Resistance to immune checkpoint inhibitors in hepatocellular carcinoma: mechanisms and combination strategies to overcome it
Abstract
Hepatocellular carcinoma (HCC) is predominantly diagnosed at an advanced stage and ranks as the third leading cause of cancer-related mortality worldwide. In recent years, the advent and clinical application of immune checkpoint inhibitors (ICIs) have markedly transformed the therapeutic landscape of HCC, yielding superior overall survival (OS) outcomes compared with conventional targeted therapies. Nevertheless, clinical benefit is limited to a subset of patients, largely due to drug resistance. Current evidence implicates both the tumor microenvironment and tumor cell-intrinsic factors as pivotal determinants of ICI resistance. Combination strategies have emerged as a central approach in HCC therapy, significantly enhancing the antitumor activity of ICIs. Notably, ICIs plus targeted therapy (anti-angiogenic agents), dual-ICI blockade, and ICI combined with locoregional therapy (transarterial chemoembolization) currently represent the most well-established and clinically validated approaches, being approved as first- or second-line treatments. In addition, complementary approaches, including traditional Chinese medicine and modulation of intestinal microbiota, demonstrate synergistic antitumor effects when integrated with ICI, representing promising avenues for further therapeutic optimization. This review mainly elucidates the underlying mechanisms of ICI resistance and examines both established and emerging combination strategies for HCC. Furthermore, it explores potential approaches to enhance the efficacy of ICI-based regimens, with the aim of improving long-term outcomes for patients with advanced HCC.
Keywords
INTRODUCTION
Liver cancer remains a highly aggressive malignancy with persistently high global incidence and mortality rates, and hepatocellular carcinoma (HCC) accounts for more than 90% of all diagnosed cases[1]. Patients with HCC often face critical clinical challenges, such as late diagnosis, high rates of recurrence, and poor prognosis. With a 5-year survival rate for advanced HCC below 20%, it ranks as the third most common cause of cancer-related death globally[2]. Therefore, the development of highly effective therapeutic strategies for HCC remains a critical and urgent clinical priority.
Traditional therapeutic strategies for HCC primarily encompass surgical resection, liver transplantation, radiotherapy, chemotherapy, and targeted therapy[3]. Surgical resection is currently the preferred treatment for liver cancer, yet approximately 70% of patients experience recurrence within five years post-surgery, significantly impacting survival time[4]. Patients who underwent liver transplantation typically exhibit high survival rates and low recurrence rates. However, due to organ shortages and risk of immune rejection, only around 20% of HCC patients are eligible for liver transplantation[5]. While radiotherapy and chemotherapy can effectively inhibit tumor cell proliferation, they often damage normal tissues and generate toxic side effects due to limited target specificity[6]. Targeted therapy primarily includes multi-kinase inhibitors such as sorafenib and lenvatinib, which can effectively suppress HCC progression and reduce side effects. However, the development of drug resistance to targeted therapy makes it unsuitable for long-term use in HCC treatment[7].
Immunotherapy, represented by immune checkpoint inhibitors (ICIs), is a novel and highly effective treatment strategy for HCC. ICIs are designed to disrupt key molecular mechanisms involved in tumor immune evasion, thereby potentiating the host’s immune response against cancer. As an inflammation-driven cancer, HCC represents a promising target for immunotherapeutic interventions. ICI targeting programmed cell death protein 1 or its ligand (PD-1/PD-L1) and cytotoxic T-lymphocyte antigen 4 (CTLA-4) have already been incorporated into clinical practice for advanced HCC, effectively improving patient survival. Moreover, a series of emerging immune checkpoints, including T-cell immunoglobulin and mucin domain-containing protein 3 (TIM-3), lymphocyte activation gene-3 (LAG-3), T-cell immunoreceptor with immunoglobulin and ITIM domains (TIGIT), V-domain Ig suppressor of T cell activation (VISTA), B7-H3 (CD276), B and T lymphocyte attenuator (BTLA), and indoleamine-2,3-dioxygenase 1 (IDO1), have been identified, and blockade of these checkpoints also demonstrates encouraging therapeutic potential in HCC patients[8-14] [Figure 1 and Table 1]. Nonetheless, emerging evidence suggests that ICI therapy is susceptible to drug resistance, posing a significant challenge to HCC immunotherapy. This review will elucidate the mechanisms underlying resistance to ICIs and explore combination strategies to overcome it, with the aim of providing new insights and therapeutic avenues to enhance ICI efficacy in HCC treatment.
Figure 1. Immune checkpoints and their ligands inducing immune escape of HCC cells. Created with BioRender. H, T. (2025) https://BioRender.com/ko8kl51. PD-1: Programmed cell death protein 1; PD-L1: programmed cell death ligand 1; TIM-3: T-cell immunoglobulin and mucin domain containing 3; CTLA-4: cytotoxic T-lymphocyte-associated protein 4; LAG-3: lymphocyte-activation gene-3; TIGIT: T-cell immunoreceptor with immunoglobulin and ITIM domains; VISTA: V-domain Ig suppressor of T cell activation; BTLA: B and T lymphocyte attenuator; B7-H3: CD276.
Immune checkpoints and inhibitors used for HCC therapy
| Target | Drug name | Approval status |
| PD-1 | Nivolumab Pembrolizumab Tislelizumab Camrelizumab Sintilimab Toripalimab Nofazinlimab | FDA approved (2017) FDA approved (2018) Approved in China (2024) Approved in China (2020) Approved in China (2021) In clinical trials In clinical trials |
| PD-L1 | Atezolizumab Durvalumab Avelumab Sugemalimab Envafolimab Adebelimumab TQB2450 | FDA approved (2017) In clinical trials In clinical trials In clinical trials In clinical trials In clinical trials In clinical trials |
| CTLA-4 | Ipilimumab Tremelimumab IBI310 | FDA approved (2020, combination therapy) FDA approved (2022, combination therapy) In clinical trials |
| VISTA | CA-170 | In clinical trials |
| TIGIT | Tiragolumab | In clinical trials |
| LAG-3 | Relatlimab | In clinical trials |
| BTLA | Tifcemalimab | In clinical trials |
| TIM-3 | Sabatolimab Cobolimab | In clinical trials In clinical trials |
| B7-H3 | Enoblituzumab | In clinical trials |
RESISTANCE MECHANISMS TO ICIS IN HCC
ICI therapy has achieved considerable success in the treatment of HCC. However, only 25%-40% of HCC patients benefit from this therapy, which is largely attributable to ICI resistance[15]. Based on the origin of factors contributing to ICI resistance, the underlying mechanisms primarily fall into two categories: the immunosuppressive tumor microenvironment (TME) [Figure 2] and tumor cell-intrinsic changes [Figure 3]. These mechanisms primarily involve immune system dysfunction, remodeling of the immune microenvironment, lack of neoantigens or impaired antigen presentation, dysregulated signaling pathways, metabolic reprogramming, expression of alternative immune checkpoints, and epigenetic modifications.
Figure 2. Immunosuppressive TME involved in ICI resistance. Created with BioRender. H, T. (2025) https://BioRender.com/1q198aq. MDSC: Myeloid-derived suppressor cells; NK: natural killer cells; Treg: regulatory T cells; DC: dendritic cells; M2: M2 macrophages; TAN: tumor-associated neutrophils; Th: helper T cells; HSC: hepatic stellate cells; LSEC: liver sinusoidal endothelial cells; CAF: cancer-associated fibroblasts; EMT: epithelial-mesenchymal transition.
Figure 3. Tumor cell-intrinsic factors involved in ICI resistance. Created with BioRender. H, T. (2025) https://BioRender.com/aezvlck. TF: Transcription factor; Ub: ubiquitination; Ace: acetylation; Lac: lactylation; Me: methylation; cAMP: cyclic adenosine monophosphate; ROS: reactive oxygen species; TMB: tumor mutational burden; MHC: major histocompatibility complex; IDO1: indoleamine-2,3-dioxygenase 1; PD-L1: programmed cell death ligand 1; B7-H3: CD276; lncRNA: long non-coding RNA.
IMMUNOSUPPRESSIVE TME
Dysfunction of antitumor immune cells
Among immune cells, cytotoxic CD8+ T cells, dendritic cells (DCs), and natural killer (NK) cells typically exert antitumor effects, and their dysfunction significantly impairs the efficacy of immunotherapy[16]. Liver functions as a central immunomodulatory organ that generally maintains an immune-tolerant state to ensure local and systemic protection. Studies have shown that some liver cancers are “cold tumors”, characterized by limited T-cell infiltration, a feature that contributes significantly to primary ICI resistance[17]. Defects in antigen-presenting cells (APCs), impaired T-cell activation, and T-cell exclusion further led to T-cell deficiency in HCC tissues[18]. Moreover, the absence of CD8+ T cells in liver cancer is often accompanied by an increased frequency of exhausted CD8+ T cells. These exhausted cells typically overexpress immune checkpoints such as PD-1, CTLA-4, TIM-3, LAG-3, and TIGIT, resulting in immune tolerance. DCs contribute to antitumor immunity by presenting antigens to T cells. However, immature DCs may promote tumor tolerance through the induction of regulatory T cells (Tregs) and suppression of effector T cells[19]. Even mature DCs sometimes facilitate immune evasion by promoting Treg differentiation and secreting immunosuppressive cytokines[20]. Additionally, abnormal proportions or functional impairments in NK cells also drive ICI resistance. For example, HCC patients exhibit an increased population of Siglec-9+ NK cells with reduced cytotoxicity and disrupted homeostasis[21]. The expression of immune checkpoints such as TIGIT and TIM-3 on NK cells, along with diminished production of cytokines such as interferon (IFN)-γ and tumor necrosis factor (TNF)-α, further attenuates the effectiveness of ICI therapy[22].
Infiltration of immunosuppressive cells
Immunosuppressive cells within the TME represent a major mechanism underlying resistance to ICIs. These cells primarily include Kupffer cells, tumor-associated macrophages (TAMs), Tregs, and myeloid-derived suppressor cells (MDSCs). Tregs act as key negative regulators of CD8+ T cells, suppressing immune responses through expression of immune-inhibitory molecules such as CTLA-4, CD39, and CD73, as well as the secretion of immunosuppressive cytokines including interleukin (IL)-10 and transforming growth factor (TGF)-β[23]. Recent studies also indicate that Tregs can induce immunosuppression via interactions with type 2 conventional DCs[24]. Other immunosuppressive cells, including Kupffer cells, TAMs, and MDSCs, promote immune tolerance through multiple mechanisms: they facilitate the proliferation, infiltration, and activation of Tregs; upregulate checkpoint ligands (e.g., PD-L1) and immunosuppressive factors such as vascular endothelial growth factor (VEGF), arginase-1 (ARG-1), and IDO1[25,26]; inhibit the activation of CD8+ T cells and NK cells; and reduce the expression of major histocompatibility complex class II (MHC-II) and co-stimulatory molecules. Additionally, TAMs have been shown to confer ICI resistance by promoting epithelial-mesenchymal transition (EMT) of HCC cells or by forming macrophage-coated tumor clusters (MCTCs) that impede the infiltration of cytotoxic T cells[27,28]. Furthermore, emerging evidence suggests that other immune cells, including B cells, monocytes, helper T cells (Th), and tumor-associated neutrophils (TANs) also contribute to ICI resistance[29]. These cells dampen antitumor immunity by expressing immune checkpoints such as PD-1, TIM-3, CTLA-4, and chemokine receptor 4/6 (CCR4/CCR6), or accelerating the exhaustion of CD8+ T cells.
Non-immune cells and factors involved in TME
Besides immune cells, functional alterations in other components of TME such as hepatic stellate cells (HSCs) and liver sinusoidal endothelial cells (LSECs) also contribute to ICI resistance. Studies have shown that HSCs promote the infiltration of Tregs and MDSCs into HCC tissues through the secretion of hepatocyte growth factor (HGF). Meanwhile, LSECs can suppress immune responses by expressing PD-L1 and facilitating Treg recruitment[30]. Cancer-associated fibroblasts (CAFs) represent another critical element within the TME, which secrete various cytokines, exosomes, and other effector molecules that enable tumor cells to evade immune surveillance[31]. For instance, CAFs recruit immunosuppressive cells and inhibit T cell responses through the production of TGF-β, IL-6, and chemokine ligand (CCL)2[32,33]. Furthermore, CAFs interact with TAMs to promote M2 polarization, which supports HCC proliferation and fosters an immunosuppressive environment[34]. Additionally, other TME features such as extracellular matrix remodeling, angiogenesis, the presence of immunosuppressive soluble factors, hypoxia, and the acidic conditions within tumor tissues collectively contribute to the development of ICI resistance[35,36].
TUMOR CELL-INTRINSIC FACTORS
Defects in antigen presentation
MHC ligands on tumor cells are directly recognized by T-cell receptors (TCRs) on CD8+ or CD4+ T cells and play a crucial role in antitumor immune responses. Reduced expression or impaired binding affinity of MHC ligands disrupts antigen presentation, representing a key mechanism of resistance to ICIs[37]. Studies have reported that downregulation of MHC ligands leads to diminished antitumor T cell activity[38]. Furthermore, HCC patients with low expression of MHC ligands often remain unresponsive to ICIs. Additionally, tumor mutational burden (TMB), defined as the total number of somatic mutations per megabase within the coding regions of the tumor genome, serves as a quantitative biomarker for neoantigen load and mutation frequency[39]. Theoretically, a higher TMB increases the diversity and immunogenicity of neoantigens, thereby enhancing T-cell-mediated tumor killing[40]. As a result, elevated TMB is associated with improved response to ICIs, whereas tumors with low TMB frequently exhibit reduced immunogenicity, which constitutes a major mechanism of primary resistance to ICIs in HCC patients[40].
Abnormal immune checkpoint expression
Tumor cells often exploit immune checkpoints (e.g., PD-L1, CTLA-4) to evade cytotoxic T-cell-mediated antitumor immunity[41]. However, the absence or low expression of immune checkpoints leads to a lack of response to ICI therapy, a phenomenon known as primary resistance. Additionally, compensatory upregulation of alternative immune checkpoints also undermines the efficacy of ICIs, typically contributing to acquired resistance. For example, treatment of HCC patients with PD-1/PD-L1 inhibitors has been shown to upregulate the major ligand of LAG-3, resulting in immune resistance[13]. Other immune checkpoints such as TIM-3, TIGIT, and BTLA have been reported to promote T-cell exhaustion under conditions of chronic antigen stimulation or down-regulation of IFN-γ, further enhancing resistance to PD-1/PD-L1 inhibitors in HCC[10,12,14]. Moreover, increased expression of IDO1 in HCC prevents phagocytosis of macrophages, and its inhibition enhances the efficacy of anti-PD-1 therapy[8]. Recently, two emerging immune checkpoints, including B7-H3 and VISTA (PD-1 homolog), have also been shown to promote immune escape by inhibiting the infiltration and activation of T cells in HCC[11,42].
Dysregulation of immune-associated signaling pathways
The TGF-β-induced pathway represents one of the most extensively studied mechanisms underlying resistance to ICIs in HCC. First, TGF-β1 upregulates the expression of PD-L1 and B7-H3 during EMT in HCC cells[43]. Additionally, TGF-β secreted by HCC cells not only exerts inhibitory effects on NK cells and DCs, but also suppresses CD8+ T cell activity and promotes its exhaustion by downregulating IFN-γ and upregulating PD-1[44,45]. Furthermore, TGF-β enhances the immunosuppressive microenvironment within HCC tissues by promoting eosinophil and mast cell chemotaxis, inducing M2-type macrophage polarization, and amplifying Tregs activity[46,47]. Abnormal activation of the WNT/β-catenin pathway is another key contributor to ICI resistance in HCC. Approximately 30% of HCC patients harbor catenin beta-1 gene (CTNNB1) mutations (encoding β-catenin), which are associated with non-response to ICIs[48]. β-Catenin activation also reduces the expression of chemokines such as CCL1 and CCL4, impairs the infiltration of DCs and antigen-specific T cells, and suppresses NK cell activity, thereby diminishing the antitumor efficacy of ICIs[49,50]. Moreover, activation of the mitogen-activated protein kinase (MAPK) signaling pathway influences both immune-cell infiltration and immune checkpoint expression. For example, the epidermal growth factor receptor (EGFR)-p38 MAPK and MAPK/nuclear Factor kappa-light-chain-enhancer of activated B cells (NF-κB) axes enhance PD-L1 and CD274 expression to facilitate immune escape[51-53]. Other signaling pathways including IFN-γ/janus kinase/signal transducer and activator of transcription (JAK/STAT), phosphoinositide 3-kinase/protein kinase B (PI3K/AKT), and NF-κB have also been implicated in ICI resistance through the regulation of cytokines and immune checkpoints (e.g., TGF-β, VEGF, type I interferon, PD-L1) expression, modulation of immune-cell function [e.g., NK cells, T helper 1 (Th1) cells, CD8+ T cells], and promotion of Treg accumulation[54-56].
Epigenetic modifications
Epigenetic modifications influence the efficacy of ICIs primarily by regulating the expression of effector genes, and the mechanisms include DNA and histone modifications, non-coding RNAs, and chromatin remodeling. Studies have shown that DNA methyltransferases (DNMTs)-induced hypermethylation suppresses MHC class I (MHC-I) expression, leading to impaired tumor antigen presentation[57]. Additionally, histone methyltransferases [e.g., enhancer of zeste 2 polycomb repressive complex 2 subunit (EZH2), SET domain bifurcated histone Llysine methyltransferase 1 (SETDB1), protein arginine methyltransferase 5 (PRMT5), and methyltransferase like 1/3 (METTL1/3)] and demethylases [e.g., lysine demethylase 1 (KDM1)] modulate the expression of PD-L1, MHC-II, and chemokines [e.g., C-X-C motif chemokine ligand 5 (CXCL5), CXCL8, CXCL10] in HCC cells. These changes inhibit the infiltration and function of NK cells and CD8+ T cells while enhancing the recruitment of MDSCs[58,59]. Furthermore, histone acetylation and deacetylation modifications mediated by enzymes such as lysine acetyltransferase 2A (KAT2A) and histone deacetylase (HDAC) 8 alter the function and abundance of Tregs and CD8+ T cells within the TME through chemokine regulation, and ultimately compromise ICI efficacy in HCC. Emerging evidence indicates that post-translational modifications (PTMs) on non-histone proteins also play important roles in modulating ICI response[60]. For instance, deacetylase silent information regulator 2 (SIRT2) and HDAC2 have been reported to promote immune escape by regulating the stability of fibrinogen-like protein 1 (FGL1) and the subcellular localization of PD-L1[61,62]. Moreover, high expression of HDAC1, HDAC2, and HDAC3 in HCC patients is associated with suppressed IFN-γ pathway activity, enhanced T-cell exclusion, and increased MDSC accumulation, contributing to ICI non-responsiveness[63]. Non-coding RNAs, including microRNAs and long non-coding RNAs (lncRNAs), represent another important class of epigenetic regulators affecting ICI efficacy in HCC. For example, miR-455 and lncRNA myocardial infarction associated transcript (MIAT) have been shown to dampen antitumor immune responses by modulating PD-L1 expression[64,65].
Tumor cell-derived metabolites and exosomes
Metabolites secreted by tumor cells influence the immune status of HCC primarily by modulating the function and infiltration of immune cells within the TME. Multiple studies indicate that enhanced aerobic glycolysis in tumor cells leads to substantial lactate release, acidifying the TME and promoting the infiltration of immunosuppressive cells. Lactate also drives M2 macrophage polarization while inhibiting the activities of CD8+ T cells, NK cells, and DCs[66]. Furthermore, dysregulated lipid metabolism in HCC reduces the abundance of CD4+ T cells and M1 macrophages and impairs NK cell function[67,68]. For instance, in obesity-associated HCC models, tumor-derived prostaglandin E2 (PGE2) binds to the prostaglandin E2 receptor 4 (EP4) receptor on immune cells, suppressing the expression of IFN-γ and TNF-α while inducing IL-10 and IL-6[69]. Additionally, HCC-secreted oleic acid (OA) promotes an immunosuppressive phenotype in macrophages, and its inhibition enhances the antitumor effects of anti-PD-1 therapy[70]. Clinical evidence suggests that elevated asparagine or glutamine metabolism in HCC patients is associated with increased infiltration of Tregs, macrophages, and memory B cells, fostering an immunosuppressive TME[71,72]. Inhibition of glutamine metabolism significantly improves response to ICIs in HCC[72]. Moreover, the accumulation of cyclic adenosine monophosphate (cAMP) has been shown to suppress CD4+ and CD8+ T cell responses while enhancing Treg activity through binding to adenosine A2A receptor (A2AR)[73]. In addition, exosomes carrying microRNAs, circular RNAs, lncRNAs, and proteins such as PD-1/PD-L1 collectively attenuate the cytotoxicity of T cells and NK cells[74]. Moreover, exosomes from tumor cells promote the infiltration of M2 macrophages, Tregs, DCs, and N2 neutrophils, as well as angiogenesis, further driving resistance to ICI therapy[75-77].
COMBINATION THERAPY FOR OVERCOMING DRUG RESISTANCE TO ICIS
Combination therapy has emerged as a key strategy to overcome resistance and improve outcomes with ICIs. Evidence demonstrates that multimodal therapeutic approaches enhance ICI efficacy through mechanisms such as counteracting immunosuppressive elements, promoting antigen release and presentation, activating and expanding effector T cells, and improving CD8+ T-cell infiltration and function [Table 2][39,78-99]. Recently, combination therapy has become a foundational approach in HCC, primarily including strategies such as ICI plus targeted therapy, dual-ICI, ICI combined with locoregional therapy, ICI integrated with traditional Chinese medicine (TCM), and ICI combined with intestinal microbiota modulation.
Combination strategies and their underlying mechanisms in promoting ICI efficacy used in clinical trials for HCC treatment
| ICI | Target | Combination (target) | Sensitization mechanism | Outcome | Trial numbers (current status) |
| Nivolumab | PD-1 | Ipilimumab (CTLA-4) | Boost the activation and proliferation of T cells | ORR: 24%, mOS: 16.4 months | NCT03222076[80] (Second-line treatment) |
| Relatlimab (LAG-3) + Bevacizumab (VEGF) | Inhibit LAG-3 checkpoint, enhance T-cell function; suppress angiogenesis, promote T-cell infiltration and efficacy | Underway | NCT05337137[81] | ||
| Lenvatinib (TKI) | Inhibit tumor angiogenesis and cancer cell proliferation, promote immune activity | ORR: 54.2%, PFS: 73.9 months | NCT04039607[82] | ||
| Cabozantinib (TKI) | Inhibit VEGF-driven angiogenesis and reduce immunosuppressive cells, promote immune cell infiltration | ORR: 19%, 1-year OSR: 12%-38.3% | NCT03299946[83] | ||
| Pembrolizumab | PD-1 | Lenvatinib (TKI) | Block tumor angiogenesis (blood vessel formation) and suppress tumor cell proliferation | mPFS: 9.3 months, mOS: 22 months | NCT03713593[84] (First-line treatment) |
| Lenvatinib (TKI) + TACE | Inhibit tumor angiogenesis and proliferation; induce localized tumor necrosis and release tumor antigens | mPFS: 14.6 months, 24-month OSR: 75% | NCT04246177[85] (First-line treatment) | ||
| Regorafenib (TKI) | Suppress tumor growth by blocking oncogenic signaling pathways and modulating the immunosuppressive TME | ORR: 29%, PFS: 6.8 months | NCT03347292[86] | ||
| Camrelizumab | PD-1 | Apatinib (VEGFR inhibitor) | Normalize tumor vasculature and suppress immunosuppressive cells | mPFS: 5.6 months, mOS: 22.1 months | NCT04297202[87] (First-line treatment) |
| Apatinib (VEGFR inhibitor) + TACE | Induce localized tumor ischemia and necrosis, release tumor antigens and promote inflammatory microenvironment; inhibit angiogenesis, improve immune cell infiltration | mPFS: 13.5 months, mOS: 22.1 months | NCT04559607[88] | ||
| Sintilimab | PD-1 | IBI305 (VEGF inhibitor) | Suppress blood vessel growth, modulate the immune-suppressive TME | mPFS: 4.6 months | NCT03794440[89] |
| IBI310 (CTLA-4 inhibitor) | Promote T-cell activation and proliferation | mPFS: 3.9 months, 6-month OSR: 93.1% | NCT04401813[90] | ||
| Bevacizumab (VEGF inhibitor) | Inhibit VEGF-driven angiogenesis, improve T-cell infiltration | ORR: 21%, PFS: 4.6 months | NCT04843943[91] (First-line treatment) | ||
| Penpulimab | PD-1 | Anlotinib (TKI) | Suppress tumor angiogenesis and proliferation | ORR: 24%, PFS: 5.4 months | NCT04344158[92] |
| Durvalumab | PD-L1 | Tremelimumab (CTLA-4 inhibitor) | Inhibit CTLA-4, promote T-cell activation and proliferation | ORR: 33%, PFS: 19.53 months | NCT03298451[93] (First-line treatment) |
| Bevacizumab (VEGF inhibitor) | Inhibit VEGF-driven angiogenesis and modulating the immunosuppressive TME | mPFS: 14.2 months | NCT02519348[94] | ||
| Bevacizumab (VEGF inhibitor) + TACE | Normalize tumor vasculature; induce localized tumor cell death, release neoantigens and promote immune cell infiltration | Underway | NCT03778957[95] | ||
| Atezolizumab | PD-L1 | Bevacizumab (VEGF inhibitor) | Inhibit VEGF to normalize tumor vasculature, reduce immunosuppression and promote T-cell infiltration | 12-month OSR: 67.2%, mPFS: 6.8 months | NCT03434379[96] (First-line treatment) |
| Bevacizumab (VEGF inhibitor) + TACE | Inhibit angiogenesis, reduce Treg activity and enhance DC maturation; induce localized tumor necrosis, release tumor antigens and promote immune cell infiltration | 13-month OSR: 75.4%, mPFS: 7.03 months | NCT05776875[97] | ||
| Cabozantinib (TKI) | Suppress tumor angiogenesis, proliferation and metastasis | Underway | NCT03755791[98] | ||
| Sorafenib (TKI) | Inhibit tumor proliferation and angiogenesis | 12-month OSR: 54.6%, mPFS: 4.3 months | NCT03434379[96] | ||
| Cobolimab | TIM-3 | Dostarlimab (PD-1) | Enhance T-cell reactivation and proliferation | Underway | NCT03680508[99] |
ICIS COMBINED WITH TARGETED THERAPY
Tyrosine kinase inhibitors
The combination of ICIs with anti-angiogenic agents represents one of the most extensively studied treatment regimens and has opened new avenues for the management of advanced HCC. Among anti-angiogenic drugs, tyrosine kinase inhibitors (TKIs) such as sorafenib, lenvatinib, regorafenib, and cabozantinib have been shown to enhance the response to PD-1 inhibitors by promoting CD8+ T-cell infiltration, suppressing M2 macrophage polarization, and reducing the infiltration of Th cells and Tregs[100-102]. Additionally, TKIs downregulate immunosuppressive cytokines including IL-10 and TGF-β, as well as immune checkpoints such as PD-1 and TIM-3, thereby helping to restore sensitivity to PD-1 blockade[103-105]. These mechanistic insights support the combined use of TKIs and ICIs as a novel therapeutic strategy in HCC. Recently, a growing number of clinical trials evaluating ICI-TKI combinations have been conducted in HCC patients, with several demonstrating superior antitumor activity compared to monotherapy. For example, the combination of cabozantinib with atezolizumab prolongs progression-free survival (PFS) of HCC patients, although no significant overall survival (OS) benefit is observed[98]. In addition, PD-1 inhibitors combined with TKIs such as regorafenib and lenvatinib yield higher objective response rates (ORR), longer PFS, and improved OS in patients with unresectable HCC[106]. Notably, the combination of pembrolizumab and lenvatinib has demonstrated significant efficacy, establishing it as a first-line treatment for advanced HCC[84].
VEGF or VEGFR inhibitors
Studies have shown that inhibitors targeting VEGF or vascular endothelial growth factor receptor (VEGFR) reduce the infiltration and activity of immunosuppressive cells. In addition, anti-VEGF/VEGFR therapy inhibits M2-type macrophage polarization and downregulates the expression of immune checkpoint (e.g., PD-L1/PD-L2) and immunosuppressive factors (e.g., IL-6, IDO1, and IL-10), while promoting DC maturation and antigen presentation[107,108]. These findings indicate that VEGF/VEGFR inhibitors can alleviate immunosuppression in the TME and possibly help to overcome resistance to ICIs. Consistently, clinical trials demonstrate that combining ICIs with anti-VEGF or anti-VEGFR agents leads to improved efficacy in HCC treatment. In particular, the combination of PD-L1 inhibitor atezolizumab with VEGF inhibitor bevacizumab has been approved as a first-line treatment for HCC[109]. Another clinical trial reveals that the VEGFR inhibitor rivoceranib (apatinib) combined with PD-1 inhibitor camrelizumab significantly improves both PFS and OS in patients with unresectable HCC[110]. Furthermore, a series of ongoing clinical trials are evaluating the efficacy of ICI-based combinations with other VEGFR inhibitors, such as anlotinib and cabozantinib, which may offer new combination strategies, particularly for patients with advanced HCC[41].
Other targeted drugs
In addition to anti-angiogenic agents, the combination of ICIs with other targeted drugs has also demonstrated improved efficacy in patients with HCC. For instance, the TGF-β receptor I inhibitor galunisertib significantly enhances the antitumor activity of PD-L1 inhibitors by suppressing TGF-β signaling[111]. Similarly, a peroxisome proliferator-activated receptor (PPAR) γ antagonist resensitizes tumors to anti-PD-L1 therapy in HCC models by inhibiting PPARγ/vascular endothelial growth factor A (VEGF-A)-mediated immunosuppression[112]. Furthermore, DNA damage repair (DDR) inhibitors are demonstrated to enhance the clinical efficacy of ICIs in HCC through mechanisms such as promoting neoantigen release, increasing TMB, and upregulating PD-L1 expression[113]. Currently, more than 80 clinical trials are investigating the therapeutic potential of combining DDR-targeted agents with ICIs, with several studies reporting favorable tolerability and significant antitumor activity[114,115]. Additionally, the combination of PD-1 blockade with the phosphatidylserine-targeting agent bavituximab has been shown to prolong the PFS and OS of HCC patients[116]. Preclinical studies also indicate that PEGylated IFN-α enhances the efficacy of PD-1 inhibitors by promoting the infiltration and activation of CD8+ T cells[117]. Epigenetic inhibitors targeting HDACs or DNMTs are reported to improve ICI response by modulating the infiltration of CD8+ T cells and IL-17-producing Th cells, as well as PD-1/PD-L1 expression, providing a rationale for combining epigenetic therapies with ICIs[118,119]. Recently, inhibitors of carbonic anhydrase XII, a novel class of anticancer agents, have shown the ability to remodel the immunosuppressive TME, suggesting another promising combination strategy for HCC treatment[120].
In conclusion, therapeutic approaches that combine targeted agents with ICIs to overcome ICI resistance are strongly supported by robust clinical trial evidence. In particular, the combination of ICIs with anti-angiogenic agents has gained approval in multiple countries as first‑line and second‑line treatments for HCC, demonstrating considerable promise for clinical application.
DUAL-ICIS COMBINATION THERAPY
The combination of PD-1/PD-L1 inhibitors and CTLA-4 inhibitors represents one of the most frequently evaluated strategies in clinical trials, as it enhances antitumor immunity through complementary mechanisms of T-cell activation. For example, the combination of nivolumab (PD-1 inhibitor) and ipilimumab (CTLA-4 inhibitor) significantly improves ORR and OS in patients with HCC[121]. This regimen received Food and Drug Administration (FDA) approval as a second-line treatment for advanced HCC previously treated with sorafenib. Similarly, clinical trials evaluating durvalumab (PD-L1 inhibitor) in combination with tremelimumab (CTLA-4 inhibitor) demonstrate promising therapeutic outcomes[122], and this combination was approved in 2022 for the first-line treatment of unresectable HCC. Recent findings from a phase III trial indicate that tremelimumab plus durvalumab significantly extends OS of HCC patients compared to monotherapy[123]. Additionally, the combination of sintilimab (PD-1 inhibitor) and IBI310 (CTLA-4 inhibitor) has shown encouraging efficacy in clinical trials. Based on these findings, this combination was approved in 2021 as a first-line treatment for unresectable or metastatic HCC[90].
In addition to CTLA-4 inhibitors, combinations of PD-1/PD-L1 inhibitors and other ICIs are also under investigation. For instance, clinical evidence suggests that PD-L1 inhibitors sometimes induce complete tumor eradication in HCC patients who have developed acquired resistance to PD-1 inhibitors[124]. The combination of the TIM-3 inhibitor cobolimab with dostarlimab has demonstrated acceptable safety and encouraging clinical activity as a first-line treatment for advanced HCC[125]. Furthermore, the LAG-3 inhibitor relatlimab and IDO1 inhibitor abrine significantly enhance antitumor efficacy when combined with anti-PD-1 therapy[8]. Recently, several clinical trials evaluating inhibitors targeting TIM-3, LAG-3, or TIGIT in combination with other ICIs for HCC treatment are currently underway[126,127]. Positive results from preclinical HCC models may offer promising new dual-ICI therapeutic options for HCC.
As one of the well-established combination strategies, dual-ICI therapy is backed by substantial clinical trial evidence, laying a solid foundation for the clinical application of PD-1/PD-L1 inhibitors combination with CTLA-4 inhibitors in HCC treatment. In the future, with the discovery of novel ICIs and corresponding inhibitors, an increasing number of ICI-based combinations are expected to enter clinical practice for HCC management.
ICIS COMBINED WITH LOCOREGIONAL THERAPY
Locoregional therapies constitute a group of liver-directed interventions that augment the immunogenicity of residual tumor cells and potentiate their susceptibility to immunotherapy. Accumulating evidence indicates that locoregional modalities enhance responsiveness to ICIs through mechanisms including the upregulation of tumor-associated antigens (TAAs) and enhanced immunogenicity, promotion of DC and CD8+ T-cell infiltration, and release of pro-inflammatory cytokines (e.g., TNF-α, IL-12)[128]. In addition, locoregional therapies mitigate the activities of immunosuppressive cells and elicit immunogenic cell death (ICD)[129]. Together, these mechanisms underpin the rational combination of locoregional therapy with ICIs as a promising strategy to amplify antitumor immunity[130].
In recent years, multiple studies have evaluated the efficacy and safety of combining ICIs with transarterial chemoembolization (TACE) for the treatment of HCC. A phase III trial demonstrates that combining lenvatinib, pembrolizumab, and TACE significantly prolongs PFS of patients with unresectable HCC, with some achieving complete tumor regression[131]. Another study shows that adding anti-PD-L1 and targeted drugs to TACE improves both OS and ORR of HCC patients[132]. A clinical trial conducted by Roche showed that atezolizumab plus bevacizumab combined with TACE led to significant benefits in unresectable HCC, highlighting the synergy between ICIs, anti-angiogenic therapy, and locoregional treatment[97]. Studies on hepatic arterial infusion chemotherapy (HAIC) combined with ICIs have also shown promise in improving the antitumor efficacy. A retrospective analysis finds that HAIC combined with ICIs and TKIs offers a favorable safety profile and better liver function preservation in intermediate and advanced HCC[133]. Furthermore, an ongoing trial is investigating concurrent hepatic arterial delivery of a PD-L1 antibody with FOLFOX chemotherapy to enhance local immune blockade and suppress tumor immune evasion[134]. Radiotherapy eliminates tumor cells within the irradiated field through high-energy radiation and elicits systemic immune effects, making it a promising partner for immunotherapy[135]. Supported by positive outcomes from phase I/II trials, the combination of radiotherapy [e.g., Yttrium-90 (Y90)] with ICIs is being increasingly explored in advanced HCC[136]. Similarly, thermal ablation techniques including microwave ablation (MWA) and radiofrequency ablation (RFA) can also mitigate local immunosuppression and synergize effectively with ICIs[130].
In summary, locoregional therapies represented by TACE combined with ICIs have accumulated abundant clinical trial evidence in the management of HCC. Approved for clinical application in HCC treatment, this combination regimen has emerged as an effective therapeutic strategy to overcome ICI resistance.
ICIS COMBINED WITH TCM
The combination of PD-1/PD-L1 inhibitors with TCM has shown superior efficacy compared to ICI monotherapy in the treatment of HCC. For instance, ginseng-derived nanoparticles (GDNPs) and Huaier enhance the antitumor effect of PD-1/PD-L1 inhibitors by promoting M1 macrophage polarization and recruiting CD8+ T cells into the TME[137,138]. Similarly, natural compounds such as bufalin and curcumin (isolated from turmeric) potentiate the activity of anti-PD-L1 antibodies by shifting TAMs from M2 to M1 phenotype and downregulating PD-1 expression on CD8+ T cells[139,140]. Other TCM-derived agents, including icaritin, luteolin, baicalein and baicalin, and osthole have been shown to induce antitumor immune responses through regulating immune cell function[141-143]. Additionally, YIV-906, a standardized formulation derived from Huang Qin Tang, reduces M2 polarization and enhances antitumor activity when combined with PD-1 inhibition[144]. Besides, a clinical trial is currently underway to evaluate the efficacy of Huaier granules in combination with atezolizumab (anti-PD-L1) and bevacizumab in patients with HCC[145].
Furthermore, the emergence of new immune checkpoints opens new avenues for combining TCM with ICIs in the treatment of HCC. For example, network pharmacological analyses suggest that the herbal medicine Yinchen may activate immune cells by suppressing CTLA-4 and LAG-3[146]. Abrine, a compound derived from Abrus cantoniensis, is identified as a specific inhibitor of IDO1. Studies also demonstrate that abrine synergizes with PD-1 inhibitors to suppress HCC progression by downregulating IDO1 and PD-L1 expression while enhancing CD8+ T-cell infiltration within tumors[8]. Moreover, the YANGYIN Fuzheng Jiedu prescription has been shown to inhibit tumor growth in HCC models by alleviating exhausted T cells and reducing the expression of immune checkpoints, including PD-1, TIM-3, and TIGIT[147,148]. These mechanisms highlight the potential of TCM agents as promising candidates for combination strategies with ICIs in HCC therapy.
Although TCM has exhibited considerable application potential in enhancing the efficacy of ICI therapy, most relevant studies are still in the preclinical exploration stage. In the future, more mechanistic research and clinical trials are needed to clarify its prospects for clinical application in HCC treatment.
ICIS COMBINED WITH INTESTINAL MICROBIOTA
Increasing evidence indicates a strong correlation between the composition of the gut microbiome and clinical outcomes following ICI treatment in patients with HCC. For instance, a higher abundance of Lachnospiraceae bacterium-GAM79, Alistipes sp. Marseille-P5997, Bifidobacterium, Coprococcus, Akkermansia, and Acidaminococcus is associated with improved responses to ICIs in HCC[149-152]. Studies indicate that specific intestinal bacteria, including Bifidobacterium and Bacteroides fragilis, restore response to ICIs by promoting the activation of DCs, CD8+ T cells, and Th1 cells[153,154]. Additionally, Akkermansia muciniphila, whose abundance decreases during metabolic dysfunction-associated fatty liver disease (MAFLD)-promoted hepatocarcinogenesis, has been shown to exert maximal tumor-suppressive effects in HCC models when combined with PD-1 inhibition[149]. Moreover, metabolites such as ursodeoxycholic acid and ursocholic acid, which correlate with the abundance of Lachnoclostridium, are significantly enriched in fecal samples from HCC patients who respond to ICI therapy[155]. These findings underscore the potential of combining ICIs with microbiota-targeted interventions to enhance the efficacy of immunotherapy in HCC.
Probiotic supplementation, fecal microbiota transplantation (FMT), and antibiotic modulation have emerged as promising strategies to enhance the efficacy of ICIs in HCC. For instance, probiotic strains such as Lactobacillus rhamnosus Probio-M9, Lachnospiraceae bacterium-GAM79, Ruminococcus, and Bifidobacterium bifidum are associated with improved ICI efficacy[153,156,157]. Moreover, several studies showed that FMT enhances antitumor efficacy of ICIs, and combining FMT with atezolizumab or bevacizumab prolonged both PFS and OS of HCC patients[158,159]. The use of antibiotics in combination with ICIs represents another emerging approach, and vancomycin has been shown to improve the efficacy of nivolumab in patients with refractory primary or metastatic HCC[160]. However, the impact of antibiotics on ICI therapy remains controversial and warrants further investigation.
In summary, combining ICIs with gut microbiota modulation has demonstrated promising efficacy in overcoming ICI resistance in HCC. Nevertheless, its clinical translation is hampered by insufficient understanding of the underlying mechanisms and a lack of robust clinical evidence. Further well‑designed trials are essential to advance this therapeutic strategy toward clinical application.
CONCLUSION AND FUTURE PERSPECTIVES
ICIs have ushered in a paradigm shift in the treatment of HCC, offering the potential for durable antitumor responses. However, the emergence of drug resistance remains a formidable challenge, undermining clinical outcomes and restricting the broader application of ICIs in HCC. In recent years, combination therapies have emerged as the most promising and widely adopted approaches to counteract ICI resistance. ICIs plus targeted therapy and dual-ICI blockade currently represent the most well-established and clinically validated approaches, with robust evidence from clinical trials supporting their approval as first- or second-line treatments. Moreover, the combination of ICIs with locoregional therapies has gained relatively broad application in HCC management. Notably, the combination of TACE with ICIs has demonstrated significant efficacy in delaying HCC progression in clinical trials, and the regimen of TACE plus lenvatinib and pembrolizumab was approved in China in 2025 for the treatment of unresectable non-metastatic HCC. In contrast, combinations of ICIs with TCM or microbiota modulators currently rely mainly on exploratory evidence and have not yet been substantiated for routine clinical use.
However, these combination strategies still face distinct challenges in clinical practice. For ICI-based targeted therapy combinations, key issues include the need for high-quality clinical data to support diverse regimens, lengthy trial and approval cycles, increased risks of combined drug toxicity, and the imperative for precise patient stratification. Safety profiles for dual-ICI blockade regimens are still being accumulated, and the absence of unified guidelines for managing immune-related adverse events (irAEs) complicates monitoring. Furthermore, the lack of reliable predictive biomarkers hinders the accurate identification of patients most likely to benefit. The diversity of locoregional treatment modalities further complicates the standardization of combination regimens with ICIs and the accurate assessment of therapeutic outcomes. For example, there is no consensus on the sequence, interval, or dose adjustment between locoregional and systemic therapies, which affects the consistency of treatment efficacy. The complex composition of TCM preparations poses significant challenges for quality control, making it difficult to establish unified clinical trial protocols and evaluation systems. As for microbiota-based combination therapies, major limitations currently include the lack of HCC-specific guidelines for probiotic strain selection, dosage, and treatment duration, as well as the absence of a dedicated regulatory pathway for FMT combined with ICIs.
Moreover, incompletely elucidated mechanisms of ICI resistance are another important reason for the limited therapeutic efficacy and clinical application of combination strategies. For example, while ICIs and targeted drugs have well-defined targets such as PD-1/PD-L1 and VEGF, the mechanisms underlying the production of these drug resistance-related factors remain incompletely understood. Consequently, combination therapy cannot completely overcome ICI resistance. According to the two categories of factors inducing ICI resistance displayed in this review, tumor cell intrinsic factors appear to play a dominant role. In addition to altering their own functions, tumor cells can secrete factors including cytokines and metabolites that influence TME, thereby coordinating multiple processes to evade immune surveillance. Therefore, elucidating the molecular mechanisms in tumor cells that drive ICI resistance is critical to the optimization of HCC immunotherapy. Besides, considering the importance of heterogeneity in influencing the functions of tumor cells, systematic evaluation of intra- and inter-patient heterogeneity may clarify the specific causes of ICI resistance. Moreover, intestinal microbiota has emerged as a key regulator of antitumor immunity[161]. Whether tumor cells escape immunotherapy through the intestinal microbiota requires further investigation, which will expand our understanding of ICI resistance mechanisms.
Given the complex and variable mechanisms that induce ICI resistance, staged combination therapy represents a promising direction for HCC. Beyond currently established regimens, innovative approaches such as cellular immunotherapies [e.g., chimeric antigen receptor T (CAR-T) cells, chimeric antigen receptor (CAR)-NK, and CAR-macrophage (CAR-M)][162-164], inhibitors targeting immunosuppressive metabolites (e.g., adenosine and lactate)[165,166], and oncolytic viruses [e.g., talimogene laherparepvec and pexastimogene devacirepvec (JX-594)] show promise in augmenting ICI efficacy[167,168]. Additionally, tumor vaccines represent a promising modality for enhancing tumor-specific immune responses through targeted delivery of tumor antigens, offering a new avenue to combat ICI resistance[169]. For instance, DC vaccines and peptide vaccines targeting alpha-fetoprotein (AFP) or glypican-3 (GPC-3) have demonstrated acceptable safety profiles in HCC patients[170]. Furthermore, precision or personalized immunotherapy, in which regimens are tailored to individual tumor biology and host characteristics, offers a compelling avenue to maximize therapeutic impact.
Predictive biomarkers are indispensable for guiding ICI therapy, enabling identification of likely responders, monitoring therapeutic efficacy, assessing recurrence risk, and anticipating irAEs, thereby advancing precision immunotherapy for HCC. Currently, biomarkers under investigation encompass host-related factors (e.g., AFP, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, gut microbiota), and tumor-derived markers [e.g., PD-L1 expression, TMB, microsatellite instability, circulating tumor DNA (ctDNA), tumor-infiltrating lymphocytes, and driver gene mutations [e.g., tumor protein p53 (TP53)][171-173]. Although most remain in the investigational phase and are not yet embedded in routine clinical workflows, these markers have considerably deepened our understanding of ICI responsiveness and hold substantial potential for future integration into clinical practice.
DECLARATIONS
Acknowledgment
The Graphical Abstract was created with BioRender. H, T. (2025) https://BioRender.com/96dg8hw.
Authors’ contributions
Conception, design, and editing of the manuscript: Zhao Q
Collection and interpretation of data, writing the initial draft: Zhang L, He T
Drawing figures: He T
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool DeepSeek (web version) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Kim E, Viatour P. Hepatocellular carcinoma: old friends and new tricks. Exp Mol Med. 2020;52:1898-907.
2. Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63.
3. Brown ZJ, Tsilimigras DI, Ruff SM, et al. Management of hepatocellular carcinoma: a review. JAMA Surg. 2023;158:410-20.
4. Ju MR, Yopp AC. Surgical resection of early stage hepatocellular carcinoma: balancing tumor biology with the host liver. Chin Clin Oncol. 2021;10:5.
5. Feng S, Roll GR, Rouhani FJ, Sanchez Fueyo A. The future of liver transplantation. Hepatology. 2024;80:674-97.
6. Yang GY, He ZW, Tang YC, et al. Unraveling the efficacy network: a network meta-analysis of adjuvant external beam radiation therapy methods after hepatectomy. World J Gastrointest Surg. 2024;16:205-14.
7. Wang Y, Deng B. Hepatocellular carcinoma: molecular mechanism, targeted therapy, and biomarkers. Cancer Metastasis Rev. 2023;42:629-52.
8. Liang X, Gao H, Xiao J, et al. Abrine, an IDO1 inhibitor, suppresses the immune escape and enhances the immunotherapy of anti-PD-1 antibody in hepatocellular carcinoma. Front Immunol. 2023;14:1185985.
9. Cao D, Chen MK, Zhang QF, et al. Identification of immunological subtypes of hepatocellular carcinoma with expression profiling of immune-modulating genes. Aging. 2020;12:12187-205.
10. Zhao Q, Huang ZL, He M, Gao Z, Kuang DM. BTLA identifies dysfunctional PD-1-expressing CD4+ T cells in human hepatocellular carcinoma. Oncoimmunology. 2016;5:e1254855.
11. Zhang M, Pang HJ, Zhao W, et al. VISTA expression associated with CD8 confers a favorable immune microenvironment and better overall survival in hepatocellular carcinoma. BMC Cancer. 2018;18:511.
12. Chiu DK, Yuen VW, Cheu JW, et al. Hepatocellular carcinoma cells up-regulate PVRL1, stabilizing PVR and inhibiting the cytotoxic T-cell response via TIGIT to mediate tumor resistance to PD1 inhibitors in mice. Gastroenterology. 2020;159:609-23.
13. Cheung CCL, Seah YHJ, Fang J, et al. Immunohistochemical scoring of LAG-3 in conjunction with CD8 in the tumor microenvironment predicts response to immunotherapy in hepatocellular carcinoma. Front Immunol. 2023;14:1150985.
14. Tang Z, Wang W, Gao B, et al. Unveiling Tim-3 immune checkpoint expression in hepatocellular carcinoma through abdominal contrast-enhanced CT habitat radiomics. Front Oncol. 2024;14:1456748.
15. Pinter M, Jain RK, Duda DG. The current landscape of immune checkpoint blockade in hepatocellular carcinoma: a review. JAMA Oncol. 2021;7:113-23.
16. Yu W, He J, Wang F, et al. NR4A1 mediates NK-cell dysfunction in hepatocellular carcinoma via the IFN-γ/p-STAT1/IRF1 pathway. Immunology. 2023;169:69-82.
17. Yuen VW, Chiu DK, Law CT, et al. Using mouse liver cancer models based on somatic genome editing to predict immune checkpoint inhibitor responses. J Hepatol. 2023;78:376-89.
18. Bonaventura P, Shekarian T, Alcazer V, et al. Cold tumors: a therapeutic challenge for immunotherapy. Front Immunol. 2019;10:168.
19. Lurje I, Hammerich L, Tacke F. Dendritic cell and t cell crosstalk in liver fibrogenesis and hepatocarcinogenesis: implications for prevention and therapy of liver cancer. Int J Mol Sci. 2020;21:7378.
20. Han Y, Chen Z, Yang Y, et al. Human CD14+ CTLA-4+ regulatory dendritic cells suppress T-cell response by cytotoxic T-lymphocyte antigen-4-dependent IL-10 and indoleamine-2,3-dioxygenase production in hepatocellular carcinoma. Hepatology. 2014;59:567-79.
21. Xiao R, Tian Y, Zhang J, et al. Increased Siglec-9/Siglec-9L interactions on NK cells predict poor HCC prognosis and present a targetable checkpoint for immunotherapy. J Hepatol. 2024;80:792-804.
22. Wei H, Suo C, Gu X, et al. AKR1D1 suppresses liver cancer progression by promoting bile acid metabolism-mediated NK cell cytotoxicity. Cell Metab. 2025;37:1103-18.e7.
23. Prawira A, Xu H, Mei Y, et al. Targeting Treg-fibroblast interaction to enhance immunotherapy in steatotic liver disease-related hepatocellular carcinoma. Gut. 2025;75:105-18.
24. Suthen S, Lim CJ, Nguyen PHD, et al. Hypoxia-driven immunosuppression by Treg and type-2 conventional dendritic cells in HCC. Hepatology. 2022;76:1329-44.
25. Cheng K, Cai N, Yang X, et al. Short-term starvation boosts anti-PD-L1 therapy by reshaping tumor-associated macrophages in hepatocellular carcinoma. Hepatology. 2025;82:1414-31.
26. Tang B, Zhu J, Wang Y, et al. Targeted xCT-mediated ferroptosis and protumoral polarization of macrophages is effective against HCC and enhances the efficacy of the anti-PD-1/L1 response. Adv Sci. 2023;10:e2203973.
27. Zhang Y, Rao Y, Lu J, et al. The influence of biophysical niche on tumor-associated macrophages in liver cancer. Hepatol Commun. 2024;8:e0569.
28. Ning J, Ye Y, Shen H, et al. Macrophage-coated tumor cluster aggravates hepatoma invasion and immunotherapy resistance via generating local immune deprivation. Cell Rep Med. 2024;5:101505.
29. Xie P, Yu M, Zhang B, et al. CRKL dictates anti-PD-1 resistance by mediating tumor-associated neutrophil infiltration in hepatocellular carcinoma. J Hepatol. 2024;81:93-107.
30. Sangro B, Sarobe P, Hervás-Stubbs S, Melero I. Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol. 2021;18:525-43.
32. Kato T, Noma K, Ohara T, et al. Cancer-associated fibroblasts affect intratumoral CD8+ and FoxP3+ T cells via IL6 in the tumor microenvironment. Clin Cancer Res. 2018;24:4820-33.
33. Yang X, Lin Y, Shi Y, et al. FAP promotes immunosuppression by cancer-associated fibroblasts in the tumor microenvironment via STAT3-CCL2 signaling. Cancer Res. 2016;76:4124-35.
34. Timperi E, Romano E. Stromal circuits involving tumor-associated macrophages and cancer-associated fibroblasts. Front Immunol. 2023;14:1194642.
35. Han S, Bao X, Zou Y, et al. d-lactate modulates M2 tumor-associated macrophages and remodels immunosuppressive tumor microenvironment for hepatocellular carcinoma. Sci Adv. 2023;9:eadg2697.
36. Shen XT, Xie SZ, Zheng X, et al. Cirrhotic-extracellular matrix attenuates aPD-1 treatment response by initiating immunosuppressive neutrophil extracellular traps formation in hepatocellular carcinoma. Exp Hematol Oncol. 2024;13:20.
37. Zeng W, Liu F, Liu Y, et al. Targeting TM4SF1 promotes tumor senescence enhancing CD8+ T cell cytotoxic function in hepatocellular carcinoma. Clin Mol Hepatol. 2025;31:489-508.
38. Wu B, Wang Q, Li B, Jiang M. LAMTOR1 degrades MHC-II via the endocytic in hepatocellular carcinoma. Carcinogenesis. 2022;43:1059-70.
39. Wang Z, Wang Y, Gao P, Ding J. Immune checkpoint inhibitor resistance in hepatocellular carcinoma. Cancer Lett. 2023;555:216038.
40. Chan TA, Yarchoan M, Jaffee E, et al. Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic. Ann Oncol. 2019;30:44-56.
41. Xie Q, Zhang P, Wang Y, Mei W, Zeng C. Overcoming resistance to immune checkpoint inhibitors in hepatocellular carcinoma: Challenges and opportunities. Front Oncol. 2022;12:958720.
42. Zhou Z, Yu X, Chen Y, et al. Inhibition of the B7-H3 immune checkpoint limits hepatocellular carcinoma progression by enhancing T lymphocyte-mediated immune cytotoxicity in vitro and in vivo. Clin Transl Oncol. 2023;25:1067-79.
43. Shrestha R, Bridle KR, Crawford DHG, Jayachandran A. Immune checkpoint molecules are regulated by transforming growth factor (TGF)-β1-induced epithelial-to-mesenchymal transition in hepatocellular carcinoma. Int J Med Sci. 2021;18:2466-79.
44. David CJ, Massagué J. Contextual determinants of TGFβ action in development, immunity and cancer. Nat Rev Mol Cell Biol. 2018;19:419-35.
45. Park BV, Freeman ZT, Ghasemzadeh A, et al. TGFβ1-mediated SMAD3 enhances PD-1 expression on antigen-specific T cells in cancer. Cancer Discov. 2016;6:1366-81.
46. Chen J, Gingold JA, Su X. Immunomodulatory TGF-β signaling in hepatocellular carcinoma. Trends Mol Med. 2019;25:1010-23.
47. de Gramont A, Faivre S, Raymond E. Novel TGF-β inhibitors ready for prime time in onco-immunology. Oncoimmunology. 2017;6:e1257453.
48. Rebouissou S, Franconi A, Calderaro J, et al. Genotype-phenotype correlation of CTNNB1 mutations reveals different ß-catenin activity associated with liver tumor progression. Hepatology. 2016;64:2047-61.
49. Ruiz de Galarreta M, Bresnahan E, Molina-Sánchez P, et al. β-Catenin activation promotes immune escape and resistance to anti-PD-1 therapy in hepatocellular carcinoma. Cancer Discov. 2019;9:1124-41.
50. Lehrich BM, Delgado ER, Yasaka TM, et al. Precision targeting of β-catenin induces tumor reprogramming and immunity in hepatocellular cancers. Nat Commun. 2025;16:5009.
51. Liu Z, Ning F, Cai Y, et al. The EGFR-P38 MAPK axis up-regulates PD-L1 through miR-675-5p and down-regulates HLA-ABC via hexokinase-2 in hepatocellular carcinoma cells. Cancer Commun. 2021;41:62-78.
52. Xing S, Chen S, Yang X, Huang W. Role of MAPK activity in PD-L1 expression in hepatocellular carcinoma cells. J buon. 2020;25:1875-82.
53. Chen Y, Liu YC, Sung YC, et al. Overcoming sorafenib evasion in hepatocellular carcinoma using CXCR4-targeted nanoparticles to co-deliver MEK-inhibitors. Sci Rep. 2017;7:44123.
54. Lin Z, Huang L, Li SL, Gu J, Cui X, Zhou Y. PTEN loss correlates with T cell exclusion across human cancers. BMC Cancer. 2021;21:429.
55. Vidotto T, Melo CM, Castelli E, Koti M, Dos Reis RB, Squire JA. Emerging role of PTEN loss in evasion of the immune response to tumours. Br J Cancer. 2020;122:1732-43.
56. Zhang S, Gan X, Qiu J, et al. IL-10 derived from Hepatocarcinoma cells improves human induced regulatory T cells function via JAK1/STAT5 pathway in tumor microenvironment. Mol Immunol. 2021;133:163-72.
57. Fernandez-Barrena MG, Uriarte I, Sarobe P, Avila MA. Epigenetic mechanisms in HCC immune landscape: therapeutic implications. Semin Immunol. 2025;79:101980.
58. Tong Y, Chen Z, Wu J, et al. METTL3 promotes an immunosuppressive microenvironment in bladder cancer via m6A-dependent CXCL5/CCL5 regulation. J Immunother Cancer. 2025;13:e011108.
59. Wang DX, Long JY, Li RZ, et al. Mutation status of the KMT2 family associated with immune checkpoint inhibitors (ICIs) therapy and implicating diverse tumor microenvironments. Mol Cancer. 2024;23:15.
60. Jiang Y, Yu Z, Wang J, et al. Targeting USP47 enhances immunotherapy in hepatocellular carcinoma by destabilizing PD-L1. Int Immunopharmacol. 2025;161:115024.
61. Gao Y, Nihira NT, Bu X, et al. Acetylation-dependent regulation of PD-L1 nuclear translocation dictates the efficacy of anti-PD-1 immunotherapy. Nat Cell Biol. 2020;22:1064-75.
62. Lin M, He J, Zhang X, et al. Targeting fibrinogen-like protein 1 enhances immunotherapy in hepatocellular carcinoma. J Clin Invest. 2023;133:e164528.
63. Tu Y, Wu H, Zhong C, et al. Pharmacological activation of STAT1-GSDME pyroptotic circuitry reinforces epigenetic immunotherapy for hepatocellular carcinoma. Gut. 2025;74:613-27.
64. Zeng C, Ye S, Chen Y, et al. HOXA-AS3 promotes proliferation and migration of hepatocellular carcinoma cells via the miR-455-5p/PD-L1 axis. J Immunol Res. 2021;2021:9289719.
65. Zhang X, Pan B, Qiu J, et al. lncRNA MIAT targets miR-411-5p/STAT3/PD-L1 axis mediating hepatocellular carcinoma immune response. Int J Exp Pathol. 2022;103:102-11.
66. Peng X, He Z, Yuan D, Liu Z, Rong P. Lactic acid: the culprit behind the immunosuppressive microenvironment in hepatocellular carcinoma. Biochim Biophys Acta Rev Cancer. 2024;1879:189164.
67. Fu J, Zhang Z, Zhou L, et al. Impairment of CD4+ cytotoxic T cells predicts poor survival and high recurrence rates in patients with hepatocellular carcinoma. Hepatology. 2013;58:139-49.
68. Tang W, Zhou J, Yang W, et al. Aberrant cholesterol metabolic signaling impairs antitumor immunosurveillance through natural killer T cell dysfunction in obese liver. Cell Mol Immunol. 2022;19:834-47.
69. Qin WH, Yang ZS, Li M, et al. High serum levels of cholesterol increase antitumor functions of nature killer cells and reduce growth of liver tumors in mice. Gastroenterology. 2020;158:1713-27.
70. Tu X, Lin W, Zhai X, et al. Oleanolic acid inhibits M2 macrophage polarization and potentiates anti-PD-1 therapy in hepatocellular carcinoma by targeting miR-130b-3p-PTEN-PI3K-Akt signaling and glycolysis. Phytomedicine. 2025;141:156750.
71. Bai J, Tang R, Zhou K, et al. An asparagine metabolism-based classification reveals the metabolic and immune heterogeneity of hepatocellular carcinoma. BMC Med Genomics. 2022;15:222.
72. Chen J, Wang R, Liu Z, et al. Unbalanced glutamine partitioning between CD8T cells and cancer cells accompanied by immune cell dysfunction in hepatocellular carcinoma. Cells. 2022;11:3924.
73. Sun C, Wang B, Hao S. Adenosine-A2A receptor pathway in cancer immunotherapy. Front Immunol. 2022;13:837230.
74. Hu Z, Chen G, Zhao Y, et al. Exosome-derived circCCAR1 promotes CD8 + T-cell dysfunction and anti-PD1 resistance in hepatocellular carcinoma. Mol Cancer. 2023;22:55.
75. Zhang PF, Gao C, Huang XY, et al. Cancer cell-derived exosomal circUHRF1 induces natural killer cell exhaustion and may cause resistance to anti-PD1 therapy in hepatocellular carcinoma. Mol Cancer. 2020;19:110.
76. Yang Y, Wu T, Wang Y, et al. Hypoxic tumour-derived exosomal miR-1290 exacerbates the suppression of CD8+ T cells by promoting M2 macrophage polarization. Immunology. 2024;173:672-88.
77. Chen J, Lin Z, Liu L, et al. GOLM1 exacerbates CD8+ T cell suppression in hepatocellular carcinoma by promoting exosomal PD-L1 transport into tumor-associated macrophages. Signal Transduct Target Ther. 2021;6:397.
78. Kou L, Xie X, Chen X, Li B, Li J, Li Y. The progress of research on immune checkpoint inhibitor resistance and reversal strategies for hepatocellular carcinoma. Cancer Immunol Immunother. 2023;72:3953-69.
79. Ruff SM, Manne A, Cloyd JM, Dillhoff M, Ejaz A, Pawlik TM. Current landscape of immune checkpoint inhibitor therapy for hepatocellular carcinoma. Curr Oncol. 2023;30:5863-75.
80. Kaseb A, Vence L, Blando J, et al. PD-009 - Randomized, open-label, perioperative phase II study evaluating nivolumab alone versus nivolumab plus ipilimumab in patients with resectable HCC. Ann Oncol. 2019;30:iv112. Available from: https://www.annalsofoncology.org/article/S0923-7534(19)30920-2/fulltext. [Last accessed on 14 May 2026].
81. Sangro B, Yau T, Harding JJ, et al. RELATIVITY-106: a phase 1/2 trial of nivolumab (NIVO) + relatlimab (RELA) in combination with bevacizumab (BEV) in first-line (1L) hepatocellular carcinoma (HCC). JCO. 2023;41:TPS636.
82. Yau T, Galle PR, Decaens T, et al.; CheckMate 9DW investigators. Nivolumab plus ipilimumab versus lenvatinib or sorafenib as first-line treatment for unresectable hepatocellular carcinoma (CheckMate 9DW): an open-label, randomised, phase 3 trial. Lancet. 2025;405:1851-64.
83. Yarchoan M, Zhu Q, Durham JN, et al. Feasibility and efficacy of neoadjuvant cabozantinib and nivolumab in patients with borderline resectable or locally advanced hepatocellular carcinoma (HCC). JCO. 2021;39:335.
84. Llovet JM, Kudo M, Cheng A, et al. Lenvatinib (len) plus pembrolizumab (pembro) for the first-line treatment of patients (pts) with advanced hepatocellular carcinoma (HCC): phase 3 LEAP-002 study. JCO. 2019;37:TPS4152.
85. El-khoueiry AB, Llovet JM, Vogel A, et al. LEAP-012 trial in progress: transarterial chemoembolization (TACE) with or without lenvatinib plus pembrolizumab for intermediate-stage hepatocellular carcinoma (HCC). JCO. 2022;40:TPS494.
86. Galle P, Kim R, Sung M, et al. 990P Updated results of a phase Ib study of regorafenib (REG) plus pembrolizumab (PEMBRO) for first-line treatment of advanced hepatocellular carcinoma (HCC). Ann Oncol. 2020;31:S691-2.
87. Xia Y, Wang P, Pu L, et al. Preliminary efficacy and safety of perioperative treatment of camrelizumab combined with apatinib in resectable hepatocellular carcinoma (HCC): a prospective phase II study. JCO. 2021;39:4082.
88. Zhu H, Teng G, Fan W, et al. Transarterial chemoembolization (TACE) combined with camrelizumab and apatinib versus TACE alone in the treatment of unresectable hepatocellular carcinoma eligible for embolization: a multicenter, open-label, randomized, phase 2 study (CAP-ACE). JCO. 2025;43:LBA522.
89. Ren Z, Xu J, Bai Y, et al.; ORIENT-32 study group. Sintilimab plus a bevacizumab biosimilar (IBI305) versus sorafenib in unresectable hepatocellular carcinoma (ORIENT-32): a randomised, open-label, phase 2-3 study. Lancet Oncol. 2021;22:977-90.
90. Zhou J, Shi Y, Liu B, et al. A phase Ib, multicenter, open-label study to assess the safety, tolerability, and preliminary efficacy of sintilimab plus IBI310 (anti-CTLA4 mAb) in patients with advanced hepatocellular carcinoma. JCO. 2022;40:421.
91. Sun HC, Zhu XD, Wang ZY, et al. Sintilimab plus bevacizumab followed by resection in intermediate-stage hepatocellular carcinoma: a phase Ib clinical trial with biomarker analysis. BMJ Oncol. 2024;3:e000578.
92. Zhou J, Bai L, Luo J, et al.; APOLLO Study Group. Anlotinib plus penpulimab versus sorafenib in the first-line treatment of unresectable hepatocellular carcinoma (APOLLO): a randomised, controlled, phase 3 trial. Lancet Oncol. 2025;26:719-31.
93. Sangro B, Chan SL, Kelley RK, et al.; HIMALAYA investigators. Four-year overall survival update from the phase III HIMALAYA study of tremelimumab plus durvalumab in unresectable hepatocellular carcinoma. Ann Oncol. 2024;35:448-57.
94. Lim HY, Heo J, Kim T, et al. Safety and efficacy of durvalumab plus bevacizumab in unresectable hepatocellular carcinoma: results from the phase 2 study 22 (NCT02519348). JCO. 2022;40:436-436.
95. Heo J, Okusaka T, Yoon J, et al. Outcomes by transarterial chemoembolization (TACE) modality from participants (pts) with embolization-eligible hepatocellular carcinoma (HCC) treated with durvalumab (D) + bevacizumab (B) + TACE and placebos (PBO) + TACE: EMERALD-1 subgroup analysis. JCO. 2025;43:575.
96. Finn RS, Qin S, Ikeda M, et al.; IMbrave150 Investigators. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382:1894-905.
97. Stein S, Cheng W, Wiess C, et al. Atezolizumab and bevacizumab in combination with TACE for patients with BCLC B HCC. JCO. 2024;42:TPS584.
98. Rimassa L, Cheng A, Braiteh F, et al. Phase 3 (COSMIC-312) study of cabozantinib in combination with atezolizumab vs sorafenib in patients with advanced hepatocellular carcinoma (aHCC) who have not received previous systemic anticancer therapy. Ann Oncol. 2019;30:iv65-6.
99. Acoba JD, Fukaya E, Goodyear SM, Kardosh A. Cobolimab and dostarlimab in the first-line treatment of unresectable hepatoma: a multi-center, single arm, phase 2 trial. JCO. 2025;43:4099-4099.
100. Kato Y, Tabata K, Kimura T, et al. Lenvatinib plus anti-PD-1 antibody combination treatment activates CD8+ T cells through reduction of tumor-associated macrophage and activation of the interferon pathway. PLoS One. 2019;14:e0212513.
101. Kimura T, Kato Y, Ozawa Y, et al. Immunomodulatory activity of lenvatinib contributes to antitumor activity in the Hepa1-6 hepatocellular carcinoma model. Cancer Sci. 2018;109:3993-4002.
102. Abou-Elkacem L, Arns S, Brix G, et al. Regorafenib inhibits growth, angiogenesis, and metastasis in a highly aggressive, orthotopic colon cancer model. Mol Cancer Ther. 2013;12:1322-31.
103. Kwilas AR, Ardiani A, Donahue RN, Aftab DT, Hodge JW. Dual effects of a targeted small-molecule inhibitor (cabozantinib) on immune-mediated killing of tumor cells and immune tumor microenvironment permissiveness when combined with a cancer vaccine. J Transl Med. 2014;12:294.
104. Zhu XD, Huang C, Shen YH, et al. Downstaging and resection of initially unresectable hepatocellular carcinoma with tyrosine kinase inhibitor and anti-PD-1 antibody combinations. Liver Cancer. 2021;10:320-9.
106. Xu Y, Fu S, Shang K, Zeng J, Mao Y. PD-1 inhibitors plus lenvatinib versus PD-1 inhibitors plus regorafenib in patients with advanced hepatocellular carcinoma after failure of sorafenib. Front Oncol. 2022;12:958869.
107. Liu JKH, Irvine AF, Jones RL, Samson A. Immunotherapies for hepatocellular carcinoma. Cancer Med. 2022;11:571-91.
108. Kudo M. Scientific rationale for combined immunotherapy with PD-1/PD-L1 antibodies and VEGF inhibitors in advanced hepatocellular carcinoma. Cancers. 2020;12:1089.
109. Finn RS, Qin S, Ikeda M, et al.; IMbrave150 Investigators. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382:1894-905.
110. Qin S, Chan SL, Gu S, et al.; CARES-310 Study Group. Camrelizumab plus rivoceranib versus sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet. 2023;402:1133-46.
111. Holmgaard RB, Schaer DA, Li Y, et al. Targeting the TGFβ pathway with galunisertib, a TGFβRI small molecule inhibitor, promotes anti-tumor immunity leading to durable, complete responses, as monotherapy and in combination with checkpoint blockade. J Immunother Cancer. 2018;6:47.
112. Xiong Z, Chan SL, Zhou J, et al. Targeting PPAR-gamma counteracts tumour adaptation to immune-checkpoint blockade in hepatocellular carcinoma. Gut. 2023;72:1758-73.
113. Yang C, Tao T, Wu Y, Wang W, Liu S. Research advances in immunotherapy combined with DNA damage response inhibitors for liver cancer therapy. J Cancer Metastasis Treat. 2024;10:14.
114. Cheng B, Pan W, Xing Y, Xiao Y, Chen J, Xu Z. Recent advances in DDR (DNA damage response) inhibitors for cancer therapy. Eur J Med Chem. 2022;230:114109.
115. Shi C, Qin K, Lin A, et al. The role of DNA damage repair (DDR) system in response to immune checkpoint inhibitor (ICI) therapy. J Exp Clin Cancer Res. 2022;41:268.
116. Hsiehchen D, Beg MS, Kainthla R, et al. The phosphatidylserine targeting antibody bavituximab plus pembrolizumab in unresectable hepatocellular carcinoma: a phase 2 trial. Nat Commun. 2024;15:2178.
117. Zhu Y, Chen M, Xu D, et al. The combination of PD-1 blockade with interferon-α has a synergistic effect on hepatocellular carcinoma. Cell Mol Immunol. 2022;19:726-37.
118. Yang W, Feng Y, Zhou J, et al. A selective HDAC8 inhibitor potentiates antitumor immunity and efficacy of immune checkpoint blockade in hepatocellular carcinoma. Sci Transl Med. 2021;13:eaaz6804.
119. Liu H, Zeng X, Ren X, et al. Targeting tumour-intrinsic N(7)-methylguanosine tRNA modification inhibits MDSC recruitment and improves anti-PD-1 efficacy. Gut. 2023;72:1555-67.
120. Ning WR, Jiang D, Liu XC, et al. Carbonic anhydrase XII mediates the survival and prometastatic functions of macrophages in human hepatocellular carcinoma. J Clin Invest. 2022;132:e153110.
121. Kaseb AO, Tran Cao HS, Mohamed YI, et al.; Kaseb’s. Final results of a randomized, open label, perioperative phase II study evaluating nivolumab alone or nivolumab plus ipilimumab in patients with resectable HCC. JCO. 2020;38:4599.
122. Abou-alfa GK, Sangro B, Morse M, et al.; 022 Study Group. Phase 1/2 study of durvalumab and tremelimumab as monotherapy and in combination in patients with unresectable hepatocellular carcinoma (HCC). JCO. 2016;34:TPS3103.
123. Rimassa L, Chan SL, Sangro B, et al. Five-year overall survival update from the HIMALAYA study of tremelimumab plus durvalumab in unresectable HCC. J Hepatol. 2025;83:899-908.
124. Duan J, Cui L, Wang G, et al. Immunotherapy efficacy and safety of programmed cell death 1 (PD-1) versus programmed death ligand-1 (PD-L1) inhibitors in pan-cancer: a systematic review and meta-analysis. JCO. 2019;37:e14113.
125. Chan SL, Tan BR, Jimenez E, et al. AMBER part 2F: cobolimab in combination with dostarlimab in treatment-naïve patients with locally advanced/metastatic and/or unresectable hepatocellular carcinoma (HCC). JCO. 2025;43:TPS650.
126. Qin S, Xu L, Yi M, Yu S, Wu K, Luo S. Novel immune checkpoint targets: moving beyond PD-1 and CTLA-4. Mol Cancer. 2019;18:155.
127. Leone P, Solimando AG, Fasano R, et al. The evolving role of immune checkpoint inhibitors in hepatocellular carcinoma treatment. Vaccines. 2021;9:532.
128. Tamai Y, Fujiwara N, Tanaka T, Mizuno S, Nakagawa H. Combination therapy of immune checkpoint inhibitors with locoregional therapy for hepatocellular carcinoma. Cancers. 2023;15:5072.
129. Brandi N, Renzulli M. The synergistic effect of interventional locoregional treatments and immunotherapy for the treatment of hepatocellular carcinoma. Int J Mol Sci. 2023;24:8598.
130. Greten TF, Mauda-Havakuk M, Heinrich B, Korangy F, Wood BJ. Combined locoregional-immunotherapy for liver cancer. J Hepatol. 2019;70:999-1007.
131. Kudo M, Ren Z, Guo Y, et al.; LEAP-012 investigators. Transarterial chemoembolisation combined with lenvatinib plus pembrolizumab versus dual placebo for unresectable, non-metastatic hepatocellular carcinoma (LEAP-012): a multicentre, randomised, double-blind, phase 3 study. Lancet. 2025;405:203-15.
132. Xue M, Wu Y, Tang Y, et al. Efficacy and safety of TACE combined with lenvatinib and PD-1 Inhibitor in intermediate-stage HCC exceeding the up-7 criteria: a retrospective cohort study. Front Immunol. 2025;16:1560750.
133. Chen J, Hu C, Chen Y, Lee Y, Wang C, Lee Y. Tu1536: hepatic arterial infusion chemotherapy (HAIC) with targeted agents and immune checkpoint inhibitors (ICIs) in advanced hepatocellular carcinoma (HCC): systematic review and meta-analysis. Gastroenterology. 2025;169:S-1446.
134. Yunxiuxiu X, Peng L, Chen Y, et al. Apatinib and camrelizumab plus Intravenous FOLFOX or hepatic arterial infusion chemotherapy with FOLFOX for advanced HCC: a multicenter, prospective, randomized phase III trial. JCO. 2024;42:TPS4193.
135. Shannon AH, Manne A, Diaz Pardo DA, Pawlik TM. Combined radiotherapy and immune checkpoint inhibition for the treatment of advanced hepatocellular carcinoma. Front Oncol. 2023;13:1193762.
136. Iyer RV, Petroziello M, Parikh N, et al. A phase II study of atezolizumab (ATEZO) and bevacizumab (BEV) in combination with Y90 TARE in patients (pts) with hepatocellular carcinoma (HCC): Y90+/- BEAT. JCO. 2023;41:TPS629.
137. Li H, You J, Wei Y, et al. Huaier improves the efficacy of anti-PD-L1 Ab in the treatment of hepatocellular carcinoma by regulating tumor immune microenvironment. Phytomedicine. 2024;123:155189.
138. Cao M, Yan H, Han X, et al. Ginseng-derived nanoparticles alter macrophage polarization to inhibit melanoma growth. J Immunother Cancer. 2019;7:326.
139. Yu Z, Li Y, Li Y, et al. Bufalin stimulates antitumor immune response by driving tumor-infiltrating macrophage toward M1 phenotype in hepatocellular carcinoma. J Immunother Cancer. 2022;10:e004297.
140. Guo L, Li H, Fan T, Ma Y, Wang L. Synergistic efficacy of curcumin and anti-programmed cell death-1 in hepatocellular carcinoma. Life Sci. 2021;279:119359.
141. Ke M, Zhang Z, Xu B, et al. Baicalein and baicalin promote antitumor immunity by suppressing PD-L1 expression in hepatocellular carcinoma cells. Int Immunopharmacol. 2019;75:105824.
142. Cai S, Gou Y, Chen Y, et al. Luteolin exerts anti-tumour immunity in hepatocellular carcinoma by accelerating CD8+ T lymphocyte infiltration. J Cell Mol Med. 2024;28:e18535.
143. Zhang L, Jiang G, Yao F, et al. Osthole promotes anti-tumor immune responses in tumor-bearing mice with hepatocellular carcinoma. Immunopharmacol Immunotoxicol. 2015;37:301-7.
144. Yang X, Lam W, Jiang Z, et al. YIV-906 potentiated anti-PD1 action against hepatocellular carcinoma by enhancing adaptive and innate immunity in the tumor microenvironment. Sci Rep. 2021;11:13482.
145. Yu YX, Wang S, Liu ZN, et al. Traditional Chinese medicine in the era of immune checkpoint inhibitor: theory, development, and future directions. Chin Med. 2023;18:59.
146. Mo Z, Cao Z, Yu L, et al. An integrative analysis reveals the potential mechanism between herbal medicine yinchen and immunoregulation in hepatocellular carcinoma. Biomed Res Int. 2020;2020:8886914.
147. Xie Y, Yan F, Wang X, et al. Mechanisms and network pharmacological analysis of Yangyin Fuzheng Jiedu prescription in the treatment of hepatocellular carcinoma. Cancer Med. 2023;12:3237-59.
148. Yan F, Wang X, Xie Y, et al. Yangyin Fuzheng Jiedu Prescription exerts anti-tumor immunity in hepatocellular carcinoma by alleviating exhausted T cells. Phytomedicine. 2021;91:153722.
149. Wu XQ, Ying F, Chung KPS, et al. Intestinal Akkermansia muciniphila complements the efficacy of PD1 therapy in MAFLD-related hepatocellular carcinoma. Cell Rep Med. 2025;6:101900.
150. Ren Z, Li A, Jiang J, et al. Gut microbiome analysis as a tool towards targeted non-invasive biomarkers for early hepatocellular carcinoma. Gut. 2019;68:1014-23.
151. Wu H, Zheng X, Pan T, et al. Dynamic microbiome and metabolome analyses reveal the interaction between gut microbiota and anti-PD-1 based immunotherapy in hepatocellular carcinoma. Int J Cancer. 2022;151:1321-34.
152. Ponziani FR, De Luca A, Picca A, et al. Gut dysbiosis and fecal calprotectin predict response to immune checkpoint inhibitors in patients with hepatocellular carcinoma. Hepatol Commun. 2022;6:1492-501.
153. Sivan A, Corrales L, Hubert N, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. 2015;350:1084-9.
154. Vétizou M, Pitt JM, Daillère R, et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science. 2015;350:1079-84.
155. Lee PC, Wu CJ, Hung YW, et al. Gut microbiota and metabolites associate with outcomes of immune checkpoint inhibitor-treated unresectable hepatocellular carcinoma. J Immunother Cancer. 2022;10:e004779.
156. Yu J, Zhu P, Shi L, et al. Bifidobacterium longum promotes postoperative liver function recovery in patients with hepatocellular carcinoma. Cell Host Microbe. 2024;32:131-44.e6.
157. Russo E, Fiorindi C, Giudici F, Amedei A. Immunomodulation by probiotics and prebiotics in hepatocellular carcinoma. World J Hepatol. 2022;14:372-85.
158. Rauber C, Roberti MP, Vehreschild MJ, et al. Protocol: faecal microbiota transfer in liver cancer to overcome resistance to atezolizumab/bevacizumab - a multicentre, randomised, placebo-controlled, double-blind phase II trial (the FLORA trial). BMJ Open. 2025;15:e097802.
159. Mao J, Wang D, Long J, et al. Gut microbiome is associated with the clinical response to anti-PD-1 based immunotherapy in hepatobiliary cancers. J Immunother Cancer. 2021;9:e003334.
160. Ma C, Awosika JA, Monge C, et al. Altering the gut microbiome and tumor microenvironment in advanced liver cancer: a phase II study of nivolumab, tadalafil and oral vancomycin in patients with refractory primary hepatocellular carcinoma or liver dominant metastatic cancer from colorectal or pancreatic cancers. Cancer Lett. 2025;632:217975.
161. Xiao K, Li K, Xiao K, Yang J, Zhou L. Gut microbiota and hepatocellular carcinoma: metabolic products and immunotherapy modulation. Cancer Med. 2025;14:e70914.
162. Xie D, Liu Y, Xu F, et al. Immune microenvironment and immunotherapy in hepatocellular carcinoma: mechanisms and advances. Front Immunol. 2025;16:1581098.
163. Zhu W, Fan C, Zhao Y, Liu Y, Cheng Y, Zhou W. Breaking bottlenecks: the future of hepatocellular carcinoma clinical trials and therapeutic targets. Hepatol Int. 2025;19:888-902.
164. Guan L, Wu S, Zhu Q, et al. GPC3-targeted CAR-M cells exhibit potent antitumor activity against hepatocellular carcinoma. Biochem Biophys Rep. 2024;39:101741.
165. Myojin Y, McCallen JD, Ma C, et al. Adenosine A2a receptor inhibition increases the anti-tumor efficacy of anti-PD1 treatment in murine hepatobiliary cancers. JHEP Rep. 2024;6:100959.
166. Zhang J, Dong K, Zhang X, Li C, Yu J, Wang W. Characteristics of lactate metabolism phenotype in hepatocellular carcinoma. Sci Rep. 2023;13:19674.
167. Hecht JR, Chan A, Martin M, et al. Phase Ib study of talimogene laherparepvec (T-VEC) injection into liver metastases (LMs) in combination with intravenous (IV) atezolizumab in patients (pts) with metastatic triple-negative breast cancer (TNBC) or colorectal cancer (CRC). JCO. 2019;37:TPS725.
168. Heo J, Reid T, Ruo L, et al. Randomized dose-finding clinical trial of oncolytic immunotherapeutic vaccinia JX-594 in liver cancer. Nat Med. 2013;19:329-36.
169. Zhao J, Chen Y, Ding ZY, Liu JY. Safety and efficacy of therapeutic cancer vaccines alone or in combination with immune checkpoint inhibitors in cancer treatment. Front Pharmacol. 2019;10:1184.
170. Zanotta S, Galati D, De Filippi R, Pinto A. Enhancing dendritic cell cancer vaccination: the synergy of immune checkpoint inhibitors in combined therapies. Int J Mol Sci. 2024;25:7509.
171. Greten TF, Villanueva A, Korangy F, et al. Biomarkers for immunotherapy of hepatocellular carcinoma. Nat Rev Clin Oncol. 2023;20:780-98.
172. Chan YT, Zhang C, Wu J, et al. Biomarkers for diagnosis and therapeutic options in hepatocellular carcinoma. Mol Cancer. 2024;23:189.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






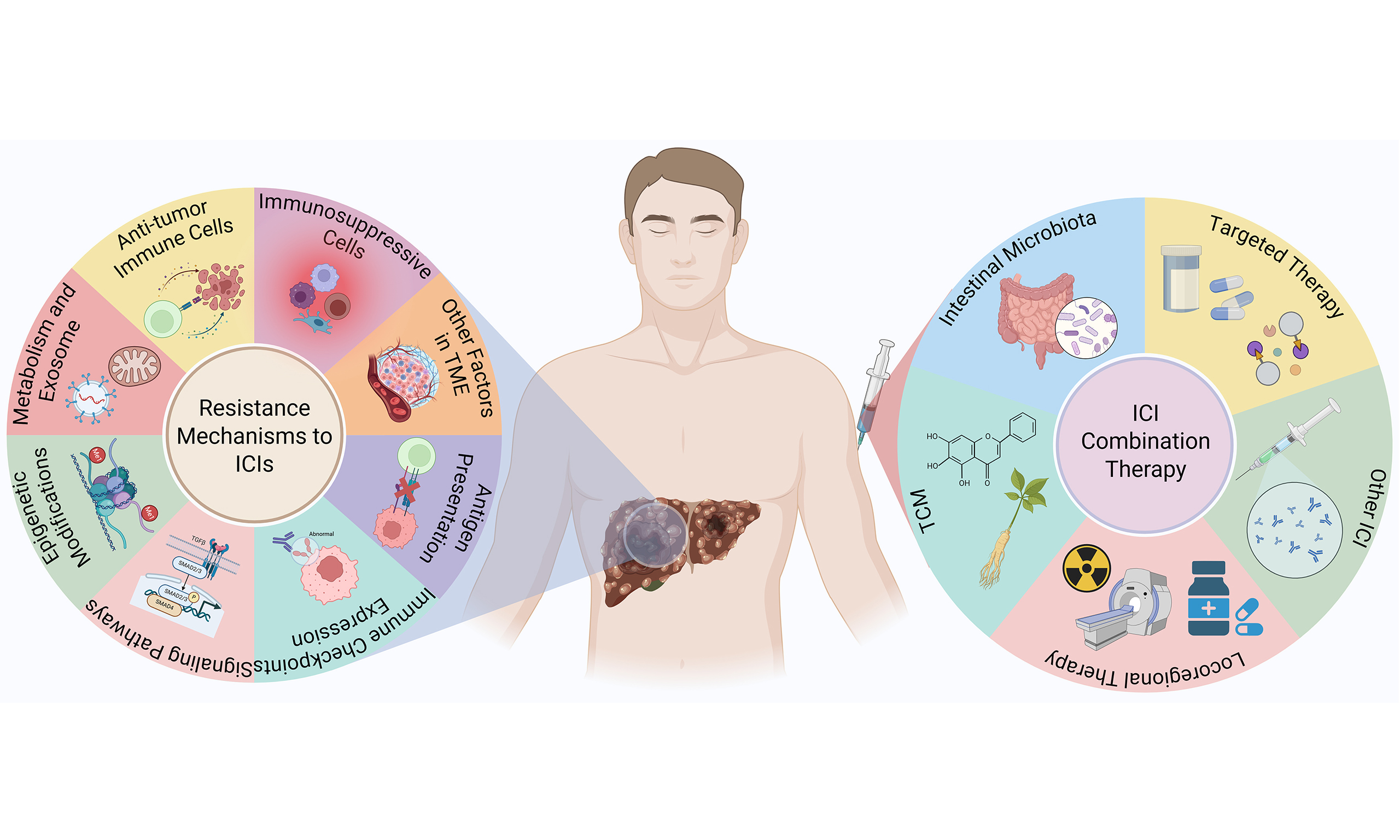
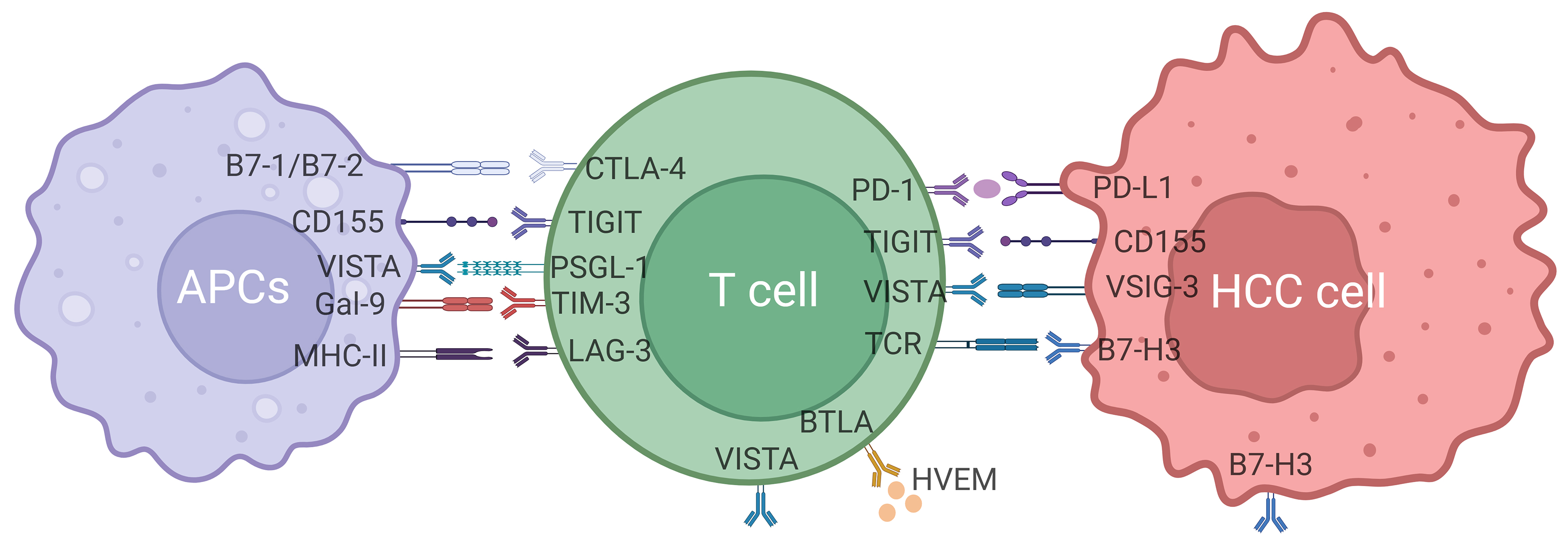
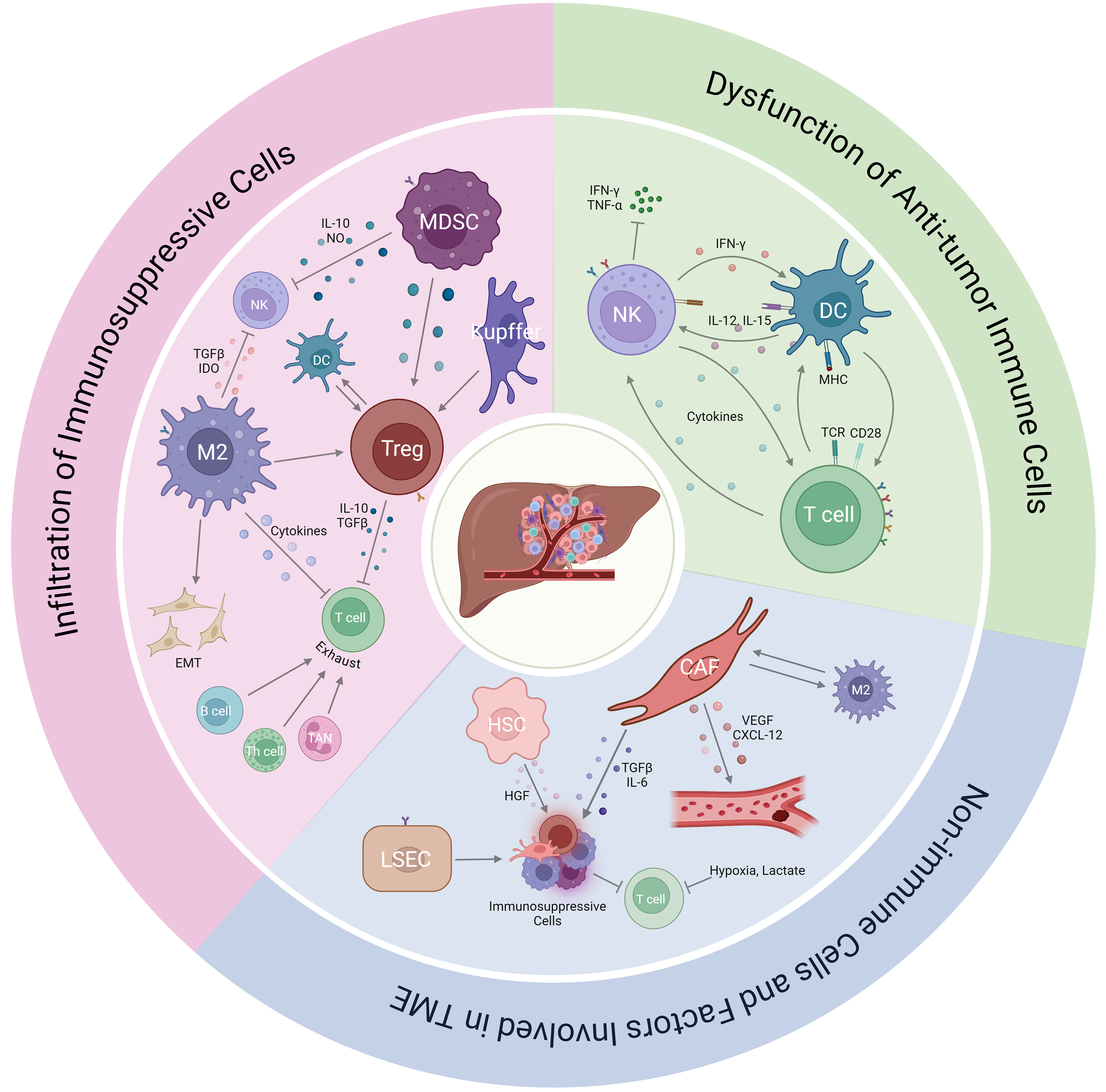
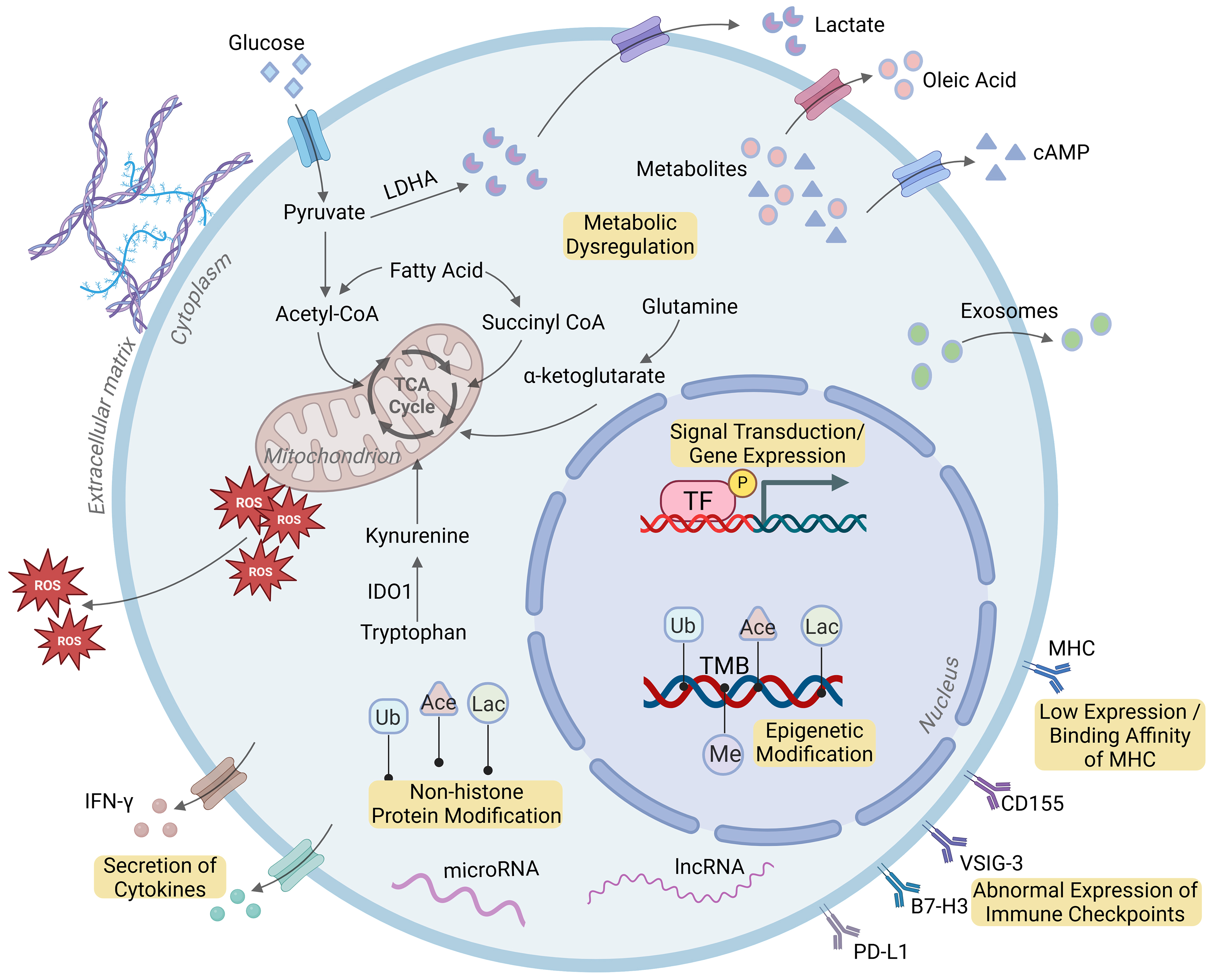










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].