Metal-organic frameworks for antibacterial therapy: a mini-review on microenvironment-responsive strategies and recent advances
Abstract
Non-antibiotic antibacterial agents can significantly reduce the risk of bacterial drug resistance, but they face limitations such as poor tissue penetration, insufficient in vivo stability, and significant side effects. Metal-organic frameworks (MOFs), as a novel class of porous functional materials, exhibit broad application prospects in the antibacterial field due to their highly tunable structures, large specific surface areas, and precisely controllable pore systems. This review introduces the antibacterial mechanisms of MOFs, explores their responsive antibacterial strategies within the bacterial infection microenvironment, and summarizes their applications in biofilm eradication, immuno-synergistic antibacterial therapy, and implant surface modification. Finally, it discusses the biocompatibility and biosafety of MOFs, addresses the existing deficiencies in this field, and outlines future directions.
Keywords
INTRODUCTION
Bacterial infection represents a major global public health challenge, posing a severe threat to human health[1,2]. Broad-spectrum antibiotics are widely used to treat such infections; however, their overuse can induce bacterial drug resistance, reducing antibacterial efficacy and increasing the prevalence of drug-resistant infections, which imposes a heavy economic burden on global healthcare systems[3-6]. Therefore, there is an urgent need to develop safe and effective novel antibacterial strategies.
In recent years, non-antibiotic antibacterial agents, such as metal-based nanoparticles (NPs), have garnered increasing attention from researchers. While they lower the risk of bacterial drug resistance and enhance antibacterial efficiency, they suffer from limitations such as poor tissue penetration and insufficient in vivo stability[7-12]. More critically, the microenvironment of in vivo bacterial infection is complex and dynamic, yet current antibacterial agents generally lack microenvironment-responsive capabilities, resulting in poor therapeutic controllability and low treatment efficacy[13]. Furthermore, the formation of bacterial biofilms further complicates treatment, as their dense extracellular polymeric substances significantly reduce the penetration efficiency of antibacterial drugs, providing a protective barrier for the bacteria[14,15].
Metal-organic frameworks (MOFs) are a novel class of porous functional materials formed by the self-assembly of metal centers and organic linkers through coordination bonds. They feature highly tunable topological structures, large specific surface areas, and precisely controllable pore systems, offering unique advantages in antibacterial applications[16]. Their intrinsic antibacterial properties, cargo-loading capabilities, and responsiveness to infection microenvironments facilitate effective bacterial elimination, thereby offering a multidimensional strategy to overcome drug-resistant infections and enhance therapeutic efficacy[17-20].
Given the advantages and promising prospects of MOFs in antibacterial therapy, this review introduces their antibacterial mechanisms and analyzes their responsive therapeutic strategies in physical and biochemical microenvironments. It also summarizes recent innovative antibacterial applications of MOF composite materials and discusses their biocompatibility and biosafety. While several reviews have summarized the landscape of antibacterial MOFs, they often focus on drug delivery. This review provides a more clinically oriented perspective on in vivo infection microenvironments, highlighting recent progress toward practical applications.
ANTIBACTERIAL MECHANISMS OF MOFS
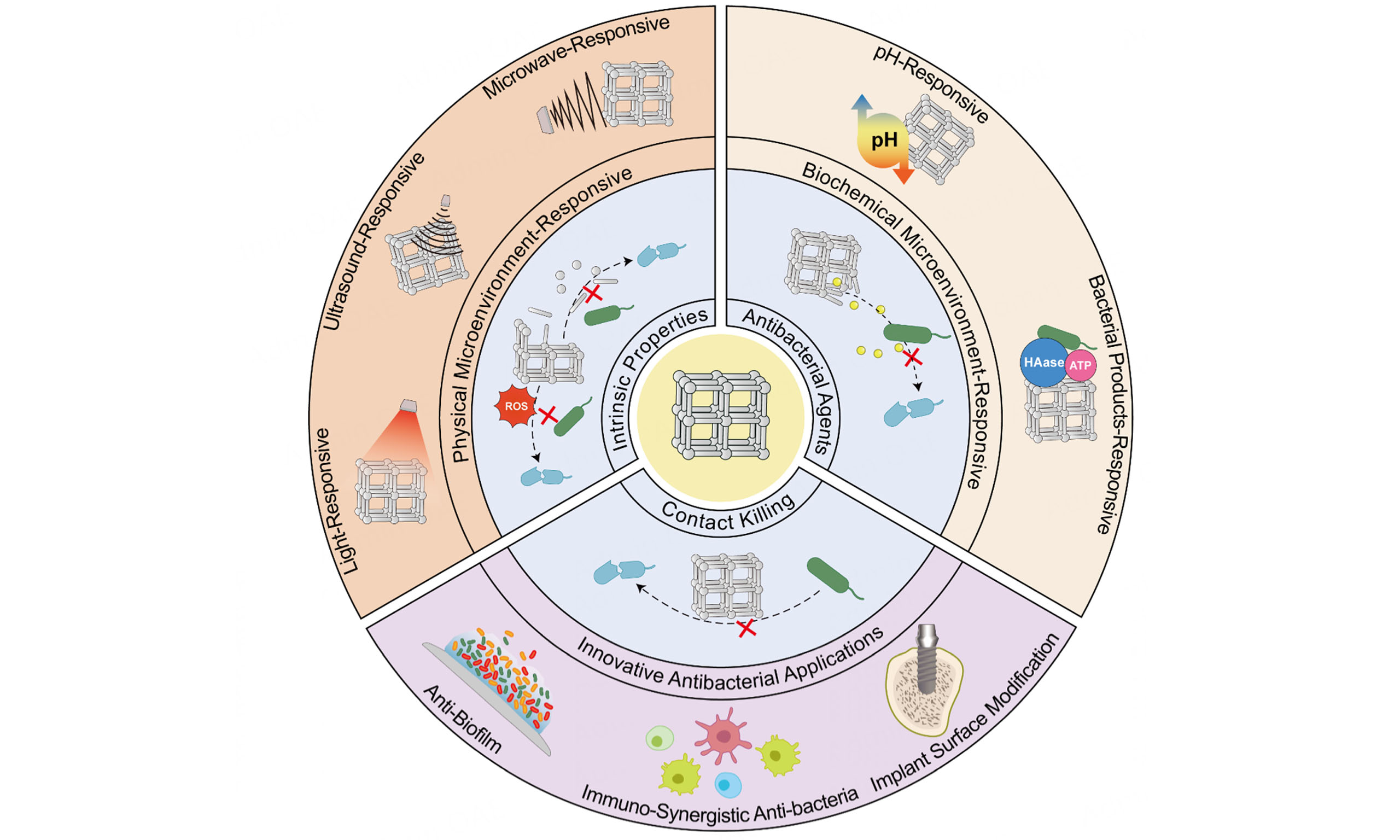
The antibacterial efficacy of MOFs is mediated through three mechanisms: intrinsic antibacterial properties, loaded antibacterial agents, and contact killing.
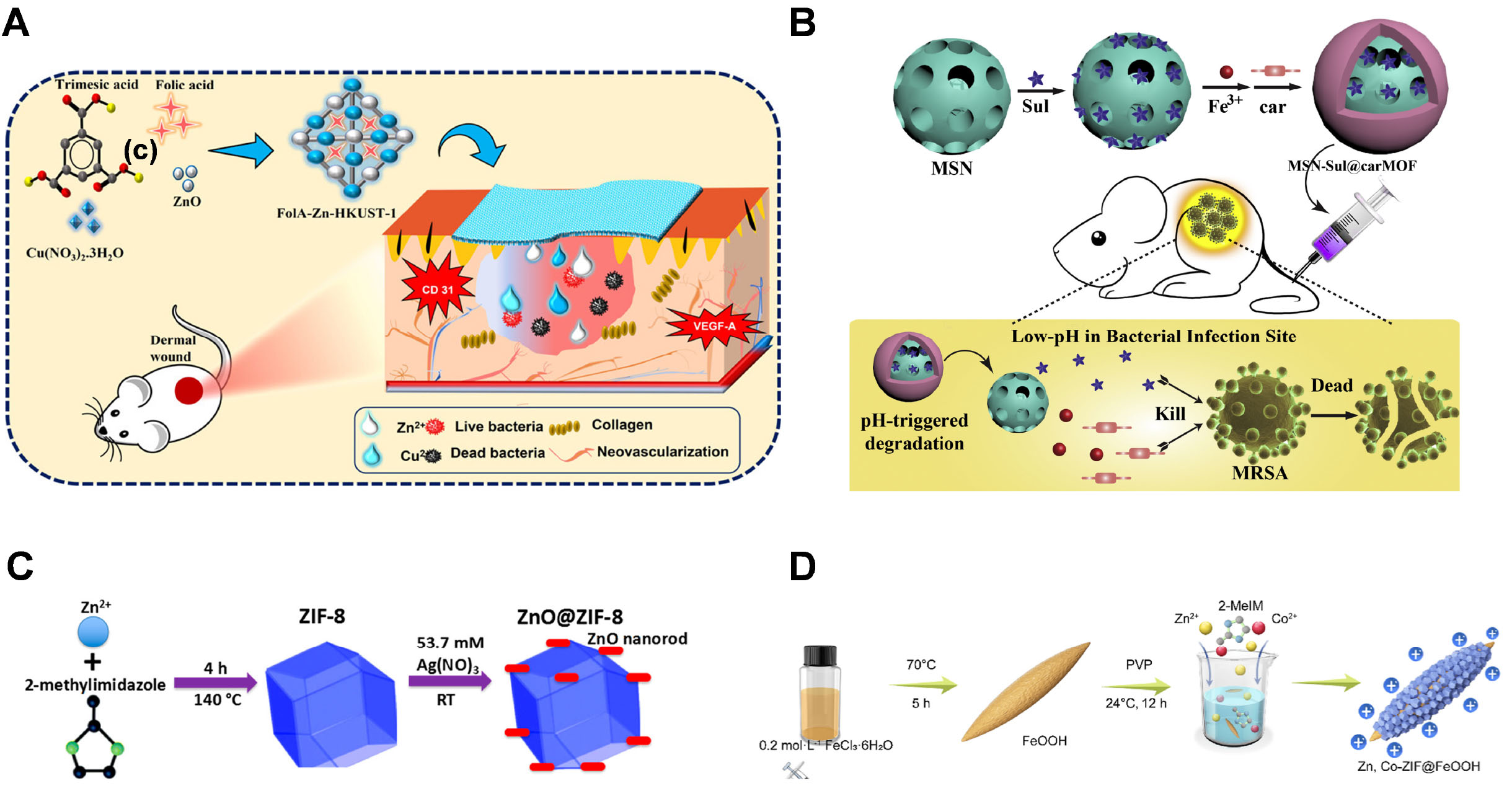
The antibacterial activity of MOFs frequently originates from their intrinsic physicochemical properties. First, MOFs can serve as sophisticated reservoirs for the controlled release of antibacterial metal ions (e.g., Zn2+, Cu2+, Ag+) or organic ligands. Amer et al. synthesized a bimetallic MOF, FolA-Zn-HKUST, that releases both Zn2+ and Cu2+, demonstrating significant antibacterial efficacy[21]. [Figure 1A] Duan et al. constructed carMOF using the antibiotic carbenicillin (car) as the organic ligand[22]. The carMOF was then coated onto mesoporous silica nanoparticles (MSNs) loaded with a β-lactamase inhibitor, creating a co-delivery system (MSN@carMOF) that synergistically enhanced the antibacterial effect against Methicillin-resistant Staphylococcus aureus (MRSA)[22] [Figure 1B]. Moreover, certain MOFs display photodynamic antibacterial activity arising from their unique frameworks. Chen et al. synthesized PCN-224(Zr/Ti), which generates reactive oxygen species (ROS) under visible light, thereby achieving potent antibacterial effects[23].
Figure 1. (A) Schematic illustration of the synthesis and structure of FolA-Zn-HKUST. Reproduced with permission[21]. Copyright 2023, American Chemical Society. (B) Schematic illustration of the synthesis and antibacterial strategy of MSN@carMOF. Reproduced with permission[22]. Copyright 2021, Elsevier. (C) Schematic illustration of the synthesis and structure of ZnO@ZIF-8. Reproduced with permission[25]. Copyright 2018, American Chemical Society. (D) Schematic illustration of fabrication process of Zn, Co-ZIF@FeOOH. Reproduced with permission[26]. Copyright 2024, American Chemical Society.
Second, leveraging their large specific surface area and regular pore structure, MOFs possess high guest-loading capacity and can serve as delivery vehicles for antibacterial agents such as antibiotics and metal-based NPs. Acharya et al. synthesized BIO-MOF-100, which exhibits efficient co-loading capability for three anti-tuberculosis drugs, overcoming the limitations of traditional drug delivery systems in co-loading hydrophilic/hydrophobic drugs[24]. Redfern et al. fabricated ZnO@ZIF-8 by embedding zinc oxide (ZnO) nanorods into Zeolitic imidazolate framework-8 (ZIF-8)[25]. ZnO@ZIF-8 demonstrated potent activity against uropathogens and can be incorporated into silicone to fabricate antibacterial catheters[25] [Figure 1C].
Third, MOFs can exert a bactericidal effect through "contact killing". This mechanism can be categorized into two primary modes of bacterial membrane disruption: direct physical damage and physicochemical interference. Yi et al. developed Zn, Co-ZIF@FeOOH. This material's sharp nanostructures can physically pierce bacterial cell membranes, while the Zn, Co-ZIF NPs on the surface of FeOOH can release antibacterial metal ions (Zn2+ and Co2+) and induce charge perturbation in the membrane, further enhancing the antibacterial effect[26] [Figure 1D].
MICROENVIRONMENT-RESPONSIVE ANTIBACTERIAL STRATEGIES OF MOFS
The microenvironment of bacterial infection is a dynamic system including diverse physical and biochemical factors, regulated by endogenous biomediators and external conditions[13,27]. Bacterial activity can markedly alter the local microenvironment. A neutral milieu may shift to weak acidity due to accumulated metabolic by-products[28]. Moreover, Bacteria also secrete enzymes such as lipases and hyaluronidase (HAase), along with energy sources such as adenosine triphosphate (ATP)[13,29,30]. These microenvironmental changes can cleave specific chemical bonds or functional groups, offering a basis for designing stimulus-responsive systems[31].
This chapter divides the microenvironment into the physical microenvironment (light, ultrasound, and microwave) and the biochemical microenvironment (pH and bacterial products), respectively, elucidating the responsive therapeutic strategies of MOFs and analyzing how they improve antibacterial efficiency and biocompatibility.
Physical microenvironment-responsive MOFs
Light-responsive MOFs
Light-responsive MOFs based on photodynamic therapy:
Photodynamic Therapy (PDT) involves irradiating a photosensitizer with a specific wavelength of light to generate ROS, which have antibacterial capabilities[32,33]. ROS are a class of highly chemically reactive oxygen-containing species, primarily including superoxide anions (O2•-), hydrogen peroxide (H2O2), hydroxyl radicals (•OH), and singlet oxygen (1O2), which have significant oxidative damaging effects on bacteria and other microorganisms[34]. The essential elements for achieving PDT are the photosensitizer, light of a specific wavelength, and an oxygen substrate. Photosensitizers play a core role in PDT; however, most of them have poor water solubility and stability, and are prone to aggregation, self-quenching, and degradation in physiological media, which greatly limits the efficiency of PDT[11,12,35]. Therefore, to achieve efficient delivery and protection of photosensitizers, researchers have constructed PDT-based light-responsive MOFs.
Porphyrins and their derivatives are among the most widely used photosensitizers[19,33]. PCN-224 is a widely studied porphyrin MOF, composed of zirconium (Zr) and tetrakis (4-carboxyphenyl) porphyrin (TCPP). Research has found that PCN-224 not only possesses excellent PDT performance but also exhibits good loading capacity for vancomycin (Van). Under visible-light irradiation, Van/PCN can generate ROS and simultaneously release vancomycin, demonstrating a synergistic antibacterial effect between the antibiotic and PDT[19].
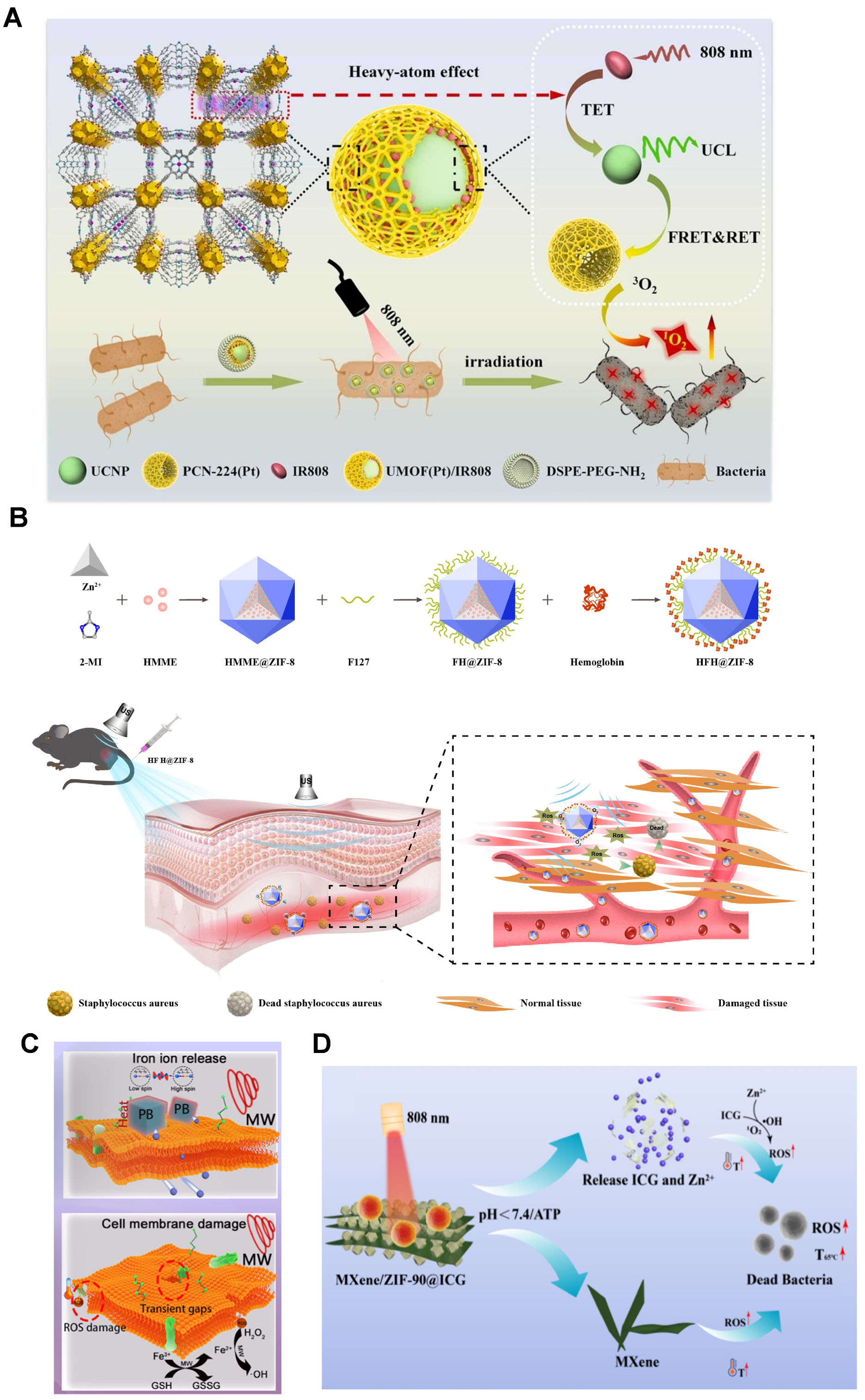
Near-infrared (NIR) light has strong tissue penetration and good biocompatibility. However, due to its lower energy, its ability to activate photosensitizers is relatively poor[36-39]. Upconversion nanoparticles (UCNPs) have unique anti-Stokes luminescence properties, capable of emitting high-energy ultraviolet and visible light upon excitation by low-energy NIR light, thereby enhancing the activation of photosensitizers[40]. Tan et al. combined UCNPs and PCN-224(Pt) to form a core-shell structure and loaded it with the NIR dye IR808, constructing an NIR light-responsive PDT nanoplatform (UMOF(Pt)/IR808)[41]. PCN-224(Pt) is formed by the coordination of Zr and Pt-TCPP. Through cascaded energy transfer from IR808 to PCN-224(Pt), UMOF(Pt)/IR808 exhibited excellent ROS generation capability under low-power-density (0.4 W/cm2) NIR light irradiation, showing outstanding antibacterial effects against MRSA[41] [Figure 2A].
Figure 2. (A) Schematic illustration of the structure and antibacterial strategy of UMOF(Pt)/IR808. Reproduced with permission[41]. Copyright 2023, Elsevier. (B) Schematic illustration of the synthesis and antibacterial strategy of HFH@ZIF-8. Reproduced with permission[56]. Copyright 2022, Elsevier. (C) Schematic illustration of the microwave-responsive antibacterial strategy of PB. Reproduced with permission[64]. Copyright 2023, Elsevier. (D) Schematic illustration of the antibacterial strategy of MXene/ZIF-90@ICG. Reproduced with permission[70]. Copyright 2024, American Chemical Society.
Light-responsive MOFs based on photothermal therapy:
Photothermal therapy (PTT) involves using a photothermal agent to convert absorbed light energy into heat, causing localized hyperthermia that denatures bacterial proteins, thereby rapidly destroying the structure and function of bacteria. It has advantages such as low side effects, non-invasiveness, and low propensity for inducing drug resistance, giving it broad clinical application prospects[42,43].
Polydopamine (PDA) is a widely used photothermal agent. It is a high-molecular-weight biopolymer formed by the oxidative polymerization of dopamine (DA) monomers, possessing high photothermal conversion efficiency and good biocompatibility[44-46]. He et al. synthesized ultrafine silver NPs (AgNPs) within the porous structure of cyclodextrin-based MOFs (CD-MOF) and coated the surface with a PDA shell[47]. Under 808 nm NIR light irradiation, the PDA shell not only generated localized hyperthermia but also accelerated the release of Ag+ through the photothermal effect, thus achieving synergistic antibacterial effects of PTT and Ag+[47].
Fang et al. grew HKUST-1 in situ on the surface of Ti3C2Tx to obtain an HKUST-1@Ti3C2Tx (HK@MX). HK@MX exhibited synergistic antibacterial effects from both PTT and Cu2+[48]. Furthermore, the numerous slit structures on its surface enhanced the adsorption of bacteria, further improving its antibacterial performance[48].
Ultrasound-responsive MOFs
Ultrasound is a mechanical wave with a frequency higher than 20 kHz. Its non-invasiveness, deep penetration (≥ 10 cm), high controllability, and low cost make it a highly promising external stimulus[49,50]. Sonodynamic Therapy (SDT) involves activating a sonosensitizer accumulated at the infection site with ultrasound, inducing ROS generation to exert an antibacterial effect[51]. SDT significantly overcomes the limitation of poor tissue penetration (≤ 1 cm) in PDT, demonstrating unique advantages in the treatment of deep-seated infections[52]. However, traditional sonosensitizers suffer from low bioavailability, poor stability, and insufficient ROS generation efficiency, which severely restricts their in vivo application[52-55]. MOFs can effectively load and protect sonosensitizers, preventing their degradation in the physiological environment and thus fully unleashing the therapeutic potential of SDT.
Geng et al. loaded the sonosensitizer hematoporphyrin monomethyl ether (HMME) into ZIF-8 and modified its surface with hemoglobin, constructing the HFH@ZIF-8 nanoplatform[56]. ZIF-8 can effectively prevent the degradation of HMME in vivo, while hemoglobin can transport and supply oxygen, providing sufficient oxygen for SDT and thereby enhancing the efficiency of ROS generation[56] [Figure 2B].
Wang et al. combined a porphyrin MOF (HNTM) with Ti3C2, constructing a hybrid nanomaterial HN-Ti3C2 with a Schottky heterojunction[57]. HNTM can respond to ultrasound to generate abundant electron-hole pairs, while Ti3C2 facilitates the rapid transfer of electrons, leading to the generation of a large amount of ROS. It demonstrated an antibacterial efficiency of up to 99.75% against MRSA[57].
Microwave-responsive MOFs
Microwave is an electromagnetic wave with a wavelength between 1 mm and 1 m. It has advantages such as strong operability, deep penetration, and minimal side effects, making it suitable for treating deep tissue infections[58,59]. Microwave-responsive antibacterial MOFs have good application potential in the biomedical field due to their ability to absorb and convert microwave energy. It exhibits two therapeutic mechanisms: (1) Microwave Dynamic Therapy (MDT): Catalyzing ROS generation; (2) Microwave Thermal Therapy (MTT): Generating localized hyperthermia at the infection site[60,61].
Due to the low energy of microwaves (10-3 eV), it is difficult to directly induce electron transitions to catalyze ROS generation[62]. Zheng et al. found that under the synergistic effect of Na+ and pulsed microwave radiation, the MDT performance of a cerium-based porphyrin MOF (CeTCPP) in a NaCl solution was significantly enhanced[63]. Under the constantly changing microwave field of pulsed microwave radiation, the tribovoltaic effect occurs between CeTCPP and water molecules, thereby inducing electron transitions and generating ROS[63].
Wei et al. developed a Na+-inserted Prussian blue (PB) microwave-responsive material, which demonstrated a synergistic effect of MTT and MDT[64]. PB is a mesoporous MOF that can achieve efficient antibacterial effects through dielectric loss under microwave irradiation. After the insertion of Na+ into PB from a NaCl solution, the spin state of iron ions in PB changed irreversibly, which accelerates the release of iron ions during microwave irradiation. Furthermore, microwave irradiation can also increase the permeability of the bacterial membrane, promoting the entry of released iron ions into the bacteria. These iron ions then react with intracellular H2O2 through a Fenton-like reaction to generate ROS, while simultaneously accelerating the consumption of bacterial glutathione (GSH), achieving a highly efficient antibacterial effect[64]
Biochemical microenvironment-responsive MOFs
pH-responsive MOFs
Under physiological conditions, the pH of blood and normal tissue microenvironments is maintained at neutral (pH ≈ 7.4). However, at the site of a bacterial infection, enhanced bacterial metabolic activity releases lactic acid, carbon dioxide, and other acidic metabolites, causing the local microenvironment to become weakly acidic (pH ≈ 5.5)[28,65]. Based on this, researchers have constructed pH-responsive MOFs that can precisely release antibacterial components in response to changes in pH.
ZIF-8 is one of the most commonly used pH-responsive MOFs[66]. Soltani et al. synthesized gentamicin-loaded ZIF-8 (GEN@NZIF-8) NPs[20]. Under acidic conditions, the 2-methylimidazole ligand of ZIF-8 undergoes protonation, which weakens its coordination with Zn2+, thereby accelerating the collapse of the ZIF-8 framework and the release of the guest molecules. Experiments showed that the release rate of gentamicin in acetate buffer solution (pH = 5.0) was significantly higher than that in phosphate buffer solution (pH = 7.4), demonstrating excellent pH-responsive properties[20].
Wang et al. used a hierarchically porous B-UiO-66 to load the natural antibacterial substance eugenol (Eu) and further chelated it with Zn2+ to construct Eu@B-UiO-66/Zn[67]. Eu@B-UiO-66/Zn uses the chelation ability between eugenol and Zn2+ as a pH-responsive switch: in an alkaline environment (pH = 8.0), the phenolic hydroxyl group of eugenol is deprotonated, enhancing the electron-donating ability of the oxygen atom and thus promoting stable coordination between Zn2+ and eugenol. In an acidic environment
Bacterial products-responsive MOFs
During an infection, bacteria secrete various specific products into the surrounding environment (e.g., HAase, ATP), which can serve as "biological switches" to regulate drug delivery.
Zhang et al. constructed a PCN-224-Ag-HA system responsive to bacterial HAase. The Ag+-loaded PCN-224 possesses both Ag+ release and PDT capabilities[29]. At the site of bacterial infection, the HA is degraded by the bacteria-secreted HAase, exposing the positively charged PCN-224-Ag+. This allows it to bind to bacteria through electrostatic interactions, thereby fully exerting the synergistic antibacterial effects of Ag+ release and PDT[29].
ATP is the most abundant energy source for microbial metabolism and an essential metabolite for maintaining the life activities of viable microorganisms[30]. Some zeolitic imidazolate frameworks (ZIFs) have a unique responsiveness to ATP. As an unstable high-energy compound, ATP has a strong coordination ability and can compete with imidazole ligands for coordination with Zn2+, thereby triggering an ATP-responsive structural collapse[68-70]. Based on this, Ding et al. loaded the photosensitizer indocyanine green (ICG) into ZIF-90 and combined it with the photothermal agent MXene-NH2 nanosheets, constructing a pH/ATP dual-responsive MXene/ZIF-90@ICG composite material[70]. Under NIR light irradiation, an acidic microenvironment (pH = 5.5) and an ATP microenvironment (0.5 mM) can accelerate the collapse of ZIF-90, releasing Zn2+ and ICG. MXene/ZIF-90@ICG can be used to prepare a nano-antibacterial spray for responsive therapy in a bacterial infection microenvironment[70] [Figure 2D].
INNOVATIVE ANTIBACTERIAL APPLICATIONS OF MOF COMPOSITE MATERIALS
MOF composite materials represent a highly versatile platform for addressing the challenge of clinical bacterial infections. Their multifunctionality renders them potent candidates for engineering antibacterial therapeutic systems. Among the numerous applications, anti-biofilm therapy, immuno-synergistic antibacterial therapy, and implant surface modification have garnered significant attention and demonstrate more promising potential for clinical translation.
MOFs in anti-biofilm therapy
Biofilm is a widespread, dynamic, and complex multicellular community of surface-attached microorganisms embedded in an extracellular polymeric substance[71,72]. It provides effective protection for bacteria and enhances their resistance to antibacterial agents[14,15].
Inhibiting initial bacterial adhesion is the primary defense against the development of biofilms. By designing various MOF coatings or films, it is possible to effectively prevent initial bacterial adhesion and kill bacteria that have already attached, thereby preventing biofilm formation. Skvortsova et al. covalently immobilized CuBTC on a polypropylene (PP) film surface, successfully constructing a smart, self-activating antibacterial coating[73]. This coating can respond to the bacterial infection marker GSNO by releasing the antibacterial free radical NO•, providing a solution for preventing catheter-related infections on surfaces such as intravenous or urinary catheters[73].
Extracellular Polymeric Substances (EPS) are the core structure of a biofilm, composed of lipids, extracellular DNA (eDNA), proteins, and polysaccharides. In addition to providing structural support, they can also form a physical barrier and enhance the drug resistance of the biofilm[71]. Therefore, developing MOF composite material that can effectively degrade or penetrate the EPS barrier has become an important strategy in anti-biofilm therapy.
eDNA is a key component that connects bacteria and other EPS components, playing a crucial role in biofilm formation, structural stability, and drug resistance[74-76]. Gokhale et al. encapsulated selenium (Se) NPs in ZIF-8 to construct Se@ZIF-8 NPs, which can degrade eDNA within the EPS, effectively disrupting the biofilm structure of Serratia marcescens[77].
To address the issue of poor penetration of •OH and other ROS in the EPS, Jia et al. designed a H2O2 self-supplying nanoplatform, CaO2/HKUST-1@l-Arg[78]. They encapsulated CaO2 NPs within the HKUST-1 framework and loaded the NO donor drug L-Arginine (l-Arg) onto its surface. CaO2 NPs react with water to continuously generate a large amount of H2O2, which then generates •OH through Fenton-like reaction. Meanwhile, H2O2 catalyzes the conversion of l-Arg to NO, which further generates the more potent antibacterial agent ONOO-. Additionally, NO also inhibits the synthesis and stability of EPS by reducing c-di-GMP levels, thereby weakening the protective effect of EPS[78].
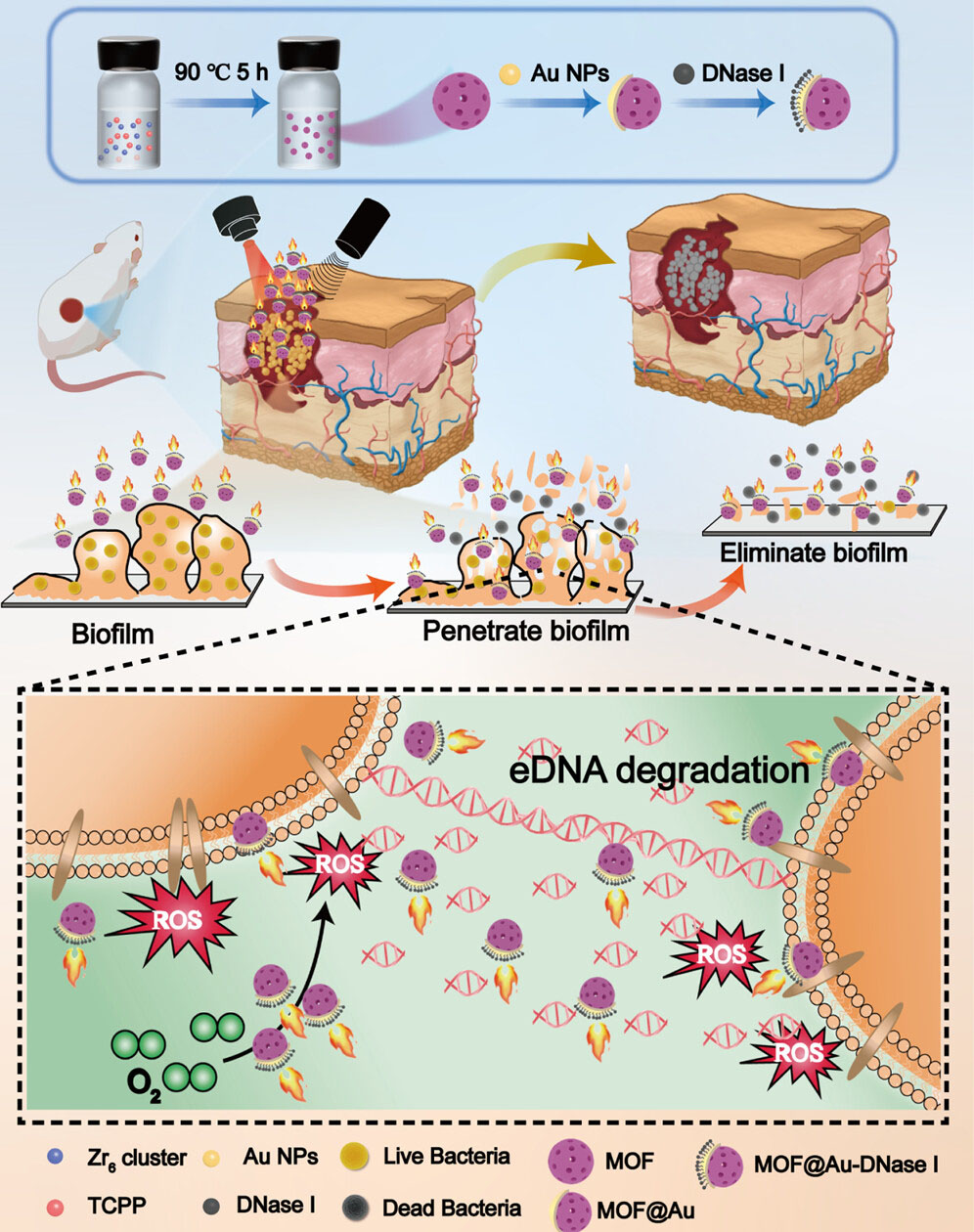
Nanomotors are nanoscale devices that achieve active movement in various liquid environments by converting different forms of energy into mechanical motion[79]. Guo et al. deposited Au NPs onto one half of the surface of a zirconium-based porphyrin MOF and then covalently attached deoxyribonuclease I
Figure 3. Schematic illustration of the synthesis and anti-biofilm strategy of MOF@Au-DNase I NPs. Reproduced with permission[74]. Copyright 2023, American Chemical Society.
MOFs in immuno-synergistic antibacterial therapy
The biofilm microenvironment has immunosuppressive properties[80]. The encapsulated bacteria-associated antigens (BAAs) are difficult to expose, leading to impaired antigen recognition and dysfunctional antigen-presenting cells (APCs), which in turn results in a failed immune response[81,82]. Furthermore, metabolic products within the biofilm, along with the acidic and hypoxic microenvironment, further induce immune cell dysfunction. For example, they polarize pro-inflammatory M1 macrophages towards anti-inflammatory M2 macrophages, significantly weakening the phagocytic and bactericidal capacities of macrophages[82-84].
Luo et al. reported a MnO2-loaded Cu-MOF (MCM). MnO2 can catalyze endogenous H2O2 to generate O2, thereby reversing the hypoxic microenvironment within biofilms[85]. This reoxygenation shifts bacterial metabolism from glycolysis to aerobic respiration, enhancing bacterial cuproptosis-like death. The reversal of the hypoxic microenvironment not only promotes dendritic cell maturation but also induces macrophage polarization toward the pro-inflammatory M1 phenotype, significantly enhancing the phagocytic activity and chemotactic response of immune cells, thereby reversing the immunosuppressive state and improving antibacterial efficacy[85].
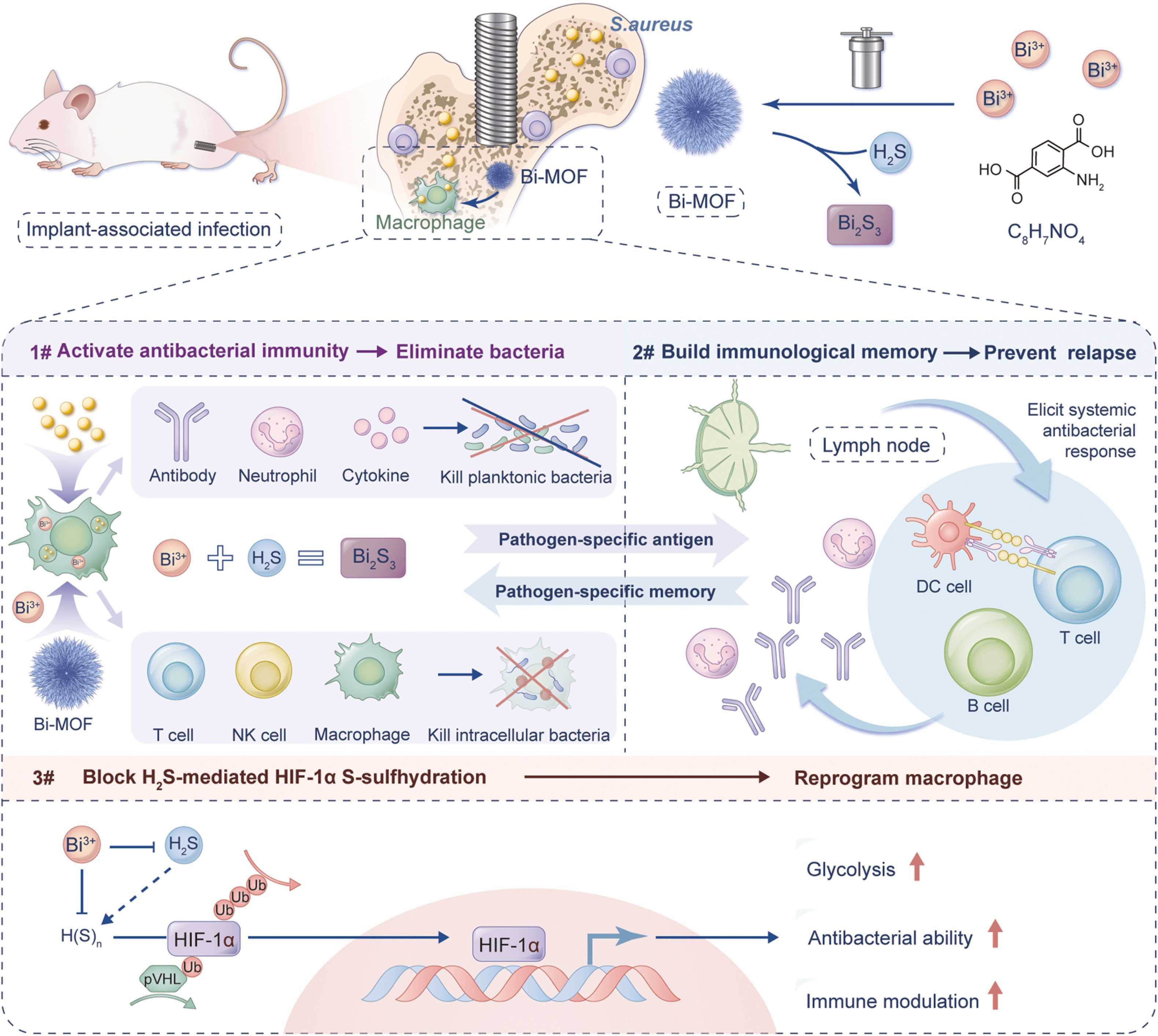
Hydrogen sulfide (H2S), a key gas-signaling molecule involved in host-pathogen interactions, has emerged as a crucial therapeutic target for immunomodulation[86]. Yang et al. reported a bismuth-based MOF (Bi-MOF). Based on the high affinity of bismuth for sulfur, Bi-MOF can efficiently scavenge intracellular H2S, blocking the S-sulfhydration of HIF-1α in macrophages, thereby inhibiting HIF-1α degradation and enhancing its post-transcriptional stability[87]. Experiments showed that Bi-MOF can induce metabolic reprogramming in macrophages, significantly enhancing their antibacterial and immunomodulatory functions. It reverses the immunosuppressive state in a macrophage-dependent manner, ultimately establishing immune memory against bacterial infections[87] [Figure 4].
Figure 4. Schematic illustration of the synthesis and immuno-synergistic antibacterial strategy of Bi-MOF. Reproduced with permission[87]. Copyright 2024, Elsevier.
Research shows that bacteria can inhibit oxidative stress by synthesizing H2S to evade immune killing[88,89]. To address this immune evasion mechanism, Ge et al. designed hafnium-based UiO-66 NPs loaded with aurintricarboxylic acid (A@UHf NPs)[90]. The piezoelectrically enhanced hafnium-based UiO-66 boosts photocatalytic ROS generation by tilting the band structure, while aurintricarboxylic acid blocks endogenous H2S production by inhibiting cystathionine γ-cleaving enzyme. The combined effect of ROS burst and H2S inhibition disrupts the redox homeostasis of the bacterial biofilm, inducing ferroptosis-like death in bacteria. Simultaneously, it activates the polarization of neutrophils in the infection microenvironment towards the N1 phenotype, thereby remodeling the antibacterial immune response[90].
MOFs in implant surface modification
Titanium has excellent biocompatibility and mechanical properties, making it a commonly used implant material in orthopedics and dentistry. To enhance the antibacterial and osseointegration capabilities of titanium implants, surface modification based on MOFs has recently become a research hotspot. However, titanium is biologically inert, and its surface lacks the organic groups necessary for binding with MOFs[91]. Therefore, various pre-treatment strategies are employed to improve the bonding of MOFs to the titanium surface.
Alkali-heat treatment, which involves immersing titanium in a sodium hydroxide solution at high temperature, can create a sponge-like nanostructure on its surface and form Ti-OH groups, thereby increasing the loading sites for MOFs[92,93]. Studies show that an alkali-heat treatment enables the in-situ growth of a ZIF-8 film on titanium substrates, ensuring a stable bond[94]. Additionally, micro-arc oxidation is another common pretreatment method. In this technique, titanium is used as the anode to create a porous ceramic oxide coating via micro-arc discharge in an electrolyte at high voltage[95]. Experiments have shown that micro-arc oxidation can increase the effective loading sites of the titanium surface, facilitating efficient, uniform loading of ZIF-8[96].
In addition to pretreating the titanium surface, an intermediate layer can be introduced between the titanium and MOFs to enhance their bonding stability. PDA, for instance, spontaneously polymerizes on the titanium surface and promotes the adhesion of MOFs through its excellent adhesive properties[97]. Si et al. immobilized methyl vanillate (MV)-loaded ZIF-8 onto a PDA-modified titanium alloy surface, which exhibited excellent tribocorrosion resistance, antibacterial activity, and osteogenic effects[97].
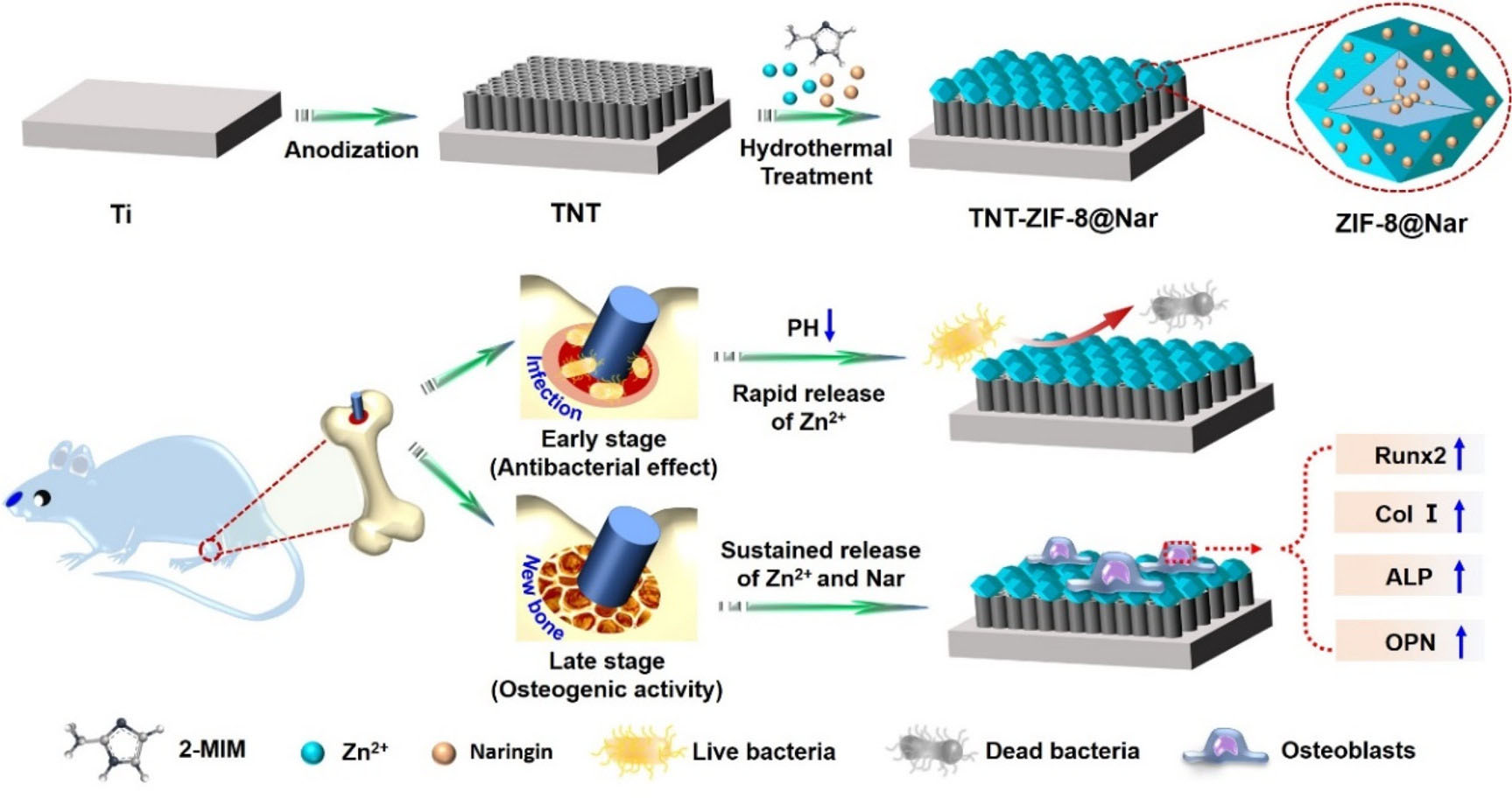
Titania nanotubes (TNTs) possess a controllable nano-topography and excellent drug-loading capacity, along with inherent antibacterial and osteogenic activity[98,99]. Wang et al. fabricated TNTs on a titanium surface via electrochemical anodization and subsequently modified them with naringin (Nar)-loaded ZIF-8. The resulting TNT-ZIF-8@Nar coating demonstrated significant antibacterial performance and effectively promoted osteogenic differentiation and implant osseointegration[100] [Figure 5].
Figure 5. Schematic illustration of the synthesis and antibacterial strategy of TNT-ZIF-8@Nar. Reproduced with permission[100]. Copyright 2022, American Chemical Society.
BIOCOMPATIBILITY AND BIOSAFETY OF ANTIBACTERIAL MOFS
Despite the excellent antibacterial performance of MOFs demonstrated in vitro and in animal models, their clinical translation still faces significant biosafety challenges[101]. Therefore, advancing the clinical application of antibacterial MOFs hinges on a comprehensive understanding of their biosafety. This involves defining their dose-dependent therapeutic window, investigating potential sources and mechanisms of toxicity, and developing effective strategies to enhance their biocompatibility.
Potential sources of MOF-associated toxicity
Both the therapeutic efficacy and toxic side effects of antibacterial MOFs exhibit concentration effects, requiring an optimal balance between the effective antibacterial concentration and a safe concentration - a concept known as the "therapeutic window"[102,103]. Ideally, an antibacterial MOF should exhibit minimal toxicity to host cells at concentrations effective for pathogen eradication, thereby presenting a wide therapeutic window. Zhu et al. reported that 3D-printed scaffolds incorporating Cu-MOF-74 achieved an optimal balance between antibacterial efficacy and osteogenic potential at Cu-MOF-74 concentrations of 0.05%-0.2%. However, at concentrations ≥ 0.5%, cytotoxic effects were observed[104]. Similar conclusions have been drawn from studies on Ag-MOFs. In vitro cytotoxicity testing on Vero cell lines demonstrated a concentration-dependent decrease in cell viability, suggesting their safe application is confined to specific concentration ranges[105].
The width of the therapeutic window for antibacterial MOFs is influenced by several factors. First, the type of metal center and its release kinetics are critical. While metal ions with high antibacterial activity (e.g., Cu2+, Ag+) are highly effective, their excessive and rapid release can lead to cytotoxicity. The toxic mechanism often involves the generation of reactive oxygen or nitrogen species, which damage cell membranes, DNA, and proteins, ultimately leading to cell death. Moreover, it may disrupt normal ionic homeostasis of the cells and interfere with intracellular physiological processes, thereby leading to cytotoxicity[106,107].
The effect of organic ligands on biosafety should not be overlooked. Many antibacterial MOFs, such as HKUST-1 and ZIF-8, employ artificially synthesized ligands, including 1,3,5-benzenetricarboxylic acid (1,3,5-H3BTC) and 2-methylimidazole[108,109]. Upon the degradation of these MOFs, the biosafety and metabolic pathways of these small-molecule ligands must be carefully considered.
Strategies for enhancing biocompatibility
Selecting low-toxicity metals is a primary strategy for improving the biosafety of MOFs. Ettlinger et al. conducted a comprehensive review and roughly ranked the cytotoxicity of MOFs containing various metals[110]. MOFs containing metals such as Ca, Bi, and Eu were identified as the least cytotoxic, followed by those containing Ti, Fe, Co, Al, and Cr, which exhibited slightly higher toxicity. Moderately cytotoxic MOFs containing Zr, Mg, Gd, Ni, and Zn, whereas those containing Cu and Mn demonstrated the highest levels of cytotoxicity[110].
The use of bio-derived or biocompatible molecules as ligands to synthesize MOFs, known as Bio-MOFs, offers a promising approach to address the potential toxicity[111]. Organic ligands such as gallic acid[112], fumaric acid[113], tannic acid[114] and cyclodextrin[115] have been successfully used to synthesize various Bio-MOFs with excellent biocompatibility. CD-MOF, which are renewable, non-toxic, and edible, have demonstrated high safety profiles in the field of drug delivery[115].
Furthermore, surface modification, which involves coating MOFs with a layer of biocompatible molecules, can effectively shield them from adverse interactions with biological systems, thereby reducing cytotoxicity and immunogenicity. Modification of MOFs with materials such as chitosan[116] and hyaluronic acid[117], can effectively mitigate cytotoxic effects and improve both dispersibility and structural stability.
CONCLUSIONS
With their tunable structures and capacity for functional integration, MOFs offer unique advantages in the antibacterial field and hold significant potential for addressing various infectious diseases. Despite remarkable achievements, challenges still exist: (1) The degradation products of some MOFs may exhibit cytotoxicity. Current research has primarily focused on their short-term biocompatibility in animal models, while their long-term safety and clinical suitability in humans require further investigation. (2) The interactions between MOFs and immune cells are complex and not yet fully elucidated. (3) Although antibacterial MOFs have shown excellent efficacy against drug-resistant bacteria, their impact on the expression of drug-resistance genes needs further exploration.
The translation from laboratory to clinical practice presents significant challenges. For instance, the large-scale, standardized, and cost-effective production of MOFs remains a major barrier, demanding the development of greener and more efficient synthesis protocols that ensure stability and reproducibility[118]. Furthermore, the in vivo fate of MOFs is critically influenced by their interaction with the physiological environment. In a biological environment, NPs adsorb various biomolecules onto their surface, forming a layer known as protein corona[119]. The protein corona can alter the stability of MOFs and reduce the targeting capability, thereby severely limiting therapeutic efficacy[120].
In summary, MOFs offer a broad research and development landscape in the antibacterial field. Future studies should focus on enhancing their biosafety, conducting in-depth analyses of their antibacterial mechanisms, and advancing their clinical translation. These efforts will further promote them as a critical platform to address the drug-resistance crisis and complex infections.
DECLARATIONS
Authors’ contributions
Conception, organization, and initial manuscript drafting: Li, Z.; Zhao, N.
Conceived the work: Wang, X.; Yu, T.; Li, Z.; Zhao, N.
Organized the content and prepared the draft manuscript: Li, Z.; Zhao, N.; Hu, N.; Zhang, Y.
Supervised the work: Han, B.; Wang, X.; Yu, T.;
Revised the manuscript: Han, B.; Wang, X.; Yu, T.; Zhao, N; Hu, N.; Zhang, Y.
All authors contributed to the writing and revision of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This project is supported by the National Key R&D Program of China (2022YFA1206100 to Yu, T., 2024YFC2510702 to Han, B.), the National Natural Science Foundation of China (82370983 to Wang, X., 52373123 to Yu, T., 52473120 to Han, B., U21A2055 to Han, B.), the Beijing Physician Scientist Training Project (BJPSTP-2025-01 to Yu, T.), and the Beijing Natural Science Foundation F252067 (Yu, T.).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Zhao, Y.; Liu, S.; Shi, Z.; Zhu, H.; Li, M.; Yu, Q. Pathogen infection-responsive nanoplatform targeting macrophage endoplasmic reticulum for treating life-threatening systemic infection. Nano. Res. 2022, 15, 6243-55.
2. Guo, Z.; Chen, Y.; Wang, Y.; Jiang, H.; Wang, X. Advances and challenges in metallic nanomaterial synthesis and antibacterial applications. J. Mater. Chem. B. 2020, 8, 4764-77.
4. Hou, J.; Long, X.; Wang, X.; et al. Global trend of antimicrobial resistance in common bacterial pathogens in response to antibiotic consumption. J. Hazard. Mater. 2023, 442, 130042.
5. Zhou, Z.; Li, B.; Liu, X.; et al. Recent progress in photocatalytic antibacterial. ACS. Appl. Bio. Mater. 2021, 4, 3909-36.
6. Rappuoli, R.; Bloom, D. E.; Black, S. Deploy vaccines to fight superbugs. Nature 2017, 552, 165-7.
7. Lemire, J. A.; Harrison, J. J.; Turner, R. J. Antimicrobial activity of metals: mechanisms, molecular targets and applications. Nat. Rev. Microbiol. 2013, 11, 371-84.
8. Lucky, S. S.; Soo, K. C.; Zhang, Y. Nanoparticles in photodynamic therapy. Chem. Rev. 2015, 115, 1990-2042.
9. Yang, Z.; Liu, Z. W.; Allaker, R. P.; et al. A review of nanoparticle functionality and toxicity on the central nervous system. J. R. Soc. Interface. 2010, 7 Suppl 4, S411-22.
10. Wang, T. Y.; Zhu, X. Y.; Wu, F. G. Antibacterial gas therapy: Strategies, advances, and prospects. Bioact. Mater. 2023, 23, 129-55.
11. Alves, S. R.; Calori, I. R.; Tedesco, A. C. Photosensitizer-based metal-organic frameworks for highly effective photodynamic therapy. Mater. Sci. Eng. C. Mater. Biol. Appl. 2021, 131, 112514.
12. Saxena, V.; Sadoqi, M.; Shao, J. Degradation kinetics of indocyanine green in aqueous solution. J. Pharm. Sci. 2003, 92, 2090-7.
13. Quek, J.; Uroro, E.; Goswami, N.; Vasilev, K. Design principles for bacteria-responsive antimicrobial nanomaterials. Mater. Today. Chem. 2022, 23, 100606.
14. Hall-Stoodley, L.; Costerton, J. W.; Stoodley, P. Bacterial biofilms: from the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95-108.
15. Townsley, L.; Shank, E. A. Natural-product antibiotics: cues for modulating bacterial biofilm formation. Trends. Microbiol. 2017, 25, 1016-26.
16. Yan, L.; Gopal, A.; Kashif, S.; et al. Metal organic frameworks for antibacterial applications. Chem. Eng. J. 2022, 435, 134975.
17. Lu, X.; Ye, J.; Zhang, D.; et al. Silver carboxylate metal-organic frameworks with highly antibacterial activity and biocompatibility. J. Inorg. Biochem. 2014, 138, 114-21.
18. Wei, Y.; Chen, C.; Zhai, S.; et al. Enrofloxacin/florfenicol loaded cyclodextrin metal-organic-framework for drug delivery and controlled release. Drug. Delivery. 2021, 28, 372-9.
19. Wang, H.; Wu, Y.; Wang, Y.; Zou, H.; Wang, H.; Zhou, M. Vancomycin-loaded porphyrinic MOF nanoparticles for killing pathogenic bacteria. ACS. Appl. Nano. Mater. 2023, 6, 9874-83.
20. Soltani, B.; Nabipour, H.; Nasab, N. A. Efficient storage of gentamicin in nanoscale zeolitic imidazolate framework-8 nanocarrier for pH-responsive drug release. J. Inorg. Organomet. Polym. Mater. 2017, 28, 1090-7.
21. Amer, T. A. M.; Palanisamy, S.; So, P. B.; et al. Sustained releasable copper and zinc biogenic ions Co-assembled in metal-organic frameworks reinforced bacterial eradication and wound mitigation in diabetic mice. Bioconjugate. Chem. 2023, 34, 1688-703.
22. Duan, F.; Feng, X.; Jin, Y.; et al. Metal-carbenicillin framework-based nanoantibiotics with enhanced penetration and highly efficient inhibition of MRSA. Biomaterials 2017, 144, 155-65.
23. Chen, M.; Long, Z.; Dong, R.; et al. Titanium incorporation into Zr-porphyrinic metal-organic frameworks with enhanced antibacterial activity against multidrug-resistant pathogens. Small 2020, 16, e1906240.
24. Acharya, A. P.; Sezginel, K. B.; Gideon, H. P.; et al. In silico identification and synthesis of a multi-drug loaded MOF for treating tuberculosis. J. Control. Release. 2022, 352, 242-55.
25. Redfern, J.; Geerts, L.; Seo, J. W.; Verran, J.; Tosheva, L.; Wee, L. H. Toxicity and antimicrobial properties of ZnO@ZIF-8 embedded silicone against planktonic and biofilm catheter-associated pathogens. ACS. Appl. Nano. Mater. 2018, 1, 1657-65.
26. Yi, J.; Pei, C.; Zhang, T.; et al. Nanoscale multipatterning Zn,Co-ZIF@FeOOH for eradication of multidrug-resistant bacteria and antibacterial treatment of wounds. ACS. Appl. Mater. Interfaces. 2024, 16, 58217-25.
27. Li, H.; Li, B.; Lv, D.; Li, W.; Lu, Y.; Luo, G. Biomaterials releasing drug responsively to promote wound healing via regulation of pathological microenvironment. Adv. Drug. Delivery. Rev. 2023, 196, 114778.
28. Xiang, Y. L.; Tang, D. Y.; Yan, L. L.; et al. Poly-l-lysine modified MOF nanoparticles with pH/ROS sensitive CIP release and CUR triggered photodynamic therapy against drug-resistant bacterial infection. Int. J. Biol. Macromol. 2024, 266, 131330.
29. Zhang, Y.; Sun, P.; Zhang, L.; et al. Silver-infused porphyrinic metal-organic framework: surface-adaptive, on-demand nanoplatform for synergistic bacteria killing and wound disinfection. Adv. Funct. Mater. 2019, 29, 1808594.
30. Ding, N.; Qin, M.; Sun, Y.; et al. Universal near-infrared fluorescent nanoprobes for detection and real-time imaging of ATP in real food samples, living cells, and bacteria. J. Agric. Food. Chem. 2023, 71, 12070-9.
31. Qiao, E.; Qiu, F.; Guo, X.; et al. Biomaterial-mediated modulation of tumor microenvironment and therapeutic sensitization. EngMedicine 2025, 2, 100092.
32. Sobotta, L.; Skupin-Mrugalska, P.; Piskorz, J.; Mielcarek, J. Porphyrinoid photosensitizers mediated photodynamic inactivation against bacteria. Eur. J. Med. Chem. 2019, 175, 72-106.
33. Tian, J.; Huang, B.; Nawaz, M. H.; Zhang, W. Recent advances of multi-dimensional porphyrin-based functional materials in photodynamic therapy. Coord. Chem. Rev. 2020, 420, 213410.
34. Sies, H.; Belousov, V. V.; Chandel, N. S.; et al. Defining roles of specific reactive oxygen species (ROS) in cell biology and physiology. Nat. Rev. Mol. Cell. Biol. 2022, 23, 499-515.
35. Lan, G.; Ni, K.; Lin, W. Nanoscale metal-organic frameworks for phototherapy of cancer. Coord. Chem. Rev. 2019, 379, 65-81.
36. Zhou, Z.; Song, J.; Nie, L.; Chen, X. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016, 45, 6597-626.
37. Sun, J.; Song, L.; Fan, Y.; et al. Synergistic photodynamic and photothermal antibacterial nanocomposite membrane triggered by single NIR light source. ACS. Appl. Mater. Interfaces. 2019, 11, 26581-9.
38. Agostinis, P.; Berg, K.; Cengel, K. A.; et al. Photodynamic therapy of cancer: an update. CA. Cancer. J. Clin. 2011, 61, 250-81.
39. Chen, Y.; Gao, Y.; Chen, Y.; Liu, L.; Mo, A.; Peng, Q. Nanomaterials-based photothermal therapy and its potentials in antibacterial treatment. J. Control. Release. 2020, 328, 251-62.
40. Du, J.; Jia, T.; Zhang, J.; Chen, G. Heterostructures combining upconversion nanoparticles and metal-organic framework: fundamental, classification, and theranostic applications. Adv. Opt. Mater. 2023, 11, 2202122.
41. Tan, C.; Li, X.; Li, Z.; et al. Near-infrared-responsive nanoplatforms integrating dye-sensitized upconversion and heavy-atom effect for enhanced photodynamic therapy efficacy. Nano. Today. 2024, 54, 102089.
42. Huang, J.; Feng, X.; Zhao, Y.; et al. Coral-like AgNPs hybrided MOFs modulated with biopolymer polydopamine for synergistic antibacterial and biofilm eradication. Int. J. Biol. Macromol. 2024, 282, 137080.
43. Huo, J.; Jia, Q.; Huang, H.; et al. Emerging photothermal-derived multimodal synergistic therapy in combating bacterial infections. Chem. Soc. Rev. 2021, 50, 8762-89.
44. Yang, Q.; Zhao, J.; Muhammad, A.; et al. Biopolymer coating for particle surface engineering and their biomedical applications. Mater. Today. Bio. 2022, 16, 100407.
45. Li, H.; Yin, D.; Li, W.; Tang, Q.; Zou, L.; Peng, Q. Polydopamine-based nanomaterials and their potentials in advanced drug delivery and therapy. Colloids. Surf. B. Biointerfaces. 2021, 199, 111502.
46. Jin, A.; Wang, Y.; Lin, K.; Jiang, L. Nanoparticles modified by polydopamine: working as "drug" carriers. Bioact. Mater. 2020, 5, 522-41.
47. He, Y.; Wang, X.; Zhang, C.; Sun, J.; Xu, J.; Li, D. Near-infrared light-mediated cyclodextrin metal-organic frameworks for synergistic antibacterial and anti-biofilm therapies. Small 2023, 19, e2300199.
48. Fang, L.; Chen, Y.; Shan, W.; et al. HKUST-1 loaded few-layer Ti3C2Tx for synergistic chemo-photothermal effects to enhance antibacterial activity. J. Mater. Chem. B. 2023, 11, 3929-40.
49. Pan, X.; Wang, W.; Huang, Z.; et al. MOF-derived double-layer hollow nanoparticles with oxygen generation ability for multimodal imaging-guided sonodynamic therapy. Angew. Chem. Int. Ed. 2020, 59, 13557-61.
50. Son, S.; Kim, J. H.; Wang, X.; et al. Multifunctional sonosensitizers in sonodynamic cancer therapy. Chem. Soc. Rev. 2020, 49, 3244-61.
51. Wang, R.; Liu, Q.; Gao, A.; et al. Recent developments of sonodynamic therapy in antibacterial application. Nanoscale 2022, 14, 12999-3017.
52. Xiao, Z.; Chen, Q.; Yang, Y.; et al. State of the art advancements in sonodynamic therapy (SDT): metal-Organic frameworks for SDT. Chem. Eng. J. 2022, 449, 137889.
53. Zhang, H.; Shi, R.; Xie, A.; et al. Novel TiO2/PEGDA hybrid hydrogel prepared in situ on tumor cells for effective photodynamic therapy. ACS. Appl. Mater. Interfaces. 2013, 5, 12317-22.
54. Zhou, Y.; Wang, M.; Dai, Z. The molecular design of and challenges relating to sensitizers for cancer sonodynamic therapy. Mater. Chem. Front. 2020, 4, 2223-34.
55. Ouyang, J.; Tang, Z.; Farokhzad, N.; et al. Ultrasound mediated therapy: recent progress and challenges in nanoscience. Nano. Today. 2020, 35, 100949.
56. Geng, X.; Chen, Y.; Chen, Z.; Wei, X.; Dai, Y.; Yuan, Z. Oxygen-carrying biomimetic nanoplatform for sonodynamic killing of bacteria and treatment of infection diseases. Ultrason. Sonochem. 2022, 84, 105972.
57. Wang, H.; Mu, N.; He, Y.; et al. Ultrasound-controlled MXene-based Schottky heterojunction improves anti-infection and osteogenesis properties. Theranostics 2023, 13, 1669-83.
58. Fu, C.; Zhou, H.; Tan, L.; et al. Microwave-activated Mn-doped zirconium metal-organic framework nanocubes for highly effective combination of microwave dynamic and thermal therapies against cancer. ACS. Nano. 2018, 12, 2201-10.
59. Majdzadeh-Ardakani, K.; Banaszak, Holl. M. M. Nanostructured materials for microwave receptors. Prog. Mater. Sci. 2017, 87, 221-45.
60. Du, Y.; Zhou, J.; He, F.; et al. A bright future: advanced nanotechnology-assisted microwave therapy. Nano. Today. 2023, 52, 101963.
61. Qiao, Y.; Wu, S.; Zheng, Y.; et al. Enhancing microwave dynamic effects via surface states of ultrasmall 2D MOF triggered by interface confinement for antibiotics-free therapy. Adv. Sci. 2023, 10, e2300084.
62. Wu, Q.; Xia, N.; Long, D.; et al. Dual-functional supernanoparticles with microwave dynamic therapy and microwave thermal therapy. Nano. Lett. 2019, 19, 5277-86.
63. Zheng, Q.; Liu, X.; Mao, C.; et al. Tribovoltaic effect strengthened microwave catalytic antibacterial composite hydrogel. Adv. Healthc. Mater. 2024, 13, e2402266.
64. Wei, S.; Qiao, Y.; Wu, Z.; et al. Na+ inserted metal-organic framework for rapid therapy of bacteria-infected osteomyelitis through microwave strengthened Fenton reaction and thermal effects. Nano. Today. 2021, 37, 101090.
65. Tan, L.; Li, J.; Liu, X.; et al. Rapid biofilm eradication on bone implants using red phosphorus and near-infrared light. Adv. Mater. 2018, 30, e1801808.
66. Ke, Q.; Jiang, K.; Li, H.; Zhang, L.; Chen, B. Hierarchically micro-, meso-, and macro-porous MOF Nanosystems for localized cross-scale dual-biomolecule loading and guest-carrier cooperative anticancer therapy. ACS. Nano. 2024, 18, 21911-24.
67. Wang, J.; Li, L.; Hu, X.; Zhou, L.; Hu, J. pH-responsive on-demand release of eugenol from metal-organic frameworks for synergistic bacterial killing. Dalton. Trans. 2024, 53, 2826-32.
68. Deng, J.; Wang, K.; Wang, M.; Yu, P.; Mao, L. Mitochondria targeted nanoscale zeolitic imidazole framework-90 for ATP imaging in live cells. J. Am. Chem. Soc. 2017, 139, 5877-82.
69. Jiang, Z.; Wang, Y.; Sun, L.; et al. Dual ATP and pH responsive ZIF-90 nanosystem with favorable biocompatibility and facile post-modification improves therapeutic outcomes of triple negative breast cancer in vivo. Biomaterials 2019, 197, 41-50.
70. Ding, N.; Zhang, B.; Khan, I. M.; et al. Dual pH- and ATP-responsive antibacterial nanospray: on-demand release of antibacterial factors, imaging monitoring, and accelerated healing of bacteria-infected wounds under NIR activation. ACS. Appl. Mater. Interfaces. 2024, 16, 30728-41.
71. Fulaz, S.; Vitale, S.; Quinn, L.; Casey, E. Nanoparticle-biofilm interactions: the role of the EPS matrix. Trends. Microbiol. 2019, 27, 915-26.
72. Flemming, H. C.; Wuertz, S. Bacteria and archaea on Earth and their abundance in biofilms. Nat. Rev. Microbiol. 2019, 17, 247-60.
73. Skvortsova, A.; Kocianova, A.; Guselnikova, O.; et al. Self-activated antibacterial MOF-based coating on medically relevant polypropylene. Appl. Surf. Sci. 2023, 623, 157048.
74. Guo, W.; Wang, Y.; Zhang, K.; et al. Near-infrared light-propelled MOF@Au nanomotors for enhanced penetration and sonodynamic therapy of bacterial biofilms. Chem. Mater. 2023, 35, 6853-64.
75. Hu, H.; Kang, X.; Shan, Z.; et al. A DNase-mimetic artificial enzyme for the eradication of drug-resistant bacterial biofilm infections. Nanoscale 2022, 14, 2676-85.
76. Chen, Z.; Ji, H.; Liu, C.; Bing, W.; Wang, Z.; Qu, X. A multinuclear metal complex based DNase-mimetic artificial enzyme: matrix cleavage for combating bacterial biofilms. Angew. Chem. 2016, 128, 10890-4.
77. Gokhale, K. M.; Patravale, V.; Pingale, R.; Pandey, P.; Vavilala, S. L. Se-functionalized ZIF-8 nanoparticles: synthesis, characterization and disruption of biofilms and quorum sensing inSerratia marcescens. Biomed. Mater. 2024, 19, 055020.
78. Jia, D.; Zou, Y.; Zhang, Y.; et al. A self-supplied hydrogen peroxide and nitric oxide-generating nanoplatform enhances the efficacy of chemodynamic therapy for biofilm eradication. J. Colloid. Interface. Sci. 2025, 678, 20-9.
79. Ye, J.; Fan, Y.; Niu, G.; Zhou, B.; Kang, Y.; Ji, X. Intelligent micro/nanomotors: fabrication, propulsion, and biomedical applications. Nano. Today. 2024, 55, 102212.
80. Yang, C.; Luo, Y.; Shen, H.; et al. Inorganic nanosheets facilitate humoral immunity against medical implant infections by modulating immune co-stimulatory pathways. Nat. Commun. 2022, 13, 4866.
81. Hu, X.; Ma, R.; Zhang, P.; et al. Biomimetic metal-organic framework combats biofilm-associated infections via hyperthermia-enhanced bacterial metabolic interference and autophagy-promoted adaptive immunity. Adv. Funct. Mater. 2023, 34, 2310509.
82. Thurlow, L. R.; Hanke, M. L.; Fritz, T.; et al. Staphylococcus aureus biofilms prevent macrophage phagocytosis and attenuate inflammation in vivo. J. Immunol. 2011, 186, 6585-96.
83. Arciola, C. R.; Campoccia, D.; Montanaro, L. Implant infections: adhesion, biofilm formation and immune evasion. Nat. Rev. Microbiol. 2018, 16, 397-409.
84. Su, Z.; Kong, L.; Mei, J.; et al. Enzymatic bionanocatalysts for combating peri-implant biofilm infections by specific heat-amplified chemodynamic therapy and innate immunomodulation. Drug. Resist. Updat. 2023, 67, 100917.
85. Luo, Z.; Lu, R.; Shi, T.; et al. Enhanced bacterial cuproptosis-like death via reversal of hypoxia microenvironment for biofilm infection treatment. Adv. Sci. 2024, 11, e2308850.
86. Wolfson, S. J.; Hitchings, R.; Peregrina, K.; et al. Bacterial hydrogen sulfide drives cryptic redox chemistry in gut microbial communities. Nat. Metab. 2022, 4, 1260-70.
87. Yang, Y.; Huang, K.; Yuan, K.; et al. A novel antibacterial immune activator: Bi-MOF acts as H2S scavenger to suppress HIF-1α S-sulfhydration and alleviate implant-associated infection. Nano. Today. 2024, 57, 102334.
88. Barayeu, U.; Schilling, D.; Eid, M.; et al. Hydropersulfides inhibit lipid peroxidation and ferroptosis by scavenging radicals. Nat. Chem. Biol. 2023, 19, 28-37.
89. Lewis, M. L.; Surewaard, B. G. J. Neutrophil evasion strategies by Streptococcus pneumoniae and Staphylococcus aureus. Cell. Tissue. Res. 2018, 371, 489-503.
90. Ge, M.; Zhu, W.; Mei, J.; et al. Piezoelectric-enhanced nanocatalysts trigger neutrophil N1 polarization against bacterial biofilm by disrupting redox homeostasis. Adv. Mater. 2025, 37, e2409633.
91. Si, Y.; Liu, H.; Yu, H.; Jiang, X.; Sun, D. MOF-derived CuO@ZnO modified titanium implant for synergistic antibacterial ability, osteogenesis and angiogenesis. Colloids. Surf. B. Biointerfaces. 2022, 219, 112840.
92. Shen, X.; Zhang, Y.; Ma, P.; et al. Fabrication of magnesium/zinc-metal organic framework on titanium implants to inhibit bacterial infection and promote bone regeneration. Biomaterials 2019, 212, 1-16.
93. Chen, X. B.; Li, Y. C.; Hodgson, P. D.; Wen, C. The importance of particle size in porous titanium and nonporous counterparts for surface energy and its impact on apatite formation. Acta. Biomater. 2009, 5, 2290-302.
94. Chen, J.; Zhang, X.; Huang, C.; et al. Osteogenic activity and antibacterial effect of porous titanium modified with metal-organic framework films. J. Biomed. Mater. Res. A. 2017, 105, 834-46.
95. Wen, X.; Liu, Y.; Xi, F.; Zhang, X.; Kang, Y. Micro-arc oxidation (MAO) and its potential for improving the performance of titanium implants in biomedical applications. Front. Bioeng. Biotechnol. 2023, 11, 1282590.
96. Teng, W.; Zhang, Z.; Wang, Y.; et al. Iodine immobilized metal-organic framework for NIR-triggered antibacterial therapy on orthopedic implants. Small 2021, 17, e2102315.
97. Si, Y.; Liu, H.; Li, M.; Jiang, X.; Yu, H.; Sun, D. An efficient metal-organic framework-based drug delivery platform for synergistic antibacterial activity and osteogenesis. J. Colloid. Interface. Sci. 2023, 640, 521-39.
98. Cheng, Y.; Yang, H.; Yang, Y.; et al. Progress in TiO2 nanotube coatings for biomedical applications: a review. J. Mater. Chem. B. 2018, 6, 1862-86.
99. Yu, Y.; Ran, Q.; Shen, X.; Zheng, H.; Cai, K. Enzyme responsive titanium substrates with antibacterial property and osteo/angio-genic differentiation potentials. Colloids. Surf. B. Biointerfaces. 2020, 185, 110592.
100. Wang, L.; Dai, F.; Yang, Y.; Zhang, Z. Zeolitic imidazolate framework-8 with encapsulated naringin synergistically improves antibacterial and osteogenic properties of Ti implants for osseointegration. ACS. Biomater. Sci. Eng. 2022, 8, 3797-809.
101. Zhao, X.; Chen, Z.; Zhang, S.; et al. Application of metal-organic frameworks in infectious wound healing. J. Nanobiotechnol. 2024, 22, 387.
102. Parizi, M. K.; Doll, K.; Rahim, M. I.; Mikolai, C.; Winkel, A.; Stiesch, M. Antibacterial and cytocompatible: combining silver nitrate with strontium acetate increases the therapeutic window. Int. J. Mol. Sci. 2022, 23, 8058.
103. Cecotto, L.; Stapels, D. A. C.; van, Kessel. K. P. M.; et al. Evaluation of silver bio-functionality in a multicellular in vitro model: towards reduced animal usage in implant-associated infection research. Front. Cell. Infect. Microbiol. 2023, 13, 1186936.
104. Zhu, T.; Ni, Q.; Wang, W.; et al. Cu-MOF-decorated 3D-printed scaffolds for infection control and bone regeneration. J. Funct. Biomater. 2025, 16, 83.
105. Desai, S. S.; Deepika, Roy. K. A.; Abishad, P.; et al. Harnessing in vitro cytotoxicity and antibacterial potential of a novel silver-DABCO framework against multi-drug-resistant pathogens. RSC. Adv. 2025, 15, 8180-8.
106. Yu, Z.; Lepoitevin, M.; Serre, C. Iron-MOFs for biomedical applications. Adv. Healthc. Mater. 2025, 14, e2402630.
107. Zhou, F.; Xu, Z.; Wang, Q.; et al. Nitric oxide-driven copper homeostasis and osteogenesis in cranial defect regeneration using l-arginine-loaded HKUST-1. Biomaterials 2026, 325, 123557.
108. Wu, M. X.; Yang, Y. W. Metal-organic framework (MOF)-based drug/cargo delivery and cancer therapy. Adv. Mater. 2017, 29, 1606134.
109. Ouyang, B.; Ouyang, P.; Shi, M.; et al. Low toxicity of metal-organic framework MOF-199 to bacteria Escherichia coli and Staphylococcus aureus. J. Hazard. Mater. Adv. 2021, 1, 100002.
110. Ettlinger, R.; Lächelt, U.; Gref, R.; et al. Toxicity of metal-organic framework nanoparticles: from essential analyses to potential applications. Chem. Soc. Rev. 2022, 51, 464-84.
111. Zhao, F.; Wan, X.; Liang, Y.; et al. Prospects, advances, and applications of BioMOF-based platforms. Top. Curr. Chem. 2025, 383, 34.
112. Luo, Z.; Ma, J.; Wang, Y.; et al. Application of Mg-MOF-loaded gelatin microspheres with osteogenic, angiogenic, and ROS scavenging capabilities in bone defect repair. Int. J. Biol. Macromol. 2024, 280, 135721.
113. Huang, L.; Guo, R.; Mao, Y.; Xu, Z.; Chi, Y. In situ encapsulation of capsaicinoids in MIL-88A as a food-grade nanopreservative for meat safety. Food. Chem. 2024, 460, 140738.
114. Sahiner, N.; Guven, O.; Demirci, S.; et al. Tannic acid-based bio-MOFs with antibacterial and antioxidant properties acquiring non-hemolytic and non-cytotoxic characteristics. Colloids. Surf. B. Biointerfaces. 2025, 252, 114669.
115. Si, Y.; Luo, H.; Zhang, P.; et al. CD-MOFs: from preparation to drug delivery and therapeutic application. Carbohydr. Polym. 2024, 323, 121424.
116. Hu, Z.; He, F.; Liu, A.; et al. Engineering a biocompatible chitosan/MOF aerogel for hyperbilirubinemia treatment in liver failure. J. Mater. Chem. B. 2025, 13, 11210-23.
117. Obaid, E. A. M. S.; Yu, C.; Ma, Y.; Yang, H.; Fu, Z. Exploring the efficacy of pH-responsive vancomycin/Ag/ZIF-8 nanoparticles modified with hyaluronic acid for enhanced antibacterial therapy and wound healing. ACS. Appl. Mater. Interfaces. 2024, 16, 65942-54.
118. Chakraborty, D.; Yurdusen, A.; Mouchaham, G.; Nouar, F.; Serre, C. Large-scale production of metal-organic frameworks. Adv. Funct. Mater. 2023, 34, 2309089.
119. Dai, Q.; Walkey, C.; Chan, W. C. Polyethylene glycol backfilling mitigates the negative impact of the protein corona on nanoparticle cell targeting. Angew. Chem. Int. Ed. 2014, 53, 5093-6.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].