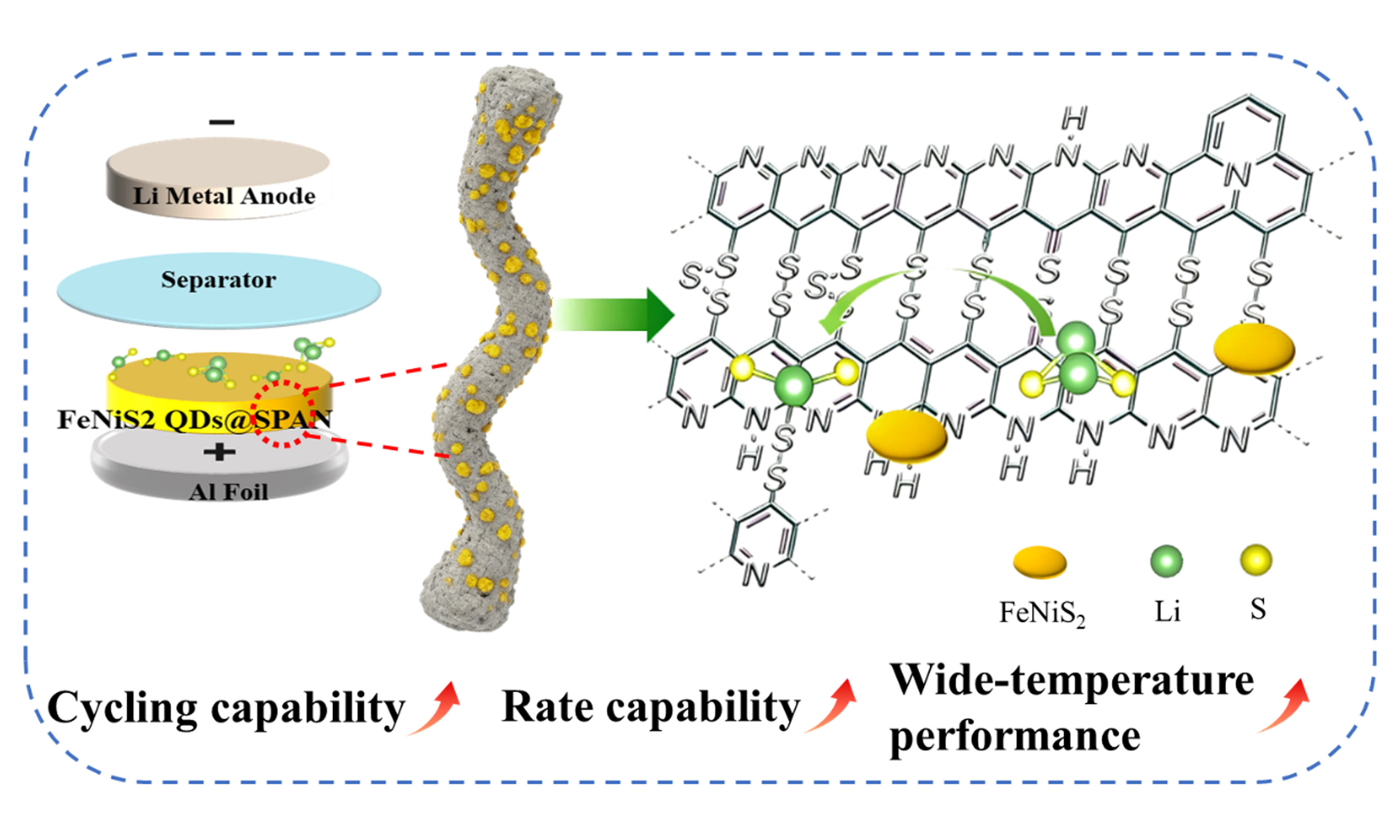
FeNiS2 quantum dot integration boosts enhanced reaction kinetics and cycle stability of SPAN nanofiber cathode for Li-S batteries
Abstract
Sulfurized polyacrylonitrile (SPAN) has been regarded as one of the most competitive cathode candidates for lithium-sulfur (Li-S) batteries, owing to its outstanding theoretical energy density, excellent structural durability, and minor self-discharge. Nevertheless, the intrinsically slow reaction kinetics of SPAN results in insufficient active sulfur utilization at high current rates, which severely restricts its rate performance and long-cycle stability. This study introduces FeNiS2 Quantum Dots (QDs) as catalyst embedding in SPAN nanofibers (FeNiS2 QDs@SPAN). Taking advantages of the ultra-small size, superior dispersibility and abundant catalytic sites of FeNiS2 QDs, the redox kinetics and cycle performance of SPAN are significantly enhanced. Kinetic analyses and theoretical calculation demonstrate the uniformly dispersed FeNiS2 QDs effectively reduce charge transfer resistance and facilitate conversion reaction. FeNiS2 QDs@SPAN material exhibits high reversible capacity of 1,213 mAh g-1 and an ultralow capacity decay of 0.034% per cycle over 1,000 cycles at 1 C. Remarkably, even at high rate of 5 C (8.37 A g-1), it delivers a stable long-cycle capacity of 720 mAh g-1 and demonstrates excellent cycling capability with a low fade rate of 0.029% per cycle over 450 cycles. FeNiS2 QDs@SPAN material maintains good performance even under lean electrolyte conditions and a wide temperature range. This work underscores the significant potential of FeNiS2 QDs as catalyst for achieving high performance sulfur cathode and advanced Li-S batteries.
Keywords
INTRODUCTION
Li-S batteries have emerged as a highly competitive electrochemical energy storage technology, mainly owing to their remarkable energy density (2,600 Wh kg-1) and the low cost of their raw materials[1-3]. However, the widespread commercialization of Li-S batteries is still hindered by several intrinsic drawbacks. The sulfur itself is electronically insulated, and the reaction between sulfur and lithium sulfide (Li2S) is sluggish. Particularly, the intermediate product lithium polysulfides (LiPSs) dissolve and diffuse in the ether electrolyte, and sulfur electrode suffers from severe shuttle effect, which leads to suboptimal utilization of active materials, poor cycle stability and even battery failure of the Li-S batteries[4-7]. Current research endeavors are aiming to mitigate the aforementioned concerns. Strategies such as physical confinement[8-10] and chemical absorption[11,12] of sulfur were used to suppress the shuttle effect. By introducing heterogeneous catalyst into the sulfur cathode, the redox reaction kinetics are accelerated[13,14]. Nevertheless, the unavoidable dissolution of long-chain LiPSs in ether-based electrolytes remains a critical issue.
As a sulfur-based cathode material, sulfurized polyacrylonitrile (SPAN) represents a promising candidate for Li-S batteries, which can be readily fabricated via heat treatment of polyacrylonitrile (PAN) under sulfur vapor[15,16]. Different from conventional sulfur-carbon composites prepared by physical blending, sulfur species in SPAN are covalently bonded to the conductive framework generated from PAN pyrolysis[17,18]. Although the precise molecular structure of SPAN has not been fully elucidated, it is widely accepted that sulfur is anchored onto the PAN-derived carbon support through covalent linkages. Accordingly, only short-chain LiPSs (Li2Sn, n ≤ 4) are involved in the electrochemical redox reactions upon cycling[19,20]. Thus, the notorious dissolution of LiPSs into electrolyte and shuttle effect of the sulfur cathode can be avoided. However, the slow kinetics of the solid-solid conversion reaction of SPAN would deteriorate the utilization of the sulfur and result in capacity fading of SPAN while cycling. In addition, the rate performance and cycle performance of the SPAN electrode under high rates are impacted as well[21]. To address the issue, electrocatalysts were explored and integrated into SPAN, which effectively anchors polysulfides and significantly improves the redox kinetics of SPAN electrode[22-24].
To optimize the cathode performance of Li-SPAN batteries, various catalytic materials have been developed to enhance the redox kinetics, including transition metal carbides, transition metal oxides, and transition metal sulfides. Among these candidates, transition metal sulfides presents combined merits, such as relatively higher electronic conductivity and good polar affinity for sulfur species, thus enabling dual functions of anchoring short-chain Li2Sn and catalyzing their solid-solid conversion reactions during the operation of Li-S batteries[25-28]. For instance, Li et al.[29] incorporated cobalt sulfide (CoS2) into SPAN, which enhanced its reactive activity and cycling stability, allowing the material to sustain over 500 cycles at 1 C. Liu et al.[30] synthesized NiS2-SPAN powders using a simple co-heating method, which exhibited superior rate capabilities with a NiS2 content of less than 3 wt%. Although transition metal sulfides are the most widely investigated category of metal sulfides for SPAN cathode modification, binary transition metal sulfides exhibit more abundant electrochemical active sites and more reactive polar surfaces compared with single transition metal sulfides[31,32]. This enables stable, rapid charge transport and sustained catalytic effects through multicomponent synergy and regulation of electronic band structures, which would boost the reaction kinetics[33,34]. Furthermore, to maximize the adsorption sites and catalytic efficiency for LiPSs, quantum dots (QDs), characterized by ultra-small size and superior dispensability, are particularly effective[35]. These QDs would significantly enhance the host-guest interactions, lower the reaction energy barrier, and provide exceptional catalytic effects on LiPSs[36,37]. Yet, the application of binary-TMS (transition metal sulfides)-based QDs to modify SPAN for boosting the electrochemical properties of Li-SPAN batteries remains rarely reported.
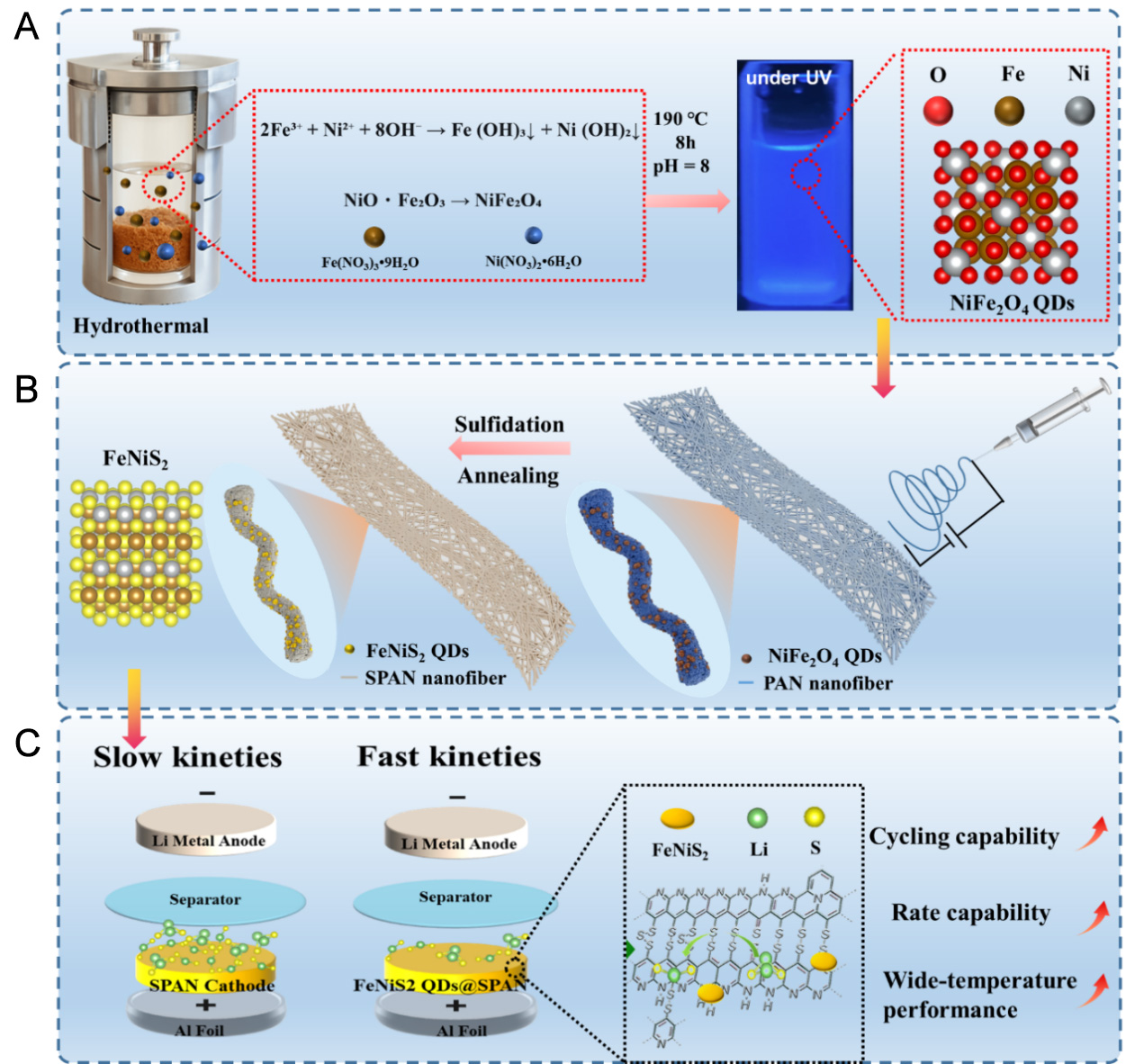
In this study, we developed a novel FeNiS2 QDs@SPAN composite by blending PAN/NiFe2O4 QDs nanofibers incorporated with sulfur powder, followed by a heat treatment. This process initiates polymerization and dehydrogenation reactions between PAN and sulfur, leading to the formation of pyridine ring structures. In the meantime, the NiFe2O4 QDs is sulfurized as FeNiS2. The uniform dispersion of FeNiS2 QDs throughout the composite increases the number of active sites, which enhances the charge transfer and accelerates the conversion of short-chain polysulfides. These improvements are crucial for optimizing the redox reaction kinetics. Meanwhile, the one-dimensional (1D) nanofibers maintain the structural integrity of the composite and provide short-distance diffusion pathways for ions. Benefiting from these synergistic advantages, the FeNiS2 QDs@SPAN nanofibers exhibit excellent cycling stability and rate performance. Notably, the FeNiS2 QDs@SPAN electrode achieves superior electrochemical performance at high rates, which is rarely reported for SPAN-based electrodes. It also maintains outstanding performance metrics across a wide temperature range and under lean electrolyte conditions, demonstrating great potential for practical application.
EXPERIMENTAL
Materials and methods
Preparation of NiFe2O4 QDs
All chemicals were of analytical grade or higher and used without further purification. [Fe(NO3)3·9H2O,
Approximately 10 mM of Fe(NO3)3·9H2O and 5 mM of Ni(NO3)2·6H2O were dissolved in 80 mL of deionized water. This solution was ultrasonicated for 30 min. Under ambient conditions, concentrated ammonia was added dropwise during magnetic stirring until the pH reached 8[35]. After an additional 10 min of stirring, the mixture was transferred to a Polytetrafluoroethylene (PTFE)-lined autoclave and heated at 190 °C for 8 h. Once cooled to room temperature, the upper layer of the resultant brown transparent liquid was collected via centrifugation and freeze-dried for later use.
Synthesis of PAN/NiFe2O4 QDs nanofibers
A mixture of 50 mg of NiFe2O4 QDs and 1 g of PAN was dispersed in 8.5 g of DMF and ultrasonicated for
Synthesis of FeNiS2 QDs@SPAN composites
Sublimed sulfur and PAN/NiFe2O4 QDs nanofibers were placed into a Flange high-pressure reaction tank. The mixture was heated under an argon atmosphere at 155 °C for 1 h, 350 °C for 2 h, and 450 °C for 5 h. To remove any excess sulfur, the sample was further heated at 200 °C for 2 h. A control sample without
Material characterization
Elemental analysis (EA) was performed using an Elementar Vario EL analyzer (Elementar, Germany). Morphological characterization was carried out using a Carl Zeiss Supra 55 field-emission scanning electron microscope (SEM) equipped with an energy-dispersive X-ray spectroscopy (EDS) detector (Carl Zeiss, Germany). X-ray diffraction (XRD) patterns were recorded on a Rigaku SmartLab diffractometer with Cu-Kα radiation (Rigaku, Japan). Raman spectra were collected using a HORIBA LabRAM HR Evolution confocal Raman microscope (HORIBA Scientific, France).
Electrochemical measurement
The working cathode slurry was formulated by uniformly blending the FeNiS2 QDs@SPAN composite with acetylene black and LA133 binder in a weight ratio of 80:10:10 in deionized water. This slurry was cast onto aluminum foil and dried under vacuum at 60 °C for 24 h. The dried electrode films were punched into discs with a diameter of 10 mm. The loading of FeNiS2 QDs@SPAN on each electrode was approximately
Theoretical calculations
Density Functional Theory (DFT) computations in this study were performed using the Vienna Ab Initio Simulation Package (VASP). Projector-augmented wave (PAW) potentials[38-40] and the Perdew-Burke-Ernzerhof (PBE) functional within the generalized gradient approximation (GGA)[41] were employed in the simulation. A 3 × 3 supercell of FeNiS2 (102) surface was built. The vacuum space along the z-direction was set to 25 Å, while the plane-wave cutoff energy was set to 550 eV. The Brillouin zone of the supercell was sampled using a 2 × 2 × 1 k-point sampling grid, and the convergence tolerances for energy and force were set to 1.0 × 10-5 eV per atom and 10-2 eV Å-1, respectively. Spin polarization was included to describe the ferromagnetic properties of FeNiS2. The binding energy (Eb) of LiPSs on FeNiS2 (102) surfaces was calculated using the following equation[13]:
where
RESULTS AND DISCUSSION
Figure 1A and B illustrates the detailed procedure for synthesizing FeNiS2 QDs@SPAN nanofibers. Initially, NiFe2O4 was synthesized using a hydrothermal method, and its aqueous dispersion exhibited bright blue luminescence under 365 nm Ultraviolet (UV) irradiation, confirming its quantum dot nature. A mixture of PAN and NiFe2O4 QDs was dispersed in DMF and stirred overnight to ensure solution homogeneity. This homogeneous solution was then loaded into a syringe for electrospinning, yielding NiFe2O4 QDs/PAN nanofiber precursors. Subsequently, these nanofibers were combined with sublimed sulfur in a high-pressure reaction vessel and subjected to heat treatment at 450 °C under an argon atmosphere. During this process, elemental sulfur (S8) decomposed into smaller sulfur chains (-Sx-, where x < 8), facilitating the dehydrogenation and cyclization of PAN. Finally, a further heating step at 200 °C in a quartz tube furnace under argon flow was employed to remove any adsorbed excess sulfur, resulting in the formation of
Figure 1. Schematic illustration for synthetic procedure of FeNiS2 QDs@SPAN. QDs: Quantum dots; SPAN: sulfurized polyacrylonitrile; PAN: polyacrylonitrile; UV: ultraviolet.
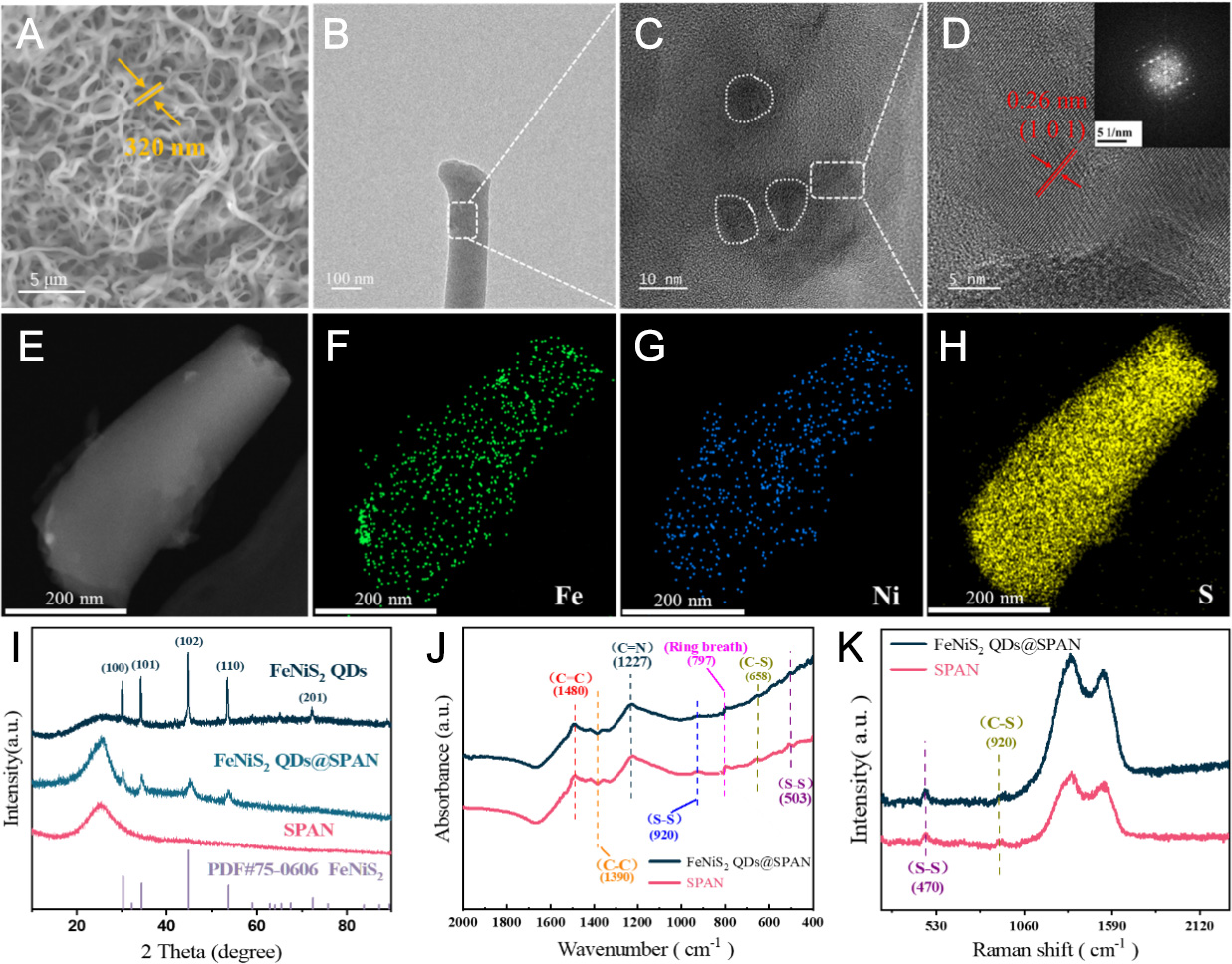
SEM images of NiFe2O4 QDs@PAN and FeNiS2 QDs@SPAN are depicted in Supplementary Figure 1 and Figure 2A, respectively. The NiFe2O4 QDs@PAN sample displays a nanofiber mesh structure, characterized by long, uniformly interconnected fibers possessing an average diameter of roughly 165 nm. For
Figure 2. Characterization of FeNiS2 QDs@SPAN and SPAN. SEM images of (A) FeNiS2 QDs@SPAN, (B and C) HR-TEM image of
The High-Resolution Transmission Electron Microscopy (HR-TEM) images reveal the FeNiS2 QDs@SPAN contains both crystalline and high-contrast disordered regions [Figure 2B and C]. A lattice spacing of
XRD measurements are performed to characterize the phase composition and crystalline properties of the resultant materials. The XRD patterns for various samples, including NiFe2O4 QDs, FeNiS2 QDs, SPAN, and FeNiS2 QDs@SPAN, are illustrated in Figure 2I and Supplementary Figure 3. The XRD spectra for
The Fourier Transform Infrared Spectroscopy (FT-IR) spectroscopic analysis of the as-prepared samples is presented in Figure 2J. Two distinct absorption peaks appearing at 503 and 920 cm-1 correspond to the stretching vibrations of disulfide (S-S) linkages, while the absorption signal at 658 cm-1 is derived from the stretching vibration of carbon-sulfur (C-S) bonds. Additionally, the characteristic peaks at 1,480 and
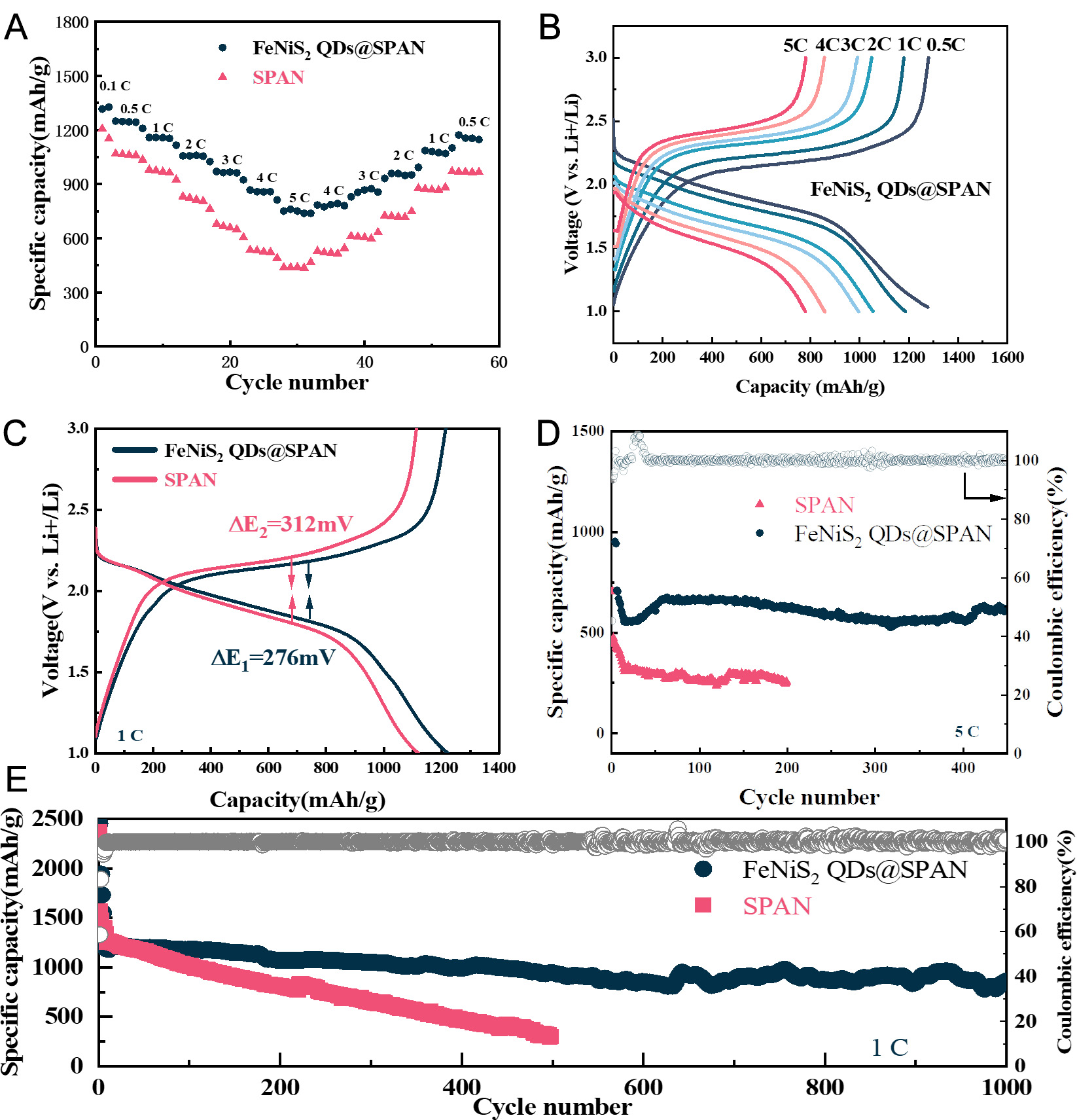
To assess the impact of FeNiS2 QDs on Li-S battery electrochemistry, corresponding cells were assembled with a metallic lithium anode and FeNiS2 QDs@SPAN/SPAN cathodes for performance investigation. Rate capability is an important parameter for practical rechargeable battery systems. As illustrated in Figure 3A, the rate performance of two cathodes was evaluated by conducting charge/discharge cycles with varying rates from 0.5 C to 5 C. Both specific capacities and current rates are normalized with respect to the sulfur mass. The FeNiS2 QDs@SPAN cathode delivers specific discharge capacities of 1,300, 1,180, 1,050, 1,005, and
Figure 3. Electrochemical properties of FeNiS2 QDs@SPAN cathode. (A) rate performance of both FeNiS2 QDs@SPAN and SPAN cathodes; (B) charge/discharge profiles of the FeNiS2 QDs@SPAN cathode across various current rates from 0.5 C to 5 C; (C) Galvanostatic charge-discharge profiles for both FeNiS2 QDs@SPAN and SPAN cathodes at 1 C; (D) Cycling stability performance of both the FeNiS2 QDs@SPAN and SPAN at 5 C; (E) Cyclic performance of the FeNiS2 QDs@SPAN and SPAN cathode at 1 C. QDs: Quantum dots; SPAN: sulfurized polyacrylonitrile.
The charge-discharge profiles of the FeNiS2 QDs@SPAN cathode at various current densities are presented in Figure 3B. Apparently, even under an ultra-high rate of 5 C, all discharge curves show a stable and flat voltage platform. In contrast, it is clear that the SPAN cathode experiences severe capacity degradation and greater polarization as the current density increases [Supplementary Figure 5]. The potential difference (discharge mid-voltage minus charge mid-voltage equals ΔE) between the discharge and charge curves represents the degree of polarization. As shown in Figure 3c, compared to SPAN (ΔE2 = 312 mV), the
To evaluate the long-term cycling performance of Li-S batteries under high current densities, both cathodes were cycled at a fixed current rate of 5 C [Figure 3D]. SPAN experiences a structural rearrangement during the first discharge process[46], therefore the SPAN and FeNiS2 QDs@SPAN electrodes were activated at 0.1 C for 2 cycles to complete the structural reorganization. After activating the FeNiS2 QDs@SPAN electrode, it exhibits a discharge specific capacity of 720 mAh g-1 when the current density is switched to 5 C (8.37 A g-1). Impressively, after 450 cycles, the FeNiS2 QDs@SPAN electrode maintains 86.6% capacity retention
The long-term cycling test results in Figure 3E reveal that the FeNiS2 QDs@SPAN cathode delivers an initial specific discharge capacity as high as 1,210 mAh g-1 under 1 C, with an ultra-low average capacity decay rate of only 0.034% across 1,000 cycles. After 1,000 cycles, the Coulombic Efficiency (CE) of the
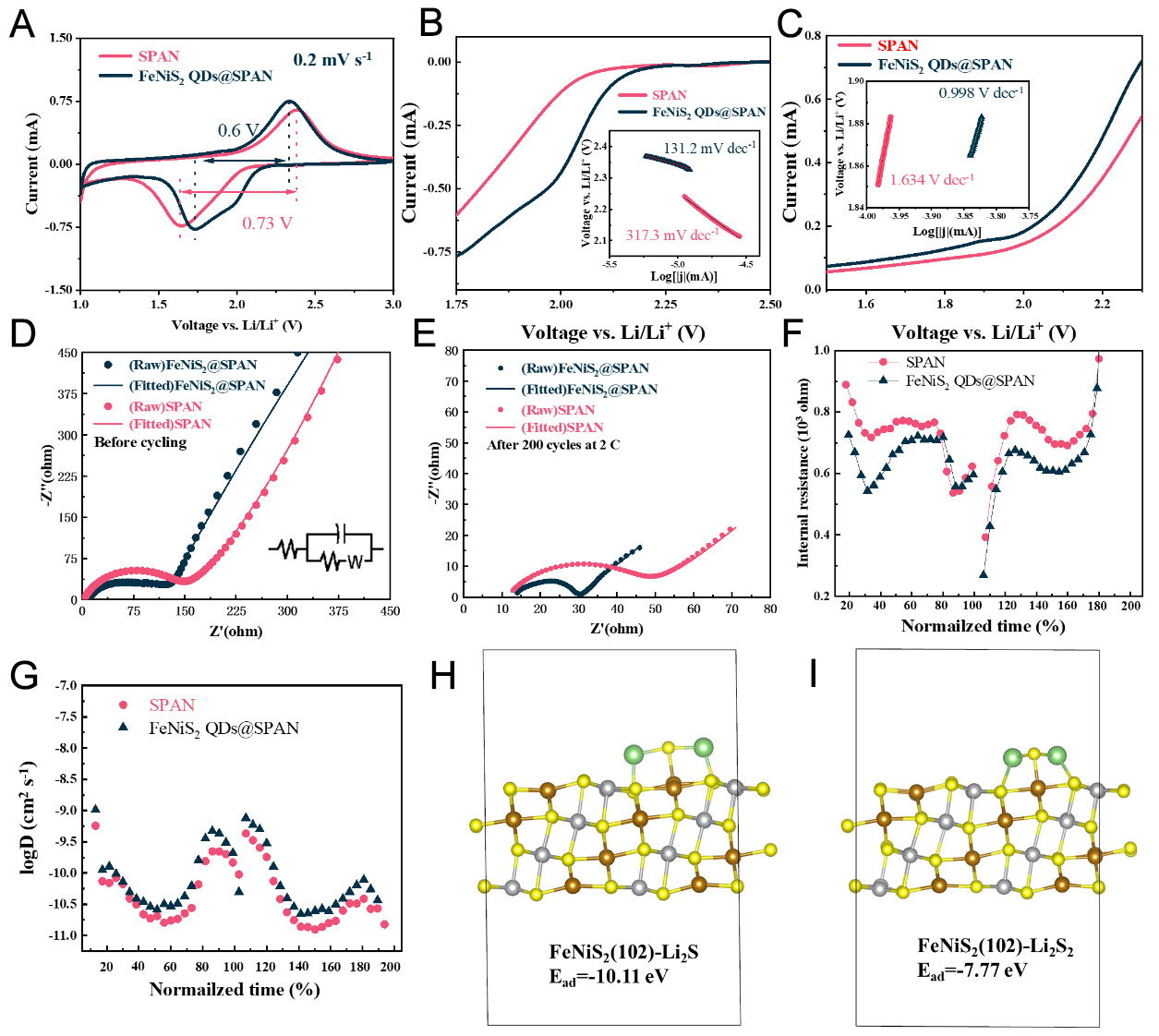
To investigate the catalytic properties of the FeNiS2 QDs@SPAN composite in lithium-sulfur reactions, the corresponding electrocatalytic performance was systematically investigated by combining CV with Tafel measurements. The typical CV curves of the two electrodes, obtained at a scan rate of 0.2 mV s-1, are illustrated in Figure 4A. Two well-resolved broad cathodic peaks at 2.08 and 1.73 V can be clearly observed for the FeNiS2 QDs@SPAN nanofiber electrode, originating from the stepwise electrochemical reduction of short-chain sulfur to Li2S2 and Li2S, in sequence[29]. For SPAN, merely a single broad cathodic peak emerges at 1.62 V, a feature indicative of pronounced polarization along with a shift toward lower potentials. These results confirm that FeNiS2 QDs can expedite the reduction reaction of SPAN and enhance the reaction kinetics during the discharging process. To better understand the catalytic activity of FeNiS2 QDs, we compared the Tafel plots for a single cathodic and anodic process, respectively, as shown in Figure 4B and C. For the reduction process, the estimated Tafel slope for the FeNiS2 QDs@SPAN cathode is 131.2 mV dec-1, which is significantly smaller than that of the SPAN electrode (317.3 mV dec-1). Similarly, for the oxidation process, the FeNiS2 QDs@SPAN cathode exhibits a lower Tafel slope of 0.998 V dec-1 in comparison with the SPAN electrode (1.634 V dec-1). This indicates that with the FeNiS2 QDs, lower polarization and enhanced redox conversion between LiPS and Li2S of the FeNiS2 QDs@SPAN electrode can be realized, in comparison to the SPAN electrode.
Figure 4. Reaction kinetics investigation of FeNiS2 QDs@SPAN and SPAN electrodes. (A) The comparison of peak voltages for
To further understand the improved kinetics of FeNiS2 QDs@SPAN, EIS measurements were conducted. The results demonstrate significantly enhanced charge transfer capability of the FeNiS2 QDs@SPAN composite before and after cycling. Before cycling, the Nyquist plots [Figure 4D] exhibited characteristic impedance spectra, which comprise a distinct high-frequency semicircle attributed to the interfacial charge transfer resistance (Rct), as well as a diagonal line at low frequency related to the Warburg impedance (Wo) of Li+ diffusion in the electrode. Analysis of the fitting data shows that FeNiS2 QDs@SPAN exhibits substantially lower Rct (124.5 Ω) than the SPAN electrode (198.7 Ω) before cycling [Supplementary Table 3]. Furthermore, Figure 4E confirms that the Rct of FeNiS2 QDs@SPAN after cycling remains markedly lower than that of SPAN, which is attributable to the composite's superior catalytic properties in promoting Li2S2/Li2S conversion kinetics [Supplementary Table 3]. Additionally, Variable-scan-rate CV (0.2-1 mV s-1) and Galvanostatic Intermittent Titration Technique (GITT) investigation further confirm the fast reaction kinetics of FeNiS2 QDs@SPAN, as manifested by enhanced Li diffusion coefficient obtained via the two methods (details see Figure 4F and G, Supplementary Figures 7 and 8).
The adsorption behavior of Li2Sn (n = 1, 2) on FeNiS2 was further investigated by DFT calculations. The (102) crystal plane is selected for the DFT calculation since the XRD pattern of the synthesized FeNiS2 shows that (102) is the strongest peak [Figure 2I]. Figure 4H and I, Supplementary Figure 9 illustrate the optimized adsorption configurations and the corresponding top views of Li2S and Li2S2 on the FeNiS2 (102) surface, respectively. The adsorption energies of Li2S2 and Li2S on the FeNiS2 (102) surface are computed to be -7.77 and -10.11 eV, respectively. These values are significantly more negative than those on pyridine nitrogen sites (in N-doped carbon)[57,58], demonstrating that the introduction of FeNiS2 provides much stronger anchoring for Li2S2 and Li2S. It might lead to the formation of Li2S2 and Li2S close to the homogeneously dispersed FeNiS2 during the reduction reaction, which further facilitates the conversion reaction between LiPS and Li2S2/Li2S.
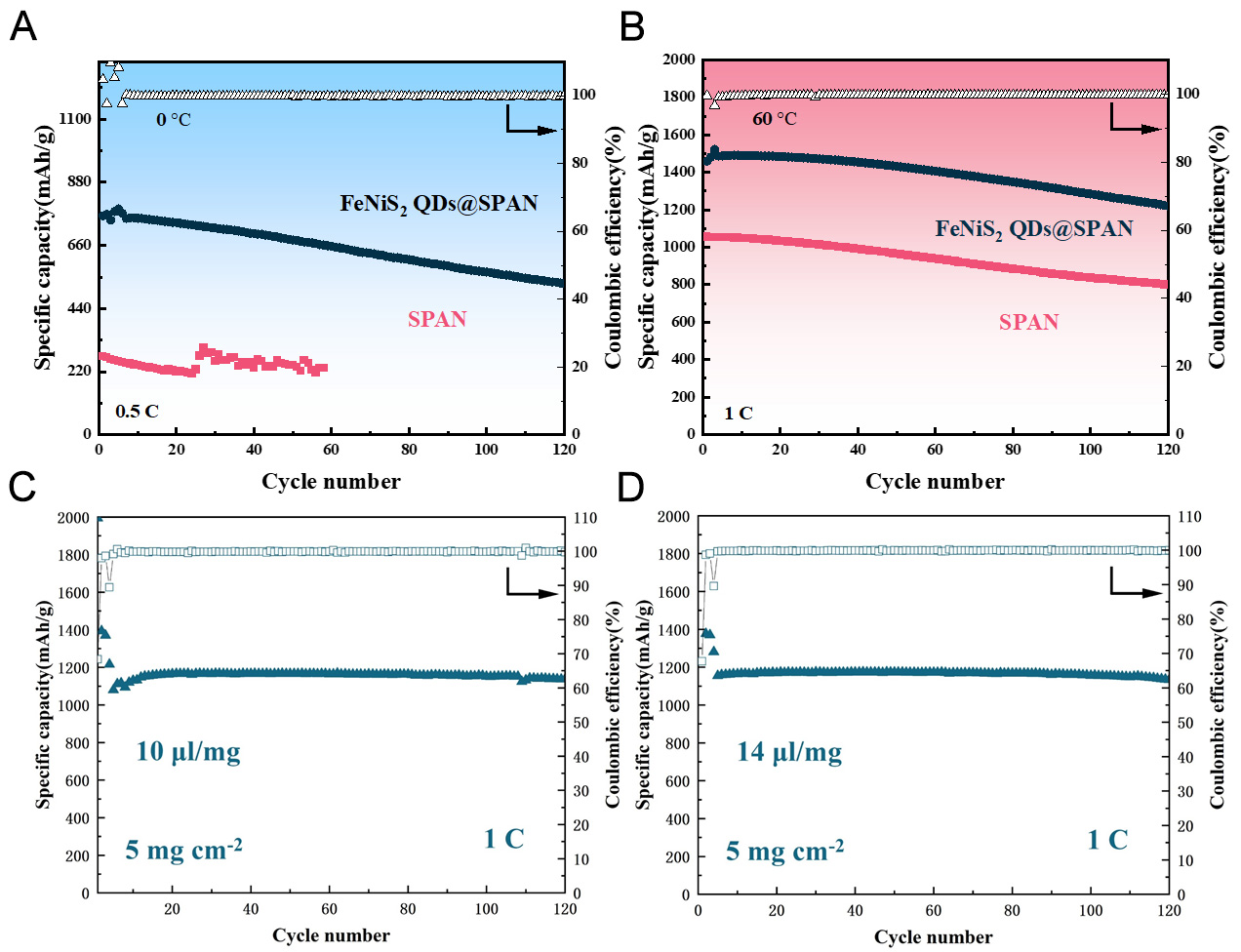
To investigate the feasibility of FeNiS2 QDs@SPAN for extreme environmental conditions, the performance of SPAN electrodes across a broad temperature range was further evaluated.As shown in Figure 5A and Supplementary Figure 10A, the FeNiS2 QDs@SPAN cathode delivered a remarkable specific discharge capacity of 751 mAh g-1 at 0.5 C under 0 °C. Despite the increased electrolyte viscosity and sluggish reaction kinetics at 0 °C, the FeNiS2 QDs@SPAN cathode still maintained 71% of its initial capacity after 120 cycles. This outstanding cycle stability of FeNiS2 QDs@SPAN cathode at 0 °C arises from the high electrocatalytic activity of FeNiS2 QDs, which effectively promotes polysulfide conversion and enhances the reaction kinetics. In stark contrast, SPAN electrode shows an initial capacity of merely 440 mAh/g at 0 °C. It suffers rapid capacity decay and short-circuit after only 60 cycles, demonstrating very poor low-temperature tolerance. The high-temperature cycling performance at 60 °C is presented in Figure 5B and Supplementary Figure 10B. After two activation cycles at 0.1 C, the FeNiS2 QDs@SPAN composite cathode achieved an impressive specific discharge capacity of 1,487 mAh g-1 at a current rate of 1 C, and retained a capacity retention of 83% after 120 cycles. By comparison, the SPAN cathode only delivered a much lower specific capacity of
Figure 5. Electrochemical cycling performance of the FeNiS2 QDs@SPAN composite under different temperatures and conditions. (A) Cycling performance of
To demonstrate the commercial viability of FeNiS2 QDs@SPAN material, its performance was tested under high sulfur loading and lean electrolyte environments. Coin cells with 5 mg cm-2 FeNiS2 QDs@SPAN electrode and different electrolyte-to-sulfur (E/S) ratios of 7, 10, 14, and 18 µL mg-1 were assembled and their performance was evaluated. All cells exhibit similar galvanostatic charge-discharge profiles, and maintain the characteristic SPAN charge-discharge curves [Supplementary Figure 11]. With a lower E/S ratio of 7 µL mg-1, the polarization is slightly increased. Cycle stability under varied E/S ratios was further investigated. As shown in Figure 5C and D, Supplementary Figure 12, the initial specific capacities of FeNiS2 QDs@SPAN in all cells are almost same, which confirms the sulfur utilization is unaffected with different E/S ratios. Specifically, the capacities of FeNiS2 QDs@SPAN in the cells with E/S ratios of 7, 10, 14, and 18 µL mg-1 are 1,173, 1,159, 1,174, and 1,169 mAh g-1, respectively. For the cells with E/S ratios of 10, 14, 18 µL mg-1, they show excellent cycle stability, and the capacity retention is 97.9%, 96.1%, and 98.7% over 120 cycles, respectively. When tested at a lean electrolyte condition with an E/S ratio as low as 7 µL mg-1, the cell exhibited a gradual capacity decay, and retained 70.6% of its initial capacity after 100 cycles. This performance degradation is mainly ascribed to the increased voltage polarization, as illustrated in Supplementary Figure 10.The results suggest that FeNiS2 QDs@SPAN electrode with 5 mg cm-2 mass loading exhibits excellent cycling performance when the E/S ratio is above 10 µL mg-1. It demonstrates promising application potential of FeNiS2 QDs@SPAN material for lithium sulfur batteries.
CONCLUSIONS
In summary, this work aims to address the intrinsically slow reaction kinetics of SPAN cathodes for Li-S batteries by fabricating FeNiS2 QDs@SPAN nanofibers via electrospinning coupled with sulfurization heat treatment. The embedded FeNiS2 QDs act as efficient catalytic sites, which reduce the cathode charge transfer resistance and accelerate the conversion of short-chain polysulfides to Li2S, thereby effectively boosting the intrinsic reaction kinetics of SPAN. This catalytic modification endows the FeNiS2 QDs@SPAN cathode with excellent rate performance, cycling stability, and reliable electrochemical performance under harsh working conditions including a wide temperature range and lean electrolyte. Further optimization of electrode structure may enhance the performance of Li//FeNiS2 QDs@SPAN battery under even leaner electrolyte conditions. This study verifies that integrating FeNiS2 QDs catalysts into SPAN nanofibers is a feasible and effective strategy for advancing SPAN-based Li-S batteries, and it provides a valuable reference for the design of high-performance catalytic electrodes for high-energy-density Li-S batteries.
DECLARATIONS
Authors’ contributions
Methodology, software, investigation, writing original draft: Li, J.
Validation, formal analysis, visualization: Niu, R.
Writing-review and editing: Qi, H.; Song, J.
Writing-reviewing, discussion: Wu, Y. (Yunling Wu); Liu, L.; Li, G.
Conceptualization, writing-review and editing, supervision, data curation, project administration: Fu, L.
Writing-reviewing, discussion, resources: Wu, Y. (Yuping Wu)
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (52122209, 52373289, 52403001).
Conflicts of interest
Yuping Wu serves as the Editor-in-Chief of the journal Energy Materials; however, he was not involved in any stage of the editorial process for this manuscript, including reviewer selection, manuscript handling, or decision-making. The other authors declare that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Fang, R.; Zhao, S.; Sun, Z.; Wang, D. W.; Cheng, H. M.; Li, F. More reliable lithium-sulfur batteries: status, solutions and prospects. Adv. Mater. 2017, 29, 1606823.
2. Deng, R.; Wang, M.; Yu, H.; et al. Recent advances and applications toward emerging lithium-sulfur batteries: working principles and opportunities. Energy. Environ. Materials. 2022, 5, 777-99.
3. Wang, J.; He, Y. S.; Yang, J. Sulfur-based composite cathode materials for high-energy rechargeable lithium batteries. Adv. Mater. 2015, 27, 569-75.
4. Sun, C.; Sheng, J.; Zhang, Q.; et al. Self-extinguishing Janus separator with high safety for flexible lithium-sulfur batteries. Sci. China. Mater. 2022, 65, 2169-78.
5. Liu, B.; Fang, R.; Xie, D.; et al. Revisiting scientific issues for industrial applications of lithium-sulfur batteries. Energy. Environ. Mater. 2018, 1, 196-208.
6. Xue, W.; Shi, Z.; Suo, L.; et al. Intercalation-conversion hybrid cathodes enabling Li-S full-cell architectures with jointly superior gravimetric and volumetric energy densities. Nat. Energy. 2019, 4, 374-82.
7. Li, H.; Li, Y.; Zhang, L. Designing principles of advanced sulfur cathodes toward practical lithium‐sulfur batteries. SusMat 2022, 2, 34-64.
8. Li, J.; Li, X.; Fan, X.; et al. Holey graphene anchoring of the monodispersed nano-sulfur with covalently-grafted polyaniline for lithium sulfur batteries. Carbon 2022, 188, 155-65.
9. Li, S.; Fan, Z. Encapsulation methods of sulfur particles for lithium-sulfur batteries: a review. Energy. Storage. Mater. 2021, 34, 107-27.
10. Feng, L.; Yu, P.; Fu, X.; et al. Regulating polysulfide diffusion and deposition via rational design of core-shell active materials in Li-S batteries. ACS. Nano. 2022, 16, 7982-92.
11. Zhou, W.; Chen, M.; Zhao, D.; et al. Confined Co9S8 nanocrystals into N/S-Co-doped carbon nanofibers as a chainmail-like electrocatalyst for high-performance lithium-sulfur batteries with high sulfur loading. J. Colloid. Interface. Sci. 2022, 625, 187-96.
12. Xiao, Z.; Li, Z.; Li, P.; Meng, X.; Wang, R. Ultrafine Ti3C2 MXene nanodots-interspersed nanosheet for high-energy-density lithium-sulfur batteries. ACS. Nano. 2019, 13, 3608-17.
13. Chu, R.; Nguyen, T. T.; Bai, Y.; Kim, N. H.; Lee, J. H. Uniformly controlled treble boundary using enriched adsorption sites and accelerated catalyst cathode for robust lithium-sulfur batteries. Adv. Energy. Mater. 2022, 12, 2102805.
14. Yuan, S.; Xia, M.; Liu, Z.; et al. Dual synergistic effects between Co and Mo2C in Co/Mo2C heterostructure for electrocatalytic overall water splitting. Chem. Eng. J. 2022, 430, 132697.
15. Wang, J.; Yang, J.; Xie, J.; Xu, N. A Novel conductive polymer-sulfur composite cathode material for rechargeable lithium batteries. Adv. Mater. 2002, 14, 963-5.
16. Wei, S.; Ma, L.; Hendrickson, K. E.; Tu, Z.; Archer, L. A. Metal-sulfur battery cathodes based on PAN-sulfur composites. J. Am. Chem. Soc. 2015, 137, 12143-52.
17. Zhao, X.; Wang, C.; Li, Z.; Hu, X.; Abdul Razzaq, A.; Deng, Z. Sulfurized polyacrylonitrile for high-performance lithium sulfur batteries: advances and prospects. J. Mater. Chem. A. 2021, 9, 19282-97.
18. Xie, J.; Chen, J.; Guo, L.; et al. Deciphering the sulfur-involved bonding interactions in sulfurized polyacrylonitrile: the formation thermodynamics and the roles in electrochemical characteristics. ACS. Nano. 2025, 19, 3931-43.
19. Liu, J.; Wang, M.; Xu, N.; Qian, T.; Yan, C. Progress and perspective of organosulfur polymers as cathode materials for advanced lithium-sulfur batteries. Energy. Storage. Mater. 2018, 15, 53-64.
20. Chen, W. J.; Li, B. Q.; Zhao, C. X.; et al. Electrolyte regulation towards stable lithium-metal anodes in lithium-sulfur batteries with sulfurized polyacrylonitrile cathodes. Angew. Chem. Int. Ed. 2020, 59, 10732-45.
21. Doan, T. N. L.; Ghaznavi, M.; Zhao, Y.; et al. Binding mechanism of sulfur and dehydrogenated polyacrylonitrile in sulfur/polymer composite cathode. J. Power. Sources. 2013, 241, 61-9.
22. Zhang, Y.; Zhao, Y.; Yermukhambetova, A.; Bakenov, Z.; Chen, P. Ternary sulfur/polyacrylonitrile/Mg0.6Ni0.4O composite cathodes for high performance lithium/sulfur batteries. J. Mater. Chem. A. 2013, 1, 295-301.
23. Fang, L.; Xu, W.; Lyu, X.; et al. Suppressing the shuttle effects with FeCo/SPAN cathodes and high-concentration electrolytes for high-performance lithium-sulfur Batteries. ACS. Appl. Energy. Mater. 2023, 6, 795-801.
24. He, R.; Li, Y.; Wei, S.; et al. Construction of high-performance sulfurized poly(acrylonitrile) cathodes for lithium-sulfur batteries via catalytic and conductive regulation. J. Alloys. Compd. 2022, 919, 165838.
25. Wu, S.; Wang, W.; Shan, J.; et al. Conductive 1T-VS2-MXene heterostructured bidirectional electrocatalyst enabling compact Li-S batteries with high volumetric and areal capacity. Energy. Storage. Mater. 2022, 49, 153-63.
26. Wang, N.; Chen, B.; Qin, K.; et al. Octopus-inspired design of apical NiS2 nanoparticles supported on hierarchical carbon composites as an efficient host for lithium sulfur batteries with high sulfur loading. ACS. Appl. Mater. Interfaces. 2020, 12, 17528-37.
27. Li, Z.; Sami, I.; Yang, J.; Li, J.; Kumar, R. V.; Chhowalla, M. Lithiated metallic molybdenum disulfide nanosheets for high-performance lithium-sulfur batteries. Nat. Energy. 2023, 8, 84-93.
28. Li, D.; Li, Y.; Xu, C.; Zhao, S.; Zhang, Y.; Huo, P. Enhanced polysulfide adsorption and conversion on FeS2@CNF-modified separator for Li-S batteries. New. J. Chem. 2026, 50, 2891-5.
29. Li, Y.; He, R.; Liu, H.; et al. Construction of CoS2 reduction accelerator-modified sulfurized polyacrylonitrile nanofibers as high-performance cathode materials for practical lithium-sulfur batteries. ACS. Appl. Energy. Mater. 2023, 6, 8466-78.
30. Liu, Y.; Wang, W.; Wang, A.; Jin, Z.; Zhao, H.; Yang, Y. A polysulfide reduction accelerator - NiS2-modified sulfurized polyacrylonitrile as a high performance cathode material for lithium-sulfur batteries. J. Mater. Chem. A. 2017, 5, 22120-4.
31. Yang, W.; Zhao, H.; Chen, L.; et al. Ferrous sulfide-assisted hollow carbon spheres as sulfur host for advanced lithium-sulfur batteries. Chem. Eng. J. 2017, 326, 1040-7.
32. Lu, X.; Zhang, Q.; Wang, J.; et al. High performance bimetal sulfides for lithium-sulfur batteries. Chem. Eng. J. 2019, 358, 955-61.
33. Ren, X.; Wang, Q.; Pu, Y.; Sun, Q.; Sun, W.; Lu, L. Synergizing spatial confinement and dual-metal catalysis to boost sulfur kinetics in lithium-sulfur batteries. Adv. Mater. 2023, 35, e2304120.
34. Dong, H.; Ji, Y.; Wang, L.; et al. Bimetallic coupling strategy modulating electronic construction to accelerate sulfur redox reaction kinetics for high-energy flexible Li-S batteries. Small 2024, 20, e2406565.
35. Li, N.; Meng, T.; Ma, L.; et al. Curtailing carbon usage with addition of functionalized NiFe2O4 quantum dots: toward more practical S cathodes for Li-S cells. NanoMicro. Lett. 2020, 12, 145.
36. Wang, Z.; Song, C.; Shen, H.; Ma, S.; Li, G.; Li, Y. RuOx quantum dots loaded on graphdiyne for high-performance lithium-sulfur batteries. Adv. Mater. 2024, 36, e2307786.
37. Xu, Z. L.; Lin, S.; Onofrio, N.; et al. Exceptional catalytic effects of black phosphorus quantum dots in shuttling-free lithium sulfur batteries. Nat. Commun. 2018, 9, 4164.
38. Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15-50.
39. Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B. 1996, 54, 11169.
40. Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 1999, 59, 1758.
41. Perdew, J. P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple [Phys. Rev. Lett. 77, 3865 (1996)]. Phys. Rev. Lett. 1997, 78, 1396.
42. Babu, B.; Koutavarapu, R.; Shim, J.; Kim, J.; Yoo, K. Improved sunlight-driven photocatalytic abatement of tetracycline and photoelectrocatalytic water oxidation by tin oxide quantum dots anchored on nickel ferrite nanoplates. J. Electroanal. Chem. 2021, 900, 115699.
43. Miah, M.; Hota, P.; Mondal, T. K.; Chen, R.; Saha, S. K. Mixed metal sulfides (FeNiS2) nanosheets decorated reduced graphene oxide for efficient electrode materials for supercapacitors. J. Alloys. Compd. 2023, 933, 167648.
44. Liu, H.; Zhang, Y.; Li, Y.; Han, N.; Liu, H.; Zhang, X. Solid-state transformations of active materials in the pores of sulfurized-polyacrylonitrile fiber membranes via nucleophilic reactions for high-loading and free-standing lithium-sulfur battery cathodes. Adv. Fiber. Mater. 2024, 6, 772-85.
45. Hwang, T. H.; Jung, D. S.; Kim, J. S.; Kim, B. G.; Choi, J. W. One-dimensional carbon-sulfur composite fibers for Na-S rechargeable batteries operating at room temperature. Nano. Lett. 2013, 13, 4532-8.
46. Wang, X.; Qian, Y.; Wang, L.; et al. Sulfurized polyacrylonitrile cathodes with high compatibility in both ether and carbonate electrolytes for ultrastable lithium-sulfur batteries. Adv. Funct. Mater. 2019, 29, 1902929.
47. Chen, X.; Peng, L.; Wang, L.; et al. Ether-compatible sulfurized polyacrylonitrile cathode with excellent performance enabled by fast kinetics via selenium doping. Nat. Commun. 2019, 10, 1021.
48. Lu, J.; Zhang, Y.; Huang, J.; et al. Melamine foam-derived N-doped carbon framework and graphene-supported sulfurized polyacrylonitrile for high performance lithiumsulfur battery cathode. J. Energy. Storage. 2025, 118, 116330.
49. Abdul Razzaq, A.; Chen, G.; Zhao, X.; et al. Cobalt coordination with pyridines in sulfurized polyacrylonitrile cathodes to form conductive pathways and catalytic M-N4S sites for accelerated Li-S kinetics. J. Energy. Chem. 2021, 61, 170-8.
50. Lu, J.; Zhang, Y.; Huang, J.; et al. A free-standing sulfide polyacrylonitrile/reduced graphene oxide film cathode with nacre-like architecture for high-performance lithium-sulfur batteries. J. Power. Sources. 2025, 629, 235916.
51. Shao, J.; Huang, C.; Zhu, Q.; et al. Flexible CNT-interpenetrating hierarchically porous sulfurized polyacrylonitrile (CIHP-SPAN) electrodes for high-rate lithium-sulfur (Li-S) batteries. Nanomaterials. 2024, 14, 1155.
52. Hao, C.; Liu, J.; Wang, Q.; et al. High volumetric capacity FeS2/SPAN composite with promoted kinetics for Li-S battery. ACS. Nano. 2025, 19, 25385-94.
53. Wang, K.; Guan, Y.; Jin, Z.; Wang, W.; Wang, A. Te0.045S0.955PAN composite with high average discharge voltage for Li-S battery. J. Energy. Chem. 2019, 39, 249-55.
54. Haridas, A. K.; Heo, J.; Li, X.; et al. A flexible and free-standing FeS/sulfurized polyacrylonitrile hybrid anode material for high-rate sodium-ion storage. Chem. Eng. J. 2020, 385, 123453.
55. Liu, H.; Xu, Q.; Zhang, Y.; Han, N.; Liu, H.; Zhang, X. Multifunctional heterostructure CoS2/FeS2 catalysts for enhancing high-performance lithium-sulfurized polyacrylonitrile batteries through intrinsic electric fields. Chin. Chem. Lett. 2025, 111084.
56. Wang, J.; Du, Z.; Lv, G.; et al. Enhancing the backbone regularity of sulfurized polyacrylonitrile for long-life Li-SPAN batteries. J. Mater. Chem. A. 2025, 13, 21545-54.
57. Gao, G.; Zheng, F.; Pan, F.; Wang, L. W. Theoretical investigation of 2D conductive microporous coordination polymers as Li-S battery cathode with ultrahigh energy density. Adv. Energy. Mater. 2018, 8, 1801823.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].