Efficient degradation of carbamazepine by polydopamine-decorated Co/N@ZS activated peroxymonosulfate: performance and mechanism
Abstract
The development of high-performance catalysts for the activation of peroxymonosulfate remains a persistent challenge in the degradation of contaminants in waste water. Herin, a catalyst, Co/N@ZS-polydopamine, with excellent recovery and recyclability, was synthesized by employing the structurally ordered Co/N@ZS and polydopamine with strong adhesion. This catalyst effectively activated 0.30 g/L peroxymonosulfate for the degradation of 20 mg/L carbamazepine, demonstrating a remarkable degradation rate of 94.46%. The combination of electron paramagnetic resonance analysis and reactive oxygen species quenching research revealed that carbamazepine degradation is caused by both radical (SO4•- and O2•-) and non-radical (1O2 and electron transfer) processes. In addition, the possible degradation pathways were proposed based on the identification of intermediate products through liquid chromatography-mass spectrometry analyses. More importantly, the catalyst-assisted thin films exhibit exceptional stability and long-term efficacy in dynamic water treatment. The present study provides innovative perspectives on the application of highly efficient and stable catalysts in water treatment procedures for advanced oxidation processes.
Keywords
INTRODUCTION
The rapid development of industries has led to a gradual increase in the discharge of urban domestic sewage and industrial wastewater, making water environmental protection an issue of widespread concern in human society[1,2]. The wastewater contains numerous recalcitrant organic compounds, which exhibit bioaccumulation potential and pose risks to environmental integrity and human well-being. The utilization of advanced oxidation technologies (AOPs) based on peroxymonosulfate (PMS) represents a highly effective approach for the degradation of organic pollutants, as it generates extremely[3-5] reactive activated species. The activation of PMS is typically triggered by a range of stimuli including heat, ultrasound, ultraviolet radiation, alkali substances, carbon materials, and transition metal catalysts[6-8]. Consequently, active species generated from PMS include hydroxyl radicals (•OH), sulfate radicals (SO4•-), superoxide radicals (O2•-), and singlet oxygen (1O2)[9-11]. These species exhibit remarkable efficacy in degrading toxic compounds present in complex water environments, such as phenol, pesticides, antibiotics, and endocrine disruptors, thereby holding significant potential for broad applications in wastewater treatment[12-14].
The effectiveness of transition metal cobalt-based catalysts in activating PMS toward AOPs for the degradation of organic pollutants has been demonstrated by numerous studies[15,16]. For instance, Chen et al. synthesized a Co3O4-BC catalyst using straw biochar as a support via straightforward pyrolysis and subsequent modification process, enabling efficient activation of ofloxacin (OFX)[17]. Yao et al. employed lotus root starch as a precursor to fabricate carbon aerogels for the efficient activation of PMS on the cobalt species in order to degrade carbamazepine (CBZ) via non-radical pathways[18]. However, the leaching of high concentrations of Co ions remains a critical issue that hinders the practical application of Co-based catalysts in pollution degradation towards AOPs. Tuning the electronic field of Co sites by the promoters can enhance the stability of Co species. Cai et al. doped nickel into Co3O4 to form a bimetallic oxide, which promoted the stability and electron transfer pathway of the catalyst and improved the catalytic activity[12]. Guo et al. prepared Co-Mn spinel oxides through the utilization of the Co-Mn bimetallic synergistic effect, which exhibited remarkable efficiency and stability in pollutant degradation[19]. Additionally, Luo et al. have developed a non-metallic modification strategy to enhance the stability of Co species by harnessing N electronic confinement effects in the Co/N@Zr-SBA-15 (ZS) catalyst, thereby showcasing good durability and activation performance during the bisphenol A (BPA) degradation process[20].
In recent years, the surface film modification strategy has gained significant attention in research due to its ability to enhance the strong interaction between metal and support while preventing the leaching of metal ions[21,22]. The polydopamine (PDA) layer exhibits a multitude of functional groups, facilitating the formation of a robust chelating moiety and coordination with metal ions and metal oxides through covalent bonds and hydrogen interactions with diverse solid materials[23,24]. Additionally, surface film modification of PDA can also alter the electronic structure and coordination environment of active metal species, thereby exhibiting remarkable catalytic ability for the selective activation of PMS[25,26]. Furthermore, PDA does not require pretreatment and can adhere well to various substrates under alkaline conditions, effectively avoiding the leaching of metal ions. Therefore, the development of surface thin film decoration catalysts is expected to facilitate enhanced activity and stability in PMS activation towards AOPs.
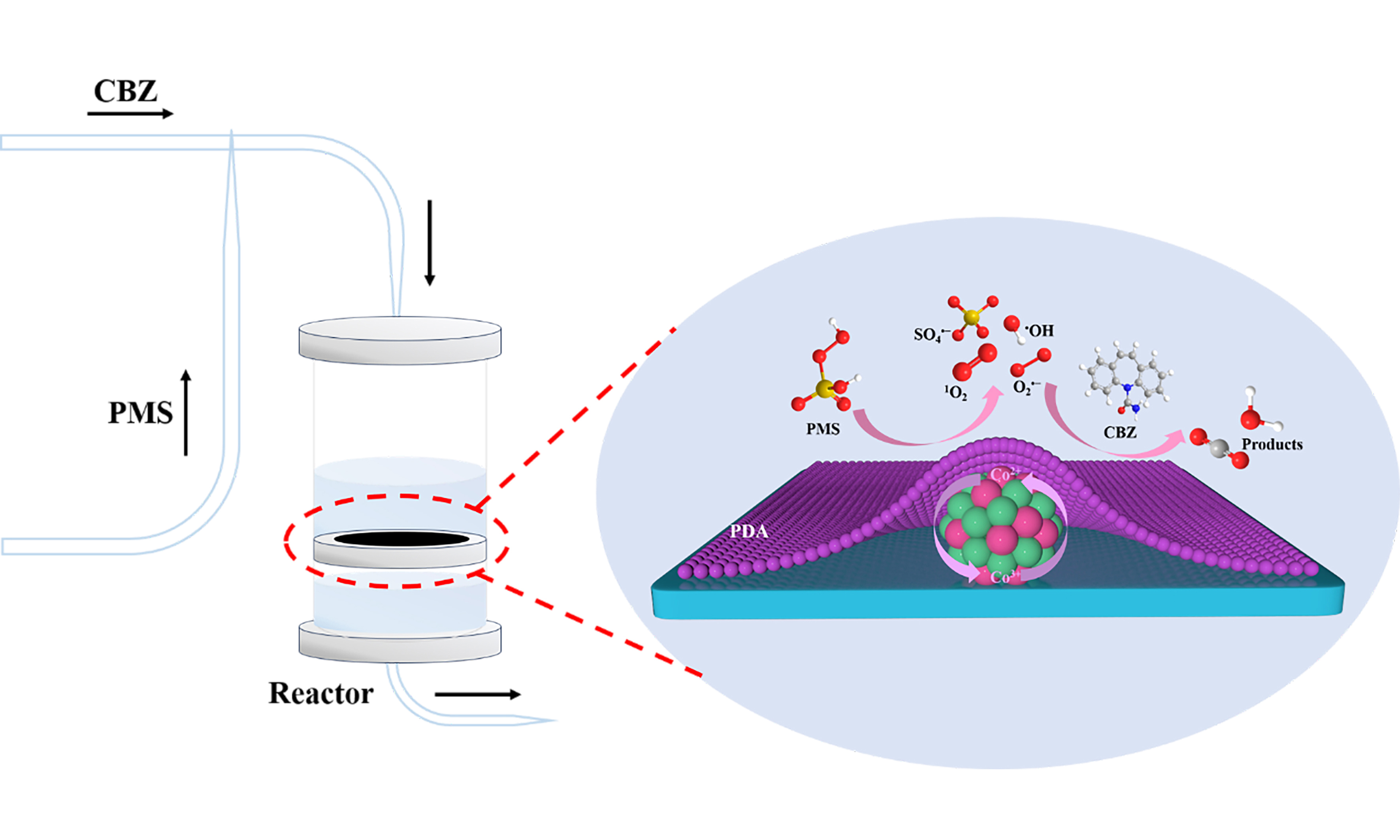
In this study, we presented a highly efficient and remarkably stable Co/N@ZS-PDA catalyst for the activation of PMS in order to degrade CBZ, achieved through a surface film modification strategy utilizing dopamine (DA) and Co/N@ZS as precursors. By examining the degradation of CBZ using various systems, the preliminary discussion focused on the mode and mechanism of PMS activation by Co/N@ZS-PDA. The elements and groups in Co/N@ZS-PDA were investigated using X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FT-IR) techniques, enabling a comprehensive analysis of the complexation between surface functional groups of Co/N@ZS-PDA and metal cobalt species. The Co/N@ZS-PDA films were prepared to investigate their dynamic degradation properties and assess their practical application value. The impact of the initial pH of the solution, catalyst dosage, PMS concentration, and coexisting ions on the activation process was investigated. This study aims to elucidate the relationship between the surface thin film decoration catalysts and structure-activity, while also providing novel concepts and insights for enhancing comprehension of high-performance Co-based catalyst design.
EXPERIMENTAL
Reagents
The chemical reagents were listed in the Supplementary Text 1.
Synthesis of the catalysts
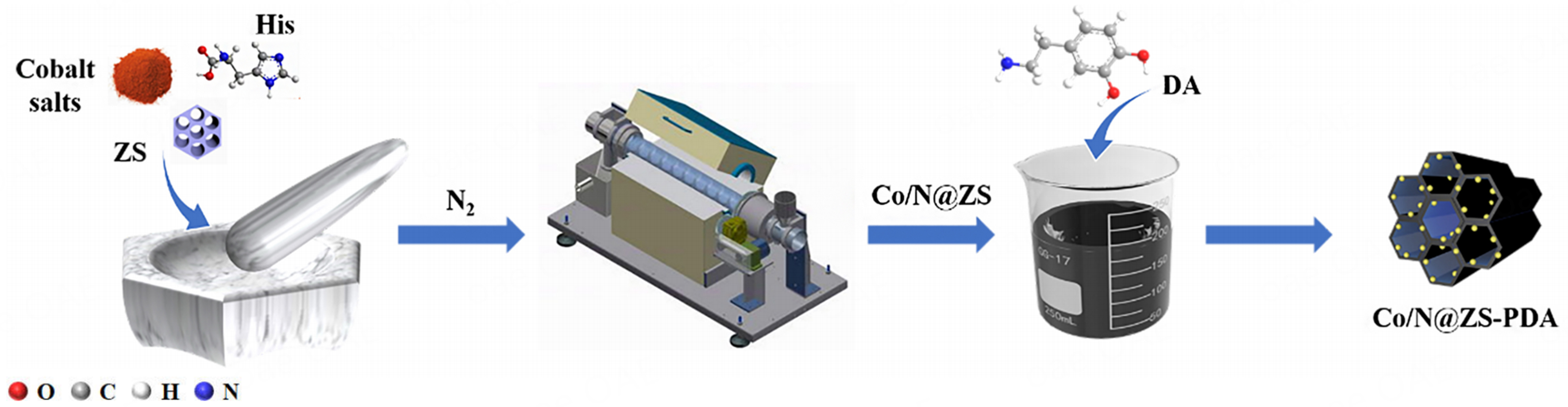
The synthetic procedures for the preparation of ZS and Co/N@ZS were described in Supplementary Text 2. In brief, a mixture of Co/N@ZS (0.1 g) and DA hydrochloride (0.141 g) was prepared in 100 mL of ultrapure water, and the pH was adjusted to 8.5 using tris (0.12 g). The suspension was agitated at a speed of 180 rpm/min for 12 h under an ambient air atmosphere. Subsequently, the resulting mixture was separated using a centrifuge and subjected to three rounds of washing with deionized water and ethanol (EtOH). Finally, the Co/N@ZS-PDA material was dried in an oven set at 60°C for future utilization.
Experimental procedure
The prepared catalysts were evaluated by assessing their performance in the decomposition of CBZ, ciprofloxacin (CIP), norfloxacin (NOR), and tetracycline (TC). All experiments were conducted in 250 mL beakers. Specifically, precise amounts of catalyst and PMS were added to 100 mL of CBZ solution, with a consistent stirring rate maintained at 180 rpm/min for 60 min (T = 25°C). As the reaction progressed, 2 mL aliquots of the solution were periodically sampled and filtered through a 0.22 μm Teflon syringe filter for analysis. Each experiment was performed in triplicate, with error bars indicating the standard deviation of the results. (Supplementary Table 1 and Supplementary Text 3 for detailed information).
Analytical methods
The methods for characterization are provided in Supplementary Text 4.
RESULTS AND DISCUSSION
Characterization of catalyst
The surface morphology of the ZS, Co/N@ZS and Co/N@ZS-PDA were characterized by scanning electron microscopy (SEM) [Scheme 1]. As shown in Figure 1A and B, ZS with a clear lamellar structure was successfully synthesized. The incorporation of Co and N species did not lead to distinct alterations in morphology [Figure 1C and D], indicating the well-preserved flake structure of Co/N@ZS. The SEM images of Co/N@ZS-PDA are presented in Figure 1E and F, illustrating the morphological changes following the self-polymerization of DA on the surface of the Co/N@ZS material. Consequently, the composite material exhibited a smooth surface due to its tightly interconnected layered structures, which can be attributed to the robust adhesion properties of PDA. Moreover, PDA also facilitated the stabilization of metal on the catalyst. The EDS elemental mapping spectrum [Supplementary Figure 1] revealed a uniform distribution of elements. In addition, the introduction of the PDA layer increased the nitrogen content on the Co/N@ZS from 3.83 wt.% to 6.83 wt.% [Supplementary Table 2]. The ordered mesoporous structural features and hexagonal sheet shape of the ZS were clearly demonstrated in Figure 1G and H. The transmission electron microscopy (TEM) images of Co/N@ZS-PDA [Figure 1I-L] revealed a surface layer coated with PDA, which had an average thickness of 5 nm. Furthermore, it can be observed that the mesoporous structure remains well-preserved even after the material is coated with PDA.
Figure 1. The SEM images depict the morphology of (A and B) ZS, (C and D) Co/N@ZS, and (E and F) Co/N@ZS-PDA. It should be noted that images (B), (D), and (F) provide magnified views of the rectangular regions in images (A), (C), and (E), respectively, with the inset showing a digital photo of the sample; The TEM images of (G and H) ZS and (I-L) Co/N@ZS-PDA are presented, with the magnified images (H), (J), and (L) corresponding to the rectangular regions in (G), (I), and (K), respectively. ZS: Zr-SBA-15; SEM: scanning electron microscopy; PDA: polydopamine; TEM: transmission electron microscopy.
Scheme 1. Preparation diagram of Co/N@ZS-PDA samples. ZS: Zr-SBA-15; DA: dopamine; PDA: polydopamine.
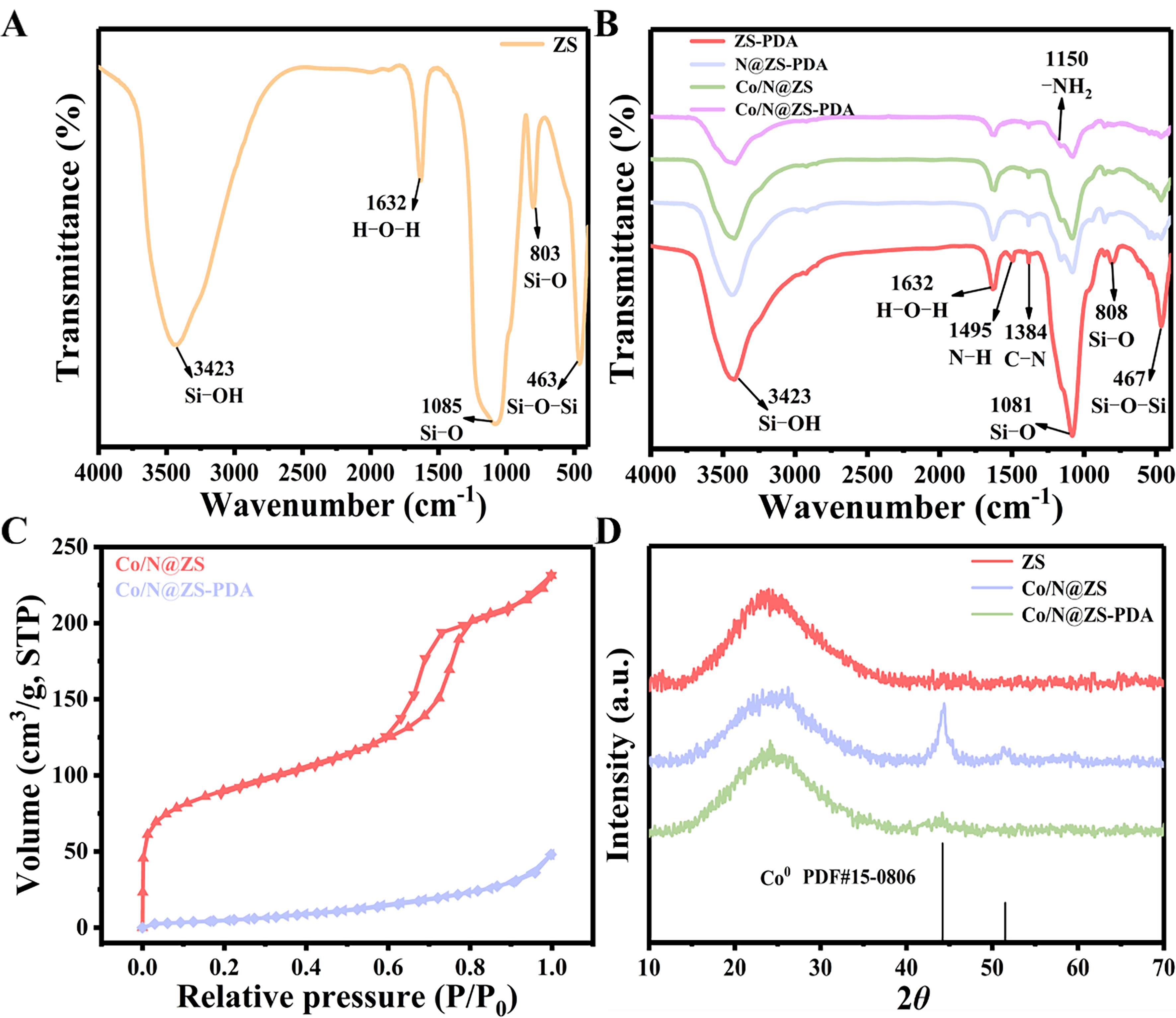
The FT-IR spectra of different materials are present in Figure 2A and B. The spectrum of ZS was shown in Figure 2A, with the presence of a peak at 3,423 cm-1 attributed to the stretching vibration of Si–OH[21]. Additionally, another peak at 1,632 cm-1 is observed, which can be ascribed to the adsorption of water on the surface. The observed bands at 803 cm-1, 1,085 cm-1 and 467 cm-1 are attributed to the antisymmetric stretching vibration peak of Si–O, stretching vibration of Si–O, and bending vibration of Si–O–Si, respectively. Notably, the band was observed at 1,384 cm-1 [Figure 2B] upon addition of L-His, which can be attributed to the C–N bond formation. In addition, a noticeable shift in bands occurred at 1,495 cm-1 and 1,150 cm-1, corresponding to N–H bonding and –NH2 bonding, respectively, indicating successful surface modification by PDA[27].
Figure 2. (A) FT-IR spectra of ZS and (B) as-prepared composites; (C) N2 adsorption/desorption isotherms; (D) XRD spectra of as-prepared catalysts. ZS: Zr-SBA-15; PDA: polydopamine; FT-IR: Fourier transform infrared spectroscopy; XRD: X-ray diffraction.
The N2 adsorption-desorption isotherm spectrum in Figure 2C reveals that Co/N@ZS possesses the characteristic features of a typical mesoporous structure (type IV isotherm) with distinct hysteresis loops. After PDA coating, Co/N@ZS-PDA exhibited a typical type II isotherm, suggesting a characteristic physical adsorption process on non-porous or macroporous adsorbents. The pore structure of the material transformed from mesoporous to non-porous or macropores after coating, attributed to the formation of a dense PDA film as the coating that blocked the pore channels on the surface of the Co/N@ZS, resulting in a reduction in the specific surface area of Co/N@ZS-PDA material.
The X-ray diffraction (XRD) spectra of various materials are presented in Figure 2D. It can be observed that within the range of 15° to 30°, there is a relatively broad "steamed bread peak" indicative of the amorphous structure of SiO2[20]. Similarly, following the introduction of Co species and utilization of PDA-modified materials, the steamed bread peak persisted, thereby further substantiating the presence of amorphous SiO2 in Co/N@ZS-PDA. The presence of two distinct diffraction peaks at 2θ = 44.2° and 51.5°, corresponding to the (111) and (200) crystallographic planes of the cobalt species with a crystalline structure (PDF#15-0806), respectively, suggests the presence of Co0 species on the catalyst. However, with the PDA coating in the Co/N@ZS, the peak of Co0 decreased, which may be the conversion of Co0 to CoOx species. The implementation of PDA tunes the chemical states of Co species, thereby enhancing the interaction between Co and the support and ultimately bolstering the catalyst’s stability.
Catalytic activity
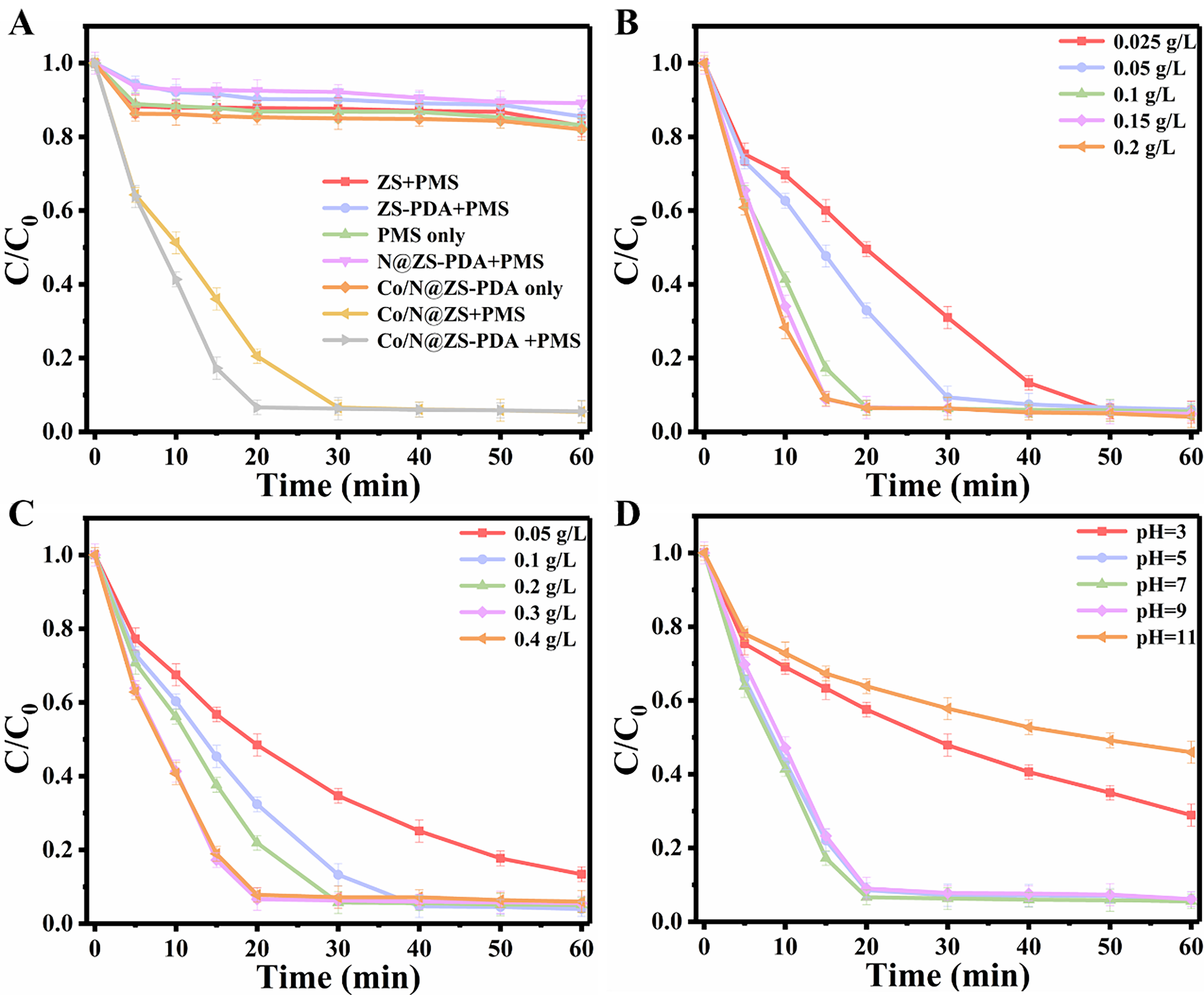
The degradation performance of various catalyst systems on pollutants was investigated [Figure 3A], including PMS, ZS/PMS, ZS-PDA/PMS, N@ZA-PDA/PMS, Co/N@ZS-PDA, Co/N@ZS/PMS, and Co/N@ZS-PDA/PMS systems. If only PMS or catalyst was present, only a limited amount of CBZ was degraded. When PMS existed alone, it remains inactive without the presence of a catalyst, resulting in fewer active free radicals and limited degradation of pollutants through self-decomposition of PMS. In the presence of only the catalyst, pollutants were primarily removed through static adsorption by the catalyst. The degradation performance of ZS/PMS, ZS-PDA/PMS, and N@ZA-PDA/PMS for CBZ remained low in the catalytic system of undoped cobalt, indicating that Co species played a crucial role in activating PMS. The Co/N@ZS/PMS and Co/N@ZS-PDA/PMS systems exhibit a CBZ removal efficiency of over 94% within 30 min. Following PDA coating, the catalyst’s performance was further enhanced
Figure 3. The impact of operational parameters on system performance: (A) various systems; (B) catalysts quality; (C) PMS dosage, and (D) pH value. Experimental conditions: [CBZ] = 20 mg/L, [PMS] = 0.30 g/L for conditions (A), (B), and (D); [Cat.] = 0.10 g/L for experiments (A), (C), and (D); and initial pH = 6.38 for tests (A), (B), and (C). Each experiment was performed in triplicate, with error bars indicating the standard deviation of the results. ZS: Zr-SBA-15; PDA: polydopamine; PMS: peroxymonosulfate; CBZ: carbamazepine; Cat.: catalyst.
Effect of parameters
Effects of Co/N@ZS-PDA and PMS dose
The removal efficiency of CBZ reached 94.47% and the reaction rate increased from 0.03265 to 0.14895 min-1 with a rise in catalyst dosage from 0.025 g/L to 0.15 g/L, as illustrated in Figure 3B and Supplementary Figure 3. This can be explained by the synergistic degradation reaction occurring at multiple sites on the surface of the PDA layer, resulting in an increase in active sites that clearly reduces the catalytic energy barrierand promotes rapid reaction kinetics[21]. However, when the catalyst dosage was further increased from 0.1 to 0.2 g/L, there was no evident improvement observed in both removal rate and reaction rate of CBZ, implying that the active sites present on the surface of a Co/N@ZS-PDA catalyst with a dosage of 0.10 g/L were sufficient for facilitating this reaction.
The impact of PMS dosage on the system was investigated as shown in Figure 3C and Supplementary Figure 4. As expected, as the concentration of PMS increased from 0.05 to 0.3 g/L, the removal efficiency of CBZ improved from 86.60% to 94.47%, accompanied by a rise in the reaction rate to 0.13467 min-1. This can be attributed to the enhanced contact between Co/N@ZS-PDA’s active sites and PMS at higher dosages, resulting in more generation of active free radicals that improve degradation efficiency[30]. However, further increasing the dosage of PMS resulted in a slight decrease in degradation efficiency and reaction rate due to potential self-quenching effects among excessive free radicals within the system, thereby affecting the catalytic performance of Co/N@ZS-PDA[31], as determined by
HSO5- + SO4•- → SO5•- + HSO4- (2)
SO4•- + SO4•- → S2O82- + SO4•- (3)
SO4•- + S2O82- → SO42- + S2O8•- (4)
Therefore, a PMS dosage of 0.3 g/L was selected for subsequent experiments.
Effect of initial pH
The investigation into the impact of the initial pH of the solution on the catalytic degradation of organic pollutants in the system is also pivotal [Figure 3D]. The Co/N@ZS-PDA demonstrated nearly identical CBZ removal efficiency within the pH range of 5-9. However, the degradation rate of CBZ was hindered at pH = 3 and 11. The primary interaction between CBZ and PDA predominantly occurs through π-π stacking on the catalyst surface, resulting in consistent adsorption capacity across a wide range of pH values (3-11) due to the electrically neutral distribution of CBZ molecules under this reaction condition. Consequently, the degradation rate of CBZ remains relatively stable within this pH range. In an acidic environment (pH = 3), a substantial amount of H+ forms hydrogen bonds with the peroxide bond (–O–O–) of HSO5- in PMS, thereby impeding its interaction with the catalyst and resulting in a reduction in degradation efficiency[32]. Under highly alkaline conditions (pH = 11), excessive OH- promotes the conversion of SO4•- to •OH[19], as given in
SO4•- + OH- → SO42- + •OH (5)
SO5- + •OH → SO4•- + OH- (6)
Moreover, hydroxylation by OH- on the surface of Co/N@ZS-PDA enhances electrostatic repulsion between PMS and the material surface, consequently hindering electron transfer processes. Additionally, the precipitation formation of Co(OH)2 results in a decrease in catalytic performance.
Effect of inorganic anions and NOM
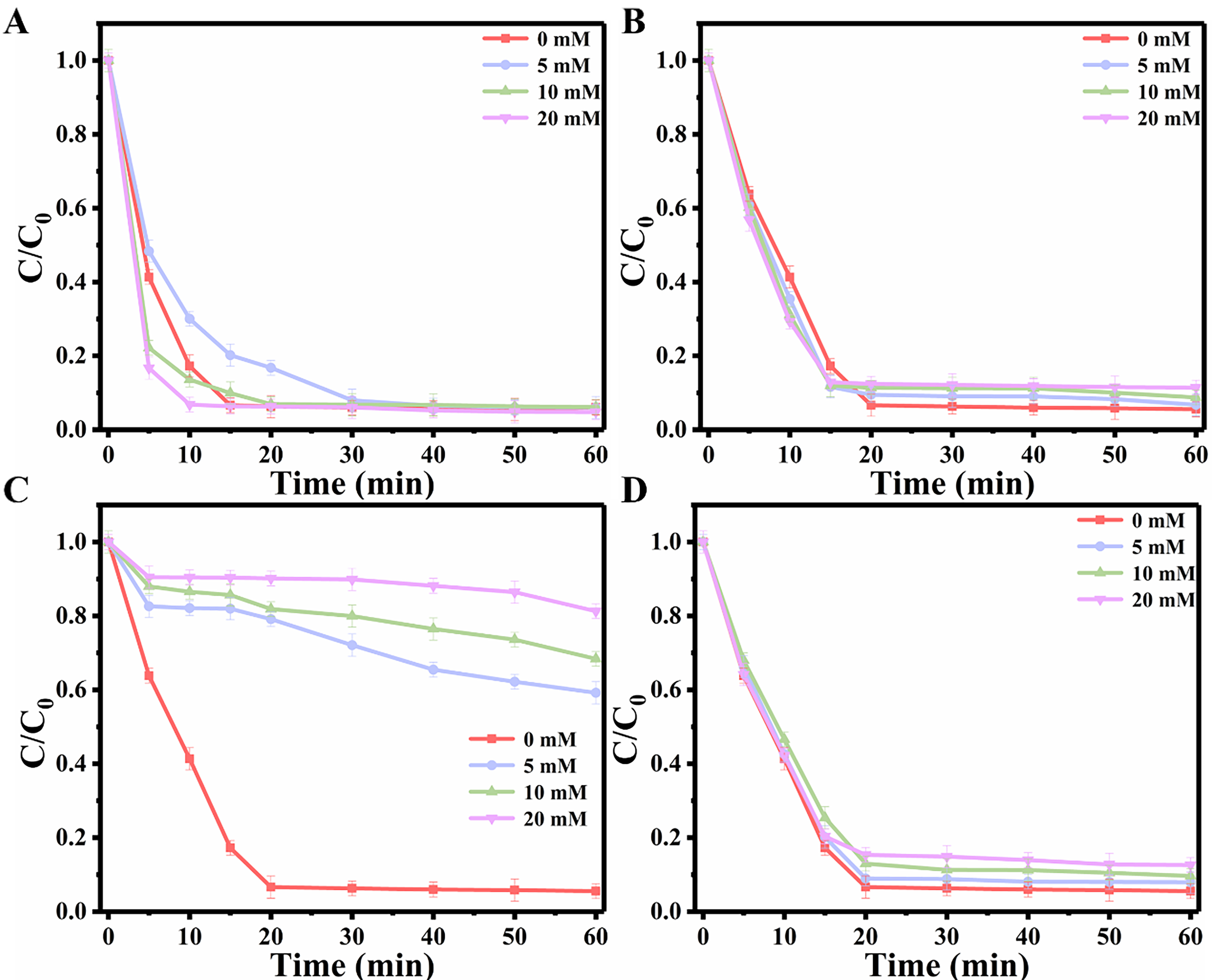
The actual water contains a terrific amount of inorganic anions (Cl-, HCO3-, H2PO4-, and NO3-), which can function as scavengers of free radicals and influence the catalytic efficiency. Hence, the investigation of the impact of inorganic anions on CBZ degradation is deemed necessary. As shown in Figure 4A, the presence of Cl- at low concentrations slightly hindered the efficiency of CBZ degradation. The decline in catalytic performance can be attributed to the reaction between Cl- and SO4•- and •OH, resulting in the formation of weakly oxidizing species such as Cl•, ClOH•- and Cl2•-[33], as given in
Figure 4. Effect of (A) Cl-, (B) H2PO4-, (C) HCO3-, and (D) NO3- on the CBZ degradation under specified experimental conditions: [CBZ] = 20 mg/L, [PMS] = 0.30 g/L, Initial pH = 6.38, [Cat.] = 0.10 g/L. Each experiment was performed in triplicate, with error bars indicating the standard deviation of the results. PMS: Peroxymonosulfate; CBZ: carbamazepine; Cat.: catalyst.
Cl- + SO4•- → Cl• + SO42- (7)
Cl- + Cl• → Cl2•- (8)
Cl- + •OH → ClOH•- (9)
ClOH•- + H+ → Cl• + H2O (10)
However, at higher concentrations (20 mM), the presence of Cl- accelerated the oxidation process, resulting in a remarkable degradation rate of CBZ exceeding 93% within just 10 min. This can be attributed to the synergistic degradation effect exerted by reactive chlorine radicals generated during the reaction[34-36]. With the increase of H2PO4- concentration in the reaction system, the degradation of CBZ was slightly inhibited [Figure 4B], which can be attributed to the competition between H2PO4- and pollutants for SO4•- and •OH is responsible for influencing the degradation efficiency[37], as given in
H2PO4- + SO4•- → H2PO4• + SO42- (11)
H2PO4- + •OH → H2PO4• + OH- (12)
The presence of HCO3- in the reaction system exhibited a pronounced inhibitory effect, and this inhibition increased with higher concentrations of HCO3-. The observed phenomenon can be ascribed to the quenching of radicals [Figure 4C] and reactions given in[38]
HCO3- + SO4•- → HCO3• + SO42- (13)
HCO3- + •OH → CO3•- + H2O (14)
Specifically, the HCO3- not only reacts with SO4•- and •OH to generate radicals (CO3-) with low oxidation potential, but also interacts with 1O2 to impede the degradation pathway of non-free radicals, thereby hindering the overall degradation process. Similar to H2PO4-, the degradation efficiency of CBZ was hindered by the negative correlation between the concentration of NO3-, as illustrated in Figure 4D.
The presence of natural organic matter (NOM) in aqueous environments hinders the degradation efficiency of target pollutants by quenching free radicals and occupying active sites on catalyst surfaces. Supplementary Figure 5 illustrates the impact of humic acid (HA), which is the most representative NOM, on degradation performance. As the HA concentration was elevated from 0 to 20 mg/L, inhibition also increased accordingly. Upon the addition of 20 mg/L HA, the CBZ degradation efficiency decreased to 81.41%. This can be attributed to the adherence of HA to the Co/N@ZS-PDA surface through π-π linkages, thereby inhibiting interactions between the catalyst, CBZ, and PMS[39,40]. Furthermore, HA competes with CBZ for radicals, consequently reducing the overall degradation efficiency of the reaction system.
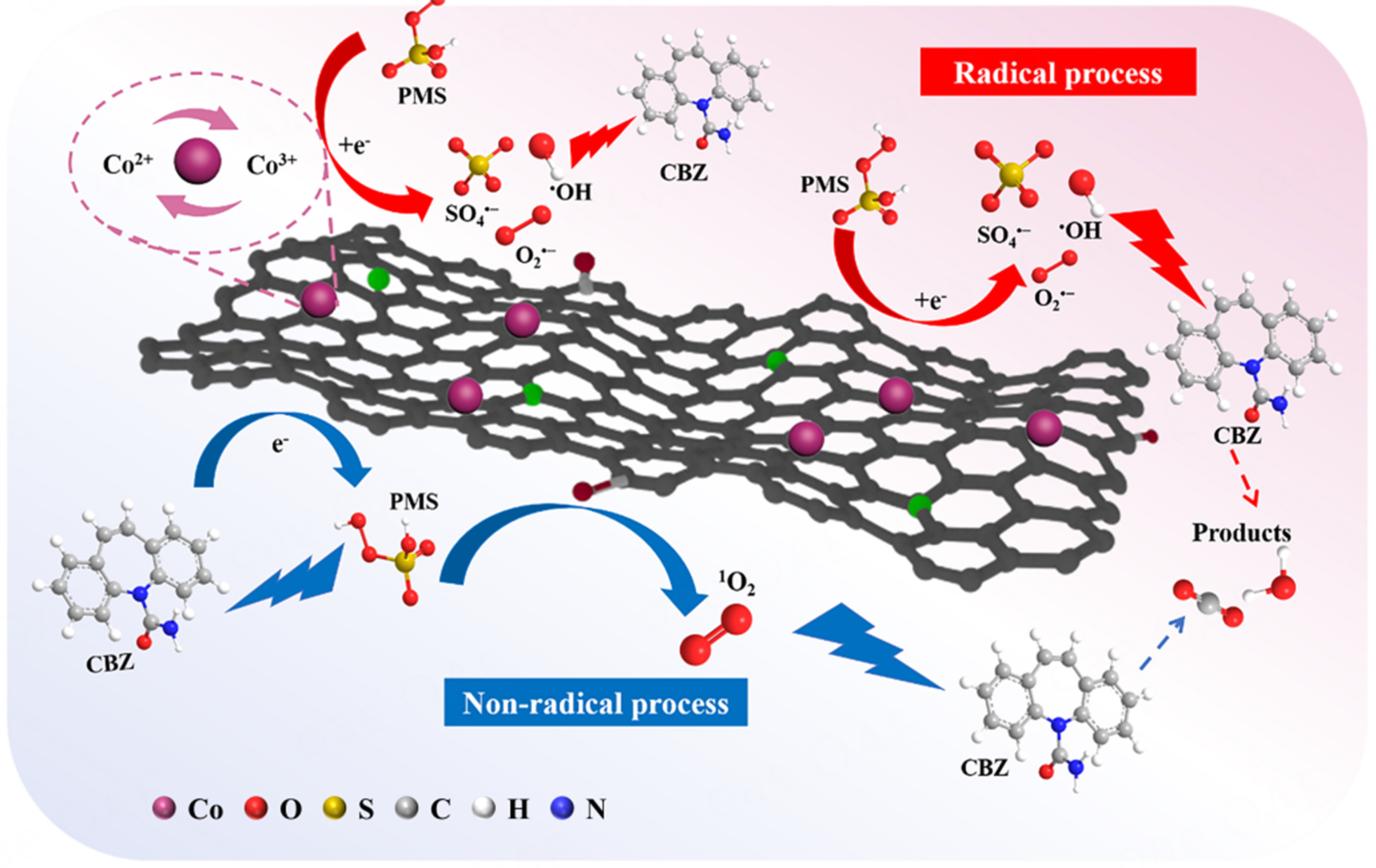
Probable activation mechanism
ROS scavenging experiments
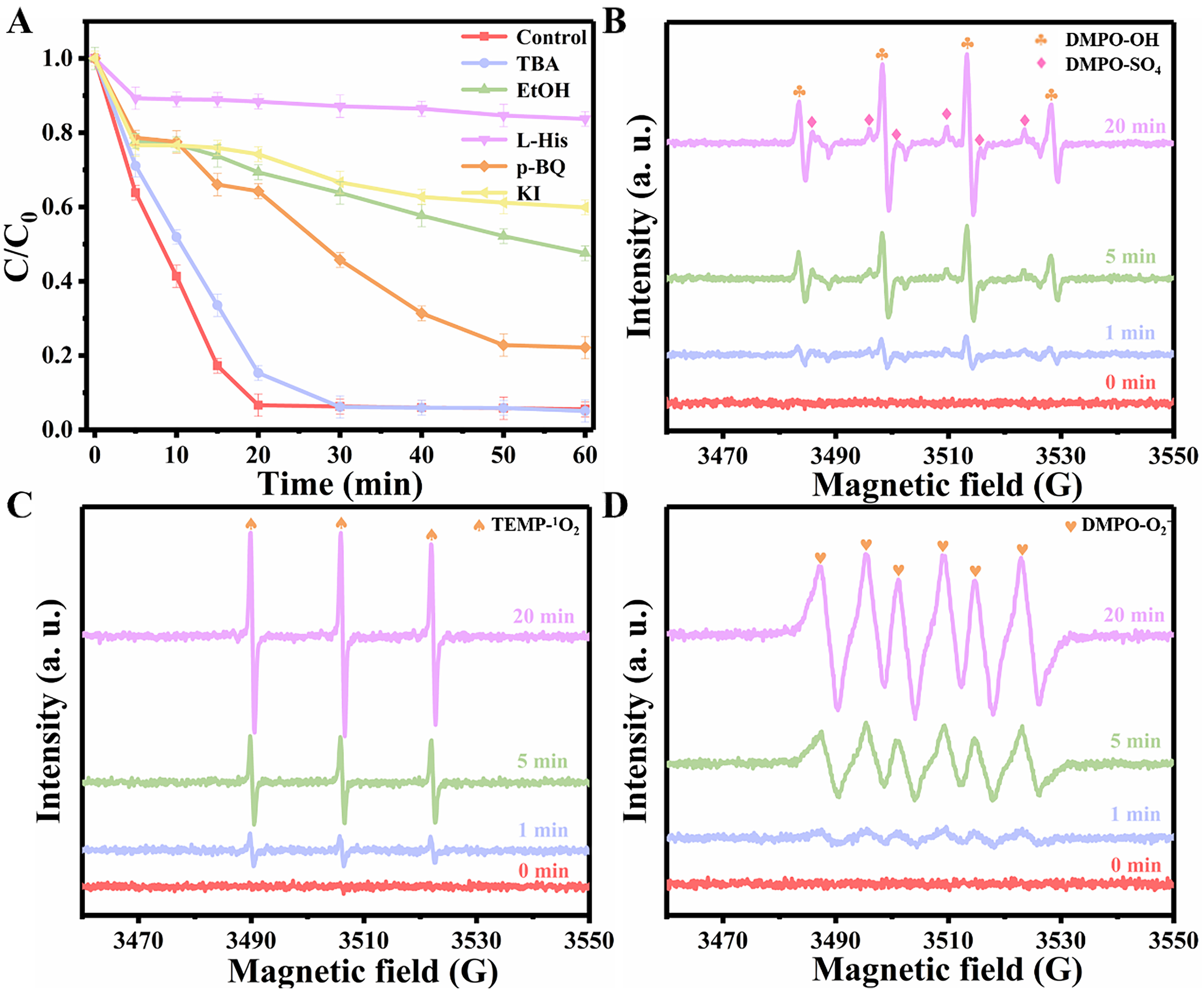
The main reactive species in the reaction system were determined through various quenching experiments to elucidate the active species generated in the Co/N@ZS-PDA/PMS system. According to the literature, EtOH, which contains α-H, can undergo simultaneous reactions with SO4•- and •OH. In contrast, tert-butanol (TBA) without α-H only can quench •OH[41-43]. Additionally, 1, 4-Benzoquinone (p-BQ) and L-histidine (L-His) can respectively act as scavengers for O2•- and 1O2[44]. As illustrated in Figure 5A, CBZ degradation had minimal impact with 200 mM of TBA added, whereas the introduction of 200 mM EtOH obviously decreased degradation efficiency with only 52.45% removal of CBZ observed, indicating a predominant role of SO4•- over •OH in the degradation process. Although the inclusion of p-BQ (0.5 mM) also exhibited some inhibitory effects on the reaction the addition of 5 mM L-His impeded the degradationof CBZ and demonstrated a more pronounced inhibitory effect compared to other quenchers employed. Potassium iodide (KI) was commonly used to quench surface-bound free radicals. The removal rate of CBZ was only 40.11% with 1 mM KI, indicating that the CBZ adsorbed on the surface of Co/N@ZS-PDA can be used as an electron donor to transfer electrons to PMS, thereby accelerating its decomposition. Henceforth, it can be hypothesized that reactive oxygen species (ROS) (SO4•-, O2•-, and 1O2) collectively participate in the degradation mechanism of CBZ within the Co/N@ZS-PDA/PMS/CBZ system where 1O2 plays a dominant role.
Figure 5. (A) Quenching experiment; EPR spectra of (B) •OH and SO4•-, (C) 1O2, and (D) O2•-. Experimental conditions: [EtOH] = [TBA] = 200 mM, [L-His] = [p-BQ] = 0.5 mM, [KI] = 1 mM, [DMPO] = [TEMP] = 10 mM, [CBZ] = 20 mg/L, [Cat.] = 0.10 g/L, [PMS] = 0.30 g/L, initial pH = 6.38. Each experiment was performed in triplicate, with error bars indicating the standard deviation of the results. EtOH: Ethanol; TBA: tert-butanol; L-His: L-histidine; p-BQ: para-benzoquinone; KI: potassium iodide; DMPO: 5,5-Dimethyl-1-pyrroline N-oxide; TEMP: 2,2,6,6-Tetramethylpiperidine; CBZ: carbamazepine; Cat.: catalyst; PMS: peroxymonosulfate; EPR: electron paramagnetic resonance; •OH: hydroxyl radical; SO4•-: sulfate radical; 1O2: singlet oxygen; O2•-: superoxide radical.
EPR analysis
The ROS (SO4•-, •OH, O2•-, and 1O2) detected by electron paramagnetic resonance (EPR) were used to further verify the active species in the reaction system [Figure 5B-D]. The 5,5-dimethyl-1-pyrroline N-oxide (DMPO) is usually combined with SO4•-, •OH, and O2•- to form DMPO-X adducts, making it an effective radical trapping agent. As shown in Figure 5B, the absence of characteristic peaks at 0 min indicates the non-occurrence of SO4•- and •OH formation in the absence of a catalyst. As the reaction progressed, the characteristic peaks of DMPO-OH and DMPO-SO4- adducts emerged, accompanied by an increase in signal peak intensity[45]. This observation suggests an elevation in both the generation and concentration of •OH and SO4•- species throughout the reaction. The compound 2,2,6,6-tetramethyl-4-piperidone (TEMP) acted as a traping agent for 1O2, exhibiting a characteristic peak ratio of 1:1:1 in the high-intensity triplet EPR spectrum. At the initial stage of the reaction, distinct signal peaks corresponding to the formation of TEMP-1O2 adducts were clearly observed [Figure 5C][46]. Similarly, the quadruple characteristic peak signal attributed to the DMPO-O2- adducts can be observed in Figure 5D, thereby confirming the presence of 1O2 and O2•- in the catalytic reaction. The results obtained from EPR analysis further validate the coexistence of SO4•-, •OH, O2•-, and 1O2 in the reaction system.
Electrochemical analysis
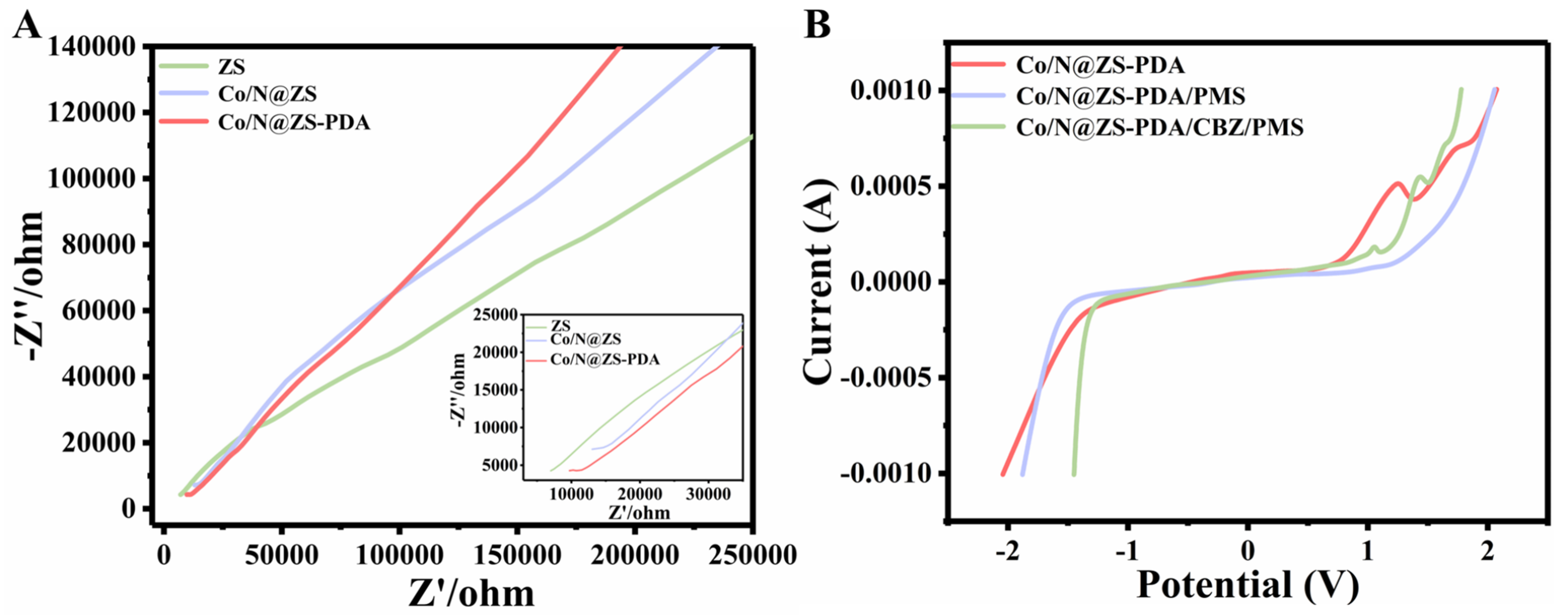
The Co/N@ZS-PDA/PMS system has also demonstrated the presence of an electron transfer mechanism, which operates as a non-free radical reaction pathway. To characterize the electron transfer on the catalyst, electrochemical impedance spectroscopy (EIS), linear sweep voltammetry (LSV), and open circuit potential-time technology (OCPT) techniques were employed [47,48].
The Nyquist semicircle of the Co/N@ZS-PDA in the high-frequency region, as depicted in Figure 6A, exhibits a small diameter indicating a reduced charge transfer resistance (Rct). The high conductivity of the material was beneficial for facilitating the activation of PMS and the degradation of contaminants. Following surface modification with PDA, the catalyst Co/N@ZS-PDA exhibited an abundance of catechol structures, which can generate hemiquinone and quinone structures under neutral conditions, thereby serving as electron acceptors by receiving electrons and protons. In addition, in the low-frequency region, the steep slope observed for Co/N@ZS-PDA indicated its low Warburg impedance and excellent ionic diffusivity in the electrolyte.
Figure 6. (A) EIS analysis of various catalysts with an inset providing an enlarged view of the high-frequency region; (B) LSV curves of Co/N@ZS-PDA under different conditions. ZS: Zr-SBA-15; PDA: polydopamine; EIS: electrochemical impedance spectroscopy; LSV: linear sweep voltammetry; CBZ: carbamazepine; PMS: peroxymonosulfate.
In the LSV curve [Figure 6B], Co/N@ZS-PDA/PMS/CBZ exhibited the highest current, indicating that Co/N@ZS-PDA and PMS interact to form a transferable complex, thereby accelerating the electron transfer process. The verification of PMS complex formation can be achieved by measuring OCPT [Supplementary Figure 6]. After the addition of PMS, there was a obvious increase in the OPCT of the system, suggesting an enhanced oxidation capacity of the formed Co/N@ZS-PDA/PMS complex and providing further evidence for the non-free radical oxidation pathway in the electron transfer process.
Possible activation mechanism
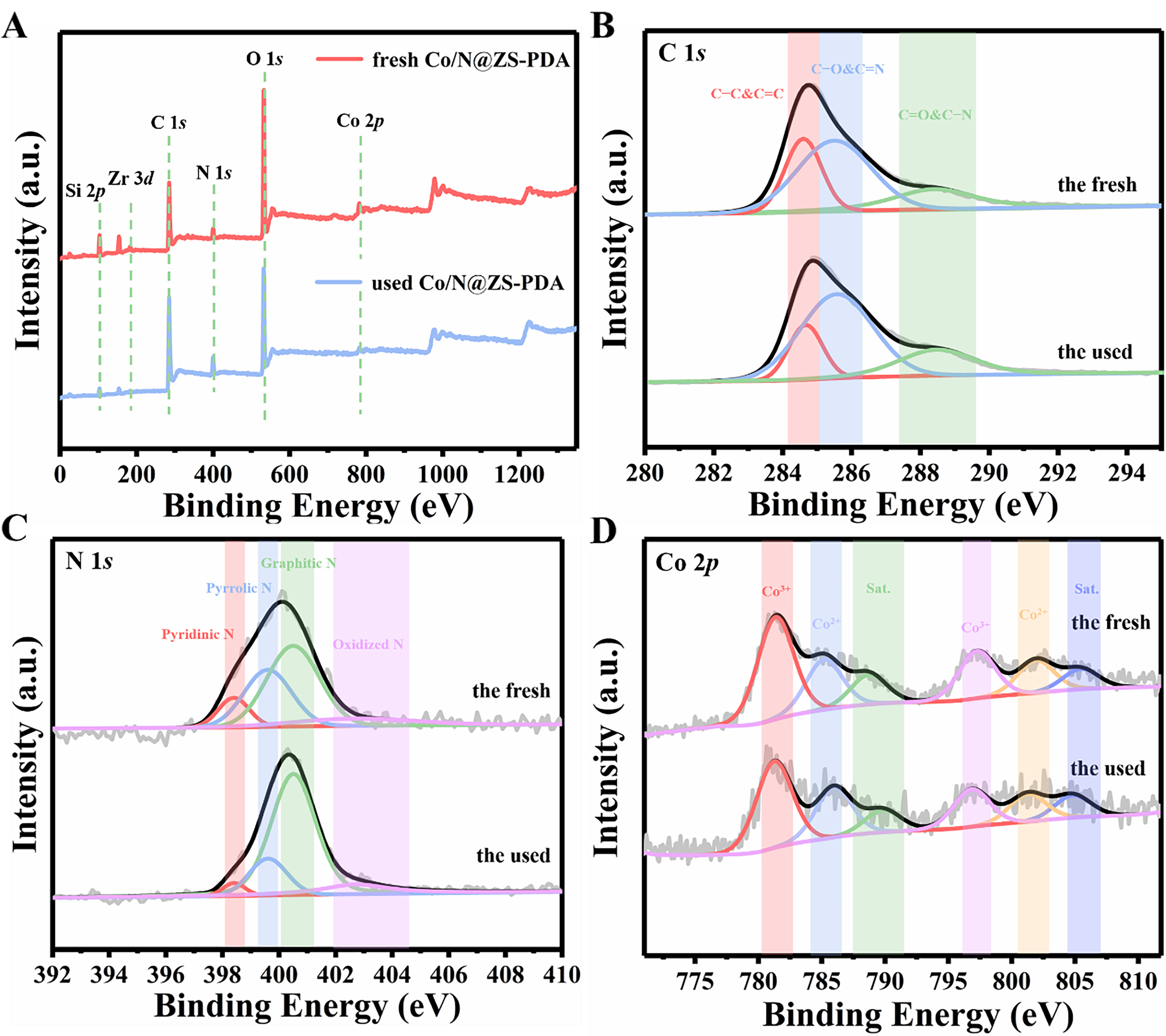
To clarify the possible reaction mechanisms within the system, the XPS spectra of Co/N@ZS-PDA (fresh and used) were studied. As shown in Figure 7A, the characteristic peaks of Co 2p, O 1s, N 1s, C 1s, Zr 3d, and Si 2p were present in the survey XPS spectra of both the fresh and used catalysts, demonstrating the presence of elements Co, O, N, C, Zr, and Si.
Figure 7. XPS analysis of the Co/N@ZS-PDA catalyst, comparing fresh and used samples, is presented for (A) full-range scans, (B) C 1s, (C) N 1s, and (D) Co 2p. ZS: Zr-SBA-15; PDA: polydopamine; XPS: X-ray photoelectron spectroscopy.
In the XPS fitting spectra of C 1s [Figure 7B], the convolution peaks at 284.6, 285.5, and 288.4 eV were assigned to the C–C/C=C, C–O/C=N, and C=O/C–N bonds, respectively. The relative proportion of
The N 1s spectra [Figure 7C] of the fresh and used Co/N@ZS-PDA were resolved into four fitted peaks at 398.4 (pyridinic N), 399.6 (pyrrolic N), 400.5 (graphite N), and 402.7 eV (oxidized N), respectively. The observed decline in pyridinic N content on the used catalyst (from 10.47% to 3.30%), whereas the graphite N content notably increased (48.52% to 69.58%), indicating that the conversion of pyridinic N and graphite N occurred during the degradation process, thereby promoting the electron transfer on the surface of the catalyst[51].
The Co 2p spectra of Co/N@ZS-PDA can be deconvoluted into six peaks [Figure 7D]. The peaks centered at 781.4, 785.2, and 788.7 eV can be attributed to the Co3+ (Co 2p3/2), Co2+ (Co 2p3/2), and satellite peaks, respectively. Three peaks at 797.3, 801.9, and 805.4 eV corresponded to Co3+ (Co 2p1/2), Co2+ (Co 2p1/2), and satellite peaks, respectively. The Co3+ concentration reduced from 52.87% to 50.30%, and the ratio of Co2+ increased (from 29.41% to 31.54%) after the catalytic reaction, indicating that the Co2+/Co3+ redox was involved in the reaction. The peroxy bond (–O–O–) in PMS can be broken by Co2+ and Co3+ through an electron transfer reaction.
From the above results and analysis, a possible mechanism for catalytic degradation was proposed [Scheme 2]. In the radical processes, Co2+ reacted with -O-O- of PMS to form SO4•- and •OH, which were oxidized to form Co3+[52], expressed as
Scheme 2. The possible mechanisms of Co/N@ZS-PDA/PMS system. ZS: Zr-SBA-15; DA: dopamine; PDA: polydopamine; PMS: peroxymonosulfate; CBZ: carbamazepine.
Co2+ + HSO5- → Co3+ + SO4•- + OH- (15)
Co2+ + HSO5- → Co3+ + SO42- + •OH (16)
The Co3+ contact with HSO5- to regenerate Co2+[37], as given in
Co3+ + HSO5- → Co2+ + SO5•- + H+ (17)
O2•- can be formed by the reaction of the generated SO52- with H2O, as given in
HSO5- → SO52- + H+ (18)
SO52- + H2O → SO42- + O2•- + H+ (19)
In addition, Co3+ and Co2+ were regenerated through redox recycling with the presence of O2•-, which was conducive to the continuous generation of SO4•- and •OH, as determined by
Co3+ + O2•- →Co2+ + O2 (20)
The SO4•- generated in the system can also react directly with OH- to form •OH, as given in
SO4•- + OH- → SO42- + •OH (21)
The radicals produced in the system can be continuously degraded into CBZ as intermediates, and eventually into small molecule intermediates, CO2 and H2O. In the non-radical processes, PMS would be adsorbed on the active sites on the surface of the Co/N@ZS-PDA in the reaction system, and the PMS would be activated through non-radical pathways to degrade CBZ. The electron-rich oxides (C=O) on the surface of the material were important active sites for the activation of PMS. Hence, in addition to self-quenching by PMS through
HSO5- + SO52- → SO42- + HSO4- + 1O2 (22)
1O2 can also be produced by C=O and -OH on the surface of the catalysts. CBZ, Co/N@ZS-PDA, and PMS were used as electron donors, electron transport media, and electron acceptors degraded pollutants through electron transfer, respectively[53].
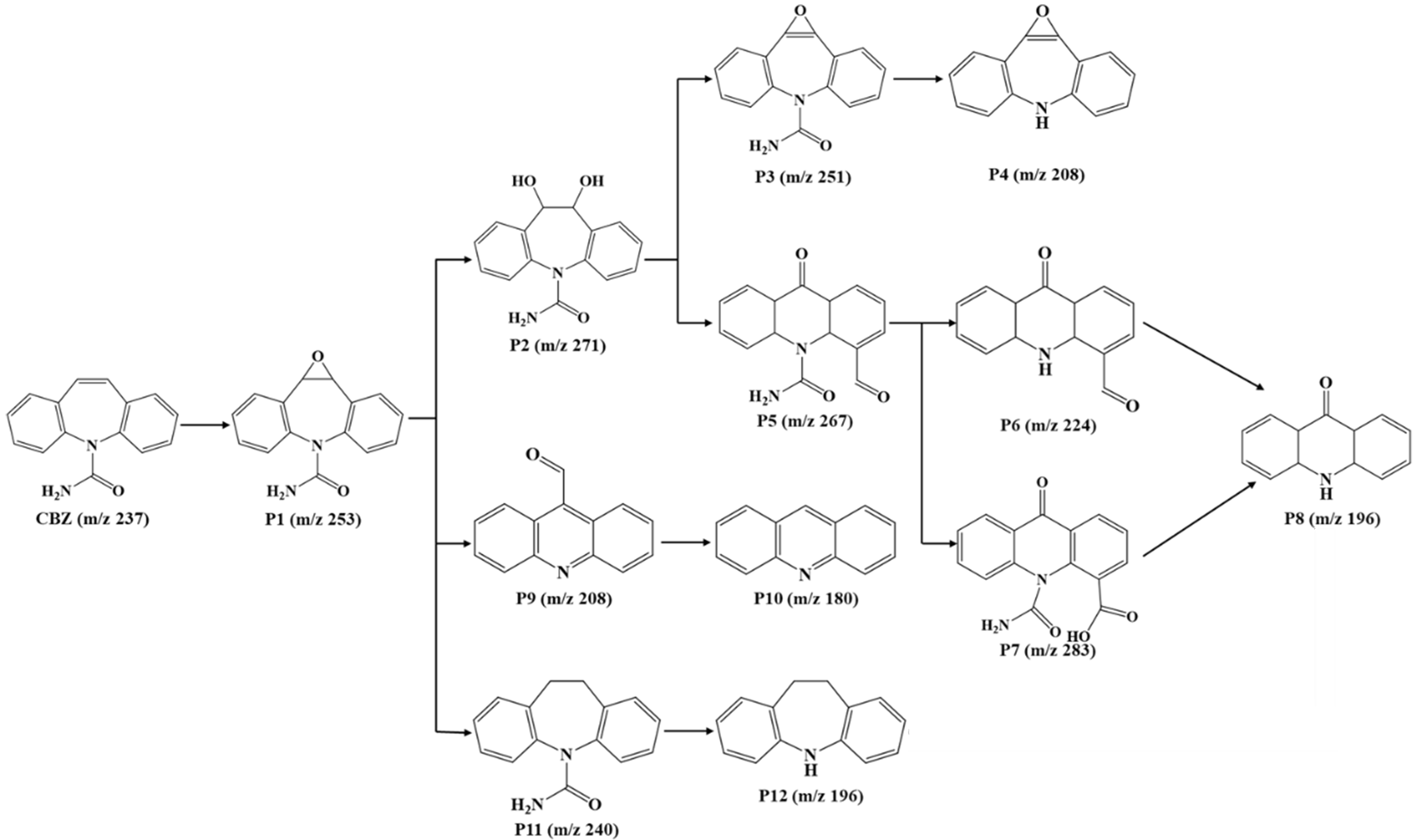
Possible degradation pathways
The intermediates generated in the system were further identified using liquid chromatography-mass spectrometry, as shown in Supplementary Table 3. Based on this, four possible degradation routes for CBZ were suggested [Figure 8]. The central heterocycle of CBZ underwent continuous attack by ROS, resulting in the epoxidation of its double bond and the formation of P1 (m/z 253). P1 underwent attack by •OH, resulting in the opening of the epoxy ring and yielding the P2 (m/z 271). In particular, it was possible that re-epoxidation or oxidation of P2 occurred, resulting in the formation of P3 (m/z 251) and P5 (m/z 267), respectively. The subsequent reaction led to the fragmentation of the amide group in P3, leading to its conversion into P4 (m/z 208). Meanwhile, the cleavage of the amide group or the oxidation of the aldehyde group in P5 resulted in P6 (m/z 224) and P7 (m/z 283), respectively[54]. The aldehyde and amide/carboxyl functional groups in P6 and P7 broke down further to produce P8 (m/z 196). Additionally, it was possible for P1 to undergo ring contraction, amide cleavage, and further oxidation to form P9 (m/z 208), and then lose the aldehyde groups to form P10 (m/z 180). The last route involved the generation of P11 (m/z 240) through ring opening to form P1, which was subsequently converted into P12 (m/z 196) by amide bond cleavage.
Stability and application
The applicability and cycling stability of the catalyst were key factors in assessing its practical performance. The versatility of the catalyst was studied by conducting degradation experiments on other pollutants in water, such as CIP, NOR, and TC in the water by the Co/N@ZS-PDA/PMS system. The degradation rate of pollutants such as CBZ, CIP, NOR, and TC can reach more than 80% in the Co/N@ZS-PDA/PMS system
Continuous operation
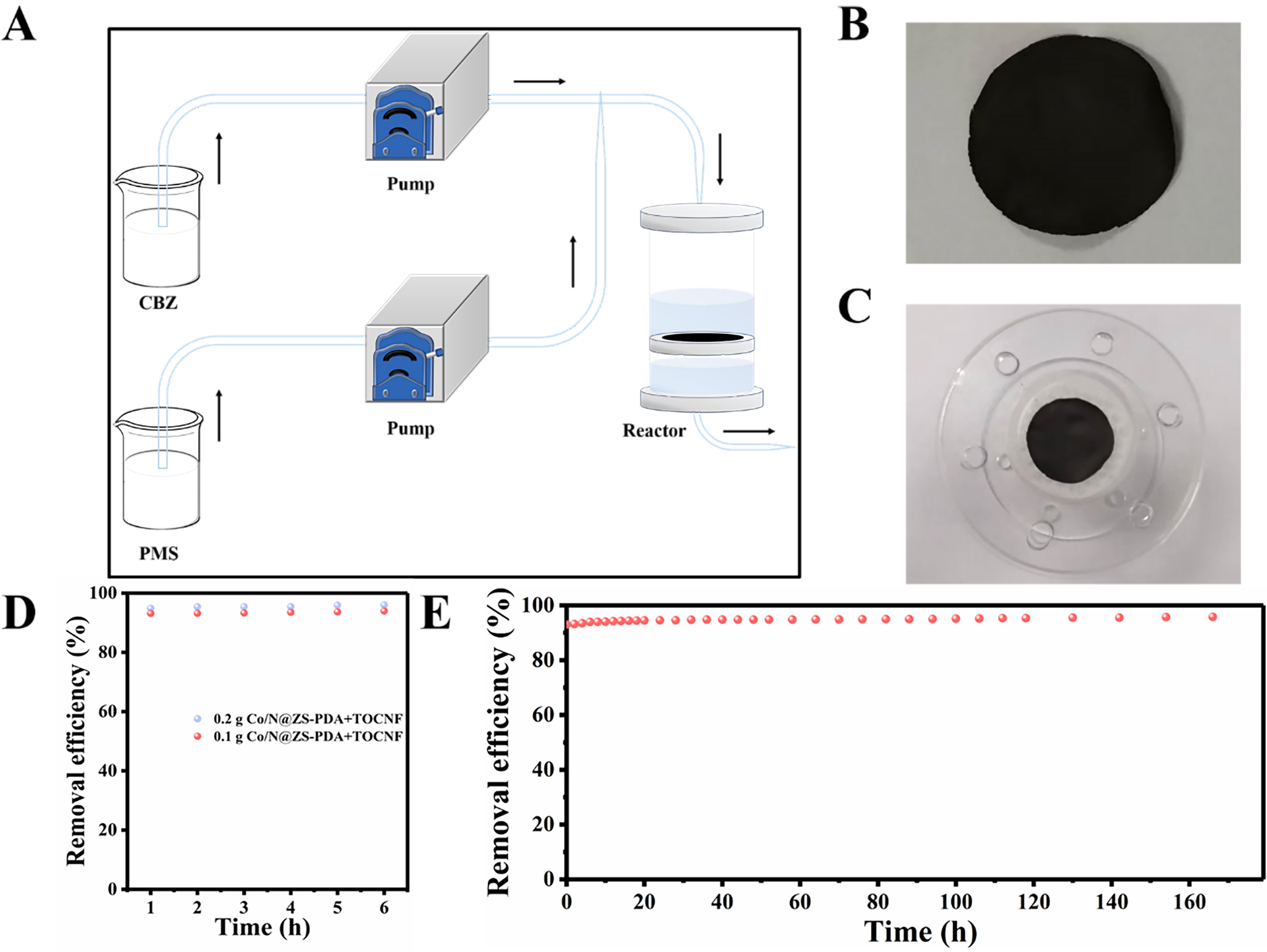
To further evaluate the practical applicability value of Co/N@ZS-PDA, a dynamic flow reactor composed of Co/N@ZS-PDA membranes was designed for achieving long-term degradation of CBZ, as shown in Figure 9A-C and Supplementary Figure 9. The Co/N@ZS-PDA membranes with flexibility were prepared by filtering a mixture suspension of cellulose oxidized by TEMPO (TOCNF) and Co/N@ZS-PDA catalyst. Subsequently, a mixed solution containing 20 mg/L CBZ and 0.3 mg/L PMS was passed through the membrane dynamics device at a 0.1 mL/min flow rate. Notably, the removal rate of CBZ remained above 94% after 6 h of operation with a TOCNF membrane filter loaded with 0.2 g of Co/N@ZS-PDA [Figure 9D]. Similarly, the membrane filter loaded with 0.1 g Co/N@ZS-PDA reached CBZ degradation rate of more than 93%. The higher the catalyst content, the more active sites were provided for the reaction. Therefore, long-term dynamic experiment was conducted on the membrane loaded with 0.1 g of Co/N@ZS-PDA. As shown in Figure 9E, the removal efficiency of CBZ was maintained at more than 90% and can be operated continuously for more than
CONCLUSIONS
In summary, a highly efficient and stable Co/N@ZS-PDA catalyst was synthesized via a PDA film decoration strategy, affording a degradation rate of over 90% for CBZ within 20 min. The long-term dynamic experiment over the Co/N@ZS-PDA membrane can be operated continuously for more than 160 h, while maintaining a CBZ removal efficiency of over 90%. The PDA decoration Co/N@ZS catalyst facilitates a strong interaction between Co species and the support by employing an electronic transfer mechanism from Co0 sites to CoOx species, endowing remarkable stability and good activity for CBZ degradation. By combining controlled experiments and in-depth characterization, the degradation of CBZ in the Co/N@ZS-PDA/PMS system involves both radical and non-radical pathways, with the non-radical pathways primarily driven by 1O2 playing a predominant role. This work provides a new strategy for addressing the Co leaching and elucidates the reaction mechanism, thereby paving the avenue for potential practical applications in pollution degradation toward AOPs.
DECLARATIONS
Acknowledgments
The authors greatly appreciate the quality service offered by the State Key Laboratory of Catalysis in Dalian Institute of Chemical Physics, Liaoning Collaborative Innovation Center for Lignocellulosic Biorefinery, and Henan Key Laboratory of Water Pollution Control and Rehabilitation Technology.
Authors’ contributions
Made substantial contributions to conception and design of the study and performed data analysis and interpretation: Li, Y.; Li, X.
Performed data acquisition and provided administrative, technical, and material support: Jiang, Z.; Zhai, S.; Wang, C.
Discussed and revised the manuscript: Shan, S.; Zhu, K.; Peng, R.
Finalized the manuscript: Li, X.; Zhai, S.; Wang, C.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (21776026, 22075034, and 22178037).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Wang, Q.; Lu, J.; Jiang, Y.; Yang, S.; Yang, Y.; Wang, Z. FeCo bimetallic metal organic framework nanosheets as peroxymonosulfate activator for selective oxidation of organic pollutants. Chem. Eng. J. 2022, 443, 136483.
2. Qing, Y.; Zhou, L.; Liu, Y.; et al. Advancing water treatment: the emerging role of NIR-driven BiO2-x catalysts. Sep. Purif. Technol. 2024, 350, 127900.
3. Zeng, H.; Zhu, H.; Deng, J.; et al. New insight into peroxymonosulfate activation by CoAl-LDH derived CoOOH: oxygen vacancies rather than Co species redox pairs induced process. Chem. Eng. J. 2022, 442, 136251.
4. Lei, Y.; Guo, X.; Jiang, M.; et al. Co-ZIF reinforced cow manure biochar (CMB) as an effective peroxymonosulfate activator for degradation of carbamazepine. Appl. Catal. B. 2022, 319, 121932.
5. Zhou, L.; Liu, Y.; Shi, H.; et al. Molecular oxygen activation: Innovative techniques for environmental remediation. Water. Res. 2024, 250, 121075.
6. Luo, J.; Bo, S.; Qin, Y.; An, Q.; Xiao, Z.; Zhai, S. Transforming goat manure into surface-loaded cobalt/biochar as PMS activator for highly efficient ciprofloxacin degradation. Chem. Eng. J. 2020, 395, 125063.
7. Pang, S.; Zhou, C.; Sun, Y.; et al. Natural wood-derived charcoal embedded with bimetallic iron/cobalt sites to promote ciprofloxacin degradation. J. Clean. Prod. 2023, 414, 137569.
8. Zhu, J.; Wang, S.; Li, H.; Qian, J.; Lv, L.; Pan, B. Degradation of phosphonates in Co(II)/peroxymonosulfate process: performance and mechanism. Water. Res. 2021, 202, 117397.
9. Sun, Y.; Cao, J.; Li, Q.; Li, D.; Ao, Z. Identifying key factors of peroxymonosulfate activation on single-atom M–N–C catalysts: a combined density functional theory and machine learning study. J. Mater. Chem. A. 2023, 11, 16586-94.
10. Zhou, C.; Guo, Y.; Du, S.; Sui, M. Ultrafast Fenton-like reaction using a peroxymonosulfate-mediated confined-Fe0 catalyst for the degradation of sulfamethoxazole. Appl. Catal. B-Environ. 2024, 358, 124442.
11. Zhou, J.; Li, X.; Yuan, J.; Wang, Z. Efficient degradation and toxicity reduction of tetracycline by recyclable ferroferric oxide doped powdered activated charcoal via peroxymonosulfate (PMS) activation. Chem. Eng. J. 2022, 441, 136061.
12. Cai, P.; Zhang, X.; Yang, S.; et al. NiCo2N nanosheets catalyzed peroxymonosulfate activation to generate 1O2 and SO4•- for efficient pollutant degradation: the role of nitrogen atoms. Appl. Catal. B. 2024, 342, 123446.
13. Zhou, Q.; Song, C.; Wang, P.; Zhao, Z.; Li, Y.; Zhan, S. Generating dual-active species by triple-atom sites through peroxymonosulfate activation for treating micropollutants in complex water. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2300085120.
14. Li, B.; Pan, T.; Yu, S.; et al. Synergistic enhancement of singlet oxygen generation in Au and oxygen vacancy co-modified Bi2MoO6 ultrathin nanosheets for efficient ciprofloxacin degradation. Sep. Purif. Technol. 2025, 354, 129321.
15. Zhu, X.; Li, J.; Wang, Y.; et al. Tetracycline degradation by the Co3O4/peroxymonosulfate system: effect of calcination temperature. Chem. Phys. Impact. 2024, 8, 100505.
16. Zhang, X.; Yan, X.; Hu, X.; Feng, R.; Zhou, M.; Wang, L. Efficient removal of organic pollutants by a Co/N/S-doped yolk-shell carbon catalyst via peroxymonosulfate activation. J. Hazard. Mater. 2022, 421, 126726.
17. Chen, L.; Yang, S.; Zuo, X.; Huang, Y.; Cai, T.; Ding, D. Biochar modification significantly promotes the activity of Co3O4 towards heterogeneous activation of peroxymonosulfate. Chem. Eng. J. 2018, 354, 856-65.
18. Yao, C.; Qin, Y.; Li, Y.; et al. Activation of peroxymonosulfate by cobalt-embedded carbon aerogels: preparation and singlet oxygen-dominated catalytic degradation insight. Sep. Purif. Technol. 2023, 307, 122728.
19. Guo, Z. Y.; Li, C. X.; Gao, M.; et al. Mn-O covalency governs the intrinsic activity of Co-Mn spinel oxides for boosted peroxymonosulfate activation. Angew. Chem. Int. Ed. Engl. 2021, 60, 274-80.
20. Luo, J.; Bo, S.; An, Q.; et al. Designing ordered composites with confined Co-N/C layers for efficient pollutant degradation: structure-dependent performance and PMS activation mechanism. Microporous. Mesoporous. Mater. 2020, 293, 109810.
21. Zhang, M.; Zhang, H.; Li, F.; Yao, L.; Peng, W.; Zhang, J. Ru supported on activated carbon and coated with a polydopamine layer for effective acetylene hydrochlorination. Catal. Sci. Technol. 2022, 12, 4255-65.
22. Lin, C.; Wen, Y.; Huang, X.; et al. Tuning the mechanical performance efficiently of various LLM-105 based PBXs via bioinspired interfacial reinforcement of polydopamine modification. Compos. Part. B. Eng. 2020, 186, 107824.
23. Fan, L.; Xie, J.; Zhang, Z.; Zheng, Y.; Yao, D.; Li, T. Magnetically recoverable Fe3O4@polydopamine nanocomposite as an excellent co-catalyst for Fe3+ reduction in advanced oxidation processes. J. Environ. Sci. 2020, 92, 69-78.
24. Liu, F.; Xiang, M.; Wang, A.; Wang, C.; Hu, B. Efficient adsorption and reduction of Cr(VI) and U(VI) by nanoscale zero-valent iron supported on polydopamine-decorated SBA-15. Appl. Surf. Sci. 2021, 568, 150931.
25. Song, Y.; Jiang, H.; Wang, B.; Kong, Y.; Chen, J. Silver-incorporated mussel-inspired polydopamine coatings on mesoporous silica as an efficient nanocatalyst and antimicrobial agent. ACS. Appl. Mater. Interfaces. 2018, 10, 1792-801.
26. Liu, G.; Shi, J.; Zhu, M.; et al. Ultra-thin free-standing sulfide solid electrolyte film for cell-level high energy density all-solid-state lithium batteries. Energy. Storage. Mater. 2021, 38, 249-54.
27. Zhang, P.; Li, W.; Wang, L.; et al. Polydopamine-modified sulfonated polyhedral oligomeric silsesquioxane: an appealing nanofiller to address the trade-off between conductivity and stabilities for proton exchange membrane. J. Membr. Sci. 2020, 596, 117734.
28. Lin, H.; Zhong, X.; Ciotonea, C.; et al. Efficient degradation of clofibric acid by electro-enhanced peroxydisulfate activation with Fe-Cu/SBA-15 catalyst. Appl. Catal. B. 2018, 230, 1-10.
29. Zhang, Z. J.; Deng, G. L.; Huang, X.; Wang, X.; Xue, J. M.; Chen, X. Y. Highly boosting the supercapacitor performance by polydopamine-induced surface modification of carbon materials and use of hydroquinone as an electrolyte additive. Electrochim. Acta. 2020, 339, 135940.
30. Xiao, S.; Cheng, M.; Zhong, H.; et al. Iron-mediated activation of persulfate and peroxymonosulfate in both homogeneous and heterogeneous ways: a review. Chem. Eng. J. 2020, 384, 123265.
31. Luo, X.; Bai, L.; Xing, J.; et al. Ordered mesoporous cobalt containing perovskite as a high-performance heterogeneous catalyst in activation of peroxymonosulfate. ACS. Appl. Mater. Interfaces. 2019, 11, 35720-8.
32. Li, W.; Zhang, Y.; Zhao, P.; et al. Enhanced kinetic performance of peroxymonosulfate/ZVI system with the addition of copper ions: reactivity, mechanism, and degradation pathways. J. Hazard. Mater. 2020, 393, 122399.
33. Fan, X.; Yu, Y.; Dong, S.; et al. Heteroatoms-doped biochar derived from deciduous resource as persulfate catalysts for efficient degradation of phenol. J. Water. Process. Eng. 2022, 48, 102866.
34. Cai, S.; Zhang, Q.; Wang, Z.; et al. Pyrrolic N-rich biochar without exogenous nitrogen doping as a functional material for bisphenol A removal: performance and mechanism. Appl. Catal. B. 2021, 291, 120093.
35. Liu, L.; Mi, H.; Zhang, M.; et al. Efficient moxifloxacin degradation by CoFe2O4 magnetic nanoparticles activated peroxymonosulfate: kinetics, pathways and mechanisms. Chem. Eng. J. 2021, 407, 127201.
36. Tang, L.; Liu, Y.; Wang, J.; et al. Enhanced activation process of persulfate by mesoporous carbon for degradation of aqueous organic pollutants: electron transfer mechanism. Appl. Catal. B. 2018, 231, 1-10.
37. Li, M.; Tong, S.; Lin, J.; Lin, K. A.; Lin, Y. Electrospun Co3O4 nanofiber as an efficient heterogeneous catalyst for activating peroxymonosulfate in water. J. Taiwan. Inst. Chem. Eng. 2020, 106, 110-7.
38. Nie, M.; Yang, Y.; Zhang, Z.; et al. Degradation of chloramphenicol by thermally activated persulfate in aqueous solution. Chem. Eng. J. 2014, 246, 373-82.
39. Pang, Y.; Zhou, J.; Yang, X.; Lan, Y.; Chen, C. Rationally designed Co3O4-SnO2 activated peroxymonosulfate for the elimination of chloramphenicol. Chem. Eng. J. 2021, 418, 129401.
40. Wang, B.; Li, Q.; Lv, Y.; et al. Insights into the mechanism of peroxydisulfate activated by magnetic spinel CuFe2O4/SBC as a heterogeneous catalyst for bisphenol S degradation. Chem. Eng. J. 2021, 416, 129162.
41. Chen, X.; Oh, W.; Lim, T. Graphene- and CNTs-based carbocatalysts in persulfates activation: material design and catalytic mechanisms. Chem. Eng. J. 2018, 354, 941-76.
42. Fang, X.; Gan, L.; Wang, L.; et al. Enhanced degradation of bisphenol A by mixed ZIF derived CoZn oxide encapsulated N-doped carbon via peroxymonosulfate activation: the importance of N doping amount. J. Hazard. Mater. 2021, 419, 126363.
43. Bao, C.; Wang, H.; Wang, C.; et al. Cooperation of oxygen vacancy and FeIII/FeII sites in H2-reduced Fe-MIL-101 for enhanced Fenton-like degradation of organic pollutants. J. Hazard. Mater. 2023, 441, 129922.
44. Shao, P.; Jing, Y.; Duan, X.; et al. Revisiting the graphitized nanodiamond-mediated activation of peroxymonosulfate: singlet oxygenation versus electron transfer. Environ. Sci. Technol. 2021, 55, 16078-87.
45. Ma, D.; Yang, Y.; Liu, B.; et al. Zero-valent iron and biochar composite with high specific surface area via K2FeO4 fabrication enhances sulfadiazine removal by persulfate activation. Chem. Eng. J. 2021, 408, 127992.
46. Zhou, C.; Zhu, L.; Deng, L.; Zhang, H.; Zeng, H.; Shi, Z. Efficient activation of peroxymonosulfate on CuS@MIL-101(Fe) spheres featured with abundant sulfur vacancies for coumarin degradation: performance and mechanisms. Sep. Purif. Technol. 2021, 276, 119404.
47. Fu, H.; Zhao, P.; Xu, S.; et al. Fabrication of Fe3O4 and graphitized porous biochar composites for activating peroxymonosulfate to degrade p-hydroxybenzoic acid: insights on the mechanism. Chem. Eng. J. 2019, 375, 121980.
48. Zhu, S.; Huang, X.; Ma, F.; Wang, L.; Duan, X.; Wang, S. Catalytic removal of aqueous contaminants on N-doped graphitic biochars: inherent roles of adsorption and nonradical mechanisms. Environ. Sci. Technol. 2018, 52, 8649-58.
49. Wang, S.; Wang, J. Synergistic effect of PMS activation by Fe0@Fe3O4 anchored on N, S, O co-doped carbon composite for degradation of sulfamethoxazole. Chem. Eng. J. 2022, 427, 131960.
50. Dai, X.; Fan, H.; Yi, C.; Dong, B.; Yuan, S. Solvent-free synthesis of a 2D biochar stabilized nanoscale zerovalent iron composite for the oxidative degradation of organic pollutants. J. Mater. Chem. A. 2019, 7, 6849-58.
51. Peng, X.; Wu, J.; Zhao, Z.; et al. Activation of peroxymonosulfate by single atom Co-N-C catalysts for high-efficient removal of chloroquine phosphate via non-radical pathways: electron-transfer mechanism. Chem. Eng. J. 2022, 429, 132245.
52. Hong, Y.; Zhou, H.; Xiong, Z.; Liu, Y.; Yao, G.; Lai, B. Heterogeneous activation of peroxymonosulfate by CoMgFe-LDO for degradation of carbamazepine: efficiency, mechanism and degradation pathways. Chem. Eng. J. 2020, 391, 123604.
53. Dong, Z.; Niu, C.; Guo, H.; et al. Anchoring CuFe2O4 nanoparticles into N-doped carbon nanosheets for peroxymonosulfate activation: built-in electric field dominated radical and non-radical process. Chem. Eng. J. 2021, 426, 130850.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].