Structure-activity relationships of Fe/N-doped carbon catalysts for electrochemical hydrodechlorination
Abstract
Electrochemical hydrodechlorination (EHDC) offers a sustainable approach to convert highly toxic chlorinated volatile organic compounds into value-added hydrocarbons. Although Fe/N-doped carbon (NC) catalysts are effective EHDC catalysts, their structure-activity relationships remain unclear. In this work, Fe/NC catalysts with tunable Fe loading and structure, from Fe−N−C single-atom catalysts (SACs) to Fe3O4 nanoparticles/NC, were systematically prepared through an active-site imprinting strategy. They were used as electrodes for EHDC of chloroform (CHCl3). It is found that the EHDC performance of these Fe/NC catalysts follows a volcano trend with Fe content. Among them, the Fe/NC-10 catalyst with a high content of atomically dispersed Fe−N4 sites delivers optimal performance, achieving a methane (CH4) Faradaic efficiency of 44.4% and a yield rate of 60.1 μmol·h-1·cm-2, which are much higher than those of Fe3O4 nanoparticles/NC catalysts. Mechanistic studies reveal that the adsorbed atomic hydrogen (H*) species play a key role in the stepwise EHDC, and Fe−N4 sites can effectively promote the generation of H*. Density functional theory calculations further confirmed that Fe−N4 sites show a lower free energy barrier for EHDC and a stronger electron supply capacity than Fe3O4 nanoparticles. Additionally, the excellent catalytic performance can be maintained in a wide range of CHCl3 concentrations and pH values. This work clearly clarifies the structure-activity relationship of Fe/NC catalysts for EHDC and provides a foundation for rational design of advanced electrocatalysts.
Keywords
INTRODUCTION
Chlorinated volatile organic compounds (CVOCs) have been extensively released into environmental media due to human production activities[1,2]. The highly toxic CVOCs exhibit acute health hazards and endanger the atmospheric environment[3,4]. Various techniques have been applied for the removal of these pollutants, including adsorption[5], catalytic combustion[6], advanced oxidation processes[7], and photocatalysis[8]. Alternatively, electrochemical hydrodechlorination (EHDC) provides a sustainable approach to transform CVOCs via breaking C−Cl bonds while producing higher-value hydrocarbon compounds[9,10]. However, the reduction potential of EHDC is very negative due to the high bond energy of C−Cl. This results in a very low Faradaic efficiency (FE) of EHDC, where the competition of hydrogen evolution reaction (HER) is very severe[11,12]. Therefore, it is imperative to develop advanced catalysts that can effectively activate the C−Cl bond.
Fe/N-doped carbon (NC) catalysts are widely recognized as prospective catalysts for electrochemical reactions due to their good catalytic activity, low cost, and environmental friendliness[13,14]. A large number of Fe/NC catalysts, including Fe3O4 nanoparticles/NC[15], and Fe−N−C single-atom catalysts (SACs)[16], have been reported to possess excellent performance in the EHDC process. Although all of these catalysts show outstanding catalytic performance, they have different structures. As we all know, different structures can lead to changes in material properties, including electronic structure, coordination structure, crystal structure, etc.[17]. These changes will further affect the catalytic performance, and they may either enhance the performance or have the opposite effect. Traditional Fe-based nanoparticles/NC materials possess certain advantages, such as high dispersion of active components, simple preparation methods, and ease of industrialization[18,19]. However, numerous Fe atoms are deeply embedded within the lattice and thus inaccessible to reactants, leading to low atomic utilization[20,21]. Furthermore, compositional inhomogeneity and surface structural complexity of Fe nanomaterials lead to indeterminate reaction pathways and fluctuating activities[22,23]. Fe−N−C SACs, characterized by atomically dispersed metal centers, ensure uniformity of active sites and enable precise regulation of their coordination environments[24-27], offering an ideal platform to optimize the adsorbed atomic hydrogen (H*) generation capability. However, the increase in metal loading, that is, the density of active sites, has always been a challenge[28]. Recent advances in Fe/NC catalysis have centered on structural modulation strategies, including coordination environment engineering, active site density regulation and morphology optimization, to boost C−Cl bond activation and suppress the competitive HER[16,21,29]. At present, little progress has been made in the systematic study of the influence of catalyst structure on EHDC performance. Thus, investigating the structure-activity relationship of Fe/NC catalysts for EHDC is crucial, as well as for designing robust and highly efficient carbon-supported metal catalysts.
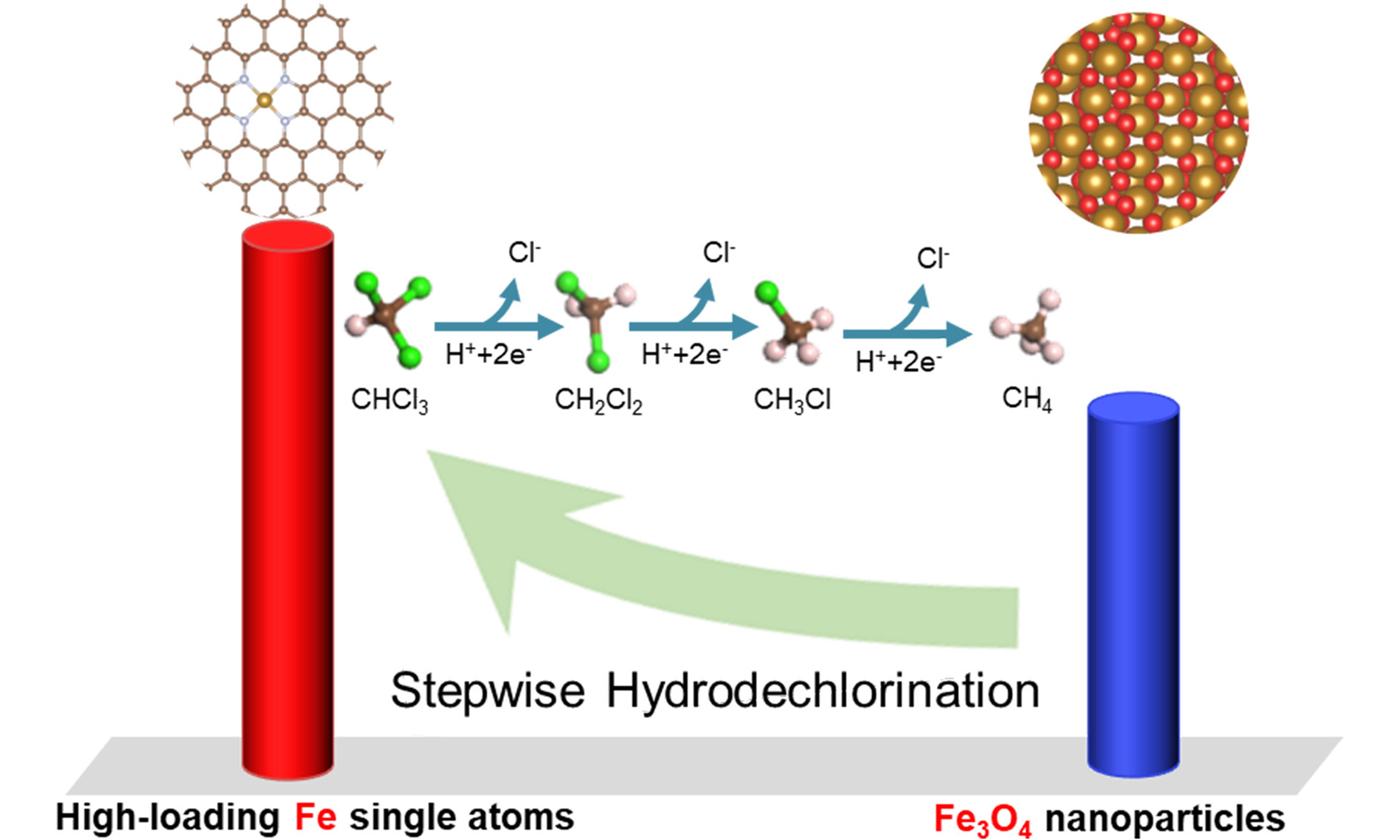
In this work, a set of Fe/NC catalysts with various Fe loadings and structures, including Fe–N–C SACs and Fe3O4 nanoparticles/NC, were prepared through active-site imprinting, in which the amount of Fe species added was carefully adjusted (1-20 wt.%). These materials were applied as electrocatalysts to systematically investigate the structure-activity relationship of Fe/NC catalysts for EHDC of chloroform (CHCl3). With increasing Fe addition, the EHDC performance follows a volcano-shaped trend. Among them, Fe/NC-10 with abundant Fe−N4 sites exhibits the optimal EHDC performance. Experimental tests and density functional theory (DFT) calculations confirm that H* acts as the key active species, and the EHDC of CHCl3 follows a stepwise hydrodechlorination reaction pathway. Fe−N4 sites in Fe−N−C SACs display a lower kinetic barrier for the rate-limiting step. Notably, Fe/NC-10 maintains excellent performance across a broad range of CHCl3 concentrations and pH values. This work not only establishes a clear proof for the structure-activity relationship of Fe/NC catalysts for EHDC, but also provides theoretical guidance for designing advanced catalysts.
EXPERIMENTAL
Chemicals
Ethanol absolute (C2H6O, > 99%), methanol (CH4O, ≥ 99.9%), zinc nitrate hexahydrate (AR, ≥ 99.99%), 2-methylimidazole (AR, ≥ 98%), ferrous chloride tetrahydrate (FeCl2⋅4H2O, > 98%), sodium sulfate (Na2SO4, 98%), sodium hydroxide (NaOH, > 98%), 5,5-dimethyl-1-pyrroline-N-oxide (DMPO, > 95%), anhydrous ethanol (C2H6O, > 99%), tert-butanol (TBA, ≥ 99%), and sulfuric acid (H2SO4, 99%) were purchased from Aladdin Reagent Co., Ltd. Nafion solution (5 wt.%) was obtained from Sigma-Aldrich (USA). Nafion N117 cation exchange membrane was purchased from DuPont Co., Ltd. (USA). All the chemicals were in analytical grade and used as received without further purification.
Synthesis of Fe/NC
A NC framework with Zn–Nx sites was prepared via the pyrolysis of zeolitic imidazolate framework-8 (ZIF-8), based on reported protocols[29,30]. Zn(NO3)2·6H2O (2.97 g, 10.0 mmol) was dissolved in 200 mL of methanol. Subsequently, this solution was added dropwise into 200 mL of methanol containing 3.28 g of 2-methylimidazole (40 mmol) under magnetic stirring at 400 rpm at room temperature, followed by maintaining the mixture at 50 °C for 3 h. Subsequently, the solid was washed with methanol, and dried at 60 °C in a vacuum to generate ZIF-8. The ZIF-8 was annealed at 700 °C (3 °C·min-1) for 1 h under N2 gas flow to form Zn/NC. The Zn/NC was acid-leached under reflux in a 2 M H2SO4 solution for 18 h to obtain NC. Afterwards, to synthesize the Fe/NC catalysts, a novel two-step annealing method was applied[31]. NC (100 mg) was added into 20 mL of methanol with a certain amount of FeCl2⋅4H2O. After 10 min of sonication, the product was dried by rotary evaporation and further in an oven at 80 °C. The dry powders were annealed under N2 gas flow at 300 °C (5 °C·min-1). After thorough washing with H2O-EtOH and drying at 80 °C, the product was annealed at a high temperature of 550 °C (2 °C·min-1) for 5 h in a N2 flow. The as-prepared catalysts were denoted as Fe/NC-x (x = 0, 1, 5, 10, 15, 20), with x representing the mass fraction of iron in weight percent.
Electrochemical measurements
EHDC measurements were performed in a typical H-cell separated by a cation-exchange membrane (Nafion N117). An Ag/AgCl electrode and a platinum sheet served as the reference and counter electrodes. Carbon cloth loaded with the as-prepared catalyst was used as the working electrode. The catalyst ink was prepared by sonicating 5 mg of catalyst in a mixture of 0.95 mL isopropyl alcohol and 50 μL Nafion. A 100 μL suspension was coated onto the carbon cloth with a geometric area of 1.0 cm2, achieving a catalyst loading of 0.5 mg·cm-2. For the EHDC, 0.1 M Na2SO4 electrolyte was added equally into the cathode and anode compartments. Before the electrochemical tests, Ar gas was bubbled through the solution to eliminate dissolved oxygen. The working electrode was activated by cyclic voltammetry (CV) scans at a scan rate of
RESULTS AND DISCUSSION
Preparation and characterization of Fe/NC
The preparation route of Fe/NC is shown in Figure 1A. The active-site imprinting strategy not only facilitates the regulation of the Fe loading amount, but also effectively eliminates the influence of the carbon support. Therefore, the materials prepared by this method are highly conducive to the study of the structure-activity relationship of catalysts. Scanning electron microscopy (SEM) reveals the retained rhombic dodecahedral morphology of prepared Fe/NC after multiple calcination steps [Figure 1B and Supplementary Figure 1], which is further proved using transmission electron microscopy (TEM) [Figure 1C and D]. In addition, TEM images indicate that no nanoparticle exists in the Fe/NC-10, while clear metal nanoparticles are found to adhere closely to the NC surface in the Fe/NC-15. The measured lattice spacing of the nanoparticle is
Figure 1. (A) Schematic illustration for the synthesis of Fe/NC; (B) SEM image, and (C) TEM images of Fe/NC-10; (D and E) TEM images of Fe/NC-15; (F) HAADF-STEM image of Fe/NC-10; (G) EDS mapping of Fe/NC-10, C (red), N (orange), and Fe (yellow). Three-dimensional structural models were constructed using 3ds Max 2018. SEM: Scanning electron microscopy; TEM: transmission electron microscopy; HAADF-STEM: high-angle annular dark-field scanning transmission electron microscopy; EDS: energy-dispersive spectroscopy.
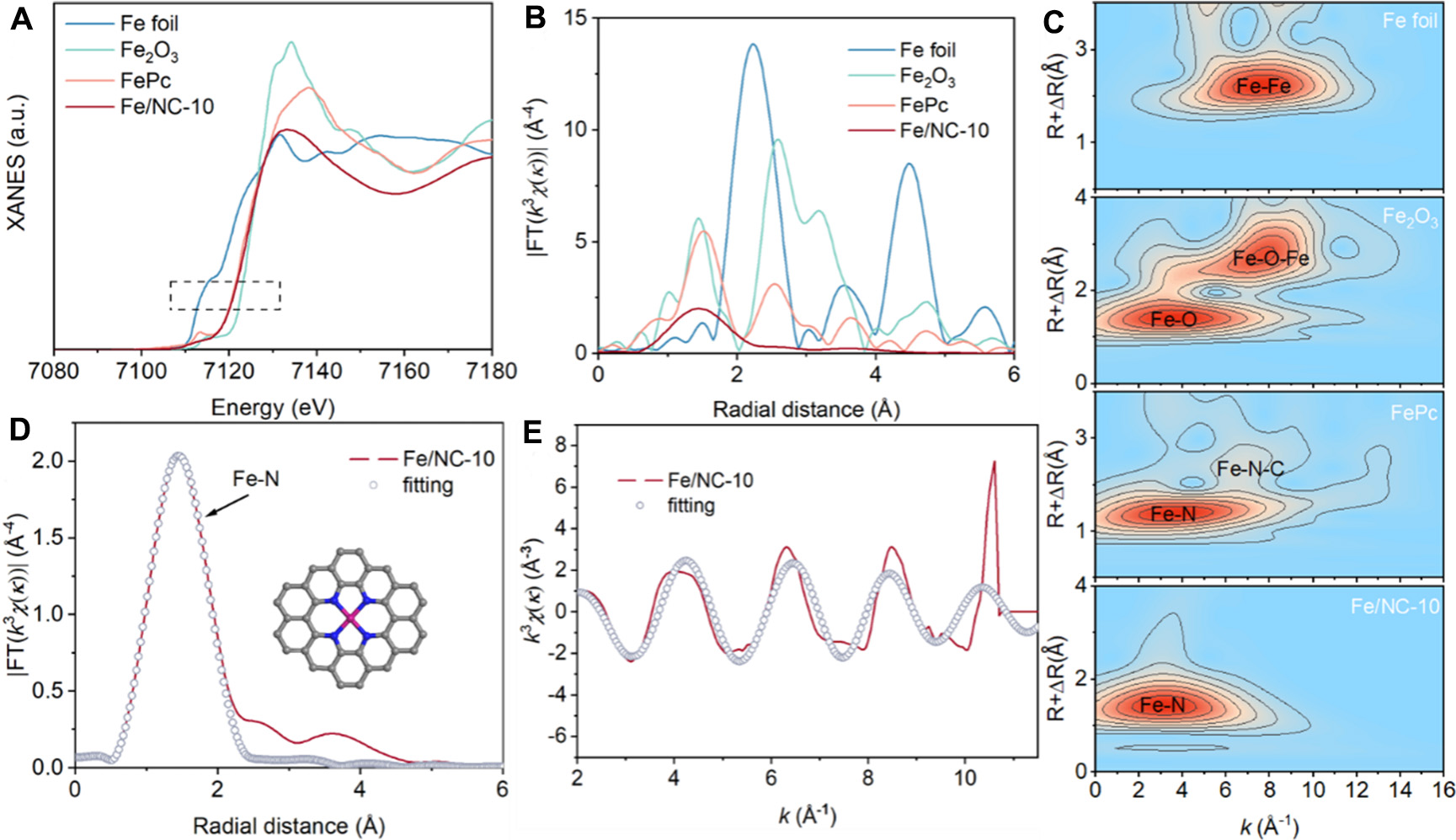
To validate the coordination structure of Fe in Fe/NC-10, X-ray absorption spectroscopy (XAS) analysis was employed. In the Fe K-edge X-ray absorption near-edge structure (XANES) spectra, the energy absorption edge is located between that of Fe foil and Fe2O3, suggesting that the oxidation state of Fe is between +2 and +3 [Figure 2A]. The Fourier transformed X-ray absorption fine structure (FT-EXAFS) spectrum of Fe/NC-10 exhibits a characteristic peak at ~1.48 Å, ascribed to the Fe−N coordination [Figure 2B]. Besides, the wavelet transform (WT) of Fe K-edge EXAFS oscillations shows only one intensity maximum at ~4 Å-1, confirming the atomic dispersion of Fe sites [Figure 2C]. This is further proved by the FT-EXAFS analysis in k space [Supplementary Figure 3]. Furthermore, FT-EXAFS fitting in R space and k space indicates that each Fe atom is coordinated with 4 N atoms, suggesting that the proposed coordination structure of Fe/NC-10 is Fe−N4 [Figure 2D and E]. The detailed EXAFS fitting parameters for Fe/NC-10 and reference samples are summarized in Supplementary Table 1. The oxidation state and composition of Fe in Fe/NC-15 were also measured by X-ray photoelectron spectroscopy (XPS). The Fe 2p spectra show a pair of peaks at 710.9 and 714.0 eV, which are assigned to Fe2+ and Fe3+ species [Supplementary Figure 4][32], further confirming the formation of Fe3O4 nanoparticles. The actual Fe contents in Fe/NC-1, Fe/NC-5, Fe/NC-10, Fe/NC-15, and Fe/NC-20 determined by Inductively Coupled Plasma Atomic Emission Spectroscopy (ICP-AES, Supplementary Table 2) are 1.04, 4.06, 8.48, 10.48, and 11.08 wt.%, respectively. Based on the above results, the Fe species in Fe/NC-1, Fe/NC-5 and Fe/NC-10 exist in the form of atomically dispersed Fe−N4 structure, and Fe species in Fe/NC-15 and Fe/NC-20 exist as Fe3O4 nanoparticles. The BET (Brunauer-Emmett-Teller) specific surface areas of Fe/NC-10 and Fe/NC-15 are 682.47 and 669.75 m2·g-1, respectively. The pore size distribution curves [Supplementary Figure 5] show that both catalysts possess a dominant mesopore size centered at ~4 nm, which promotes the mass transfer of reactants during the EHDC process.
Figure 2. (A) Fe K-edge XANES spectra, (B) FT-EXAFS spectra in R space, and (C) WT-EXAFS spectra of Fe foil, Fe2O3, FePc, and Fe/NC-10. (D and E) FT-EXAFS fitting curves of Fe/NC-10 in (d) R space and (E) k space. The inset is the schematic model of Fe−N4 SACs: Fe (purple), N (blue), and C (gray). Data plotting was performed using Origin 2022. XANES: X-ray absorption near-edge structure; FT-EXAFS: Fourier transformed X-ray absorption fine structure; WT: wavelet transform; SACs: single-atom catalysts.
EHDC performances of Fe/NC
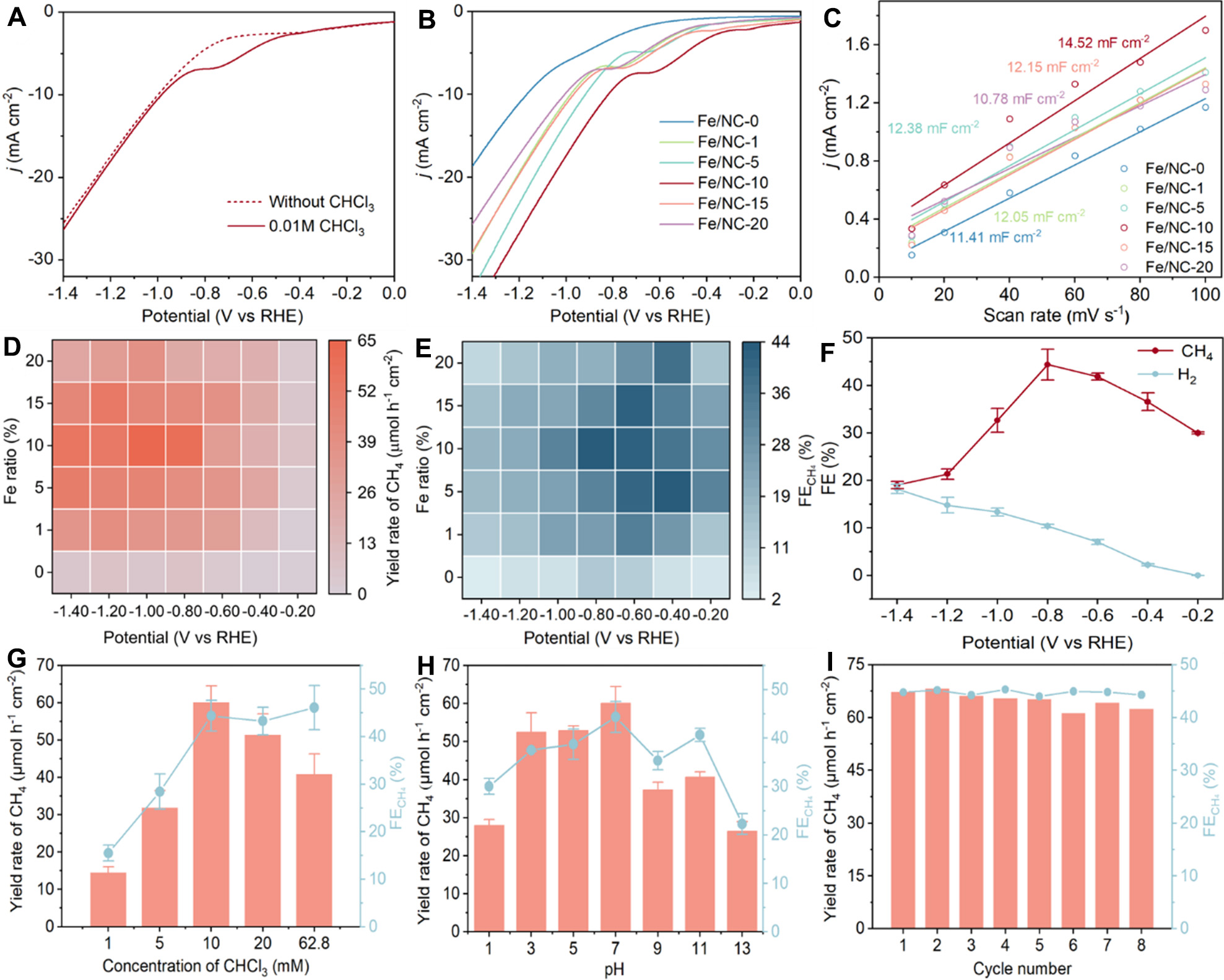
The EHDC performances were measured with 0.1 M Na2SO4 electrolyte. Firstly, in the linear sweep voltammetry (LSV) curves of Fe/NC-10, an obvious reduction peak appears in the presence of 0.01 M CHCl3, which can be assigned to the hydrodechlorination of CHCl3 [Figure 3A]. Meanwhile, among all Fe/NC catalysts, Fe/NC-10 demonstrated the lowest onset potential and the highest current density in the presence of CHCl3, indicating its superior electrocatalytic activity [Figure 3B]. Despite their similar physical surface areas, the electrochemical active surface areas (ECSA) differ substantially. Fe/NC-10 exhibits the largest electrochemical double layer capacitance (Cdl) value of 14.52 mF·cm-2 compared to other catalysts [Figure 3C and Supplementary Figure 6], indicating the largest electrochemical surface area and the most exposed active sites, thus promoting increased activity[33]. In contrast, Fe/NC-15 shows a significantly lower Cdl of
Figure 3. (A) LSV curves of Fe/NC-10 in the presence or absence of CHCl3; (B) LSV curves, (C) electrochemical active surface areas, heatmaps of (D) yield rate and (E) FE of Fe/NC catalysts with CHCl3; (F) FE of CH4 and H2 over Fe/NC-10. CH4 yield rate and FE of Fe/NC-10 at different (G) concentrations of CHCl3, (H) pH, and (I) cycling experiments. Error bars in (F-I) represent the standard deviation of three independent replicate experiments. Data plotting and fitting were performed using Origin 2022. LSV: Linear sweep voltammetry; FE: Faradaic efficiency; RHE: reversible hydrogen electrode.
Motivated by the above results, the catalytic activities were further evaluated by the constant potential tests. Heatmaps in Figure 3D and E clearly show the CHCl3 yield rate and FE over different Fe/NC catalysts at different potentials. Obviously, both the CH4 FE and the yield rate of Fe/NC catalysts also follow a volcano trend. Among them, Fe/NC-10 exhibits optimal performance, which achieves the highest FE of 44.4% with a corresponding yield rate of 60.1 μmol·h-1·cm-2 at -0.8 V (vs. RHE). The FE of Fe/NC-10 for the EHDC reaches 44.4%, which outperforms most reported electrocatalysts in the field of EHDC [Supplementary Table 3]. Despite the higher metal loading, Fe/NC-15 and Fe/NC-20 exhibit inferior catalytic activity, highlighting the advantages of single-atom Fe sites over Fe nanoparticles [Supplementary Table 2]. After normalization by the ECSA, the activity trend remains consistent, further demonstrating the superior performance of single-atom Fe sites [Supplementary Figure 7]. Meanwhile, the FE of HER during the EHDC process was also evaluated [Figure 3F]. The FE of H2 increased with increasing potential, which explains the decrease in EHDC FE at high potentials.
To further evaluate their industrial potential, a series of measurements were performed at -0.8 V (vs. RHE) using Fe/NC-10. Firstly, the effect of CHCl3 concentrations (1-62.8 mM) on the EHDC performance was investigated, revealing that high activity remained robust over this wide range [Figure 3G]. However, the CH4 yield rate is decreased when the concentration of CHCl3 exceeded 10 mM, because the high concentration of CHCl3 on the surface of the electrode led to insufficient production of H*. Subsequently, the EHDC performance at different pH values was also investigated [Figure 3H]. It is found that the yield and FE of CH4 remained at a relatively high level when the pH was 3. However, the performance declined significantly when the pH dropped to 1, because the high H⁺ concentration significantly enhanced HER competition. Additionally, the performance is also decreased under alkaline conditions, possibly because of the insufficient production of H*. The long-term electrochemical test indicates a satisfactory stability over
Reaction mechanism of EHDC over Fe/NC
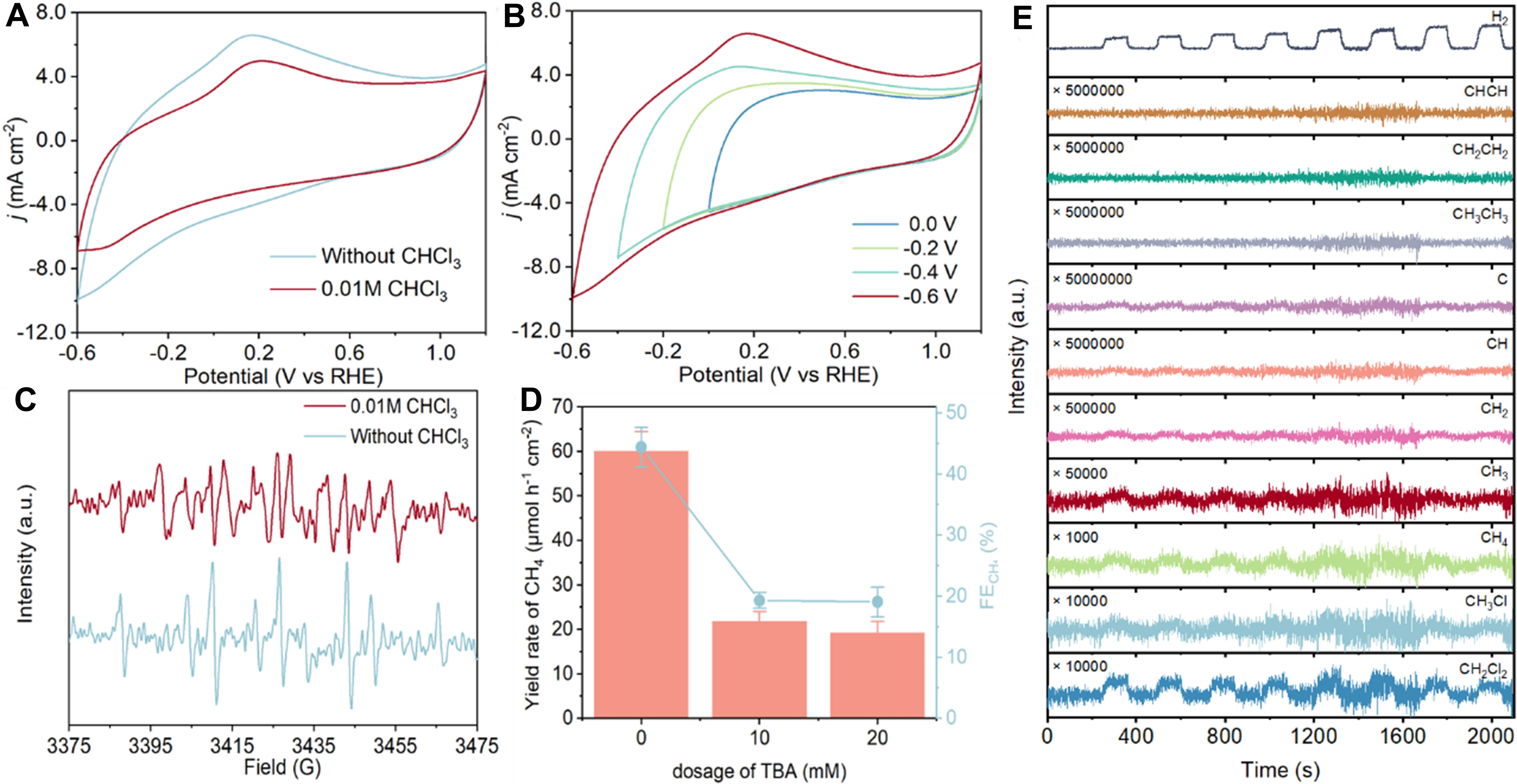
The reaction mechanism of EHDC over Fe/NC was carefully investigated by the combination of experiments and theoretical calculations. First of all, active species involved in the EHDC were identified by CV, electron paramagnetic resonance (EPR), and quenching experiments. The occurrence of the Volmer reaction was displayed through the peak of H* at around 0.2 V vs. RHE [Figure 4A][34]. With the presence of 0.01 M CHCl3, the intensity of the oxidation peak is decreased, indicating that H* participated in the EHDC. The mechanistic behavior of Fe/NC-10 was further probed through the CV at different ending potentials [Figure 4B][35]. An increasing trend in the intensity of the H* peak is observed with the ending potentials becoming more negative, suggesting that the negative potentials are beneficial to the production of H*. EPR analysis further suggests the significant role of H* in the EHDC process using DMPO as the capture agent, where the intensity of nine characteristic peaks is decreased in the electrolyte with 0.01 M CHCl3 [Figure 4C][36]. Further, the FE and yield rate of CH4 are decreased to 19.3% and 21.8 μmol·h-1·cm-2 in the quenching tests with the addition of 10 mM TBA [Figure 4D]. All of the above results suggest that H* acts as an important active species during the EHDC process. However, the hydrodechlorination process cannot be completely stopped even with the addition of a large amount of capturing agents, indicating the co-contribution from the direct reduction mechanism of electrons.
Figure 4. (A and B) CV curves of Fe/NC-10 in 0.1 M Na2SO4 electrolyte (A) with and without CHCl3, (B) with different ending potentials; (C) EPR spectra of Fe/NC-10 in 0.1 M Na2SO4 electrolyte with and without CHCl3 at -0.8 V vs. RHE; (D) CH4 yield rate and FE of Fe/NC-10 with the addition of TBA; (E) DEMS results for the EHDC of CHCl3 over Fe/NC-10. Error bars in (D) represent the standard deviation of three independent replicate experiments. Data plotting and fitting were performed using Origin 2022. CV: Cyclic voltammetry; EPR: electron paramagnetic resonance; RHE: reversible hydrogen electrode; FE: Faradaic efficiency; TBA: tert-butanol; DEMS: differential electrochemical mass spectrometry; EHDC: electrochemical hydrodechlorination.
To further determine the reaction mechanism, online differential electrochemical mass spectrometry (DEMS) was applied to identify the key intermediates and products of the EHDC over Fe/NC-10 [Figure 4E]. The clear signals for the m/z of 84.93, 50.49, and 16, corresponding to CH2Cl2, CH3Cl, and CH4, respectively, are detected. This indicates that the electrocatalytic transformation of CHCl3 is a stepwise hydrodechlorination process with no coupling reactions or dimerization reactions observed.
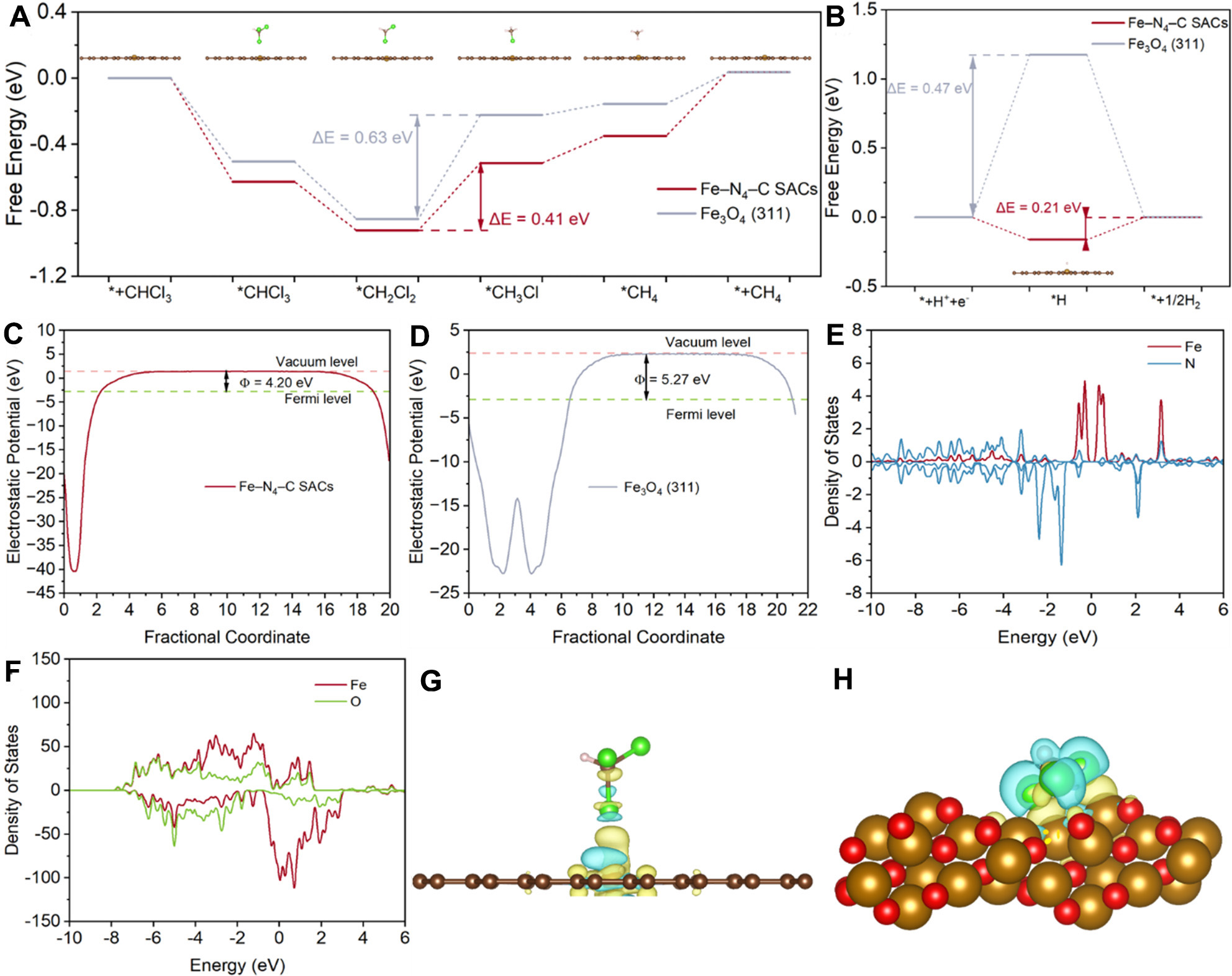
DFT calculations are employed to further investigate the structure-activity relationships of Fe/NC-based catalysts on EHDC of CHCl3. The geometric structure of Fe−N4−C SACs and Fe3O4 (311) was constructed [Figure 5A and Supplementary Figure 9]. The energy profile for EHDC of CHCl3 suggests that *CH2Cl2 → *CH3Cl is the rate-limiting step for Fe−N−C SACs and Fe3O4 nanoparticles. The energy barrier of Fe−N4 site is only 0.41 eV, which is relatively lower than that of Fe3O4 (311) [Figure 5A and Supplementary Table 4]. This is consistent with the high EHDC performance of Fe−N4−C SACs. Meanwhile, the energy profile for HER indicates that the generation of H* is more facile on Fe−N4−C SACs than that on Fe3O4 nanoparticles [Figure 5B], where H* has been demonstrated as the main active species above.
Figure 5. (A and B) The calculated energy diagrams for (A) EHDC of CHCl3 to CH4, and (B) HER on Fe−N4−C SACs and Fe3O4 (311); (C and D) The work functions of (C) Fe−N4−C SACs and (D) Fe3O4 (311); (E and F) The density of states of (E) Fe−N4−C SACs and (F) Fe3O4 (311); (G and H) The charge density difference mappings of (G) Fe−N4−C SACs and (H) Fe3O4 (311). Data plotting and fitting were performed using Origin 2022. Crystal structure visualization was performed using VESTA. EHDC: Electrochemical hydrodechlorination; HER: hydrogen evolution reaction; SACs: single-atom catalysts; VESTA: Visualization for Electronic and Structural Analysis.
The work functions of Fe−N4−C SACs and Fe3O4 (311) are 4.20 and 5.27 eV, respectively [Figure 5C and D]. A lower work function suggests stronger chemical activity and higher electron energy, facilitating the generation of H*. Additionally, this is consistent with the partial density of states (PDOS; Figure 5E and F), which shows a sharper and more intense density of states near the Fermi level for Fe−N4−C SACs. This electronic configuration signifies a greater population of readily available active electrons, which are crucial for the initial cleavage of the C−Cl bond. The finding is also proved by the calculated charge density difference mappings for EHDC of CHCl3 [Figure 5G and H]. Clearly, more electrons (yellow) are located between the Cl atom and the Fe site, which implies a strong interaction between them. Therefore, Fe−N4−C SACs show a much higher EHDC performance than Fe3O4 nanoparticles/NC.
CONCLUSIONS
In conclusion, to clearly investigate the structure-activity relationships of Fe/NC catalysts on EHDC of CHCl3, Fe/NC catalysts with different Fe loading and structure, including the Fe−N−C SACs and Fe3O4 nanoparticles/NC, were prepared and applied as electrodes for EHDC. The EHDC performance follows a volcano trend with the increase of Fe content, and the Fe/NC-10 with abundant Fe−N4 sites exhibits optimal performance for the EHDC of CHCl3 to CH4, which follows a stepwise hydrodechlorination pathway, and H* served as the key active species in this process. Moreover, theoretical calculations suggest that the energy barrier of EHDC over Fe−N4 sites is much lower than that on Fe3O4 nanoparticles, and the electron supply capacity of Fe−N4 sites is also much stronger than that of the Fe site in Fe3O4, which are the intrinsic reasons for the performance differences of Fe−N−C SACs and Fe3O4 nanoparticles/NC. Additionally, the excellent catalytic performance can be maintained in a wide range of CHCl3 concentrations and pH values. This work clearly clarifies the structure-activity relationship of Fe/NC catalysts for EHDC and provides a theoretical basis for the rational design of advanced electrocatalysts.
DECLARATIONS
Authors’ contributions
Made substantial contributions to conception and design of the study and performed data analysis and interpretation: Li, G.; Cao, X.; Wang, L.; Chen, P.; Lu, Y.; Liu, Z.; Sun, Y.; Ye, L.
Performed data acquisition and provided administrative, technical, and material support: Gan, G.; Hou, H.; Feng, X.; Bai, B.; He, C.
Availability of data and materials
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author(s).
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Nos. 52301261, 22476157, 22276145), the China Postdoctoral Science Foundation (Nos. 2025M771222, 2025T180017), the Postdoctoral Research Project Funding of Shaanxi Province (No. 2024BSHSDZZ214), and the Fundamental Research Funds for the Central Universities, CHD (No. 300102295301).
Conflicts of interest
He, C. is an Editorial Board Member of the journal Greenverse Science. He, C. was not involved in any step of the editorial process, notably including reviewer selection, manuscript handling, or decision-making. The other authors declare no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Chen, D.; Si, W.; Wang, R.; et al. Enhanced Ru–Ce interface electron transfer and reverse oxygen spillover promote chlorobenzene catalytic oxidation. Appl. Catal. B. Environ. Energy. 2025, 373, 125315.
2. Lin, F.; Chen, Z.; Gong, H.; Wang, X.; Qin, Y. A synergistic catalyst of Ni-β-Mo2C/γ-Al2O3 for robust syngas production via catalytic steam reforming of chlorinated volatile organic wastes. Appl. Catal. B. Environ. Energy. 2025, 370, 125181.
3. Zhao, Y.; Ye, Y.; Wu, Z.; et al. Manipulating weak interactions between host/guest and analytes in Cu(I)-cluster-based MOF for fluorescent gas sensing towards chlorinated volatile organic compounds. Chem. Eng. J. 2025, 506, 159923.
4. Ding, M.; Zhang, Y.; Guo, Y.; et al. Selective adsorption of chlorine species on RuO2 sites for efficient elimination of vinyl chloride on the Ru/SnO2 catalyst. Environ. Sci. Technol. 2025, 59, 956-67.
5. Zhao, Z.; Wu, W.; Li, W.; et al. Adsorption recovery of chlorinated volatile organic compounds on coffee ground-based activated carbon of tunable porosity. Sep. Purif. Technol. 2025, 354, 129271.
6. Ding, M.; Wang, Y.; Li, M.; et al. Sulfur-engineered Ru/SnO2 catalysts for highly efficient catalytic combustion of 1,2-dichloroethane. Environ. Sci. Technol. 2025, 59, 15445-56.
7. Xie, X.; Xiao, F.; Zhan, S.; et al. Deep oxidation of chlorinated VOCs by efficient catalytic peroxide activation over nanoconfined Co@NCNT catalysts. Environ. Sci. Technol. 2024, 58, 1625-35.
8. Wu, Q.; Yuan, D.; Wang, H.; et al. Ambient sunlight-driven high performance chlorinated volatile organic compound oxidation by Cu0.15Mn0.15Ce0.7Ox hollow spheres. Catal. Sci. Technol. 2023, 13, 1173-9.
9. Choi, C.; Wang, X.; Kwon, S.; et al. Efficient electrocatalytic valorization of chlorinated organic water pollutant to ethylene. Nat. Nanotechnol. 2023, 18, 160-7.
10. Gao, Y.; Zhang, W.; Choi, C.; et al. Effective electrochemical trichloroethylene removal from water enabled by selective molecular catalysis. Carbon. Futur. 2024, 1, 9200015.
11. Yu, W.; Jiang, H.; Fang, J.; Song, S. Designing an electron-deficient Pd/NiCo2O4 bifunctional electrocatalyst with an enhanced hydrodechlorination activity to reduce the consumption of Pd. Environ. Sci. Technol. 2021, 55, 10087-96.
12. Cheon, S.; Zhu, S.; Gao, Y.; et al. Neighboring catalytic sites are essential for electrochemical dechlorination of 2-chlorophenol. J. Am. Chem. Soc. 2024, 146, 25151-7.
13. Zhao, Z.; Yao, X.; Yu, R.; et al. Enhanced electrocatalytic hydrodechlorination by modulating metal-support interaction and H generation of single-Pd-atom anchored NiFeP electrode. Chem. Eng. J. 2024, 492, 152340.
14. Shen, X.; Xiao, F.; Zhao, H.; et al. In situ-formed PdFe nanoalloy and carbon defects in cathode for synergic reduction-oxidation of chlorinated pollutants in electro-fenton process. Environ. Sci. Technol. 2020, 54, 4564-72.
15. Xiao, Q.; Li, W.; Xie, S.; Wang, L.; Tang, C. Y. Ultrafast complete dechlorination enabled by ferrous oxide/graphene oxide catalytic membranes via nanoconfinement advanced reduction. Nat. Commun. 2024, 15, 9607.
16. Gan, G.; Li, X.; Wang, L.; et al. Active sites in single-atom Fe-Nx-C nanosheets for selective electrochemical dechlorination of 1,2-dichloroethane to ethylene. ACS. Nano. 2020, 14, 9929-37.
17. Koolen, C. D.; Luo, W.; Züttel, A. From single crystal to single atom catalysts: structural factors influencing the performance of metal catalysts for CO2 electroreduction. ACS. Catal. 2022, 13, 948-73.
18. Zhang, H.; Wang, C.; Luo, H.; Chen, J.; Kuang, M.; Yang, J. Iron nanoparticles protected by chainmail-structured graphene for durable electrocatalytic nitrate reduction to nitrogen. Angew. Chem. Int. Ed. Engl. 2023, 62, e202217071.
19. Xia, H.; Yang, M.; Zhou, X.; et al. Reshape iron nanoparticles using a zinc oxide nanowire array for high efficiency and stable electrocatalytic nitrogen fixation. ACS. Appl. Mater. Interfaces. 2025, 17, 7607-18.
20. Feijóo, J.; Yang, Y.; Fonseca Guzman, M. V.; et al. Operando high-energy-resolution X-ray spectroscopy of evolving Cu nanoparticle electrocatalysts for CO2 reduction. J. Am. Chem. Soc. 2023, 145, 20208-13.
21. Zhang, J.; Ma, Y.; Yang, J.; et al. Reexploring size-dependent catalytic performance under same metal loadings and identifying real active species: from single atom, cluster to nanoparticle. ACS. Nano. 2025, 19, 39701-13.
22. Peng, M.; Dong, C.; Gao, R.; Xiao, D.; Liu, H.; Ma, D. Fully exposed cluster catalyst (FECC): toward rich surface sites and full atom utilization efficiency. ACS. Cent. Sci. 2021, 7, 262-73.
23. Yang, S.; Liu, X.; Wang, X.; et al. High-entropy type Fe-Ni-P-O-C amorphous Nanospheres: remarkable Fe-ion migration induced efficient surface reconstruction for oxygen evolution reaction. Adv. Powder. Mater. 2025, 4, 100329.
24. Qiao, B.; Wang, A.; Yang, X.; et al. Single-atom catalysis of CO oxidation using Pt1/FeOx. Nature. Chem. 2011, 3, 634-41.
26. Raziq, F.; Feng, C.; Hu, M.; et al. Isolated Ni atoms enable near-unity CH4 selectivity for photothermal CO2 hydrogenation. J. Am. Chem. Soc. 2024, 146, 21008-16.
27. Kaiser, S. K.; Chen, Z.; Faust Akl, D.; Mitchell, S.; Pérez-Ramírez, J. Single-atom catalysts across the periodic table. Chem. Rev. 2020, 120, 11703-809.
28. Jiang, Y.; Chen, Z.; Peng, T.; et al. Single-atom Fe catalysts with improved metal loading for efficient ammonia synthesis under mild conditions. Angew. Chem. Int. Ed. Engl. 2025, 64, e202501190.
29. Mehmood, A.; Gong, M.; Jaouen, F.; et al. High loading of single atomic iron sites in Fe–NC oxygen reduction catalysts for proton exchange membrane fuel cells. Nat. Catal. 2022, 5, 311-23.
30. Menga, D.; Ruiz‐zepeda, F.; Moriau, L.; et al. Active-site imprinting: preparation of Fe–N–C catalysts from zinc ion–templated ionothermal nitrogen-doped carbons. Adv. Energy. Mater. 2019, 9, 1902412.
31. Hai, X.; Xi, S.; Mitchell, S.; et al. Scalable two-step annealing method for preparing ultra-high-density single-atom catalyst libraries. Nat. Nanotechnol. 2022, 17, 174-81.
32. Xiao, F.; Xu, G.; Sun, C.; et al. Nitrogen-coordinated single iron atom catalysts derived from metal organic frameworks for oxygen reduction reaction. Nano. Energy. 2019, 61, 60-8.
33. Sun, J.; Tang, T.; Zhang, S.; et al. A dual-atom La2 catalyst for the oxygen reduction reaction. Angew. Chem. Int. Ed. Engl. 2025, 64, e202509063.
34. Liu, R.; Zhao, H.; Zhao, X.; et al. Defect sites in ultrathin pd nanowires facilitate the highly efficient electrochemical hydrodechlorination of pollutants by H*ads. Environ. Sci. Technol. 2018, 52, 9992-10002.
35. Zeng, H.; Zhang, G.; Ji, Q.; et al. pH-independent production of hydroxyl radical from atomic H*-mediated electrocatalytic H2O2 reduction: a green fenton process without byproducts. Environ. Sci. Technol. 2020, 54, 14725-31.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].