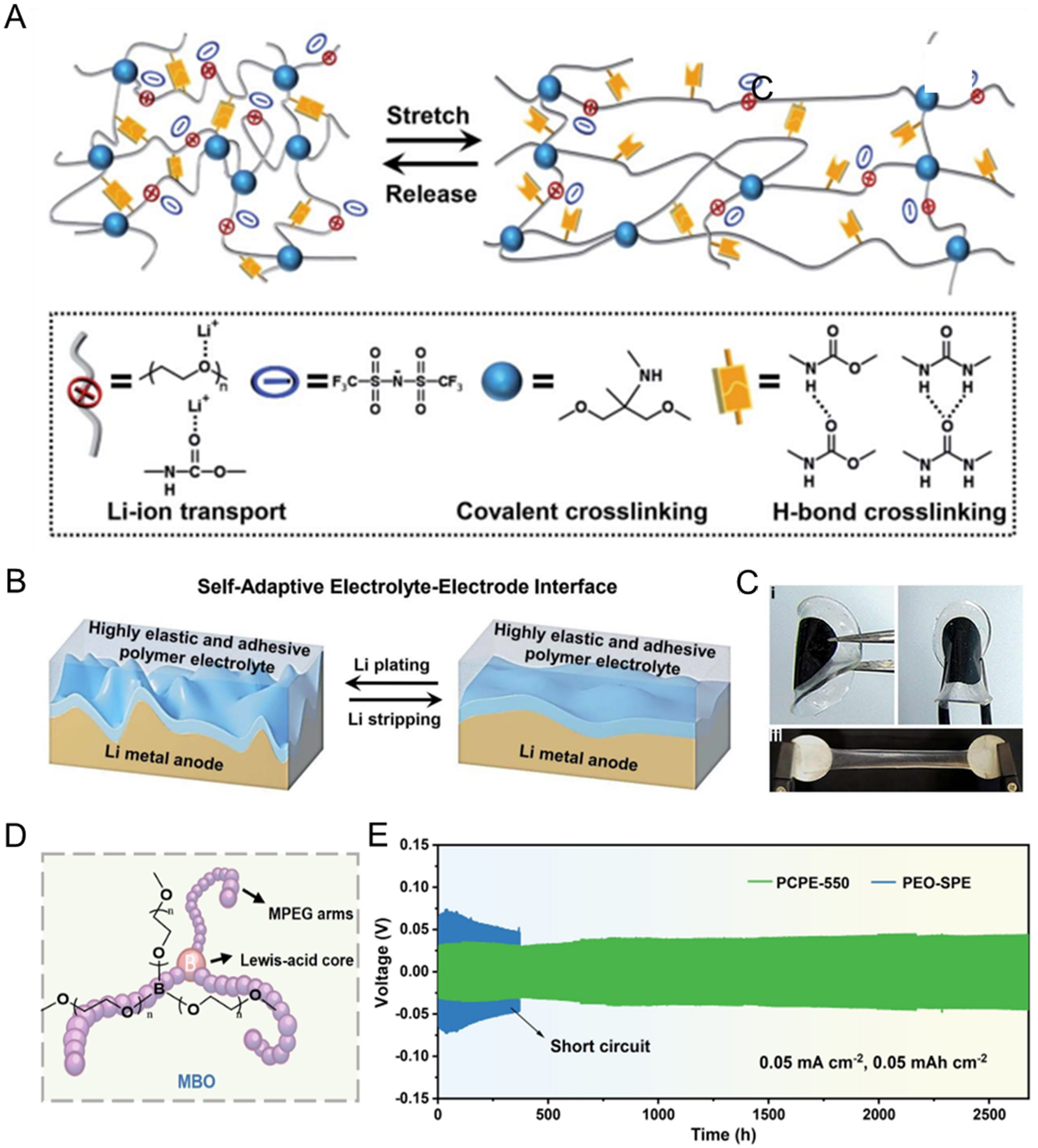
fig5
Figure 5. (A) Schematic illustration of the elastomeric polymer network of the PU electrolyte; (B) Schematic illustration of the self-adaptive electrolyte-electrode interface; (C) Photographs showing the adhesion of the PU-EO12/LiTFSI/TEG41% electrolyte to LiFePO4 cathode material. Figure 5A-C is reprinted with permission from Ref. [84], Copyright © 2021 Royal Society of Chemistry; (D) The chemical structure of electrolyte; (E) Long-term cycling performance of Li/Li symmetric cells using PCPE-550 and PEO-SPE at