioaccumulation of MPs and NPs make them increasingly severe environmental pollutants, and their impact on ecosystems and human health requires urgent attention[10].
The removal of MPs/NPs can be broadly categorized into physical capture[11] and chemical degradation[12]. Compared with degradation strategies, capture-based approaches are generally more efficient and practically viable in real water treatment systems, particularly for large-scale applications[13]. Capture techniques include adsorption, coagulation-flocculation, magnetic separation, flotation, and membrane filtration. These methods aim to directly remove MPs/NPs from water without altering their chemical structure, thereby avoiding the formation of potentially more mobile and hazardous by-products[14]. Among these approaches, membrane filtration has attracted increasing attention due to its high removal efficiency, operational stability, and compatibility with existing water treatment infrastructure[15]. Membranes can be categorized into organic and inorganic types. Organic membranes, with advantages such as low cost, have played a significant role in removing MPs and NPs from water[16]. However, recent studies have found that organic membranes, after prolonged use and chemical cleaning, may release MPs and NPs, presenting a “dual role” of both mitigating pollution and potentially becoming a pollution source[17,18]. This issue requires systematic investigation.
In contrast, the rapid development of ceramic membranes, a type of inorganic membrane with rigid pores formed by the accumulation of ceramic particles, has proven to be a promising solution[19]. Due to their stable chemical structure, acid and alkali resistance, and resistance to chemical corrosion, ceramic membranes fundamentally avoid the release of MPs and NPs. Furthermore, ceramic membranes have a surface rich in hydroxyl groups, offering stronger hydrophilicity, and their pure water permeability is typically two to three times that of polymeric membranes[20,21]. Therefore, ceramic membranes provide higher separation efficiency and longer service life in treating MPs and NPs in water, significantly improving the reliability and sustainability of water treatment.
Given the importance of these advancements, this review summarizes the application of ceramic membranes for the removal of MPs and NPs from water [Figure 1]. The performance of ceramic membrane technologies is systematically evaluated, with particular emphasis on removal efficiency across different pore sizes and the associated operational challenges. In addition, the pollution and fouling mechanisms of MPs and NPs are comprehensively analyzed. Finally, recent advances in piezoelectric ceramic membranes and the integration of ceramic membranes with advanced oxidation processes are discussed as promising strategies for fouling mitigation.
SOURCES OF MPs AND NPs IN AQUATIC ENVIRONMENTS
MPs in aquatic environments mainly originate from two pathways: primary inputs and secondary generation[7,22]. Primary MPs refer to plastic particles that already exist at the micro- to millimeter scale during production or use. Their environmental sources mainly include the leakage of pre-production plastic pellets during manufacturing, transportation, and processing, as well as microplastic components intentionally added to certain products[23]. In contrast, secondary MPs are generated through the continuous fragmentation of larger plastic items during use and environmental exposure[24]. Major input pathways include microfibers released from synthetic textiles during washing and daily wear, tire wear particles generated by road traffic, weathering products of surface coatings such as building and marine paints and road markings, and the aging and fragmentation of plastic debris driven by ultraviolet (UV) radiation, thermal oxidation, and mechanical stress[25] [Figure 2].
NPs are generally considered to originate predominantly from the further fragmentation of MPs in the environment and represent a continuous size-down evolution of plastic particles. Recent studies have further demonstrated that wastewater treatment plants are not only important sinks and transport nodes for MPs but also significant secondary sources of NPs in aquatic systems[1,26]. During wastewater treatment, MPs present in the influent are exposed to strong shear forces generated by pumping, aeration, and mixing operations, which can promote further fragmentation and lead to the formation of NPs that are subsequently discharged with treated effluents[27]. In tertiary treatment, membrane filtration is widely applied and is generally capable of achieving high removal efficiencies for MPs and NPs from water and wastewater[28]. At present, polymeric membranes such as polyvinylidene fluoride (PVDF), polysulfone (PSF), and polyether sulfone (PES) remain widely used in wastewater treatment for microplastic removal. However, recent studies have noted that during long-term operation and exposure to typical pretreatment and cleaning conditions, these membranes may gradually undergo material aging, which can lead to the release of small polymeric fragments into treated water[18,29,30]. For instance, recent experimental evidence has shown that microplastic concentrations in permeate can even exceed those in feed water under certain conditions, resulting in negative removal efficiencies, indicating the occurrence of membrane-derived microplastic release[18]. This phenomenon has been attributed to multiple stress factors, including mechanical abrasion, hydraulic shear, and chemical oxidation during membrane cleaning, which can accelerate polymer degradation and fragment detachment. Although the reported concentrations vary across operating conditions, these observations suggest that polymeric membrane systems may contribute trace levels of secondary MPs under certain circumstances.
CERAMIC MEMBRANE TECHNOLOGY
Membrane separation is essentially a selective transport process governed by membrane materials, in which a physical barrier is constructed between two phases to regulate the migration behavior of different components. Within the current separation technology landscape, membrane materials can be categorized into various types according to their composition and physical form. Among them, ceramic membranes composed primarily of inorganic oxides occupy a distinct position in water treatment applications owing to their excellent structural stability and environmental adaptability[31]. In studies focusing on the removal of MPs/NPs from water, ceramic membranes are generally regarded as structurally robust physical separation units capable of long-term operation. Unlike polymeric membranes, ceramic membranes do not rely on flexible polymer chains, allowing them to maintain structural integrity under harsh conditions such as elevated temperatures, strong oxidative environments, or complex water matrices. This intrinsic stability fundamentally eliminates the risk of secondary release of MPs/NPs during filtration processes, rendering ceramic membranes particularly advantageous for treating waters containing particulate pollutants and emerging contaminants simultaneously.
Ceramic membranes used in water treatment typically adopt an asymmetric multilayer architecture, which generally consists of a porous support, one or more intermediate layers, and a thin selective separation layer [Figure 3A-D][19,32-34]. The porous support primarily provides mechanical strength and is characterized by relatively large pore sizes (1-20 μm) and high porosity (30%-60%), thereby enhancing permeability and reducing hydraulic resistance. The intermediate layer is positioned between the porous support and the selective layer, serving to prevent penetration of fine particles into the support pores during membrane fabrication and operation, which would otherwise result in internal pore blockage and flux loss[15]. Owing to the presence of the intermediate layer, the support can be designed with larger pores while the selective layer can be made thinner, leading to a lower overall transport resistance and higher permeance. Depending on design requirements, the intermediate layer may consist of one or multiple sublayers with gradually decreasing pore sizes to ensure structural compatibility with the selective layer. Typically, the pore size of intermediate layers ranges from 0.05 to 0.5 μm, with individual layer thicknesses generally below 40 μm[35]. The selective layer, which is deposited onto the support or intermediate layer through various coating or deposition techniques, is the functional layer where separation predominantly occurs. Its thickness usually falls within the range of 0.2-10 μm, and ongoing research efforts have enabled the laboratory-scale fabrication of ultrathin selective layers with thicknesses down to several tens of nanometers.
Figure 3. Ceramic membrane technology: (A) the schematic of the ceramic membrane asymmetric multilayer architecture, Adapted with permission from Ref.[32], Copyright 2017, Elsevier; (B) performance comparison of different ceramic materials, Adapted with permission from Ref.[19], Copyright 2021, Elsevier; (C) fabrication of ceramic membranes using the sol-gel method, Reprinted with permission from Ref.[33], Copyright 2025, Elsevier; (D) Fabrication of ceramic membranes using 3D printing technology, Reprinted with permission from Ref.[34], Copyright 2024, Elsevier. 3D: Three-dimensional.
The geometric configuration of ceramic membranes in engineering applications is primarily determined by support design, with tubular, hollow-fiber, and flat-sheet formats being the most common. These configurations differ markedly in membrane packing density, mechanical strength, and operating modes. Tubular and hollow-fiber ceramic membranes are generally preferred for continuous-flow systems with high shear and complex water matrices due to their superior pressure resistance, whereas flat-sheet membranes are more commonly used in submerged systems, where air scouring and module replacement are more convenient[36,37]. According to selective-layer pore size, ceramic membranes are classified as microfiltration (MF, > 100 nm), ultrafiltration (UF, 2-100 nm), and nanofiltration (NF, < 2 nm) membranes. MF membranes are mainly applied for removing suspended solids and large particulates, UF membranes enable effective rejection of colloids, macromolecules, and part of MPs, while NF membranes are suitable for separating dissolved species and potentially retaining smaller NPs. Ceramic membranes are predominantly fabricated from inorganic materials such as Al2O3, ZrO2, TiO2, and SiC, which fundamentally eliminate the risk of secondary microplastic release during operation. Their distinct properties in terms of mechanical strength, chemical resistance, and thermal stability provide broad operational flexibility. From an industrial perspective, a relatively mature supply chain has been established, supporting applications from laboratory research to full-scale deployment[33] [Figure 3B]. Notably, MF and UF membranes have already been implemented at full scale in several countries, including Singapore, Japan, China, and the United States, demonstrating their technological maturity and industrial reliability[19]. In contrast, ceramic NF membranes are currently at an intermediate stage between pilot-scale validation and large-scale application. Existing studies indicate that ceramic NF membranes have been successfully applied in small- to medium-scale systems (10-100 m2 per plant) for applications such as hot wastewater recycling in industrial processes. For example, membrane elements have been progressively scaled up from conventional 0.25 to 1.3, 4.5 m2, and even up to 10 m2 per element, accompanied by corresponding developments in support design, coating processes, and quality control[38]. Pilot-scale systems with a total membrane area exceeding 200 m2 have already been demonstrated under continuous operation, confirming the technical feasibility of ceramic NF membranes in real industrial scenarios.
Ceramic membrane fabrication integrates techniques originally developed for ceramic and metallic materials processing. Porous supports are commonly prepared by extrusion or slip casting[39], while intermediate MF layers are typically formed via particulate suspension coating[40]. The top selective layers of asymmetric membranes, including UF and NF layers, are mainly fabricated using sol-gel[41,42] or solid-state sintering methods[43-45]. Despite variations in specific routes, fabrication generally involves three key steps: slurry preparation, structural shaping, and high-temperature sintering, with slurry composition, forming method, and sintering conditions jointly determining membrane pore structure and mechanical properties. Membranes fabricated via solid-state sintering primarily rely on particle packing behavior to form their porous structure. This approach is commonly employed for ceramic MF membranes, where the surface chemical properties are largely governed by the intrinsic characteristics of the raw materials, enabling efficient removal of MPs through size exclusion. In contrast, for UF and NF membranes, the sol-gel method is one of the most important fabrication routes due to its excellent controllability over composition and microstructure [Figure 3C]. In this process, metal alkoxides or inorganic salts undergo controlled hydrolysis and condensation to form a stable sol[46], which is deposited onto a support and subsequently transformed into a ceramic membrane after drying and thermal treatment[47]. This method typically yields separation layers with narrower pore size distributions and higher surface hydroxyl group densities, thereby improving water permeability and facilitating higher flux during MPs/NPs removal. In parallel, advances in additive manufacturing are driving a transition toward digitally controlled membrane fabrication. Three-dimensional (3D) printing enables precise design of membrane geometry and pore architecture through layer-by-layer construction[34], offering new opportunities for fabricating complex, asymmetric, and structure-function integrated ceramic membranes[48] [Figure 3D].
REMOVAL OF MPs/NPs BY CERAMIC MEMBRANE TECHNOLOGIES
Microfiltration
MF ceramic membranes are widely used low-pressure membrane separation technologies in water treatment systems. They are mainly applied for the removal of fine particles and suspended solids from water. The separation process is pressure-driven and is sensitive to changes in operating pressure. In microplastic removal, the dominant mechanism of MF membranes is size sieving. The pore size of MF membranes typically ranges from 0.1 to 5 μm. Therefore, they are mainly effective for larger microplastic particles. When the microplastic size is significantly larger than the membrane pores, particles are geometrically restricted during transport and are retained on the membrane surface, resulting in high removal efficiency[49].
In municipal wastewater treatment systems, MPs in secondary effluent are mainly distributed in the size range of 10-20 μm. Conventional activated sludge processes show limited removal efficiency for MPs smaller than 20 μm. Takeuchi et al.[50] employed ceramic MF membranes as a tertiary treatment unit for secondary effluent. By introducing ceramic MF membranes with pore sizes of 0.18 and 0.8 μm, the additional removal efficiency of residual MPs exceeded 72%. As a result, the overall removal efficiency of the combined process increased to more than 96%.
When treating industrial laundry wastewater dominated by fibrous MPs, MF membranes can also achieve high removal efficiency. Previous studies have shown that silicon carbide (SiC)[51], alumina (Al2O3)[3] and zirconia-titania(ZrO2-TiO2)[52]. MF membranes can remove more than 98% of fibrous MPs from real industrial laundry wastewater. However, under conditions of high fiber content and high suspended solid loading, significant permeability decline is commonly observed during continuous operation. After approximately four days of filtration, membrane permeability can decrease to about 5% of its initial value[51]. This indicates that although size exclusion ensures high retention efficiency, continuous particle accumulation accelerates cake layer formation and leads to a pronounced increase in filtration resistance.
In addition, ceramic MF membranes have demonstrated remarkable advantages in the removal of MPs from rainwater. Song et al.[53] systematically investigated the interception and fouling behaviors of MPs with different morphologies - predominantly fibrous and granular polyethylene terephthalate (PET) MPs - in roofing rainwater using a gravity-driven ceramic MF membrane reactor. The results showed that a ceramic MF membrane with a pore size of 0.14 μm could efficiently retain MPs with particle sizes of 100-300 μm. In actual roofing rainwater, where microplastic concentrations ranged from 21 to 29 items L-1, no deterioration in permeate quality attributable to microplastic breakthrough was observed during long-term operation. The effluent concentrations of NH3-N and total organic carbon (TOC) remained consistently low, indicating the high reliability of ceramic MF membranes for microplastic removal in rainwater reuse systems[53].
Ultrafiltration
UF ceramic membranes occupy an intermediate position between MF and NF in particulate separation processes. Although UF membranes remove MPs primarily through the same size-exclusion mechanism as MF, their smaller pore size and denser separation layer fundamentally alter fouling development during filtration.
Under operating conditions where microplastic rejection approaches quantitative removal, UF ceramic membranes generally exhibit more moderate flux decline and higher operational stability than MF membranes. In high-load systems dominated by fibrous and fragmented plastics, MF membranes can experience near-complete permeability loss (> 90%) within a few days of continuous operation, whereas UF membranes show a much smaller flux reduction (30%-40%) over the same operating period and maintain stable filtration performance[51]. Similar hydraulic behavior is also observed in complex water matrices such as municipal wastewater, where UF ceramic membranes sustain higher stable permeate fluxes. This behavior can be attributed to differences in fouling evolution. During the initial stage of operation, UF ceramic membranes may undergo a certain degree of internal pore deposition, particularly in the presence of low-molecular-weight organics or colloidal foulants. However, as filtration proceeds, internal pore blocking does not continue to intensify. Instead, fouling gradually evolves toward a regime dominated by surface deposition and cake layer formation. Compared with MF membranes, the smaller pore size of UF membranes effectively suppresses the penetration of large microplastic particles and fibers into membrane pores[54]. This substantially reduces the accumulation of deep pore blocking and contributes to improved long-term operational stability.
Consequently, compared with MF, UF ceramic membranes provide a broader stable operating window for microplastic removal. High rejection efficiency is maintained while resistance to fouling-induced performance deterioration is significantly improved. This makes UF particularly suitable for high-load conditions or long-term continuous operation. It should be noted, however, that despite pore sizes approaching the sub-100 nm range, UF membranes do not form absolute barriers to true NPs (< 100 nm)[55]. Several studies have pointed out that UF membranes exhibit limited effectiveness in removing smaller MPs and even NPs[56]. For example, a ZrO2 ceramic UF membrane with an average pore size of approximately 55 nm achieved a MPs removal efficiency of about 96% when treating hospital laundry wastewater, yet approximately 4% of MPs were not completely retained[57]. The authors attributed the permeation of this fraction mainly to the size distribution and morphological characteristics of MPs. On the one hand, some MPs exhibit spherical or short and compact morphologies with high circularity, which facilitates their transport through membrane pores under pressure-driven filtration. On the other hand, the analytical detection limit (~20 μm) employed in the study implies that smaller MPs, or even NPs, may not have been fully identified or may have been underestimated, thereby appearing as “incomplete removal” in the apparent rejection efficiency.
Nanofiltration
NF ceramic membranes are capable of effectively retaining multivalent salts and organic molecules with molecular weights exceeding 200 Da. As the separation process is predominantly governed by size-exclusion mechanisms, NF membranes are more suitable than MF and UF membranes for the retention of smaller MPs and NPs, thereby enabling the efficient removal of NPs from aquatic systems.
The application potential of ceramic NF membranes for NPs removal in real wastewater matrices has been preliminarily demonstrated. Russo et al.[6] investigated municipal wastewater treatment plant secondary effluent using a 1 kDa TiO2 tubular ceramic NF membrane as a tertiary treatment unit, operated at a transmembrane pressure of approximately 6.5 bar. Efficient retention of MPs/NPs was achieved, with no plastic-related signals detected in the permeate, while high volume concentration factors enabled the enrichment of plastic particles in the retentate. Membrane fouling during operation was mainly attributed to particle deposition and pore blockage; however, it was predominantly reversible, as more than 90% of the initial permeance could be recovered through backwashing and hot-water cleaning, indicating good operational stability.
Furthermore, Rodrigo et al.[58] developed a hybrid ceramic membrane separation process that integrates a flat-sheet ceramic MF membrane with a tubular ceramic membrane with a nominal pore size of approximately 1 kDa for the stepwise removal of MPs and NPs from real municipal wastewater. In this system, the flat-sheet MF membrane primarily removes suspended solids and micrometer-scale plastic particles, while the tubular ceramic membrane serves as a polishing unit, selectively retaining and concentrating nanoscale plastic particles as well as their complexes with dissolved organic matter (DOM). Although the operating flux of the tubular ceramic membrane is substantially lower than that of the MF membrane (approximately 6 L·m-2·h-1), it effectively stabilizes the total suspended solids in the permeate at 1-3 mg·L-1 and further reduces the chemical oxygen demand to 6-11 mg·L-1[58]. These results demonstrate the strong water polishing capacity of ceramic tight-pore (NF-like) membranes and highlight their potential for the advanced control of NPs in wastewater treatment.
The main conditions under which the studies we found regarding the use of MF, UF, and NF ceramic membranes for removing MPs/NPs are presented in Table 1. MF, UF, and NF ceramic membranes exhibit a clear hierarchical differentiation in separation behavior, in which decreasing pore size systematically enhances selectivity toward smaller MPs/NPs while progressively constraining permeability and altering fouling characteristics.
Articles found in the literature related to MF, UF, and NF ceramic membranes for the removal of MPs/NPs
| Membrane | Aqueous matrix | Microplastic/nanoplastic | Refs. | ||||||
| Type | Material | Pore size/ MWCO | Type | Concentration | Size (μm) | Operating condition | Removal | ||
| MF | TiO2 | 800 nm | Secondary wastewater effluent | - | 25-68 MP/L | 10-20 | Cross-flow T: 25 °C TMP: 20-100 kPa | > 72% | [50] |
| MF | SiC | 247 nm | Laundry wastewater | Nylon fibers | 4.5 × 104 MP/L (22.6 mg/L) | 80 ± 36 | Cross-flow Time: 96h | 98.5% | [51] |
| UF | ZrO2 | 58 nm | 80 ± 36 | 99.2% | [51] | ||||
| UF | SiC | 100 nm | PET fibers | - | - | Suction-driven T: 20 °C TMP: 0.25 bar | 87.7%-98.9% | [3] | |
| UF | Al2O3 | 100 nm | - | - | 90.3%-97.7% | [3] | |||
| MF | ZrO2-TiO2 | 800 nm | PET fibers | - | 89.3 | Dead-end T: 25 °C TMP: 0.1 - 1.0 bar | 99.96% | [52] | |
| UF | - | 300 kDa | Eutrophic surface water | PET | 2 mg/L; | 300 | Dead-end TMP: 0.2 bar | - | [55] |
| UF | - | 50 kDa | PET | 34 mg/L | 100 | Dead-end TMP: 0.6 bar | - | [55] | |
| MF | - | 140 nm | Roof rainwater | PET | 288 MP/L | 100-300 | Gravity-driven Time: 90 days | - | [53] |
| UF | - | 50 kDa | PET | 288 MP/L | 100-300 | - | [53] | ||
| MF | SiC | 300 nm | Hospital laundry wastewater | PET | 1.4 × 106 MP/L | 100-200 | Suction-driven TMP: 0.2 bar | 87% | [57] |
| UF | ZrO2 | 60 nm | PET | 1.4 × 106 MP/L | 100-200 | Suction-driven TMP: 0.5 bar | 96% | [57] | |
| UF | Al2O3 | 100 nm | Laundry wastewater | PMPF | 10 mg/L | - | Cross-flow CFV: 100 mL/min TMP: 0.2 bar Time: 48 h | 90.4% | [56] |
| MF | - | 1.4 μm | Municipal wastewater | MPs (PE, PP, PVC, PS) | 1,900-2,224 MP/L | 56-80 | Cross-flow CFV: 0.4 L/min T: 25 °C | > 99% | [58] |
| NF | - | 1 kDa | NPs | 3,203-11,821 NP/L | < 0.1 | Cross-flow CFV: 2 L/min T: 25 °C | - | [58] | |
| NF | TiO2 | 1 kDa | PE, PVC, PS, PP | - | < 0.1 | Cross-flow TMP: 6.5 bar | - | [6] | |
POLLUTION MECHANISMS OF MPs/NPs
MPs/NPs exhibit complex fouling behaviors during membrane separation processes that are distinct from those of conventional particulate foulants. Accumulating evidence indicates that membrane fouling induced by MPs/NPs is not governed by a single mechanism but rather arises from the combined effects of physical blocking, interfacial interactions, enhanced biofouling, and synergistic interactions with organic matter.
Physical blocking
During membrane separation, MPs and NPs act as typical particulate foulants with time-dependent deposition behaviors. Strong interactions with membrane pore entrances at the early filtration stage cause rapid flux decline, whereas prolonged operation leads to a gradual transition from pore-blocking-dominated fouling to surface cake layer-controlled filtration, as supported by flux decline analyses and kinetic modeling.
Enfrin et al.[59] systematically investigated polyethylene MPs/NPs (13-690 nm) and identified three fouling stages: an initial stage dominated by intermediate and complete pore blocking (0-6 h), a transitional stage characterized by progressive pore closure (6-30 h), and a late stage in which particle accumulation on the membrane surface formed a porous cake layer, resulting in continuous flux decline without complete permeability loss [Figure 4A-D][59-61]. In the pore-blocking-dominated early stage, size matching between MPs and membrane pores is decisive. When particle size is comparable to pore size, MPs are more likely to penetrate pore entrances or become trapped within pores, markedly increasing irreversible fouling. For example, 0.25 μm polystyrene MPs (PS-MPs) induced significantly higher transmembrane pressure and irreversible resistance than larger MPs, whereas MPs of 31.6 and 225 μm were mainly retained on the membrane surface and did not enter pore structures[62]. Post-cleaning analyses confirmed that pore-scale physical entrapment constitutes a critical initiation mechanism for rapid performance deterioration.
Figure 4. Pollution mechanisms of MPs and NPs: (A) Physical blocking, Reprinted with permission from Ref.[59], Copyright 2020, Elsevier; (B): Interfacial interaction, Reprinted with permission from Ref.[60], Copyright 2021, Elsevier; (C): Synergistic interactions with organic matter and microorganisms, Reprinted with permission from Ref.[61], Copyright 2024, Elsevier. MPs: Microplastics; NPs: nanoplastics; PSF: polysulfone; SE: secondary effluent; MP: microplastic.
With extended filtration, fouling shifts toward a surface deposition-dominated regime, where fouling severity no longer varies monotonically with particle size. Instead, a critical particle size range may exist that maximizes hydraulic resistance. In UF of natural raw water (30 nm pore size), 1 μm PS-MPs caused the greatest transmembrane pressure increase, forming the densest and least porous cake layers, whereas smaller or larger MPs generated looser deposits[63]. These findings indicate that fouling severity is governed not only by particle retention but also by cake layer microstructure and compaction.
Beyond particle size, microplastic morphology further regulates fouling evolution at later stages. In gravity-driven ceramic membrane systems, fibrous MPs induced faster flux decline than particulate MPs due to their high aspect ratio, which promotes preferential orientation and entanglement, enabling rapid formation of a 3D cake skeleton[53]. In contrast, particulate MPs formed cake layers more slowly but exhibited steeper increases in specific cake resistance under compression. Collectively, these results demonstrate that particle size and morphology jointly govern MP-induced fouling by controlling pore blockage, cake layer formation, and compressibility.
Interfacial interactions
In membrane processes involving MPs/NPs, surface adsorption driven by interfacial interactions is a primary mechanism governing fouling initiation and evolution. Unlike conventional particulate fouling dominated by size exclusion, MPs/NPs exhibit strong hydrophobicity and high interfacial chemical activity, making their deposition behavior largely controlled by interfacial interaction mechanisms. Based on the XDLVO (extended Derjaguin-Landau-Verwey-Overbeek) analysis, Enfrin et al.[60] demonstrated that polar interactions play a decisive role in MP/NP-induced fouling. For unmodified PSF UF membranes with low surface polarity, weak hydration layers lead to reduced short-range polar repulsion and strong thermodynamic attraction toward polyethylene MPs/NPs (~93 nm), as evidenced by negative adhesion free energy [Figure 4B]. Under cross-flow filtration (10 mg·L-1 MPs/NPs, 1 bar), this resulted in a 38%-40% flux decline within 6 h and ~40% particle adsorption. In contrast, enhancing membrane surface polarity via polar functional groups significantly strengthened polar repulsion, reducing MP/NP adsorption by over 60% and effectively mitigating flux decline.
Notably, ceramic membranes inherently possess abundant surface hydroxyl (-OH) groups[64], which confer high polarity and hydrophilicity, promoting the formation of a stable hydration layer and strong interfacial repulsion. Furthermore, membrane hydrophilicity can be further enhanced through fabrication strategies. For example, the incorporation of hydrophilic elements (e.g., Fe) during the sol synthesis stage has been reported to reduce the water contact angle of ceramic UF membranes from approximately 30° to 18°, indicating significantly improved surface wettability[65]. This enhanced hydrophilicity effectively suppresses interfacial interactions between the membrane surface and MPs/NPs, thereby reducing particle adsorption. For example, in real laundry wastewater, the flux of a PES membrane sharply decreased from 8742 to 244 L·m-2·h-1 (≈ 97.2% decline), whereas that of a ceramic membrane decreased from 1647 to 130 L·m-2·h-1 (≈ 92.1% decline), indicating a lower fouling propensity of the ceramic membrane[52]. Consequently, ceramic membranes generally exhibit reduced MP/NP adsorption and improved resistance to flux decline during filtration of MP/NP-containing waters[66].
Synergistic interactions of MPs/NPs with organic matter and microorganisms
The impact of MPs/NPs on membrane fouling cannot be attributed solely to their intrinsic physicochemical properties or direct membrane interactions. In real water systems, MPs/NPs induce a nonlinear amplification of fouling through synergistic interactions with natural organic matter (NOM) and microorganisms. Lin et al.[61] reported that the transmembrane pressure increase caused by MPs coexisting with NOM was significantly greater than the linear sum of their individual effects, highlighting strong coupling effects [Figure 4C].
Rather than contributing primarily via direct deposition, MPs act as dynamic interfacial substrates that selectively associate with DOM, particularly aromatic and hydrophobic fractions, due to their hydrophobic and π-electron-rich polymer backbones[24]. This selective interfacial partitioning redistributes DOM from the aqueous phase to MP surfaces without reducing overall organic carbon, forming organic-enriched composite microplastic interfaces that govern subsequent transport and fouling behavior [Figure 5]. When such organic-coated MPs participate in membrane filtration, they further amplify fouling by altering the chemical availability of organic interfaces. Lin et al.[61] observed increased hydrophobicity of fouling layers in the presence of MPs during NF, despite the enrichment of proteins, polysaccharides, and humic substances, indicating shielding or complexation of polar functional groups within dense fouling structures. These results demonstrate that MPs modify fouling chemistry rather than merely increasing organic loading.
Figure 5. Schematic of micro- and nanoplastics-induced membrane fouling. MPs: Microplastics; NPs: nanoplastics; MP: microplastic; NOM: natural organic matter; EPS: extracellular polymeric substance.
In addition, MPs act as active biological modulators by promoting microbial attachment and metabolic activity, thereby stimulating extracellular polymeric substance (EPS) production[67,68]. Under prolonged filtration, excessive EPS secretion becomes a key driver of fouling evolution, leading to rapid cake-layer thickening and densification[69]. The interaction between carboxyl-rich EPS components and multivalent cations (e.g., Ca2+) promotes the formation of cross-linked gel networks (“egg-box” structures), enhancing fouling layer stability and accelerating the development of dense, low-permeability composite fouling layers[61,69,70].
EMERGING CERAMIC MEMBRANE TECHNOLOGIES FOR FOULING RESISTANCE
Membrane fouling caused by MPs/NPs poses a new challenge to the performance of water treatment facilities. In recent years, a series of novel ceramic membrane materials and technologies have been developed to mitigate fouling, including piezoelectric ceramic membranes[71] and advanced oxidation coupled ceramic membranes. These approaches either reduce the adhesion of MPs, alleviate the formation of fouling[72] layers during the filtration process, promote membrane regeneration, significantly enhance the antifouling performance of ceramic membranes and further expand their application in the removal of MPs/NPs from water.
Piezoelectric ceramic membranes
Currently, membrane fouling control is dominated by surface modification strategies, including superhydrophilic coatings, low-surface-energy layers, and charge-regulated structures, which aim to reduce foulant adsorption on membrane surfaces and within pores. However, these approaches are inherently passive, offering only temporary fouling delay rather than sustained suppression. Under long-term operation, modified surfaces inevitably become covered by pollutants, leading to a gradual decline in antifouling efficiency and necessitating frequent physical or chemical cleaning, thereby increasing operational costs and accelerating membrane aging.
Against this background, piezoelectric ceramic membranes have emerged as a new class of functional membranes with active response capabilities, attracting increasing attention for fouling mitigation. Owing to their ability to generate electrical potential under mechanical stress, piezoelectric membranes can respond to dynamic changes during filtration, enabling self-driven fouling control. Based on stress-electrical coupling, piezoelectric effects are generally categorized into the direct and inverse piezoelectric effects, with the latter being more relevant to membrane separation.
The inverse piezoelectric effect induces periodic deformation or vibration of the membrane under an applied alternating electric field, disturbing foulants on membrane surfaces and within pores and thereby promoting their detachment. Representative studies have demonstrated that structural optimization of piezoelectric ceramics enables the simultaneous enhancement of permeability and piezoelectric performance. For example, Mao et al.[71] tailored grain size and pore structure to obtain piezoelectric membranes with high permeability (814 L·m-2·h-1·bar-1) and a stable piezoelectric response, while further reducing the piezoelectric layer thickness to 500 μm to improve flux stability under an alternating electric field. Zhu et al.[73] reported BaTiO3-based piezoelectric membranes that maintained nearly constant flux during oil-water separation at 60 V [Figure 6A]. Fan et al.[74] further demonstrated that combining piezoelectric supports with robust membrane layers via optimized forming and co-sintering strategies enables effective self-cleaning, achieving high normalized stable flux and full flux recovery after cleaning [Figure 6B].
Figure 6. Piezoelectric ceramic membranes: (A) BTO piezoelectric ceramic membrane Reprinted with permission from Ref.[73], Copyright 2025, Elsevier; (B) ZrO2/Al2O3 piezoelectric ultrafiltration membrane; Reprinted with permission from Ref.[74], Copyright 2026, Elsevier; (C) MnO/BTO ceramic membrane with antifouling behavior induced by the positive piezoelectric effect. Reprinted with permission from Ref.[75], Copyright 2022, Springer Nature. BTO: Barium titanate.
The positive piezoelectric effect refers to the spontaneous generation of electrical charges on the surface of piezoelectric materials when they are subjected to external pressure or mechanical stress, thereby forming an electric potential. Zhao et al.[75] proposed a pulse water pressure-responsive piezoelectric ceramic self-cleaning membrane, utilizing the positive piezoelectric effect of BaTiO3 piezoelectric ceramics. This converts the inevitable hydraulic pressure pulses during the membrane separation process into transient electroactive responses, eliminating the need for an external electric field during operation and realizing a truly “process-induced” self-cleaning mechanism [Figure 6C]. Under periodic pressure fluctuations, the membrane generates rapid voltage oscillations and current pulses, which subsequently induce the generation of reactive oxygen species (ROS) on the membrane surface and produce dielectrophoretic repulsion. This synergistically inhibits the attachment and accumulation of organic pollutants, oil droplets, microorganisms, and inorganic colloidal particles, significantly alleviating membrane fouling.
The mechanism of generating electroactive responses using the piezoelectric effect has not only made significant progress in membrane self-cleaning but has also been shown to play an important role in microplastic degradation. Through the piezoelectric photocatalytic mechanism, the Mo5Fe5 heterostructure generates H2O2 and ROS, effectively degrading PS-MPs. Similar to the ROS generated during membrane cleaning, the Mo5Fe5 catalyst, under light and ultrasonic excitation, utilizes its piezoelectric effect to generate an internal electric field, enhancing photocatalytic efficiency and significantly promoting the degradation of MPs[76]. This technology has broad application potential when combined with ceramic membranes. By integrating membrane self-cleaning with electroactive responses, the membrane material can actively generate electroactive substances during operation, improving its antifouling capability. This innovative mechanism helps extend the membrane's service life and significantly enhances the efficiency of treating micro and NPs in water, providing a novel solution for controlling membrane fouling and addressing microplastic pollution.
Ceramic membranes coupled with advanced oxidation technology
Advanced Oxidation Processes (AOPs) are widely used water treatment technologies for the degradation and removal of persistent pollutants in the environment, including photocatalysis, persulfate activation, and ozone catalysis[77]. The core principle of AOPs is the generation of highly oxidative reactive radicals that can oxidize and decompose refractory organic pollutants into low-toxic or non-toxic small molecules, and in some cases, even directly degrade them into carbon dioxide and water. Compared to traditional water treatment methods, AOPs offer advantages such as high reactivity, broad applicability, and more thorough treatment efficiency, making them particularly advantageous for the degradation of emerging pollutants[78]. In recent years, leveraging the high chemical and structural stability of ceramic membranes[79], researchers have increasingly focused on coupling oxidation processes with ceramic membrane filtration technology to NOM in MPs/NPs-containing systems. By degrading NOM, such integrated systems can effectively mitigate the synergistic interactions between NOM and MPs/NPs, thereby alleviating membrane fouling, improving operational stability, and extending membrane lifespan. This approach has emerged as a rapidly growing research focus.
Photocatalysis
Photocatalytic degradation, as an environmentally friendly pollutant treatment technology, has become a research focus in the field of microplastic and antibiotic pollution control in aquatic environments[80,81]. Titanium dioxide (TiO2) is a commonly used UV photocatalyst, and many researchers have recently coupled it with ceramic membranes to mitigate membrane fouling. Song et al.[82] designed a hybrid system combining tubular ceramic MF membranes with TiO2-loaded PES microspheres, aimed at treating simulated water containing humic acid and MPs. The study found that as the humic acid (HA) concentration increased, the membrane fouling resistance significantly rose, with DOM primarily causing irreversible fouling through adsorption and pore blockage [Figure 7A]. In this hybrid system, the TiO2 photocatalytic oxidation on PES microspheres removed approximately 46.9% of DOM, significantly reducing the organic load reaching the membrane surface, thereby lowering the membrane fouling resistance from 0.393 × 10-9 kg/m2s in the MF process alone to 0.242 × 10-9 kg/m2s in the MF+PES+UV process[82]. This strategy not only alleviated membrane fouling but also improved the overall operational stability of the system by reducing the encapsulation of MPs by organic matter.
TiO2 is inherently a ceramic material, and when prepared into a membrane, it not only serves as a filtration membrane but also exhibits catalytic properties, thus simplifying the process[80,83]. Coelho et al.[57] embedded the photocatalyst TiO2 onto the surface of ceramic membranes to prepare Ce-Y-ZrO2/TiO2 ceramic UF membranes that were used to treat hospital laundry wastewater containing MPs and high concentrations of organic matter (Total solids 1,738 mg/L), achieving a 99.9% removal of MPs. The study found that filtration with a pure ceramic membrane alone resulted in the formation of a gel layer on the membrane surface, causing severe irreversible fouling [Figure 7B]. By employing Ce-Y-ZrO2/TiO2 UF membranes with photocatalytic activity and UV-light-emitting diode (LED) irradiation, the catalyst induced ROS production under light, which oxidized and degraded organic pollutants, thus preventing the formation of the gel layer and pore blockage. The organic fouling on the membrane surface was effectively degraded, nearly eliminating irreversible fouling. The study noted that membrane flux decay under illumination was approximately 12% lower than under dark reaction conditions, and the flux recovery rate after backwashing was significantly higher, confirming that photocatalysis imparted excellent “self-cleaning” ability to the ceramic membrane. Additionally, the superhydrophilicity of TiO2 under UV light reduced the adsorption of hydrophobic pollutants on the membrane surface, further reducing fouling resistance.
Persulfate activation
Persulfate activation, through the generation of highly oxidative radicals, enables efficient degradation of emerging micropollutants, and its integration with ceramic membranes offers a promising strategy for simultaneously enhancing removal efficiency and mitigating membrane fouling. Bai et al.[84] fabricated ceramic membranes loaded with bimetallic layered metal oxides [CoAl@AM, CuAl@AM, and MnAl@AM (AM = alumina ceramic substrate membranes)] and demonstrated that in situ peroxymonosulfate (PMS) activation effectively mitigates NOM-induced membrane fouling. PMS addition markedly increased the normalized flux of all membranes, with the CoAl@AM/PMS system showing the greatest enhancement, where the normalized flux increased from 0.28 to 0.62. Interfacial free energy analysis revealed that PMS activation significantly increased the total interfacial free energy of the membrane-NOM system, dominated by changes in Lewis acid-base interactions, thereby strengthening membrane-NOM repulsion and reducing organic foulant adsorption [Figure 8].
Figure 8. Schematic diagram of ceramic membrane coupled with persulfate activation for mitigating membrane fouling, Reprinted with permission from Ref.[84], Copyright 2022, Elsevier. UV254: Ultraviolet absorbance at 254 nm; TOC: total organic carbon; LMO: layered metal oxide; NOM: natural organic matter.
However, the effectiveness of catalytic oxidation in fouling mitigation is strongly dependent on oxidation intensity, as insufficient oxidative capacity may even aggravate irreversible fouling. Although PMS addition reduced reversible fouling, the irreversible fouling resistance increased in the MnAl@AM and CuAl@AM systems, which was attributed to their weaker oxidation ability that caused partial degradation of high-molecular-weight NOM into pore-penetrating intermediates, thereby promoting internal pore blocking[84]. In contrast, the CoAl@AM system, with stronger ROS generation and mineralization capability, enabled more complete organic degradation and achieved the highest hydraulic cleaning efficiency. Consistently, Chen et al.[85] demonstrated that increasing PMS dosage (0.01-0.3 mM) markedly suppressed fouling, as evidenced by a reduction in net transmembrane pressure increment from 1.69 to 0.12 kPa, highlighting the critical role of sufficient oxidation intensity in fouling control.
To address the inherent susceptibility of conventional catalytic membranes to pore blockage and fouling during operation, refined membrane pore structure engineering has emerged as an effective strategy to enhance long-term operational stability. Yang et al.[86] incorporated single-atom catalytic systems, characterized by high atomic utilization efficiency and superior reaction kinetics, into the pore channels of ceramic membranes, thereby achieving a deep integration of catalytic reactions and membrane separation through nanoconfinement. A systematic comparison between ceramic membranes loaded with manganese oxide nanoparticles (Mn-NP@CM) and single-atom-confined ceramic membranes (Mn-SA@CM) revealed pronounced differences in fouling behavior and operational performance[86]. In the Mn-NP@CM system, manganese species existed predominantly in nanoparticulate form and were prone to aggregation, resulting in substantially lower reaction kinetics than those of the single-atom counterpart. Consequently, a dense foulant layer formed on the membrane surface after approximately 3 h of continuous operation. By contrast, the Mn-SA@CM constructed via a hierarchical cross-scale assembly strategy exploited the synergistic confinement of MOF-derived micropores and ceramic membrane nanopores, enabling highly efficient reaction-mass transfer coupling and imparting a pronounced self-cleaning functionality[86]. During continuous filtration over 168 h and treatment of real hospital wastewater, Mn-SA@CM exhibited negligible foulant deposition on the membrane surface, while maintaining a stable permeate flux. This outstanding antifouling performance was primarily attributed to the up to 105-fold enhancement in reaction kinetics under nanoconfinement, which allowed organic pollutants to be rapidly degraded within millisecond-scale residence times, thereby effectively preventing their accumulation and blockage within membrane pores. Overall, the integration of advanced oxidation processes into membrane systems offers substantial benefits for fouling control.
CHALLENGES AND FUTURE PERSPECTIVES
Ceramic membrane technology shows great potential for microplastic removal, especially in high-load, long-term operation water treatment systems. However, there are still several challenges that need to be overcome:
(1) Development of low-cost ceramic membranes. The high cost of ceramic membranes remains a significant barrier to their wider adoption in the market. Currently, the price of Al2O3 ceramic membranes ranges from $500 to $3,000 per square meter[87,88], while polymeric membranes typically cost between $20 and $200 per square meter[89,90]. Therefore, the development of advanced technologies for low-cost ceramic membranes could greatly promote their industrial application. The high cost of ceramic membranes mainly arises from two aspects: the relatively expensive raw materials and the high energy consumption associated with high-temperature processing during fabrication. To address these challenges, cost reduction strategies can focus on both material selection and process optimization. On the one hand, the use of inexpensive raw materials, such as low-cost natural minerals containing naturally occurring silicates[91] and inorganic industrial solid wastes[92], offers a promising pathway to lower material costs. On the other hand, the development of cost-effective fabrication techniques is also critical. This includes the adoption of novel forming methods (e.g., centrifugal casting[93] and phase inversion[94]) as well as advanced sintering strategies (e.g., low-temperature sintering[95,96] and co-sintering[74,97]), which together contribute to more efficient and economically viable membrane production.
(2) Coupling ceramic membranes with advanced oxidation processes can significantly enhance the degradation efficiency of organic pollutants. However, it should be noted that incomplete mineralization during oxidation may lead to the formation of a series of complex intermediates, including aldehydes, ketones, and carboxylic acids, as well as many NPs[98,99]. These transformation products may exhibit higher mobility and bioavailability, enabling them to more readily penetrate biological barriers, accumulate in organs, and induce oxidative stress and inflammatory responses, thereby posing potential health risks that cannot be overlooked[100]. Therefore, future research should place greater emphasis on elucidating the composition, toxicity, and transmembrane transport behavior of these oxidation by-products. In addition, optimizing membrane materials, structural design, and catalytic efficiency is essential to minimize the generation and permeation of these potentially harmful substances.
(3) Detection of NPs and the balance between detection accuracy and cost remain critical challenges in this field. The detection of NPs is inherently difficult due to their extremely small particle size, low environmental concentration, and the lack of standardized analytical methods. Although conventional techniques such as electron microscopy and vibrational spectroscopy can achieve reliable identification under laboratory conditions, they are often associated with high costs, complex sample preparation, and low throughput, limiting their applicability for large-scale environmental monitoring[101]. In recent years, several novel analytical approaches have been developed to improve nanoplastic detection and quantification. High-resolution techniques, such as scanning probe microscopy coupled with vibrational spectroscopy [e.g., AFM-IR (atomic force microscopy-based infrared spectroscopy)[102,103]] and optical tweezers-enabled Raman spectroscopy[104], enable chemical identification at the single-particle level with nanoscale resolution. Meanwhile, advanced mass spectrometry-based methods, including TD-PTR/MS (thermal desorption-proton transfer reaction mass spectrometry)[105] and MALDI-ToF MS (matrix-assisted laser desorption/ionization time-of-flight mass spectrometry)[106], provide high sensitivity and molecular-level characterization, although they are typically destructive and lack particle-specific information. In addition, emerging preconcentration strategies, such as cloud point extraction[107], have been introduced to enhance detection sensitivity for NPs present at ultralow concentrations. Despite these advances, most novel techniques remain costly, time-consuming, and difficult to standardize[108]. On the other hand, fluorescence staining methods, which are relatively low-cost and widely used for microplastic detection, still face limitations in sensitivity and specificity when applied to NPs[109]. Therefore, future efforts should focus on integrating low-cost detection strategies with advanced data analysis tools, such as artificial intelligence[110], to improve detection accuracy, reduce false positives, and ultimately achieve a practical balance between analytical performance and economic feasibility.
CONCLUSION
This review systematically summarizes the application of ceramic membrane technologies for the removal of MPs/NPs from water and wastewater. Ceramic MF, UF, and NF membranes exhibit a progressive transition from high permeability to high selectivity, enabling effective removal across different particle size ranges, while MPs/NPs-induced fouling is governed by coupled mechanisms including physical blocking, interfacial interactions, and synergistic effects with organic matter and microorganisms. Emerging strategies, such as piezoelectric ceramic membranes and membrane-advanced oxidation process coupling, demonstrate strong potential for fouling mitigation and performance enhancement. Future efforts should focus on reducing material costs, improving nanoplastic detection, and optimizing integrated membrane systems to facilitate large-scale and sustainable applications.
DECLARATIONS
Authors’ contributions
Writing-original draft: Liu, M.
Data curation, validation, visualization: Liu, M.; Da, X.
Writing-review and editing: Qiu, M.; Chen, X.; Fu, K.
Supervision, writing-review and funding acquisition: Fan, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Key Research and Development Program of China (No. 2022YFB3805000).
Conflicts of interest
Da, X. is affiliated with Nanjing Membrane Materials Industrial Technology Research Institute Co., Ltd., while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Evangeliou, N.; Grythe, H.; Klimont, Z.; et al. Atmospheric transport is a major pathway of microplastics to remote regions. Nat. Commun. 2020, 11, 3381.
2. Iftikhar, A.; Qaiser, Z.; Sarfraz, W.; et al. Understanding the leaching of plastic additives and subsequent risks to ecosystems. Water. Emerg. Contam. Nanoplastics. 2024, 3, 5.
3. Hyeon, Y.; Kim, S.; Ok, E.; Park, C. A fluid imaging flow cytometry for rapid characterization and realistic evaluation of microplastic fiber transport in ceramic membranes for laundry wastewater treatment. Chem. Eng. J. 2023, 454, 140028.
4. Golgoli, M.; Khiadani, M.; Shafieian, A.; et al. Microplastics fouling and interaction with polymeric membranes: a review. Chemosphere 2021, 283, 131185.
5. Hou, L.; Kumar, D.; Yoo, C. G.; Gitsov, I.; Majumder, E. L. Conversion and removal strategies for microplastics in wastewater treatment plants and landfills. Chem. Eng. J. 2021, 406, 126715.
6. Russo, B.; Lavorato, C.; Argurio, P.; et al. Nanofiltration as an effective tertiary treatment for the removal of micro- and nanoplastics from municipal water effluent. Sep. Purif. Technol. 2025, 376, 134121.
7. Thompson, R. C.; Courtene-jones, W.; Boucher, J.; Pahl, S.; Raubenheimer, K.; Koelmans, A. A. Twenty years of microplastic pollution research - what have we learned? Science 2024, 386, eadl2746.
8. Mintenig, S. M.; Bäuerlein, P. S.; Koelmans, A. A.; Dekker, S. C.; Van Wezel, A. P. Closing the gap between small and smaller: towards a framework to analyse nano- and microplastics in aqueous environmental samples. Environ. Sci. Nano. 2018, 5, 1640-9.
9. Leslie, H. A.; Van Velzen, M. J.; Brandsma, S. H.; Vethaak, A. D.; Garcia-vallejo, J. J.; Lamoree, M. H. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199.
10. Sawma, M. J.; Zayyat, R. M.; Ghaddar, R.; Ayoub, G. M. Environmental management of microplastics and additives: a critical review of treatment technologies and their impact. Water. Emerg. Contam. Nanoplastics. 2024, 3, 21.
11. García-rollán, M.; Sanz-santos, E.; Belver, C.; Bedia, J. Key adsorbents and influencing factors in the adsorption of micro- and nanoplastics: a review. J. Environ. Manage. 2025, 383, 125394.
12. Piao, Z.; Agyei Boakye, A. A.; Yao, Y. Environmental impacts of biodegradable microplastics. Nat. Chem. Eng. 2024, 1, 661-9.
13. Chen, Z.; Wei, W.; Ni, B. Prioritizing capture and utilization for microplastic management in water systems. Nat. Rev. Clean. Technol. 2025, 1, 525-7.
14. Divya, V.; Deivayanai, V.; Anbarasu, K.; Saravanan, A.; Vickram, A. A review on advances in hybrid magnetic nanoparticles for microplastics removal: mechanistic insights and emerging prospects. Environ. Res. 2025, 285, 122554.
15. Gu, Q.; Ng, T. C. A.; Bao, Y.; Ng, H. Y.; Tan, S. C.; Wang, J. Developing better ceramic membranes for water and wastewater treatment: where microstructure integrates with chemistry and functionalities. Chem. Eng. J. 2022, 428, 130456.
16. Lin, Z.; Hu, X.; Lin, H.; et al. Membrane technology for microplastic removal: Microplastic occurrence, challenges, and innovations of process and materials. Chem. Eng. J. 2025, 520, 166183.
17. Ding, H.; Zhang, J.; He, H.; et al. Do membrane filtration systems in drinking water treatment plants release nano/microplastics? Sci. Total. Environ. 2021, 755, 142658.
18. Maliwan, T.; Hu, J. Release of microplastics from polymeric ultrafiltration membrane system for drinking water treatment under different operating conditions. Water. Res. 2025, 274, 123047.
19. Asif, M. B.; Zhang, Z. Ceramic membrane technology for water and wastewater treatment: a critical review of performance, full-scale applications, membrane fouling and prospects. Chem. Eng. J. 2021, 418, 129481.
20. Kim, S.; Park, C. Potential of ceramic ultrafiltration membranes for the treatment of anionic surfactants in laundry wastewater for greywater reuse. J. Water. Process. Eng. 2021, 44, 102373.
21. Hyeon, Y.; Kim, S.; Park, C. Exploring the transformation of polyethylene and polyamide microplastics during membrane filtration through FlowCam analysis. Sep. Purif. Technol. 2024, 334, 126036.
22. Obbard, R. W.; Sadri, S.; Wong, Y. Q.; Khitun, A. A.; Baker, I.; Thompson, R. C. Global warming releases microplastic legacy frozen in Arctic Sea ice. Earth’s. Future. 2014, 2, 315-20.
23. Ragu Prasath, A.; Sudhakar, C.; Selvam, K. Microplastics in the environment: types, sources, and impact on human and aquatic systems. Bioresour. Technol. Rep. 2025, 29, 102055.
24. Ding, L.; Luo, Y.; Yu, X.; Ouyang, Z.; Liu, P.; Guo, X. Insight into interactions of polystyrene microplastics with different types and compositions of dissolved organic matter. Sci. Total. Environ. 2022, 824, 153883.
25. De Falco, F.; Cocca, M.; Avella, M.; Thompson, R. C. Microfiber release to water, via laundering, and to air, via everyday use: a comparison between polyester clothing with differing textile parameters. Environ. Sci. Technol. 2020, 54, 3288-96.
26. Ziajahromi, S.; Neale, P. A.; Rintoul, L.; Leusch, F. D. Wastewater treatment plants as a pathway for microplastics: development of a new approach to sample wastewater-based microplastics. Water. Res. 2017, 112, 93-9.
27. Magni, S.; Binelli, A.; Pittura, L.; et al. The fate of microplastics in an Italian Wastewater Treatment Plant. Sci. Total. Environ. 2019, 652, 602-10.
28. Yang, L.; Li, K.; Cui, S.; Kang, Y.; An, L.; Lei, K. Removal of microplastics in municipal sewage from China’s largest water reclamation plant. Water. Res. 2019, 155, 175-81.
29. Gan, X.; Lin, T.; Jiang, F.; Zhang, X. Impacts on characteristics and effluent safety of PVDF ultrafiltration membranes aged by different chemical cleaning types. J. Membr. Sci. 2021, 640, 119770.
30. Yu, S.; Xie, S.; Zhu, Y.; et al. Purifying or polluting? Tracing membrane-based microplastics release for long-term drinking water safety. Water. Res. 2026, 289, 124991.
31. Zhang, M.; Xu, D.; Wang, Y.; et al. Fabrication of cost-effective and antifouling Al2O3-SiO2 membrane for efficient oil emulsion separation. J. Water. Process. Eng. 2024, 68, 106505.
32. Qiu, M., Chen, X., Fan, Y., Xing, W. 1.11 ceramic membranes. In Comprehensive Membrane Science and Engineering, 2nd ed.; Elsevier, 2017; pp 270-97.
33. Zhang, Y.; Chen, T.; Chen, X.; Fu, K.; Qiu, M.; Fan, Y. The application of pressure-driven ceramic-based membrane for the treatment of saline wastewater and desalination - a review. Desalination 2025, 597, 118327.
34. Chen, T.; Xu, P.; Qiu, M.; Chen, X.; Zhong, Z.; Fan, Y. Construction of anti-fouling ceramic tubular membranes with corrugated inner surfaces using DLP 3D printing. J. Membr. Sci. 2024, 706, 122941.
35. Zhao, L.; Bram, M.; Buchkremer, H.; Stover, D.; Li, Z. Preparation of TiO composite microfiltration membranes by the wet powder spraying method. J. Membr. Sci. 2004, 244, 107-15.
36. Samaei, S. M.; Gato-trinidad, S.; Altaee, A. The application of pressure-driven ceramic membrane technology for the treatment of industrial wastewaters - a review. Sep. Purif. Technol. 2018, 200, 198-220.
37. Da Silva Biron, D., Dos Santos, V., Zeni, M. Applications of ceramic membranes. In Ceramic Membranes Applied in Separation Processes; Topics in Mining, Metallurgy and Materials Engineering; Springer International Publishing, 2017; pp 67-80.
38. Voigt, I.; Richter, H.; Stahn, M.; et al. Scale-up of ceramic nanofiltration membranes to meet large scale applications. Sep. Purif. Technol. 2019, 215, 329-34.
39. Falamaki, C.; Naimi, M.; Aghaie, A. Dual behavior of CaCO3 as a porosifier and sintering aid in the manufacture of alumina membrane/catalyst supports. J. Eur. Ceram. Soc. 2004, 24, 3195-201.
40. Kakui, T.; Miyauchi, T.; Kamiya, H. Analysis of the action mechanism of polymer dispersant on dense ethanol alumina suspension using colloidal probe AFM. J. Eur. Ceram. Soc. 2005, 25, 655-61.
41. Li, S.; Wei, C.; Zhou, L.; Wang, P.; Meng, Q.; Xie, Z. Sol-gel derived zirconia membrane on silicon carbide substrate. J. Eur. Ceram. Soc. 2019, 39, 3804-11.
42. Facciotti, M.; Boffa, V.; Magnacca, G.; et al. Deposition of thin ultrafiltration membranes on commercial SiC microfiltration tubes. Ceram. Int. 2014, 40, 3277-85.
43. Feng, X.; Jiang, L. Y.; Matsuura, T.; Wu, P. Fabrication of hydrophobic/hydrophilic composite hollow fibers for DCMD: influence of dope formulation and external coagulant. Desalination 2017, 401, 53-63.
44. Liu, B.; Wang, Z.; Tan, X.; Liu, S. Ultra-high flux ceramic hollow fiber membranes for nanofiltration. J. Membr. Sci. 2024, 709, 123104.
45. De Jong, J.; Benes, N.; Koops, G.; Wessling, M. Towards single step production of multi-layer inorganic hollow fibers. J. Membr. Sci. 2004, 239, 265-9.
46. Puthai, W.; Kanezashi, M.; Nagasawa, H.; Tsuru, T. Enhancement of water permeability in SiO2-ZrO2 nanofiltration membranes via adding organic solutes into the SiO2-ZrO2 sols. Sep. Purif. Technol. 2024, 333, 125851.
47. Puthai, W.; Kanezashi, M.; Nagasawa, H.; Wakamura, K.; Ohnishi, H.; Tsuru, T. Effect of firing temperature on the water permeability of SiO2-ZrO2 membranes for nanofiltration. J. Membr. Sci. 2016, 497, 348-56.
48. Zeng, Y.; Chen, X.; Sun, L.; Yao, H.; Chen, J. Effect of different sintering additives type on Vat photopolymerization 3D printing of Al2O3 ceramics. J. Manuf. Processes. 2022, 83, 414-26.
49. Yahyanezhad, N.; Bardi, M. J.; Aminirad, H. An evaluation of microplastics fate in the wastewater treatment plants: frequency and removal of microplastics by microfiltration membrane. Water. Pract. Technol. 2021, 16, 782-92.
50. Takeuchi, H.; Tanaka, S.; Koyuncu, C. Z.; Nakada, N. Removal of microplastics in wastewater by ceramic microfiltration. J. Water. Process. Eng. 2023, 54, 104010.
51. Luogo, B. D. P.; Salim, T.; Zhang, W.; Hartmann, N. B.; Malpei, F.; Candelario, V. M. Reuse of water in laundry applications with micro- and ultrafiltration ceramic membrane. Membranes 2022, 12, 223.
52. Kim, S.; Hyeon, Y.; Rho, H.; Park, C. Ceramic membranes as a potential high-performance alternative to microplastic filters for household washing machines. Sep. Purif. Technol. 2024, 344, 127278.
53. Song, W.; Li, J.; Liu, C.; et al. Mechanistic analysis of membrane fouling by microplastics in a gravity-driven ceramic membrane reactor for roofing rainwater reuse. J. Membr. Sci. 2024, 707, 122995.
54. Kook, H.; Park, C. Engineered approaches to facile identification of tiny microplastics in polymeric and ceramic membrane filtrations for wastewater treatment. Membranes 2022, 12, 565.
55. Lin, D.; Zhuang, Z.; Yu, N.; Wang, Z.; Song, W.; Du, X. Comprehensive effects of microplastics on algae-laden surface water treatment by coagulation-ultrafiltration combined process: algae cultivation, coagulation performance and membrane fouling development. Sci. Total. Environ. 2024, 924, 171553.
56. Biao, W.; Awanis Hashim, N.; Rabuni, M. F. B.; Lide, O.; Ullah, A. An innovative strategy for polyester microplastic fiber elimination from laundry wastewater via coupled separation and degradation using TiO2-based photocatalytic membrane reactor. Sep. Purif. Technol. 2025, 356, 129929.
57. Bortot Coelho, F. E.; Sohn, S. I.; Candelario, V. M.; Hartmann, N. I. B.; Hélix-nielsen, C.; Zhang, W. Microplastics removal from a hospital laundry wastewater combining ceramic membranes and a photocatalytic membrane reactor: fouling mitigation, water reuse, and cost estimation. J. Membr. Sci. 2025, 715, 123485.
58. Rodrigo, P. M.; Sims, E. M.; Kommalapati, R. R. Application of hybrid ceramic membranes for microplastic and nanoplastic separation and improved wastewater treatment. Microplastics 2025, 4, 76.
59. Enfrin, M.; Lee, J.; Le-clech, P.; Dumée, L. F. Kinetic and mechanistic aspects of ultrafiltration membrane fouling by nano- and microplastics. J. Membr. Sci. 2020, 601, 117890.
60. Enfrin, M.; Wang, J.; Merenda, A.; Dumée, L. F.; Lee, J. Mitigation of membrane fouling by nano/microplastics via surface chemistry control. J. Membr. Sci. 2021, 633, 119379.
61. Lin, D.; Lai, C.; Wang, X.; et al. Enhanced membrane fouling by microplastics during nanofiltration of secondary effluent considering secretion, interaction and deposition of extracellular polymeric substances. Sci. Total. Environ. 2024, 906, 167110.
62. Ko, J.; Kim, H.; Cha, H.; Kim, S.; Oh, H. Effect of microplastic size on biofouling in membrane bioreactors. J. Water. Process. Eng. 2025, 69, 106664.
63. Li, J.; Wang, B.; Chen, Z.; Ma, B.; Chen, J. Ultrafiltration membrane fouling by microplastics with raw water: behaviors and alleviation methods. Chem. Eng. J. 2021, 410, 128174.
64. Moritz, T.; Benfer, S.; Árki, P.; Tomandl, G. Influence of the surface charge on the permeate flux in the dead-end filtration with ceramic membranes. Sep. Purif. Technol. 2001, 25, 501-8.
65. Wang, J.; Shi, W.; Qiu, M.; Wen, J.; Chen, X.; Fan, Y. Construction of a Hydrophilic α-Al2O3 tight ultrafiltration membrane with a narrow pore size via Fe-lattice doping. ACS. Appl. Mater. Interfaces. 2026, acsami.5c20623.
66. So, Y.; Kim, S. Y.; Kim, S.; Park, C. Innovative approaches to high-speed ceramic membrane filtration for microplastic mitigation in urban wastewater treatment facilities. Sep. Purif. Technol. 2025, 363, 132013.
67. Xiong, X.; Bond, T.; Saboor Siddique, M.; Yu, W. The stimulation of microbial activity by microplastic contributes to membrane fouling in ultrafiltration. J. Membr. Sci. 2021, 635, 119477.
68. Xiong, X.; Siddique, M. S.; Graham, N. J.; Yu, W. Towards microplastics contribution for membrane biofouling and disinfection by-products precursors: the effect on microbes. J. Hazard. Mater. 2022, 426, 127797.
69. Maliwan, T.; Pungrasmi, W.; Lohwacharin, J. Effects of microplastic accumulation on floc characteristics and fouling behavior in a membrane bioreactor. J. Hazard. Mater. 2021, 411, 124991.
70. Wang, B.; Liu, X.; Chen, J.; Peng, D.; He, F. Composition and functional group characterization of extracellular polymeric substances (EPS) in activated sludge: the impacts of polymerization degree of proteinaceous substrates. Water. Res. 2018, 129, 133-42.
71. Mao, H.; Bu, J.; Da, X.; et al. High-performance self-cleaning piezoelectric membrane integrated with in-situ ultrasound for wastewater treatment. J. Eur. Ceram. Soc. 2020, 40, 3632-41.
72. Zhao, P.; Gao, Y.; Xu, G.; Zhang, E.; Liu, L.; Jin, S. Fabrication of Cu1.4Mn1.6O4 modified catalytic ozonation ceramic membrane for membrane fouling elimination and micro‐pollutant degradation. J. Am. Ceram. Soc. 2023, 106, 4089-102.
73. Zhu, Z.; Fan, C.; Qiu, M.; Chen, X.; Fan, Y. Construction of self-cleaning alumina ultrafiltration membrane on build-in ultrasonic porous barium titanate. Sep. Purif. Technol. 2025, 366, 132794.
74. Fan, C.; Lv, L.; Liu, M.; et al. Co-sintering of ZrO2/Al2O3 bi-layer ultrafiltration membranes supported on built-in piezoelectric quartz. J. Membr. Sci. 2026, 739, 124913.
75. Zhao, Y.; Gu, Y.; Liu, B.; et al. Pulsed hydraulic-pressure-responsive self-cleaning membrane. Nature 2022, 608, 69-73.
76. Lu, Y.; Dong, Y.; Liu, W.; Jin, Q.; Lin, H. Piezo-photocatalytic enhanced microplastic degradation on hetero-interpenetrated Fe1-xS/FeMoO4/MoS2 by producing H2O2 and self-Fenton action. Chem. Eng. J. 2025, 508, 160935.
77. Zhuang, S.; Wang, J. Interaction between antibiotics and microplastics: recent advances and perspective. Sci. Total. Environ. 2023, 897, 165414.
78. Hedtke, T.; Zhang, Y.; Beebe, M.; et al. Copper single-atom catalyst on nanoconfined ceramic membranes for Fenton-like removal of organic contaminants. ACS. EST. Eng. 2025, 5, 1171-9.
79. Lin, B.; Deng, X.; Chen, J.; Low, Z.; Zhong, Z.; Xing, W. Integration of oxidation processes and ceramic membrane filtration for advanced water treatment: a review of foulant-membrane interactions. Adv. Membr. 2025, 5, 100138.
80. Bortot Coelho, F. E.; Deemter, D.; Candelario, V. M.; Boffa, V.; Malato, S.; Magnacca, G. Development of a photocatalytic zirconia-titania ultrafiltration membrane with anti-fouling and self-cleaning properties. J. Environ. Chem. Eng. 2021, 9, 106671.
81. Sioulas, S.; Rapti, I.; Kosma, C.; Konstantinou, I.; Albanis, T. Photocatalytic degradation of the antidepressant drug Paroxetine using TiO2 P-25 under lab and pilot scales in aqueous substrates. Water. Emerg. Contam. Nanoplastics. 2025, 4, 5.
82. Song, M.; Park, J. Y. Removal of microplastics in a hybrid treatment process of ceramic Microfiltration and photocatalyst-mounted PES spheres with air backwashing. Membranes 2024, 14, 169.
83. Deemter, D.; Coelho, F. E. B.; Oller, I.; Malato, S.; Amat, A. M. Assessment of a novel photocatalytic TiO2-zirconia ultrafiltration membrane and combination with solar photo-Fenton tertiary treatment of urban wastewater. Catalysts 2022, 12, 552.
84. Bai, Z.; Gao, S.; Yu, H.; Liu, X.; Tian, J. Layered metal oxides loaded ceramic membrane activating peroxymonosulfate for mitigation of NOM membrane fouling. Water. Res. 2022, 222, 118928.
85. Chen, L.; Maqbool, T.; Nazir, G.; et al. Peroxymonosulfate activated by composite ceramic membrane for the removal of pharmaceuticals and personal care products (PPCPs) mixture: Insights of catalytic and noncatalytic oxidation. Water. Res. 2023, 229, 119444.
86. Yang, Y.; Li, H.; Fu, W.; et al. Large-scale deployment of single-atom catalysts via cross-scale confinement in ceramic membranes for advanced water treatment. Nat. Water. 2025, 3, 1281-90.
87. Mestre, S.; Gozalbo, A.; Lorente-ayza, M.; Sánchez, E. Low-cost ceramic membranes: a research opportunity for industrial application. J. Eur. Ceram. Soc. 2019, 39, 3392-407.
88. Nandi, B.; Uppaluri, R.; Purkait, M. Identification of optimal membrane morphological parameters during microfiltration of mosambi juice using low cost ceramic membranes. LWT. -. Food. Sci. Technol. 2011, 44, 214-23.
89. Bhide, B.; Stern, S. A new evaluation of membrane processes for the oxygen-enrichment of air. II. Effects of economic parameters and membrane properties. J. Membr. Sci. 1991, 62, 37-58.
90. Vasanth, D.; Uppaluri, R.; Pugazhenthi, G. Influence of sintering temperature on the properties of porous ceramic support prepared by uniaxial dry compaction method using low-cost raw materials for membrane applications. Sep. Sci. Technol. 2011, 46, 1241-9.
91. Elgamouz, A.; Tijani, N. From a naturally occurring-clay mineral to the production of porous ceramic membranes. Microporous. Mesoporous. Mater. 2018, 271, 52-8.
92. Fu, M.; Liu, J.; Dong, X.; Zhu, L.; Dong, Y.; Hampshire, S. Waste recycling of coal fly ash for design of highly porous whisker-structured mullite ceramic membranes. J. Eur. Ceram. Soc. 2019, 39, 5320-31.
93. Bissett, H.; Zah, J.; Krieg, H. Manufacture and optimization of tubular ceramic membrane supports. Powder. Technol. 2008, 181, 57-66.
94. Hubadillah, S. K.; Harun, Z.; Othman, M. H. D.; et al. Preparation and characterization of low cost porous ceramic membrane support from kaolin using phase inversion/sintering technique for gas separation: effect of kaolin content and non-solvent coagulant bath. Chem. Eng. Res. Des. 2016, 112, 24-35.
95. Achiou, B.; Elomari, H.; Bouazizi, A.; et al. Manufacturing of tubular ceramic microfiltration membrane based on natural pozzolan for pretreatment of seawater desalination. Desalination 2017, 419, 181-7.
96. Wang, S.; Tian, J.; Wang, Q.; Zhao, Z.; Cui, F.; Li, G. Low-temperature sintered high-strength CuO doped ceramic hollow fiber membrane: preparation, characterization and catalytic activity. J. Membr. Sci. 2019, 570-571, 333-42.
97. Zhu, W.; Liu, Y.; Guan, K.; Peng, C.; Qiu, W.; Wu, J. Integrated preparation of alumina microfiltration membrane with super permeability and high selectivity. J. Eur. Ceram. Soc. 2019, 39, 1316-23.
98. Hermabessiere, L.; Dehaut, A.; Paul-pont, I.; et al. Occurrence and effects of plastic additives on marine environments and organisms: a review. Chemosphere 2017, 182, 781-93.
99. Gottschalk, F.; Nowack, B. The release of engineered nanomaterials to the environment. J. Environ. Monit. 2011, 13, 1145.
100. Wu, N.; Yu, H.; Liu, Z.; et al. The underestimated environmental risk of tris (2-chloroethyl) phosphate photodegradation in aqueous environment induced by polystyrene microplastics. Water. Res. 2025, 273, 123048.
101. Fischer, M.; Scholz-böttcher, B. M. Simultaneous trace identification and quantification of common types of microplastics in environmental samples by pyrolysis-gas chromatography-mass spectrometry. Environ. Sci. Technol. 2017, 51, 5052-60.
102. Dazzi, A.; Prater, C. B. AFM-IR: Technology and applications in nanoscale infrared spectroscopy and chemical imaging. Chem. Rev. 2016, 117, 5146-73.
103. Kurouski, D.; Dazzi, A.; Zenobi, R.; Centrone, A. Infrared and Raman chemical imaging and spectroscopy at the nanoscale. Chem. Soc. Rev. 2020, 49, 3315-47.
104. Gillibert, R.; Balakrishnan, G.; Deshoules, Q.; et al. Raman Tweezers for small microplastics and nanoplastics identification in seawater. Environ. Sci. Technol. 2019, 53, 9003-13.
105. Materić, D.; Kasper-giebl, A.; Kau, D.; et al. Micro- and nanoplastics in Alpine snow: a new method for chemical identification and (semi)quantification in the nanogram range. Environ. Sci. Technol. 2020, 54, 2353-9.
106. Lin, Y.; Huang, X.; Liu, Q.; Lin, Z.; Jiang, G. Thermal fragmentation enhanced identification and quantification of polystyrene micro/nanoplastics in complex media. Talanta 2020, 208, 120478.
107. Zhou, X. X.; Hao, L. T.; Wang, H. Y. Z.; Li, Y. J.; Liu, J. F. Cloud-point extraction combined with thermal degradation for nanoplastic analysis using pyrolysis gas chromatography-mass spectrometry. Anal. Chem. 2018, 91, 1785-90.
108. Ivleva, N. P. Chemical analysis of microplastics and nanoplastics: challenges, advanced methods, and perspectives. Chem. Rev. 2021, 121, 11886-936.
109. Marine Anthropogenic Litter; Bergmann, M., Gutow, L., Klages, M., Eds.; Springer International Publishing, 2015.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data




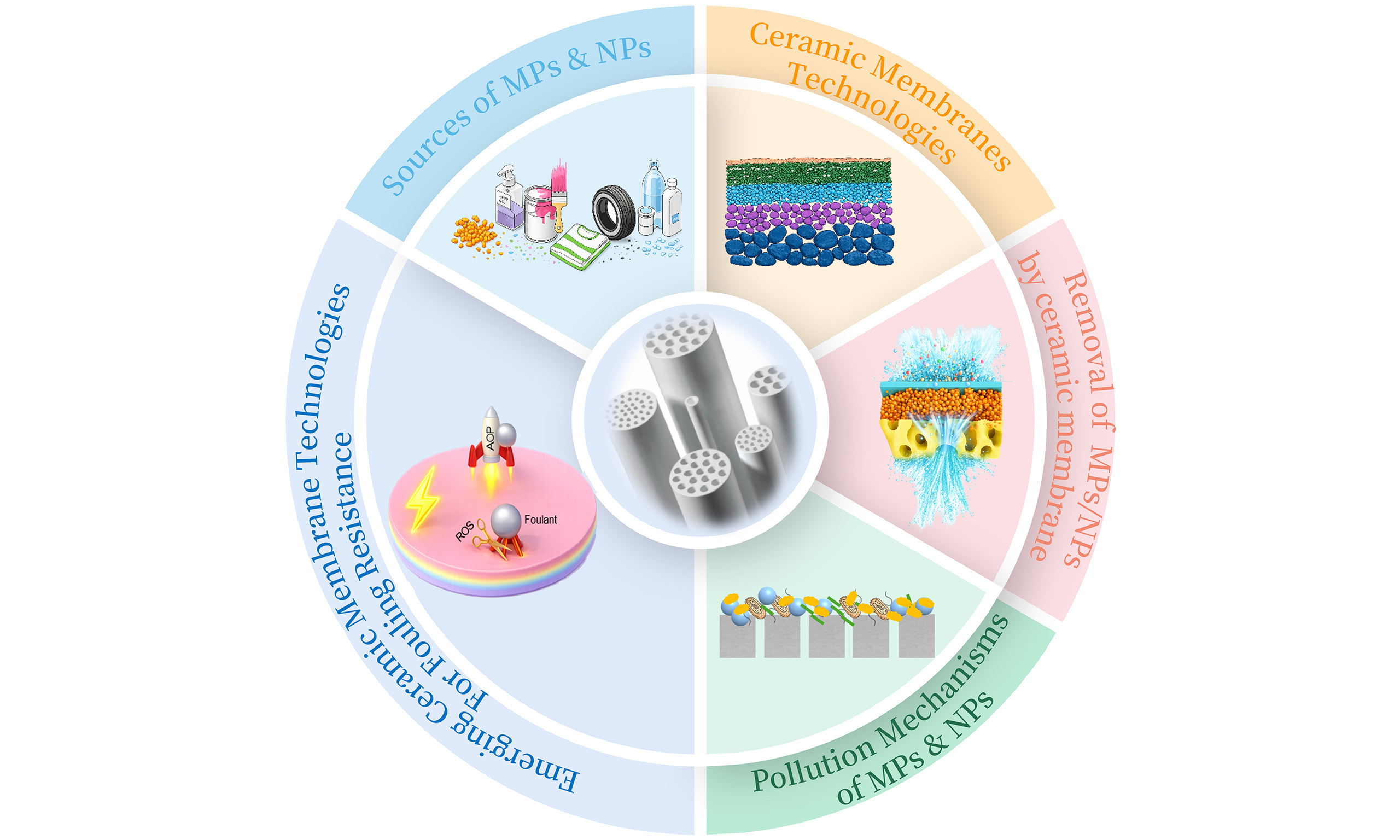
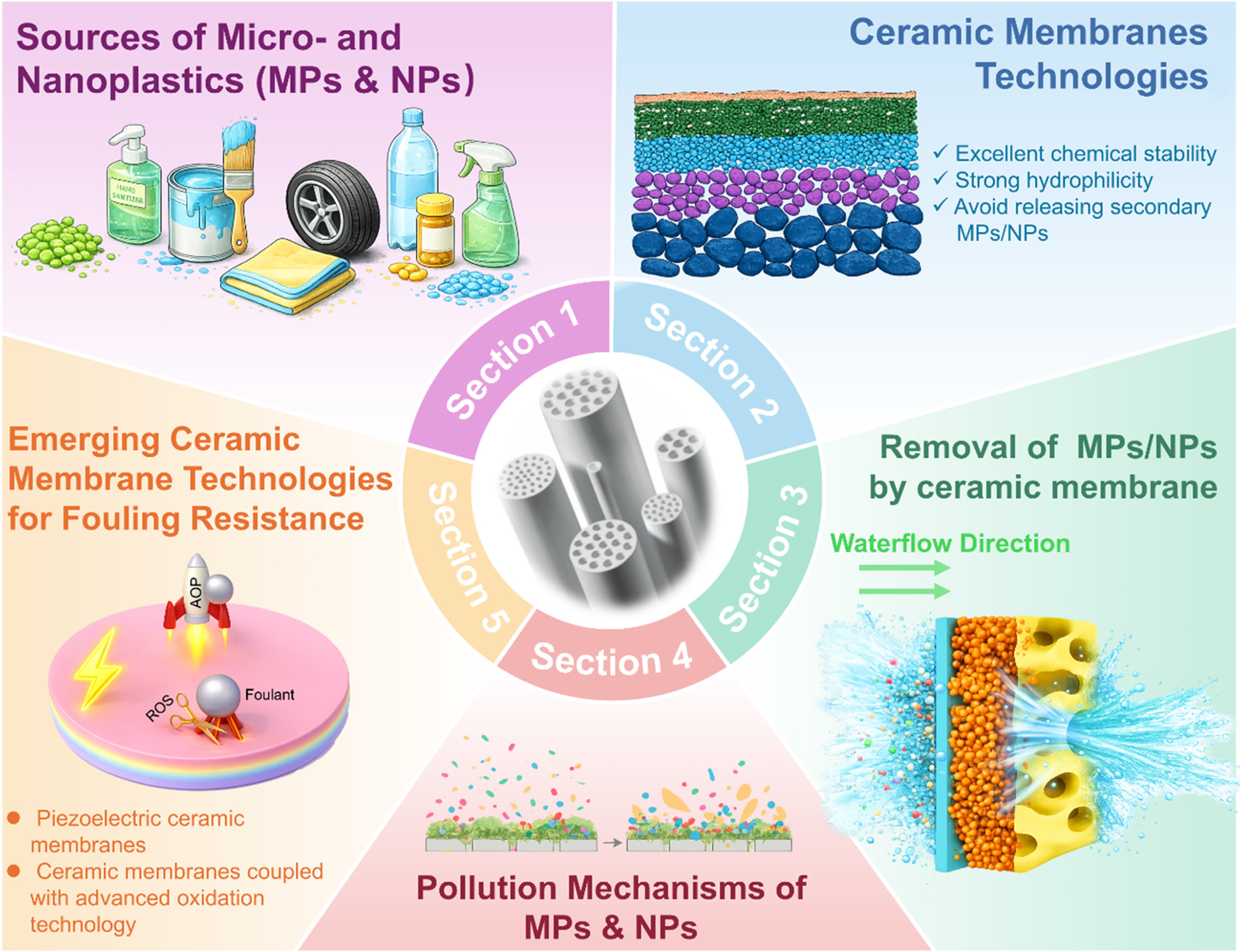
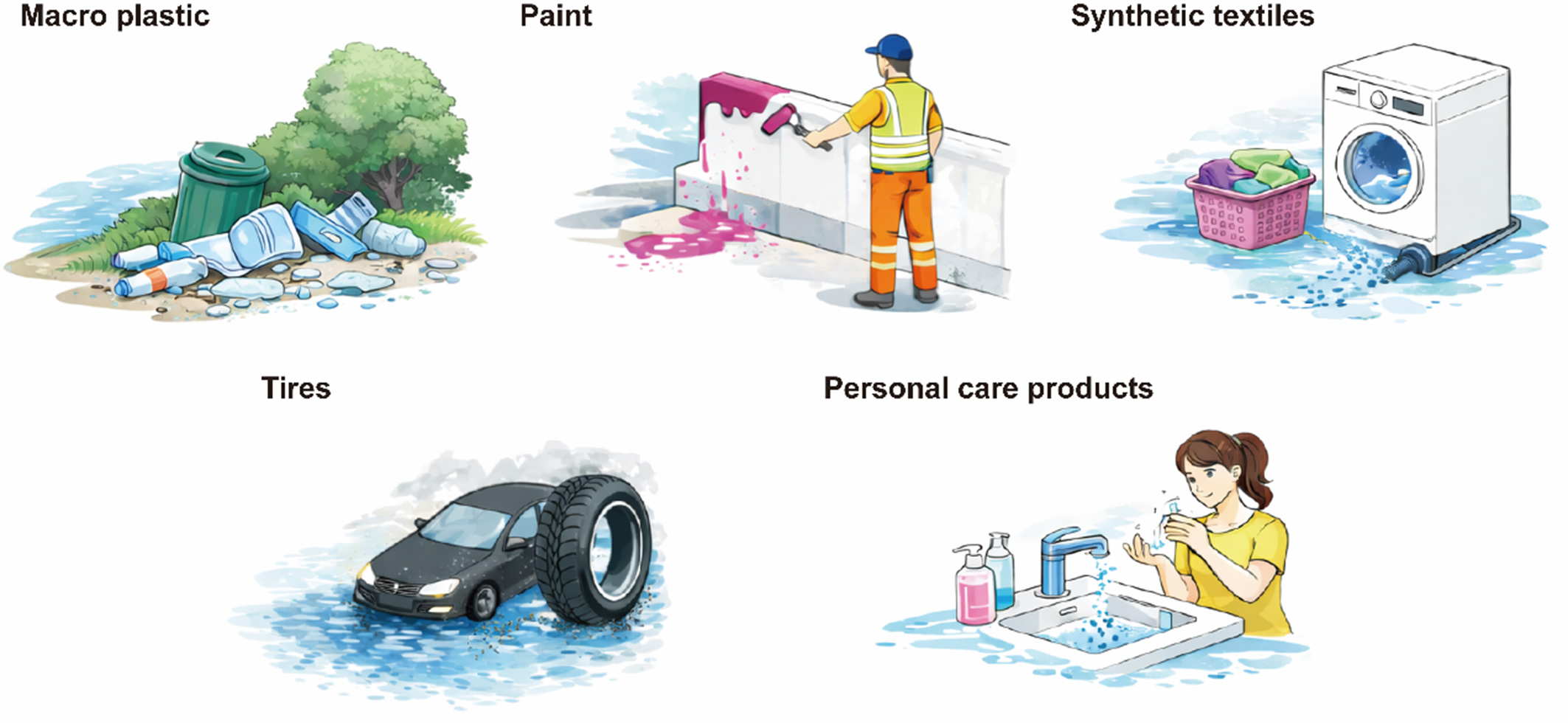
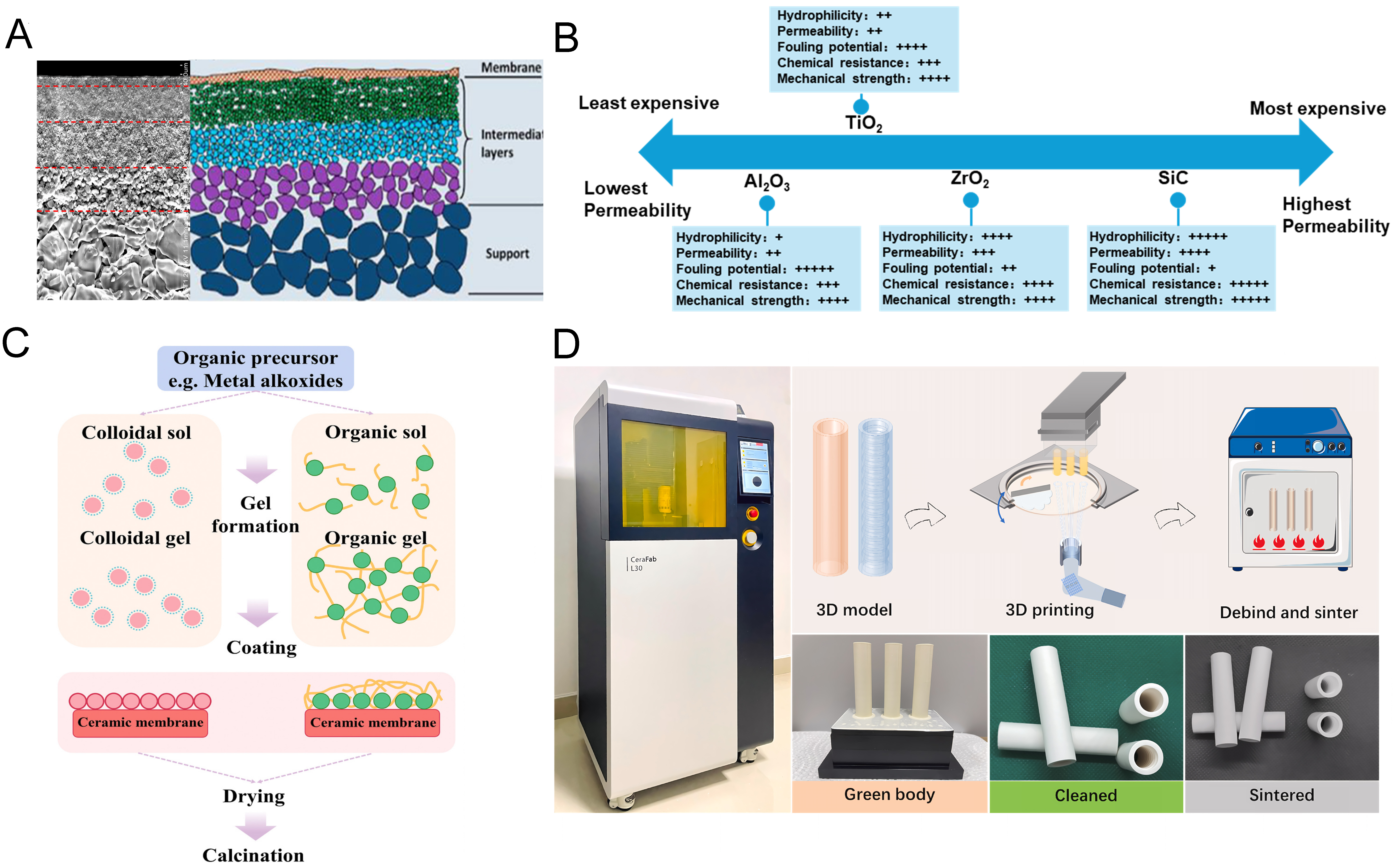
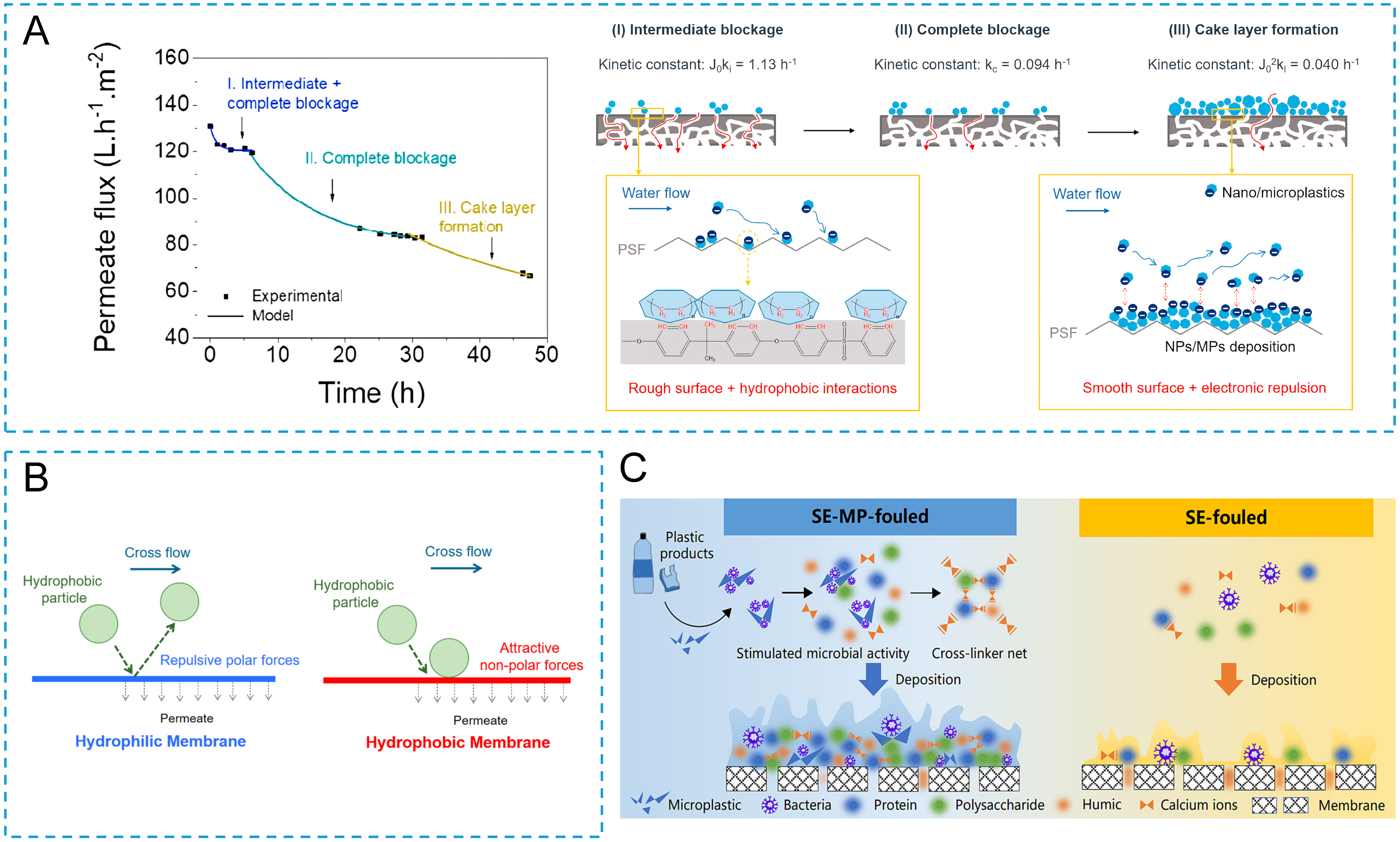
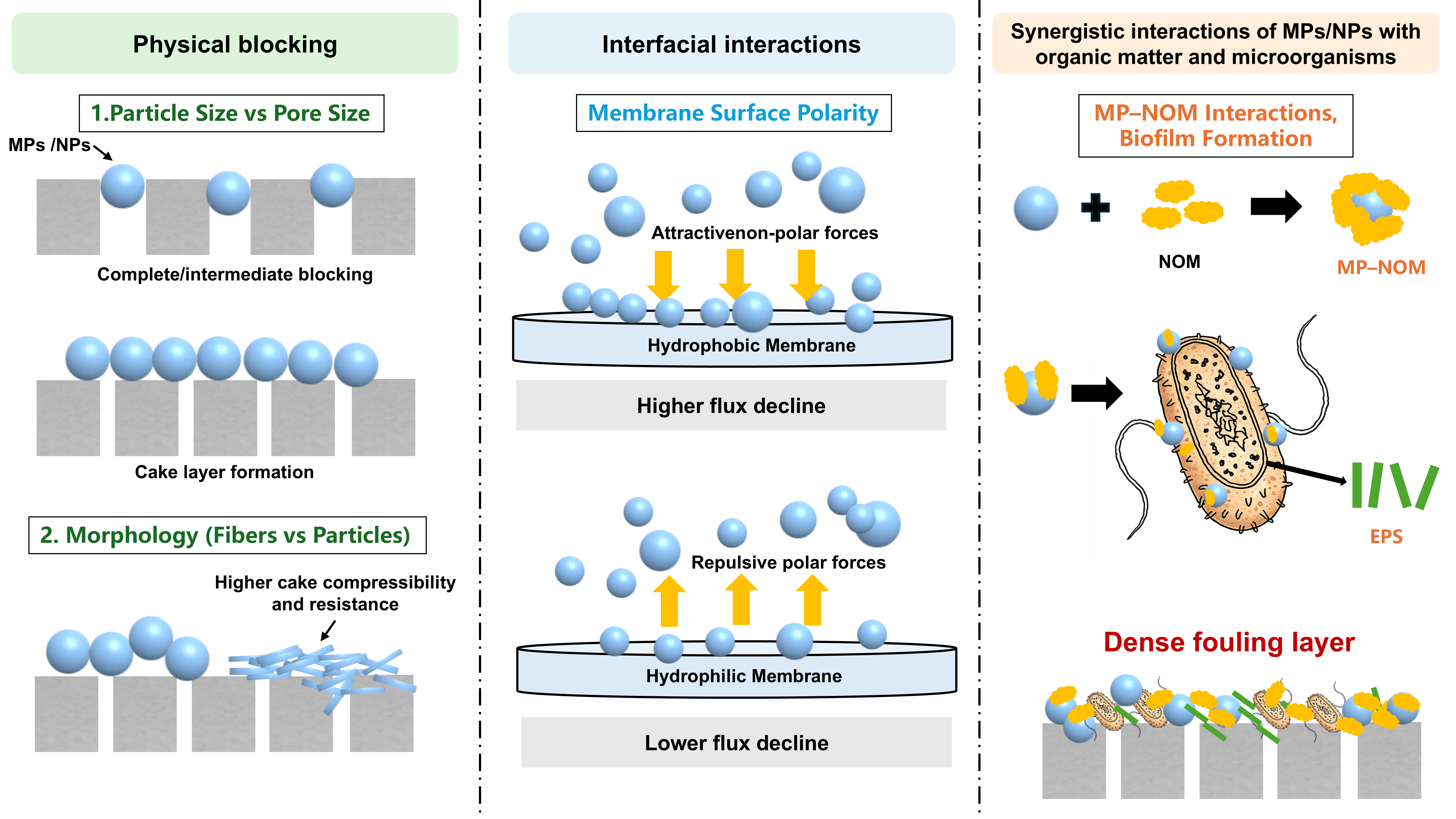
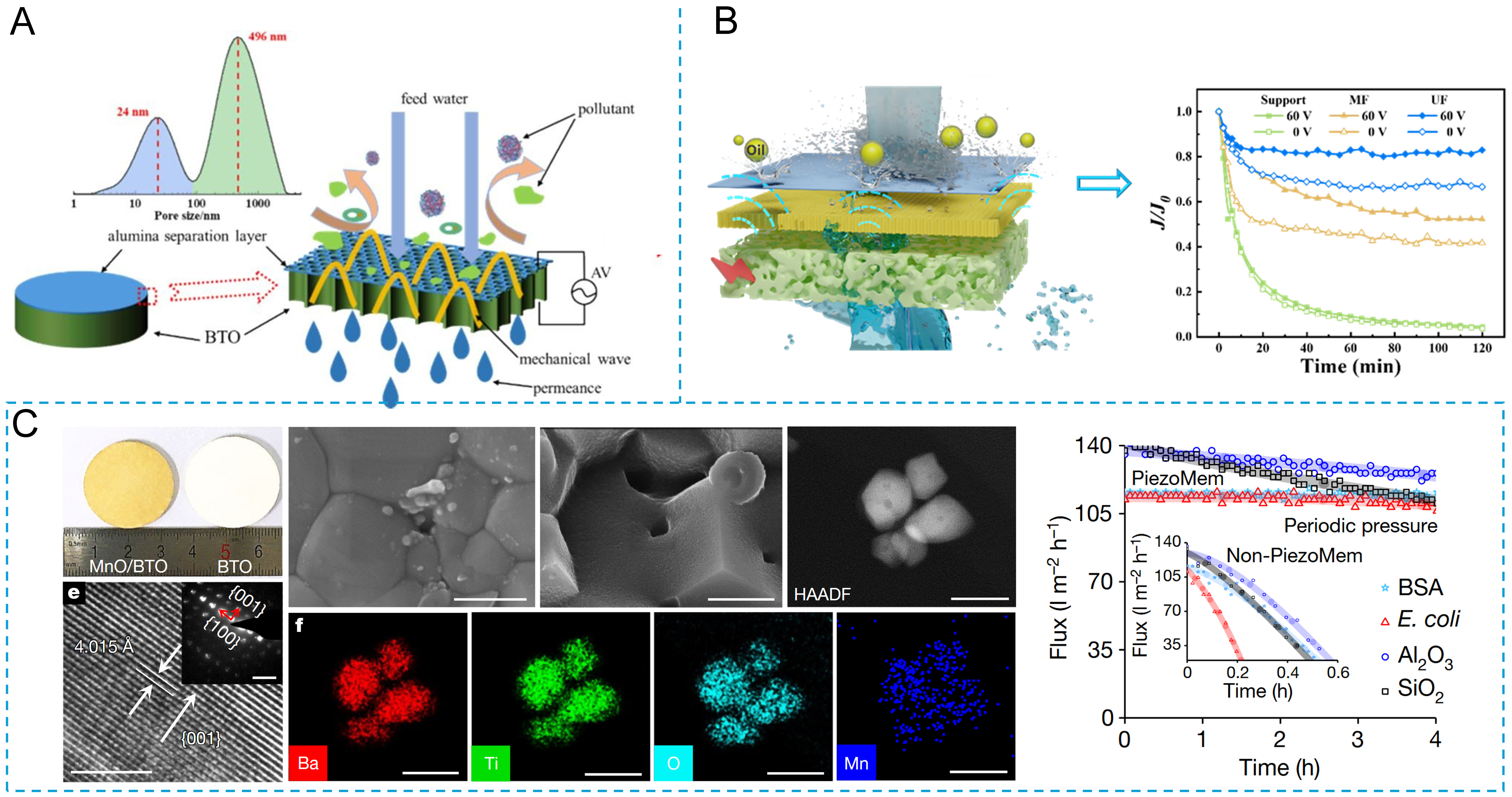
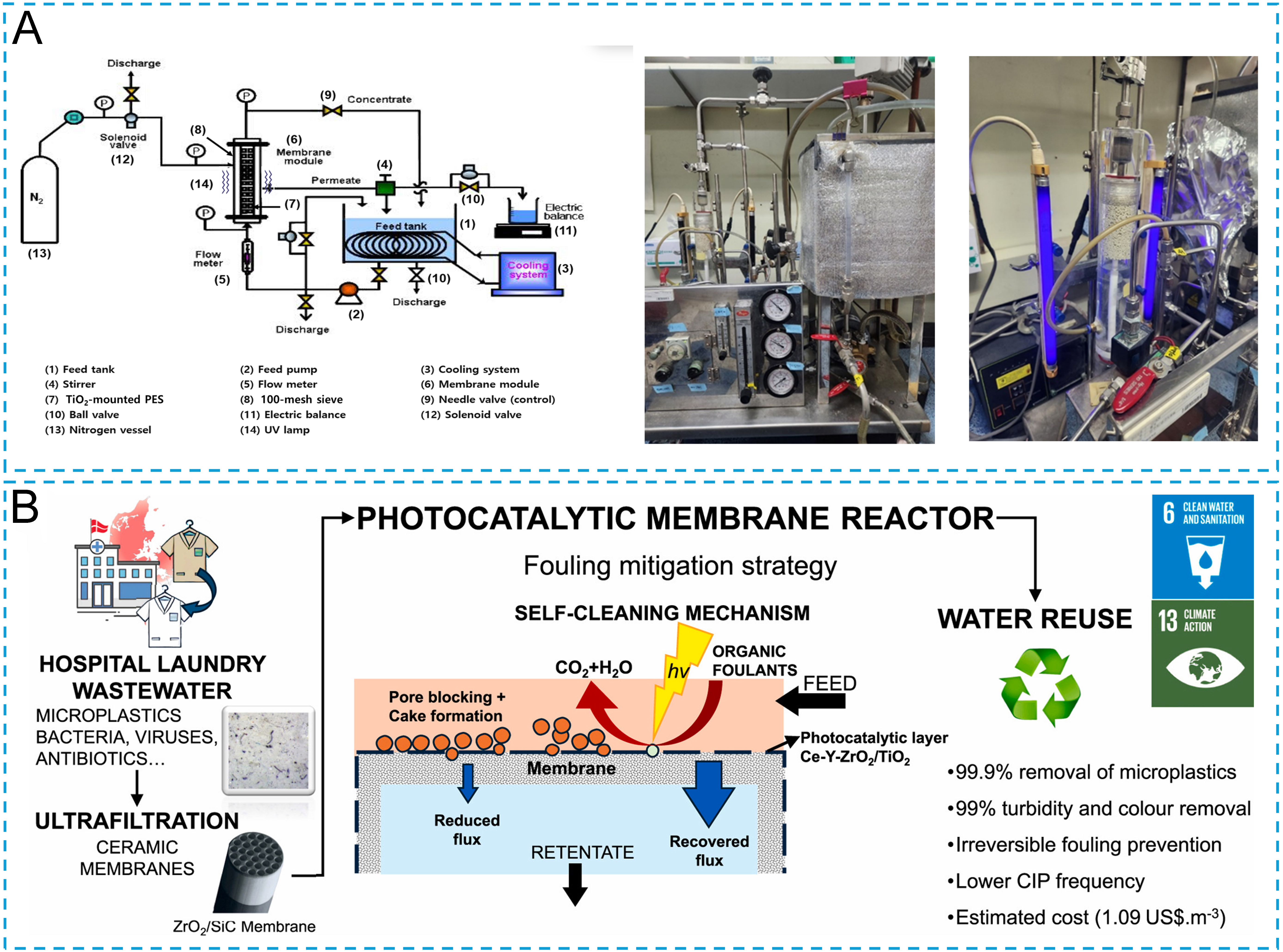
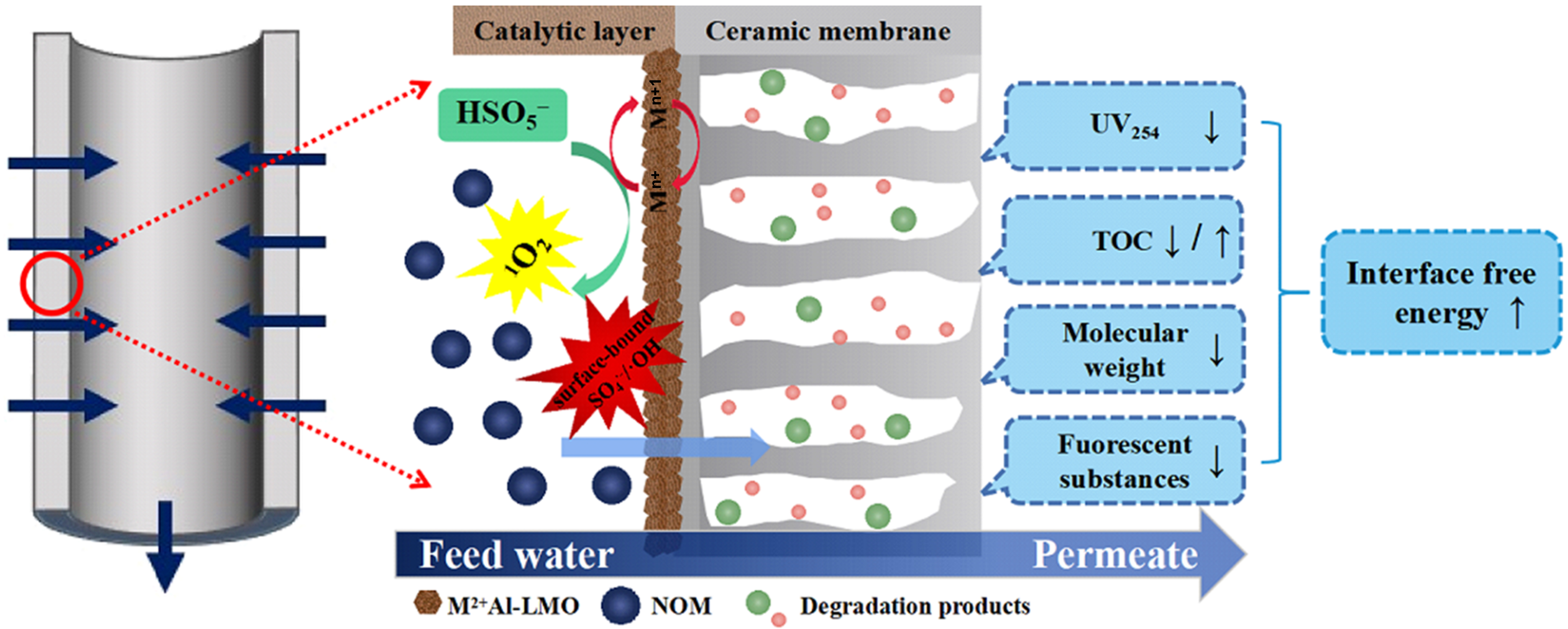











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].