Multifunctional origami-structured triboelectric nanogenerators based on zinc coordination polymers for self-powered photoinduced oxidation systems toward green energy harvesting
Abstract
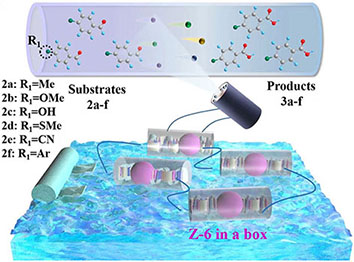
Development of multifunctional triboelectric nanogenerators (TENGs) capable of efficiently harvesting diverse low-frequency mechanical energies for self-powered systems remains a significant challenge. To address this issue, we designed and fabricated a zigzag-origami-structured TENG based on composite films by integrating a zinc coordination polymer (Zn-CP) with ethylcellulose (EC), aiming to convert human-motion and water-wave energies into electricity to drive a self-powered photo-induced oxidation system. A series of flexible Zn-CP@EC composite films with varying Zn-CP contents were prepared, among which the 10% Zn-CP@EC composite film exhibited the best triboelectric performance. By scaling the film dimensions and integrating multiple origami-structured 10% Zn-CP@EC-TENGs (Z-TENGs), the output performance was further enhanced, with the six-unit device (Z-6) showing the best performance under palm pressure. The Z-6 device, encapsulated in a plastic enclosure, was deployed in an oscillating water tank to harvest wave energy, which successfully powered LEDs as light sources for the photo-induced oxidation of aldehydes to carboxylic acids with high selectivity and efficiency. This work demonstrates that CP-based composite films can serve as effective triboelectric materials for scalable TENGs, enabling the realization of self-powered photochemical systems driven by diverse environmental mechanical energies.
Keywords
INTRODUCTION
With the continuous and rapid exploitation of fossil energy resources, energy-related challenges have become increasingly pronounced, leading to severe ecological environment degradation[1-4]. Consequently, the development of self-sustaining power generation systems that utilize renewable and clean energy sources has attracted growing attention and become an urgent societal and scientific demand[5-9]. As one of the most abundant and clean renewable energy sources on earth, mechanical energy offers significant potential for sustainable power generation, spurring the demand for advanced flexible and eco-friendly energy-harvesting devices[10-13]. Triboelectric nanogenerators (TENGs), based on electrostatic induction and contact electrification, have emerged as a promising technology for converting environmental mechanical energy into electricity, owing to their simple fabrication, readily available materials, and scalability[14-21]. Various strategies, including material selection and functionalization, and micro/nano- structural engineering, have been explored to enhance TENG output performance[22-26]. However, existing TENG devices still face a critical limitation: their insufficient efficiency in harvesting human motion and water-wave energy, which are characterized by low, random, and unpredictable frequencies[16,17,27-31]. This limitation underscores the critical need for targeted structural design and material optimization to improve TENG performance.
Hybrid organic-inorganic materials have been recognized as advanced triboelectric layers for TENGs, as they synergistically tune carrier diffusion lengths, band gap, and dielectric characteristics by combining organic and inorganic components[32-37]. Coordination polymers (CPs), as a class of hybrid organic-inorganic materials with highly ordered reticular structures, possess high structural designability, unique porous properties and high specific surface areas, providing robust platforms for triboelectric layers[38,39]. Their well-defined frameworks with precise distributions of metal nodes and organic ligands enable regulation of electron transport, polarity, and charge storage, thereby enhancing triboelectric performance[40-42]. However, the practical applications of CP-based triboelectric materials are hindered by their inherent deficiencies, such as poor stability, difficulty in large-scale preparation, and insufficient mechanical strength[38,41]. To address these issues, recent efforts have focused on developing CP-based self-supporting composites with excellent mechanical stability and outstanding processability[43,44]. In particular, integrating CPs with organic polymers has been proven effective in improving mechanical robustness and processability. However, most reported CP-based composite films are still fabricated into conventional planar TENG structures, which suffer from low space utilization and restricted contact area - key bottlenecks for efficient harvesting of low-frequency mechanical energy[45-56]. Thus, it is necessary to develop novel CP-based composite films with optimized structures for scalable low-frequency energy collection[57-59].
In this study, we designed a suitable and rationally designed zigzag-origami TENG that integrates numerous parts within a limited space and enables them to operate under different types of low-frequency mechanical energy. To achieve higher output performance and simulate practical applications in real-world conditions, Zn-CP@EC composite films were prepared by introducing a zinc coordination polymer (Zn-CP) into the organic polymer ethylcellulose (EC) at varying ratios, identifying the optimal Zn-CP content (10%) for peak triboelectric performance. These films were used to construct a scalable zigzag-origami structured 10% Zn-CP@EC-based TENG (Z-TENGs), capable of harvesting human movement and water-wave energy. To further augment performance and demonstrate scalability, we constructed multi-unit integrated devices, denoted as Z-2, Z-4, and Z-6, corresponding to systems comprising two, four, and six 10% Zn-CP@EC-TENG units, respectively. Moreover, the Z-6 device can be enclosed in a box to simulate water waves and harvest wave energy, converting it into electricity, which can continuously and sustainably drive the light-emitting diodes (LEDs) to provide illumination for executing self-powered photo-induced reactions without any energy devices. This self-powered system exhibited outstanding ability to promote the photo-induced oxidation of aldehydes to carboxylic acids.
EXPERIMENTAL
Materials. 4,5-di(2H-tetrazole-5-yl)-1H-imidazole-2-amino (NH2-H3idbt) was synthesized according to improved methods in the literature[60]. 2-Amino-4,5-dicyano-1H-imidazole (97%) was purchased from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). Triethylamine hydrochloride (99%) was bought from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). N, N-dimethylformamide (DMF) and Zinc sulfate heptahydrate (ZnSO4·7H2O) were obtained from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). P-Tolualdehyde (2a, 97%), Anisic aldehyde (2b, 98%), 4-Hydroxybenzaldehyde (2c, 97%), 4-(Methylthio) benzaldehyde (2d, 98%), 4-cyanobenzaldehyde (2e, 98%), and p-phenylbenzaldehyde (2f, 98%) were bought from Anhui Zesheng Technologies Co., Ltd. (Anhui, China). All reagents and solvents were commercially available without further purification.
Characterizations. The detailed characterization instruments, such as powder X-ray diffraction (PXRD), thermal gravimetric analyzer (TGA), and X-ray photoelectron spectroscopy (XPS) of Zn-CP and composite materials were given in the Supplementary Materials. Meanwhile, the Single-Crystal X-ray diffraction (SCXRD) test and crystal data were provided in the Supplementary Tables 1 and 2.
Synthesis of {[Zn(NH2-H3idbt)(H2O)]·H2O}n (Zn-CP). NH2-H3idbt (22 mg, 0.1 mmol), ZnSO4·7H2O
Assembly of Zn-CP@EC-TENGs. 3.75 g of polyvinylidene fluoride (PVDF), 8.5 g of DMF and 12.75 g of acetone were charged into a 50 mL round-bottom flask and stirred at 60 °C for 30 min. After cooling to room temperature, the mixed solution was spin-coated onto a Kapton film at 2,000 rpm for 90 s, then baked in an oven at 80 °C for 4 h to obtain the PVDF film. The back side of the Kapton film was subsequently attached to the adhesive side of a Cu tape, and a copper wire was bonded to the rear of the Cu tape with conductive silver epoxy to complete the negative electrode.
Dissolve powder EC (0.4 g) in DMF (6 mL) and magnetically stir at 100 °C for 30 min to obtain a homogeneous EC solution. In a separate centrifuge tube, disperse Zn-CP in 6 mL DMF at mass fractions of 1%, 5%, 10%, 15% and 20%, and sonicate the mixture for 30 min to ensure uniform dispersion. Combine the two solutions and continue stirring for 4 h to obtain a mixed solution. Cast the solution onto a clean glass plate and dry in an oven at 80 °C for 30 min to form a film. After peeling off the film, cut it into 5 cm × 5 cm squares and adhere each square to the adhesive side of a Cu tape. A copper wire was then bonded to the back of the copper tape with conductive silver epoxy, yielding Zn-CP@EC composite-film electrodes containing different mass fractions of Zn-CP. Spin-coated PVDF film and different-proportion Zn-CP@EC composite films were used as the negative and positive friction materials, respectively. They were combined to fabricate Zn-CP@EC-TENG devices, working in the vertical contact-separation mode with an effective contact area of 5 × 5 cm2.
Preparation of Z-2 device with different contact areas. Two PET films were cut into different size rectangles (1.5 × 5, 2.5 × 8, 3.5 × 11, 4.5 × 14, and 5.5 × 17 cm2, respectively). Two positive electrodes (1 × 1, 2 × 2, 3 × 3,
Preparation of Z-2, Z-4, Z-6 measured at 5 × 5 cm2. Two PET films were cut into different size rectangles (5.5 × 12, 5.5 × 22, and 5.5 × 32 cm2, respectively). Two, four, or six 5 × 5 cm2 positive electrodes and an equal number of 5 × 5 cm2 negative electrodes were attached to the upper and lower sides of each of the two PET films, respectively. The two PET films were then positioned perpendicular to each other, bonded together, alternately folded, and finally sealed to complete the Z-2, Z-4, Z-6 device.
RESULTS AND DISCUSSION
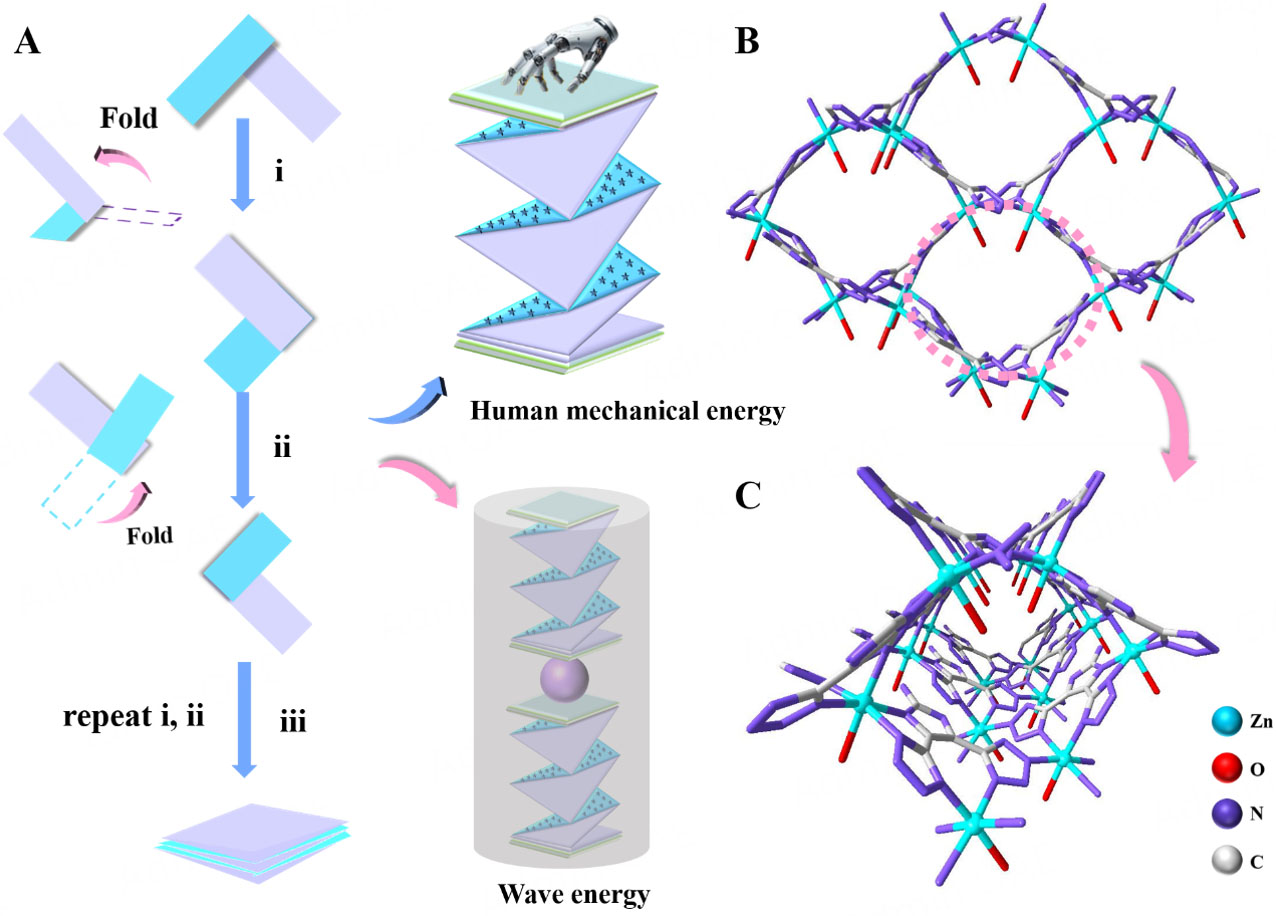
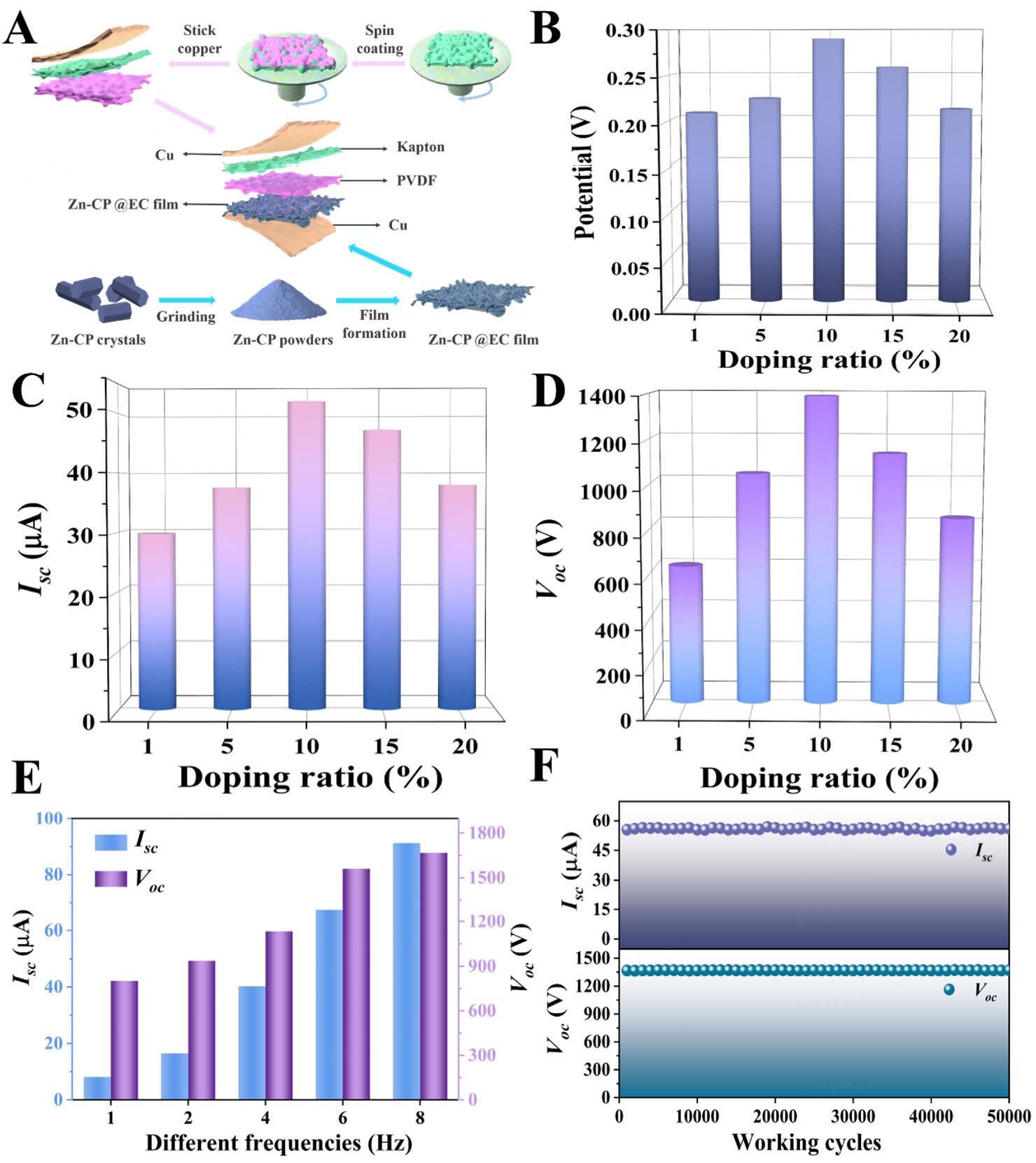
This study aims to design and fabricate a zigzag-origami-structured triboelectric nanogenerator [Figure 1A] based on Zn-CP@EC composite films for efficiently harvesting human-motion and water-wave energies to drive a self-powered photo-induced oxidation system. To that end, Zn-CP was synthesized by reacting NH2-H3idbt ligand and ZnSO4·7H2O to yield colorless single crystals, which served as photocatalyst and triboelectric material to explore their bifunctional properties. Single-crystal X-ray diffraction analysis (SCXRD) of Zn-CP, which has the space group Pna21, showed an orthorhombic net with a diamond-shaped framework containing hexanuclear ZnII secondary building units (Zn-SBUs). The corresponding formula for Zn-CP was obtained by SCXRD, XPS, TGA, elemental analysis (EA), and charge-balance analyses [Supplementary Figure 1-3]. In Zn-CP, the asymmetric unit comprised one NH2-Hidbt2- ligand, one ZnII ion, and one coordinated H2O molecule, respectively. The ZnII ion had the characteristic octahedral geometry, ligated by one O atom from one coordinated H2O molecule and five N atoms from three NH2-Hidbt2- ligands [Supplementary Figure 4]. The fully deprotonated tetrazole groups had bridging and chelating coordination modes to connect with ZnII ions, generating a hexanuclear ZnII secondary building unit (Zn-SBUs) [Supplementary Figure 5]. Furthermore, these Zn-SBUs were interconnected by the coordination bonds of tetrazole-N atoms and Zn ions to form a diamond-shaped channel. These channels were bridged by tetrazole rings to construct a three-dimensional (3D) architecture [Figure 1B and C, Supplementary Figure 6].
Figure 1. (A) Schematic illustration of CP-based TENG for the self-powered photoinduced applications. (B) A view of the 3D network of Zn-CP. (C) Perspective view of the open rhomboid channel.
To achieve precise molecular insights into the influence of the Zn-CP-based TENG output performance, the NH2-H3idbt ligand and Zn-CP were milled and assembled as positive-polarity triboelectrodes with negative-polarity polyvinylidene fluoride (PVDF) to fabricate Zn-CP-TENG and NH2-H3idbt-TENG, respectively. As shown in Supplementary Figure 7, the operation mode of the TENGs was regular contact-separation, where the flow of electrons was driven by the positive and negative triboelectric materials in the external circuit. The triboelectric output signals indicated that the Zn-CP-TENG demonstrated a much higher output performance, with short-circuit current (Isc) and open-circuit voltage (Voc) approximately two times higher than those of NH2-H3idbt-TENG [Supplementary Figure 8]. The higher outputs were mainly due to Zn-CP with a periodic and uniform arrangement of metal centers and ligands in the 3D network, which could improve the electron-donating capability to generate electrons significantly more easily and produce much more charge to enhance the Zn-CP-TENG outputs[61,62].
To enhance TENG output and improve the adaptability of CPs as triboelectric materials, organic polymers were integrated with CPs, the surface of the CPs was periodically modified, and well-proportioned composite films were fabricated. These films allow surface charge density (σ) to accumulate rapidly to a high initial value to provide a stable and high current in a brief period to increase the TENG outputs. Owing to its excellent electron-donating properties, film-forming capability, and flexibility, the organic polymer EC, which ranks just below Nylon-6 in the triboelectric series, was used to fabricate a series of Zn-CP@EC composite films [Figure 2A]. To assess the effect of Zn-CP on TENG output, Zn-CP@EC films with different Zn-CP contents (ranging from 1% to 20%) were prepared. These films were used as triboelectric layers to assemble Zn-CP@EC-TENGs. The scanning electron microscopy (SEM) images indicate that Zn-CP was firmly and uniformly loaded onto the EC to successfully induce Zn-CP@EC composite films
Figure 2. (A) Assembly process of Zn-CP@EC-based TENG. (B) KPFM surface potential of Zn-CP @EC with different weight ratios, respectively. (C and D) Isc and Voc of the TENGs based on Zn-CP@EC with different doping ratios, respectively. (E) Isc and Voc of the TENGs based on Zn-CP@EC under different operating frequencies. (F) Isc and Voc of 10% Zn-CP@EC-based TENG after working 50,000 cycles.
Furthermore, the output of Zn-CP@EC-TENG with different proportions was explored at 5 Hz under identical test conditions. As shown in Figure 2C and D, when the Zn-CP contents in EC increased from 1% to 10%, the Isc and Voc values of Zn-CP@EC-TENG enhanced from 29.8 μA and 626.9 V to 51.9 μA and 1,393.8 V, respectively. This was caused by the enhancement of the tribopositive polarity of the Zn-CP@EC composite films through the interfacial polarization between Zn-CP and EC in the composite films[63,64]. Nevertheless, the outputs declined as the Zn-CP content rose to 15% or 20%, and the corresponding Isc and Voc peaks for 15% Zn-CP@EC-TENG and 20% Zn-CP@EC-TENG were 46.7 μA and 1,141.5 V and 37.3 μA and 826.7 V, respectively. This deterioration was attributed to the coacervation of Zn-CP within the Zn-CP@EC films, which resulted in a poor triboelectric performance and weak mechanical properties[65,66]. These results revealed that the output of Zn-CP@EC-TENG was closely related to the Zn-CP content of the Zn-CP@EC films. The incorporation of low Zn-CP concentration improved the Zn-CP@EC-TENG output, and the output performance of Zn-CP@EC-TENG achieved the largest values when the percentage of Zn-CP in Zn-CP@EC film was 10%, which was consistent with the results of KPFM surface potentials.
Having successfully built a connection between the Zn-CP content in EC and Zn-CP@EC-TENG outputs, we next sought to study the environmental suitability and stability by using the optimal 10% Zn-CP@EC-TENG device. It could be seen that the outputs (Isc and Voc) of 10% Zn-CP@EC-TENG gradually increased as the operating frequency (1-8 Hz) increased, with the largest values at 8 Hz. This observation could be attributed to the reduction in the 10% Zn-CP@EC-TENG impedance because of the increased strain [Figure 2E]. The output power density of 10% Zn-CP@EC-TENG was examined by employing the formula P = I2R, and the maximum power densities were 4151.2 mW m-2 under a load of 30 MΩ [Supplementary Figure 13]. The corresponding outputs of 10% Zn-CP@EC-TENG were larger than those of CPs composites or CP-based TENGs [Supplementary Table 3][67-70]. Accordingly, stability and durability tests showed that the 10% Zn-CP@EC-TENG output exhibited hardly any significant decrease after 50,000 working cycles [Figure 2F]. In particular, the 10% Zn-CP@EC-TENG maintained its original performance even after being retested after two months [Supplementary Figure 14]. Moreover, the 10% Zn-CP@EC-TENG worked continuously for 36 h at 5 Hz (equivalent to 648,000 cycles), and the corresponding output performance did not decline significantly [Supplementary Figure 15]. These results confirm that the 10% Zn-CP@EC-TENG has excellent cycle performance and stable output, making it suitable for further research.
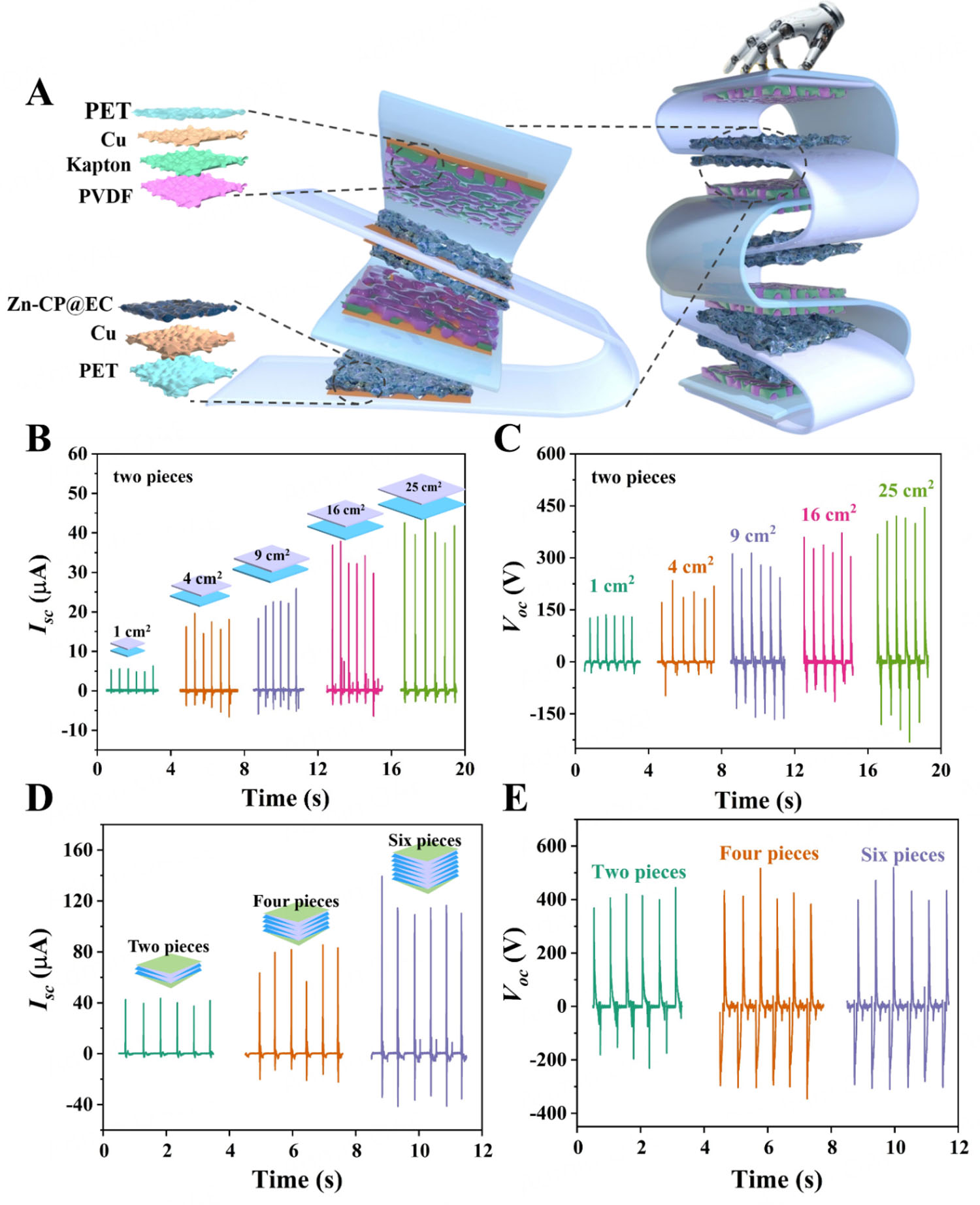
Subsequently, the corresponding 10% Zn-CP@EC composite film and PVDF of the assembled 10% Zn-CP@EC-TENG were employed to integrate the zigzag-origami 3D structure devices, which had the flexibility and stretchability to integrate a large number of TENG units for enhancing the output performance. As shown in Figure 3A and Supplementary Figure 16, the zigzag-origami-structured 10% Zn-CP@EC-TENG was fabricated from two highly elastic strips that were repeatedly folded to create the compact, elastic device. In Z-TENGs, one strip as the tribonegative layer was formed from PVDF, Kapton, Cu, and FEP (fluorinated ethylene propylene) units. The other strip as the tribopositive layer consisted of the 10% Zn-CP@EC, Cu, and FEP units. These two flexible equal-length strips could be cyclically folded and staggered to create a zigzag-origami structure with a clear crease pattern, which was beneficial for freeing space to construct a series of multilayered Z-TENGs. Furthermore, the Z-TENGs displayed good stretching and compression performance during the linear-reciprocating contact-separation process, indicating that only a very small driving force was required to achieve and harvest mechanical energy. As a result, the different contact areas and numbers of origami monomer contact separation Z-TENGs composed of two 10% Zn-CP@EC-TENG units, named Z-2, were designed to explore and optimize output performance by monitoring the hand palm pressing. A series of Z-2 with different dimensions (1 × 1, 2 × 2, 3 × 3, 4 × 4, and
Figure 3. (A) Schematic diagram of Z-6 device. (B and C) Isc and Voc of 1, 4, 9, 16, and 25 cm2 Z-2, respectively. (D and E) Isc and Voc of Z-2, Z-4, Z-6 measured at 25 cm2.
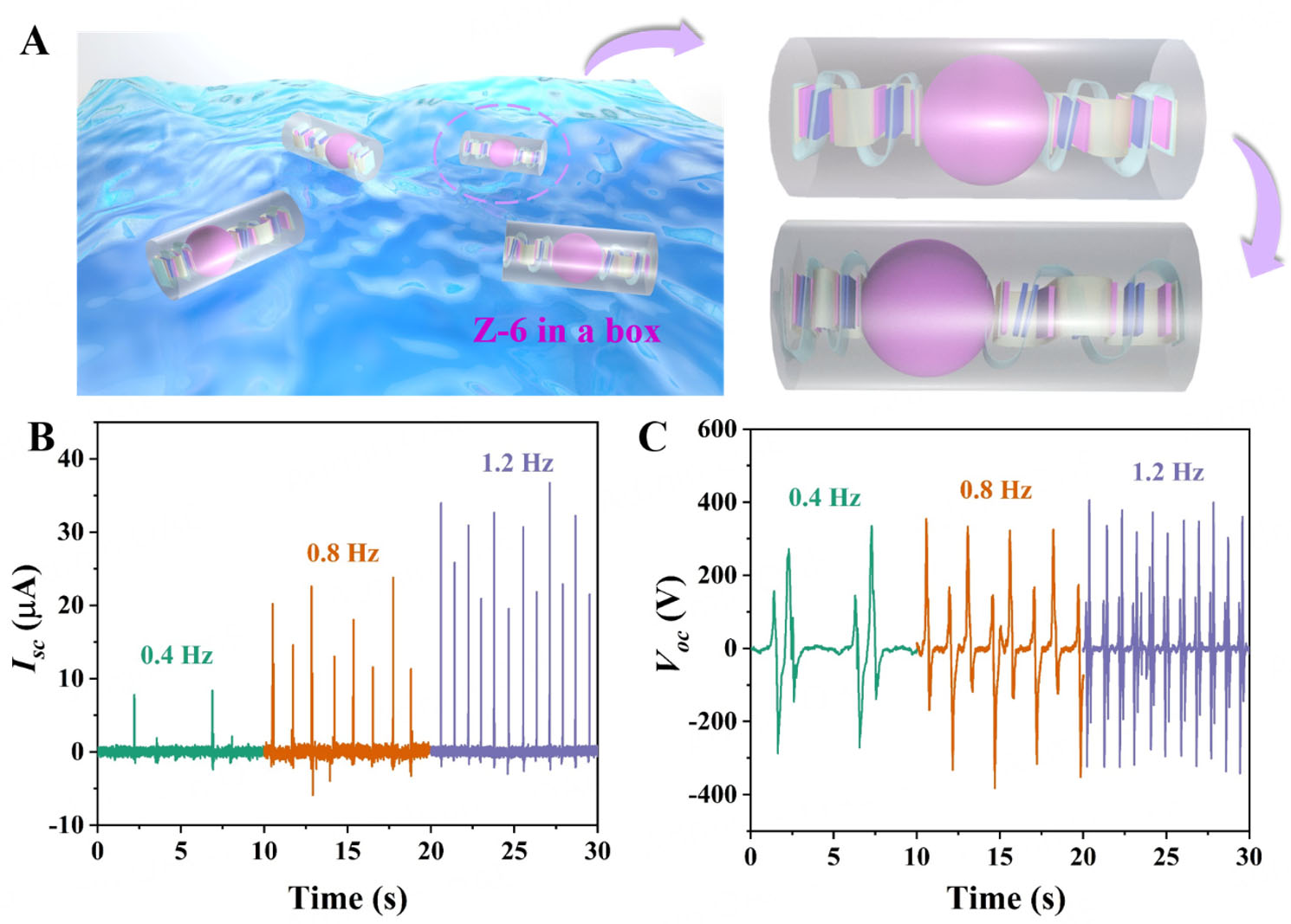
To further simulate and measure the actual application of the Z-6 device for harvesting environmental energy, a plastic box containing two Z-6 devices with a copper ball was placed in a water tank to imitate wave motions in the laboratory. As shown in Figure 4A, a motor was used to generate water waves to drive the copper ball back and forth, which leveraged the fluctuation of the waves to realize the contact and separation of the origami Z-6 device for harnessing wave energy. The corresponding outputs were examined at different water-wave frequencies [Figure 4B and C]. As the operating frequencies increased from 0.4 to 1.2 Hz, the Isc and Voc increased, peaking at 30.9 μA and 372.3 V under 1.2 Hz water waves. The results indicated that as the operating frequency increased, the triboelectric layer contact of Z-6 in the motion process became more sufficient, resulting in improved output performance. Furthermore, the electronic devices of the thermohygrometer and calculators could be effectively driven by continuously harvesting water-wave energy [Supplementary Videos 1 and 2], demonstrating the outstanding energy supply capacity of self-powered systems without additional storage units [Supplementary Figure 19]. Notably, by collecting water-wave energy, Z-6 devices quickly lit up a series of LEDs without any energy storage unit [Supplementary Figure 20 and Supplementary Video 3], making them a power supply to produce a successive light source for exploring a self-powered photochemical system.
Figure 4. (A) Schematic illustration of water-wave energy harvesting using the Z-6 device. (B and C) Isc and Voc of water-wave energy harvesting at different frequencies (0.4, 0.8, and 1.2 Hz, respectively).
Subsequently, a self-powered and sustainable photoinduced system was designed by employing origami Z-6 in an oscillating device to power LEDs, which could serve as a light source for photo-induced reactions. This simple, economical, and eco-friendly self-powered setup, harvests human movement energy, as well as collects wave energy in an oscillating device to convert electrical energy into driving continuous lighting simultaneously. Z-6 was used in this self-powered photo-induced system to harvest sustainable wave energy and drive LEDs as a light source for performing selective photo-induced reactions. p-Tolualdehyde (2a) was selected to evaluate the feasibility of the self-powered photo-induced selective oxidation reaction by combining it with Z-6 as a passive optical device. Preliminary screening experiments demonstrated that oxygen (O2) exhibited higher selectivity and efficiency than the other oxidants [e.g., air, I2, H2O2, or CF3SO2Na, respectively, Supplementary Table 4, entries 1-5]. Based on these results, a range of solvents, atmospheres, and light irradiation were explored [Supplementary Table 4, entries 6-10]. 2a could be stably converted into the single oxidation product, p-toluic acid (3a), in the presence of O2 in H2O, producing the desired carboxylic acid product 3a in a comparable yield (98%). Therefore, the most selective self-powered photo-induced oxidation reaction proceeded smoothly under O2/H2O/light-irradiation.
The self-powered photo-induced oxidation of 2a was monitored using 1H NMR spectroscopy [Supplementary Figure 21]. The peak of δ = 9.96 ppm for singlet hydrogen signal was assigned to the aldehyde group (CHO), which weakened step by step, confirming that self-powered photo-induced oxidation reaction from 2a to 3a proceeded stably. Correspondingly, the new singlet signal at δ = 12.80 ppm appeared and strengthened, which was attributable to the carboxyl group. The results demonstrated that product 3a was generated by prolonging the illumination time, suggesting that the self-powered photo-induced oxidation process was completed.
Under standard photocatalytic conditions, various functionalized benzaldehyde-based substrates (2a-f) were screened to evaluate their self-powered photo-induced activity [Table 1]. With Z-6 as the light source, 2a-f worked well and generated the corresponding carboxylic acid frameworks (3a-f) in acceptable-to-good yields. Notably, functional groups such as methyl, methoxy, and hydroxyl as electron-donating groups at the para-substitution sites on benzaldehyde substrates 2a-c, were well tolerated under similar reaction conditions and gave the sole benzoic acid products (3a-c) in excellent yields [Table 1, entries 1-3]. Importantly, 4-(methylthio)benzaldehyde (2d) and 4-cyanobenzaldehyde (2e), which bear oxidizable and base-labile groups on their aromatic rings, showed high reactivity and selectivity for desired products 3d and 3e
Procedure for the light-induced oxidation of aldehydes to carboxylic acids driven by Z-6 device
 | ||
| Entry | Substrates 2a-f | Yield of 3a-f (%)b |
| 1 | 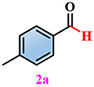 | 98% |
| 2 | 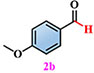 | 92% |
| 3 | 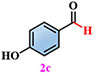 | 96% |
| 4 | 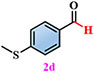 | 92% |
| 5 | 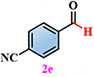 | 94% |
| 6 |  | 90% |
Furthermore, a selective self-powered photo-induced oxidation reaction was conducted using 2a as the substrate to explore the relationship between the Z-6 outputs and conversion efficiencies at 1.2 Hz and different reaction times. As shown in Supplementary Figure 22, the conversion efficiency of 2a gradually increased from 0% to 19% under light source at 4 h owing to the continuous and stable output of Z-6. When the illumination time (8, 12, 16, 20, 24, and 28 h, respectively) was extended, the conversion efficiency of 2a continued to increase. In contrast, 2a did not undergo any conversion without Z-6 to drive the LEDs light source for 28 h. Subsequently, the self-powered photo-induced oxidation process was investigated at different frequencies (0.4, 0.8, and 1.2 Hz, respectively) under similar conditions
To illustrate the mechanism of this oxidation reaction, control measurements were conducted with 2a. Normally, the results of light irradiation on/off tests revealed that the consecutive light irradiation of LEDs was necessary for the self-powered oxidation of 2a to 3a [Supplementary Figure 24]. Furthermore, the inhibition was examined by introducing the radical inhibitor 2, 2, 6, 6-tetramethylpiperidine-1-oxyl (TEMPO), and the corresponding target product 3a was not produced, which is consistent with the commonly accepted free radical chain process. A plausible mechanism is proposed based on the above results. Originally, the aldehydes were excited and converted to active species I under light irradiation. I could further generate radical pairs II and III through hydrogen atom transfer (HAT), which is known to participate in the reaction processes[73,74]. Subsequently, II interacted with O2 to provide peroxy radical IV, which could further perform HAT with aldehydes derivatives to generate peracetic acid V and radical II. The corresponding carboxylic acids were then produced by reacting aldehydes and V through a Baeyer-Villiger rearrangement. Meanwhile, radical III reacted with O2 to give peroxy radical VI, which could undergo HAT processes to transform into radical II and intermediate peroxide VII. VII added to the aldehydes to provide intermediate product VIII, which underwent intramolecular rearrangement to produce the desired carboxylic acids and aldehydes [Supplementary Scheme 1].
CONCLUSIONS
In summary, we designed and manufactured a flexible origami-structured TENG based on Zn-CP@EC composite films, realizing efficient harvesting of low-frequency mechanical energies from human motion and water waves for self-powered selective photo-induced systems. By integrating Zn-CP into EC, composite films with tunable triboelectric performance were obtained, among which the 10% Zn-CP@EC film delivered optimal and stable electrical output. By optimizing the film dimensions (25 cm2 as the optimal dimension) and stacking multiple origami units, the six-unit Z-6 device achieved superior output performance under palm pressure. Its encapsulated configuration efficiently captured wave energy, generating stable electricity to light LEDs as a continuous light source for the photo-induced oxidation of aldehydes to carboxylic acids with high selectivity and efficiency. This study underscores the potential of CP-based composite films as scalable triboelectric materials for multifunctional energy-harvesting applications. Future research will focus on the structural optimization and enhancement of the environmental stability of CPs as well as the expansion of their photochemical applications to advance the practical implementation of CP-based TENGs in green energy conversion and sustainable chemistry.
DECLARATIONS
Authors’ contributions
Investigation, methodology, data curation, writing - original draft: Zhao, C.
Supervision, formal analysis, writing - original draft: Zhang, Y. Y.
Validation, visualization: Cui, L.
Project administration, visualization: Wang, D.
Resources, formal analysis: Shao, Z.
Funding acquisition, supervision: Qin, Q.
Resources, writing - review & editing: Hou, H.
Conceptualization, supervision, funding acquisition: Huang, C.
Availability of data and materials
The data presented in this study are available on request from the corresponding author.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (52503283), Program for Innovative Research Team (in Science and Technology) in Universities of Henan Province (26IRTSTHN041), Program for Science & Technology Innovation Talents in Universities of Henan Province (22HASTIT030), Natural Science Foundation of Henan (252300420270), Program for Science & Technology Innovation Talents in Zhongyuan University of Technology (K2023YXRC02), Training Program for Young Backbone Teachers in Higher Education Institutions in Henan Province (2024GGJS101), Natural Science Foundation of Zhongyuan University of Technology (K2026ZD013), Program for Innovative Research Team of Zhongyuan University of Technology (K2025TD006), Postgraduate Education Reform and Quality Improvement Project of Henan Province (YJS2026AL071), and Postgraduate Education Reform and Quality Improvement Project of Zhongyuan University of Technologye (ALK202511).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Carley, S.; Konisky, D. M. The justice and equity implications of the clean energy transition. Nat. Energy. 2020, 5, 569-77.
2. Ye, J.; He, C.; Gong, X.; Zhang, H.; Li, X. Blue energy harvesting based on triboelectric nanogenerators (TENG): structural design, performance optimization, and application prospects. J. Alloys. Compd. 2025, 1014, 178710.
3. Dong, W.; Gao, Z.; Duan, Z.; et al. Sliding-mode cement-based triboelectric nanogenerators in intelligent infrastructure for a new energy harvesting paradigm. Mater. Today. Energy. 2025, 52, 101943.
4. Wu, X.; Cai, T.; Wu, Q.; et al. Droplet-based triboelectric nanogenerators with needle electrodes for efficient water energy harvesting. ACS. Appl. Mater. Interfaces. 2025, 17, 13762-72.
5. Yang, Y.; Wang, J.; Qian, W.; Wang, Z. L. Triboelectric nanogenerators as a clean energy scavenging technology. Mater. Today. 2025, 88, 839-54.
6. Zhang, C.; Hao, Y.; Yang, J.; et al. Magnetic suspension damped hybrid nanogenerator for water wave energy harvesting. Adv. Energy. Mater. 2025, 15, 2500130.
7. Heo, D.; Hur, J.; Cho, H.; et al. Nano-oil-barrier-based fluttering triboelectric nanogenerator. Adv. Sci. 2025, 12, e02278.
8. Davis, S. J.; Lewis, N. S.; Shaner, M.; et al. Net-zero emissions energy systems. Science 2018, 360, 6396.
9. Sohn, S. H.; Choi, G. J.; On, B. D.; Park, I. K. Synergistic coupling of tribovoltaic and moisture-enabled electricity generation in layered-double hydroxides. Adv. Energy. Mater. 2024, 14, 2304206.
10. Pan, X.; Yang, X.; Yu, M.; et al. 2D MXenes polar catalysts for multi-renewable energy harvesting applications. Nat. Commun. 2023, 14, 4183.
11. Wu, H.; Kang, Y.; Li, K.; et al. Secondary power generation via charge recombination in triboelectric nanogenerators. ACS. Energy. Lett. 2024, 9, 4516-23.
12. Rani, S.; Khandelwal, G.; Kumar, S.; et al. Flexible self-powered supercapacitors integrated with triboelectric nanogenerators. Energy. Storage. Mater. 2025, 74, 103977.
13. Gong, S.; Tang, B.; Liu, C.; et al. A synergistically enhanced triboelectric-electromagnetic hybrid generator enabled by multifunctional amorphous alloy for highly efficient self-powered system. Adv. Mater. 2025, 37, e2412856.
14. Nie, J.; Wang, Z.; Ren, Z.; Li, S.; Chen, X.; Lin, Wang. Z. Power generation from the interaction of a liquid droplet and a liquid membrane. Nat. Commun. 2019, 10, 2264.
15. Wu, C.; Wang, A. C.; Ding, W.; Guo, H.; Wang, Z. L. Triboelectric nanogenerator: a foundation of the energy for the new era. Adv. Energy. Mater. 2018, 9, 1802906.
16. Xiang, H.; Peng, L.; Yang, Q.; Wang, Z. L.; Cao, X. Triboelectric nanogenerator for high-entropy energy, self-powered sensors, and popular education. Sci. Adv. 2024, 10, eads2291.
17. Rui, P.; Zhang, W.; Wang, P. Super-durable and highly efficient electrostatic induced nanogenerator circulation network initially charged by a triboelectric nanogenerator for harvesting environmental energy. ACS. Nano. 2021, 15, 6949-60.
18. Zhang, Q.; Li, Y.; Cai, H.; et al. A single-droplet electricity generator achieves an ultrahigh output over 100 V without pre-charging. Adv. Mater. 2021, 33, e2105761.
19. Zhang, C.; Hao, Y.; Lu, X.; et al. Advances in TENGs for marine energy harvesting and in situ electrochemistry. Nanomicro. Lett. 2025, 17, 124.
20. Chi, M.; Cai, C.; Liu, Y.; et al. Aramid triboelectric materials: opportunities for self-powered wearable personal protective electronics. Adv. Funct. Mater. 2024, 34, 2411020.
21. Wang, T.; Shen, Y.; Chen, L.; et al. Large-scale production of the 3D warp knitted terry fabric triboelectric nanogenerators for motion monitoring and energy harvesting. Nano. Energy. 2023, 109, 108309.
22. Hou, K.; Dai, X.; Zhao, S.; Huang, L.; Li, C. A damage-tolerant, self-healing and multifunctional triboelectric nanogenerator. Nano. Energy. 2023, 116, 108739.
23. Kovalska, E.; Lam, H. T.; Saadi, Z.; et al. Textile beeswax triboelectric nanogenerator as self-powered sound detectors and mechano-acoustic energy harvesters. Nano. Energy. 2024, 120, 109109.
24. Zhang, F.; Zheng, L.; Li, H.; et al. Multifunctional triboelectric nanogenerator for wind energy harvesting and mist catching. Chem. Eng. J. 2024, 488, 150875.
25. Yang, R. A multifunctional triboelectric nanogenerator based on PDMS/MXene for bio-mechanical energy harvesting and volleyball training monitoring. Heliyon 2024, 10, e32361.
26. Yun, J.; Kim, D. Self-powered triboelectricity-driven multiple-input-single-output occupancy detection system using a triboelectric nanogenerator for energy management. Polymers 2024, 17, 34.
27. Han, J.; Dai, X.; Huang, L.; Hao, J. Optimizing electrical output performance of triboelectric nanogenerators by micro-/nano-morphology design and fabrication. Int. J. Extrem. Manuf. 2025, 7, 032008.
28. Liu, D.; Yang, P.; Gao, Y.; et al. A dual-mode triboelectric nanogenerator for efficiently harvesting droplet energy. Small 2024, 20, e2400698.
29. Lou, Y.; Li, M.; Yu, A.; Zhai, J.; Wang, Z. L. From wave energy to electricity: functional design and performance analysis of triboelectric nanogenerators. Nanomicro. Lett. 2025, 17, 298.
30. Kim, W. G.; Kim, D. W.; Tcho, I. W.; Kim, J. K.; Kim, M. S.; Choi, Y. K. Triboelectric nanogenerator: structure, mechanism, and applications. ACS. Nano. 2021, 15, 258-87.
31. Liang, X.; Jiang, T.; Liu, G.; Feng, Y.; Zhang, C.; Wang, Z. L. Spherical triboelectric nanogenerator integrated with power management module for harvesting multidirectional water wave energy. Energy. Environ. Sci. 2020, 13, 277-85.
32. Alghamdi, M. S.; Morgan, J. J.; Walsh, K.; et al. Triboelectric nanogenerator based on cellulose nanocrystals and graphene for energy harvesting from piano playing motion. Nano. Energy. 2025, 138, 110816.
33. Jiao, Y.; Lin, Z.; Guo, X.; et al. Compositional engineering of hybrid organic-inorganic lead-halide perovskite and PVDF-graphene for high-performance triboelectric nanogenerators. ACS. Appl. Mater. Interfaces. 2024, 16, 3532-41.
34. Fan, J.; Yang, R.; Du, Y.; et al. A triboelectric nanogenerator based on MXene/TPU composite films with excellent stretchability for self-powered flexible sensing. Nano. Energy. 2024, 129, 109999.
35. Tan, X.; Wang, S.; You, Z.; Zheng, J.; Liu, Y. High performance porous triboelectric nanogenerator based on silk fibroin@MXene composite aerogel and PDMS sponge. ACS. Materials. Lett. 2023, 5, 1929-37.
36. Rani, G. M.; Kim, H.; Pammi, S. V. N.; Umapathi, R.; Huh, Y. S. Next-generation flexible and wearable triboelectric nanogenerator based on PVDF-HFP@GCN composite for mechanical energy scavenging and sleep pattern detection. Adv. Funct. Mater. 2025, 36, e19594.
37. Gajula, P.; Yoon, J. U.; Woo, I.; Bae, J. W. Harnessing mechanical energy for green hydrogen: pioneering high-performance triboelectric nanogenerators. Adv. Funct. Mater. 2025, 35, 2501074.
38. Shao, Z.; Chen, J.; Xie, Q.; Mi, L. Functional metal/covalent organic framework materials for triboelectric nanogenerator. Coord. Chem. Rev. 2023, 486, 215118.
39. Wen, R.; Feng, R.; Zhao, B.; Song, J.; Fan, L.; Zhai, J. Controllable design of high-efficiency triboelectric materials by functionalized metal - organic frameworks with a large electron-withdrawing functional group. Nano. Res. 2022, 15, 9386-91.
40. Collins, S. P.; Daff, T. D.; Piotrkowski, S. S.; Woo, T. K. Materials design by evolutionary optimization of functional groups in metal-organic frameworks. Sci. Adv. 2016, 2, e1600954.
41. Lin, C.; Sun, L.; Meng, X.; et al. Covalent organic frameworks with tailored functionalities for modulating surface potentials in triboelectric nanogenerators. Angew. Chem. Int. Ed. 2022, 61, e202211601.
42. Yu, X.; Ge, J.; Wang, Z.; et al. High-performance triboelectric nanogenerator with synchronization mechanism by charge handling. Energy. Convers. Manag. 2022, 263, 115655.
43. Zhang, Y.; Zhang, Z.; Peng, W.; et al. Bionic wood-inspired structure enables aerogel film triboelectric material with humidity adaptation. Chem. Eng. J. 2024, 497, 154906.
44. Fu, C.; Han, X.; Zhang, Y.; Hu, J.; Du, J.; Wang, H. Synergistic functionality of metal-organic framework-cellulose composites structures for advanced high-performance energy materials. Chem. Eng. J. 2025, 503, 158384.
45. Pander, M.; Gil-San-Millan, R.; Delgado, P.; et al. MOF/polymer hybrids through in situ free radical polymerization in metal-organic frameworks. Mater. Horiz. 2023, 10, 1301-8.
46. Su, X.; Cheng, L.; Yan, X.; et al. In situ construction of amide-functionalized 2D conjugated metal-organic frameworks with multiple active sites for high-performance potassium-ion batteries. J. Am. Chem. Soc. 2025, 147, 18338-48.
47. Esposito, M. R.; Newar, R.; Kim, M.; Cohen, S. M. Incorporation of low-valent metal ions in metal-organic frameworks and coordination polymers. Coord. Chem. Rev. 2025, 531, 216491.
48. Ma, R.; Hou, Y.; Zhang, W.; Pan, Y.; Huo, S.; Shi, C. MOF-based nanocomposites in polymer matrix: progress and prospects. Adv. Compos. Hybrid. Mater. 2025, 8, 351.
49. Salahshoori, I.; Golriz, M.; Namayandeh, Jorabchi. M.; Moghari, S.; Khonakdar, H. A. Recent progress in MOF-based biopolymer composites for wastewater detoxification. Coord. Chem. Rev. 2025, 543, 216919.
50. Rana SM, Faruk O, Selim Reza M, Robiul Islam M, Kim H, Yeong Park J. All porous Ecoflex and SEBS-based stretchable high-performance triboelectric nanogenerator for self-powered human activity monitoring. Chem. Eng. J. 2024, 488, 151050.
51. Wan, H.; Fang, Y.; Hu, M.; et al. Interpretable machine-learning and big data mining to predict the CO2 separation in polymer-MOF mixed matrix membranes. Adv. Sci. 2025, 12, e2405905.
52. Feng, H.; Li, H.; Xu, J.; et al. Triboelectric nanogenerator based on direct image lithography and surface fluorination for biomechanical energy harvesting and self-powered sterilization. Nano. Energy. 2022, 98, 107279.
53. Wang, F.; Zhang, Y.; Li, S.; et al. Integration of cobalt coordination polymer-based triboelectric nanogenerators as sustainable power sources for self-driven selective photocatalytic reactions. Chem. Eng. J. 2025, 503, 158194.
54. Wang, X.; Ye, C.; Chen, P.; et al. Achieving high power density and durability of multilayered swing-structured triboelectric nanogenerator toward marine environmental protection. Adv. Funct. Mater. 2023, 34, 2311196.
55. Higoshi, H.; Naritomi, D.; Kamiyauchi, S.; et al. Self-folded corrugated origami sensor based on triboelectric nanogenerator for a smart cushioning device. Adv. Mater. Technol. 2025, 10, 2500032.
56. Wu, G.; Xu, M.; Lei, M.; et al. Full-fiber triboelectric nanogenerators with knitted origami structures for high impact resistance intelligent protection fabric. Mater. Horiz. 2025, 12, 1246-54.
57. Hong, H.; Yang, X.; Cui, H.; et al. Self-powered seesaw structured spherical buoys based on a hybrid triboelectric-electromagnetic nanogenerator for sea surface wireless positioning. Energy. Environ. Sci. 2022, 15, 621-32.
58. Shan, C.; He, W.; Wu, H.; et al. Dual mode TENG with self-voltage multiplying circuit for blue energy harvesting and water wave monitoring. Adv. Funct. Mater. 2023, 33, 2305768.
59. Zheng, Z.; Xia, J.; Wang, B.; Guo, Y. Hierarchically designed nanocomposites for triboelectric nanogenerator toward biomechanical energy harvester and smart home system. Nano. Energy. 2022, 95, 107047.
60. Huang, C.; Zhu, K.; Zhang, Y.; et al. Surfactant-assisted assembly of nanoscale zinc coordination compounds to enhance tandem conversion reactions in water. Dalton. Trans. 2019, 48, 16008-16.
61. Xiang, L.; Tang, C.; Xu, Z.; Xu, F.; Li, C.; Mai, Y. Functional porous cubosomes: synthesis and applications in energy storage and conversion. Acc. Mater. Res. 2025, 6, 939-51.
62. Heine, T.; Dinca, M.; Zhu, G. Physical phenomena in porous frameworks. Acc. Chem. Res. 2025, 58, 327-9.
63. Si, H.; Lee, D.; Park, D.; et al. Size effects of polydopamine-coated BaTiO3 nanoparticles on the piezoelectric performance of P(VDF-TrFE)/BaTiO3 composite. J. Materiomics. 2024, 10, 857-69.
64. Luo, J.; Wang, Y. Effect of hierarchical layer structure design on the sensing performances of NaNbO3/P(VDF-TrFE) piezoelectric composite films. J. Alloys. Compd. 2025, 1017, 179071.
65. Lee, C. H.; Min, K. A.; Park, H. B.; Hong, Y. T.; Jung, B. O.; Lee, Y. M. Sulfonated poly(arylene ether sulfone)-silica nanocomposite membrane for direct methanol fuel cell (DMFC). J. Membr. Sci. 2007, 303, 258-66.
66. Shalu, S.; Kar, P.; Krupka, J.; Dasgupta, Ghosh. B. Synthesis, characterization, thermal, dynamic mechanical, and dielectric studies of Ba0.7Sr0.3TiO3/polystyrene composites. Polym. Compos. 2018, 39, 1714-24.
67. Bochu, L.; Babu, A.; Potu, S.; Khanapuram, U. K.; Rajaboina, R. K.; Kodali, P. Enhancing triboelectric nanogenerator performance with metal-organic framework composite nanofibers: applications in public transit monitoring, staircase alerts, and security. ACS. Appl. Polym. Mater. 2025, 7, 4132-41.
68. Graham, S. A.; Kurakula, A.; Kavarthapu, V. S.; et al. Metal-organic framework embedded electrospun fibrous membranes-based hybrid nanogenerators with hierarchical modified polyamide films for mechanical energy harvesting and IoT applications. Adv. Funct. Mater. 2025, 36, e07125.
69. Salauddin, M.; Rana, S. S.; Sharifuzzaman, M.; et al. Laser-carbonized MXene/ZiF-67 nanocomposite as an intermediate layer for boosting the output performance of fabric-based triboelectric nanogenerator. Nano. Energy. 2022, 100, 107462.
70. Xiong, J.; Cui, P.; Chen, X.; et al. Skin-touch-actuated textile-based triboelectric nanogenerator with black phosphorus for durable biomechanical energy harvesting. Nat. Commun. 2018, 9, 4280.
71. Wu, Y.; Zhang, D.; Zhang, Y.; et al. Water wave energy-harvesting accordion structure triboelectric nanogenerators for self-driven corrosion protection. Nano. Energy. 2025, 142, 111207.
72. Xu, L.; Pang, Y.; Zhang, C.; et al. Integrated triboelectric nanogenerator array based on air-driven membrane structures for water wave energy harvesting. Nano. Energy. 2017, 31, 351-8.
73. Xu, J.; Yue, X.; He, L.; et al. Photoinduced protocol for aerobic oxidation of aldehydes to carboxylic acids under mild conditions. ACS. Sustain. Chem. Eng. 2022, 10, 14119-25.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].