fig1
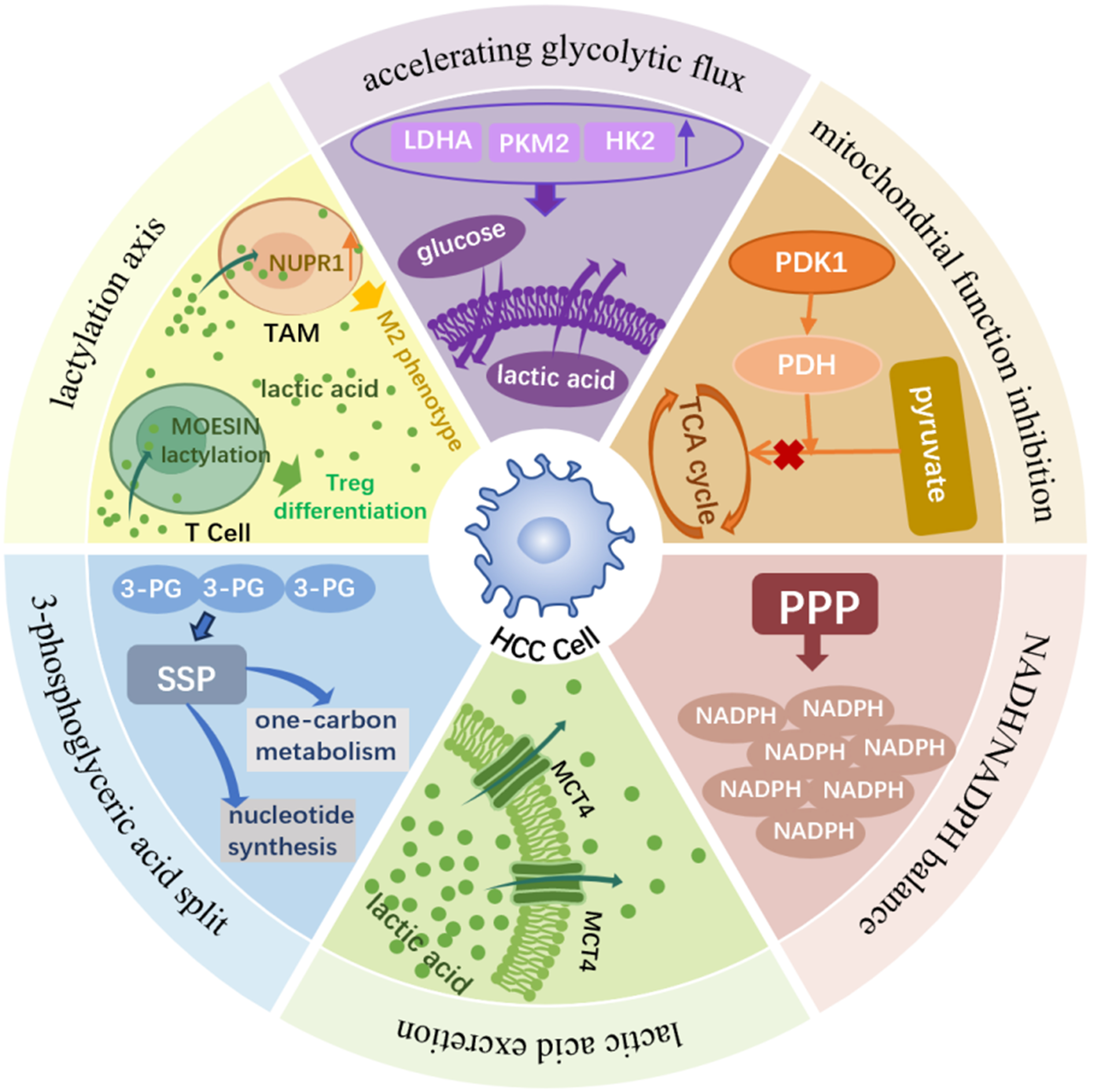
Figure 1. The Warburg effect in HCC: mechanisms and functional consequences. HCC cells preferentially rely on aerobic glycolysis - the Warburg effect - even in the presence of oxygen and functional mitochondria. Upregulation of key glycolytic enzymes (HK2, PKM2, LDHA) enhances glucose uptake, glycolytic flux, and lactate production/export. PDH inhibition diverts pyruvate away from the TCA cycle, reinforcing glycolytic dependency. Glycolytic intermediates are channeled into anabolic pathways: G6P fuels the PPP to generate NADPH for redox balance, while 3-PG supports serine and one-carbon metabolism for nucleotide biosynthesis and proliferation. Excess lactate is exported via MCTs, accumulates in the TME, and acts as a signaling molecule that induces lysine lactylation. Lactylation promotes HCC progression by activating stemness-related transcriptional programs and reshaping the immune microenvironment - driving M2 polarization of TAMs via NUPR1 upregulation and facilitating regulatory T-cell differentiation through MOESIN K72 lactylation. Hypoxia further amplifies these processes via HIF-1α–mediated transcriptional activation of glycolytic genes, collectively establishing a metabolic–epigenetic landscape that supports HCC survival, immune evasion, and progression. HCC: Hepatocellular carcinoma; HK2: hexokinase 2; PKM2: pyruvate kinase M2; LDHA: lactate dehydrogenase A; PDH: pyruvate dehydrogenase; TCA: tricarboxylic acid; G6P: glucose-6-phosphate; PPP: pentose phosphate pathway; NADPH: nicotinamide adenine dinucleotide phosphate; 3-PG: 3-phosphoglycerate; MCTs: monocarboxylate transporters; TME: tumor microenvironment; TAMs: tumor-associated macrophages; NUPR1: nuclear protein 1; HIF-1α: hypoxia-inducible factor 1α; PDK1: pyruvate dehydrogenase kinase 1; NADH: nicotinamide adenine dinucleotide; SSP: serine synthesis pathway; Treg: regulatory T cell.