Synergistic optimization of NCM9055/AC composites enabling superior performance in hybrid battery-supercapacitors
Abstract
Owing to sluggish ion migration, structural instability, and elevated interfacial impedance, ternary lithium nickel cobalt manganese oxide (NCM) systems often fail to meet the requirements of rapid charge-discharge, resulting in low power densities in lithium-ion batteries (LIBs). This study investigates the electrochemical performance of cathodes fabricated via the integration of three types of ternary NCM material (namely NCM523, NCM811, and NCM9055) composited with activated carbon (AC) for hybrid battery supercapacitors (HBS). Attributed to the highly uniform morphology, enhanced structural stability, and high Ni percentage, NCM9055/AC composite cathodes not only a superior specific capacity (231 mAh/g at 0.1 C) but also exceptional rate charge and discharge performance and long cycling stability. At a current density of 0.5 C, the NCM9055/AC composite cathode maintained a high capacity of 222 mAh/g. Even at a high current density of 5 C, NCM9055/AC composite cathodes delivered a reversible capacity of
Keywords
INTRODUCTION
With the accelerating electrification of transportation and the proliferation of portable electronics, there is an escalating demand for energy storage systems that combine high energy density with rapid response capabilities[1]. Among the numerous energy storage technologies, lithium-ion batteries (LIBs) and supercapacitors represent two typical technical routes characterized by high energy and power densities, respectively. However, the former is limited by relatively low power output and cycle life, while the latter fails to satisfy long duration energy storage needs due to low energy density[2,3]. To address these performance limitations, hybrid battery supercapacitors (HBS) have been developed, integrating the benefits of both technologies. By incorporating both battery-type and capacitor-type electrodes within a single device, HBS effectively achieve a synergistic improvement in both energy density and power density[4-7]. In recent years, HBS have demonstrated significant potential for application in various domains, including smart grid frequency regulation, regenerative energy recovery, and high-power consumer electronics. This potential is attributed to their capability to perform rapid charge-discharge cycles within minutes and sustain cycle lives extending to tens of thousands of cycles[8-10].
By leveraging unique storage mechanisms and structural architectures, HBS combines the characteristics of secondary batteries and supercapacitors, achieving high energy density of secondary batteries and high power density, rapid charge-discharge capabilities, and longer cycle lives of supercapacitors[11-14]. High-performance HBSs offer balanced high power output and energy storage performance, showing great potential for widespread application in consumer electronics, unmanned mining vehicles, industrial equipment, rail transit, and power systems[3,15-19]. The cathode active material serves as the foundational and a critical component of high-performance HBS. Its performance directly influences the overall energy storage mechanism and electrochemical performance, setting the lower limits for energy density and manufacturing costs. Enhancing high-performance cathode materials and refining production processes can substantially mitigate the prevailing high-cost challenges while simultaneously augmenting the overall performance of HBS[20,21]. Ternary materials such as nickel cobalt manganese oxide (NCM) and lithium nickel cobalt aluminum oxide (NCA), and lithium iron phosphate (LiFePO4) have become mainstream cathode materials in advanced energy storage systems due to their high specific capacity and excellent cycling stability[16,22,23]. Sun et al. systematically studied NCM523 and activated carbon (AC) composite cathodes[24]. To ascertain the regulatory mechanisms governing the capacitance of LIBs within the composite cathode active material, the research team adjusted the composite cathodes using seven distinct mass ratios of NCM523. For the hybrid battery, they achieved a specific energy of 36.2 Wh/kg at an average specific power of 39.1 W/kg. Han et al. compared NCM622 with the addition of 0 wt% and 5 wt% AC and found that adding 5 wt% AC[25] alleviated concentration polarization, obtained a uniform reaction distribution with faster reaction kinetics, and demonstrated excellent electrochemical performance in full cells. Chen et al. conducted a similar controlled variable comparison, using carbon aerogel (CA) instead of AC with NCM622 as a hybrid cathode in Li+ hybrid capacitors[26]. By controlling the CA ratio (3%, 6%, and 9%), they explored the synergistic effects of the two electrode materials and the impact of different material ratios on electrochemical performance. CA possesses a high specific surface area, high porosity (80%-98%), and controllable pore size distribution
Nonetheless, the advancement of HBSs is still impeded by three critical cathode-related bottlenecks: the limited specific capacity of traditional capacitive materials restricts overall energy density; battery-type cathode materials are susceptible to structural instability during cycling, thereby restricting the cycle life of devices; and the sluggish electrode reaction kinetics results in suboptimal rate capability, hindering the achievement of rapid charge-discharge processes[23,28-32]. To address these impediments, this study proposes a structural synergistic optimization strategy for constructing high-performance composite cathodes. High-capacity layered NCM is employed as a battery type active component to serve as the high energy reservoir, while high surface area AC is integrated to induce rapid surface capacitive behavior and establish a conductive three-dimensional (3D) networks. Three types of ternary NCM material, including NCM523, NCM811, and NCM9055, composited with AC were investigated as HBS cathodes.
EXPERIMENTAL
Preparation of NCM/AC composite cathode
LiNi0.5-0.9Co0.05-0.2Mn0.05-0.3O2 powder and AC powder were used as the cathode active materials. During electrode preparation, the active material, conductive agent, and polyvinylidene Fluoride (PVDF) were mixed in a mass ratio of 8:1:1 and dispersed in NMP.
To address the potential agglomeration arising from the distinct architectures of conductive carbon black (SP) and graphene (GN), an optimized dispersion protocol was employed. Specifically, a vacuum planetary mixer was utilized to generate high-shear forces, ensuring the mixture was stirred until uniform. In this system, the SP particles act as spacers between the GN sheets, effectively preventing the restacking of GN layers and facilitating the construction of an integrated point-plane conductive network[33]. Subsequently, the homogeneous slurry was coated onto a carbon-coated aluminum foil. Crucially, to prevent component segregation during the solvent removal phase, a two-step drying process was applied. The coated electrodes were immediately subjected to pre-drying at 110 °C. This rapid solvent evaporation strategy immobilizes the solid particles, thereby suppressing binder migration and density-driven phase separation[34,35]. Following this pre-drying step, the electrodes were thoroughly vacuum-dried at 120 °C for 12 h and then cut into circular discs with a diameter of 12 mm to serve as electrode sheets.
The electrode slurry was prepared using a vacuum mixer (MSK-SFM-16), and the coating process was performed on an automatic coating machine (MSK-AFA-EI200). Afterward, the electrodes were calendered using a rolling machine (MSK-2300A) and cut using an automatic punching machine (MSK-180-AM). The cell assembly was completed using a vacuum sealing machine (MSK-115A-MS). All the aforementioned instruments were manufactured by Shenzhen Kejing Star Technology Co., Ltd. (China).
Material characterization
The microstructure and morphology of the samples were tested using scanning electron microscopy (SEM) on a Thermo Scientific Apreo S HiVac. The particle size values were obtained by measuring the diameters of randomly selected particles in the SEM images using ImageJ Software, followed by statistical analysis to determine the particle dimensions.The crystal and phase structures of all samples were confirmed using X-ray Diffraction (XRD) on a Bruker D8 Advance. Raman analysis was performed using a Thermo Scientific DXR2. The absorption spectra were recorded using a spectrophotometer.
Assembly of half-cells/pouch cells
The half-cells were assembled in a glove box. The NCM/AC composite material was used as the cathode, LiNi0.5-0.9Co0.05-0.2Mn0.05-0.3O2 were obtained from Ningbo Ronbay New Energy Technology Co., Ltd. (China). The employed AC is a commercial product (model: YP-50F) purchased from Kuraray Co., Ltd. (Japan). Regarding the conductive additives, SP was supplied by TIMCAL (Switzerland), and the GN slurry (model: XF181-Graphene electric oily slurry) was sourced from Jiangsu XFNANO Materials Tech Co., Ltd. (China). The PVDF binder was purchased from Arkema (France). All materials were used as received without further purification. Li metal as the anode, and the electrolyte was 1.0 M LiPF6 in Ethylene Carbonate:Dimethyl Carbonate (EC:DMC) = 1:1 vol%. A polypropylene (PP) separator was used, and the cells were assembled in an LIR2032 coin-cell configuration. Pouch HBS were assembled via manual stacking in a glove box using a 1+2 stacking method. The cathode, anode, and separator were alternately stacked in the following order: anode → separator → cathode → separator → anode. Subsequently, the stack was placed in an aluminum-plastic film, injected with electrolyte, and vacuum-sealed to produce pouch cells.
To evaluate the practical application potential, the energy density (Ed)[36] and maximum power density (Pmax)[37] of the pouch cell were calculated based on the total device mass (mtotal = 26 g), which includes the cathode, anode, separator, electrolyte, current collectors, tabs, and packaging materials.
The gravimetric energy densities (Ed) was calculated using the discharge capacity (Q, 392 mAh) and nominal voltage (U, 3.7 V) under standard testing conditions:
Theoretical maximum specific power density (Pmax) was calculated based on the maximum power transfer theorem, using the measured internal resistance (R = 72 mΩ) of the pouch cell:
Electrochemical testing
Cyclic voltammetry (CV) measurements were conducted on a multichannel electrochemical workstation (VMP3, Bruker, Germany) with a potential window of 2.5-4.2 V and scan rates varying from 0.1 to 2 mV s-1. Electrochemical impedance spectroscopy (EIS) was performed using the same instrument. Galvanostatic intermittent titration technique (GITT) tests were conducted using a battery test system (CT-4008-5V1A-S1, Neware, Shenzhen, China). Galvanostatic charge discharge tests were performed between 2.5 and 4.2 V at various current densities at 25 °C using a Neware battery test system (Neware Technology Ltd., Shenzhen). The reported specific capacities were normalized only to the active mass of NCM. The masses of the conductive agents and inactive components were excluded from the calculations.
RESULTS AND DISCUSSION
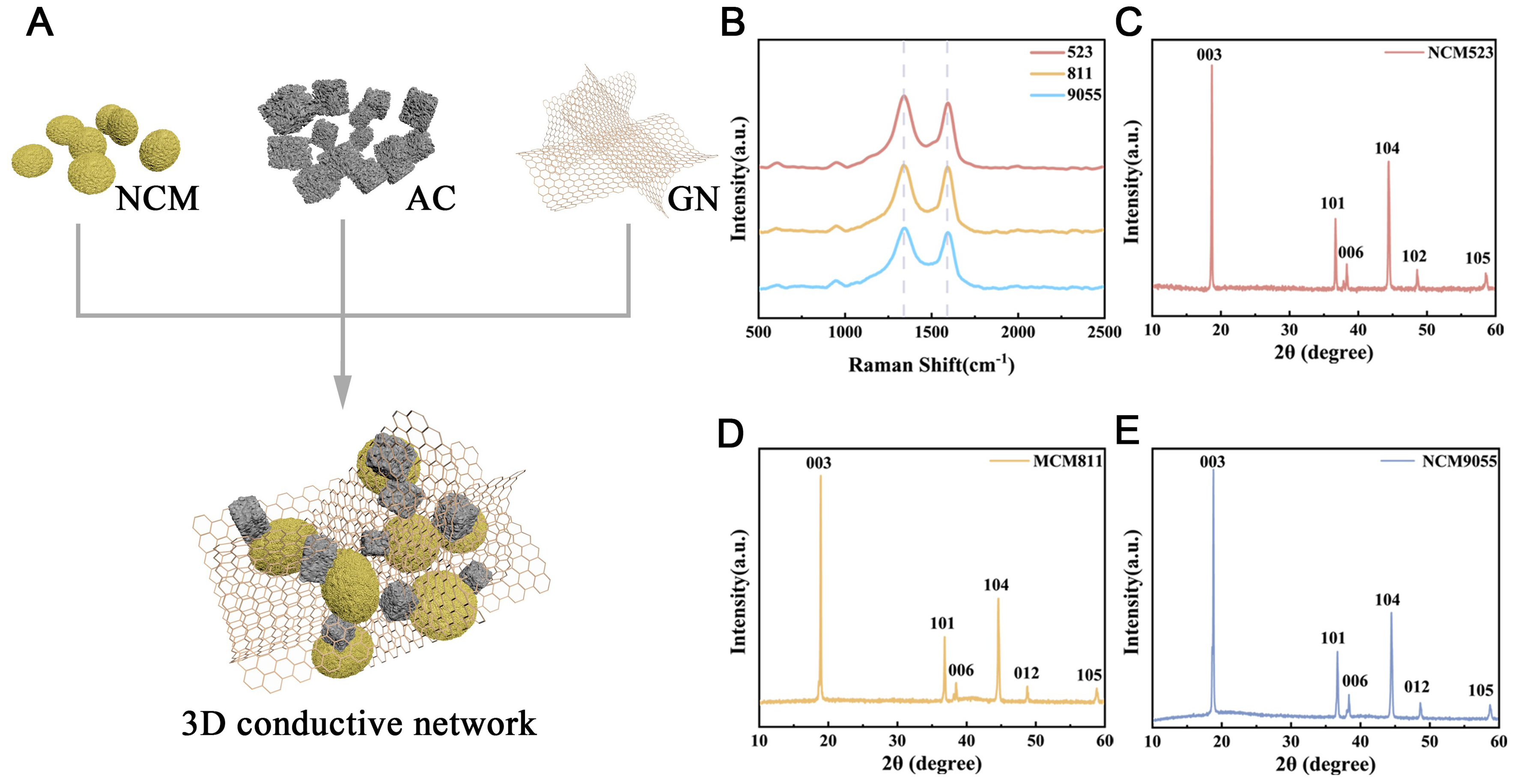
As shown in Figure 1A, by combining NCM with AC and incorporating conductive additives, such as SP and GN, a robust 3D conductive network was constructed to fabricate high performance HBC electrodes. This process employs a strategy of prioritizing the construction of a conductive network before introducing active components, enabling GN/SP to form efficient electron transport pathways between NCM and AC particles[38]. This structure significantly improves the electron transport efficiency and effectively inhibits the agglomeration of active materials, guaranteeing structural uniformity and interface stability under high mass loading, thereby laying a solid structural foundation for achieving high capacity, excellent rate performance, and long cycle life[39,40].
Figure 1. Schematic diagram for preparation of NCM/AC composite cathode and structural characterization of three NCM powders. (A) Schematic diagram for preparation of HBS cathode; (B) Raman spectra of three NCM samples; (C) XRD pattern of NCM523 powder; (D) XRD pattern of NCM811 powder; (E) XRD pattern of NCM9055 powder. NCM: Lithium nickel cobalt manganese oxide; AC: activated carbon; GN: graphene; HBS: hierarchical binder system; XRD: X-ray diffraction; a.u.: arbitrary units.
XRD and Raman data in Figure 1 systematically reveal the crystal structures and ordering of the three NCM materials. Complementing the XRD results, Raman spectroscopy [Figure 1B] was employed to probe the short range structural order and local cationic arrangement. The A1g peak of NCM 9055 was sharper than that of NCM 811, and the shoulder peak characterizing cation mixing was significantly reduced, confirming that its local chemical environment was optimized[41]. The mutual corroboration of XRD and Raman results strongly indicates that NCM 9055 successfully achieves a unity of high Ni content and low cation mixing[42]. XRD patterns in Figures 1C-E show that all the materials possessed a standard layered structure with good phase purity. Crucially, the intensity ratio of the (003) to (104) diffraction peaks indicated that the ordering of the layered structure followed the trend NCM 9055 ≈ NCM 523 > NCM 811. This proves that the prepared NCM 9055 maintains an extremely low degree of Li/Ni cation mixing despite its high Ni content; its long range structural ordering is comparable to that of stable NCM 523 and significantly superior to that of NCM 811[25].
To probe the local structural environment, Raman spectra were collected. To facilitate a detailed comparison of the local lattice fidelity among the three materials, the characteristic region (500-850 cm-1) is presented in a magnified view in Supplementary Figure 1. The vibrational mode at ~600 cm-1, assigned to the A1g vibration, corresponds to the symmetric stretching of M–O bonds within the MO6 octahedra. It is well established that in high-Ni cathodes, aggravated Li/Ni cation mixing and local lattice distortions typically break the Raman selection rules, resulting in significant A1g peak broadening or splitting[43]. However, in this enlarged spectrum, the NCM9055 sample, despite its high Ni content, retains a highly symmetrical and well-defined A1g band shape, comparable to that of the structurally stable NCM523[44]. The absence of conspicuous shoulder peaks or severe broadening confirms the preservation of local cationic ordering and the structural integrity of the layered lattice[45].
SEM images in Figure 2 systematically reveal the microstructural evolution from the pristine NCM materials to the NCM/AC composite electrodes. As shown in Figure 2A and B, E and F, I and J, the three NCM materials exhibited a regular spherical secondary particle morphology, indicating their excellent fundamental quality as active electrode materials. Notably, the NCM9055 particles were particularly intact and uniform, laying a physical foundation for their superior performance. As illustrated in Supplementary Figure 2, NCM9055 exhibits notable advantages in particle size control. Although its average particle size (D = 9.43 ± 1.41 µm) is marginally smaller than that of NCM523 (10.17 ± 3.14 µm) and NCM811 (9.86 ± 3.19 µm), its particle size distribution is highly concentrated, demonstrating excellent uniformity. The standard deviation is merely 1.41, which is significantly lower than those of the other two materials[46]. This physical feature makes the electrode easy to process and keeps the density even, which helps increase the battery’s energy storage. Besides, it improves the paths for ions and electrons and strengthens the structure, effectively suppressing particle pulverization and crack propagation during cycling[46].
Figure 2. SEM images of the pristine NCM particles and NCM/AC/GN composites. (A and B) pristine NCM523 particles; (C and D) NCM523/AC/GN composites; (E and F) pristine NCM811 particles; (G and H): NCM811/AC/GN composites; (I and J) pristine NCM9055 particles; (K and L) NCM9055/AC/GN composites. SEM: Scanning electron microscopy; NCM: lithium nickel cobalt manganese oxide; AC: activated carbon; GN: graphene.
Figures 2C and D, G and H, K and L deliver the architecture of the composite electrode, wherein the pristine NCM spherical particles are encapsulated, segmented, and integrated within the AC/conductive network. At low magnification, the NCM spheres were wrapped in the AC network. At high magnification, this encapsulation relationship is more evident, with the surface of the NCM particles covered by finer AC and conductive agent particles. The porous AC structure can provide abundant passageways for electrolyte infiltration and ion transport, and the NCM particles can be tightly wrapped by the conductive carbon network, ensuring good contact between the active particle and the conductive network, thereby greatly reducing the interfacial resistance. This structure intuitively explains how the NCM/AC composite electrode achieves both high rate performance and long cycle life, providing direct microstructural evidence for the superior electrochemical performance[47]. More detailed SEM images can be found in Supplementary Figures 3-5 in the Supplementary Materials.
Beyond the external conductive network, the intrinsic structural characteristics of the active material, particularly the secondary particle size distribution, profoundly govern electrochemical reaction kinetics. Statistical analysis of the SEM images [Supplementary Table 1 and Supplementary Figure 2] reveals distinct particle size distribution differences among the three NCM cathodes. NCM523 and NCM811 exhibit broad distributions with a notable fraction of oversized agglomerates (> 13 μm), while NCM9055 displays a highly uniform morphological feature, with the majority of secondary particles concentrated in the 8-10 μm range. This uniform particle size offers a fundamental kinetic advantage for Li+ transport. Governed by the diffusion relationship τ = L2/DLi, the reduced and uniform secondary particle size of NCM9055 effectively shortens the solid-state Li+ transport path[48,49]. Consequently, this intrinsic morphological optimization works synergistically with the extrinsic 3D AC conductive network to minimize concentration polarization and enhance high-rate capability.
NCM523, as shown in Supplementary Figures 6 and 7 and Supplementary Table 2, represents a traditional high stability material system. The secondary spherical morphology was intact, and the distribution of the transition metal elements was uniform, conforming to the standard stoichiometric ratio. NCM811, shown in Supplementary Figures 8 and 9 and Supplementary Table 3, serves as a key step towards achieving high energy density by significantly increasing the Ni content, resulting in challenges in terms of particle uniformity and increased residual Li compounds on the surface. NCM9055, shown in Supplementary Figures 10 and 11 and Supplementary Table 4, achieved comprehensive optimization and surpassed the previous two generations of materials. While retaining the high Ni content of NCM811 to achieve high energy, it successfully overcame the accompanying uniformity and interface issues. The secondary sphere regularity and particle size uniformity were significantly better than those of NCM811 and even surpassed those of NCM523, guaranteeing electrode processing consistency and mechanical stability. Notably, the uniform distribution of transition metals, particularly Ni, attests to the precise synthesis. This structural integrity minimizes component segregation and surface residues, thereby effectively suppressing side reactions.
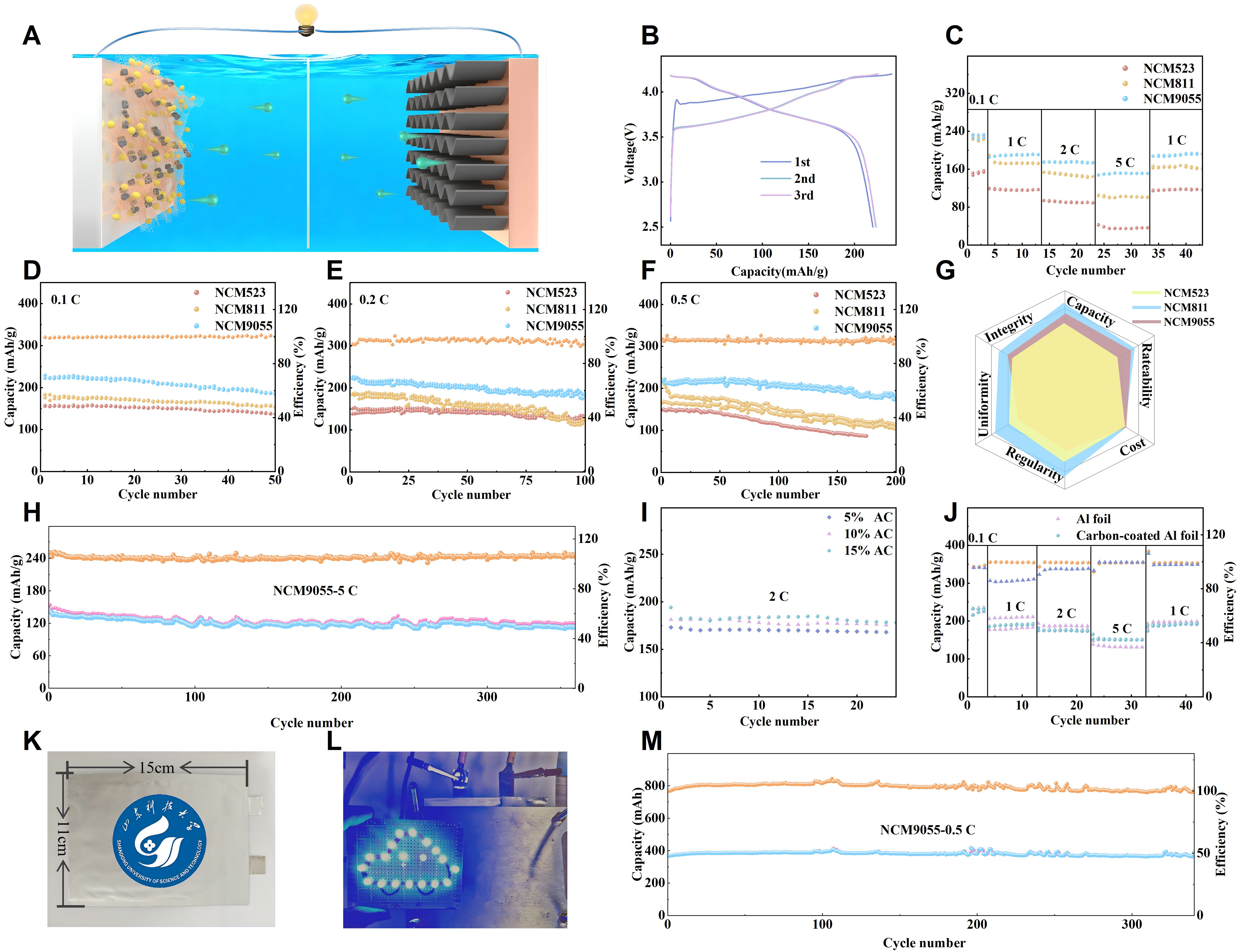
As illustrated in Figure 3A, NCM/AC composite was used as the cathode, while pure Li foil was adopted as the anode to assemble a coin type cell for testing the performance of the as prepared NCM/AC cathode. Given the highly reactive delithiated NCM9055 surface and the large specific surface area of AC, the hybrid system is prone to parasitic side reactions. To clarify the electrolyte effects and identify an optimal formulation, we evaluated the electrochemical compatibility of three carbonate based electrolytes (1 M LiPF6 with different carbonate solvents) for the hybrid electrode, with the rate performance comparison shown in Supplementary Figure 12. The system utilizing 1M LiPF6 in EC:DMC (1:1 vol%) exhibited the most superior rate capability and stability among the tested formulations. Mechanistically, this enhanced performance is attributed to the formation of a robust cathode electrolyte interphase (CEI) derived from the preferential oxidation of EC/DMC[50]. This CEI layer acts as a physical and chemical barrier, isolating the highly oxidized Ni species on the NCM surface from the bulk solvent, thereby suppressing the continuous oxidative decomposition of electrolyte molecules[51]. In contrast, electrolytes containing Propylene Carbonate (PC) or Diethyl Carbonate (DEC) trigger severe capacity degradation, which is attributed to their lower anodic stability on high Ni surfaces and the formation of thicker, electronically insulating byproducts on the AC pores[52].
Figure 3. Electrochemical performances of NCM/AC composite cathodes. (A) Schematic simulation of the charge-discharge process in NCM cathodes; (B) Charge-discharge profiles of NCM9055/AC cathode during the initial three cycles at 0.2 C; (C) Rate performance of three NCM/AC cathodes; (D) Cycling performance of NCM/AC cathodes at 0.1 C; (E) Cycling performance of NCM/AC cathodes at 0.2 C; (F) Cycling performance of NCM/AC cathodes at 0.5 C; (G) Radar chart comparison of three NCM/AC composite cathode; (H) Cycling performance of NCM9055/AC cathode at 5 C; (I) Comparison of different AC proportions; (J) Rate performance comparison of the NCM9055/AC/GN composite electrode deposited on a bare aluminum foil and a carbon-coated aluminum foil; (K) Digital photograph taken by the authors of the pouch HBS featuring an internal one-cathode and two-anodes configuration and dimensions of 15 cm × 11 cm, encapsulated in aluminum-laminated film; (L) Digital photograph taken by the authors showing the pouch HBS powering a set of light bulbs; (M) Cycling performance of the as-prepared pouch HBS at 0.5 C. NCM: Lithium nickel cobalt manganese oxide; AC: activated carbon; GN: graphene; HBS: hierarchical binder system; C: C-rate (charge/discharge rate); Al: aluminum.
To elucidate the specific enhancement induced by GN, a comparative study was conducted between SP-only and SP+GN, as shown in Supplementary Figure 13. AC serves as the primary capacitive component that defines the HBS system, while GN acts as a critical electron transport bridge in the conductive network. Comparative rate performance indicates that the SP+GN-based electrode exhibits superior capacity retention at high-current densities. This enhancement is attributed to the distinct geometry of GN unlike the discrete point-to-point contact of SP nanoparticles, GN nanosheets provide continuous point-plane contact with the NCM and AC particles. This architecture effectively minimizes interparticle contact resistance and ensures rapid electron supply to the active sites during fast charging/discharging[53-55].
With the optimized electrolyte, composition, and conductive network,in Figure 3B, the first cycle Coulombic efficiency of NCM9055/AC based HBS reached 91.7%, which is an exceptional value for high Ni cathode materials. The high initial efficiency implies that irreversible Li loss during the formation of the NCM9055/AC CEI is controlled at a low level, laying a robust foundation for the long term cycling stability of the as prepared HBS. Beginning with the second cycle, the electrochemical performance rapidly stabilizes, with efficiencies in the second and third cycles exceeding 99.5%, and the voltage platforms nearly completely overlapping. This observation confirms that Li+ intercalation and deintercalation within the layered structure is a highly reversible process[46,47].
The rate performance test results in Figure 3C clearly display a performance hierarchy among the three NCM/AC cathodes. NCM523/AC based cathode displays a capacity of 157 mAh/g at 0.1 C and a poor rate performance, with capacity retention dropping sharply to 23% at 5 C, mainly due to its intrinsically slow Li+ diffusion and low electronic conductivity causing severe polarization. NCM811/AC based cathode, representing high Ni materials, shows a high capacity of 219 mAh/g at 0.1 C and a better retention of 45.7% at 5 C, benefiting from the enhanced intrinsic conductivity and wider Li transmission channels provided by the higher Ni content. NCM9055/AC based cathode achieves the highest specific capacity of 231 mAh/g at 0.1 C, and the capacity retention at 5 C is significantly improved to 66.2%, which can be attributed to the superior spherical morphology, higher Ni percentage, and improved electrode microstructure[56]. These advantages of the NCM9055/AC cathode can shorten the solid phase diffusion path of Li+, increase the electrode/electrolyte contact area, and facilitate the construction of a more perfect conductive network. These improvements collectively promote rapid ion and electron transport, mitigate polarization at high rates, and achieve a more effective balance between high capacity and power output[46]. These three sets of comparative data demonstrate that NCM9055 and AC does not enhance the capacity by simply increasing the Ni content. Instead, through optimized material design, the inherent drawbacks of poor cycling stability and rate performance in high Ni materials were successfully addressed, providing both a high energy density and guaranteed long service life under high rates[25]. The specific capacities reported in this work are normalized solely to the mass of the NCM active material. Since the AC component (a minor weight fraction) acts as a conductive and capacitive buffer analogous, this calculation approach allows for a direct assessment of the intrinsic utilization rate of the NCM material. This methodology aligns with that adopted in previous studies on hybrid composite cathodes[57,58], ensuring the reported electrochemical values faithfully reflect the intrinsic performance of the high Ni active material.
Figure 3D shows that at a low rate of 0.1 C, NCM9055 based cathode delivers the highest capacity. The initial discharge specific capacity was 239 mAh/g, far exceeding those of NCM811 and NCM523. While materials with high Ni content often encounter challenges related to cycling stability, the findings presented in Figures 3E and F indicate that cathodes based on uniform NCM9055 can effectively integrate high capacity with favorable cycle life. As the rate increased, the gap in the capacity retention between NCM9055 and NCM811/NCM523 widened. At 0.2 C, the initial discharge capacity of the NCM9055 based cathode was
Figure 3G presents a comprehensive comparison across five key dimensions. While NCM523 benefits from the inherent stability of its Mn rich framework and NCM811 suffers from instability due to its high Ni content, NCM9055 achieves a remarkable balance. Crucially, the radar chart highlights that NCM9055 possesses the highest degree of morphological regularity among the samples. This superior particle uniformity and sphericity effectively mitigated the local stress concentration and maintained the lattice integrity during Li+ extraction/insertion. Consequently, this morphological advantage directly translates to its leadership in both structural stability and rate capability, far exceeding the performance limits of conventional high Ni materials.
The robustness provided by the optimized morphology is further evidenced in Figure 3H. Even under an extreme rate of 5 C, the NCM9055/AC composite cathode delivered a high initial discharge capacity of
Because AC has good conductivity but contributes to poor capacity, varying the AC percentage affects the NCM cathode performance. Increasing AC content facilitates the construction of a continuous electron/ion transport network, thereby enhancing the rate capability of the composite cathode. Since the specific capacity and tap density of porous AC are intrinsically lower than dense NCM material, excessive AC loading inevitably dilutes the overall gravimetric and volumetric energy densities of the eletrode. As shown in Figure 3I, by exploring the impact of different AC ratios (5%, 10%, and 15%) on the NCM9055 cathode performance, we found a significant correlation between the AC content and discharge specific capacity at different current densities. At 2 C, as the AC ratio increased from 5% to 15%, the specific capacity increased from 173 to 182 mAh/g; when the AC content increased to 15%, the discharge capacity increased to
In the application of NCM9055/AC cathode materials, carbon coated aluminum foil exhibits significant performance differences compared to conventional Al foil. Figure 3J shows that the NCM9055/AC cathode with a carbon coated foil demonstrates superior rate performance. At current densities of 0.1 C, 1 C, 2 C, and 5 C, the discharge specific capacities reached 231, 189, 175, and 153 mAh/g, respectively, whereas the conventional foil yielded only 216, 180, 177, and 131 mAh/g, respectively. The carbon coated foil leads comprehensively, whether in terms of capacity output or charge/discharge efficiency, particularly at high rates. The carbon coating constructs an efficient conductive network, reducing the interfacial impedance between the current collector and cathode material, effectively alleviating polarization at high rates, reducing the area for initial side reactions, preserving more active Li, and exhibiting higher Coulombic efficiency.
As shown in Figure 3K, a soft pack HBS was fabricated using one NCM9055/AC composite cathode
To evaluate the competitiveness of the proposed architecture, we introduced Supplementary Table 5 to benchmark the NCM9055 and AC composite against advanced energy storage systems reported in literature in recent years. Regarding power capability, the device delivers a high power density of 1,828 W·kg-1. This represents a threefold increase compared to conventional high Ni cathodes which are typically limited to approximately 550 W·kg-1 due to sluggish kinetics[60]. In terms of energy storage, the system maintains an energy density of 55.8 Wh·kg-1. This value notably surpasses that of standard Li+ capacitors and other capacitive devices reported[61,62], which generally fall below 46 Wh·kg-1. The system exhibits exceptional durability with 99% capacity retention after 340 cycles. This stability significantly outperforms similar hybrid modification strategies[63] that often suffer from rapid decay owing to interface instability.
Although the current energy and power densities are not yet exceptional, these results are achieved with a configuration consisting of one cathode layer and two anode layers. With an increase in the number of cathode and anode layers, both energy and power densities are anticipated to improve significantly, thereby enhancing the system’s potential for promising applications.
To further elucidate the origin of the superior cycling stability, the surface chemistry of the NCM9055/AC electrode after long-term 5 C cycling was characterized via XPS. As shown in Supplementary Figure 16, XPS results confirm the formation of a stable CEI film on the electrode surface. Specifically, the F 1s spectrum reveals a dominant peak at ~685.0 eV, corresponding to LiF, an inorganic species derived from the decomposition of the LiPF6[64]. which is widely recognized as a critical species for stabilizing the electrode surface by facilitating Li+ diffusion while suppressing continuous electrolyte decomposition. The C 1s spectrum displays peaks assigned to conductive carbon (C-C, 284.8 eV) and organic carbonate species with C–O and C=O bonds, which are consistent with the presence of organic carbonate decomposition products detected in the O 1s spectrum. The coexistence of this inorganic LiF rich layer and the organic carbonate matrix constructs a robust and protective CEI layer, which effectively mitigates side reactions and preserves the structural integrity of the high Ni cathode during high rate cycling.
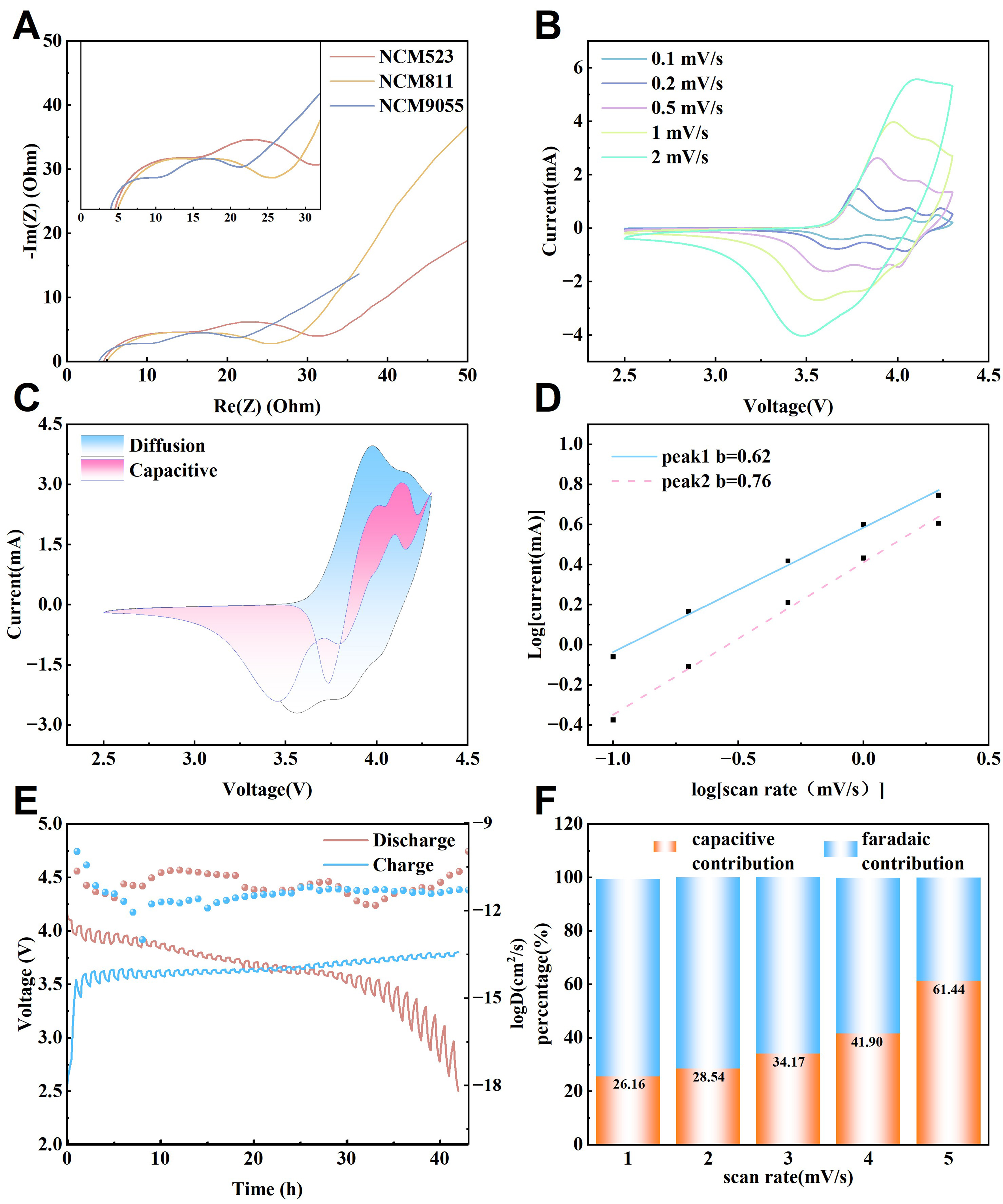
Subsequently, EIS, CV, and GITT tests, along with capacitive contribution studies, were conducted on these three NCM based cathodes, including NCM523, NCM811, and NCM9055. A comprehensive comparison of these three materials was performed to screen for the HBS cathode active material with superior performance. As shown in Figure 4A, the EIS results for the three NCM materials revealed significant differences in the charge transfer resistance, following the order NCM9055 < NCM811 < NCM523. Notably, NCM9055 exhibited the smallest semicircle diameter, indicating the lowest interfacial charge transfer impedance. This implies the lowest reaction energy barrier for Li+ at the electrode/electrolyte interface, resulting in the most rapid charge transfer process. In contrast, NCM523 exhibited the highest interfacial impedance, severely limiting its electrochemical performance at high rates. This result explains the intrinsic reason for the superior rate performance of NCM9055 from the perspective of the interfacial kinetics. The fact that the plot does not start at zero is attributed to the unavoidable system resistance, such as the internal contact resistance of the electrode or the contact resistance between the current collector and the coating[25,65].
Figure 4. Quantitative analysis of the capacitive contribution of NCM/AC cathodes. (A) Impedance comparison of three NCM/AC composite cahodes; (B) CV curves of NCM9055/AC at different scan rates; (C) Relationship between the capacitive contribution and scan rate for NCM9055/AC cathode; (D) Logarithmic relationship between the peak current and scan rate for NCM9055/AC cathode; (E) GITT curves of NCM9055/AC cathode; (F) Capacitive contribution ratios of NCM9055/AC cathode at different scan rates. NCM: Lithium nickel cobalt manganese oxide; AC: activated carbon; CV: cyclic voltammetry; GITT: galvanostatic intermittent titration technique.
In Supplementary Figure 17, the NCM9055 electrode exhibits significantly lower Surface Film Resistance (Rsf) and Charge Transfer Resistance (Rct) values compared to the control samples. This reduction suggests that the constructed conductive network not only facilitates the formation of a thinner, more stable SEI film but also drastically accelerates electron and ion transfer kinetics during redox reactions.
The CV test results clearly reflect the differences in the electrochemical behaviors of the three materials. In Supplementary Figure 18A, NCM523 exhibited typical battery type material CV characteristics; its oxidation/reduction peaks were distinctly separated and broad, indicating that its reaction kinetics were limited by the bulk diffusion of Li+, resulting in significant polarization. In Supplementary Figure 19A, the CV curve of NCM811 shows the most severe polarization phenomenon, with the largest peak potential difference and an asymmetric peak shape. This is closely related to the severe cation mixing and interfacial instability caused by the high Ni content, which hinders the progress of the reversible reactions. Conversely, the CV curve of NCM9055 in Figure 4B is the best, featuring the sharpest and most symmetric oxidation/reduction peaks with the smallest peak potential difference. This indicates the highest reaction reversibility and fastest charge transfer kinetics, laying the foundation for a high rate performance[25].
In Supplementary Figure 18B, the charge storage of NCM523/AC cathode mainly stems from a diffusion controlled battery type process. Its capacitive contribution ratio was the lowest and increased slightly even at higher scan rates, which determined the bottleneck in its rate performance. As shown in Supplementary Figure 19B, the capacitive contribution of NCM811/AC cathode was higher than that of NCM523/AC, stemming from the more significant Faradaic pseudocapacitance behavior on the surface of the high Ni materials. However, its overall capacity and absolute contribution are limited by its intrinsically poor bulk diffusion capability. NCM9055/AC cathode (in Figure 4C) demonstrated the highest capacitive contribution ratio at all scan rates, which intuitively explains its superior rate performance. While maintaining a high capacitive contribution, its diffusion controlled battery-type capacity contribution is far higher than those of the other two, achieving synergy between high capacity and high power. The wing-like intersection in the figure reflects the competition and switching of the charge storage mechanisms in the NCM9055/AC composite electrode at high rates. On the left side of the oxidation peak and the right side of the reduction peak, the current was dominated by non Faradaic electric double layer charging and a trace pseudocapacitive current. Because the capacitive current of AC increases linearly with the scan rate and has little dependence on the potential, the current value of the high scan rate curve may exceed that of the low scan rate curve in these potential regions. Near the peak potential, the diffusion controlled Faradaic current at low scan rates was strong, whereas at high scan rates, it was limited by diffusion, resulting in slower peak growth. Consequently, the baseline of the high scan rate curve crossed above the low scan rate curve but remained lower at the peak summit, thereby forming intersections on both sides of the peak. This proves that in the non peak potential regions, capacitive behavior dominates with a rapid response, whereas in the peak potential regions, the diffusion kinetics of the battery material limits the current at high scan rates. The NCM/AC composite cathodes achieved an effective combination of two energy storage mechanisms, retaining the high capacity of the battery material while utilizing AC to provide the surface capacitance characteristics required for high power[25,26].
In Supplementary Figure 18C, the b values for both the oxidation and reduction peaks of NCM523 based cathode are the lowest, confirming that the energy storage process is dominated by diffusion control. The b value of NCM811/AC in Supplementary Figure 19C increases, indicating an increased proportion of surface controlled behavior, but it still does not break free from the limitations of diffusion control. In Figure 4D, the b value of NCM9055/AC is closest to 1, providing a solid mathematical basis for capacitive contribution analysis. This strongly proves that its charge storage mechanism is dominated by surface controlled processes, which is the fundamental reason for its ability to achieve a capacitor like rapid charge discharge response.
NCM523/AC in Supplementary Figure 20A exhibits typical diffusion limited characteristics. Its GITT curve displays large voltage steps and slow relaxation, indicating a high resistance to Li+ migration within the lattice and a low diffusion coefficient. This directly leads to poor rate performance and severe capacity decay at high rates. NCM811/AC [Supplementary Figure 20B] showed improved kinetics with reduced voltage steps and faster relaxation. The relaxation was incomplete, and the plateau stability was insufficient, which indicates that high Ni content materials still encounter significant diffusion barriers owing to factors such as phase transitions during Li intercalation/deintercalation, limiting the performance under extreme conditions. In comparison, the GITT curve of NCM9055/AC in Figure 4E demonstrates superior kinetic properties, the smallest voltage steps and fastest relaxation speeds prove that it possesses the highest Li+ diffusion coefficient. While achieving a Ni content of approximately 90%, NCM9055 constructs unobstructed Li+ transport channels by optimizing the crystal structure (e.g., by reducing cation mixing). The chemical Li+ diffusion coefficients (DLi+) derived from GITT are presented in Supplementary Figure 21. To clearly visualize the variation of DLi+ which spans across orders of magnitude during the phase transitions, the values are plotted on a logarithmic scale (logD), consistent with previous literature[66-68]. The NCM9055 sample demonstrates faster kinetic behavior, its DLi+ values fall in the range of 1.073 × 10-10 to 1.218 × 10-12 cm2·s-1, corresponding to logD values of approximately -9.9 to -13.0, which are notably higher than those of NCM811 and NCM523 across the entire voltage window. As a result, NCM9055 not only possesses bulk ionic conductivity surpassing those of NCM523 and NCM811, providing the core guarantee for its superior rate performance and fast charging capability, but also supports an ultralong cycle life through its structural robustness, achieving a unification of energy density, power density, and cycling stability[46].
Moreover, as displayed in Figure 4F, with an increase in the scan rate, the proportion of the capacitive contribution increased from 34.17% to 61.44%. This proves that at high scan rates, the rapid surface capacitive process becomes dominant, thereby avoiding the severe capacity decay caused by slow ion diffusion.
The evolution and breakthrough of the energy storage mechanisms from NCM523 to NCM9055 can be clearly observed through the analysis of the three capacitive contribution figures. NCM523/AC cathode [Supplementary Figure 18D] was dominated by slow bulk diffusion control, resulting in limited rate performance. NCM811/AC in Supplementary Figure 19D shows some improvement but remains limited by diffusion at high rates. In contrast, NCM9055/AC in Figure 4F achieves a fundamental shift towards capacitive behavior dominance[25]. As summarized in Supplementary Table 6, distinct kinetic trends are observed among the three cathodes. The cathode based on NCM9055 displays the highest b values, in contrast to the relatively low b values of NCM523 and NCM811, suggesting a significant shift towards fast, surface dominated kinetics. This trend is further quantified by separating the capacitive controlled current (k1v) from the diffusion-controlled current (k2v1/2). The NCM9055 electrode demonstrates the highest capacitive contribution ratio of 61.44% at 2 mV/s, which is markedly higher than those of NCM811 (25.54%) and NCM523 (33.44%).The enhanced pseudocapacitive behavior of the NCM9055 system is attributed to the synergistic coupling of the high surface area AC network and the optimized NCM surface, which facilitates rapid ion adsorption/desorption at the electrode-electrolyte interface and effectively mitigates the polarization typically induced by bulk Li+ diffusion[69,70]. A comprehensive comparison confirms that NCM9055 is not merely a high capacity material but also a kinetic powerhouse. It overcomes the diffusion limitations of NCM523 and the stability issues of NCM811, achieving a unification of fast surface-dominated kinetics and efficient bulk Li+ transport.
As shown in Figures 5A-E, after 50 cycles, the spherical structure of NCM9055 remained relatively intact, with a smooth surface and no obvious large cracks. After 100 cycles, microcracks appeared along the radial direction or grain boundaries in some particles, which resulted from stress accumulation caused by lattice volume changes during repeated Li+ intercalation/deintercalation. After 200 cycles, crack propagation became evident, and some particles fractured into smaller ones, leading to an increased specific surface area that exacerbated side reactions and electrolyte decomposition. The SEM image after 360 cycles shows that the pristine spherical morphology nearly disappeared, and the particles began to fracture[71]. Supplementary Figure 22 reveals that after 200 cycles, although the secondary spherical morphology of NCM523 remained recognizable, the particle surface exhibited obvious corrosion marks and roughening. In Supplementary Figure 23, NCM811 demonstrated more pronounced failure characteristics after 200 cycles. The secondary sphere structure experienced significant cracking and pulverization, resulting in the near disappearance of particle outlines and the formation of substantial debris and irregular agglomerates. This highlights the superior structural stability and electrochemical reliability of the uniformed NCM9055 material. Focusing on the early stage of cycling (in Supplementary Figure 24), the secondary spherical morphology of NCM9055 was maintained remarkably well. This indicates that the material possessed excellent mechanical integrity during the initial cycles, effectively delaying the initiation and propagation of cracks. Owing to this robust structure, HBS based on NCM9055/AC cathodes can deliver an extremely stable and reliable performance, and the tolerance far exceeds that of conventional high Ni materials, demonstrating the competitiveness as a high performance cathode material[72,73].
Figure 5. SEM images and elemental mapping results of NCM9055/AC cathode after different cycles. (A) SEM image of NCM9055/AC cathode after 50 cycles at 5 C; (B) SEM image of NCM9055/AC cathode after 100 cycles at 5 C; (C) SEM image caught under EDS mode of NCM9055/AC cathode after 360 cycles at 5 C; (D) SEM image of NCM9055/AC cathode after 200 cycles at 5 C; (E) SEM image of NCM9055/AC cathode after 360 cycles at 5 C; (F) EDS mapping of Li, C, and O for NCM9055/AC cathode. SEM: Scanning electron microscopy; EDS: energy-dispersive X-ray spectroscopy; NCM: lithium nickel cobalt manganese oxide; AC: activated carbon; Li: lithium; C: carbon; O: oxygen; C: C-rate (charge/discharge rate).
As shown in Supplementary Figure 25, NCM9055/AC cathode exhibited superior structural and interfacial stability after 360 cycles at an ultra high rate of 5C. The intensity stable phosphate characteristic peak at ~1,100 cm-1 indicates that the material rapidly constructed a dense CEI film under high rate conditions, effectively suppressing continuous side reactions and the growth of impedance. Meanwhile, the characteristic peaks at ~1,350 cm-1 (D band), ~1,580 cm-1 (G band), and ~2,700 cm-1 (2D band) remained highly stable throughout the long term cycling, proving the AC conductive skeleton did not degrade, and ensuring consistently efficient electron transport pathways. In Supplementary Figure 26, the NCM523 secondary spheres have clear outlines, but a broad particle size distribution and relatively rough surfaces. The elemental distribution was generally uniform, but with a weaker signal intensity, reflecting the stability and limitations of traditional materials. In Supplementary Figure 27, NCM811 exhibited significant particle refinement, and the micropores and initial cracks appeared on the surface. NCM9055 [Supplementary Figure 28] presented a regular secondary spherical morphology, highly uniform particle size, and a smooth and dense surface, indicating that the preparation process achieved excellent morphology control. Its elemental mapping, particularly the Ni and O signals, exhibited clear features that highly coincided with the particle morphology. This proves that the transition metals and oxygen achieved an atomic level uniform distribution within and between particles, with no obvious component segregation or surface enrichment.
The Energy Dispersive X-ray Spectroscopy (EDS) elemental mapping in Figure 5F reveals that the major metal elements (Ni, Co, and Mn) maintained a highly uniform spatial distribution within the composite electrode. This observation is further corroborated by the EDS sum spectrum in Supplementary Figure 29, which confirms the stable preservation of the distinctive chemical composition after 360 cycles. Collectively, these results indicate that no significant elemental segregation, loss, or phase separation (particularly of Ni) occurred. Furthermore, the corresponding original SEM images presented in Supplementary Figure 30 (related to the magnified regions in Figure 5) visually validate that the NCM9055/AC cathode retained superior structural integrity without severe particle pulverization or collapse. A significant enrichment of F and C elements appeared on the particle surface, representing the conductive carbon network (from AC and SP) in the electrode and potentially formed carbonate decomposition products. The continuous network structure proves that the conductive skeleton remained intact after cycling, ensuring the continuity of electron conduction. F mainly originates from the decomposition of the electrolyte Li salts (such as LiPF6). Its weak and uniform signal distribution is a positive indicator, suggesting that the formed CEI is thin, stable, and dense, without severe electrolyte decomposition side reactions. These changes lead to increased interfacial impedance, loss of active material, and capacity fading, which are consistent with the degradation characteristics of high Ni layered materials during long term cycling[74].
CONCLUSIONS
Through systematic investigation, this study successfully developed a HBS cathode system that integrates high energy density, high power density, and ultra-long cycle life by compositing high-Ni NCM9055 with AC. The NCM9055/AC composite cathodes delivered a peak specific capacity of 231 mAh/g at 0.1 C and exhibited superior rate capability, achieving a reversible capacity of 153 mAh/g at 5 C with 79.4% retention after 360 cycles. This performance is mainly attributed to the high Ni content, uniform morphology, and enhanced structural stability. Furthermore, the assembled pouch HBS (featuring one NCM9055/AC cathode and two graphite anodes) achieved an energy density of 55 Wh/kg and a power density of 1,828 W/kg, while maintaining a capacity retention of 99% after 340 cycles at 0.5 C. Mechanistically, the incorporation of AC not only establishes a robust 3D conductive network to reduce charge transfer impedance but also provides unobstructed Li+ diffusion channels. This structural advantage effectively mitigates stress accumulation and leverages a surface-dominated storage mechanism to boost rate performance. Post-cycling SEM and EDS analyses further confirmed that the electrode maintains intact structural morphology and chemical stability, demonstrating superior mechanical and interfacial durability. Consequently, this study confirms that the NCM9055/AC composite bridges the performance gap between traditional batteries and supercapacitors, offering a viable material solution and technical pathway for next-generation high-performance hybrid energy storage devices.
DECLARATIONS
Authors’ contributions
Conceptualization: Qiu, F.
Visualization, writing - original draft: Wang, X.
Data curation: Wang, X.; Shi, H.
Methodology: Qiu, F.; Wang, C.
Investigation: Wang, X.; Shi, H.; Song, Y.; Zheng, X.; Liu, R.; He, Y.; Lu, X.
Supervision: Qiu, F.; Yang, Z.; Wang, C.
Writing - review and editing: Qiu, F.; Yang, Z.; Wang, C.
Availability of data and materials
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author(s).
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Science and Technology Projects of China Southern Power Grid (YNKJXM20240030, YNKJXM20240033), the Shandong Province Natural Science Foundation (ZR2024ME026, ZR2024QC122), and Qingdao Natural Science Foundation (24-4-4-zrjj-177-jch).
Conflicts of interest
Qiu, F., Wang, X., Song, Y., Zheng, X., Liu, R., and He, Y. are affiliated with Electric Power Research Institute of Yunnan Power, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Yu, C.; Luo, H.; Jin, Z.; et al. High-efficiency energy storage: high entropy materials design and interface regulation mechanism. Adv. Funct. Mater. 2025, 36, e11308.
2. Thomas, S. A.; Cherusseri, J.; Rajendran, D. N. Rapid synthesis of hierarchical cobalt disulfide nanostructures by microwave-assisted hydrothermal method for high performance supercapatteries. ACS. Appl. Electron. Mater. 2024, 6, 4321-35.
3. Park, K. J.; Choi, M. J.; Maglia, F.; et al. High-capacity concentration gradient Li[Ni0.865Co0.120Al0.015]O2 cathode for lithium-ion batteries. Adv. Energy. Mater. 2018, 8, 1703612.
4. He, T.; Kang, X.; Wang, F.; et al. Designing capacitive contribution in hard carbon materials for balancing energy and power under high current density for sodium-ion batteries. ACS. Appl. Energy. Mater. 2023, 6, 8149-57.
5. Liu, L.; Zhang, X.; Liu, Y.; Gong, X. Electrochemical energy storage devices - batteries, supercapacitors, and battery-supercapacitor hybrid devices. ACS. Appl. Electron. Mater. 2025, 7, 2233-70.
6. Wang, X.; Chen, W.; Shi, X.; et al. Microfluidics-assisted fabrication of all-flexible substrate-free micro-supercapacitors with customizable configuration and high performance. Adv. Energy. Mater. 2023, 13, 2203535.
7. Zhu, Q.; Zhao, D.; Cheng, M.; et al. A new view of supercapacitors: integrated supercapacitors. Adv. Energy. Mater. 2019, 9, 1901081.
8. Gao, D.; Luo, Z.; Liu, C.; Fan, S. A survey of hybrid energy devices based on supercapacitors. Green. Energy. Environ. 2023, 8, 972-88.
9. Xing, F.; Bi, Z.; Su, F.; Liu, F.; Wu, Z. S. Unraveling the design principles of battery-supercapacitor hybrid devices: from fundamental mechanisms to microstructure engineering and challenging perspectives. Adv. Energy. Mater. 2022, 12, 2200594.
10. Li, X.; Lou, D.; Wang, H.; Sun, X.; Li, J.; Liu, Y. N. Flexible supercapacitor based on organohydrogel electrolyte with long-term anti-freezing and anti-drying property. Adv. Funct. Mater. 2020, 30, 2007291.
11. Anjana, P.; Aminabhavi, T. M. Supercapattery: energy storage devices combining functionalities of battery electrodes and supercapacitor electrodes. J. Energy. Storage. 2025, 134, 118265.
12. Zhang, G.; Hu, J.; Nie, Y.; et al. Integrating flexible ultralight 3D Ni micromesh current collector with NiCo bimetallic hydroxide for smart hybrid supercapacitors. Adv. Funct. Mater. 2021, 31, 2100290.
13. Ye, F.; Yang, W.; Liao, X.; Dong, C.; Xu, L.; Mai, L. A micro battery supercapacitor hybrid device with ultrahigh cycle lifespan and power density enabled by Bi-functional coating design. Adv. Funct. Mater. 2024, 35, 2413379.
14. Zhou, X.; Zheng, X.; Dong, X.; et al. Recent advances in wearable fiber-shaped supercapacitors: materials, design, and applications. Energy. Mater. 2025, 5, 500048.
15. Hu, Y.; Wan, Z.; Song, H.; et al. Ultralong cycle stability poly(benzodifurandione)/Ti3C2Tx films as novel self-supporting electrodes for supercapacitors. J. Energy. Storage. 2025, 135, 118279.
16. Myung, S.; Maglia, F.; Park, K.; et al. Nickel-rich layered cathode materials for automotive lithium-ion batteries: achievements and perspectives. ACS. Energy. Lett. 2016, 2, 196-223.
17. Dong, H.; Zhang, L.; Liao, Y.; et al. Floating catalyst chemical vapor deposition patterning nitrogen-doped single-walled carbon nanotubes for shape tailorable and flexible micro-supercapacitors. Adv. Funct. Mater. 2023, 33, 2301103.
18. Liu, T.; Yan, R.; Huang, H.; et al. A micromolding method for transparent and flexible thin-film supercapacitors and hybrid supercapacitors. Adv. Funct. Mater. 2020, 30, 2004410.
19. Chen, Y.; Wang, Z.; Mei, X.; Wang, Q.; Xie, H.; Li, Y. Nitrogen and oxygen-codoped dense porous carbon enhancing ion adsorption for high-volumetric performance supercapacitors. Energy. Mater. 2025, 5, 500146.
20. Xu, J.; Dou, S.; Zhou, W.; et al. Scalable waste-plastic-derived carbon nanosheets with high contents of inbuilt nitrogen/sulfur sites for high performance potassium-ion hybrid capacitors. Nano. Energy. 2022, 95, 107015.
21. Cai, S.; Zhou, X.; Wang, Y.; Lu, X. Advanced carbonaceous materials for Zn-ion hybrid supercapacitors: status and perspectives. Energy. Mater. 2025, 5, 500085.
22. Agustiana, D.; Daraz, U.; Siburian, D. M.; Lyu, P.; Liu, H. Cathode materials for advanced lithium-ion batteries: developments, challenges, and emerging trends. Mater. Res. Bull. 2026, 195, 113842.
23. Kim, U.; Myung, S.; Yoon, C. S.; Sun, Y. Extending the battery life using an Al-doped Li[Ni0.76Co0.09Mn0.15]O2 cathode with concentration gradients for lithium ion batteries. ACS. Energy. Lett. 2017, 2, 1848-54.
24. Sun, X.; Zhang, X.; Huang, B.; Zhang, H.; Zhang, D.; Ma, Y. (LiNi0.5Co0.2Mn0.3O2 + AC)/graphite hybrid energy storage device with high specific energy and high rate capability. J. Power. Sources. 2013, 243, 361-8.
25. Han, Y.; Wang, Z.; Xie, L.; et al. Revealing the accelerated reaction kinetic of Ni-rich cathodes by activated carbons for high performance lithium-ion batteries. Carbon 2023, 203, 445-54.
26. Chen, X.; Mu, Y.; Cao, G.; et al. Structure-activity relationship of carbon additives in cathodes for advanced capacitor batteries. Electrochim. Acta. 2022, 413, 140165.
27. Su, F.; Qin, J.; Das, P.; Zhou, F.; Wu, Z. A high-performance rocking-chair lithium-ion battery-supercapacitor hybrid device boosted by doubly matched capacity and kinetics of the faradaic electrodes. Energy. Environ. Sci. 2021, 14, 2269-77.
28. Iqbal, M. Z.; Aziz, U. Supercapattery: merging of battery-supercapacitor electrodes for hybrid energy storage devices. J. Energy. Storage. 2022, 46, 103823.
29. Li, H.; Zhang, W.; Sun, K.; et al. Manganese-based materials for rechargeable batteries beyond lithium-ion. Adv. Energy. Mater. 2021, 11, 2100867.
30. Yan, Z.; Luo, S.; Li, Q.; Wu, Z. S.; Liu, S. F. Recent advances in flexible wearable supercapacitors: properties, fabrication, and applications. Adv. Sci. 2024, 11, e2302172.
31. Yang, E.; Shi, X.; Wu, L.; et al. A low-cost moderate-concentration hybrid electrolyte of introducing CaCl2 and ethylene glycerol enables low-temperature and high-voltage micro-supercapacitors. Adv. Funct. Mater. 2024, 34, 2313395.
32. Zhao, W.; Jiang, M.; Wang, W.; Liu, S.; Huang, W.; Zhao, Q. Flexible transparent supercapacitors: materials and devices. Adv. Funct. Mater. 2020, 31, 2009136.
33. Wang, B.; Guo, R.; Zheng, M.; et al. Embedded binary functional materials/cellulose-based paper as freestanding anode for lithium ion batteries. Electrochim. Acta. 2018, 260, 1-10.
34. Zhang, Y. S.; Courtier, N. E.; Zhang, Z.; et al. A review of lithium-ion battery electrode drying: mechanisms and metrology. Adv. Energy. Mater. 2021, 12, 2102233.
35. Saqib, K. S.; Choi, J. H.; Park, S.; et al. High-mass loading nickel-rich cathode electrode design incorporating multidimensional carbon conductive additives to minimize ohmic contact resistance for high-performance lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 48429-39.
37. Stoller, M. D.; Park, S.; Zhu, Y.; An, J.; Ruoff, R. S. Graphene-based ultracapacitors. Nano. Lett. 2008, 8, 3498-502.
38. Hsu, T. H.; Liu, W. R. Effects of graphene nanosheets with different lateral sizes as conductive additives on the electrochemical performance of LiNi0.5Co0.2Mn0.3O2 cathode materials for li ion batteries. Polymers 2020, 12, 1162.
39. Mu, J.; Zhang, L.; He, R.; et al. Enhancing the electrochemical performance of LiNi0.5Mn1.5O4 cathode material by a conductive LaCoO3 coating. J. Alloys. Compd. 2021, 865, 158629.
40. Li, Y.; Wu, X.; Wang, J.; Gao, X.; Hu, Y.; Wen, Z. Ni-less cathode with 3D free-standing conductive network for planar Na-NiCl2 batteries. Chem. Eng. J. 2020, 387, 124059.
41. Kim, J. H.; Kim, H.; Choi, W.; Park, M. S. Bifunctional surface coating of LiNbO3 on high-Ni layered cathode materials for lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2020, 12, 35098-104.
42. Ryu, H.; Park, K.; Yoon, C. S.; Sun, Y. Capacity fading of Ni-Rich Li[NixCoyMn1-x-y]O2 (0.6 ≤ x ≤ 0.95) cathodes for high-energy-density lithium-ion batteries: bulk or surface degradation? Chem. Mater. 2018, 30, 1155-63.
43. Ko, D. S.; Park, J. H.; Yu, B. Y.; et al. Degradation of high-nickel-layered oxide cathodes from surface to bulk: a comprehensive structural, chemical, and electrical analysis. Adv. Energy. Mater. 2020, 10, 2001035.
44. Gao, X.; Li, B.; Rousse, G.; et al. Achieving high-voltage stability in Li-Rich Ni-Rich oxides with local W/Ni(Li) superstructure. Adv. Energy. Mater. 2024, 15, 2402793.
45. Vivekanantha, M.; Sundhar Arul Saravanan, R.; Kumar Nayak, P.; Prakash, R.; Kamala Bharathi, K. Synergistic-effect of high Ni content and Na dopant towards developing a highly stable Li-Rich cathode in Li-ion batteries. Chem. Eng. J. 2022, 444, 136503.
46. Wu, Z.; Zhang, C.; Yuan, F.; et al. Ni-rich cathode materials for stable high-energy lithium-ion batteries. Nano. Energy. 2024, 126, 109620.
47. Park, G. T.; Park, N. Y.; Ryu, H. H.; Sun, H. H.; Hwang, J. Y.; Sun, Y. K. Nano-rods in Ni-rich layered cathodes for practical batteries. Chem. Soc. Rev. 2024, 53, 11462-518.
48. Kim, E. J.; Tatara, R.; Hosaka, T.; Kubota, K.; Kumakura, S.; Komaba, S. Effects of particle size and polytype on the redox reversibility of the layered Na0.76Ni0.38Mn0.62O2 electrode. ACS. Appl. Energy. Mater. 2024, 7, 1015-26.
49. Taleghani, S. T.; Marcos, B.; Zaghib, K.; Lantagne, G. A study on the effect of porosity and particles size distribution on Li-ion battery performance. J. Electrochem. Soc. 2017, 164, E3179-89.
50. Ma, S.; Zhang, X.; Wu, S.; et al. Unraveling the nonlinear capacity fading mechanisms of Ni-rich layered oxide cathode. Energy. Storage. Materials. 2023, 55, 556-65.
51. Takahashi, I.; Kiuchi, H.; Ohma, A.; Fukunaga, T.; Matsubara, E. Cathode electrolyte interphase formation and electrolyte oxidation mechanism for Ni-Rich cathode materials. J. Phys. Chem. C. 2020, 124, 9243-8.
52. Li, B.; Zheng, J.; Zhang, H.; et al. Electrode materials, electrolytes, and challenges in nonaqueous lithium-ion capacitors. Adv. Mater. 2018, 30, e1705670.
53. Han, C.; Shi, F.; Wei, D.; et al. A three-dimensional points-lines-planes interweaved conductive additive with ultrahigh electron transfer for high-performance supercapacitors. ACS. Appl. Energy. Mater. 2024, 7, 4462-71.
54. Shi, Y.; Wen, L.; Pei, S.; Wu, M.; Li, F. Choice for graphene as conductive additive for cathode of lithium-ion batteries. J. Energy. Chem. 2019, 30, 19-26.
55. Hwang, J.; Do, K.; Ahn, H. Highly conductive 3D structural carbon network-encapsulated Ni-rich LiNi0.8Co0.1Mn0.1O2 as depolarized and passivated cathode for lithium-ion batteries. Chem. Eng. J. 2021, 406, 126813.
56. Zhao, W.; Zhang, R.; Ren, F.; et al. Protective coating of single-crystalline Ni-Rich cathode enables fast charging in all-solid-state batteries. ACS. Nano. 2025, 19, 8595-607.
57. Shellikeri, A.; Yturriaga, S.; Zheng, J.; et al. Hybrid lithium-ion capacitor with LiFePO4/AC composite cathode - long term cycle life study, rate effect and charge sharing analysis. J. Power. Sources. 2018, 392, 285-95.
58. Liao, J.; Manthiram, A. Surface-modified concentration-gradient Ni-rich layered oxide cathodes for high-energy lithium-ion batteries. J. Power. Sources. 2015, 282, 429-36.
59. Chen, H.; Xia, X.; Ma, J. Comprehensive review of Li-Rich Mn-based layered oxide cathode materials for lithium-ion batteries: theories, challenges, strategies and perspectives. ChemSusChem 2024, 17, e202401120.
60. Sun, X.; Zhang, X.; Zhang, H.; Xu, N.; Wang, K.; Ma, Y. High performance lithium-ion hybrid capacitors with pre-lithiated hard carbon anodes and bifunctional cathode electrodes. J. Power. Sources. 2014, 270, 318-25.
61. Bhowmik, S.; Dutta, J.; Ansari, T. A.; Martha, S. K. Design and performance analysis of high surface area carbon and LiNi0.8Mn0.1Co0.1O2 composite as cathode for lithium-ion battery capacitors. Electrochim. Acta. 2025, 515, 145703.
62. Eleri, O. E.; Lou, F.; Yu, Z. Lithium-ion capacitors: a review of strategies toward enhancing the performance of the activated carbon cathode. Batteries 2023, 9, 533.
63. Dong, S.; Yao, H.; Qin, Z.; Li, W.; Liu, Y.; Zhao, Y. Multifunctional surface engineering of Ni-Rich layered cathodes for ultra-stable lithium-ion batteries. Chemistry 2026, 32, e02108.
64. Jiao, T.; Liu, G.; Zou, Y.; et al. A novel trimethylsilyl 2-(fluorosulfonyl)difluoroacetate additive for stabilizing the Ni-rich LiNi0.9Co0.05Mn0.05O2/electrolyte interface. J. Power. Sources. 2021, 515, 230618.
65. Liu, Z.; Wang, D.; Kou, X.; et al. High-performance oxygen reduction electrocatalysts derived from bimetal-organic framework and sulfur-doped precursors for use in microbial fuel cells. J. Power. Sources. 2022, 521, 230944.
66. Liu, K.; Yang, J.; Wang, H.; et al. Achieving fast charging and superior cycling stability single-crystal Ni-Rich cathodes by ultrafast aqueous washing. Adv. Sci. 2026, 13, e17421.
67. Chu, M.; Huang, Z.; Zhang, T.; et al. Enhancing the electrochemical performance and structural stability of Ni-Rich layered cathode materials via dual-site doping. ACS. Appl. Mater. Interfaces. 2021, 13, 19950-8.
68. Xiang, H.; Ren, H.; Liu, Y.; Yang, D.; Feng, X. High loading electrode with superior electron/ion transport network for high performance lithium-ion batteries. J. Power. Sources. 2024, 609, 234672.
69. Zhou, S.; Huang, P.; Xiong, T.; et al. Sub-thick electrodes with enhanced transport kinetics via in situ epitaxial heterogeneous interfaces for high areal-capacity lithium ion batteries. Small 2021, 17, e2100778.
70. Qiu, L.; Zhang, M.; Hua, W.; et al. Unveiling surface reconstruction as the primary trigger for capacity loss in ultra-high nickel cathodes. Angew. Chem. Int. Ed. Engl. 2025, 64, e202417278.
71. Wang, G.; Huang, C.; Hu, C.; et al. Dynamic pinning by embedded La4LiNiO8 for novel and advanced high-nickel cathode materials of LiNi0.9Co0.085Ti0.015O2. Chem. Eng. J. 2025, 523, 168403.
72. Tang, W.; Shu, Z.; Li, A.; Huang, X.; Li, W. Enhancing structural integrity and long-term cycling stability of high-voltage single-crystalline Ni-rich cathodes via surface/subsurface dual-functional modification engineering. Energy. Storage. Materials. 2025, 77, 104185.
73. Liao, Q.; Guo, S.; Qi, M.; et al. The genesis and control of microcracks in nickel-rich cathode materials for lithium-ion batteries. Sustainable. Energy. Fuels. 2023, 7, 4805-24.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data




















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].