Polychlorinated biphenyls (PCBs) in water and sediments from the Udu River, Niger Delta, Nigeria: concentration, distribution and risk assessment
Abstract
In this study, five surface water and sediment samples were collected from five different points along the course of the Udu River, Niger Delta, Nigeria, which were near the human population. Water samples were liquid-liquid extracted with dichloromethane in a separatory funnel, while sediment samples were Soxhlet extracted using a mixture of acetone, dichloromethane, and n-hexane. Quantification of polychlorinated biphenyls (PCBs) in the extracts was done using gas chromatography-mass spectrometry. The ecological risk of PCBs was assessed by comparing the determined PCB concentrations with established guideline values, while health risk was evaluated using non-cancer and total cancer risk models. From the results obtained, only one congener (PCB-167) was present in water samples, with concentrations ranging from 20-1860 ng L-1. For sediment samples, 28 congeners were detected. The concentration of Ʃ28PCBs and the 12 dioxin-like PCBs (dl-PCBs) ranged from 5.34-16.1 and 1.07-5.36 ng g-1, respectively. The toxicity equivalence values for dl-PCBs varied from 0.0065-0.018. Compared to guideline values for both water and sediment, the PCB concentration obtained in this study does not pose any ecological risk at all except for one sampled point. Similarly, the hazard index values for non-cancer risk evaluation were < 1 at all but one point, while total cancer risk values were between 1 × 10-6 and 1 × 10-4 at all but one sampling point, indicating no potential risk of developing cancer associated with PCBs in water and sediments of the Udu River.
Keywords
INTRODUCTION
Polychlorinated biphenyls (PCBs) are a class of chemicals comprising a biphenyl ring structure with 2-10 chlorine atoms attached to it[1]. There are 209 congeners of PCBs, which can be grouped into two types based on their toxicological properties[2]. The first group comprises 12 congeners (PCBs 77, 81, 105, 114, 118, 123, 126, 156, 157, 167, 169, and 189) and are called dioxin-like PCBs (dl-PCBs) due to their similar toxicities with those of dioxins[3], e.g., immunotoxicity, developmental toxicity, carcinogenesis, neurotoxicity, etc.[4]. The second group of PCBs does not show any dioxin-like toxicity but exhibits other toxicological properties[5] such as neurobehavioral effects, including impaired auditory functions (PCBs 52 and 180) and altered sexually dimorphic behaviors (PCB-180)[6].
Anthropogenic activities such as emissions from commercial PCB products (e.g., agricultural chemicals and electrical equipment)[7] and industrial activities (thermal processes, and production of paints and chlorinated solvent)[8] are major contributors to PCBs in environmental matrices. However, PCBs have been found in remote areas devoid of industrial activities[9]. Even though PCBs were banned by the United Nations Stockholm Convention several years ago, they still find useful applications in many developing countries[10,11]. The PCBs in these developing countries originate mainly from electrical transformers containing PCB oil[12,13]. Thus, PCBs can become present in the environment through leakages and illegal dumping of PCB-containing equipment, transformer fluids, and emissions resulting from the incineration of PCB wastes[14]. Atmospheric transport is also reported to be the main distribution route for PCBs, carrying them great distances from the emission sources, where they are deposited on different environmental matrices, including soil, water, and biota[15-17].
The presence of PCBs in water bodies is an emerging issue for water quality[18]. This is because, once PCBs reach a lake or river, they concentrate in the sediments by attaching to the organic matter[19]. Aquatic organisms such as crustaceans, snails, and phytoplankton that live in the sediments ingest these PCB-contaminated organic materials[20], and fish can then accumulate PCBs by consuming these organisms[21]. Fish contaminated with PCBs can then be transferred to humans via ingestion; since PCBs are lipophilic, they continue to bioaccumulate in human fatty tissues with time. There are several reports on elevated PCB levels in water from Nigerian rivers[22-29]. Beyond Nigeria, elevated levels of PCBs have also been reported in surface water from the Bay of Bengal coast of Bangladesh[30] and rivers in Shanghai[31], while relatively low levels were reported in surface water of the Yangtze River Delta in China[32].
Accidental exposure or consumption of fish contaminated with PCBs has been reported to alter endocrine, immune and nervous systems, particularly in developing children[33,34]. Studies on both human and laboratory mammals provide strong evidence of the carcinogenic and reproductive toxicities of PCBs[35]. An approximate three-fold increase in the risk of developing breast cancer in postpartum women exposed to PCBs (PCBs 167, 187, and 203 in particular) has been reported[36]. Early PCBs exposure in humans may influence the development of breast cancer, thereby leading to harmful health consequences[37]. There is, therefore, the need for monitoring the levels of PCBs in aquatic environments and the probable risk to humans through either consumption of or dermal contact with contaminated water and sediments.
The Udu River is a large tributary of the Warri River in Nigeria’s Niger Delta region. It is essential to the inhabitants of Udu community, as it serves as a source of sea foods and water for farming, domestic, and recreational purposes. Although the microbial load[38] and physicochemical and heavy metals status[39] of the river water have been previously reported, data on the concentration and potential health risk of PCBs in the river are limited. Additionally, the fact that water from this river is largely used for domestic and recreational purposes, and the environmental persistence and bioaccumulation potential of PCBs, necessitated this study. Therefore, the objective of this study was to determine the concentration and distribution of PCBs in water and sediment samples from the Udu River. Risk exposure assessment was also carried out to evaluate the ecological and probable health risks to humans using the toxic equivalency and cancer and non-cancer risk models.
MATERIALS AND METHODS
Study area
The Udu River is situated in Udu town in Delta State, Nigeria, and extends more than 48 km2[38]. The river is sourced from Aboh and lies between longitude 6.20°N and latitude 5.45°E[39]. It flows southwest to Warri, where it joins several creeks, then empties into the brackish Forcados River, and finally into the Atlantic Ocean[40]. The river aids several communities, such as Ekete Waterside, Aladja, and Ovwian, where fishing is a major livelihood for residents of these communities. Activities such as washing clothes, bathing, swimming, dumping sawdust, and slaughtering cattle by the riverbank also take place. The area covered in this study spanned from the Udu Bridge down to a few meters after the jetty, where the most human activity takes place. Maps of Nigeria showing Delta State and the sampling points on the Udu River are shown in Figure 1A and B, respectively.
Chemicals and solvents
Dichloromethane (LC grade) and alumina were obtained from BDH (Poole, Dorset, UK). Acetone and n-hexane were purchased from Sigma-Aldrich (St. Louis, Missouri, USA). The mixed standard of surrogate
Sampling and sample preparation
Five samples of water and sediment were collected from five different points along the course of the Udu River. Sampling was done between 8:00 and 10:00 a.m., in April 2021, just after sunrise. A locally fabricated Ekman grab sampler was used for collecting sediment samples, while glass bottles were used for collecting water samples. Water samples were collected just below the water surface (up to 1 m deep) using pre-cleaned bottles. The samples were then transferred into pre-cleaned and labeled opaque glass bottles, which were also rinsed with the sampled water[41]. These were kept in an ice chest and transported to the laboratory. Collected sediment samples were placed in properly labeled aluminum foils, kept in an ice chest, and taken to the laboratory. Replicate samples were collected at each site, and the final sample was a composite of all replicate samples via thorough mixing. In the laboratory, water samples were preserved at a temperature of 4 °C. Sediment samples were dried in the open at ambient temperature, passed through a sieve (2 mm mesh), ground into smaller particles using an agate mortar and pestle, and refrigerated until analysis.
Extraction and clean-up of PCBs from water samples
Extraction of PCBs was done according to the USEPA Method 1668B[42]. First, a 200 mL of water sample was liquid-liquid extracted in a separatory funnel with 75 mL of dichloromethane (DCM). The funnel was agitated for 2 min with intermittent venting to release vapor pressure from the funnel. The organic layer was allowed to separate for 10 min and recovered into a 500 mL flask. The aqueous layer was re-extracted twice with 75 mL of the extracted solvent. The two extracts were combined and dried with anhydrous
Extraction and clean-up of PCBs from sediment samples
Extraction was carried out in accordance with the USEPA Method 3540C[43] using a Soxhlet apparatus. First, 10 g of sediment samples was extracted with 150 mL of a mixture of acetone, DCM and n-hexane (1:1:1 v/v) in a water bath at 65 °C for 18 h. The surrogate 13C12-labeled PCBs were also added to the samples prior to extraction. After extraction, 3 g of anhydrous Na2SO4 and 1 g of activated copper granules were added to remove water and sulfur, respectively. The extract was then concentrated to about 2 mL in a rotary evaporator. Clean-up of the extract was done in a similar manner as described above for water samples.
Quantification of PCBs
A gas chromatograph-mass spectrometer (Agilent 7890A, Agilent 5975C, Palo Alto, CA, USA) was used for the quantification of PCBs. The operating parameters of the equipment have been previously described[29]. The column type used was a DB-5 column (30 m length × 0.25 mm internal diameter, 0.25 μm film thickness). The carrier gas was high-purity helium gas flowing at a rate of 2 mL min-1. The initial temperature of the column was set at 80 °C for 2 min, then increased to 180 °C at a rate of 10 °C min-1, and lastly increased to 280 °C at a rate of 5 °C min-1 for 20 min, giving a run-time of 33 min.
Given a sample concentration of C µg L-1, a final volume of 2 mL (0.002 L) and an initial weight of
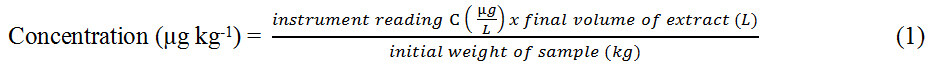
Since 1 µg = 1000 ng and 1 kg = 0.001 g, Equation (1) can be re-arranged and expressed in units of ng g-1, as shown in Equation (2):
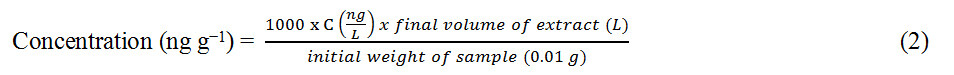
The final concentration unit of ng g-1 was used in this study to ensure uniformity with units expressed largely in the literature.
Quality assurance/quality control measures
Quality control measures are described in an earlier publication on PCBs[44], which included the use of procedural blanks, spikes, and surrogate 13C12-labeled PCBs. Analysis of procedural blanks was used to check for reagent purity, interference and potential contaminations from the laboratory. None of the determined PCB congeners were detected in the procedural blank samples. Recovery studies were performed to validate the efficiency of the chosen analytical procedure. Recovery studies were carried out by adding known concentrations of PCB standards to selected samples that had been initially quantified for PCBs, and all the analytical procedure was repeated. The calculated recoveries ranged from 85.7% to 102.3%. The recoveries of the surrogate 13C12-labeled standards ranged from 83.6% to 94.9%. The limits of detection and quantification (LOD and LOQ, respectively) of the instrument were evaluated based on the background noise from the analysis of blank samples. The LOD and LOQ values were 0.01 and 0.03 ng g-1, respectively.
Data analysis
Analysis of variance (ANOVA) was used to test for significant differences in PCB concentrations across the studied sites at P = 0.05. All statistical calculations, including unit conversions and health risk assessments, were done using SPSS version 15.1 software.
Ecological risk assessment
The ecological risk of PCBs in sediments was done by comparing the PCB concentration obtained in this study with stipulated guideline values[45]. The guidelines used for comparison in this study include the International Sediment Quality Guidelines (ISQGs); the effect range approach, including the effect range low (ERL) and the effect range medium (ERM) concentrations; and the effect level approach, including the threshold effect level (TEL) and the probable effect level (PEL)[46].
Human health risk assessment
Evaluation of toxicity equivalence (TEQ) of dl-PCBs
In the TEQ approach for assessing mixtures of dl-PCBs, the concentrations of individual dl-PCBs are converted to the toxicity equivalence concentration of 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) using a toxicity equivalence factor (TEF)[1]. The TCDD is assigned a TEF value of 1, while other dioxin-like compounds are assigned values less than 1[47]. The TEQ was evaluated using the expression[48]:

where Ci is the determined concentration of dl-PCBs and TEFi is the toxicity equivalence factor. The TEF values of dl-PCBs used in this study were obtained from Van den Berg et al.: 1 × 10-4 for PCB-77, 3 × 10-4 for PCB-81, 3 × 10-5 for PCB-105, 3 × 10-5 for PCB-114, 3 × 10-5 for PCB-118, 3 × 10-5 for PCB-123 and 1 × 10-1 for PCB-126, 3 × 10-5 for PCB-156, 3 × 10-5 for PCB-157, 3 × 10-5 for PCB-167, and 3 × 10-5 for PCB-189[49].
Health risk assessment
In the aquatic environment, human exposure to PCBs can occur mainly by ingestion (intentional ingestion, when water is used for drinking, or incidental ingestion, during swimming) and dermal contact[50].
For intentional ingestion of PCBs in surface water, the following expression was used[51]:

where CDI is the chronic daily intake (mg kg-1-day); C is the measured concentration (mg L-1) of PCBs in water; IngR* is the ingestion rate of drinking water (L day-1), 1.5 L day-1 for adults and 1 L for children; F* is the resident exposure frequency (days year-1), 365 days; ED is the exposure duration (years), 55 years for adults and 21 years for children; BW* is the body weight (kg), 70 kg for adults and 15 kg for children; and AT* is the average time (days), ED 365 (*adapted from Eslami et al.[52]).
Similarly, for incidental ingestion of PCBs in surface waters while swimming, two scenarios were considered in this study. In the first scenario (chronic exposure), it was assumed that exposures may have occurred over an entire lifetime, such as is obtainable in a rural or tribal community. In the second scenario (intermediate exposure), a three-month vacation period of the exposed individual was assumed.
The following expression was used for both chronic and intermediate exposures[53]:

where CDI is the chronic exposure dose (mg kg-1 day-1); C is the measured concentration (mg L-1) of PCBs in water; IngR** is the ingestion intake rate (L hour-1), 0.12 L hour-1 for children and 0.071 L hour-1 for adults; EV** is the site-specific event frequency (events day-1), 1 event day-1; tevent** is the time-specific event duration (hour event-1), 2 h event-1; F** is the exposure frequency (days week-1 week year-1), 7 days week-1 52 weeks year-1 for chronic exposures and 5 days week-1 13 weeks year-1 for intermediate exposures; ED is the exposure duration (years), 55 years for adults and 21 years for children; AT is the averaging time (days), F ED; and BW is the body weight (kg) (**values were adapted from ATSDR[53]).
For non-cancer risk, the hazard quotient (HQ) was calculated using Equation (5)[53,54]:

where RfDo is the oral reference dose (mg kg-1 day-1), 3.3 10-5 for PCB-167[54]. HQ values greater than 1 indicate high potential non-cancer risk, while HQ values less than 1 indicate low potential non-carcinogenic risk[55]
The total cancer risk was evaluated using the expression:

where SFO is the oral slope factor (mg kg-1 day-1). The values of SFO used for the ingestion pathway were 0.4 and 2.0 for adults and children, respectively[56].
RESULTS AND DISCUSSION
Concentration of PCBs in water
The concentrations of PCBs in water samples from Udu River are shown in Table 1. Out of the 28 PCBs quantified, only PCB-167 was identified across the sites, with concentrations ranging from 20-1860 ng L-1. The maximum concentration was at Point 3 (1860 ng L-1), while the minimum concentration was at Point 4 (20 ng L-1). The elevated concentration of PCB-167 at Point 3 relative to other sampling points could be due to emissions of particulates from jetty boats located nearby. Although there are limited health concerns about PCB-167, this congener has been reported to be associated with a low risk of breast cancer in women[36].
Concentration of PCBs in Udu River water
| Concentration (ng L-1) | |||||
| PCB congener | Point 1 | Point 2 | Point 3 | Point 4 | Point 5 |
| PCB-8 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-18 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-28 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-44 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-52 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-66 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-77 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-81 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-101 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-105 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-114 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-118 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-123 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-126 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-128 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-138 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-153 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-156 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-157 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-167 | 290 | 240 | 1860 | 20 | 40 |
| PCB-169 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-170 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-180 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-187 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-189 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-195 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-206 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| PCB-209 | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| Total | 290 | 240 | 1860 | 20 | 40 |
| ƩIndicator PCBs | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
| Ʃdl- PCBs | < 0.01 | < 0.01 | < 0.01 | < 0.01 | < 0.01 |
The 12 dl-PCBs and 7 indicator PCBs were not detected in water samples. This could be attributed to their inherently low concentrations, which were probably caused by biological and photo degradation. Additionally, the generally low detection of most PCB congeners in this study could be partly attributed to the fact that PCBs in the aquatic ecosystem usually become adsorbed onto particles in the water and settle on sediments[57]. Industrial effluents from food, textiles, fertilizers, detergents, and soap production are reported to be the main industrial activities responsible for elevated levels of PCBs in river waters[58], and such activities are not common around the studied river.
The results of this study are comparable with reports for PCBs in river waters available in the literature, as shown in Table 2. The absence of indicator and dl-PCBs in surface waters in this study compared with surface waters in nearby areas such as Warri, Ethiope, and Benin Rivers indicates the existence of some significant point and non-point sources of PCBs in the latter[9].
Comparison of the PCB concentrations in water obtained in this study with those found in the existing studies
| S/No. | Location | Number of PCB congeners quantified | Concentration of PCBs (ng L-1) | Reference |
| 1 | Warri river | ND-3900 | [9] | |
| 2 | Ethiope river, Nigeria | 8 | ND-5600 | [23] |
| 3 | Benin river, Nigeria | 8 | ND-11,300 | [23] |
| 4 | Bay of Bengal coast of Bangladesh | 209 | 32.1-199.4 | [30] |
| 5 | Shanghai rivers, China | 14 | ND-34.8 | [31] |
| 6 | Yamuna river, India | 27 | 10-779 | [57] |
| 7 | River Nile, Egypt | 10 | 14,000-20,000 | [58] |
| 8 | Bay of Bengal, India | NS | 1.93-4.48 | [59] |
| 9 | Hudson river estuary, USA | NS | 0.86-6.0 | [60] |
| 10 | Southern Moravia region, Czech Republic | 13 | 5.2-190.8 | [61] |
| 11 | Ogun river, Nigeria | 9 | 400-1,100 | [62] |
| 12 | Udu River, Nigeria | 1 | 20-1860 | This study |
Concentration of PCBs in sediments
Sediments give a better reflection of the contamination status of an aquatic environment compared to surface waters[9,63,64]. The concentration of PCBs in sediment samples is presented in Table 3. In total, 28 PCB congeners were identified across the sampled locations. The total concentration (Ʃ28PCBs) ranged from 5.34 to 16.1 ng g-1 dry weight. The highest concentration was found at Point 4, where all 28 quantified congeners were detected. The lowest concentration was obtained at Point 1, with only 17 congeners identified. Out of the 28 PCBs identified, PCBs 8, 18, 44, 81, and 167 were dominant across the sampling points, accounting for 11.3%, 8.90%, 15.6%, 6.10%, and 18.6% of the total PCB burden in the sediments, respectively. The lower chlorinated congeners (i.e., congeners with PCB numbers < 101) represented 76% at Point 1, 50.6% at Point 2, 28.7% at Point 3, 48.1% at Point 4, and 60.3% at Point 5 of the total PCB concentration. The dominance of lower chlorinated congeners at Points 1, 2, and 5 could be an indicator that there were no significant point sources of PCBs at these locations, since lower chlorinated congeners are transported to longer distances from their points of contamination[65].
Concentration of PCBs in Udu River sediments
| Concentration (ng g-1) | Average | |||||
| PCB congener | Point 1 | Point 2 | Point 3 | Point 4 | Point 5 | |
| PCB-8 | 1.21 | 0.49 | 0.17 | 2.02 | 0.79 | 0.94 |
| PCB-18 | 0.67 | 0.27 | 0.094 | 2.26 | 0.84 | 0.83 |
| PCB-28 | < 0.01 | < 0.01 | < 0.01 | 0.11 | 0.18 | 0.059 |
| PCB-44 | 1.47 | 1.14 | 0.91 | 1.32 | 0.54 | 1.07 |
| PCB-52 | 0.44 | 0.34 | 0.27 | 0.24 | 0.73 | 0.40 |
| PCB-66 | < 0.01 | < 0.01 | < 0.01 | 0.65 | 0.25 | 0.18 |
| PCB-77 | 0.074 | 0.29 | 0.20 | 0.024 | 0.69 | 0.25 |
| PCB-81 | 0.20 | 0.76 | 0.51 | 1.12 | 0.09 | 0.54 |
| PCB-101 | 0.20 | 0.23 | 0.30 | 1.37 | 0.088 | 0.44 |
| PCB-105 | 0.23 | 0.26 | 0.35 | 0.19 | 0.02 | 0.21 |
| PCB-114 | < 0.01 | 0.068 | 0.058 | 1.23 | 0.17 | 0.31 |
| PCB-118 | < 0.01 | 0.19 | 0.10 | 0.37 | 0.11 | 0.15 |
| PCB-123 | < 0.01 | < 0.01 | 0.038 | 0.10 | 0.17 | 0.063 |
| PCB-126 | 0.10 | 0.18 | 0.17 | 0.09 | 0.064 | 0.12 |
| PCB-128 | 0.026 | 0.016 | < 0.01 | 0.088 | 0.060 | 0.038 |
| PCB-138 | 0.048 | 0.11 | 0.098 | 0.13 | 0.076 | 0.092 |
| PCB-153 | < 0.01 | < 0.01 | < 0.01 | 0.17 | 0.10 | 0.056 |
| PCB-156 | < 0.01 | 0.064 | 0.06 | 0.078 | 0.068 | 0.054 |
| PCB-157 | 0.098 | 0.082 | 0.16 | 0.32 | 0.21 | 0.17 |
| PCB-167 | 0.36 | 1.59 | 3.51 | 1.74 | 0.27 | 1.49 |
| PCB-169 | 0.11 | 0.086 | 0.18 | 0.056 | < 0.01 | 0.085 |
| PCB-170 | 0.054 | 0.084 | 0.076 | 0.10 | 0.068 | 0.077 |
| PCB-180 | 0.058 | < 0.01 | 0.068 | 0.27 | 0.10 | 0.10 |
| PCB-187 | < 0.01 | 0.064 | 0.078 | 0.33 | 0.042 | 0.10 |
| PCB-189 | < 0.01 | < 0.01 | 0.084 | 0.13 | 0.21 | 0.085 |
| PCB-195 | < 0.01 | 0.11 | < 0.01 | 0.11 | 0.13 | 0.071 |
| PCB-206 | < 0.01 | 0.094 | < 0.01 | 0.45 | 0.094 | 0.123 |
| PCB-209 | < 0.01 | < 0.01 | < 0.01 | 1.03 | 0.62 | 0.33 |
| Total | 5.34 | 6.55 | 7.49 | 16.1 | 6.81 | 8.45 |
| ƩIndicator PCBs | 0.74 | 0.86 | 0.84 | 2.67 | 1.38 | 1.29 |
| Ʃdl- PCBs | 1.07 | 3.42 | 5.23 | 5.36 | 2.03 | 3.42 |
| Ʃnon-ortho dl-PCBs | 0.38 | 1.21 | 0.95 | 1.28 | 0.84 | 0.93 |
The levels of PCBs in sediments can be influenced by water flow and organic matter, which may influence several physical properties of PCBs, such as transport, migration, and adsorption[66-68]. The concentration of PCBs at Point 4 was significantly (P < 0.05) greater than at the other sampling points. However, the generally low level of PCBs in sediments obtained in this study can be attributed to minimal industrial activities and municipal effluent discharges into the river[69]. The resulting concentrations of this study are very low when compared with reports for PCBs in sediments in nearby rivers, including Warri River[9], Orogodo River[19], Ethiope and Benin Rivers[23], and River Niger[27]. Table 4 gives a comparison of PCB concentrations in sediment obtained in this study with some existing studies.
Comparison of the PCB concentrations in sediments obtained in this study with those found in existing studies
| S/No. | Location | Number of PCB congeners quantified | Concentration of PCBs (ng g-1) | Reference |
| 1 | Benin river, Nigeria | 8 | ND-40.9 | [9] |
| 2 | Ethiope river, Nigeria | 8 | 0.33-22.9 | [9] |
| 3 | Asalouyeh harbour, Iran | 18 | 0.0095-14.14 | [17] |
| 4 | River Ona, Nigeria | 17 | 589-1353.6 | [25] |
| 5 | Rivers Ase, Niger and Forcados, Nigeria | 28 | ND-1,633 | [29] |
| 6 | Shanghai rivers, China | 14 | 1.46-46.11 | [31] |
| 7 | Danube river, Serbia | 6 | 0.41 | [41] |
| 8 | River Ogun, Nigeria | 9 | 7.8-10,000 | [63] |
| 9 | River Nile, Egypt | 11 | 1.67-2.46 | [70] |
| 10 | River Beijing, China | NS | 0.78-8.47 | [71] |
| 11 | Liaohe river, China | 12 | 0.0245-0.365 | [72] |
| 12 | New Calabar river, Nigeria | 8 | 210-6090 | [73] |
| 13 | Udu River, Nigeria | 28 | 5340-16100 | This study |
The indicator PCBs detected in sediments in this study include PCBs 28, 52, 101, 118, 138, 153, and 180, accounting for 13.8%, 13.3%, 11.2%, 16.3%, and 20.4% of the total PCB concentrations across the five sampling points, respectively. The concentration of the indicator PCBs varied between 0.74 and 2.67 ng g-1. Indicator PCBs 52, 101, and 138 were the dominant congeners identified in all sampling points. The concentration of these PCBs in the river sediments was within the ecological risk criteria value of
The mean concentration of the 12 detected dl-PCBs ranged from 1.07 ng g-1 at Point 1 to 5.36 ng g-1 at Point 4. The average concentration followed the sequence: PCB-167 > PCB-81 > PCB-114 > PCB-77 > PCB-105 > PCB-157 > PCB-118 > PCB-126 > PCB-169 > PCB-189 > PCB-123 > PCB-156. PCB-105 was detected at all sampling points, and its presence in sediments has been attributed to its use in paint additives[75]. PCB-167 was also detected at all sampling points, with Point 3 having the highest concentration. The elevated concentration of PCB-167 at Point 3 could be attributed to emissions from nearby jetty boats. The non-ortho dl-PCBs (i.e., PCBs 77, 81, 126, and 169) are PCB congeners that produce dioxin-like toxicity in fish, birds, and mammals, while the mono-ortho dl-PCBs cause dioxin-like toxicity in mammals and birds but generally do not produce the same effect in fish[1]. This means that the concentrations of non-ortho dl-PCBs are of importance in aquatic ecosystems. In this study, there was an observed dominance of mono-ortho dl-PCBs. This also suggests minimal harm to aquatic organisms and a potentially reduced toxicity in the food chain due to bioaccumulation.
Distribution of PCB homologs in sediments
The degree of chlorination of a PCB congener determines its physicochemical characteristics[76]. As a consequence, their distribution pattern will provide data on the environmental consequence and likely origin of PCBs[77]. The patterns of occurrence and distribution of PCB homologs in sediments from the Udu River are shown in Figure 2 and followed the order: Tetra-PCBs > Hexa-PCBs > Penta-PCBs > Di-PCBs > Tri-PCBs > Hepta-PCBs > Deca-PCBs > Nona-PCBs > Octa-PCBs. The di-PCB homolog in the sediment samples ranged from 0.17 to 2.02 ng g-1, accounting for 2%-22% of the total PCBs. For tri-PCBs homolog, the concentrations varied between 0.10 and 2.37 ng g-1, accounting for 1%-15% of the total PCBs. The concentration of the tetra-PCBs was greatest at Points 1, 2, 4, and 5, ranging from 1.89-3.36 ng g-1 and accounting for 20%-40% of the total PCBs. The presence of di-PCB through to octa-PCBs suggests the possibility of varied sources of PCBs[9].
Ecological risk assessment of PCBs
Comparison with guideline values
Nigeria has no guidelines for evaluating the ecological risk of PCBs. However, there are guidelines for PCBs established by some international agencies. According to the US EPA guideline, the concentration of PCBs in surface water should not exceed 0.0005 mg L-1 (500 ng L-1), a concentration below which they are not considered to pose any hazard to aquatic life or human health[31]. Additionally, PCBs have a specific maximum contaminant limit (MCL) value of 0.5 µg L-1 (500 ng L-1) for drinking water, as established by the New York State Department of Health[78]. In this study, only one congener was detected in surface water, and the mean concentration across the sampling sites was low compared to the guideline value. However, considering the individual sampling points, the concentration at Point 3 exceeded the guideline value.
For sediment samples, the total concentration of PCBs obtained in this study was within the ISQG values of 34.1 and 21.5 ng g-1 for PCBs in freshwater and marine/estuarine, respectively[79]. When compared with the German Legislation for individual indicator PCBs in sediments, with a target value of 20 ng g-1[80], none of the individual indicator PCB congeners surpassed it. The total PCB concentrations in this study were also within the 187 ng g-1 probable effect level (PEL)[81] and the 274 ng g-1 consensus-based probable effect concentration (PEC)[82]. However, since PCBs are lipophilic, they possess good bioaccumulation tendencies in fatty tissues of aquatic organisms[1]. Therefore, there might be a potential tendency for PCBs to move up the food chain over time. For instance, very low PCB concentrations in water and sediments in rivers of Southern Moravia region were found, but high contamination was observed in fish from the same rivers[61].
Toxicity equivalence (TEQ) of dl-PCBs
The TEQ values for dl-PCBs are shown in Table 5. The values varied between 0.0065 and 0.018 ng TEQ g-1. The average contribution of PCB congeners to the TEQ values across the 5 locations followed the order: PCB-156 > PCB-123 > PCB-189 > PCB-118 > PCB-157 > PCB-105 > PCB-114 > PCB-77 > PCB-167 > PCB-81 > PCB-126. PCB-126 congener had the greatest influence on the TEQ values in sediments from the studied river. A similar study also reported that PCB-126 greatly impacted the TEQ values of dl-PCBs in river sediments[29]. This congener has been established to induce hepatotoxicity and hepatic micronutrient disruption in animals[83]. However, the TEQ values of dl-PCBs in the studied sediment samples were within the sediment quality value of 0.0215 ng TEQ g-1[72], which suggests minimal risks linked with PCB exposure.
Toxicity equivalence values of PCBs in the Udu River
| Site | TEQ (ng TEQ-1) |
| 1 | 0.01 |
| 2 | 0.018 |
| 3 | 0.017 |
| 4 | 0.0095 |
| 5 | 0.00065 |
Health risk assessment of PCBs
Non-cancer risk
The non-cancer risks due to the presence of PCBs in water samples from Udu River are shown in Table 6. The HI values of PCBs in the water samples via intentional ingestion ranged from 1.30 × 10-2 to 1.21 and from 4.04 × 10-2 to 3.76 for adults and children, respectively. For incidental ingestion of water, as expected, the chronic exposure scenario results were all greater than those of the intermediate exposure scenario. The HI values for incidental chronic exposure ranged from 1.23 × 10-3 to 1.14 × 10-1 and from 9.70 × 10-3 to
Non-cancer and cancer risks of PCB-167 in Udu River water
| Sampling points | Human group | Exposure route | CDI | HI | TCR |
| 1 | Adults | Intentional ingestion | 6.20 × 10-6 | 1.88 × 10-1 | 2.49 × 10-6 |
| 2 | 5.14 × 10-6 | 1.56 × 10-1 | 2.06 × 10-6 | ||
| 3 | 4.00 × 10-5 | 1.21 | 1.59 × 10-5 | ||
| 4 | 4.28 × 10-7 | 1.30 × 10-2 | 1.71 × 10-7 | ||
| 5 | 8.57 × 10-7 | 2.60 × 10-2 | 3.43 × 10-7 | ||
| 1 | Children | Intentional ingestion | 1.99 × 10-5 | 5.86 × 10-1 | 3.87 × 10-5 |
| 2 | 1.60 × 10-5 | 4.85 × 10-1 | 3.20 × 10-5 | ||
| 3 | 1.24 × 10-4 | 3.76 | 2.48 × 10-4 | ||
| 4 | 1.33 × 10-6 | 4.04 × 10-2 | 2.67 × 10-6 | ||
| 5 | 2.66 × 10-6 | 8.08 × 10-1 | 5.33 × 10-6 | ||
| 1 | Adults | Incidental ingestion | 1.93 × 10-5 | 1.78 × 10-2 | 2.35 × 10-7 |
| 2 | (chronic exposure) | 1.60 × 10-6 | 1.48 × 10-2 | 1.95 × 10-7 | |
| 3 | 1.24 × 10-4 | 1.14 × 10-1 | 1.51 × 10-6 | ||
| 4 | 1.33 × 10-6 | 1.23 × 10-3 | 1.62 × 10-8 | ||
| 5 | 2.66 × 10-6 | 2.46 × 10-3 | 3.25 × 10-8 | ||
| 1 | Children | Incidental ingestion | 4.64 × 10-6 | 1.41 × 10-1 | 9.28 × 10-6 |
| 2 | (chronic exposure) | 3.84 × 10-6 | 1.16 × 10-1 | 7.68 × 10-6 | |
| 3 | 2.97 × 10-5 | 9.02 × 10-1 | 5.99 × 10-5 | ||
| 4 | 3.20 × 10-7 | 9.70 × 10-3 | 6.40 × 10-7 | ||
| 5 | 6.40 × 10-7 | 1.94 × 10-2 | 1.28 × 10-6 | ||
| 1 | Adults | Incidental ingestion | 4.20 × 10-7 | 1.27 × 10-2 | 1.68 × 10-7 |
| 2 | (intermediate exposure) | 3.46 × 10-7 | 1.05 × 10-2 | 1.39 × 10-7 | |
| 3 | 2.69 × 10-6 | 8.18 × 10-2 | 1.08 × 10-6 | ||
| 4 | 2.89 × 10-8 | 8.78 × 10-4 | 1.16 × 10-8 | ||
| 5 | 5.79 × 10-8 | 1.78 × 10-3 | 2.32 × 10-8 | ||
| 1 | Children | Incidental ingestion | 3.31 × 10-6 | 1.00 × 10-1 | 6.63 × 10-6 |
| 2 | (intermediate exposure) | 2.74 × 10-6 | 8.31 × 10-2 | 5.48 × 10-6 | |
| 3 | 2.12 × 10-5 | 6.44 × 10-1 | 4.25 × 10-5 | ||
| 4 | 2.28 × 10-7 | 6.92 × 10-3 | 4.57 × 10-7 | ||
| 5 | 4.56 × 10-7 | 1.38 × 10-2 | 9.14 × 10-7 |
Total cancer risk
The total cancer risks (TCR) due to PCBs in Udu River water are also shown in Table 6. The TCR values for intentional ingestion of water ranged from 1.71 × 10-7 to 1.59 × 10-5 for adult exposure and from 2.67 × 10-6 to 2.48 × 10-4 for child exposure. Similarly, the TCR values for chronic incidental ingestion of water ranged from 1.62 × 10-8 to 1.51 × 10-6 for adult exposure and from 6.40 × 10-7 to 5.99 × 10-5 for child exposure. For intermediate incidental exposure, the TCR ranged from 1.16 × 10-8 to 1.08 × 10-6 and from 4.57 × 10-7 to
CONCLUSION
This study provided useful information on the concentration, distribution, and risks of PCBs in water and sediments from the Udu River, Niger Delta, Nigeria. The results show that only PCB-167 was present in surface water, with concentrations greater than the guideline value at one of the sampling points. For sediment samples, all determined concentrations were low relative to established guideline values. The results also reveal that the water and sediments from the Udu River were not significantly impacted by PCBs, and that the potential ecological and health risks associated with PCBs in the river water were very low (except for surface water at one sampling point). Even though there were limited health concerns about PCB-167 in the water, further investigation of its source and monitoring is necessary. Similarly, with scarce data available on the pollution status of this river, there is a need for further monitoring of the river with respect to other types of pollutants, including polycyclic aromatic hydrocarbons (PAHs) and organochlorine pesticides (OCPs). Additionally, there is a need to monitor the concentrations of PCBs, PAHs, and OCPs in aquatic organisms, especially fish, in the river.
DECLARATIONS
Acknowledgements
The authors are grateful to Mr Jude C. Ossai and the Management of Apex Analytics, Port Harcourt, Nigeria, for their assistance in chemical analyses.
Authors’ contributions
Conception and design of the study: Kpomah ED
Materials preparation, sampling, data collection and analysis: Iniaghe PO, Kpomah ED
Interpretation of results and writing of first draft of the manuscript: Iniaghe PO
Availability of data and materials
All data generated in this study are included in the manuscript.
Financial support and sponsorship
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Conflicts of Interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2022.
REFERENCES
1. Henry TR, DeVito MJ. Non-dioxin-like PCBs: effects and consideration in ecological risk assessment. Available from: https://www.epa.gov/sites/default/files/2015-11/documents/1340-erasc-003.pdf [Last accessed on 19 Sep 2022].
2. Skrbić B, Durisić-Mladenović N. Principal component analysis for soil contamination with organochlorine compounds. Chemosphere 2007;68:2144-52.
3. Ogura I, Masunaga S, Nakanishi J. Quantitative source identification of dioxin-like PCBs in Yokohama, Japan, by temperature dependence of their atmospheric concentrations. Environ Sci Technol 2004;38:3279-85.
4. White SS, Birnbaum LS. An overview of the effects of dioxins and dioxin-like compounds on vertebrates, as documented in human and ecological epidemiology. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 2009;27:197-211.
5. Agency for Toxic Substances and Disease Registry (ATSDR); Toxicological Profile for Polychlorinated Biphenyls. U.S. Department of Health and Human Services 2000; 1-758. Available from: https://citeseerx.ist.psu.edu/viewdoc/summary?doi=10.1.1.649.7533 [Last accessed on 19 Sep 2022].
6. Viluksela M, van der Ven LTM, Schrenk D, et al. Biological and toxicological effects of non-dioxin-like PCBs. Acta Vet Scand 2012:54.
7. Hu D, Hornbuckle KC. Inadvertent polychlorinated biphenyls in commercial paint pigments. Environ Sci Technol 2010;44:2822-7.
8. Gerven T, Geysen D, Vandecasteele C. Estimation of the contribution of a municipal waste incinerator to the overall emission and human intake of PCBs in Wilrijk, Flanders. Chemosphere 2004;54:1303-8.
9. Ezemonye L. Levels of polychlorinated biphenyls residues in Warri river, Nigeria. Glo Jnl Env Res 2005:4.
10. Mahmood A, Malik RN, Li J, Zhang G. Levels, distribution profile, and risk assessment of polychlorinated biphenyls (PCBs) in water and sediment from two tributaries of the River Chenab, Pakistan. Environ Sci Pollut Res Int 2014;21:7847-55.
11. Men B, He M, Tan L, Lin C. Distributions of polychlorinated biphenyls in the Daliao River estuary of Liaodong Bay, Bohai Sea (China). Mar Environ Res 2014;78:77-84.
12. Chen X, Yao X, Yu C, et al. Hydrodechlorination of polychlorinated biphenyls in contaminated soil from an e-waste recycling area, using nanoscale zerovalent iron and Pd/Fe bimetallic nanoparticles. Environ Sci Pollut Res Int 2014;21:5201-10.
13. Debela SA, Sheriff I, Wu J, et al. Occurrences, distribution of PCBs in urban soil and management of old transformers dumpsite in Addis Ababa, Ethiopia. Sci Afr 2020;8:e00329.
14. Sohail M, Eqani SAMAS, Podgorski J, et al. Persistent organic pollutant emission via dust deposition throughout Pakistan: Spatial patterns, regional cycling and their implication for human health risks. Sci Total Environ 2018;618:829-37.
15. Hirai Y, Masunaga S, Nakanishi J, Owari M. A case study of aerial PCB pollution in and around PCB waste storage. 11th Symposium on Environmental Chemistry Programs and Abstracts, Hakone, Japan, June 3-5,2002; pp.484-5. Available from: http://risk.kan.ynu.ac.jp/publish/hirai/hirai200206.pdf [Last accessed on 22 Sep 2022].
16. Wania F. Assessing the potential of persistent organic chemicals for long-range transport and accumulation in Polar regions. Environ Sci Technol 2003; 37:1344-51.
17. Arfaeinia H, Asadgol Z, Ahmadi E, et al. Characteristics, distribution and sources of polychlorinated biphenyls (PCBs) in coastal sediments from the heavily industrialized area of Asalouyeh, Iran. Water Sci Technol 2017;76:3340-50.
18. Gerbersdorf SU, Cimatoribus C, Class H, et al. Anthropogenic Trace Compounds (ATCs) in aquatic habitats - research needs on sources, fate, detection and toxicity to ensure timely elimination strategies and risk management. Environ Int 2015;79:85-105.
19. Edjere O, Iyekowa O. Assessment of the levels of polychlorinated biphenyls (PCBs) in Orogodo River sediments Agbor, Delta State, Nigeria. Ovidius Univ Ann Chem 2017;28:25-9.
20. Filipkowska A. Determining the PCBs in fish and sediment samples related to intercomparison studies. Pol J Environ Stud 2013;22:1341-7.
21. James MO, Kleinow KM. Seasonal influences on PCB retention and biotransformation in fish. Environ Sci Pollut Res Int 2014;21:6324-33.
22. Osibanjo O, Bamgbose O. Chlorinated hydrocarbons in marine fish and shellfish of Nigeria. Mar Pollut Bull 1990;21:581-6.
23. Ezemonye LIN. Polychlorinated biphenyls (PCBs) levels and distribution in Ethiope and Benin Rivers of the Niger Delta, Nigeria: surface water and sediments. Int J Environ. Stud 2005;62:491-504.
24. Adeyemi D, Ukpo G, Anyakora C, Uyimadu JP. Polychlorinated biphenyl in fish samples from Lagos Lagoon, Nigeria. Afr J Biotech 2009;8:2811-5. Available from: https://www.ajol.info/index.php/ajb/article/view/60880 [Last accessed on 20 Sep 2022]
25. Adeogun AO, Babatunde TA, Chukwuka AV. Spatial and temporal variations in water and sediment quality of Ona River, Ibadan, Southwest Nigeria. Eur J Sci Res 2012;74:186-204.
26. Adeogun AO, Chukwuka AV, Okoli CP, Arukwe A. Concentration of polychlorinated biphenyl (PCB) congeners in the muscle of Clarias gariepinus and sediment from inland rivers of southwestern Nigeria and estimated potential human health consequences. J Toxicol Environ Health A 2016;79:969-83.
27. Iwegbue CMA. Distribution and ecological risks of polychlorinated biphenyls (PCBs) in surface sediment of the Forcados River, Niger Delta, Nigeria. Afr J Aquat Sci 2016;41:51-6.
28. Unyimadu JP, Osibanjo O, Babayemi JO. Polychlorinated biphenyls (PCBs) in River Niger, Nigeria: Occurrence, distribution and composition profiles. Toxicol Ind Health 2018;34:54-67.
29. Irerhievwie GO, Iwegbue CMA, Lari B, et al. Spatial characteristics, sources, and ecological and human health risks of polychlorinated biphenyls in sediments from some river systems in the Niger Delta, Nigeria. Mar Pollut Bull 2020;160:111605.
30. Habibullah-Al-Mamun M, Kawser Ahmed M, Saiful Islam M, Tokumura M, Masunaga S. Occurrence, distribution and possible sources of polychlorinated biphenyls (PCBs) in the surface water from the Bay of Bengal coast of Bangladesh. Ecotoxicol Environ Saf 2019;167:450-8.
31. Wang X, Han J, Bi C, Huang X, Jia J, Chen Z. Distribution, sources, and risk assessment of polychlorinated biphenyls in surface waters and sediments of rivers in Shanghai, China. Front Earth Sci 2017;11:283-96.
32. Zhang L, Shi S, Dong L, et al. Concentrations and possible sources of polychlorinated biphenyls in the surface water of the Yangtze River Delta, China. Chemosphere 2011;85:399-405.
33. Faroon O, Jones D, de Rosa C. Effects of polychlorinated biphenyls on the nervous system. Toxicol Ind Health 2000;16:305-33.
34. Pinson A, Franssen D, Gérard A, Parent AS, Bourguignon JP. Neuroendocrine disruption without direct endocrine mode of action: Polychloro-biphenyls (PCBs) and bisphenol A (BPA) as case studies. C R Biol 2017;340:432-8.
35. Fagbote EO, Olanipekun EO. Levels of polycyclic aromatic hydrocarbons and polychlorinated biphenyls in sediment of bitumen deposit impacted area. Int J Environ Sci Technol 2010;7:561-70.
36. Cohn BA, Terry MB, Plumb M, Cirillo PM. Exposure to polychlorinated biphenyl (PCB) congeners measured shortly after giving birth and subsequent risk of maternal breast cancer before age 50. Breast Cancer Res Treat 2012;136:267-75.
37. Morgan M, Deoraj A, Felty Q, Roy D. Environmental estrogen-like endocrine disrupting chemicals and breast cancer. Mol Cell Endocrinol 2017;457:89-102.
38. Aluyi HSA, Ekhaise FO, Adelusi DM, Oviasogie FE. Effect of human activities and oil pollution on the microbiological and physicochemical quality of UDU river, Warri, Nigeria. Estud Biol 2006:28.
39. Okobiebi O, Okobiebi B. Physicochemical and heavy metal assessment of the Udu River, Delta state, Nigeria. jasem 2021;25:665-7.
41. Škrbić BD, Kadokami K, Antić I, Jovanović G. Micro-pollutants in sediment samples in the middle Danube region, Serbia: occurrence and risk assessment. Environ Sci Pollut Res Int 2018;25:260-73.
42. The United States Environmental Protection Agency (US EPA). Method 1668B: chlorinated biphenyl congeners in water, soil, sediment, biosolids, and tissue by HRGC/HRMS. Available from: https://ntrl.ntis.gov/NTRL/dashboard/searchResults/titleDetail/PB2009105236.xhtml [Last accessed on 20 Sep 2022].
43. United States Environmental Protection Agency (US EPA). SW-846 test method 3540C: soxhlet extraction Available from: https://www.epa.gov/hw-sw846/sw-846-test-method-3540c-soxhlet-extraction [Last accessed on 20 Sep 2022].
44. Tesi GO, Iniaghe PO. Polychlorinated biphenyls in canned sardines in Nigeria and health risk assessment. Food Addit Contam Part B Surveill 2020;13:200-6.
45. MacDonald DD, Ingersoll CG, Smorong DE, et al. Development and evaluation of numerical sediment quality assessment guidelines for Florida Inland waters. Technical Report, Florida Department of Environmental Protection 2003, Florida.
46. Long ER; Morgan LG; The potential for biological effects of sediment-sorbed contaminants tested in the National Status and Trends Program. In: NOAA Technical Memorandum NOS OMA-52. US Department of Commerce, Coastal and Estuarine Assessment Branch, NOAA, Seattle 1990. Available from: https://udspace.udel.edu/handle/19716/1562 [Last accessed on 20 Sep 2022].
47. Minnesota Department of Health (MDH). MDH risk assessment: guidance for dioxins. Available from: https://www.health.state.mn.us/communities/environment/risk/docs/guidance/dioxinmemo1.pdf [Last accessed on 19 Sep 2022].
48. Bhavsar SP, Reiner EJ, Hayton A, Fletcher R, MacPherson K. Converting toxic equivalents (TEQ) of dioxins and dioxin-like compounds in fish from one toxic equivalency factor (TEF) scheme to another. Environ Int 2008;34:915-21.
49. Van den Berg M, Birnbaum LS, Denison M, et al. The 2005 World Health Organization reevaluation of human and Mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol Sci 2006;93:223-41.
50. Duodu GO, Ogogo KN, Mummullage S, et al. Source apportionment and risk assessment of PAHs in Brisbane River sediment, Australia. Ecol Indic 2017;73:784 -99.
51. United States Environmental Protection Agency (US EPA). Risk assessment guidance for Superfund, Vol. I: human health evaluation manual. Office of Solid Waste and Emergency Response, 1989, EPA/540/1-89/002.
52. Eslami H, Esmaeili A, Razaeian M, et al. Potentially toxic metal concentration, spatial distribution, and health risk assessment in drinking water resources of southeast Iran. Geosci Front 2022:101276.
53. ATSDR, Agency for Toxic Substances and Disease Registry. Exposure dose guidance for dermal and ingestion exposure to surface water. Atlanta, GA: U.S. Department of Health and Human Services, Public Health Service, 2018.
54. United State Environmental Protection Agency (US EPA). Risk assessment guidance for Superfund (RAGS), Vol. III-part a, process for conducting probabilistic risk assessment, EPA 540-R-02-002. Office of Emergency and Remedial Response, Washington, DC, 2001.
55. United States Environmental Protection Agency (US EPA). Guidelines for carcinogen risk assessment, 2005.
56. United States Department of Energy (US DOE). The risk assessment information system (RAIS). US Department of Energy, Oak Ridge Operations (ORO) Office: Oak Ridge, TN, USA, 2011.
57. Kumar B, Singh SK, Mishra M, Kumar S, Sharma CS. Assessment of polychlorinated biphenyls and organochlorine pesticides in water samples from the Yamuna River. J Xenobiotics 2012;2:6.
58. Megahed AM, Dahshan H, Abd-El-Kader MA, et al. Polychlorinated biphenyls water pollution along the river Nile, Egypt. Sci World J 2015:7.
59. Rajendran RB, Imagawa T, Tao H, Ramesh R. Distribution of PCBs, HCHs and DDTs, and their ecotoxicological implications in Bay of Bengal, India. Environ Int 2005;31:503-12.
60. Yan S, Rodenburg LA, Dachs J, Eisenreich SJ. Seasonal air-water exchange fluxes of polychlorinated biphenyls in the Hudson River Estuary. Environ Pollut 2008;152:443-51.
61. Lána R, Vávrová M, Cáslavský J, et al. PCBs in samples from the environment of the Southern Moravia Region, Czech Republic. Bull Environ Contam Toxicol 2008;81:574-7.
62. Biphenyl Contamination in Water and Sediment Samples in Upper River Ogun, Lagos State, Nigeria. IJEE 2018. Available from: https://ir.unilag.edu.ng/handle/123456789/6183 [Last accessed on 22 Sep 2022]
63. Odiete WO. Environmental Physiology of animals and pollution. Diversified resources, Lagos, 1999.
64. Aremu MO, Atolaiye BO, Gav BL, et al. Metal concentrations in sediments and water from Rivers Doma, Farinruwa and Mada in Nassarawa State, Nigeria. J Environ Chem Ecotoxicol 2011;3:242-9.
65. Salem DMS, Khaled A, El Nemr A. Assessment of pesticides and polychlorinated biphenyls (PCBs) in sediments of the Egyptian Mediterranean Coast. Egyptian J Aquat Res 2013;39:141-152.
66. Yang Z, Shen Z, Gao F, Tang Z, Niu J. Occurrence and possible sources of polychlorinated biphenyls in surface sediments from the Wuhan reach of the Yangtze River, China. Chemosphere 2009;74:1522-30.
67. Yang H, Zhuo S, Xue B, Zhang C, Liu W. Distribution, historical trends and inventories of polychlorinated biphenyls in sediments from Yangtze River Estuary and adjacent East China Sea. Environ Pollut 2012;169:20-6.
68. Cui S, Fu Q, Guo L, et al. Spatial-temporal variation, possible source and ecological risk of PCBs in sediments from Songhua River, China: Effects of PCB elimination policy and reverse management framework. Mar Pollut Bull 2016;106:109-18.
69. Biljana S. Assessment of the Serbian population exposure to polychlorinated biphenyls by crops. Environ Toxicol Pharmacol 2008;25:171-5.
70. Omar WA, Mahmoud HM. Risk assessment of polychlorinated biphenyls (PCBs) and trace metals in River Nile up- and downstream of a densely populated area. Environ Geochem Health 2017;39:125-37.
71. Zhang Z, Huang J, Yu G, Hong H. Occurrence of PAHs, PCBs and organochlorine pesticides in the Tonghui River of Beijing, China. Environ Pollut 2004;130:249-61.
72. Zhang Y, Wang C, Du L. Distribution, source and potential risk assessment of polychlorinated biphenyls (PCBs) in sediments from the Liaohe River protected area, China. Sustainability 2021;13:10750.
73. Ilechukwu I, Mgbemena NM, Inagbor PO, Ndukwe GI. Assessment of the levels of polychlorinated biphenyls in sediments of new Calabar River, Niger Delta Region, Nigeria. Ovidus Univ Ann Chem 2018;29:34-40.
74. OSPAR Commission. OSPAR Commission for the Protection of the marine environment of the North-East Atlantic, quality status report 2000. OSPAR Commission, London, pp.56.
75. Yao T, He C, Zhang P, Gao H, Zhou C. Distribution and Sources of Polychlorinated Biphenyls (PCBs) and Organochlorine Pesticides (OCPs) in Surface Waters of Jinzhou Bay in China. Proc Envir Sci 2013;18:317-22.
76. Fiedler H. Polychlorinated biphenyls (PCBs): uses and environment release. Paper presented at the sub-regional awareness raising workshop on Persistent Organic Pollutants (POP), Kranjska, Gora, Slovenia, May 1998.
77. Combi T, Miserocchi S, Langone L, Guerra R. Polychlorinated biphenyls (PCBs) in sediments from the western Adriatic Sea: Sources, historical trends and inventories. Sci Total Environ 2016;562:580-7.
78. New York State Department of Environmental Conservation, Division of Water. SJSAmbient water quality value for protection of sources of potable water - polychlorinated biphenyls (PCBs). New York State Human health fact sheet: 1998.
79. Canadian Council of Ministers of Environment (CCME). Canadian soil quality guidelines for the protection of environmental and human health, 6th ed., 2007. Canadian Council of Ministers of the Environment, Winnipeg, Canada.
80. FGG BLBE. Fachberichte. Available from: http://www.fgg-elbe.de/dokumente/gewaesserguete.html [Last accessed on 19 Sep 2022].
81. Macdonald DD, Carr RS, Calder FD, Long ER, Ingersoll CG. Development and evaluation of sediment quality guidelines for Florida coastal waters. Ecotoxicology 1996;5:253-78.
82. Gómez-Gutiérrez A, Garnacho E, Bayona JM, Albaigés J. Screening ecological risk assessment of persistent organic pollutants in Mediterranean sea sediments. Environ Int 2007;33:867-76.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].