fig5
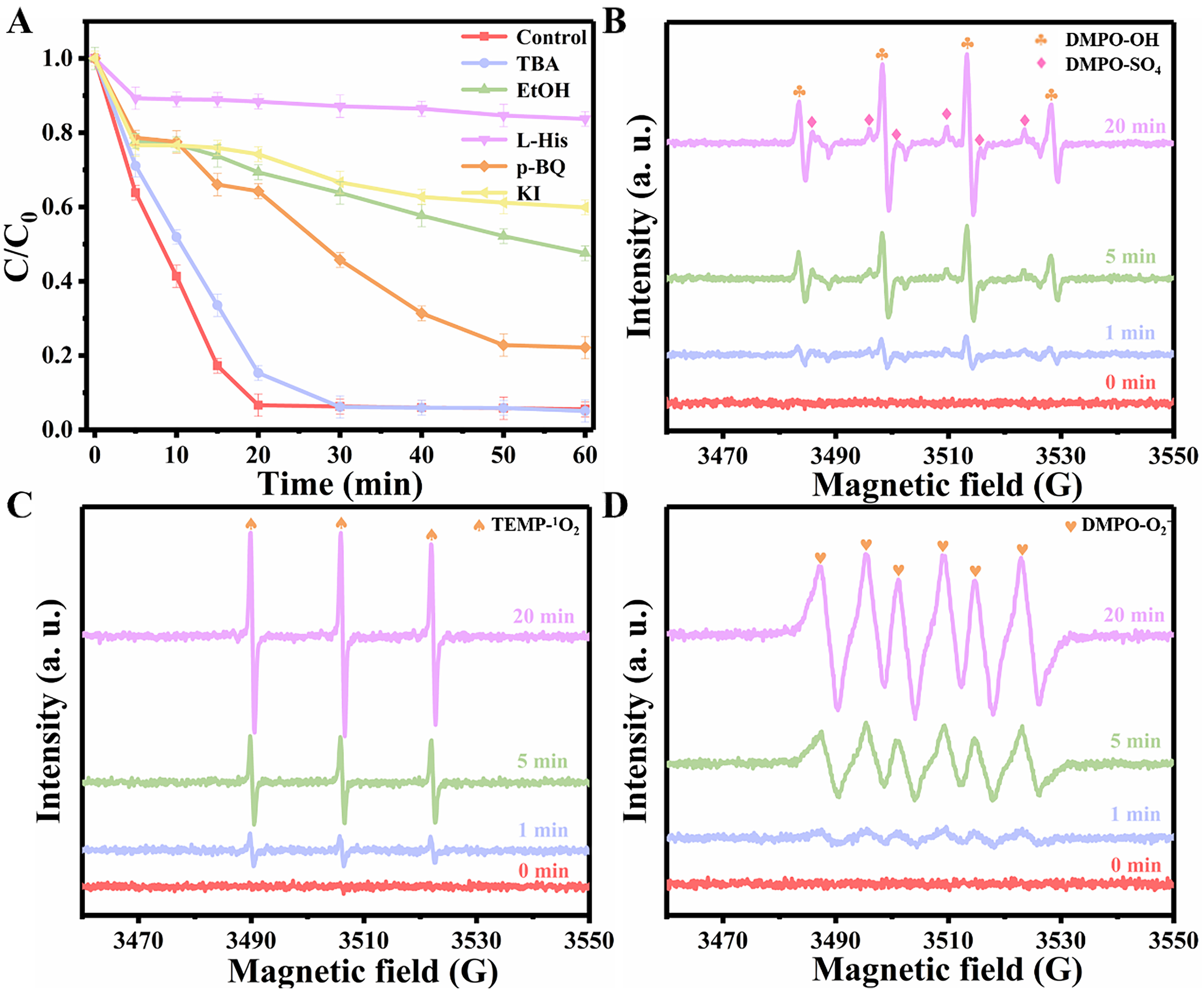
Figure 5. (A) Quenching experiment; EPR spectra of (B) •OH and SO4•-, (C) 1O2, and (D) O2•-. Experimental conditions: [EtOH] = [TBA] = 200 mM, [L-His] = [p-BQ] = 0.5 mM, [KI] = 1 mM, [DMPO] = [TEMP] = 10 mM, [CBZ] = 20 mg/L, [Cat.] = 0.10 g/L, [PMS] = 0.30 g/L, initial pH = 6.38. Each experiment was performed in triplicate, with error bars indicating the standard deviation of the results. EtOH: Ethanol; TBA: tert-butanol; L-His: L-histidine; p-BQ: para-benzoquinone; KI: potassium iodide; DMPO: 5,5-Dimethyl-1-pyrroline N-oxide; TEMP: 2,2,6,6-Tetramethylpiperidine; CBZ: carbamazepine; Cat.: catalyst; PMS: peroxymonosulfate; EPR: electron paramagnetic resonance; •OH: hydroxyl radical; SO4•-: sulfate radical; 1O2: singlet oxygen; O2•-: superoxide radical.








