fig2
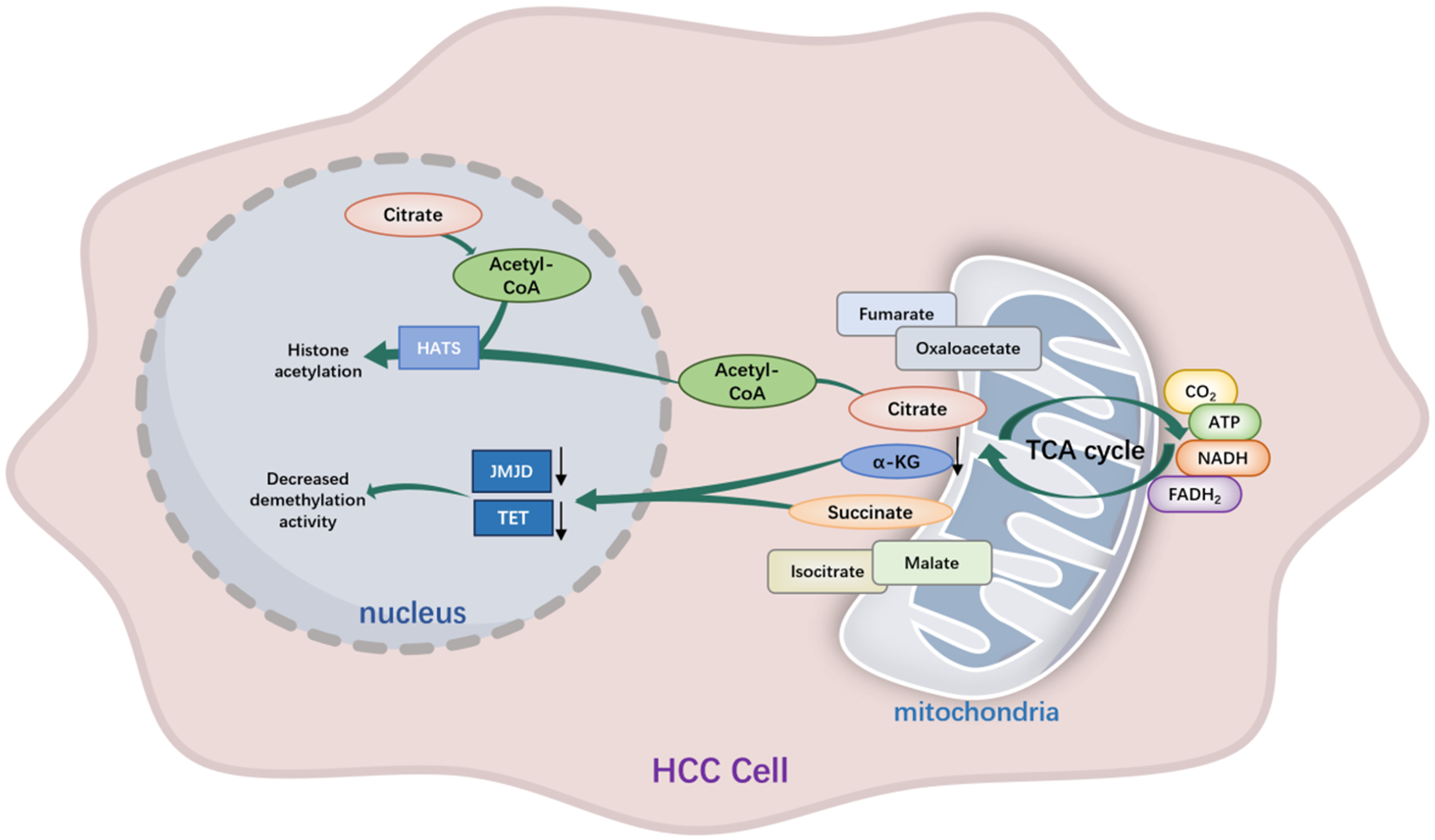
Figure 2. Metabolic–epigenetic coupling of the TCA cycle in HCC. Reprogrammed TCA cycle flux in HCC generates metabolic signals that directly regulate nuclear chromatin states. Succinate accumulation inhibits α-KG–dependent TET and JMJD demethylases, promoting DNA and histone hypermethylation, whereas reduced α-KG availability further constrains demethylation and limits enhancer plasticity. In parallel, mitochondrial-derived citrate is exported and converted to acetyl-CoA to fuel histone acetyltransferase activity, reinforcing oncogenic transcriptional programs. TCA: Tricarboxylic acid; HCC: hepatocellular carcinoma; α-KG: α-ketoglutarate; TET: ten-eleven translocation; JMJD: Jumonji C domain-containing; HATs: histone acetyltransferases; ATP: adenosine triphosphate; NADH: nicotinamide adenine dinucleotide; FADH2: flavin adenine dinucleotide.