The role of “treatment unfeasibility” in the multiparametric, multidisciplinary, and expert evaluation of HCC patients
Abstract
The Multiparametric Therapeutic Hierarchy (MTH) has been proposed as an evolution of the Barcelona Clinic Liver Cancer (BCLC) algorithm to better reflect the complexity of real-world hepatocellular carcinoma (HCC) management. While the BCLC often assumes idealised conditions, the MTH explicitly incorporates “treatment unfeasibility” as a variable in clinical decision-making, a concept this review seeks to examine and refine. We propose that unfeasibility should not be limited to absolute contraindications but should be understood as a multidimensional construct comprising four interacting dimensions: (1) Technical Feasibility; (2) Resources; (3) Equity; and (4) Values and Acceptability. This concept of unfeasibility adapts the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Evidence-to-Decision (EtD) framework, originally designed for population-level guideline development, to individual-level clinical reasoning. At the point of care, the five EtD domains (Feasibility, Resources, Acceptability, Equity, and Values and Preferences) converge into a unified assessment of treatment feasibility for the specific patient. A precise mapping between our four dimensions and the original GRADE domains is provided. This approach is complementary to, rather than a departure from, GRADE methodology and is particularly relevant in settings characterised by low or very low certainty of evidence. Our analysis reveals a recurring inverse relationship between therapeutic efficacy and feasibility: the most effective treatments, such as liver transplantation, are often constrained by technical, resource-related, and systemic barriers. This tension results in widespread undertreatment across the HCC population. By defining unfeasibility as a key element of expert reasoning, this framework aims to enhance transparency in how multidisciplinary tumour boards weigh multiple considerations when making individualised treatment decisions under uncertainty, without prescribing decision rules or treatment recommendations.
Keywords
INTRODUCTION
Hepatocellular carcinoma (HCC) is the most prevalent primary liver malignancy and represents a significant global health challenge due to its high incidence, frequent association with underlying liver disease, and persistently elevated mortality rates despite recent therapeutic advances[1]. The therapeutic landscape has become increasingly complex, encompassing curative options such as liver transplantation (LT), liver resection (LR), and local ablation (LA), alongside intra-arterial therapies (IATs), external beam radiation therapy (EBRT), and systemic treatments (STs)[2]. To navigate this complexity, systems such as the Barcelona Clinic Liver Cancer (BCLC) algorithm[3] and, more recently, the Multiparametric Therapeutic Hierarchy (MTH)[4] framework have been developed to support treatment selection based on tumour burden, liver function, and patient general health conditions. One of the main differences between the MTH and the BCLC algorithm is that the BCLC often assumes an idealised context, thereby underestimating real-world barriers that may render a theoretically appropriate treatment unfeasible. This limitation is particularly relevant in clinical scenarios where the certainty of evidence supporting one treatment option over another is low or very low, and where guidelines appropriately offer conditional recommendations or multiple reasonable alternatives rather than a single preferred strategy[5]. Notably, the most recent 2025 update of the BCLC system explicitly acknowledges these limitations by introducing the CUSE framework, Complexity, Uncertainty, Subjectivity, and Emotion, as a conceptual recognition that treatment decision-making in HCC cannot be fully captured by rigid, stage-based algorithms alone[6]. This evolution reflects an increasing awareness that uncertainty is inherent to HCC management and that individual values, contextual factors, and expert judgment inevitably influence clinical decisions. Conversely, among the variables explicitly incorporated into the MTH model is treatment unfeasibility, a concept that this paper seeks to explore and refine. Rather than limiting the notion of unfeasibility to the presence of absolute contraindications, we propose a broader, more granular interpretation that reflects the true complexity of HCC management in clinical practice.
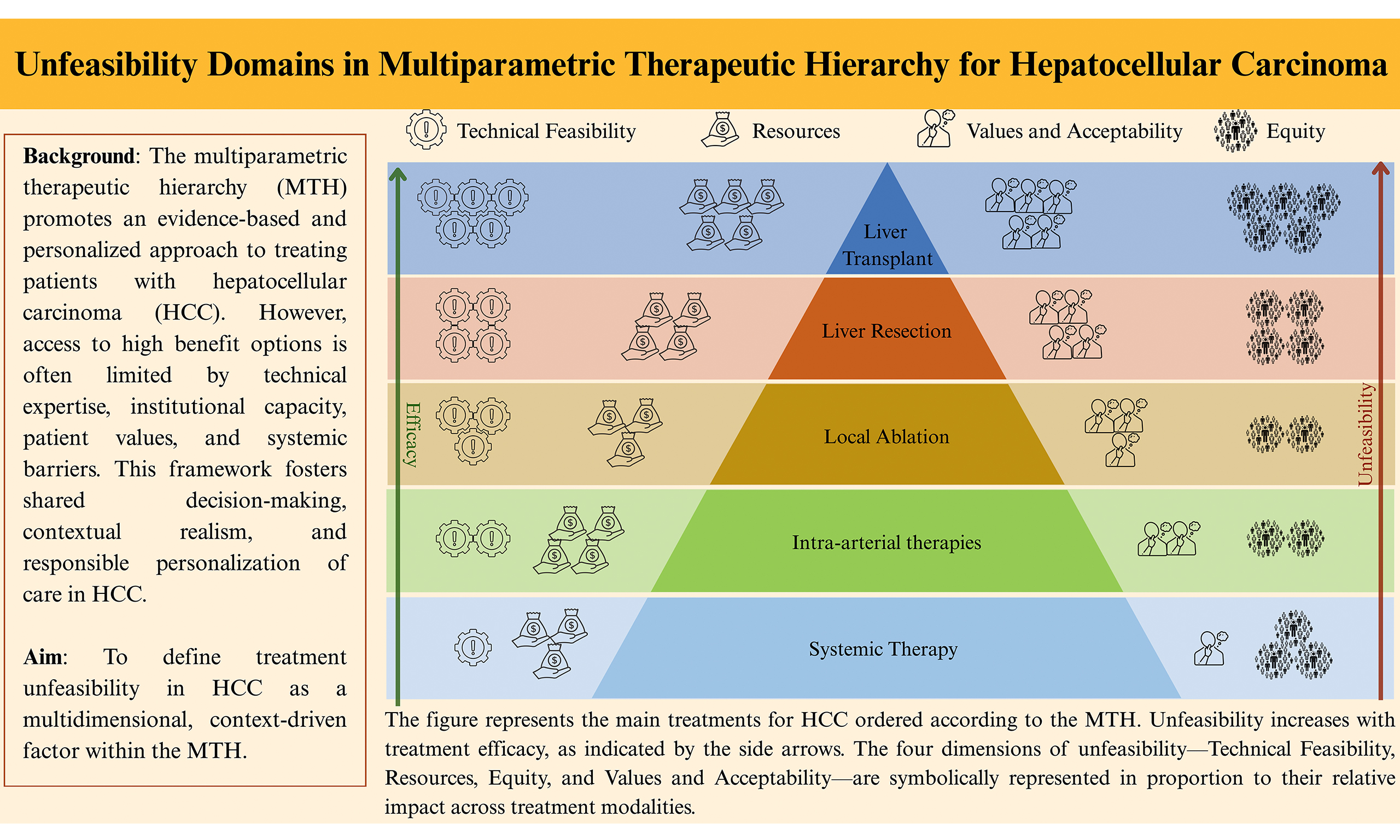
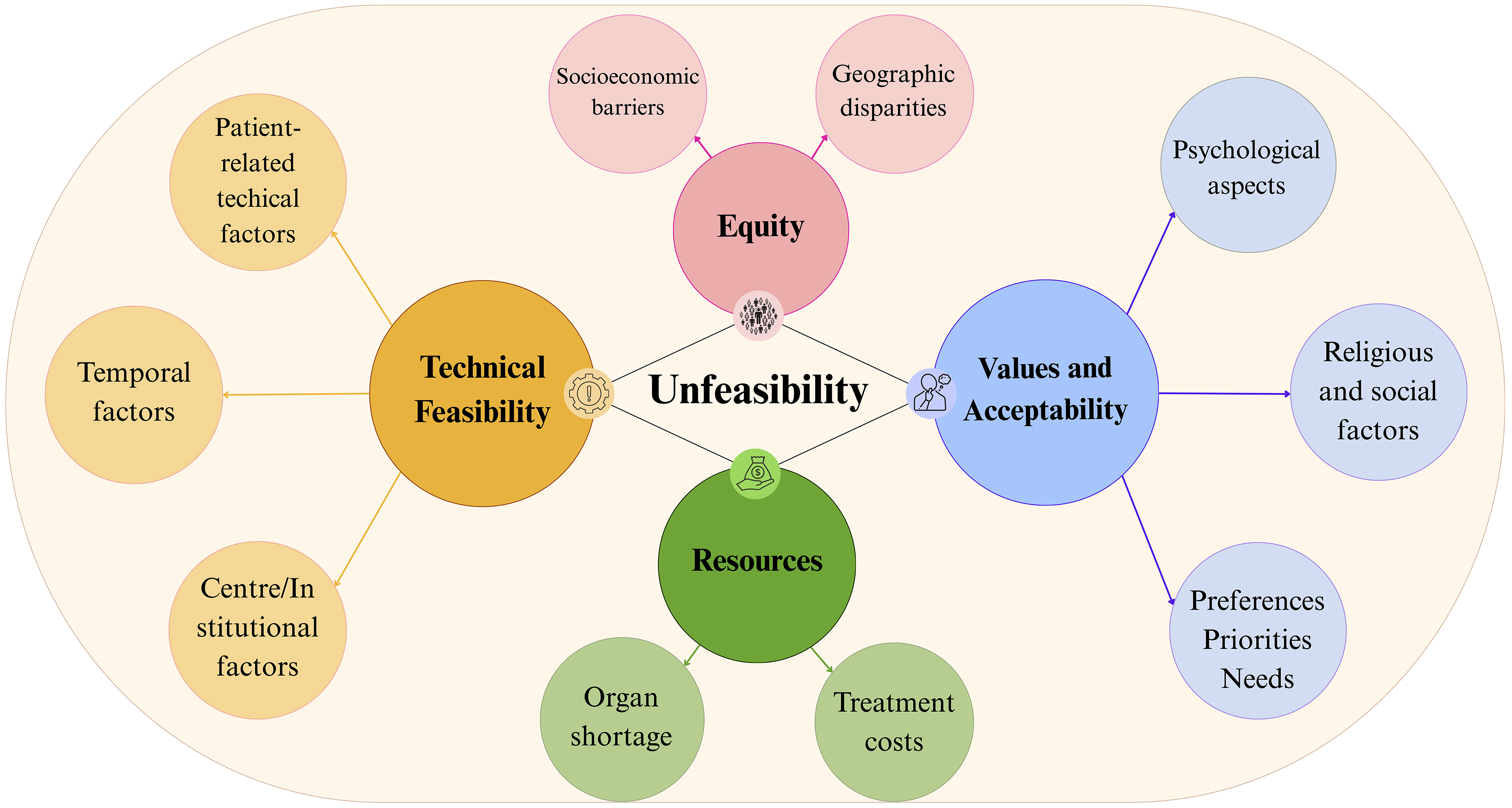
This review aims to provide a conceptual framework for understanding and categorising the multiple dimensions of treatment unfeasibility in HCC [Figure 1], with the intent of enhancing transparency in individualised, multidisciplinary decision-making under conditions of uncertainty, without proposing decision rules or treatment recommendations.
Figure 1. Unfeasibility results from the interaction of four dimensions: Technical Feasibility (including patient-related technical factors, institutional factors, and temporal factors), Resources, Equity, and Values and Acceptability. These dimensions capture barriers to treatment realisation that emerge after a patient has been deemed clinically eligible through MTH criteria (Unfitness, Liver dysfunction, Critical tumour features). This integrative construct adapts the five GRADE EtD domains to individual-level clinical decision-making (see Supplementary Materials, Supplementary Table 1 for the complete mapping and Supplementary Table 2 for the expanded Technical Feasibility domain). This image was created by the authors with Canva. MTH: Multiparametric Therapeutic Hierarchy; GRADE: Grading of Recommendations Assessment, Development and Evaluation; EtD: Evidence-to-Decision.
In routine multidisciplinary tumour board (MDT) discussions, factors influencing treatment unfeasibility are implicitly weighed by expert judgement. Yet these factors are rarely made explicit or systematically documented, potentially limiting the transparency and reproducibility of individualised decisions[7].
To address this gap, we also propose a structured feasibility assessment checklist for MDT use (see Supplementary Materials).
LIVER TRANSPLANTATION
High-volume, experienced centres are associated with improved patient and graft survival, lower waitlist mortality, and fewer complications following LT[8,9]. LT is the first-line treatment for many HCC patients; however, technical challenges in LT remain formidable, particularly in the presence of underlying cirrhosis, portal vein thrombosis, vascular anomalies in the donor, extensive visceral adhesions, and in retransplantation cases[10]. These conditions markedly increase intraoperative complexity, blood loss, and perioperative complication rates[11]. All these challenges, therefore, make the feasibility of transplantation closely dependent on the centre’s surgical experience.
Another key factor that often contributes to the LT unfeasibility is the global limitation of resources, encompassing both economic constraints and limited organ availability. The economic burden arises from the high costs associated with preoperative evaluation, surgery, postoperative care, immunosuppression, and long-term follow-up[12,13]. Historically, organ shortages have represented a critical challenge in the field of LT. While it remains a global issue, its severity is particularly pronounced in certain regions, shaped by country-specific factors, healthcare infrastructure, and donation systems. Recent changes in the United Network for Organ Sharing (UNOS) liver allocation policy and increased reliance on organ sharing have further inflated per-transplant costs, disproportionately affecting patients in rural or underserved areas[14]. While many Asian countries, constrained by cultural limitations on deceased donation, address organ scarcity through living donor liver transplantation (LDLT) programs, the adoption of donation after cardiac death (DCD) protocols and machine perfusion (MP) technologies has markedly expanded the donor pool, mainly in numerous Western nations. However, their implementation remains limited in resource-constrained settings, where infrastructural and economic barriers continue to pose substantial challenges[15,16,17]. Compared to static cold storage, MP entails markedly higher costs due to disposable components, device infrastructure, and specialised expertise[18]. Although these technologies may be cost-effective in selected high-volume centres, their availability and implementation are highly context-dependent and remain uneven across healthcare systems[19].
The distribution and prevalence of LT centres vary significantly across the world, and global disparities in access to LT have long posed a significant ethical challenge. Transplant centres are predominantly concentrated in high-income nations, particularly in the United States, Western Europe, and selected regions of East Asia[20,21]. In low- and middle-income countries, the availability of transplant centres remains severely limited due to inadequate financial resources, infrastructural deficiencies, and poorly organised organ donation programs. Even within high-income countries, disparities persist, driven by geographic, socioeconomic, ethnic, and insurance-related factors, all of which influence both the likelihood of being listed and the probability of receiving a transplant[22,23]. In the United States, around 15% of the population lives in rural areas, whereas transplant centres are predominantly located in urban regions. This disparity in access contributes to a higher risk of overall mortality[24]. In summary, worldwide, less than 10% of patients who require a LT actually undergo the procedure[25,26].
Recently, the European Society for Organ Transplantation (ESOT) and The Lancet have established a Commission on Transplantation with the goal of advancing policy reforms that underline the priority of transplant services, the development of innovative solutions to reduce global inequities in transplantation and promote equitable access to both standard and innovative therapies, including advanced technologies[27]. Jaure et al. identify governance, data infrastructure, workforce development, financing, service delivery, and technology access as key areas requiring systemic support. Countries should implement context-specific strategies that promote public trust, enhance organ donation, and allocate resources effectively. Priority actions include incorporating equity metrics into transplant registries, ensuring financial neutrality for living donors, and expanding telemedicine to improve care in underserved regions[25].
All the aforementioned inequities may lead to prolonged delays and increased waiting list dropout, ultimately resulting in transplantation unfeasibility. Importantly, in many cases, this unfeasibility does not reflect a lack of clinical indication or expected benefit, but rather the interaction between resource constraints, organisational factors, and inequitable access to care. The resulting higher rates of hospitalisation for complications and additional costs associated with prolonged waiting impose significant economic, clinical, and psychological burdens on both patients and the healthcare system[28,29,30].
These systemic challenges not only affect clinical outcomes but also increase patients’ vulnerability, stress, and anxiety, highlighting the critical role of comprehensive psychological and social support throughout the entire transplant journey.
Evaluation for LT is complex and often overwhelming, involving comprehensive medical, surgical, and psychosocial assessments to determine a patient’s eligibility. Many patients find it difficult to manage these overlapping steps, which can lead to frustration, withdrawal from the process, and in some cases, deterioration in health or even death before they can be listed for transplant[31]. The American Association for the Study of Liver Diseases (AASLD) and the American Society of Transplantation recommend a rigorous, comprehensive assessment - covering treatment adherence, substance use, mental‐health stability and social support - as a prerequisite for successful LT and favourable long‐term results[32]. Since lifelong follow-up and immunosuppression are mandatory, this evaluation identifies patients at risk of nonadherence, unrecognised psychiatric disorders or insufficient support systems, each of which can increase the likelihood of graft loss or mortality after LT[13,33].
These assessments may also reveal patient values, preferences, and psychosocial factors that influence the acceptability of transplantation and the patient’s willingness or ability to engage with the proposed care pathway[31,33].
Moreover, it gives patients a structured opportunity to express their preferences, clarify any misconceptions, and fears, potential common drivers of transplant refusal or a preference for less invasive alternatives. Early identification and intervention for psychological or social barriers can improve patient engagement, optimise candidacy, and facilitate shared decision-making[33,34]. Psychological assessment is also essential to a patient’s acceptance of an organ from a deceased donor, as this scenario can be difficult to embrace and may trigger depression, responsibility toward the donor, guilt, or a sense of indebtedness[35,36].
The overall fragility of these patients is critical throughout the transplant journey, emphasising the importance of social support, which is associated with improved post-transplant outcomes[37]. Caregivers play a pivotal role in the early post-transplant phase, when patients are particularly vulnerable to complications, by coordinating logistics, managing medications, and ensuring adherence to complex post-LT regimens, including immunosuppressive therapy and follow-up care[38]. Additionally, robust social support provides both emotional reassurance and practical assistance, helping patients navigate the entire transplant journey, ultimately reducing depression and anxiety, fostering post-traumatic growth, and improving overall health-related quality of life (HRQoL) and survival following LT[39].
Taken together, these elements illustrate that transplantation feasibility is shaped not only by technical and biological factors but also by resource availability, organisational capacity, equity of access, and patient-related acceptability considerations, particularly in contexts of unavoidable uncertainty[5].
LIVER RESECTION
Technical factors are critical in determining the unfeasibility of LR, largely depending on the experience of both the centre and the surgeon. A volume threshold of over 25 LRs per year is generally associated with a reduction in postoperative complications, suggesting that higher experience improves outcomes[40,41]. Minimally invasive approaches offer additional advantages in selected patients[42], particularly in the elderly and fragile ones[43], but require at least 20 laparoscopic resections (LLRs) in complex cases to minimise conversion and morbidity rates[44]. Multidisciplinary teams - including hepatology, oncology, conventional and interventional radiology, intensive care, pathology, and digestive endoscopy - are crucial for optimising perioperative care, particularly in cirrhotic patients[45]. According to National Comprehensive Cancer Network (NCCN) and AASLD guidelines, the resectability of liver tumours requires an adequate evaluation of liver anatomy, function, and the relationship of the tumour to major vessels and bile ducts. Resection is considered technically unfeasible if the future liver remnant (FLR) is insufficient to sustain postoperative liver function - particularly in patients with cirrhosis or advanced steatosis, where the FLR should exceed 40% and 30%, respectively. Analogously, comorbidities that contraindicate major surgery or anaesthesia are also causes of technical unfeasibility[45]. These criteria should be interpreted as context-dependent considerations rather than absolute decision rules, particularly in settings where multiple treatment options may offer comparable benefit under conditions of limited or uncertain evidence[5].
Economic limitations associated with LR increase further with the complexity of the procedure, and the incidence of postoperative complications, which often result in longer hospital stays[46]. Minimally invasive techniques, such as laparoscopic liver resection (LLR), provide a more cost-effective alternative to conventional open surgery, as they are associated with reduced morbidity, shorter hospital stays, and lower readmission rates[47]. Although they entail higher upfront costs due to specialised equipment and
Given the inherent risks of LR, the presence of a LT program within a Hepato-Pancreato-Biliary (HPB) department has been shown to reduce the risk of post-hepatectomy liver failure (PHLF)[51]. It also offers a crucial therapeutic option for high-risk patients, including the potential for curative salvage liver transplantation (SLT) in the event of recurrence[52]. Nowadays, preoperative virtual planning software is available to accurately predict the postoperative FLR volume following LR, with early liver regeneration detectable as soon as five days after surgery[53]. Limited access to such planning tools may therefore influence unfeasibility judgments, particularly in complex cases. Techniques such as portal vein embolisation (PVE), Associating Liver Partition and Portal vein Ligation for Staged Hepatectomy (ALPPS), total venous deprivation (TVD), two-stage hepatectomy and radioembolisation may allow sufficient FLR hypertrophy for resection[54]. These techniques require advanced surgical expertise and substantial institutional resources, and are therefore performed almost exclusively in specialised centres. Consequently, their applicability varies widely across healthcare systems and should be considered in light of local resource availability and organisational constraints.
However, referral centres are not universally available, thereby contributing to disparities in access to care, similar to those observed in LT. As reported in a recent study involving more than 140,000 patients, factors such as lower income, ethnicity, older age, socioeconomic status, geographic location, absence of private insurance, treatment at non-academic institutions, and rural origin profoundly affect access to LR, in particular, minimally invasive ones[55]. Additionally, cultural beliefs, distrust of the healthcare system, and limited language proficiency can contribute to delayed diagnosis or reluctance to pursue surgical intervention. These disparities highlight how equity-related considerations may influence real-world unfeasibility independently of clinical indications[56].
The choice among surgical, locoregional, systemic, or supportive strategies is not determined exclusively by medical appropriateness, but also by the patient’s emotional disposition and capacity to engage with the proposed care pathway. Patients’ preferences are crucial in determining the feasibility of surgical intervention, making it essential to provide a comprehensive risk-benefit assessment during the consultation. Patients often prefer minimally invasive approaches, as they are associated with reduced morbidity, less postoperative pain, and shorter hospital stays, without compromising oncologic outcomes[57]. Their involvement in multidisciplinary discussions and in a shared decision-making process should be essential, as patients consistently value transparent communication about risks, benefits, and alternatives[58].
Similarly, social support plays a crucial role in the perioperative management. Caregivers contribute significantly to reducing psychological distress and promoting functional recovery, HRQoL, and overall well-being[59]. Higher levels of perceived family support are associated with improved postoperative recovery, reduced stress (e.g., lower cortisol levels), and better physical and mental health[60]. Caregivers help mitigate anxiety, depression, and helplessness[61], while also assisting with practical needs such as medication adherence and transportation. As recommended in recent guidelines, their involvement is recognised as a key element of comprehensive patient care[45,62].
In summary, the application of the MTH frameworks cannot be reduced to algorithmic matching of clinical criteria; rather, it requires an individualised approach that considers what is acceptable, tolerable, and meaningful for each patient. Encouraging active involvement of patients and caregivers, while fostering open and respectful dialogue, are essential components of a truly patient-centred approach. Unfeasibility arises from the interaction among technical factors, institutional resources, patient values and preferences, and equity-related constraints, each of which may carry different weights across individual clinical contexts characterised by unavoidable uncertainty (see Supplementary Materials).
LOCAL ABLATION
In LA, tumour and patient characteristics can determine technical limitations. Tumour size (according to major guidelines, LA is curative for tumours ≤ 3 cm[45,63-65]) and location (e.g., tumours adjacent to bile ducts, gastrointestinal structures, or subcapsular or subdiaphragmatic nodules[66]) are considered as part of the “critical tumour features” within MTH. They play a key role in determining the unfeasibility for LA. Moreover, macrovascular invasion or extensive portal vein thrombosis often precludes LA due to limited efficacy[67].
Patients’ coagulopathic status is crucial in LA procedures, as severe thrombocytopenia (< 50,000/µL) and prolonged PT-International Normalized Ratio (INR) (≥ 1.5) increase the risk of bleeding and may render the procedure unfeasible[68]. Chronic kidney disease (CKD) further complicates the procedure due to capillary fragility, even when platelets are normal[69].
Finally, another important aspect to consider in determining LA unfeasibility is a history of biliary surgery or endoscopic procedures. Anatomical changes such as adhesions, biliary prostheses, or bilioenteric anastomoses can complicate needle positioning and increase the risk of complications such as cholangitis, abscess formation, or biliary injury[70-72]. Patients with a history of biliary surgery or endoscopic sphincterotomy are at higher risk of post-procedural cholangitis, primarily due to incomplete biliary drainage, and require rigorous antimicrobial prophylaxis[72].
Another important aspect in this field concerns resources and logistical limitations. LA is more cost-effective compared to surgical options, particularly for early-stage HCC[73,74]. However, resource constraints, such as limited access to high-cost technologies (e.g., microwave ablation, advanced imaging systems, and intraoperative navigation), can restrict the capacity to treat technically challenging tumours[72]. High-volume centres benefit from advanced imaging technologies - such as fusion imaging, Computed Tomography (CT) guidance, cone-beam CT, and electromagnetic navigation - which improve lesion targeting, needle placement, and overall procedural safety[75-79]. The lack of software for centring and margin control, as well as the non-use of contrast agents, should be considered a relative contraindication, as these tools are essential to minimise procedural risks and improve treatment efficacy.
Economic and geographic disparities limit access to locoregional therapies, as patients in rural or low-resource areas may face infrastructure gaps or travel costs, potentially reducing the use of cost-effective LA and shifting treatment toward less effective or more invasive options[45,80].
LA shows clear limitations when patient preferences are considered. In cases of borderline liver function or unfavourable tumour characteristics, patients who value minimally invasive procedures, shorter recovery times, or the avoidance of surgery may be inclined to accept higher procedural risks or modest efficacy. While LA meets these expectations, it offers less durable disease control, creating a trade-off between reduced invasiveness and long-term outcomes. For this reason, patients who prioritise oncologic radicality or have low tolerance for uncertainty are less likely to consider LA appropriate, even when technically feasible[45,81]. These trade-offs between invasiveness, durability of disease control, and tolerance for uncertainty exemplify how patient values and preferences directly shape treatment feasibility in individual clinical contexts. Patient values and preferences are a key Evidence-to-Decision (EtD) criterion (see Supplementary Materials).
Although the quality of life (QoL) following thermal ablation is generally favourable, individuals lacking adequate social support are at higher risk for diminished HRQoL and increased psychological distress, particularly in the social and emotional domains[82,83].
Geographic distance, lack of caregiver support, and indirect costs delay or prevent timely treatment, worsening survival disparities[55]. While less complex than LR, LA remains largely inaccessible outside major urban hospitals, often limited to private or tertiary centres.
IATs AND RADIATION THERAPY
IATs - such as Transarterial Chemoembolisation (TACE), Transarterial Radioembolisation (TARE), and Hepatic Arterial Infusion Chemotherapy (HAIC) - are contraindicated or technically unfeasible in several scenarios. Severely reduced portal vein flow, particularly due to tumour thrombosis, increases the risk of ischemic liver damage, making the hepatic artery the sole supply[84-86]. Vascular anomalies (untreatable arteriovenous fistulae, arterial occlusions) hinder catheterisation and increase non-target embolisation risk. The AASLD points out that technical challenges, such as being unable to access the artery that feeds the tumour due to these vascular anomalies, are valid reasons for deeming TACE and other IATs unsuitable for HCC[45]. Lung shunt occurs when blood and treatments from the hepatic arterial system pass into the lungs, potentially causing treatments such as TARE and TACE to miss their intended targets[87]. A lung shunt resulting in a dose over 25 Gray in one session or 30 Gray across multiple sessions is a contraindication for Y-90 radioembolisation due to the risk of radiation pneumonitis[88]. Biliary tract abnormalities, especially in patients with biliary surgery or hepaticojejunostomy, increase the risk of bile duct necrosis, cholangitis, and abscess[89]. These therapies also face significant economic barriers, with costs varying greatly by treatment method and geographic location. They are often dependent on the expertise and resources available at the treating centre, as not all hospitals have the interventional radiology experience or the necessary tools to perform them. Additionally, the limited availability of these therapies further compounds the inequity in access, as patients in low-resource settings may be unable to access them, which can lead to suboptimal treatment choices[55]. Even though IATs are becoming increasingly important in clinical settings, most people remain unfamiliar with them. This knowledge gap can lead to misunderstandings and hesitations. A Japanese cross-sectional web-based survey investigated patient preferences for the treatment of intermediate or advanced HCC. It specifically compared TACE, HAIC, and STs. The standout favourite was “oral medication”, while the least liked features were “risk of liver damage” and “risk of catheter-related complications.” This study emphasises that factors such as treatment type, how the treatment works, and the associated risks play a significant role in what patients prefer, and these should be considered during shared decision-making for managing HCC[90]. Such findings underscore how acceptability, as defined in the Grading of Recommendations Assessment, Development and Evaluation (GRADE) EtD framework (see Supplementary Materials), varies across populations and can independently influence the real-world feasibility of otherwise technically available treatments.
Although these therapies are generally associated with a lower risk of severe complications compared to LT or LR, patients selected for these treatments frequently present with advanced-stage disease and are often clinically and psychologically vulnerable[62]. As a result, any complications could significantly influence patient well-being, particularly by negatively influencing perceived QoL. In this context, perceived social support has been shown to be highly beneficial and is positively correlated with improved overall HRQoL in these patients[91-93]. Regarding EBRT, it presents its own limitations. The main challenge lies in balancing effective tumoricidal dose with protection of healthy liver tissue and adjacent organs[94]. In patients with poor liver function[45] or tumours near gastrointestinal structures, high-dose radiation may cause ulcers or perforation[95]. Radiotherapy, including techniques such as stereotactic body radiation therapy (SBRT) and proton beam therapy (PBT), requires specialised equipment (precise imaging, managing respiratory motion, and daily image guidance) that is both expensive to purchase and to maintain[96-98]. These therapies are primarily available at high-volume centres, and their high cost can thus limit access, especially in low-resource settings where the infrastructure to support such technologies is lacking. Together, these barriers illustrate how the unfeasibility of intra-arterial and radiation therapies is shaped by the interaction of resources (costs, insurance coverage), acceptability (patient readiness, mistrust), and equity (urban-rural disparities, socioeconomic access). These domains, according to GRADE, deserve explicit consideration in guideline development and individual treatment decisions[99].
SYSTEMIC THERAPIES
The landscape of systemic therapy for advanced HCC has expanded significantly with U.S. Food and Drug Administration (FDA)-approved immunotherapies and tyrosine kinase inhibitors (TKIs), offering improved survival over prior standards[100]. First-line options feature immunotherapy combinations such as atezolizumab plus bevacizumab and the dual checkpoint inhibitor nivolumab plus ipilimumab, approved in April 2025 based on the CheckMate-9DW trial showing superior overall survival versus TKIs[101]. For patients ineligible for immunotherapy, TKIs such as sorafenib and lenvatinib are used. In contrast, second-line and subsequent therapies incorporate additional TKIs such as regorafenib and cabozantinib, along with further immunotherapy combinations, selected based on prior treatment, performance status, and liver function.
The 2025 BCLC guidelines update integrates these advances, recommending personalised first-line systemic therapy for BCLC stage C with immunotherapy combinations as preferred options while preserving stage-linked evidence-based strategies[6].
This framework recognises the high degree of clinical heterogeneity among patients, together with multiple factors that may determine the unfeasibility of individual treatment strategies. This complexity makes MDT discussion essential. Key determinants of treatment unfeasibility include poor performance status or advanced liver dysfunction (Child-Pugh B/C), as well as additional factors such as comorbidities, residual organ function, and treatment tolerance[4]. Real-world studies suggest limited benefit and high toxicity [21% for TKIs and 28% for immune checkpoint inhibitors (ICIs)], with increased risk of severe hepatotoxicity in combined treatments[102]. Autoimmune diseases can make immunotherapy unfeasible due to the increased risk of immune-related adverse events, requiring careful assessment of the severity of the autoimmune condition and treatment alternatives[103,104]. Anti-angiogenic agents also have significant contraindications, including active gastrointestinal bleeding, high-risk esophageal varices, uncontrolled hypertension, and recent major surgery[105]. These therapies require careful evaluation and individualised risk assessment, as conditions such as proteinuria, coagulopathy, and thromboembolic events can be exacerbated[45,106]. Systemic therapies for HCC pose significant economic and logistical barriers. The high cost of drugs, limited insurance coverage, and out-of-pocket expenses are common obstacles to starting and adhering to treatment, particularly in countries without universal healthcare coverage or with inconsistent reimbursement policies[107]. Moreover, the management and administration of STs require a multidisciplinary team, with the hepatologist playing a central role[108]. From a logistical standpoint, STs often require administration in specialised oncology centres for proper monitoring and toxicity management[45], necessitating travel for patients from rural or underserved areas. This represents a considerable barrier for patients with advanced liver disease who may already face physical challenges[109]. It is fundamental to have an informed, collaborative decision process so that patients truly grasp all the treatment choices available to them, along with their benefits, risks, and limitations[110]. Genuine patient involvement goes beyond just clear communication; it also requires active participation. Without the patient’s commitment and mental readiness, STs, especially those that are lengthy, demanding, and do not provide immediate rewards, are unlikely to be effective[111]. Finally, emotional support and continuous, compassionate communication are vital components of an effective therapeutic approach in HCC care. Mistrust in new therapies, concerns about side effects, or misunderstanding of the treatment intent may lead to delayed initiation or refusal. The demanding nature of treatment schedules, potential side effects, and the emotional burden of a malignancy diagnosis can significantly impair a patient’s autonomy and daily functioning[112]. In this context, the presence of a robust support system is critical. Such support not only facilitates adherence to treatment protocols but also contributes to psychological well-being, nutritional maintenance, and transportation to healthcare facilities[113,114]. Therefore, a comprehensive approach that includes assessing and addressing the patient’s support network is essential for optimising the effectiveness and tolerability of STs[115,116].
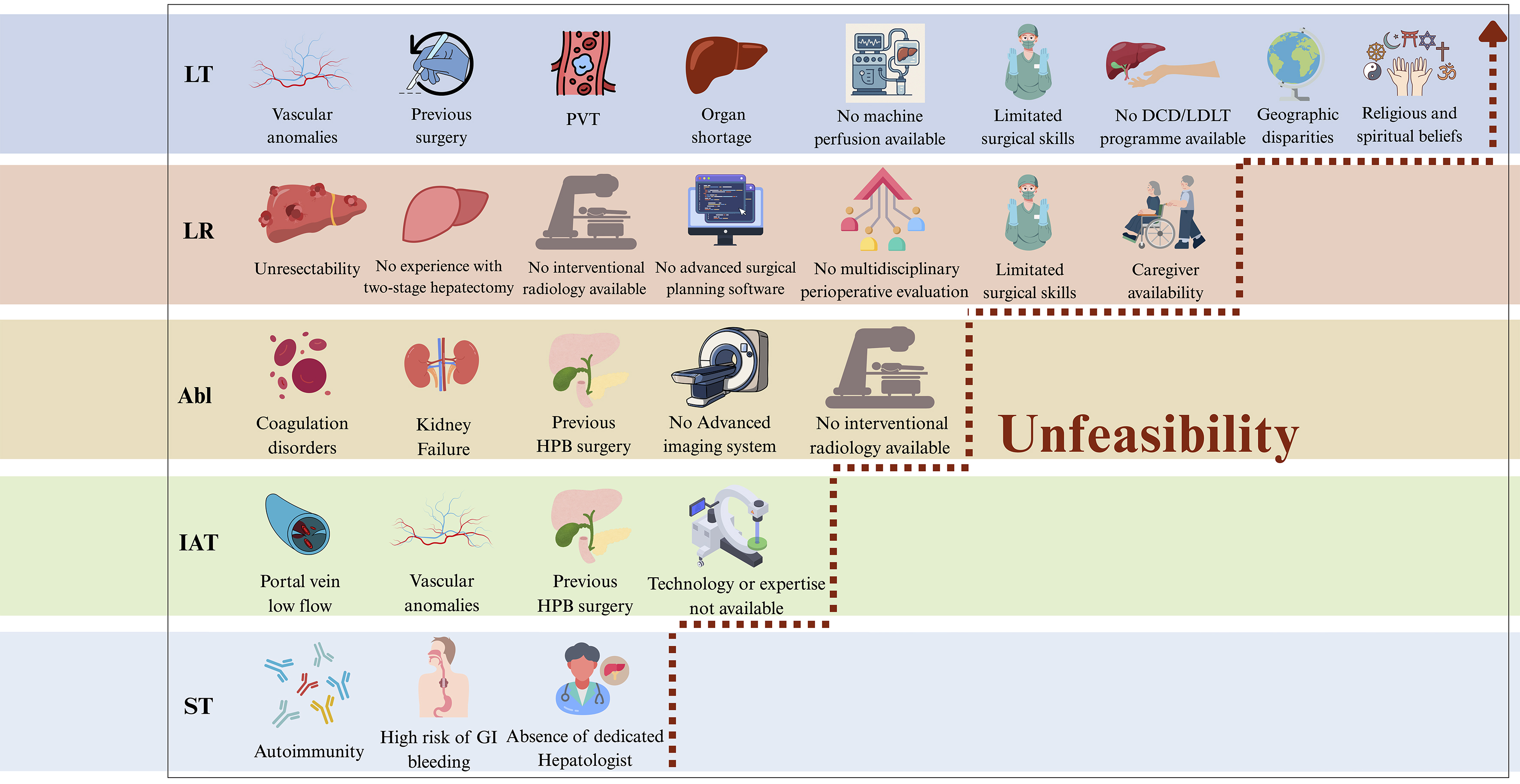
The limitations of ST and other treatments, described above, are summarised in Figure 2.
Figure 2. Examples of limitations, associated with each therapeutic approach, contributing to treatment unfeasibility in HCC across the four dimensions: Technical Feasibility, Resources, Equity, and Values and Acceptability. Unfeasibility, indicated by the arrow, progressively increases with the curative potential of the treatment, such as in liver resection and transplantation. This image was created by the authors with Canva. HCC: Hepatocellular carcinoma; LT: Liver transplantation; LR: liver resection; LA: local ablation; IAT: intra-arterial therapies; ST: systemic therapies; HPB: hepatopancreatobiliary; PVT: portal vein thrombosis; DCD: donation after circulatory death; LDLT: living donor liver transplantation; GI: gastrointestinal.
CONCLUSION
This review has highlighted how the MTH, while conceptually robust, collides with the realities of unfeasibility across HCC treatment modalities. By systematically examining the limitations of transplantation, resection, ablation, IATs, radiotherapy, and STs, we demonstrated that unfeasibility acts as a silent filter that ultimately determines real-world access to care. What clearly emerged from our analysis is the frequent inverse relationship between therapeutic efficacy and unfeasibility [Figure 2], a tension that translates into widespread undertreatment of the HCC population. The most effective strategies, such as LT, are among the most frequently obstructed by barriers, including organ shortages, limited access to enabling technologies, and global disparities in healthcare infrastructure. Conversely, more widely available options tend to have lower curative potential, creating a persistent gap between what could be ideally offered and what is actually delivered to patients. The consequence is a pattern of undertreatment that transcends geographical borders and healthcare systems. Recognising this reality obliges us not only to select the best option for each individual patient but also to confront the systemic, technical, and organisational factors that render therapy unfeasible. While our use of “unfeasibility” as an integrative concept should not obscure the methodological distinction between GRADE EtD domains, it provides a pragmatic framework for MDTs to explicitly document how the four dimensions - Technical Feasibility, Resources, Equity, and Values and Acceptability - converge in shaping treatment choices for individual patients.
Moreover, the unfeasibility of the various treatments within the MTH approach should be understood as a dynamic variable that may change over time. As illustrated in the clinical case reported in the Supplementary Materials, conversion therapy toward more curative interventions can be successfully achieved. Nevertheless, institutional factors - such as access to novel therapies and available capabilities (e.g., interventional radiology) - may also evolve over time, influencing treatment accessibility. Beyond individual decision-making, the oncology community has the duty to effectively address the underlying causes of therapeutic unfeasibility, ensuring that the principle of MTH is not merely theoretical but becomes a standard of care consistently applied in practice.
DECLARATIONS
Acknowledgments
All Figures (Figures 1 and 2 and the Graphical Abstract) were created by the authors using Canva. The copyright of all figures belongs to the authors. All graphical elements were either original or obtained from Canva under its standard content license, which allows use in academic publications.
Govoni I, Padoan V, Brolese M, and Vitale A wish to sincerely thank their mentor, Professor Umberto Cillo, for his enduring guidance, clinical vision, and intellectual inspiration throughout the development of this work.
Members of the Associazione Italiana per lo Studio del Fegato (AISF) HCC Special Interest Group: ASST Papa Giovanni XXIII Bergamo, Italy: Mauro Viganò; Campus Bio-Medico University Rome, Italy: Giovanni Galati; Cardarelli Hospital Naples, Italy: Marco Guarracino, Raffaella Tortora; Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Italy: Nicoletta De Matthaeis, Simone Famularo, Felice Giuliante, Luca Miele, Francesca R. Ponziani; Foggia University Hospital, Italy: Rodolfo Sacco; Hospital of Faenza, Italy: Francesco G. Foschi; Humanitas University Milan, Italy: Luca Viganò; Imperial College London, UK: David J. Pinato; IS.ME.T.T. Palermo, Italy: Salvatore Gruttadauria, Duilio Pagano; Istituto Tumori Milano, Italy: Sherrie Bhoori, Carlo Sposito; National Institute of Gastroenterology Saverio de Bellis Bari, Italy: Raffaele Cozzolongo; Niguarda Ca’ Granda Hospital Milan, Italy: Leonardo Centonze, Chiara Mazzarelli; Polytechnic University of Marche: Daniele Nicolini; University of Rome, Italy: Quirino Lai, Fabio Melandro; University Hospital of Verona, Italy: Paola Violi; University of Bari, Italy: Francesco D’Amico; University of Bologna, Italy: Matteo Cescon, Fabio Piscaglia, Matteo Renzulli, Nicolò Brandi, Francesco Tovoli, Franco Trevisani; University of Genoa, Italy: Edoardo G. Giannini, Giulia Pieri, Maria Corina Platz Torres; University of Milan, Italy: Massimo Iavarone, Angelo Sangiovanni; University of Milano Bicocca, Italy: Fabrizio Romano; University of Modena and Reggio Emilia, Italy: Stefano Di Sandro; University of Naples Federico II, Italy: Maria Guarino, Filomena Morisco; University of Padua, Italy: Patrizia Burra, Umberto Cillo, Fabio Farinati, Martina Gambato, Filippo Pelizzaro, Francesco P. Russo; University of Palermo, Italy: Giuseppe Cabibbo, Ciro Celsa; University of Pisa, Italy: Laura Crocetti, Davide Ghinolfi; University of Salerno, Italy: Mario Masarone, Marcello Persico; University of Tor Vergata Rome, Italy: Ilaria Lenci, Tommaso M. Manzia, Bruno Sensi; University of Udine, Italy: Umberto Baccarani, Riccardo Pravisani; Vita-Salute San Raffaele University Milan, Italy: Andrea Casadei-Gardini; Università degli Studi Link Campus University Roma: Giuseppina Brancaccio.
Authors’ contributions
Responsible for the conception, design, analysis, and writing of the study: Vitale A, Govoni I, Brolese M
Involved with the collection and interpretation of data: Brolese M, Vitale A, Govoni I, Rendina M, Levi Sandri GB, Padoan V
Participated in data management, review and editing of the manuscript: Vitale A, Govoni I, Padoan V, Rendina M, Levi Sandri GB, Finotti M, Brolese M
All authors have reviewed and approved the final version of the manuscript prior to submission.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Vitale A reports consulting and lecture fees from AstraZeneca, and Roche. The other authors declare no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
2. Krupa K, Fudalej M, Cencelewicz-Lesikow A, Badowska-Kozakiewicz A, Czerw A, Deptała A. Current treatment methods in hepatocellular carcinoma. Cancers 2024;16:4059.
3. Reig M, Forner A, Rimola J, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol 2022;76:681-93.
4. Vitale A, Cabibbo G, Iavarone M, et al.; HCC Special Interest Group of the Italian Association for the Study of the Liver. Personalised management of patients with hepatocellular carcinoma: a multiparametric therapeutic hierarchy concept. Lancet Oncol. 2023;24:e312-22.
5. Guyatt GH, Oxman AD, Vist GE, et al.; GRADE Working Group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924-6.
6. Reig M, Sanduzzi-Zamparelli M, Forner A, et al. BCLC strategy for prognosis prediction and treatment recommendations: the 2026 update. J Hepatol 2026;84:631-54.
7. Haynes RB, Devereaux PJ, Guyatt GH. Clinical expertise in the era of evidence-based medicine and patient choice. BMJ Evid Based Med 2002;83:383-6.
8. Hsieh CE, Hsu YL, Lin KH, et al. Association between surgical volumes and hospital mortality in patients: a living donor liver transplantation single center experience. BMC Gastroenterol 2021;21:228.
9. Alqahtani SA, Stepanova M, Kabbara KW, Younossi I, Mishra A, Younossi Z. Liver transplant center size and the impact on clinical outcomes and resource utilization. Transplantation 2022;106:988-96.
10. Azoulay D, Salloum C, Llado L, et al. Defining surgical difficulty of liver transplantation. Ann Surg 2023;277:144-50.
12. Axelrod DA, Gheorghian A, Schnitzler MA, et al. The economic implications of broader sharing of liver allografts. Am J Transplant 2011;11:798-807.
14. Ahmed O, Doyle MBM, Abouljoud MS, et al. Liver transplant costs and activity after united network for organ sharing allocation policy changes. JAMA Surg 2024;159:939-47.
15. Hibi T, Wei Chieh AK, Chi-Yan Chan A, Bhangui P. Current status of liver transplantation in Asia. Int J Surg 2020;82S:4-8.
16. Canizares S, Montalvan A, Chumdermpadetsuk R, Modest A, Eckhoff D, Lee DD. Liver machine perfusion technology: expanding the donor pool to improve access to liver transplantation. Am J Transplant 2024;24:1664-74.
17. Tingle SJ, Dobbins JJ, Thompson ER, et al. Machine perfusion in liver transplantation. Cochrane Database Syst Rev 2023;9:CD014685.
18. Boteon YL, Hessheimer AJ, Brüggenwirth IMA, et al. The economic impact of machine perfusion technology in liver transplantation. Artif Organs 2022;46:191-200.
19. Li Z, Pfister M, Huwyler F, et al. Revolutionizing liver transplantation: transitioning to an elective procedure through ex situ normothermic machine perfusion - a benefit analysis. Ann Surg 2024;280:887-95.
20. Yilma M, Dalal N, Wadhwani SI, Hirose R, Mehta N. Geographic disparities in access to liver transplantation. Liver Transpl 2023;29:987-97.
21. Rinella NS, Charlton W, Reddy G, McLean Diaz P, Charlton MR. Current burden of and geographic disparities in liver mortality and access to liver transplant. JAMA Netw Open 2024;7:e2439846.
22. Ross-Driscoll K, Gunasti J, Ayuk-Arrey AT, et al. Identifying and understanding variation in population-based access to liver transplantation in the United States. Am J Transplant 2023;23:1401-10.
23. Wahid NA, Rosenblatt R, Brown RS Jr. A review of the current state of liver transplantation disparities. Liver Transpl 2021;27:434-43.
24. Goldberg DS, Newcomb C, Gilroy R, et al. Increased distance to a liver transplant center is associated with higher mortality for patients with chronic liver failure. Clin Gastroenterol Hepatol 2017;15:958-60.
25. Jaure A, Jha V, McElroy LM, et al. Policy innovations to advance equity in solid organ transplantation. Lancet 2025;406:403-16.
26. Hill AL, Khan M, Kiani AZ, et al. Global liver transplantation: emerging trends and ethical challenges. Langenbecks Arch Surg 2023;408:418.
27. Oniscu GC, Muller E, Wilson C, Horton R; ESOT-Lancet Commission on Transplantation. The ESOT-lancet commission on transplantation: a new vision for global sustainability, innovation, and equity in organ transplantation. Lancet. 2025;406:313-5.
28. Brand DA, Viola D, Rampersaud P, Patrick PA, Rosenthal WS, Wolf DC. Waiting for a liver--hidden costs of the organ shortage. Liver Transpl 2004;10:1001-10.
29. Yi Z, Mayorga ME, Orman ES, Wheeler SB, Hayashi PH, Barritt AS Th. Trends in characteristics of patients listed for liver transplantation will lead to higher rates of waitlist removal due to clinical deterioration. Transplantation 2017;101:2368-74.
30. Dar FS, Arsalan M, Haq IU, et al. Liver transplantation: a right or a privilege? Sustainable liver transplant financing with an innovative model for the developing world. Transplant Proc 2023;55:402-7.
31. Strauss AT, Brundage J, Sidoti CN, et al. Patient perspectives on liver transplant evaluation: a qualitative study. Patient Educ Couns 2024;127:108346.
32. Martin P, DiMartini A, Feng S, Brown R Jr
33. Bailey P, Vergis N, Allison M, Riddell A, Massey E. Psychosocial evaluation of candidates for solid organ transplantation. Transplantation 2021;105:e292-302.
34. Cooper KM, Colletta A, Amin D, Herscovici DM, Devuni D. Psychosocial trauma history negatively impacts liver transplant access in women with chronic liver disease. J Transplant 2024;2024:2455942.
35. Tarabeih M, Bokek-Cohen Y, Azuri P. Health-related quality of life of transplant recipients: a comparison between lung, kidney, heart, and liver recipients. Qual Life Res 2020;29:1631-9.
36. Saiyu G, Shali W, Yanfang L, Jingping Z. Mental health of recipients after cadaveric liver transplantation: a perspective from positive psychology. J Clin Nurs 2023;32:4710-8.
37. Benson AA, Rowe M, Eid A, et al. Pre-liver transplant psychosocial evaluation predicts post-transplantation outcomes. Psychol Health Med 2018;23:788-96.
38. Kisielska A, Schneider-Matyka D, Rachubińska K, et al. Adherence to therapy among liver transplant patients as related to depression, anxiety, acceptance of the disease, and social support. Eur Rev Med Pharmacol Sci 2023;27:6850-9.
39. Yıldız E, Demir B, Sarıtaş S. Perceived social support associated with posttraumatic growth in liver transplant recipients: a cross-sectional study. Transpl Immunol 2022;72:101562.
40. Magnin J, Bernard A, Cottenet J, et al. Impact of hospital volume in liver surgery on postoperative mortality and morbidity: nationwide study. Br J Surg 2023;110:441-8.
41. Koh YX, Zhao Y, Tan IE, et al. The impact of hospital volume on liver resection: a systematic review and Bayesian network meta-analysis. Surgery 2024;175:393-403.
42. Ciria R, Cherqui D, Geller DA, Briceno J, Wakabayashi G. Comparative short-term benefits of laparoscopic liver resection: 9000 cases and climbing. Ann Surg 2016;263:761-77.
43. Brolese A, Rigoni M, Vitale A, et al. Role of laparoscopic and robotic liver resection compared to open surgery in elderly hepatocellular carcinoma patients: a systematic review and meta-analysis. Hepatoma Res 2020;6:34.
44. Ghamarnejad O, Sahan LA, Kardassis D, et al. Technical aspects and learning curve of complex laparoscopic hepatectomy: how we do it. Surg Endosc 2024;38:4583-93.
45. Singal AG, Llovet JM, Yarchoan M, et al. AASLD practice guidance on prevention, diagnosis, and treatment of hepatocellular carcinoma. Hepatology 2023;78:1922-65.
46. Cosic L, Ma R, Churilov L, et al. The financial impact of postoperative complications following liver resection. Medicine 2019;98:e16054.
47. Koh YX, Zhao Y, Tan IE, et al. Cost-effectiveness of laparoscopic vs open liver resection: a propensity score-matched single-center analysis of 920 cases. J Am Coll Surg 2025;241:203-19.
48. Aziz H, Hanna K, Lashkari N, Ahmad NU, Genyk Y, Sheikh MR. Hospitalization costs and outcomes of open, laparoscopic, and robotic liver resections. Am Surg 2022;88:2331-7.
49. Miller HP, Hakim A, Kellish A, et al. Cost-benefit analysis of robotic vs. laparoscopic hepatectomy: a propensity-matched retrospective cohort study of american college of surgeons national surgical quality improvement program database. Am Surg 2022;88:2886-92.
50. Koh YX, Zhao Y, Tan IE, et al. Comparative cost-effectiveness of open, laparoscopic, and robotic liver resection: a systematic review and network meta-analysis. Surgery 2024;176:11-23.
51. Serenari M, Lenzi J, Cucchetti A, et al.; HE.RC.O.LE.S. Group. The effect of a liver transplant program on the outcomes of resectable hepatocellular carcinoma: a nationwide multicenter analysis. Ann Surg. 2023;277:664-71.
52. Vitale A, Brolese M, Govoni I, et al. Laparoscopic microwave ablation and salvage liver transplantation in patients with hepatocellular carcinoma. Cancers 2025;17:2248.
53. Simpson AL, Geller DA, Hemming AW, et al. Liver planning software accurately predicts postoperative liver volume and measures early regeneration. J Am Coll Surg 2014;219:199-207.
54. Wong P, Vien P, Kessler J, Lafaro K, Wei A, Melstrom LG. Augmenting the future liver remnant prior to major hepatectomy: a review of options on the menu. Ann Surg Oncol 2025;32:5694-709.
55. Beltrán Ponce S, Gokun Y, Douglass F, et al. Disparities in outcomes and access to therapy options in hepatocellular carcinoma. J Natl Cancer Inst 2024;116:264-74.
56. Welch VA, Akl EA, Guyatt G, et al. GRADE equity guidelines 1: considering health equity in GRADE guideline development: introduction and rationale. J Clin Epidemiol 2017;90:59-67.
57. Coletta D, De Padua C, Parrino C, et al. Laparoscopic liver surgery: what are the advantages in patients with cirrhosis and portal hypertension? Systematic review and meta-analysis with personal experience. J Laparoendosc Adv Surg Tech A 2020;30:1054-65.
58. Nathan H, Bridges JF, Schulick RD, et al. Understanding surgical decision making in early hepatocellular carcinoma. J Clin Oncol 2011;29:619-25.
59. Archer S, Pinto A, Vuik S, et al. Surgery, complications, and quality of life: a longitudinal cohort study exploring the role of psychosocial factors. Ann Surg 2019;270:95-101.
60. Cardoso-Moreno MJ, Tomás-Aragones L. The influence of perceived family support on post surgery recovery. Psychol Health Med 2017;22:121-8.
61. Li L, Zhang HZ, Ge Y, et al. The perioperative experience and needs of hepatocellular carcinoma patients in interventional therapy: a phenomenological qualitative study. Eur J Gastroenterol Hepatol 2024;36:423-9.
62. Association for the Study of the Liver. EASL clinical practice guidelines on the management of hepatocellular carcinoma. J Hepatol 2025;82:315-74.
63. Omata M, Cheng AL, Kokudo N, et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int 2017;11:317-70.
64. NCCN Guidelines for patients: liver cancer. Available from: https://www.nccn.org/patients/guidelines/content/PDF/liver-hp-patient.pdf. [Last accessed on 15 Apr 2026].
65. Goh MJ, Kang W, Kim KM, et al. Incidence and risk factors for development of hepatocellular carcinoma at young age in patients with chronic hepatitis B. Scand J Gastroenterol 2022;57:70-7.
66. Crocetti L, de Baére T, Pereira PL, Tarantino FP. CIRSE standards of practice on thermal ablation of liver tumours. Cardiovasc Intervent Radiol 2020;43:951-62.
67. Kang TW, Lim HK, Cha DI. Percutaneous ablation for perivascular hepatocellular carcinoma: Refining the current status based on emerging evidence and future perspectives. World J Gastroenterol 2018;24:5331-7.
68. Goto E, Tateishi R, Shiina S, et al. Hemorrhagic complications of percutaneous radiofrequency ablation for liver tumors. J Clin Gastroenterol 2010;44:374-80.
69. Ocak G, Rookmaaker MB, Algra A, et al.; SMART Study Group. Chronic kidney disease and bleeding risk in patients at high cardiovascular risk: a cohort study. J Thromb Haemost. 2018;16:65-73.
70. Choi D, Lim HK, Kim MJ, et al. Liver abscess after percutaneous radiofrequency ablation for hepatocellular carcinomas: frequency and risk factors. AJR Am J Roentgenol 2005;184:1860-7.
71. Nault JC, Sutter O, Nahon P, Ganne-Carrié N, Séror O. Percutaneous treatment of hepatocellular carcinoma: state of the art and innovations. J Hepatol 2018;68:783-97.
72. Minami Y, Aoki T, Hagiwara S, Kudo M. Tips for preparing and practicing thermal ablation therapy of hepatocellular carcinoma. Cancers 2023;15:4763.
73. Charalel RA, Mushlin AI, Zheng X, et al. Short-term out-of-pocket and total costs of care after ablation, resection, or transplant for early-stage hepatocellular carcinoma: a national SEER-medicare cost comparison. AJR Am J Roentgenol 2024;223:e2431272.
74. Cucchetti A, Piscaglia F, Cescon M, et al. Cost-effectiveness of hepatic resection versus percutaneous radiofrequency ablation for early hepatocellular carcinoma. J Hepatol 2013;59:300-7.
75. Kato K, Abe H, Ika M, et al. Efficacy of fusion imaging and cone-beam computed tomography-guided radiofrequency ablation for hepatocellular carcinoma poorly visualized on ultrasonography. Oncology 2026;104:270-82.
76. Ahn SJ, Lee JM, Lee DH, et al. Real-time US-CT/MR fusion imaging for percutaneous radiofrequency ablation of hepatocellular carcinoma. J Hepatol 2017;66:347-54.
77. Calandri M, Mauri G, Yevich S, et al. Fusion imaging and virtual navigation to guide percutaneous thermal ablation of hepatocellular carcinoma: a review of the literature. Cardiovasc Intervent Radiol 2019;42:639-47.
78. Huang ZM, Zuo MX, Gu YK, et al. Computed tomography-guided radiofrequency ablation combined with transarterial embolization assisted by a three-dimensional visualization ablation planning system for hepatocellular carcinoma in challenging locations: a preliminary study. Abdom Radiol 2020;45:1181-92.
79. Volpi S, Tsoumakidou G, Loriaud A, Hocquelet A, Duran R, Denys A. Electromagnetic navigation system combined with High-Frequency-Jet-Ventilation for CT-guided hepatic ablation of small US-Undetectable and difficult to access lesions. Int J Hyperthermia 2019;36:1051-7.
80. Ramtohul T, Vilgrain V, Soubrane O, et al. Impact of extended use of ablation techniques in cirrhotic patients with hepatocellular carcinoma: a cost-effectiveness analysis. Cancers 2022;14:2634.
81. Gervais DA, Goldberg SN, Brown DB, Soulen MC, Millward SF, Rajan DK. Society of Interventional Radiology position statement on percutaneous radiofrequency ablation for the treatment of liver tumors. J Vasc Interv Radiol 2009;20:S342-7.
82. Huang G, Chen X, Lau WY, et al. Quality of life after surgical resection compared with radiofrequency ablation for small hepatocellular carcinomas. Br J Surg 2014;101:1006-15.
83. Toro A, Pulvirenti E, Palermo F, Di Carlo I. Health-related quality of life in patients with hepatocellular carcinoma after hepatic resection, transcatheter arterial chemoembolization, radiofrequency ablation or no treatment. Surg Oncol 2012;21:e23-30.
84. Tan ZB, Zhang J. Recent advances in treatment strategies for hepatocellular carcinoma with portal vein cancer thrombus. Eur Rev Med Pharmacol Sci 2023;27:8119-34.
85. Garg T, Shrigiriwar A, Habibollahi P, et al. Intraarterial therapies for the management of hepatocellular carcinoma. Cancers 2022;14:3351.
86. Ebeling Barbier C, Heindryckx F, Lennernäs H. Limitations and possibilities of transarterial chemotherapeutic treatment of hepatocellular carcinoma. Int J Mol Sci 2021;22:13051.
87. Ward TJ, Tamrazi A, Lam MG, et al. Management of high hepatopulmonary shunting in patients undergoing hepatic radioembolization. J Vasc Interv Radiol 2015;26:1751-60.
88. Busse NC, Al-Ghazi MSAL, Abi-Jaoudeh N, et al. AAPM medical physics practice guideline 14.a: Yttrium-90 microsphere radioembolization. J Appl Clin Med Phys 2024;25:e14157.
89. Brown ZJ, Tsilimigras DI, Ruff SM, et al. Management of hepatocellular carcinoma: a review. JAMA Surg 2023;158:410-20.
90. Chiba T, Hiraoka A, Mikami S, et al. Japanese patient preferences regarding intermediate to advanced hepatocellular carcinoma treatments. Patient Prefer Adherence 2019;13:637-47.
91. Loffroy R, Ronot M, Greget M, et al.; CIRT-FR Principal Investigators. Short-term safety and quality of life outcomes following radioembolization in primary and secondary liver tumours: a multi-centre analysis of 200 patients in France. Cardiovasc Intervent Radiol. 2021;44:36-49.
92. Zhang WZ, Han JQ, Chin KY, Zakaria R, Hassan NH. Determinants of health-related quality of life after transarterial chemoembolization in hepatocellular carcinoma patients: a systematic review. J Clin Med 2025;14:3941.
93. Man KH, Law HK, Tam SY. Psychosocial needs of post-radiotherapy cancer survivors and their direct caregivers - a systematic review. Front Oncol 2023;13:1246844.
94. Apisarnthanarax S, Barry A, Cao M, et al. External beam radiation therapy for primary liver cancers: an ASTRO clinical practice guideline. Pract Radiat Oncol 2022;12:28-51.
95. Tsurugai Y, Takeda A, Eriguchi T, Sanuki N, Aoki Y. Hypofractionated radiotherapy for hepatocellular carcinomas adjacent to the gastrointestinal tract. Hepatol Res 2021;51:294-302.
96. Knavel Koepsel EM, Smolock AR, Pinchot JW, et al.; Expert Panel on Interventional Radiology. ACR appropriateness Criteria® management of liver cancer: 2022 update. J Am Coll Radiol. 2022;19:S390-408.
97. van de Lindt TN, Fast MF, van den Wollenberg W, et al. Validation of a 4D-MRI guided liver stereotactic body radiation therapy strategy for implementation on the MR-linac. Phys Med Biol 2021;66:105010.
98. Dhont J, Harden SV, Chee LYS, Aitken K, Hanna GG, Bertholet J. Image-guided radiotherapy to manage respiratory motion: lung and liver. Clin Oncol 2020;32:792-804.
99. Pottie K, Welch V, Morton R, et al. GRADE equity guidelines 4: considering health equity in GRADE guideline development: evidence to decision process. J Clin Epidemiol 2017;90:84-91.
100. Selene II, Ozen M, Patel RA. Hepatocellular carcinoma: advances in systemic therapy. Semin Intervent Radiol 2024;41:56-62.
101. Yau T, Galle PR, Decaens T, et al.; CheckMate 9DW investigators. Nivolumab plus ipilimumab versus lenvatinib or sorafenib as first-line treatment for unresectable hepatocellular carcinoma (CheckMate 9DW): an open-label, randomised, phase 3 trial. Lancet. 2025;405:1851-64.
102. Griffiths CD, Zhang B, Tywonek K, Meyers BM, Serrano PE. Toxicity profiles of systemic therapies for advanced hepatocellular carcinoma: a systematic review and meta-analysis. JAMA Netw Open 2022;5:e2222721.
103. Sangro B, Sarobe P, Hervás-Stubbs S, Melero I. Advances in immunotherapy for hepatocellular carcinoma. Nat Rev Gastroenterol Hepatol 2021;18:525-43.
104. Foerster F, Gairing SJ, Ilyas SI, Galle PR. Emerging immunotherapy for HCC: a guide for hepatologists. Hepatology 2022;75:1604-26.
105. Su GL, Altayar O, O’Shea R, et al. AGA clinical practice guideline on systemic therapy for hepatocellular carcinoma. Gastroenterology 2022;162:920-34.
106. De Gaetano V, Pallozzi M, Cerrito L, et al. Management of portal hypertension in patients with hepatocellular carcinoma on systemic treatment: current evidence and future perspectives. Cancers 2024;16:1388.
107. Gordan JD, Kennedy EB, Abou-Alfa GK, et al. Systemic Therapy for Advanced Hepatocellular Carcinoma: ASCO Guideline Update. J Clin Oncol 2024;42:1830-50.
108. Cabibbo G, Aghemo A, Lai Q, Masarone M, Montagnese S, Ponziani FR; Italian Association for the Study of the Liver (AISF). Optimizing systemic therapy for advanced hepatocellular carcinoma: the key role of liver function. Dig Liver Dis. 2022;54:452-60.
109. Likhitsup A, Parikh ND. Economic implications of hepatocellular carcinoma surveillance and treatment: a guide for clinicians. Pharmacoeconomics 2020;38:5-24.
110. Noteboom EA, May AM, van der Wall E, de Wit NJ, Helsper CW. Patients’ preferred and perceived level of involvement in decision making for cancer treatment: a systematic review. Psychooncology 2021;30:1663-79.
111. Nizet P, Grivel C, Rabeau P, et al. Patients’ preferences in therapeutic decision-making in digestive oncology: a single centre cross-sectional observational study. Sci Rep 2023;13:8534.
112. Graf J, Stengel A. Psychological burden and psycho-oncological interventions for patients with hepatobiliary cancers-a systematic review. Front Psychol 2021;12:662777.
113. Ruiz-Rodríguez I, Hombrados-Mendieta I, Melguizo-Garín A, Martos-Méndez MJ. The importance of social support, optimism and resilience on the quality of life of cancer patients. Front Psychol 2022;13:833176.
114. Pasek M, Goździalska A, Jochymek M, Caruso R. Social support in a cancer patient-informal caregiver dyad: a scoping review. Cancers 2023;15:1754.
115. Olver I, Keefe D, Herrstedt J, Warr D, Roila F, Ripamonti CI. Supportive care in cancer-a MASCC perspective. Support Care Cancer 2020;28:3467-75.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].