Transseptal transcatheter mitral valve replacement with dedicated prosthesis: current devices and early results
Abstract
Transcatheter mitral valve replacement (TMVR) has emerged as a transformative intervention for patients with severe mitral valve disease who are at high or prohibitive surgical risk. Among the various access routes, the transseptal approach has gained prominence due to its minimally invasive nature and favorable hemodynamic profile. The transseptal route offers direct access to the mitral valve while avoiding thoracotomy and cardiopulmonary bypass, making it particularly suitable for frail and comorbid patients. However, it poses unique technical challenges, including precise transseptal puncture, complex valve anchoring in the dynamic mitral annulus, and management of left ventricular outflow tract obstruction risk. Multimodal imaging, especially real-time 3D transesophageal echocardiography and cardiac computed tomography, plays a pivotal role in procedural planning and execution. This review underscores the transseptal TMVR approach as a rapidly advancing frontier in structural heart disease treatment, emphasizing the need for further randomized trials and long-term data to fully define its role in the therapeutic landscape of mitral valve disease.
Keywords
INTRODUCTION
Mitral regurgitation (MR) is one of the most prevalent valvular heart diseases in developed countries, with frequency increasing with age[1,2]. MR can be categorized as primary (PMR), when caused by intrinsic structural abnormalities of the mitral valve (MV) complex, or secondary (SMR), arising from left ventricular or atrial remodeling. The management of MR is complex and influenced by multiple factors, including its underlying etiology, pathophysiological mechanisms, natural history, and the anticipated benefit of therapeutic interventions. Surgical intervention has been the gold standard for many decades; however, it has been noted that a significant proportion of patients are not offered intervention due to age or comorbidities. This is especially relevant in those patients with SMR, where a prognostic benefit of conventional surgery has never been proved and surgical repair techniques offer suboptimal results[3,4]. These patients experience poor outcomes, with high rates of mortality and hospitalizations[5].
Transcatheter mitral therapies have emerged to fill the large unmet need of MR management in high-risk populations or suboptimal surgical candidates[6]. Percutaneous edge-to-edge repair is most commonly performed to mimic the surgical edge-to-edge technique (mitral transcatheter edge-to-edge repair, M-TEER) conceptualized by Alfieri, using either the MitraClip (Abbott Vascular) device or the PASCAL system (Edwards Lifesciences). Recently, another transcatheter edge-to-edge repair (TEER) device, the Dragonfly (Valgen Ltd., China), received Conformité Européenne (CE) mark approval, and additional prototypes are currently in the early development phase[7-9]. However, edge-to-edge repair in the mitral space can be less effective in certain complex anatomies, such as wide coaptation gaps, device-related regurgitation, or severe leaflet tethering. These challenges have encouraged the development of alternative therapies, most notably transcatheter mitral valve replacement (TMVR). This review aims to discuss the current transseptal TMVR technologies using dedicated devices for the treatment of native, nonoperated MV disease. Other transseptal applications, such as valve-in-valve or valve-in-mitral annular calcification procedures, are not covered here, as they fall outside the scope of the present work.
CURRENT RESULTS OF MITRAL TRANSCATHETER EDGE-TO-EDGE REPAIR
In PMR, current guidelines recommend M-TEER in symptomatic patients with severe disease, favorable MV anatomy, and high or prohibitive surgical risk[4,6]. The EVEREST (Endovascular Valve Edge-to-Edge Repair Study) II trial (NCT00209274) demonstrated comparable 5-year mortality between MitraClip and surgery in a predominantly PMR population, though percutaneous repair was associated with more frequent reinterventions[10]. However, these findings are less applicable today due to advancements in device technology and operator experience. The EXPAND G4 registry (NCT04177394), evaluating the latest-generation MitraClip G4, reported excellent outcomes in real-world PMR patients, with optimal MR reduction (MR ≤ 1+) in 88.8% and acceptable reduction (MR ≤ 2+) in 97.7% at one year[11]. Similarly, the CLASP IID trial (NCT03706833) showed non-inferiority of the PASCAL system compared to MitraClip, with high rates of MR reduction and no significant difference in adverse events. The successful implantation rate was similar between groups (98.5% vs. 98.9%), and the primary effectiveness endpoint (MR ≤ 2+ at 6 months) was achieved by 97.9% in the PASCAL group and 95.7% in the MitraClip group[12]. Finally, the DRAGONFLY-DMR trial (NCT04734756) included 120 patients with prohibitive surgical risk and PMR ≥ 3+. It was a prospective, single-arm, multicenter study on the safety and effectiveness of the DragonFly system. The findings were promising, with a procedural success of 99.2% and a 92.0% rate of MR ≤ 2+ at 1 year of follow-up. Also, significant improvements in the patients’ functional and quality-of-life outcomes were reported[9].
In SMR, the evidence for M-TEER as first-line therapy is even more robust. The COAPT trial (NCT01626079) showed that, in patients with symptomatic SMR and optimized guideline-directed medical therapy (GDMT), M-TEER significantly improved survival and reduced heart failure (HF) hospitalizations, with durable MR reduction over two years[13]. Similarly, the RESHAPE2HF trial (NCT02444338) confirmed that M-TEER led to lower rates of cardiovascular death or HF hospitalization and better quality-of-life outcomes compared to medical therapy alone[14]. Acute procedural success resulted in MR of ≤ 1+ in 74.5% of patients and ≤ 2+ in 92.2%. At 1 year, the MR ≤ 2+ rate was 90.4% compared with a 36.1% rate in the conservative cohort. These results have positioned M-TEER as a key therapeutic strategy in patients with SMR and LV dysfunction not eligible for surgical intervention.
Real-world data further support these findings. The EuroSMR registry (DRKS00017428) demonstrated that optimal MR reduction (MR ≤ 1+) with M-TEER correlates with improved survival, especially when HF is not severely advanced[15]. In the EXPAND registry (NCT03502811), an optimal result (MR ≤ 1+) at 1 year was achieved in 89.5% of enrolled patients[16], whereas in the EXPAND G4 study, this rate increased to 95.3%[11].
Furthermore, the available evidence also supports the use of the PASCAL device in SMR. In the prospective cohort of the CLASP study (NCT03170349), an optimal result (MR ≤ 1+) was achieved in 77% and 71% of cases at 1-year and 3-year follow-up, respectively, while an acceptable result (MR ≤ 2+) was observed in 96% and 93%[17,18]. These findings suggest that PASCAL offers a durable therapeutic option for patients with SMR, with consistent outcomes over mid-term follow-up.
All this evidence supports the American and European guidelines’ decision to designate M-TEER as a class IIa indication for the treatment of SMR[4,6]. It is now almost standard practice in high-volume centers to use M-TEER as a first-line option in patients with left ventricular dysfunction and no coronary disease suitable for surgical grafting[19].
ROLE OF ACHIEVING OPTIMAL AND DURABLE RESULTS
All positive outcomes of M-TEER have been linked to achieving optimal procedural success, defined as residual MR 0-1+ without significant stenosis and maintaining this result over time[20-22]. Thus, appropriate anatomical selection and operator experience are critical when identifying candidates for M-TEER. Recently, Hausleiter et al. proposed an anatomical classification based on increasing procedural complexity, which correlates with a progressively lower likelihood of achieving optimal outcomes with M-TEER[23]. As anatomical challenges accumulate, the probability that patients will derive greater benefit from valve replacement rather than repair increases. Table 1 summarizes the clinical impact of having recurrent MR at follow-up.
Clinical impact of recurrent mitral regurgitation after M-TEER
| Study | MR Reduction | MR reintervention and/or outcomes |
| Sugiura et al.[24] | MR ≤ 2+ at 1 year, 90.2% vs. MR ≥ 3+, 9.8% | HFH or NYHA III/IV: 37.8% vs. 54.1% (P = 0.018) Repeat MV intervention: 2.2% vs. 9.8% (P = 0.005) Mortality: 16.1% vs. 42.3% (P = 0.08) |
| Sorajja et al.[25] | MR ≤ 2+ post-procedural, 93.0% vs. MR ≥ 3+, 7.0% | MVS: 1.7% vs. 6.1% (P < 0.001) Redo M-TEER: 5.1% vs. 17.9% (P < 0.001) Mortality: 23.9% vs. 48.9% (P < 0.001) |
| Adamo et al.[26] | Post-procedural MR 0/1+, 67% vs. MR 2+, 33% | HFH: 24.0% vs. 30.0% (P = 0.035) CV death: 16.3% vs. 24.8% (P = 0.003) Mortality: 25.7% vs. 40% (P < 0.001) |
| Orban et al.[27] | MR ≤ 1+ at discharge, 92.0% vs. MR ≥ 2+, 8.0% | Mortality or HFH: 29.7% vs. 69.6% (P < 0.001) HFH: 22.2% vs. 62.2% (P < 0.001) Mortality: 15.8% vs. 29.6% (P < 0.001) |
| Higuchi et al.[20] | Post-procedural MR 0/1+, 67% vs. MR 2+ 26% vs. MR, ≥ 3+ 7% | Mortality: 29% vs. 36% vs. 41% |
RATIONALE OF TRANSCATHETER MITRAL VALVE REPLACEMENT
Although M-TEER has transformed the management of MR and is now widely used in high-risk patients, it is not a universal solution. Its success depends heavily on anatomy: patients with very dilated ventricles, severe leaflet tethering, broad coaptation gaps, or complex prolapse often achieve only partial or temporary benefit. Even when the initial result looks good, recurrent regurgitation is not uncommon, especially in those with challenging anatomy or advanced ventricular disease[24].
Follow-up studies have taught us that durability is another important concern. While M-TEER reliably improves symptoms and reduces hospitalizations in the short term, up to 20% of patients may develop significant MR again within a few years[25,29]. This late recurrence carries a clear penalty in terms of survival and quality of life, and it raises difficult questions when treating younger patients whose life expectancy exceeds the durability of the repair.
These realities explain why TMVR has emerged as a natural next step. By replacing the valve rather than repairing it, TMVR has the potential to bypass many of the anatomical and pathophysiological barriers that limit M-TEER. In this sense, TMVR is not a competitor but a complement to M-TEER, helping expand our ability to tailor therapy to the needs of each individual patient.
Transcatheter mitral valve replacement
TMVR is an alternative to transcatheter MV repair and offers several potential advantages. First, the pathophysiology of MV disease is complex, resulting in a heterogeneous anatomical spectrum that may be difficult to treat with current transcatheter MV repair devices. A TMVR device capable of overcoming this heterogeneity and addressing numerous anatomical variations would therefore represent a tangible advantage. TMVR would provide a standardized universal treatment with a more predictable reduction in MR than MV repair while remaining less invasive than current surgical techniques[30]. Importantly, successful TMVR also relies on an optimal transseptal approach, which typically requires a higher and more central puncture in the left atrium (LA, around 3.5 to 4.5 cm above the mitral annulus) to accommodate the larger and less flexible delivery systems.
Most current experience and evidence regarding TMVR is based on the transapical Tendyne device (Abbott Vascular, Santa Clara, USA). The Tendyne global early feasibility study (EFS) (NCT02321514) showed high procedural success rates and substantial reduction of MR in nearly all patients, with a level of periprocedural adverse events considered acceptable[31]. Two-year follow-up data from this cohort confirmed sustained MR reduction and reverse remodeling, although mortality rates remained high, likely reflecting characteristics of the patient population selected for this therapy[32,33]. These results have been replicated in the real-world setting. The TENDER registry (NCT04898335) confirmed the previous results and extended the benefit of those patients with an off-label indication[34]. Despite its potential benefits, TMVR faces major challenges, including high screen-failure rates [mainly due to annular size or increased risk of left ventricular outflow tract (LVOT) obstruction], long-term anticoagulation requirements, valve durability concerns, and the heart’s response to a sudden increase in afterload. In the CHOICEMI registry (NCT04688190), only one-third of screened patients were eligible for TMVR, and adverse event rates remained high at one year[35]. Notably, transseptal TMVR demonstrated better outcomes than transapical approaches, guiding future device development[36]. With new platforms aimed at reducing screening failures and favoring a default transseptal approach, the field of TMVR may broaden in the near future[37]. In the next section, we review the most advanced transseptal TMVR platforms [Figure 1].
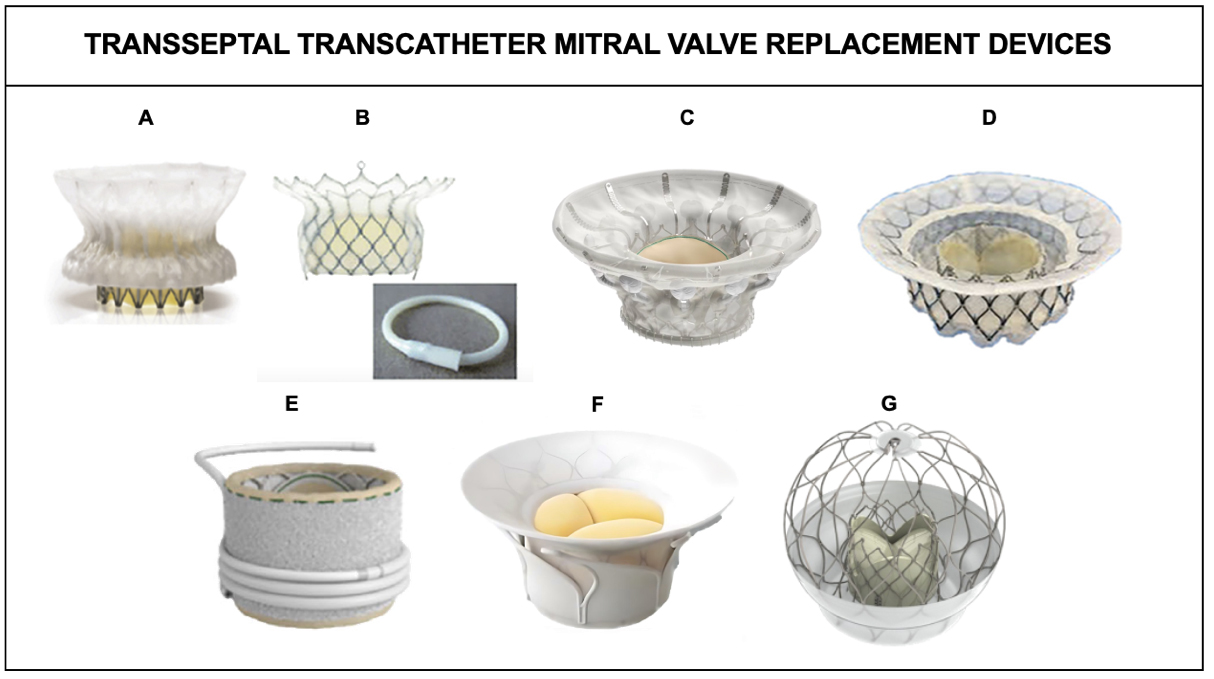
Figure 1. Transseptal Transcatheter Mitral Valve Replacement Devices[38]. (A) Cephea; (B) HighLife; (C) Cardiovalve; (D) Intrepid; (E) Sapien M3 and dock; (F) Innovalve; (G) AltaValve.
Tables 2 and 3 depict an overview of the TMVR devices presented in this review.
Structural, functional, and clinical evidence overview of the most commonly used transseptal TMVR devices
| Device | Delivery size | Anchoring mechanism | PVL prevention | Study |
| Intrepid | 35Fr | Radial force from oversized outer frame + cleats | PET skirt | Early feasibility study[39] (N = 33) |
| HighLife | 18Fr | Subannular mitral ring; atrial flange | Subannular implantation | Early feasibility study[40] (N = 30) |
| Cardiovalve | 28Fr | Self-expanding frame + radial force + sealing | Inflatable cuff | Case report[41] (N = 5) |
| AltaValve | 29Fr | Atrial fixation | PET skirt | Early feasibility study[42] (N = 17) |
| Cephea | 32Fr | Axial compression | Atrial disc | Initial experience[43] (N = 24) |
| Sapien M3 | 20Fr | Self-expanding nitinol dock + balloon-expandable valve | PET skirt | Early feasibility study[44] (N = 35) |
| Innovalve | 32Fr | Subvalvular leaflet grasping with encircling fixation | Atrial flange | Early feasibility study[45] (N = 10) |
Procedural performance, clinical outcomes, and ongoing trials of the most commonly used transseptal TMVR devices
| Device | Procedural success | Severe bleeding | Major vascular complications | LVOT obstruction | MV reintervention | Death | Ongoing studies |
| Intrepid | 93.9% | 30.9% | 24.2% | 4 patients | 3% | 6.7% | APOLLO Trial |
| HighLife | 90% | 23% | 13% | 0% | 3% | 17% | EFS Trials (US/EU) |
| Cardiovalve | 100% | 2 patients | NA | 0% | NA | 3 patients | AHEAD US/EU |
| AltaValve | 100% | 0% | NA | 0% | 0% | 0% | Pivotal Trial |
| Cephea | 95.8% | 0% | NA | 8.3% | 0% | 4.2% | Feasibility Trial |
| Sapien M3 | 88.6% | 0% | NA | NA | NA | 2.9% | ENCIRCLE Trial |
| Innovalve | 100% | 0% | 0% | 0% | 0% | 0% | TWIST‑EFS |
INTREPID TRANSCATHETER MITRAL VALVE
The Intrepid TMVR system (Medtronic) features a 27 mm trileaflet bovine pericardial valve within a self-expanding nitinol frame, composed of an inner stent housing the valve and a flexible, atrial-facing outer stent for anchoring to the MV complex[39]. Designed to preserve geometry and adapt to cardiac motion, the system incorporates a compliant atrial brim and a stiffer ventricular portion for structural support. Fixation is achieved via annular oversizing (10%-30% relative to annular dimensions measured by cardiac computed tomography) and leaflet-engaging cleats[46]. Initially delivered transapically, the system now uses a 29 French (Fr) transfemoral transseptal approach with outer frame sizes of 42 mm and 48 mm.
Procedural steps
After transseptal access, the transseptal sheath is advanced at least 2 cm into the LA. The valve system is then introduced through the sheath into the LA, with the combined TS sheath and valve capsule positioned approximately 5 cm inside the LA. At this point, the device is steered across the MV to establish a coaxial trajectory with the MV apparatus. Once coaxial alignment is achieved, the pressurized indeflator is actuated to expose the brim over the MV. The capsule is then advanced until the brim is apposed to the MV annulus, and, under rapid pacing and breath-hold, the valve is fully deployed.
Clinical evidence
Experience with the 35 Fr transseptal device has been reported in the EFS study at both 30 days and 1 year[39,47]. Fifteen patients were included in the 30-day study, with a median age of 80 years. Procedure was successful in all but 1 case, with MR elimination in all cases and no deaths, strokes or reinterventions. Access site bleeding was the most common adverse event. One-year data have also been reported in 33 patients, showing a success rate of 93% and a 1-year mortality of 6.7%, with no residual MR in any case and significant improvements in quality of life and New York Heart Association (NYHA) functional class.
The APOLLO trial (Transcatheter Mitral Valve Replacement with the Medtronic Intrepid TMVR System in Patients with Severe Symptomatic Mitral Regurgitation; NCT03242642) aims to evaluate TMVR with the Intrepid system. An addition of a single-arm substudy for patients exhibiting significant mitral annulus calcification (MAC) is also ongoing.
HIGHLIFE TRANSCATHETER MITRAL VALVE
The HighLife device (HighLife SAS, Paris, France) consists of a 30 mm nitinol capsule housing a 28 mm trileaflet bovine pericardial valve. The subannular implant (SAI) is secured with a polymer ring and nitinol anchoring elements, stabilizing the system while minimizing the risk of displacement or LVOT obstruction. Once positioned, it traps the native leaflets between the implant and the prosthetic valve. Each part is covered with Dacron and becomes endothelialized over time, promoting integration and stability. The prosthesis is available in one size and is delivered via transfemoral venous access and transseptal puncture, while the ring is delivered via transfemoral arterial approach[48].
Procedural steps
Initial access is established via the femoral artery, where an 18 Fr introducer sheath is positioned to facilitate the introduction of a specialized catheter designed for loop placement (LPC) and SAI delivery. Secondary access through the femoral vein allows for transseptal entry for valve delivery. The LPC is then advanced retrogradely across the aortic valve into the left ventricle (LV). Two subannular tubes are deployed to circumnavigate the subannular structure, and a guidewire is retrieved with a snare, externalized through the femoral access, thus forming a continuous guidewire loop that passes from and returns to the femoral site, encircling the subannular apparatus. Subsequently, the SAI is delivered and closed using a dedicated 18 Fr SAI delivery catheter (SDC), which accommodates both the SAI and the two ends of the pre-placed guidewire loop. The SDC is advanced over the guidewire loop ends into the LV, where the SAI is secured around the loop. TMVR deployment proceeds via the transseptal approach, with the delivery catheter traversing the native mitral leaflets and the SAI already in position. The outflow end of the TMVR device is deployed distal to the SAI, after which the system is carefully retracted toward the LA to position the SAI adjacent to the native annulus. The inflow end of the TMVR is then released from the delivery catheter. The guidewire loop is removed from the SAI and the SAI closing catheter (SCC) by withdrawing one end, followed by removal of the SCC and introducer sheath from the femoral site. Hemostasis is then achieved at the femoral access point.
Clinical evidence
The results of a 1-year EFS study have been presented enrolling 30 patients in 13 sites. The mean age of participants was 75.6 years, and 90% had SMR with a mean ejection fraction (EF) of 43%. Technical success was achieved in 90%, and at one year, no patient had more than mild residual MR. The one-year mortality was 17%, and there were no cases of LVOT obstruction[40].
CARDIOVALVE TRANSCATHETER MITRAL VALVE
The Cardiovalve TMVR system (Cardiovalve) is a self-expandable trileaflet valve available in three sizes (range: 35-55 mm) for delivery via a transfemoral approach. It has a symmetrical design with 12 ventricular legs designed for leaflet capture and an atrial flange for sandwiching leaflets and avoiding paravalvular leak. Following deployment, it extends just 12 mm into the LV, maintaining a minimal ventricular footprint[49].
Procedural steps
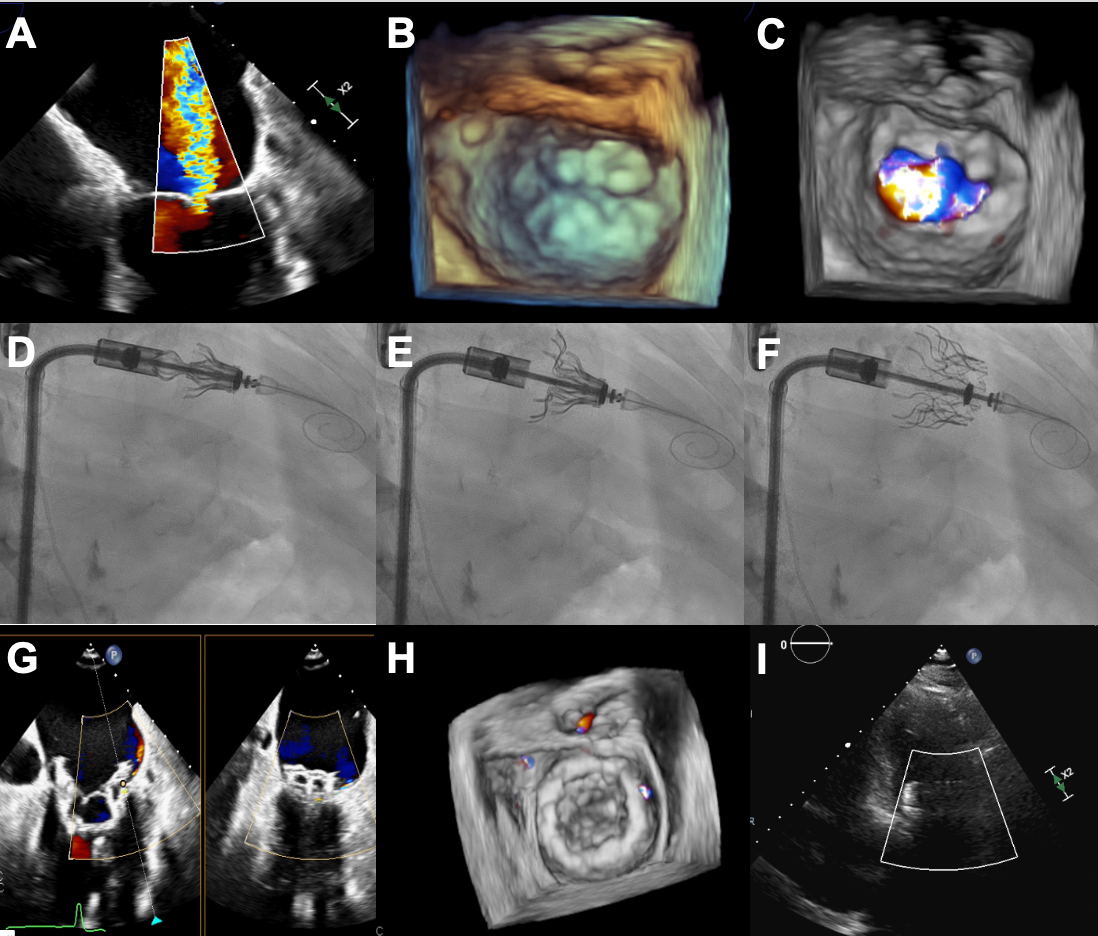
Following transseptal access, a Safari XS guidewire is positioned in the LV apex via a deflectable sheath. Valve flossing with a 12-15 mm balloon ensures a chord-free path. Septostomy is performed with a 12 mm balloon, after which the Cardiovalve delivery system is advanced into the LA using progressive flexion. Posterior rotation of the transseptal sheath in the LA raises the system above the MV; further posterior positioning is achieved by advancing the curve between the transseptal sheath and valve capsule. Once the valve is coaxially placed with adequate height over the MV leaflets, the ventricular legs are deployed and the implant catheter is advanced into the LV. Retracting the catheter engages all leaflets, allowing full deployment of the atrial flange and subsequent expansion of the valve, thereby eliminating MR [Figure 2].
Figure 2. Transseptal Transcatheter Mitral Valve Replacement with Cardiovalve system. (A-C) Severe MR with MV annulus dilated and thickened leaflets; (D) Fluoroscopic image of the Cardiovalve device with open grasp legs into the LV; (E) Fluoroscopic image showing the atrial flange and ventricular part deployment; (F) Fully deployed Cardiovalve device; (G-I) Echocardiographic images showing good expansion and stable position with mild MR and trace PVL. Figure provided by the authors. MR: Mitral regurgitation; LV: left ventricle; MV: mitral valve; PVL: paravalvular leak.
Clinical evidence
Early use of the Cardiovalve TMVR system in five patients demonstrated normal hemodynamics, complete elimination of MR, and no evidence of LVOT obstruction. Although three of the five patients died at 30 days due to issues with bleeding or vascular access, the surviving two have demonstrated promising outcomes, including sustained elimination of MR without further complications at 1 year[41]. Currently, the delivery system has been completely redesigned to reduce screen failures and to safely deploy the implant into the MV apparatus in a coaxial fashion. Several cases with the enhanced system have been performed under compassionate use with 100% success and no adverse events during follow-up, thereby showing promising results[49,50]. The AHEAD trial (European Feasibility Study of High Surgical Risk Patients with Severe Mitral Regurgitation Treated with the Cardiovalve Transfemoral Mitral Valve System, NCT03339115) will enroll thirty patients and evaluate the 30-day safety and performance and long-term clinical outcomes over a follow-up period of 2 years.
ALTAVALVE TRANSCATHETER MITRAL VALVE
The AltaValve device (4C Medical, Minnesota, USA) is designed for repositioning and retrieval, featuring an atrial fixation mechanism, and is implanted at the annular site via a transfemoral approach. The valve consists of two different structures: the chimney, which hosts a 27 mm trileaflet bovine pericardial valve, and the cage. The chimney is compressed by a snare to allow crimping and is released during valve implantation. The annular part of the cage has 3 sizes to accommodate different annular sizes with a degree of oversize and is covered with fabric to prevent paravalvular leak[51,52]. The cage can also be manufactured in different sizes to achieve oversizing in both the axial and sagittal planes, ensuring the valve is held securely in place. Currently, the LA sizes that can be accommodated by the cage range from 45 mm to 85 mm in height and from 55 mm to 85 mm in width. The transseptal delivery system is 29Fr. Interestingly, the cells of the cage are 24 Fr, meaning that reaccess to LA for any procedure is warranted.
Procedural steps
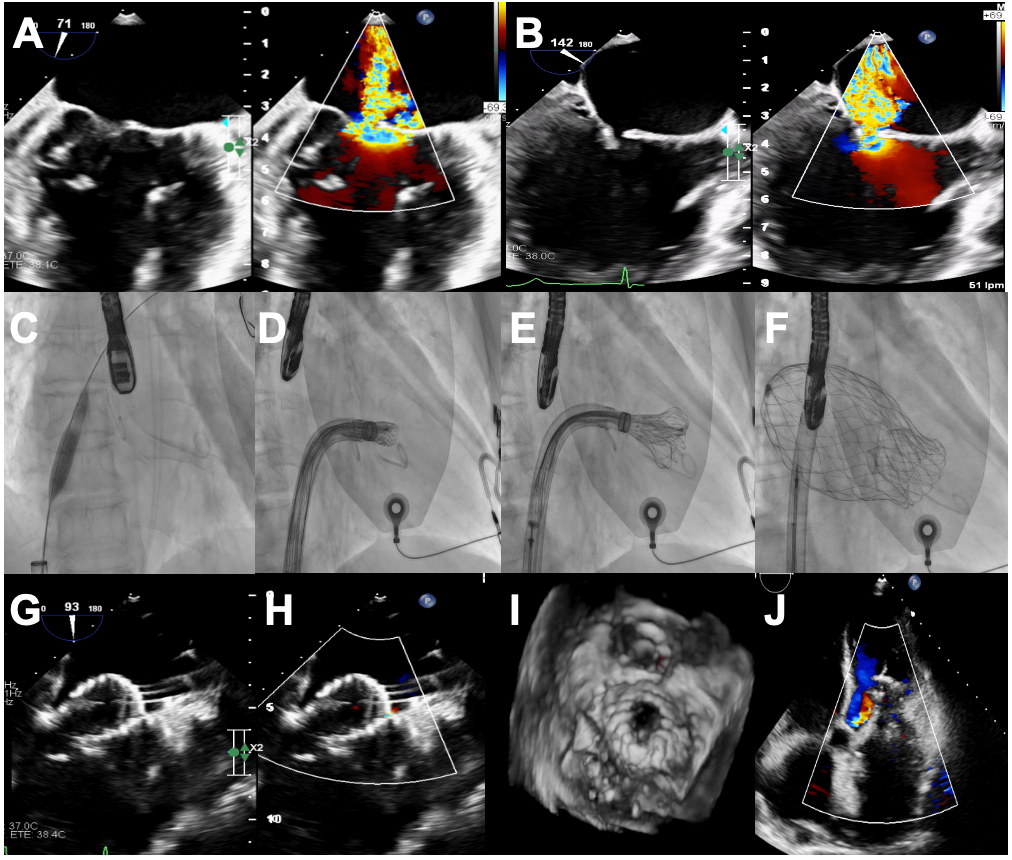
After performing the transseptal puncture 3.5-4.5 cm over the annular plane and centered over the medial commissure, the delivery system is advanced into the LA. It is then flexed to reach the annular plane, and by withdrawing both the transseptal sheath and the implant sheath, the lower part of the valve is exposed inside the MV annulus at an intended depth of 15 mm below the annular plane. Lateral engagement of the annulus is then required, and when both catheters are 1 cm above the superior part of the chimney, the snare is opened and the valve expands and begins functioning. Finally, the cage is completely deployed into the LA [Figure 3]. After retrieving the transseptal access, a decision must be made regarding closure of the residual atrial septal defect. Current expert opinion recommends closure with a patent foramen ovale device to maintain adequate LV filling and prevent any possibility of LVOT obstruction.
Figure 3. Transseptal Transcatheter Mitral Valve Replacement with AltaValve system. (A and B) Severe atrial functional MR with severe gap and posterior leaflet restriction; (C) Transseptal access; (D) Fluoroscopic image of the AltaValve system navigating to the MV plane; (E) Starting deployment; (F) Fully deployed AltaValve device; (G-J) Echocardiographic images showing good expansion and stable position with abolished MR and trace lateral PVL. Figure provided by the authors. MR: Mitral regurgitation; MV: mitral valve; PVL: paravalvular leak.
Clinical evidence
Due to its special fixation and anatomical requirements, AltaValve has a lower screen-failure rate compared with other TMVR platforms. Early experiences with the device via transseptal showed high technical success and clinical improvements in different anatomical scenarios including MAC, which is particularly appealing due to the transcatheter aortic valve implantation-like interaction of the frame with the calcified annulus[51]. Thirty high-surgical-risk patients (mean age ~ 77 years; 63% female) with symptomatic (NYHA ≥ II), grade 3+ MR - 43% degenerative, 50% functional, 7% mixed, and one-third with LVEF ≤ 50% - were treated in the AltaValve Early Feasibility Study via transseptal (57%) or transapical (43%) access. Technical success was achieved in 97% of cases, with all patients demonstrating none/trace residual MR (mean gradient 2.1 ± 1.0 mmHg) immediately postimplant and no major periprocedural safety events[42]. At 6 months, 86% of survivors improved to NYHA I/II. Sustained MR reduction was confirmed by an independent core laboratory. Quality of life, assessed by the Kansas City Cardiomyopathy Questionnaire (KCCQ), increased from 57.6 ± 20.7 to 67.1 ± 22.0, and exercise capacity improved from 237.9 ± 98.5 m to 298.0 ± 108.4 m. The pivotal ATLAS trial (NCT06465745) is currently enrolling patients. It comprises two nonrandomized cohorts: (1) patients without significant MAC, and (2) patients with moderate-to-severe MAC.
CEPHEA TRANSCATHETER MITRAL VALVE
The Cephea TMVR system (Abbott Vascular) is constructed for transseptal delivery and contains a self-expanding double-disc stent structure that incorporates a trileaflet pericardial valve. The ventricular disc serves to anchor the system in the subannular region, while the atrial disc is positioned at the base of the LA, securing the valve through axial force without requiring additional sub-valve anchors. Its design enables accommodation of diverse anatomical variations, while the centrally isolated core supporting the valve helps prevent dynamic compression of the leaflets by the annulus. The valve frame’s low profile is intended to minimize the risk of LVOT obstruction, including in cases with complex anatomy[53].
Procedural steps
Following septal crossing, the ventricular disc is released beginning at the distal edge of the anterior mitral leaflet; once it is fully expanded and seated against the annulus, the atrial disc is gradually deployed, achieving full valve apposition within the mitral annulus.
Clinical evidence
The first reported case involved a patient with HF secondary to severe MR resulting from a flail leaflet and prolapse of the P3 scallop[54]. Successful Cephea valve implantation resulted in immediate abolition of MR and significant improvement of HF symptoms (NYHA class I) at 28-week follow-up with normal valve function and sustained LVOT. Following the first in-human case, successful implantation was reported in 3 patients, demonstrating normal valve function at 6 months and no LVOT obstruction. All patients showed improvements in functional class and quality of life[55]. However, due to significant anatomical exclusions, a Cephea Gen2 (second-generation) has already been developed. This new generation includes additional sizes (28, 32, 36, and 40 mm) to accommodate different annular dimensions and configurations. The valve is stored dry without glutaraldehyde, and the ventricular portion has been redesigned with a fabric skirt to prevent paravalvular leak and open-cell frames to reduce the risk of LVOT obstruction. With these modifications, a screen-pass rate of approximately 50% is expected. Likewise, a Gen3 (3rd-generation) is already in development, with an expected acceptance rate of 78%.
The Cephea Gen2 EFS included 24 patients with a mean age of 75 years. PMR was the most frequent etiology, with a Society of Thoracic Surgeons Predicted Risk of Mortality (STS-PROM) score of 5.9% and a mean EF of 53.7%. Procedural success was obtained in all but one patient with MR equal to or less than mild in 100%. No procedural mortality was noted. At 30 days, mortality occurred in one patient, and the majority of patients were in NYHA functional class I-II[43]. The Cephea Early Feasibility Study is currently ongoing and aims to enroll 50 patients (NCT05061004).
SAPIEN M3 TRANSCATHETER MITRAL VALVE
The Sapien M3 transseptal TMVR system (Edwards Lifesciences, California, USA) is derived from the S3 valve for transcatheter aortic valve replacement, featuring a 29 mm frame with a polyethylene terephthalate skirt for paravalvular sealing[56]. The procedure uses a spiral nitinol dock - two wide turns secure the native leaflets and atrial position, with central turns anchoring the implant - followed by balloon-expanded valve delivery via the Edwards Commander system.
Procedural steps
While attached to the delivery catheter, the dock remains completely retrievable. The balloon-expandable valve is then deployed into this dock under rapid pacing, using a technique analogous to that of the Sapien 3 valve in treating degenerated mitral prostheses.
Clinical evidence
Following the initial first-in-human report[57], 35 high-risk patients with severe MR were treated with the Sapien M3 system as part of the US Early Feasibility Study (NCT03230747)[44]. Procedural success was accomplished in 31 patients. At 30 days, there was one death, one disabling stroke and one valve thrombosis. Most patients (93.8%) had minimal or no MR and showed improved functional status.
Although the established durability and widespread clinical experience of the Sapien valve platform may offer advantages over other TMVR systems, the intricate relationship between the docking implant and subvalvular anatomy can increase procedural difficulty. Husain et al. reported a 5-year follow-up of a TMVR using the SAPIEN M3 system in an 81-year-old woman treated for severe, mixedetiology MR[58]. Despite initial procedural success with only a trivial paravalvular leak, she developed moderate-to-severe PVL and NYHA II-III symptoms at 5 years; imaging showed a well-functioning prosthesis with intact leaflets and a stable mean gradient of 4 mmHg, with minor leaflet mobility at the paravalvular site. The paravalvular leak was successfully closed percutaneously via transseptal Amplatzer Vascular Plug deployment, reducing regurgitation to trivial levels. The etiology of the delayed paravalvular remains unclear, with possible contributions from native leaflet degeneration, mechanical leaflet injury, or reverse annular remodeling postTMVR. The ENCIRCLE trial (NCT04153292) is an ongoing, prospective, single-arm, multicenter study designed to evaluate the safety and effectiveness of the SAPIEN M3 system in patients with symptomatic MR of at least grade 3+, for whom commercially available surgical or transcatheter treatment options are considered unsuitable.
INNOVALVE TRANSCATHETER MITRAL VALVE
The Innovalve Transcatheter Mitral Valve Replacement System (Innovalve Bio Medical Ltd., Ramat Gan, Israel) is a novel prosthetic valve composed of an atrial skirt, a cylinder with three bovine pericardium cusps, and six arms that extend radially within the subvalvular apparatus. The arms are gradually rotated to engage the native MV apparatus with several fixation mechanisms, leading to robust prosthesis anchoring and sealing and providing a stable prosthesis and prevention of paravalvular leaks while holding the native anterior leaflet away from the LVOT. This is complemented by a posterior annular position with a small ventricular presence (≤ 13 mm) and posterior canting resulting from the posterior leaflet being relatively shorter.
Procedural steps
The valve is delivered over a stiff guidewire and positioned below the valvular plane between the leaflets and the papillary muscle tips; the capsule is opened and its arms exposed by gradual clockwise rotation until the anterior and posterior leaflets are wrapped with reduced mobility. The valve is then released by stepwise opening of the cylinder and atrial flange.
Clinical evidence
In the initial results of the TWISTGE pilot study (NCT05682066), ten high-surgical-risk or transcatheterpreferring patients with symptomatic moderate-to-severe or severe MR were enrolled across three centers in Tbilisi, Georgia[45]. All had NYHA class III symptoms and, after transseptal Innovalve implantation (median procedure time 49 ± 14 min), achieved technical success with no periprocedural mortality, stroke, myocardial infarction, LVOT obstruction, major bleeding, or vascular complications. At 30 days, 90% had ≤ mild paravalvular MR, one required uncomplicated surgery for worsening MR, and left ventricular EF trended upward to 49.3%. Functional capacity (6MWT +69 m) and KCCQ12 scores (+18.1 points, P < 0.05) improved in all patients.
UNMET CLINICAL NEEDS
Although the body of evidence on transseptal TMVR is growing rapidly, it remains important to acknowledge the limitations that shape current knowledge. Most of the available data derive from small, single-arm feasibility studies or multicenter registries. While these studies have been crucial in demonstrating proof of concept and early safety signals, the absence of randomized comparisons and the modest sample sizes inevitably limit the strength of the conclusions that can be drawn. Moreover, high rates of screening failure[59,60], often exceeding half of the referred population, underscore the challenges of patient selection and the restricted applicability of TMVR to real-world practice.
Selection bias further complicates interpretation of outcomes. Patients who undergo TMVR are typically carefully chosen by highly specialized centers, which may result in better procedural success and lower complication rates than would be observed in broader practice. As such, current reports may not fully capture the risks associated with more complex anatomies or less experienced operators. Longer-term data are also scarce, and the durability of these devices over many years remains uncertain. This is of particular relevance in younger or lower-risk patients, for whom questions of valve longevity and reintervention strategies are especially critical.
Another area of uncertainty relates to post-procedural management. Anticoagulation strategies vary widely across studies, and optimal regimens to prevent valve thrombosis while minimizing bleeding risk are not yet established. In addition, while TMVR almost universally eliminates MR, whether this translates into consistent improvements in survival and reductions in heart-failure hospitalization compared with TEER has not been conclusively determined. Comparative effectiveness studies are lacking, leaving clinicians to extrapolate from non-randomized experiences and observational insights.
CONCLUSIONS
In conclusion, transseptal TMVR is progressing, but not all devices are advancing at the same pace. Some platforms have generated only limited early experience and have not expanded much in recent years. In contrast, newer systems are being studied in actively recruiting clinical trials, aiming to provide more robust data on safety, efficacy, and long-term durability. This landscape shows a field in transition: while certain devices remain in a proof-of-concept stage, others are moving into broader evaluation and may soon shape clinical practice. Ultimately, larger registries and randomized studies will be essential to define the role of transseptal TMVR in routine care.
DECLARATIONS
Acknowledgments
We acknowledge that the heart and human figure illustrations in the Graphical Abstract were created with BioRender.com.
Authors’ contributions
Substantial contributions to study conception and design, and data analysis and interpretation: Echarte-Morales J, Reyes-Mitre A, Ávila EM, Estévez-Loureiro R
Data acquisition and provision of administrative, technical, or material support: Baz-Alonso JA, Barreiro-Pérez M, Gonzalez-Ferreiro R, Caneiro-Queija B, Parada-Barcia JA
Drafting of the manuscript: Estévez-Loureiro R, Echarte-Morales J
Critical revision of the manuscript: Estévez-Loureiro R, Iñiguez-Romo A
Final approval of the manuscript: All authors
Availability of data and materials
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable. Written consent was obtained from the patients shown in Figure 2 and Figure 3.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Iung B, Delgado V, Rosenhek R, et al. ; EORP VHD II Investigators. Contemporary Presentation and Management of Valvular Heart disease: The EUR observational research programme valvular heart disease II survey. Circulation. 2019;140:1156-69.
2. Unger P, Tribouilloy C. Aortic stenosis with other concomitant valvular disease: aortic regurgitation, mitral regurgitation, mitral stenosis, or tricuspid regurgitation. Cardiol Clin. 2020;38:33-46.
3. Badhwar V, Chikwe J, Gillinov AM, et al. Risk of surgical mitral valve repair for primary mitral regurgitation. J Am Coll Cardiol. 2023;81:636-48.
4. Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the management of patients with valvular heart disease: executive summary: a report of the American college of cardiology/American heart association joint committee on clinical practice guidelines. Circulation. 2021;143:e35-71.
5. Deja MA, Malinowski M, Widenka K, et al. Repair or replacement for secondary mitral regurgitation: results from polish national registry. Ann Thorac Surg. 2022;113:146-56.
6. Vahanian A, Beyersdorf F, Praz F, et al. ; ESC/EACTS Scientific Document Group. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2022;43:561-632.
7. Gavazzoni M, Taramasso M, Zuber M, et al. Conceiving MitraClip as a tool: percutaneous edge-to-edge repair in complex mitral valve anatomies. Eur Heart J Cardiovasc Imaging. 2020;21:1059-67.
8. Elbadawi A, Dang AT, Hamed M, et al. Transcatheter edge-to-edge repair for mitral regurgitation using PASCAL or MitraClip. Catheter Cardiovasc Interv. 2023;102:521-7.
9. Wang J, Liu X, Pu Z, et al. Safety and efficacy of the DragonFly system for transcatheter valve repair of degenerative mitral regurgitation: one-year results of the DRAGONFLY-DMR trial. EuroIntervention. 2024;20:e239-49.
10. Feldman T, Foster E, Glower DD, et al. ; EVEREST II Investigators. Percutaneous repair or surgery for mitral regurgitation. N Engl J Med. 2011;364:1395-406.
11. von Bardeleben RS, Mahoney P, Morse MA, et al. 1-year outcomes with fourth-generation mitral valve transcatheter edge-to-edge repair from the EXPAND G4 Study. JACC Cardiovasc Interv. 2023;16:2600-10.
12. Zahr F, Smith RL, Gillam LD, et al. 1-year outcomes from the CLASP IID randomized trial for degenerative mitral regurgitation. 2023;16:2803-16.
13. Stone GW, Lindenfeld J, Abraham WT, et al. ; COAPT Investigators. Transcatheter mitral-valve repair in patients with heart failure. N Engl J Med. 2018;379:2307-18.
14. Anker SD, Friede T, von Bardeleben RS, et al. ; RESHAPE-HF2 Investigators. Transcatheter valve repair in heart failure with moderate to severe mitral regurgitation. N Engl J Med. 2024;391:1799-809.
15. Stocker TJ, Stolz L, Karam N, et al. ; EuroSMR Investigators. Long-term outcomes after edge-to-edge repair of secondary mitral regurgitation: 5-year results from the EuroSMR registry. JACC Cardiovasc Interv. 2024;17:2543-54.
16. Kar S, von Bardeleben RS, Rottbauer W, et al. Contemporary outcomes following transcatheter edge-to-edge repair: 1-year results from the EXPAND study. JACC Cardiovasc Interv. 2023;16:589-602.
17. Webb JG, Hensey M, Szerlip M, et al. 1-year outcomes for transcatheter repair in patients with mitral regurgitation from the CLASP study. JACC Cardiovasc Interv. 2020;13:2344-57.
18. Spargias K, Lim DS, Makkar R, et al. Three-year outcomes for transcatheter repair in patients with mitral regurgitation from the CLASP study. Catheter Cardiovasc Interv. 2023;102:145-54.
19. Taramasso M, Maisano F, Latib A, et al. Clinical outcomes of MitraClip for the treatment of functional mitral regurgitation. EuroIntervention. 2014;10:746-52.
20. Higuchi S, Orban M, Stolz L, et al. Impact of residual mitral regurgitation on survival after transcatheter edge-to-edge repair for secondary mitral regurgitation. JACC Cardiovasc Interv. 2021;14:1243-53.
21. Boekstegers P, Hausleiter J, Schmitz T, et al. ; MITRA-PRO Investigators. Intraprocedural residual mitral regurgitation and survival after transcatheter edge-to-edge repair: prospective German multicenter registry (MITRA-PRO). JACC Cardiovasc Interv. 2023;16:574-85.
22. Singh GD, Price MJ, Shuvy M, et al. Combined impact of residual mitral regurgitation and gradient after mitral valve transcatheter edge-to-edge repair. JACC Cardiovasc Interv. 2024;17:2530-40.
23. Hausleiter J, Stocker TJ, Adamo M, Karam N, Swaans MJ, Praz F. Mitral valve transcatheter edge-to-edge repair. EuroIntervention. 2023;18:957-76.
24. Sugiura A, Kavsur R, Spieker M, et al. Recurrent mitral regurgitation after MitraClip: predictive factors, morphology, and clinical implication. Circ Cardiovasc Interv. 2022;15:e010895.
25. Sorajja P, Vemulapalli S, Feldman T, et al. Outcomes with transcatheter mitral valve repair in the United States: An STS/ACC TVT registry report. J Am Coll Cardiol. 2017;70:2315-27.
26. Adamo M, Pagnesi M, Popolo Rubbio A, et al. Predictors of optimal procedural result after transcatheter edge-to-edge mitral valve repair in secondary mitral regurgitation. Catheter Cardiovasc Interv. 2022;99:1626-35.
27. Orban M, Rottbauer W, Williams M, et al. Transcatheter edge-to-edge repair for secondary mitral regurgitation with third-generation devices in heart failure patients - results from the Global EXPAND Post-Market study. Eur J Heart Fail. 2023;25:411-21.
28. Kubo S, Yamamoto M, Saji M, et al. ; OCEAN‐Mitral Investigators. One-year outcomes and their relationship to residual mitral regurgitation after transcatheter edge-to-edge repair with MitraClip device: insights from the OCEAN-mitral registry. J Am Heart Assoc. 2023;12:e030747.
29. Chhatriwalla AK, Vemulapalli S, Szerlip M, et al. Operator experience and outcomes of transcatheter mitral valve repair in the United States. J Am Coll Cardiol. 2019;74:2955-65.
30. Taramasso M, Gavazzoni M, Nickenig G, Maisano F. Transcatheter mitral repair and replacement: which procedure for which patient? EuroIntervention. 2019;15:867-74.
31. Muller DWM, Farivar RS, Jansz P, et al. ; Tendyne global feasibility trial investigators. Transcatheter mitral valve replacement for patients with symptomatic mitral regurgitation: a global feasibility trial. J Am Coll Cardiol. 2017;69:381-91.
32. Fukui M, Sorajja P, Gössl M, et al. Left ventricular remodeling after transcatheter mitral valve replacement with Tendyne: new insights from computed tomography. JACC Cardiovasc Interv. 2020;13:2038-48.
33. Muller DWM, Sorajja P, Duncan A, et al. 2-year outcomes of transcatheter mitral valve replacement in patients with severe symptomatic mitral regurgitation. J Am Coll Cardiol. 2021;78:1847-59.
34. Hell MM, Wild MG, Baldus S, et al. ; TENDER Investigators. Transapical mitral valve replacement: 1-year results of the real-world Tendyne European experience registry. JACC Cardiovasc Interv. 2024;17:648-61.
35. Ben Ali W, Ludwig S, Duncan A, et al. ; CHOICE-MI Investigators. Characteristics and outcomes of patients screened for transcatheter mitral valve implantation: 1-year results from the CHOICE-MI registry. Eur J Heart Fail. 2022;24:887-98.
36. Al-Tawil M, Butt S, Reap S, et al. Transseptal vs transapical transcatheter mitral valve-in-valve and valve-in-ring implantation: a systematic review and meta-analysis. Curr Probl Cardiol. 2023;48:101684.
37. Anselmi A, Leurent G, Donal E. Transcatheter mitral valve replacement: technology update. advances in cardiovascular technology. Elsevier; 2022. pp. 319-34.
38. Tartaglia F, Antonelli G, Gitto M, et al. TMVR for the treatment of mitral regurgitation: a state-of-the-art review. Circ Cardiovasc Interv. 2025;18:e015298.
39. Zahr F, Song HK, Chadderdon S, et al. 1-year outcomes following transfemoral transseptal transcatheter mitral valve replacement: intrepid TMVR early feasibility study results. JACC Cardiovasc Interv. 2023;16:2868-79.
40. Schneider LM, Worthley S, Nickenig G, et al. 1-year outcomes following transfemoral transseptal transcatheter mitral valve replacement: The HighLife TSMVR feasibility study. JACC Cardiovasc Interv. 2023;16:2854-65.
41. Maisano F. Cardiovalve device attributes, implant procedure and early results. Presented at EuroPCR 2019; May 22, 2019; Paris, France. https://www.tctmd.com/slide/cardiovalve-device-attributes-implant-procedure-and-early-results. (accessed 7 January 2026).
42. Kaneko et al. AltaValve Early Feasibility Study Results. Presented at: PCR Tokyo Valves 2025. T. AltaValve System Early Feasibility Study Results. AltaValve Early Feasibility Study Results Presented at: PCR Tokyo Valves 2025. https://www.pcronline.com/Cases-resources-images/Resources/Course-videos-slides/2025/PCR-Tokyo-Valves/Late-Breaking-Trial-session. (accessed 7 January 2026).
43. Granada JF. Cephea TMVR Program Technology and Clinical Update. Presented at: New York Valves; June 26, 2025; New York, USA. Available from: https://d14d5nk8lue86f.cloudfront.net/public/2025-06/5dba363c-2b20-470c-b9e0-79974d7a5983.pdf. (accessed 30 December 2025).
44. Makkar R, O'neill W, Whisenant B, et al. TCT-8 updated 30-day outcomes for the U.S. early feasibility study of the SAPIEN M3 transcatheter mitral valve replacement system. J Am Coll Cardiol. 2019;74:B8.
45. Meerkin D, Guetta V, Shaburishvili T, et al. Early experience with the innovalve transcatheter mitral valve replacement system. JACC Cardiovasc Interv. 2024;17:712-4.
46. Meredith I, Bapat V, Morriss J, McLean M, Prendergast B. Intrepid transcatheter mitral valve replacement system: technical and product description. EuroIntervention. 2016;12:Y78-80.
47. Zahr F, Song HK, Chadderdon SM, et al. 30-day outcomes following transfemoral transseptal transcatheter mitral valve replacement: intrepid TMVR early feasibility study results. JACC Cardiovasc Interv. 2022;15:80-9.
48. Lange R, Piazza N. The HighLife transcatheter mitral valve implantation system. EuroIntervention. 2015;11 Suppl W:W82-3.
49. Maisano F, Benetis R, Rumbinaite E, et al. 2-year follow-up after transseptal transcatheter mitral valve replacement with the cardiovalve. JACC Cardiovasc Interv. 2020;13:e163-4.
50. Sherif M, Sündermann SH, Maisano F, et al. Cardiovalve in mitral valve position-additional solution for valve replacement. Front Cardiovasc Med. 2022;9:960849.
51. Généreux P, Wróbel K, Rinaldi MJ, et al. AltaValve atrial fixation system for the treatment of severe mitral regurgitation and mitral annular calcification. Struct Heart. 2024;8:100294.
52. Généreux P, Wróbel K, Ninios V, et al. 6-month outcomes of a supra-annular transcatheter mitral valve replacement: an early feasibility study. JACC Cardiovasc Interv. 2025;18:2584-92.
53. Coisne A, Rodés-Cabau J, Ludwig S, et al. Impact of a dedicated transseptal transcatheter mitral valve replacement system on cardiac remodeling and hemodynamics. JACC Cardiovasc Interv. 2023;16:1310-2.
54. Modine T, Vahl TP, Khalique OK, et al. First-in-human implant of the Cephea transseptal mitral valve replacement system. Circ Cardiovasc Interv. 2019;12:e008003.
55. Alperi A, Dagenais F, Del Val D, et al. Early experience with a novel transfemoral mitral valve implantation system in complex degenerative mitral regurgitation. JACC Cardiovasc Interv. 2020;13:2427-37.
56. Penteris M, Lampropoulos K. The SAPIEN M3 system for transcatheter mitral valve replacement: a new era begins. Cardiovasc Revasc Med. 2025;79:120-5.
57. Webb JG, Murdoch DJ, Boone RH, et al. Percutaneous transcatheter mitral valve replacement: first-in-human experience with a new transseptal system. J Am Coll Cardiol. 2019;73:1239-46.
58. Husain A, Meier D, Dundas J, et al. Transcatheter mitral valve replacement: 5 years later. JACC Cardiovasc Interv. 2023;16:2920-2.
59. Niikura H, Gössl M, Kshettry V, et al. Causes and clinical outcomes of patients who are ineligible for transcatheter mitral valve replacement. JACC Cardiovasc Interv. 2019;12:196-204.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].