Sulfur poisoning mechanism of LSCF cathode material in the presence of SO2: a computational and experimental study
Abstract
Aiming at the comprehensive understanding of the single sulfur poisoning effect and, eventually, the multiple impurities poisoning phenomena on the SOFC (Solid Oxide Fuel Cell) cathode materials, the sulfur poisoning effect on the (La0.6Sr0.4)0.95Co0.2Fe0.8O3 (LSCF-6428) has been investigated in the presence of 10 ppm SO2 at 800, 900, and 1,000 °C, respectively, with a combined computational and experimental approach. The good agreement between the CALPHAD (Computer Coupling of Phase Diagrams and Thermochemistry) simulations and the XRD (X-Ray Diffraction), SEM (Scanning Electron Microscopy), and TEM (Transmission Electron Microscopy) characterization results support the reliability of the CALPHAD approach in the SOFC field. Furthermore, comprehensive simulations were made to understand the impact of temperature, P(SO2), P(O2), and Sr concentration on the threshold of SrSO4 stability. Results showed that the formation of SrSO4 is thermodynamically favored at lower temperatures, higher P(SO2), higher P(O2), and higher Sr concentration. Finally, comparisons were also made between LSCF-6428 and LSM20 (La0.8Sr0.2MnO3) using simulations, which confirmed that LSCF-6428 is a poor sulfur-tolerant cathode, in agreement with the literature.
Keywords
INTRODUCTION
Mixed Ionic and Electronic Conducting (MIEC) (La, Sr)(Co, Fe)O3 (LSCF) is one of the most promising Solid Oxide Fuel Cell (SOFC) cathodes due to its comprehensive electrochemical properties. It has been widely applied at intermediate conditions in the energy conversion from fossil fuels to electricity to tackle the penalty of CO2 emission and poor efficiency from traditional oxy-fuel combustion[1,2]. However, SOFC cathode materials suffer from thermodynamic instability in their long-term durability in the presence of several impurities, of which SO2 is known to be a concern[3,4]. Thus, developing a comprehensive understanding of the sulfur poisoning of LSCF cathodes in the presence of SO2 impurity is necessary to address the long-term degradation of the SOFC cathode systems at operating conditions.
It is reported that a trace amount of SO2 in air, as low as in part per million (ppm) level, can still cause a performance drop and shorten the service life of LSCF cathodes due to the formation of the detrimental secondary phases[5]. Liu et al.[6] first reported the sulfur poisoning of an LSCF cathode in the presence of 1 and 20 ppm of SO2 at 800 °C for 1,000 h. Results suggested that the LSCF has a low tolerance to sulfur poisoning even with 1 ppm SO2. Moreover, the degradation rate of LSCF was 8 times higher than that of LSM at 20 ppm SO2 because of the formation of SrSO4 in LSCF. In the same year, Wang et al.[7] reported the formation of SrSO4, La2O2SO4, and CoFe2O4 in 100 ppm SO2 at 800 °C for 24 h, SrSO4 and CoFe2O4 in 10 and 1 ppm SO2 at 800 °C for 24 h, respectively. The cell was free from sulfur poisoning in 0.1 ppm SO2 for 24 h and SO2-free air for 100 h at 800 °C, respectively, indicating a thermodynamic threshold or a diffusion-controlled mechanism for the sulfur poisoning process. Wang et al.[8] reported the formation of SrSO4 in the presence of 20mg/L SO2 at 800 °C for 500 h and concluded that LSCF exhibited poorer durability than LSM against SO2 poisoning. Later, Wang et al.[9] investigated the effect of Sr concentration on the sulfur poisoning of LSCF cathodes, namely (La0.6Sr0.4)(Co0.2Fe0.8)O3 (LSCF-6428) and (La0.8Sr0.2)(Co0.2Fe0.8)O3 (LSCF-8228), at 800 °C with 1 ppm SO2. They concluded that the sulfur poisoning is much more severe for the less active LSCF-8228 cathode, even though the volume of SrSO4 formed is much larger in LSCF-6428 due to it having more available electrochemically active sites. Moreover, Wang et al.[10] evaluated the SO2 poisoning phenomena of LSCF-6428 cathodes with 0.1 ppm SO2-air at 800 °C for 100 h by varying the flow rate (25, 50, and 90 mL min-1). They concluded that SrSO4 formed on the surface of the LSCF-6428 first due to the fast absorption of the SO2 and then concentrated in the vicinity of the triple phase boundaries (TPBs) under a polarization condition. Subsequently, Wang and Jiang[11] reviewed the mechanism of SO2 poisoning on both LSCF and LSM cathodes. They reported that the sulfur poisoning effect was more pronounced at lower temperatures for both LSCF and LSM cathodes. More importantly, it was most pronounced on the surface of the LSCF cathode and more evident on the interface of LSM/YSZ TPBs. Wang et al.[12] investigated the interrelation between sulfur poisoning and performance degradation of LSCF in the presence of 1 ppm SO2 at 700 °C, 750 °C, and 800 °C, respectively. They found that the performance degradation was more severe at either 700 °C or 750 °C, even though no significant sulfur was detected in either of these two samples.
Up to now, there are two principal methods to investigate the relationship between the stabilities of the electrodes and the long-term degradation under SOFC applications, CALPHAD[13-17] and DFT (Density-Functional Theory)[18,19]. Both can provide a potential approach to designing highly efficient and stable electrode materials with excellent impurity tolerance for SOFCs. Darvish et al.[15] reported the impacts of temperatures, SO2 partial pressure (P(SO2)), oxygen partial pressure (P(O2)), and Sr content on the sulfur poisoning phenomena of the LSCF cathode using the CALPHAD approach and electrochemical analysis. They concluded that the formation of SrSO4 and spinel phase is thermodynamically in favor of higher P(SO2), higher P(O2), lower temperatures, and higher Sr content. Later, Wang et al.[5] reviewed the sulfur poisoning of SOFCs, especially the LSCF cathode, using a very detailed literature review with a corresponding chemical reaction sequence taking into account the parameters like P(SO2), temperature, P(O2), and Sr content. The primary poisoning mechanism proposed was the formation of SrSO4 from the reaction of SO2 with SrO and O2 to form SrSO4. Subsequent reactions would start to form other sulfur-containing phases like La2O2SO4 and La2(SO4)3 through a reaction with the La components. However, much remains unknown as to how these poisoning reactions are affected by the structure and composition of the cathode material, which limits our ability to design cathodes with improved long-term stability in the presence of natural SO2 impurities. Moreover, a comprehensive understanding of the relationship between the long-term degradation and single or eventually multiple gas impurities poisoning effects is still needed.
In this work, we have utilized a combined computational and experimental approach to understand the sulfur poisoning mechanism(s) and improve the long-term durability of LSCF cathodes in order to develop better-performing sulfur-tolerant cathode materials in the future. We first examined the agreement between the simulation approach and the experimental observations at various conditions, confirming the reliability of CALPHAD for poisoning simulations in these cathode systems. Using this approach, we then investigated the accelerated testing protocol, which has become the standard method for conducting sulfur poisoning experiments involving LSCF cathode materials. We found that our simulation approach is able to predict in which systems (cathode material and treatment environment) the accelerated testing protocols reflect actual operating conditions. Finally, in-depth simulations were done for LSCF and compared with those for LSM to help understand the LSCF cell system and suggest directions toward alternative cathode materials that have superior sulfur tolerance.
EXPERIMENTAL PROCEDURE
The as-received (La0.6Sr0.4)0.95Co0.2Fe0.8O3 (denoted as LSCF-6428) powders (FuelCellMaterials) were shaped into several pellets with thickness and diameter of around 1 and 13 mm, respectively. Afterward, the pellets were pre-sintered in the ambient air at 1,200 °C for 2h to facilitate handling and remove the binders. Later, the pre-sintered pellets were heat-treated at 800 °C, 900 °C, and 1,000 °C under SO2-containing dry air (10ppm SO2 balanced with dry air, Airgas) or dry Argon (10ppm SO2 balanced with Argon, Airgas) in the tube furnace (OTF-1500X-III, MTI Corporation) for 2 days. The furnace was equipped with 3 independent temperature zones, which allowed the above experiments with the same atmospheric but different temperature conditions to be run in the same batch. Finally, the samples were collected after furnace cooling for further characterization.
The crystal structures of all the samples, including the commercial powder, pre-sintered and heat-treated pellets, were examined by X-ray Diffraction with a Cu tube (PANalytical EMPYREAN) in the range of 20-60°. Later, the microstructure and elemental distributions of the heat-treated pellets were investigated by Scanning Electron Microscopy (SEM) (JEOL JSM-7000F) coupled with Energy-dispersive X-ray Spectroscopy (EDS) (Oxford Instrument X-MAXN) to characterize the secondary phases. Transmission electron microscope (TEM) samples, ca. 100 nm thick cross-sections mounted on Cu support grids, were prepared using a Thermo Fisher Helios 460F1 dual-beam focused ion beam (FIB-SEM) using a Ga ion beam and standard FIB lift-out procedures to minimize ion damage/implantation. The electron beam was used to deposit a carbon protective layer on the top surface of each lift-out lamella, and the voltage of the cutting ion beam was progressively lowered as each lift-out lamella was thinned down to ca. 100 nm and cleaned. Each FIB-prepared TEM sample was characterized by TEM imaging, diffraction, and spectroscopy (elemental mapping).
Bright Field TEM images and diffraction patterns were obtained for each FIB-prepared TEM sample using a Thermo Fisher Talos TEM equipped with a CETA-M camera, operating at 200 keV, gun lens = 4,
Scanning-TEM (STEM) images and STEM-EDS elemental maps were obtained for each FIB-prepared TEM sample using a Thermo Fisher Titan Themis TEM operating at 300 keV, gun lens = 3, spot size = 3,
COMPUTATIONAL PROCEDURE
The CALPHAD approach is considered the computational tool in the present study, which is highly dependent on the reliability of the thermodynamic databases that are usually assessed and constructed using existing literature data. With the corresponding database ready, thermodynamic simulations can thus be conducted using the minimization of the Gibbs free energy of the current system in the Thermo-Calc software. So far, the CALPHAD approach has already been considered and utilized extensively and proven to be a significantly effective tool in the field of SOFCs[13-16,20-24].
To perform the simulations in the current LSCF system, the thermodynamic database established by Zhang[25] has been employed in the present work, where detailed thermodynamic parameters can be found. Based on the compound energy formalism, the LSCF perovskite phase is modeled with three sublattices:
Perovskite: (La3+, Sr2+, Va)1(Co2+, Co3+, Co4+, Fe2+, Fe3+, Fe4+, Va)1(O2-, Va)3
where Va stands for Vacancy. There are, in total, 42 end-members in this perovskite phase, such as (La3+)1(Fe3+)1(O2-)3, where most of them are not charge neutral. The charge neutrality will be considered and maintained when doing simulations in Thermo-calc. Thus, the Gibbs energy per mole molecule can be described via the CALPHAD formalism:
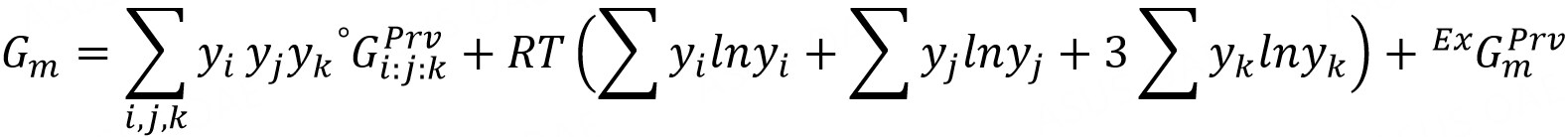
where yi, yj, and yk represent the site fraction of each species in A-, B-, and oxygen-site, respectively.
Halite: (Co2+, Fe2+, Fe3+, Va)1(O2-)1
Spinel: (Co2+, Co3+, Fe2+, Fe3+)1(Co2+, Co3+, Fe2+, Fe3+, Va)2(O2-)4
Similar to the treatment in our previous works[15,17], the Gibbs free energy of the missing sulfur-containing secondary phases La2O2S[26], La2O2SO4[27] and La2(SO4)3[28] were added together with the SSUB5 database, which did not consider these phases in the database. With this, the final obtained LSCF + S database is able to perform thermodynamic simulations of the sulfur poisoning phenomena in LSCF cathode materials. More importantly, to make thermodynamic predictions, a fixed number of thermodynamic conditions are needed to obtain reliable results, which is based on the following concerns. Firstly, the simulation conditions should reflect the current experimental operating conditions as shown above, which include a temperature range from 800 up to 1,000 °C, a P(O2) range from Argon (10-5 atm) up to ambient atmosphere (0.21 atm) and a fixed 10 ppm P(SO2) based on the concentration of the actual gas we received from Airgas. Only in this way can the simulation results be comparable to our experimental observations. In addition, simulations should also mimic the general sintering and operation conditions under the actual or accelerating testing circumstances. Here, P(SO2), ranging from ppb level (~100 ppb in the atmosphere) to ppm level (accelerated testing condition), with P(O2), from ambient air on the surface down to the reducing conditions on TPBs due to the polarization effect[29] is considered as the atmospheric conditions in the simulations. And the temperature is from 600 to 800 °C as the general operating temperature for IT-SOFCs (Intermediate-Solid Oxide Fuel Cells). Finally, the influence of the Sr content is considered in the current simulation to investigate the relationship between the Sr content and sulfur poisoning phenomenon, of which LSCF-8228, LSCF-7328 and LSCF-6428 are chosen. Finally, the sulfur poisoning results of the current LSCF cathode were cross-compared with the most widely-used LSM20 (La0.8Sr0.2MnO3)[17] cathode in our previous literature to better understand the sulfur-tolerant cathode in the future.
RESULTS AND DISCUSSIONS
The CALPHAD simulation approach introduced above was applied to the LSCF-6428 cathode in 10 ppm SO2 conditions, shown in Figure 1 as a function of temperature, where Figure 1A is for dry air and Figure 1 is for Argon. It should be noted that the simulation is based on 0.1 mole molecule of the LSCF-6428 cathode. More importantly, the dry air condition is for the surface of the LSCF cathode (the cathode-gas interface), while the argon condition is for the TPBs (the cathode-electrolyte-gas interface) due to the polarization effect[14]. It can be seen that the sulfur-containing phase, SrSO4, will be thermodynamically stable over the temperature range (600-1,000 °C) in both dry air and argon atmospheric conditions. However, at higher temperatures, the amount of SrSO4 formed drops more severely in the argon condition than in dry air, indicating that the SrSO4 secondary phase is more thermodynamically favorable in higher P(O2), which agrees well with the literature[5,12] and suggests that the formation of the sulfates necessitates the oxidation of SO2. Furthermore, the stability of the sulfate phase decreases with increasing temperature in both dry air and Argon, suggesting that sulfate formation favors lower temperature conditions. In addition, a halite phase, instead of the spinel phase, shows up as the stable Co-Fe oxide phase at high temperatures in Argon [Figure 1B], but not in dry air, which is also intuitive due to the lower valance of the cations in reducing conditions. It is also worth mentioning that in both conditions, the structure of the spinel phase will change with increasing temperature from a Co-rich spinel (spinel #2) to a Fe-rich spinel (spinel #1) due to the miscibility gap of the spinel phase. The stability of two spinel structures is predicted thermodynamically but may be different from the actual experimental observations.
Figure 1. Computational results of the stability of LSCF-6428 under 10 ppm SO2 in terms of temperature in a) dry air and b) argon.
Results from the experimental characterization of LSCF-6428 samples heat-treated in both dry air and Argon were then cross-compared with the simulation results for empirical validation. The XRD spectra of the pre-sintered LSCF-6428 and the heat-treated samples are shown in Figure 2. It is apparent from the figure that some secondary phases do form during sulfur poisoning heat treatment experiments, which mainly correspond to the SrSO4 phase. Meanwhile, a few isolated peaks cannot be identified due to their limited signals, which the simulations suggest could be related to the formation of other oxide phases. More importantly, it can be seen from Figure 2 that the peaks of the SrSO4 phase are more pronounced in the dry air condition and at higher temperatures, indicating that the process for the formation of the SrSO4 phase is governed both thermodynamically and kinetically, as its formation is thermodynamically favored at higher oxygen partial pressure and lower temperature conditions.
Figure 2. XRD spectra of the LSCF-6428 pellet (denoted as LSCF-6428) and heat-treated samples under the corresponding experimental conditions.
To further identify and verify the phases formed during the LSCF-6428 heat-treatment experiments, electron microscopy analysis was used to image and spectroscopically measure the spatial elemental distribution of the samples heat-treated in 10 ppm SO2 containing atmosphere for 2 days, shown in Figure 3, where Figure 3A is for dry air at 1,000 °C, Figure 3B is for dry air at 800 °C, and Figure 3C is for Argon at 1,000 °C, respectively. In 1,000 °C dry air [Figure 3A], there are two different grain morphologies besides the LSCF matrix based on the SEM-EDS mapping results: larger grains correspond to the Sr, S, and O-rich phases, while smaller ones are rich in Co, Fe, and O, which agrees well with the thermodynamic predictions from Figure 1. Similarly, as shown in Figure 3B, two different secondary phases were observed in the 800 °C dry air sample. The large but limited grains were a Co, Fe, and O-rich phase, while the tiny but well-distributed grains along the larger grain boundaries and on the surfaces of the LSCF matrix cannot be characterized through SEM-EDS due to resolution/signal limitations. However, they were still believed to be the SrSO4 phase based on the 1,000 °C dry air SEM-EDS and the predictions, where the SrSO4 phase is thermodynamically more favorable at the lower temperature condition. The SEM results also indicate that the formation of the SrSO4 is a kinetically limited process due to the apparent particle size difference (smaller SrSO4 particles at a lower temperature), while the formation of the spinel phase is more likely governed by thermodynamics as there is no apparent grain size effect between the two temperature conditions. Finally, SEM-EDS results for the sample in Argon at 1,000 °C [Figure 3C] show that the average size of the SrSO4 phase grained formed at 1,000 °C in Argon is between those formed under the conditions of 1,000 °C in dry air and 800 °C in dry air, indicating that the formation of the SrSO4 phase is also governed by P(O2) when kinetics are not constrained by lower temperature. In general, the SEM-EDS results support the predictions from our thermodynamic simulations regarding the formation of the secondary phases of the LSCF cathode under SO2 poisoning conditions. However, further electron microscopy characterization at higher resolution is also needed to draw determining conclusions about the exact identity of the secondary phases formed, especially the crystal structure of the oxides present.
Figure 3. SEM micro-image and the corresponding EDS mapping results of the selected region of heat-treated LSCF-6428 for 2 days under 10 ppm SO2 balanced with (A) dry air at 1,000 °C; (B) dry air at 800 °C, and (C) argon at 1,000 °C.
To characterize the nano-scale features on the surface of the LSCF samples, particularly the S-rich and Co-rich phases, TEM imaging, diffraction, and spectroscopic elemental mapping (STEM-EDS) were performed on lift-out samples prepared by focused ion beam (FIB), shown in Figures 4 and 5 for the sample treated at 800 °C in dry air (same sample as Figure 3B), and in Figures 6 and 7 for the sample treated at 1,000 °C in Argon (same sample as Figure 3C). The regions of interest (ROIs) in Figure 4 and Figure 6 were chosen for containing large Co-rich phase crystals, and the ROIs in Figure 5 and Figure 7 were chosen for containing several tiny S-rich phase crystals. For LCSF at 800 °C in the air [Figure 4], the surface crystal (ROI-4A) only contains Cr and O (and trace Fe), at ca 3:4 ([Co/Fe]:O) atomic ratio by STEM-EDS analysis [Table 1]. Based on TEM diffraction of the same surface crystal [Figure 4B], the measured d-spacing values are consistent with Co3O4, spinel #1, which fits the EDS composition results and agrees well with our modeling predictions. While the small surface crystal (ROI-5A) in Figure 5 only contains Sr, S, and O, at ca 1:1:2.5 atomic ratio by STEM-EDS analysis [Table 2]. Based on the TEM diffraction of the surface crystal, shown in Figure 5B, the crystal’s measured d-spacing values are consistent with SrSO4. Based on the STEM-EDX and TEM diffraction analysis of the LSCF sample treated in dry air at 800 °C, it can be concluded that the Co/O rich phase is primarily composed of the spinel #1 phase ([Co/Fe]3O4), while the small S-rich surface grains are SrSO4, in agreement with the simulation predictions [Figure 1A].
Figure 4. TEM characterization of a Co-rich region of the LSCF surface after treatment in dry air at 800 °C. (A) Dark field STEM image of Co-rich surface nanocrystal. White circle marks the location of the SAD aperture used to acquire a diffraction pattern of the nanocrystal, and the cyan box marks the location of the STEM EDS elemental map (C-I); (B) TEM diffraction pattern of nanocrystal; d-spacing values match those of Co3O4 for d111; (C-H) STEM-EDX elemental maps for Sr, S, La, Fe, Co, and O, displayed as relative atomic composition for each element; (I) combined elemental map for atomic fractions of Sr, S, La, Co, and Fe. ROI-4A and ROI-4B mark the regions where atomic composition was quantified using the STEM-EDS data, shown in Table 1.
Figure 5. TEM characterization of a Sulfur-rich region of the LSCF surface after treatment in dry air at 800 °C. (A) Dark field STEM image of S-rich surface nanocrystals. White circle marks the location of the SAD aperture used to acquire a diffraction pattern of the nanocrystal, and the cyan box marks the location of the STEM EDS elemental map (C-I); (B) TEM diffraction pattern of nanocrystal; d-spacing values match those of SrSO4 for d220 and d303; (C-H) STEM-EDX elemental maps for Sr, S, La, Fe, Co, and O, displayed as relative atomic composition for each element; (I) combined elemental map for atomic fractions of Sr, S, La, Co, and Fe. ROI-5A and ROI-5B mark the regions where atomic composition was quantified using the STEM-EDS data, shown in Table 2.
Figure 6. TEM characterization of a Co-rich region of the LSCF surface after treatment in Argon at 1,000 °C. (A) Dark field STEM image of Co-rich nanocrystal. White circle marks the location of the SAD aperture used to acquire a diffraction pattern of the nanocrystal, and the cyan box marks the location of the STEM EDS elemental map (C-I); (B) TEM diffraction pattern of nanocrystal; d-spacing values match those of Co1O1 for d022 and d133; (C-H) STEM-EDX elemental maps for Sr, S, La, Fe, Co, and O, displayed as relative atomic composition for each element; (I) combined elemental map for atomic fractions of Sr, S, La, Co, and Fe. ROI-6A and ROI-6B mark regions where atomic composition was quantified using the STEM-EDS data, shown in Table 3.
Figure 7. TEM characterization of a Sulfur-rich region of the LSCF surface after treatment in Argon at 1,000 °C. (A) Dark field STEM image of S-rich surface nanocrystals. White circle marks the location of the SAD aperture used to acquire a diffraction pattern of the nanocrystal, and the cyan box marks the location of the STEM EDS elemental map (C-I); (B) TEM diffraction pattern of nanocrystal; d-spacing values match those of SrSO4 for d102, d210, and d401; (C-H) STEM-EDX elemental maps for Sr, S, La, Fe, Co, and O, displayed as relative atomic composition for each element; (I) combined elemental map for atomic fractions of Sr, S, La, Co, and Fe. ROI-7A, ROI-7B, and ROI-7C mark the regions where atomic composition was quantified using the STEM-EDS data, shown in Table 4.
Quantification results from STEM-EDX analysis of LSCF treated at 800 °C in dry air from the ROIs in Figure 4
| ROI-4A (atomic %) | (% error) | ROI-4B (atomic %) | (% error) | |
| O | 58.01 | 0.17 | 60.30 | 0.03 |
| S | 0.04 | 13.72 | 0.05 | 13.49 |
| Fe | 3.91 | 0.11 | 17.25 | 0.12 |
| Co | 37.95 | 0.04 | 3.71 | 0.30 |
| Sr | 0.04 | 8.22 | 7.96 | 0.19 |
| La | 0.05 | 2.51 | 10.72 | 0.10 |
Quantification results from STEM-EDX analysis of LSCF treated at 800 °C in dry air from the ROIs in Figure 5
| ROI-5A (atomic %) | (% error) | ROI-5B (atomic %) | (% error) | |
| O | 50.31 | 1.49 | 46.14 | 0.34 |
| S | 22.98 | 1.15 | 0.04 | 10.15 |
| Fe | 0.88 | 7.58 | 23.17 | 0.05 |
| Co | 0.20 | 26.91 | 5.12 | 0.09 |
| Sr | 24.96 | 1.12 | 11.17 | 0.16 |
| La | 0.67 | 5.38 | 14.35 | 0.09 |
For the LSCF sample treated in Argon at 1,000 °C, the near-surface crystal (ROI-6A) in Figure 6 only contains Cr and O (and trace Fe), at ca 1:1 ([Co/Fe]:O) atomic ratio by STEM-EDS analysis [Table 3]. Based on TEM diffraction of the same crystal [Figure 6B], the measured d-spacing values are consistent with
Quantification results from STEM-EDX analysis of LSCF treated at 1,000 °C in Argon from the ROIs in Figure 6
| ROI-6A (atomic %) | (% error) | ROI-6B (atomic %) | (% error) | |
| O | 51.36 | 3.28 | 60.31 | 1.71 |
| S | 0.12 | 0.02 | 0.02 | 0.00 |
| Fe | 0.27 | 0.04 | 17.35 | 1.93 |
| Co | 48.06 | 3.32 | 3.45 | 0.45 |
| Sr | 0.04 | 0.01 | 7.72 | 0.94 |
| La | 0.15 | 0.02 | 11.15 | 1.10 |
Quantification results from STEM-EDX analysis of LSCF treated at 1,000 °C in Argon from the ROIs in Figure 7
| ROI-7A (atomic %) | (% error) | ROI-7B (atomic %) | (% error) | ROI-7c (atomic %) | (% error) | |
| O | 59.57 | 0.29 | 66.28 | 0.21 | 51.14 | 0.43 |
| S | 0.04 | 12.67 | 16.13 | 0.10 | 0.11 | 18.78 |
| Fe | 19.88 | 0.08 | 0.18 | 2.97 | 3.91 | 0.61 |
| Co | 1.90 | 0.29 | 0.06 | 7.56 | 41.41 | 0.10 |
| Sr | 4.70 | 0.37 | 17.29 | 0.18 | 0.55 | 5.78 |
| La | 13.90 | 0.04 | 0.06 | 3.85 | 2.88 | 0.44 |
So far, the combined approach of CALPHAD simulation and XRD, SEM, and TEM characterization has provided a deeper insight into the sulfur poisoning phenomena of LSCF cathode material in the presence of SO2 under various treatment temperatures and gas environments. Furthermore, the good agreement between the thermodynamic predictions and the experimental observations for LSCF highlights the utility of computational tools to study materials degradation phenomena in SOFC systems. Thus, further simulations were conducted to address more specific details, such as the influence of the LSCF cathode’s Sr-composition on its sulfur poisoning, the validity of the SO2 accelerated testing method for LSCF cathodes, and the long-term degradation mechanism(s) during the sulfur poisoning of LSCF cathodes.
To test the effect of Sr concentration on the sulfur poisoning phenomena of LSCF cathode materials, three commonly used compositions, LSCF-6428, LSCF-7328, and LSCF-8228, listed from highest to lowest Sr content, respectively, were chosen as candidates for investigation using simulations. The thresholds for the formation of SrSO4 with these three candidates are plotted in Figure 8 as a function of P(SO2) and temperature, where Figure 8A is for treatment in dry air while Figure 8B is for treatment in Argon. The SrSO4 stable region infers that the SrSO4 phase is thermodynamically stable under given conditions, while the SrSO4 free region suggests the chemical instability of the SrSO4 phase. Consequently, the optimal operating condition for each composition would be in the SrSO4 free region to prevent the formation of the detrimental SrSO4 secondary phase. The threshold diagrams show that the SrSO4 phase is thermodynamically more favorable at lower temperatures, higher P(SO2), higher P(O2), and for higher Sr composition electrodes. This agrees well with our experimental observations for LSCF-6428, shown in Figure 3. It also matches well with prior studies by Wang et al.[7,11], where the formation of SrSO4 was observed in the presence of 1, 10, and 100 ppm SO2 but not in 0.1ppm SO2, and where sulfur poisoning was more pronounced at lower temperatures. In addition, Wang et al.[9] investigated the effect of Sr concentration on the sulfur poisoning of LSCF by mainly using LSCF-6428 and LSCF-8228. Their results showed that the thickness of SrSO4 formed in LSCF-6428 was almost four times thicker than that in LSCF-8228, which has the lower relative Sr composition of the two LSCF materials.
Figure 8. SrSO4 threshold diagrams in terms of the P(SO2) and temperatures for LSCF-6428 (red), LSCF-7328 (blue) and LSCF-8228 (green) under (A) dry air and (B) Argon.
Under typical P(SO2) atmospheric conditions, the only sulfur-containing secondary phase observed to form in LSCF cathodes is SrSO4[6-12,30,31]. It is widely accepted that the formation of SrSO4 is responsible for the long-term degradation of the LSCF cathode materials[5]. Based on the simulation results in Figure 8, the threshold for SrSO4 formation in LSCF-6428 at 800 °C is around 10-10 atm in air and 10-8 atm under reducing conditions. The typical (SO2) content in the ambient air is at the level of 10-7 atm, indicating that ambient SO2 in the air is sufficient to drive long-term degradation of the LSCF-6428 cathode. Therefore, sulfur poisoning results from accelerated testing, which is done at elevated SO2 levels, should mimic the natural sulfur poisoning that occurs during cathode operation at standard testing conditions with atmospheric SO2, as in both cases, there is excess SO2. Thus, accelerated testing will not only reduce the total time for running poisoning experiments but is also capable of reproducing the sulfur poisoning phenomena that occur in the presence of ambient SO2 during standard testing conditions. In systems where the CALPHAD simulations predict that ambient SO2 will be insufficient to drive SrSO4 stability, the use of accelerated testing can potentially generate erroneous experimental results regarding SrSO4 poisoning related to standard testing conditions[17]. Using this simulation approach, the reliability of accelerated testing can be scrutinized for different cathode materials under different environmental conditions.
The threshold diagrams of the LSCF cathode suggest that LSCF has poor sulfur tolerance under operating conditions, and finding an alternative sulfur-resistant cathode material thus remains of great importance. To better understand the sulfur poisoning of LSCF cathode in the context of other potential cathode materials, CALPHAD threshold diagrams were created for LSM20[17] and compared to LSCF-6428 in terms of P(SO2) and temperature under air and argon atmospheric conditions, as shown in Figure 9. Simulations predict that LSM20 has superior sulfur resistance to LSCF-6428 both in the air and argon conditions. Specifically, the threshold of SrSO4 stability for LSM20 is around 10-7 atm at 800 °C in air, while it is 10-10 atm for LSCF-6428. Unlike LSCF-6428, LSM20 could be free from sulfur poisoning at ambient SO2. This sulfur resistance behavior was also experimentally observed by Liu et al.[6], where LSM20 was found to be free from sulfur poisoning even following exposure to 1ppm of SO2 for 1,000 h, and the degradation rate at 20 ppm SO2 for LSCF-6428 was 8 times higher than that of LSM20. These computational and experimental results confirmed that the LSCF cathode is a poor sulfur-tolerant material in comparison with LSM. One potential solution for the poor sulfur resistance of LSCF is to place an additional sulfur filter before contacting the LSCF cathode to purge the sulfur concentration to below the threshold concentration. Another solution would be to utilize a core-shell structure[32], having LSM as the sulfur-protecting shell and LSCF cathode as the core.
CONCLUSIONS
In this work, we have evaluated the sulfur poisoning behavior of LSCF cathode materials in the presence of SO2 with a combined computational and experimental approach and tested the reliability of the simulation method for predicting sulfur poisoning behavior under different environmental conditions. The results from CALPHAD simulations regarding the stability of the secondary phases were validated using the experimental characterization, XRD, SEM, and TEM, of the same cathode materials following operations at the simulation conditions. Further simulation predictions were made to better understand the effects that other determining factors, such as temperature, P(O2), P(SO2), and cathode Sr-composition, have on the formation of SrSO4 and the overall sulfur poisoning behavior. We find that the formation of SrSO4 on LSCF cathodes (sulfur poisoning) is more thermodynamically favorable at lower temperatures, higher P(SO2), higher P(O2), and higher Sr composition. Finally, comparisons were made between LSCF-6428 and LSM20 cathode materials, which confirmed that LSCF-6428 has a much lower sulfur tolerance than LSM20, in agreement with recent literature. The CALPHAD simulation approach used here can be extended to other potential cathode systems to theoretically test their tolerance to poisoning at different environmental conditions, which has the potential to accelerate the experimental development of novel poisoning-resistant cathode materials.
DECLARATIONS
Authors’ ContributionsMade substantial contributions to conception and design of this research, data analysis, writing the draft and editing: Wang R
Performed data analysis, figures preparation, writing-review and editing: Parent LR
Made substantial contributions to conception and design of this research, data analysis, funding acquisition, writing and editing: Zhong Y
Availability of data and materialsNot Applicable.
Financial support and sponsorshipThis material is based upon work supported by the Department of Energy under Award Number (DE-FE0031652). The authors would like to thank the support and guidance from the DOE National Energy Technology Laboratory program managers, Jason Montgomery, Venkat Venkataraman and Andrew O’Connell.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2023.
REFERENCES
1. Skinner SJ. Recent advances in Perovskite-type materials for solid oxide fuel cell cathodes. Int J Inorg Mater 2001;3:113-21.
2. Stambouli A, Traversa E. Solid oxide fuel cells (SOFCs): a review of an environmentally clean and efficient source of energy. Renew Sustain Energy Rev 2002;6:433-55.
3. Schuler AJ, Wuillemin Z, Hessler-wyser A, Van Herle J. Sulfur as pollutant species on the cathode side of a SOFC system. ECS Trans 2009;25:2845-52.
4. Xiong Y, Yamaji K, Horita T, et al. Sulfur poisoning of SOFC cathodes. J Electrochem Soc 2009;156:B588-92.
5. Wang F, Kishimoto H, Ishiyama T, et al. A review of sulfur poisoning of solid oxide fuel cell cathode materials for solid oxide fuel cells. J Power Sources 2020;478:228763.
6. Liu RR, Taniguchi S, Shiratori Y, Ito K, Sasaki K. Influence of SO2 on the long-term durability of SOFC cathodes. ECS Trans 2011;35:2255-60.
7. Wang F, Yamaji K, Cho D, et al. Sulfur poisoning on La0.6Sr0.4Co0.2Fe0.8O3 cathode for SOFCs. J Electrochem Soc 2011;158:B1391-7.
8. Wang DJ, Jing L. Effect of SO2 on performance of solid oxide fuel cell cathodes. Available from: https://www.semanticscholar.org/paper/Effect-of-SO2-on-Performance-of-Solid-Oxide-Fuel-De-jun-Jing/82c3339827f4bedfe62b0270ecab17a1aac3d0ca#citing-papers [Last accessed on 9 Mar 2023].
9. Wang F, Yamaji K, Cho D, et al. Effect of strontium concentration on sulfur poisoning of LSCF cathodes. Solid State Ionics 2012;225:157-60.
10. Wang F, Yamaji K, Cho D, et al. Evaluation of sulfur dioxide poisoning for LSCF cathodes. Fuel Cells 2013;13:520-5.
11. Wang C, Jiang SP. Mechanism of SO2 poisoning on the electrochemical activity of LSCF and LSM electrodes. ECS Trans 2015;68:1023-9.
12. Wang F, Kishimoto H, Develos-bagarinao K, Yamaji K, Horita T, Yokokawa H. Interrelation between sulfur poisoning and performance degradation of LSCF cathode for SOFCs. J Electrochem Soc 2016;163:F899-904.
13. Darvish S, Asadikiya M, Hu B, Singh P, Zhong Y. Thermodynamic prediction of the effect of CO2 to the stability of (La0.8Sr0.2)0.98MnO3±δ system. Int J Hydrogen Energy 2016;41:10239-48.
14. Darvish S, Gopalan S, Zhong Y. Thermodynamic stability maps for the La0.6Sr0.4Co0.2Fe0.8O3±δ-CO2-O2 system for application in solid oxide fuel cells. J Power Sources 2016;336:351-9.
15. Darvish S, Wang CC, Jiang SP, Zhong Y. Thermodynamic stability mapping and electrochemical study of La1-xSrxCo0.2Fe0.8O3±δ (x = 0.2-0.4) as a cathode of solid oxide fuel cells in the presence of SO2. Electrochim Acta 2018;287:68-77.
16. Darvish S, Hu B, Singh P, Zhong Y. Thermodynamic and experimental evaluation of La1-xSrxMnO3±δ cathode in presence of Cr-containing humidified air. JOM 2019;71:3814-24.
17. Wang R, Parent LR, Gopalan S, Zhong Y. Experimental and computational investigations on the SO2 poisoning of (La0.8Sr0.2)0.95MnO3 cathode materials. Adv Powder Mater 2023;2:100062.
18. Walker E, Ammal SC, Suthirakun S, Chen F, Terejanu GA, Heyden A. Mechanism of sulfur poisoning of Sr2Fe1.5Mo0.5O6-δ perovskite anode under solid oxide fuel cell conditions. J Phys Chem C 2014;118:23545-52.
19. Su M, Huan D, Hu X, Zhu K, Peng R, Xia C. Understanding the favorable CO2 tolerance of Ca-doped LaFeO3 perovskite cathode for solid oxide fuel cells. J Power Sources 2022;521:230907.
20. Ta N, Chen M, Zhang L, et al. Numerical simulation of kinetic demixing and decomposition in a LaCoO3-δ oxygen membrane under an oxygen potential gradient. J Membr Sci 2018;548:526-39.
21. Wang CC, Darvish S, Chen K, et al. Combined Cr and S poisoning of La0.8Sr0.2MnO3-δ (LSM) cathode of solid oxide fuel cells. Electrochim Acta 2019;312:202-12.
22. Xu H, Cheng K, Chen M, Zhang L, Brodersen K, Du Y. Interdiffusion between gadolinia doped ceria and yttria stabilized zirconia in solid oxide fuel cells: experimental investigation and kinetic modeling. J Power Sources 2019;441:227152.
23. Sabarou H, Wang R, Zhong Y. The origin of the phase separation in (La0.8Sr0.2)0.95(CrxFe1-x)O3±δ perovskites for oxygen transport membranes applications. Solid State Ion 2020;349:115293.
24. Cheng K, Xu H, Zhang L, et al. Computational engineering of the oxygen electrode-electrolyte interface in solid oxide fuel cells. NPJ Comput Mater 2021;7:119.
25. Zhang W, Barfod R. Investigation of degradation mechanisms of LSCF based SOFC cathodes-by CALPHAD modeling and experiments. Available from: https://orbit.dtu.dk/en/publications/investigation-of-degradation-mechanisms-of-lscf-based-sofc-cathod [Last accessed on 9 Mar 2023].
26. Kumar RV, Kay DAR. Thermodynamics of the Ca-S-O, Mg-S-O, and La-S-O systems at high temperatures. Metall Trans B 1985;16B:287-94.
27. Dwivedi RK, Kay DAR. Thermodynamics of the oxidation of rare earth oxysulfides at high temperatures. Metall Trans B 1984;15B:523-8.
28. Kellogg HH. A critical review of sulfation equilibira. Available from: https://archive.org/details/sim_american-institute-of-mining-metallurgical-petroleum_1964-12_230_7/page/n131/mode/2up [Last accessed on 9 Mar 2023].
29. Levy C, Zhong Y, Morel C, Marlin S. Thermodynamic stabilities of La2Zr2O7 and SrZrO3 in SOFC and their relationship with LSM synthesis processes. J Electrochem Soc 2010;157:B1597-601.
30. Gao J, Li L, Yin Z, Zhang J, Lu S, Tan X. Poisoning effect of SO2 on the oxygen permeation behavior of La0.6Sr0.4Co0.2Fe0.8O3-δ perovskite hollow fiber membranes. J Membr Sci 2014;455:341-8.
31. Liu RR, Wang DJ, Jing L. Effect of SO2 on the performance of LSCF cathode. Adv Mater Res 2014;902:41-4.
32. Gopalan S, Levitas B. Core-shell heterostructures as functional materials for solid oxide fuel cell (SOFC) electrodes. Available from: https://www.osti.gov/biblio/1872369/ [Last accessed on 9 Mar 2023].
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data



























Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].