Phase engineering enhanced upconversion luminescence in ultrasmall Li+-doped Cs2ZrF6:Yb/Er nanocrystals for ratiometric optical thermometry
Abstract
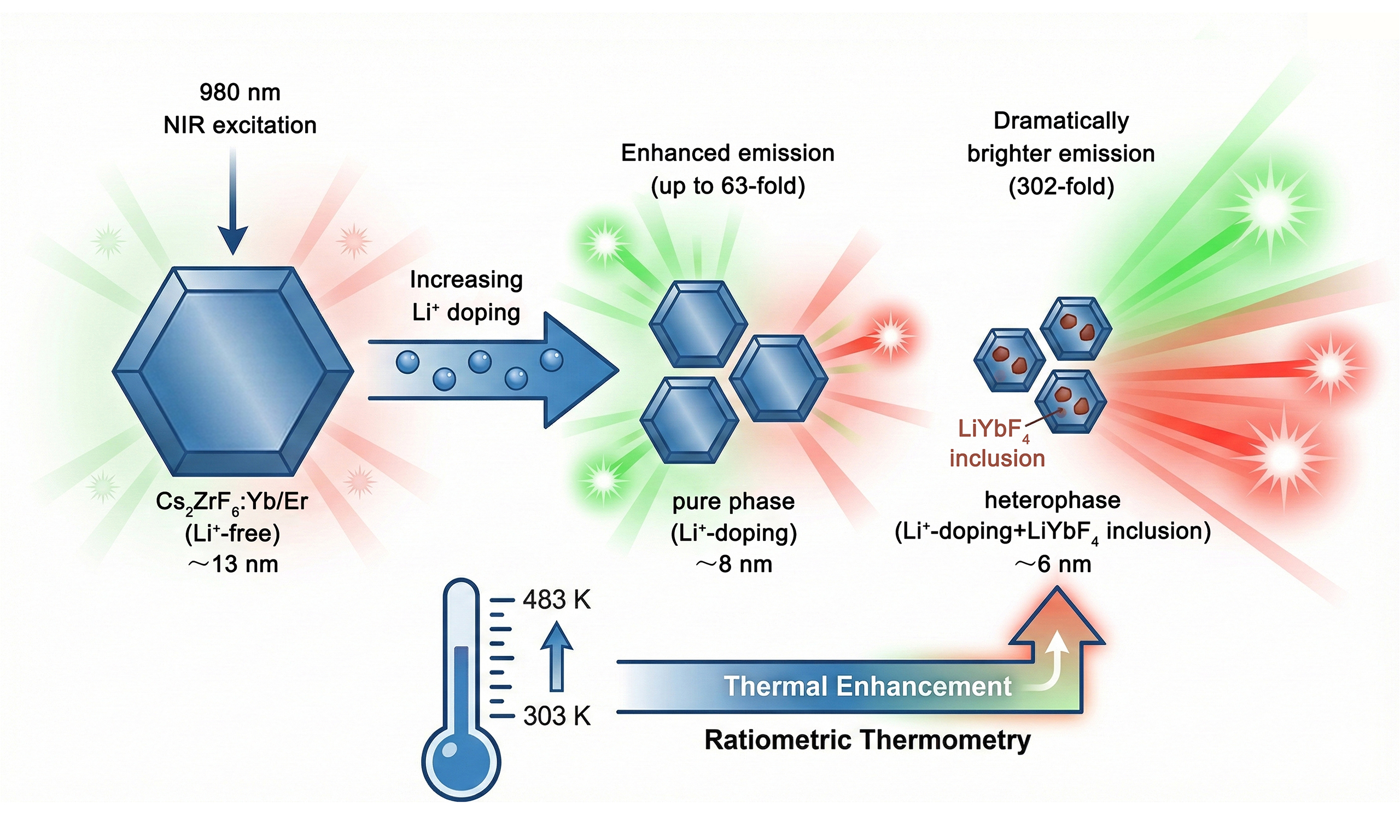
Lanthanide-doped ultrasmall upconversion nanocrystals exhibit unique optical properties that differ significantly from bulk materials, making them highly promising for diverse applications. However, their widespread applications have long been impeded by extremely weak photoluminescence due to severe surface quenching effect. Herein, we present a Li+-doping-induced phase engineering strategy to achieve significantly enhanced upconversion luminescence in ultrasmall Cs2ZrF6:Yb/Er nanocrystals (NCs). While initial Li+ doping improves crystallinity, yielding up to a 63-fold emission increase, further doping triggers a phase transition to form heterophase nanocrystals consisting of trigonal Cs2ZrF6 and tetragonal LiYbF4. The synergistic effect of improved crystallinity and reduced local symmetry around lanthanide ions results in a 302-fold boost in emission intensity, even as the particle size decreases to ~6.1 nm. Moreover, these ultrasmall nanocrystals display anomalous anti-thermal quenching behavior, with luminescence intensity increasing as temperature rises from 303 to 483 K, driven by defect-mediated energy repopulation. This work not only offers a robust approach for fabricating ultrasmall, high-brightness NCs but also establishes phase engineering as a pivotal mechanism for modulating local crystal field, paving the way for high-performance, thermally stable ratiometric nanothermometry.
Keywords
INTRODUCTION
Lanthanide (Ln3+)-doped inorganic nanocrystals (NCs) possess exceptional optical properties including large Stokes shifts, narrow emission bands, long photoluminescence lifetimes and high photostability, outperforming conventional luminescent materials like organic dyes and quantum dots[1-5]. In particular, Ln3+-doped upconversion NCs can convert long-wavelength near-infrared (NIR) excitation into short-wavelength visible or NIR emissions, rendering them highly promising for a broad range of applications such as light-induced switching, data storage, solar cells, anti-counterfeiting, temperature sensing and bioimaging[6-13]. Among NCs of various dimensions, sub-10 nm ultrasmall NCs have attracted considerable interest for both fundamental research and practical use, owing to their unique optical properties that are distinctly different from those of their larger counterparts[14-19]. Despite these significant advances, Ln3+-doped NCs usually suffer from severe luminescence quenching, leading to quite low quantum yields that greatly hinder their practical application. This issue is especially pronounced in sub-10 nm ultrasmall NCs due to the well-known surface quenching effect associated with their high surface-to-volume ratio[20]. Therefore, it is essential to develop strategies that mitigate the detrimental surface quenching effects while retaining the beneficial properties of ultrasmall NCs.
Compared to numerous approaches that require either complicated synthetic processes or tedious post-treatments, impurity doping has proven to be a simple yet effective strategy for achieving luminescence enhancement of Ln3+-doped NCs[21-25]. This method involves the intentional introduction of foreign ions into the host lattice, enabling precise control over the morphology, size, phase structure, as well as optical property of NCs simply by varying the dopant concentration. Among various dopants, lithium ion (Li+) has attracted significant attention due to its small ionic radius, which allows facile incorporation into the crystal lattice, either substitutionally or interstitially[26-29]. By incorporating Li+ into the crystal lattice, it is possible to modulate the morphology and size, improve the particle crystallinity and modify the crystal-field environment of Ln3+ emitters, thereby achieving upconversion luminescence (UCL) enhancement of Ln3+-doped NCs[30-36]. For example, Wang et al. demonstrated that doping with 60 mol% Li+ in KSc2F7:Yb/Er NCs resulted in an approximately 21 times luminescence enhancement relative to the Li+-free NCs[30]. The UCL enhancement originated primarily from a variation in the local crystal field of Ln3+ dopants due to Li+ incorporation, along with a contribution from the increased particle size. Dou et al. reported that Li+ doping in NaYF4:Yb/Er NCs triggered a phase evolution from hexagonal to cubic NaYF4 and finally to tetragonal LiYF4, concomitant with significant alterations in their size, shape and luminescent performance[31]. Since then, Li+-doping-induced alterations in structure and emission spectra have been reported in various rare earth fluorides, such as NaLnF4 and LnF3[28,32-36]. Beyond fluorides, the versatility and broad applicability of Li+ doping have also been validated in various host matrices, such as oxides, phosphates, molybdates, vanadates, titanates, and tungstates[37-43]. Nonetheless, simultaneously achieving precise size control below 10 nm and UCL enhancement in ultrasmall NCs remains a formidable challenge.
In this work, we developed a Li+-doping-induced phase engineering strategy to achieve enhanced UCL in ultrasmall Cs2ZrF6:Yb/Er NCs. Through detailed structural characterization, the influence of Li+ doping on the morphology, size, and phase structure of the NCs was systematically investigated. With increasing Li+ doping concentration up to 35 mol%, the NCs retained the pure Cs2ZrF6 phase and showed a gradual enhancement in UCL intensity. Further increasing the Li+ content to 38 mol% resulted in the formation of heterophase NCs, consisting of both Cs2ZrF6 and LiYbF4 phases. This phase transition led to a remarkable 302-fold enhancement in emission intensity compared to the Li+-free counterparts, despite a reduction in size of over 50%. In addition, abnormal UCL thermal enhancement was observed in these ultrasmall NCs, enabling the development of high-performance luminescent nanothermometers with excellent optical thermometric properties. This study provides an effective strategy for markedly improving the UCL performance of ultrasmall NCs, thereby promoting their potential applications in various areas such as optical nanothermometry and bioimaging.
MATERIALS AND METHODS
Materials
Cs2CO3, CH3COOLi and NH4F were purchased from Aladdin (China). Y(CH3CO2)3·4H2O (99.999%), Yb(CH3CO2)3·4H2O (99.999%), Er(CH3CO2)3·4H2O (99.99%), Zr(CH3COO)4, oleic acid (OA) and 1-octadecene (ODE) were purchased from Sigma-Aldrich (China). Cyclohexane, methanol, and ethanol were purchased from Sinopharm Chemical Reagent Co., China. All chemical reagents were used directly without further purification.
Synthesis of Cs2ZrF6:Yb/Er NCs with and without Li+-doping
In a typical procedure for the synthesis of Cs2ZrF6:Yb/Er NCs with and without Li+-doping (CZF-Li-X NCs), a mixture of (1.5-x)/2 mmol Cs2CO3, x mmol CH3COOLi (x = 0, 0.15, 0.3, 0.45, 0.525, and 0.57), 0.39 mmol Zr(CH3COO)4, 0.1 mmol Yb(CH3COO)3·4H2O, and 0.01 mmol Er(CH3CO2)3·4H2O was added to a 100 mL three-necked round-bottom flask containing a solvent mixture of OA (8 mL) and ODE (16 mL). The mixture was heated to 150 °C and stirred continuously for 60 min under a nitrogen atmosphere to form a clear solution, which was subsequently cooled to room temperature. Then, 10 mL of a methanol solution containing 3 mmol NH4F was introduced, and the resulting mixture was stirred for an additional 30 min. After evaporating the methanol, the reaction was carried out at 280 °C for 60 min under vigorous stirring and nitrogen flow. The final product was cooled to room temperature, precipitated with ethanol, collected via centrifugation, washed several times with ethanol, and ultimately redispersed in cyclohexane for further use.
Characterization and measurement
Powder X-ray diffraction (XRD) patterns were collected on a Rigaku MiniFlex2 diffractometer (Cu Kα radiation, λ = 1.5418 Å). In situ high-temperature XRD measurements were performed on a Bruker D8 Advance diffractometer (Cu Kα source, λ = 1.5406 Å) operated at 40 kV and 40 mA. X-ray photoelectron spectroscopy (XPS) analysis was conducted on a Thermo Scientific ESCALAB 250Xi system. Transmission electron microscopy (TEM) images were acquired using an FEI Tecnai F20 microscope. The actual doping concentrations of Yb3+ and Er3+ were determined by inductively coupled plasma atomic emission spectroscopy (ICP-AES) on a PerkinElmer Optima 8300. UCL spectra and decay curves were measured with an Edinburgh FLS980 spectrophotometer, employing a 980 nm diode laser as the excitation source. The absolute UCQY of the samples was measured with a custom UCL spectroscopy system at room-temperature upon 980 nm diode laser excitation at a power density of 100 W/cm2, and the UCL peaks from the Er3+ ions in the spectral range of 500-700 nm were integrated for the UCQY determination. Temperature-dependent UCL measurements were carried out on the FLS980 system coupled with a Linkam THMS600E temperature stage. All spectra were corrected for the instrumental response.
RESULTS AND DISCUSSION
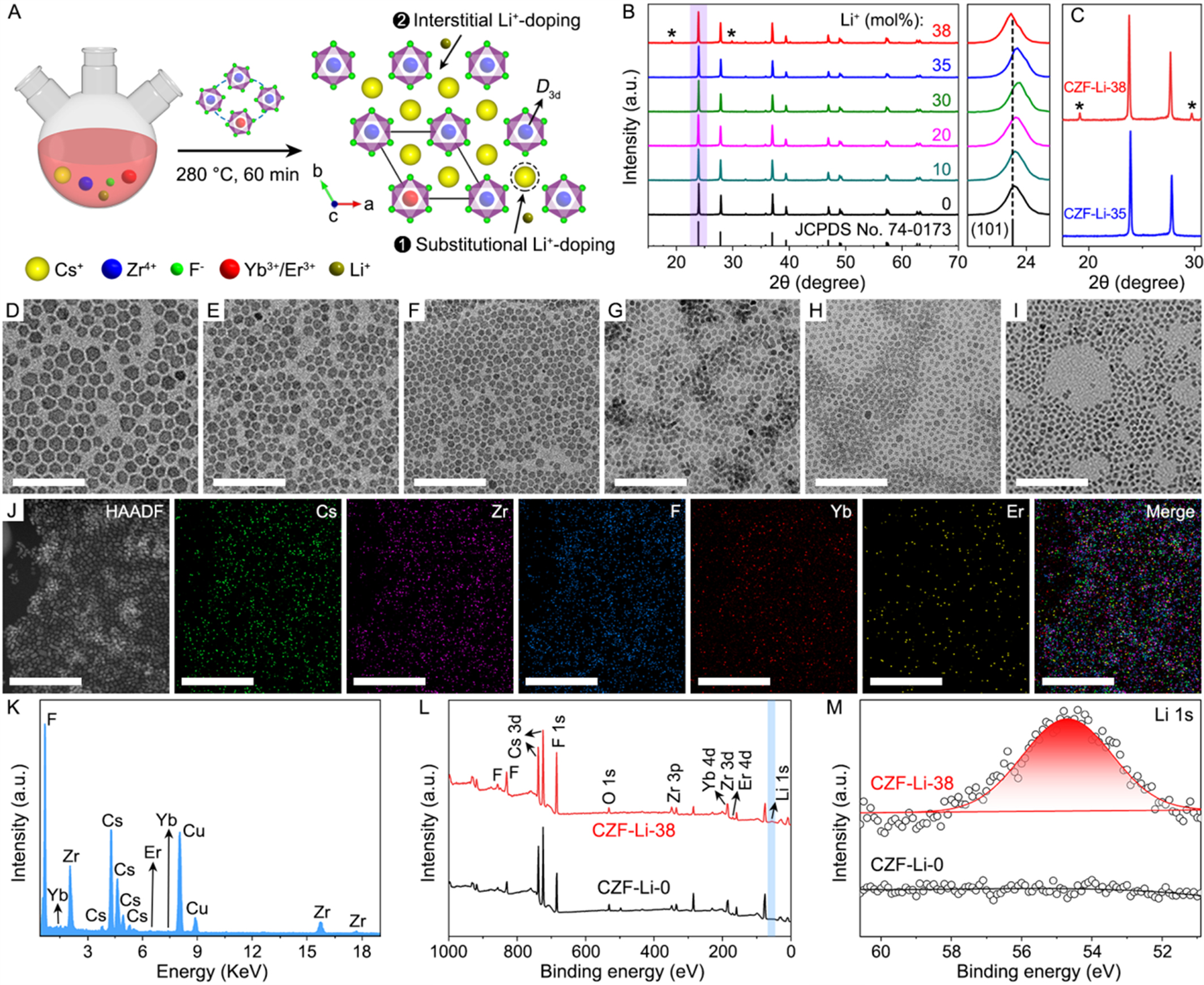
A series of Cs2ZrF6:20%Yb/2%Er/x%Li+ (x = 0, 10, 20, 30, 35 and 38) NCs were synthesized by a high-temperature co-precipitation method, using oleic acid as surfactant and 1-octadecene as solvent [Figure 1A]. In the structure of the Cs2ZrF6 host, Zr ions occupied an octahedral coordination environment, where each Zr ion was surrounded by six F ions with a site symmetry of D3d. The X-ray diffraction (XRD) patterns of 0-35 mol% Li+-doped samples primarily exhibited diffraction peaks of (101), (110), (021), (012), (211), (202), and (122) planes, matching well with the trigonal-phase Cs2ZrF6 crystal [Joint Committee on Powder Diffraction Standards (JCPDS) No. 74-0173] [Figure 1B]. However, raising the Li+ content to 38 mol% led to the identification of two noticeable impurity peaks at 19.21° and 29.85°, highlighted by asterisks, suggesting that a secondary phase has formed in conjunction with the main Cs2ZrF6 phase. To achieve a more precise analysis of this secondary phase, the zoomed-in XRD patterns of the samples with Li+ contents of 35 and
Figure 1. (A) Schematic illustration of the synthesis of Li+-doped Cs2ZrF6:Yb/Er NCs; (B) XRD patterns of Cs2ZrF6:Yb/Er NCs doped with different Li+ content, and the right panel displays the evolution of (101) diffraction peak; (C) Zoomed-in XRD patterns of the CZF-Li-35 and CZF-Li-38 NCs. TEM images of Cs2ZrF6:Yb/Er NCs doped with (D) 0, (E) 10, (F) 20, (G) 30, (H) 35, and (I) 38 mol% Li+, respectively; (J) HAADF-STEM image and corresponding elemental mapping images of Cs, Zr, F, Yb, and Er for the CZF-Li-38 NCs; (K) EDS spectrum of the CZF-Li-38 NCs. Comparison of XPS spectra for the CZF-Li-0 and CZF-Li-38 NCs: (L) full survey and (M) Li 1s. Scale bars in (D-J):
Given the significant size mismatch between Li+ (ionic radius: 0.68 Å) and Cs+ (ionic radius: 1.69 Å), Li+ dopants in Cs2ZrF6:Yb/Er NCs are likely to occupy two distinct sites: substitutional (replacing Cs+) and interstitial [Figure 1A]. This suggests a plausible transition from substitutional to interstitial site with increasing Li+ doping content. At Li+ concentrations below 30 mol%, substitutional doping may mainly occur, potentially leading to lattice contraction and thus the high-angle shift of XRD peaks. Conversely, when the Li+ content exceeds 35 mol%, Li+ ions are more likely to occupy interstitial sites, possibly resulting in lattice expansion and consequently the low-angle shift of XRD peaks. Due to lack of direct sensitivity to light elements like Li+ in XRD analysis, future studies using neutron diffraction and pair distribution function analysis could provide direct evidence for Li+ occupancy. Furthermore, the site symmetry of Yb3+/Er3+ in the Li+-doped Cs2ZrF6:Yb/Er NCs would significantly alter after the heterophase formation, comprising both high-symmetry D3d for the Cs2ZrF6 phase and low-symmetry S4 for the LiYbF4 phase [Figure 1A and Supplementary Figure 1]. For simplicity, Li+-doped Cs2ZrF6:Yb/Er NCs are hereafter referred to as CZF-Li-X, where X is the nominal doping concentration of Li+ (0, 10, 20, 30, 35, and 38 mol%).
Representative transmission electron microscopy (TEM) images revealed approximately hexagonal morphologies for the as-synthesized CZF-Li-X NCs, demonstrating a progressive decrease in average size with increasing Li+ content [Figure 1D-I]. Quantitative analysis determined the average sizes of 12.8 ± 1.7, 10.1 ± 1.2, 8.5 ± 1.3, 8.1 ± 0.8, 7.5 ± 1.0, and 6.1 ± 0.8 nm, respectively [Supplementary Figure 2]. Their corresponding high-resolution TEM images showed clear lattice fringes with an identical d-spacing of
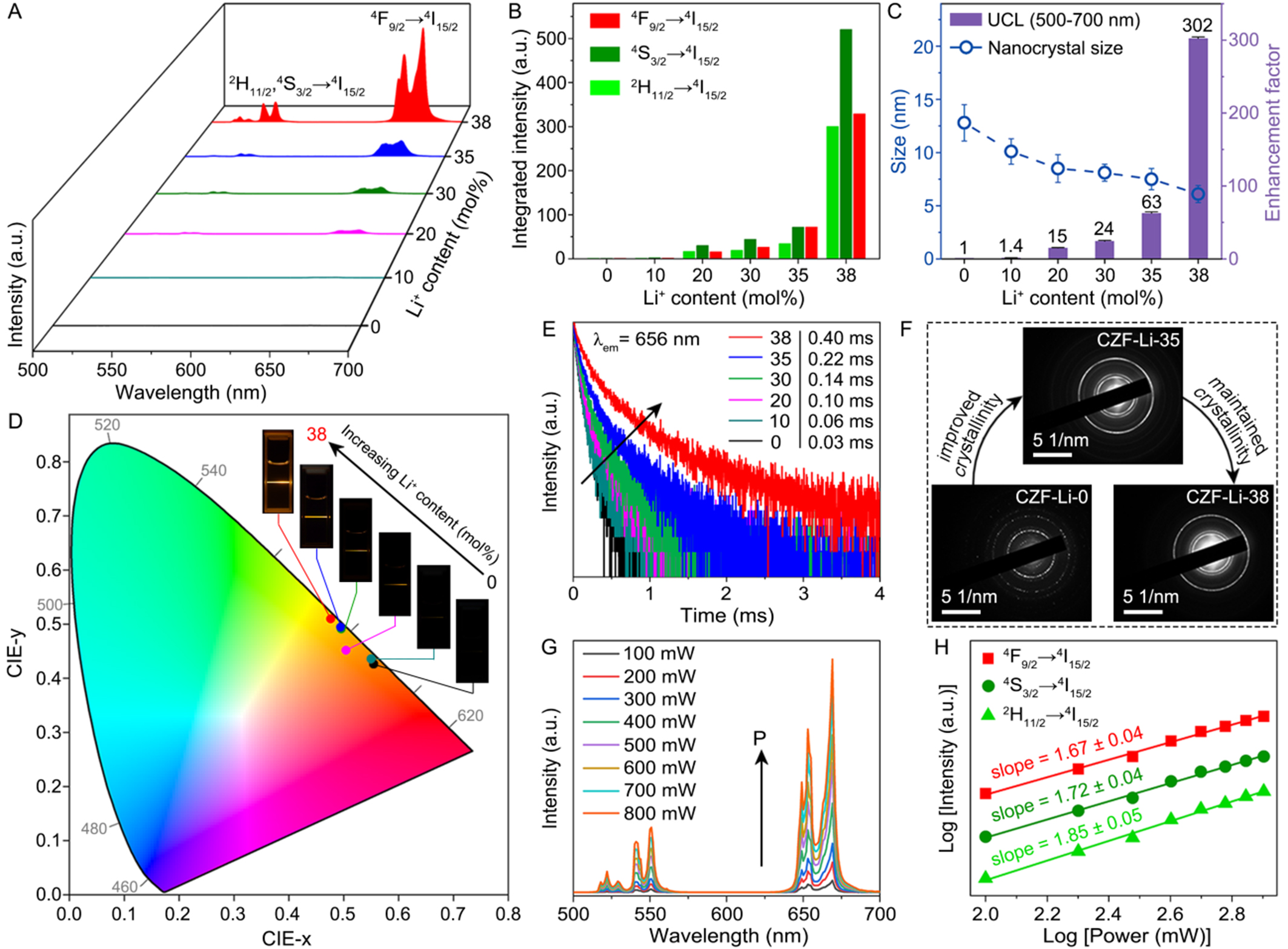
Figure 2A depicts the room-temperature UCL spectra of the CZF-Li-X NCs under 980 nm diode laser excitation at a power density of 10 W·cm-2. As can be seen, all the samples displayed a strong red emission at ~656 nm and two relatively weak green emissions at ~525 and ~545 nm, corresponding to the 4F9/2→4I15/2 and 2H11/2, 4S3/2→4I15/2 transitions of Er3+ ions, respectively. Although the nanocrystal size decreased progressively with increasing Li+ content, the green, red, and overall emission intensities of the CZF-Li-X NCs were markedly enhanced [Figure 2B and C], departing significantly from conventional enhancement strategies such as core-shell engineering. Specifically, the UCL intensities of pure-phase CZF-Li-X NCs (with 10 to
Figure 2. (A) Room-temperature UCL spectra of Cs2ZrF6:Yb/Er NCs doped with different Li+ content under 980 nm excitation; (B) Integrated green and red emission intensity of Cs2ZrF6:Yb/Er NCs doped with different Li+ content; (C) Evolution of the UCL enhancement factor for the CZF-Li-X NCs and their corresponding nanocrystal sizes as a function of Li+ doping content; (D) CIE (x,y) chromaticity diagram showing the color change with increasing Li+ content from 0 to 38 mol%. The inset shows their corresponding photographs obtained by a Canon digital camera; (E) Lifetimes of Er3+ monitored at 656 nm for the CZF-Li-X NCs under 980 nm excitation; (F) Comparison of SAED patterns for the CZF-Li-0, CZF-Li-35, and CZF-Li-38 NCs, illustrating different degrees of crystallinity improvement with increasing Li+ doping; (G) Power-dependent UCL spectra of the CZF-Li-38 NCs and (H) corresponding pump power dependence of green and red emission intensities. UCL: Upconversion luminescence; NCs: nanocrystals; CZF-Li-X NCs: Cs2ZrF6:Yb/Er NCs with and without Li+-doping; CIE: Commission Internationale de l’Éclairage; SAED: selected area electron diffraction.
Such substantial Li+-doping induced UCL enhancement in the CZF-Li-X NCs can be attributed to two synergistic factors: (i) improved crystallinity and (ii) reduced local symmetry around the Ln3+ ions. The improved crystallinity was directly evidenced by the selected area electron diffraction (SAED) patterns[46] [Figure 2F], while the reduced local symmetry was corroborated by the phase transition from high-symmetry D3d to low-symmetry S4 observed in the XRD patterns [Figure 1C and Supplementary Figure 1]. Despite their reduced nanoparticle sizes, the CZF-Li-35 and CZF-Li-38 NCs exhibited notably sharper diffraction rings compared to the Li+-free counterparts [Figure 2F]. Nevertheless, the exceptional UCL enhancement in the CZF-Li-38 NCs could not be explained solely by the crystallinity improvements, since SAED patterns showed minimal changes between 35 and 38 mol% Li+ doping. Instead, this enhancement arose primarily from the pronounced reduction of local symmetry around Ln3+ ions, induced by the phase transition from the high-symmetry D3d in pure-phase Cs2ZrF6 to the heterophase containing the low-symmetry S4 in LiYbF4. These results indicate that the dramatic UCL enhancement in the CZF-Li-38 NCs originated from the combined effects of crystallinity improvement and local symmetry reduction.
Complementary to the spectral analysis, the pump power dependence of the UCL intensity for the CZF-Li-38 NCs was investigated. As shown in Figure 2G, the emission intensity (I) increased progressively with pump power (P) over the range of 100-800 mW. The relationship follows the power law I ∝ Pn, where n represents the number of photons involved in each upconversion (UC) process. Linear fitting yielded slopes (n values) of 1.85, 1.72, and 1.67 for the 2H11/2→4I15/2, 4S3/2→4I15/2, and 4F9/2→4I15/2 transitions of Er3+, respectively [Figure 2H]. These n values, all close to 2, collectively demonstrated that the green and red emissions arose predominantly from two-photon UC processes. In addition, the photostability of the CZF-Li-38 NCs was assessed by monitoring the UCL intensity under continuous 980 nm laser irradiation over time [Supplementary Figure 7]. It was observed that the UCL intensity remained almost constant after 60 min of exposure at a power density of ~10 W/cm2, demonstrating the excellent photostability of the obtained NCs. This remarkable photostability implies that the CZF-Li-38 NCs can serve as excellent probes for long-term detection, particularly in temperature sensing.
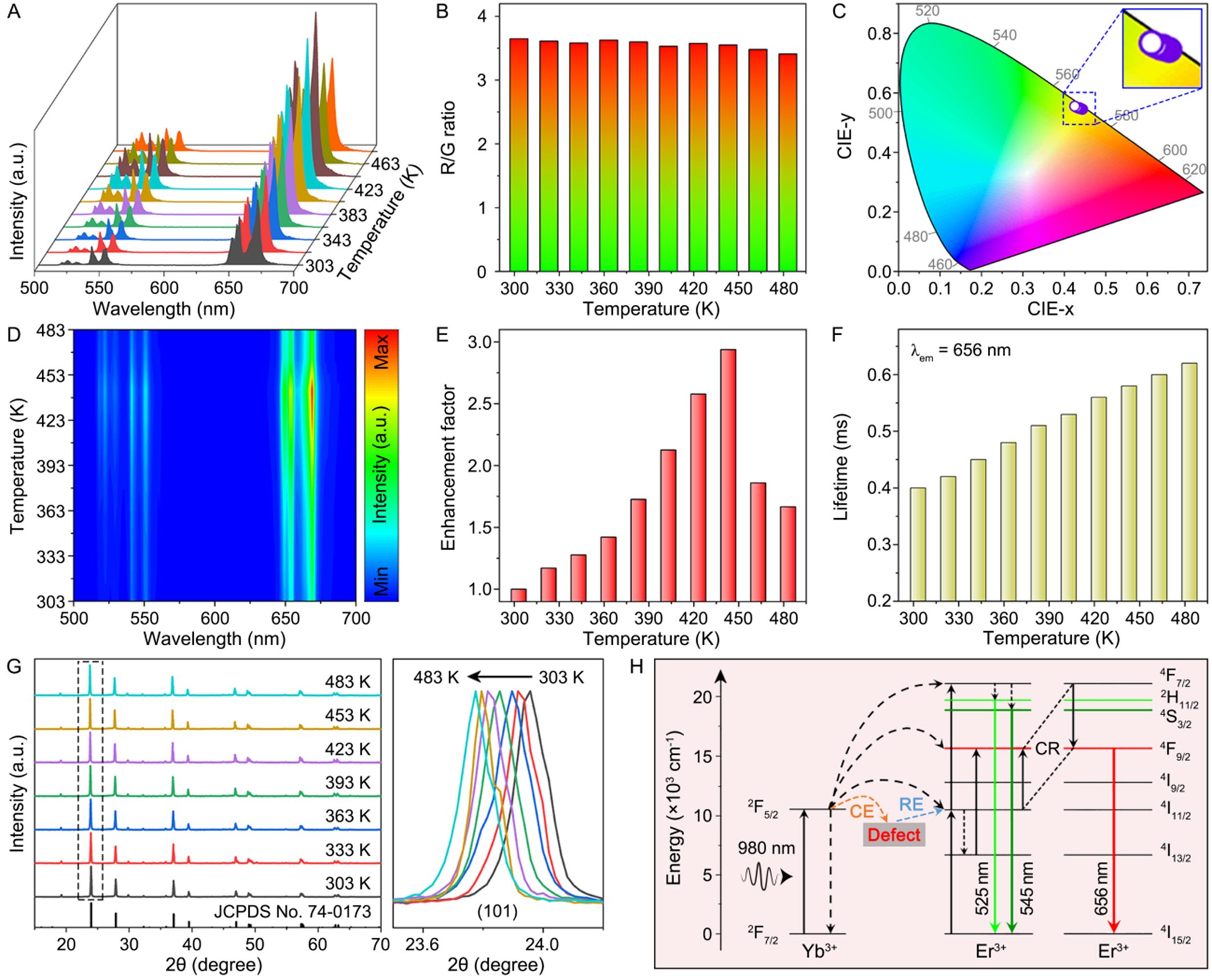
To shed more light on the luminescent behavior of the CZF-Li-38 NCs, temperature-dependent spectra were acquired from 303 to 483 K under 980 nm excitation with a power density of ~100 W·cm-2 [Figure 3A]. The results revealed that the UCL R/G ratios of Er3+ for these samples exhibited good stability with increasing temperature, consistently producing yellow emissions [Figure 3B and C]. To quantitatively assess the temperature-dependent emission color, the corresponding color purity was calculated using the following formula[47]:
Figure 3. (A) UCL spectra of the CZF-Li-38 NCs as a function of temperature in the range of 303-483 K under 980 nm excitation and their corresponding (B) UCL R/G ratios; (C) CIE (x,y) and corresponding zoomed-in chromaticity diagrams of the emission colors from the CZF-Li-38 NCs with an increase in temperature; (D) Contour map of thermal evolution spectra and (E) temperature dependence of the luminescent enhancement factor for the CZF-Li-38 NCs; (F) Lifetimes of Er3+ emission monitored at 656 nm under 980 nm excitation; (G) In situ temperature-dependent XRD patterns of the CZF-Li-38 NCs from 303 to 483 K, and the right panel shows the evolution of (101) diffraction peak; (H) Energy level diagram revealing the proposed energy transfer processes for the CZF-Li-38 NCs under 980 nm excitation. UCL: Upconversion luminescence; CZF-Li-X NCs: Cs2ZrF6:Yb/Er NCs with and without Li+-doping; NCs: nanocrystals; XRD: X-ray diffraction; CE: captured electrons; RE: released electrons; CR: cross relaxation; CIE: Commission Internationale de l’Éclairage.
where (X, Y) are the Commission Internationale de l’Éclairage (CIE) coordinates of the CZF-Li-38 NCs measured at different temperatures, (Xi, Yi) = (0.310, 0.316) is the fixed chromaticity coordinate of the white illuminant point, and (Xd, Yd) are the CIE coordinates of the dominant wavelengths. The calculated color purities for the CZF-Li-38 NCs remained highly consistent across the temperature range of 303-483 K [Supplementary Table 3], corresponding well to the observed color stability. More intriguingly, the CZF-Li-38 NCs exhibited anomalous thermal enhancement of UCL with increasing temperature, reaching maximum intensity at 443 K. This finding was further corroborated by the UCL spectral contour map, depicting the evolution of emission intensity as a function of temperature [Figure 3D]. As temperature increased from 303 K to 483 K, the UCL intensity of the CZF-Li-38 NCs progressively increased, achieving a maximum enhancement factor of 2.94 at 443 K [Figure 3E]. Concurrently, the 4F9/2→4I15/2 transition lifetime of Er3+ (monitored at 656 nm) gradually increased from 0.40 to 0.62 ms [Figure 3F]. Notably, the thermal enhancement of UCL in the CZF-Li-38 NCs was further verified under high-power 980 nm laser irradiation [~100 W·cm-2, Supplementary Figure 8]. The UCL intensity exhibited a gradual increase during the initial
In general, F- vacancies inevitably emerge due to the inequivalent substitution of Zr4+ by Yb3+/Er3+ in the Cs2ZrF6 lattice for maintaining the charge balance, which thereby act as defect states favoring the thermally enhanced UCL in the CZF-Li-38 NCs. To validate this mechanism, temperature-dependent structure evolution of the sample was first examined to exclude the possibility of negative lattice expansion, one conceivable explanation for thermal enhancement of UCL reported in the literature[48-50]. The in situ XRD patterns presented almost no change in the characteristic diffraction profiles except for a slight diffraction peak shift, such as a ~0.2° shift to small angle for the (101) plane as temperature increased from 303 to
In view of the significantly enhanced UCL from Li+-doping and anti-thermal quenching luminescence, the CZF-Li-38 NCs are expected to be promising candidates as high-performance nanothermometers for temperature sensing. Figure 4A reveals an identical variation trend of green (525 and 545 nm) and red
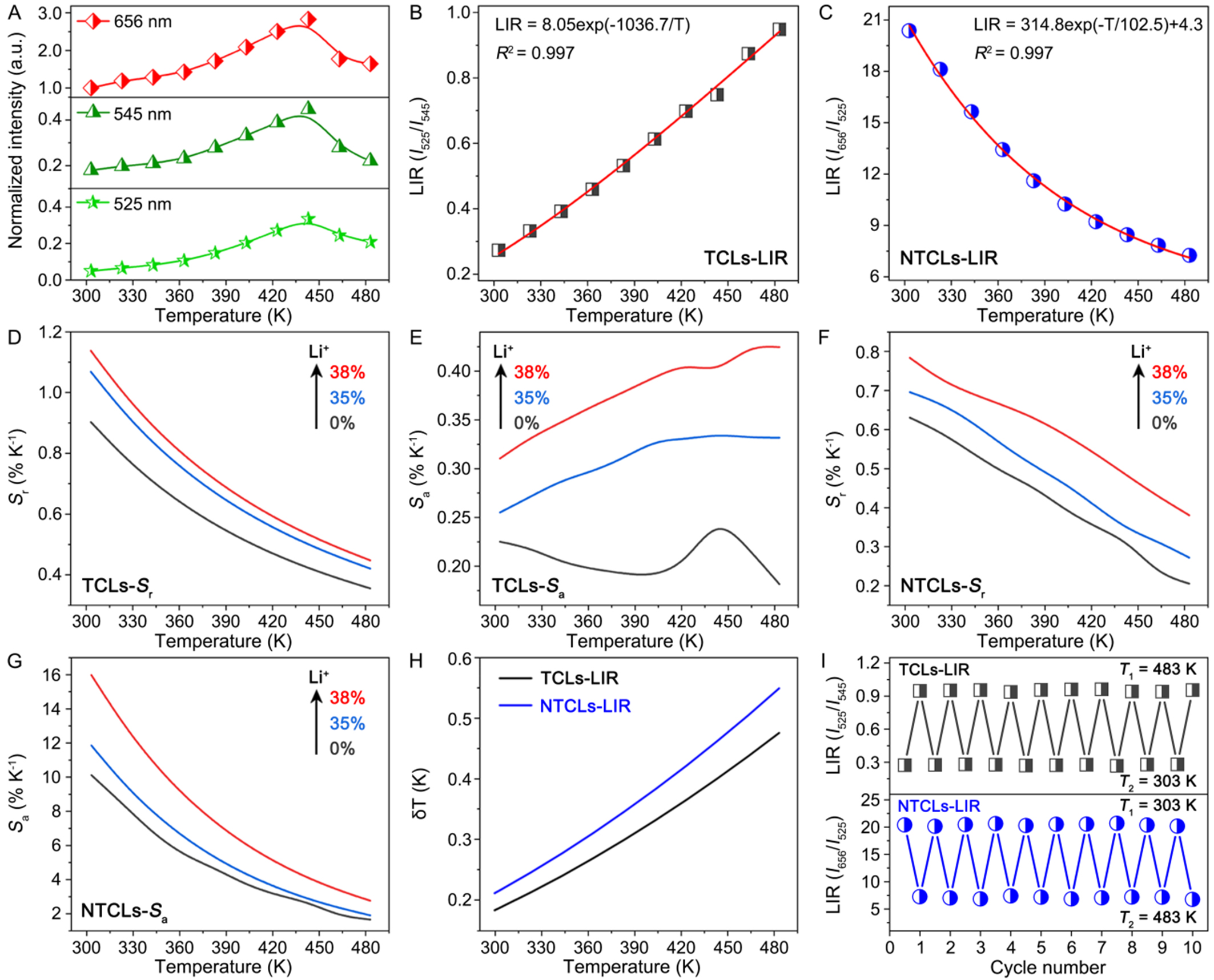
Figure 4. (A) Normalized intensities of UC emissions centered at 525, 545, and 656 nm for the CZF-Li-38 NCs as a function of temperature. Fitting curves of temperature-dependent (B) LIR(I525/I545) and (C) LIR(I656/I525). Comparison of (D) relative (Sr) and (E) absolute (Sa) sensitivities of the CZF-Li-0, CZF-Li-35, and CZF-Li-38 NCs based on the TCLs-LIR method. Comparison of (F) Sr and (G) Sa of the CZF-Li-0, CZF-Li-35, and CZF-Li-38 NCs based on the NTCLs-LIR method; (H) Temperature uncertainty for the CZF-Li-38 NCs at different temperatures based on the TCLs- and NTCLs-LIR methods; (I) Thermal cycling measurements for the LIR(I525/I545) and LIR(I656/I525) of the CZF-Li-38 NCs between 303 and 483 K. UC: Upconversion; CZF-Li-X NCs: Cs2ZrF6:Yb/Er NCs with and without Li+-doping; NCs: nanocrystals; TCLs: thermally coupled levels; LIR: luminescence intensity ratio; NTCLs: non-thermally coupled levels.
As shown in Figure 4D-G, the maximum Sr and Sa values achieved in the CZF-Li-38 NCs were up to 1.14% and 0.42% K-1 using the TCLs-LIR method, and 0.79% and 16% K-1 using the NTCLs-LIR method, respectively. These maximum sensitivities were significantly higher than those observed for the pure-phase CZF-Li-0 and CZF-Li-35 NCs, demonstrating the effectiveness of our phase engineering strategy in producing ultrasmall, bright Ln3+-doped NCs as high-performance luminescent nanothermometers.
The temperature uncertainty (δT) is a key parameter to assess the performance of an optical thermometer, which contains not only the relative sensitivity but also the error on the luminescence intensity ratio (δLIR). δT is determined as follows[52]:
Where δLIR/LIR is the uncertainty in the calculation of LIR. Temperature uncertainty for the CZF-Li-38 NCs at different temperatures is presented in Figure 4H. The minimum values of δT were determined to be 0.18 and 0.21 K at 303 K using the TCLs- and NTCLs-LIR methods, respectively. In addition to thermal sensitivities and temperature uncertainty, reliable LIR repeatability across temperature cycles serves as another vital indicator for evaluating luminescent nanothermometers. Over ten consecutive thermal cycles between 303 and 483 K, both LIR of I525/I545 and I656/I525 demonstrated exceptional stability with signal variations below 0.5% [Figure 4I]. This remarkable repeatability confirmed the structural robustness of the CZF-Li-38 NCs as luminescent nanothermometers. Collectively, these findings underscore unique advantages of the Cs2ZrF6 host for precision temperature sensing, enabling sub-10 nm size with anti-thermal quenching that cannot be achieved in the conventional fluorides like NaYF4[53].
CONCLUSIONS
In summary, a phase engineering strategy via Li+ doping was developed to synthesize ultrasmall and high-brightness Cs2ZrF6:Yb/Er NCs. Within the Li+ doping range of 0-35 mol%, the NCs maintained the pure Cs2ZrF6 phase, exhibiting progressively enhanced UCL intensity owing to improved crystallinity. At
DECLARATIONS
Authors’ contributions
Conceptualization: Fu, H.; Liu, M.; Wu, A.; Su, L.
Investigation and methodology: Fu, H.; Liu, M.; Qian, Y.; Zhang, Y.; Lu, J.; Xiong, Q.; Jiang, D.
Data curation and writing - original draft: Fu, H.; Liu, M.
Writing - review and editing and supervision: Wu, A.; Su, L.; Liu, M.; Fu, H.
Funding acquisition: Kou, H.; Fu, H.; Liu, M.
Validation and software: Qian, Y.; Zhang, Y.; Lu, J.; Xiong, Q.; Jiang, D.; Kou, H.
Availability of data and materials
The detailed materials and methods in the experiment are presented in the Supplementary Materials. Other raw data that support the findings are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This research was financially supported by the National Natural Science Foundation of China (Nos. 62275255 and 22101180), the National Key Research and Development Project (No. 2016YFA0400904), and the Scientific Research Foundation for the Talented Young Scholars of Shanghai Institute of Technology (Nos. YJ2021-12 and ZQ2020-7).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Wan, S.; Xia, X.; Gao, Y.; et al. Curvature-guided depletion stabilizes Kagome superlattices of nanocrystals. Science 2025, 387, 978-84.
2. Ming, J.; Chen, Y.; Miao, H.; et al. High-brightness transition metal-sensitized lanthanide near-infrared luminescent nanoparticles. Nat. Photon. 2024, 18, 1254-62.
3. Szalkowski, M.; Kotulska, A.; Dudek, M.; et al. Advances in the photon avalanche luminescence of inorganic lanthanide-doped nanomaterials. Chem. Soc. Rev. 2025, 54, 983-1026.
4. Li, M.; You, W.; Liu, S.; et al. Unveiling the dynamics of frenkel defects in fluoride materials for X-ray-induced persistent luminescence and advanced imaging applications. Laser. Photon. Rev. 2025, 19, 2500092.
5. Ou, X.; Qin, X.; Huang, B.; et al. High-resolution X-ray luminescence extension imaging. Nature 2021, 590, 410-5.
6. Wang, X.; Wu, W.; Yun, B.; et al. An emerging toolkit of Ho3+ sensitized lanthanide nanocrystals with NIR-II excitation and emission for in vivo bioimaging. J. Am. Chem. Soc. 2025, 147, 2182-92.
7. Dong, H.; Guan, L.; Qiao, S.; et al. Parallel photon avalanche nanoparticles for tunable emission and multicolour sub-diffraction microscopy. Nat. Photon. 2025, 19, 692-700.
8. Wang, E.; Yuan, J.; Niu, L.; et al. Cross relaxation controlled ultralow temperature detection in high-level-doping upconversion thermometer. Laser. Photon. Rev. 2025, 19, 2402222.
9. Liu, C.; Zhang, X.; Chen, X.; Liang, L. Emerging advances in lanthanide photon avalanche nanophotonics. Nano. Lett. 2024, 24, 15489-500.
10. Xu, W.; Zheng, Y.; Liu, X.; et al. Manipulation of coupled X-ray-excited persistent luminescence and upconversion in Er3+ doped fluoride nanoparticles for multifaceted applications. Laser. Photon. Rev. 2024, 18, 2400147.
11. Brites, C. D. S.; Marin, R.; Suta, M.; et al. Spotlight on luminescence thermometry: basics, challenges, and cutting-edge applications. Adv. Mater. 2023, 35, e2302749.
12. Li, K.; Li, D.; Jia, M.; et al. Li-based nanoprobes with boosted photoluminescence for temperature visualization in NIR imaging-guided drug release. Nano. Lett. 2025, 25, 776-85.
13. Wei, H.; Cai, Z.; Huang, H.; Wei, G.; Zhou, B. Ultra-sensitive low-temperature upconversion via interfacial energy transfer toward visual cryogenic nanothermometry. Adv. Funct. Mater. 2025, 35, e10764.
14. Liu, M.; Pan, J.; Shi, X.; Zou, B.; Pan, D. Room-temperature and ultrafast synthesis of sub-4 nm Yb3+/Nd3+ ions-doped CaMoO4 nanocrystals with near-infrared photoluminescence quantum yield of 40%. Laser. Photon. Rev. 2025, 19, 2401259.
15. Gálico, D. A.; Rodrigues, E. M.; Halimi, I.; et al. Confining single Er3+ ions in sub-3 nm NaYF4 nanoparticles to induce slow relaxation of the magnetisation. Nat. Commun. 2024, 15, 3498.
16. Zou, H.; Zhu, W.; Zhao, J.; Zhou, S.; Xu, S.; Lei, L. Sub-10 nm lanthanide-doped Lu6O5F8 nanoscintillators for real-time high-resolution dynamic 3D X-ray imaging. Adv. Funct. Mater. 2024, 34, 2409156.
17. Hu, C.; Li, X.; Xie, W.; et al. Thermal enhancement of upconversion in sub-10 nm Yb3+/Er3+/Na+ tridoped Cs2ZrF6 nanocrystals for ratiometric temperature sensing. Adv. Optical. Mater. 2024, 12, 2400073.
18. Zhao, F.; Ling, H.; Zhang, W.; Zhang, Y.; Liu, Q. Dye-to-Er3+ direct energy transfer for enhancing up- and down-conversion luminescence in sub-10 nm NaErF4. Nano. Lett. 2024, 24, 14838-46.
19. Huang, F.; Labrador-páez, L.; Ågren, H.; et al. Transient energy trapping as a size-conserving surface passivation strategy for producing bright ultrasmall upconversion nanoprobes. Nano. Energy. 2023, 105, 108015.
20. Zhang, Y.; Yu, Z.; Li, J.; et al. Ultrasmall-superbright neodymium-upconversion nanoparticles via energy migration manipulation and lattice modification: 808 nm-activated drug release. ACS. Nano. 2017, 11, 2846-57.
21. Huang, L.; Xue, M.; Wei, Y.; Wang, J. Improving persistent luminescence by defect engineering. Coord. Chem. Rev. 2026, 546, 217088.
22. Yadav, J.; Pushpendra; Samal, S. K.; Achary, S. N.; Naidu, B. S. Sr2BiF7: a new bismuth-based host material for lanthanide ions doping: synthesis, downshifting, and upconversion luminescence properties for multimode anticounterfeiting. ACS. Appl. Mater. Interfaces. 2024, 16, 51028-36.
23. Li, G.; Jiang, S.; Liu, A.; et al. Proof of crystal-field-perturbation-enhanced luminescence of lanthanide-doped nanocrystals through interstitial H+ doping. Nat. Commun. 2023, 14, 5870.
24. Zheng, B.; Fan, J.; Chen, B.; et al. Rare-earth doping in nanostructured inorganic materials. Chem. Rev. 2022, 122, 5519-603.
25. Hu, J.; Bian, X.; Wang, R.; et al. Giant enhancement in upconversion luminescence of β-Ba2ScAlO5:Yb3+/Er3+ phosphor by the intermediate band through Ca2+ doping. Chem. Mater. 2022, 34, 3089-98.
26. Pandey, P.; Tripathi, S.; Singh, M. N.; Sharma, R. K.; Giri, S. Behavior of microstrain in Nd3+-sensitized near-infrared upconverting core-shell nanocrystals for defect-induced tailoring of luminescence intensity. Nano. Lett. 2024, 24, 6320-9.
27. Li, H.; Sun, Y.; Zhang, J.; et al. Enhancement of ultraviolet upconversion photoluminescence of Y7O6F9:Pr,Gd induced by Li+ codoping. J. Rare. Earth. 2025, 43, 684-90.
28. Som, S.; Yang, C.; Lu, C.; Das, S. Synthesis of Li+-ion activated NaYF4: Er3+/Yb3+ phosphors via a modified solid-state process for latent fingerprint detection. Ceram. Int. 2019, 45, 5703-9.
29. Debasu, M. L.; Riedl, J. C.; Rocha, J.; et al. The role of Li+ in the upconversion emission enhancement of (YYbEr)2O3 nanoparticles. Nanoscale 2018, 10, 15799-808.
30. Wang, Y.; Wei, T.; Cheng, X.; et al. Insights into Li+-induced morphology evolution and upconversion luminescence enhancement of KSc2F7:Yb/Er nanocrystals. J. Mater. Chem. C. 2017, 5, 3503-8.
31. Dou, Q.; Zhang, Y. Tuning of the structure and emission spectra of upconversion nanocrystals by alkali ion doping. Langmuir 2011, 27, 13236-41.
32. Yin, W.; Zhao, L.; Zhou, L.; et al. Enhanced red emission from GdF3:Yb3+,Er3+ upconversion nanocrystals by Li+ doping and their application for bioimaging. Chem. Eur. J. 2012, 18, 9239-45.
33. Cheng, Q.; Sui, J.; Cai, W. Enhanced upconversion emission in Yb3+ and Er3+ codoped NaGdF4 nanocrystals by introducing Li+ ions. Nanoscale 2012, 4, 779-84.
34. Mao, C.; Yang, X.; Zhao, L. Simultaneous morphology control and upconversion fluorescence enhancement of NaYF4:Yb,Er crystals through alkali ions doping. Chem. Eng. J. 2013, 229, 429-35.
35. Zhao, C.; Kong, X.; Liu, X.; et al. Li+ ion doping: an approach for improving the crystallinity and upconversion emissions of NaYF4:Yb3+, Tm3+ nanoparticles. Nanoscale 2013, 5, 8084-9.
36. Hu M.; Ma D.; Cheng Y.; et al. Synergistically enhanced upconversion luminescence in Li+-doped core-shell-structured ultrasmall nanoprobes for dual-mode deep tissue fluorescence/CT imaging. J. Mater. Chem. B. 2017, 5, 2662-70.
37. Monika; Yadav, R. S.; Bahadur, A.; Rai, S. B. Near-infrared light excited highly pure green upconversion photoluminescence and intrinsic optical bistability sensing in a Ho3+/Yb3+ co-doped ZnGa2O4 phosphor through Li+ doping. J. Phys. Chem. C. 2020, 124, 10117-28.
38. Mondal, M.; Kumar Rai, V.; Srivastava, C.; Sarkar, S.; Akash, R. Enhanced frequency upconversion in Ho3+/Yb3+/Li+:YMoO4 nanophosphors for photonic and security ink applications. J. Appl. Phys. 2016, 120, 233101.
39. Mukhopadhyay, L.; Rai, V. K.; Bokolia, R.; Sreenivas, K. 980 nm excited Er3+/Yb3+/Li+/Ba2+:NaZnPO4 upconverting phosphors in optical thermometry. J. Lumin. 2017, 187, 368-77.
40. Yu, X.; Duan, L.; Ni, L.; Wang, Z. Fabrication and luminescence behavior of phosphate glass ceramics co-doped with Er3+ and Yb3+. Opt. Commun. 2012, 285, 3805-8.
41. Mahalingam, V.; Naccache, R.; Vetrone, F.; Capobianco, J. A. Enhancing upconverted white light in Tm3+/Yb3+/Ho3+-doped GdVO4 nanocrystals via incorporation of Li+ ions. Opt. Express. 2012, 20, 111-9.
42. Sun, Q.; Chen, X.; Liu, Z.; Wang, F.; Jiang, Z.; Wang, C. Enhancement of the upconversion luminescence intensity in Er3+ doped BaTiO3 nanocrystals by codoping with Li+ ions. J. Alloys. Compd. 2011, 509, 5336-40.
43. Kim, D.; Ryu, J. H.; Ho Chung, J.; Shim, K. B.; Cho, S. Green upconversion luminescence of Li+∕Er3+∕Yb3+ tridoped CaWO4 and effects of Er3+ concentration. J. Electrochem. Soc. 2011, 158, J345.
44. Zhang, Z.; Lu, S.; Huang, G.; et al. Carbon-coated Li4Ti5O12 optimized by fluorine regulation strategy for high-rate lithium-ion batteries with mixed diffusion and capacitive effects. Carbon 2024, 221, 118885.
45. Boyer, J. C.; van Veggel, F. C. Absolute quantum yield measurements of colloidal NaYF4: Er3+, Yb3+ upconverting nanoparticles. Nanoscale 2010, 2, 1417-9.
46. Zheng, W.; Zhou, S.; Chen, Z.; et al. Sub-10 nm lanthanide-doped CaF2 nanoprobes for time-resolved luminescent biodetection. Angew. Chem. 2013, 125, 6803-8.
47. Shin, S.; Lim, J. P.; Hong, Y. K.; et al. Multicolor fine-tunable upconversion luminescence from a single nanoparticle for full-color displays with a wide color gamut. Adv. Funct. Mater. 2025, 35, 2415687.
48. Chen, B.; Wang, F. Emerging frontiers of upconversion nanoparticles. Trends. Chem. 2020, 2, 427-39.
49. Fu, B.; Yan, H.; Li, R.; et al. Heterovalent Zr4+/Nb5+-cosubstituted negative thermal expansion luminescent materials with anti-thermal quenching luminescence. Laser. Photon. Rev. 2024, 18, 2400739.
50. Wang, Y.; Rui, J.; Song, H.; et al. Antithermal quenching upconversion luminescence via suppressed multiphonon relaxation in positive/negative thermal expansion core/shell NaYF4:Yb/Ho@ScF3 nanoparticles. J. Am. Chem. Soc. 2024, 146, 6530-5.
51. Fu, H.; Xie, W.; Qian, Y.; et al. Ultrafast room-temperature synthesis of Yb3+/Er3+ codoped K3ZrF7 nanocrystals for thermal enhancement of upconversion. Laser. Photon. Rev. 2024, 19, 2401522.
52. Brites, C. D. S.; Balabhadra, S.; Carlos, L. D. Lanthanide-based thermometers: at the cutting-edge of luminescence thermometry. Adv. Optical. Mater. 2018, 7, 1801239.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].