Reconstructing the lining in full-thickness nasal defects: techniques, challenges, and innovations
Abstract
Full-thickness nasal defects pose significant reconstructive challenges due to the complex anatomical and functional requirements of nasal lining reconstruction. The lining is crucial for maintaining nasal patency, humidification, and overall structural integrity. This review explores the existing literature on nasal lining reconstruction, highlighting common etiologies, reconstructive principles, techniques, outcomes, and complications associated with various approaches. Techniques discussed range from simple local mucosal flaps suitable for small defects to more complex regional flaps, such as septal mucosal flaps and inferior turbinate flaps, ideal for moderate defects. Additionally, advanced approaches, including microvascular free tissue transfer - such as the radial forearm free flap - are evaluated for their effectiveness in larger or composite defects. Emerging strategies involving tissue engineering, biomaterials, and prefabricated composite grafts are explored as potential advancements that could mitigate current limitations. Critical appraisal of the advantages, complications, and limitations of each method highlights the importance of tailored reconstruction based on defect size, location, and patient-specific factors such as prior radiation therapy or surgical history. This review underscores the necessity for meticulous surgical planning, adherence to reconstructive principles, and consideration of innovative technologies to improve patient satisfaction and functional outcomes. By summarizing current evidence and identifying critical gaps, this paper aims to guide future research toward refining nasal lining reconstruction practices.
Keywords
INTRODUCTION
Full-thickness nasal defects represent a unique reconstructive challenge due to the need to restore the nose’s three-layered structure: external skin, cartilaginous or bony framework, and internal mucosal lining. Among these, lining reconstruction is often the most technically demanding and prone to failure if not performed correctly, particularly in defects resulting from oncologic resection, trauma, or infection. An insufficient or nonviable internal lining can lead to flap failure, contracture, nasal airway obstruction, and long-term functional compromise[1].
The nasal lining must be thin, well-vascularized, and pliable to maintain airway patency and support the overlying framework[2]. Unlike external skin, the lining cannot tolerate desiccation or inadequate perfusion. Despite its importance, lining reconstruction often receives less attention in the literature than structural or skin coverage techniques. Inadequate planning or inappropriate technique selection can result in devastating long-term consequences, including nostril stenosis or collapse[1].
Numerous reconstructive options have been described, ranging from local mucosal advancement and hinge flaps to more complex regional flaps such as the inferior turbinate or septal mucosal flap[3,4]. In cases of large or composite defects, free tissue transfer - particularly with thin fasciocutaneous flaps such as the radial forearm free flap (RFFF) - may be required[5]. The folded forehead flap has also emerged as a solution for reconstructing both internal and external lining for large full-thickness defects[6]. Each approach must be tailored to defect size, location, and the patient’s prior surgical history and comorbidities, including prior radiation therapy.
The goal of this review is to provide an in-depth overview of current techniques for reconstructing the nasal lining in full-thickness nasal defects, emphasizing evidence-based practices, technical pearls, limitations of existing approaches, and emerging innovations. By highlighting both historical context and contemporary refinements, we aim to equip surgeons with a practical and comprehensive understanding of this critical aspect of nasal reconstruction.
This is a narrative review. To minimize selection bias, we conducted a structured literature search in PubMed/MEDLINE, Embase, and Google Scholar for the years 1990-2025. Search terms included combinations of: nasal lining reconstruction, septal mucosal flap, inferior turbinate flap, hinge flap, paramedian forehead flap, folded paramedian forehead flap, RFFF, anterolateral thigh (ALT), ALT fascia flap, and composite graft. Both Medical Subject Headings (MeSH) terms and keywords were used. We included English-language, human studies that focused on reconstructive techniques, outcomes, or innovations for full-thickness nasal lining defects. Given the heterogeneity of study types and lack of randomized trials, we elected a narrative review approach rather than a systematic review. This review focuses on adult nasal lining reconstruction; pediatric-specific protocols are beyond its scope, and oncologic timing is considered only in relation to its impact on reconstructive staging.
The current evidence base for nasal lining reconstruction is composed almost entirely of retrospective case series, single-surgeon experiences, and small cohort studies, with no randomized or comparative trials available. As such, the strength of evidence supporting each technique is limited, and reported outcomes must be interpreted within the context of low-level study designs. Standardized functional reporting - such as Nasal Obstruction Symptom Evaluation (NOSE) or Standardized Cosmesis and Health Nasal Outcomes Survey (SCHNOS) scores - is rarely included, highlighting the need for prospective data collection and consistent patient-reported outcome measures in future studies. These limitations underscore the importance of individualized clinical judgment when extrapolating results across reconstructive approaches.
ANATOMY AND PHYSIOLOGY OF NASAL LINING
The nasal lining is composed primarily of respiratory epithelium - a ciliated pseudostratified columnar epithelium rich in goblet cells - that plays an essential role in humidifying, filtering, and warming inspired air. Submucosal glands secrete mucus that traps particulates and microbes, while the ciliary apparatus facilitates mucociliary clearance[7]. Thus, the nasal mucosa not only produces these secretions, but also plays a critical role in their natural clearance. The lining is also vital for maintaining airway resistance, regulating airflow turbulence, and supporting olfactory function[8].
The nasal cavity’s internal surface is richly vascularized, primarily via branches of the sphenopalatine, anterior ethmoidal, and superior labial arteries. This robust vascularity allows for the viability of local flaps and enhances healing but also increases the risk of bleeding during surgery[9]. Understanding this intricate anatomy is crucial in planning flaps for lining reconstruction, as certain regions (e.g., the septum, inferior turbinate and surrounding skin) serve as reliable donor sites.
ETIOLOGIES AND DEFECT CLASSIFICATION
Full-thickness nasal defects can result from oncologic resection (particularly of skin cancers such as basal/squamous cell carcinoma or melanoma), trauma, iatrogenic injury, or infection. In rare cases, congenital anomalies or autoimmune diseases such as granulomatosis with polyangiitis may contribute[7].
When cancer is the proximate cause of the defect, it is critical to ensure that repair is not undertaken until oncologic resection to negative margins is confirmed[10]. This is best accomplished by Mohs micrographic excision or by delayed reconstruction after final pathology by permanent section is complete. Frozen sections should not be relied upon, as the need to re-resect may be difficult after initiation of the reconstruction.
Defect classification traditionally follows the subunit principle described by Burget and Menick, segmenting the nose into aesthetic zones: dorsum, tip, alae, sidewalls, and soft tissue triangles[11]. This principle is critical in nasal reconstruction because repairs that respect subunit boundaries tend to yield superior aesthetic outcomes and more predictable contour restoration. For internal lining reconstruction, defects are further characterized by:
· Location: septal, lateral wall, vestibular lining, columella or floor
· Aesthetic subunit involvement: whether lining loss affects specific overlying subunits such as the ala, tip, or columella, which directly impacts flap selection and framework planning
· Size: small (< 1.5 cm2), medium (1.5-3 cm2), or large (> 3 cm2)
· Depth: partial-thickness (mucosal or soft tissue loss only) vs. full-thickness (involving all three nasal layers)
· Laterality: unilateral or bilateral defects
PRINCIPLES OF LINING RECONSTRUCTION
Tissue requirements
An ideal lining substitute must meet several key requirements:
· Thin, to prevent bulkiness that could obstruct airflow
· Well-vascularized, to promote rapid healing and minimize infection risk
· Flexible, to conform to the nasal contour
· Durable, to resist post-operative contraction and maintain long-term airway patency
The internal nasal environment is inherently a moist environment and poorly tolerant to nonviable or ischemic tissue, making strict adherence to vascular principles and layered reconstruction essential for success[9].
The principle of “like with like” applies strongly in lining reconstruction: whenever feasible, native nasal mucosa should be replaced with similar respiratory epithelium, as many local options (such as hinge flaps or skin-based flaps) introduce keratinized epithelium that can lead to dryness, crusting, and desiccation over time[9]. Poor patient comfort and long-term hygiene limitations can compromise even the most natural-appearing external reconstruction. The highest quality reconstruction of nasal lining therefore takes into account the limitations of the donor tissue, selecting thin, vascularized, pliable tissue when possible and favoring mucosa over skin-based reconstruction in order to optimize the functional, as well as aesthetic, result.
Staging and sequencing
Lining reconstruction is typically performed before or concurrently with restoration of the nasal framework and external skin envelope. In complex or radiated defects, a staged approach is often recommended to maximize flap perfusion, reduce ischemic risk, and allow time for vascular delay procedures or flap prefabrication[2].
Staging also facilitates gradual structural build-up, allowing surgeons to first establish a stable internal lining that can support cartilage grafting and overlying skin coverage in subsequent phases.
RECONSTRUCTIVE TECHNIQUES
Nasal lining reconstruction draws on a wide range of techniques, which can be grouped into free grafts, local flaps, regional flaps, and microvascular free tissue transfer. The choice of technique depends on the size, location, depth, and complexity of the defect, as well as patient-specific factors such as prior surgeries, radiation exposure, and comorbidities. See Table 1 for a summary of reconstructive techniques.
Summary table
| Technique | Ideal indication | Advantages | Limitations | Key vessel/Pedicle |
| Secondary intention | Small defects | Simple; no surgery needed | Contracture and synechiae | N/A (healing by granulation) |
| Skin/mucosal grafts (FTSG, STSG, hard palate) | Small, well-vascularized defects | Simple; readily available donor sites | Non-vascularized; prone to contraction, failure | N/A (graft survival dependent on recipient bed) |
| Composite grafts (skin + cartilage) | Alar rim reconstruction | Provides structural support; less contraction | High metabolic demand; size limited (< 1.5 cm) | N/A (graft survival dependent on recipient bed) |
| Hinge flaps | Small vestibular defects | Local; immediate vascularized coverage | Bulk; hair-bearing risk; limited reach | Subdermal plexus of adjacent skin |
| Vestibular advancement flaps | Small anterior lining defects | Preserves native mucosa; minimal donor morbidity | Fragile; insufficient for larger defects | Septal branches of the superior labial artery |
| Septal mucosal flaps | Moderate-sized anterior or tip defects | Thin, robust mucosal tissue; wide arc of rotation | Requires septal integrity; risk of septal perforation | Superior labial/columellar artery (anterior), ethmoidal branches (superior), sphenopalatine artery (posterior) |
| Inferior turbinate flaps | Posterior or lateral lining defects | Vascularized mucosa; endoscopic harvest possible | Donor site crusting; risk of atrophic rhinitis | Sphenopalatine artery (posterior) or angular artery (anterior) |
| Pericranial flaps | Scaffold for secondary mucosalization | Thin, pliable; axial pattern | Requires tunneling; acts as a substrate, not lining | Supratrochlear and supraorbital arteries |
| Melolabial flaps | Alar defects (lining + cover) | Can reconstruct internal + external components | Limited width; reliant on subdermal plexus | Angular artery branches |
| FAMM flaps | Complex cases lacking intranasal mucosa | Provides vascularized oral mucosa | Bulk; hair-bearing risk; limited reach | Facial artery (axial pattern) |
| RFFF | Large or composite lining defects | Thin, versatile; can be folded/tubed; osteocutaneous option | Microsurgical demands; operative time; donor site morbidity | Radial artery (long pedicle, venae comitantes) |
| Other free flaps (ALT, MSAP, MFC) | Large or complex reconstructions needing bone/cartilage | Versatility; tissue-specific options | Bulk; requires expert microvascular skills | Descending branch of LCFA; medial sural artery; descending genicular artery |
Secondary intention
Secondary Intention must always be considered as an option, consistent with the principle of the reconstructive ladder. Because of the robust regenerative potential of nasal mucosa, as well as the fact that vascularized tissue is commonly employed for the corollary skin defect, small internal lining defects may be effectively managed with secondary intention alone. When employed, key factors for success include providing a moist wound healing environment, vascularized wound bed, and minimizing bacterial burden[4].
Potential limitations of healing by secondary intention include the significant potential for contracture of the wound. This, in turn, may create intranasal synechiae/stenosis, or distort the external reconstruction of pivotal structures such as the ala and soft tissue triangle. This is particularly true for defects within the lower third of the nose. More definitive reconstruction of nasal lining is therefore preferred for larger and/or more distal lining defects[4].
Free grafts
Skin grafts
Skin Grafts [full- or split-thickness; full thickness skin graft (FTSG) and split thickness skin graft (STSG)] can be used for limited lining defects but are inherently non-vascularized and rely entirely on the underlying recipient bed for survival. They are prone to contraction and failure unless placed on a well-vascularized bed[12]. Even when placed appropriately, transferring keratinized epithelium to a natively moist environment can result in chronic dryness and desiccation[9]. As a result, skin grafting is typically reserved for small sidewall defects in which nasal valve integrity is preserved[13].
When skin grafting is undertaken, meticulous technique is essential. A light bolster or conservative quilting suture technique should be used to ensure contact between the graft and vascular bed, promoting take without undue compression[14]. If additional structural support is needed, it can be added during a second stage operation.
Haack et al. describe a method in which a precisely measured full-thickness skin graft is harvested, inset into the defect with everted edges, and subsequently used as a bed for forehead flap inset. In this technique, the graft is secured to the forehead flap using quilting sutures and a small foam bolster placed into the nasal vestibule to maintain contact and shape during healing[12].
Mucosal grafts
Mucosal grafts can also be used for nasal lining reconstruction and share many of the same indications and limitations as skin grafts. Hard palate mucosa, in particular, can be harvested along with its periosteum, offering some intrinsic structural support to the reconstructed area. However, graft shrinkage remains a significant concern, with reports of up to 11%-35% contraction when used to reconstruct critical structures such as the alar margin. Additionally, the donor site can be associated with considerable postoperative discomfort and typically heals by secondary intention, leading to a prolonged recovery period[15].
Composite grafts
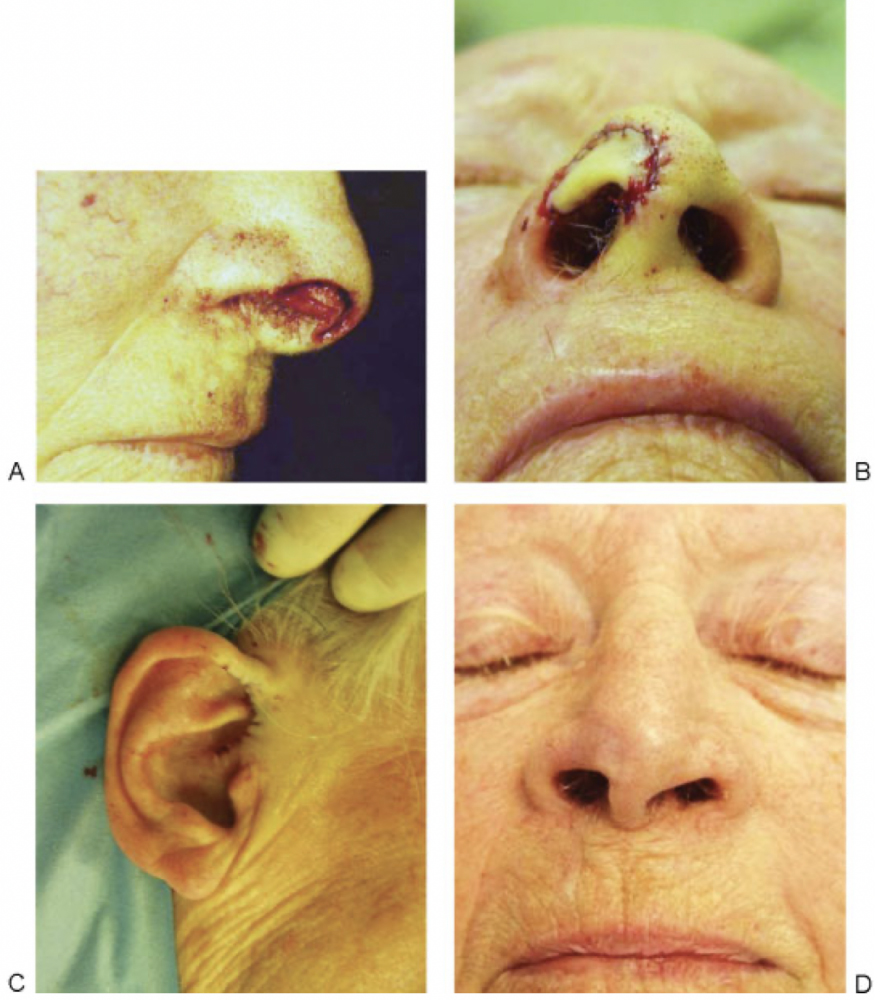
Composite grafts are particularly valuable for alar reconstruction, where structural support is critical to prevent alar notching and maintain contour [Figure 1][12]. Compared to skin grafts, composite grafts contract less and provide more structure; however, they have a higher metabolic demand and consequently a greater risk of failure. Partial or complete necrosis has been reported at more than 5%-11% in some series[16,17].
Figure 1. (A) Full thickness defect of nasal soft tissue triangle; (B) Composite graft in position along soft tissue triangle; (C) Right ear where composite graft harvested from; (D) Composite graft healed. Reprinted with permission from[12]. Copyright © 2026 Georg Thieme Verlag KG.
To optimize chance of success, careful attention must be given to graft size. The maximum size of a composite graft that can reliably survive remains somewhat controversial, but it is generally accepted that grafts larger than 1.5 cm in diameter are at increased risk for necrosis. The most critical determinant of survival is the distance from the vascularized recipient bed to the most distal portion of the graft[12].
Two-layer composite grafts, when covered by a vascularized cutaneous flap, demonstrate improved survival rates compared to three-layer reconstructions that attempt to replace internal lining, framework, and external skin simultaneously[17]. The vascular support provided by the overlying flap is essential for optimizing graft take and long-term viability.
Dermal regeneration templates and prefabricated composite grafts
Dermal regeneration templates (e.g., Integra) and prefabricated composite grafts are under investigation for lining purposes, particularly in complex reconstructions or in irradiated fields. These materials offer the potential to reduce donor site morbidity and create ready-to-use composite constructs, but further clinical research is needed to validate their long-term performance and integration compared to traditional grafts and flaps.
Local flaps
Cutaneous hinge flaps
Cutaneous hinge flaps are pedicled skin flaps elevated from the nasal dorsum, sidewall, or medial cheek and folded inward to resurface small vestibular or intranasal defects. They are relatively simple to raise, typically in a subdermal plane, and provide immediate vascularized coverage. However, they have several limitations, including the risk of transferring hair-bearing skin, excess bulk, and limited compliance, which can compromise long-term airway patency[4,12].
Flap survival is generally excellent, but hinge flaps are at greater risk of marginal vascularity when the length exceeds 1.5 cm. Techniques such as dermabrasion of the wound edge to bleeding tissue have been shown to extend flap viability safely by over 2 cm[18]. Since cutaneous hinge flaps achieve maximal vascularity after a period of epithelialization at the wound edges, they may require a prolonged delay between injury and definitive reconstruction - often three weeks or longer - which is a notable drawback[19]. In some cases, particularly when using the flap for isolated alar reconstruction, a single inferiorly based cutaneous flap can be folded to simultaneously reconstruct the internal lining and external alar margin, with the donor site closed primarily in a V-Y advancement[20].
Significant risk factors for necrosis include pre-existing microvascular disease and disruption of the angular artery or surrounding vasculature from prior oncologic resection or trauma[21]. Despite these limitations, hinge flaps remain a versatile and convenient option for select small to moderate nasal lining defects.
A specific version of the cutaneous hinge flap is the so-called hingeover flap that takes advantage of the initial healing process[22]. When full-thickness defects are allowed to heal by secondary intention or are grafted primarily, a scar naturally forms to connect the nasal lining defect to the cutaneous defect. By 6-8 weeks afterwards, these scars can be excised and turned inward to provide lining that already articulates with the border of the nasal lining defect. Limitations of the hingeover flap include bulkiness and poor vascularity; for this reason, they should only be used for subcentimeter defects and caution should be exercised in conjunction with nonvascularized tissue such as cartilage grafts. Elevation and delay of the hingeover flaps may increase their vascularity; however, this further delays definitive reconstruction, making this option less palatable in many cases.
Vestibular advancement flaps
Vestibular advancement flaps can be used for small anterior lining defects by mobilizing native vestibular mucosa. These flaps are considered delicate and can be prone to tearing, making inset difficult; they are therefore insufficient for larger or deeper defects. They are best suited for lining defects < 1 cm in vertical height, measured cranio-caudad, of the tip or alar subunits[23].
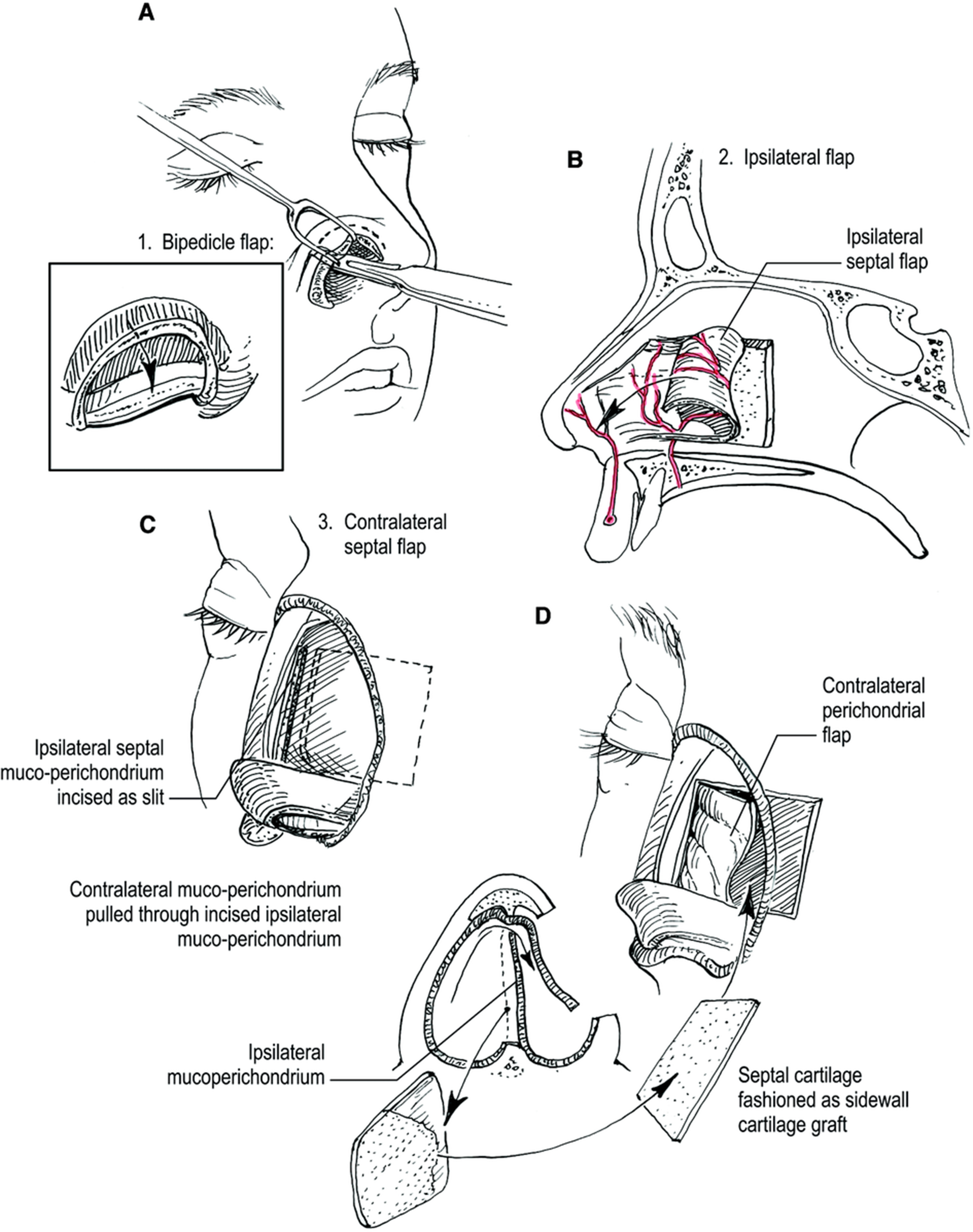
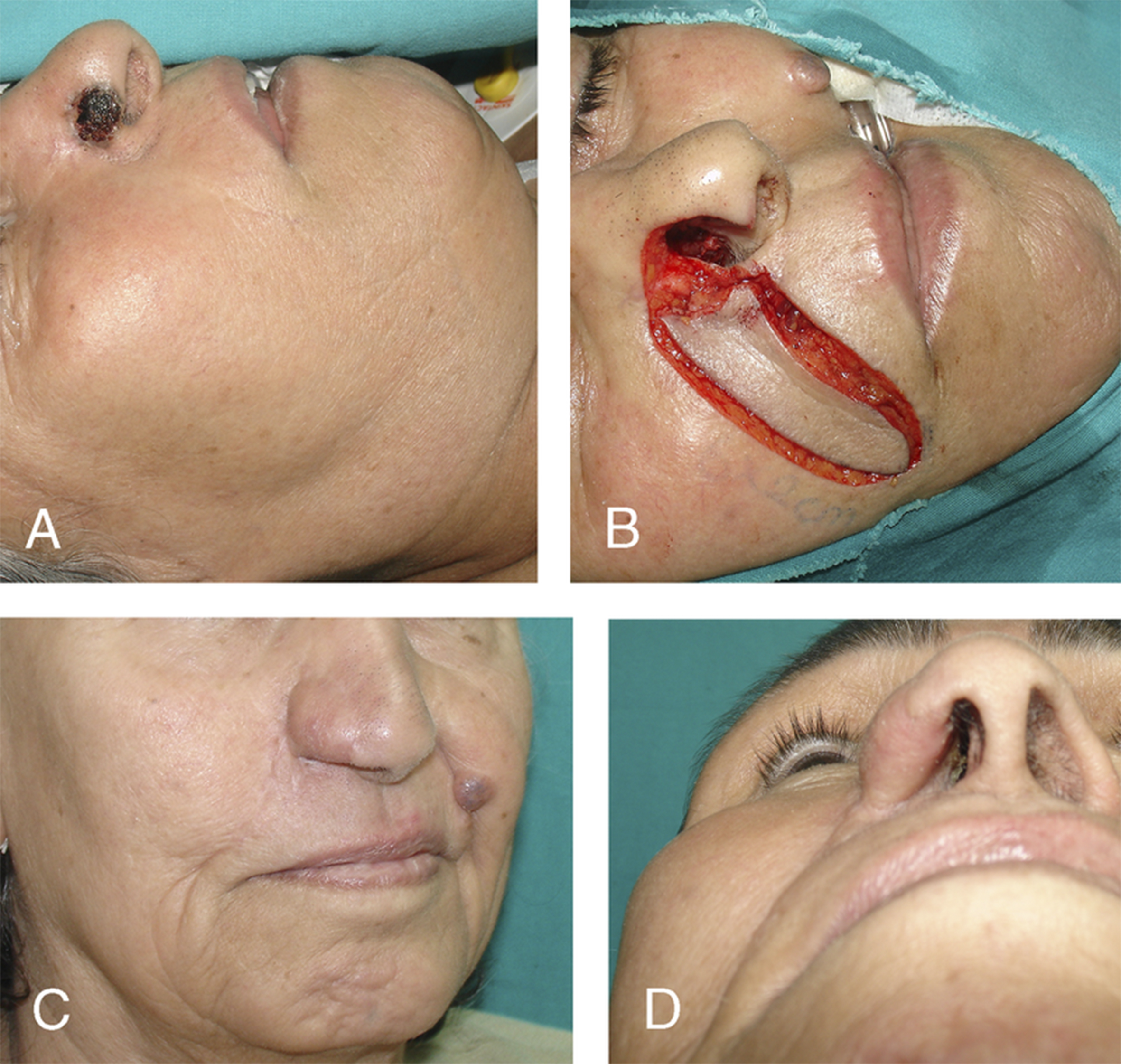
Bipedicled advancement flaps of residual vestibular lining can be incised at the intercartilaginous line and advanced inferiorly to line the nostril margin [Figure 2][4]. These flaps are based on the septum medially and the alar base laterally. Their donor defect can be skin grafted, resurfaced with another lining flap or can be left to heal secondarily[2].
Figure 2. (A) Bipedicled flap created from marginal and intercartilaginous incision; (B) Anteriorly based ipsilateral septal hinge flap; (C) Contralateral septal hinge flap utilized by creating an ipsilateral incision along septum and pulling contralateral mucoperichondrium through incision; (D) Contralateral septal cartilage can also be harvested to provide sidewall support. Reprinted with permission from[4]. Copyright © 2026 American Society of Plastic Surgeons.
Regional flaps
Septal mucosal flaps
Septal mucosal flaps are the workhorses for intermediate-sized nasal lining reconstructions [Figure 2][4]. These flaps are thin, well-vascularized, and offer a broad arc of rotation, capable of reaching the columella, nasal tip, or alar rim. They can be tunneled or transposed through pre-drilled cartilage windows to access anterior or lateral defects[4,12,23]. The three dominant pedicle orientations - anterior, superior, and posterior - enable tailored reconstruction: anteriorly based flaps (facial artery supply) are ideal for anterior and columellar defects; superiorly based flaps (ethmoidal branches) reach the upper vault; and posteriorly based flaps (sphenopalatine supply) are well suited to posterior and lateral reconstructions.
A generous amount of mucosa can be harvested making it a reliable donor site for lining replacement[12]. In cases where bilateral septal flaps are harvested, a midline dorsal strut must be preserved to maintain septal integrity and prevent postoperative nasal collapse[9]. The main disadvantage is donor site morbidity with crusting and nasal airway obstruction, as well as risk of septal perforation.
Inferior turbinate flaps
Inferior turbinate flaps are robust mucoperiosteal flaps supplied by the sphenopalatine artery if pedicled posteriorly, and the angular artery if pedicled anteriorly. They are ideal for posterior or lateral nasal wall defects and can be harvested endoscopically, though donor site crusting and atrophy may occur[3]. After medialization of the turbinate, subperiosteal elevation - facilitated by “postage stamping” the conchal bone - yields approximately 3-8 cm2 of thin, pliable mucosa that can be advanced anteriorly[3]. The nasal floor mucosa may also be raised in continuity with the inferior turbinate, which can increase the flap width for combined columellar and alar defects[1]. Survival of the flap may be improved by avoiding the overdrying effect of nasal airflow in the early post-operative period, employing light postoperative packing and emollients such as petroleum jelly for several weeks after inset[3]. Limitations include temporary nasal obstruction, possibly secondary procedures for pedicle division and a risk for post-operative atrophic rhinitis. Some component of atrophic rhinitis is expected due to removal of conchal bone and inferior turbinate mucosa for flap harvest; diligent humidification and postoperative stenting help mitigate these issues[3].
Pericranial flaps (PCFs)
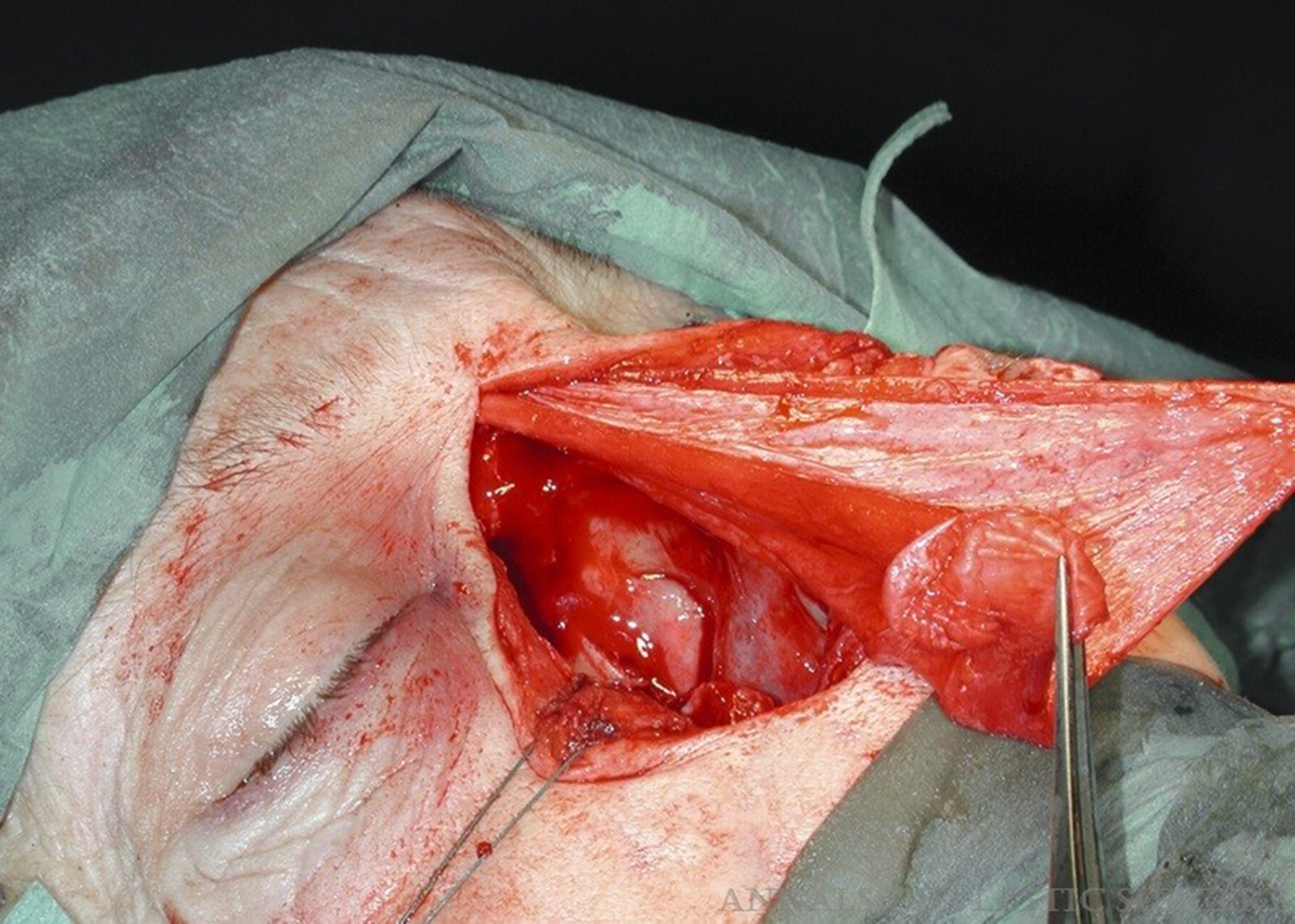
Pericranial flaps (PCFs) are broad, thin, and pliable axial flaps composed of the forehead periosteum and overlying galea[Figure 3][24]. They are based on the ipsilateral deep branches of the supratrochlear and supraorbital arteries, providing a reliable vascular supply. The flap is typically templated and elevated with a 2.5 cm base located approximately 2.2 cm from the midline at the level of the superior orbital rim. Care is taken to avoid crossing the midline during harvest, as the flap relies on a unilateral pedicle, although it may be transposed to either side of the nose for reconstruction.
Once elevated, the PCF is tunneled subcutaneously through the nasal root and inset into the recipient site[25]. Its arc of rotation allows it to extend 8–12 cm superiorly, enabling coverage of distal nasal subunits, including the tip and columella[24,26]. PCFs may also be elevated endoscopically, offering a minimally invasive alternative[27,28].
In select cases, PCFs have been used in combination with a contralateral paramedian forehead flap (PMFF), facilitated by a hemicoronal incision extending between the superomedial forehead defect and the ipsilateral temporal line. Functionally, PCFs and other non-cutaneous vascularized flaps (e.g., fascia, perichondrium, and muscle) do not directly replace nasal mucosa but instead serve as a vascularized scaffold that supports secondary mucosalization[29].
Figure 3. Pericranial flap for internal lining. Reprinted with permission from[24]. Copyright © 2025 Wolters Kluwer Health, Inc.
Facial artery musculomucosal (FAMM)
Facial Artery Musculomucosal (FAMM) flaps offer a mucosalized surface but are often too thick for internal nasal use. FAMM flaps are axial pattern flaps based on the facial artery, that can be transposed to the nasal cavity to provide moderate quantities of vascularized mucosal tissue. This is a pedicled flap involving oral mucosa, buccinator muscle, and facial artery that’s tunneled under the lip for inset into the nasal cavity[30]. These have been successfully used in Wegener’s noses where there is a significant lack of healthy intranasal mucosa. The major disadvantage to this flap is bulk, limited reach and the inability to line the entire nose[9].
Melolabial flap
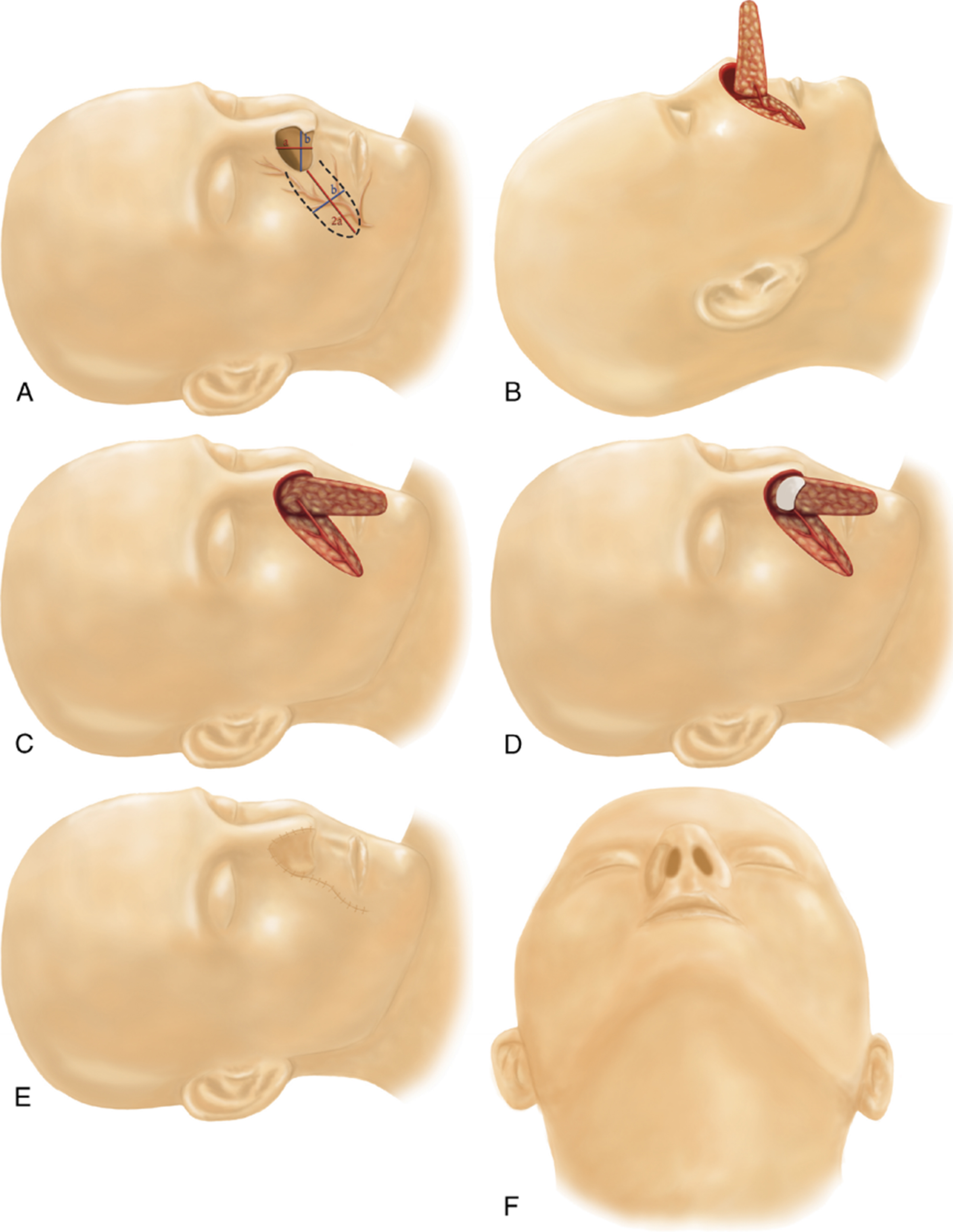
Melolabial flap (also referred to in some resources as “nasolabial flap”)[31,32] is a pedicled random-pattern flap based primarily on branches of the angular artery [Figures 4 and 5][31]. It is particularly useful for reconstruction of alar defects and can be employed for either external or internal nasal lining reconstruction. For internal lining, the cutaneous surface of the flap is inverted to face the nasal cavity. Melolabial flaps can be completed either as single-stage island flaps or as interpolated flaps requiring subsequent division of the pedicle.
Figure 4. (A) Design of the nasolabial flap. Flap width is matched to the vertical height of the defect, and flap length is planned to be at least twice the defect width; (B) Elevation and mobilization of the flap on its perforator; (C) After the medial aspect of the flap is inset internally to create the nasal lining, the flap is rotated approximately 90 degrees for closure of the external defect; (D) Placement of septal cartilage support onto the flap in cases requiring structural reinforcement; (E) Inset view demonstrating the medial edge of the flap folded over the cartilage graft, with primary closure of the donor site visible; (F) Inferior view of the reconstructed alar subunit following completion of reconstruction. Reprinted with permission from[31]. Copyright © 2025 Wolters Kluwer Health, Inc.
Figure 5. (A) Preoperative appearance of the skin tumor on the right ala; (B) Post-excision defect with planning of the reconstructive flap; (C) Appearance of the repair following reconstruction with a cartilage-supported nasolabial perforator flap, shown at 12 months postoperatively; (D) Inferior view of the reconstructed ala showing good contour and symmetry. Reprinted with permission from[31]. Copyright © 2025 Wolters Kluwer Health, Inc.
Although large nasal defects can be lined with melolabial flaps, the technique is limited by the maximum flap width that can be harvested without causing significant cosmetic distortion - typically around 3 cm[32]. For moderate-sized defects, both the external cover and internal lining can be reconstructed using a single melolabial flap. Depending on flap design, either the distal or proximal portion of the flap can be used for internal lining[32].
If the alar rim has been preserved, partial de-epithelialization of the melolabial flap can allow simultaneous reconstruction of both the internal lining and external skin envelope. When attempting to increase the arc of rotation, a handheld Doppler can be used to design the flap as a perforator-based island flap[31].
The principal disadvantage of the melolabial flap is its reliance on the subdermal plexus rather than a true axial vessel, which increases the risk of partial flap necrosis. For this reason, it is generally contraindicated in patients who are actively smoking or have significant microvascular disease[33]. This flap often incorporates hair bearing skin which can cause intranasal hair growth and patient discomfort.
PMFF
Folded
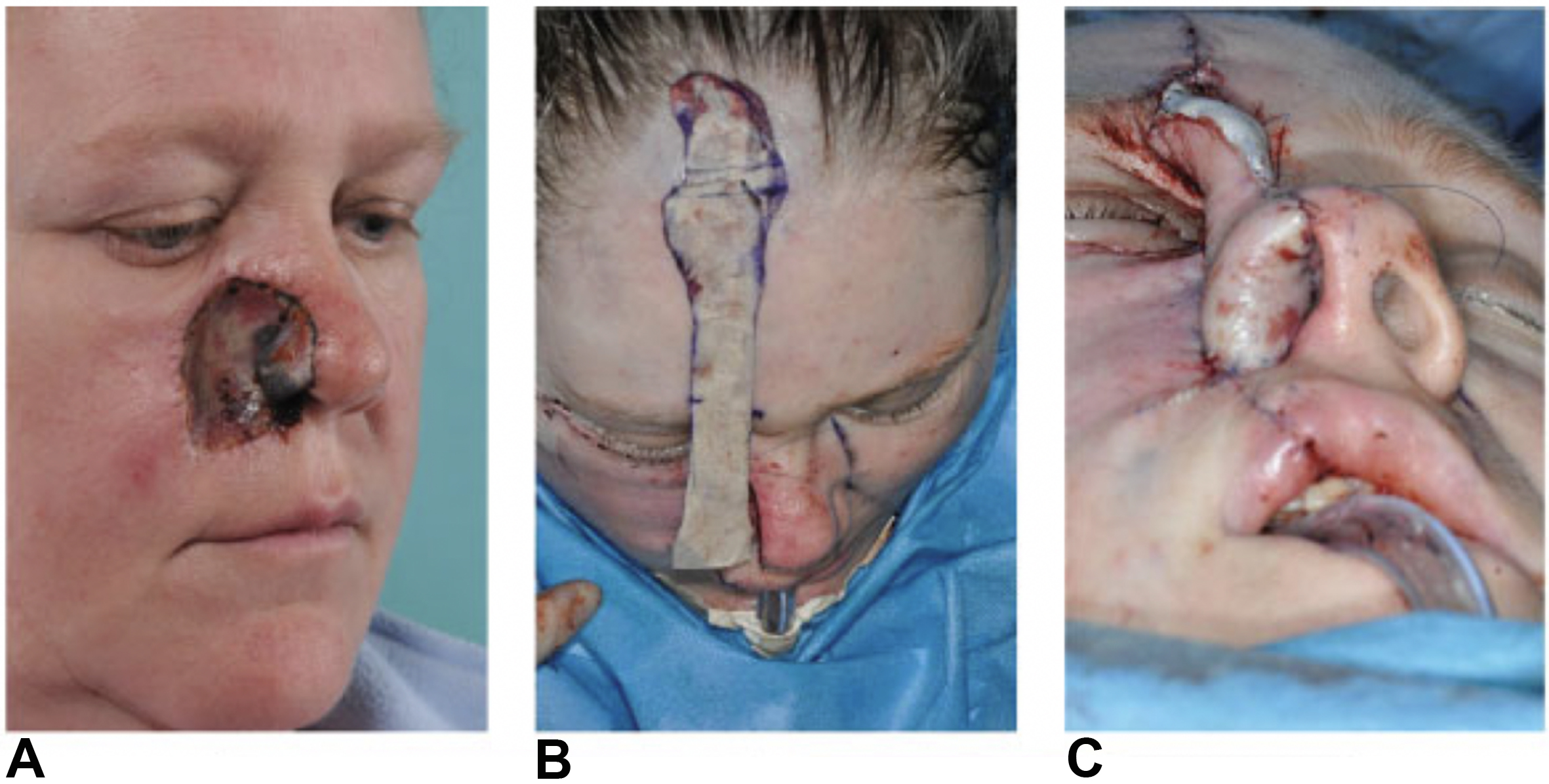
PMFF can be folded to simultaneously reconstruct both the internal lining and external skin envelope [Figure 6][12]. An exact template of the missing lining is created and placed distally along the trajectory of the supratrochlear artery, with 3-5 mm of additional tissue between the lining and skin templates to allow for effective folding. The internal portion of the flap is secured to the residual intranasal lining, and the flap is then folded, with the external component sutured to the surrounding skin. This technique typically requires an intermediate stage for flap thinning, contouring, and the addition of structural support as needed[2,4,12].
Figure 6. (A) Defect of the right lateral nose, medial cheek and upper lip; (B) Creating a template for external and internal lining, with an extra 3-5 mm for folding; (C) Inset of folded paramedian forehead flap reconstructing nasal ala. Reprinted with permission from[12]. Copyright © 2026 Georg Thieme Verlag KG.
Prelaminated
Prelamination of a paramedian forehead flap can be used to reconstruct both the external skin and internal lining in cases where local intranasal options are unavailable or insufficient. Templates are created for both the external skin and internal lining defects. A full- or split-thickness skin graft is harvested - commonly from the postauricular, supraclavicular or thigh regions - and secured to the undersurface of a full-thickness forehead flap in the location corresponding to the future internal lining. The flap is then laid back down onto the forehead and allowed to mature in situ for approximately four weeks, providing time for the graft to vascularize and adhere. After this period, the prelaminated flap is elevated and transferred to the nasal defect, allowing for single-stage reconstruction of both the internal lining and external skin envelope[4].
Folded vs. prelaminated PMFF
The folded PMFF provides immediate two-layer reconstruction by turning the distal portion of the flap inward to serve as lining while the external surface restores cutaneous cover. This technique is relatively straightforward but tends to create bulk intranasally, as adequate thinning is not possible at the initial stage. Therefore, these flaps involve, an intermediate stage of thinning and delayed placement of structural grafts, typically prior to pedicle division. Advocates of this technique suggest that with these additional stages, appropriately thin intranasal lining can be achieved[22].
By contrast, prelamination involves inset of a skin graft onto the undersurface of the forehead flap prior to transfer, which allows a thinner, vascularized lining to be delivered in a single stage. However, this approach requires additional time for graft maturation before transfer and enlarges the donor-site footprint. In practice, folded flaps are favored for their reliability and versatility in large defects, acknowledging the morbidity of prolonged pedicle duration and potential for transferring hair-bearing skin into the nose. In contrast, prelaminated flaps are selected when minimizing intranasal bulk and permitting earlier grafting of cartilage or bone is prioritized, accepting the trade-off of a staged delay and an additional donor site for skin grafting.
Second
In select cases - particularly in patients with extensive intranasal scarring, prior radiation therapy, or a smoking history precluding local flaps or skin grafts - a second PMFF may be used exclusively for internal lining reconstruction. While effective, this approach significantly increases the donor site defect, can result in more prominent forehead scarring, and may preclude the use of another forehead flap for future reconstruction in cases of recurrence or salvage[2,30].
Free tissue transfer
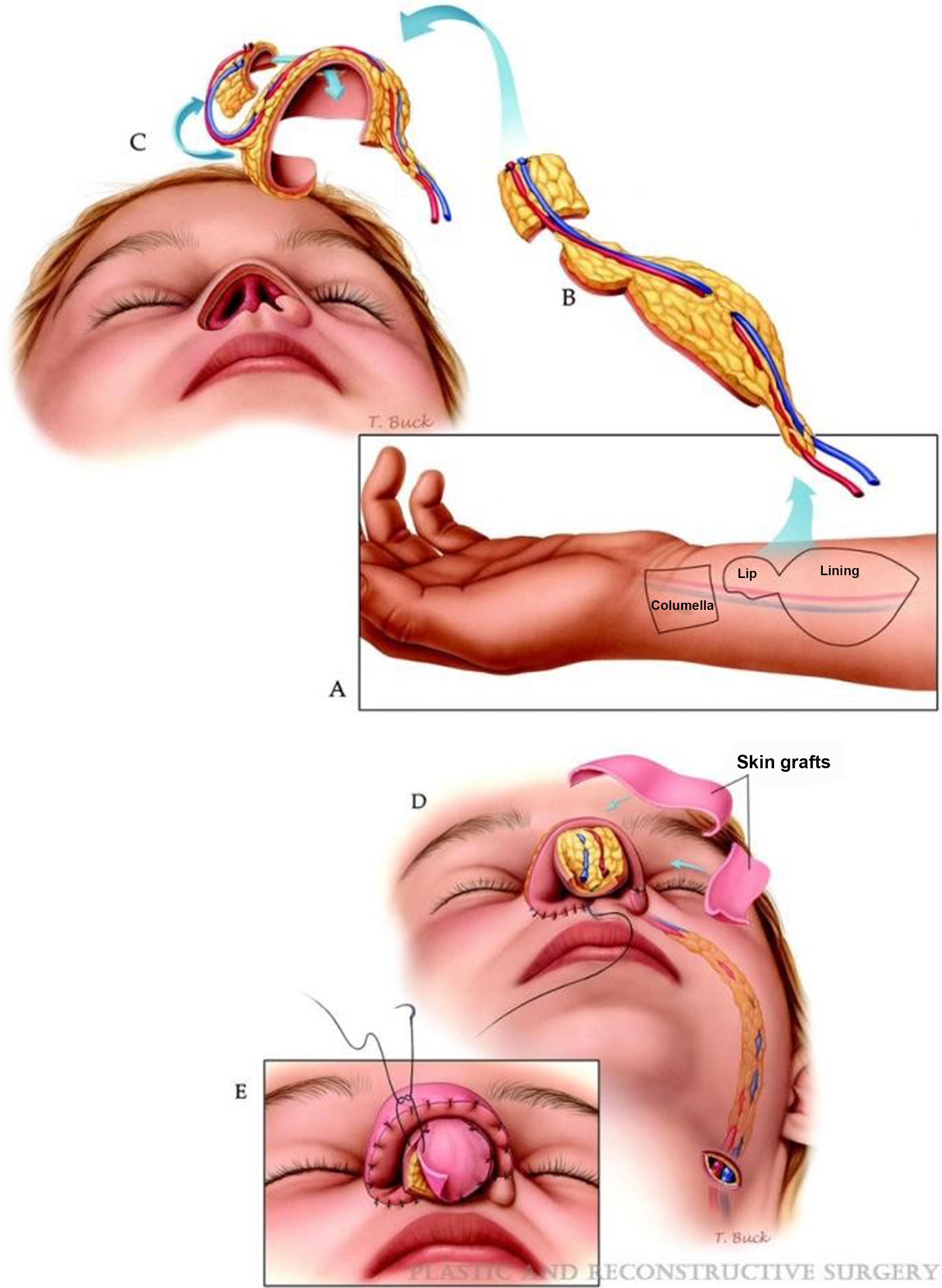
Microvascular free tissue transfer is an essential option for reconstructing extensive or composite nasal lining defects, particularly when local or regional tissues are unavailable or unreliable due to prior radiation, trauma, intranasal drug use, or oncologic resection [Figure 7][34]. Among available options, the RFFF remains the most widely used due to its thin, pliable, well-vascularized skin and long vascular pedicle[5,34]. Its versatility allows for reconstruction of the nasal vestibule, columella, floor, and even adjacent lip or cheek defects. RFFF can also be harvested as an osteocutaneous flap when bony reconstruction of the nasal dorsum is needed, such as in total or subtotal rhinectomy.
Figure 7. (A) Radial forearm free flap is designed for repair of nose and upper lip with three islands; (B) Flap is raised with pedicle; (C) Flap is oriented appropriately for inset; (D) The vascular pedicle is tunneled through the cheek and its vessels are connected to the facial vessels; (E) full thickness skin grafts are applied to the exposed fat of the paddles. Reprinted with permission from[34]. Copyright © 2025 Wolters Kluwer Health, Inc.
The flap can be folded, tubed, or designed with multiple skin paddles to conform to complex internal nasal geometry while maintaining airway patency. Careful positioning of the flap behind the pyriform aperture helps prevent intranasal bulk and airflow obstruction[34]. In selected cases, prelamination on the forearm with full-thickness skin grafts and cartilage grafts allows for the creation of a vascularized, anatomically contoured airway unit. While this technique can provide excellent outcomes, it requires multiple stages and longer recovery times[5].
Microsurgical access to the nasal cavity is typically achieved via a transnaris approach or re-elevation of a prior forehead flap. Vascular anastomoses are most often performed to the facial artery and vein through a small submandibular incision.
Other free tissue transfers have also been described in the literature. Potential sources of vascularized tissue for full-thickness defects or total nasal reconstruction are wide-ranging and include auricular helical rim, ALT, first dorsal metacarpal, dorsalis pedis, and femoral condyle flaps[29,35]. Other authors have suggested that free fascial flaps, which allow for mucosalization of the fascial surface, may be optimal for reconstructing the intranasal lining[29]. The free fascia lata flap as described by Seth et al. has the advantage of a discreet donor site, thin fascial material to replicate nasal lining, while also being able to be harvested simultaneously with ALT for cases requiring additional tissue (including restoration of bulk to midface or skull base)[36]. Recently, the medial femoral condyle corticoperiosteal flap has been championed for internal lining when bony structural support is also desired. Use of the periosteum allows for mucosalization of the internal lining, and external coverage can be provided by a traditional paramedian forehead flap in staged fashion[29].
Among free flap options, the RFFF remains the most versatile, offering a long pedicle, thin pliable skin, and reliable mucosalization, though donor-site morbidity with forearm grafting is a drawback. The ALT flap provides abundant tissue and the option of harvesting fascia lata; however, its bulk can obstruct airflow unless carefully thinned, and pedicle length may be shorter than the RFFF. The medial femoral condyle (MFC) flap is valuable when vascularized bone and lining are both needed, though its technical demands and limited harvest size constrain routine use. Fascia-only flaps (such as free fascia lata) offer the thinnest option with excellent mucosalization and discreet donor sites, but they lack immediate structural support. In terms of design, folded flaps are favored when opposing intranasal surfaces must be resurfaced, while tubed flaps are advantageous for reconstructing neovestibular airways or circumferential lining defects. Selection therefore depends on balancing defect geometry with the trade-offs of flap thickness, vascular reach, and donor-site morbidity.
Although microvascular reconstruction is technically challenging and requires longer operative time and hospitalization, it remains the most reliable option for restoring nasal lining in large, radiated, or scarred defects. When combined with thoughtful structural support and external coverage, free flap reconstruction can achieve excellent long-term airway function and aesthetic outcomes.
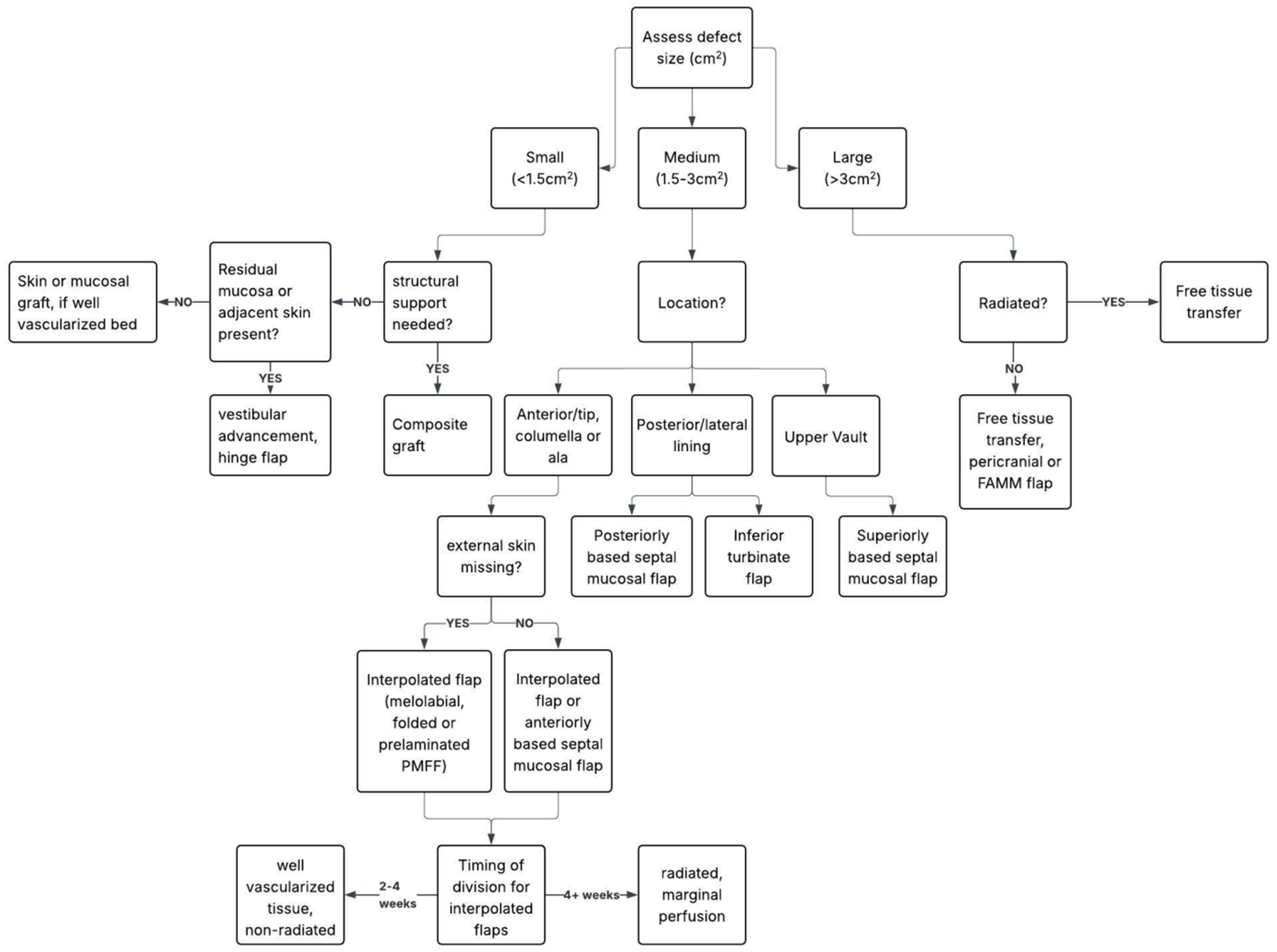
DECISION MAKING ALGORITHMS
Matching the defect to the most appropriate lining technique is essential for success. See decision making flowchart below [Figure 8]. The decision-making algorithm provides a stepwise framework for selecting the most appropriate lining technique based on defect size, location, depth, and patient-specific factors such as prior radiation or available donor tissue.
OUTCOMES AND COMPLICATIONS
Across reconstructive categories, traditional mucosal flaps (septal and turbinate) continue to demonstrate the most favorable balance of long-term airway patency, low bulk, and minimal donor morbidity, particularly when compared with fascia-based and free flap options. Fascia-only flaps and fasciocutaneous free flaps expand possibilities for large or radiated defects but introduce trade-offs in donor-site burden, intranasal bulk, and variability in mucosalization time. Although formal patient-reported outcome data remain limited, available series consistently note greater comfort and reduced crusting with mucosalized intranasal lining compared with skin-based or fasciocutaneous alternatives.
Septal and inferior turbinate mucosal flaps generally demonstrate high viability (> 90%-95% in most series) with durable airway patency and low revision needs when structural support is adequate; donor morbidity is typically limited to transient crusting and congestion[3,4]. Folded PMFF are reliable for large full-thickness defects but often require staged thinning due to bulk and may increase intranasal crusting compared with mucosa[6]. Among free options, the RFFF offers very high survival (> 95% in experienced centers) and excellent conformability (folded or tubed), balanced against forearm donor-site grafting and potential sensory changes[5]. Fascia-based approaches (e.g., ALT fascia lata) minimize intranasal bulk and allow secondary mucosalization, which can be advantageous in tight airways or radiated beds, though mucosalization time varies by tissue quality[36]. The medial femoral condyle corticoperiosteal flap is useful when vascularized bone and lining are both required, but harvest is technically demanding and flap size is limited[31]. Technique selection should weigh bulk vs. mucosalization behavior, pedicle length/reach, expected donor morbidity, and whether a folded (dual-surface) or tubed (circumferential airway) design best matches the defect.
Taken together, these comparative trends underscore that outcomes of nasal lining reconstruction depend on flap viability, tissue match, and surgeon experience. Successful lining provides long-term airway patency, supports framework, and resists contracture. When complications occur, they can be difficult to manage due to the complex 3D anatomy of the nose and the relative scarcity of nearby tissue for salvage. The emotional toll (to both patient and surgeon) of complications, especially if they require re-operation, is often significant. Nonetheless, even in the context of complications, successful salvage of the reconstruction is often possible with thoughtful, patient management.
In addition to these considerations, long-term functional performance of the reconstructed lining is central to defining success. Optimal outcomes depend on maintaining low airflow resistance, preserving mucociliary clearance, and preventing contracture that narrows the nasal airway over time. Mucosalized flaps, such as septal or turbinate mucosa, tend to demonstrate superior humidification and ciliary function compared with skin-based or fasciocutaneous options, which may contribute to patient-reported improvements in breathing comfort. Long-term patency can still be threatened by scarring, stenosis, or intranasal bulk, underscoring the importance of vigilant postoperative stenting and humidification. However, objective assessments of nasal airflow and mucociliary function remain inconsistently reported in the literature, highlighting a need for standardized functional outcome measures in future studies.
To improve consistency and comparability in future research, adoption of standardized outcome measures is strongly recommended. These should include validated patient-reported instruments assessing nasal obstruction and quality of life, objective functional assessments of airflow (such as rhinomanometry or acoustic rhinometry), and standardized reporting of complication profiles, need for revision, and time to mucosalization. Routine incorporation of such tools would enhance the reliability of outcome reporting, facilitate meaningful comparison across reconstructive techniques, and strengthen the evidence base guiding nasal lining reconstruction.
Complications include:
1. Flap/graft necrosis
(1) Risk factors[4,37]:
- Prior radiation to the surgical field (recipient or donor site)
- Extensive scar tissue
- History of alloplastic implant
- History of prior reconstructions
- Previous infection
- Smoking
- Pedicle occlusion/poor geometry
(2) Treatment:
- Debridement of affected tissue
- Removal of cartilage grafts in the area
- Full thickness skin graft if healthy underlying vascular bed (e.g. full-thickness forehead flap)
- May require unplanned surgeries or delay of planned subsequent stages
2. Intranasal crusting
(1) Less common when lining repaired with mucosal source, rather than skin[29]
(2) Treatment with standard nasal hygiene protocols
3. Delayed healing
4. Obstruction or stenosis
(1) May need to operatively debulk, or can trial conservative measurements such as nasal cones during healing
5. Infection
(1) Risk factors[38]:
- Graft or flap necrosis
- History of prior infection [especially methicillin resistant staph aureus (MRSA)]
- Break in sterile technique
- Patient factors (e.g., nose blowing)
(2) Treatment[38]:
- Culture-directed antibiotics
- Drainage/debridement as needed
6. Poor cosmesis due to contraction, asymmetry, or flap bulk
7. Donor site morbidity
(1) Need for additional grafting to donor site
(2) Nasal septal perforation
(3) Intranasal synechiae
(4) Crusting or empty nose from turbinate flaps
(5) Numbness
(6) Alopecia
SALVAGE AND REVISION STRATEGIES IN HIGH-RISK FIELDS
Reconstruction in previously irradiated, infected, or multiply operated noses is especially challenging due to impaired vascularity, scarring, and limited local tissue options. In these settings, reliance on vascularized mucosa or fascia-based flaps is strongly preferred over non-vascularized grafts, which can have poor survival. A delayed or staged approach may improve flap reliability, allowing vascular conditioning before definitive inset[4]. For large or composite salvage defects, free tissue transfer (e.g., RFFF, ALT with fascia lata, or fascia-only flaps) often provides the most dependable lining with sufficient vascular supply[5,36]. When the external skin envelope is tenuous, negative-pressure dressings can be used as a temporizing measure to optimize the wound bed before definitive coverage. Although there is no direct literature supporting negative-pressure therapy in nasal reconstruction, it is well studied in other domains of head and neck reconstruction, where it has demonstrated improved wound conditioning and reduced infection rates; it therefore represents a reasonable and potentially valuable adjunct in this setting[39]. Surgeons should anticipate a higher likelihood of revision procedures, maintain a low threshold for re-stenting to prevent stenosis, and plan reconstructions with maximal redundancy of vascularized tissue. This strategy emphasizes durability and airway preservation while accepting the trade-off of additional stages and donor-site morbidity.
PERI AND POST-OPERATIVE PROTOCOLS
Successful nasal lining reconstruction depends not only on flap selection but also on meticulous peri- and postoperative care. Patients are routinely maintained on humidification and saline irrigations to prevent crusting and desiccation of the reconstructed lining. Topical decongestants may be used selectively to reduce edema and maintain airway patency, while antibiotic ointment applied to suture lines and intranasal bolster sites helps reduce bacterial burden. Intranasal splints such as Doyle stents or custom conformers are frequently employed to prevent synechiae formation and preserve airway contour; these are typically left in place for 2-3 weeks, with longer duration considered in radiated fields or when vascular delay is desired. Pedicle division for interpolated flaps is generally performed at 3-4 weeks, though delayed division beyond 4 weeks may be necessary in compromised vascular beds. Postoperative scar care - including silicone sheeting, massage, and sun protection - is emphasized to optimize external cosmesis. In cases of incipient synechiae, early recognition and office-based lysis with re-stenting is critical to avoid long-term stenosis. A structured postoperative regimen therefore plays a central role in preserving both function and form after lining reconstruction.
INNOVATIONS AND FUTURE DESIGNS
Recent innovations
Recent innovations have the potential to significantly advance nasal lining reconstruction by improving surgical precision, reducing complication rates, and expanding reconstructive options for complex or high-risk patients.
3D surgical planning
Advances in imaging and 3D printing allow for preoperative flap shaping and intraoperative precision[40].
The complex 3-dimensional anatomy of the nose is among the facets that creates significant challenge for reconstruction. This includes both internal lining and external contour. When adequate tissue is present for comparison, it is common to mimic the anatomy of the contralateral side to create a template for missing anatomical subunits. However, when considering total or subtotal rhinectomy, it can be difficult to create fully absent anatomy. 3D printing based on either preoperative/pre-deformity imaging or based on generated data from other patients can help create a template when none exists on the surgical field.
When possible, the patient’s native anatomy should be utilized for 3D template creation[40]. Typically, computed tomography (CT) images are used to digitally create the 3D model. This can be printed to create a mold which can be sterilized and placed onto the surgical field. Templates can then be created in standard fashion from Telfa, suture foil, silicone sheeting, or other flexible materials in order to transpose them to the forehead for use. To date, this method has mostly been described for the creation of templates for external lining; however, 3D printed molds could be envisioned that might more fully mimic the desired shape of the internal lining, enabling more fidelity in design of internal lining flaps and grafts.
In cases where no pre-morbid CT exists, possibility of a “standardized” 3D printed mold or a “composite” mold based on a conglomerate of digital images could be considered. The 3D imaging files such as generated on the VECTRA M3 system (Canfield Scientific, Parsippany NJ, USA) or other 3D imaging systems can also be utilized to create 3D printed images, potentially omitting the need for ionized radiation when not otherwise clinically indicated[41].
Conversion of digital images, whether from CT [Digital Imaging and Communications in Medicine (DICOM)] or 3D imaging systems such as VECTRA, typically involves an intermediate step to create an Standard Tessellation Language (STL) file. Many research-quality 3D printers are able to directly print 3D models from a formatted STL file, and commercially available printers increasingly also have this capacity. However, specialized software may be required to complete the transition and subsequent modeling. Mold materials can range from traditional plastics/composites to medical-grade silicone, some of which can be sterilized for use intraoperatively.
Prefabricated flaps
Delayed flaps or staged composite grafts are under investigation for high-risk patients or irradiated fields. These advances may reduce complication rates, improve aesthetics, reduce operative time, and enable more single-stage reconstructions in the future.
Potential future directions
Tissue engineering
Research is ongoing into engineered mucosal grafts with pre-vascularized scaffolds seeded with autologous stem cells.
Tissue engineering for replacement of native tissue relies on the so-called “tissue engineering triad:” self-regenerating cells (stem cells), a scaffold, and induction signals[42]. A type of composite graft can be created in which appropriate cell types are implanted into a biomaterial that will subsequently degrade, which serves as the scaffold. This composite material can be further cultured or implanted directly into a defect as a graft. As the scaffold degrades, new extracellular matrix integrates and helps promote tissue growth[42].
Cell sheets of mesenchymal stem cells (MSCs) preserve the extracellular matrix in addition to the cells themselves, and have been successful in helping regenerate corneal, myocardial, periodontal, and bony tissues[42]. Human ethmoid sinus mucosa MSCs (hESMSCs) have been shown to possess multipotent differentiation as well as self-renewal, making them a promising source for tissue engineering[43]. In addition to their potential for regeneration, they may also demonstrate neuroprotective, anti-inflammatory, and immune reactions. hESMSCs have even demonstrated the ability to promote osteogenesis in calvarial defects in rat models. Maxillary sinus mucosal stem cells have also shown promise, but have slightly less proliferation capacity and colony-forming propensity[43]. To date, no literature exists on the direct application of hESMSCs cell sheets for repair of nasal lining defects, but there is great potential for further investigation in this area.
Human amnion contains multipotent/pluripotent stem cells and is a promising potential source for regeneration of nasal lining[44]. Amnion is not vascularized, but is nonetheless highly metabolically active. When applied, it prevents desiccation of the wound bed, promotes epithelialization, and inhibits scarring. It therefore may be used to enhance flap/graft survival or may serve as a scaffold for ingrowth of mucosal tissue. In addition, it exhibits anti-inflammatory and anti-bacterial properties. Limitations exist currently on expanding the applications of human amnion, largely based on ethical/religious and financial barriers.
Engineered tissue poses exciting potential but is as yet not well vetted in clinical practice. Potential limitations to their usefulness include lack of evidence on the viability of prevascularized scaffold in irradiated beds, inconsistent time to mucosalization compared to traditional skin grafts, and high associated cost and resource use. However, the benefits of providing mucosal tissue for lining, particularly for extensive defects where mucosal donor sites are lacking, make engineered tissue replacements a worthwhile area of continued investigation.
Synthetic scaffolds and biomaterials
Synthetic scaffolds and biomaterials, such as collagen-elastin matrices and nanofiber scaffolds, are being evaluated for other mucosal applications and may offer off-the-shelf alternatives in nasal reconstruction. These materials aim to replicate native extracellular matrix architecture and support host cell ingrowth and neovascularization. Their potential to reduce donor site morbidity and expand options for salvage cases warrants further study[45].
Minimally invasive flap harvest techniques
Minimally invasive flap harvest techniques, including endoscopic or robotic-assisted dissection of septal, inferior turbinate, or PCFs, may reduce donor site morbidity and improve precision in flap inset. Endoscopic-assisted pericranial flap elevation has already been described and may be particularly useful in patients undergoing simultaneous intracranial procedures or in cases requiring minimally disruptive access[25].
Looking forward, further development of engineered mucosal scaffolds will likely require stepwise preclinical validation, including assessment of vascular integration, mucociliary function, and long-term resistance to contraction in animal models. As digital design continues to mature, integration with robotic or endoscopic-assisted flap harvest may offer greater precision in septal or turbinate flap elevation while minimizing donor morbidity. A potential future workflow could involve preoperative imaging, STL-based digital modeling, and intraoperative use of printed templates to guide flap shaping and inset. These concepts remain investigational but outline a feasible roadmap for the next phase of innovation in nasal lining reconstruction. Future research should focus on long-term functional outcomes, patient-reported satisfaction, and cost-effectiveness across reconstructive strategies.
CONCLUSION
Reconstruction of the nasal lining in full-thickness defects remains one of the most complex and technically demanding aspects of facial reconstruction. Successful outcomes hinge on careful assessment of defect size, location, and patient-specific factors such as prior radiation or surgical history. A wide range of reconstructive options - ranging from local mucosal flaps to microvascular free tissue transfer - offers versatility, but each carries distinct advantages and limitations. Emerging technologies, such as 3D surgical planning and tissue-engineered grafts, may further improve surgical precision, reduce donor site morbidity, and expand options for high-risk patients.
This review underscores the importance of restoring a thin, well-vascularized, and durable lining to support nasal function and aesthetic outcome. Future research should focus on optimizing flap design, expanding the application of regenerative materials, and developing standardized outcome measures to refine techniques and enhance patient care.
DECLARATIONS
Authors’ contributions
Contributed to the conception and design of the manuscript, conducted literature review, and were involved in drafting and revising the article for intellectual content: Gossett KA, Waters CM
Read and approved the final manuscript: Gossett KA, Waters CM
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (Open AI) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
None.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
3. Murakami CS, Kriet JD, Ierokomos AP. Nasal reconstruction using the inferior turbinate mucosal flap. Arch Facial Plast Surg. 1999;1:97-100.
5. Walton RL, Burget GC, Beahm EK. Microsurgical reconstruction of the nasal lining. Plast Reconstr Surg. 2005;115:1813-29.
7. Tollefson TT, Kriet JD. Complex nasal defects: structure and internal lining. Facial Plast Surg Clin North Am. 2005;13:333-43.
8. Sozansky J, Houser SM. The physiological mechanism for sensing nasal airflow: a literature review. Int Forum Allergy Rhinol. 2014;4:834-8.
9. Cannady SB, Cook TA, Wax MK. The total nasal defect and reconstruction. Facial Plast Surg Clin North Am. 2009;17:189-201.
11. Burget GC, Menick FJ. The subunit principle in nasal reconstruction. Plast Reconstr Surg. 1985;76:239-47.
12. Haack S, Fischer H, Gubisch W. Lining in nasal reconstruction. Facial Plast Surg. 2014;30:287-99.
13. Goreshi R, Hendi A. Tissue conserving technique for reconstruction of nasal lining defects. Dermatol Surg. 2016;42:124-6.
14. Iacobucci JJ, Stevenson TR. Bolster dressing to support a full-thickness skin graft. Ann Plast Surg. 1987;18:550-1.
15. Hatoko M, Tanaka A, Kuwahara M, Tada H, Imai K, Muramatsu T. An evaluation of hard palate mucosa graft as a lining material in alar reconstruction: a 7-year experience applied to the full-thickness alar defect. Plast Reconstr Surg. 2000;105:1940-7.
16. Teltzrow T, Arens A, Schwipper V. One-stage reconstruction of nasal defects: evaluation of the use of modified auricular composite grafts. Facial Plast Surg. 2011;27:243-8.
17. Keck T, Lindemann J, Kühnemann S, Sigg O. Healing of composite chondrocutaneous auricular grafts covered by skin flaps in nasal reconstructive surgery. Laryngoscope. 2003;113:248-53.
18. Bashir MM, Khan BA, Abbas M, Khan FA. Outcome of modified turn in flaps for the lining with primary cartilage support in nasal reconstruction. J Craniofac Surg. 2013;24:454-7.
19. Hessam S, Sand M, Georgas D, Kassa T, Bechara FG. Scar hinge flap for inner lining reconstruction of full-thickness defects on the ala of the nose. Rhinology. 2015;53:167-70.
20. Lee KK, Gorman AK, Swanson NA. Hinged turnover flap: a one-stage reconstruction of a full-thickness nasal ala defect. Dermatol Surg. 2004;30:479-81.
21. Ulug BT, Kuran I. Nasal reconstruction based on subunit principle combined with turn-over island nasal skin flap for nasal lining restoration. Ann Plast Surg. 2008;61:521-6.
22. Menick FJ. Nasal reconstruction: art and practice. Elsevier Health Sciences; 2008. Available from: https://www.amazon.com/Nasal-Reconstruction-Practice-Frederick-Menick-ebook/dp/B005IX89DE. [Last accessed on 18 Mar 2026].
23. Baker S. Local flaps in facial reconstruction. Elsevier/Saunders; 2014. Available from: https://www.sciencedirect.com/book/edited-volume/9780323036849/local-flaps-in-facial-reconstruction. [Last accessed on 18 Mar 2026].
24. Sertel S, Pasche P. Pericranial flap for inner lining in nasal reconstruction. Ann Plast Surg. 2016;77:425-32.
25. Farias N, Moore C. Full thickness nasal reconstruction with paired pericranial and paramedian forehead flaps. J Craniofac Surg. 2024;Epub ahead of print.
26. Yoshioka N, Kishimoto S. Anteriorly based pericranial flap: an anatomic study of feeding arteries. Skull Base Surg. 1991;1:161-4.
27. Bastaninejad S, Karimi E, Saeedi N, Amirizad E. Endoscopic pericranial flap design for the restoration of nasal mid-vault lining defects. Int J Oral Maxillofac Surg. 2018;47:865-8.
28. Waters CM, Zanation AM, Thorp BD, Shockley WW, Clark JM. Repair of septal perforation with endoscopic-assisted pericranial flap harvest and open rhinoplasty approach. Facial Plast Surg Aesthet Med. 2020;22:225-6.
29. Howard BE, Patel S, Shockley WW, Clark JM. Total nasal reconstruction: advances in free tissue transfer for internal lining and structural support. Facial Plast Surg Clin North Am. 2024;32:247-59.
30. Rahpeyma A, Khajehahmadi S. Facial artery musculomucosal (FAMM) flap for nasal lining in reconstruction of large full thickness lateral nasal defects. Ann Med Surg. 2015;4:351-4.
31. Durgun M, Özakpınar HR, Selçuk CT, Sari E, Seven E, İnözü E. Repair of full-thickness nasal alar defects using nasolabial perforator flaps. Ann Plast Surg. 2015;75:414-7.
32. Iwao F. Alar reconstruction with subcutaneous pedicled nasolabial flap: difficulties, considerations, and conclusions for this procedure. Dermatol Surg. 2005;31:1351-4.
33. Yellin SA, Nugent A. Melolabial flaps for nasal reconstruction. Facial Plast Surg Clin North Am. 2011;19:123-9.
34. Burget GC, Walton RL. Optimal use of microvascular free flaps, cartilage grafts, and a paramedian forehead flap for aesthetic reconstruction of the nose and adjacent facial units. Plast Reconstr Surg. 2007;120:1171-207.
35. Antunes MB, Chalian AA. Microvascular reconstruction of nasal defects. Facial Plast Surg Clin North Am. 2011;19:157-62.
36. Seth R, Revenaugh PC, Scharpf J, Shipchandler TZ, Fritz MA. Free anterolateral thigh fascia lata flap for complex nasal lining defects. JAMA Facial Plast Surg. 2013;15:21-8.
37. Weber SM, Wang TD. Options for internal lining in nasal reconstruction. Facial Plast Surg Clin North Am. 2011;19:163-73.
39. Rosenthal EL, Blackwell KE, McGrew B, Carroll WR, Peters GE. Use of negative pressure dressings in head and neck reconstruction. Head Neck J Sci Spec Head Neck. 2005;27:970-5.
40. Ziegler JP, Oyer SL. Prelaminated paramedian forehead flap for subtotal nasal reconstruction using three-dimensional printing. BMJ Case Rep. 2021;14:e238146.
41. Denour E, Woo AS, Crozier J, Van Dongen C. The use of three-dimensional photography and printing in the fabrication of a nasal prosthesis. J Craniofac Surg. 2020;31:e488-91.
42. Scheller E, Krebsbach P, Kohn D. Tissue engineering: state of the art in oral rehabilitation. J Oral Rehabil. 2009;36:368-9.
43. Xie Q, Wang Z, Huang Y, et al. Characterization of human ethmoid sinus mucosa derived mesenchymal stem cells (hESMSCs) and the application of hESMSCs cell sheets in bone regeneration. Biomaterials. 2015;66:67-82.
44. Fairbairn NG, Randolph MA, Redmond RW. The clinical applications of human amnion in plastic surgery. J Plast Reconstr Aesthet Surg. 2014;67:662-75.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].