Ecological and health risks of polycyclic aromatic hydrocarbons in the sediment core of Phayao Lake, Thailand
Abstract
Polycyclic aromatic hydrocarbons (PAHs) in sediment cores from Phayao Lake were investigated in terms of their concentration, distribution, and potential effects on the environment and human health. The concentration of
Keywords
INTRODUCTION
Polycyclic aromatic hydrocarbons (PAHs) are persistent organic pollutants (POPs) with two or more benzene rings in their chemical structure[1-3]. The main origin of PAHs is the incomplete combustion of organic compounds and fossil fuels through human activities, such as transportation and industrial facilities[4-7]. The majority of PAHs are insoluble and long-lasting in the environment. Both direct and indirect exposure of living creatures to PAHs results in accumulation and transmission. According to previous studies, some of the increased cases of cancer may be linked to environmental exposure and/or chemical absorption of PAHs through the food chain[3,8,9]. PAHs are hazardous to health and the environment[10], potentially causing cancer (carcinogen)[11,12] and inducing mutations (mutagen) or embryo defects. Moreover, PAHs have the potential to be teratogenic in both humans and animals[13].
PAHs have the potential to spread and accumulate in lakes and rivers[14,15]. Wind and rivers carry sediments and other particles to lakes. PAHs adsorbed on small solid particles (aerosols) in dust in the atmosphere move from their primary sources and can either fall into lakes or combine with precipitation[16,17]. Water buffers sediments and particles that have settled to the bottom of water bodies from wind and other physical forces. As a result, sediments are maintained in lakes on a very fine scale. PAHs can also leach from the ground, such as wastewater from oil refinery plants, lubricant spills from industrial plants, water from agricultural fields, and water from communities, and then be carried into lakes through adsorption to soil particles[18-21]. Because PAHs are discharged from industrial, municipal, and natural surface run-offs around lakes, lake sediments serve as a significant reservoir of pollutants. The degree of PAH contamination and the main PAH source in lake sediments have been evaluated. In particular, nations that are more developed are more vulnerable to the impacts of PAH pollution arising from different industrial activities. Yuan et al. obtained sediment samples from Yangzong Lake, China at a depth of 20 cm for a study on the genesis and history of PAHs[22]. They found that between 1950 and 1990, there were 200.7 to 1913.7 ng g-1 of PAHs, the majority of which were produced by the burning of petroleum.
The bioactivity of PAHs has been investigated. For example, benzo[a]pyrene (B[a]P) is physiologically activated by enzymes to produce carcinogenic and mutagenic epoxides[23]. Sayer discovered that ellagic acid, which is abundant in green plants, degrades the diol epoxide form of B[a]P, reducing its potential to induce cancer or mutations[24]. PAHs have been identified in the following environments: lake water[25], marine air, soil/sediment, snow, mosses, lichen, krill, penguin, and skua blood. An exhaustive investigation into the presence of POPs in biotic and abiotic components has been conducted on national and international scales[26]. PAHs in sediment may be volatilized into the atmosphere and adsorbed by aquatic animals as well as aquatic crops through roots and leaves. The accumulation of PAHs in humans and other species is therefore possible if food chains are involved[27]. As a result, monitoring the concentration of PAHs in sediments is critical to understand the course of PAHs through the ecosystem.
Phayao Lake, the largest freshwater lake in northern Thailand, is a significant water supply for human consumption. Furthermore, Phayao Lake supports a large number of aquatic species. Hence, lake sediments commonly contain fossils, including fossils of microscopic creatures that live in the water. The presence or absence of these fossils can reveal information about the water and lake, e.g., lake depth, water temperature, and whether the water is salty or fresh. The sediments in cores, such as sand and clay, can reveal when the lake depth decreases (due to a drier environment) or increases (during times of a wetter climate). All the information assists in determining the settings and climates in which ancestors lived.
Because of the ecological importance and high socioeconomic value of Phayao Lake, investigations of the causes, trends, concentrations, distributions, and toxicity of pollutants in the sediment are required to evaluate the effects of oil exploration, transportation, waste disposal, industrial operations, and pollution in the area. Previous investigations of dated sediment cores from Phayao Lake (covering the past ~150 years) revealed that the greatest flow and composition changes of PAHs follow local industrialization. The concentrations of Σ23PAHs, namely all parent and alkyl PAHs (excluding perylene, which has a predominantly in situ biogenic diagenesis origin), low molecular weight (LMW, 2-3 ring)-PAHs, and high molecular weight (HMW, 4 ring)-PAHs, were low and fluctuated minimally until ~1980s. PAH concentrations and fluxes grew until ~1996 and then declined but remained higher than those before ~1980, reflecting the emission history of the Mae Moh power plant[17]. The atmosphere in Chiang-Mai province, located in northern Thailand and close to Phayao Lake, was analyzed during 2017-2018, revealing the presence of 19 PAHs: acenaphthene (Ace), fluorene (Fl), phenanthrene (Phe), anthracene (An), fluoranthene (Fluo), pyrene (Pyr), benzo[a]anthracene (B[a]A), chrysene (Chry), benzo[b]fluoranthene (B[b]F), benzo[k]fluoranthene (B[k]F), benzo[a]fluoranthene (B[a]F), benzo[e]pyrene (B[e]P), benzo[a]pyrene (B[a]P), perylene (Per), indeno[1,2,3-cd]pyrene (Ind), benzo[g,h,i]perylene (B[g,h,i]P), dibenz[a,h]anthracene (D[a,h]A), coronene (Cor), and dibenzo[a,e]pyrene (D[a,e]P). According to this study, during 2017-2018, the concentration of total PAHs was 2.361 ± 2.154 mg m-3, and the important sources of PAHs in northern Thailand were vehicular exhaust, biomass burning (BB), and diesel emissions[28]. Therefore, there is a scarcity of health risk assessments of Phayao Lake sediments. Han et al. determined the concentrations and fluxes of BC, char, soot, and PAHs in aged sediment cores obtained along the lake[17]. Nevertheless, no research has been conducted to assess the ecological and health risks of PAHs. Therefore, in this study, 81 cm long sediment cores from Phayao Lake were analyzed to determine the vertical trends, distributions, profiles, and toxicity equivalent concentrations (TEQs) of PAHs. This is essential for risk evaluations and hazardous action plans used to protect human health and the environment.
EXPERIMENTAL
Sampling and methods
Sampling site
Phayao Lake is the largest lake in northern Thailand and the fourth-largest lake in Thailand. It is centrally located in the city of Phayao, with mountains in the background. It is formed by water flowing from 18 local streams, with an average annual water volume of 29.40 million m3. It covers 20.5 km2 of land. Additionally, it serves as a spawning habitat for many different fish species. Phayao Lake’s surrounding landscape makes it a natural attraction to visitors[29]. Phayao Lake is exposed to a variety of local and regional pollution as well as transboundary haze pollution, including urban run-off, industrial waste, vehicular emissions, and biomass emissions, such as the cooking of food and burning of agricultural waste[17]. A gravity corer (Uwitec, Austria) was used to obtain sediment cores from the southern part of the lake (N 19°9.8’,
Chemical analysis of PAHs
Extraction and analysis of the sediment core (PY) followed the method by Han et al.[17]. Briefly, 2 g of sediment was spiked with internal standards (mixture of 7 deuterated PAHs and 2 deuterated OPAHs) and then extracted using an ultrasonic technique with organic solvents. The extracts were then cleaned and fractionated using column chromatography. For identification and quantification of PAHs, the final extract was fed into a gas chromatography coupled to a mass spectrometer (GC/MS, Agilent, Santa Clara, CA, USA). On the dry sediment mass basis, the concentration of each target component was calculated. Quality assurance/quality control (QA/QC) analyses followed the method by Han et al.[17]. Briefly, internal standards were used to quantify the component concentrations in the samples, which were then adjusted by subtracting the mean blank values. Spike and recovery studies were used to comprehensively evaluate the analytical method’s accuracy and precision.
Ecotoxicological risk and health risk assessment
Toxicity equivalent concentration
In this study, all samples were subjected to health risk assessments for both children and adults using the Toxicity equivalent concentration (TEQ) formula shown in Equation 1. This formula is used to calculate the health risk from exposure to the carcinogen in various substances, which has been compared with the health risk of exposure to the toxin B[a]P[30].

where Ci is the concentration of PAH and TEFs is the toxic equivalency factor used to compare the toxicity of each PAH relative to B[a]P in accordance with the United States Environmental Protection Agency (USEPA) standards[30].
*TEFs: B[a]P and D[a,h]A was 1; B[a]A, B[b]F, B[k]F, and Ind was 0.1; An, Chry, and B[g,h,i]P was 0.01; and the last group containing Phe, Fluo, and Pyr was 0.001.
Ecological risk assessment of PAHs in sediments
In the environment, PAHs can be found in various ecosystems, particularly in sediments and water[30,31]. Therefore, it is very important to monitor and assess PAHs in the ecosystem, especially in sediments, because they are major sources of many pollutants released into lakes and water resources. The risk of PAHs in the sediment escaping to the environment was evaluated using a risk quotient (RQ). The unfavorable impacts of environmental pollution on ecosystems can be evaluated by measuring the ratio of the concentration of specific pollutants to the corresponding concentrations of pollutants in the medium, as shown in Equation 2[27,30]. Hence, risk assessment using the RQ formula was first applied to evaluate the risk of the sediment to the ecosystem.

where CPAHs and CQV are the concentration of different PAHs and the corresponding level of PAHs in the sediments, respectively.
Second, the negligible (NC) and maximum (MPC) concentrations of PAHs in sediments were calculated using Equations 3 and 4[32]. In this study, we used RQNCs and RQMPCs for the calculations, as shown below:

where CQV(NCs) is the quality value of the NCs for PAHs and CQV(MPCs) is the quality value of the MPCs for PAHs.
Finally, an ecological risk of only nine PAHs, namely naphthalene, anthracene, phenanthrene, fluoranthene, benzo[a]anthracene, chrysene, benzo[k]fluoranthene, benzo[a]pyrene, and benzo[g,h,i]perylene, was evaluated in accordance with USEPA standards for each PAH.
Additionally, the technique by Cao et al. was used to determine the ecological risk of PAHs in the ecosystem[33]. The concern that the ecological risk of specific PAHs is disregarded can be addressed with this method. The following definitions apply to RQΣPAHs, RQΣPAHs(NCs), and RQΣPAHs(MPCs):

In this study, the ecological risk assessment of certain PAHs was conducted by referring to studies conducted by Cao et al., Wang et al., and Sun et al.[33,27,31]. To determine the RQPAHs(NCs) and RQPAHs(MPCs) values of individual PAH, the values of RQ(NCs) and RQ(MPCs) that were not less than 1 were added. According to this metric, each PAH may not be of significant concern if RQ(NCs) is less than 1. There is a high chance of contamination from a single PAH if RQ(MPCs) is greater than 1. If RQ(NCs) is larger than 1 and RQ(MPCs) is less than 1, contamination by specific PAHs poses a moderate risk.
Health risk assessment
In accordance with the Human Health Evaluation Manual[34], the incremental lifetime cancer risk (ILCR) was used to quantify the health risk from exposure to environmental PAHs[35]. The population in this study was separated into two groups according to the demarcation between the ages of children and adults. The models used to analyze the ILCRs account for the three major exposure paths: ingestion, skin contact, and inhalation[36]. PAHs are related to the food chain, such as when humans cook aquatic animals for human consumption. Additionally, people in the area can breathe in the pollutants released from the sediment into the air. ILCR was calculated using Equations 8, 9, and 10:
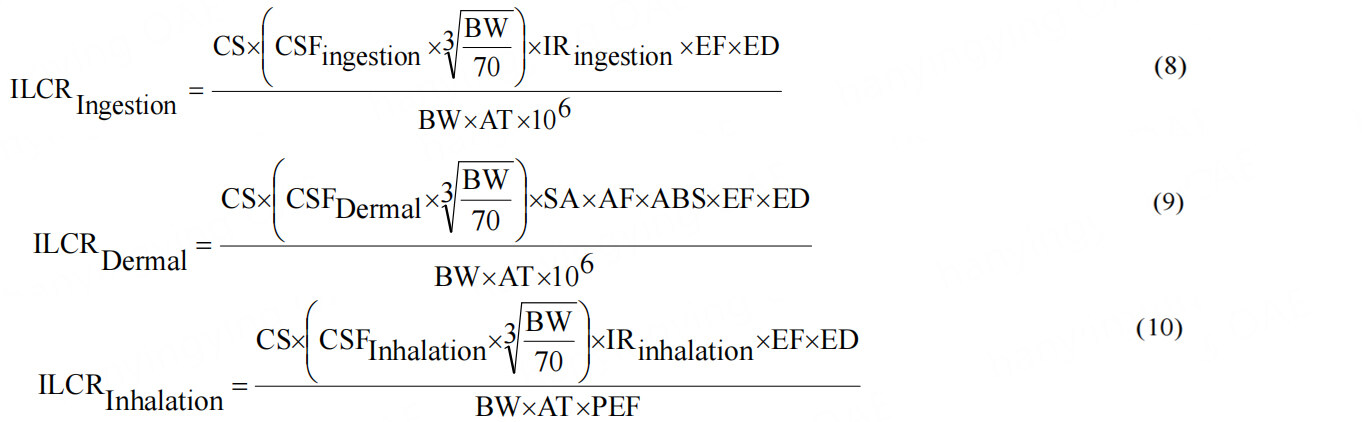
where CS represents the converted PAH concentration based on TEQ measurements, ABS is the dermal adsorption fraction (0.13 for children and adult)[37], AF is the dermal adherence factor (0.2 mg cm-2 h-1 for children and 0.07 mg cm-2 h-1 for adult)[37], AT is the average life span (25,550 days for children and adult)[38], BW is the body weight (15 kg for children and 70 for adult)[34], CSFDermal is the dermal cancer slope factor of B[a]P (25 kg d mg-1 for children and adult)[38], CSFIngestion is the ingestion cancer slope factor of
RESULTS AND DISCUSSION
The concentration and character of PAHs in the sediment core
The concentrations of 16 different PAHs identified in the sediment core from Phayao Lake, namely Nap, Acy, Ace, Flu, Phe, An, Fluo, Pyr, B[a]A, Chry, B[b]F, B[k]F, B[a]P, B[g,h,i]P, Ind, and D[ah]A, and
Figure 2. (A) Vertical profiles of the concentrations (ng g-1) of different Polycyclic aromatic hydrocarbons (PAH) congeners and
We also measured the concentrations of 16 PAHs by depth (see Supplementary Table 1), revealing that the concentration of Σ16PAHs was highest at a depth of 34 cm (1251.10 ± 30.55 ng g-1). The PAH levels in the sediment from Phayao Lake at a depth of 34 cm were relatively high compared with those in other samples from other nations (high levels: 1000 to 5000 ng g-1)[41]. The depth distribution of PAHs in the sediment core from Phayao Lake showed that LMW-PAHs were dominant at every depth (see Figure 3), indicating that the main source of PAHs is the incomplete combustion of organic sources, including pyrogenic, petrogenic, and biological matter, and thus PAHs are discharged into Phayao Lake through pyrogenic processes[40].
Figure 3. Depth distribution of Polycyclic aromatic hydrocarbons (PAH) in the sediment core from Phayao Lake, Thailand.
The amounts of various PAHs contained in sediment are categorized as follows: low levels: 0 to 100 ng g-1, moderate levels: 100 to 1000 ng g-1, high levels: 1000 to 5000 ng g-1, and extremely high levels:
Comparison of polycyclic aromatic hydrocarbons (PAH) concentrations in sediments from different areas in the world
| Sampling sites | Nature of sample | Number of PAHs | Concentration (ng g-1) | References |
| Phayao Lake, Thailand | Core sediment | 16 | 77.65-1251.10 | This study |
| Samsun Coast, Turkey | Sediment (~5 cm) | 16 | 71.64-1186.87 | [42] |
| Han River, China | Sediment | 16 | 137.1-1478.4 | [43] |
| Black Sea | Sediment | 16 | 28.47-444.36 | [44] |
| Gorgan Bay, Iran | Surface sediments | 16 | 13.70-23.68 | [45] |
| Escravos River Basin, Nigeria | Surface sediments | 16 | 750-213,000 | [46] |
| Pearl River Estuary, China | Surface sediment | 16 | 73.68-933.25 | [47] |
| Peninsular, Malaysia | Surface sediments (top 5 cm layer) | 16 | 57.7-19,300 | [48] |
| Burrard Inlet, Canada | Sediment | 15 | 20-6840 | [49] |
| Bering Sea | Surface sediments | 16 | 49.84-65.38 | [50] |
| Canadian Basin | Surface sediments | 16 | 27.66-167.48 | [50] |
| Chukchi Sea | Surface sediments | 16 | 52.40-91.25 | [50] |
| Nzhelele River, South Africa | Surface sediments | 16 | 206-13,710 | [51] |
Ecological Risks of PAHs in the sediment
PAHs that accumulate in sediments might be used by benthic creatures, such as phytoplanktons and zooplanktons, which would then enter the food chain. Therefore, the aquatic ecology may be at risk from PAHs found in sediment samples. An effective approach for assessing the ecological risk of PAHs to organisms and the ecosystem is an ecological risk assessment[52]. The risk quotient (RQ) was developed by Kalf et al. to measure the risk of organic compounds[32]. The proposed approach was used to evaluate the ecological risk posed by samples of contaminated silt collected from Phayao Lake.
According to the contamination levels used by Maliszewska-Kordybach[53] to classify soil pollution, soil is considered not contaminated at levels below 200 ng g-1, slightly contaminated at levels between 200 and
Toxic equivalent quantities (TEQs) of polycyclic aromatic hydrocarbons (PAHs) in sediment cores collected from Phayao Lake, Thailand
| PAHs | TEFs (US-EPA) | Mean | Stdev | Range |
| Naphthalene | 0.001 | 0.12 | 0.05 | 0.02-0.20 |
| Acenaphthylene | 0.001 | 0.02 | 0.01 | ND-0.04 |
| Acenaphthene | 0.001 | 0.02 | 0.02 | ND-0.08 |
| Fluorene | 0.001 | 0.04 | 0.03 | 0.01-0.11 |
| Phenanthrene | 0.001 | 0.10 | 0.06 | 0.02-0.23 |
| Anthracene | 0.01 | 0.08 | 0.13 | ND-0.39 |
| Fluoranthene | 0.001 | 0.04 | 0.03 | ND-0.12 |
| Pyrene | 0.001 | 0.02 | 0.02 | ND-0.08 |
| Benz[a]anthracene | 0.1 | 1.20 | 1.29 | 0.18-4.38 |
| Chrysene | 0.001 | 0.02 | 0.03 | ND-0.09 |
| Benzo[b]fluoranthene | 0.1 | 1.98 | 3.07 | 0.09-11.99 |
| Benzo[k]fluoranthene | 0.1 | 2.12 | 3.64 | 0.10-10.94 |
| Benzo[a]pyrene | 1 | 16.93 | 25.49 | ND-81.92 |
| Indeno[1,2,3-cd]pyrene | 0.1 | 1.56 | 1.78 | 0.11-5.90 |
| Dibenz[a,h]anthracene | 1 | 13.89 | 15.37 | 1.49-50.47 |
| Benzo[g,h,i]perylene | 0.01 | 0.15 | 0.21 | 0.01-0.77 |
| Σ16PAHs | 38.29 | 46.69 | 2.67-155.09 | |
| Σ9PAHs | 20.77 | 30.63 | 0.58-97.65 |
Risk classification for different polycyclic aromatic hydrocarbons (PAHs) and ΣPAHs
| ΣPAHs | Individual PAHs | ||||
| ΣPAHs | RQ(NCs) | RQ(MPCs) | Individual PAHs | RQ(NCs) | RQ(MPCs) |
| Risk-free | 0 | Risk-free | 0 | ||
| Low-risk | ≥ 1; < 800 | 0 | Moderate-risk | ≥ 1 | < 1 |
| Moderate-risk1 | ≥ 800 | 0 | High-risk | ≥ 1 | |
| Moderate-risk2 | < 800 | ≥ 1 | |||
| High-risk | ≥ 800 | ≥ 1 | |||
To calculate the toxic equivalency factors (TEFs), the toxic equivalent quantities (TEQs) of 16 PAHs were calculated. In Phayao Lake, the TEQs of 16 PAHs varied from 2.67 to 155.09 ng g-1, with an average of
If RQ(NCs) < 1 and RQ(MPCs) < 1 for certain PAHs, then the risk may be of minimal concern. However, if RQ(NCs) ≥ 1 and RQ(MPCs) < 1, then the risk is at a moderate level, and it may be essential to exercise control or implement remedial actions. Importantly, RQ(MPCs) ≥ 1 indicates a high-risk situation that necessitates quick action[31,32]. Categorization of the ecological risk of PAHs based on RQ(NCs) and RQ(MPCs) is shown in Table 3. The results of this study showed that RQ(NCs) < 1 and RQ(MPCs) < 1 for individual PAHs, indicating that the risk may be of minimal concern.
Table 4 and Figure 4 show how much each PAH increased the ecological risk of the sediment in Phayao Lake.The RQ(NCs) of ΣPAHs(RQΣPAHs(NCs)) ranged from 1.65 × 10-4 to 6.27 × 10-1 with an average value of
Figure 4. Ecological risks based on RQ(NCs) and RQ(MPCs) for specific Depth distribution of Polycyclic aromatic hydrocarbons (PAH) congeners in sediment samples from Phayao Lake as a percentage contribution.
Statistical parameters of RQ(NCs) and RQ(MPCs) of PAHs in sediments from Phayao Lake, Thailand
| PAHs | NCs | MPCs | RQ(NCs) | RQ(MPCs) | ||||
| Range | Mean | Stdev | Range | Mean | Stdev | |||
| Naphthalene | 1.4 | 140 | 1.49 × 10-2-1.46 × 10-1 | 8.93 × 10-2 | 3.44 × 10-2 | 1.49 × 10-4-1.46 × 10-3 | 8.93 × 10-4 | 3.44 × 10-4 |
| Acenaphthene | 1.2 | 120 | 1.14 × 10-3-6.26 × 10-2 | 1.71 × 10-2 | 1.93 × 10-2 | 1.14 × 10-5-6.26 × 10-4 | 1.71 × 10-4 | 1.93 × 10-4 |
| Phenanthrene | 5.1 | 510 | 3.05 × 10-3-4.59 × 10-2 | 2.02 × 10-2 | 1.08 × 10-2 | 3.05 × 10-5-4.59 × 10-4 | 2.02 × 10-4 | 1.08 × 10-4 |
| Fluoranthene | 26 | 2600 | 1.88 × 10-5-4.56 × 10-3 | 1.68 × 10-3 | 1.22 × 10-3 | 1.88 × 10-7-4.56 × 10-5 | 1.68 × 10-5 | 1.22 × 10-5 |
| Benz[a]anthracene | 3.6 | 360 | 5.09 × 10-2-1.22 × 100 | 3.34 × 10-1 | 3.60 × 10-1 | 5.09 × 10-4-1.22 × 10-2 | 3.34 × 10-3 | 3.60 × 10-3 |
| Chrysene | 107 | 10700 | 8.17 × 10-4-1.81 × 10-5 | 1.65 × 10-4 | 2.35 × 10-4 | 1.81 × 10-7-8.17 × 10-6 | 1.65 × 10-6 | 2.35 × 10-6 |
| Benzo[k]fluoranthene | 24 | 2400 | 4.03 × 10-3-4.56 × 10-1 | 8.83 × 10-2 | 1.52 × 10-1 | 4.03 × 10-5-4.56 × 10-3 | 8.83 × 10-4 | 1.52 × 10-3 |
| Benzo[a]pyrene | 27 | 2700 | ND-3.03 × 100 | 6.27 × 10-1 | 9.44 × 10-1 | ND-3.03 × 10-2 | 6.27 × 10-3 | 9.44 × 10-3 |
| Benzo[g,h,i]perylene | 75 | 7500 | 7.80 × 10-5-1.02 × 10-2 | 2.06 × 10-3 | 2.81 × 10-3 | 7.80 × 10-7-1.02 × 10-4 | 2.06 × 10-5 | 2.81 × 10-5 |
| Σ9PAHs | 1.18 × 100 | 1.52 × 100 | 1.18 × 10-2 | 1.52 × 10-2 | ||||
Health risk assessment of PAH contamination
It is well known that PAHs are found in the food chain because humans cook aquatic animals for consumption. Additionally, people in the area can breathe in the pollutants released from the sediment into the air. Therefore, ILCR was used in this study to determine the potential health risks for people exposed to PAHs released from the sediment in Phayao Lake. Investigation of the health risks included three exposure pathways: inhalation (ILCRInhalation), dermal contact (ILCRDermal), and direct ingestion (ILCRIngestion). Children and adults were divided into two age groups. Supplementary Table 2 contains statistical parameters of all ILCRs from sediment in Phayao Lake. The results revealed that the mean values of ILCRinhalation of all samples for children and adults were 7.62 × 10-13 ± 1.02 × 10-12 and 8.48 × 10-12 ± 1.14 × 10-11, respectively. The average values of ILCRDermal were 2.28 × 10-7 ± 3.06 × 10-7 for children and 1.94×10-7 ± 2.60 × 10-7 for adults. Moreover, the mean value of ILCRIngestion were 1.83×10-7 ± 2.45 × 10-7 for children and 1.09 × 10-7 ± 1.46 × 10-7 for adults, which were lower than the reference values, as shown in Figure 5. These findings indicated that dermal contact, followed by ingestion and inhalation, was the primary route of exposure to PAH in the sediment. These results agreed with those of previous studies in that analyses of samples collected from other Asian countries, such as India, Korea, and China, showed large increases in cancer risk as a result of soil-bound PAHs, with dermal absorption identified as the main route of exposure, followed by ingestion and inhalation[55-57].
The ILCR values of each of the investigated PAHs were less than 10-6 for both groups of humans (children and adults), which were lower than the baseline value[38,39], indicating that each PAH poses a minimal risk of cancer to humans. A previous study presented values of the hazard index (HI) for the non-carcinogenic effects of PAHs on humans: 1.68 ×10-6 for adults and 1.10 × 10-5 for children[58]. HI values greater than 1.0 constitute a serious health concern. Nap and Ant has the greatest and least contribution to non-carcinogenic effects on humans, with HI values of 5.87 × 10-7 for Nap and 2.08 × 10-8 for Ant. Therefore, our results showed that the cumulative lifetime cancer risk for both children and adults from exposure to PAHs in sediments from Phayao Lake was less than 10-6, which was below the baseline set for safe levels of PAHs.
CONCLUSIONS
Sediment cores from Phayao Lake were used in this work to examine the vertical trends, distributions, profiles, and hazardous ratios of PAHs. The results showed that LMW-PAHs (two or three fused aromatic rings) were dominant, and the depth distribution of PAHs in sediment cores showed that LMW-PAHs were dominant at every depth, indicating that the main source of PAHs is the incomplete combustion of organic sources, including pyrogenic, petrogenic, and biological matter. Therefore, PAHs are discharged into Phayao Lake through pyrogenic processes. In the present study, RQ(NCs) < 1 and RQ(MPCs) < 1 for individual PAHs, indicating that the risk may be of minimal concern. However, it may be necessary to exert control or take corrective action. RQ(NCs) of ΣPAHs (RQΣPAHs(NCs)) ranged from 1.65 × 10-4 to 6.27 × 10-1 with an average value of 1.18×100 ± 1.52 × 100 (Σ9PAHs), indicating a low risk to the environment, plants, and living organisms. All the values obtained in this study were lower than the values determined for samples from China (RQΣPAHs(NCs) > 800). These findings demonstrate that total PAHs present a significant level of ecological concern owing to petroleum pollution. Furthermore, the results showed the sediment-bound PAHs with the most prevalent exposure routes in the order of dermal absorption > ingestion > inhalation. The ILCR values of the investigated PAHs were less than 10-6 for children and adults, which were lower than the baseline value, indicating that each PAH poses a minimal risk of cancer to humans.
DECLARATIONS
AcknowledgementsThe authors acknowledge the Institute of Earth Environment, Chinese Academy of Sciences, Xian, China for supporting this study.
Authors’ contributionsMade significant contributions to the study’s conception and design as well as the data processing and interpretation: Han Y, Choochuay C
Conceptualization, writing - original draft preparation, review and editing, visualization: Deelaman W, Pongpiachan S
Availability of data and materialsAdditional data and information can be made available at request from the corresponding author.
Financial support and sponsorshipThe financial support was provided by the NSFC (41625015 and 41991250), the Strategic 398 Priority Research Program of Chinese Academy of Sciences (XDB40000000), and the 399 CAS (QYZDY-SSW-DQC001 and ZDBS-SSW-DQC001).
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2023.
Supplementary MaterialsREFERENCES
1. Bouloubassi I, Roussiez V, Azzoug M, et al. Sources, dispersal pathways and mass budget of sedimentary polycyclic aromatic hydrocarbons (PAH) in the NW Mediterranean margin, Gulf of Lions. Mar Chem 20121;142-144:18-28.
2. Bragato M, Joshi K, Carlson JB, Tenório JA, Levendis YA. Combustion of coal, bagasse and blends thereof. Fuel 2012;96:51-8.
3. Davis E, Walker TR, Adams M, Willis R, Norris GA, Henry RC. Source apportionment of polycyclic aromatic hydrocarbons (PAHs) in small craft harbor (SCH) surficial sediments in Nova Scotia, Canada. Sci Total Environ 2019;691:528-37.
4. Yunker MB, Macdonald RW, Vingarzan R, Mitchell RH, Goyette D, Sylvestre S. PAHs in the Fraser River basin: a critical appraisal of PAH ratios as indicators of PAH source and composition. Org Geochem 2002;33:489-515.
5. Jiang JJ, Lee CL, Fang MD, Liu JT. Polycyclic aromatic hydrocarbons in coastal sediments of southwest Taiwan: an appraisal of diagnostic ratios in source recognition. Mar Pollut Bull 2009;58:752-60.
6. Tongo I, Ezemonye L, Akpeh K. Distribution, characterization, and human health risk assessment of polycyclic aromatic hydrocarbons (PAHs) in Ovia River, Southern Nigeria. Environ Monit Assess 2017;189:247.
7. Awe AA, Opeolu BO, Olatunji O, et al. Occurrence and probabilistic risk assessment of PAHs in water and sediment samples of the Diep River, South Africa. Heliyon 2020;6:e04306.
8. Choochuay C, Pongpiachan S, Tipmanee D, et al. Impacts of PM2.5 sources on variations in particulate chemical compounds in ambient air of Bangkok, Thailand. Atmospheric Pollut Res 2020;11:1657-67.
9. Deelaman W, Pongpiachan S, Tipmanee D, et al. Source apportionment of polycyclic aromatic hydrocarbons in the terrestrial soils of King George Island, Antarctica. J South Am Earth Sci 2020;104:102832.
10. Mittal AK, Van Grieken R. Health risk assessment of urban suspended particulate matter with special reference to polycyclic aromatic hydrocarbons: a review. Rev Environ Health 2001;16:169-89.
11. Ravindra K, Sokhi R, Vangrieken R. Atmospheric polycyclic aromatic hydrocarbons: source attribution, emission factors and regulation. Atmospheric Environ 2008;42:2895-921.
12. Dumanoglu Y, Gaga EO, Gungormus E, Sofuoglu SC, Odabasi M. Spatial and seasonal variations, sources, air-soil exchange, and carcinogenic risk assessment for PAHs and PCBs in air and soil of Kutahya, Turkey, the province of thermal power plants. Sci Total Environ 2017;580:920-35.
13. Zhi H, Zhao Z, Zhang L. The fate of polycyclic aromatic hydrocarbons (PAHs) and organochlorine pesticides (OCPs) in water from Poyang Lake, the largest freshwater lake in China. Chemosphere 2015;119:1134-40.
14. Pongpiachan S. Application of cloud point extraction for the determination of pyrene in natural water. Southeast Asian journal of tropical medicine and public health 2009;40:392-400.
15. Pongpiachan S, Tipmanee D, Deelaman W, Muprasit J, Feldens P, Schwarzer K. Risk assessment of the presence of polycyclic aromatic hydrocarbons (PAHs) in coastal areas of Thailand affected by the 2004 tsunami. Mar Pollut Bull 2013;76:370-8.
16. Guzzella L, De Paolis A. Polycyclic aromatic hydrocarbons in sediments of the Adriatic Sea. Mar Pollut Bull 1994;28:159-65.
17. Han Y, Bandowe BAM, Schneider T, et al. A 150-year record of black carbon (soot and char) and polycyclic aromatic compounds deposition in Lake Phayao, north Thailand. Environ Pollut 2021;269:116148.
18. EPRI. Literature review of background polycyclic aromatic hydrocarbons. Available from: https://www.epri.com/research/products/TR-114755 [Last accessed on 9 Dec 2022].
19. Dong J, Li F, Xie K. Study on the source of polycyclic aromatic hydrocarbons (PAHs) during coal pyrolysis by PY-GC-MS. J Hazard Mater 2012;243:80-5.
20. Rinawati, Koike T, Koike H, et al. Distribution, source identification, and historical trends of organic micropollutants in coastal sediment in Jakarta Bay, Indonesia. J Hazard Mater 2012;217-218:208-16.
21. Tehrani GM, Hashim R, Sulaiman AH, et al. Distribution of total petroleum hydrocarbons and polycyclic aromatic hy-drocarbons in Musa Bay sediments (northwest of the Persian Gulf). ENVIRON PROT ENG 2013:39.
22. Yuan B, Brüchert V, Sobek A, de Wit CA. Temporal trends of C8-C36 chlorinated paraffins in Swedish coastal sediment cores over the past 80 years. Environ Sci Technol 2017;51:14199-208.
23. Levin W, Wood AW, Wislocki PG, et al. Carcinogenicity of Benzo-Ring Derivatives of Benzo(a)pyrene on Mouse Skin. Cancer Research 1977;37:3356-61.
24. Sayer JM, H Yagi, Wood AW, et al. Extremely facile reaction between the ultimate carcinogen benzo(a)pyrene-7,8-diol 9,10-epoxide and ellagic acid. J Am Chem. Soc 1982;104:5562-64.
25. Audere AK, Lindberg ZY, Smirnov GA, Shabad LM. Experiences in the study on the effect of an airport situated within the city limits on the level of environmental contamination with benz(a)pyrene. Gig Sanit 1973;38:90-2.
26. Falk HL, Markul I, Kotin P. Aromatic hydro carbons. IV. Their fate following emission into atmosphere and experimental exposure to washed air and synthetic smog. Arch Ind Health 1956;13:13-7. Available from: https://www.cabdirect.org/cabdirect/abstract/19562701970 [Last accessed on 9 Dec 2022]
27. Wang D, Ma J, Li H, Zhang X. Concentration and potential ecological risk of PAHs in different layers of soil in the petroleum-contaminated areas of the loess plateau, China. Int J Environ Res Public Health 2018;15:1785.
28. Choochuay C, Pongpiachan S, Tipmanee D, et al. Effects of agricultural waste burning on pm2.5-bound polycyclic aromatic hydrocarbons, carbonaceous compositions, and water-soluble ionic species in the ambient air of Chiang-Mai, Thailand. Polycycl Aromat Compd 2022;42:749-70.
29. Chompoorat T, Thepumong T, Taesinlapachai S, Likitlersuang S. Repurposing of stabilised dredged lakebed sediment in road base construction. J Soils Sediments 2021;21:2719-30.
30. Deelaman W, Pongpiachan S, Tipmanee D, et al. Ecotoxicological risk and health risk characterization of polycyclic aromatic hydrocarbons (PAHs) in terrestrial soils of King George Island, Antarctica. Polar Science 2021;29:100715.
31. Sun Y, Zhang S, Xie Z, et al. Characteristics and ecological risk assessment of polycyclic aromatic hydrocarbons in soil seepage water in karst terrains, southwest China. Ecotoxicol Environ Saf 2020;190:110122.
32. Kalf DF, Crommentuijn T, van de Plassche EJ. Environmental quality objectives for 10 polycyclic aromatic hydrocarbons (PAHs). Ecotoxicol Environ Saf 1997;36:89-97.
33. Cao Z, Liu J, Luan Y, et al. Distribution and ecosystem risk assessment of polycyclic aromatic hydrocarbons in the Luan River, China. Ecotoxicology 2010;19:827-37.
34. USEPA. Risk assessment guidance for superfund, volume 1, human health evaluation manual (Part B, Development of Risk-Based Preliminary Remediation Goals). Available from: https://semspub.epa.gov/work/HQ/192.pdf [Last accessed on 9 Dec 2022].
35. Wang J, Zhang X, Ling W, et al. Contamination and health risk assessment of PAHs in soils and crops in industrial areas of the Yangtze River Delta region, China. Chemosphere 2017;168:976-87.
36. Yang W, Lang Y, Li G. Cancer risk of polycyclic aromatic hydrocarbons (PAHs) in the soils from Jiaozhou Bay wetland. Chemosphere 2014; 112:289-295.
37. USEPA. Exposure Factors Handbook. 2011 edition. Final report. Environmental Protection Agency, Washington, DC EPA/600/R-09/052F. Available from: https://www.nrc.gov/docs/ML1400/ML14007A666.pdf [Last accessed on 9 Dec 2022].
38. Mihankhah T, Saeedi M, Karbassi A. Contamination and cancer risk assessment of polycyclic aromatic hydrocarbons (PAHs) in urban dust from different land-uses in the most populated city of Iran. Ecotoxicol Environ Saf 2020;187:109838.
39. USEPA. Human health evaluation manual, supplemental guidance: update of standard default exposure factors. 2014; OSWER Directive 9200, 1-120. Available from: https://www.epa.gov/sites/default/files/2015-11/documents/oswer_directive_9200.1-120_exposurefactors_corrected2.pdf [Last accessed on 9 Dec 2022].
40. Abdel-shafy HI, Mansour MS. A review on polycyclic aromatic hydrocarbons: source, environmental impact, effect on human health and remediation. Egypt J Pet 2016;25:107-23.
41. Baumard P, Budzinski H, Garrigues P. Polycyclic aromatic hydrocarbons in sediments and mussels of the western Mediterranean sea. Environ Toxicol Chem 1998;17:765-76.
42. Tepe Y, Taştekin Ö. Spatiotemporal PAH levels in the coastal sediment of Samsun, a Metropolis between Turkey's two largest deltas. Mar Pollut Bull 2022;181:113907.
43. Dong L, Lin L, He J, et al. PAHs in the surface water and sediments of the middle and lower reaches of the Han River, China: occurrence, source, and probabilistic risk assessment. Process Saf Environ 2022;164:208-18.
44. Tepe Y, Aydın H, Ustaoğlu F, et al. Seasonal distribution and risk assessment of polycyclic aromatic hydrocarbons (PAHs) in surface sediments from the Giresun coast of southeastern Black Sea. Mar Pollut Bull 2022;178:113585.
45. Zafarani GG, Karbalaei S, Golshani R, Pustokhina I, Walker TR. Baseline occurrence, distribution and sources of PAHs, TPH, and OCPs in surface sediments in Gorgan Bay, Iran. Mar Pollut Bull 2022;175:113346.
46. Iwegbue CMA, Bebenimibo E, Obi G, et al. Distribution and sources of n-alkanes and polycyclic aromatic hydrocarbons in sediments around oil production facilities in the Escravos River Basin, Niger Delta, Nigeria. Arch Environ Contam Toxicol 2021;80:474-89.
47. Wang YS, Wu FX, Gu YG, Huang HH, Gong XY, Liao XL. Polycyclic aromatic hydrocarbons (PAHs) in the intertidal sediments of Pearl River Estuary: characterization, source diagnostics, and ecological risk assessment. Mar Pollut Bull 2021;173:113140.
48. Vaezzadeh V, Yi X, Rais F. R, et al. Distribution of black carbon and PAHs in sediments of peninsular Malaysia. Mar Pollut Bull 2021; 172:112871.
49. Yang ZY, Shah K, Laforest S, et al. Occurrence, characterization, and ecological assessment of petroleum-related hydrocarbons in intertidal marine sediments of Burrard Inlet, Vancouver, British Columbia, Canada. Mar Pollut Bull 2020;157:111304.
50. Chen F, Lin Y, Cai M, et al. Occurrence and risk assessment of PAHs in surface sediments from western arctic and subarctic oceans. Int J Environ Res Public Health 2018;15:734.
51. Edokpayi J, Odiyo J, Popoola O, Msagati T. Assessment of trace metals contamination of surface water and sediment: a case study of Mvudi River, South Africa. Sustainability 2016;8:135.
52. Dudhagara DR, Rajpara RK, Bhatt JK, Gosai HB, Sachaniya BK, Dave BP. Distribution, sources and ecological risk assessment of PAHs in historically contaminated surface sediments at Bhavnagar coast, Gujarat, India. Environ Pollut 2016;213:338-46.
53. Maliszewska-kordybach B. Polycyclic aromatic hydrocarbons in agricultural soils in Poland: preliminary proposals for criteria to evaluate the level of soil contamination. Applied Geochemistry 1996;11:121-7.
54. Canadian Council of Ministers of the Environment (CCME). Canadian soil quality guidelines for the protection of environmental and human health. 2007; Dioxins and Furans. Available from: https://ccme.ca/en/resources/sediment [Last accessed on 9 Dec 2022].
55. Tarafdar A, Sinha A. Cancer Risk Assessment of polycyclic aromatic hydrocarbons in the soils and sediments of India: a meta-analysis. Environ Manage 2017;60:784-95.
56. Kim SJ, Park MK, Lee SE, et al. Impact of traffic volumes on levels, patterns, and toxicity of polycyclic aromatic hydrocarbons in roadside soils. Environ Sci Process Impacts 2019;21:174-82.
57. Shi R, Li X, Yang Y, Fan Y, Zhao Z. Contamination and human health risks of polycyclic aromatic hydrocarbons in surface soils from Tianjin coastal new region, China. Environ Pollut 2021;268:115938.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.





















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].