Microstructure-engineered electrocaloric P(VDF-TrFE-CFE) terpolymer-based nanocomposites with enhanced interfacial coupling for rapid thermal switching
Abstract
Electrocaloric (EC) cooling represents a promising solid-state approach for next-generation thermal management. However, achieving substantial temperature modulation remains a challenge due to intrinsic material limitations and inefficient energy conversion. Herein, we focus on microstructure regulation to enhance thermal conductivity and EC performance. A hydroxyl-functionalized Ba0.63Sr0.37Zr0.01(Ti0.999Mn0.001)0.99O3 (BSZMT-OH) and h-BNNS-OH composite was designed, with enhanced interfacial hydrogen bonding to optimize electric field response in a P(VDF-TrFE-CFE) matrix. Finite element analysis (FEA) and piezo response force microscopy (PFM) reveal strengthened interfacial coupling, which facilitates rapid domain switching kinetics by amplifying tetragonal P4mm phase responses. These enhancements yield a peak EC temperature change (ΔT) of 1.59 K under a low electric field of 40 MV/m in the optimized 6% BSZMT-OH@4 h-BNNS-OH (6@4BNNS) composite. Integrated into a double-layer four-section electrocaloric cooling (DL 4 EC) device, it cools from 70 to 23 °C in 23 s, outperforming water cooling. Our findings offer insights into EC mechanisms and present a high-performance thermal management strategy.
Keywords
INTRODUCTION
The thermal management challenges in modern microelectronic devices represent a critical bottleneck for technological advancement. As device dimensions continue to shrink and power densities grow exponentially[1,2], conventional cooling methods are increasingly challenged to meet the demands of compact, high-performance systems. Traditional air cooling systems are limited by fundamental thermal diffusion properties, while vapor compression technologies encounter significant challenges including their excessive dimension, adverse environmental impact, and reduced efficiency when scaled down[2,3]. Electrocaloric (EC) cooling has emerged as a promising solution, offering efficient and compact thermal management capabilities that are stable for microelectronic applications. Solid-state EC cooling devices provide significant advantages, including enhanced performance, reduced energy consumption, and improved coefficient of performance (COP)[4]. Ceramic-based EC materials, such as PbSc0.5Ti0.5O3 ferroelectric ceramics, have demonstrated remarkable potential for effective thermal regulation by leveraging their unique EC properties at moderate electric fields[3,5]. However, conventional ceramic-based EC materials often suffer from inherent limitations, including mechanical rigidity and reliance on external driving components, which hinder their practical application. Although ferroelectric films can achieve a large EC effect under high electric fields, their total cooling capacity is constrained by the limited volume of the materials[6]. Additionally, challenges such as interfacial thermal resistance and inefficient domain switching could further restrict EC performance. Thus, optimized interfacial interactions and enhanced domain switching are essential for developing low-electric-field-driven solid-state cooling solutions with improved heat exchange efficiency and effective sub-ambient temperature modulation[7].
Significant progress has been made in EC materials and devices through recent advancements. High-performance ferroelectric polymers, particularly P(VDF-TrFE-CFE), have exhibited remarkable EC properties, with a calculated adiabatic temperature change of 12 K under an electric field of 307 MV/m[7]. Numerous performance enhancement strategies have been developed, including the incorporation of inorganic particles[8-12], optimization of thermal treatment[13], and introduction of conjugated double bonds[14,15], all of which have improved EC performance and also contributed to a reduction in the required electric field. Among them, incorporating functional inorganic nanofillers has shown remarkable effects. For example, Ca/Sn co-doped BaTiO3 (BCST) particles in P(VDF-TrFE) copolymers led to enhanced dielectric behavior and a ΔT of 6.96 K under 900 kV/cm[16]. Rare-earth-doped BaTiO3 nanopowders introduced into P(VDF-TrFE-CFE) improved EC strength (7-21 µK·m·kV-1) across 25-60 °C[17]. Additionally, boron nitride nanosheets (BNNS) dispersed in the terpolymer matrix significantly boosted breakdown strength and charge-discharge efficiency, offering a promising route to improve polymer EC composites[18]. Innovative device architectures have emerged as a promising approach. Single-unit electrostatic EC coolers have achieved 2.8 K temperature spans[19], while cascaded systems have demonstrated enhanced performance, achieving temperature spans of 4.8 K and 8.7 K in dual-unit and quad-unit configurations, respectively. Despite these advancements, achieving efficient thermal management in practical applications remains a critical challenge, particularly in enhancing interfacial heat transfer and optimizing fluid interactions[19-22].
A feasible solution to overcome these challenges involves the integration of EC cooling with microfluidic technology. Recent achievements in cascade EC cooling tubes have demonstrated substantial improvements, including temperature drops up to 4.6 K and temperature spans of 4.2 K at 10 mm/s flow rates[23]. Moreover, EC architecture featuring microfluidic channels incorporating electronic components has achieved cooling powers exceeding 1.7 kW/cm2[24]. Freestanding flexible EC capillaries have shown maximum temperature reductions of 3.53 K, with corresponding power densities of 702.1 mW/cm3[10]. Customized interfacial interactions, which accelerate ferroelectric phase switching and enhance heat exchange efficiency, are crucial for developing low-electric-field-driven solid-state cooling solutions.
Herein, we design a hydroxyl-functionalized composite that optimizes interfacial interactions within the P(VDF-TrFE-CFE) polymer matrix, enabling a synergistic balance between high thermal conductivity and enhanced EC performance. Finite element analysis (FEA) modeling and piezo response force microscopy (PFM) in situ characterization confirm that hydroxyl modification strengthens interfacial coupling, accelerating ferroelectric domain switching by amplifying the response of tetragonal P4mm phase domains under an electric field. As a result, the optimized system achieves improved electrocaloric temperature changes and thermal conductivity compared to the pure polymer. Moreover, the composite shows high cooling efficiency within a prototype device configuration. Such results highlight the role of microstructure design in advancing electrocaloric materials for future solid-state cooling applications.
MATERIALS AND METHODS
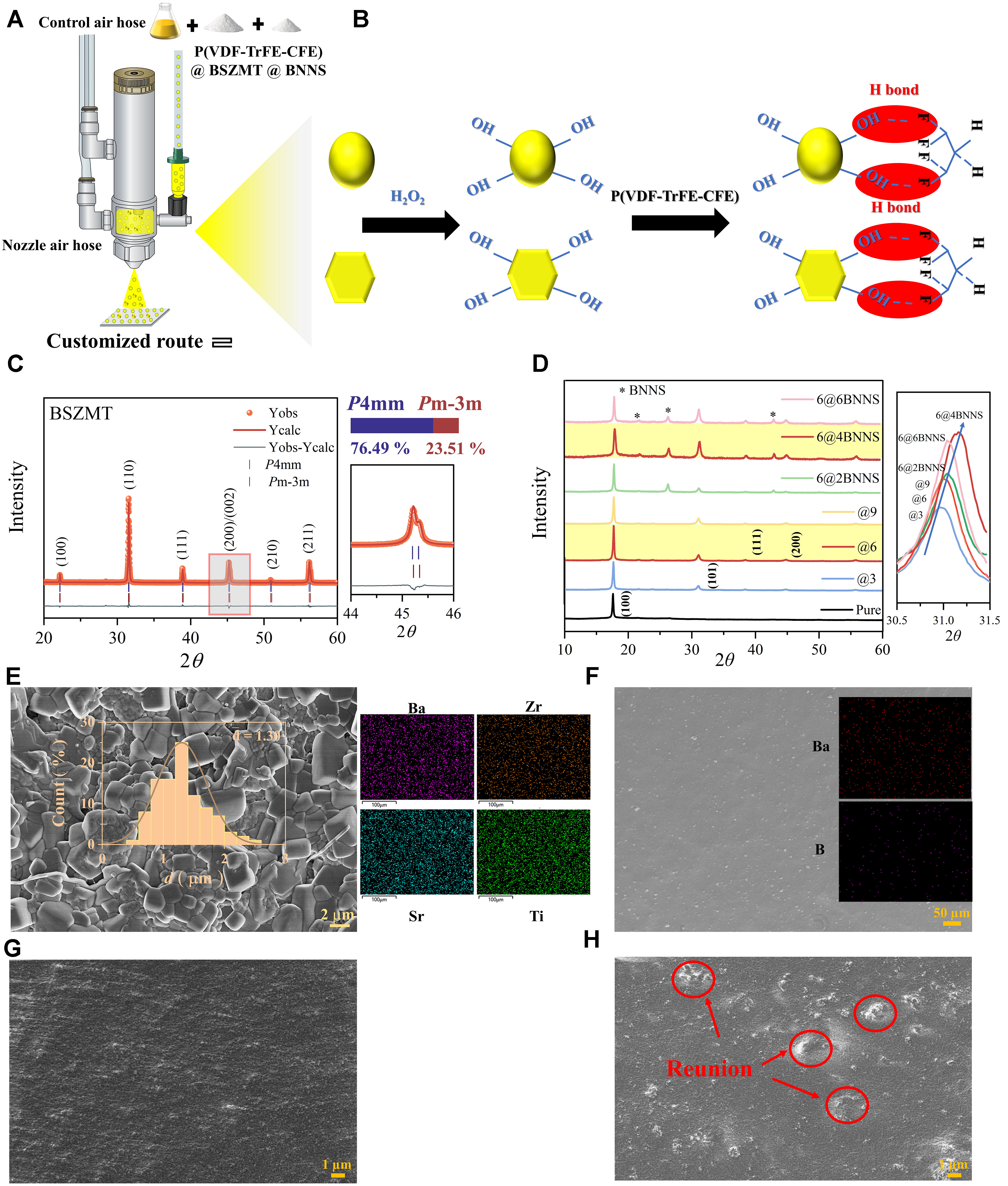
Ba0.63Sr0.37Zr0.01(Ti0.999Mn0.001)0.99O3 (BSZMT) ceramics were synthesized via a solid-state reaction method involving high-purity BaCO3, ZrO2, SrCO3, TiO2, and MnO2 (purity ≥ 99.0%, Aladdin Biochemical Technology Co., Ltd., Shanghai, China). The obtained powders were subjected to high-energy ball milling for 24 h to produce nanoscale particles (BSZMT-NPS), thereby enhancing the homogeneity of the microstructure. Detailed information on the synthesis process can be found in our previous work[25]. BSZMT-OH and hexagonal boron nitride nanosheets (h-BNNS) were then vacuum-dried at 120 °C for 12 h [Figure 1A and B]. BSZMT-NPS and hexagonal boron nitride nanosheets (h-BNNS, Model XFBN03-1, CAS 10043-11-5, XFNANO Materials Tech Co., Ltd., Nanjing, China) were individually dispersed in 35% hydrogen peroxide solution (300 mL, Nanjing Chemical Reagent Co., Ltd., Nanjing, China) and reacted at
Figure 1. (A) Schematic illustration of the customized fabrication route; (B) Hydroxyl functionalization process of BSZMT-NPs and h-BNNS; (C) XRD patterns of BSZMT ceramics, the inset showing diffraction peaks in the 44°-46°; (D) XRD patterns of nanocomposites with varying content, the inset showing peaks at ~31°; (E) SEM surface morphology and EDS of BSZMT ceramics, with the inset showing an average grain size; (F) SEM surface morphology and EDS of the 6@4BNNS nanocomposite; (G) SEM image of the hydroxyl-functionalized 6@4BNNS composite; (H) SEM image of the unfunctionalized 6@4BNNS composite surface. BSZMT: Ba0.63Sr0.37Zr0.01
Subsequently, a high-precision pressure spray-coating technique with customizable deposition paths was employed to fabricate P(VDF-TrFE-CFE)/BSZMT-OH composite films. Initially, 0.5 g of P(VDF-TrFE-CFE) (Piezotech Arkema, 67/27.4/5.6 mol) was divided into six portions, each mixed with BSZMT-OH at concentrations of 3, 6, and 9 vol.%, as well as h-BNNS-OH at 2, 4, and 6 vol.%. The volume percentages of BSZMT-OH and h-BNNS-OH were calculated based on the total volume of the polymer and fillers. For instance, a 3 vol% BSZMT-OH composite contains 3 vol% BSZMT-OH and 97 vol% P(VDF-TrFE-CFE). Similarly, hybrid compositions such as 6@2BNNS refer to a mixture of 6 vol% BSZMT-OH and 2 vol% h-BNNS-OH in total volume. The mixtures were uniformly dispersed in N,N-dimethylformamide (DMF) and stirred at room temperature for 6 h. The prepared solutions were then sprayed onto pre-cleaned glass substrates under a pressure of 15-20 MPa, forming uniform films with a thickness of approximately 40 µm. The films were subsequently dried at 90 °C to remove residual DMF, followed by annealing at 120 °C for 12 h to enhance crystallinity (3@, 6@, 9@, 6@2BNNS, 6@4BNNS, and 6@6BNNS, where the number before “@” denotes the vol% of BSZMT-OH, and the number before “BNNS” denotes the vol% of h-BNNS). Finally, the composite films were carefully peeled off from the glass substrates for further characterization and testing. The prepared 6@4BNNS composite was uniformly spray-coated onto both the front and back surfaces of a
The composites were prepared using a Nordson 781S series spray valve, and the phase structure was determined by X-ray diffraction (Rigaku Ultima IV), while the surface morphology was examined by SEM (ZEISS Crossbeam 350). Dielectric properties were measured with a dielectric spectrometer (DMS-20005PBVG, BALAB, Agilent E4990A), and the polarization-electric field (P-E) hysteresis loops were tested using a ferroelectric analyzer (TF-2000 E, Aix ACCT), and piezoelectric effects were analyzed with PFM (Oxford Instruments). For PFM measurements, we used an ASYELEC-01-R2 conductive AFM probe (Oxford Instruments) with a Ti/Ir (5/20 nm) coating on both the tip and lever. The tip radius is 25 ± 10 nm, and the spring constant is 2.8 N/m (range: 1.4-5.8 N/m), with a resonance frequency of ~75 kHz (range: 58-97 kHz). Dual AC resonance tracking (DART-PFM) mode was employed to enhance the signal-to-noise ratio and sensitivity to the local piezoelectric response. The scanning rate was maintained at 0.5-1 Hz to ensure stable contact and minimize drift during imaging. FTIR spectra were recorded using a Bruker INVENIO R Hyperion 1,000 spectrometer. Electrocaloric effect (ECE) performance was evaluated by collecting infrared (IR) temperature data with a HIKMICRO HM-TD203SH-4/X. A DC voltage was applied using a function generator and high-voltage amplifier (Aigtek, ATA-7025), alongside a compression testing machine (ZQ-990LB/0-5000N). The heat flux variations induced by ECE were measured using a heat flux sensor (Omega HFS-5). When voltage was applied or removed, the flux signal fluctuated due to the heat generated by ECE. TA software was used to extract Q values, which were then used to calculate ΔT based on
RESULTS AND DISCUSSION
A schematic diagram of the nanocomposite preparation is illustrated in Figure 1A and B, clearly showing the structural composition of the synthesized materials. The detailed fabrication procedure is provided in the Experimental Section. The XRD pattern confirms the phase purity of the BSZMT ceramic, with no residual precursor phases or secondary crystallization byproducts detected [Figure 1C]. The sharp diffraction peaks indicate that the BSZMT ceramic primarily exhibits a P4mm ferroelectric phase, accounting for 76.49%, while the pseudocubic Pm-3m phase at 23.51%. This phase coexistence arises from lattice distortion induced by Sr2+, which enhances the relaxor behavior. The successful preparation of a P4mm-dominated ceramic lays a robust foundation for the high ferroelectric performance of the ensuing nanocomposites, as corroborated by Rietveld refinement results (Rwp and Rp < 10.00%).
Further XRD analysis validates the structural integrity of the nanocomposites [Figure 1D]. The inset highlights a characteristic peak at ~31°, reflecting the crystalline phase evolution with increasing BNNS content. Notably, the 6@4BNNS composite exhibits the most pronounced tetragonal phase peak intensity, suggesting that optimized interfacial interactions enhance dipole alignment - a key factor in improving EC performance[13,26].
SEM imaging reveals a dense and uniform ceramic microstructure with an average grain size of ~1.30 µm [Figure 1E]. The corresponding EDS elemental mapping demonstrates the homogeneous distribution of Ba, Sr, Zr, and Ti, indicating well-integrated phases that promote superior interfacial compatibility. Additionally, SEM surface analysis and EDS mapping of the 6@4BNNS nanocomposite confirm the uniform dispersion of Ba and B elements [Figure 1F], reinforcing the hypothesis that optimized filler distribution plays a pivotal role in enhancing electrothermal performance. These findings underscore the structural and compositional advantages of the 6@4BNNS composite for next-generation EC applications. The presence of surface hydroxyl groups on BSZMT and h-BNNS was confirmed by Fourier-transform infrared (FTIR) spectroscopy [Supplementary Figure 1]. For both hydroxylated BSZMT and h-BNNS-OH, broad absorption bands centered around 3,400 cm-1 were observed, which are characteristic of -OH stretching vibrations, indicating successful grafting of hydroxyl groups during the chemical treatment process[27,28]. To further validate the impact of hydroxyl functionalization, Figure 1G presents the SEM image of the hydroxylated 6@4BNNS composite, revealing a highly uniform microstructure with minimal aggregation. In contrast, the unfunctionalized 6@4BNNS sample shown in Figure 1H exhibits noticeable filler agglomeration (marked by red circles), indicating poor dispersion of BNNS within the matrix. This comparison clearly demonstrates that hydroxyl functionalization significantly improves filler distribution, thereby contributing to enhanced structural integrity.
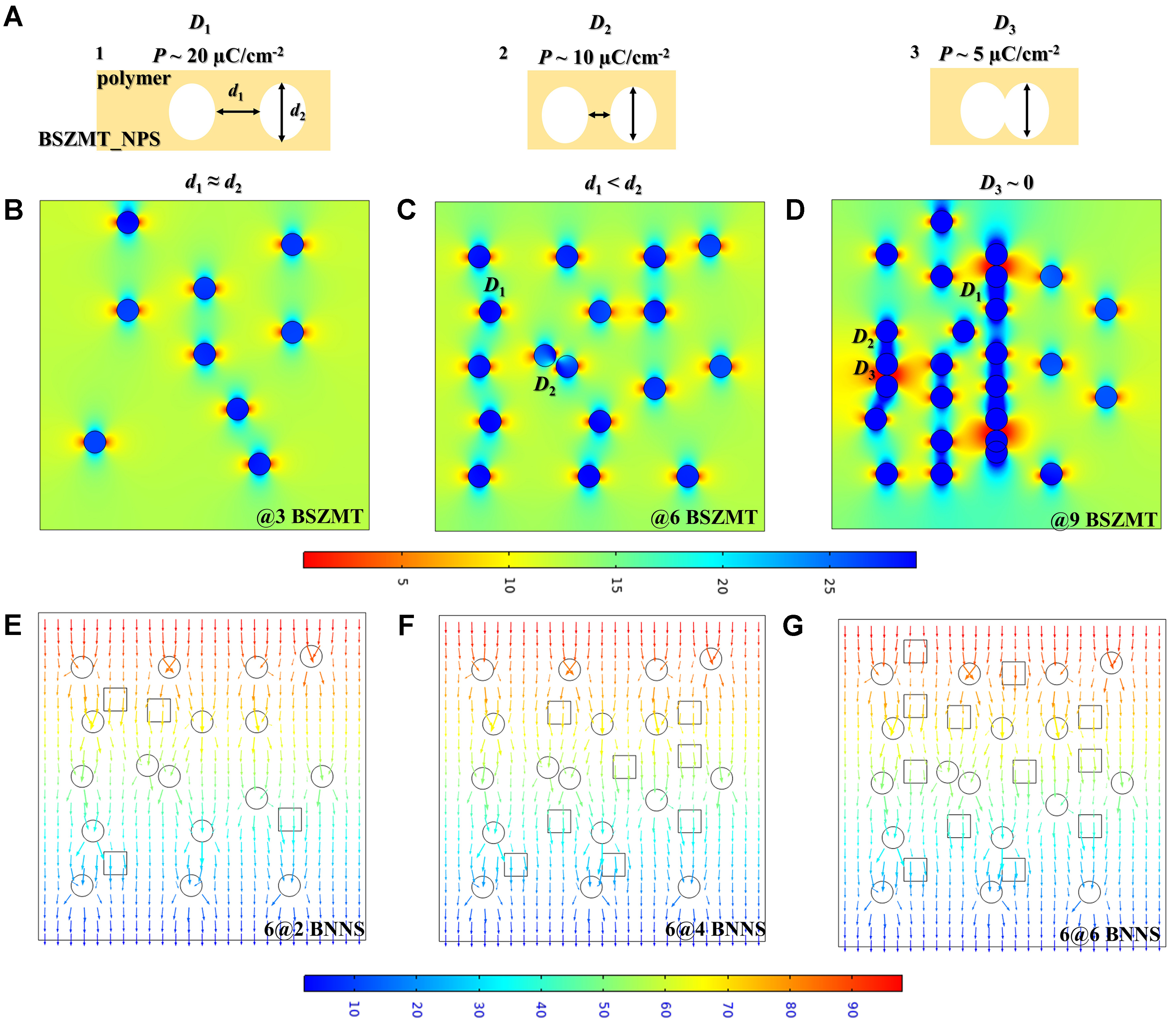
Phase-field simulations of ferroelectric behavior in BSZMT-terpolymer systems were conducted using FEA in COMSOL Multiphysics. The model is based on the Time-Dependent Landau-Ginzburg-Devonshire (TLGD) theory, describing polarization evolution through a semilinear parabolic partial differential equation. The local polarization vector P = (P1, P2, P3) serves as the order parameter governing ferroelectric domain formation and interactions. The polarization evolution follows the TLGD equation[26]:
where L is the kinetic coefficient. The total free energy F includes bulk, gradient, elastic, and electrostatic contributions:
The bulk energy follows a Landau polynomial expansion:
where α coefficients are material-dependent. The gradient energy term accounts for domain wall interactions:
Elastic energy incorporates strain effects:
where cijkl is the elastic stiffness tensor and Qijkl represents electrostrictive coupling. Finally, electrostatic energy follows:
To analyze the polarization response, a sinusoidal alternating electric field E(t) = E(0)sin(ωt) is applied along the Z-axis. Simulation results reveal that dipoles in the BSZMT system respond rapidly to the applied field, inducing pronounced variations in Pz (unit: C/m²). This fast response is attributed to the enhanced dipole alignment at the matrix-filler interfaces, which facilitates efficient polarization switching under the alternating electric field. Meanwhile, h-BNNS particles contribute to a more uniform electric field distribution (unit: MV/m), as their two-dimensional structure effectively screens the local field fluctuations and reduces the electric field gradient within the filler clusters.
As shown in Figure 2, the polarization strength and electric field intensity are predominantly concentrated at the matrix-filler interfaces, with the weakest fields observed inside the fillers. Among all configurations, the 6@4BNNS system exhibits the most significant polarization enhancement. This enhancement is primarily due to the optimized interfacial coupling between the hydroxyl-functionalized BSZMT nanoparticles and the h-BNNS nanosheets, which creates a favorable environment for uniform polarization switching. Further analysis indicates that isolated nanoparticles (D1, D2) form well-defined polar interfaces, enabling faster polarization responses. The distinct boundary conditions at these interfaces promote the formation of coherent polar domains, which enhance the overall polarization magnitude. However, as inter-particle distances decrease, interface overlap weakens the coherent polar configurations, thereby reducing the polarization strength (D3)[29]. This behavior aligns with percolation theory, which provides a framework for understanding the evolution of EC and dielectric properties in the composite system. To ensure accurate finite element modeling, representative material parameters were selected and are summarized in Supplementary Table 1. These include the Landau coefficients, dielectric constants, elastic stiffness values, and electrostriction constants for the P(VDF-TrFE-CFE) matrix, BSZMT ceramic nanoparticles, and h-BNNS fillers. These parameters serve as the physical basis for simulating the polarization behavior and field distribution within the composite system.
Figure 2. (A) Schematic diagram of polarization variation at different interparticle distances; (B-D) Polarization distribution Pz at different BSZMT (circle) loadings; (E-G) Electric field intensity distribution at different BNNS (square) contents. BSZMT: Ba0.63Sr0.37Zr0.01(Ti0.999Mn0.001)0.99O3; BNNS: boron nitride nanosheets.
Notably, 6 vol.% serves as a critical percolation threshold. Below this concentration, the internal electric field strength increases gradually with filler loading, as the individual nanoparticles begin to establish localized polar regions. Beyond 6 vol.% (e.g., 9 vol.%), a sharp increase in internal field strength is observed, driven by the formation of interconnected conductive pathways through aligned BNNS nanosheets. These pathways facilitate the redistribution of the electric field and enhance the coupling between the BSZMT nanoparticles and the h-BNNS nanosheets. This multi-scale structural optimization enhances the composite’s EC performance by improving both polarization magnitude and field uniformity, which are critical for achieving high energy efficiency and rapid thermal regulation in EC applications.
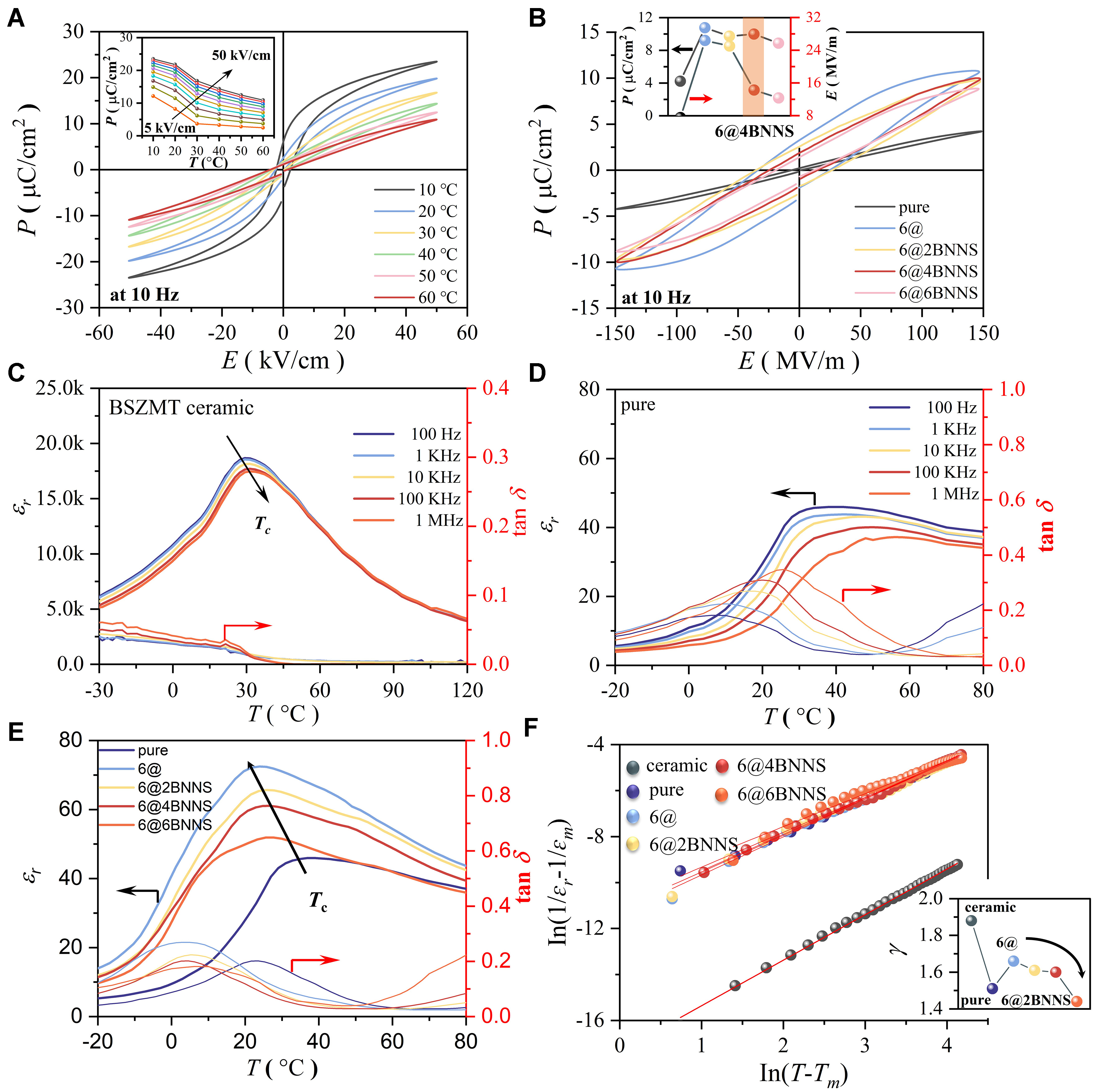
Figure 3 presents the electrical properties of different systems, focusing on polarization behavior, dielectric characteristics, and the degree of phase transition diffuseness. In Figure 3A, the pristine BSZMT ceramic demonstrates a relaxor-like ferroelectric signature, characterized by a slim P-E hysteresis loop with a high polarization strength of 23.50 µC/cm2 at 10 Hz and an ultralow coercive field. The temperature-dependent polarization inset reveals a sharp transition near the Curie temperature (Tc ≈ 25 °C), where the polarization variation (ΔP = 12.25 µC/cm2 between 10 and 60 °C at 50 KV/cm) underpins its high EC effect performance. Figure 3B contrasts the P-E responses of pure P(VDF-TrFE-CFE) and nanocomposites. While the pure polymer exhibits a modest saturation polarization of 4.23 µC/cm², the 6 vol% BSZMT composite (6@) achieves a 2.5-fold enhancement polarization strength to 10.77 µC/cm², albeit with an increased coercive field (26.37 MV/m) due to interfacial charge trapping. Strikingly, the synergistic incorporation of h-BNNS-OH nanosheets (6@4BNNS system) optimizes this trade-off: polarization strength stabilizes at 9.99 µC/cm² with a 46% reduction in the coercive field (14.26 MV/m), attributable to hydroxyl-mediated defect passivation and strain homogenization[28,30]. This interfacial tailoring not only reduces the driving field for polarization switching but also improves fatigue endurance, positioning the composite as a robust candidate for high-efficiency EC cooling.
Figure 3. (A) P-E hysteresis loops of BSZMT ceramics at 10 Hz, with the inset showing polarization variation from 10-60 °C; (B) Polarization properties of pure P(VDF-TrFE-CFE) and composite systems; (C) Temperature-dependent dielectric constant (εr) and dielectric loss (tanδ) of BSZMT ceramics at different frequencies; (D) Temperature-dependent dielectric constant and dielectric loss of pure P(VDF-TrFE-CFE); (E) Temperature-dependent dielectric constant and loss of composite systems at 100 Hz; (F) Phase transition diffuseness analysis of composite systems at 100 Hz, with the inset showing the diffusion index γ. BSZMT: Ba0.63Sr0.37Zr0.01
Figure 3C displays the temperature-dependent dielectric constant of BSZMT ceramics. At 100 Hz, the dielectric constant (εr) is approximately 18.68 K, slightly decreasing to 17.47 K at 1M Hz. With increasing frequency, the Tc shifts toward higher temperatures, reflecting a typical relaxor ferroelectric characteristic. This suggests that the phase transition process in this material is more gradual compared to conventional ferroelectrics, improving its temperature adaptability. Figure 3D illustrates the temperature-dependent dielectric constant and dielectric loss of pure P(VDF-TrFE-CFE). The results indicate that this system exhibits a relatively low dielectric constant near the phase transition temperature but is accompanied by significant dielectric loss, suggesting high internal dissipation during ferroelectric polarization switching.
Figure 3E further analyzes the temperature-dependent dielectric constant and loss characteristics of different systems at 100 Hz. Notably, the 6@ system exhibits the highest relative dielectric constant, reaching ~72.5 at 24 °C, indicating enhanced polarization under low electric fields. However, this also correlates with increased dielectric loss near the Curie temperature. Upon incorporation of hydroxyl-functionalized
Further analysis is conducted on the phase transition diffuseness coefficient (γ), which is calculated based on the modified Curie-Weiss law to characterize the diffuseness of the ferroelectric-to-paraelectric phase transition in different systems Figure 3F. The relationship is expressed as[31]:
where εr represents the maximum dielectric constant, and Tm is the corresponding temperature. A value of
In the undoped system, the γ value is relatively low, indicating a sharp phase transition. Upon incorporating BSZMT ceramic nanoparticles, γ increases significantly to 1.66, placing it within the characteristic relaxor ferroelectric range (1.5-2.0). This enhancement in relaxor behavior promotes a broadened phase transition over an extended temperature range. However, with the introduction of h-BNNS-OH nanosheets, γ decreases slightly, suggesting a reduction in phase transition diffuseness. Nevertheless, the 6@4BNNS system retains its relaxor ferroelectric nature with a γ value of 1.60, ensuring stable EC performance across a wide temperature range.
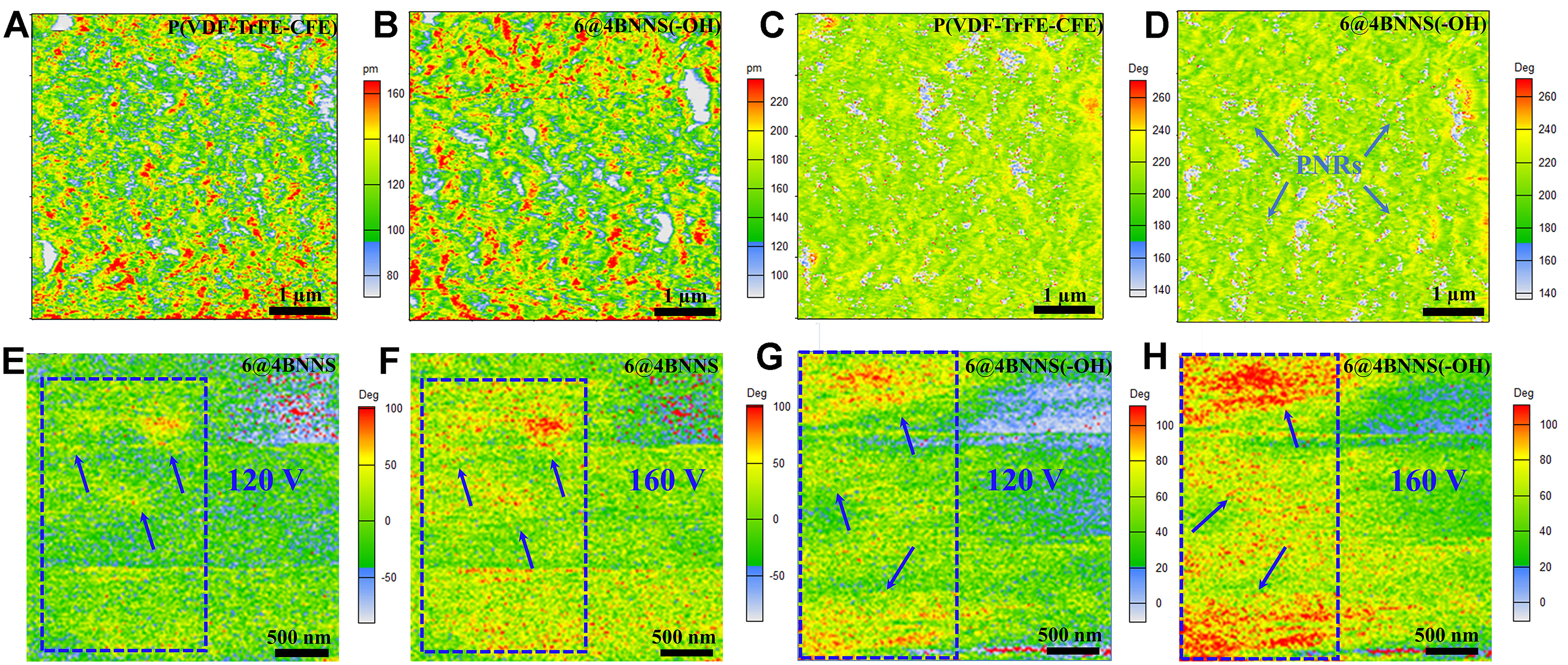
PFM was used to elucidate the correlation between microscopic mechanisms and electrothermal performance [Figure 4]. The PFM amplitude and phase images of pure P(VDF-TrFE-CFE) and the 6@4BNNS system provide insight into the ferroelectric domain structures [Figure 4A-D]. The introduction of 6 vol% BSZMT-OH nanoceramics significantly alters the domain morphology of the composite. Specifically, the incorporation of BSZMT nanoparticles disrupts the long-range ordered domains within the PVDF matrix, inducing the formation of uniformly distributed polar nanoregions (PNRs). This structural transition is clearly reflected in the more homogeneous contrast observed in both phase and amplitude images. Such a transformation from large domains to nanoscale PNRs plays a crucial role in enhancing the EC performance, as domain fragmentation leads to more relaxor-like polarization behavior, directly correlating with improved dielectric and EC properties. Notably, with the inclusion of ceramic nanoparticles, the PFM amplitude signal increases from the initial range of 80-180 to 100-220 pm, indicating an enhanced polarization response.
Figure 4. (A-D) PFM amplitude and phase images of pure P(VDF-TrFE-CFE) and 6@4BNNS composite; (E-H) Domain evolution under applied electric fields of 30 and 40 MV/m before and after hydroxylation. PFM: Piezo response force microscopy; BNNS: boron nitride nanosheets.
The electric field-dependent domain evolution Figure 4E-G further decouples the roles of hydroxylation in polarization switching. When an external field of 30 MV/m (120 V) was applied, domain walls began to move and expand, with new domains nucleating and growing by consuming adjacent structures. As shown in Figure 4E-H, the single-headed arrows mark regions where such domain switching behavior is evident, reflecting polarization reorientation under the applied field. As indicated by the previous XRD analysis, the switching mechanism in this process was primarily governed by the tetragonal P4mm phase. With an increased electric field of 40 MV/m (160 V), continuous nucleation and growth phenomena were observed. Notably, hydroxylation [6@4BNNS(-OH)] led to a visibly accelerated domain evolution rate at lower fields, as evidenced by denser domain reorientation regions (highlighted by arrows), indicating a faster polarization response. This suggests that the rapid response of the P4mm tetragonal phase plays a crucial role in enhancing the EC effect.
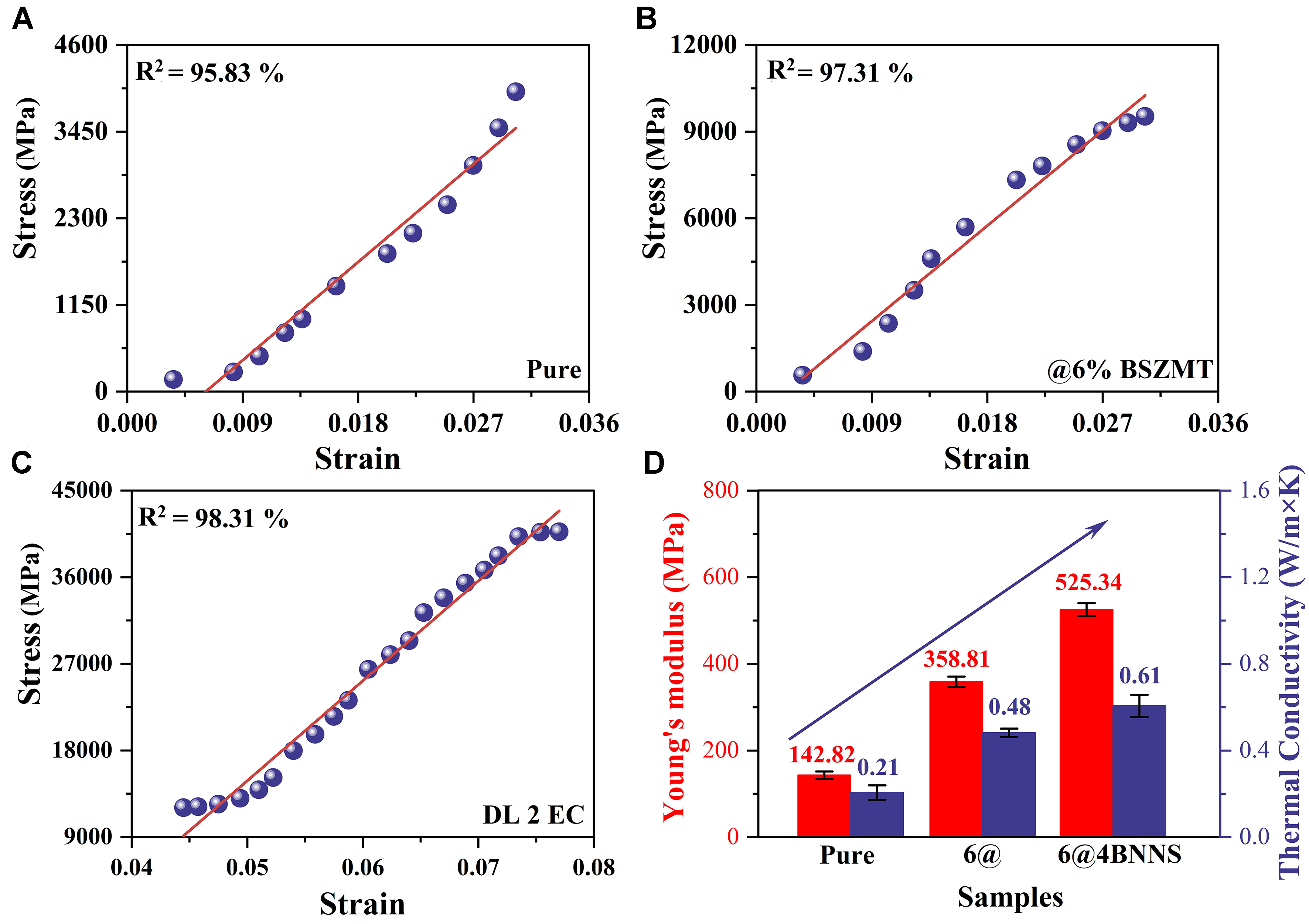
Figure 5 illustrates the mechanical and thermal conductivity properties of the materials. Figure 5A-C presents the stress-strain curves of pure P(VDF-TrFE-CFE), 6% BSZMT-OH composite, and the 6@4BNNS system, respectively, demonstrating a significant enhancement in mechanical strength upon nanofiller incorporation. Notably, the 6@4BNNS composite exhibits the highest peak stress (~25,000 MPa) and the best linear fit (R2 = 98.32%), highlighting the effect of interfacial reinforcement. Figure 5D further quantifies the variations in Young’s modulus and thermal conductivity, where the 6@4BNNS composite achieves a Young’s modulus of 519.17 MPa and a thermal conductivity of 0.68 W/m·K, both substantially higher than those of the pure polymer. These improvements underscore the material’s potential for mechanical reinforcement and thermal management, paving the way for high-efficiency EC cooling applications.
Figure 5. (A-C) Stress-strain curves for the different samples; (D) Young’s modulus values and Thermal conductivity measurements for the different samples. BSZMT: Ba0.63Sr0.37Zr0.01(Ti0.999Mn0.001)0.99O3; BNNS: boron nitride nanosheets.
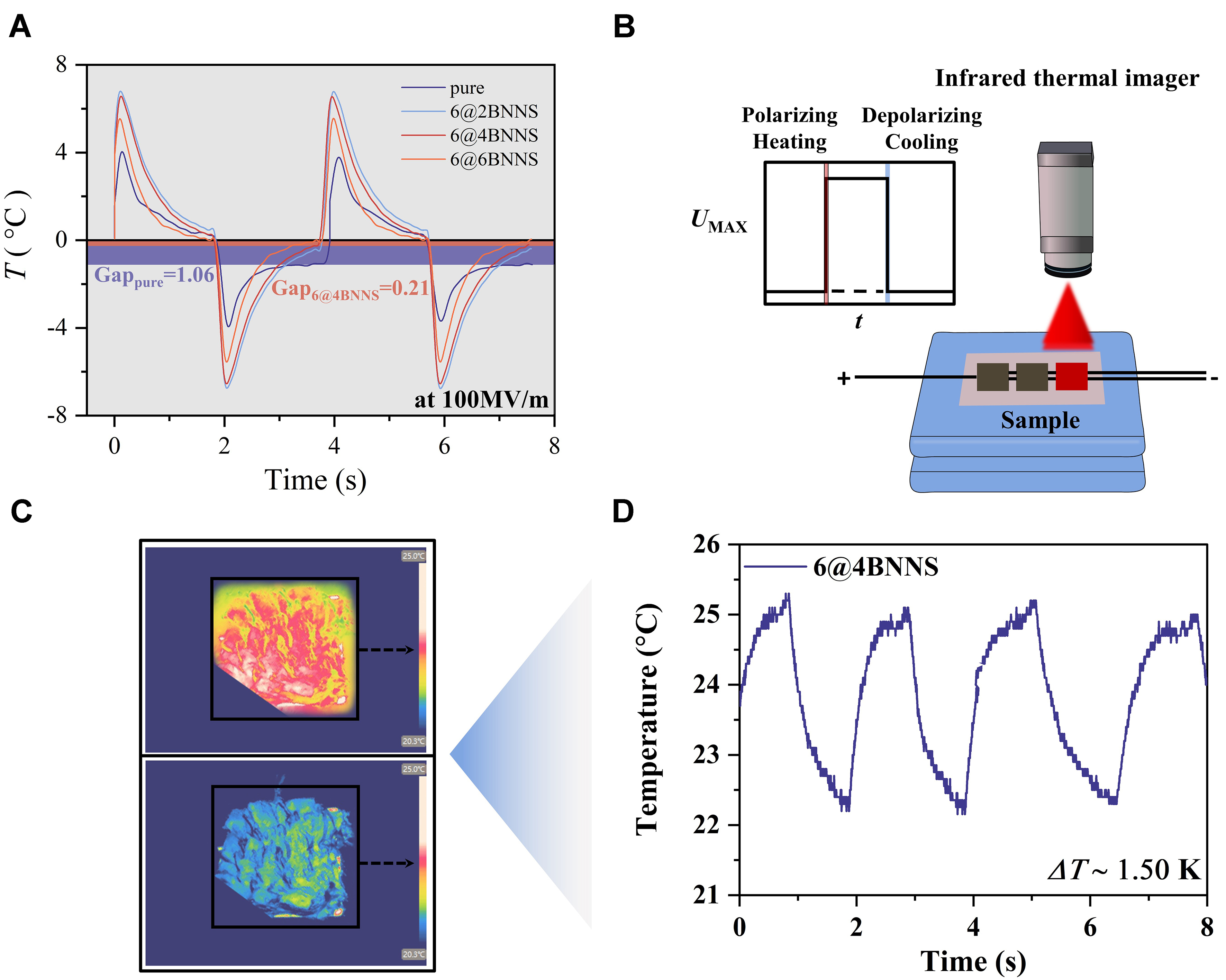
Figure 6 illustrates the EC effect in the 6@4BNNS composite, underscoring its potential for solid-state cooling applications. Figure 6A presents the direct EC response of the nanocomposite film under
Figure 6. (A) Thermodynamic cycling of EC films; (B) Schematic illustration of temperature measurement conducted using a thermocouple; (C) A macro IR image of the 6@4BNNS nanocomposite under 40 MV/m, showing applied (up) and removed (down) fields; (D) Time-temperature response curves of the 6@4BNNS nanocomposite under 40 MV/m at 0.5 Hz. EC: Electrocaloric; BNNS: boron nitride nanosheets; IR: infrared.
Figure 6B presents a schematic of the IR imaging setup used to track temperature evolution. The macroscopic IR images in Figure 6C further depict the temperature distribution under a 40 MV/m electric field, where the upper image represents the heating state and the lower image corresponds to the cooling state[23]. Notably, due to factors such as emissivity and surface reflectivity, the observed temperature may be lower than the actual value. Figure 6D displays the time-dependent temperature response under a 40 MV/m alternating field at 0.5 Hz, revealing a temperature oscillation of approximately 1.50 K, with values fluctuating between 21~26 °C. This dynamic thermal behavior highlights the potential of 6@4BNNS for thermal management applications, especially in scenarios requiring low electric field operation and efficient heat exchange near room temperature.
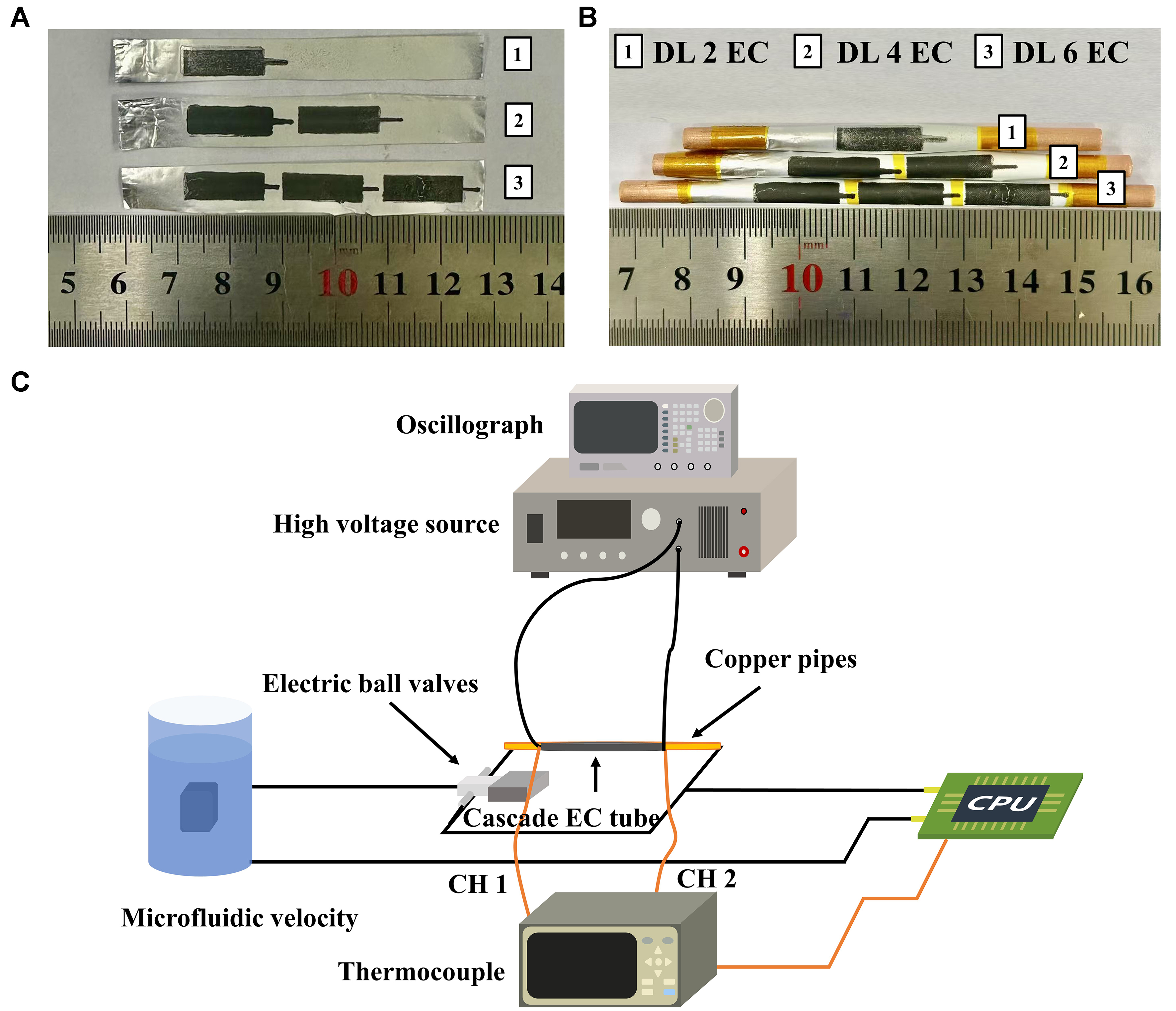
The coated foil was carefully rolled onto a copper tube to construct a double-layer EC device with varying section numbers (DL 2EC, DL 4EC, and DL 6EC), forming a cylindrical structure with dimensions of φ
Figure 7. (A) Electrocaloric devices with different layer numbers (DL 2EC, DL 4EC and DL 6EC); (B) Side view of the assembled devices; (C) Schematic of the thermal performance testing setup. DL: Double-layer; EC: electrocaloric.
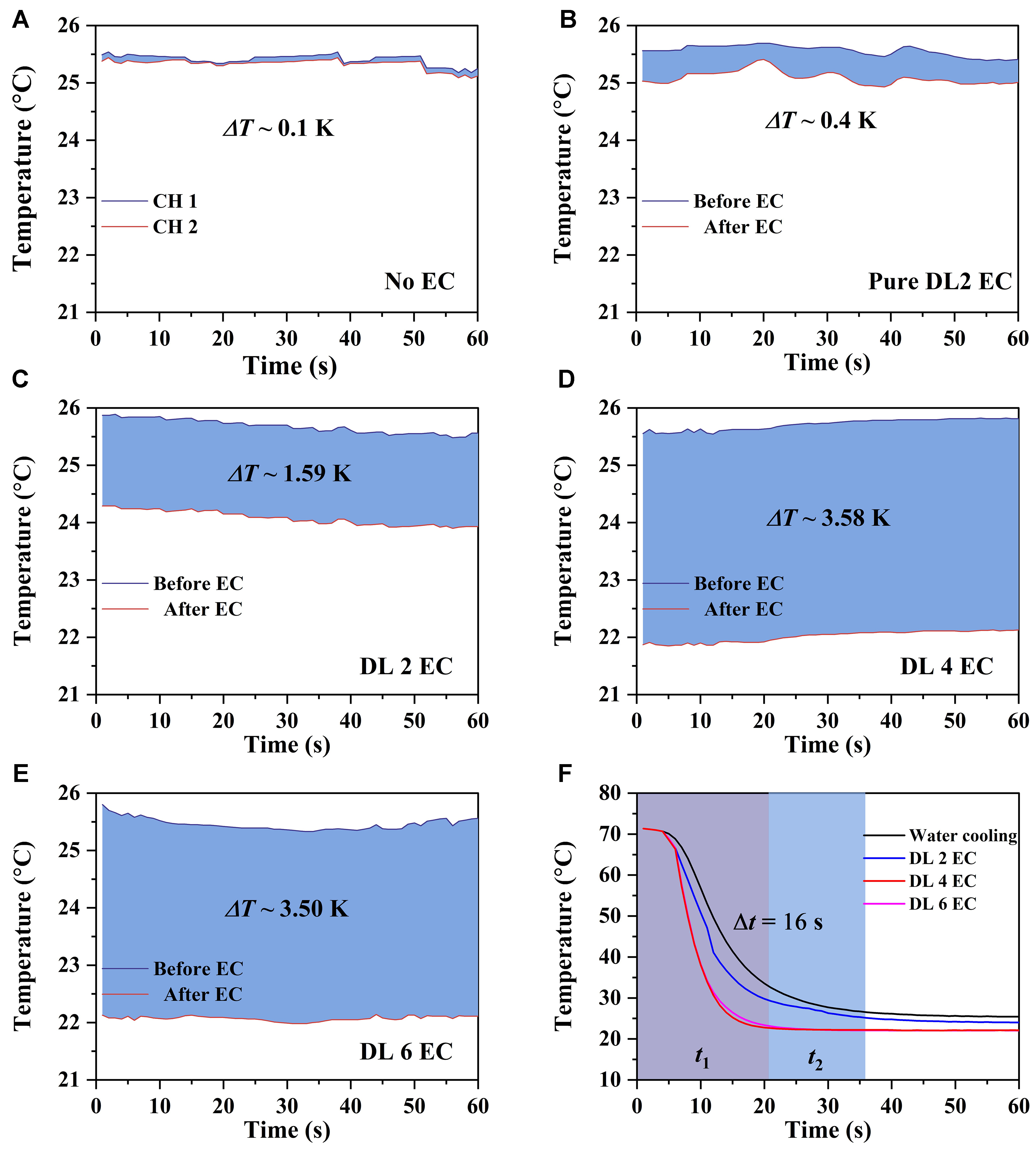
Figure 8 shows the temperature variation of EC cooling at a low electric field of 40 MV/m using a thermocouple, demonstrating the performance of the DL EC device based on the modification of 6@4BNNS material (detailed data provided in Supplementary Table 2). The measurements were conducted at a working frequency of 0.5 Hz. Figure 8A shows the baseline measurement without EC cooling (ΔT ~ 0.1 K), confirming the reliability of the thermocouple data. Figure 8B illustrates the cooling performance of the pure DL 2 EC configuration, which achieves a modest temperature drop (ΔT ~ 0.4 K), reflecting the inherent limitations of the polymer-only system.
Figure 8. (A) Baseline temperature profile without electrocaloric effect; (B) Temperature response for the pure DL 2 EC configuration; (C) Performance of the DL 2 EC sample; (D) Temperature response for the DL 4 EC configuration; (E) Performance of the DL 4 EC configuration; (F) Comprehensive overview of temperature evolution across all samples. EC: Electrocaloric; DL: double-layer.
With the integration of the 6@4BNNS composite, a significant enhancement in EC cooling performance is observed. Figures 8C-E depict the cooling effects of DL 2 EC, DL 4 EC, and DL 6 EC, achieving temperature reductions of ΔT ~ 1.59, 3.58, and 3.50 K, respectively. The thermocouple data (CH1, CH2) record the temperature changes before and after the fluid enters the DL EC device, confirming effective heat transfer. Notably, the DL 4 EC system achieves the highest cooling efficiency, while the DL 6 EC system exhibits a slight decrease in cooling effectiveness, indicating that increasing the number of layers does not result in a strictly linear improvement due to heat transfer limitations in the fluid medium[23].
Figure 8F provides a comparative analysis of various EC configurations alongside water cooling. The DL 4 EC system demonstrates the most rapid cooling capability, reducing CPU temperature by 3.58 K and achieving this result 16 s faster than water cooling. These findings validate the superior cooling performance of 6@4BNNS-based EC devices, establishing them as a scalable and efficient solution for high-performance thermal management in microelectronics.
In conclusion, this study demonstrates a hydroxyl-functionalized BSZMT-OH@h-BNNS-OH composite with tailored interfacial hydrogen bonding, enabling enhanced dipole response and improved EC performance in a P(VDF-TrFE-CFE) matrix. FEA and in situ PFM confirm strengthened interfacial coupling and accelerated domain switching kinetics, leading to a peak ΔT of 1.59 K under 40 MV/m in the optimized 6@4BNNS system. These findings establish an effective strategy for microstructure-driven EC enhancement by optimizing interfacial interactions and domain dynamics. To validate its practical feasibility, the composite was integrated into a DL 4 EC device, demonstrating rapid cooling from 70 to
CONCLUSIONS
The objective of this study was to design a microstructure-engineered nanocomposite with enhanced interfacial coupling to optimize both thermal conductivity and electrocaloric performance. Our results demonstrate that hydroxyl functionalization strengthens interfacial hydrogen bonding, which accelerates ferroelectric domain switching kinetics and improves heat exchange efficiency. This work provides a scalable and efficient strategy for developing next-generation solid-state thermal management solutions in high-performance microelectronics.
DECLARATIONS
Authors’ contributions
Design, manuscript revision and supervision: Liu, L.; Ke, Q.
Experiments and data collection: Tang, S.; Meng, Y.; Li, D.; Zhang, G.
Data analysis: Tang, S.
Manuscript writing: Tang, S.
All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Guangdong Province International Science and Technology Cooperation Research Project (2023A0505050101), the Science and Technology Plan of Guangxi (AA23023027), the Key Research and Development Program of Shandong Province (2022CXGC020203), and the Bagui Scholar Project.
Conflicts of interest
Liu, L. is the Guest Editor of the Special Issue “Role of Microstructures in the High-performance of Advanced Dielectric Materials and Devices” but was not involved in any steps of editorial processing, notably including reviewer selection, manuscript handling, or decision making. The other authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Velders, G. J.; Fahey, D. W.; Daniel, J. S.; McFarland, M.; Andersen, S. O. The large contribution of projected HFC emissions to future climate forcing. Proc. Natl. Acad. Sci. U. S. A. 2009, 106, 10949-54.
2. Shah, N. K.; Wei, M.; Letschert, V.; Phadke, A. A. Benefits of leapfrogging to superefficiency and low global warming potential refrigerants in room air conditioning. https://www.osti.gov/biblio/1397235 (accessed 2025-06-06).
3. Torelló, A.; Lheritier, P.; Usui, T.; et al. Giant temperature span in electrocaloric regenerator. Science 2020, 370, 125-9.
4. Hou, H.; Qian, S.; Takeuchi, I. Materials, physics and systems for multicaloric cooling. Nat. Rev. Mater. 2022, 7, 633-52.
5. Li, J.; Torelló, A.; Kovacova, V.; et al. High cooling performance in a double-loop electrocaloric heat pump. Science 2023, 382, 801-5.
6. Hu, Q.; Tian, Y.; Zhu, Q.; et al. Achieve ultrahigh energy storage performance in BaTiO3-Bi(Mg1/2Ti1/2)O3 relaxor ferroelectric ceramics via nano-scale polarization mismatch and reconstruction. Nano. Energy. 2020, 67, 104264.
7. Neese, B.; Chu, B.; Lu, S. G.; Wang, Y.; Furman, E.; Zhang, Q. M. Large electrocaloric effect in ferroelectric polymers near room temperature. Science 2008, 321, 821-3.
8. Zhang, G.; Li, Q.; Gu, H.; et al. Ferroelectric polymer nanocomposites for room-temperature electrocaloric refrigeration. Adv. Mater. 2015, 27, 1450-4.
9. Zhang, G.; Zhang, X.; Yang, T.; et al. Colossal room-temperature electrocaloric effect in ferroelectric polymer nanocomposites using nanostructured barium strontium titanates. ACS. Nano. 2015, 9, 7164-74.
10. Cui, H.; Zhang, Q.; Bo, Y.; et al. Flexible microfluidic electrocaloric cooling capillary tube with giant specific device cooling power density. Joule 2022, 6, 258-68.
11. Qian, J.; Peng, R.; Shen, Z.; et al. Interfacial coupling boosts giant electrocaloric effects in relaxor polymer nanocomposites: in situ characterization and phase-field simulation. Adv. Mater. 2019, 31, e1801949.
12. Lu, S. G.; Rožič, B.; Zhang, Q. M.; Kutnjak, Z.; Neese, B. Enhanced electrocaloric effect in ferroelectric poly(vinylidene-fluoride/trifluoroethylene) 55/45 mol % copolymer at ferroelectric-paraelectric transition. Appl. Phys. Lett. 2011, 98, 122906.
13. Qian, J.; Jiang, J.; Shen, Y. Enhanced electrocaloric strength in P(VDF-TrFE-CFE) by decreasing the crystalline size. J. Materiomics. 2019, 5, 357-62.
14. Qian, X.; Han, D.; Zheng, L.; et al. High-entropy polymer produces a giant electrocaloric effect at low fields. Nature 2021, 600, 664-9.
15. Le Goupil, F.; Kallitsis, K.; Tencé-girault, S.; et al. Enhanced electrocaloric response of vinylidene fluoride-based polymers via one-step molecular engineering. Adv. Funct. Mater. 2021, 31, 2007043.
16. Tokkan, M.; Demir, M. M.; Adem, U. Enhanced electrocaloric effect of P(VDF-TrFE)-based nanocomposites with Ca and Sn co-doped BaTiO3 particles. Ceram. Int. 2023, 49, 2904-10.
17. Wang, Z.; Gao, Y.; Ma, Y.; Xie, X.; Yang, M.; Zhang, H. Enhanced electrocaloric effect within a broad temperature range in lead-free polymer composite films by blending the rare-earth doped BaTiO3 nanopowders. Adv. Compos. Hybrid. Mater. 2021, 4, 469-77.
18. Shang, Y.; Feng, Y.; Zhang, C.; Zhang, T.; Chi, Q. Improvement of energy storage density of P(VDF-TrFE-CFE) composites by filling with two dimensional BNNS nanosheets. IET. Conf. Proc. 2022, 2021, 1939-43.
19. Ma, R.; Zhang, Z.; Tong, K.; et al. Highly efficient electrocaloric cooling with electrostatic actuation. Science 2017, 357, 1130-4.
20. Bo, Y.; Zhang, Q.; Cui, H.; et al. Electrostatic actuating double‐unit electrocaloric cooling device with high efficiency. Adv. Energy. Mater. 2021, 11, 2003771.
21. Kalizan, J.; Tušek, J. Caloric micro-cooling: numerical modelling and parametric investigation. Energy. Convers. Manag. 2020, 225, 113421.
22. Wang, H.; Meng, Y.; Zhang, Z.; et al. Self-actuating electrocaloric cooling fibers. Adv. Energy. Mater. 2020, 10, 1903902.
23. Bai, P.; Cui, H.; Zhang, D.; Bo, Y.; Liu, L.; Ma, R. A highly efficient cascade electrocaloric cooling tube with enhanced temperature change by sawtooth voltage. Next. Materials. 2023, 1, 100001.
24. van Erp R, Soleimanzadeh R, Nela L, Kampitsis G, Matioli E. Co-designing electronics with microfluidics for more sustainable cooling. Nature 2020, 585, 211-6.
25. Tang, S.; Huan, C.; Zhou, X.; et al. Selective forming process for microstructures engineering of BST-based ceramics to achieve promoted electrocaloric performance. Ceram. Int. 2025, 51, 10174-83.
26. Zhao, M.; Sun, X.; Ji, P.; et al. Giant electrocaloric effect and breakdown field strength in ferroelectric terpolymer nanocomposites by morphologically diverse nanostructures. Nano. Energy. 2024, 128, 109948.
27. Ravichandran, V.; Chandrashekar, A.; Prabhu, T. N.; Varrla, E. SPI-modified h-BN nanosheets-based thermal interface materials for thermal management applications. ACS. Appl. Mater. Interfaces. 2024, 16, 34367-76.
28. Liu, S.; Xiu, S.; Shen, B.; Zhai, J.; Kong, L. B. Dielectric properties and energy storage densities of poly(vinylidenefluoride) nanocomposite with surface hydroxylated cube shaped Ba0.6Sr0.4TiO3 nanoparticles. Polymers 2016, 8, 45.
29. Li, X.; He, S.; Jiang, Y.; et al. Unraveling bilayer interfacial features and their effects in polar polymer nanocomposites. Nat. Commun. 2023, 14, 5707.
30. Zhao, Y.; Zhang, R.; Chen, Y.; et al. Interfacial lattice coupling engineering in all-inorganic coupled flexible films for dielectric energy storage. Acta. Mater. 2024, 264, 119554.
31. Maiti, T.; Guo, R.; Bhalla, A. S. Structure-property phase diagram of BaZrxTi1-xO3 system. J. Am. Ceram. Soc. 2008, 91, 1769-80.
32. Liu, Y.; Scott, J. F.; Dkhil, B. Direct and indirect measurements on electrocaloric effect: recent developments and perspectives. Appl. Phys. Rev. 2016, 3, 031102.
33. Moya, X.; Kar-Narayan, S.; Mathur, N. D. Caloric materials near ferroic phase transitions. Nat. Mater. 2014, 13, 439-50.
34. Nair, B.; Usui, T.; Crossley, S.; et al. Large electrocaloric effects in oxide multilayer capacitors over a wide temperature range. Nature 2019, 575, 468-72.
35. Qian, X.; Ye, H.; Zhang, Y.; et al. Giant electrocaloric response over a broad temperature range in modified BaTiO3 ceramics. Adv. Funct. Mater. 2014, 24, 1300-5.
36. Shi, J.; Han, D.; Li, Z.; et al. Electrocaloric cooling materials and devices for zero-global-warming-potential, high-efficiency refrigeration. Joule 2019, 3, 1200-25.
37. Wang, Y.; Zhang, Z.; Usui, T.; et al. A high-performance solid-state electrocaloric cooling system. Science 2020, 370, 129-33.
38. Kitanovski, A.; Plaznik, U.; Tomc, U.; Poredoš, A. Present and future caloric refrigeration and heat-pump technologies. Int. J. Refrig. 2015, 57, 288-98.
39. Mañosa, L.; Planes, A. Materials with giant mechanocaloric effects: cooling by strength. Adv. Mater. 2017, 29, 1603607.
40. Pu, Y.; Zhang, Q.; Li, R.; Chen, M.; Du, X.; Zhou, S. Dielectric properties and electrocaloric effect of high-entropy (Na0.2Bi0.2Ba0.2Sr0.2Ca0.2)TiO3 ceramic. Appl. Phys. Lett. 2019, 115, 223901.
41. Peng, B.; Tang, S.; Lu, L.; et al. Low-temperature-poling awakened high dielectric breakdown strength and outstanding improvement of discharge energy density of (Pb,La)(Zr,Sn,Ti)O3 relaxor thin film. Nano. Energy. 2020, 77, 105132.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].