Converse therapeutic hierarchy in hepatocellular carcinoma
Abstract
The therapeutic landscape of hepatocellular carcinoma (HCC) is undergoing a substantial transformation driven by advances in systemic therapies and locoregional treatments. High response rates observed with immune checkpoint inhibitors and combination regimens have opened the door to conversion therapy. Initially unresectable or non-curable patients can achieve a tumour downsizing to access potentially curative options such as surgery, ablation, or transplantation. This evolving strategy is framed within the concept of the converse therapeutic hierarchy, which promotes a dynamic, response-guided approach. In this model, every treatment is no longer a terminal option but a potential gateway to curative interventions. This review explores the clinical rationale and current evidence supporting conversion therapy in HCC, detailing systemic regimens, transarterial and percutaneous treatments, and their integration into multimodal strategies. Emphasis is placed on response-guided treatment reassessment, perioperative immunotherapy, and the potential of tailored sequencing to redefine clinical practice in HCC. Barriers such as biological heterogeneity, the lack of predictive biomarkers, and organisational gaps in multidisciplinary coordination remain significant. At the same time, improvements in systemic efficacy, advances in locoregional techniques, and new evidence from real-world data point toward a future in which therapeutic intent is no longer fixed but can evolve according to patient response. Conversion therapy, once aspirational, is becoming a realistic and strategic objective in modern HCC care.
Keywords
INTRODUCTION
Hepatocellular carcinoma (HCC) is a significant and complex cancer with worldwide relevance, marked by both clinical and biological diversity, and frequently linked to an unfavourable prognosis[1]. Assessing outcomes and selecting an appropriate treatment approach is incredibly challenging in individuals with HCC due to the prognostic influence of underlying liver conditions and the common occurrence of clinical frailty[2-4].
In clinical practice, HCC treatments are often applied individually. Examples include liver resection (LR) or thermal ablation for solitary tumours, and systemic therapies (STs) for metastatic disease. However, in real-world settings, several modalities are frequently combined to manage complex cases such as multinodular or locally advanced HCC[5]. These combined approaches may involve simultaneous therapies [such as LR with ablation (ABL) or ABL with chemoembolisation] or sequential treatment strategies[6].
In oncology, the concept of conversion may initially seem straightforward; however, in the context of HCC, it typically refers to transforming an unresectable tumour into a resectable one through specific interventions. Conversely, neoadjuvant therapy encompasses treatments designed to optimise surgical conditions and enhance postoperative outcomes in patients with tumours initially deemed resectable. Meanwhile, adjuvant therapy includes any intervention administered following curative treatment (such as surgery or ablation) to minimise the risk of recurrence by targeting residual microscopic disease.
However, applying these concepts to patients with HCC is still controversial. First, several treatment strategies are available for HCC. Some of these, such as liver transplantation (LT), LR, and percutaneous or surgical ABL, are potentially curative. Other therapies, including intra-arterial treatments (IAT) and STs, can extend survival in HCC patients[7]. Consequently, the aim of conversion in patients with HCC cannot be limited only to “resectability” but, more in general, should be extended to “suitability for curative treatments” (i.e., LT and ABL). Second, most importantly, “suitable for curative therapies” is highly heterogeneous since it refers to two main clinical and pathophysiological fields: oncological/biological and technical/functional. On the one hand, oncological (biological) unresectability refers to the initial tumour stage or its inherent biological aggressiveness. From this perspective, a patient with HCC can be considered unsuitable for curative-intent therapy because the tumour is judged to have an unacceptably high risk of post-treatment recurrence[8,9].
On the other hand, technical (functional) unresectability mainly refers to the safety profile of a potentially curative-intent therapy[10]. From this perspective, a patient with HCC is technically unsuitable for curative treatment when the intrinsic postoperative risks of the procedure are considered too high compared to the potential treatment benefits. Although there is general agreement on the definition of insufficient remnant liver volume in patients with or without cirrhosis, surgical skills and availability of techniques to reduce the extent of LR and induce hypertrophy of remnant liver are heterogeneous, inevitably affecting surgical indications[11].
Nevertheless, both these definitions - oncologically and technically unsuitable for curative therapy - are characterised by a large grey area of uncertainty. For example, some clinicians consider a technically resectable multinodular HCC oncologically unresectable, while others can regard the same tumour as oncologically resectable[12]. Based on the definitions provided above, any preoperative therapy can fall into one of two categories. It is defined as a conversion intent therapy when the patient is oncologically unresectable but technically resectable. Conversely, it is considered neoadjuvant intent when the patient is both oncologically and technically resectable.
Third, the availability of effective systemic treatments represents a fundamental prerequisite of conversion or adjuvant approaches. This process relies on modifying the tumour’s biology to address all tumour cells, including potential microscopic metastases[13]. However, new systemic treatments only offered notable responses in patients with HCC in recent years, showing improved response rates [objective response rate (ORR)] of 25%-30% with Lenvatinib or immunotherapies, compared to earlier treatments such as sorafenib, which had 10%-11% ORRs[14].
Fourth, significant heterogeneity exists in assessing radiological treatment response, depending on the criteria used to evaluate the response to therapy for solid tumours (RECIST). The RECIST 1.1 criteria are appropriate for assessing an absolute reduction in tumour burden, while the modified RECIST (mRECIST) criteria facilitate the evaluation of a decrease in contrast-enhancing tumours[13].
For these reasons, which create significant uncertainty in defining sequential treatment strategies for patients with HCC, the concept of the “converse therapeutic hierarchy” has been recently introduced[7,14].
Traditional treatment algorithms for HCC, such as the Barcelona Clinic Liver Cancer (BCLC) system, are based on a static linear model. In this model, available options are arranged from curative to palliative intent according to tumour stage, liver function, and performance status. In contrast, the converse therapeutic hierarchy represents a dynamic, response-guided framework that allows therapeutic intent to evolve bidirectionally. Within this model, treatments are not terminal endpoints but potential gateways to higher-order curative strategies, depending on biological response and patient reassessment. The converse therapeutic hierarchy therefore complements rather than replaces the ordinal hierarchy: it introduces the concept of upward mobility across treatment tiers, guided by real-time evaluation of tumour biology, liver function, and multidisciplinary judgement.
This “umbrella concept” includes all the strategies aimed at increasing feasibility (conversion approach) or the effectiveness (neoadjuvant or adjuvant approaches) of radical treatments for HCC. Although the concept of conversion to both LT and LR using locoregional therapies has been recently endorsed by the guidelines of the European Association for the Study of the Liver (EASL)[15], the same guidelines underline the scarcity of solid evidence about conversion after ST or neoadjuvant/adjuvant strategies. The concept of the converse therapeutic hierarchy is illustrated in Figure 1. The left axis represents the conventional sequence of therapies, from curative to palliative. The right axis shows the converse pathway, indicating the potential to return toward curative intent after an effective response. The horizontal dimension links these therapeutic directions within a multiparametric decision-making process that incorporates oncologic, functional, and technical factors[7]. From this perspective, a converse therapeutic hierarchy means a flexible container that needs to be filled with new evidence in the context of the complex scenario of sequential treatment strategies.
Figure 1. The concept of “multiparametric therapeutic hierarchy” is based on three main pillars: (a) the ordinal therapeutic hierarchy (left vertical axis); (b) the multiparametric multidisciplinary expert decision (horizontal axis); (c) the converse therapeutic hierarchy (right axis). Adapted from Ref.[14]. © 2023 Elsevier. Reprinted/adapted with permission from Elsevier. All rights reserved. PS: Performance status; AFP: alpha-fetoprotein; SD: stable disease; PD: progressive disease; LDLT: living donor liver transplantation; DCD: donation after circulatory death; WT: waiting time; CRPH: clinically relevant portal hypertension; MELD: Model for End-stage Liver Disease; TACE: transarterial chemoembolisation; PVT: portal vein thrombosis.
TREATMENT MODALITIES IN CONVERSION THERAPIES: STS
Current scenario
Over the past few years, the treatment landscape of unresectable HCC has undergone a paradigm shift, with immune-based strategies progressively replacing multikinase inhibitors [tyrosine kinase inhibitors (TKIs)] as the frontline standard. Initial experiences with single-agent checkpoint inhibitors targeting programmed death-1 (PD-1) or programmed death ligand 1 (PD-L1) revealed modest antitumour activity, prompting the development of combinatory regimens to enhance therapeutic efficacy. ORR is one of the most relevant endpoints used to assess these new treatment combinations. It reflects both tumour shrinkage and the potential for durable clinical benefit.
Three major categories of combination strategies have been investigated in phase 3 trials: immune checkpoint inhibitors (ICIs) paired with anti-vascular endothelial growth factor (VEGF) antibodies, dual immune checkpoint blockade, and regimens combining ICIs with TKIs[16]. All these approaches have shown improvements over sorafenib, the long-standing standard of care, and several have achieved regulatory approval across different regions [Table 1].
Phase 3 trials evaluating combination STs in advanced HCC
| Trial | Regimen | Comparator | Population (n) | ORR (%) | Median OS (months) | Comments |
| IMbrave150[17,18] | Atezolizumab + Bevacizumab (anti-PD-L1 + anti-VEGF) | Sorafenib | 501 | 30 vs. 11 | 19.2 vs. 13.4 | Established the first immune combination standard of care |
| HIMALAYA (STRIDE)[19,20] | Durvalumab + Tremelimumab (anti-PD-L1 + anti-CTLA-4) | Sorafenib | 782 | 20.1 vs. 5.1 | 16.4 vs. 13.8 | 20% of patients alive at 5 years |
| CheckMate-9DW[21] | Nivolumab + Ipilimumab (anti-PD-1 + anti-CTLA-4) | Sorafenib or Lenvatinib | 668 | 36 vs. 15 | 23.7 vs. 20.6 | In the comparator arm 85% patients received lenvatinib; requires close toxicity management |
| CARES-310[22] | Camrelizumab + Rivoceranib (anti-PD-1 + TKI) | Sorafenib | 543 | 25 vs. 6 | 22.1 vs. 15.2 | > 80% patients with HBV-related HCC |
The atezolizumab–bevacizumab combination (anti-PD-L1 + anti-VEGF) was the first regimen to demonstrate a clinically meaningful survival advantage, establishing a new benchmark for first-line therapy. Its success validated the concept of immunotherapy–angiogenesis synergy, where VEGF blockade enhances immune infiltration and activity within the tumour microenvironment[17,18].
Dual checkpoint inhibition represented the next conceptual advance. The HIMALAYA trial (NCT03298451)[19], using a single tremelimumab [anti-cytotoxic T-lymphocyte antigen-4 (CTLA-4)] priming dose followed by durvalumab (anti-PD-L1) maintenance, produced durable responses and unprecedented 5-year survival for approximately one in five patients[20]. More recently, the CheckMate-9DW trial (NCT04039607) with nivolumab plus ipilimumab reported the highest ORR (36%) observed in any phase 3 HCC trial[21]. These findings confirm that the simultaneous PD-(L)1 and CTLA-4 blockade can induce deep, long-lasting tumour regression in a subset of patients.
Parallel to these advances, combinations of ICIs with TKIs have also been pursued. Among these, the CARES-310 trial (NCT03764293) stands out, evaluating camrelizumab (anti-PD-1) with rivoceranib [a VEGF receptor-2 (VEGFR2)-selective TKI][22]. In this phase 3 study, the combination achieved an ORR of 25%, surpassing the 6% response rate observed in the sorafenib arm[22].
These combination strategies represent biologically synergistic regimens capable of achieving meaningful tumour regression and, in selected cases, enabling curative conversion. At present, no head-to-head trials directly compare these regimens, and apparent differences in efficacy between studies should not be interpreted through cross-trial comparisons. In current practice, treatment choice is largely influenced by comorbidities, physician experience, and local drug availability rather than by objective biological criteria, underscoring the need for a personalised and evidence-based selection framework.
STs in conversion
The emergence of combinations that elicit deep and sustained responses in a substantial subset of patients offers the rationale for tailoring treatment based on patient-specific goals, including downstaging and downsizing.
Real-world studies have begun to highlight the potential of ST as a bridge to resection, ablation, or embolisation techniques. A proof-of-concept study by Kudo et al. reported that 35% of patients with intermediate-stage, transarterial chemoembolisation (TACE)-unsuitable HCC achieved either clinical or pathological complete response (CR) after treatment with Atezo/Bev followed by a curative procedure[23]. Among those achieving CR, two-thirds remained drug-free, with no evidence of recurrence during follow-up, emphasising the possibly durable nature of conversion. Additional support comes from retrospective analyses and registry-based data. In a Japanese multicentre cohort of 244 patients treated with either Atezo/Bev or lenvatinib, 4.9% underwent conversion therapy, most of whom had intermediate-stage disease and well-compensated liver function[24].
These data suggest that the conversion population remains a minority, but its identification may provide substantial clinical benefit. Prospective observational studies have begun to identify predictive factors for successful conversion, including low tumour burden, early radiological response, good liver function, and immunologic biomarkers, such as a low neutrophil-to-lymphocyte ratio[25].
Aggressive multimodal approaches are also emerging. Triple-combination regimens involving ICIs, TKIs, and either TACE or hepatic arterial infusion chemotherapy (HAIC) have been explored in patients with bulky or multifocal disease. A recent meta-analysis included 741 patients treated with triple-combination regimens combining ICIs, TKIs, and either TACE or HAIC. This study reported a pooled conversion rate of 35.9 percent and significant improvements in ORR, progression-free survival (PFS), and overall survival (OS) compared with dual-therapy combinations[26]. It should be emphasised, however, that these impressive conversion rates largely derive from highly selected patient populations, typically including individuals with favourable baseline characteristics. Most studies were retrospective and single-centre, often conducted in East Asian cohorts where multidisciplinary infrastructure and local expertise in combined interventional and systemic approaches are particularly advanced. As a result, these findings may not be directly generalizable to broader, real-world populations, and the potential for selection and publication bias should be acknowledged. Nonetheless, the consistency of positive signals across studies reinforces the conceptual validity of multimodal conversion strategies and highlights the urgent need for prospective, standardised definitions of eligibility and response.
Despite the promise of this approach, several challenges persist. One major concern is timing. Prolonged treatment may allow for deeper responses but carries the risk of immune escape or disease progression. Conversely, intervening too early may forsake the full benefit of continued systemic control. Some authors advocate an “early-switch” strategy, transitioning to surgery or ablation once partial or complete responses are documented radiographically[27].
Another unresolved issue is patient selection. Retrospective studies suggest that specific morphologic patterns are more amenable to shrinkage and downstaging[25]. Other predictive factors include early tumour shrinkage on imaging, low alpha-fetoprotein levels at baseline, and preserved hepatic function. However, prospective validation and standardised conversion criteria are lacking, and real-world registries remain sparse. Harmonised response metrics and decision-making frameworks will be essential to advance this field.
In conclusion, STs function not only as a last line of defence but also as an engine of upward therapeutic mobility. This dynamic, bidirectional use of ST reflects a more nuanced understanding of cancer as an evolving biological entity rather than a static stage.
Recent clinical guidelines are beginning to reflect this shift. The 2022 Chinese national guidelines explicitly endorse multimodal conversion strategies - including Atezo/Bev combined with HAIC or TACE - for technically unresectable but potentially convertible patients [China Liver Cancer (CNLC) stage IIa-IIIa][28]. Japanese and European consensus statements are likewise evolving toward a multidisciplinary, longitudinal evaluation of patients, thus accommodating therapeutic conversion, although alignment between staging systems and practical definitions remains a work in progress[15,29,30].
THERAPEUTIC MODALITIES IN CONVERSION THERAPY: TRANSARTERIAL TREATMENTS
Transarterial techniques
Transarterial therapies exploit the distinct blood supply of HCC compared with normal liver tissue: normal liver is primarily supplied by the portal vein, whereas HCC is mainly supplied by the hepatic artery. The distinct arterial supply to tumours allows for selective delivery of therapeutic agents, minimising damage to healthy liver tissue, potentially reducing side effects and preserving liver function. Intra-arterial strategies include:
• Transarterial embolisation - TAE: delivery of bland embolic agents
• Transarterial chemoembolisation - TACE: delivery of cytotoxic drug plus embolic agent(s)
• Hepatic arterial infusion chemotherapy - HAIC: intra-arterial delivery of chemotherapy
• Transarterial radioembolisation - TARE: delivery of radioactive particles
TACE is generally favoured over TAE in HCC treatment, combining targeted chemotherapy and embolisation techniques. For patients with multinodular HCC, leading guidelines consistently recommend TACE as the primary treatment option. While doxorubicin remains the primary chemotherapeutic agent in TACE procedures, interventional radiologists may also utilise alternatives such as epirubicin, idarubicin, cisplatin, mitomycin C, or 5-fluorouracil. Two main TACE approaches exist: conventional TACE (cTACE) and drug-eluting bead TACE (DEB-TACE). The traditional cTACE method uses a mixture of Lipiodol and chemotherapy drugs, followed by gel-foam embolisation. In contrast, DEB-TACE employs specialised particles that provide a controlled release of chemotherapy drugs into the tumour.
Although DEB-TACE demonstrates reduced systemic toxicity and enhanced safety compared to cTACE, studies have not shown significant improvements in local tumour control. Consequently, both techniques remain widely used, with the choice between them typically depending on the physician’s experience and the availability of embolisation products at different medical centres[31].
HAIC is a locoregional therapy that directly delivers chemotherapeutic agents into tumour-associated arterial branches at increased local drug concentrations. Because of a greater first-pass effect in the liver, HAIC provides stronger antitumour efficacy and lower systemic toxicity than ST[32]. HAIC may be performed via pump implant or repeated catheterisation and is widely offered in Asian countries, where it has been endorsed in clinical guidelines before the advent of immune-checkpoint inhibitors and newer TKIs[33].
TARE is a local therapy that delivers radioactive microspheres containing either Yttrium-90 (Y90) or Holmium-166 (166Ho), through the hepatic artery to treat liver tumours. These microspheres lodge themselves at the arteriolar level, where they deliver localised radiation - beta radiation from Y90 or both beta and gamma radiation from 166Ho[34,35]. This treatment modality is designed to create a minimal embolic effect while preserving blood flow to the treated liver segments, maximising the tumour-destroying effects of radiation.
Before performing TARE, proper patient evaluation is essential and includes a diagnostic arteriogram with technetium-99m macroaggregated albumin (MAA). This preliminary step is crucial for identifying potential non-target areas and calculating the lung shunt fraction. Treatment exclusion criteria include significant lung shunting (exceeding 25 Gray in a single treatment or 30 Gray cumulatively across multiple treatments) and the presence of gastric or duodenal branches in the infusion zone that cannot be successfully embolised[36].
TAE/TACE and HAIC as conversion treatments
Evidence from both prospective and retrospective studies has explored the potential of transarterial as a conversion or downstaging strategy.
In Eastern countries, where there is a high prevalence of HCC in non-cirrhotic hepatitis B virus (HBV) patients and LT is less available, TACE and HAIC are widely used to convert patients to surgery. In recent years, many reports have explored the combination of TACE/HAIC, ICIs, and TKIs (especially lenvatinib) to convert patients to surgery. Conversion rates widely vary in different studies, ranging from 13% to 40%[37-39].
Recently, the results of a randomised clinical trial (RCT) regarding a combination of ICIs and TACE in the intermediate stage showed promise in terms of LR-free survival[40]. Similarly, better LR-free survival has been demonstrated for the regimen lenvatinib + ICIs + TACE vs. TACE alone[41]. Data regarding potential conversion to surgery are not available in these preliminary reports; however, in an observational study with propensity score matching, a cohort of 32 patients with initially unresectable HCC who were converted to resection through TACE, lenvatinib, and camrelizumab had the same OS as patients with initially resectable HCC who underwent surgery. The safety profiles of the two groups were similar, and, interestingly, the conversion surgery group had a lower incidence of microvascular invasion than the initially resected group (3.1% vs. 50.4%, P < 0.001)[42].
TACE can effectively downstage tumours in a significant proportion of patients[43]. Patients who are successfully downstaged to meet transplant criteria have better post-transplant survival than those who are not transplanted[44]. TACE is the most common form of locoregional therapy, comprising 55.6% and 63.0% of first-downstaging procedures in all comers and United Network for Organ Sharing Down-Staging (UNOS-DS), respectively, as shown by a recent data analysis of the Multicenter Evaluation of Reduction in Tumor Size before Liver Transplantation (MERITS-LT) consortium[45]. In particular, in a study where data about DEB-TACE as the only locoregional treatment to downstage tumours initially exceeding the Milan criteria and bridging patients within Milan criteria were collected, post-transplant survival and HCC recurrence-free probability at five years were similar in the two cohorts of patients[46].
Taken together, these findings highlight the complementary role of transarterial treatments within the broader concept of conversion therapy. The wide variability across studies underscores the need for harmonised response criteria and prospective validation in well-defined patient populations.
TARE as conversion treatment
The evolution of TARE has shifted its use from palliative treatment of advanced disease to a selective approach targeting specific liver segments or lesions, broadening its therapeutic scope beyond palliation.
Radiation lobectomy (RL) was developed following observations that TARE treatment of a single liver lobe not only targets tumours but also triggers an atrophy-hypertrophy response: the treated, tumour-bearing lobe shrinks, while the untreated portion of the liver experiences compensatory growth. This effect can increase the future liver remnant (FLR), potentially enabling surgical resection in previously inoperable cases.
For patients with limited disease, radiation segmentectomy (RS) offers a more focused approach by delivering high-dose radiation to a restricted area, typically encompassing one or two Couinaud segments. This technique aims to achieve ablative effects while minimising impact on healthy liver tissue. Building on these advances, modified RL combines the targeted, high-dose approach of RS with the FLR-promoting benefits of traditional RL, creating an integrated treatment strategy to enhance patient outcomes[34].
Since 2009, evidence has accumulated from two complementary directions: dedicated studies have explored TARE as a conversion therapy leading to surgery or transplantation, while comparative trials with TACE have defined their relative efficacy and situational advantages within the treatment hierarchy [Table 2][47-54].
Key studies enploring TARE as a conversion therapy or downstaging modality in HCC
| Study/year | Design | Population (n) | Treatment | Conversion/downstaging success (%) | OS (%) | Key findings/notes |
| Foundational studies | ||||||
| Gabr et al.[47] (2018) | Retrospective, single-centre | 31 | TARE before resection | 100 | 86 (3 years) | First evidence of feasibility of resection after TARE; contralateral hypertrophy enabled surgery |
| Salem et al. (LEGACY trial)[48] (2021) | Prospective, multicenter (single-nodule HCC > 5 cm) | 162 | TARE | 6.8 (resection); 22.2 (transplant) | 92.8 (3 years) | Durable tumour control; subgroup successfully rescued to surgery |
| Aliseda et al.[49] (2023) | Retrospective | 26 | TARE before resection | 100 | 84.6 (3 years), 76.9 (5 years), 57.0 (10 years) | Outcome similar to that of resected Milan-in HCC |
| Tzedakis et al.[50] (2023) | Prospective, single centre (single-nodule HCC > 5 cm) | 72 | TARE before surgery vs. upfront surgery | 27.7 | 83 vs. 94 (1 year), 60 vs. 86 (3 years), 47 vs. 55 (5 years) | Similar OS between groups; better OS for TARE after matching; Comparable DFS |
| Comparative TARE vs. TACE studies | ||||||
| Lewandowski et al.[51] (2009) | Retrospective, single-centre | 86 | TARE-Y90 vs TACE (downstaging before LT) | 58 (TARE) vs. 31 (TACE) | Median OS: 35.7 vs. 18.7 months (P = 0.18) | Downstaging to UNOS T2 criteria: 58% vs. 31% Event-free survival: 17.7 vs. 7.1 months, P = 0.0017 |
| Mehta et al. (MERITS-LT consortium)[52] (2021) | Multicentre, prospective | 262 | TARE-Y90 vs. TACE | > 80 (both) | - | Similar overall success rate; Complete pathological response: 30.8% vs. 20.5% Microvascular invasion:7.7% vs. 20.5% |
| Studies with ITT-approach for outcome | ||||||
| Berardi et al.[53] (2025) | Prospective | 73 | TARE (downstaging before LT) | 30 | - | Unfavourable baseline characteristics |
| Regnault et al.[54] (2024) | Retrospective | 46 | TARE (downstaging before LT) | 39 | - | Unfavourable baseline characteristics |
Taken together, these studies provided consistent evidence that TARE can achieve not only effective local control but also oncologic conversion in selected patients. When adequate dosimetry is applied, the procedure combines sustained tumour irradiation with preservation of hepatic function, creating the physiological and volumetric conditions required for curative resection or transplantation. Reported conversion rates varied widely, reflecting differences in study design, patient selection, and disease stage. In the transplant setting, multicentre studies have confirmed that both TACE and TARE can successfully downstage advanced tumours to transplant eligibility with comparable efficacy[46]. However, TARE consistently produces higher rates of complete necrosis and lower microvascular invasion, especially in patients with vascular involvement or large tumour burden. Long-term outcomes after TARE-assisted surgery are remarkably similar to those achieved with upfront resection in early HCC[55]. These findings support the integration of TARE into multidisciplinary conversion strategies aimed at bridging biological response with curative intent.
The recently published EASL guidelines recommend that the choice of downstaging strategy before LT should depend on patient and tumour characteristics. No single locoregional approach is currently preferred over the other[15]. TARE, by delivering radiation with minimal embolic impact, is particularly suited for patients with unilobar or macrovascular disease and for those being considered for downstaging or bridging to curative therapies. Instead, TACE remains the standard of care for patients with intermediate-stage, multinodular disease and preserved portal flow, offering well-established efficacy and widespread availability. In practice, therefore, TACE and TARE should be viewed not as competing modalities but as complementary tools within a response-guided continuum, their selection driven by tumour morphology, vascular anatomy, and hepatic functional reserve.
The question of whether patients with macrovascular invasion can be converted to resection or transplantation remains open. A multicentre study published in 2020 reported promising results in a cohort of 30 patients downstaged with TACE, TARE and surgery and with a radiological absence of remaining macrovascular invasion. Those patients had a 5-year survival rate of 60% and a recurrence rate of 11%[56]. In a population of 18 patients with large, multifocal or portal vein-invading HCC (15/18 patients had tumoural portal vein thrombosis), complete pathological necrosis was achieved after TARE-Y90 in a significant number of tumours (36%). Of note, all complete pathological necrosis occurred in HCC invading portal vein branches, suggesting that portal vein tumour thrombosis can be completely cured by TARE using personalised dosimetry[57]. Higher rates of complete pathological necrosis (47%-68%) were reported in pathological series after RS, but only early HCCs were involved[58].
THERAPEUTIC MODALITIES IN CONVERSION THERAPY: PERCUTANEOUS PROCEDURES
Overview of percutaneous therapies
Percutaneous interventions (PIs) have become firmly established in the therapeutic scenario for HCC, particularly in early-stage disease. These procedures aim to induce localised tumour destruction via thermal or non-thermal mechanisms, offering a minimally invasive approach with potential curative intent in selected patients[59,60].
Radiofrequency ablation (RFA) and microwave ablation (MWA) are the most extensively utilised thermal techniques. Both methods are designed to achieve coagulative necrosis, and their efficacy depends heavily on appropriate tumour and peritumoural tissue coverage[59]. A safety margin of at least 0.5-1 cm beyond the visible tumour is generally advised to reduce the risk of local recurrence and address possible microscopic disease. RFA delivers high-frequency alternating current (375-480 kHz) to produce tissue heat, typically around
Beyond thermal modalities, irreversible electroporation (IRE) has gained attention for its ability to preserve extracellular structures while triggering apoptosis via high-voltage electrical pulses. This characteristic makes it especially advantageous near bile ducts or blood vessels, where thermal ablation carries greater risk[63,64]. Cryoablation (CA), which employs freezing and thawing cycles, offers another alternative. Though CA may promote an immunogenic microenvironment and has demonstrated feasibility in HCC, it is limited by its complexity and potential for severe systemic adverse events, such as cryoshock[60]. Although not widely adopted, high-intensity focused ultrasound (HIFU) has shown feasibility in small tumours and is sometimes considered within combined treatment protocols[65].
In general, PIs are considered safe. Serious complications are infrequent, occurring in roughly 5% of patients, and include liver abscesses, hemoperitoneum, pneumothorax, and biliary injury. Minor post-procedural effects such as fever, pain, or nausea are more common but typically self-limiting[60-66]. Notably, these interventions can be repeated in the case of incomplete response or recurrence, and laparoscopic thermal ablation (LTA) is an alternative when percutaneous access is unfeasible. LTA has been shown to provide outcomes comparable to standard PIs and superior to TACE, occupying an intermediate position in the treatment hierarchy[67].
Integration into combined strategies
PIs are incorporated into broader treatment strategies, particularly in patients with intermediate or unresectable HCC. Their use alongside locoregional or STs reflects a shift toward multimodal care tailored to tumour burden, liver function, and patient fitness.
A well-documented approach involves the combination of TACE and MWA, which is particularly effective in multifocal disease or tumours measuring 5-10 cm. This combination has demonstrated promising survival outcomes, with 1- and 3-year OS rates often exceeding 80% and 45%, respectively, in retrospective and meta-analytic studies[68].
The advent of immunotherapy has further underscored the relevance of PIs in systemic treatment combinations. RFA is known to modulate the tumour immune microenvironment by releasing antigens and proinflammatory cytokines into circulation. This process can prime the immune system, creating a more favourable context for checkpoint inhibition. Data suggest that these immune-enhancing effects are most pronounced in partial or incomplete ablation areas, reinforcing the rationale for sequencing ablation before or during ICI administration[69]. From a clinical perspective, some studies have explored ST induction followed by local ablation to consolidate response. The underlying principle is that modest reductions in tumour burden achieved through systemic agents can improve the efficacy and feasibility of local consolidation, particularly when liver function preservation is a concern.
Despite encouraging findings, the optimal integration of PIs within combined regimens remains under investigation. Questions persist regarding ideal sequencing, duration, and agent selection, as well as identifying biomarkers to guide patient selection. Nevertheless, the flexibility and repeatability of PIs make them attractive tools in comprehensive treatment planning, particularly when coordinated within a multidisciplinary team[68].
Role in therapeutic conversion
Within the conceptual model of the converse therapeutic hierarchy, PIs are increasingly recognised not only as definitive options for early-stage HCC but also as enabling tools that facilitate transitions toward other hierarchically superior, curative interventions[69].
One of the most well-known applications of PIs in this context is their use as bridging therapies for LT. Patients awaiting LT are at risk of tumour progression, which may exclude them from eligibility based on criteria such as Milan or University of California San Francisco (UCSF) guidelines[70,71]. By maintaining disease stability, thermal ablation can delay or prevent transplant ineligibility, especially in regions with limited organ availability and prolonged waiting times.
In addition to bridging, PIs play a central role in downstaging protocols. Patients with tumour burden exceeding conventional transplant criteria may undergo ablative treatment (often combined with TACE) to achieve radiologic and biological regression, requalifying them for LT. Post-transplant outcomes in such patients are increasingly reported as comparable to those treated within criteria from the outset, reinforcing the value of this adaptive strategy[72,73].
Beyond transplantation, PIs contribute to surgical conversion, transforming borderline or unresectable lesions into operable diseases[74]. For instance, in cases where tumours are situated near critical structures or involve high surgical risk, initial ablation can debulk or devascularise the lesion, paving the way for delayed resection. Similarly, in patients with impaired hepatic reserve, PIs may allow for local disease control without compromising liver function, creating time for hepatic recovery or hypertrophy through associated procedures such as portal vein embolisation (PVE).
Sequential therapeutic strategies also illustrate the bidirectional nature of PI within the therapeutic hierarchy[69]. For example, a patient may receive initial TACE to reduce tumour size, followed by PI for residual nodules, ultimately achieving curative conversion. Conversely, a patient initially treated with PI who develops recurrence may be re-evaluated for ST, resection, or even transplantation, depending on disease evolution[75].
OTHER THERAPEUTIC MODALITIES IN CONVERSION THERAPY
Overview
The concept of conversion therapy in HCC extends beyond tumour treatment. In patients initially considered ineligible for curative surgery, a wide spectrum of interventional and medical strategies can be employed to modify either anatomical or functional constraints. These include techniques designed to increase the FLR, thereby improving the technical feasibility of resection, as well as interventions aimed at stabilising or reversing the underlying liver disease to enhance hepatic functional reserve. Together, these complementary strategies embody the “converse” dimension of the therapeutic hierarchy, where treatment intent evolves dynamically according to both tumour and host factors.
Strategies to increase FLR
In patients with liver malignancies, achieving an adequate FLR is essential to maintain postoperative liver function and to prevent post-hepatectomy liver failure (PHLF). The minimum required FLR varies based on the patient’s baseline liver function and other factors. A FLR of 20% is generally adequate in patients with normal liver function. However, the FLR requirement is higher for patients who are exposed to systemic chemotherapy (FLR, > 30%) or have underlying chronic liver disease (FLR, > 40%)[76]. Therefore, assessing the perioperative risk factors associated with the development of PHLF is crucial for optimising patient selection for hepatectomy and for applying strategies to prevent the occurrence of PHLF.
PVE
PVE can increase liver volume over a defined period, making surgery feasible. Studies have indicated that the success rate of conversion with this method ranges from 60% to 80%, with a complication rate of 10% to 20%[77-81]. PVE, first introduced in 1986 by the Osaka City University group in Japan, remains a primary method for inducing hypertrophy of the FLR by occluding the portal vein to promote atrophy in the embolised liver. This process is possible due to the dual blood supply from the hepatic artery and portal vein, which allows for safe embolisation without ischemia of the remaining healthy liver tissue[82]. Additionally, TACE is often used in combination with PVE to promote FLR hypertrophy further and to control tumour progression during the waiting period for hepatectomy. Studies suggest that combining PVE with TACE results in a significantly greater increase in the percentage of liver volume compared to PVE alone, although the overall morbidity rates were similar between the two groups. TACE followed by PVE is typically performed sequentially, with TACE done first, followed by PVE 2-3 weeks later. However, some clinicians have recommended the opposite sequence - PVE followed by TACE - to avoid the risk of liver congestion due to extensive arterial obstruction secondary to TACE[83-85]. Furthermore, PVE also offers additional oncological benefits, such as preventing intraportal metastases, limiting the portal vein tumour thrombosis progression, and enhancing the antitumour effects of TACE[80,82,86]. Contraindications for PVE are severe portal hypertension, Vp3-Vp4 tumour thrombus, and extensive tumour metastasis[80].
Combined PVE and hepatic vein embolisation
Combined PVE and hepatic vein embolisation (HPVE) has gained attention as an effective strategy to promote FLR hypertrophy, particularly in patients with colorectal liver metastases and perihilar cancers[87,88]. The benefit of HPVE has been highlighted, as it ensures a more controlled approach and avoids complications such as liver ischemia[89,90]. A recent systematic review of HPVE has shown a significant increase in FLR volume (33%-63.3%) and liver function (64%) following the procedure, with no reported mortality or major morbidity[89]. However, HPVE requires careful consideration in cirrhotic patients, as the procedure can increase the risk of portal hypertension-related complications such as ascites or variceal bleeding. As a result, sequential HPVE is recommended only in patients with insufficient FLR after PVE and with compensated liver disease.
Associated liver partition and portal vein ligation for staged hepatectomy
Associated liver partition and portal vein ligation for staged hepatectomy (ALPPS) is a relatively novel surgical procedure aimed at achieving rapid FLR hypertrophy in patients with insufficient liver volume for resection. It involves partitioning the liver and ligating the portal vein, with a staged hepatectomy performed after a period of hypertrophy. ALPPS is particularly beneficial in patients with cirrhosis or advanced liver disease and has been recommended as a “rescue” strategy in patients with inadequate FLR after PVE or RL. It has been demonstrated that ALPPS results in more rapid hypertrophy and a shorter waiting time to hepatectomy compared to PVE, but it is associated with a higher risk of morbidity[91-93].
Cell-based therapies
In addition to surgical treatment, cell-based therapy holds significant promise for promoting liver regeneration. Advances in stem cell bioengineering and organoid generation have accelerated the development of cell therapies for liver injury[94]. Stem cells can proliferate and differentiate into mature cells, facilitating tissue or organ restoration and repair in vivo without the risk of immune rejection[95]. Various types of stem cells, including induced pluripotent stem cells (iPSCs), mesenchymal stem cells (MSCs), embryonic stem cells (ESCs), endothelial progenitor cells (EPCs), and liver progenitor cells (LPCs), have been shown to differentiate into hepatocytes or hepatocyte-like cells in preclinical and clinical studies on liver diseases[96]. However, while some studies suggested that liver function may improve with stem cell therapy, the overall efficacy remains controversial[97].
Control of underlying liver disease
Factors predicting the recurrence of HCC after resection have been extensively studied[98-101]. The influence of underlying liver disease on long-term survival after HCC resection has also been noted[102,103]. Shirabe et al. demonstrated that patients with better liver function, as assessed by preoperative indocyanine green retention, had a higher likelihood of surviving for 10 years after surgery than those with poor liver function[104]. Published data obtained from large meta-analyses have shown that patients with positive serology for HBV and hepatitis C virus (HCV) undergoing resection for HCC had a poor prognosis compared to patients with negative serology[105]. Multiple studies have demonstrated that HCC recurrence rates were similar between patients with and without cirrhosis up to five years after resection. However, after this period, patients with cirrhosis continued to experience mortality due to HCC, while those without cirrhosis were less likely to develop recurrence[106-108]. Two potential explanations account for the negative impact of existing fibrosis on long-term survival. First, advanced fibrosis and cirrhosis may act as a “field of cancerisation”, increasing the risk of developing metachronous HCC after resection. Second, patients with advanced fibrosis or cirrhosis may have an increased risk of death related to liver disease complications[109]. Consequently, addressing the aetiology of liver disease offers substantial prognostic benefits for patients with cirrhosis. Cirrhosis has long been considered an irreversible endpoint of chronic liver injury. However, accumulating evidence indicates that even established cirrhosis may undergo regression. Regression reflects not only a halt in disease progression but also the activation of biological mechanisms that promote collagen degradation and partial restoration of hepatic architecture[110]. Although complete architectural normalisation is rare, partial reversal can meaningfully enhance hepatic reserve, lower portal pressure, and improve tolerance to major surgery or locoregional interventions. The regression of cirrhosis exemplifies that conversion therapy need not be confined to tumour shrinkage alone. Rather, modifying the biological substrate of liver disease itself represents a parallel and equally critical axis of therapeutic progress within the converse therapeutic hierarchy.
HBV infection
Nucleos(t)ide analogues (NAs) have revolutionised the management of HBV-related liver disease, offering significant benefits in terms of suppressing HBV levels, improving liver histology, and preventing disease progression[111-114]. Achieving viral suppression in patients with HBV-related cirrhosis provided improved OS, a reduced incidence of HCC, and an independent association with a lower risk of late recurrence following local curative-intent treatment for HCC[115]. Furthermore, the sustained suppression of HBV replication reduces the risk of progression to end-stage liver disease and the need for LT[115-118].
HCV infection
HCV infection has long been the leading cause of liver disease underlying HCC in the West and Japan[119,120]. The introduction of direct-acting antiviral (DAA) therapies, which effectively cure HCV, stands as one of the major successes of modern medicine. Prior to the introduction of effective antiviral therapies, HCV infection was a significant predictor of poor outcomes for patients with HCC, both in terms of surgical resection and LT[121]. Historically, the lower survival rate after HCC resection in patients with active HCV, compared to those with HBV, was attributed to increased mortality from both tumour- and non-tumour-related causes. The higher recurrence of HCC observed in HCV patients was primarily due to the development of de novo HCC, while non-tumour-related deaths were mainly a result of cirrhosis progression[122,123]. With the advent of DAA treatment, both outcomes have been improved[124-128]. Concerns were briefly raised in 2016 regarding the potential impact of HCV eradication on increasing the risk of HCC recurrence following resection or ablation[129-131]. However, subsequent prospective studies have not confirmed this alert[132]. Regardless, complete resolution of fibrosis is rare, and significant fibrosis remains a substantial risk factor for HCC. Consequently, patients with advanced fibrosis who are cured of HCV continue to face an ongoing risk of developing HCC and should be regularly screened for HCC[133-135].
Nonviral etiologies
Prolonged abstinence from alcohol abuse improves prognosis and reduces decompensation risk and mortality in alcohol-associated liver disease (ALD)[136]. Alcohol abstinence reduces the risk of HCC in patients with ALD cirrhosis, but this benefit is primarily observed in those without a history of decompensated liver disease[137].
The treatment of metabolic dysfunction-associated steatotic liver disease (MASLD), an increasing global cause of cirrhosis, largely relies on lifestyle modifications, bariatric surgery, and management of comorbidities[138]. Weight loss, dietary modifications, and increased physical activity have been shown to reduce the risk of fibrosis progression and, consequently, HCC development[139]. Bariatric surgery also reduces the risk of HCC by improving liver fibrosis but is contraindicated in patients with decompensated cirrhosis[140]. Recent studies have highlighted the potential role of statins as protective factors against HCC occurrence in patients with MASLD, but confirmation is needed[139,141,142].
PERIOPERATIVE STRATEGIES: ADJUVANT AND NEOADJUVANT IMMUNOTHERAPY
Adjuvant immunotherapy: rationale and current evidence
The concept of adjuvant therapy in oncology stems from the need to consolidate the efficacy of a curative treatment (typically surgical resection) by mitigating residual microscopic disease and minimising recurrence risk. While this principle has succeeded across several solid tumours, including breast and lung cancers, its application to HCC has historically encountered unique obstacles[143].
A defining feature of HCC is its frequent coexistence with underlying cirrhosis, which contributes independently to the risk of recurrence through de novo carcinogenesis. As a result, recurrence in HCC may arise from both microscopic dissemination and ongoing oncogenesis driven by liver disease, complicating the interpretation of what constitutes a meaningful benefit from adjuvant therapy[144].
Before the advent of ICIs, several efforts explored the adjuvant use of antiviral agents, vitamin K analogues, and STs such as sorafenib. Randomised trials involving interferon-α (IFN-α and IFN-α2b) showed no benefit in recurrence-free survival (RFS)[145,146]. Likewise, the STORM trial (NCT00692770), a landmark phase III study assessing adjuvant sorafenib after curative therapy, failed to demonstrate RFS or OS benefit[147].
The introduction of ICIs reignited interest in this field. Their mechanism of action and their generally tolerable safety profile make them theoretically well-suited for perioperative use. One of the earliest signs of efficacy came from a randomised trial evaluating adoptive cell therapy using cytokine-induced killer (CIK) cells, which share some common characteristics with ICIs. In that study, patients treated with adjuvant CIK therapy after curative treatment showed significantly prolonged RFS and cancer-specific survival, with durable benefits extending beyond nine years of follow-up[148].
A milestone in this setting was the IMbrave050 trial, which tested Atezo/Bev in patients with high-risk features following resection or ablation. This study demonstrated a modest but significant reduction in recurrence risk at one year and over the treatment course[149]. However, updated analyses revealed that the benefit in RFS may diminish over time, and no OS advantage has been established to date[150].
Additional exploratory evidence comes from a phase II trial of sintilimab (ChiCTR2000037655), conducted in a population of patients with microvascular invasion after curative resection. While the combination demonstrated a 46.6% reduction in risk of recurrence or death, the OS difference remained statistically inconclusive, although trending favourably[151].
Taken together, these findings suggest that while adjuvant ICI-based strategies hold promise, their definitive role in resectable HCC remains to be established.
Neoadjuvant immunotherapy: emerging approaches and challenges
Neoadjuvant immunotherapy has gained increasing traction, leveraging the immunological advantages of treating intact tumours before LR. This approach, successfully explored in other solid tumours, rests on the premise that exposing a biologically active tumour to immune-modulating agents may elicit a stronger, more sustained antitumour response[143].
By initiating treatment while the tumour is still present, ICIs may promote neoantigen release, intratumoural T-cell priming, and systemic immunity[143,152]. Unlike conversion therapy, where systemic agents are used to render inoperable tumours resectable, neoadjuvant strategies in HCC aim to target tumours that are already operable.
Two U.S.-based phase II trials (NCT03916627 and NCT03222076) laid the groundwork for this approach in HCC. In the first, patients received two cycles of cemiplimab, followed by surgery[153]; in the second, nivolumab with or without ipilimumab was administered preoperatively[154]. The observed major pathological response (MPR) rates were 20% and 33%, respectively, and were associated with increased immune cell infiltration. These data underscore the potential for MPR to serve as a biological indicator of treatment efficacy.
A pooled analysis by D’Alessio et al. recently reinforced this notion, showing that patients achieving MPR had significantly longer RFS than those who did not. The analysis proposed a threshold of ≥ 90% tumour regression in post-surgical specimens as an optimal predictor of outcome[155].
Through published studies, treatment-related surgical delays have been rare, with toxicity-related withdrawals virtually absent. These findings suggest that neoadjuvant ICI may be feasible and safe, although long-term validation in larger cohorts is required.
Perspective
The integration of perioperative immunotherapy into HCC management is still evolving. While RFS remains a commonly reported endpoint, its clinical relevance relative to OS continues to be debated. Whether a durable delay in recurrence is sufficient to justify therapy without OS gain remains unresolved.
From a strategic standpoint, biomarker-guided approaches, including pathological response and radiomic features, may help refine patient selection, increasing the likelihood of benefit. Moreover, ongoing comparative studies will be critical in determining whether monotherapy or combination regimens are preferable and whether neoadjuvant or adjuvant administration yields superior outcomes.
BARRIERS TO CONVERSION
Despite growing enthusiasm surrounding the concept of conversion therapy, biological, technical, systemic, and organisational barriers limit its widespread application in clinical practice.
The intrinsic heterogeneity of HCC is one of the most pervasive problems. The absence of predictive biomarkers complicates the identification of patients who are less likely to respond to systemic or locoregional therapy, limiting opportunities for downstaging or resection.
Logistical and organisational barriers also play a role. Conversion strategies often require tight coordination among hepatologists, surgeons, radiologists, and oncologists. However, the availability of multidisciplinary tumour boards and centralised expertise varies substantially across institutions and regions. Without a structured, dynamic reassessment framework, patients with early signs of response may miss the therapeutic window for radical intervention.
Finally, current evidence largely stems from retrospective series or early-phase studies, often with heterogeneous populations and inconsistent endpoints. The lack of standardisation in defining “conversion” and the absence of prospective registries limit the generalizability and reproducibility of reported outcomes. From the perspective of the converse therapeutic hierarchy, these barriers do not merely represent operational limitations but structural constraints that hinder vertical mobility within the treatment continuum. Biological heterogeneity and the lack of predictive biomarkers make it difficult to identify which patients are most likely to respond. This limitation often prevents timely progression from non-curative to curative intent. Technical barriers obstruct comparability across studies and make it difficult to translate individual responses into shared algorithms of therapeutic escalation. Finally, organisational barriers disrupt the feedback loop that is fundamental to the converse model. Without coordinated decision-making and real-time evaluation, the window for curative conversion may close before it is recognised. Collectively, these factors underscore the need for harmonised criteria, predictive tools, and structured care pathways to translate the promise of conversion therapy into broader clinical benefit.
Addressing these barriers is therefore essential to operationalise the converse hierarchy: it requires a shift from static, stage-based frameworks toward adaptive, response-guided care pathways supported by biomarkers, imaging analytics, and institutional infrastructure for multidisciplinary review.
CONCLUSIONS
The management of HCC is undergoing a rapid and profound transformation. The emerging concept of the converse therapeutic hierarchy provides a new interpretative framework in which treatments are no longer regarded as terminal endpoints but as potential gateways to curative intent. This paradigm reflects a dynamic and biologically driven view of HCC therapy, emphasising that both tumour control and liver function can evolve through multidisciplinary intervention.
The emergence of ST with high ORR and innovative locoregional treatments has opened the door to a therapeutic and cultural shift. Conversion therapy is now supported by data demonstrating that a subset of patients can achieve sufficient tumour regression to undergo LR, ABL, or LT. Furthermore, the perioperative use of immunotherapy is redefining the goals of therapy in patients initially deemed resectable, aiming to prolong disease-free survival and potentially reduce recurrence.
These advances are not without challenges. The absence of standardised criteria for defining conversion, the heterogeneity of available data, and the limited prospective validation all underscore the need for cautious optimism. Integrating advanced therapies into clinical workflows requires not only scientific innovation but also system-level organisation and institutional commitment.
Looking ahead, the translation of the converse therapeutic hierarchy into daily clinical practice will require not only conceptual endorsement but methodological innovation.
First, establishing standardised conversion criteria is crucial to transform “conversion” from a qualitative notion into a measurable outcome. This process should rely on consensus-building initiatives promoted by leading scientific societies integrating radiologic, biochemical, and functional endpoints. Second, biomarker validation will be fundamental to identify “conversion responders”. Prospective multicentre registries and nested biobanking studies can enable the integration of serum, imaging, and molecular biomarkers, linking dynamic biological changes with eligibility for curative interventions. Artificial intelligence–driven imaging analysis and multi-omics profiling could provide early predictors of response and fibrosis regression, helping to time the transition from systemic to local or surgical therapy. Third, adaptive platform trials represent the most suitable research design to test this model prospectively. In contrast to fixed-arm trials, they would allow real-time treatment reassignment based on predefined response thresholds - mirroring the flexibility of the converse hierarchy itself. Incorporating radiologic, functional, and molecular endpoints within shared master protocols would accelerate evidence generation and harmonise conversion criteria across centres. Finally, clinical implementation requires a structural rethinking of multidisciplinary management. A dynamic MDT should include a comprehensive range of specialists, ensuring that all therapeutic options, from systemic to curative, are continuously accessible within the same decision-making environment. Rather than following fixed schedules, the MDT should operate as a flexible, response-driven forum where treatment decisions are revisited whenever new clinical or radiologic information emerges. The adoption of structured checklists could help standardise discussions and ensure that, in patients showing meaningful tumour or functional response, the possibility of transitioning to therapies with curative intent is always considered.
By coordinating these efforts, the converse therapeutic hierarchy could move from a theoretical concept to a practical clinical model. This transformation would turn the treatment of HCC into a continuous, evidence-based process of therapeutic evolution.
DECLARATIONS
Acknowledgements
The authors would like to acknowledge the members of the Associazione Italiana per lo Studio del Fegato (AISF) HCC Special Interest Group for their valuable contributions: Mauro Viganò (ASST Papa Giovanni XXIII Bergamo, Italy); Giovanni Galati (Campus Bio-Medico University Rome, Italy); Marco Guarracino and Raffaella Tortora (Cardarelli Hospital Naples, Italy); Nicoletta De Matthaeis, Simone Famularo, Felice Giuliante, Luca Miele, and Francesca R. Ponziani (Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Italy); Rodolfo Sacco (Foggia University Hospital, Italy); Francesco G. Foschi (Hospital of Faenza, Italy); Luca Viganò (Humanitas University Milan, Italy); David J. Pinato (Imperial College London, UK); Salvatore Gruttadauria and Duilio Pagano (IS.ME.T.T. Palermo, Italy); Sherrie Bhoori and Carlo Sposito (Istituto Tumori Milano, Italy); Raffaele Cozzolongo (National Institute of Gastroenterology Saverio de Bellis Bari, Italy); Leonardo Centonze and Chiara Mazzarelli (Niguarda Ca’ Granda Hospital Milan, Italy); Daniele Nicolini (Polytechnic University of Marche, Italy); Quirino Lai and Fabio Melandro (Sapienza University of Rome, Italy); Paola Violi (University Hospital of Verona, Italy); Maria Rendina and Francesco D’Amico (University of Bari, Italy); Matteo Cescon, Fabio Piscaglia, Matteo Renzulli, Nicolò Brandi, Francesco Tovoli, and Franco Trevisani (University of Bologna, Italy); Edoardo G. Giannini, Giulia Pieri, and Maria Corina Platz Torres (University of Genoa, Italy); Massimo Iavarone and Angelo Sangiovanni (University of Milan, Italy); Fabrizio Romano (University of Milano Bicocca, Italy); Stefano Di Sandro (University of Modena and Reggio Emilia, Italy); Maria Guarino and Filomena Morisco (University of Naples Federico II, Italy); Patrizia Burra, Umberto Cillo, Fabio Farinati, Michele Finotti, Martina Gambato, Filippo Pelizzaro, Francesco P. Russo, and Alessandro Vitale (University of Padua, Italy); Giuseppe Cabibbo and Ciro Celsa (University of Palermo, Italy); Paola Carrai, Laura Crocetti, and Davide Ghinolfi (University of Pisa, Italy); Mario Masarone and Marcello Persico (University of Salerno, Italy); Ilaria Lenci, Tommaso M. Manzia, and Bruno Sensi (University of Tor Vergata Rome, Italy); Umberto Baccarani and Riccardo Pravisani (University of Udine, Italy); Andrea Casadei Gardini (Vita-Salute San Raffaele University Milan, Italy); Giuseppina Brancaccio (Università degli Studi Link Campus University Rome, Italy); and Giovanni Battista Levi Sandri (Dipartimento di Chirurgia, Frosinone, Italy).
Authors’ contributions
Methodology, project administration, writing - review and editing: Tovoli F
Investigation, writing - original draft: Crocetti L, Mazzarelli C, Foschi FG, Tortora R, Gambato M
Conceptualisation, investigation, supervision, writing - review and editing: Vitale A
All authors reviewed and approved the final version of the manuscript and agree to be accountable for all aspects of the work.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Tovoli F is a Junior Editorial Board Member of Hepatoma Research. Vitale A is the Guest Editor of the Special Issue “Multidisciplinary Management of HCC” of Hepatoma Research and the Editorial Board Member of Hepatoma Research. They were not involved in any stage of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision making. The other authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR. A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol. 2019;16:589-604.
2. Llovet JM, Pinyol R, Kelley RK, et al. Molecular pathogenesis and systemic therapies for hepatocellular carcinoma. Nat Cancer. 2022;3:386-401.
3. Iavarone M, Nault JC, Cabibbo G, Torres F, Reig M. Indolent cancer and pattern of progression: two missing parameters in trial design for hepatology. Hepatology. 2024;79:1452-62.
4. Cabibbo G, Enea M, Attanasio M, Bruix J, Craxì A, Cammà C. A meta-analysis of survival rates of untreated patients in randomized clinical trials of hepatocellular carcinoma. Hepatology. 2010;51:1274-83.
5. Reig M, Forner A, Rimola J, et al. BCLC strategy for prognosis prediction and treatment recommendation: the 2022 update. J Hepatol. 2022;76:681-93.
6. Saltz LB. Curative-intent treatment for colorectal liver metastases: a medical oncologist’s perspective. Am Soc Clin Oncol Educ Book. 2012;32:205-8.
7. Trevisani F, Vitale A, Kudo M, et al. Merits and boundaries of the BCLC staging and treatment algorithm: learning from the past to improve the future with a novel proposal. J Hepatol. 2024;80:661-9.
8. Chen QF, Chen S, Chen M, Lyu N, Zhao M. Improving the conversion success rate of hepatocellular carcinoma: focus on the use of combination therapy with a high objective response rate. J Clin Transl Hepatol. 2024;12:298-304.
9. Sun HC, Zhou J, Wang Z, et al.; Alliance of Liver Cancer Conversion Therapy, Committee of Liver Cancer of the Chinese Anti-Cancer Association. Chinese expert consensus on conversion therapy for hepatocellular carcinoma (2021 edition). Hepatobiliary Surg Nutr. 2022;11:227-52.
10. Sun J, Guo R, Bi X, et al. Guidelines for diagnosis and treatment of hepatocellular carcinoma with portal vein tumor thrombus in China (2021 Edition). Liver Cancer. 2022;11:315-28.
11. Chan A, Zhang WY, Chok K, et al. ALPPS versus portal vein embolization for hepatitis-related hepatocellular carcinoma: a changing paradigm in modulation of future liver remnant before major hepatectomy. Ann Surg. 2021;273:957-65.
12. Ma Z, Xiao Z, Yin P, et al. Comparison of survival benefit and safety between surgery following conversion therapy versus surgery alone in patients with surgically resectable hepatocellular carcinoma at CNLC IIb/IIIa stage: a propensity score matching study. Int J Surg. 2024;110:2910-21.
13. Tran NH, Muñoz S, Thompson S, Hallemeier CL, Bruix J. Hepatocellular carcinoma downstaging for liver transplantation in the era of systemic combined therapy with anti-VEGF/TKI and immunotherapy. Hepatology. 2022;76:1203-18.
14. Vitale A, Cabibbo G, Iavarone M, et al.; HCC Special Interest Group of the Italian Association for the Study of the Liver. Personalised management of patients with hepatocellular carcinoma: a multiparametric therapeutic hierarchy concept. Lancet Oncol. 2023;24:e312-22.
15. European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J Hepatol. 2025;82:315-74.
16. Ascari S, Chen R, Vivaldi C, et al. Advancements in immunotherapy for hepatocellular carcinoma. Expert Rev Anticancer Ther. 2025;25:151-65.
17. Finn RS, Qin S, Ikeda M, et al.; IMbrave150 Investigators. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma. N Engl J Med. 2020;382:1894-905.
18. Cheng AL, Qin S, Ikeda M, et al. Updated efficacy and safety data from IMbrave150: atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J Hepatol. 2022;76:862-73.
19. Abou-Alfa GK, Lau G, Kudo M, et al. Tremelimumab plus durvalumab in unresectable hepatocellular carcinoma. NEJM Evid. 2022;1:EVIDoa2100070.
20. Rimassa L, Chan SL, Sangro B, et al. Five-year overall survival update from the HIMALAYA study of tremelimumab plus durvalumab in unresectable HCC. J Hepatol. 2025;83:899-908.
21. Galle PR, Decaens T, Kudo M, et al. Nivolumab (NIVO) plus ipilimumab (IPI) vs lenvatinib (LEN) or sorafenib (SOR) as first-line treatment for unresectable hepatocellular carcinoma (uHCC): first results from CheckMate 9DW. J Clin Oncol. 2024;42:LBA4008.
22. Qin S, Chan SL, Gu S, et al.; CARES-310 Study Group. Camrelizumab plus rivoceranib versus sorafenib as first-line therapy for unresectable hepatocellular carcinoma (CARES-310): a randomised, open-label, international phase 3 study. Lancet. 2023;402:1133-46.
23. Kudo M, Aoki T, Ueshima K, et al. Achievement of complete response and drug-free status by atezolizumab plus bevacizumab combined with or without curative conversion in patients with transarterial chemoembolization-unsuitable, intermediate-stage hepatocellular carcinoma: a multicenter proof-of-concept study. Liver Cancer. 2023;12:321-38.
24. Tomonari T, Tani J, Sato Y, et al. Clinical features and outcomes of conversion therapy in patients with unresectable hepatocellular carcinoma. Cancers. 2023;15:5221.
25. Shimose S, Iwamoto H, Shirono T, et al. The impact of curative conversion therapy aimed at a cancer-free state in patients with hepatocellular carcinoma treated with atezolizumab plus bevacizumab. Cancer Med. 2023;12:12325-35.
26. Ke Q, Xin F, Fang H, Zeng Y, Wang L, Liu J. The significance of transarterial chemo(embolization) combined with tyrosine kinase inhibitors and immune checkpoint inhibitors for unresectable hepatocellular carcinoma in the era of systemic therapy: a systematic review. Front Immunol. 2022;13:913464.
27. Sanduzzi-Zamparelli M, Cabibbo G. The “shell game” after objective response in patients with advanced HCC treated with immunotherapy. Hepatology. 2025;81:1635-7.
28. Xie DY, Zhu K, Ren ZG, Zhou J, Fan J, Gao Q. A review of 2022 Chinese clinical guidelines on the management of hepatocellular carcinoma: updates and insights. Hepatobiliary Surg Nutr. 2023;12:216-28.
29. Hasegawa K, Takemura N, Yamashita T, et al.; committee for Revision of the Clinical Practice Guidelines for Hepatocellular Carcinoma, Tokyo, Japan. Clinical Practice Guidelines for hepatocellular carcinoma: the Japan Society of Hepatology 2021 version (5th JSH-HCC Guidelines). Hepatol Res. 2023;53:383-90.
30. Kudo M. Current therapeutic strategies for hepatocellular carcinoma in Japan. Liver Cancer. 2023;12:497-509.
31. Lucatelli P, Burrel M, Guiu B, de Rubeis G, van Delden O, Helmberger T. CIRSE Standards of practice on hepatic transarterial chemoembolisation. Cardiovasc Intervent Radiol. 2021;44:1851-67.
32. Bartkowski R, Berger MR, Aguiar JL, et al. Experiments on the efficacy and toxicity of locoregional chemotherapy of liver tumors with 5-fluoro-2’-deoxyuridine (FUDR) and 5-fluorouracil (5-FU) in an animal model. J Cancer Res Clin Oncol. 1986;111:42-6.
33. Chen LT, Martinelli E, Cheng AL, et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with intermediate and advanced/relapsed hepatocellular carcinoma: a TOS-ESMO initiative endorsed by CSCO, ISMPO, JSMO, KSMO, MOS and SSO. Ann Oncol. 2020;31:334-51.
34. Miller FH, Lopes Vendrami C, Gabr A, et al. Evolution of radioembolization in treatment of hepatocellular carcinoma: a pictorial review. Radiographics. 2021;41:1802-18.
35. Garin E, Guiu B, Edeline J, Rolland Y, Palard X. Trans-arterial radioembolization dosimetry in 2022. Cardiovasc Intervent Radiol. 2022;45:1608-21.
36. Vitellius C, Desjonqueres E, Lequoy M, et al. MASLD-related HCC: multicenter study comparing patients with and without cirrhosis. JHEP Rep. 2024;6:101160.
37. Huang Z, Chen T, Li W, et al. Atezolizumab and bevacizumab plus transarterial chemoembolization and hepatic arterial infusion chemotherapy for patients with high tumor burden unresectable hepatocellular carcinoma: a multi-center cohort study. Int Immunopharmacol. 2024;139:112711.
38. Gan L, Lang M, Tian X, et al. A retrospective analysis of conversion therapy with lenvatinib, sintilimab, and arterially-directed therapy in patients with initially unresectable hepatocellular carcinoma. J Hepatocell Carcinoma. 2023;10:673-86.
39. Zhao W, Liu C, Wu Y, et al. Transarterial chemoembolization (TACE)-hepatic arterial infusion chemotherapy (HAIC) combined with PD-1 inhibitors plus lenvatinib as a preoperative conversion therapy for nonmetastatic advanced hepatocellular carcinoma: a single center experience. Transl Cancer Res. 2024;13:2315-31.
40. Lencioni R, Kudo M, Erinjeri J, et al. EMERALD-1: a phase 3, randomized, placebo-controlled study of transarterial chemoembolization combined with durvalumab with or without bevacizumab in participants with unresectable hepatocellular carcinoma eligible for embolization. J Clin Oncol. 2024;42:LBA432.
41. Kudo M, Ren Z, Guo Y, et al.; LEAP-012 investigators. Transarterial chemoembolisation combined with lenvatinib plus pembrolizumab versus dual placebo for unresectable, non-metastatic hepatocellular carcinoma (LEAP-012): a multicentre, randomised, double-blind, phase 3 study. Lancet. 2025;405:203-15.
42. Li X, Wang X, Bai T, et al. Conversion surgery for initially unresectable hepatocellular carcinoma using lenvatinib combined with TACE plus PD-1 inhibitor: a real-world observational study. Dig Liver Dis. 2024;56:1078-86.
43. Sun HC, Zhu XD. Downstaging conversion therapy in patients with initially unresectable advanced hepatocellular carcinoma: an overview. Front Oncol. 2021;11:772195.
44. Mazzaferro V, Citterio D, Bhoori S, et al. Liver transplantation in hepatocellular carcinoma after tumour downstaging (XXL): a randomised, controlled, phase 2b/3 trial. Lancet Oncol. 2020;21:947-56.
45. Natarajan B, Tabrizian P, Hoteit M, et al. Downstaging hepatocellular carcinoma before liver transplantation: a multicenter analysis of the “all-comers” protocol in the Multicenter Evaluation of Reduction in Tumor Size before Liver Transplantation (MERITS-LT) consortium. Am J Transplant. 2023;23:1771-80.
46. Affonso BB, Galastri FL, da Motta Leal Filho JM, et al. Long-term outcomes of hepatocellular carcinoma that underwent chemoembolization for bridging or downstaging. World J Gastroenterol. 2019;25:5687-701.
47. Gabr A, Abouchaleh N, Ali R, et al. Outcomes of surgical resection after radioembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2018;29:1502-10.e1.
48. Salem R, Johnson GE, Kim E, et al. Yttrium-90 radioembolization for the treatment of solitary, unresectable HCC: the LEGACY study. Hepatology. 2021;74:2342-52.
49. Aliseda D, Martí-Cruchaga P, Zozaya G, et al. Liver resection and transplantation following yttrium-90 radioembolization for primary malignant liver tumors: a 15-year single-center experience. Cancers. 2023;15:733.
50. Tzedakis S, Sebai A, Jeddou H, et al. Resection postradioembolization in patients with single large hepatocellular carcinoma. Ann Surg. 2023;278:756-62.
51. Lewandowski RJ, Kulik LM, Riaz A, et al. A comparative analysis of transarterial downstaging for hepatocellular carcinoma: chemoembolization versus radioembolization. Am J Transplant. 2009;9:1920-8.
52. Mehta N, Frenette C, Tabrizian P, et al. Downstaging outcomes for hepatocellular carcinoma: results from the Multicenter Evaluation of Reduction in Tumor Size before Liver Transplantation (MERITS-LT) Consortium. Gastroenterology. 2021;161:1502-12.
53. Berardi G, Guglielmo N, Cucchetti A, et al. Transarterial radioembolization can downstage intermediate and advanced hepatocellular carcinoma to liver transplantation. Transplantation. 2025;109:e54-63.
54. Regnault H, Chalaye J, Galetto-Pregliasco A, et al. Selective internal radiation therapy for unresectable HCC: the SIRT downstaging study. Hepatol Commun. 2024;8:e0475.
55. Lim KC, Chow PK, Allen JC, Siddiqui FJ, Chan ES, Tan SB. Systematic review of outcomes of liver resection for early hepatocellular carcinoma within the Milan criteria. Br J Surg. 2012;99:1622-9.
56. Assalino M, Terraz S, Grat M, et al. Liver transplantation for hepatocellular carcinoma after successful treatment of macrovascular invasion - a multi-center retrospective cohort study. Transpl Int. 2020;33:567-75.
57. Meerun MA, Allimant C, Rivière B, et al. Large, multifocal or portal vein-invading hepatocellular carcinoma (HCC) downstaged by Y90 using personalized dosimetry: safety, pathological results and outcomes after surgery. Hepatobiliary Surg Nutr. 2023;12:351-65.
58. De la Garza-Ramos C, Toskich BB. Radioembolization for the treatment of hepatocellular carcinoma: the road to personalized dosimetry and ablative practice. Semin Intervent Radiol. 2021;38:466-71.
59. Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A. Hepatocellular carcinoma. Lancet. 2022;400:1345-62.
60. Huber TC, Bochnakova T, Koethe Y, Park B, Farsad K. Percutaneous therapies for hepatocellular carcinoma: evolution of liver directed therapies. J Hepatocell Carcinoma. 2021;8:1181-93.
61. Ahmed M, Brace CL, Lee FT Jr, Goldberg SN. Principles of and advances in percutaneous ablation. Radiology. 2011;258:351-69.
62. Di Costanzo GG, Tortora R, D’Adamo G, et al. Radiofrequency ablation versus laser ablation for the treatment of small hepatocellular carcinoma in cirrhosis: a randomized trial. J Gastroenterol Hepatol. 2015;30:559-65.
63. Tasu JP, Tougeron D, Rols MP. Irreversible electroporation and electrochemotherapy in oncology: state of the art. Diagn Interv Imaging. 2022;103:499-509.
64. Sutter O, Calvo J, N’Kontchou G, et al. Safety and efficacy of irreversible electroporation for the treatment of hepatocellular carcinoma not amenable to thermal ablation techniques: a retrospective single-center case series. Radiology. 2017;284:877-86.
65. Wu R, Shi Q, Liao D, et al. Efficacy and safety of antiangiogenic agents combined with HIFU in the treatment of advanced liver cancer. Front Oncol. 2025;15:1652434.
66. Han S, Sung PS, Park SY, et al. Local ablation for hepatocellular carcinoma: 2024 Expert Consensus-Based Practical Recommendations of the Korean Liver Cancer Association. Gut Liver. 2024;18:789-802.
67. Cillo U, Caregari S, Barabino M, et al. Hierarchically positioning laparoscopic microwave ablation in the therapeutic span of early hepatocellular carcinoma: a real-life comparative analysis. Ann Surg Oncol. 2025;32:1063-72.
68. Liu Q, Zhang R, Shen W. Advancements in locoregional therapy for advanced hepatocellular carcinoma: emerging perspectives on combined treatment strategies. Eur J Surg Oncol. 2025;51:109502.
69. Vitale A, Cabibbo G, Rimassa L, et al. The concept of "converse therapeutic hierarchy" for patients with hepatocellular carcinoma. Liver Cancer. 2025;14:743-57.
70. Yao FY, Ferrell L, Bass NM, et al. Liver transplantation for hepatocellular carcinoma: expansion of the tumor size limits does not adversely impact survival. Hepatology. 2001;33:1394-403.
71. Mazzaferro V, Regalia E, Doci R, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334:693-9.
72. Yao FY, Mehta N, Flemming J, et al. Downstaging of hepatocellular cancer before liver transplant: long-term outcome compared to tumors within Milan criteria. Hepatology. 2015;61:1968-77.
73. Tabrizian P, Holzner ML, Mehta N, et al. Ten-year outcomes of liver transplant and downstaging for hepatocellular carcinoma. JAMA Surg. 2022;157:779-88.
74. Sun HC, Zhu XD. Downstaging conversion therapy in patients with initially unresectable advanced hepatocellular carcinoma: an overview. Front Oncol. 2021;11:772195.
75. Duan Y, Zhang H, Tan T, et al. The immune response of hepatocellular carcinoma after locoregional and systemic therapies: the available combination option for immunotherapy. Biosci Trends. 2024;17:427-44.
76. Memeo R, Conticchio M, Deshayes E, et al. Optimization of the future remnant liver: review of the current strategies in Europe. Hepatobiliary Surg Nutr. 2021;10:350-63.
77. Charles J, Nezami N, Loya M, et al. Portal vein embolization: rationale, techniques, and outcomes to maximize remnant liver hypertrophy with a focus on contemporary strategies. Life. 2023;13:279.
78. Chansangrat J, Keeratibharat N. Portal vein embolization: rationale, techniques, outcomes and novel strategies. Hepat Oncol. 2021;8:HEP42.
79. Shindoh J, Tzeng CWD, Vauthey JN. Portal vein embolization for hepatocellular carcinoma. Liver Cancer. 2012;1:159-67.
80. Aoki T, Kubota K. Preoperative portal vein embolization for hepatocellular carcinoma: consensus and controversy. World J Hepatol. 2016;8:439-45.
81. Glantzounis GK, Tokidis E, Basourakos SP, Ntzani EE, Lianos GD, Pentheroudakis G. The role of portal vein embolization in the surgical management of primary hepatobiliary cancers. a systematic review. Eur J Surg Oncol. 2017;43:32-41.
82. Beppu T, Yamamura K, Okabe H, Imai K, Hayashi H. Oncological benefits of portal vein embolization for patients with hepatocellular carcinoma. Ann Gastroenterol Surg. 2021;5:287-95.
83. Araki K, Shibuya K, Harimoto N, et al. A prospective study of sequential hepatic vein embolization after portal vein embolization in patients scheduled for right-sided major hepatectomy: results of feasibility and surgical strategy using functional liver assessment. J Hepatobiliary Pancreat Sci. 2023;30:91-101.
84. Park GC, Lee SG, Yoon YI, et al. Sequential transcatheter arterial chemoembolization and portal vein embolization before right hemihepatectomy in patients with hepatocellular carcinoma. Hepatobiliary Pancreat Dis Int. 2020;19:244-51.
85. Zhang CW, Dou CW, Zhang XL, et al. Simultaneous transcatheter arterial chemoembolization and portal vein embolization for patients with large hepatocellular carcinoma before major hepatectomy. World J Gastroenterol. 2020;26:4489-500.
86. Terasawa M, Allard MA, Golse N, et al. Sequential transcatheter arterial chemoembolization and portal vein embolization versus portal vein embolization alone before major hepatectomy for patients with large hepatocellular carcinoma: an intent-to-treat analysis. Surgery. 2020;167:425-31.
87. Niekamp AS, Huang SY, Mahvash A, et al. Hepatic vein embolization after portal vein embolization to induce additional liver hypertrophy in patients with metastatic colorectal carcinoma. Eur Radiol. 2020;30:3862-8.
88. Smits J, Chau S, James S, et al.; EuroLVD and DRAGON Trials Collaborative. Combined portal and hepatic vein embolisation in perihilar cholangiocarcinoma. HPB. 2024;26:1458-66.
89. Esposito F, Lim C, Lahat E, et al. Combined hepatic and portal vein embolization as preparation for major hepatectomy: a systematic review. HPB. 2019;21:1099-106.
90. Schadde E. Combined portal vein and hepatic vein embolization-finally the platinum procedure of regenerative liver surgery? Hepatobiliary Surg Nutr. 2020;9:92-4.
91. Baili E, Tsilimigras DI, Moris D, Sahara K, Pawlik TM. Technical modifications and outcomes after associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) for primary liver malignancies: a systematic review. Surg Oncol. 2020;33:70-80.
92. Lau WY, Lai EC, Lau SH. Associating liver partition and portal vein ligation for staged hepatectomy: the current role and development. Hepatobiliary Pancreat Dis Int. 2017;16:17-26.
93. Xiang F, Hu ZM. Chance and challenge of associating liver partition and portal vein ligation for staged hepatectomy. Hepatobiliary Pancreat Dis Int. 2019;18:214-22.
94. Nikokiraki C, Psaraki A, Roubelakis MG. The potential clinical use of stem/progenitor cells and organoids in liver diseases. Cells. 2022;11:1410.
95. So J, Kim A, Lee SH, Shin D. Liver progenitor cell-driven liver regeneration. Exp Mol Med. 2020;52:1230-8.
96. Cardinale V, Lanthier N, Baptista PM, et al. Cell transplantation-based regenerative medicine in liver diseases. Stem Cell Reports. 2023;18:1555-72.
97. Zahmatkesh E, Khoshdel Rad N, Hossein-Khannazer N, et al. Cell and cell-derivative-based therapy for liver diseases: current approaches and future promises. Expert Rev Gastroenterol Hepatol. 2023;17:237-49.
98. Nevola R, Ruocco R, Criscuolo L, et al. Predictors of early and late hepatocellular carcinoma recurrence. World J Gastroenterol. 2023;29:1243-60.
99. Xu XF, Xing H, Han J, et al. Risk factors, patterns, and outcomes of late recurrence after liver resection for hepatocellular carcinoma: a multicenter study from China. JAMA Surg. 2019;154:209-17.
100. Fuster-Anglada C, Mauro E, Ferrer-Fàbrega J, et al. Histological predictors of aggressive recurrence of hepatocellular carcinoma after liver resection. J Hepatol. 2024;81:995-1004.
101. Poon R, Fan ST, Wong J. Risk factors, prevention, and management of postoperative recurrence after resection of hepatocellular carcinoma. Ann Surg. 2000;232:10-24.
102. Yamanaka N, Tanaka T, Tanaka W, et al. Correlation of hepatitis virus serologic status with clinicopathologic features in patients undergoing hepatectomy for hepatocellular carcinoma. Cancer. 1997;79:1509-15.
103. Fung AKY, Cheng NMY, Chong CCN, et al. Single-center experience on actual mid-term (≥5 years) and long-term (≥10 years) survival outcome in patients with hepatocellular carcinoma after curative hepatectomy: a bimodal distribution. Medicine. 2020;99:e23358.
104. Shirabe K, Sugimachi K, Harada N, et al. Favorable prognosis in patients with sustained virological response to antiviral therapy, including interferon, for chronic hepatitis C before hepatic resection for hepatocellular carcinoma. Anticancer Res. 2015;35:6963-9.
105. Zhou Y, Si X, Wu L, Su X, Li B, Zhang Z. Influence of viral hepatitis status on prognosis in patients undergoing hepatic resection for hepatocellular carcinoma: a meta-analysis of observational studies. World J Surg Oncol. 2011;9:108.
106. Arnaoutakis DJ, Mavros MN, Shen F, et al. Recurrence patterns and prognostic factors in patients with hepatocellular carcinoma in noncirrhotic liver: a multi-institutional analysis. Ann Surg Oncol. 2014;21:147-54.
107. Kim JM, Kwon CH, Joh JW, et al. Differences between hepatocellular carcinoma and hepatitis B virus infection in patients with and without cirrhosis. Ann Surg Oncol. 2014;21:458-65.
108. Lee HW, Choi GH, Kim DY, et al. Less fibrotic burden differently affects the long-term outcomes of hepatocellular carcinoma after curative resection. Oncology. 2017;93:224-32.
109. D’Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: a systematic review of 118 studies. J Hepatol. 2006;44:217-31.
110. Piano S, Reiberger T, Bosch J. Mechanisms and implications of recompensation in cirrhosis. JHEP Rep. 2024;6:101233.
111. Lampertico P, Invernizzi F, Viganò M, et al. The long-term benefits of nucleos(t)ide analogs in compensated HBV cirrhotic patients with no or small esophageal varices: a 12-year prospective cohort study. J Hepatol. 2015;63:1118-25.
112. Nguyen MH, Keeffe EB. Chronic hepatitis B: early viral suppression and long-term outcomes of therapy with oral nucleos(t)ides. J Viral Hepat. 2009;16:149-55.
113. Jang JW, Choi JY, Kim YS, et al. Long-term effect of antiviral therapy on disease course after decompensation in patients with hepatitis B virus-related cirrhosis. Hepatology. 2015;61:1809-20.
114. Cholongitas E, Papatheodoridis GV, Goulis J, et al. The impact of newer nucleos(t)ide analogues on patients with hepatitis B decompensated cirrhosis. Ann Gastroenterol. 2015;28:109-17.
115. Zhang Y, Liu X, Li S, et al. Risk of HCC decreases in HBV-related patients with cirrhosis acquired recompensation: a retrospective study based on Baveno VII criteria. Hepatol Commun. 2024;8:e0355.
116. Yu SJ, Kim YJ. Hepatitis B viral load affects prognosis of hepatocellular carcinoma. World J Gastroenterol. 2014;20:12039-44.
117. Shim JJ. Long-term suppression of viral replication in chronic hepatitis B: outcomes and future directions. Gut Liver. 2015;9:265-6.
118. Llovet JM, Kelley RK, Villanueva A, et al. Hepatocellular carcinoma. Nat Rev Dis Primers. 2021;7:6.
119. Singal AG, Kanwal F, Llovet JM. Global trends in hepatocellular carcinoma epidemiology: implications for screening, prevention and therapy. Nat Rev Clin Oncol. 2023;20:864-84.
120. Kim DY. Changing etiology and epidemiology of hepatocellular carcinoma: Asia and worldwide. J Liver Cancer. 2024;24:62-70.
121. Vibert E, Schwartz M, Olthoff KM. Advances in resection and transplantation for hepatocellular carcinoma. J Hepatol. 2020;72:262-76.
122. Roayaie S, Haim MB, Emre S, et al. Comparison of surgical outcomes for hepatocellular carcinoma in patients with hepatitis B versus hepatitis C: a western experience. Ann Surg Oncol. 2000;7:764-70.
123. Franssen B, Alshebeeb K, Tabrizian P, et al. Differences in surgical outcomes between hepatitis B- and hepatitis C-related hepatocellular carcinoma: a retrospective analysis of a single North American center. Ann Surg. 2014;260:650-8.
124. Singal AG, Rich NE, Mehta N, et al. Direct-acting antiviral therapy for hepatitis C virus infection is associated with increased survival in patients with a history of hepatocellular carcinoma. Gastroenterology. 2019;157:1253-63.e2.
125. Singal AG, Rich NE, Mehta N, et al. Direct-acting antiviral therapy not associated with recurrence of hepatocellular carcinoma in a multicenter North American cohort study. Gastroenterology. 2019;156:1683-92.e1.
126. Cabibbo G, Celsa C, Calvaruso V, et al.; Rete Sicilia Selezione Terapia - HCV (RESIST-HCV) and Italian Liver Cancer (ITA.LI.CA.) Group. Direct-acting antivirals after successful treatment of early hepatocellular carcinoma improve survival in HCV-cirrhotic patients. J Hepatol. 2019;71:265-73.
127. Kamp WM, Sellers CM, Stein S, Lim JK, Kim HS. Direct-acting antivirals improve overall survival in interventional oncology patients with hepatitis C and hepatocellular carcinoma. J Vasc Interv Radiol. 2020;31:953-60.
128. Gorgen A, Galvin Z, Huang AC, et al. The impact of direct-acting antivirals on overall mortality and tumoral recurrence in patients with hepatocellular carcinoma listed for liver transplantation: an international multicenter study. Transplantation. 2020;104:2087-96.
129. Reig M, Mariño Z, Perelló C, et al. Unexpected high rate of early tumor recurrence in patients with HCV-related HCC undergoing interferon-free therapy. J Hepatol. 2016;65:719-26.
130. Conti F, Buonfiglioli F, Scuteri A, et al. Early occurrence and recurrence of hepatocellular carcinoma in HCV-related cirrhosis treated with direct-acting antivirals. J Hepatol. 2016;65:727-33.
131. Tsai PC, Huang CF, Yu ML. Unexpected early tumor recurrence in patients with hepatitis C virus-related hepatocellular carcinoma undergoing interferon-free therapy: issue of the interval between HCC treatment and antiviral therapy. J Hepatol. 2017;66:464.
132. Lui FH, Moosvi Z, Patel A, et al. Decreased risk of hepatocellular carcinoma recurrence with direct-acting antivirals compared with no treatment for hepatitis C: a meta-analysis. Ann Gastroenterol. 2020;33:293-8.
133. Reiberger T, Lens S, Cabibbo G, et al. EASL position paper on clinical follow-up after HCV cure. J Hepatol. 2024;81:326-44.
134. Mezzacappa C, Kim NJ, Vutien P, Kaplan DE, Ioannou GN, Taddei TH. Screening for hepatocellular carcinoma and survival in patients with cirrhosis after hepatitis C virus cure. JAMA Netw Open. 2024;7:e2420963.
135. Semmler G, Meyer EL, Kozbial K, et al. HCC risk stratification after cure of hepatitis C in patients with compensated advanced chronic liver disease. J Hepatol. 2022;76:812-21.
136. Hofer BS, Simbrunner B, Hartl L, et al. Alcohol abstinence improves prognosis across all stages of portal hypertension in alcohol-related cirrhosis. Clin Gastroenterol Hepatol. 2023;21:2308-17.e7.
137. Rodríguez M, González-Diéguez ML, Varela M, et al. Impact of alcohol abstinence on the risk of hepatocellular carcinoma in patients with alcohol-related liver cirrhosis. Am J Gastroenterol. 2021;116:2390-8.
138. European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). Obes Facts. 2024;17:374-444.
139. Lange NF, Radu P, Dufour JF. Prevention of NAFLD-associated HCC: role of lifestyle and chemoprevention. J Hepatol. 2021;75:1217-27.
140. Njei B, McCarty TR, Sharma P, et al. Bariatric surgery and hepatocellular carcinoma: a propensity score-matched analysis. Obes Surg. 2018;28:3880-9.
141. Zou B, Odden MC, Nguyen MH. Statin use and reduced hepatocellular carcinoma risk in patients with nonalcoholic fatty liver disease. Clin Gastroenterol Hepatol. 2023;21:435-44.e6.
142. Pinyopornpanish K, Al-Yaman W, Butler RS, Carey W, McCullough A, Romero-Marrero C. Chemopreventive effect of statin on hepatocellular carcinoma in patients with nonalcoholic steatohepatitis cirrhosis. Am J Gastroenterol. 2021;116:2258-69.
143. Stefanini B, Manfredi GF, D’Alessio A, et al. Delivering adjuvant and neoadjuvant treatments in the early stages of hepatocellular carcinoma. Expert Rev Gastroenterol Hepatol. 2024;18:647-60.
144. Cabibbo G, Celsa C, Rimassa L, et al. Navigating the landscape of liver cancer management: Study designs in clinical trials and clinical practice. J Hepatol. 2024;80:957-66.
145. Mazzaferro V, Romito R, Schiavo M, et al.; HCC Italian Task Force. Prevention of hepatocellular carcinoma recurrence with alpha-interferon after liver resection in HCV cirrhosis. Hepatology. 2006;44:1543-54.
146. Chen LT, Chen MF, Li LA, et al.; Disease Committee of Adjuvant Therapy for Postoperative Hepatocellular Carcinoma, Taiwan; Cooperative Oncology Group, National Health Research Institutes, Zhunan, Taiwan. Long-term results of a randomized, observation-controlled, phase III trial of adjuvant interferon Alfa-2b in hepatocellular carcinoma after curative resection. Ann Surg. 2012;255:8-17.
147. Bruix J, Takayama T, Mazzaferro V, et al.; STORM investigators. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16:1344-54.
148. Lee JH, Lee JH, Lim YS, et al. Adjuvant immunotherapy with autologous cytokine-induced killer cells for hepatocellular carcinoma. Gastroenterology. 2015;148:1383-91.e6.
149. Qin S, Chen M, Cheng AL, et al.; IMbrave050 investigators. Atezolizumab plus bevacizumab versus active surveillance in patients with resected or ablated high-risk hepatocellular carcinoma (IMbrave050): a randomised, open-label, multicentre, phase 3 trial. Lancet. 2023;402:1835-47.
150. Yopp A, Kudo M, Chen M, et al. LBA39 Updated efficacy and safety data from IMbrave050: Phase III study of adjuvant atezolizumab (atezo) + bevacizumab (bev) vs active surveillance in patients (pts) with resected or ablated high-risk hepatocellular carcinoma (HCC). Ann Oncol. 2024;35:S1230.
151. Wang K, Xiang YJ, Yu HM, et al. Adjuvant sintilimab in resected high-risk hepatocellular carcinoma: a randomized, controlled, phase 2 trial. Nat Med. 2024;30:708-15.
152. Zhang S, Yuan L, Danilova L, et al. Spatial transcriptomics analysis of neoadjuvant cabozantinib and nivolumab in advanced hepatocellular carcinoma identifies independent mechanisms of resistance and recurrence. Genome Med. 2023;15:72.
153. Marron TU, Fiel MI, Hamon P, et al. Neoadjuvant cemiplimab for resectable hepatocellular carcinoma: a single-arm, open-label, phase 2 trial. Lancet Gastroenterol Hepatol. 2022;7:219-29.
154. LaPelusa M, Chamseddine S, Tran Cao HS, et al. Tissue and imaging biomarkers of response to neoadjuvant nivolumab or nivolumab plus ipilimumab in patients with resectable hepatocellular carcinoma. Oncology. 2025;103:490-7.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






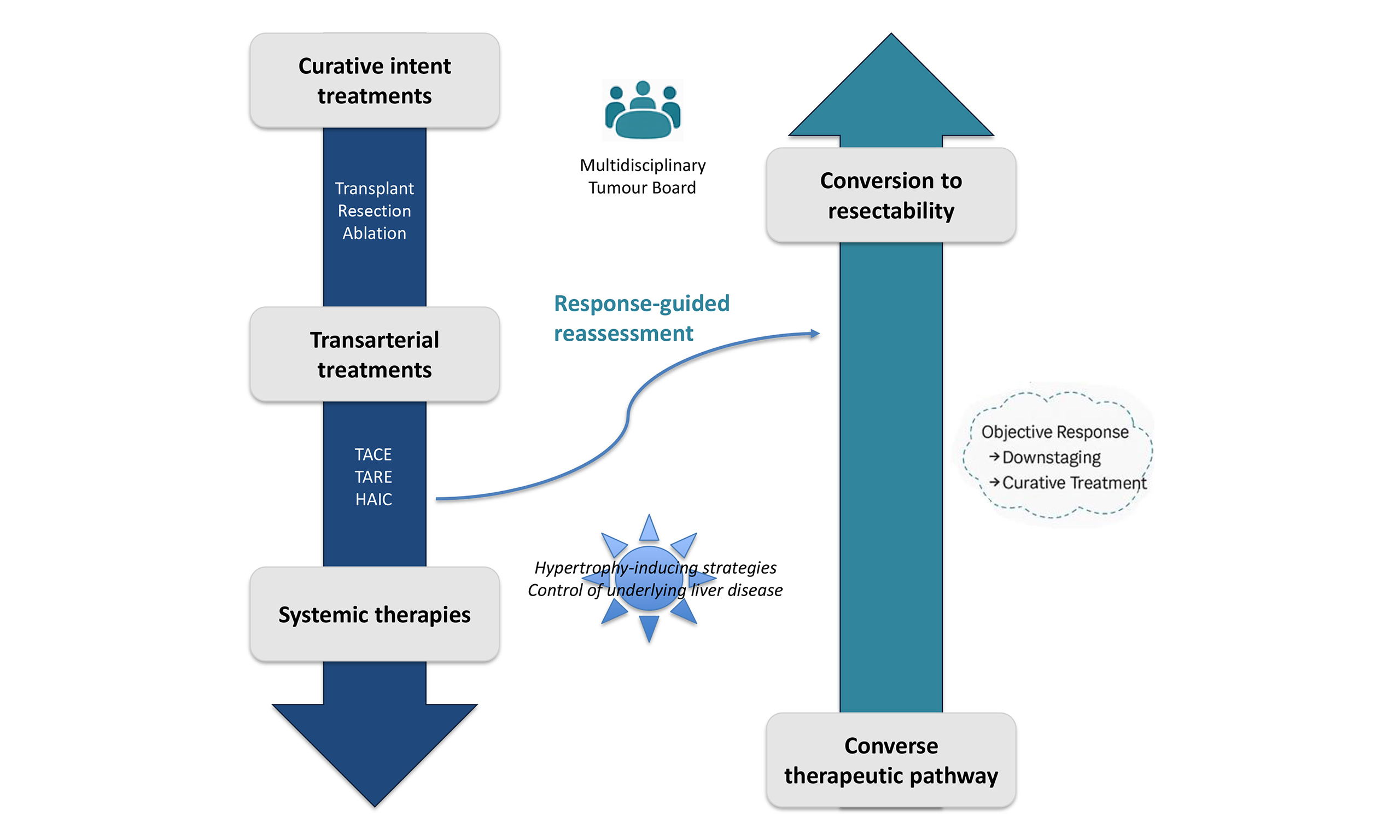
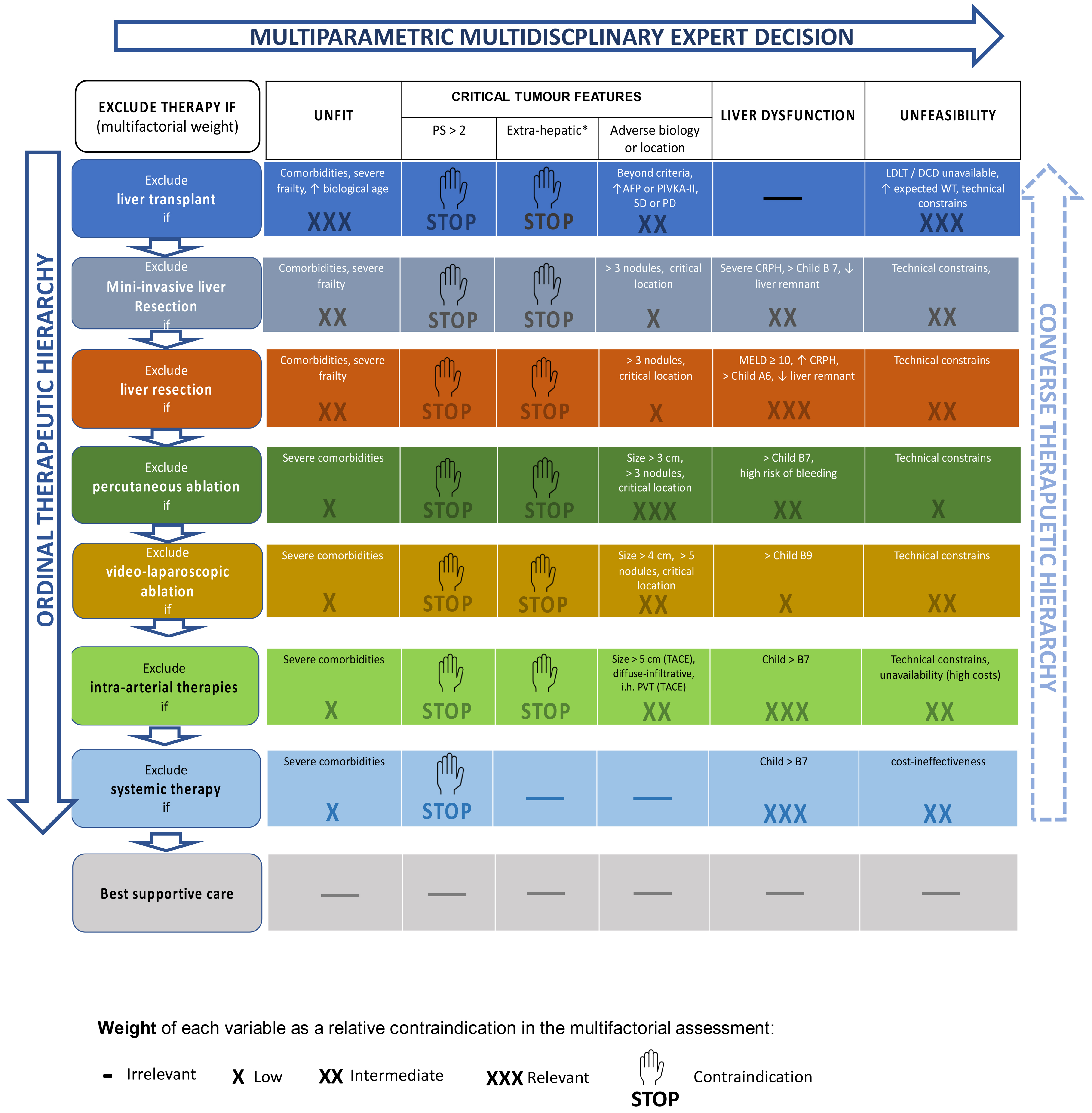










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].