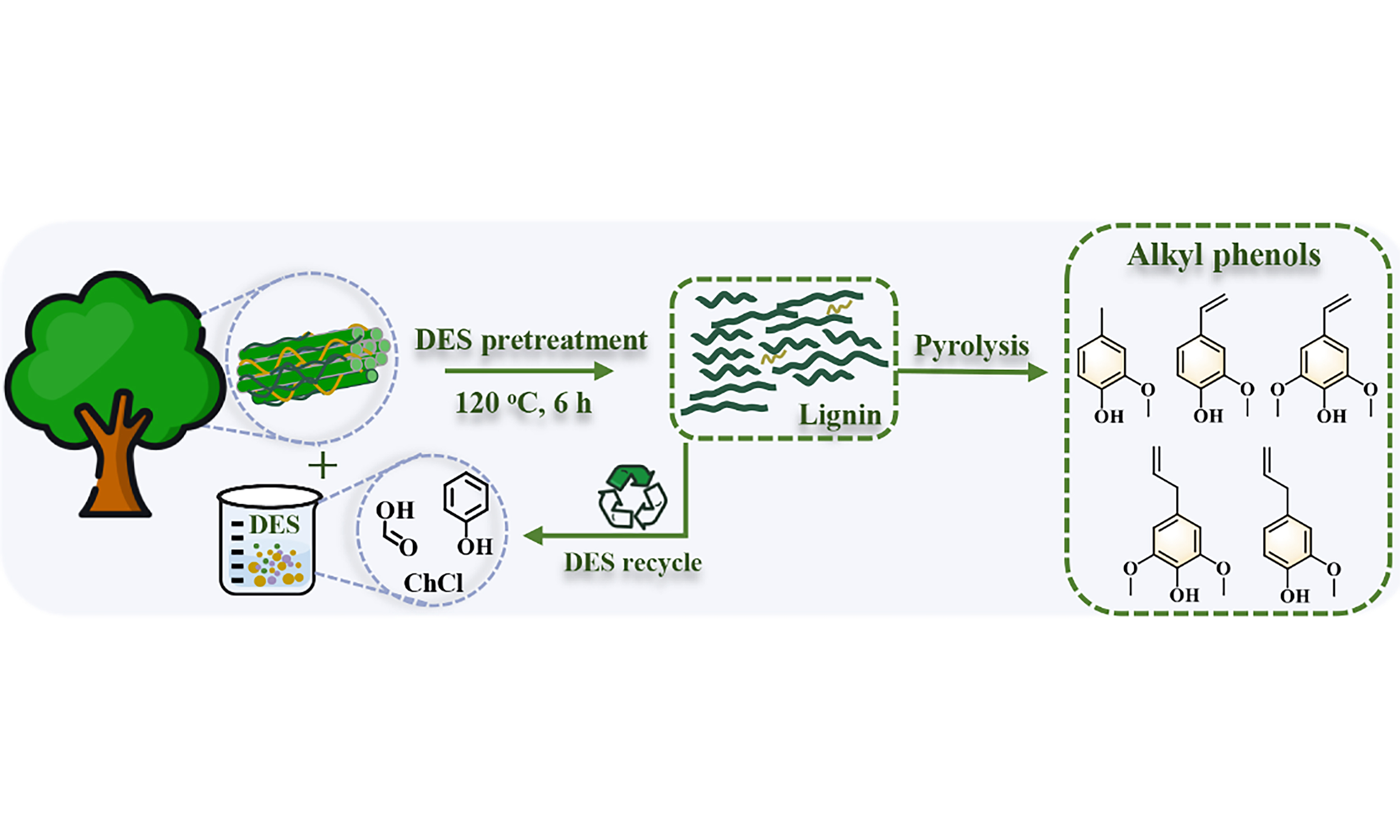
Lignin separation and tailoring with phenol-assisted deep eutectic solvents for enhancing alkyl phenols yield in pyrolysis
Abstract
Deep eutectic solvents (DESs) offer a sustainable and effective strategy for lignin fractionation from biomass, improving the efficiency of enzymatic hydrolysis. However, the downstream utilization of the lignin extracted in high yield through DES is a difficult problem. Herein, this work employed a novel acidic DES with phenolic modifiers to investigate their dual role in lignin extraction and structural modification, aiming to optimize the production of phenolic compounds via pyrolysis. The result showed that the choline chloride/formic acid/phenol DES with a molar ratio of 1:2:0.05 exhibited an excellent lignin extraction efficiency when pretreated at 120 °C for 6 h, and the recovered lignin maintained a high β-O-4 content, decreased molecular weight, and low char yield. High yield and selectivity of alkyl phenols were obtained by pyrolysis of the regenerated lignin extracted under the optimal pretreatment conditions. The low-condensed lignin easily generated phenolic compounds after pyrolysis. After five cycles of reuse, the recycled DES maintained a superior delignification effect but significantly decreased pyrolysis efficiency compared to the fresh DES. This indicated that the highly condensed lignin with a large molecular weight is not favorable for producing phenolic compounds by pyrolysis. These highlight the potential of DES-based strategies for the efficient extraction and structural tailoring of lignin to maximize the production of value-added phenolic compounds.
Keywords
INTRODUCTION
Due to serious environmental pollution and a sharp decline in fossil reserves caused by the over-exploitation and consumption of fossil resources, the development of green sustainable biofuels using lignocellulose as feedstock has gained wide attention[1-4]. Lignin, one of the primary components of lignocellulose, is a natural aromatic heteropolymer linked by a variety of chemical bonds, accounting for 15-35 wt% of lignocellulose[5]. The unique aromatic structures of lignin make it great and promising for producing aromatic platform chemicals. To date, a variety of conversion approaches including gasification, liquefaction, and pyrolysis have been applied for the high-value conversion of lignin[6-8]. Among these approaches, the pyrolysis of lignin is an attractive and powerful method for producing phenolic products due to its easy operation and low capital investment[9]. However, the production of pyrolytic products is profoundly influenced by the structural features of lignin which are highly determined by lignin extraction conditions. Therefore, pursuing an excellent extraction technology for separating high-yield lignin with a suitable structure is a prerequisite for promoting lignin pyrolysis.
Most studies have dedicated efforts to exploring the effect of the lignin extraction method on the pyrolysis behavior of lignin[10-12]. For example, the pyrolysis products of four lignin fractions, including milled wood lignin, alkali lignin (AL), and sulfonate lignin, were compared[13]. The results showed that sulfonate lignin was converted into furan and sulfur compounds after pyrolysis, but the pyrolysis of the other three lignins mainly generated phenols. This indicated the significance of selecting a suitable lignin extraction method for customizing the specific pyrolysis products. Subsequently, γ-valerolactone (GVL)/diluted sulfuric acid-based binary solvent system was used to pretreat pine for the extraction of the GVL-lignin which mainly produced phenols and alkoxy phenols after pyrolysis[14]. Meanwhile, increasing sulfuric acid concentration and reaction temperature applied in the GVL-lignin extraction could enhance the amount of phenols in pyrolysis. However, these methods were accompanied by high temperatures, long duration, inorganic acid/alkali environment, and difficult solvent recovery, inevitably causing the introduction of new functional groups and severe side reactions of lignin, such as condensation reaction, which could affect pyrolysis efficiency.
Deep eutectic solvent (DES), a green designer solvent consisting of a hydrogen bond acceptor (HBA) and a hydrogen bond donor (HBD), has received wide attention in lignin extraction from lignocellulose due to its selective dissolution on lignin, available feedstock, and recyclability[15,16]. It was revealed that acidic DES with acid HBD usually exhibits better lignin extraction efficiency compared to other DES systems[17,18]. Above 80% of lignin was extracted from miscanthus during the choline chloride (ChCl)/formic acid (FA) DES pretreatment[19,20]. Although the acidic DES pretreatment can extract high-yield lignin, the extensive cleavage of unstable ether bonds and severe condensation reactions are not conducive to the following utilization of the extracted lignin[21,22]. Most research disclosed that the structural properties of lignin could be properly regulated by selecting suitable DES components and treatment conditions[23,24]. For example, a binary acid-alcohol DES was synthesized to modify the structure of soft kraft lignin, and the effect of different DES pretreatments on lignin pyrolysis products was discussed[25]. The result showed that the yield of the DES-treated lignin pyrolysis products was enhanced compared to the untreated lignin. Most studies revealed that the pyrolysis of the DES-modified lignin could produce abundant phenolic compounds[26,27]. However, few studies have focused on how the structure of the DES-extracted lignin affects the distribution and efficiency of lignin pyrolysis products.
In the previous study, we designed a phenols-based acidic DES for biomass pretreatment and lignin separation to improve the accessibility of enzymes, thus achieving a high enzymatic hydrolysis efficiency of cellulose-rich residue[28]. Nevertheless, very limited information was available about the structure of the high-yield lignin extracted by the phenols-based DES, which posed challenges for its downstream high-value applications. Herein, the phenols-based acidic DES was used to extract lignin from eucalyptus, aiming to evaluate the impact of DES processing conditions on structural characterizations and pyrolysis behaviors of lignin. FA-based ternary DES containing various HBAs and phenol compounds was synthesized and used to pretreat eucalyptus at different temperatures and times for lignin extraction. The structural features of the regenerated lignin, including functional groups, internal linkage, structural units, molecular weight distribution, and thermal-degradation properties, were extensively investigated. Furthermore, the efficiency and selectivity of lignin pyrolysis were comprehensively explored. This study could provide a novel and sustainable pathway forward for the valorization of the DES-extracted lignin.
EXPERIMENTAL
Material and DES synthesis
Corn stalk, collected from Shaanxi, China, was processed by crushing and subsequently screened to obtain a powder with a particle size of 40-60 mesh before use. Corn stalk was composed of 45.11% cellulose, 20.10% hemicelluloses, and 21.26% lignin. The following chemicals were provided by Shanghai Aladdin Biochemical Technology Co., Ltd., China: monoethanolamine (MEA, 99% purity), ChCl (98%), NH4Cl (98%), tetra-n-butyl ammonium chloride (TBAC, 98%), ethanol (95%), and anhydrous sodium acetate
For the preparation of DES, three HBAs, including ChCl, NH4Cl, and TBAC, were mixed with MEA at varying molar ratios. The mixtures were then heated to 85 °C and stirred for 6 h to produce a uniform and clear solvent. Prior to use, the prepared DES was transferred to a desiccator for storage.
DES pretreatment
First, 3.0 g of eucalyptus was blended with 30 mL of DES, and then heated at varying temperatures (100-
Once the lignin had been separated, the remaining liquid was gathered and concentrated using a rotary evaporator to remove water, bringing it back to the initial volume of the DES. Taking into account the FA lost during rotary evaporation, the DES solution that was reclaimed was then brought back to the pH of the fresh DES by adding FA, and it was subsequently employed for another round of pretreatment. The lignin fractions obtained from each cycle were labeled as 1C, 2C, 3C, 4C, and 5C, corresponding to their respective cycle numbers.
Lignin pyrolysis
The lignin samples were analyzed using pyrolysis-gas chromatography/mass spectrometry (Py-GC/MS) techniques with a CDS 6150 Series pyrolyzer coupled to a Perkin Elmer Clarus 560 GC/MS system. For each analysis, a precise amount of lignin (0.1 mg) was loaded into a quartz tube and pyrolyzed at 400 °C for 20 s, achieving a heating rate of 20,000 °C/s. The identification of pyrolysis products was carried out using the NIST17.L mass spectrometry database[29].
NMR analysis
All nuclear magnetic resonance (NMR) spectra of lignin were recorded on a Bruker AVIII 400 MHz (Bruker, Germany) spectrometer at 25 °C. The liquid was dissolved in 0.2 mL of dimethyl sulfoxide (DMSO)-d6. The peak (2.49 ppm) of DMSO-d6 was used as internal standard. The 1H NMR spectra were acquired according to previous papers[5,6]. For the 2D heteronuclear single quantum coherence (HSQC) NMR, 30 mg of lignin was dissolved in 0.5 mL of DMSO-d6. The data was processed using standard Bruker Topspin-NMR software and the DMSO-d6 was used as an internal standard (δC/δH 39.5/2.49). For 31P NMR, 20 mg of lignin was dissolved in a solvent mixture composed of anhydrous pyridine and deuterated chloroform, with cyclohexanol as an internal standard, chromium (III) acetylacetonate solution as relaxation reagent and 2-chloro-1, 3, 2-dioxaphospholane as the phosphitylating reagent.
RESULTS AND DISCUSSION
Delignification effectiveness at different DES systems
In this study, eucalyptus was pretreated by using FA-based DES with various phenols under different conditions (HBA types, phenol types, molar ratio, temperature, and time). The delignification rate and lignin extraction yield under DES pretreatment are shown in Supplementary Figure 1. For the DESs containing different HBA types, the delignification rate after the ChCl-based DES pretreatment was about three times that after the TBAC-based DES pretreatment and six times that after the Bet-based DES pretreatment [Supplementary Figure 1A]. Moreover, the ChCl-based DES exhibited the highest lignin extraction yield (61.58%). This result indicated that ChCl was the best HBA in the synthesis of DES for achieving efficient delignification. When various phenols were used as an enhancer in DES, the delignification rate and lignin extraction yield after phenol-enhanced DES pretreatment were greater than those of the eucalyptus pretreated with the pure ChCl/FA DES pretreatment (69.97%). Moreover, the Ph-based DES presented superior delignification rate and lignin extraction yield compared to the Sy- and Gu-based DES [Supplementary Figure 1B]. Within the ChCl/Ph/FA DES system, where the molar ratio of Ph remained constant, an increase in FA content and a decrease in ChCl content corresponded to a higher delignification rate and lignin extraction yield [Supplementary Figure 1C]. Moreover, the highest delignification rate and lignin extraction yield were achieved when the molar ratio of ChCl/Ph/FA DES was 1:0.05:2. The delignification rate elevated from 31.75% to 85.48% with increasing temperature
Structure analysis of the regenerated lignin
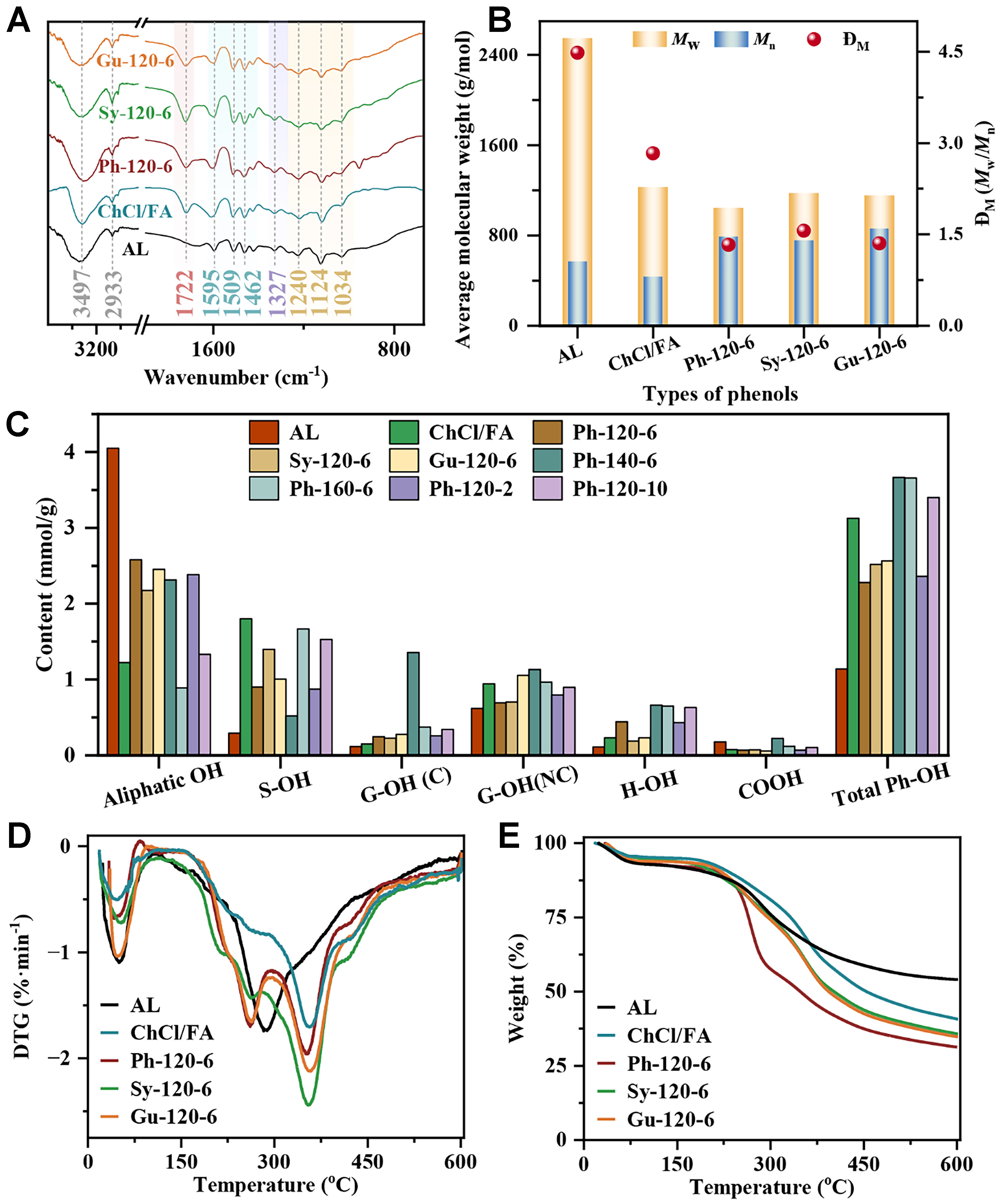
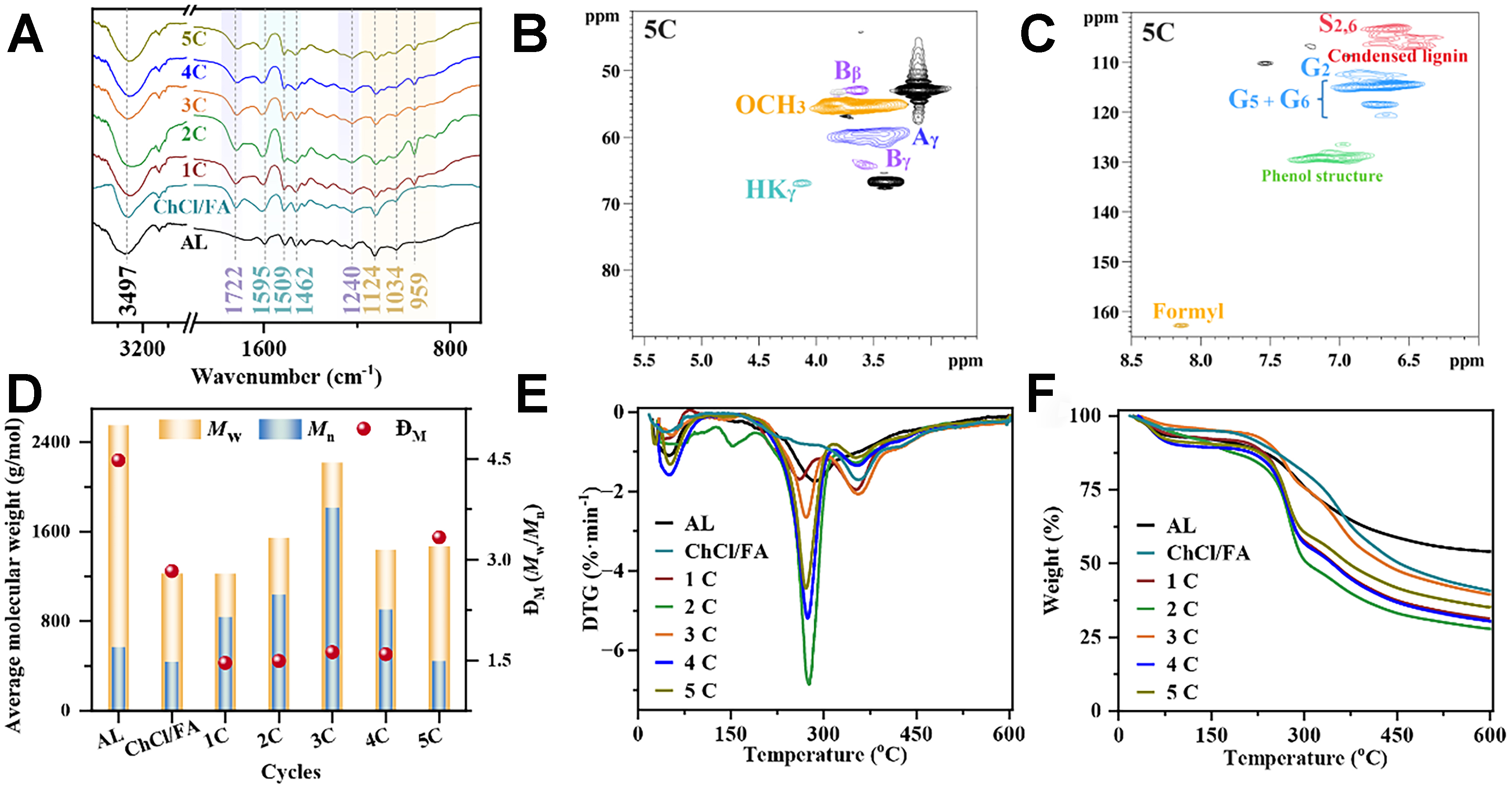
The delignification rate is primarily related to the ChCl/Ph/FA DES treatment conditions, which could reflect the damage degree of DES to the cell wall of biomass. Notably, the structural features of the DES regenerated lignin can markedly determine the final lignin-based product quality. Considering the prospective use of these lignin fractions in pyrolysis, the functional groups of the lignin had a great effect on the selectivities and contents of pyrolysis products. Herein, the structural changes of AL and the regenerated lignin fractions after pretreatment were observed using Fourier transform infrared spectroscopy (FT-IR) analysis [Supplementary Figure 2 and Figure 1A]. The peak at 3,497 cm-1 was assigned to the stretching vibrations of hydroxyl groups (OH) including the aliphatic and phenolic OH. This peak exhibited higher intensity in pure ChCl/FA lignin than phenols-based DES lignin [Supplementary Figure 2 and Figure 1A]. The higher OH content in pure ChCl/FA lignin was mostly due to the essential fragmentation of β-O-4 linkages[9]; instead, the incorporation of a phenol compound in DES can decrease the extent of ether linkages fracture via the explicit arylation with phenols[30]. Besides, the intensity of 3,497 cm-1 peak in
Figure 1. Structure properties of lignin fractions. (A) FT-IR curves; (B) molecular weight; (C) content of various OH groups detected by 31P NMR; (D) TG curves; (E) DTG curves. FT-IR: Fourier transform infrared spectroscopy; 31P NMR: phosphorus nuclear magnetic resonance; TG: thermogravimetric; DTG: derivative thermogravimetric.
During DES pretreatment, variation in the molecular weight of lignin inevitably occurred, resulting from both depolymerization and condensation reactions[32]. The weight average molecular (Mw) and number average molecular (Mn) of the lignin fragments were detected based on acetylated lignin using gel permeation chromatography (GPC) analysis, and dispersity (ĐM) is equal to Mw divided by Mn
The differences in OH groups in AL and the regenerated lignin were analyzed by 31P NMR. Figure 1C illustrates that the aliphatic OH content in AL far exceeds that of the regenerated lignin. The substantially reduced aliphatic OH content indicated that the dehydration and acylation reaction could occur in the DES system. Besides, phenol types in DES had a minimal impact on the aliphatic OH content. However, elevated reaction temperatures and extended reaction times led to a considerable decrease in the aliphatic hydroxyl group content. The S-OH content in the Sy-120-6 sample was higher than that of the
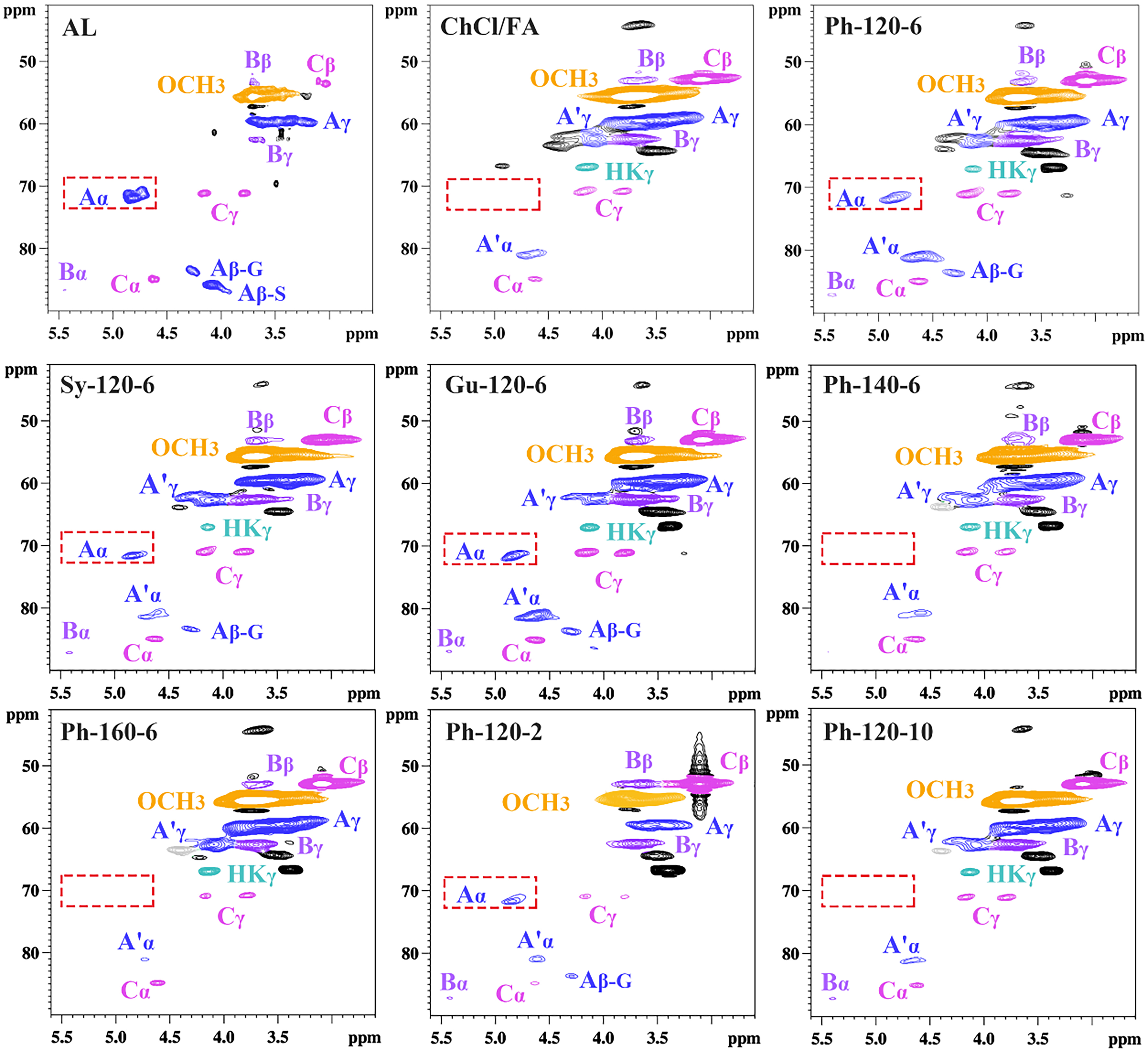
To delve into structure changes of the regenerated lignin during DES pretreatment, the type and content of the internal linkages in the lignin were examined using 2D HSQC NMR, with the principal lignin substructures depicted in Supplementary Figure 4. The signals were primarily divided into the aliphatic and aromatic regions [Figures 2 and 3]. In the aliphatic regions, the dominating cross-signals including methoxyl groups (OCH3), β-O-4 (A), β-5 (B), and β-β (C) linkages were identified [Figure 2][34]. After DES pretreatment, it was noted that the Aα signal in the ChCl/FA sample disappeared completely; on the contrary, the Aα signal was still present in Ph-120-6, Sy-120-6, and Gu-120-6 samples but the signal intensity was significantly reduced compared to that in AL. The result proved that the introduction of phenol compounds in the DES system could prevent the breakage of β-O-4 linkage, consistent with 31P NMR result. Nevertheless, after the pretreatment of ChCl/FA/Ph DES, it was found that the Aα signal could be retained at a low temperature (120 °C) and short time (2 and 6 h), but completely disappeared at a high temperature (140 and 160 °C) and long time (10 h). Moreover, the intensity of signals relating to β-5 and β-β linkages progressively diminished as the reaction temperature and duration increased. The result showed that β-O-4 linkages were susceptible to reaction temperature and time. Additionally, two novel signals A’α (4.63/81.0 ppm) and A’γ (4.32/62.0 ppm) were observed in the regenerated lignin. These signals indicated the formation of the acylated β-O-4 substructure resulting from the acylation reaction between carboxyl group in FA and OH groups in the lignin side chain[35]. Meanwhile, it can be seen that the regenerated lignin shows the signal of Hibbert’s ketone, suggesting the breakage of ether linkages[36].
Figure 2. Comparison of aliphatic regions of lignin fractions in 2D HSQC spectra. HSQC: Heteronuclear single quantum coherence.
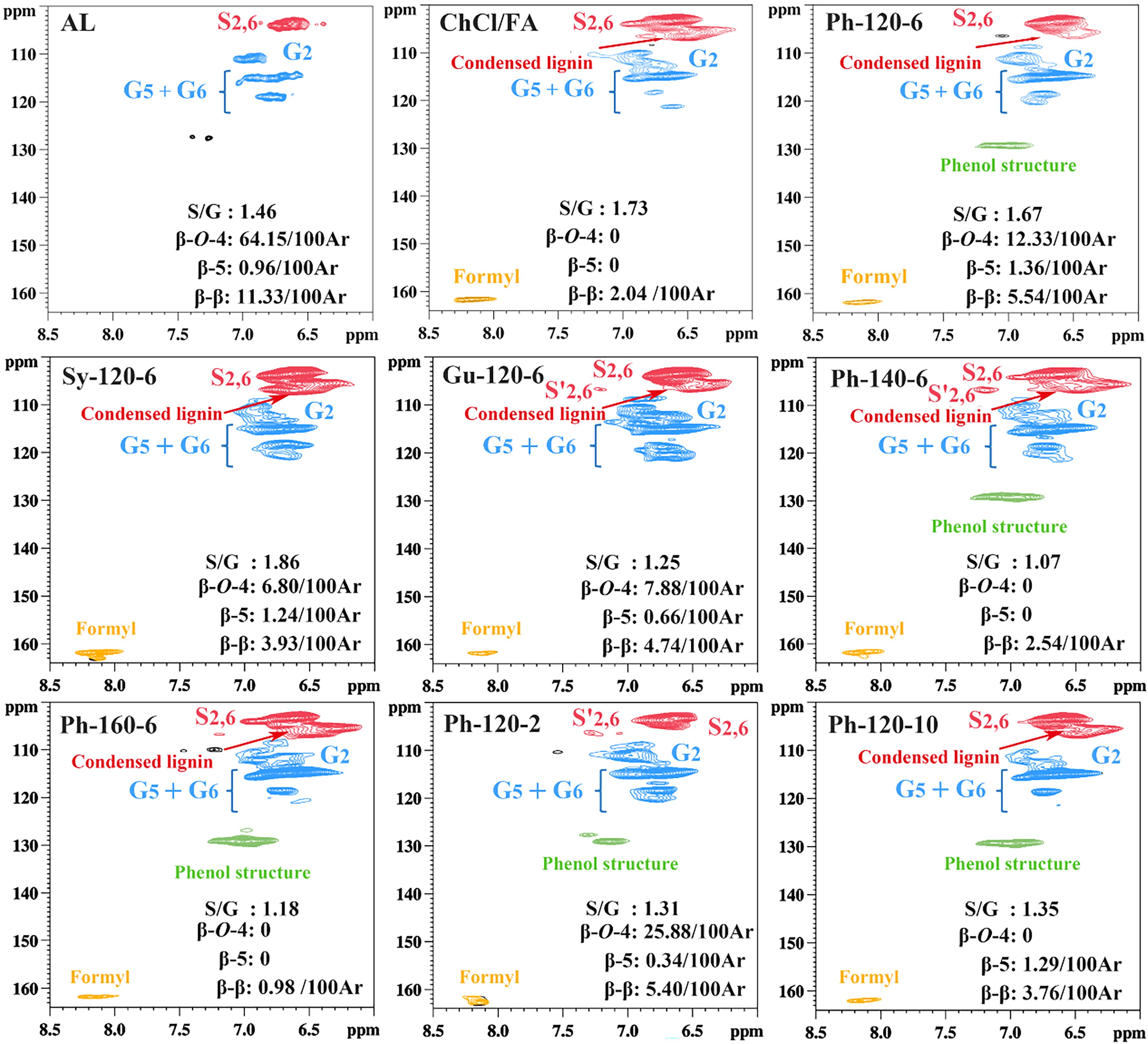
Figure 3. Comparison of aromatic regions of lignin fractions in 2D HSQC spectra. HSQC: Heteronuclear single quantum coherence.
In the aromatic domain, the signals of the S and G units were presented [Figure 3]. Compared to AL, the G signal in the ChCl/FA sample caused severe condensation, but the signal in the Ph-120-6, Sy-120-6, and Gu-120-6 samples was only slightly changed. The result indicated that the introduction of the phenol compounds in the DES system can protect the lignin structure to some degree[30]. However, in the ChCl/FA/Ph DES system, when the reaction temperature and time exceeded 120 oC and 6 h, respectively, the G units underwent condensation. Furthermore, Moreover, the intensity of signals corresponding to S-type lignin units progressively rose, whereas the signals associated with G units became weak, indicating that the condensation of lignin occurred[37]. Besides, the formyl signal (8.10/161.7 ppm) was observed in all DES regenerated lignin samples, which was caused by FA in the DES system, consistent with the previous literature[30].
The relative abundances of the internal linkages and S/G ratio in lignin were determined based on the number per 100 aromatic rings (Ar) by integrating the related signals. Figure 3 illustrates that the β-O-4 linkage content in the regenerated lignin is rather lower than that in AL, indicating the substantial cleavage of β-O-4 linkages in the DES. Moreover, the β-O-4 linkage content was remarkably reduced in the ChCl/FA sample compared to that in the Ph-120-6, Sy-120-6, and Gu-120-6 samples, proving that the introduction of a phenol compound to the DES could decrease the degree of ether linkage breakage. Besides, the β-O-4 linkage content of the Ph-120-6 sample (12.33/100 Ar) was slightly higher than that of the Sy-120-6 (6.80/100 Ar) and Gu-120-6 (7.88/100 Ar) samples. The result indicated that the Ph had a better protective effect on lignin structure compared to the Sy and Gu, which could be attributed to the low steric hindrance of Ph. Additionally, it was seen that the increase in the reaction temperature and time resulted in the decrease in the internal bonds including β–O–4, β–5, and β–β. The result implied that a relatively high temperature and long duration during DES processing usually caused significant damage to the lignin structure. When the pretreatment condition was the same, the highest S/G ratio was detected in Sy-120-6 (1.86), followed by ChCl/FA (1.73), Ph-120-6 (1.67), AL (1.46), and Gu-120-6 (1.25). The highest S/G ratio in Sy-120-6 could be due to the grafting of Sy onto the lignin structure, causing the increase of S unit content. Similarly, the lowest S/G ratio in Gu-120-6 was ascribed to the introduction of Gu onto the lignin structure, thus enhancing the content of G units in lignin. During the ChCl/FA/Ph DES pretreatment, the reaction temperature had a greater effect on the S/G ratio than time.
Thermogravimetric (TG) analysis is a usable method to evaluate thermal stability, which will be conducive to predicting the pyrolysis behavior of lignin that follows[25]. The thermochemical degradation of lignin as a heterogeneous polymer with various functional groups, occurred across a wide temperature range. The thermal degradation characteristics of AL and the regenerated lignin were investigated through TG and derivative thermogravimetric (DTG) analysis [Figure 1D and Supplementary Figure 5]. The AL showed three main degradation stages, while the decomposition process of the regenerated lignin was mainly divided into four stages. All lignin fractions exhibited the first degradation stage at 30-100 °C, which was ascribed to moisture evaporation. The AL and ChCl/FA samples only exhibited one large decomposition at 100-470 °C, and the maximum weight loss rate temperature of the ChCl/FA sample (356 °C) was higher than that of AL (286 °C). This result indicated that the thermal stability of the ChCl/FA lignin was largely enhanced due to the considerable breakage of unstable ether linkages after pretreatment. Comparatively, the phenols-based DES lignin samples appeared with two primary degradation peaks of 100-300 °C and 300-
Impact of DES reuse on lignin structure
In this study, the optimal pretreatment parameters (ChCl/FA/Ph DES with a 1:2:0.05 molar ratio, 120 °C,
Figure 4. Structure characterization of AL and lignin fractions after cycle. (A) FT-IR curves; (B) aliphatic domain of 2D HSQC spectrum; (C) aromatic domain of 2D HSQC spectrum; (D) molecular weight; (E) DTG curves; (F) TG curves. AL: Alkaline lignin; FT-IR: Fourier transform infrared spectroscopy; HSQC: heteronuclear single quantum coherence; DTG: derivative thermogravimetry; TG: thermogravimetry.
The 2D HSQC NMR spectrum in the regenerated lignin after the fifth cycle showed that the β-O-4 linkage signal disappeared completely, but the signal of Hibbert’s ketone was observed in the aliphatic domain
The GPC results of the regenerated lignin after different cycles are shown in Figure 4D. The Mw and ĐM of the regenerated lignin after cycles were lower than those of AL but were higher than those of the 1C sample. The result implied that the recycled DES showed an excellent ability to improve the cleavage of the labile linkages. The higher Mw and ĐM of the regenerated lignin compared to the 1C sample were likely due to the precipitation of small molecular lignin fractions dissolved in the previous cycles onto the lignin structure. The DTG curves showed that the weight loss rate temperatures of the regenerated lignin after cycles were close to those of the 1C sample [Figure 4E]. The TG curves revealed that the char yield of the 5C sample (35.28%) was slightly higher than that of the 1C sample (31.28%) [Figure 4F]. This suggested that the recycled DES was not conducive to suppressing the formation of coke from lignin pyrolysis.
Pyrolysis behaviors of lignin
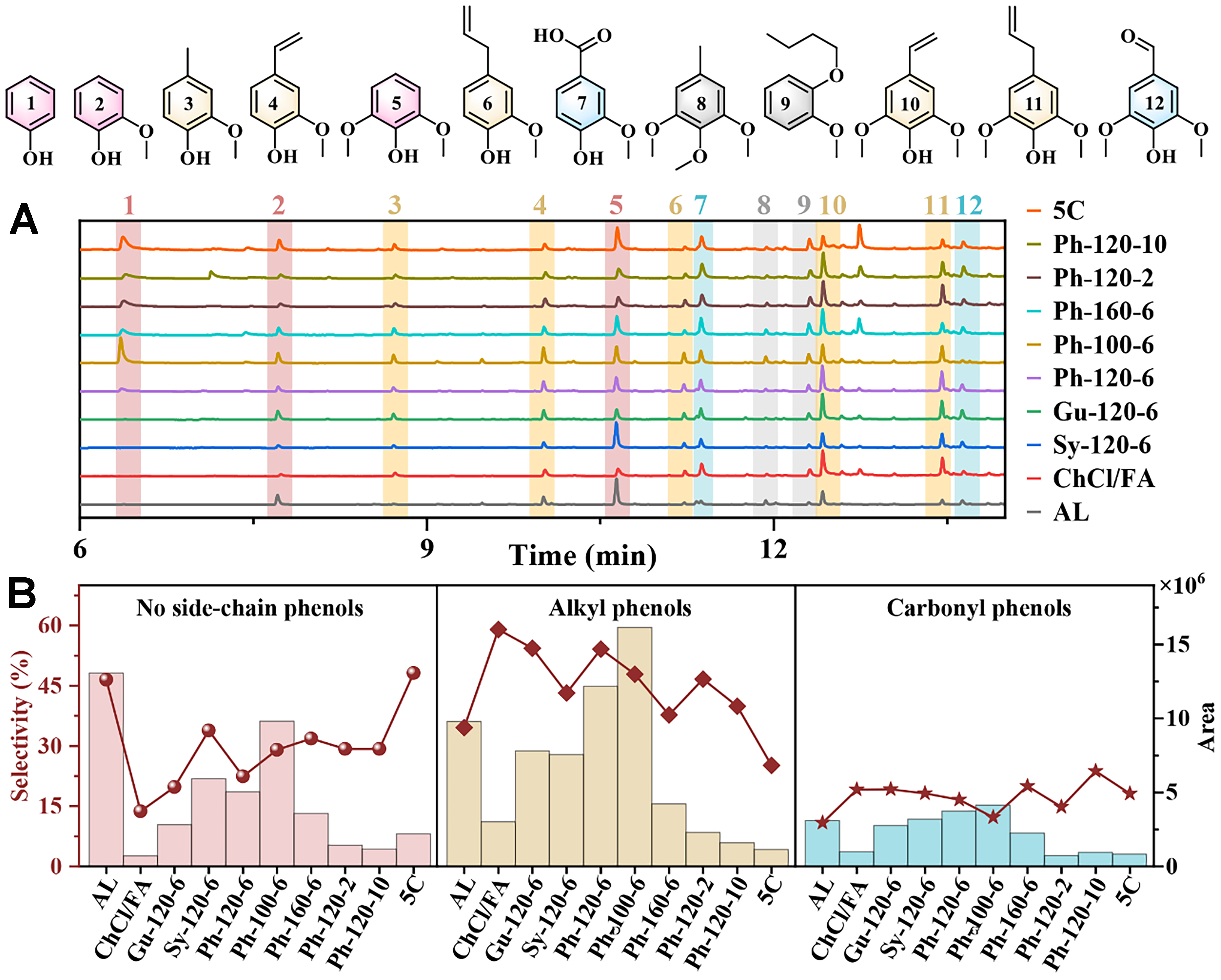
To achieve the value-added conversion of the regenerated lignin, a fast pyrolysis experiment was conducted at 400 °C by Py-GC/MS. The chromatograms of AL and regenerated lignin after the DES pretreatment are delineated in Figure 5A, and twelve lignin pyrolysis products were identified. Notably, the pyrolysis curves of all Ph-based DES lignin observed a phenol peak, while no phenol peak was detected in the pyrolysis curves of the AL, ChCl/FA, Sy-120-6, and Gu-120-6 samples. This result indicated the successful introduction of the Ph in the ChCl/FA/Ph DES system onto the lignin structure, aligned with the NMR results. The areas and selectivities of different lignin pyrolysis products are shown in Supplementary Tables 1 and 2, respectively.
Figure 5. Pyrolysis products obtained from AL and DES regenerated lignin. (A) Py-GC/MS chromatograms and (B) selectivity (point plot) and area (bar graph). Py: Pyrolysis; GC/MS: gas chromatography/mass spectrometry; AL: alkali lignin; DES: deep eutectic solvent.
For better comparison, the phenolic products after lignin pyrolysis were divided into three major categories: no side-chain phenols, alkyl phenols, and carbonyl phenols according to their side-chain structures in aromatic compounds. The areas and selectivities of the three kinds of phenolic compounds are presented in Figure 5B. Among these phenolic compounds, the area and selectivity of the carbonyl phenols were the lowest. The AL pyrolysis produced the largest area of the no-side-chain phenols, while the ChCl/FA lignin pyrolysis produced the smallest area. Comparatively, the area of the no-side-chain phenols from the regenerated lignin after the phenol-containing DES pretreatment fell between the above two lignin fractions. This could result from the retention of partial β–O–4 bonds and the grafting of phenols in the DES onto lignin, which was confirmed by NMR spectra. The largest alkyl phenols area does not necessarily correspond to the optimal overall yield. The previous research also revealed that more β-O-4 linkages were conducive to facilitating the pyrolysis of lignin to various phenolic compounds[40]. Besides, the pyrolysis of the phenols-containing DES lignin fractions generated more alkyl phenols than the no- side-chain phenols except for the 5C lignin. Furthermore, the largest area of the alkyl phenols was observed in the Ph-100-6 lignin, following that in the Ph-120-6 lignin. On the contrary, the selectivity of the alkyl phenols in the Ph-120-6 lignin was the highest among the regenerated lignin after the phenols-containing DES pretreatment. Although the pyrolysis of the Ph-100-6 lignin obtained the largest alkyl phenols area, the delignification efficiency was extremely weak at 100 °C during the ChCl/FA/Ph DES pretreatment, which will tremendously limit the final total yield of alkyl phenols. In short, the pyrolysis of the Ph-120-6 lignin would produce the high-yield alkyl phenols with high selectivity. Furthermore, the recycled DES significantly decreased pyrolysis efficiency compared to the fresh DES, likely attributed to the high condensation of lignin in the recycled DES.
CONCLUSIONS
This study aimed to elucidate the structural characteristics of the regenerated lignin after the phenol-based DES pretreatment and their influence on subsequent pyrolysis product distribution. The result showed that after the phenol-based DES pretreatment, the regenerated lignin displayed less β-O-4 linkage cleavage, lower molecular weight, and weaker thermal stability. The pyrolysis of the regenerated lignin generated more alkyl phenols than the no-side-chain phenols. After five recycling processes, the regenerated lignin, with a highly condensed degree and a large molecular weight, is not conducive to the production of mono-phenolics by pyrolysis. This study could promote insights into the lignin structure and motivate high-value utilization of lignin in downstream processing.
DECLARATIONS
Authors’ contributions
Investigation, methodology, data acquisition: Wang, T.; Wang, D.
Investigation, methodology: Chen, Z.; Wang, C.
Investigation: Wang, J.; Xie, C.
Supervision, funding acquisition, writing-review and editing: Zhang, C.
Investigation, validation, writing-original draft: Hong, S.
Supervision, validation, funding acquisition, writing-review and editing: Shen, X.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Undergraduate Innovation and Entrepreneurship Program of China (202310022083), the National Natural Science Foundation of China (22308029) and the Foundation of Key Laboratory of Pulp and Paper Science and Technology of the Ministry of Education of China (No.KF202202).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Yuan, J. S.; Pavlovich, M. J.; Ragauskas, A. J.; Han, B. Biotechnology for a sustainable future: biomass and beyond. Trends. Biotechnol. 2022, 40, 1395-8.
2. Tong, Z.; Li, X.; Dong, J.; et al. Adsorption configuration-determined selective hydrogenative ring opening and ring rearrangement of furfural over metal phosphate. ACS. Catal. 2021, 11, 6406-15.
3. Deng, Q.; Hou, X.; Zhong, Y.; et al. 2D MOF with compact catalytic sites for the one-pot synthesis of 2,5-dimethylfuran from saccharides via tandem catalysis. Angew. Chem. Int. Ed. Engl. 2022, 61, e202205453.
4. Deng, Q.; Li, X.; Gao, R.; et al. Hydrogen-catalyzed acid transformation for the hydration of alkenes and epoxy alkanes over Co-N frustrated lewis pair surfaces. J. Am. Chem. Soc. 2021, 143, 21294-301.
5. Mennani, M.; Kasbaji, M.; Ait, Benhamou. A.; et al. Current approaches, emerging developments and functional prospects for lignin-based catalysts - a review. Green. Chem. 2023, 25, 2896-929.
6. Singh-Morgan, A.; Puente-Urbina, A.; van, Bokhoven. J. A. Technology overview of fast pyrolysis of lignin: current state and potential for scale-up. ChemSusChem 2022, 15, e202200343.
7. Castro Garcia A, Cheng S, Cross JS. Lignin gasification: current and future viability. Energies 2022, 15, 9062.
8. Hernández-ramos, F.; Novi, V.; Alriols, M. G.; Labidi, J.; Erdocia, X. Optimisation of lignin liquefaction with polyethylene glycol/ glycerol through response surface methodology modelling. Ind. Crops. Prod. 2023, 198, 116729.
9. Yue, X.; Lin, J.; Suopajärvi, T.; et al. Conversion of highly polymerized lignin into monophenolic products via pyrolysis: a comparative study of acidic and alkaline depolymerization pretreatments using deep eutectic solvents. Chem. Eng. J. 2023, 478, 147368.
10. Miao, G.; Wong, J. L.; Chew, J. J.; Khaerudini, D. S.; Sunarso, J.; Xu, F. Deep eutectic solvent pretreatment of oil palm biomass: promoted lignin pyrolysis and enzymatic digestibility of solid residues. Int. J. Biol. Macromol. 2025, 293, 138847.
11. Tan, Y. T.; Chua, A. S. M.; Ngoh, G. C. Evaluation on the properties of deep eutectic solvent-extracted lignin for potential aromatic bio-products conversion. Ind. Crops. Prod. 2020, 154, 112729.
12. Wang, H.; Sun, Y.; Wang, B.; et al. Insights into the structural changes and potentials of lignin from bagasse during the integrated delignification process. ACS. Sustainable. Chem. Eng. 2019, 7, 13886-97.
13. Lin, X.; Sui, S.; Tan, S.; Pittman, C.; Sun, J.; Zhang, Z. Fast pyrolysis of four lignins from different isolation processes using Py-GC/MS. Energies 2015, 8, 5107-21.
14. Jampa, S.; Puente-urbina, A.; Ma, Z.; Wongkasemjit, S.; Luterbacher, J. S.; van, Bokhoven. J. A. Optimization of lignin extraction from pine wood for fast pyrolysis by using a γ-valerolactone-based binary solvent system. ACS. Sustainable. Chem. Eng. 2019, 7, 4058-68.
15. Zhou, M.; Fakayode, O. A.; Ahmed, Yagoub. A. E.; Ji, Q.; Zhou, C. Lignin fractionation from lignocellulosic biomass using deep eutectic solvents and its valorization. Renew. Sustain. Energy. Rev. 2022, 156, 111986.
16. Satlewal, A.; Agrawal, R.; Bhagia, S.; Sangoro, J.; Ragauskas, A. J. Natural deep eutectic solvents for lignocellulosic biomass pretreatment: recent developments, challenges and novel opportunities. Biotechnol. Adv. 2018, 36, 2032-50.
17. Hou, X. D.; Li, A. L.; Lin, K. P.; Wang, Y. Y.; Kuang, Z. Y.; Cao, S. L. Insight into the structure-function relationships of deep eutectic solvents during rice straw pretreatment. Bioresour. Technol. 2018, 249, 261-7.
18. Xiao, T.; Hou, M.; Guo, X.; et al. Recent progress in deep eutectic solvent(DES) fractionation of lignocellulosic components: a review. Renew. Sustain. Energy. Rev. 2024, 192, 114243.
19. Kohli, K.; Katuwal, S.; Biswas, A.; Sharma, B. K. Effective delignification of lignocellulosic biomass by microwave assisted deep eutectic solvents. Bioresour. Technol. 2020, 303, 122897.
20. Bai, Y.; Zhang, X. F.; Ma, J.; et al. Comprehensive utilization of poplar sawdust for glucose, xylose and lignin production using aluminum salt-based deep eutectic solvent. Int. J. Biol. Macromol. 2025, 308, 142657.
21. Liu, Y.; Deak, N.; Wang, Z.; et al. Tunable and functional deep eutectic solvents for lignocellulose valorization. Nat. Commun. 2021, 12, 5424.
22. Long, X.; Yao, M.; Wang, S.; et al. Efficient separation of poplar lignin using a new carboxylic acid-based deep eutectic solvents - choline chloride/malonic acid. ChemSusChem 2025, 18, e202402345.
23. Yu, Y.; Cheng, W.; Li, Y.; et al. Tailored one-pot lignocellulose fractionation to maximize biorefinery toward versatile xylochemicals and nanomaterials. Green. Chem. 2022, 24, 3257-68.
24. Chen, Z.; Bai, X.; A, L.; Zhang, H.; Wan, C. Insights into structural changes of lignin toward tailored properties during deep eutectic solvent pretreatment. ACS. Sustainable. Chem. Eng. 2020, 8, 9783-93.
25. Li, P.; Du, X.; Wu, S. Effect of acid-alcohol deep eutectic solvents-regulated lignin structure on subsequent pyrolysis products distribution. Fuel 2024, 378, 132879.
26. Li, T.; Yin, Y.; Wu, S.; Du, X. Effect of deep eutectic solvents-regulated lignin structure on subsequent pyrolysis products selectivity. Bioresour. Technol. 2022, 343, 126120.
27. Lu, M.; Sun, S.; Li, X.; Liu, Q.; Zhang, W.; Qiu, X. Fractionation of lignin from corncob with high-yield p - coumaric acid production in deep eutectic solvents followed by pyrolysis to produce monophenols. Int. J. Biol. Macromol. 2025, 285, 138331.
28. Wang, D.; Hong, S.; Chen, Z.; et al. Sustainable recycling of phenol-assisted deep eutectic solvent for efficient lignocellulose fractionation and enzymatic hydrolysis. Ind. Crops. Prod. 2025, 223, 120173.
29. Wen, J.; Sun, S.; Yuan, T.; Sun, R. Structural elucidation of whole lignin from Eucalyptus based on preswelling and enzymatic hydrolysis. Green. Chem. 2015, 17, 1589-96.
30. Li, N.; Yan, K.; Rukkijakan, T.; et al. Selective lignin arylation for biomass fractionation and benign bisphenols. Nature 2024, 630, 381-6.
31. Lu, C.; Xu, J.; Xie, J.; et al. Preparation, characterization of light-colored lignin from corn stover by new ternary deep eutectic solvent extraction. Int. J. Biol. Macromol. 2022, 222, 2512-22.
32. Wang, S.; Li, H.; Xiao, L.; Song, G. Unraveling the structural transformation of wood lignin during deep eutectic solvent treatment. Front. Energy. Res. 2020, 8, 48.
33. Zhao, X.; Yang, Y.; Xu, J.; et al. Lignin condensation inhibition and antioxidant activity improvement in a reductive ternary DES fractionation microenvironment by thiourea dioxide self-decomposition. New. J. Chem. 2022, 46, 8892-900.
34. Xu, Y.; Sun, S.; Zhang, C.; Ma, C.; Wen, J.; Yuan, T. Complete valorization of bamboo biomass into multifunctional nanomaterials by reactive deep eutectic solvent pretreatment: towards a waste-free biorefinery. Chem. Eng. J. 2023, 462, 142213.
35. van Erven G, Boerkamp VJP, van Groenestijn JW, Gosselink RJA. Choline and lactic acid covalently incorporate into the lignin structure during deep eutectic solvent pulping. Green. Chem. 2024, 26, 7101-12.
36. Hong, S.; Li, H. Y.; Shen, X. J.; Sun, S. N.; Sun, Z.; Yuan, T. Q. Unveiling the migration and transformation mechanism of lignin in eucalyptus during deep eutectic solvent pretreatment. ChemSusChem 2022, 15, e202200553.
37. Yao, X.; Pan, Y.; Ma, X.; Yin, S.; Zhu, M. Efficient separation and production of high-quality rubber, lignin nanoparticles and fermentable sugars from Eucommia ulmoides pericarp via deep eutectic solvent pretreatment. Int. J. Biol. Macromol. 2023, 253, 127221.
38. Moustaqim, M. E.; Kaihal, A. E.; Marouani, M. E.; et al. Thermal and thermomechanical analyses of lignin. Sustain. Chem. Pharm. 2018, 9, 63-8.
39. Ji, Q.; Yu, X.; Wu, P.; et al. Pretreatment of sugarcane bagasse with deep eutectic solvents affect the structure and morphology of lignin. Ind. Crops. Prod. 2021, 173, 114108.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].