Rotational reconfiguration for single-position radical nephroureterectomy with bladder cuff excision on the Xi platform
Abstract
Aim: To establish and describe a standardized single-position workflow for robot-assisted radical nephroureterectomy with bladder cuff excision (RANU + BCE) using the da Vinci Xi platform. This approach features a dual-rotation reconfiguration, allowing a seamless transition from the renal to the pelvic field without repositioning the patient.
Methods: A retrospective, single-center series of 37 patients with upper tract urothelial carcinoma (UTUC) undergoing transperitoneal Xi RANU + BCE. The rotational reconfiguration commenced with a 90° axial rotation of the multichannel single-site platform - clockwise for left-sided and counterclockwise for right-sided UTUC (as observed externally on the platform). This was followed by coordinated patient-cart axis rotation and overhead-boom adjustment to achieve a pelvic suturing view (12 → 3 for left-sided; 12 → 9 for right-sided), without altering port sites or patient position.
Results: All procedures were completed without repositioning; there were no conversions or reoperations. The median docking time was 15.00 min [interquartile range (IQR) 13.0-19.5], and the median console time was 150.0 min (IQR 121.50-175.50). Estimated blood loss was 50 mL (IQR 20.00-50.00). Hemoglobin reduction was 11.48% on postoperative day (POD) 1 and 10.20% on POD 3. Serum creatinine increased by 8.09% on POD 1 and 18.80% on POD 3. Postoperative complications occurred in 2/37 patients (5.41%), with no complications graded Clavien–Dindo ≥ III.
Conclusion: This integrated Xi workflow may enable a reproducible renal-to-pelvic transition and may help maintain continuous exposure for bladder cuff management without patient repositioning. The straightforward disengage–rotate–re-engage arm choreography allows flexible traction under surgeon discretion and facilitates team adoption and process standardization.
Keywords
INTRODUCTION
Upper tract urothelial carcinoma (UTUC) is an aggressive malignancy for which radical nephroureterectomy (RNU) with complete bladder cuff excision (BCE) remains the gold standard. Current guidelines recommend RNU + BCE for high-risk disease and report comparable oncologic outcomes among open, laparoscopic, and robotic approaches[1-3]. While open surgery provides reliable cancer control, it entails substantial trauma and prolonged convalescence. Laparoscopic nephroureterectomy offers a minimally invasive alternative but can be technically demanding in complex anatomy. By contrast, multiple series suggest that robot-assisted RNU (RANU) achieves reduced blood loss and shorter hospitalization with oncologic outcomes comparable to open and laparoscopy[4]. UTUC predominantly affects elderly patients who often present with impaired renal reserve and multiple comorbidities. Preserving perioperative renal function and minimizing surgical stress are critical, as postoperative renal decline may preclude adjuvant or systemic therapies. Consequently, surgical strategies that reduce anesthesia duration and intraoperative repositioning are of particular value in this population.
Over the past decade, robotic technology has revolutionized multi-quadrant urologic surgery. The da Vinci Xi system offers a compact architecture, extended instrument reach, and a multi-directional overhead boom that enables seamless access to both upper-abdominal and pelvic fields without cart relocation. These capabilities make the Xi platform particularly advantageous for RNU, which requires sequential exposure of renal and pelvic regions. However, the optimal configuration to leverage this capability while maintaining oncologic precision remains under development. A persistent limitation of many robotic workflows is the need for patient repositioning to transition from the renal to the pelvic field, which can add 30-60 min of operative time, interrupt working triangulation, and exacerbate instrument/port interference[5]. To mitigate these inefficiencies, several groups have described single-setup or single-docking RANU techniques that avoid repositioning while enabling formal bladder cuff management[6-8].
Against this backdrop, we present our center’s experience with a transperitoneal, single-position da Vinci Xi RANU with BCE that eliminates patient repositioning by employing a dual-rotation reconfiguration (90° access-platform rotation first, then ~90° boom swing with cart axis rotation) to shift from the upper abdomen to the pelvis. We sought to optimize and describe an integrated, single-position workflow for RANU + BCE on the Xi platform[9,10], aiming to improve procedural continuity, minimize anesthesia duration, and enhance perioperative stability for patients with high surgical risk.
METHODS
Study population
We retrospectively reviewed 37 consecutive patients with UTUC who underwent transperitoneal, single-position da Vinci Xi RANU with BCE (RANU + BCE) at the First Affiliated Hospital of Guangzhou Medical University between August 2022 and November 2024. All eligible patients scheduled for RNU with curative intent during this period were considered for the single-position dual-rotation Xi workflow. Patients with radiologically confirmed distant metastases and those deemed unfit for general anesthesia were not considered candidates for RANU + BCE; no exclusions were applied based on body mass index (BMI), clinical T stage, or prior abdominal surgery. Follow-up data were available for 30 patients (81.1%), with 7 patients (18.9%) lost to oncologic follow-up. This retrospective study was approved by the Ethics Committee of the First Affiliated Hospital of Guangzhou Medical University (Guangzhou, China; Approval No. ES-2024-102-01). Written informed consent for surgery and use of de-identified clinical data for research purposes was obtained from all patients in accordance with institutional and ethical guidelines.
Preoperative preparation and intraoperative management
Preoperative assessment included chest–abdominal computed tomography (CT) urography and cystoscopy to rule out concomitant bladder lesions. Routine laboratory testing comprised complete blood count, serum chemistry, and coagulation profile. In patients with moderate chronic kidney disease, perioperative hydration and avoidance of nephrotoxic medications were emphasized to preserve renal function. Bowel preparation consisted of a clear liquid diet on the day before surgery. A single dose of a second-generation cephalosporin or an equivalent broad-spectrum antibiotic was administered within 30 min before skin incision. All procedures were performed under general anesthesia with endotracheal intubation. Patients were placed in a lateral decubitus position and secured with adequate padding to prevent pressure injuries. All pressure points were checked after final positioning to ensure proper ventilation mechanics and to avoid nerve compression during prolonged lateral decubitus procedures.
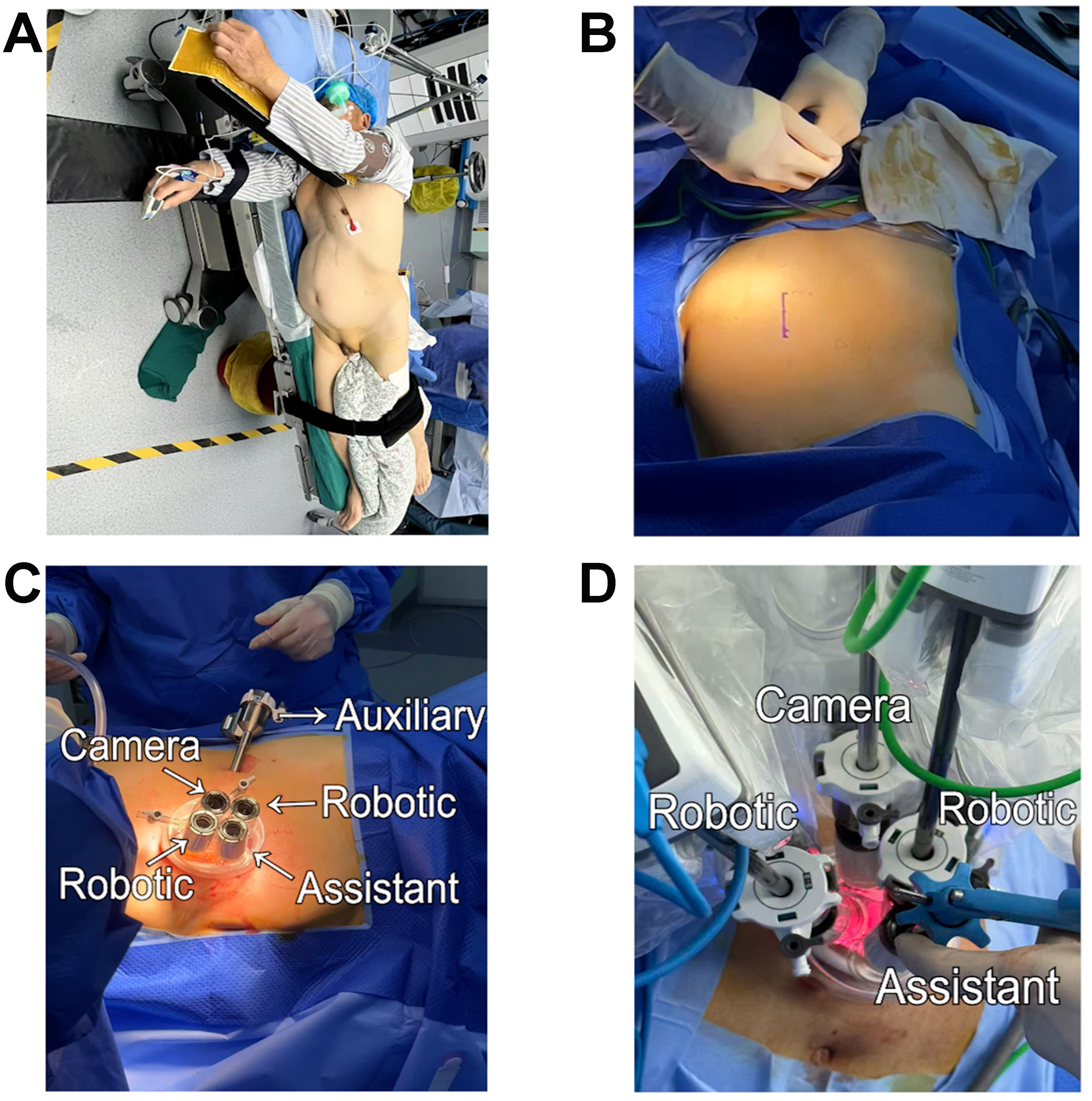
The robotic setup adhered to a predefined team protocol. All procedures were performed by a single experienced console surgeon (Prof. Di Gu) to ensure technical consistency and eliminate inter-operator variability, with a dedicated bedside assistant team participating throughout the series. The console surgeon controlled the camera and dominant working arms, while the assistant surgeon managed suction, retraction, and stapling. A scrub nurse trained in robotic docking coordinated instrument exchanges and monitored cable tension during rotational maneuvers. This structured division of roles minimized arm collision and improved operative efficiency.
During the dual-rotation reconfiguration, all instrument arms were temporarily disengaged and parked in a neutral position before rotation to avoid torque transmission. Pneumoperitoneum was maintained at 12-20 mmHg, and real-time hemodynamic and ventilatory parameters - including heart rate, blood pressure, end-tidal CO2, and peak airway pressure - were continuously monitored to ensure cardiorespiratory stability. No clinically significant hemodynamic or ventilatory derangements requiring intervention were observed during the maneuver.
Surgical technique
Overview
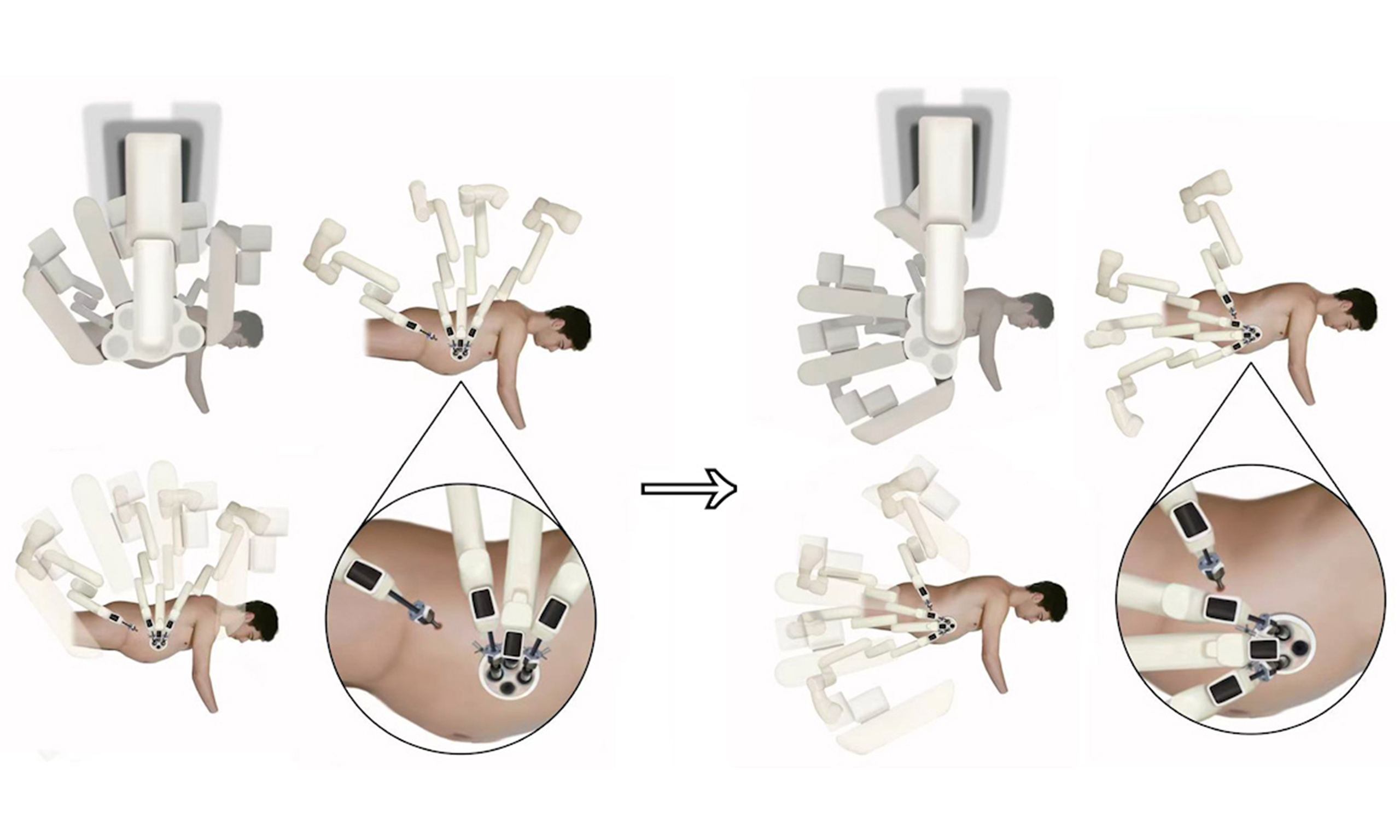
The patient was positioned in a lateral decubitus position with a 70° tilt. A 5 cm incision was made along the lateral edge of the rectus abdominis muscle on the affected side, through which a multichannel single-site system was placed. Quadrant transition from the renal/upper-ureteral field to the pelvis was achieved through a standardized rotational reconfiguration, starting with a 90° rotation of the multichannel single-site platform [Figure 1] - clockwise for left-sided UTUC and counterclockwise for right-sided UTUC (as observed externally on the platform). This was followed by coordinated rotation of the patient cart axis and adjustment of the overhead boom, all while maintaining fixed camera/arm assignments and unchanged port sites.
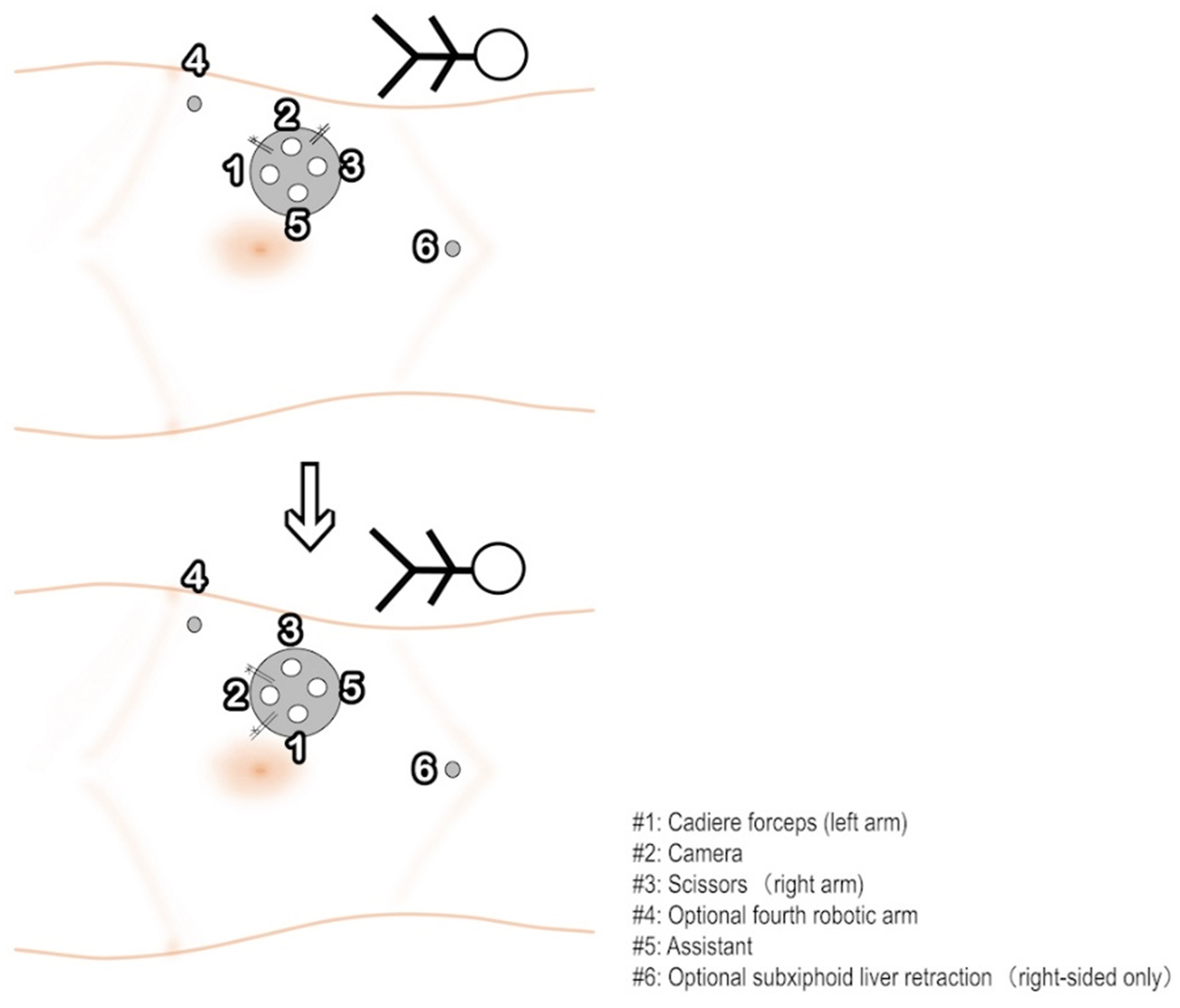
Figure 1. Patient positioning and port configuration for left-sided UTUC. (A) The patient was positioned in a lateral decubitus position with a 70° tilt; (B) A 5-cm incision is made along the lateral border of the rectus abdominis, with the inferior edge aligned with the umbilicus; (C) A multichannel single-site port is placed through this incision. An additional 8-mm incision is created 2 cm cranial to the ASIS to place a trocar for the robotic fourth arm, providing bowel/peritoneal retraction to reduce visual interference; this accessory port is optional and used at the primary surgeon’s discretion; (D) The 12-mm camera is assigned to the platform’s 12-o’clock channel and maintained in the 30° “up” orientation throughout. The 6-o’clock 12-mm channel serves as the assistant port. The 3- and 9-o’clock channels dock the working robotic arms: right arm monopolar curved scissors, left arm Cadiere forceps. UTUC: Upper tract urothelial carcinoma; ASIS: anterior superior iliac spine.
Access and port placement
A 5-cm multichannel single-site platform was routinely established as standardized access. When additional exposure was judged necessary, an 8-mm port 2 cm cranial to the ipsilateral anterior superior iliac spine (ASIS) (Port #4) could be placed to dock the fourth arm for peritoneal traction and gentle bowel sweeping. For right-sided upper-tract tumors where the liver obscured the upper pole or hilum, a subxiphoid 8-mm assistant trocar (Port #6; not robot-docked) could be added temporarily for liver elevation. Both Port #4 and Port #6 were optional (not mandatory) and were placed at the surgeon’s discretion based on exposure [Figure 2].
Figure 2. Port layout and 90° rotational reconfiguration for right-sided UTUC. A 5-cm multichannel single-site platform is used. Instrument assignment: port #1 Cadiere forceps (left working arm), port #3 scissors (right working arm), and port #2 camera (initially oriented at 12-o’clock), port #5 assistant port. Two accessory incisions may be added at the surgeon’s discretion: port #4 (8 mm), 2 cm cranial to the ipsilateral ASIS, docks the fourth arm for sustained peritoneal traction and gentle bowel sweeping; port #6 (8 mm), subxiphoid, is used only in right-sided cases when hepatic retraction is required. Neither port is mandatory, and their placement does not alter the single-position workflow. Prior to distal ureter dissection and cuff excision, the first step is a 90° axial rotation of the single-site platform - clockwise for left-sided and counterclockwise for right-sided UTUC - redirecting the camera caudally (12 → 3 left-sided; 12 → 9 right-sided). This is then followed by patient-cart axis rotation and an overhead-boom swing into the pelvic quadrant. UTUC: Upper tract urothelial carcinoma; ASIS: anterior superior iliac spine.
Renal/upper-ureteral phase
A hemlock clamp was applied to the proximal ureter immediately after exposing the ureter, prior to performing renal hilum dissection. The clamp was used to secure the ureter and prevent retrograde urine leakage. After the ureter was exposed and clamped, renal hilum control was then performed. The fourth arm, if docked, was parked in a neutral position when sustained traction was unnecessary, while temporary retraction was provided manually through the assistant port as needed.
Arm handling during rotation
All instrument arms were temporarily undocked and parked in a neutral position before rotation to prevent torque transmission to the abdominal wall or viscera. After reconfiguration, the same arms were re-engaged into their original trocars, and all port sites remained unchanged.
Rotational reconfiguration
After proximal control, rotational reconfiguration was performed without patient repositioning, maintaining fixed camera/arm assignments and unchanged port sites. The sequence began with a 90° axial rotation of the single-site platform (clockwise for left-sided, counterclockwise for right-sided UTUC), followed by patient-cart axis rotation and an overhead-boom swing as follows:
Left-sided cases: rotate the patient-cart axis caudally toward the left pelvic quadrant and swing the boom to the left lower quadrant, yielding a camera orientation of 12 → 3 o’clock.
Right-sided cases: rotate the patient-cart axis caudally toward the right pelvic quadrant and swing the boom to the right lower quadrant, yielding a camera orientation of 12 → 9 o’clock.
If the bowel or mesentery was anticipated to limit exposure during the transition, Port #4 traction (when present) was maintained until a stable, triangulated pelvic suturing view was achieved [Figure 2].
Pelvic phase
The operation then proceeded with distal ureter mobilization, BCE, and watertight closure per the center’s standard technique. Selective augmentation through Port #4 (if present) could be used to maintain peritoneal traction or gentle bowel sweeping; otherwise, the bedside assistant provided brief manual retraction via the assistant port. Temporary use of Port #6 for liver elevation in right-sided cases did not alter the camera angle, arm assignments, or rotational sequence.
Note on optional ports
Port #4 and Port #6 were not part of the standardized access and were used only when the operating surgeon deemed them necessary for exposure; their use did not require patient repositioning and remained fully compatible with the fixed camera/arm assignments.
Outcomes and definitions
Preoperative variables included age, sex, BMI, tumor location (renal pelvis vs. ureter), laterality, tumor volume, and baseline estimated glomerular filtration rate (eGFR). Tumor volume was calculated as (π/6) × length × width × height and reported in cm3.
Intraoperative variables included docking time, console time, estimated blood loss, operative time and conversion to open.
Postoperative variables included drainage volume, postoperative length of stay (LOS), abdominal drain duration, hemoglobin reduction on postoperative day (POD) 1 and POD 3, and serum creatinine change on POD 1 and POD 3.
Complications were graded using the Clavien–Dindo classification; major complications were defined as Grade ≥ III.
Pathologic variables included final pathologic T stage, histologic grade, lymph node dissection and status, and surgical margin status, as well as the occurrence of tumor recurrence during follow-up.
Follow-up duration was calculated from the index surgery to last contact. We report median [interquartile range (IQR)] for time-to-follow-up.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics, version 26.0 (IBM Corp., Armonk, NY, USA). Continuous variables are presented as median values with IQRs, and categorical variables as absolute numbers and percentages. Missing follow-up data were handled by available-case analysis without imputation.
RESULTS
Baseline characteristics
Among 37 patients, the median age was 72.00 years (IQR 64.00-76.00) and median BMI 22.41 kg/m2 (IQR 20.76-25.00). There were 21/37 (56.80%) males and 16/37 (43.20%) females. Tumors arose in the renal pelvis in 19 (51.40%) and ureter in 18/37 (48.60%); laterality was left 23/37 (62.20%) and right 14/37 (37.80%). Baseline eGFR was 55.00 mL/min (IQR 48.23-65.09). Tumor volume was 5.48 cm3 (IQR 0.83-31.12) for renal pelvis tumors and 2.47 cm3 (IQR 0.65-4.48) for ureteral tumors [Table 1].
Preoperative variables
| Variables | Value |
| Age, years (Median [IQR]) | 72.00 (64.00, 76.00) |
| BMI, kg/m2 (Median [IQR]) | 22.41 (20.76, 25.00) |
| Tumor location, n (%) | |
| Renal pelvis | 19 (51.40%) |
| Ureter | 18 (48.60%) |
| Tumor volume, cm3 (Median [IQR]) | |
| Renal pelvis | 5.48 (0.83, 31.12) |
| Ureter | 2.47 (0.65, 4.48) |
| eGFR, mL/min (Median [IQR]) | 55.00 (48.23, 65.09) |
| Sex, n (%) | |
| Male | 21 (56.80%) |
| Female | 16 (43.20%) |
| Laterality (n, %) | |
| Left | 23 (62.20%) |
| Right | 14 (37.80%) |
Intraoperative outcomes
Median docking time was 15.00 min (IQR 13.00-19.50) and console time 150.00 min (IQR 121.50-175.50). Estimated blood loss was 50.00 mL (IQR 20.00-50.00). No intraoperative conversions and no postoperative reoperations occurred [Table 2]. A gradual reduction in console time was observed with case accumulation, indicating increasing procedural familiarity. Docking time remained consistent after the initial cases, suggesting early stabilization of team coordination. The median total operative time, including docking and console phases, was 163.00 min (IQR 137.00-191.00), which appears comparable to previously published series of robotic nephroureterectomy requiring patient repositioning[11]. No intraoperative complications occurred.
Operative variables
| Variables | Value |
| Docking time, min (Median [IQR]) | 15.00 (13.00, 19.50) |
| Console time, min (Median [IQR]) | 150.00 (121.50, 175.50) |
| Operative time, min (Median [IQR]) | 163.00 (137.00, 191.00) |
| Estimated blood loss, mL (Median [IQR]) | 50.00 (20.00, 50.00) |
| Conversion to open (n, %) | 0 (0.00%) |
Early postoperative course
Median hemoglobin reduction was 11.48% (IQR 9.10-16.99) on POD 1 and 10.20% (IQR 6.73-18.65) on POD 3. Median serum creatinine increase was 8.09% (IQR -6.49~14.95) on POD 1 and 18.80% (IQR 2.73-38.66) on POD 3; drainage volume was 207.00 mL (IQR 15.00-393.50). Postoperative LOS was 6.00 days (IQR 4.00-7.00), and abdominal drain duration was 2.00 days (IQR 1.00-4.00) [Table 3].
Postoperative variables
| Variables | Value |
| Postoperative LOS, days (Median [IQR]) | 6.00 (4.00, 7.00) |
| Duration of abdominal drainage, days (Median [IQR]) | 2.00 (1.00, 4.00) |
| Drainage volume, mL (Median [IQR]) | 207.00 (15.00, 393.50) |
| Hemoglobin reduction on Day 1, % (Median [IQR]) | 11.48 (9.10, 16.99) |
| Hemoglobin reduction on Day 3, % (Median [IQR]) | 10.20 (6.73, 18.65) |
| Creatinine increase on POD 1, % (Median [IQR]) | 8.09 (-6.49, 14.95) |
| Creatinine increase on POD 3, % (Median [IQR]) | 18.80 (2.73, 38.66) |
| Postoperative complications (n, %) | |
| Clavien–Dindo < III | 2 (5.41%) |
| Clavien–Dindo ≥ III | 0 (0.00%) |
| Follow-up (n, %) | |
| Completed | 30 (81.08%) |
| Lost | 7 (18.92%) |
| Follow-up days (Median [IQR]) | 129.00 (67.50, 530.50) |
Postoperative complications and follow-up
Postoperative complications occurred in 2/37 patients (5.41%), all low-grade (Clavien–Dindo I-II). No major complications (Clavien–Dindo ≥ III) were observed. Events comprised left inguinal numbness (n = 1), and voiding difficulty/urinary retention (n = 1); all were self-limited without specific intervention. At data cut-off, 30/37 (81.08%) had completed follow-up and 7/37 (18.92%) were lost; median follow-up was 129.00 days (IQR 67.50-530.50) [Table 3].
Pathologic and oncologic outcomes
Final pathologic tumor stage (pT) showed carcinoma in situ (pTis) in 1 patient (2.70%), noninvasive papillary carcinoma (pTa) in 6 (16.22%), tumor invading subepithelial connective tissue (pT1) in 4 (10.81%), tumor invading muscularis (pT2) in 17 (45.95%), tumor invading peripelvic or periureteric fat (pT3) in 7 (18.92%), and tumor invading adjacent organs (pT4) in 2 patients (5.41%). Histologically, high-grade invasive tumors accounted for the majority of cases (23/37, 62.16%), followed by high-grade non-invasive (5/37, 13.51%), low-grade non-invasive (8/37, 21.62%), and low-grade invasive tumors (1/37, 2.70%). Lymph node dissection or sampling was performed in 14 patients (37.84%), with a total of 96 lymph nodes removed. Three patients (8.11%) had lymph node metastasis, with four positive nodes identified. All 37 patients had negative surgical margins. During follow-up, 2/30 patients experienced recurrence, both presenting as intravesical recurrence [Table 4].
Pathologic and oncologic variables
| Variables | Value |
| Pathologic T stage (n, %) | |
| pTis | 1 (2.70%) |
| pTa | 6 (16.22%) |
| pT1 | 4 (10.81%) |
| pT2 | 17 (45.95%) |
| pT3 | 7 (18.92%) |
| pT4 | 2 (5.41%) |
| Histologic grade (n, %) | |
| High-grade invasive | 23 (62.16%) |
| High-grade non-invasive | 5 (13.51%) |
| Low-grade invasive | 1 (2.70%) |
| Low-grade non-invasive | 8 (21.62%) |
| Lymph node dissection (n, %) | 14 (37.84%) |
| Lymph node positive patients (n, %) | 3 (8.11%) |
| Total positive nodes (n) | 4 |
| Total lymph nodes removed (n) | 96 |
| Positive surgical margin (n, %) | 0 (0.00%) |
| Recurrence during follow-up (n, %) | 2/30 (6.67%) |
DISCUSSION
This study describes a transperitoneal Xi RANU with BCE that avoids patient repositioning by employing a dual-rotation reconfiguration, consisting of a 90° access-platform rotation followed by a ~90° overhead boom adjustment. This innovation addresses a long-standing limitation in robotic RNU workflows - the need for patient repositioning to transition from the upper abdomen to the pelvis. In accordance with current guidelines recommending RNU with formal BCE as the standard for high-risk UTUC[1-3], our approach refines procedural efficiency without altering oncologic principles. This refinement streamlines operative workflow and provides a structured foundation for defining standardized robotic strategies applicable across centers. As the global adoption of robotic nephroureterectomy accelerates, consistent procedural geometry and reproducible team choreography are increasingly vital for maintaining cross-institutional comparability. Establishing such standardized pathways not only enhances surgical safety and efficiency but also facilitates data aggregation for future multicenter outcome analyses, addressing one of the major gaps in current robotic oncology practice.
The rationale behind this dual-rotation design lies in preserving triangulation and instrument geometry while achieving full pelvic access. The overhead boom and extended arm reach of the Xi platform theoretically enable multi-quadrant exposure[9,10], but their practical application has been inconsistent across centers. Our structured reconfiguration standardizes this transition by defining two reproducible rotational axes (access platform → cart axis) and fixed camera orientation (12 → 3 for left-sided, 12 → 9 for right-sided cases). As a result, the entire workflow proceeds in a single position, maintaining spatial orientation and avoiding instrument cross-over or external arm collisions. Maintaining geometric alignment ensures seamless continuity between upper-tract and pelvic dissection, reducing the need for frequent camera or arm reorientation. This continuity preserves visual orientation and instrument economy, both critical for minimizing cognitive load and sustaining precision during extended console time. This stable spatial configuration preserves tension balance on the ureter and perivesical tissues, mitigating the risk of traction-related injury while enabling precise suturing angles during bladder cuff closure. Consistent maintenance of spatial geometry during this transition minimizes the need for reorientation by the console surgeon, which can otherwise lead to dissection inaccuracies or prolong the operation. By preserving a continuous visual and instrument axis, the workflow supports intuitive surgical navigation and maintains stable ergonomics even during complex pelvic suturing. This predictable setup also enhances team coordination, allowing each member to anticipate sequential movements with minimal verbal cueing.
During the operation, the decision to utilize a fourth robotic arm is left to the discretion of the primary surgeon. When not using the fourth arm, the multi-channel configuration of the single-site platform allows the camera and the two operating arms to form a stable triangular structure. This setup significantly reduces the likelihood of instrument collision and ensures smooth instrument movement, particularly during the dissection of the kidney and proximal ureter, which mirrors the setup used in standard robotic nephrectomy procedures. For distal ureter and BCE, the rotational capacity of the da Vinci Xi platform is fully leveraged. A sequential rotation of the boom followed by the access port maintains the triangulation between the camera and the two working instruments, allowing precise suturing under stable visualization. Additionally, the lateral decubitus positioning of the patient enables gravitational displacement of the bowel, reducing interference in the pelvic field. The assistant’s suction instrument can further retract the peritoneum, improving exposure of the bladder and facilitating effective closure. When the fourth robotic arm is deployed, it reduces the primary surgeon’s reliance on the bedside assistant. It provides stable and autonomous tissue retraction, particularly valuable during bladder cuff management. Importantly, the trocar for the fourth arm is positioned at the caudal aspect of the single-site platform, forming a quadrilateral structure with the existing camera and working arms. Despite this added complexity, instrument collision does not increase.
Compared with previously reported single-docking or single-setup techniques[6-8], the present configuration emphasizes both reproducibility and scalability. Earlier methods often relied on unconventional port geometries or manual arm realignment, limiting their generalizability. Patient repositioning will not only increase the complexity and duration of the procedure but also affect the fluidity and precision of the surgery. Our study, through the adoption of a dual-rotation configuration, avoids these common operational steps and allows for a smooth transition from the renal area to the pelvic region without altering the port positions or patient posture. In our experience, this approach was associated with relatively short operative times and hospital stays, but formal comparative studies will be required to determine whether it truly reduces surgical time or postoperative hospitalization compared with standard repositioning workflows. In multi-port robotic surgery, in addition to the initial trocar incision, an additional incision is often required to retrieve the gross specimen after the completion of the procedure. In contrast, our center’s single-site robotic-assisted RNU for renal pelvic and ureteral cancer involves only one incision. Compared to multi-port approaches, this results in a more aesthetically pleasing postoperative scar and a reduction in the number of incisions[12]. Furthermore, the use of a single incision allows for faster abdominal closure, further reducing surgical time and anesthesia duration, which contributes to improved postoperative recovery[13]. In the study by Veccia et al., the postoperative complication rate for single-stage Xi® robotic nephroureterectomy was 17.7%, with 2.7% classified as major complications (Clavien-Dindo ≥ 3)[14,15]. In comparison, our study reported a lower complication rate of 5.41%, with no major complications. These findings raise the hypothesis that a single-position approach might contribute to a lower complication burden, although this requires confirmation in larger, controlled cohorts.
From a technical perspective, three key ergonomic elements appear to underlie the success of this workflow. First, the 12-o’clock camera orientation during the renal and upper-ureteral phase aligns the optical axis with the gonadal and renal venous trajectory, minimizing cross-arm interference. Second, the dual-rotation sequence (access-platform 90° rotation followed by ~90° boom swing) enables a seamless shift of the optical field toward the pelvis without altering port sites or patient position. Third, selective use of a fourth stabilizer arm for peritoneal or bladder traction maintains exposure and suturing angles during distal ureteral dissection, reducing dependence on the bedside assistant.
Our workflow focuses on process standardization rather than oncologic endpoints; nonetheless, it preserves the oncologic tenets emphasized in contemporary guidelines, including formal BCE and avoidance of distal manipulation before proximal control[1-3]. By maintaining triangulation and a stable suturing view in the pelvis, the configuration facilitates a circumferential excision and watertight closure. It also minimizes urine backflow and potential tumor seeding - oncologic principles echoed by prior comparative and narrative RANU reports[4-6].
Maintaining a single lateral decubitus position provides measurable physiological advantages, particularly for elderly patients with cardiopulmonary comorbidities. By avoiding repeated Trendelenburg adjustments or body rotations, this approach minimizes hemodynamic fluctuations and respiratory compromise, enhancing anesthetic safety. Continuous positioning not only simplifies airway and line management but also reduces the anesthetic workload and stabilizes intraoperative monitoring. The absence of hemodynamic instability during the dual-rotation phase further supports the safety of this method. These findings align with the principles of enhanced recovery after surgery (ERAS)[1,3], emphasizing that ergonomic workflow optimization can directly improve perioperative stability. Additionally, the dual-rotation principle demonstrated in nephroureterectomy has potential applications beyond this specific procedure. The same disengage–rotate–re-engage choreography could be applied to other multi-quadrant robotic procedures, such as nephrectomy combined with contralateral adrenalectomy or extended lymphadenectomy.
Bang et al. described a retroperitoneal single-port (SP) robotic nephroureterectomy with BCE, performed entirely through a single incision[16]. The robot was repositioned to enable BCE, and the docking port was adjusted to direct toward the bladder beyond the psoas muscle, all without requiring re-docking. This approach avoids concerns regarding force transmission between multiple instruments during rotation. Recent reviews have also summarized the emerging role of SP robotic platforms in upper tract surgery and highlighted both their ergonomic advantages and current limitations[17].
In contrast, our Xi robotic platform employs a similar instrument distribution and workflow but differs in how it manages rotation. Due to safety concerns about force transmission to the internal organs when rotating toward the bladder and pelvis, we chose to disconnect the port before rotation. This step ensures that the robotic arms do not exert unintended force on the patient’s internal organs, requiring a brief re-docking to continue the procedure. While this adds a small amount of time, it significantly enhances safety and allows for precise maneuvering during the critical phase of BCE.
While SP robotic systems are increasingly adopted, our study demonstrates that robotic nephroureterectomy with BCE (RANU + BCE) can also be effectively performed using the widely available da Vinci Xi system. This approach provides an alternative for centers lacking access to SP systems due to financial or logistical constraints. The dual-rotation reconfiguration of the Xi platform allows for comparable technical precision and surgical outcomes, making advanced robotic surgery more accessible to a wider range of institutions.
Consistent with evidence from large multicenter cohorts, which suggests that advanced pathologic stage and high tumor grade are important predictors of postoperative recurrence after RNU, both recurrence events in our study occurred in patients with high-grade invasive disease. This limited pattern may be interpreted as being more compatible with the expected natural history of biologically aggressive UTUC than with workflow-related oncologic compromise, although the small sample size precludes firm conclusions. Taken together with uniformly negative margins and regionally confined nodal metastases, these findings tentatively support the hypothesis that the dual-rotation Xi configuration can preserve oncologic principles while potentially streamlining workflow[18].
All procedures in this series were performed by a single, highly experienced robotic surgeon, which inevitably introduces performance bias. However, the core elements of the dual-rotation workflow - including fixed port geometry, predefined rotational axes, early proximal ureteral control, and standardized BCE - were designed to be process-driven rather than operator-dependent. The consistency of oncologic quality indicators observed in this cohort, such as uniformly negative surgical margins and regionally confined nodal disease, suggests that this structured approach may be transferable to centers with varying levels of robotic expertise. This is contingent on appropriate training and case selection.
However, several limitations must be noted. This was a single-center, retrospective study with a modest sample size and limited follow-up, which may not fully capture oncologic or long-term functional outcomes. Additionally, the lack of a contemporaneous control group or time-motion analysis prevents direct quantification of efficiency gains. Future prospective multicenter studies, incorporating ergonomic metrics, cost analysis, and long-term oncologic endpoints, are needed to validate and refine this workflow.
Management factors likely contributed to the favorable outcomes observed. The fixed lateral position avoided repetitive pressure adjustments, and no patient experienced cardiorespiratory instability during the procedure. Although recovery parameters were not specifically analyzed, the consistently stable intraoperative course and low complication rate underscore the technical feasibility and physiological safety of the dual-rotation configuration. With structured team training and institutional standardization, this approach can serve as a practical model for next-generation robotic nephroureterectomy workflows. The principles of continuous exposure, geometric reproducibility, and minimal patient manipulation embodied in this design may also inform future innovations in multi-quadrant robotic surgery, bridging technical refinement with oncologic rigor.
In conclusion, in our single-center experience, this single-position, dual-rotation Xi workflow for RANU with BCE may represent a practical and efficient option for managing UTUC, although further multicenter validation and comparative studies are required to confirm its generalizability and long-term oncologic safety.
DECLARATIONS
Authors’ contributions
Conceptualization and methodology: Zeng B, Liang J, Zong Z, Li M, Wang Y, Gu D
Investigation: Zeng B, Wang Y, Liang J, Zong Z, Li M, Stankov V
Formal analysis and data curation: Zeng B, Wang Y, Zong Z
Writing - original draft: Zeng B, Wang Y, Zong Z
Writing, review and editing: Zeng B, Wang Y, Zong Z, Li M, Liu Y, Cai C, Zhong J, Xie T, Liang J, Long X, Huang Y, Stankov V, Wang Y, Gu D
Visualization: Zong Z, Wang Y
Rating scale review and collection: Zhao Y
Resources, supervision, project administration: Gu D
Gu D and Zeng B had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis.
Availability of data and materials
The dataset generated and analyzed during this study is not publicly available due to ethics approval restrictions but is available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This study was supported by the Guangzhou Clinical Characteristic Technology Project (No. 2023C-TS36), the National Natural Science Foundation of China (No. 82303472), and the Guangdong Province High-Level Talents Special Support Program for Young Outstanding Talents (No. 0720240251).
Conflicts of interest
Gu D serves as a Guest Editor for the topic “Single-Port Robotic Surgery in Urology: Current Applications and Future Perspectives” for the journal Mini-invasive Surgery. Gu D was not involved in any part of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. The other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
This retrospective study was approved by the Ethics Committee of the First Affiliated Hospital of Guangzhou Medical University, Guangzhou, China (Approval No. ES-2024-102-01). Written informed consent for surgery and for the use of de-identified clinical data for research purposes was obtained from all patients in accordance with institutional and ethical guidelines.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Masson-Lecomte A, Birtle A, Pradere B, et al. European Association of Urology Guidelines on upper urinary tract urothelial carcinoma: summary of the 2025 update. Eur Urol. 2025;87:697-716.
2. Coleman JA, Clark PE, Bixler BR, et al. Diagnosis and management of non-metastatic upper tract urothelial carcinoma: AUA/SUO Guideline. J Urol. 2023;209:1071-81.
3. Pandolfo SD, Cilio S, Aveta A, et al. Upper tract urothelial cancer: guideline of guidelines. Cancers. 2024;16:1115.
4. Ji R, He Z, Fang S, et al. Robot-assisted vs. laparoscopic nephroureterectomy for upper urinary tract urothelial carcinoma: a systematic review and meta-analysis based on comparative studies. Front Oncol. 2022;12:964256.
5. Teo XL, Lim SK. Robot-assisted nephroureterectomy: current perspectives. Robot Surg. 2016;3:37-48.
6. Lee Z, Cadillo-Chavez R, Lee DI, Llukani E, Eun D. The technique of single stage pure robotic nephroureterectomy. J Endourol. 2013;27:189-95.
7. Zargar H, Krishnan J, Autorino R, et al. Robotic nephroureterectomy: a simplified approach requiring no patient repositioning or robot redocking. Eur Urol. 2014;66:769-77.
8. Ye H, Feng X, Wang Y, et al. Single-docking robotic-assisted nephroureterectomy and extravesical bladder cuff excision without intraoperative repositioning: the technique and oncological outcomes. Asian J Surg. 2020;43:978-85.
9. Argun OB, Mourmouris P, Tufek I, et al. Radical nephroureterectomy without patient or port repositioning using the Da Vinci Xi robotic system: initial experience. Urology. 2016;92:136-9.
10. Bieri U, Stihl S, Caruso J, et al. DaVinci Xi ROtation technique for NEphroureterectomy (DRONE): a retrospective single-centre cohort study and description of a novel approach with augmented range of motion. J Robot Surg. 2025;19:80.
11. Ditonno F, Franco A, Wu Z, et al. Robot-assisted nephroureterectomy: surgical and mid-term oncological outcomes in over 1100 patients (ROBUUST 2.0 collaborative group). BJU Int. 2024;134:967-75.
12. Wang Y, Li M, Yao K, et al. Extraperitoneal single-site robot-assisted radical prostatectomy with extended pelvic lymph node dissection: technique and experience. BJU Int. 2025;135:700-5.
13. Long X, Du X, Wang Y, et al. Evidence-based practice and future development of enhanced recovery after surgery (ERAS) in urology: a multidimensional assessment based on the GRADE system. J Robot Surg. 2025;19:358.
14. Veccia A, Carbonara U, Derweesh I, et al. Single-stage Xi® robotic radical nephroureterectomy for upper tract urothelial carcinoma: surgical technique and outcomes. Minerva Urol Nephrol. 2022;74:233-41.
15. Gu D, Wang Y, Chang Y, et al. Intriguing future: single-site robotic-assisted radical prostatectomy. Chin Clin Oncol. 2025;14:9.
16. Bang S, Cho HJ, Ha US, Lee JY, Hong SH. Retroperitoneal single-port robot-assisted nephroureterectomy with bladder cuff excision: initial experience and description of the technique. J Clin Med. 2023;12:6091.
17. Biasatti A, Soputro NA, Porpiglia F, et al. The current landscape of single-port robotic surgery in urology. Nat Rev Urol. 2026;23:156-73.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].