Microdroplet technology for green chemistry and sustainable energy systems
Abstract
In response to escalating global environmental challenges and carbon neutrality goals, developing efficient, eco-friendly chemical transformation platforms is central to sustainable development. Microdroplets, characterized by their immense specific surface area, ultra-strong interfacial electric fields (~109 V/m), and unique confinement effects, exhibit remarkable kinetic acceleration exceeding conventional bulk systems, providing an ideal micro-environment for sustainable molecular activation. Crucially, these interfacial phenomena align with the core principles of green chemistry by enabling high-efficiency transformations without external energy-intensive drivers. This article systematically reviews recent advances in microdroplet-mediated green chemistry. Specifically, the review highlights current applications of microdroplets across key sustainable synthesis scenarios, including CO2 utilization, artificial nitrogen fixation, green H2O2 synthesis, and the directed transformation of complex organic feedstocks such as alkanes and alcohols under mild conditions. By scrutinizing interfacial electric-field-induced charge separation and radical-mediated activation pathways, the underlying physicochemical mechanisms of microdroplet-intensified green chemical processes are comprehensively elucidated. To guide future practical implementation, key challenges regarding industrial scale-up and applications in complex catalytic systems are discussed, along with forward-looking perspectives on innovative directions for sustainable chemical technologies.
Keywords
INTRODUCTION
With the increasing global demand for sustainable development, developing efficient, eco-friendly chemical transformation technologies, such as CO2 reduction, artificial nitrogen fixation, and green fuel synthesis, has become a central focus of scientific research[1-4]. However, traditional bulk-phase reaction systems are often constrained by physical limitations, including low gas solubility, high mass transfer resistance, and poor charge separation efficiency[5]. In recent years, the emergence of microdroplet chemistry has offered a new pathway to overcome these bottlenecks. As a unique micro-reactor, microdroplets not only provide an immense specific surface area but also exhibit chemical kinetic characteristics distinct from the bulk phase due to sub-micrometer-scale confinement effects[1,6-9].
The core allure of microdroplets lies in their transient and extreme interfacial environments[10]. At the gas-liquid interface, a spontaneously generated ultra-strong electric field (up to 109 V/m) can significantly lower the activation energy barriers of inert molecules and promote the spatial separation of photogenerated charge carriers[5,7,11]. Simultaneously, contact electrification at the interface endows droplets with a reducing power comparable to alkali metals, enabling the spontaneous generation of H2O2 or the reduction of CO2 without external voltage or catalysts[12]. By utilizing water as a universal green solvent and leveraging these interfacial phenomena to replace energy-intensive industrial processes, microdroplet chemistry offers a transformative paradigm for sustainable manufacturing. This ability to transcend bulk thermodynamic limits transforms microdroplets into ideal platforms for micro-laboratories for exploring the activation mechanisms of green chemistry[13]. This review aims to systematically summarize the latest research progress of microdroplet chemistry in the field of green synthesis and sustainable transformation. The discussion primarily focuses on the application of microdroplets across diverse eco-friendly scenarios, specifically evaluating their pivotal roles in CO2 valorization, ambient nitrogen fixation, green H2O2 synthesis, and the selective functionalization of complex organic feedstocks such as alkanes and alcohols. By scrutinizing the unique interfacial properties - including ultra-strong electric fields, enhanced mass transfer, and spontaneously generated reactive oxygen species (ROS) - this work elucidates the physicochemical mechanisms that enable microdroplets to transcend the thermodynamic and kinetic constraints of bulk-phase reactions. Finally, forward-looking perspectives are provided regarding the challenges of industrial scale-up, offering a strategic roadmap for the development of future sustainable energy technologies.
DIVERSIFIED APPLICATION SCENARIOS IN GREEN CHEMISTRY
Building upon the unique physicochemical properties of the gas-liquid interface, microdroplets have emerged as highly versatile platforms for various green chemistry applications[10,14,15]. This section comprehensively explores the diversified application scenarios of microdroplet reactors in sustainable chemical transformations. Specifically, we focus on their roles in mitigating greenhouse gas emissions via CO2 valorization, promoting energy-efficient ambient nitrogen fixation, enabling the on-demand green synthesis of H2O2, and facilitating the directed functionalization of complex organic feedstocks under mild conditions. The significance of these microdroplet-mediated processes extends beyond mere laboratory-scale acceleration; they represent a fundamental paradigm shift in chemical manufacturing. By bypassing the inherent thermodynamic and kinetic limitations of bulk-phase chemistry, microdroplet systems offer a strategic pathway to address global energy and environmental challenges. Their ability to drive high-value transformations at ambient temperature and pressure, eliminating the need for energy-intensive catalysts or harsh operating environments, positions microdroplet technology as a critical cornerstone for achieving carbon neutrality and sustainable industrial development. These applications collectively demonstrate the potential of microdroplet systems to replace traditional, resource-intensive bulk processes with eco-friendly and sustainable alternatives.
Microdroplet-mediated CO2 reduction and conversion
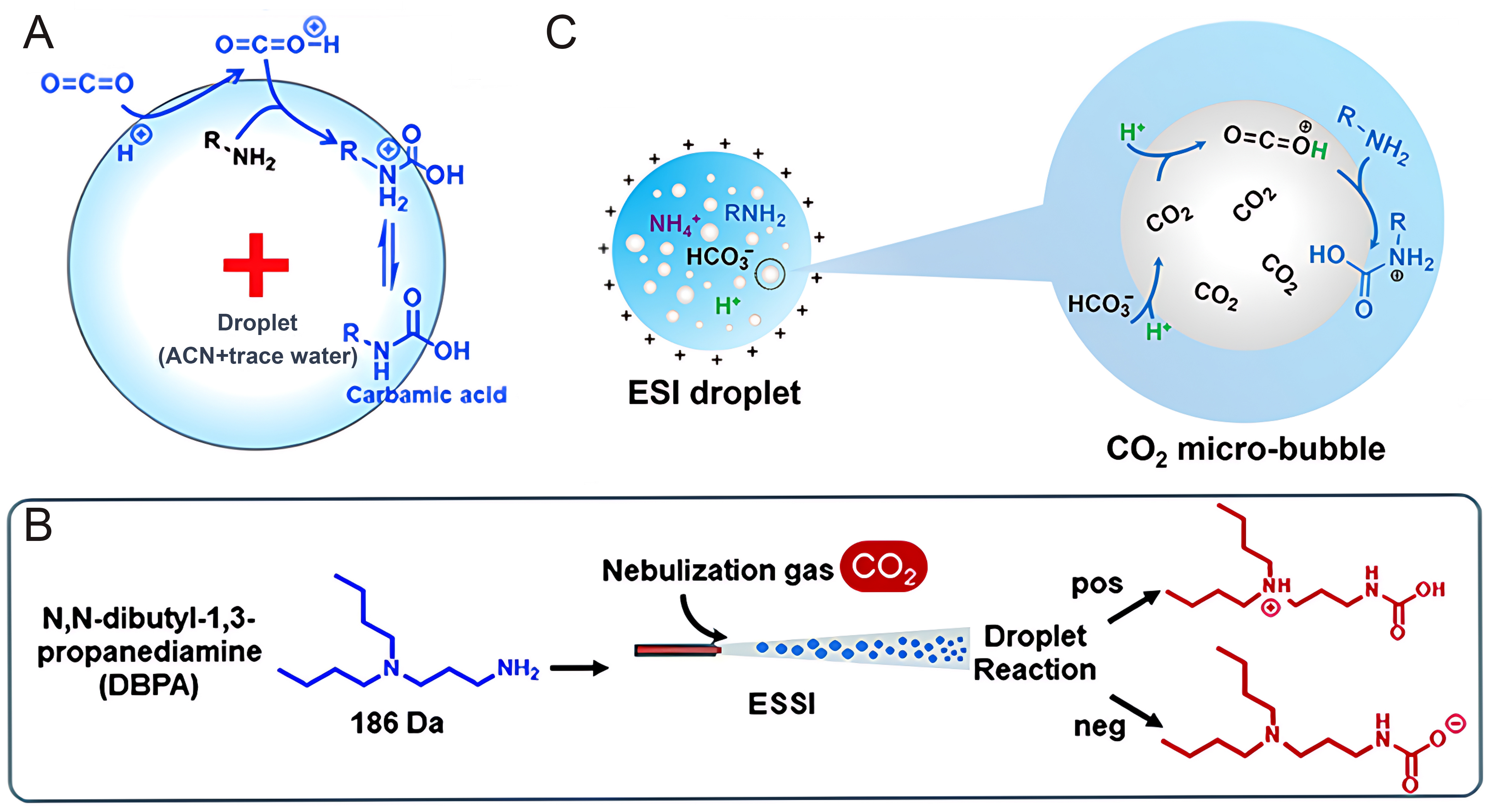
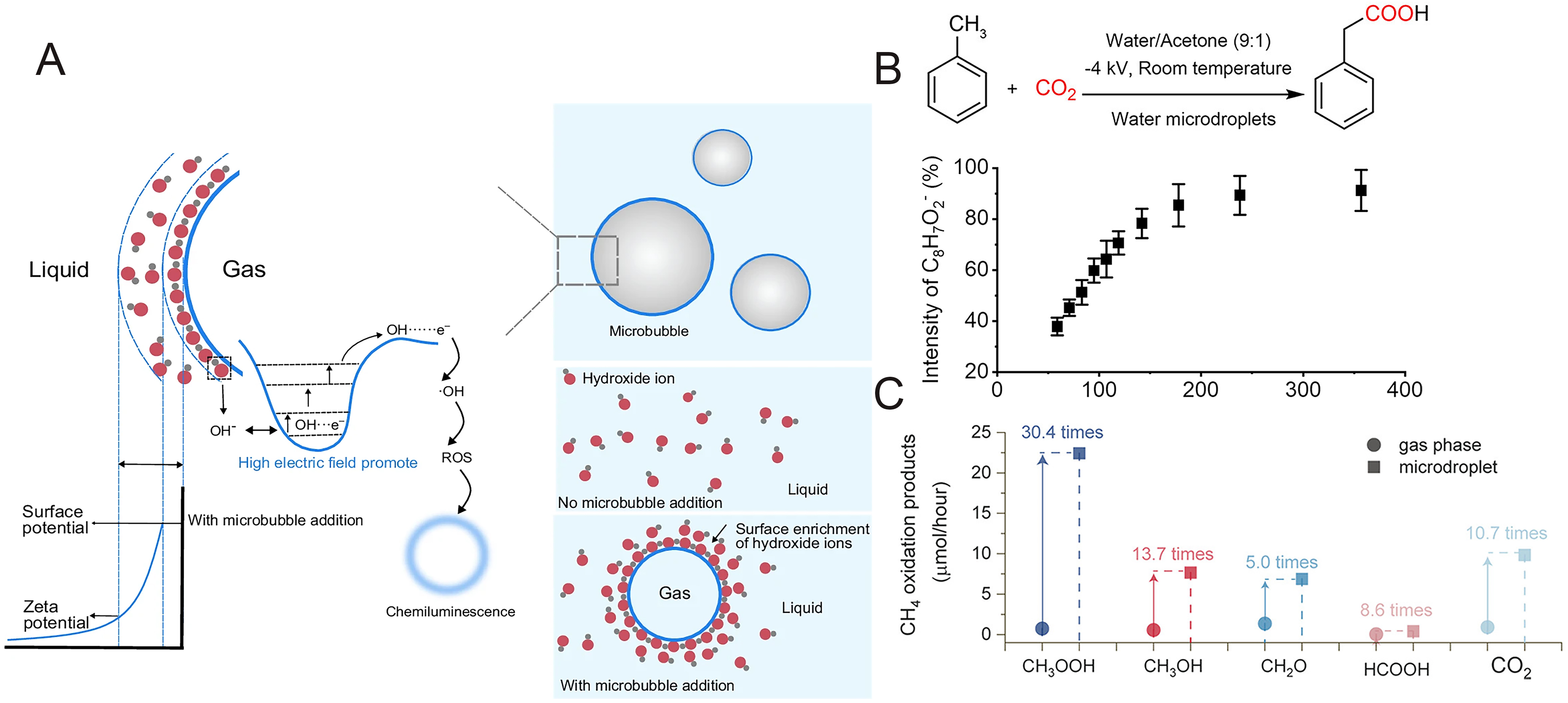
Microdroplet technology first addresses the long-standing gas-liquid mass transfer limitations in CO2 capture. Recent engineering studies have introduced cyclonic field technology; for instance, Tao et al. developed a “Pre-Self-Rotation Cyclone Absorber” (PSRCA) that utilizes strong shear forces to induce high-speed self-rotation and revolution of amine droplets[16]. This unique hydrodynamic behavior drastically renews the gas-liquid interface and thins the boundary layer, increasing the volumetric mass transfer coefficient (KLa) several times over traditional reactors and achieving a CO2 capture efficiency of over 95% within milliseconds. At the microscopic chemical level, the acceleration mechanisms for amine-CO2 reactions in microdroplets are categorized into two pathways: (1) the interfacial superacid mechanism, where Huang et al. discovered that the strong interfacial electric field induces water dissociation to generate protons that activate CO2 for nucleophilic attack by amines [Figure 1A and B][17]; and (2) the “Microbubble” mechanism, as Feng et al. found that adding ammonium bicarbonate releases endogenous bubbles via decomposition inside droplets, physically expanding the reaction interface area and enabling a conversion rate of 93.7% [Figure 1C][18].
Figure 1. CO2 capture mechanisms in aqueous microdroplets. (A) Proposed interfacial superacid mechanism for the accelerated reactions of amines with CO2 in room air[17]; (B) Reaction pathway of Amine with CO2 in microdroplets[17]; (C) Schematic of the microbubble in the microdroplet mechanism accelerated by ammonium bicarbonate[18]. Figure 1A and B is reprinted with permission from Ref.[17], available under a CC-BY-3.0. Figure 1C is reprinted with permission from Ref.[18], Copyright © 2021 American Chemical Society. ACN: Acetonitrile; ESI: electrospray ionization; ESSI: enhanced electrospray ionization.
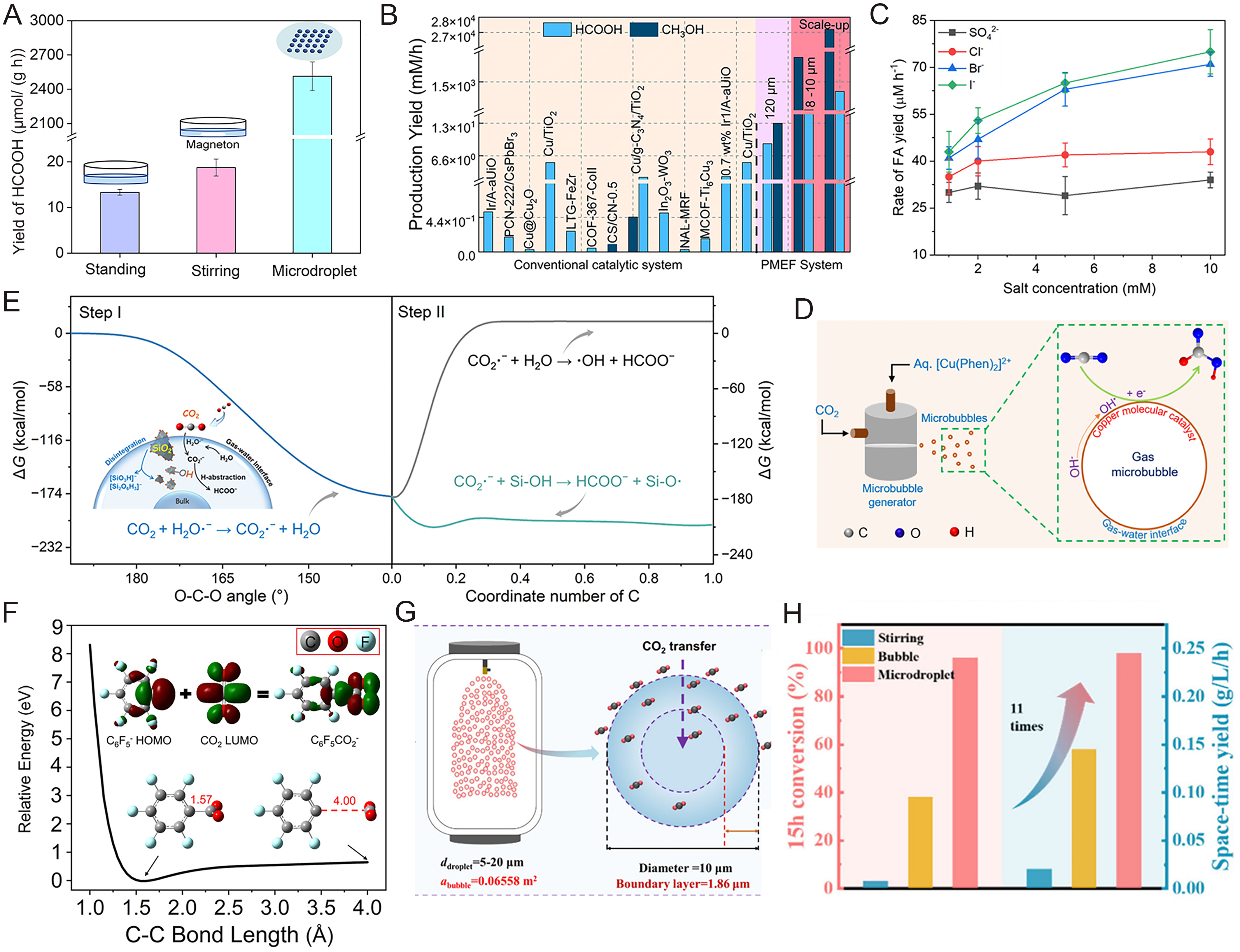
The spontaneous electric field of up to 109 V/m at the gas-liquid interface endows microdroplets with unique reactivity[13,19]. In photocatalysis, this field is utilized to effectively separate photogenerated electrons and holes, enhancing the formic acid yield of the WO3·0.33H2O reduction system by two orders of magnitude compared to the bulk phase [Figure 2A][5]. More importantly, a recent study revealed that microdroplet size is key to regulating product selectivity: as the droplet diameter decreases below 10 μm, the intensified interfacial electric field significantly prolongs the lifetime of the strong reductant hydrated electrons (eaq-) and alters intermediate energy levels, thereby elevating the selectivity for deep reduction of CO2 to methanol to ~99% [Figure 2B][1]. In molecular catalysis, copper-phenanthroline complexes show high selectivity for formic acid in microbubble systems [Figure 2C and D][20]. This reductive microenvironment facilitates the spontaneous conversion of inert molecules; for instance, silica (SiO2) nanofluids can drive CO2 reduction to formic acid and ethanol [Figure 2E][21]. In addition, 1,2,3-triazole assists CO2 conversion via complex formation [Figure 2F][22,23].
Figure 2. CO2 activation and conversion in aqueous microdroplets. (A) WO3·0.33H2O photocatalytic CO2 reduction[5]; (B) Comparisons between the yield of CO2 photoreduction in the microdroplet system (PMEF) and the previous observation in bulk solution[1]; (C) Formation of FA (HCOOH) depending on different conditions of the copper(II)-phenanthroline complex [Cu(Phen)2]2+[20]; (D) Schematic of the experimental setup for the reduction of CO2 in microbubbles[20]; (E) SiO2-driven CO2 conversion at the gas-water interface[21]; (F) Fixation of CO2 by C6F5- in microdroplets[6]; (G) Schematic diagram of scale-up reactor and CO2 transfer in microdroplet[15]; (H) Kinetics analysis of CO2 conversion in different scale-up reactors (stirring reactor, bubble reactor, and microdroplet reactor)[15]. Figure 2A is reprinted with permission from Ref.[5], Copyright © 2023 WILEY. Figure 2B is reprinted with permission from Ref.[1], available under a CC BY-NC 4.0. Figure 2C and D is reprinted with permission from Ref.[20], Copyright © 2025 American Chemical Society. Figure 2E is reprinted with permission from Ref.[21], Copyright © 2025 American Chemical Society. Figure 2F is reprinted with permission from Ref.[6], Copyright © 2023 American Chemical Society. Figure 2G and H is reprinted with permission from Ref.[15], Copyright © 2025 American Chemical Society. HOMO: Highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.
Addressing the low throughput and scale-up challenges of traditional microreactors, a 400-L industrial-scale microdroplet reactor has been developed guided by multi-scale Computational Fluid Dynamics (CFD) simulations [Figure 2G and H]. By optimizing nozzle arrays and internal flow fields, this system demonstrates superior performance in the continuous cycloaddition of CO2 and epoxides, achieving an annual capacity of 420 kg per unit with high consistency between experimental and simulated data[15]. Additionally, microdroplet intensification systems combined with deep eutectic solvents (DES) or ionic liquids have achieved substrate conversion rates as high as 85%[24]. These engineering breakthroughs confirm that microdroplet technology offers not only unique reaction pathways but also a feasible solution for overcoming mass transfer limitations, reducing energy consumption, and realizing large-scale CO2 resource utilization in industry.
Nitrogen fixation in microdroplets
The gas-liquid interface of microdroplets possesses unique physicochemical properties that can overcome the extremely stable N≡N triple bond in nitrogen gas (N2), enabling nitrogen fixation under mild conditions. Research indicates that a strong electric field of up to 109 V/m exists at the microdroplet surface, which promotes the spontaneous redox reaction of interfacial water molecules to generate reactive hydrogen species (RHS; e.g., hydrated electrons and hydrogen radicals) and ROS (e.g., hydroxyl radicals[25]). These reactive species act as electron donors and proton sources, triggering the reductive or oxidative activation of nitrogen[26]. For instance, by controlling the self-electrification of microdroplets and their charge neutralization on a metal mesh, electron transfer can be guided to regulate the reaction pathways of nitrogen transformation[27].
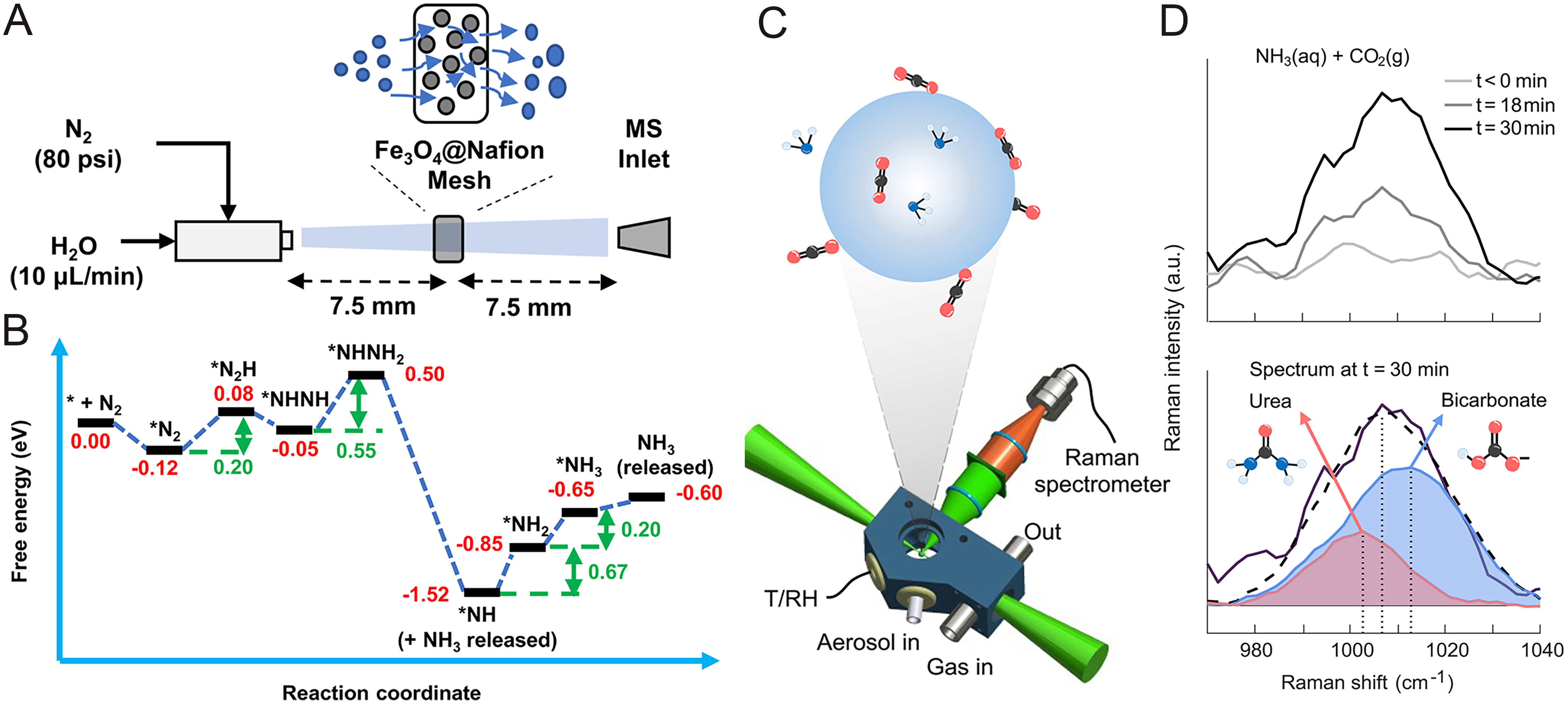
Using the mechanisms described above, researchers have successfully achieved the conversion of nitrogen gas into various inorganic nitrogenous compounds. In the reduction pathway, nitrogen in sprayed microdroplets can be hydrogenated to generate ammonia (NH3) using a Fe2O3 and Nafion-coated mesh [Figure 3A and B], where water microdroplets provide the necessary hydrogen atoms; this process occurs on a millisecond timescale without external voltage or irradiation[25]. In the oxidation pathway, the conversion of nitrogen to nitrate (NO3-) is enhanced through charge neutralization on a nickel (Ni) mesh, achieving a robust semi-continuous nitrogen fixation process[27]. Furthermore, recent studies have uncovered a novel reduction-then-oxidation (RTO) mechanism, where nitrogen is first reductively activated by interfacial hydrated electrons and subsequently reacts rapidly with oxygen or ozone to produce nitrous oxide (N2O)[28].
Figure 3. Investigation of microdroplet-mediated nitrogen fixation and subsequent carbon-nitrogen coupling. (A) Ultrasonic spray setup combined with mass spectrometry (MS) for the online monitoring of the nitrogen-to-ammonia conversion process[25] and (B) corresponding free energy changes found from DFT calculations[25]; (C) Schematic representation of a single microdroplet with ammonia and carbon dioxide molecules[10]; (D) Raman spectra recorded before and after exposure of an aqueous ammonia [NH3(aq)] droplet to CO2 gas [CO2(g)][10]. Figure 3A and B is reprinted with permission from Ref.[25], available under a CC BY-NC 4.0. Figure 3C and D is reprinted with permission from Ref.[10], Copyright © 2025 The American Association for the Advancement of Science. DFT: Density functional theory; MS: mass spectrometer; T/RH: temperature and relative humidity.
The potential of microdroplet chemistry extends to combining nitrogen fixation with carbon capture, realizing the one-step synthesis of urea, a high-value organic compound. By spraying N2 and CO2 as nebulizing gases onto a catalyst, water microdroplets serve as a bridge connecting gas-phase substrates and the solid-phase catalyst while providing protons and electrons[26]. It is noteworthy that the unique interfacial environment of microdroplets even enables C-N coupling in the absence of a catalyst[29]. Recent research demonstrates that when using NH3 and CO2 as raw materials, the pH gradient at the microdroplet surface can function as a microscopic flow reactor [Figure 3C and D]. This allows the reaction to bypass the energy barriers that are typically insurmountable in bulk phases, spontaneously generating urea through a carbamic acid intermediate[10]. Overall, these findings underscore the transformative potential of microdroplet interfaces in facilitating multi-substrate coupling for the sustainable synthesis of high-value nitrogenous compounds.
Synthesis of H2O2 in microdroplet systems
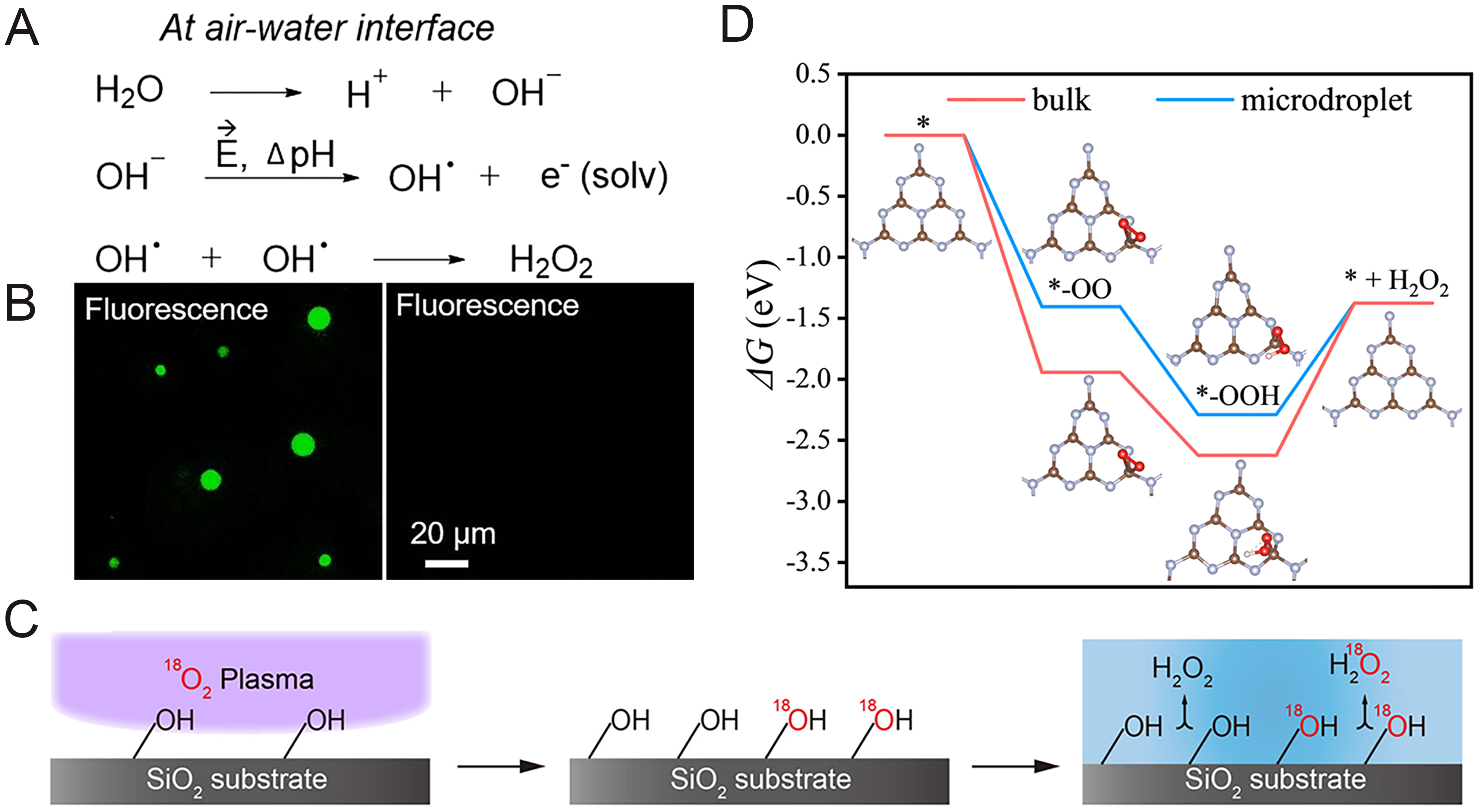
The spontaneous generation of H2O2 at the air-water interface of pure water microdroplets challenges traditional thermodynamic understanding[30-33]. By utilizing microwell confinement technology to extend droplet lifetimes, researchers have dynamically monitored H2O2 generation in individual sub-10 μm droplets using time-lapse fluorescence imaging [Figure 4A and B][32]. Experiments confirmed that the generation rate of H2O2 is directly proportional to the surface-to-volume ratio of the droplets, indicating that the reaction is strictly interface-controlled[34]. In the presence of competitive H2O2-consuming reactions, an apparent equilibrium concentration of approximately 3 μM can eventually be reached within the droplets[31]. This continuous spontaneous generation characteristic not only reveals the enrichment patterns of ROS in microscale reactors but also provides a physical foundation for catalyst-free green oxidant production. Contact electrification occurring during the formation of microdroplets is considered a key energy source driving H2O2 generation [Figure 4C][35]. Research indicates that when water and hydrophobic liquids (such as oil) form emulsion microdroplets via ultrasonic spraying, interfacial electron exchange can increase the number of charge carriers by approximately 13-fold, subsequently enhancing the yield of generated hydrogen species by 16-fold[36].
Figure 4. Synthesis of H2O2 in microdroplet systems. (A) Proposed mechanism to form H2O2 at the air-water interface of microdroplets[31]; (B) Fluorescence imaging of spontaneous generation of hydrogen peroxide in aqueous microdroplets[31]; (C) Dependence of H2O2 generation on the surface OH groups on the SiO2 substrate[35]; (D) DFT calculations of photocatalytic H2O2 production[37]. Figure 4A and B is reprinted with permission from Ref.[31], available under a CC BY-NC-ND 4.0. Figure 4C is reprinted with permission from Ref.[35], available under a CC BY-NC-ND 4.0. Figure 4D is reprinted with permission from Ref.[37], with permission from the Royal Society of Chemistry. DFT: Density functional theory.
Similarly, the radical recombination mechanism triggered by water-solid contact electrification has been confirmed as the dominant pathway for H2O2 production: OH- at the interface loses electrons to form hydroxyl radicals (·OH), which then dimerize (·OH + ·OH → H2O2). The coexistence of this spontaneously generated strong reducing and oxidizing power transforms the microdroplet into a micro-electrochemical cell capable of driving numerous redox cycles that are difficult to achieve in the bulk phase[35].
Beyond spontaneous pathways, the microdroplet environment significantly enhances the efficiency of artificially induced oxygen reduction reactions (ORR)[37]. Under photocatalysis or with external voltage assistance, the unique gas-liquid interface of microdroplets effectively overcomes the mass transfer resistance of oxygen (O2) in water, maintaining saturated O2 concentrations at the catalyst surface [Figure 4D][37]. Utilizing online electrochemical monitoring and in-situ spectroscopy, researchers have observed that by tuning the interfacial electric field strength in microdroplet systems, the selectivity of the 2e- pathway can be significantly improved, suppressing the competitive 4e- reaction that produces water[33]. This interfacial enhancement makes the direct synthesis of H2O2 from ambient O2 more efficient, providing highly promising technical solutions for distributed energy storage and in-situ water treatment.
Interfacial selective transformation of hydrocarbons and oxygenated fuels
The activation of C-H bonds in alkanes typically requires high temperatures or precious metal catalysts, whereas the microdroplet interface offers a mild solution to this challenge. It has been discovered that during the water microdroplet spraying process, spontaneously generated hydroxyl radicals (·OH) at the interface can efficiently abstract hydrogen atoms from alkanes to generate active carbon radicals, thereby initiating subsequent value-added transformation pathways [Figure 5A][38]. For instance, by applying a negative high voltage during spraying, toluene can react with gas-phase CO2 at the interface in a single step to form phenylacetic acid, achieving the synthesis of pharmaceutical intermediates under catalyst-free conditions [Figure 5B][19]. This interface-driven radical mechanism is not only applicable to toluene but also generalizable to various molecules with aryl α-C-H structures, demonstrating the versatility of microdroplets in the functionalization of complex organic molecules[13]. Furthermore, this interfacial acceleration extends to the most fundamental alkane, methane (CH4). Recent findings reveal that methane photo-oxidation at the air-water interface is enhanced by an order of magnitude compared to the bulk phase [Figure 5C], driven by enriched interfacial hydroxyl radicals and novel barrierless reaction pathways[39].
Figure 5. Interfacial radical-driven organic transformations in microdroplet systems. (A) Schematic diagram of the mechanism of ·OH generated at the gas‒liquid interface of microbubbles[38]; (B) The formation of phenylacetic acid from toluene and CO2 in water microdroplets[19]; (C) Comparison of CH4 photo-oxidation product generation rates under gas-phase and microdroplet conditions[39]. Figure 5A is reprinted with permission from Ref.[38], available under a CC BY-NC-ND 4.0. Copyright © 2026 Springer Nature. Figure 5B is reprinted with permission from Ref.[19], Copyright © 2023 American Chemical Society. Figure 5C is reprinted with permission from Ref.[39], Copyright © 2025 American Chemical Society.
The selective transformation of alcohols into aldehydes or ketones is a vital link in energy chemistry, yet bulk reactions are often limited by the over-oxidation of products into carboxylic acids[40]. The microdroplet system leverages its extremely short reaction residence time (typically on the millisecond scale) and rapid phase separation characteristics to transfer generated intermediates from the highly active interface to the droplet interior in real-time, thereby protecting the products. Research indicates that in microdroplet cascade catalytic systems, the selective conversion rates of biomass-derived alcohols (such as glycerol and ethylene glycol) are significantly enhanced, with the degree of oxidation being effectively controlled[41]. This “fast-in, fast-out” kinetic feature makes microdroplets an ideal platform for processing complex oxygenated molecules, enabling high-selectivity liquid fuel valorization under ambient conditions.
The high specific surface area of microdroplets significantly eliminates mass transfer resistance in heterogeneous catalytic processes, particularly in complex systems involving the degradation or transformation of non-polar hydrocarbons. When oil-water mixtures form emulsion microdroplets through ultrasonic spraying, contact electrification between the interfaces generates a large number of ROS, enabling the spontaneous degradation of long-chain alkanes such as hexadecane (C16H34) under anoxic conditions[14]. The observed generation of short-chain hydrocarbons (C1, C2) and H2 confirms that the microdroplet interface can effectively strip the solvation layer from catalyst surfaces, intensifying the contact between reactant molecules and active sites[42]. This interfacial enhancement effect provides a new experimental basis for developing low-energy, high-throughput technologies for the conversion of biomass and petrochemical resources.
TECHNO-ECONOMIC EVALUATION AND SCALABILITY
To further evaluate the practical viability of microdroplet technology, Table 1 provides a comprehensive quantitative comparison of the performance metrics and techno-economic potential of representative systems. The transition from laboratory-scale chemistry to industrial green manufacturing hinges on overcoming the mass-transfer and energy barriers inherent in bulk-phase processes. As summarized, microdroplet platforms exhibit extraordinary kinetic superiority, exemplified by a 103-104 fold acceleration in CO2 capture[1,17,18] and an order-of-magnitude rate enhancement in methane photo-oxidation[39]. Beyond kinetics, the economic competitiveness is underscored by spontaneous and catalyst-free pathways; for instance, the voltage-free synthesis of urea and H2O2 under ambient conditions represents an ‘ultimate green synthesis’ model that effectively eliminates external energy inputs and expensive chemical reagents[10]. Furthermore, the implementation of a numbering-up strategy utilizing modular spray reactors facilitates seamless scalability, achieving a space-time yield of 420 kg/year for cyclic carbonates[19]. This process intensification not only reduces the reactor footprint by three orders of magnitude but also drives a cliff-like reduction in capital and operational expenditures (CAPEX/OPEX). Consequently, these multidimensional advancements position the microdroplet platform as a high-efficiency, cost-effective alternative for decentralized chemical production and sustainable carbon/nitrogen utilization.
Quantitative performance metrics, reaction conditions of representative microdroplet-enhanced chemical transformations
| Application category | Target reaction/system | Key performance & advancement | Reaction conditions | Refs. |
| CO2 valorization | CO2 capture & conversion | 103-104 fold acceleration | Air-water interface; Pure water | [1,17,18] |
| Cyclic carbonate synthesis | 11x higher space-time yield; 420 kg/year per modular unit | Catalyst-free; Single-step spray reactor | [19] | |
| Nitrogen fixation | Spontaneous urea synthesis | 0.1 ms ultra-fast reaction; Yield: 12.3 mmol·g-1·h-1 | No external voltage; Pure water/gas interface | [10] |
| Green synthesis | Spontaneous H2O2 formation | 1 in 65,000 water molecules converted; 7.7 nmol/min | Ambient spray; No external reagents/energy | [31] |
| C-H activation | CH4 photo-oxidation | > 10 times faster than bulk phase (Order of magnitude) | Light-driven; Air-water interface | [39] |
| Resource valorization | Alkane degradation (C16) | Spontaneous generation of C1, C2, and H2 without oxygen | Ultrasonic spraying; Contact electrification | [42] |
CONCLUSION AND OUTLOOK
Microdroplet chemistry provides a new paradigm for addressing the kinetic and thermodynamic challenges in traditional energy conversion and sustainable chemical synthesis. This review systematically revisits the significant acceleration effects of microdroplets in fields such as CO2 reduction, nitrogen fixation, H2O2 synthesis, and complex molecule upgrading. These enhancements are attributed to the unique physicochemical properties of microdroplets: the vast specific surface area overcomes gas-liquid mass transfer resistance; the strong interfacial electric field (~109 V/m) induces the polarization of inert molecules and promotes charge carrier separation; and the spontaneously generated ROS and interfacial electrons provide potent redox driving forces[39]. These findings not only deepen our understanding of interfacial chemistry but also demonstrate the immense potential of microdroplets in constructing efficient, decentralized energy conversion and green chemical systems.
Despite the promising outlook, microdroplet energy chemistry still faces numerous challenges. Firstly, although on-line mass spectrometry and in-situ spectroscopic techniques have made significant progress, direct observation of nanosecond-scale ultrafast charge migration and molecular orientation at the droplet surface remains insufficient, limiting definitive mechanistic conclusions. Secondly, current research is predominantly concentrated at the laboratory scale (μL/min level); thus, designing efficient microdroplet array reactors for high-throughput industrial production while maintaining the unique properties of micrometer-scale interfaces remains a significant engineering barrier. Furthermore, the long-term stability of energy conversion and the mechanical and chemical degradation of catalysts during cyclic spraying processes require in-depth evaluation.
From a techno-economic perspective, the transition of microdroplet systems from fundamental research to viable industrial platforms requires a rigorous evaluation of energy efficiency and cost-effectiveness. Future studies should focus on the “energy-per-gram” metric, comparing the power consumption of high-pressure nebulization or ultrasonic atomization with the yield enhancement achieved. While microdroplets eliminate the need for bulk stirring and large volumes of hazardous solvents, the economic burden of gas compression and continuous collection of products at the micrometer scale must be systematically analyzed through comprehensive Techno-Economic Analysis (TEA). Establishing standardized protocols for life-cycle assessments will be crucial to quantify the true “greenness” of microdroplet processes, ensuring that the accelerated kinetics translate into a net reduction in both operational costs and carbon footprint compared to conventional batch reactors.
Looking ahead, microdroplet chemistry is poised for leapfrog development through deep interdisciplinary integration with materials science, fluid mechanics, and artificial intelligence (AI). Screening catalytic systems adapted to droplet interfaces using advanced AI algorithms, combined with microfluidic technology for precise regulation of droplet dynamic parameters, is expected to significantly enhance energy efficiency. More importantly, microdroplet technology can be integrated with atmospheric aerosol chemistry to provide new perspectives on understanding the natural carbon/nitrogen cycles. As a green, solvent-free, or water-based transformation technology, microdroplet chemistry is expected to play a key role in distributed hydrogen production, on-site nitrogen fertilizer synthesis, and the eco-friendly preparation of value-added chemicals, thereby contributing to the achievement of global carbon neutrality goals.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the work, performed the systematic literature search, and drafted the original manuscript: Ge, Q.; Liu, Y.
Provided administrative, technical, and material support, and assisted in figure preparation: Wang, T.
Provided financial support, supervised the research progress, and performed final review and editing of the manuscript: Zhang, L.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Some elements in the graphical abstract were generated by AI tool Gemini 3 Flash Image (version 3.1, released 2026-02-26) and do not involve copyright issues.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (No. 22376028 and No. 22176036 to Zhang, Z.; No. 223B2601 to Ge, Q.) and the Natural Science Foundation of Shanghai (No. 19ZR1471200 to Zhang, L.).
Conflicts of interest
Zhang, L. is an Editorial Board Member of Greenverse Science; he had no involvement in the review or editorial process of this manuscript, including but not limited to reviewer selection, evaluation, or the final decision. The other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Ge, Q.; Liu, Y.; You, W.; et al. Substantially improved efficiency and selectivity of carbon dioxide reduction by superior hydrated electron in microdroplet. Sci. Adv. 2025, 11, eadx5714.
2. Shi, Y.; Su, W.; Xing, Y.; et al. Research progress and future prospects of chemical utilization of CO2. Chem. Eng. J. 2025, 510, 161579.
3. Kim, D.; Surendran, S.; Janani, G.; et al. Nitrogen-impregnated carbon-coated TiO2 nanoparticles for N2 reduction to ammonia under ambient conditions. Mater. Lett. 2022, 314, 131808.
4. Jesudass, S. C.; Jo, C.; Kim, D.; et al. Achieving higher efficiency on N2 reduction reaction through Mo‐ and Bi‐based active sites for sustainable photoelectrochemical ammonia production. Solar. RRL. 2024, 8, 2400386.
5. Ge, Q.; Liu, Y.; Li, K.; et al. Significant acceleration of photocatalytic CO2 reduction at the gas-liquid interface of microdroplets. Angew. Chem. Int. Ed. Engl. 2023, 62, e202304189.
6. Chen, H.; Wang, R.; Xu, J.; et al. Spontaneous reduction by one electron on water microdroplets facilitates direct carboxylation with CO2. J. Am. Chem. Soc. 2023, 145, 2647-52.
7. Wang, R.; Wang, S.; Zhu, C.; et al. Increased acidity of nitrogen heterocyclic compounds on water microdroplets facilitates CO2 capture. Chin. Chem. Lett. 2026, 37, 111547.
8. Shi, Z.; Nie, J.; Chen, Z. C.; et al. Synergistically activating N2 and CO2 at water microdroplet interfaces. Angew. Chem. Int. Ed. Engl. 2026, 65, e19068.
9. Spoorthi, B. K.; Debnath, K.; Basuri, P.; Nagar, A.; Waghmare, U. V.; Pradeep, T. Spontaneous weathering of natural minerals in charged water microdroplets forms nanomaterials. Science 2024, 384, 1012-7.
10. Mohajer, M. A.; Basuri, P.; Evdokimov, A.; et al. Spontaneous formation of urea from carbon dioxide and ammonia in aqueous droplets. Science 2025, 388, 1426-30.
11. Shi, L.; LaCour, R. A.; Qian, N.; et al. Water structure and electric fields at the interface of oil droplets. Nature 2025, 640, 87-93.
13. Basuri, P.; Mukhopadhyay, S.; Reddy, K. S. S. V. P.; et al. Spontaneous α-C-H carboxylation of ketones by gaseous CO2 at the air-water interface of aqueous microdroplets. Angew. Chem. Int. Ed. Engl. 2024, 63, e202403229.
14. Mehndiratta, L.; Wang, J.; Slade, J. H.; Grassian, V. H. Ester formation in alcohol microdroplet sprays: enhanced reactivity of C8 to C16 carboxylic acids with C1 to C3 alcohols and the effect of water. J. Phys. Chem. A. 2025, 129, 10807-18.
15. Liu, X.; Li, X.; Wang, X.; et al. Scale-up of microdroplet reactors for efficient CO2 resource utilization. J. Am. Chem. Soc. 2025, 147, 3529-38.
16. Tao, P.; Li, Q.; Fang, H.; et al. High-speed pre-self-rotation of amine microdroplets in cyclones to enhance CO2 capture. Sep. Purif. Technol. 2025, 360, 131045.
17. Huang, K. H.; Wei, Z.; Cooks, R. G. Accelerated reactions of amines with carbon dioxide driven by superacid at the microdroplet interface. Chem. Sci. 2020, 12, 2242-50.
18. Feng, L.; Yin, X.; Tan, S.; et al. Ammonium bicarbonate significantly accelerates the microdroplet reactions of amines with carbon dioxide. Anal. Chem. 2021, 93, 15775-84.
19. Meng Y, Gnanamani E, Zare R N. One-step formation of pharmaceuticals having a phenylacetic acid core using water microdroplets. J. Am. Chem. Soc. 2023, 145, 7724-8.
20. Bose, S.; Mehrgardi, M. A.; Zare, R. N. Selective photochemical conversion of carbon dioxide to formic acid at gas-water interface of microbubbles. J. Am. Chem. Soc. 2025, 147, 27449-57.
21. Chen, W.; Liu, J.; Jiang, Q.; et al. Silica-driven CO2 reduction in water microdroplets. J. Am. Chem. Soc. 2025, 147, 25287-94.
22. Gong, K.; Meng, Y.; Zare, R. N.; Xie, J. Molecular mechanism for converting carbon dioxide surrounding water microdroplets containing 1,2,3-triazole to formic acid. J. Am. Chem. Soc. 2024, 146, 8576-84.
23. Song, X.; Meng, Y.; Zare, R. N. Spraying water microdroplets containing 1,2,3-triazole converts carbon dioxide into formic acid. J. Am. Chem. Soc. 2022, 144, 16744-8.
24. Zheng, L.; Liu, X.; Yang, G.; et al. Highly efficient synthesis of cyclic carbonates via deep eutectic solvents from CO2 at the gas-liquid interface of microdroplet under atmospheric pressure condition. Chem. Eng. Sci. 2024, 289, 119867.
25. Song, X.; Basheer, C.; Zare, R. N. Making ammonia from nitrogen and water microdroplets. Proc. Natl. Acad. Sci. U. S. A. 2023, 120, e2301206120.
26. Song, X.; Basheer, C.; Xia, Y.; et al. One-step formation of urea from carbon dioxide and nitrogen using water microdroplets. J. Am. Chem. Soc. 2023, 145, 25910-6.
27. Wu, Y.; Luo, Z.; Yang, Y.; Wu, J.; Jia, X.; Wang, F. Nitrate synthesis from charged water microdroplets and dinitrogen. J. Energy. Chem. 2025, 108, 743-8.
28. Yabushita, A.; Enami, S.; Sakamoto, Y.; Kawasaki, M.; Hoffmann, M. R.; Colussi, A. J. Anion-catalyzed dissolution of NO2 on aqueous microdroplets. J. Phys. Chem. A. 2009, 113, 4844-8.
29. Wang, Y.; Luo, J.; Fang, Y. G.; et al. Catalyst-free nitrogen fixation by microdroplets through a radical-mediated disproportionation mechanism under ambient conditions. J. Am. Chem. Soc. 2025, 147, 2756-65.
30. Eatoo, M. A.; Mishra, H. Disentangling the roles of dissolved oxygen, common salts, and pH on spontaneous hydrogen peroxide production in water: No O2, No H2O2. J. Am. Chem. Soc. 2025, 147, 35392-400.
31. Lee, J. K.; Walker, K. L.; Han, H. S.; et al. Spontaneous generation of hydrogen peroxide from aqueous microdroplets. Proc. Natl. Acad. Sci. U. S. A. 2019, 116, 19294-8.
32. Zhou, K.; Su, H.; Gao, J.; et al. Deciphering the kinetics of spontaneous generation of H2O2 in individual water microdroplets. J. Am. Chem. Soc. 2024, 146, 2445-51.
33. Krushinski, L. E.; Dick, J. E. Direct electrochemical evidence suggests that aqueous microdroplets spontaneously produce hydrogen peroxide. Proc. Natl. Acad. Sci. U. S. A. 2024, 121, e2321064121.
34. Lee, K.; Mehrgardi, M. A.; Zare, R. N. Interfacial curvature, not simply size, controls spontaneous hydrogen peroxide formation in water microdroplets. J. Am. Chem. Soc. 2025, 147, 33240-7.
35. Chen, B.; Xia, Y.; He, R.; et al. Water-solid contact electrification causes hydrogen peroxide production from hydroxyl radical recombination in sprayed microdroplets. Proc. Natl. Acad. Sci. U. S. A. 2022, 119, e2209056119.
36. Zhou, X.; Du, S.; Zhang, W.; Zheng, B. Deciphering the mechanism of hydrogen peroxide formation in ultrasound-mediated water-in-oil microdroplets. Chem. Sci. 2025, 16, 6450-7.
37. Li, K.; Ge, Q.; Liu, Y.; et al. Highly efficient photocatalytic H2O2 production in microdroplets: accelerated charge separation and transfer at interfaces. Energy. Environ. Sci. 2023, 16, 1135-45.
38. Yang, S. Y.; Wang, W.; Chen, J. J.; Francisco, J. S.; Liu, X. W. Probing catalyst-free hydroxyl radical generation at microbubble interfaces. Nat. Commun. 2025, 16, 8835.
39. Zheng, X.; Fang, Y. G.; Tan, M.; et al. Accelerated methane photo-oxidation at the air-water interface. J. Am. Chem. Soc. 2025, 147, 26635-42.
40. Yan, X.; Lai, Y. H.; Zare, R. N. Preparative microdroplet synthesis of carboxylic acids from aerobic oxidation of aldehydes. Chem. Sci. 2018, 9, 5207-11.
41. Mofidfar, M.; Mehrgardi, M. A.; Zare, R. N. Water microdroplets surrounded by alcohol vapor cause spontaneous oxidation of alcohols to organic peroxides. J. Am. Chem. Soc. 2024, 146, 18498-503.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].